Development and Characterization of a Novel Peptide-Loaded Antimicrobial Ocular Insert
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. VA-Loaded Matrices
2.2.1. Preparation and Characterization of the Freeze-Dried Inserts
2.2.2. Mucoadhesion Test
2.2.3. DSC Analysis
2.3. hLF 1-11-loaded Matrices
2.3.1. Preparation and Physicochemical Characterization
2.3.2. In Vitro Susceptibility Testing
2.4. In Vitro Drug Release Test
2.5. Analytical Methods
3. Results and Discussion
3.1. VA-Loaded Matrices
3.1.1. Preparation and Characterization of the Freeze-Dried Inserts Containing VA
3.1.2. Mucoadhesion Test
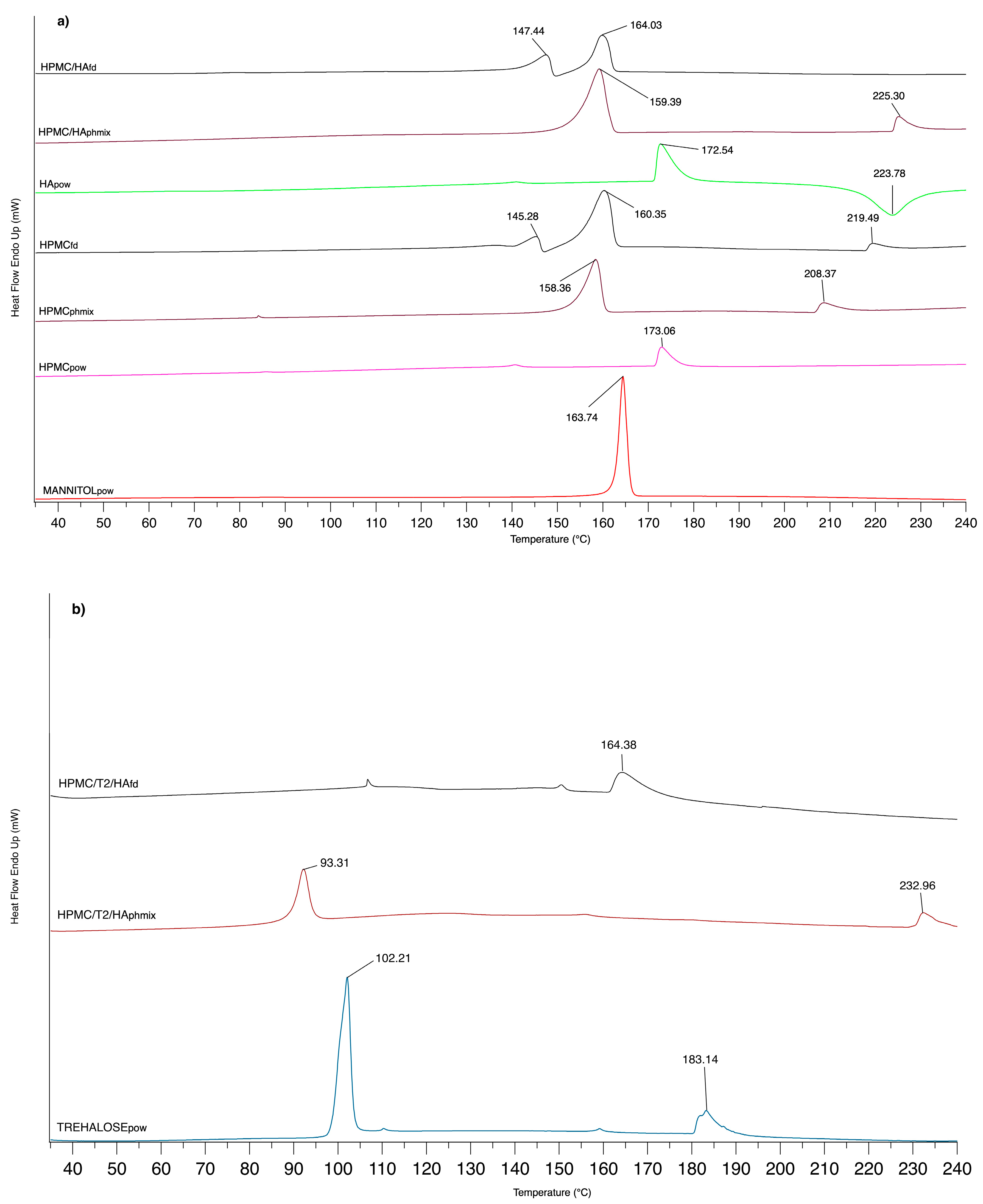
3.1.3. DSC Analysis
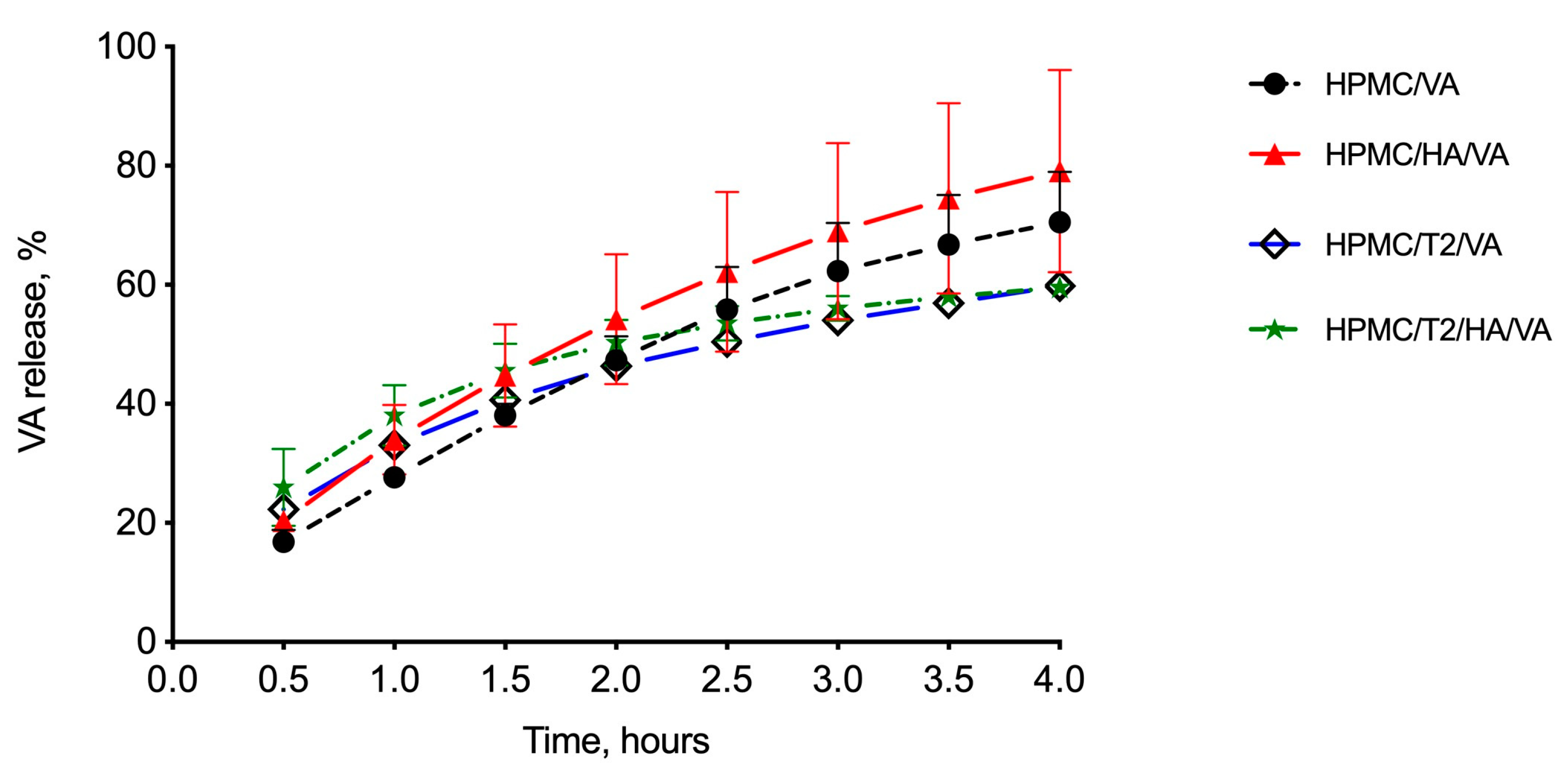
3.1.4. In Vitro Drug Release Studies
3.2. hLF 1-11-Loaded Matrices
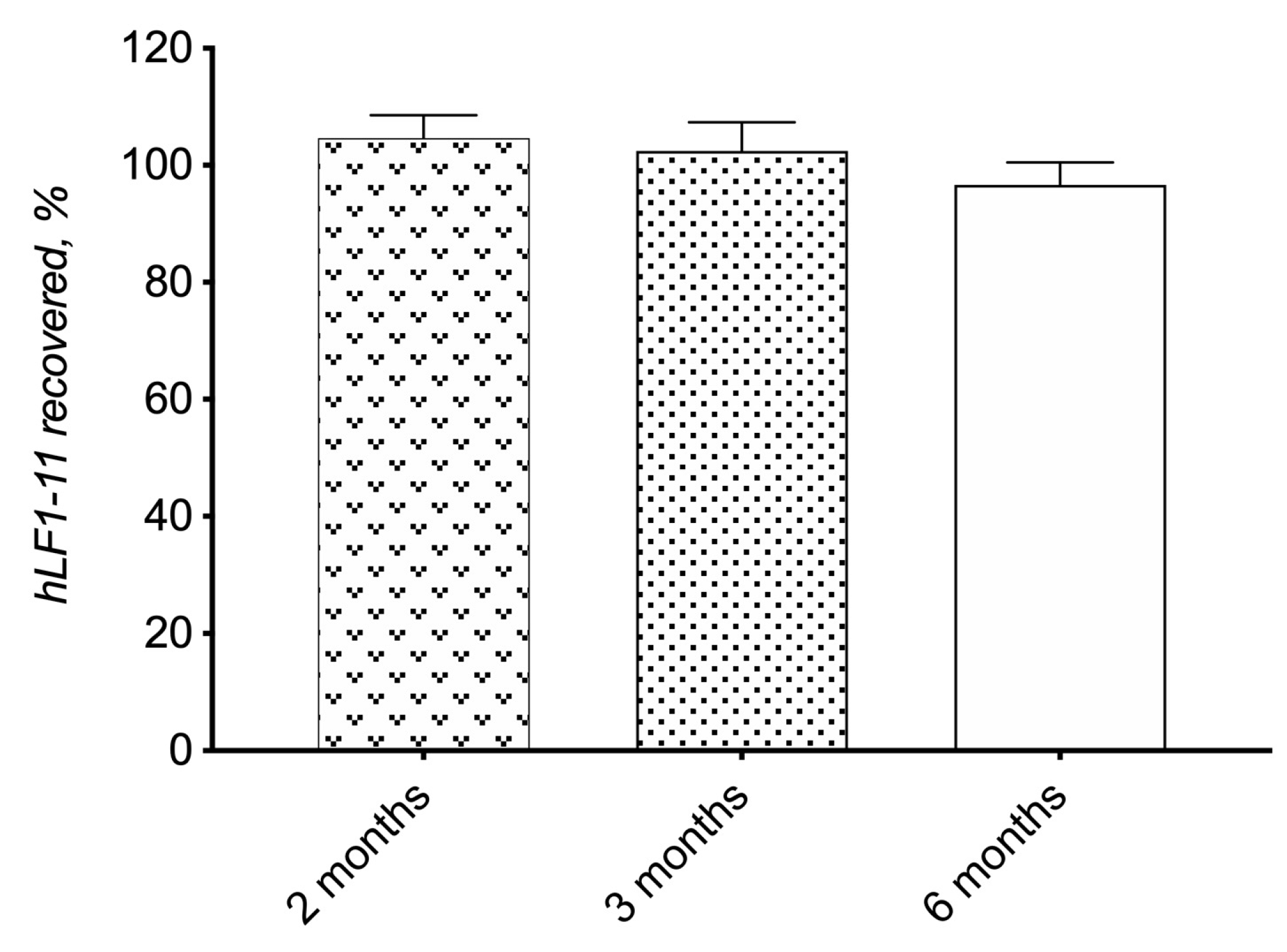
3.2.1. Preparation and Physicochemical Characterization
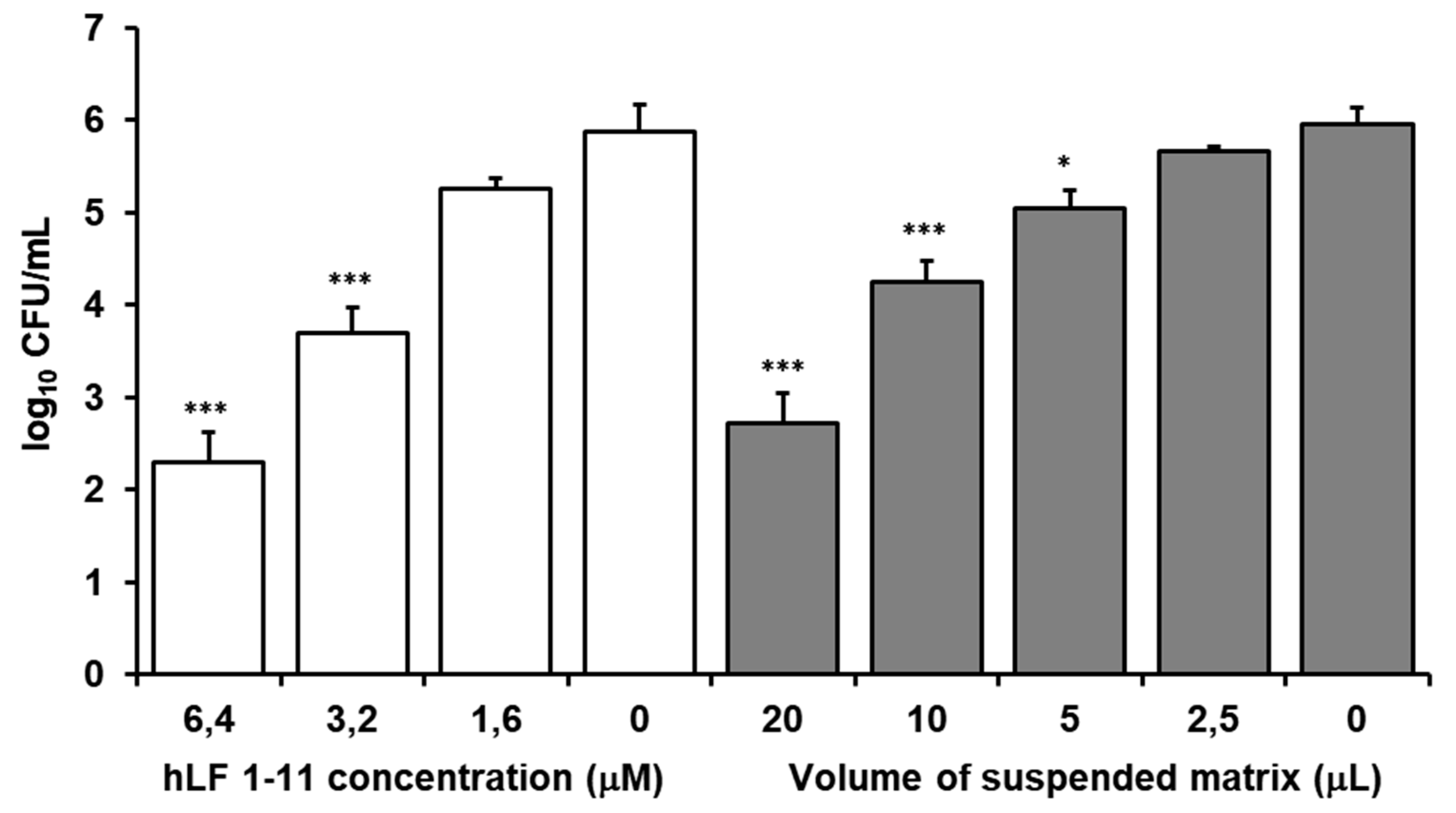
3.2.2. In Vitro Susceptibility Testing
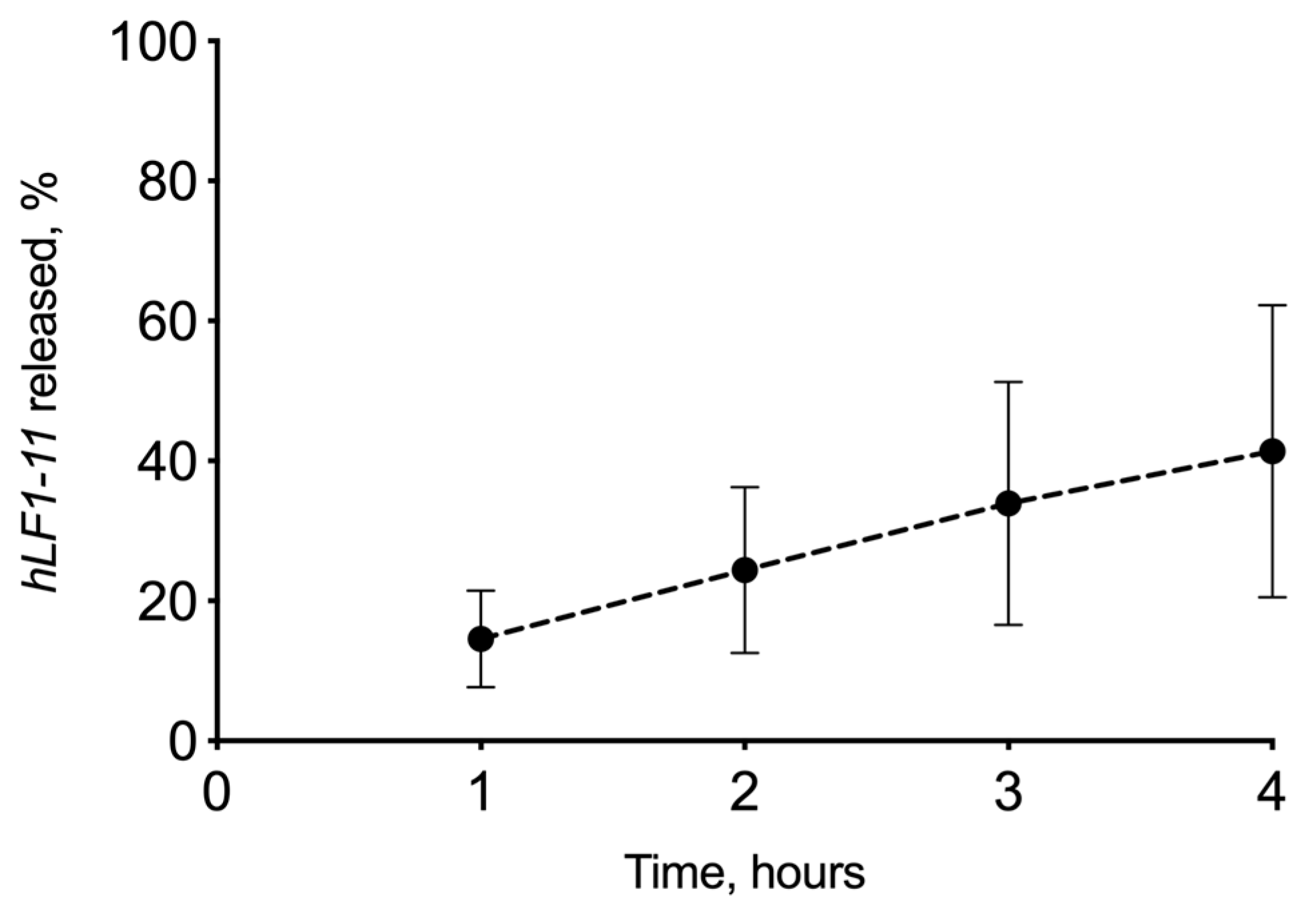
3.2.3. In Vitro Drug Release Experiment
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Austin, A.; Lietman, T.; Rose-Nussbaumer, J. Update on the Management of Infectious Keratitis. Ophthalmology 2017, 124, 1678–1689. [Google Scholar] [CrossRef] [PubMed]
- Garbutcheon-Singh, K.B.; Carnt, N.; Pattamatta, U.; Samarawickrama, C.; White, A.; Calder, V. A Review of the Cytokine IL-17 in Ocular Surface and Corneal Disease. Curr. Eye Res. 2019, 44, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Ong, H.S.; Corbett, M.C. Corneal infections in the 21st century. Postgrad. Med. J. 2015, 91, 565–571. [Google Scholar] [CrossRef] [PubMed]
- Srigyan, D.; Gupta, M.; Ahsan, S.; Behera, H. Infectious Keratitis: An Immediate Cause of Concern. Ophthalmol. Res. Int. J. 2017, 7, 1–6. [Google Scholar] [CrossRef]
- Ung, L.; Bispo, P.J.; Shanbhag, S.S.; Gilmore, M.S.; Chodosh, J. The Persistent Dilemma of Microbial Keratitis: Global Burden, Diagnosis, and Antimicrobial Resistance. Surv. Ophthalmol. 2018, 64, 255–271. [Google Scholar] [CrossRef]
- Solanki, S.; Rathi, M.; Khanduja, S.; Dhull, C.; Sachdeva, S.; Phogat, J. Recent trends: Medical management of infectious keratitis. Oman J. Ophthalmol. 2015, 8, 83–85. [Google Scholar] [CrossRef]
- Lombardi, L.; Falanga, A.; Del Genio, V.; Galdiero, S. A New Hope: Self-Assembling Peptides with Antimicrobial Activity. Pharmaceutics 2019, 11, 166. [Google Scholar] [CrossRef]
- Levay, P.F.; Viljoen, M. Lactoferrin: A general review. Haematologica 1995, 80, 252–267. [Google Scholar]
- Lonnerdal, B.; Iyer, S. Lactoferrin: Molecular structure and bio-logical function. Annu. Rev. Nutr. 1995, 15, 93–110. [Google Scholar] [CrossRef]
- Kijlstra, A.; Jeurissen, S.H.; Koning, K.M. Lactoferrin levels in normal human tears. Br. J. Ophthalmol. 1983, 67, 199–202. [Google Scholar] [CrossRef]
- Masson, P.L.; Heremas, J.F.; Dive, C. An iron-binding protein common to many external secretions. Clin. Chim. Acta 1966, 14, 735–739. [Google Scholar] [CrossRef]
- Lupetti, A.; Paulusma-Annema, A.; Welling, M.M.; Senesi, S.; Van Dissel, J.T.; Nibbering, P.H. Candidacidal activities of human lactoferrin peptides derived from the N terminus. Antimicrob. Agents Chemother. 2000, 44, 3257–3263. [Google Scholar] [CrossRef] [PubMed]
- Lupetti, A.; Paulusma-Annema, A.; Welling, M.M.; Dogterom-Ballering, H.; Brouwer, C.P.J.M.; Senesi, S.; Van Dissel, J.T.; Nibbering, P.H. Synergistic activity of the N-terminal peptide of human lactoferrin and fluconazole against Candida species. Antimicrob. Agents Chemother. 2003, 47, 262–267. [Google Scholar] [CrossRef]
- Lupetti, A.; Brouwer, C.P.J.M.; Bogaards, S.J.P.; Welling, M.M.; de Heer, E.; Campa, M.; van Dissel, J.T.; Friesen, R.H.E.; Nibbering, P.H. Human Lactoferrin-Derived Peptide’s Antifungal Activities against Disseminated Candida albicans Infection. J. Infect. Dis. 2007, 196, 1416–1424. [Google Scholar] [CrossRef] [PubMed]
- Morici, P.; Fais, R.; Rizzato, C.; Tavanti, A.; Lupetti, A. Inhibition of Candida albicans Biofilm Formation by the Synthetic Lactoferricin Derived Peptide hLF1-11. PLoS ONE 2016, 11, e0167470. [Google Scholar] [CrossRef]
- Morici, P.; Florio, W.; Rizzato, C.; Ghelardi, E.; Tavanti, A.; Rossolini, G.M.; Lupetti, A. Synergistic activity of synthetic N-terminal peptide of human lactoferrin in combination with various antibiotics against carbapenem-resistant Klebsiella pneumoniae strains. Eur. J. Clin. Microbiol. Infect. Dis. 2017, 36, 1739–1748. [Google Scholar] [CrossRef]
- Lupetti, A.; Welling, M.M.; Pauwels, E.K.; Nibbering, P.H. Radiolabelled antimicrobial peptides for infection detection. Lancet Infect. Dis. 2003, 3, 223–229. [Google Scholar] [CrossRef]
- Lupetti, A.; van Dissel, J.T.; Brouwer, C.P.J.M.; Nibbering, P.H. Human antimicrobial peptides’ antifungal activity against Aspergillus fumigatus. Eur. J. Clin. Microbiol. Infect. Dis. 2008, 27, 1125–1129. [Google Scholar] [CrossRef]
- Nibbering, P.H.; Ravensbergen, E.; Welling, M.M.; Van Berkel, L.A.; Van Berkel, P.H.; Pauwels, E.K.; Nuijens, J.H. Human lactoferrin and peptides derived from its N terminus are highly effective against infection with antibiotic-resistant bacteria. Infect. Immun. 2001, 69, 1469–1476. [Google Scholar] [CrossRef]
- Dijkshoorn, L.; Brouwer, C.P.; Bogaards, S.J.; Nemecm, A.; van den Broek, P.J.; Nibbering, P.H. The synthetic N-terminal peptide of human lactoferrin, hLF(1-11), is highly effective against experimental infection caused by multidrug-resistant Acinetobacter baumannii. Antimicrob. Agents Chemother. 2004, 48, 4919–4921. [Google Scholar] [CrossRef]
- Tanida, T.; Rao, F.; Hamada, T.; Ueta, E.; Osaki, T. Lactoferrin peptide increases the survival of Candida albicans-inoculated mice by upregulating neutrophil and macrophage functions, especially in combination with amphotericin B and granulocyte-macrophage colony-stimulating factor. Infect. Immun. 2001, 69, 3883–3890. [Google Scholar] [CrossRef] [PubMed]
- Pizzo, E.; Cafaro, V.; Di Donato, A.; Notomista, E. Cryptic Antimicrobial Peptides: Identification Methods and Current Knowledge of their Immunomodulatory Properties. Curr. Pharm. Des. 2018, 24, 1054–1066. [Google Scholar] [CrossRef] [PubMed]
- Dai, M.; Pan, P.; Li, H.; Liu, S.; Zhang, L.; Song, C.; Li, Y.; Li, Q.; Mao, Z.; Long, Y.; et al. The antimicrobial cathelicidin peptide hlF(1-11) attenuates alveolar macrophage pyroptosis induced by Acinetobacter baumannii in vivo. Exp. Cell Res. 2018, 364, 95–103. [Google Scholar] [CrossRef] [PubMed]
- Brouwer, C.P.J.M.; Rahman, M.; Welling, M.M. Discovery and development of a synthetic peptide derived from lactoferrin for clinical use. Peptides 2011, 32, 1953–1963. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, K.E.; Carter, D.A. The antifungal activity of lactoferrin and its derived peptides: Mechanisms of action and synergy with drugs against fungal pathogens. Front. Microbiol. 2017, 8, 1–10. [Google Scholar] [CrossRef]
- Brouwer, C.P.J.M.; Roscini, L.; Cardinali, G.; Corte, L.; Pierantoni, D.C.; Robert, V.; Rahman, M.; Welling, M.M. Structure-Activity Relationship Study of Synthetic Variants Derived from the Highly Potent Human Antimicrobial Peptide hLF(1-11). Cohesive J. Microbiol. Infect. Dis. 2018, 1, 1–19. [Google Scholar] [CrossRef]
- Van der Does, A.M.; Bogaards, S.J.P.; Jonk, L.; Wulferink, M.; Velders, M.P.; Nibbering, P.H. The human lactoferrin-derived peptide hLF1-11 primes monocytes for an enhanced TLR-mediated immune response. BioMetals 2010, 23, 493–505. [Google Scholar] [CrossRef]
- Fornili, S.L.; Pizzi, R.; Rebeccani, D. Conformational Analysis of a Synthetic Antimicrobial Peptide in Water and Membrane-Mimicking Solvents: A Molecular Dynamics Simulation Study. Int. J. Pept. Res. Ther. 2010, 16, 223–231. [Google Scholar] [CrossRef]
- Mandal, A.; Pal, D.; Agrahari, V.; Trinh, H.M.; Joseph, M.; Mitra, A.K. Ocular delivery of proteins and peptides: Challenges and novel formulation approaches. Adv. Drug Deliv. Rev. 2018, 126, 67–95. [Google Scholar] [CrossRef]
- Bispo, P.J.; Hofling-Lima, A.L.; Pignatari, A.C. Characterization of ocular methicillin-resistant Staphylococcus epidermidis isolates belonging predominantly to clonal complex 2 subcluster II. J. Clin. Microbiol. 2014, 52, 1412–1417. [Google Scholar] [CrossRef]
- Burgalassi, S.; Monti, D.; Nicosia, N.; Tampucci, S.; Terreni, E.; Vento, A.; Chetoni, P. Freeze-dried matrices for ocular administration of bevacizumab: A comparison between subconjunctival and intravitreal administration in rabbits. Drug Deliv. Transl. Res. 2018, 8, 461–472. [Google Scholar] [CrossRef] [PubMed]
- Burgalassi, S.; Monti, D.; Tampucci, S.; Chetoni, P. In vitro evaluation of some parameters involved in mucoadhesion of aqueous polymeric dispersions. Pharm. Dev. Technol. 2015, 20, 927–934. [Google Scholar] [CrossRef] [PubMed]
- Matejtschuk, P.; Phillips, P.; Andersen, M. Freeze-Drying of Biological Standards. In Lyophilization of Pharmaceutical and Biological Products, 2nd ed.; Rey, L., May, J.C., Eds.; CRC Press Inc.: Boca Raton, FL, USA, 2004. [Google Scholar] [CrossRef]
- Oetjen, G.-W. Industrial Freeze-Drying for Pharmaceutical Applications. In Freeze-Drying/Lyophilization of Pharmaceutical and Biological Products, 2nd ed.; CRC Press Inc. Boca Raton, FL, USA, Rey, L., May, J.C., Eds.; 2004; Volume 137, pp. 425–475. [Google Scholar] [CrossRef]
- Kumar, K.N.; Mallik, S.; Sarkar, K. Role of freeze-drying in the presence of mannitol on the echogenicity of echogenic liposomes. J. Acoust. Soc. Am. 2017, 142, 3670–3676. [Google Scholar] [CrossRef] [PubMed]
- Muzzio, C.R.; Dini, N.G.; Simionato, L.D. Determination of moisture content in lyophilized mannitol through intact glass vials using NIR micro-spectrometers. Braz. J. Pharm. Sci. 2011, 47, 289–297. [Google Scholar] [CrossRef]
- Mirfakhraei, Y.; Faghihi, H.; Zade, A.H.M.; Darabi, M.; Vatanara, A. Optimization of Stable IgG Formulation Containing Amino Acids and Trehalose During Freeze-Drying and After Storage: A Central Composite Design. AAPS PharmSciTech 2019, 20, 154. [Google Scholar] [CrossRef]
- Patel, S.M.; Nail, S.L.; Pikal, M.J.; Geidobler, R.; Winter, G.; Hawe, A.; Davagnino, J.; Rambhatla Gupta, S. Lyophilized Drug Product Cake Appearance: What Is Acceptable? J. Pharm. Sci. 2017, 106, 1706–1721. [Google Scholar] [CrossRef]
- Cowman, M.K.; Schmidt, T.A.; Raghavan, P.; Stecco, A. Viscoelastic Properties of Hyaluronan in Physiological Conditions. F1000 Res. 2015, 4, 622. [Google Scholar] [CrossRef]
- Wedlock, D.J.; Phillips, G.O.; Davies, A.; Gormally, J.; Wyn-Jones, E. Depolymerization of sodium hyaluronate during freeze drying. Int. J. Biol. Macromol. 1983, 5, 186–188. [Google Scholar] [CrossRef]
- Mensink, M.A.; Frijlink, H.W.; van der Voort Maarschalk, K.; Hinrichs, W.L.J. How sugars protect proteins in the solid state and during drying (review): Mechanisms of stabilization in relation to stress conditions. Eur. J. Pharm. Biopharm. 2017, 114, 288–295. [Google Scholar] [CrossRef]
- Bouchard, A.; Hofland, G.W.; Witkamp, G.J. Properties of sugar, polyol, and polysaccharide water-ethanol solutions. J. Chem. Eng. Data 2007, 52, 1838–1842. [Google Scholar] [CrossRef]
- Cassanelli, M.; Norton, I.; Mills, T. Interaction of Mannitol and Sucrose with Gellan Gum in Freeze-Dried Gel Systems. Food Biophys. 2018, 13, 304–315. [Google Scholar] [CrossRef] [PubMed]
- Jaipal, A.; Pandey, M.M.; Charde, S.Y.; Raut, P.P.; Prasanth, K.V.; Prasad, R.G. Effect of HPMC and mannitol on drug release and bioadhesion behavior of buccal discs of buspirone hydrochloride: In-vitro and in-vivo pharmacokinetic studies. Saudi Pharm. J. 2015, 23, 315–326. [Google Scholar] [CrossRef]
- Batool, A.; Arshad, R.; Razzaq, S.; Nousheen, K.; Kiani, H.M.; Shahnaz, G. Formulation and Evaluation of Hyaluronic Acid-based Mucoadhesive Self Nanoemulsifying Drug Delivery System (SNEDDS) of Tamoxifen for targeting breast cancer. Int. J. Biol. Macromol. 2020, 152, 503–515. [Google Scholar] [CrossRef]
- Verhoeven, N.; Neoh, T.L.; Furuta, T.; Yamamoto, C.; Ohashi, T.; Yoshii, H. Characteristics of dehydration kinetics of dihydrate trehalose to its anhydrous form in ethanol by DSC. Food Chem. 2012, 132, 1638–1643. [Google Scholar] [CrossRef]
- Imamura, K.; Yokoyama, T.; Fukushima, A.; Kinuhata, M.; Nakanishi, K. Water sorption, glass transition, and protein-stabilizing behavior of an amorphous sucrose matrix combined with various materials. J. Pharm. Sci. 2010, 99, 4669–4677. [Google Scholar] [CrossRef] [PubMed]
- Kristensen, K.; Henriksen, J.R.; Andresen, T.L. Adsorption of cationic peptides to solid surfaces of glass and plastic. PLoS ONE 2015, 10, e0122419. [Google Scholar] [CrossRef] [PubMed]
- Feng, M.; Morales, A.B.; Poot, A.; Beugeling, T.; Bantjes, A. Effects of Tween 20 on the desorption of proteins from polymer surfaces. J. Biomater. Sci. Polym. Ed. 1995, 7, 415–424. [Google Scholar] [CrossRef]
- Nordström, M.; Malmsten, M. Delivery systems for antimicrobial peptides. Adv. Colloid Interface Sci. 2017, 242, 17–34. [Google Scholar] [CrossRef] [PubMed]
| Formulation | HPMC | HA | Mannitol | Trehalose | VA |
|---|---|---|---|---|---|
| HPMC/VAfd | 24.92 | - | 74.75 | - | 0.33 |
| HPMC/HA/VAfd | 22.43 | 2.49 | 74.75 | - | 0.33 |
| HPMC/T2/VAfd | 24.92 | - | 24.92 | 49.83 | 0.33 |
| HPMC/T2/HA/VAfd | 22.43 | 2.49 | 24.92 | 49.83 | 0.33 |
| Drug | Detector | Column | Analysis | Mobile Phase | Flow Rate, mL/min | λ, nm | LOQ 1, μg/mL | RT, min |
|---|---|---|---|---|---|---|---|---|
| VA | UV | Kinetex Phenomenex® C18 100 Å (5 μm; 150 × 4.6 mm) | Isocratic | NaH2PO4/Na2HPO4 (40:60, pH = 6.95, 50 mM) | 1.0 | 210 | 0.025 | 5.0 |
| hLF 1-11 | DAD | Phenomenex® Aeris widepore XB-C8 200Å (3.6 μm; 150 × 4.6 mm) | Linear gradientfrom 5% to 70% solvent A over 25 min | Solvent A (0.05% TFA in acetonitrile); Solvent B (0.05% of TFA in water) | 1.0 | 220 | 1.0 | 5.4 |
| Matrix | Theoretical Weight, mg | Batch 1, mg | Batch 2, mg | CV 1 |
|---|---|---|---|---|
| HPMC/VAfd | 6.00 | 5.77 ± 0.03 | 5.85 ± 0.02 | 1.37 |
| HPMC/HA/VAfd | 6.00 | 5.80 ± 0.02 | 5.77 ± 0.04 | 1.44 |
| HPMC/T2/VAfd | 6.00 | 5.65 ± 0.09 | 5.67 ± 0.02 | 2.04 |
| HPMC/T2/HA/VAfd | 6.00 | 5.63 ± 0.02 | 5.73 ± 0.04 | 1.65 |
| Viscosityapp (mPa*s) | ||
|---|---|---|
| Polymeric Dispersion | Before | After |
| HPMC/VAdisp | 227.7 ± 11.82 | 212.9 ± 10.27 |
| HPMC/HA/VAdisp | 528.3 ± 2.00 | 351.7 ± 23.76 * |
| HPMC/T2/VAdisp | 265.2 ± 10.66 | 275.1 ± 4.42 |
| HPMC/T2/HA/VAdisp | 321.2 ± 5.53 | 274.8 ± 11.13 |
| Matrix | Recovery (%) ± SE |
|---|---|
| HPMC/VA | 92.46 ± 5.09 |
| HPMC/HA/VA | 91.39 ± 5.88 |
| HPMC/T2/VA | 96.02 ± 2.21 |
| HPMC/T2/HA/VA | 100.2 ± 0.16 |
| Matrix | Measured Concentration (μg/mL) | ||
|---|---|---|---|
| Before Freeze-Drying | After Freeze-Drying | Recovery (%) | |
| HPMC/HA/hLFfd | 6.96 ± 0.38 | 4.77 ± 0.31 | 68.38 ± 4.43 |
| HPMC/T2/HA/hLFfd | 13.24 ± 1.85 | 11.99 ± 0.93 | 86.74 ± 1.83 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Terreni, E.; Burgalassi, S.; Chetoni, P.; Tampucci, S.; Zucchetti, E.; Fais, R.; Ghelardi, E.; Lupetti, A.; Monti, D. Development and Characterization of a Novel Peptide-Loaded Antimicrobial Ocular Insert. Biomolecules 2020, 10, 664. https://doi.org/10.3390/biom10050664
Terreni E, Burgalassi S, Chetoni P, Tampucci S, Zucchetti E, Fais R, Ghelardi E, Lupetti A, Monti D. Development and Characterization of a Novel Peptide-Loaded Antimicrobial Ocular Insert. Biomolecules. 2020; 10(5):664. https://doi.org/10.3390/biom10050664
Chicago/Turabian StyleTerreni, Eleonora, Susi Burgalassi, Patrizia Chetoni, Silvia Tampucci, Erica Zucchetti, Roberta Fais, Emilia Ghelardi, Antonella Lupetti, and Daniela Monti. 2020. "Development and Characterization of a Novel Peptide-Loaded Antimicrobial Ocular Insert" Biomolecules 10, no. 5: 664. https://doi.org/10.3390/biom10050664
APA StyleTerreni, E., Burgalassi, S., Chetoni, P., Tampucci, S., Zucchetti, E., Fais, R., Ghelardi, E., Lupetti, A., & Monti, D. (2020). Development and Characterization of a Novel Peptide-Loaded Antimicrobial Ocular Insert. Biomolecules, 10(5), 664. https://doi.org/10.3390/biom10050664










