Discovery of the First Neurotransmitter Receptor: The Acetylcholine Nicotinic Receptor
Abstract
1. The Concept of Pharmacological Receptor
2. The Pioneers in the Unsuccessful Attempts to Identify the Nicotinic Acetylcholine Receptor
3. The Techniques or Models Applied and the Reasons for the Failures
4. The Strategies which Lead to the Identification of the nAChR
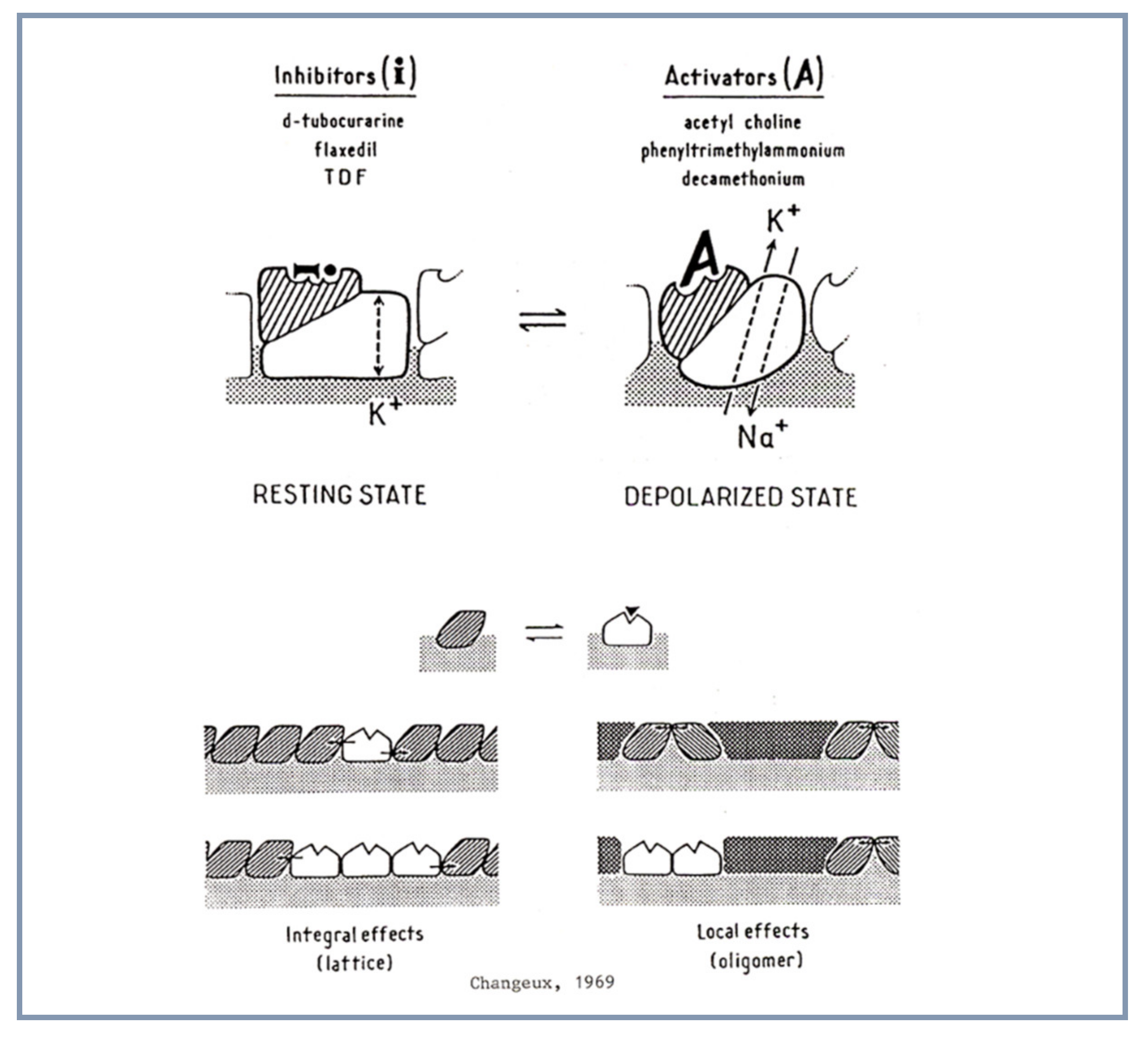
4.1. Allosteric Protein
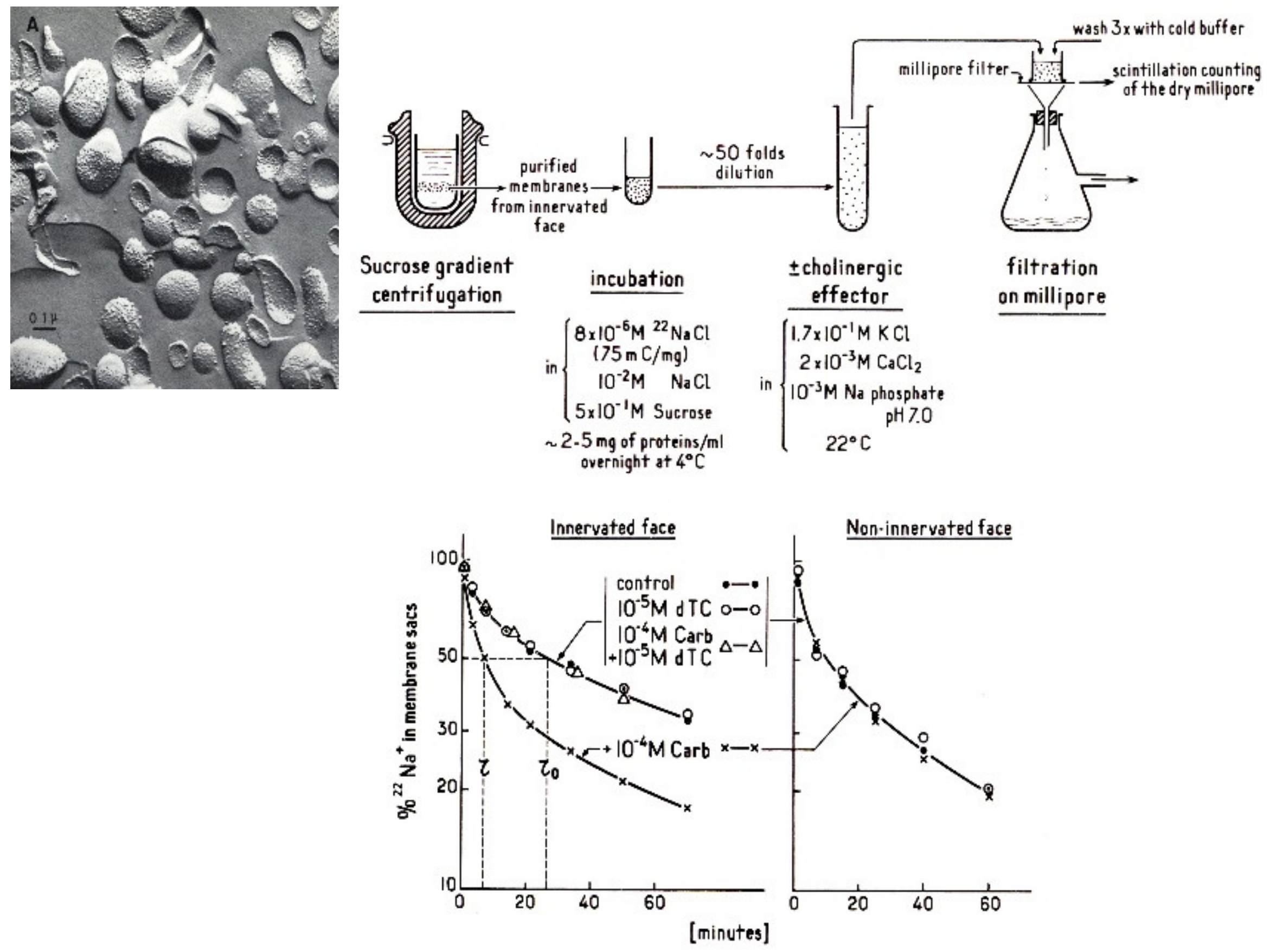
4.2. Electric Organ: the Electroplaque and the Excitable Microsacs
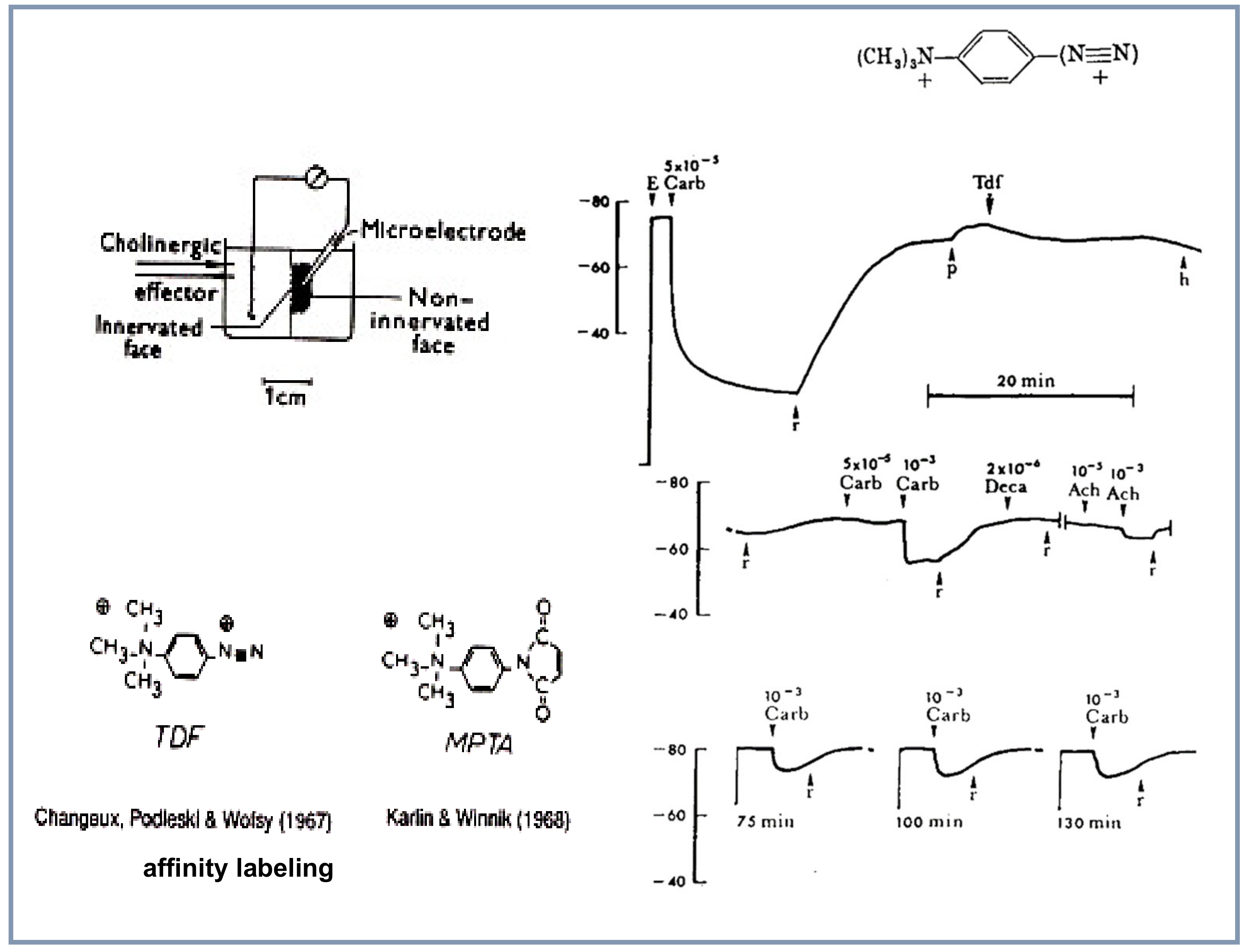
4.3. Search for Specific Ligands: Affinity Labeling
4.4. Microsacs and Equilibrium Dialysis
- (1)
- The nicotinic agonist decamethonium was used as the radioactive ligand;
- (2)
- Conditions of solubilization of the electric organ membranes were found with the detergent deoxycholate, to gently extract the membrane proteins without denaturing them;
- (3)
- Concentrations of proteins in the dialysis bag were made high enough to expect a measurable displacement of the ligand by a population of sites in the range of that of the acetylcholinesterase active sites;
- (4)
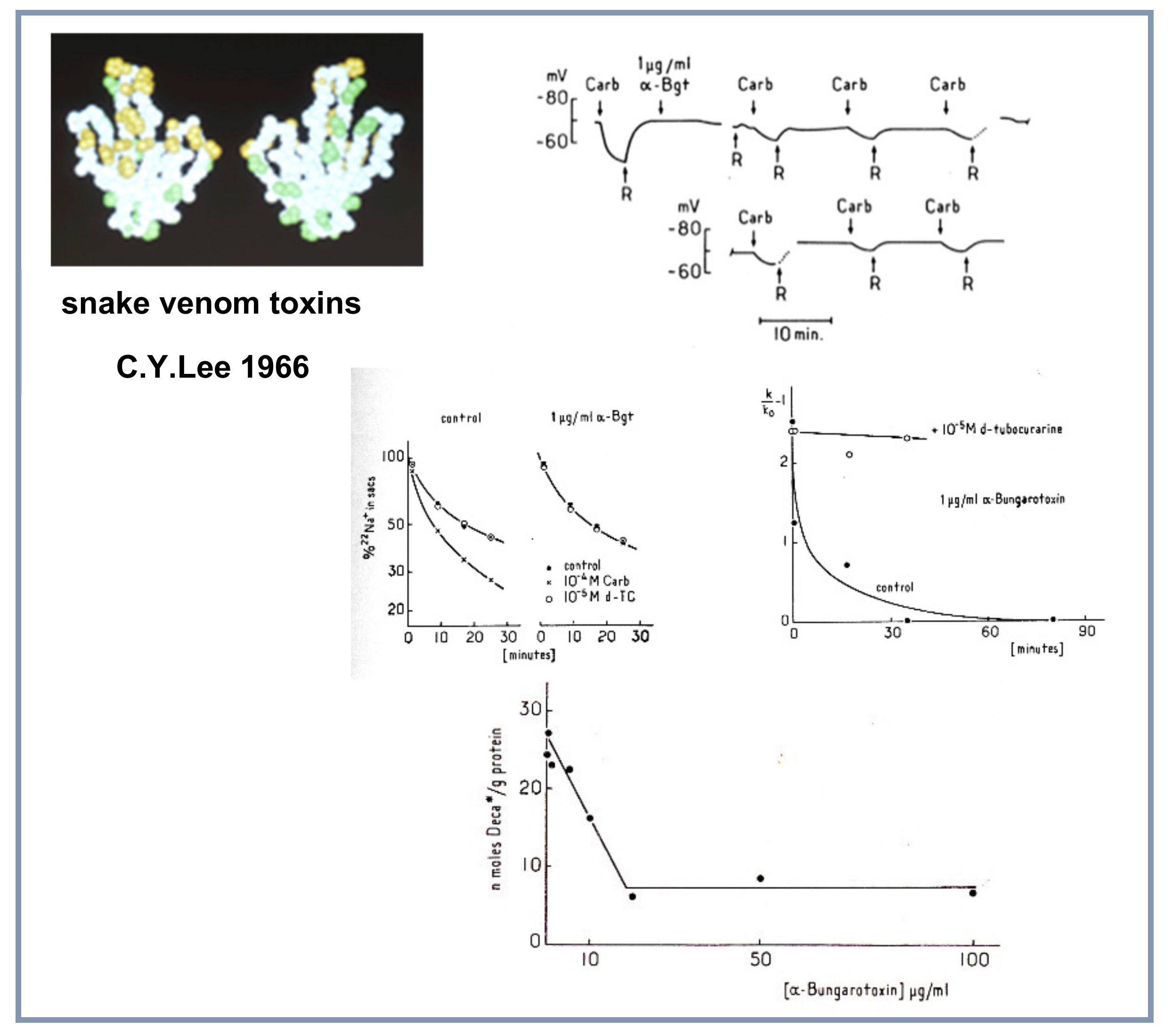
4.5. Snake Venom Toxins
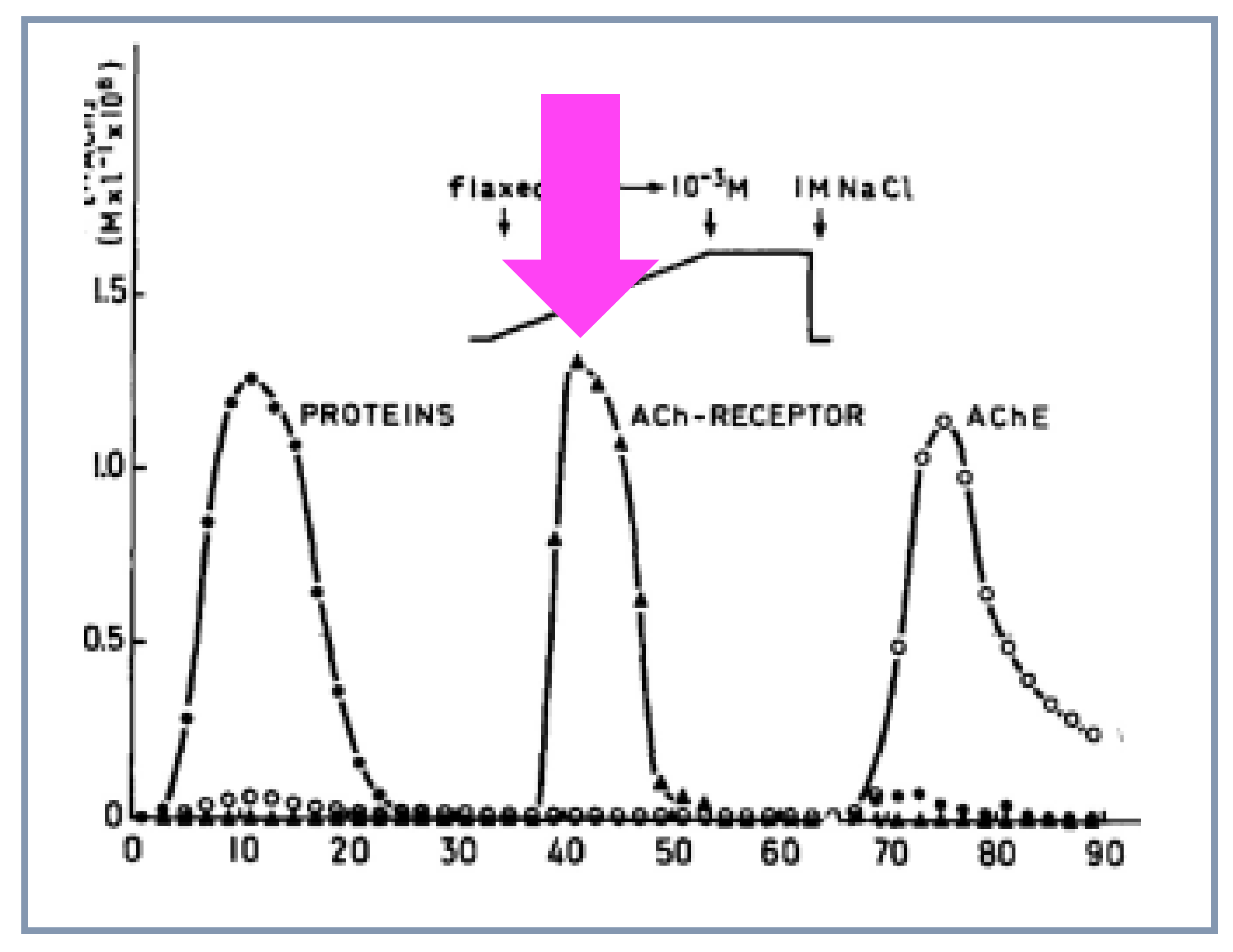
4.6. Purification
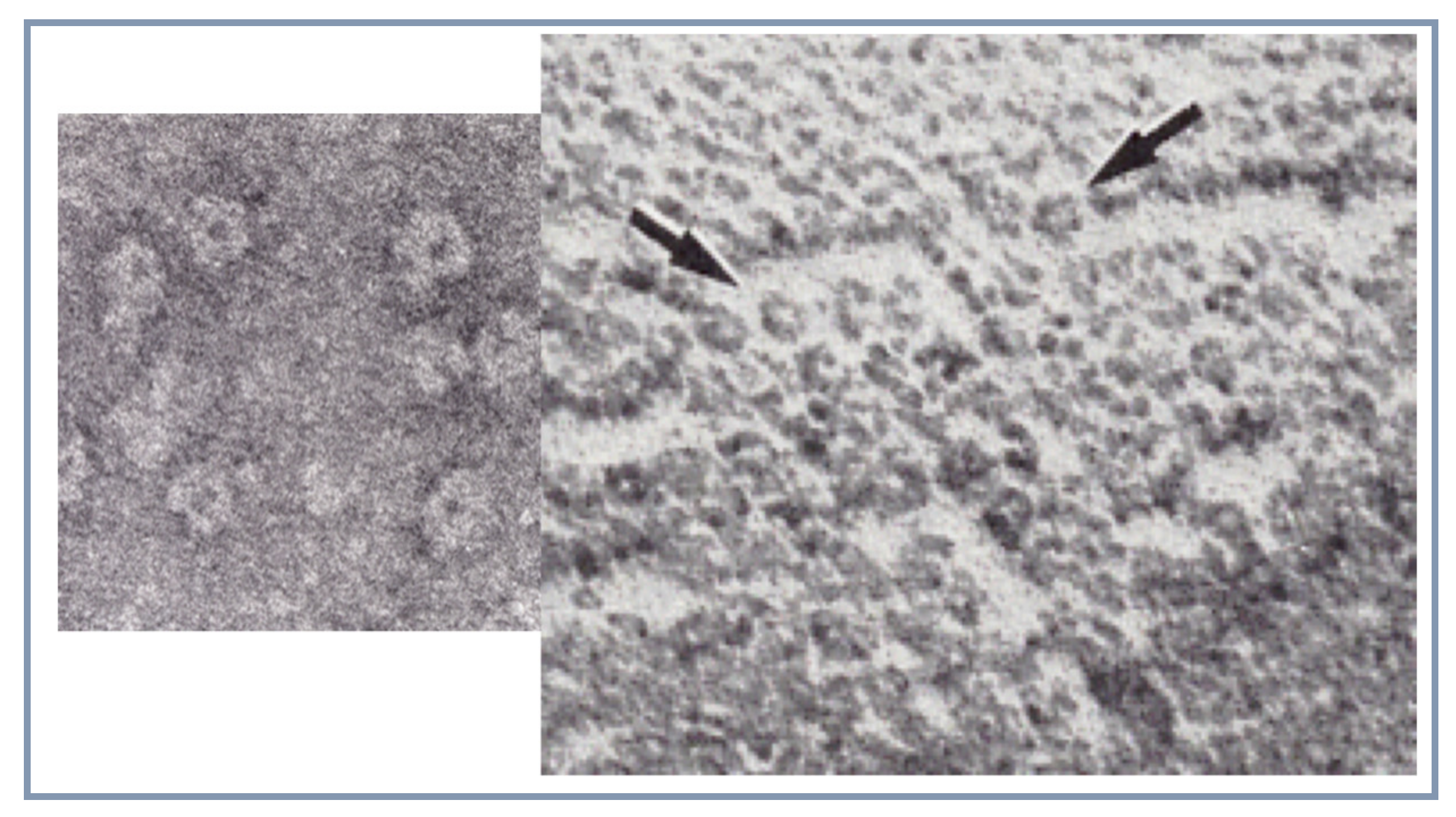
5. First Visualisation of the Receptor Protein Structure by Electron Microscopy
6. Experimental Myasthenia Gravis with Purified nAChR
7. Primary and Quaternary Structure
8. The Acetylcholine Nicotinic Receptors to-Day: Biological Functions, Relationships with Brain Diseases and Pharmacology
9. International Recognition and Importance for the History of Neuroscience
Funding
Acknowledgments
Conflicts of Interest
References
- Bernard, C. Leçons Sur Les Effets des Substances Toxiques et Médicamenteuses; Baillière: Paris, France, 1857. [Google Scholar]
- Ehrlich, P. Das Sauerstoff Bedurfnis des Organismus, Eine Farbenanalytische Studie; Hirschwald: Berlin, Germany, 1865. [Google Scholar]
- Langley, J.N. On the contraction of muscle, chiefly in relation to the presence of “receptive” substances. Physiology 1907, 36, 347–384. [Google Scholar] [CrossRef] [PubMed]
- Chagas Filho, C.; Penna-Franca, E.; Hassón-Voloch, A. Studies of the mechanism of curarization. Ann. N. Y. Acad. Sci. 1957, 81, 345–357. [Google Scholar] [CrossRef] [PubMed]
- De Robertis, E.; Sabatini, D.D. Submicroscopic analysis of the secretory process in the adrenal medulla. Fed. Proc. 1960, 19, 70–78. [Google Scholar] [PubMed]
- Nachmansohn, D. The Chemical and Molecular Basis of Nerve Activity; Academic Press: New York, NY, USA, 1959. [Google Scholar]
- Ehrenpreis, S. Interaction of curare and related substances with acetylcholine receptor-like protein. Science 1959, 129, 1613–1614. [Google Scholar] [CrossRef] [PubMed]
- Barrantes, F.J.; Changeux, J.-P.; Lunt, G.O.; Sobel, A. Differences between the detergent extracted acetylcholine receptor and the cholinergie proteolipids. Nature 1975, 256, 325–327. [Google Scholar] [CrossRef] [PubMed]
- Del Castillo, J.; Katz, B. A study of curare action with an electrical micro-method. Proc. R. Soc. Lond. 1957, 146, 339–356. [Google Scholar] [PubMed]
- Changeux, J.-P. The feedback control mechanism of biosynthetic L-threonine deaminase by L-isoleucine. Cold Spring Harb. Symp. Quant. Biol. 1961, 26, 313–318. [Google Scholar] [CrossRef] [PubMed]
- Monod, J.; Wyman, J.; Changeux, J.-P. On the nature of allosteric transitions: A plausible model. J. Mol. Biol. 1965, 12, 88–118. [Google Scholar] [CrossRef]
- Gerhart, J.C.; Pardee, A.B. The enzymology of controi by feedback inhibition. J. Biol. Chem. 1962, 237, 891–896. [Google Scholar] [PubMed]
- Gerhart, J.C.; Pardee, A.B. Aspartate transcarbamylase, an enzyme designed for feedback inhibition. Fed. Proc. 1964, 23, 727–735. [Google Scholar]
- Perutz, M.F. Mechanisms of cooperativity and allosteric regulation in proteins. Q. Rev. Biophys. 1989, 22, 139–237. [Google Scholar] [CrossRef] [PubMed]
- Changeux, J.-P. Sur les propriétés allostériques de la L·thréonine désaminase de biosynthèse. VI. Discussion générale. Bull. Soc. Chim. Biol. 1965, 47, 281–300. [Google Scholar] [PubMed]
- Changeux, J.-P.; Thiéry, J.P.; Tung, Y.; Kittel, C. On the cooperativity of biological membranes. Proc. Natl. Acad. Sci. USA 1967, 57, 335–341. [Google Scholar] [CrossRef]
- Karlin, A. On the application of a plausible model of allosteric proteins to the receptor for acetylcholine. J. Theor. Biol. 1967, 16, 306–320. [Google Scholar] [CrossRef]
- Cecchini, M.; Changeux, J.P. The nicotinic acetylcholine receptor and its prokaryotic homologues: Structure, conformational transitions & allosteric modulation. Neuropharmacology 2015, 96 Pt B, 137–149. [Google Scholar]
- Schoffeniels, E.; Nachmansohn, D. An isolated single electroplax preparation. 1. New data on the effect of acetylcholine and related compounds. Biochim. Biophys. Acta 1957, 26, 1–15. [Google Scholar] [CrossRef]
- Changeux, J.-P.; Gautron, J.; Israe, L.M.; Podleski, T. Separation of excitable membranes from the electric organ of Electrophorus electricus. Comptes Rendus Hebd. Seances Acad. Sci. D 1969, 269, 1788–1791. [Google Scholar]
- Kasai, M.; Changeux, J.-P. Demonstration of the excitation by cholinergic agonists from fractions of purified membranes in vitro. Comptes Rendus Hebd. Seances Acad. Sci. D 1970, 270, 1400–1403. [Google Scholar]
- Changeux, J.-P. Remarks on the symmetry and cooperative properties of biological membranes. In Symmetry and Functions at Biological Systems at the Macromolecular Level; Nobel Symposium N° 11; Engstrom, A., Stranberg, B., Eds.; Wiley Inter Science Division: New York, NY, USA, 1969; pp. 235–256. [Google Scholar]
- Changeux, J.-P. Responses of acetylcholinesterase from Torpedo marmorata to salts and curarizing drugs. Mol. Pharmacol. 1966, 2, 369–392. [Google Scholar]
- Singer, S.J.; Doolittle, R.F. Antibody active sites and immunoglobulin molecules. Science 1966, 153, 13–25. [Google Scholar] [CrossRef]
- Gill, E.W.; Rang, H.P. An alkylating derivative of benzilylcholine with specific and long-lasting parasympatholytic activity. Mol. Pharmacol. 1966, 2, 284–297. [Google Scholar] [PubMed]
- Fenton, J.W.; Singer, S.J. Affinity labeling of antibodies to the p-azophenyltrimethylammonium hapten and a structural relationship among antibody active sites of different specificities. Biochem. Biophys. Res. Commun. 1965, 20, 315–320. [Google Scholar] [CrossRef]
- Changeux, J.P.; Podleski, T.R.; Wofsy, L. Affinity labeling of the acetylcholine receptor. Proc. Natl. Acad. Sci. USA 1967, 58, 2063–2070. [Google Scholar] [CrossRef] [PubMed]
- Weiland, G.; Frisman, D.; Taylor, P. Affinity labeling of the subunits of the membrane associated cholinergie receptor. Mol. Pharmacol. 1979, 15, 213–226. [Google Scholar]
- Dennis, M.; Giraudat, J.; Kotzyba-Hibert, F.; Goeldner, M.; Hirth, C.; Chang, J.Y.; Lazure, C.; Chretién, M.; Changeux, J.P. Amino acids of the Torpedo marmorata acetylcholine receptor α subunit labeled by a photoaffinity ligand for the acetylcholine binding site. Biochemistry 1988, 27, 2346–2357. [Google Scholar] [CrossRef]
- Karlin, A.; Winnik, M. Reduction and specific alkylation of the receptor for acetylcholine. Proc. Natl. Acad. Sci. USA 1968, 60, 668–674. [Google Scholar] [CrossRef]
- Karlin, A.; Bartels, E. Effects of blocking sulfhydryl groups and of reducing disulfide bonds on the acetylcholine-activated permeability systems of the electroplax. Biochim. Biophys. Acta 1966, 126, 525–535. [Google Scholar] [CrossRef]
- Karlin, A.; Prives, J.; Deal, W.; Winnik, M. Counting acetylcholine receptors in the electroplax. In Molecular Properties of Drug Receptors, Ciba Foundation Symposium; J. & A. Churchill, Ltd: London, UK, 1970; pp. 247–261. [Google Scholar]
- Weill, C.L.; McNamee, M.G.; Karlin, A. Affinity labeling of purified acetylcholine receptor from Torpedo califomica. Biochem. Biophys. Res. Commun. 1974, 61, 997–1003. [Google Scholar] [CrossRef]
- Gilbert, W.; Muller-Hill, B. Isolation of the lac-repressor. Proc. Natl. Acad. Sci. USA 1966, 56, 1891–1898. [Google Scholar] [CrossRef]
- Changeux, J.-P.; Kasai, M.; Huchet, M.; Meunier, J.C. Extraction from electric tissue of Electrophorus of a protein presenting several typical properties characteristic of the physiological receptor of acetylcholine. Comptes Rendus Hebd. Seances Acad. Sci. D 1970, 270, 2864–2867. [Google Scholar]
- Changeux, J.P.; Kasai, M.; Lee, C.Y. Use of a snake venom toxin to characterize the cholinergic receptor protein. Proc. Natl. Acad. Sci. USA 1970, 67, 1241–1247. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.C.; Lee, C.Y. Isolation of neurotoxins from the venom of Bungarus multicinctus and their modes of neuro-muscular blocking action. Arch. Int. Pharmacodyn. Ther. 1963, 144, 241–257. [Google Scholar] [PubMed]
- Meunier, J.C.; Huchet, M.; Boquet, P.; Changeux, J.-P. Separation of the receptor protein of acetylcholine and acetylcholinesterase. Comptes Rendus Hebd. Seances Acad. Sci. D 1971, 272, 117–120. [Google Scholar]
- Miledi, R.; Molinoff, P.; Potter, L.T. Isolation of the cholinergic receptor protein of Torpedo electric tissue. Nature 1971, 229, 554–557. [Google Scholar] [CrossRef]
- Karlsson, E.; Heilbronn, E.; Widlund, L. Isolation of the nicotinic acetylcholine receptor by biospecific chromatography on insolubilized Naja naja neurotoxin. FEBS Lett. 1972, 28, 107–111. [Google Scholar] [CrossRef]
- Klett, R.P.; Fulpius, B.W.; Cooper, D.; Smith, M.; Reich, E.; Possani, L.D. The acetylcholine receptor. I. Purification and characterization of a macromolecule isolated from Electrophorus electricus. J. Biol. Chem. 1973, 248, 6841–6853. [Google Scholar]
- Lindstrom, J.; Patrick, J. Acetylcholine receptors. In Synaptic Transmission and Neuronal Interaction; Bennett, M.V.L., Ed.; Raven Press: New York, NY, USA, 1974; pp. 191–216. [Google Scholar]
- Olsen, R.W.; Meunier, J.C.; Changeux, J.-P. Progress in purification of the cholinergic receptor protein from Electrophorus electricus by affinity chromatography. FEBS Lett. 1972, 28, 96–100. [Google Scholar] [CrossRef]
- Meunier, J.C.; Sealock, R.; Olsen, R.; Changeux, J.P. Purification and properties of the cholinergic receptor protein from Electrophorus electricus electric tissue. Eur. J. Biochem. 1974, 45, 371–394. [Google Scholar] [CrossRef]
- Schmidt, T.J.; Raftery, M.A. Use of affinity chromatography for acetylcholine receptor purification. Biochem. Biophys. Res. Commun. 1972, 49, 572–578. [Google Scholar] [CrossRef]
- Cohen, J.B.; Weber, M.; Huchet, M.; Changeux, J.P. Purification from Torpedo marmorata electric tissue of membrane fragments particularly rich in cholinergie receptor. FEBS Lett. 1972, 26, 43–47. [Google Scholar] [CrossRef]
- Cartaud, J.; Benedetti, E.L.; Cohen, J.B.; Meunier, J.C.; Changeux, J.P. Presence of a lattice structure in membrane fragments rich in nicotinic receptor protein from the electric organ of Torpedo marmorata. FEBS Lett. 1973, 33, 109–113. [Google Scholar] [CrossRef]
- Nickel, E.; Potter, L.T. Ultrastructure of isolated membranes of Torpedo electric tissue. Brain Res. 1973, 57, 508–517. [Google Scholar] [CrossRef]
- Brisson, A.; Unwin, P.N.T. Quaternary structure of the acetylcholine receptor. Nature 1985, 315, 474–477. [Google Scholar] [CrossRef] [PubMed]
- Patrick, J.; Lindstrom, J. Autoimmune response to acetylcholine receptor. Science 1973, 180, 871–872. [Google Scholar] [CrossRef] [PubMed]
- Luo, J.; Lindstrom, J. Acetylcholine receptor-specific immunosuppressive therapy of experimental autoimmune myasthenia gravis and myasthenia gravis. Ann. N. Y. Acad. Sci. 2018, 1413, 76–81. [Google Scholar] [CrossRef] [PubMed]
- Hucho, F.; Changeux, J.P. Molecular weight and quaternary structure of the cholinergic receptor protein extracted by detergents from Electrophorus electricus electric tissue. FEBS Lett. 1973, 38, 11–15. [Google Scholar] [CrossRef]
- Karlin, A.; Weill, C.; McNamee, M.G.; Valderrama, R. Facets of the structure of acetylcholine receptor from Electrophorns and Torpedo. Cold Spring Harbor Symp. Quant. Biol. l976, 40, 203–210. [Google Scholar] [CrossRef]
- Raftery, M.A.; Vandlen, R.; Michaelson, D.; Bode, J.; Moody, T.; Chao, Y.; Reed, K.; Deutsch, J.; Duguid, J. The biochemistry of an acetylcholine receptor. J. Supramol. Struct. 1974, 2, 582–592. [Google Scholar] [CrossRef]
- Lindstrom, J.; Walter, B.; Einarson, B. Immunochemical similarities between subunits of acetylcholine receptors from Torpedo, Electrophorus, and mammalian muscle. Biochemistry 1979, 18, 4470–4480. [Google Scholar] [CrossRef]
- Saitoh, T.; Oswald, R.; Wennogle, L.P.; Changeux, J.P. Conditions for the selective labelling of the 66,000 Dalton chain of the acetylcholine receptor by the covalent non-competitive blocker 5-azido- [3H]trimethisoquin. FEBS Lett. 1980, 116, 30–36. [Google Scholar] [CrossRef]
- Devillers-Thiéry, A.; Changeux, J.P.; Paroutaud, P.; Strosberg, A.D. The amino-terminal sequence of the 40,000 molecular weight subunit of the acetylcholine receptor protein from Torpedo marmorata. FEBS Lett. 1979, 104, 99–105. [Google Scholar] [CrossRef]
- Hunkapiller, M.W.; Strader, C.D.; Hood, L.; Raftery, M.A. Amino-terminal amino acid sequence of the major polypeptide subunit of Torpedo californica acetylcholine receptor. Biochem. Biophys. Res. Commun. 1979, 91, 164–169. [Google Scholar] [CrossRef]
- Raftery, M.A.; Hunkapiller, M.W.; Strader, C.D.; Hood, L.E. Acetylcholine receptor: Complex of homologous subunits. Science 1980, 208, 1454–1456. [Google Scholar] [CrossRef] [PubMed]
- Noda, M.; Takahashi, H.; Tanabe, T.; Toyosato, M.; Furutani, Y.; Hirose, T.; Asai, M.; Inayama, S.; Miyata, T.; Numa, S. Primary structure of α-subunit precursor of Torpedo californica acetylcholine receptor deduced from cDNA sequence. Nature 1982, 299, 793–797. [Google Scholar] [CrossRef]
- Noda, M.; Takahashi, H.; Tanabe, T.; Toyosato, M.; Kikyotani, S.; Hirose, T.; Asai, M.; Takashima, H.; Inayama, S.; Miyata, T.; et al. Primary structures of β- and δ-subunit precursors of Torpedo californica acetylcholine receptor deduced from cDNA sequences. Nature 1983, 301, 251–255. [Google Scholar] [CrossRef]
- Noda, M.; Takahashi, H.; Tanabe, T.; Toyosato, M.; Kikyotani, S.; Furutani, Y.; Hirose, T.; Takashima, H.; Inayama, S.; Miyata, T.; et al. Structural homology of Torpedo californica acetylcholine receptor subunits. Nature 1983, 302, 528–553. [Google Scholar] [CrossRef]
- Ballivet, M.; Patrick, J.; Lee, J.; Heinemann, S. Molecular cloning of cDNA coding for the γ-subunit of Torpedo acetylcholine receptor. Proc. Natl. Acad. Sci. USA 1982, 79, 4466–4470. [Google Scholar] [CrossRef]
- Claudio, T.; Ballivet, M.; Patrick, J.; Heinemann, S. Nucleotide and deduced amino acid sequences of Torpedo californica acetylcholine receptor γ-subunit. Proc. Natl. Acad. Sci. USA 1983, 80, 1111–1115. [Google Scholar] [CrossRef]
- Sumikawa, K.; Houghton, M.; Smith, J.C.; Bell, L.; Richards, B.M.; Barnard, E.A. The molecular cloning and characterisation of cDNA coding for the α-subunit of the acetylcholine receptor. Nucleic Acids Res. 1982, 10, 5809–5822. [Google Scholar] [CrossRef][Green Version]
- Giraudat, J.; Devillers-Thiéry, A.; Auffray, C.; Rougeon, F.; Changeux, J.P. Identification of a cDNA clone coding for the acetylcholine binding subunit of Torpedo marmorata acetylcholine receptor. EMBO J. 1982, 1, 713–717. [Google Scholar] [CrossRef]
- Devillers-Thiéry, A.; Giraudat, J.; Bentaboulet, M.; Changeux, J.P. Complete mRNA coding sequence of the acetylcholine binding α-subunit of Torpedo marmorata acetylcholine receptor: A model for the transmembrane organization of the polypeptide chain. Proc. Natl. Acad. Sci. USA 1983, 80, 2067–2071. [Google Scholar] [CrossRef]
- Barnard, E.A.; Miledi, R.; Sumikawa, K. Translation of exogenous messenger RNA coding for nicotinic acetylcholine receptors produces functional receptors in Xenopus oocytes. Proc. R. Soc. Lond. B Biol. Sci. 1982, 215, 241–246. [Google Scholar] [PubMed]
- Mishina, M.; Kurosaki, T.; Tobimatsu, T.; Morimoto, Y.; Noda, M.; Yamamoto, T.; Terao, M.; Lindstrom, J.; Takahashi, T.; Kuno, M.; et al. Expression of functional acetylcholine receptor from cloned cDNAs. Nature 1984, 307, 604–608. [Google Scholar] [CrossRef] [PubMed]
- Hazelbauer, G.L.; Changeux, J.P. Reconstitution of a chemically excitable membrane. Proc. Natl. Acad. Sci. USA 1974, 71, 1479–1483. [Google Scholar] [CrossRef] [PubMed]
- Epstein, M.; Racker, E. Reconstitution of carbamylcholine-dependent sodium ion flux and desensitization of the acetylcholine receptor from Torpedo californica. J. Biol. Chem. 1978, 253, 6660–6662. [Google Scholar]
- Boulter, J.; Evans, K.; Goldman, D.; Martin, G.; Treco, D.; Heinemann, S.; Patrick, J. Isolation of a cDNA clone coding for a possible neural nicotinic acetylcholine receptor α-subunit. Nature 1986, 319, 368–374. [Google Scholar] [CrossRef]
- Changeux, J.P.; Edelstein, S. Nicotinic Acetylcholine Receptors: From Molecular Biology to Cognition; Odile Jacob: New York, NY, USA, 2005. [Google Scholar]
- Changeux, J.-P. Functional architecture and dynamics of the nicotinic acetylcholine receptor: An allosteric ligand-gated ion channel. In Fidia Research Foundation Neuroscience Award Lectures; Raven Press: New York, NY, USA, 1990; Volume 4, pp. 21–168. [Google Scholar]
- Tasneem, A.; Iyer, L.M.; Jakobsson, E.; Aravind, L. Identification of the prokaryotic ligand-gated ion channels and their implications for the mechanisms and origins of animal Cys loop ion channels. Genome Biol. 2004, 6, R4. [Google Scholar] [CrossRef]
- Bocquet, N.; Prado de Carvalho, L.; Cartaud, J.; Neyton, J.; Le Poupon, C.; Taly, A.; Grutter, T.; Changeux, J.-P.; Corringer, P.J. A prokaryotic proton-gated ion channel from the nicotinic acetylcholine receptor family. Nature 2007, 445, 116–119. [Google Scholar] [CrossRef]
- Changeux, J.-P. The nicotinic acetylcholine receptor: A typical ‘allosteric machine’. Philos. Trans. R. Soc. B 2018, 373, 20170174. [Google Scholar] [CrossRef]
- Changeux, J.-P. The nicotinic acetylcholine receptor: The founding father of the pentameric ligand-gated ion channel superfamily. J. Biol. Chem. 2012, 287, 40207–40215. [Google Scholar] [CrossRef]
- Changeux, J.-P. 50 years of allosteric interactions: The twists and turns of the models. Nat. Rev. Mol. Cell. Biol. 2013, 14, 819–829. [Google Scholar] [CrossRef] [PubMed]
- Changeux, J.-P. Climbing Brain Levels of Organization from Genes to Consciousness. Trends Cogn. Sci. 2017, 21, 168–181. [Google Scholar] [CrossRef] [PubMed]
- Changeux, J.P.; Corringer, P.J.; Maskos, U. The nicotinic acetylcholine receptor: From molecular biology to cognition. Neuropharmacology 2015, 96 Pt B, 135–136. [Google Scholar] [CrossRef]
- Changeux, J.-P. Nicotine addiction and nicotinic receptors: lessons from genetically modified mice. Nat. Rev. Neurosci. 2010, 11, 389–401. [Google Scholar] [CrossRef] [PubMed]
- Engel, A.G.; Ohno, K.; Sine, S.M. Congenital myasthenic syndromes: A diverse array of molecular targets. J. Neurocytol. 2003, 32, 1017–1037. [Google Scholar] [CrossRef] [PubMed]
- Steinlein, O.K. Genetics and epilepsy. Dialogues Clin. Neurosci. 2008, 10, 29–38. [Google Scholar]


© 2020 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Changeux, J.-P. Discovery of the First Neurotransmitter Receptor: The Acetylcholine Nicotinic Receptor. Biomolecules 2020, 10, 547. https://doi.org/10.3390/biom10040547
Changeux J-P. Discovery of the First Neurotransmitter Receptor: The Acetylcholine Nicotinic Receptor. Biomolecules. 2020; 10(4):547. https://doi.org/10.3390/biom10040547
Chicago/Turabian StyleChangeux, Jean-Pierre. 2020. "Discovery of the First Neurotransmitter Receptor: The Acetylcholine Nicotinic Receptor" Biomolecules 10, no. 4: 547. https://doi.org/10.3390/biom10040547
APA StyleChangeux, J.-P. (2020). Discovery of the First Neurotransmitter Receptor: The Acetylcholine Nicotinic Receptor. Biomolecules, 10(4), 547. https://doi.org/10.3390/biom10040547



