The Regulation of Fat Metabolism during Aerobic Exercise
Abstract
1. Introduction
2. Lipids as a Form of Energy during Exercise
2.1. Fat Metabolism and Endurance Training
2.1.1. The rate of Lipolysis is Modulated by Temperature
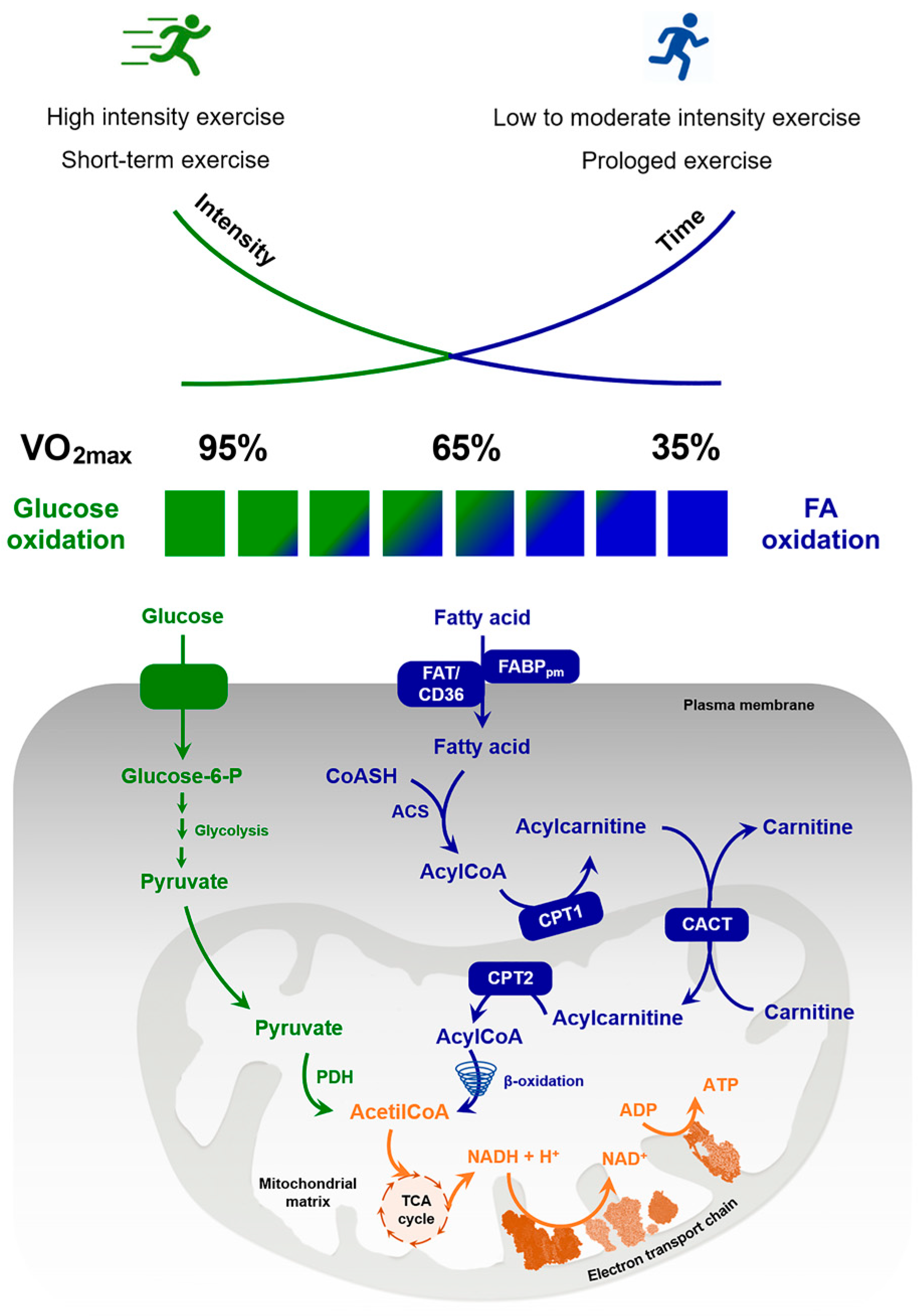
2.1.2. The Rate of Lipolysis Is Modulated by the Intensity of Physical Activities
2.2. Regulation of FAs Oxidation in Skeletal Muscle during Exercise
2.2.1. Exercise Intensities’ Effects on Beta Oxidation
2.2.2. Molecular Mechanisms Regulating FAs Oxidation
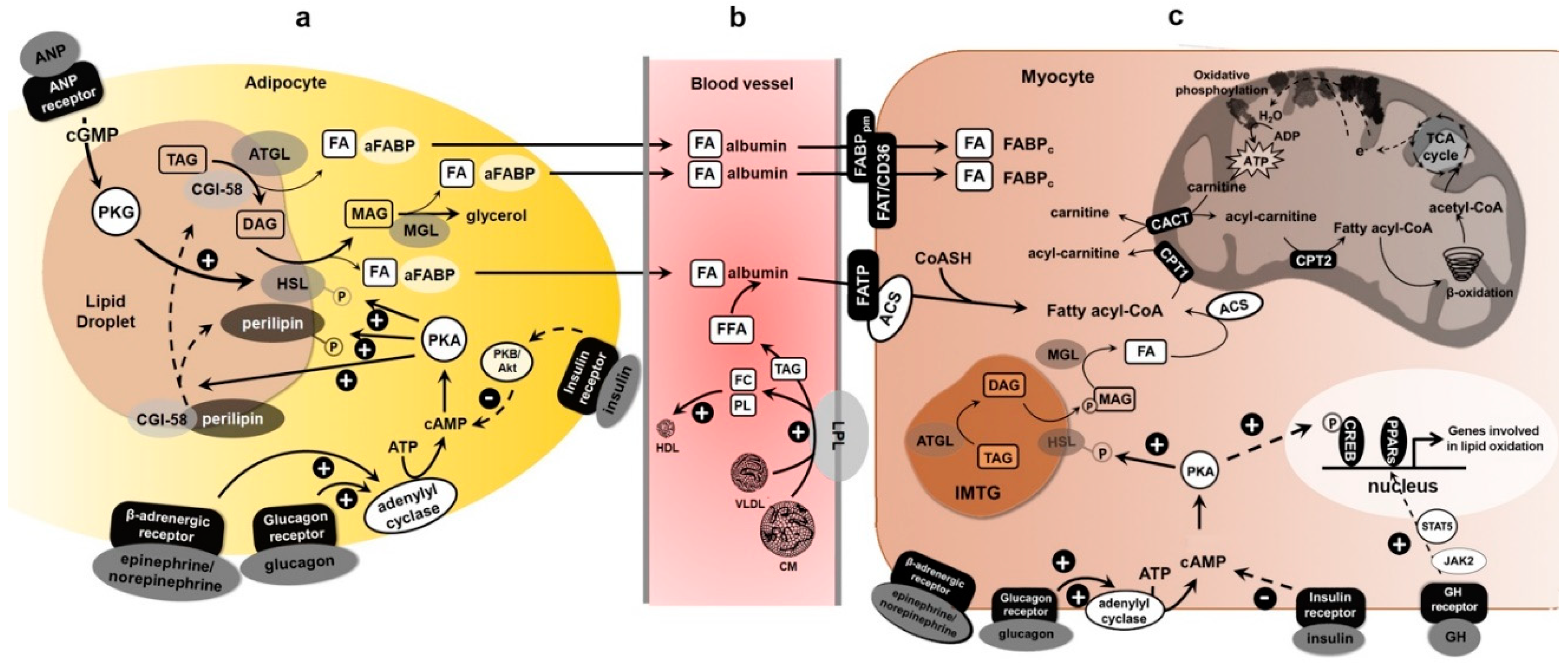
3. Transport of FAs from Adipose Tissue to Skeletal Muscle and Exercise
4. Molecular Regulation of Lipolysis and Exercise
4.1. Endocrine Regulation
4.2. Hormone-Sensitive Lipase (HSL) Functions
4.3. Intracellular Hormone-Sensitive Lipase Regulation
4.4. Adipose Triglyceride Lipase Functions
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Soci, U.P.R.; Melo, S.F.S.; Gomes, J.L.P.; Silveira, A.C.; Nóbrega, C.; de Oliveira, E.M. Exercise training and epigenetic regulation: Multilevel modification and regulation of gene expression. Adv. Exp. Med. Biol. 2017, 1000, 281–322. [Google Scholar] [PubMed]
- Mendonca, G.V.; Pezarat-Correia, P.; Vaz, J.R.; Silva, L.; Almeida, I.D.; Heffernan, K.S. Impact of exercise training on physiological measures of physical fitness in the elderly. Curr. Aging Sci. 2016, 9, 240–259. [Google Scholar] [PubMed]
- Hansen, D.; de Strijcker, D.; Calders, P. Impact of endurance exercise training in the fasted state on muscle biochemistry and metabolism in healthy subjects: Can these effects be of particular clinical benefit to type 2 diabetes mellitus and insulin-resistant patients? Sports Med. 2017, 47, 415–428. [Google Scholar] [PubMed]
- Bhati, P.; Shenoy, S.; Hussain, M.E. Exercise training and cardiac autonomic function in type 2 diabetes mellitus: A systematic review. Diabetes Metab. Syndr. 2018, 12, 69–78. [Google Scholar] [PubMed]
- Berlin, J.A.; Colditz, G.A. A meta-analysis of physical activity in the prevention of coronary heart disease. Am. J. Epidemiol. 1990, 132, 612–628. [Google Scholar]
- Powers, S.K.; Quindry, J.C.; Kavazis, A.N. Exercise-induced cardioprotection against myocardial ischemia-reperfusion injury. Free Radic. Biol. Med. 2008, 44, 193–201. [Google Scholar]
- Fedewa, M.V.; Hathaway, E.D.; Ward-Ritacco, C.L. Effect of exercise training on C reactive protein: A systematic review and meta-analysis of randomised and non-randomised controlled trials. Br. J. Sports Med. 2017, 51, 670–676. [Google Scholar]
- Tambosco, L.; Percebois-Macadré, L.; Rapin, A.; Nicomette-Bardel, J.; Boyer, F.C. Effort training in Parkinson’s disease: A systematic review. Annu. Phys. Rehabil. Med. 2014, 57, 79–104. [Google Scholar]
- Motl, R.W.; Sandroff, B.M. Benefits of exercise training in multiple sclerosis. Curr. Neurol. Neurosci. Rep. 2015, 15, 62. [Google Scholar]
- Dowman, L.M.; McDonald, C.F.; Hill, C.J.; Lee, A.L.; Barker, K.; Boote, C.; Glaspole, I.; Goh, N.S.L.; Southcott, A.M.; Burge, A.T.; et al. The evidence of benefits of exercise training in interstitial lung disease: A randomised controlled trial. Thorax 2017, 72, 610–619. [Google Scholar]
- Halabchi, F.; Alizadeh, Z.; Sahraian, M.A.; Abolhasani, M. Exercise prescription for patients with multiple sclerosis; potential benefits and practical recommendations. BMC Neurol. 2017, 17, 185. [Google Scholar] [CrossRef] [PubMed]
- Palermo, P.; Corrà, U. Exercise prescriptions for training and rehabilitation in patients with heart and lung disease. Ann. Am. Thorac. Soc. 2017, 14, 59–66. [Google Scholar] [CrossRef] [PubMed]
- Markvardsen, L.H.; Overgaard, K.; Heje, K.; Sindrup, S.H.; Christiansen, I.; Vissing, J.; Andersen, H. Resistance training and aerobic training improve muscle strength and aerobic capacity in chronic inflammatory demyelinating polyneuropathy. Muscle Nerve 2018, 57, 70–76. [Google Scholar] [CrossRef] [PubMed]
- Banfi, G.; Colombini, A.; Lombardi, G.; Lubkowska, A. Metabolic markers in sports medicine. Adv. Clin. Chem. 2012, 56, 1–54. [Google Scholar]
- Horowitz, J.F.; Klein, S. Lipid metabolism during endurance exercise. Am. J. Clin. Nutr. 2000, 72, 558S–563S. [Google Scholar] [CrossRef]
- Jocken, J.W.; Blaak, E.E. Catecholamine-induced lipolysis in adipose tissue and skeletal muscle in obesity. Physiol. Behav. 2008, 94, 219–230. [Google Scholar] [CrossRef]
- Carlson, L.A.; Ekelund, L.-G.; Froberg, S.O. Concentration of triglycerides, phospholipids and glycogen in skeletal muscle and of free fatty acids and β-hydroxybutyric acid in blood in man in response to exercise. Eur. J. Clin. Investig. 1971, 1, 248–254. [Google Scholar] [CrossRef]
- Froberg, S.O.; Mossfeldt, F. Effect of prolonged strenuous exercise on the concentration of triglycerides, phospholipids and glycogen in muscle of man. Acta Physiol. Scand. 1971, 82, 167–171. [Google Scholar] [CrossRef]
- Ranallo, R.F.; Rhodes, E.C. Lipid metabolism during exercise. Sports Med. 1998, 26, 29–42. [Google Scholar] [CrossRef]
- Romijn, J.A.; Coyle, E.F.; Sidossis, L.S.; Gastaldelli, A.; Horowitz, J.F.; Endert, E.; Wolfe, R.R. Regulation of endogenous fat and carbohydrate metabolism in relation to exercise intensity and duration. Am. J. Physiol. 1993, 265, 380–391. [Google Scholar] [CrossRef]
- Houten, S.M.; Wanders, R.J. A general introduction to the biochemistry of mitochondrial fatty acid β-oxidation. J. Inherit. Metab. Dis. 2010, 33, 469–477. [Google Scholar] [CrossRef] [PubMed]
- Chycki, J.; Zajac, A.; Michalczyk, M.; Maszczyk, A.; Langfort, J. Hormonal and metabolic substrate status in response to exercise in men of different phenotype. Endocr. Connect. 2019, 8, 814–821. [Google Scholar] [CrossRef] [PubMed]
- Denton, N.; Karpe, F. Insulin resistance and fatty acid trafficking. In Reference Module in Biomedical Sciences; Elsevier: Amsterdam, The Netherlands, 2014. [Google Scholar]
- Miller, N.E. HDL metabolism and its role in lipid transport. Eur. Heart J. 1990, 11, 1–3. [Google Scholar] [CrossRef] [PubMed]
- James, A.P.; Slivkoff-Clark, K.; Mamo, J.C. Prior exercise does not affect chylomicron particle number following a mixed meal of moderate fat content. Lipids Health Dis. 2007, 6, 8. [Google Scholar] [CrossRef]
- Slivkoff-Clark, K.M.; James, A.P.; Mamo, J.C. The chronic effects of fish oil with exercise on postprandial lipaemia and chylomicron homeostasis in insulin resistant viscerally obese men. Nutr. Metab. 2012, 7, 9. [Google Scholar] [CrossRef]
- Gavin, C.; Sigal, R.J.; Cousins, M.; Menard, M.L.; Atkinson, M.; Khandwala, F.; Kenny, G.P.; Proctor, S.; Ooi, T.C. Diabetes aerobic and resistance exercise (DARE) trial investigators. Resistance exercise but not aerobic exercise lowers remnant-like lipoprotein particle cholesterol in type 2 diabetes: A randomized controlled trial. Atherosclerosis 2010, 213, 552–557. [Google Scholar] [CrossRef]
- Motiani, P.; Teuho, J.; Saari, T.; Virtanen, K.A.; Honkala, S.M.; Middelbeek, R.J.; Goodyear, L.J.; Eskola, O.; Andersson, J.; Löyttyniemi, E.; et al. Exercise training alters lipoprotein particles independent of brown adipose tissue metabolic activity. Obes. Sci. Pract. 2019, 5, 258–272. [Google Scholar] [CrossRef]
- Bittel, A.J.; Bittel, D.C.; Mittendorfer, B.; Patterson, B.W.; Okunade, A.L.; Yoshino, J.; Porter, L.C.; Abumrad, N.A.; Reeds, D.N.; Cade, W.T. A single bout of resistance exercise improves postprandial lipid metabolism in overweight/obese men with prediabetes. Diabetologia 2019, 63, 611–623. [Google Scholar] [CrossRef]
- Slentz, C.A.; Duscha, B.D.; Johnson, J.L.; Ketchum, K.; Aiken, L.B.; Samsa, G.P.; Houmard, J.A.; Bales, C.W.; Kraus, W.E. Effects of the amount of exercise on body weight, body composition and measures of central obesity. Arch. Intern. Med. 2004, 164, 31–39. [Google Scholar] [CrossRef]
- Woudberg, N.J.; Mendham, A.E.; Katz, A.A.; Goedecke, J.H.; Lecour, S. Exercise intervention alters HDL subclass distribution and function in obese women. Lipids Health Dis. 2018, 17, 232. [Google Scholar] [CrossRef]
- Gordon, T.; Castelli, W.P.; Hjortland, M.C.; Kannel, W.B.; Dawber, T.R. High density lipoprotein as a protective factor against coronary heart disease: The Framingham study. Am. J. Med. 1977, 62, 707–714. [Google Scholar] [CrossRef]
- Argani, N.; Sharifi, G.; Golshahi, J. Comparison of the effect of different intensity exercise on a bicycle ergometer on postprandial lipidemia in type II diabetic patients. ARYA Atheroscler. 2014, 10, 147–153. [Google Scholar] [PubMed]
- Kodama, S.; Tanaka, S.; Saito, K.; Shu, M.; Sone, Y.; Onitake, F.; Suzuki, E.; Shimano, H.; Yamamoto, S.; Kondo, K.; et al. Effect of aerobic exercise training on serum levels of high-density lipoprotein cholesterol. Arch. Intern. Med. 2007, 167, 999–1008. [Google Scholar] [CrossRef] [PubMed]
- Kühnast, S.; Fiocco, M.; van der Hoorn, J.W.A.; Princen, H.M.G.; Jukema, J.W. Innovative pharmaceutical interventions in cardiovascular disease: Focusing on the contribution of non-HDL-C/LDL-C-lowering versus HDL-C-raisingA systematic review and meta-analysis of relevant preclinical studies and clinical trials. Eur. J. Pharmacol. 2015, 763, 48–63. [Google Scholar] [CrossRef]
- Woudberg, N.J.; Pedretti, S.; Lecour, S.; Schulz, R.; Vuilleumier, N.; James, R.W.; Frias, M.A. Pharmacological intervention to modulate HDL: What do we target? Front. Pharmacol. 2018, 8, 1–16. [Google Scholar] [CrossRef]
- Sarzynski, M.A.; Burton, J.; Rankinen, T.; Blair, S.N.; Church, T.S.; Després, J.-P.; Hagberg, J.M.; Landers-Ramos, R.; Leon, A.S.; Mikus, C.R.; et al. The effects of exercise on the lipoprotein subclass profile: A meta-analysis of 10 interventions. Atherosclerosis 2015, 243, 364–372. [Google Scholar] [CrossRef]
- Casella-Filho, A.; Chagas, A.C.P.; Maranhão, R.C.; Trombetta, I.C.; Cesena, F.H.Y.; Silva, V.M.; Tanus-Santos, J.E.; Negrão, C.E.; da Luz, P.L. Effect of exercise training on plasma levels and functional properties of high-density lipoprotein cholesterol in the metabolic syndrome. Am. J. Cardiol. 2011, 107, 1168–1172. [Google Scholar] [CrossRef]
- Adams, V.; Besler, C.; Fischer, T.; Riwanto, M.; Noack, F.; Höllriegel, R.; Oberbach, A.; Jehmlich, N.; Völker, U.; Winzer, E.B.; et al. Exercise training in patients with chronic heart failure promotes restoration of high-density lipoprotein functional properties. Circ. Res. 2013, 113, 1345–1355. [Google Scholar] [CrossRef]
- Iborra, R.T.; Ribeiro, I.C.; Neves, M.Q.; Charf, A.M.; Lottenberg, S.A.; Negrão, C.E.; Nakandakare, E.R.; Passarelli, M. Aerobic exercise training improves the role of high-density lipoprotein antioxidant and reduces plasma lipid peroxidation in type 2 diabetes mellitus. Scand. J. Med. Sci. Sports 2008, 18, 742–750. [Google Scholar]
- Roberts, C.K.; Ng, C.; Hama, S.; Eliseo, A.J.; Barnard, R.J. Effect of a short-term diet and exercise intervention on inflammatory/anti-inflammatory properties of HDL in overweight/obese men with cardiovascular risk factors. J. Appl. Physiol. 2006, 101, 1727–1732. [Google Scholar] [CrossRef]
- Sang, H.; Yao, S.; Zhang, L.; Li, X.; Yang, N.; Zhao, J.; Zhao, L.; Si, Y.; Zhang, Y.; Lv, X.; et al. Walk-run training improves the anti-inflammation properties of high-density lipoprotein in patients with metabolic syndrome. J. Clin. Endocrinol. Metab. 2015, 100, 870–879. [Google Scholar] [CrossRef] [PubMed]
- Sondergaard, E.; Rahbek, I.; Sørensen, L.P.; Christiansen, J.S.; Gormsen, L.C.; Jensen, M.D.; Nielsen, S. Effects of exercise on VLDL-triglyc-eride oxidation and turnover. Am. J. Physiol. Endocrinol. Metab. 2011, 300, 939–944. [Google Scholar] [CrossRef] [PubMed]
- Nellemann, B.; Christensen, B.; Vissing, K.; Thams, L.; Sieljacks, P.; Larsen, M.S.; Jørgensen, J.O.; Nielsen, S. Kinetics and utilization of lipid sources during acute exercise and acipimox. Am. J. Physiol. Endocrinol. Metab. 2014, 307, E199–E208. [Google Scholar] [CrossRef] [PubMed]
- Barter, P. Lipoprotein metabolism and CKD: Overview. Clin. Exp. Nephrol. 2014, 18, 243–246. [Google Scholar] [CrossRef]
- Brouns, F.; van der Vusse, G.J. Utilization of lipids during exercise in human subjects: Metabolic and dietary constraints. Br. J. Nutr. 1998, 79, 117–128. [Google Scholar] [CrossRef]
- Alves-Bezerra, M.; Cohen, D.E. Triglyceride metabolism in the liver. Compr. Physiol. 2017, 8, 1–8. [Google Scholar]
- Wang, Y.; Xu, D. Effects of aerobic exercise on lipids and lipoproteins. Lipids Health Dis. 2017, 16, 132. [Google Scholar] [CrossRef]
- Peric, R.; Meucci, M.; Bourdon, P.C.; Nikolovski, Z. Does the aerobic threshold correlate with the maximal fat oxidation rate in short stage treadmill tests? J. Sports Med. Phys. Fit. 2018, 58, 1412–1417. [Google Scholar]
- Febbraio, M.A.; Carey, M.F.; Snow, R.J.; Stathis, C.G.; Hargreaves, M. Influence of elevated muscle temperature on metabolism during intense, dynamic exercise. Am. J. Physiol. 1996, 271, R1251–R1255. [Google Scholar] [CrossRef]
- Hargreaves, M.; Angus, D.; Howlett, K.; Conus, N.M.; Febbraio, M.A. Effect of heat stress on glucose kinetics during exercise. J. Appl. Physiol. 1996, 81, 1594–1597. [Google Scholar] [CrossRef]
- O’Hearn, K.; Tingelstad, H.C.; Blondin, D.; Tang, V.; Filion, L.G.; Haman, F. Heat exposure increases circulating fatty acids but not lipid oxidation at rest and during exercise. J. Therm. Biol. 2016, 55, 39–46. [Google Scholar] [CrossRef] [PubMed]
- Galloway, S.D.R.; Maughan, R.J. Effects of ambient temperature on the capacity to perform prolonged cycle exercise in man. Med. Sci. Sports Exerc. 1997, 29, 1240–1249. [Google Scholar] [CrossRef] [PubMed]
- Layden, D.D.; Patterson, M.J.; Nimmo, M.A. Effects of reduced ambient temperature on fat utilization during submaximal exercise. Med. Sci. Sports Exerc. 2002, 34, 774–779. [Google Scholar] [CrossRef] [PubMed]
- Gagnon, D.D.; Rintamäki, H.; Gagnon, S.S.; Cheung, S.S.; Herzig, K.-H.; Porvari, K.; Kyröläinen, H. Cold exposure enhances fat utilization but not non-esterified fatty acids, glycerol or catecholamines availability during submaximal walking and running. Front. Physiol. 2013, 4, 99. [Google Scholar] [CrossRef] [PubMed]
- Vallerand, A.L.; Zamecnik, J.; Jones, P.J.; Jacobs, I. Cold stress increases lipolysis, FFA Ra and TG/FFA cycling in humans. Aviat. Space Environ. Med. 1999, 70, 42–50. [Google Scholar] [PubMed]
- Nimmo, M. Exercise in the cold. J. Sports Sci. 2004, 22, 898–915. [Google Scholar] [CrossRef] [PubMed]
- Purdom, T.; Kravitz, L.; Dokladny, K.; Mermier, C. Understanding the factors that effect maximal fat oxidation. J. Int. Soc. Sports Nutr. 2018, 15, 3. [Google Scholar] [CrossRef]
- Lundsgaard, A.M.; Fritzen, A.M.; Kiens, B. Molecular regulation of fatty acid oxidation in skeletal muscle during aerobic exercise. Trends Endocrinol. Metab. 2018, 29, 18–30. [Google Scholar] [CrossRef]
- Jeukendrup, A.; Gleeson, M. Sport Nutrition: An Introduction to Energy Production and Performance, 2nd ed.; Human Kinetics: Champaign, IL, USA, 2010. [Google Scholar]
- Van Loon, L.J.; Greenhaff, P.L.; Constantin-Teodosiu, D.; Saris, W.H.; Wagenmakers, A.J. The effects of increasing exercise intensity on muscle fuel utilisation in humans. J. Physiol. 2001, 536, 295–304. [Google Scholar] [CrossRef]
- Passler, S.; Bohrer, J.; Blöchinger, L.; Senner, V. Validity of wrist-worn activity trackers for estimating VO2max and energy expenditure. Int. J. Environ. Res. Public Health 2019, 16, 3037. [Google Scholar] [CrossRef]
- Klein, S.; Coyle, E.F.; Wolfe, R.R. Fat metabolism during low-intensity exercise in endurance-trained and untrained men. Am. J. Physiol. 1994, 267, E934–E940. [Google Scholar] [CrossRef] [PubMed]
- Holloszy, J.O.; Kohrt, W.M. Regulation of carbohydrate and fat metabolism during and after exercise. Ann. Rev. Nutr. 1996, 16, 121–138. [Google Scholar] [CrossRef] [PubMed]
- Achten, J.; Gleeson, M.; Jeukendrup, A.E. Determination of the exercise intensity that elicits maximal fat oxidation. Med. Sci. Sports Exerc. 2002, 34, 92–97. [Google Scholar] [CrossRef] [PubMed]
- Achten, J.; Venables, M.C.; Jeukendrup, A.E. Fat oxidation rates are higher during running compared with cycling over a wide range of intensities. Metabolism 2003, 52, 747–752. [Google Scholar] [CrossRef]
- Achten, J.; Jeukendrup, A.E. Optimizing fat oxidation through exercise and diet. Nutrition 2004, 20, 716–727. [Google Scholar] [CrossRef]
- Muscella, A.; Stefàno, E.; Marsigliante, S. The effects of exercise training on lipid metabolism and coronary heart disease. Am. J. Physiol. Heat. Circ. Physiol. 2020, 319, H76–H88. [Google Scholar]
- Petibois, C.; Cazorla, G.; Poortmans, J.R.; Déléris, G. Biochemical aspects of overtraining in endurance sports: A review. Sports Med. 2002, 32, 867–878. [Google Scholar] [CrossRef]
- Wolfe, R.R.; Klein, S.; Carraro, F.; Weber, J.M. Role of triglyceride-fatty acid cycle in controlling fat metabolism in humans during and after exercise. Am. J. Physiol. 1990, 258, 382–389. [Google Scholar] [CrossRef]
- Verboven, K.; Stinkens, R.; Hansen, D.; Wens, I.; Frederix, I.; Eijnde, B.O.; Jocken, J.W.E.; Goossens, G.H.; Blaak, E.E. Adrenergically and non-adrenergically mediated human adipose tissue lipolysis during acute exercise and exercise training. Clin. Sci. 2018, 132, 1685–1698. [Google Scholar] [CrossRef]
- Nieman, D.C.; Sha, W.; Pappan, K.L. IL-6 linkage to exercise-induced shifts in lipid-related metabolites: A metabolomics-based analysis. J. Proteome Res. 2017, 16, 970–977. [Google Scholar] [CrossRef]
- Nieman, D.C.; Shanely, R.A.; Gillitt, N.D.; Pappan, K.L.; Lila, M.A. Serum metabolic signatures induced by a three-day intensified exercise period persist after 14 h of recovery in runners. J. Proteome Res. 2013, 12, 4577–4584. [Google Scholar] [CrossRef] [PubMed]
- Laaksonen, M.S.; Kyröläinen, H.; Kemppainen, J.; Knuuti, J.; Kalliokoski, K.K. Muscle free fatty-acid uptake associates to mechanical efficiency during exercise in humans. Front. Physiol. 2018, 9, 1171. [Google Scholar] [CrossRef] [PubMed]
- Shaw, C.S.; Swinton, C.; Morales-Scholz, M.G.; McRae, N.L.; Erftemeyer, T.; Aldous, A.; Murphy, R.M.; Howlett, K.F. The impact of exercise training status on the fibre type specific abundance of proteins regulating intramuscular lipid metabolism. J. Appl. Physiol. 2020, 128, 379–389. [Google Scholar] [CrossRef] [PubMed]
- Dandanell, S.; Meinild-Lundby, A.K.; Andersen, A.B.; Lang, P.F.; Oberholzer, L.; Keiser, S.; Robach, P.; Larsen, S.; Rønnestad, B.R.; Lundby, C. Determinants of maximal whole-body fat oxidation in elite cross-country skiers: Role of skeletal muscle mitochondria. Scand. J. Med. Sci. Sports 2018, 28, 2494–2504. [Google Scholar] [CrossRef]
- Talanian, J.L.; Holloway, G.P.; Snook, L.A.; Heigenhauser, G.J.; Bonen, A.; Spriet, L.L. Exercise training increases sarcolemmal and mitochondrial fatty acid transport proteins in human skeletal muscle. Am. J. Physiol. Endocrinol. Metab. 2010, 299, 180–188. [Google Scholar] [CrossRef]
- Jeppesen, J.; Jordy, A.B.; Sjøberg, K.A.; Füllekrug, J.; Stahl, A.; Nybo, L.; Kiens, B. Enhanced fatty acid oxidation and FATP4 protein expression after endurance exercise training in human skeletal muscle. PLoS ONE 2012, 7, e29391. [Google Scholar] [CrossRef]
- Petridou, A.; Chatzinikolaou, A.; Avloniti, A.; Jamurtas, A.; Loules, G.; Papassotiriou, I.; Fatouros, I.; Mougios, V. Increased triacylglycerol lipase activity in adipose tissue of lean and obese men during endurance exercise. J. Clin. Endocrinol. Metab. 2017, 102, 3945–3952. [Google Scholar] [CrossRef]
- Emed, L.G.; Passaglia, D.G.; Guerios, S.T.; João, P.G.; Moser, A.I.; Abdalla, D.S.; Guarita-Souza, L.C.; Mikilita, E.S.; Baena, C.P.; da Costa, A.B.; et al. Acute modification in plasma lipid levels in ultramarathon runners. J. Sports Sci. 2016, 34, 1657–1661. [Google Scholar] [CrossRef]
- Arakawa, K.; Hosono, A.; Shibata, K.; Ghadimi, R.; Fuku, M.; Goto, C.; Imaeda, N.; Tokudome, Y.; Hoshino, H.; Marumoto, M.; et al. Changes in blood biochemical markers before, during, and after a 2-day ultramarathon. J. Sports Med. 2016, 7, 43–50. [Google Scholar] [CrossRef]
- Yanai, H.; Watanabe, I.; Ishii, K.; Morimoto, M.; Fujiwara, H.; Yoshida, S.; Hui, S.-P.; Matsuno, K.; Chiba, H. Attenuated aerobic exercise capacity in CD36 deficiency. J. Med. Genet. 2007, 44, 445–447. [Google Scholar] [CrossRef]
- Davis, P.G.; Bartoli, W.P.; Durstine, J.L. Effects of acute exercise intensity on plasma lipids and apolipoproteins in trained runners. J. Appl. Physiol. 1992, 72, 914–919. [Google Scholar] [CrossRef] [PubMed]
- Vuorimaa, T.; Häkkinen, K.; Vähäsöyrinki, P.; Rusko, H. Comparison of three maximal anaerobic running test protocols in marathon runners, middle-distance runners and sprinters. Int. J. Sports Med. 1996, 17, 109–113. [Google Scholar] [CrossRef] [PubMed]
- Magkos, F.; Mohammed, B.S.; Patterson, B.W.; Mittendorfer, B. Free fatty acid kinetics in the late phase of postexercise recovery: Importance of resting fatty acid metabolism and exerciseinduced energy deficit. Metabolism 2009, 58, 1248–1255. [Google Scholar] [CrossRef] [PubMed]
- Hetlelid, K.J.; Plews, D.J.; Herold, E.; Laursen, P.B.; Seiler, S. Rethinking the role of fat oxidation: Substrate utilisation during high-intensity interval training in well-trained and recreationally trained runners. BMJ Open Sport Exerc. Med. 2015, 1, e000047. [Google Scholar] [CrossRef] [PubMed]
- Aslankeser, Z.; Balcı, S.S. Re-examination of the contribution of substrates to energy expenditure during high-intensity intermittent exercise in endurance athletes. PeerJ 2017, 5, e3769. [Google Scholar] [CrossRef]
- Van Hall, G. The physiological regulation of skeletal muscle fatty acid supply and oxidation during moderate-intensity exercise. Sports Med. 2015, 45, 23–32. [Google Scholar] [CrossRef]
- Martin, W.H., III; Dalsky, G.P.; Hurley, B.F.; Matthews, D.E.; Bier, D.M.; Hagberg, J.M.; Rogers, M.A.; King, D.S.; Holloszy, J.O. Effect of endurance training on plasma free fatty acid turnover and oxidation during exercise. Am. J. Physiol. 1993, 265, E708–E714. [Google Scholar] [CrossRef]
- De Bock, K.; Richter, E.A.; Russell, A.P.; Eijnde, B.O.; Derave, W.; Ramaekers, M.; Koninckx, E.; Léger, B.; Verhaeghe, J.; Hespel, P. Exercise in the fasted state facilitates fibre type-specific intramyocellular lipid breakdown and stimulates glycogen resynthesis in humans. J. Physiol. 2005, 564, 649–660. [Google Scholar] [CrossRef]
- Holloszy, J.O. Biochemical adaptations in muscle. Effects of exercise on mitochondrial oxygen uptake and respiratory enzyme activity in skeletal muscle. J. Biol. Chem. 1967, 242, 2278–2282. [Google Scholar]
- Harris, R.A.; Bowker-Kinley, M.M.; Huang, B.; Wu, P. Regulation of the activity of the pyruvate dehydrogenase complex. Adv. Enzyme Regul. 2002, 42, 249–259. [Google Scholar] [CrossRef]
- McGarry, J.D.; Brown, N.F. The mitochondrial carnitine palmitoyltransferase system. Eur. J. Biochem. 1997, 244, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Houten, S.M.; Violante, S.; Ventura, F.V.; Wanders, R.J. The biochemistry and physiology of mitochondrial fatty acid β-oxidation and its genetic disorders. Annu. Rev. Physiol. 2016, 78, 23–44. [Google Scholar] [CrossRef] [PubMed]
- Constantin-Teodosiu, D.; Carlin, J.I.; Cederblad, G.; Harris, R.C.; Hultman, E. Acetyl group accumulation and pyruvate dehydrogenase activity in human muscle during incremental exercise. Acta Physiol. Scand. 1991, 143, 367–372. [Google Scholar] [CrossRef] [PubMed]
- Sahlin, K. Muscle carnitine metabolism during incremental dynamic exercise in humans. Acta Physiol. Scand. 1990, 138, 259–262. [Google Scholar] [CrossRef] [PubMed]
- Meienberg, F.; Loher, H.; Bucher, J.; Jenni, S.; Krüsi, M.; Kreis, R.; Boesch, C.; Betz, M.J.; Christ, E. The effect of exercise on intramyocellular acetylcarnitine (AcCtn) concentration in adult growth hormone deficiency (GHD). Sci. Rep. 2019, 9, 19431. [Google Scholar] [CrossRef] [PubMed]
- Roepstorff, C.; Vistisen, B.; Kiens, B. Intramuscular triacylglycerol in energy metabolism during exercise in humans. Exerc. Sport Sci. Rev. 2005, 33, 182–188. [Google Scholar] [CrossRef]
- Constantin-Teodosiu, D.; Cederblad, G.; Hultman, E. A sensitive radioisotopic assay of pyruvate dehydrogenase complex in human muscle tissue. Anal. Biochem. 1991, 198, 347–351. [Google Scholar] [CrossRef]
- Howlett, R.A.; Parolin, M.L.; Dyck, D.J.; Hultman, E.; Jones, N.L.; Heigenhauser, G.J.; Spriet, L.L. Regulation of skeletal muscle glycogen phosphorylase and PDH at varying exercise power outputs. Am. J. Physiol. 1998, 275, R418–R425. [Google Scholar] [CrossRef]
- Stephens, F.B.; Constantin-Teodosiu, D.; Laithwaite, D.; Simpson, E.J.; Greenhaff, P.L. An acute increase in skeletal muscle carnitine content alters fuel metabolism in resting human skeletal muscle. J. Clin. Endocrinol. Metab. 2006, 91, 5013–5018. [Google Scholar] [CrossRef] [PubMed]
- Stephens, F.B.; Wall, B.T.; Marimuthu, K.; Shannon, C.E.; Constantin-Teodosiu, D.; Macdonald, I.A.; Greenhaff, P.L. Skeletal muscle carnitine loading increases energy expenditure, modulates fuel metabolism gene networks and prevents body fat accumulation in humans. J. Physiol. 2013, 591, 4655–4666. [Google Scholar] [CrossRef]
- Wall, B.T.; Stephens, F.B.; Constantin-Teodosiu, D.; Marimuthu, K.; Macdonald, I.A.; Greenhaff, P.L. Chronic oral ingestion of L-carnitine and carbohydrate increases muscle carnitine content and alters muscle fuel metabolism during exercise in humans. J. Physiol. 2011, 589, 963–973. [Google Scholar] [CrossRef] [PubMed]
- Marriott, B.M. Food Components to Enhance Performance: An Evaluation of Potential Performance-Enhancing Food Components for Operational Rations; National Academy Press: Washington, DC, USA, 1994. [Google Scholar]
- Grivas, G.V. The role of L-carnitine in distance athletes. Int. J. Sports Sci. 2018, 8, 158–163. [Google Scholar]
- Colombani, P.; Wenk, C.; Kunz, I.; Krähenbühl, S.; Kuhnt, M.; Arnold, M.; Frey-Rindova, P.; Frey, W.; Langhans, W. Effects of L-carnitine supplementation on physical performance and energy metabolism of endurance-trained athletes: A double-blind crossover field study. Eur. J. Appl. Physiol. Occup. Physiol. 1996, 73, 434–439. [Google Scholar] [CrossRef] [PubMed]
- Stuessi, C.; Hofer, P.; Meier, C.; Boutellier, U. L-carnitine and the recovery from exhaustive endurance exercise: A randomised, double-blind, placebo-controlled trial. Eur. J. Appl. Physiol. 2005, 95, 431–435. [Google Scholar] [CrossRef]
- Giamberardino, M.A.; Dragani, L.; Valente, R.; Di Lisa, F.; Saggini, R.; Vecchiet, L. Effects of prolonged L-carnitine administration on delayed muscle pain and CK release after eccentric effort. Int. J. Sports Med. 1996, 17, 320–324. [Google Scholar] [CrossRef]
- Spiering, B.A.; Kraemer, W.J.; Vingren, J.L.; Hatfield, D.L.; Fragala, M.S.; Ho, J.Y.; Maresh, C.M.; Anderson, J.M.; Volek, J.S. Responses of criterion variables to different supplemental doses of L-carnitine L-tartrate. J. Strength Cond. Res. 2007, 21, 259–264. [Google Scholar] [CrossRef]
- Ho, J.Y.; Kraemer, W.J.; Volek, J.S.; Fragala, M.S.; Thomas, G.A.; Dunn-Lewis, C.; Coday, M.; Häkkinen, K.; Maresh, C.M. L-Carnitine L-tartrate supplementation favorably affects biochemical markers of recovery from physical exertion in middle-aged men and women. Metabolism 2010, 59, 1190–1999. [Google Scholar] [CrossRef]
- Parandak, K.; Arazi, H.; Khoshkhahesh, F.; Nakhostin-Roohi, B. The effect of two-week L-carnitine supplementation on exercise -induced oxidative stress and muscle damage. Asian J. Sports Med. 2014, 5, 123–128. [Google Scholar]
- Volek, J.S.; Kraemer, W.J.; Rubin, M.R.; Gómez, A.L.; Ratamess, N.A.; Gaynor, P. L-Carnitine L-tartrate supplementation favorably affects markers of recovery from exercise stress. Am. J. Physiol. Endocrinol. Metab. 2002, 282, 474–482. [Google Scholar] [CrossRef]
- Sidossis, L.S.; Gastaldelli, A.; Klein, S.; Wolfe, R.R. Regulation of plasma fatty acid oxidation during low- and high-intensity exercise. Am. J. Physiol. 1997, 272, 1065–1070. [Google Scholar] [CrossRef]
- Monaco, C.; Whitfield, J.; Jain, S.S.; Spriet, L.L.; Bonen, A.; Holloway, G.P. Activation of AMPKα2 is not required for mitochondrial FAT/CD36 accumulation during exercise. PLoS ONE 2015, 10, e0126122. [Google Scholar] [CrossRef] [PubMed]
- Holloway, G.P.; Bezaire, V.; Heigenhauser, G.J.F.; Tandon, N.N.; Glatz, J.F.C.; Luiken, J.J.F.P.; Bonen, A.; Spriet, L.L. Mitochondrial long chain fatty acid oxidation, fatty acid translocase/CD36 content and carnitine palmitoyltransferase I activity in human skeletal muscle during aerobic exercise. J. Physiol. 2006, 571, 201–210. [Google Scholar] [CrossRef] [PubMed]
- Smith, B.K.; Jain, S.S.; Rimbaud, S.; Dam, A.; Quadrilatero, J.; Ventura-Clapier, R.; Bonen, A.; Holloway, G.P. FAT/CD36 is located on the outer mitochondrial membrane, upstream of long-chain acyl-CoA synthetase, and regulates palmitate oxidation. Biochem. J. 2011, 437, 125–134. [Google Scholar] [CrossRef] [PubMed]
- Smith, B.K.; Bonen, A.; Holloway, G.P. A dual mechanism of action for skeletal muscle FAT/CD36 during exercise. Exerc. Sport Sci. Rev. 2012, 40, 211–217. [Google Scholar] [CrossRef] [PubMed]
- Brownsey, R.W.; Boone, A.N.; Elliott, J.E.; Kulpa, J.E.; Lee, W.M. Regulation of acetyl-CoA carboxylase. Biochem. Soc. Trans. 2006, 34, 223–227. [Google Scholar] [CrossRef] [PubMed]
- Marcinko, K.; Steinberg, G.R. The role of AMPK in controlling metabolism and mitochondrial biogenesis during exercise. Exp. Physiol. 2014, 99, 1581–1585. [Google Scholar] [CrossRef] [PubMed]
- Vavvas, D.; Apazidis, A.; Saha, A.K.; Gamble, J.; Patel, A.; Kemp, B.E.; Witters, L.A.; Ruderman, N.B. Contraction-induced changes in acetyl-CoA carboxylase and 5’-AMP-activated kinase in skeletal muscle. J. Biol. Chem. 1997, 272, 13255–13261. [Google Scholar] [CrossRef]
- Dean, D.; Daugaard, J.R.; Young, M.E.; Saha, A.; Vavvas, D.; Asp, S.; Kiens, B.; Kim, K.; Witters, L.; Richter, E.A.; et al. Exercise diminishes the activity of acetyl-CoA carboxylase in human muscle. Diabetes 2000, 49, 1295–1300. [Google Scholar] [CrossRef]
- Winder, W.W.; Hardie, D.G. Inactivation of acetyl-CoA carboxylase and activation of AMP-activated protein kinase in muscle during exercise. Am. J. Physiol. 1996, 270, 299–304. [Google Scholar] [CrossRef]
- Rasmussen, B.B.; Winder, W.W. Effect of exercise intensity on skeletal muscle malonyl-CoA and acetyl-CoA carboxylase. J. Appl. Physiol. 1997, 83, 1104–1109. [Google Scholar] [CrossRef]
- Odland, L.M.; Heigenhauser, G.J.; Lopaschuk, G.D.; Spriet, L.L. Human skeletal muscle malonyl-CoA at rest and during prolonged submaximal exercise. Am. J. Physiol. 1996, 270, 541–544. [Google Scholar] [CrossRef] [PubMed]
- Odland, L.M.; Howlett, R.A.; Heigenhauser, G.J.; Hultman, E.; Spriet, L.L. Skeletal muscle malonyl-CoA content at the onset of exercise at varying power outputs in humans. Am. J. Physiol. 1998, 274, 1080–1085. [Google Scholar] [CrossRef] [PubMed]
- Schuler, M.; Ali, F.; Chambon, C.; Duteil, D.; Bornert, J.-M.; Tardivel, A.; Desvergne, B.; Wahli, W.; Chambon, P.; Metzger, D. PGC1alpha expression is controlled in skeletal muscles by PPARbeta, whose ablation results in fiber-type switching, obesity, and type 2 diabetes. Cell Metab. 2006, 4, 407–414. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, T.; Yamamoto, J.; Iwasaki, S.; Asaba, H.; Hamura, H.; Ikeda, Y.; Watanabe, M.; Magoori, K.; Ioka, R.X.; Tachibana, K.; et al. Activation of peroxisome proliferator-activated receptor delta induces fatty acid beta-oxidation in skeletal muscle and attenuates metabolic syndrome. Proc. Natl. Acad. Sci. USA 2003, 100, 15924–15929. [Google Scholar] [CrossRef] [PubMed]
- Fritz, T.; Krämer, D.K.; Karlsson, H.K.R.; Galuska, D.; Engfeldt, P.; Zierath, J.R.; Krook, A. Low-intensity exercise increases skeletal muscle protein expression of PPARdelta and UCP3 in type 2 diabetic patients. Diabetes Metab. Res. Rev. 2006, 22, 492–498. [Google Scholar] [CrossRef]
- Mahoney, D.J.; Parise, G.; Melov, S.; Safdar, A.; Tarnopolsky, M.A. Analysis of global mRNA expression in human skeletal muscle during recovery from endurance exercise. FASEB J. 2005, 19, 1498–1500. [Google Scholar] [CrossRef]
- Greene, N.P.; Fluckey, J.D.; Lambert, B.S.; Greene, E.S.; Riechman, S.E.; Crouse, S.F. Regulators of blood lipids and lipoproteins? PPARdelta and AMPK, induced by exercise, are correlated with lipids and lipoproteins in overweight/obese men and women. Am. J. Physiol. Endocrinol. Metab. 2012, 303, E1212–E1221. [Google Scholar] [CrossRef]
- Fan, W.; Waizenegger, W.; Lin, C.S.; Sorrentino, V.; He, M.-X.; Wall, C.E.; Li, H.; Liddle, C.; Yu, R.T.; Atkins, A.R.; et al. PPARδ promotes running endurance by preserving glucose. Cell Metab. 2017, 25, 1186–1193. [Google Scholar] [CrossRef]
- World Anti-Doping Agency. The 2020 Prohibited. 2020. Available online: https://www.wada-ama.org/en/resources/science-medicine/prohibited-list-documents (accessed on 2 November 2020).
- Mansueto, G.; Armani, A.; Viscomi, C.; D’Orsi, L.; de Cegli, R.; Polishchuk, E.V.; Lamperti, C.; Di Meo, I.; Romanello, V.; Marchet, S.; et al. Transcription factor EB controls metabolic flexibility during exercise. Cell Metab. 2017, 25, 182–196. [Google Scholar] [CrossRef]
- O’Neill, H.M. AMPK and exercise: Glucose uptake and insulin sensitivity. Diabetes Metab. J. 2013, 37, 1–21. [Google Scholar] [CrossRef]
- Wang, J.; Wu, X.; Simonavicius, N.; Tian, H.; Ling, L. Medium-chain fatty acids as ligands for orphan G protein-coupled receptor GPR84. J. Biol. Chem. 2006, 281, 34457–34464. [Google Scholar] [CrossRef] [PubMed]
- Harasim, E.; Kalinowska, A.; Chabowski, A.; Stepek, T. The role of fatty-acid transport proteins (FAT/CD36, FABPpm, FATP) in lipid metabolism in skeletal muscles. Postepy Higieny Medycyny Doswiadczalnej 2008, 62, 433–441. [Google Scholar]
- Manio, M.C.C.; Matsumura, S.; Masuda, D.; Inoue, K. CD36 is essential for endurance improvement, changes in whole-body metabolism, and efficient ppar-related transcriptional responses in the muscle with exercise training. Physiol. Rep. 2017, 5, e13282. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, L.F. Mitochondrial basis for sex-differences in metabolism and exercise performance. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2018, 314, R848–R849. [Google Scholar] [CrossRef]
- Jeppesen, J.; Albers, P.H.; Rose, A.J.; Birk, J.B.; Schjerling, P.; Dzamko, N.; Steinberg, G.R.; Kiens, B. Contraction-induced skeletal muscle FAT/CD36 trafficking and FA uptake is AMPK independent. J. Lipid Res. 2011, 52, 699–711. [Google Scholar] [CrossRef]
- Hames, K.C.; Vella, A.; Kemp, B.J.; Jensen, M.D. Free fatty acid uptake in humans with CD36 deficiency. Diabetes 2014, 63, 3606–3614. [Google Scholar] [CrossRef]
- Kiens, B.; Roepstorff, C.; Glatz, J.F.; Bonen, A.; Schjerling, P.; Knudsen, J.; Nielsen, J.N. Lipidbinding proteins and lipoprotein lipase activity in human skeletal muscle: Influence of physical activity and gender. J. Appl. Physiol. 2004, 97, 1209–1218. [Google Scholar] [CrossRef]
- Simoneau, J.; Veerkamp, J.; Turcotte, L.; Kelly, D. Markers of capacity to utilize fatty acids in human skeletal muscle; relation to insulin resistance and obesity and effects of weight loss. FASEB J. 1999, 13, 2051–2060. [Google Scholar] [CrossRef]
- Tunstall, R.J.; Mehan, K.A.; Wadley, G.D.; Collier, G.R.; Bonen, A.; Hargreaves, M.; Cameron-Smith, D. Exercise training increases lipid metabolism gene expression in human skeletal muscle. Am. J. Physiol. Endocrinol. Metab. 2002, 283, 66–72. [Google Scholar] [CrossRef]
- Roepstorff, C.; Vistisen, B.; Roepstorff, K.; Kiens, B. Regulation of plasma long-chain fatty acid oxidation in relation to uptake in human skeletal muscle during exercise. Am. J. Physiol. Endocrinol. Metab. 2004, 287, 696–705. [Google Scholar] [CrossRef]
- Bradley, N.S.; Snook, L.A.; Jain, S.S.; Heigenhauser, G.J.F. Acute endurance exercise increases plasma membrane fatty acid transport proteins in rat and human skeletal muscle. Am. J. Physiol. Endocrinol. Metab. 2012, 302, E183–E189. [Google Scholar] [CrossRef] [PubMed]
- Samovski, D.; Su, X.; Xu, Y.; Abumrad, N.A.; Stahl, P.D. Insulin and AMPK regulate FA translocase/CD36 plasma membrane recruitment in cardiomyocytes via Rab GAP AS160 and Rab8a Rab GTPase. J. Lipid Res. 2012, 53, 709–717. [Google Scholar] [CrossRef] [PubMed]
- O’Neill, H.M.; Maarbjerg, S.J.; Crane, J.D.; Jeppesen, J.; Jørgensen, S.B.; Schertzer, J.D.; Shyroka, O.; Kiens, B.; van Denderen, B.J.; Tarnopolsky, M.A.; et al. AMP-activated protein kinase (AMPK) beta1beta2 muscle null mice reveal an essential role for AMPK in maintaining mitochondrial content and glucose uptake during exercise. Proc. Natl. Acad. Sci. USA 2011, 108, 16092–16097. [Google Scholar] [CrossRef] [PubMed]
- Dzamko, N.; Schertzer, J.D.; Ryall, J.G.; Steel, R.; Macaulay, S.L.; Wee, S.; Chen, Z.-P.; Michell, B.J.; Oakhill, J.S.; Watt, M.J.; et al. AMPK-independent pathways regulate skeletal muscle fatty acid oxidation. J. Physiol. 2008, 586, 5819–5831. [Google Scholar] [CrossRef] [PubMed]
- Roepstorff, C.; Thiele, M.; Hillig, T.; Pilegaard, H.; Richter, E.A.; Wojtaszewski, J.F.P.; Kiens, B. Higher skeletal muscle alpha2AMPK activation and lower energy charge and fat oxidation in men than in women during submaximal exercise. J. Physiol. 2006, 574, 125–138. [Google Scholar] [CrossRef] [PubMed]
- Jeppesen, J.; Maarbjerg, S.J.; Jordy, A.B.; Fritzen, A.M.; Pehmøller, C.; Sylow, L.; Serup, A.K.; Jessen, N.; Thorsen, K.; Prats, C.; et al. LKB1 regulates lipid oxidation during exercise independently of AMPK. Diabetes 2013, 62, 1490–1499. [Google Scholar] [CrossRef]
- Sakamoto, K.; Göransson, O.; Grahame Hardie, D.; Alessi, D.R. Activity of LKB1 and AMPK-related kinases in skeletal muscle: Effects of contraction, phenformin, and AICAR. Am. J. Physiol. Endocrinol. Metab. 2004, 287, 310–317. [Google Scholar] [CrossRef]
- Bouchard, C. Adaptation to acute and regular exercise: From reductionist approaches to integrative biology. Prog. Mol. Biol. Transl. Sci. 2015, 135, 1–15. [Google Scholar]
- Jain, S.S.; Chabowski, A.; Snook, L.A.; Schwenk, R.W.; Glatz, J.F.C.; Luiken, J.J.F.P.; Bonen, A. Additive effects of insulin and muscle contraction on fatty acid transport and fatty acid transporters, FAT/CD36, FABPpm, FATP1, 4 and 6. FEBS Lett. 2009, 583, 2294–2300. [Google Scholar] [CrossRef]
- Hoffman, N.J.; Parker, B.L.; Chaudhuri, R.; Fisher-Wellman, K.H.; Kleinert, M.; Humphrey, S.J.; Yang, P.; Holliday, M.; Trefely, S.; Fazakerley, D.J.; et al. Global phosphoproteomic analysis of human skeletal muscle reveals a network of exercise-regulated kinases and AMPK substrates. Cell Metab. 2015, 22, 922–935. [Google Scholar] [CrossRef]
- Su, X.; Abumrad, N.A. Cellular fatty acid uptake: A pathway under construction. Trends Endocrinol. Metab. 2009, 20, 72–77. [Google Scholar] [CrossRef] [PubMed]
- Abbott, M.F.; Edelman, A.M.; Turcotte, L.P. CaMKK is an upstream signal of AMP-activated protein kinase in regulation of substrate metabolism in contracting skeletal muscle. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2009, 297, R1724–R1732. [Google Scholar] [CrossRef] [PubMed]
- Raney, M.A.; Turcotte, L.P. Regulation of contraction induced FA uptake and oxidation by AMPK and ERK1/2 is intensity dependent in rodent muscle. Am. J. Physiol. Endocrinol. Metab. 2006, 291, 1220–1227. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Langfort, J.; Donsmark, M.; Ploug, T.; Holm, C.; Galbo, H. Hormone-sensitive lipase in skeletal muscle: Regulatory mechanisms. Acta Physiol. Scand. 2003, 178, 397–403. [Google Scholar] [CrossRef] [PubMed]
- Holm, C.; Osterlund, T.; Laurell, H.; Contreras, J.A. Molecular mechanisms regulating hormone-sensitive lipase and lipolysis. Annu. Rev. Nutr. 2000, 20, 365–393. [Google Scholar] [CrossRef] [PubMed]
- Shen, W.J.; Patel, S.; Natu, V.; Kraemer, F.B. Mutational analysis of structural features of rat hormone-sensitive lipase. Biochemistry 1998, 37, 8973–8979. [Google Scholar] [CrossRef]
- Anthonsen, M.W.; Ronnstrand, L.; Wernstedt, C.; Degerman, E.; Holm, C. Identification of novel phosphorylation sites in hormone-sensitive lipase that are phosphorylated in response to isoproterenol and govern activation properties in vitro. J. Biol. Chem. 1998, 273, 215–221. [Google Scholar] [CrossRef]
- Holm, C. Molecular mechanisms regulating hormone-sensitive lipase and lipolysis. Biochem. Soc. Trans. 2003, 31, 1120–1124. [Google Scholar] [CrossRef]
- Birbrair, A.; Zhang, T.; Wang, Z.M.; Messi, M.L.; Enikolopov, G.N.; Mintz, A.; Delbono, O. Role of pericytes in skeletal muscle regeneration and fat accumulation. Stem Cells Dev. 2013, 22, 2298–2314. [Google Scholar] [CrossRef]
- Lafontan, M.; Langin, D. Lipolysis and lipid mobilization in human adipose tissue. Prog. Lipid Res. 2009, 48, 275–297. [Google Scholar] [CrossRef]
- Jaworski, K.; Sarkadi-Nagy, E.; Duncan, R.E.; Ahmadian, M.; Sul, H.S. Regulation of triglyceride metabolism. IV. Hormonal regulation of lipolysis in adipose tissue. Am. J. Physiol. Gastrointest. Liver Physiol. 2007, 293, G1–G4. [Google Scholar] [CrossRef] [PubMed]
- Gollnick, P.D.; Soule, R.G.; Taylor, A.W.; Williams, C.; Ianuzzo, C.D. Exercise-induced glycogenolysis and lipolysis in the rat: Hormonal influence. Am. J. Physiol. 1970, 219, 729–733. [Google Scholar] [CrossRef] [PubMed]
- Tsiloulis, T.; Watt, M.J. Exercise and the regulation of adipose tissue metabolism. Prog. Mol. Biol. Transl. Sci. 2015, 135, 175–201. [Google Scholar] [PubMed]
- Williams, R.S.; Caron, M.G.; Daniel, K. Skeletal muscle β-adrenergic receptors: Variations due to fiber type and training. Am. J. Physiol. 1984, 246, 160–167. [Google Scholar] [CrossRef] [PubMed]
- Cleroux, J.; van Nguyen, P.; Taylor, A.W.; Leenen, F.H. Effects of Beta 1- Vs. Beta 1 + Beta 2-blockade on exercise endurance and muscle metabolism in humans. J. Appl. Physiol. 1989, 66, 548–554. [Google Scholar] [CrossRef] [PubMed]
- Oberer, M.; Boeszoermenyi, A.; Nagy, H.M.; Zechner, R. Recent insights into the structure and function of comparative gene identification-58. Curr. Opin. Lipidol. 2011, 22, 149–158. [Google Scholar] [CrossRef] [PubMed]
- Ogasawara, J.; Izawa, T.; Sakurai, T.; Sakurai, T.; Shirato, K.; Ishibashi, Y.; Ishida, H.; Ohno, H.; Kizaki, T. The molecular mechanism underlying continuous exercise training-induced adaptive changes of lipolysis in white adipose cells. J. Obes. 2015, 473, 430. [Google Scholar] [CrossRef]
- Ormsbee, M.J.; Choi, M.D.; Medlin, J.K.; Geyer, G.H.; Trantham, L.H.; Dubis, G.S.; Hickner, R.C. Regulation of fat metabolism during resistance exercise in sedentary lean and obese men. J. Appl. Physiol. 2009, 106, 1529–1537. [Google Scholar] [CrossRef]
- Large, V.; Reynisdottir, S.; Langin, D.; Fredby, K.; Klannemark, M.; Holm, C.; Arner, P. Decreased expression and function of adipocyte hormone-sensitive lipase in subcutaneous fat cells of obese subjects. J. Lipid Res. 1999, 40, 2059–2066. [Google Scholar]
- Langin, D.; Dicker, A.; Tavernier, G.; Hoffstedt, J.; Mairal, A.; Rydén, M.; Arner, E.; Sicard, A.; Jenkins, C.M.; Viguerie, N.; et al. Adipocyte lipases and defect of lipolysis in human obesity. Diabetes 2005, 54, 3190–3197. [Google Scholar] [CrossRef]
- Lafontan, M.; Moro, C.; Berlan, M.; Crampes, F.; Sengenes, C.; Galitzky, J. Control of lipolysis by natriuretic peptides and cyclic GMP. Trends Endocrinol. Metab. 2008, 19, 130–137. [Google Scholar] [CrossRef] [PubMed]
- Park, H.; Kaushik, V.K.; Constant, S.; Prentki, M.; Przybytkowski, E.; Ruderman, N.B.; Saha, A.K. Coordinate regulation of malonyl-CoA decarboxylase, sn-glycerol-3-phosphate acyltransferase, and acetyl-CoA carboxylase by AMP-activated protein kinase in rat tissues in response to exercise. J. Biol. Chem. 2002, 277, 32571–32577. [Google Scholar] [CrossRef] [PubMed]
- Daval, M.; Diot-Dupuy, F.; Bazin, R.; Hainault, I.; Viollet, B.; Vaulont, S.; Hajduch, E.; Ferre, P.; Foufelle, F. Anti-lipolytic action of AMP-activated protein kinase in rodent adipocytes. J. Biol. Chem. 2005, 280, 25250–25257. [Google Scholar] [CrossRef] [PubMed]
- Sponarova, J.; Mustard, K.J.; Horakova, O.; Flachs, P.; Rossmeisl, M.; Brauner, P.; Bardova, K.; Thomason-Hughes, M.; Braunerova, R.; Janovska, P.; et al. Involvement of AMP-activated protein kinase in fat depot-specific metabolic changes during starvation. FEBS Lett. 2005, 579, 6105–6110. [Google Scholar] [CrossRef]
- Watt, M.J.; Holmes, A.G.; Pinnamaneni, S.K.; Garnham, A.P.; Steinberg, G.R.; Kemp, B.E.; Febbraio, M.A. Regulation of HSL serine phosphorylation in skeletal muscle and adipose tissue. Am. J. Physiol. Endocrinol. Metab. 2006, 290, 500–508. [Google Scholar] [CrossRef]
- Kristensen, J.M.; Johnsen, A.B.; Birk, J.B.; Nielsen, J.N.; Jensen, B.R.; Hellsten, Y.; Richter, E.A.; Wojtaszewski, J.F.P. Absence of humoral mediated 5′AMP-activated protein kinase activation in human skeletal muscle and adipose tissue during exercise. J. Physiol. 2007, 585, 897–909. [Google Scholar] [CrossRef]
- Sengenes, C.; Moro, C.; Galitzky, J.; Berlan, M.; Lafontan, M. Natriuretic peptides: A new lipolytic pathway in human fat cells. Med. Sci. 2005, 21, 61–65. [Google Scholar]
- Sakharova, A.A.; Horowitz, J.F.; Surya, S.; Goldenberg, N.; Harber, M.P.; Symons, K.; Barkan, A. Role of growth hormone in regulating lipolysis, proteolysis, and hepatic glucose production during fasting. J. Clin. Endocrinol. Metab. 2008, 93, 2755–2759. [Google Scholar] [CrossRef]
- Grahn, T.H.; Kaur, R.; Yin, J.; Schweiger, M.; Sharma, V.M.; Lee, M.J.; Ido, Y.; Smas, C.M.; Zechner, R.; Lass, A.; et al. Fat-specific protein 27 (FSP27) interacts with adipose triglyceride lipase (ATGL) to regulate lipolysis and insulin sensitivity in human adipocytes. J. Biol. Chem. 2014, 289, 12029–12039. [Google Scholar] [CrossRef]
- Sharma, R.; Luong, Q.; Sharma, V.M.; Harberson, M.; Harper, B.; Colborn, A.; Berryman, D.E.; Jessen, N.; Jørgensen, J.O.L.; Kopchick, J.J.; et al. Growth hormone controls lipolysis by regulation of FSP27 expression. J. Endocrinol. 2018, 239, 289–301. [Google Scholar] [CrossRef]
- Chakrabarti, P.; Kim, J.Y.; Singh, M.; Shin, Y.K.; Kim, J.; Kumbrink, J.; Wu, Y.; Lee, M.J.; Kirsch, K.H.; Fried, S.K.; et al. Insulin inhibits lipolysis in adipocytes via the evolutionarily conserved mTORC1-Egr1-ATGL-mediated pathway. Mol. Cell. Biol. 2013, 33, 3659–3666. [Google Scholar] [CrossRef] [PubMed]
- Watt, M.J.; Steinberg, G.R. Regulation and function of triacylglycerol lipases in cellular metabolism. Biochem. J. 2008, 414, 313–325. [Google Scholar] [CrossRef] [PubMed]
- Jepson, C.A.; Yeaman, S.J. Inhibition of hormone-sensitive lipase by intermediary lipid metabolites. FEBS Lett. 1992, 310, 197–200. [Google Scholar] [CrossRef]
- Watt, M.J.; Stellingwerff, T.; Heigenhauser, G.J.; Spriet, L.L. Effects of plasma adrenaline on hormone-sensitive lipase at rest and during moderate exercise in human skeletal muscle. J. Physiol. 2003, 550, 325–332. [Google Scholar] [CrossRef]
- Kiens, B.; Essen-Gustavson, B.; Christensen, N.J.; Saltin, B. Skeletal muscle substrate utilization during submaximal exercise in man: Effect of endurance training. J. Physiol. 1993, 469, 459–478. [Google Scholar] [CrossRef]
- Watt, M.J.; Holmes, A.G.; Steinberg, G.R.; Mesa, J.L.; Kemp, B.E.; Febbraio, M.A. Reduced plasma FFA availability increases net triacylglycerol degradation, but not GPAT or HSL activity, in human skeletal muscle. Am. J. Physiol. Endocrinol. Metab. 2004, 287, E120–E127. [Google Scholar] [CrossRef][Green Version]
- Spriet, L.L.; Heigenhauser, G.J.; Jones, N.L. Endogenous triacylglycerol utilization by rat skeletal muscle during tetanic stimulation. J. Appl. Physiol. 1986, 60, 410–415. [Google Scholar] [CrossRef]
- Peters, S.J.; Dyck, D.J.; Bonen, A.; Spriet, L.L. Effects of epinephrine on lipid metabolism in resting skeletal muscle. Am. J. Physiol. 1998, 275, 300–309. [Google Scholar] [CrossRef]
- Langfort, J.; Ploug, T.; Ihlemann, J.; Saldo, M.; Holm, C.; Galbo, H. Expression of hormone-sensitive lipase and its regulation by adrenaline in skeletal muscle. Biochem. J. 1999, 340, 459–465. [Google Scholar] [CrossRef]
- Langfort, J.; Ploug, T.; Ihlemann, J.; Holm, C.; Galbo, H. Stimulation of hormone-sensitive lipase activity by contractions in rat skeletal muscle. Biochem. J. 2000, 351, 207–214. [Google Scholar] [CrossRef]
- Watt, M.J.; Steinberg, G.R.; Chan, S.; Garnham, A.; Kemp, B.E.; Febbraio, M.A. Beta-adrenergic stimulation of skeletal muscle HSL can be overridden by AMPK signaling. FASEB J. 2004, 18, 1445–1446. [Google Scholar] [CrossRef] [PubMed]
- Alsted, T.J.; Nybo, L.; Schweiger, M.; Fledelius, C.; Jacobsen, P.; Zimmermann, R.; Zechner, R.; Kiens, B. Adipose triglyceride lipase in human skeletal muscle is upregulated by exercise training. Am. J. Physiol. Endocrinol. Metab. 2009, 296, E445–E453. [Google Scholar] [CrossRef] [PubMed]
- Osuga, J.; Ishibashi, S.; Oka, T.; Yagyu, H.; Tozawa, R.; Fujimoto, A.; Shionoiri, F.; Yahagi, N.; Kraemer, F.B.; Tsutsumi, O.; et al. Targeted disruption of hormone-sensitive lipase results in male sterility and adipocyte hypertrophy, but not in obesity. Proc. Natl. Acad. Sci. USA 2000, 97, 787–792. [Google Scholar] [CrossRef] [PubMed]
- Haemmerle, G.; Zimmermann, R.; Hayn, M.; Theussl, C.; Waeg, G.; Wagner, E.; Sattler, W.; Magin, T.M.; Wagner, E.F.; Zechner, R. Hormone-sensitive lipase deficiency in mice causes diglyceride accumulation in adipose tissue, muscle, and testis. J. Biol. Chem. 2002, 277, 4806–4815. [Google Scholar] [CrossRef]
- Kjaer, M.; Howlett, K.; Langfort, J.; Zimmerman-Belsing, T.; Lorentsen, J.; Bulow, J.; Ihlemann, J.; Feldt-Rasmussen, U.; Galbo, H. Adrenaline and glycogenolysis in skeletal muscle during exercise: A study in adrenalectomised humans. J. Physiol. 2000, 528, 371–378. [Google Scholar] [CrossRef]
- Belfrage, P.; Jergil, B.; Stralfors, P.; Tornqvist, H. Hormonesensitive lipase of rat adipose tissue: Identification and some properties of the enzyme protein. FEBS Lett. 1977, 75, 259–264. [Google Scholar] [CrossRef]
- Garton, A.J.; Campbell, D.G.; Carling, D.; Hardie, D.G.; Colbran, R.J.; Yeaman, S.J. Phosphorylation of bovine hormone-sensitive lipase by the AMP-activated protein kinase. A possible antilipolytic mechanism. Eur. J. Biochem. 1989, 179, 249–254. [Google Scholar] [CrossRef]
- Watt, M.J.; Heigenhauser, G.J.; Spriet, L.L. Effects of dynamic exercise intensity on the activation of hormone-sensitive lipase in human skeletal muscle. J. Physiol. 2002, 547, 301–308. [Google Scholar] [CrossRef]
- Donsmark, M.; Langfort, J.; Holm, C.; Ploug, T.; Galbo, H. Contractions activate hormone-sensitive lipase in rat muscle by protein kinase C and mitogen-activated protein kinase. J. Physiol. 2003, 550, 845–854. [Google Scholar] [CrossRef]
- Greenberg, A.S.; Shen, W.; Muliro, K.; Patel, S.; Souza, S.C.; Roth, R.A.; Kraemer, F.B. Stimulation of lipolysis and hormone-sensitive lipase via the extracellular signal-regulated kinase pathway. J. Biol. Chem. 2001, 276, 45456–45461. [Google Scholar] [CrossRef]
- Talanian, J.L.; Tunstall, R.J.; Watt, M.J.; Duong, M.; Perry, C.G.R.; Steinberg, G.R.; Kemp, B.E.; Heigenhauser, G.J.F.; Spriet, L.L. Adrenergic regulation of HSL serine phosphorylation and activity in human skeletal muscle during the onset of exercise. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2006, 291, 1094–1099. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Woods, A.; Cheung, P.C.; Smith, F.C.; Davison, M.D.; Scott, J.; Beri, R.K.; Carling, D. Characterization of AMP-activated protein kinase beta and gamma subunits. Assembly of the heterotrimeric complex in vitro. J. Biol. Chem. 1996, 271, 10282–10290. [Google Scholar] [CrossRef] [PubMed]
- Stapleton, D.; Mitchelhill, K.I.; Gao, G.; Widmer, J.; Michell, B.J.; Teh, T.; House, C.M.; Fernandez, C.S.; Cox, T.; Witters, L.A.; et al. Mammalian AMP-activated protein kinase subfamily. J. Biol. Chem. 1996, 271, 611–614. [Google Scholar] [CrossRef] [PubMed]
- Woods, A.; Salt, I.; Scott, J.; Hardie, D.G.; Carling, D. The alpha1 and alpha2 isoforms of the AMP-activated protein kinase have similar activities in rat liver but exhibit differences in substrate specificity in vitro. FEBS Lett. 1996, 397, 347–351. [Google Scholar] [CrossRef]
- Chen, Z.P.; McConell, G.K.; Michell, B.J.; Snow, R.J.; Canny, B.J.; Kemp, B.E. AMPK signaling in contracting human skeletal muscle: Acetyl-CoA carboxylase and NO synthase phosphorylation. Am. J. Physiol. Endocrinol. Metab. 2000, 279, E1202–E1206. [Google Scholar] [CrossRef]
- Derave, W.; Ai, H.; Ihlemann, J.; Witters, L.A.; Kristiansen, S.; Richter, E.A.; Ploug, T. Dissociation of AMP-activated protein kinase activation and glucose transport in contracting slow-twitch muscle. Diabetes 2000, 49, 1281–1287. [Google Scholar] [CrossRef]
- Fujii, N.; Hayashi, T.; Hirshman, M.F.; Smith, J.T.; Habinowski, S.A.; Kaijser, L.; Mu, J.; Ljungqvist, O.; Birnbaum, M.J.; Witters, L.A.; et al. Exercise induces isoform-specific increase in 5AMP-activated protein kinase activity in human skeletal muscle. Biochem. Biophys. Res. Commun. 2000, 273, 1150–1155. [Google Scholar] [CrossRef]
- Stephens, T.J.; Chen, Z.P.; Canny, B.J.; Michell, B.J.; Kemp, B.E.; McConell, G.K. Progressive increase in human skeletal muscle AMPKalpha2 activity and ACC phosphorylation during exercise. Am. J. Physiol. Endocrinol. Metab. 2002, 282, 688–694. [Google Scholar] [CrossRef]
- Wojtaszewski, J.F.; MacDonald, C.; Nielsen, J.N.; Hellsten, Y.; Hardie, G.D.; Kemp, B.E.; Kiens, B.; Richter, E.A. Regulation of 5′AMP-activated protein kinase activity and substrate utilization in exercising human skeletal muscle. Am. J. Physiol. Endocrinol. Metab. 2003, 284, 813–822. [Google Scholar] [CrossRef]
- Prats, C.; Donsmark, M.; Qvortrup, K.; Londos, C.; Sztalryd, C.; Holm, C.; Galbo, H.; Ploug, T. Decrease in intramuscular lipid droplets and translocation of HSL in response to muscle contraction and epinephrine. J. Lipid Res. 2006, 47, 2392–2399. [Google Scholar] [CrossRef]
- Zimmermann, R.; Strauss, J.G.; Haemmerle, G.; Schoiswohl, G.; Birner-Gruenberger, R.; Riederer, M.; Lass, A.; Neuberger, G.; Eisenhaber, F.; Hermetter, A.; et al. Fat mobilization in adipose tissue is promoted by adipose triglyceride lipase. Science 2004, 306, 1383–1386. [Google Scholar] [CrossRef] [PubMed]
- Villena, J.A.; Roy, S.; Sarkadi-Nagy, E.; Kim, K.H.; Sul, H.S. Desnutrin, an adipocyte gene encoding a novel patatin domain-containing protein, is induced by fasting and glucocorticoids: Ectopic expression of desnutrin increases triglyceride hydrolysis. J. Biol. Chem. 2004, 279, 47066–47075. [Google Scholar] [CrossRef]
- Jenkins, C.M.; Mancuso, D.J.; Yan, W.; Sims, H.F.; Gibson, B.; Gross, R.W. Identification, cloning, expression, and purification of three novel human calcium-independent phospholipase A2 family members possessing triacylglycerol lipase and acylglycerol transacylase activities. J. Biol. Chem. 2004, 279, 48968–48975. [Google Scholar] [CrossRef]
- Jocken, J.W.; Smit, E.; Goossens, G.H.; Essers, Y.P.; van Baak, M.A.; Mensink, M.; Saris, W.H.; Blaak, E.E. Adipose triglyceride lipase (ATGL) expression in human skeletal muscle is type I (oxidative) fibre specific. Histochem. Cell Biol. 2008, 129, 535–538. [Google Scholar] [CrossRef] [PubMed]
- Badin, P.M.; Louche, K.; Mairal, A.; Liebisch, G.; Schmitz, G.; Rustan, A.C.; Smith, S.R.; Langin, D.; Moro, C. Altered skeletal muscle lipase expression and activity contribute to insulin resistance in humans. Diabetes 2011, 60, 1734–1742. [Google Scholar] [CrossRef] [PubMed]
- Chanarin, I.; Patel, A.; Slavin, G.; Wills, E.J.; Andrews, T.M.; Stewart, G. Neutral-lipid storage disease: A new disorder of lipid metabolism. Br. Med. J. 1975, 1, 553–555. [Google Scholar] [CrossRef] [PubMed]
- Lefevre, C.; Jobard, F.; Caux, F.; Bouadjar, B.; Karaduman, A.; Heilig, R.; Lakhdar, H.; Wollenberg, A.; Verret, J.L.; Weissenbach, J.; et al. Mutations in CGI-58, the gene encoding a new protein of the esterase/lipase/thioesterase subfamily, in Chanarin–Dorfman syndrome. Am. J. Hum. Genet. 2001, 69, 1002–1012. [Google Scholar] [CrossRef]
- Fischer, J.; Lefevre, C.; Morava, E.; Mussini, J.M.; Laforet, P.; Negre-Salvayre, A.; Lathrop, M.; Salvayre, R. The gene encoding adipose triglyceride lipase (PNPLA2) is mutated in neutral lipid storage disease with myopathy. Nat. Genet. 2007, 39, 28–30. [Google Scholar] [CrossRef]
- Kobayashi, K.; Inoguchi, T.; Maeda, Y.; Nakashima, N.; Kuwano, A.; Eto, E.; Ueno, N.; Sasaki, S.; Sawada, F.; Fujii, M.; et al. The lack of the C-terminal domain of adipose triglyceride lipase causes neutral lipid storage disease through impaired interactions with lipid droplets. J. Clin. Endocrinol. Metab. 2008, 93, 2877–2884. [Google Scholar] [CrossRef]
- Nielsen, T.S.; Vendelbo, M.H.; Jessen, N.; Pedersen, S.B.; Jørgensen, J.O.; Lund, S.; Møller, N. Fasting, but not exercise, increases adipose triglyceride lipase (ATGL) protein and reduces G(0)/G(1) switch gene 2 (G0S2) protein and mRNA content in human adipose tissue. J. Clin. Endocrinol. Metab. 2011, 96, E1293–E1297. [Google Scholar] [CrossRef]
- Ogasawara, J.; Sakurai, T.; Kizaki, T.; Ishibashi, Y.; Izawa, T.; Sumitani, Y.; Ishida, H.; Radak, Z.; Haga, S.; Ohno, H. Higher levels of ATGL are associated with exercise-induced enhancement of lipolysis in rat epididymal adipocytes. PLoS ONE 2012, 7, e40876. [Google Scholar] [CrossRef] [PubMed]
- Mason, R.R.; Meex, R.C.; Lee-Young, R.; Canny, B.J.; Watt, M.J. Phosphorylation of adipose triglyceride lipase Ser404 is not related to 5-AMPK activation during moderate-intensity exercise in humans. Am. J. Physiol. Endocrinol. Metab. 2012, 303, E534–E541. [Google Scholar] [CrossRef] [PubMed]
- MacPherson, R.E.; Ramos, S.V.; Vandenboom, R.; Roy, B.D.; Peters, S.J. Skeletal muscle PLIN proteins, ATGL and CGI-58, interactions at rest and following stimulated contraction. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2013, 304, R644–R650. [Google Scholar] [CrossRef] [PubMed]
- Smirnova, E.; Goldberg, E.B.; Makarova, K.S.; Lin, L.; Brown, W.J.; Jackson, C.L. ATGL has a key role in lipid droplet/adiposome degradation in mammalian cells. EMBO Rep. 2006, 7, 106–113. [Google Scholar] [CrossRef]
| References | Type of Exercise | Type of Study | Study Sample | Results |
|---|---|---|---|---|
| Low-Intensity Exercise | ||||
| Klein et al. (1994) [63] | 4 h of treadmill exercise eliciting an oxygen uptake of 20 mL/kg/min. | Glycerol and free fatty acid rate of appearance and lipid oxidation were evaluated during basal resting conditions and after 4 h of treadmill exercise and 1 h of recovery. | n = 5 endurance-trained men; n = 5 untrained men. | After 4 h of exercise, the average glycerol and free fatty acid values, was similar in both trained and untrained subjects; but during recovery, glycerol and free fatty acid values decreased more rapidly in trained than in untrained subjects. Triglyceride oxidation was greater during exercise in the trained than in the untrained group. |
| Wolfe et al. (1990) [70] | 4 h of treadmill exercise at 40% maximum O2 consumption, and 2 h of recovery. | Total fat oxidation was quantified by indirect calorimetry in response to exercise and in recovery from exercise. | n = 5 healthy male subjects. | Rate of appearance of glycerol and free fatty acids increased after 30 min and 4 h of exercise. Lipolysis decreased rapidly from the first 20 min to 2 h of recovery. |
| Verboven et al. (2018) [71] | 12-week exercise training | Abdominal subcutaneous adipose tissue (SCAT) extracellular glycerol concentration and blood flow were measured using microdialysis at rest, during low-intensity endurance-type exercise and post-exercise recovery; at the same time, the response to α-/β-adrenoceptor was evaluated. | n = 10 healthy lean insulin-sensitive men n = 10 obese insulin-sensitive men n = 10 obese insulin-resistant men. | Exercise induce an increase of extracellular glycerol in SCAT in obese IS versus lean IS men: this could be the result of a lower blood flow in subcutaneous adipose tissue in obese IS men. Nonetheless, extracellular glycerol was blunted in obese IR versus obese IS men, despite comparable local blood flow after exercise. SCAT extracellular glycerol was reduced by 60% following local α-/β-adrenoceptor blockade in obese IS but not in obese IR men; in the latter, exercise training did not affect non- adrenergically-mediated lipolysis, despite an improved metabolic profile and body composition. |
| Moderate-Intensity Exercise | ||||
| Chycki et al. (2019) [22] | Individuals belonging to the three groups were subjected to progressive exercise protocol on a treadmill at 30%, 50% and 70% VO2max, separated by 45 s of passive rest. | Venous blood was collected before, during and after exercise to determinate GH, noradrenaline, insulin, cortisol, glucose, FFA and glycerol. | n = 18 healthy trained and untrained men (32 ± 5.4 years): 6 obese subjects; 6 athletic subjects; 6 endurance-trained subjects. | Plasma glucose oxidation increased in relation to exercise intensity, especially in the athletic group, while plasma FFA level decreased with different kinetics in the three groups. Plasma GH increased immediately after exercise and remains high in all groups 45 min into recovery compared to rest. Plasma insulin decreased during exercise in all groups, but to a lesser extent in obese subjects. |
| O’Hearn et al. (2016) [52] | Two experimental trials conducted in the following way: 90 min baseline period in ambient temperature, followed by 120 min at rest and 30 min exercise at 50% VO2max at either 42 °C or 23 °C. | Metabolic data, heart rate, thermal responses and ventilation were measured throughout the baseline, passive periods and exercise period. Metabolic and ventilation measurements were recorded every 30 min. Blood samples were collected at baseline and 60 and 120 min of the passive period to determine changes in non-esterified fatty acid, TG, phospholipid and TC concentrations. | n = 8 healthy males (23–27 years). | Lipid oxidation rates were not different between heat (42 °C) and control (23 °C) conditions, as well as TG, phospholipid and TC levels. However, non-esterified fatty acid concentrations were significantly higher following passive heating (618 µM, 95% CI: 479–757) compared to control condition (391 µM, 95% CI: 270–511), and also following exercise (2036 µM, 95% CI: 1604–2469 for HEAT and 1351 µM, 95% CI: 1002–1699). * CI = confidence interval |
| Four experimental trials that consist in a baseline period of 15-min (25 °C) and 60 min of exercise (walking at 50% VO2max in 0 °C; walking at 50% VO2max in 22 °C; running at 70% VO2max in 0 °C and running at 70% VO2max in 22 °C. | Thermal, cardiovascular and oxidative responses were measured every 15 min during exercise. Blood samples for serum non-esterified fatty acids, glycerol, glucose, beta-hydroxybutyrate, plasma catecholamines and serum lipids were collected immediately prior, and at 30 and 60 min of exercise. | n = 10 moderately active males (24.3 ± 3.0 years). | During submaximal walking and running, a rise in fat utilization in the cold was seen through lower respiratory quotient (RQ) (−0.03 ± 0.02), greater fat oxidation (+0.14 ± 0.13 g·min−1) and contribution of fat to total energy expenditure (+1.62 ± 1.99 kcal·min−1). However, serum non-esterified fatty acids, glycerol or catecholamine concentrations did not increase. | |
| Petibois et al. (2002) [69] | A 10 Km run at the individual marathon velocity. | Blood triglycerides and glycerol and other biochemical parameters concentration, during exercise were analyzed. | n = 14 marathon runners (28–40 years) | Longer and/or less unsaturated blood fatty acids, a plasma triglyceride decrease, and a glycerol concentration increase were measured in the best runners |
| Nieman et al. (2017) [72] | Subjects ran on treadmills to exhaustion, with the speed set at 70% of VO2max. | Blood samples were collected before and after running to evaluate three cytokines, MCP-1, IL-6 and IL-8, and the stress hormones cortisol and epinephrine. Glycogen concentration was measured in vastus lateralis muscle biopsy. To study lipid metabolic profile was used three independent platforms: ultra-high performance liquid chromatography tandem mass spectrometry optimized for acidic or basic species and gas chromatography–mass spectrometry | n = 24 male runners (36.5 ± 1.8 years) | After running, muscle glycogen decreased (33.7% ± 4.2%), while MCP-1, IL-6 and IL-8 increased (1.4 ± 0.1-, 39.0 ± 8.8-, 2.4 ± 0.3-fold, respectively), such as cortisol and epinephrine (95.0% ± 18.9%, 158% ± 20.6%).The metabolomics analysis revealed changes in 209 metabolites, mostly long- and medium-chain fatty acids, fatty acid oxidation products (dicarboxylate and monohydroxy fatty acids, acylcarnitines) and ketone bodies. In this study, the relationship between IL-6 cytokine and adipose tissue lipolysis stimulation was not found. |
| Nieman et al. (2013) [73] | Subjects ran for 2.5 h/day on treadmills at ∼70% VO2max, for 3 days in a row. | 75 metabolites, pre-exercise, immediately and 14 h post-exercise, were identified. | n = 15 long distance runners (7 males, 8 females; 19−45 years). | Of a total of 75 metabolites, increased immediately following the 3-day running period, 22 were related to lipid and carnitine metabolism, 13 to amino acid and peptide metabolism, 4 to hemoglobin and porphyrin metabolism and 3 to Krebs cycle intermediates (succinate, fumarate, and malate). |
| Laaksonen et al. (2018) [74] | Participants were divided into efficient (EF) and inefficient (IE) groups based on their mechanical efficiency at 45% of VO2 peak intensity during submaximal bicycle ergometer test. | During exercise, muscle blood flow, uptakes of oxygen, fatty acids and glucose were measured using positron emission tomography. | n = 17 healthy physically active male (EF: 24 ± 2 years; IE: 23 ± 2 years). | The use of blood glucose and intramuscular FA and glucose appeared to be similar between the two groups. However, EF group had increased muscle FA compared to IE group during exercise which led to higher usage of plasma FA, leading to think that use of plasma FA is important for mechanical efficiency during exercise. |
| Shaw et al. (2020) [75] | The endurance trained men had regularly competed in cycling and/or triathlon events within the last year, whereas the untrained group were physically active but did not complete regular endurance-type training. | Maximal fat oxidation and maximal oxygen uptake of the two groups was evaluated after exercise test on a cycle ergometer until exhaustion. Blood samples and biopsy were collected to assessed muscle fiber type and proteins involved in intramuscular lipids utilization by immunofluorescence microscopy and immunoblotting. | n = 7 endurance trained young males n = 8 untrained young males | Endurance-trained subjects displayed a higher maximal fat oxidation rate, a greater proportion of type I muscle fibers and higher intramuscular lipids content compared to untrained individuals. ATGL, HSL, PLIN 2, PLIN 5 and HAD content was ~2–3-fold higher in type muscle fibers compared to type IIa fibers. Consequently, these last were higher in endurance trained individuals. |
| Dandanell et al. (2018) [76] | A graded exercise test was performed | Plasma maximal rates of fat oxidation and VO2max were determined. Skeletal muscle biopsies were obtained to determine fatty acid oxidation and mitochondrial volume density. | n = 8 endurance-trained male cross-country skiers (20–22 years; VO2max 71 mL/min/kg); n = 8 healthy untrained male controls (23–24 years; VO2max 48 mL/min/kg). | VO2max, plasma maximal rate of fat oxidation, fatty acid oxidation and mitochondrial volume density were higher in the endurance-trained subjects compared to untrained subjects. The mitochondrial volume density, together with central adaptations as VO2max, determined the maximal rate of fat oxidation in endurance-trained subjects. Intrinsic mitochondrial changes were not associated with augmented maximal rate of fat oxidation. |
| Talanian et al. (2010) [77] | Six weeks of high-intensity interval training. | Biopsies were taken before and following 2 and 6 weeks of training from the vastus lateralis muscle. | n = 10 untrained females (22 ± 1 years) | High-intensity interval training increases fatty acid transport protein FAT/CD36 (10%) and FABPpm (48%) content in whole muscle; FABPpm and FAT/CD36 content increase in sarcolemmal (23%) and mitochondrial (51%) membranes in human skeletal muscle, respectively. |
| Jeppesen at al. (2012) [78] | Eight week aerobic training program at 45–80% VO2max, with increasing training frequency during the weeks. | FATP1 and FATP4 protein expression in the vastus lateralis muscle. | n = 8 healthy males (30 ± 1 years) | FATP4 protein content is increased (33%), whereas FATP1 protein content is reduced (20%) in skeletal muscle. FATP4 protein expression is related to lipid oxidation during endurance exercise. |
| Petridou et al. (2017) [79] | Participants cycled for 30 min at a heart rate of 130 to 140 beats per minute. | Subcutaneous adipose tissue was sampled at baseline and 5, 10, 20 and 30 min of exercise to determinate triacylglycerol lipase activity and expression; blood was collected for glycerol, non-esterified fatty acid, glucose, lactate, insulin, and catecholamine determination. | n = 16 healthy, sedentary men (20–26 years): lean group (n = 7; body mass index BMI ≤ 25 kg/m2; body fat < 15%) and an obese group (n = 9; BMI > 30 kg/m2; body fat > 20%). | Triacylglycerol lipase activity increased at 10 min of exercise in the lean men and returned to baseline at 20 and 30 min; in the obese men, it was higher than baseline at 10, 20 and 30 min and higher than the corresponding values in the lean men at 20 and 30 min. mRNA levels did not change during exercise, but the obese men had lower mRNA levels of ATGL, HSL and CGI-58 compared with the lean men. |
| High-Intensity Exercise | ||||
| Romijin et al., (1993) [20] | Different exercise intensity was performed (25%, 65% or 85% of VO2max). | Plasma glucose tissue uptake and muscle glycogen oxidation were measured during the different exercise intensities. | n = 5 endurance-trained cyclists (24 ± 2 years; VO2max 67 ± 3 mL/min/kg) | Plasma glucose tissue uptake and muscle glycogen oxidation increased in relation to exercise intensity. During at higher intensities exercise, muscle triglyceride lipolysis was stimulated only whereas muscle glycogen and triglyceride oxidation decreased. During recovery from high-intensity exercise, the rate of lipolysis and release of fatty acids into plasma decreased. |
| Emed et al. (2016) [80] | The 24 h ultramarathon race was performed on an outdoor 400 m athletics track. | Total cholesterol, HDL, triglycerides, ApoB and ApoA1, before and after 400 m run, were assessed. | n = 14 male athletes (>18 years old). | No significant modifications in high-density lipoprotein, LDL and ApoA1 levels were measured. A reduction in ApoB levels correlated directly to the distance covered, and an increase in the LDL/ApoB ratio was observed. Lipid profile levels and oxidation of LDL were not acutely altered by prolonged physical activity. |
| Arakawa et al. (2016) [81] | 2-day, 130 km ultramarathon. | Free fatty acids levels, after 1, 3 and 5/6 days 130 km ultramarathon were measured. | n = 18 runners (52.1 ± 12.1 years; BMI 21.1 ± 1.6 kg/m2). | Free fatty acids levels significantly enhanced during the race periods and stayed elevated after the race. Triglycerides declined on day 2 and day 3, and then returned to baseline level. HDL-C elevated on day 2 and remained elevated up to day 5. T-Chol concentrations decreased on day 2 and day 3, and afterward returned to baseline level. |
| Yanai et al. (2007) [82] | Participants were subjected to an incremental work test: 3 min of pedaling on a 15 W-loaded cycle ergometer increased by 15 W every minute. | Ventilatory threshold and serum FA changes were evaluated in all participants during exercise; blood samples were obtained at rest, peak work rate and 15 min after exercise. | n = 34 healthy female students (20.0 ± 1.0 years; BMI 20.6 ± 1.9): normal participants (n = 22) and participants with CD36 deficiency (n = 12). | Subjects with CD36 deficiency showed significantly lower ventilatory threshold than normal participants that was related to percentage changes in FA at peak work rate. In normal participants, serum FA levels decreased at peak work rate; in participants with CD36 deficiency, FA levels were not decreased at peak work rate and remained at significantly higher levels than normal participants 15 min after exercise. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Muscella, A.; Stefàno, E.; Lunetti, P.; Capobianco, L.; Marsigliante, S. The Regulation of Fat Metabolism during Aerobic Exercise. Biomolecules 2020, 10, 1699. https://doi.org/10.3390/biom10121699
Muscella A, Stefàno E, Lunetti P, Capobianco L, Marsigliante S. The Regulation of Fat Metabolism during Aerobic Exercise. Biomolecules. 2020; 10(12):1699. https://doi.org/10.3390/biom10121699
Chicago/Turabian StyleMuscella, Antonella, Erika Stefàno, Paola Lunetti, Loredana Capobianco, and Santo Marsigliante. 2020. "The Regulation of Fat Metabolism during Aerobic Exercise" Biomolecules 10, no. 12: 1699. https://doi.org/10.3390/biom10121699
APA StyleMuscella, A., Stefàno, E., Lunetti, P., Capobianco, L., & Marsigliante, S. (2020). The Regulation of Fat Metabolism during Aerobic Exercise. Biomolecules, 10(12), 1699. https://doi.org/10.3390/biom10121699







