Vitamin B12b Enhances the Cytotoxicity of Diethyldithiocarbamate in a Synergistic Manner, Inducing the Paraptosis-Like Death of Human Larynx Carcinoma Cells
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Cell Culture
2.3. Cytotoxicity Assay and Drug Treatment
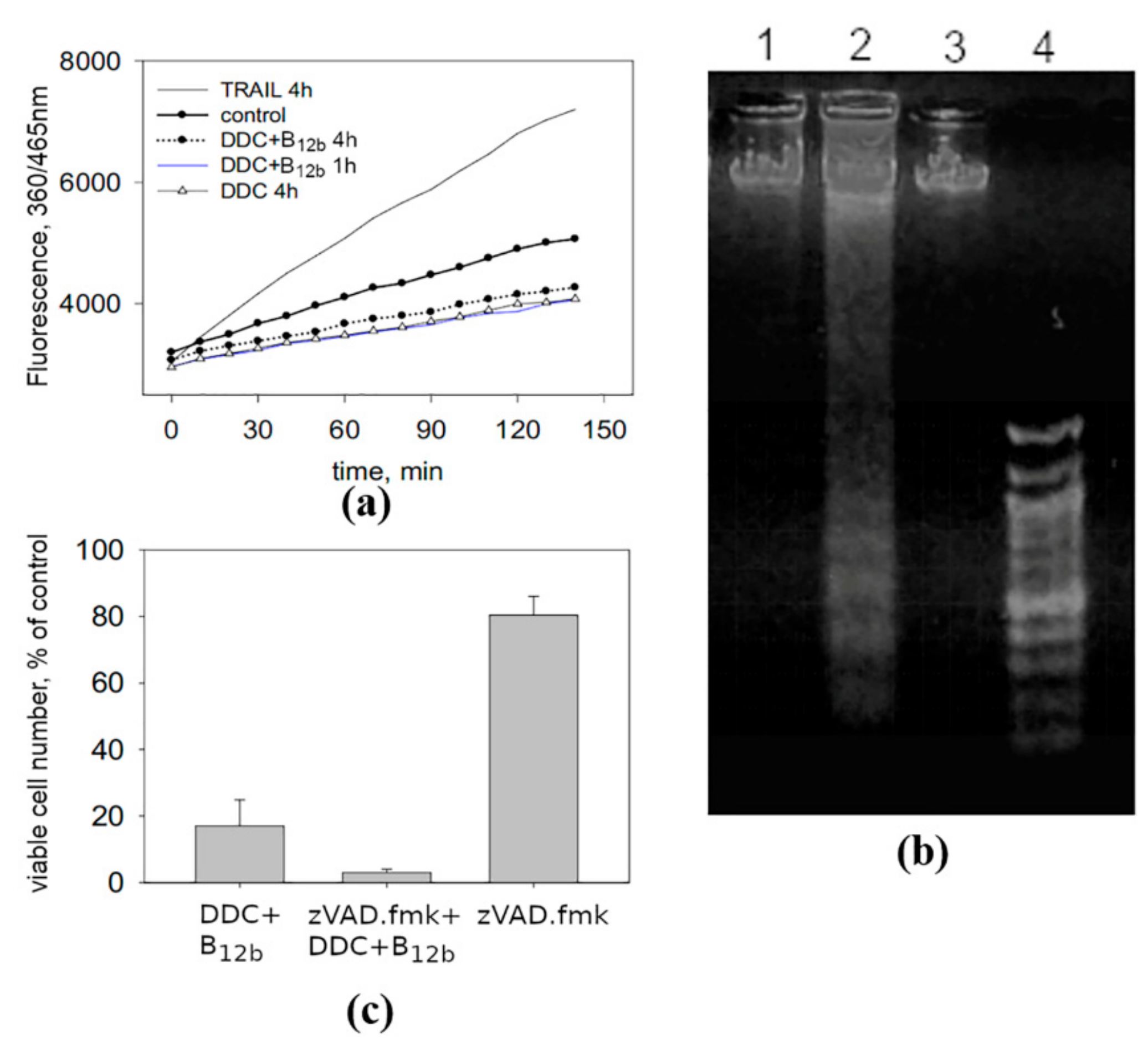
2.4. DNA Fragmentation Assay
2.5. Assay of the Activity of Caspase-3
2.6. Transmission Electron Microscopy
2.7. Fluorescent Confocal Microscopy
2.8. Flow Cytometry Assay
2.9. Ca2+-Retention Capacity of Mitochondria in Permeabilized Cells
2.10. Measurement of Intracellular ATP
2.11. GSH/GSSG Assay
2.12. mBCl Assay
2.13. Immunoblotting
2.14. Statistical Analysis
3. Results
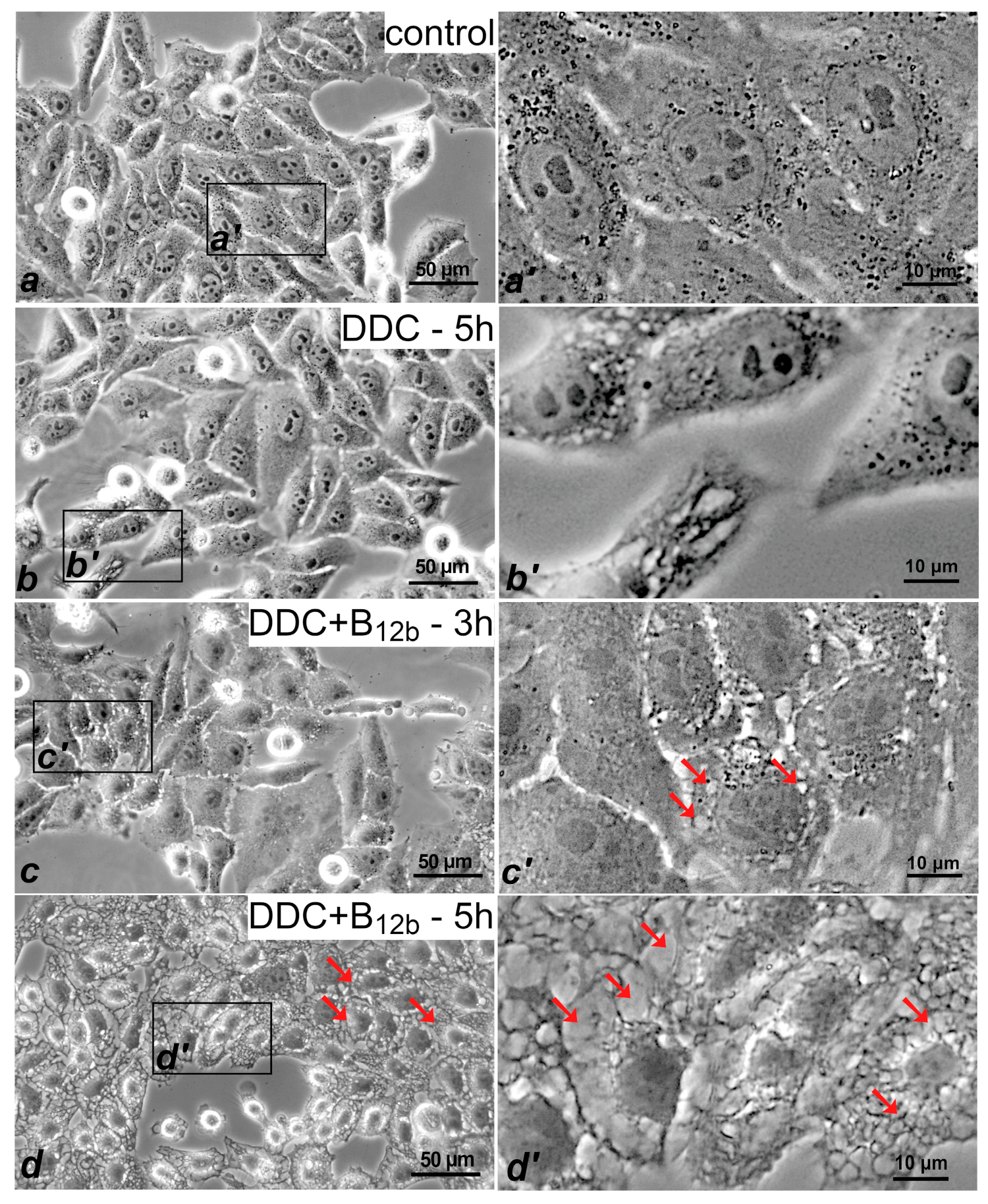
3.1. Vacuolization of the Cytoplasm and the Absence of the Signs of Apoptosis and Necrosis Upon the Initiation of Cell Death by the Combination DDC + B12b
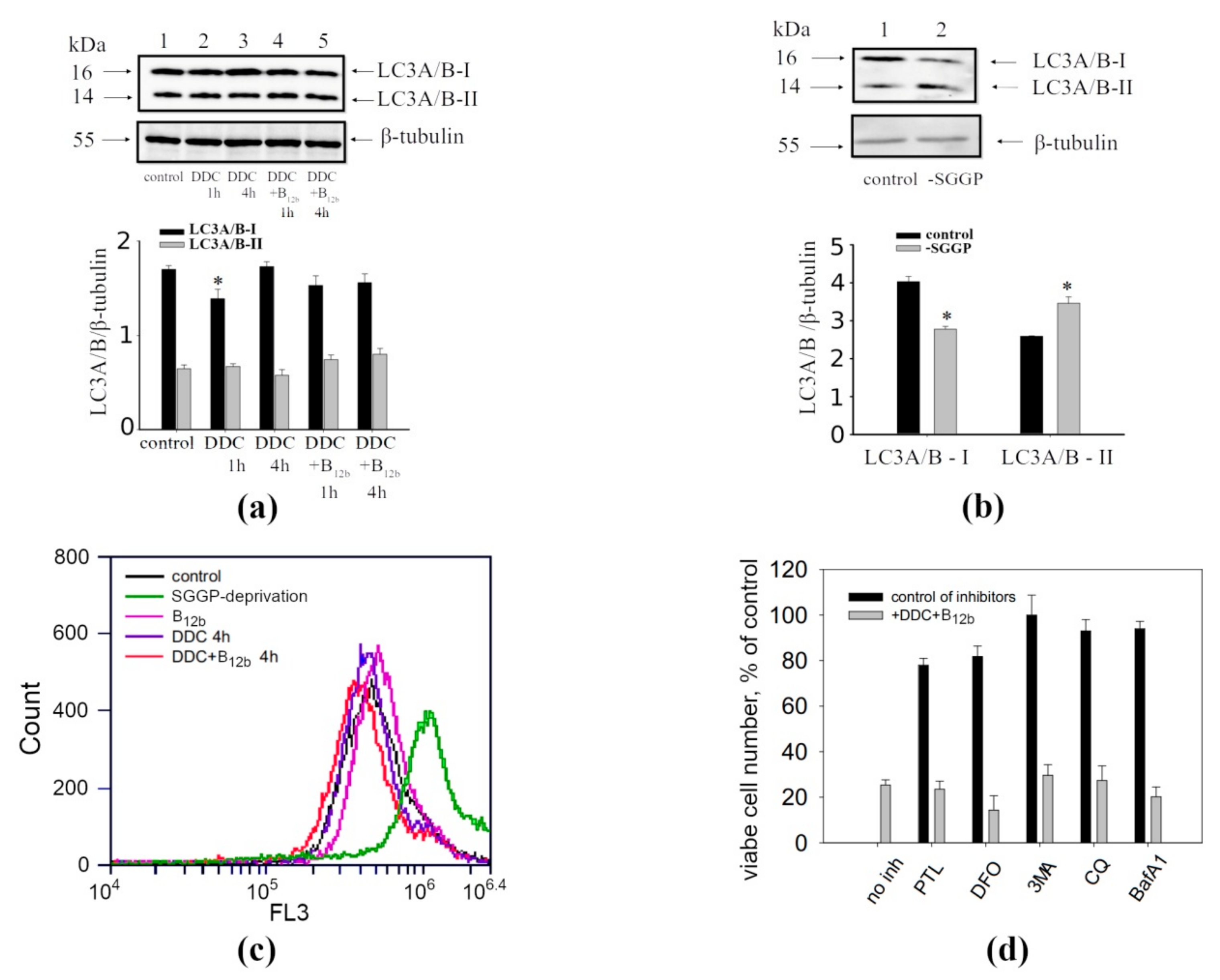
3.2. Absence of the Signs of Autophagy During Cell Vacuolization Induced by DDC + B12b
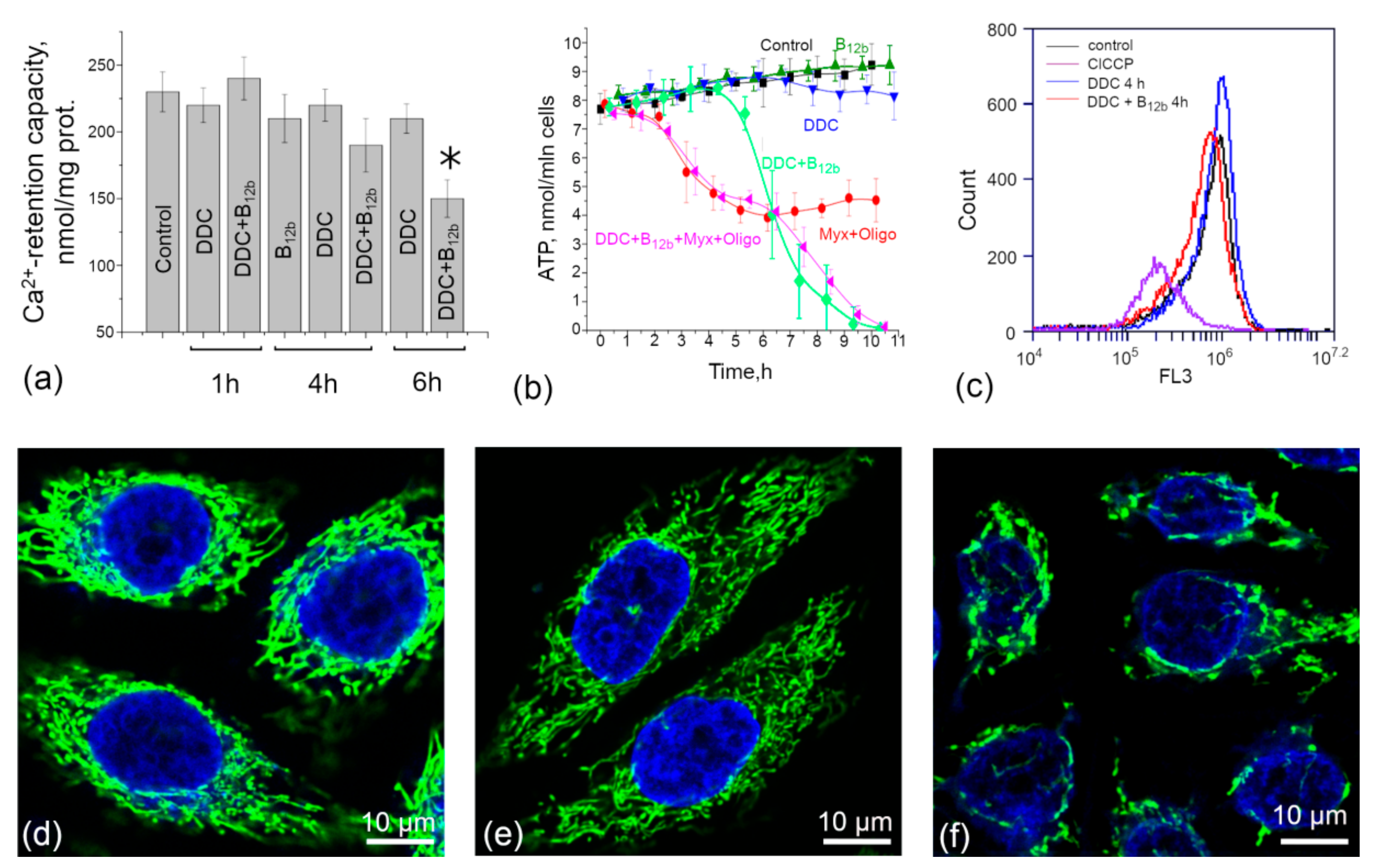
3.3. Maintenance of the Functional State of Mitochondria During the Initiation of Cell Death by DDC + B12b
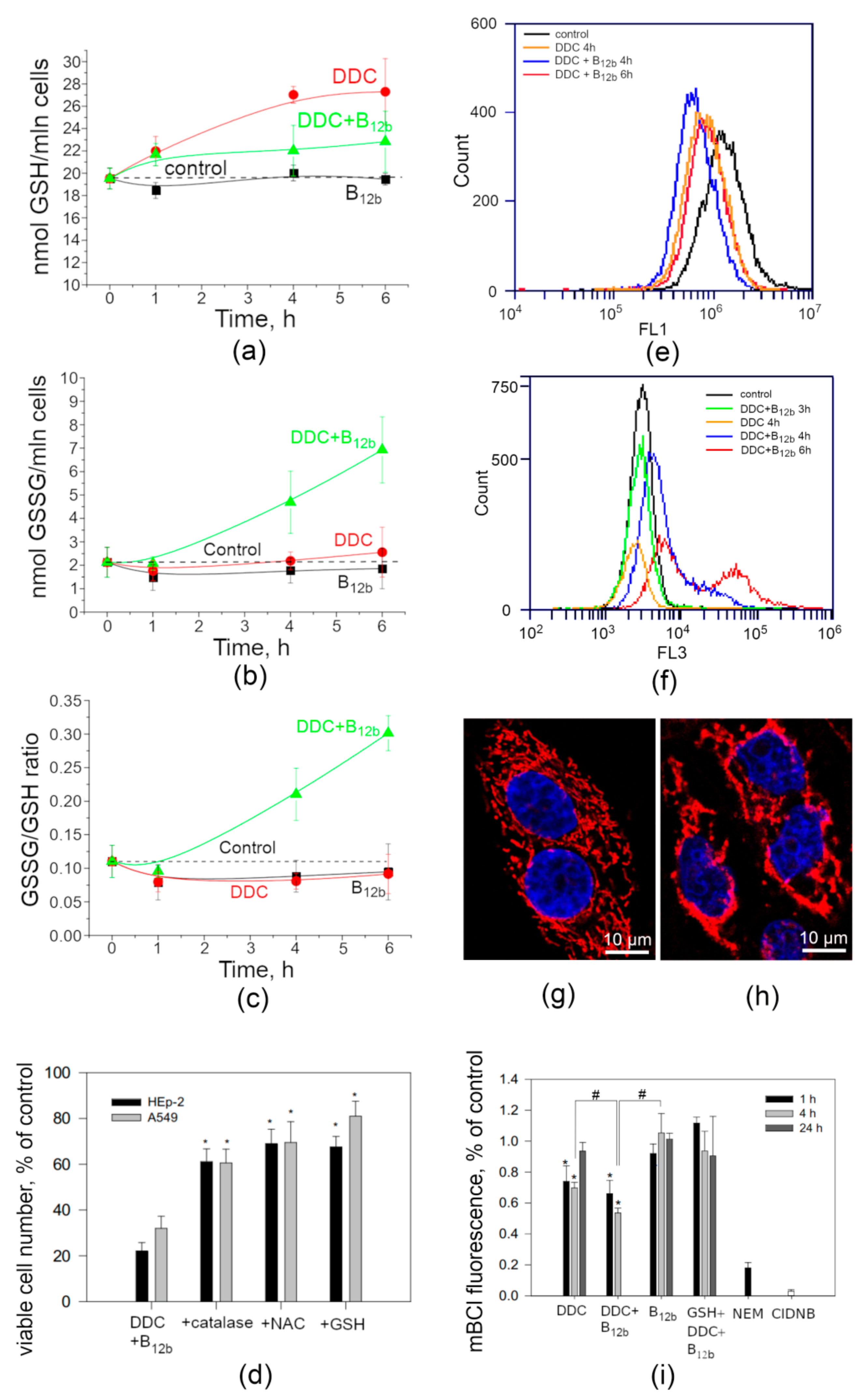
3.4. Intracellular Redox Balance During the Initiation of Cell Death by DDC + B12b
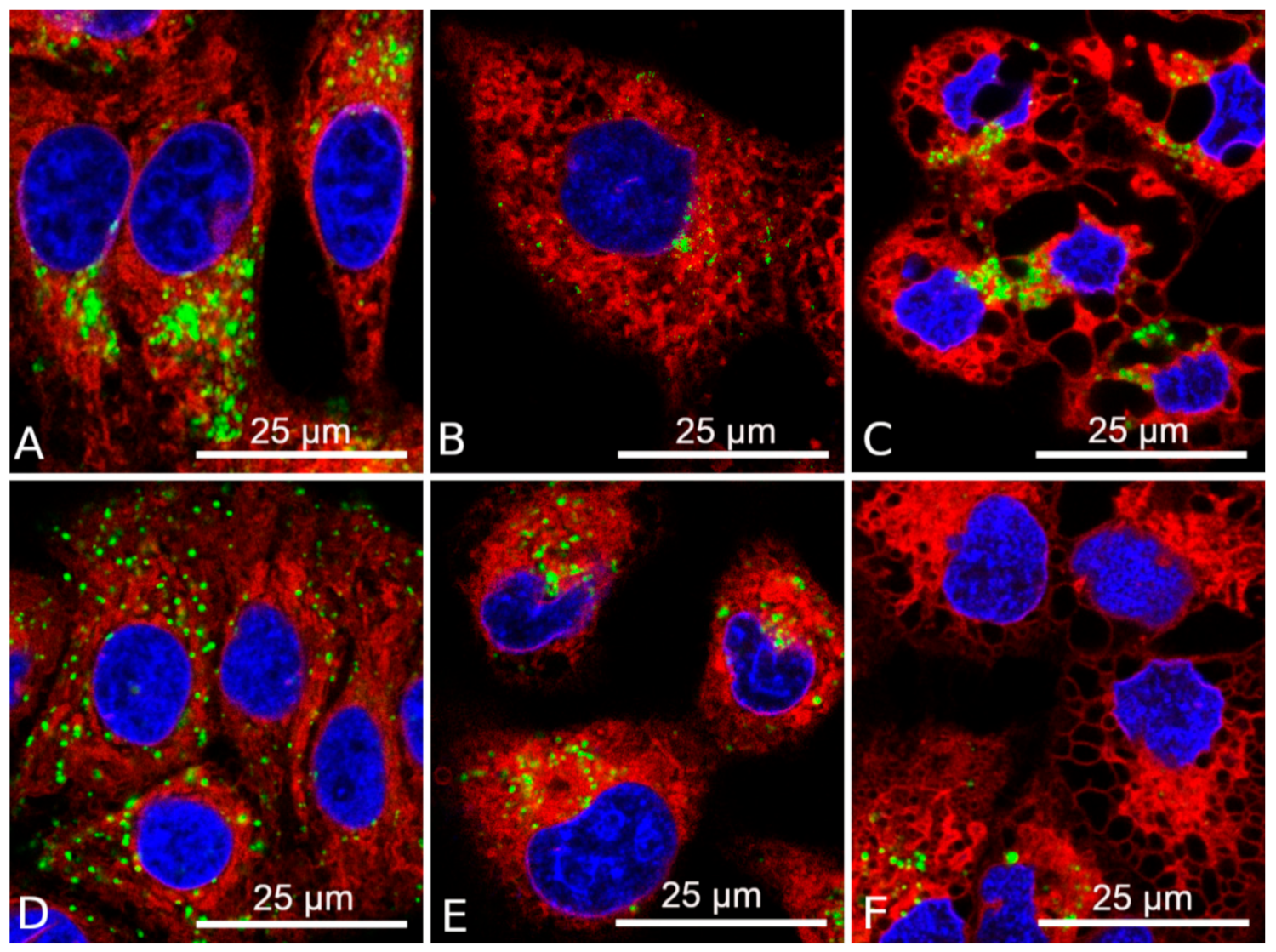
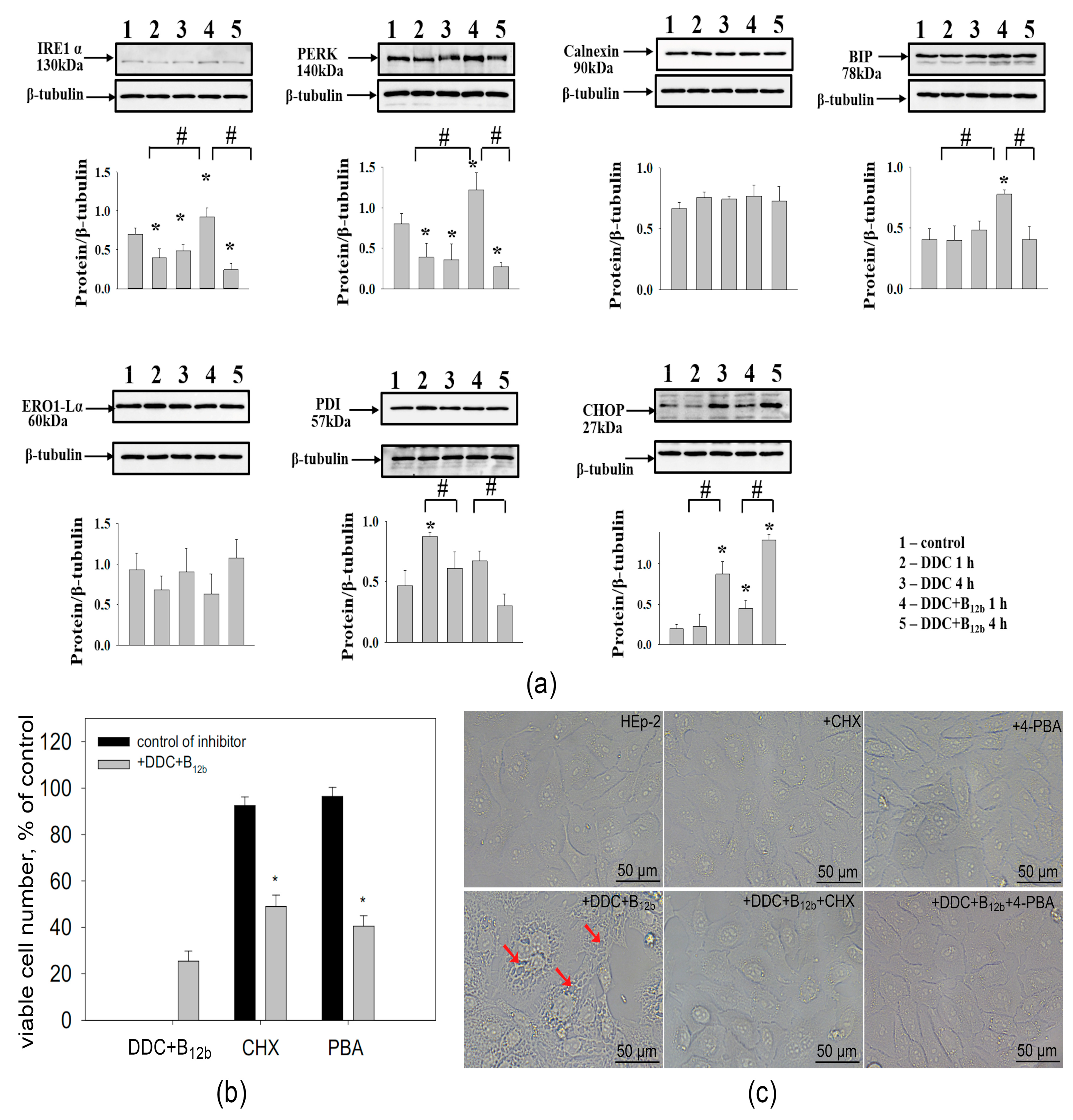
3.5. Combination DDC+B12b Induces ER Stress During the Initiation of Cell Death
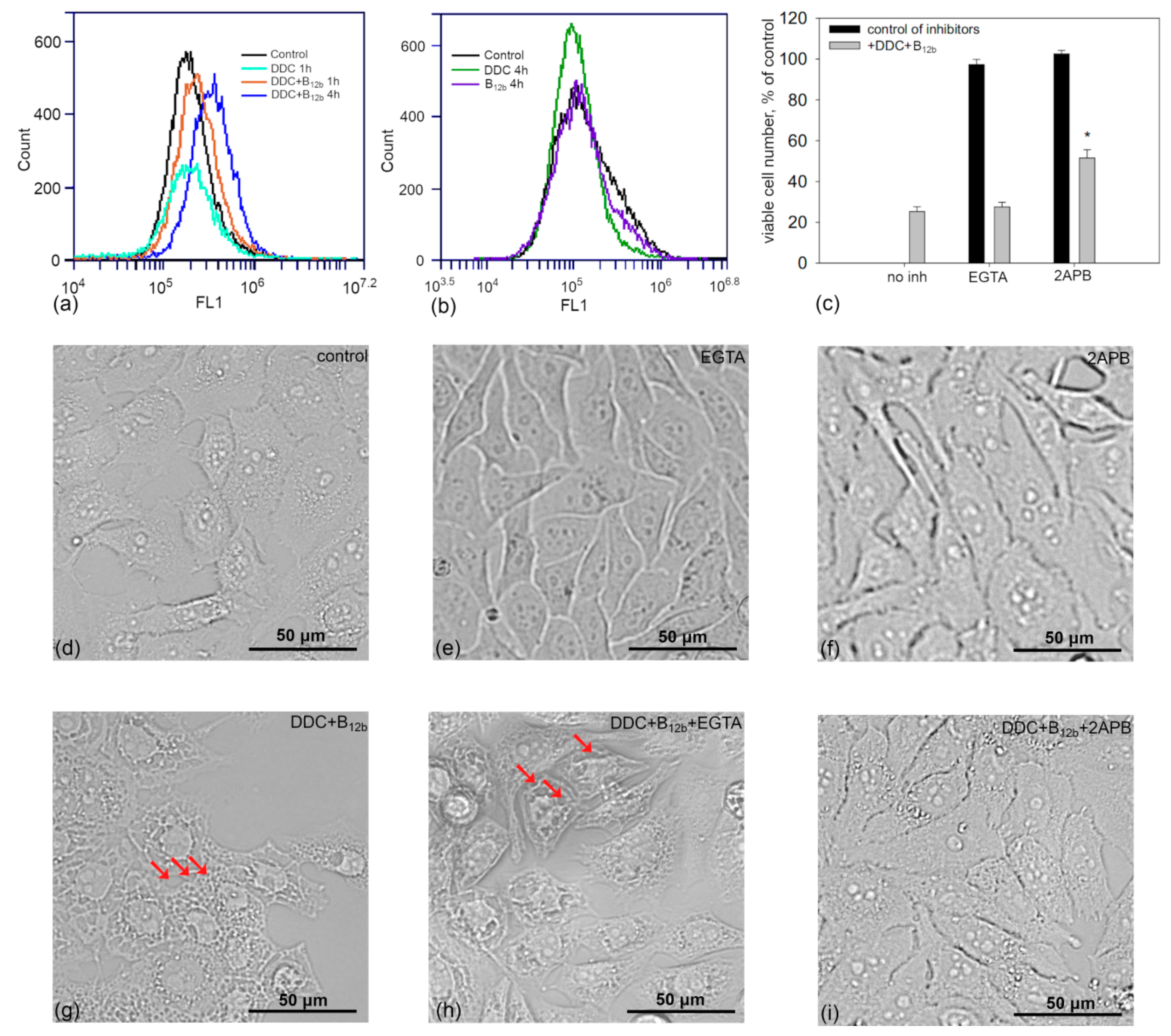
3.6. Increase in the Concentration of Intracellular Calcium During the Initiation of Cell Death by DDC + B12b
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hendren, R.L.; James, S.J.; Widjaja, F.; Lawton, B.; Rosenblatt, A.; Bent, S. Randomized, Placebo-Controlled Trial of Methyl B12 for Children with Autism. J. Child. Adolesc. Psychopharmacol. 2016, 26, 774–783. [Google Scholar] [CrossRef] [PubMed]
- Thompson, J.P.; Marrs, T.C. Hydroxocobalamin in cyanide poisoning. Clin. Toxicol. 2012, 50, 875–885. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Hodgson, N.W.; Trivedi, M.S.; Abdolmaleky, H.M.; Fournier, M.; Cuenod, M.; Do, K.Q.; Deth, R.C. Decreased brain levels of vitamin B12 in aging, autism and schizophrenia. PLoS ONE 2016. [Google Scholar] [CrossRef] [PubMed]
- Shah, P.R.; Reynolds, P.S.; Pal, N.; Tang, D.; McCarthy, H.; Spiess, B.D. Hydroxocobalamin for the treatment of cardiac surgery-associated vasoplegia: A case series. Canad. J. Anaesth. 2017, 65, 560–568. [Google Scholar] [CrossRef] [PubMed]
- Chan, W.; Almasieh, M.; Catrinescu, M.-M.; Levin, L.A. Cobalamin-associated superoxide scavenging in neuronal cells is a potential mechanism for Vitamin B12–deprivation optic neuropathy. Amer. J. Pathol. 2018, 188, 160–172. [Google Scholar] [CrossRef] [PubMed]
- Kucherenko, P.N.; Salnikov, D.S.; Bui, T.T.; Makarov, S.V. Interaction of aquacobalamin and diaquacobinamide with cyanamide. Macroheterocycles 2013, 6, 262–267. [Google Scholar] [CrossRef][Green Version]
- Marguerite, V.; Gkikopoulou, E.; Alberto, J.M.; Guéant, J.L.; Merten, M. Phospholipase D activation mediates cobalamin-induced downregulation of Multidrug Resistance-1 gene and increase in sensitivity to vinblastine in HepG2 cells. Int. J. Biochem. Cell Biol. 2013, 45, 213–220. [Google Scholar] [CrossRef]
- Waibel, R.; Treichler, H.; Schaefer, N.G.; Van Staveren, D.R.; Mundwiler, S.; Kunze, S.; Küenzi, M.; Alberto, R.; Nüesch, J.; Knuth, A.; et al. New derivatives of Vitamin B12 show preferential targeting of tumors. Cancer Res. 2008, 68, 2904–2911. [Google Scholar] [CrossRef]
- Walker, P.R.; Smith, B.; Carson, C.; LeBlanc, J.; Sikorska, M.; Woodhouse, C.S.; Morgan, A.C. Induction of apoptosis in neoplastic cells by depletion of vitamin B12. Cell Death Diff. 1997, 4, 233–241. [Google Scholar] [CrossRef]
- Tang, Z.; Bauer, J.A.; Morrison, B.; Lindner, D.J. Nitrosylcobalamin promotes cell death via S nitrosylation of Apo2L/TRAIL receptor DR4. Mol. Cell Biol. 2006, 26, 5588–5594. [Google Scholar] [CrossRef]
- Ruiz-Sánchez, P.; König, C.; Ferrari, S.; Alberto, R. Vitamin B12 as a carrier for targeted platinum delivery: In vitro cytotoxicity and mechanistic studies. J. Biol. Inorg. Chem. 2011, 16, 33. [Google Scholar] [CrossRef] [PubMed]
- Mutti, E.; Hunger, M.; Fedosov, S.; Nexo, E.; Kräutler, B. Organometallic DNA-B12 conjugates as potential oligonucleotide vectors: Synthesis and structural and binding studies with human cobalamin-transport proteins. Chembiochem 2017, 18, 2280–2291. [Google Scholar] [CrossRef] [PubMed]
- Vol’pin, M.E.; Krainova, N.Y.; Levitin, I.Y.; Mityaeva, Z.Y.; Novodarova, G.N.; Oganezov, V.K.; Chissov, V.I.; Yakubovskaya, R.I. Combinations of vitamin B12 with ascorbic acid as potential antitumor agents. Russ. Chem. J. 1998, 42, 116–127. [Google Scholar]
- Jacobsen, D.W.; Troxell, L.S.; Brown, K.L. Catalysis of thiol oxidation by cobalamins and cobinamides: Reaction products and kinetics. Biochemistry 1984, 23, 2017–2025. [Google Scholar] [CrossRef]
- Ramasamy, S.; Kundu, T.K.; Antholine, W.; Manoharan, P.T.; Rifkind, J.M. Internal spin trapping of thiyl radical during the complexation and reduction of cobalamin with glutathione and dithiothrietol. J. Porphyr. Phthalocyanines 2012, 16, 25–38. [Google Scholar] [CrossRef]
- Akatov, V.S.; Evtodienko, Y.V.; Leshchenko, V.V.; Teplova, V.V.; Potselueva, M.M.; Kruglov, A.G.; Lezhnev, E.I.; Yakubovskaya, R.I. Combined vitamins B12b and C induce the glutathione depletion and the death of epidermoid human larynx carcinoma cells HEp-2. Biosci. Rep. 2000, 20, 411–417. [Google Scholar] [CrossRef]
- Solovieva, M.E.; Soloviev, V.V.; Akatov, V.S. Vitamin B12b increases the cytotoxicity of short-time exposure to ascorbic acid, inducing oxidative burst and iron-dependent DNA damage. Eur. J. Pharmacol. 2007, 566, 206–214. [Google Scholar] [CrossRef]
- Solovieva, M.E.; Solovyev, V.V.; Kudryavtsev, A.A.; Trizna, Y.A.; Akatov, V.S. Vitamin B12b enhances the cytotoxicity of dithiothreitol. Free Rad. Biol. Med. 2008, 44, 1846–1856. [Google Scholar] [CrossRef]
- Solovyeva, M.E.; Faskhutdinova, A.A.; Solovyev, V.V.; Akatov, V.S. Thiol antioxidants in combination with vitamin B12 induce apoptotic death of human lymphocytic leukemia cells by destabilization of lysosomes with the involvement of iron ions. B. Exp. Biol. Med. 2013, 154, 449–452. [Google Scholar] [CrossRef]
- Solovieva, M.E.; Shatalin, Y.V.; Solovyev, V.V.; Sazonov, A.V.; Kutyshenko, V.P.; Akatov, V.S. Hydroxycobalamin catalyzes the oxidation of diethyldithiocarbamate and increases its cytotoxicity independently of copper ions. Redox Biol. 2019, 20, 28–37. [Google Scholar] [CrossRef]
- Brar, S.S.; Grigg, C.; Wilson, K.S.; Holder, W.D., Jr.; Dreau, D.; Austin, C.; Foster, M.; Ghio, A.J.; Whorton, A.R.; Stowell, G.W.; et al. Disulfiram inhibits activating transcription factor/cyclic AMP-responsive element binding protein and human melanoma growth in a metal-dependent manner in vitro, in mice and in a patient with metastatic disease. Mol. Cancer Ther. 2004, 3, 1049–1060. [Google Scholar] [PubMed]
- Rae, C.; Tesson, M.; Babich, J.W.; Boyd, M.; Sorensen, A.; Mairs, R.J. The role of copper in disulfiram-induced toxicity and radiosensitization of cancer cells. J. Nucl. Med. 2013. [Google Scholar] [CrossRef] [PubMed]
- Allensworth, J.L.; Evans, M.K.; Bertucci, F.; Aldrich, A.J.; Festa, R.A.; Finetti, P.; Ueno, N.T.; Safi, R.; McDonnell, D.P.; Thiele, D.J.; et al. Disulfiram (DSF) acts as a copper ionophore to induce copper-dependent oxidative stress and mediate anti-tumor efficacy in inflammatory breast cancer. Mol. Oncol. 2015, 9, 1155–1168. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.; Cui, Q.C.; Yang, H.; Dou, Q.P. Disulfiram, a clinically used anti-alcoholism drug and copper-binding agent, induces apoptotic cell death in breast cancer cultures and xenografts via inhibition of the proteasome activity. Cancer Res. 2006, 66, 10425–10433. [Google Scholar] [CrossRef]
- Erl, W.; Weber, C.; Hansson, G.K. Pyrrolidine dithiocarbamate-induced apoptosis depends on cell type, density, and the presence of Cu(2+) and Zn(2+). Am. J. Physiol. Cell Physiol. 2000, 278, C1116–C1125. [Google Scholar] [CrossRef]
- Daniel, K.G.; Chen, D.; Yan, B.; Dou, Q.P. Copper-binding compounds as proteasome inhibitors and apoptosis inducers in human cancer. Front Biosci. 2007, 12, 135–144. [Google Scholar] [CrossRef]
- Wu, X.; Xue, X.; Wang, L.; Wang, W.; Han, J.; Sun, X.; Zhang, H.; Liu, Y.; Che, X.; Yang, J.; et al. Suppressing autophagy enhances disulfiram/copper-induced apoptosis in non-small cell lung cancer. Eur. J. Pharmacol. 2018, 827, 1–12. [Google Scholar] [CrossRef]
- Ali, I.; Wania, W.A.; Saleem, K.; Hseih, M.-F. Design and synthesis of thalidomide based dithiocarbamate Cu(II), Ni(II) and Ru(III) complexes as anticancer agents. Polyhedron 2013, 56, 134–143. [Google Scholar] [CrossRef]
- Altaf, M.; Monim-ul-Mehboob, M.; Kawde, A.-N.; Corona, G.; Larcher, R.; Ogasawara, M.; Casagrande, N.; Celegato, M.; Borghese, C.; Siddik, Z.H.; et al. New bipyridine gold(III) dithiocarbamate-containing complexes exerted a potent anticancer activity against cisplatin-resistant cancer cells independent of p53 status. Oncotarget 2017, 8, 490–505. [Google Scholar] [CrossRef]
- Skrott, Z.; Mistrik, M.; Andersen, K.K.; Friis, S.; Majera, D.; Gursky, J.; Ozdian, T.; Bartkova, J.; Turi, Z.; Moudry, P.; et al. Alcohol-abuse drug disulfiram targets cancer via p97 segregase adaptor NPL4. Nature 2017, 552, 194–199. [Google Scholar] [CrossRef]
- Chou, T.C.; Talalay, P. Quantitative analysis of dose-effect relationships: The combined effects of multiple drugs or enzyme inhibitors. Adv. Enzyme Regul. 1984, 22, 27–55. [Google Scholar] [CrossRef]
- Filippi-Chiela, E.C.; Villodre, E.S.; Zamin, L.L.; Lenz, G. Autophagy interplay with apoptosis and cell cycle regulation in the growth inhibiting effect of resveratrol in glioma cells. PLoS ONE 2011, 6, e20849. [Google Scholar] [CrossRef] [PubMed]
- Kauffman, M.; Kauffman, M.; Traore, K.; Zhu, H.; Trush, M.; Jia, Z.; Li, Y. MitoSOX-based flow cytometry for detecting mitochondrial ROS. React. Oxyg. Species 2016, 2, 361–370. [Google Scholar] [CrossRef] [PubMed]
- Kharechkina, E.S.; Nikiforova, A.B.; Teplova, V.V.; Odinokova, I.V.; Krestinina, O.V.; Baburina, Y.L.; Kruglova, S.A.; Kruglov, A.G. Regulation of permeability transition pore opening in mitochondria by external NAD(H). Biochim. Biophys. Acta Gen. Subj. 2019, 1863, 771–783. [Google Scholar] [CrossRef]
- Rahman, I.; Kode, A.; Biswas, S.B. Assay for quantitative determination of glutathione and glutathione disulfide levels using enzymatic recycling method. Nat. Protoc. 2006, 1, 3159–3165. [Google Scholar] [CrossRef]
- Barhoumi, R.; Bailey, R.H.; Burghardt, R.C. Kinetic analysis of glutathione in anchored cells with monochlorobimane. Cytometry 1995, 19, 226. [Google Scholar] [CrossRef]
- Li, L.; Chen, Y.; Gibson, S.B. Starvation-induced autophagy is regulated by mitochondrial reactive oxygen species leading to AMPK activation. Cell. Signal. 2013, 25, 50–65. [Google Scholar] [CrossRef]
- Ye, Z.W.; Zhang, J.; Ancrum, T.; Manevich, Y.; Townsend, D.M.; Tew, K.D. Glutathione S-Transferase P-mediated protein s-glutathionylation of resident endoplasmic reticulum proteins influences sensitivity to drug-induced unfolded protein response. Antioxid. Redox Signal. 2017, 26, 247–261. [Google Scholar] [CrossRef]
- Maltese, W.A.; Overmeyer, J.H. Methuosis. Nonapoptotic cell death associated with vacuolization of macropinosome and endosome compartments. Am. J. Pathol. 2014, 184, 1630e1642. [Google Scholar] [CrossRef]
- Antunes, F.; Cadenas, E.; Brunk, U.T. Apoptosis induced by exposure to a low steady-state concentration of H2O2 is a consequence of lysosomal rupture. Biochem. J. 2001, 356, 549–555. [Google Scholar] [CrossRef]
- Kolb, P.S.; Ayaub, E.A.; Zhou, W.; Yum, V.; Dickhout, J.G.; Ask, K. The therapeutic effects of 4-phenylbutyric acid in maintaining proteostasis. Int. J. Biochem. Cell Biol. 2015, 61, 45–52. [Google Scholar] [CrossRef] [PubMed]
- Dumay, A.; Rincheval, V.; Trotot, P.; Mignotte, B.; Vayssière, J.L. The superoxide dismutase inhibitor diethyldithiocarbamate has antagonistic effects on apoptosis by triggering both cytochrome c release and caspase inhibition. Free Radic. Biol. Med. 2006, 40, 1377–1390. [Google Scholar] [CrossRef] [PubMed]
- Brüning, A.; Kast, R.E. Oxidizing to death. Disulfiram for cancer cell killing. Cell Cycle. 2014, 13, 1513–1514. [Google Scholar] [CrossRef] [PubMed]
- Kanno, S.; Matsukawa, E.; Miura, A.; Shouji, A.; Asou, K.; Ishikawa, M. Diethyldithiocarbamate-induced cytotoxicity and apoptosis in leukemia cell lines. Biol. Pharm. Bull. 2003, 26, 964–968. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Tardito, S.; Bassanetti, I.; Bignardi, C.; Elviri, L.; Tegoni, M.; Mucchino, C.; Bussolati, O.; Franchi-Gazzola, R.; Marchiò, L. Copper binding agents acting as copper ionophores lead to caspase inhibition and paraptotic cell death in human cancer cells. J. Am. Chem. Soc. 2011, 133, 6235–6242. [Google Scholar] [CrossRef]
- Nobel, C.S.; Kimland, M.; Nicholson, D.W.; Orrenius, S.; Slater, A.F. Disulfiram is a potent inhibitor of proteases of the caspase family. Chem. Res. Toxicol. 1997, 10, 1319–1324. [Google Scholar] [CrossRef]
- Ploemen, J.P.; van Iersel, M.L.; Wormhoudt, L.W.; Commandeur, J.N.; Vermeulen, N.P.; van Bladeren, P.J. In vitro inhibition of rat and human glutathione S-transferase isoenzymes by disulfiram and diethyldithiocarbamate. Biochem. Pharmacol. 1996, 52, 197–204. [Google Scholar] [CrossRef]
- Zhang, H.; Nakajima, S.; Kato, H.; Gu, L.; Yoshitomi, T.; Nagai, K.; Shinmori, H.; Kokubo, S.; Kitamura, M. Selective, potent blockade of the IRE1 and ATF6 pathways by 4-phenylbutyric acid analogues. Br. J. Pharmacol. 2013, 170, 822–834. [Google Scholar] [CrossRef]
- O’Brien, P.S.; Xi, Y.; Miller, J.R.; Brownell, A.L.; Zeng, Q.; Yoo, G.H.; Garshott, D.M.; O’Brien, M.B.; Galinato, A.E.; Cai, P.; et al. Disulfiram (antabuse) activates ROS-dependent ER stress and apoptosis in oral cavity squamous cell carcinoma. J. Clin. Med. 2019, 8, 611. [Google Scholar] [CrossRef]
- Huang, H.; Liao, Y.; Liu, N.; Hua, X.; Cai, J.; Yang, C.; Long, H.; Zhao, C.; Chen, X.; Lan, X.; et al. Two clinical drugs deubiquitinase inhibitor auranofin and aldehyde dehydrogenase inhibitor disulfiram trigger synergistic anti-tumor effects in vitro and in vivo. Oncotarget 2016, 19, 2796–2808. [Google Scholar] [CrossRef]
- Liu, T.; Wang, P.; Cong, M.; Zhao, X.; Zhang, D.; Xu, H.; Liu, L.; Jia, J.; You, H. Diethyldithiocarbamate, an anti-abuse drug, alleviates steatohepatitis and fibrosis in rodents through modulating lipid metabolism and oxidative stress. Br. J. Pharmacol. 2018, 175, 4480–4495. [Google Scholar] [CrossRef] [PubMed]
- Prentice, H.; Modi, J.P.; Wu, J.-Y. Mechanisms of neuronal protection against excitotoxicity, endoplasmic reticulum stress, and mitochondrial dysfunction in stroke and neurodegenerative diseases. Oxid. Med. Cell. Longev. 2015, 2015, 964518. [Google Scholar] [CrossRef] [PubMed]
- Southwood, C.M.; Fykkolodziej, B.; Maheras, K.J.; Garshott, D.M.; Estill, M.; Fribley, A.M.; Gow, A. Overexpression of CHOP in myelinating cells does not confer a significant phenotype under normal or metabolic stress conditions. J. Neurosci. 2016, 36, 6803–6819. [Google Scholar] [CrossRef] [PubMed]
- Nagendra, S.N.; Rao, K.M.; Subhash, M.N.; Shetty, K.T. Disulfiram lowers Ca 2+, Mg2+-ATPase activity of rat brain synaptosomes. Neurochem. Res. 1994, 19, 1509–1513. [Google Scholar] [CrossRef]
- Bury, M.; Girault, A.; Megalizzi, V.l.; Spiegl-Kreinecker, S.; Mathieu, V.; Berger, W.; Evidente, A.; Kornienko, A.; Gailly, P.; Vandier, C.; et al. Ophiobolin A induces paraptosis-like cell death in human glioblastoma cells by decreasing BKCa channel activity. Cell Death Dis. 2013, 4, e561. [Google Scholar] [CrossRef]
- Yoon, M.J.; Lee, A.R.; Jeong, S.A.; Kim, Y.-S.; Kim, J.-Y.; Kwon, Y.-J.; Choi, K.S. Release of Ca2+ from the endoplasmic reticulum and its subsequent influx into mitochondria trigger celastrol-induced paraptosis in cancer cells. Oncotarget 2014, 5, 6816–6831. [Google Scholar] [CrossRef]
- Seo, M.J.; Lee, D.M.; Kim, I.Y.; Lee, D.; Choi, M.-K.; Lee, J.-Y.; Park, S.S.; Jeong, S.-Y.; Choi, E.K.; Choi, K.S. Gambogic acid triggers vacuolization-associated cell death in cancer cells via disruption of thiol proteostasis. Cell Death Dis. 2019, 10, 187. [Google Scholar] [CrossRef]
- Hempel, S.L.; Buettner, G.R.; O’Malley, Y.Q.; Wessels, D.A.; Flaherty, D.M. Dihydrofluorescein diacetate is superior for detecting intracellular oxidants: Comparison with 2′,7′-dichlorodihydrofluorescein diacetate, 5(and 6)-carboxy-2′,7′-dichlorodihydrofluorescein diacetate, and dihydrorhodamine 123. Free Radic. Biol. Med. 1999, 27, 146–159. [Google Scholar] [CrossRef]




| Cell Line | IC50 for DDC Alone, mM | IC50 for DDC Combined with B12b, mM | Chou-Talalay Combination Index |
|---|---|---|---|
| A549 | 2.47 ± 0.64 * | 0.4 ± 0.05 * | 0.17 |
| A431 | 1.86 ± 0.21 * | 0.43 ± 0.04 * | 0.24 |
| HEp-2 | 2.22 ± 0.24 * | 0.46 ± 0.09 * | 0.22 |
| HT1080 | 4.79 ± 0.94 | 0.5 ± 0.07 | 0.11 |
| HT29 | 2.33 ± 0.42 | 0.53 ± 0.06 | 0.24 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Solovieva, M.; Shatalin, Y.; Fadeev, R.; Krestinina, O.; Baburina, Y.; Kruglov, A.; Kharechkina, E.; Kobyakova, M.; Rogachevsky, V.; Shishkova, E.; et al. Vitamin B12b Enhances the Cytotoxicity of Diethyldithiocarbamate in a Synergistic Manner, Inducing the Paraptosis-Like Death of Human Larynx Carcinoma Cells. Biomolecules 2020, 10, 69. https://doi.org/10.3390/biom10010069
Solovieva M, Shatalin Y, Fadeev R, Krestinina O, Baburina Y, Kruglov A, Kharechkina E, Kobyakova M, Rogachevsky V, Shishkova E, et al. Vitamin B12b Enhances the Cytotoxicity of Diethyldithiocarbamate in a Synergistic Manner, Inducing the Paraptosis-Like Death of Human Larynx Carcinoma Cells. Biomolecules. 2020; 10(1):69. https://doi.org/10.3390/biom10010069
Chicago/Turabian StyleSolovieva, Marina, Yuri Shatalin, Roman Fadeev, Olga Krestinina, Yulia Baburina, Alexey Kruglov, Ekaterina Kharechkina, Margarita Kobyakova, Vadim Rogachevsky, Elena Shishkova, and et al. 2020. "Vitamin B12b Enhances the Cytotoxicity of Diethyldithiocarbamate in a Synergistic Manner, Inducing the Paraptosis-Like Death of Human Larynx Carcinoma Cells" Biomolecules 10, no. 1: 69. https://doi.org/10.3390/biom10010069
APA StyleSolovieva, M., Shatalin, Y., Fadeev, R., Krestinina, O., Baburina, Y., Kruglov, A., Kharechkina, E., Kobyakova, M., Rogachevsky, V., Shishkova, E., & Akatov, V. (2020). Vitamin B12b Enhances the Cytotoxicity of Diethyldithiocarbamate in a Synergistic Manner, Inducing the Paraptosis-Like Death of Human Larynx Carcinoma Cells. Biomolecules, 10(1), 69. https://doi.org/10.3390/biom10010069









