2-Deoxy-d-Glucose-Induced Metabolic Alteration in Human Oral Squamous SCC15 Cells: Involvement of N-Glycosylation of Axl and Met
Abstract
:1. Introduction
2. Results
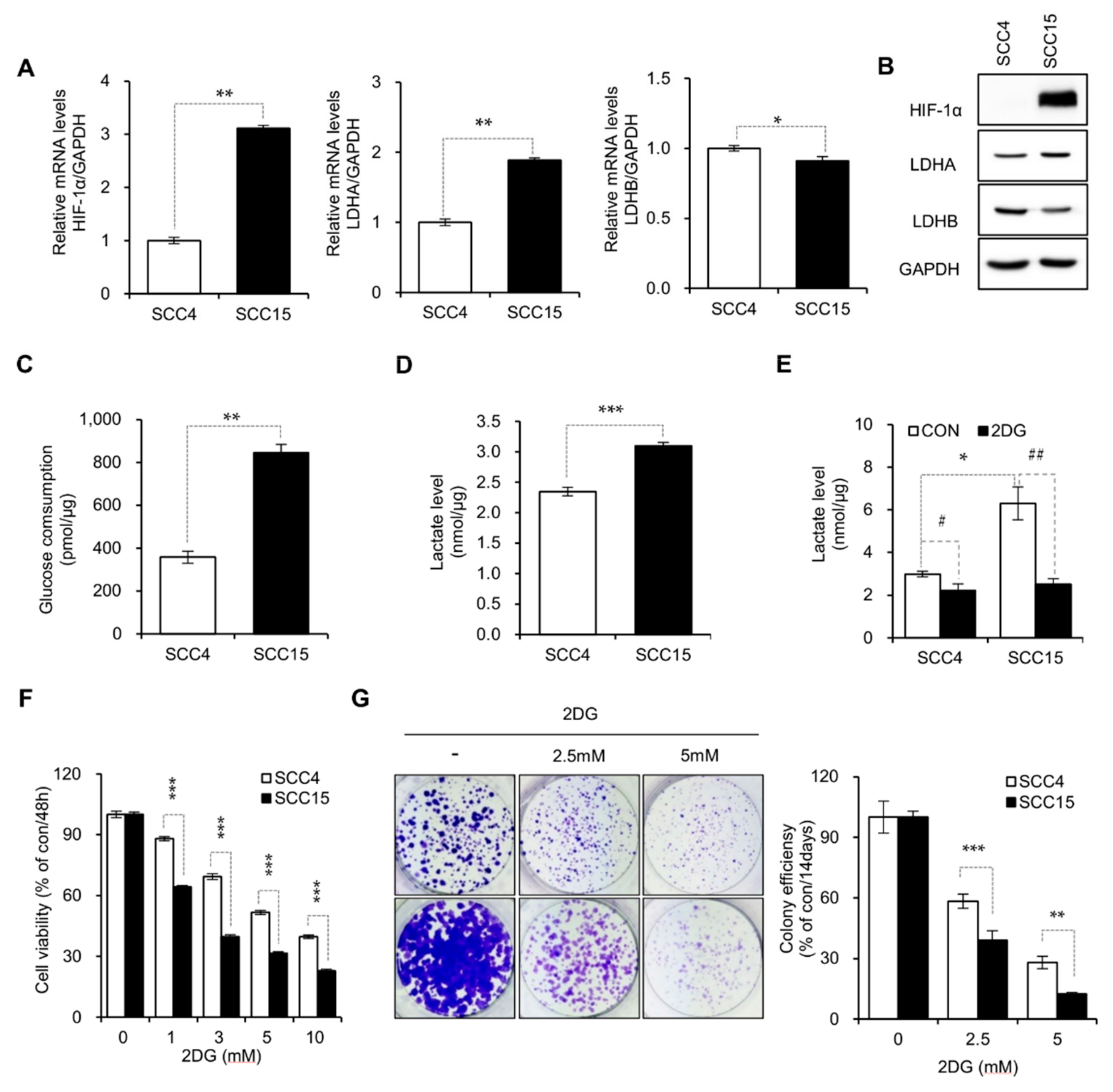
2.1. DG-Induced Inhibition of Aerobic Glycolysis in Oral Cancer Cell Lines
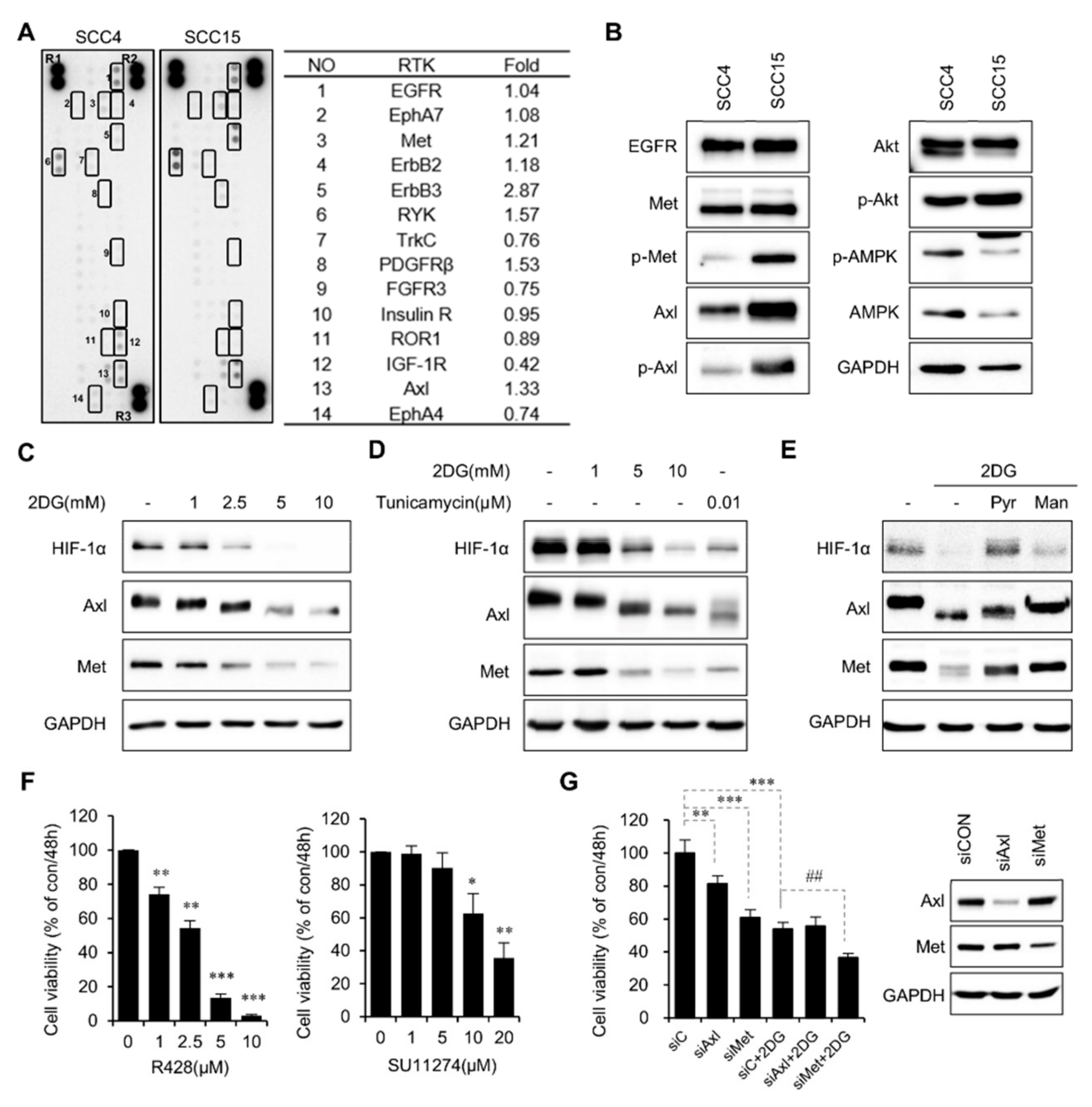
2.2. 2DG-Induced Interference of N-Linked Glycosylation of Axl and Met
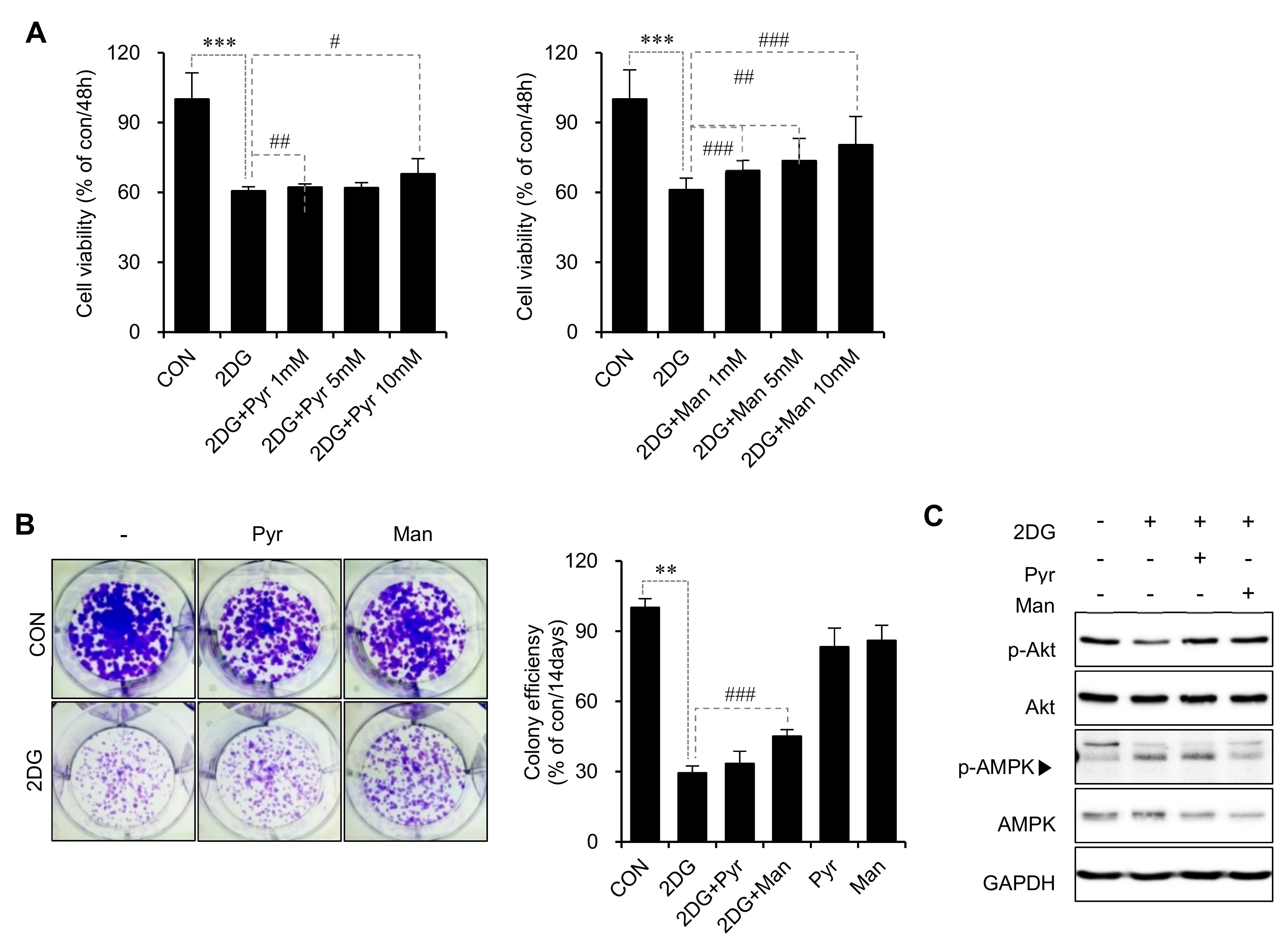
2.3. Inhibition of N-Linked Glycosylation Contributes to the Anticancer Activity of 2DG in SCC15 Cells
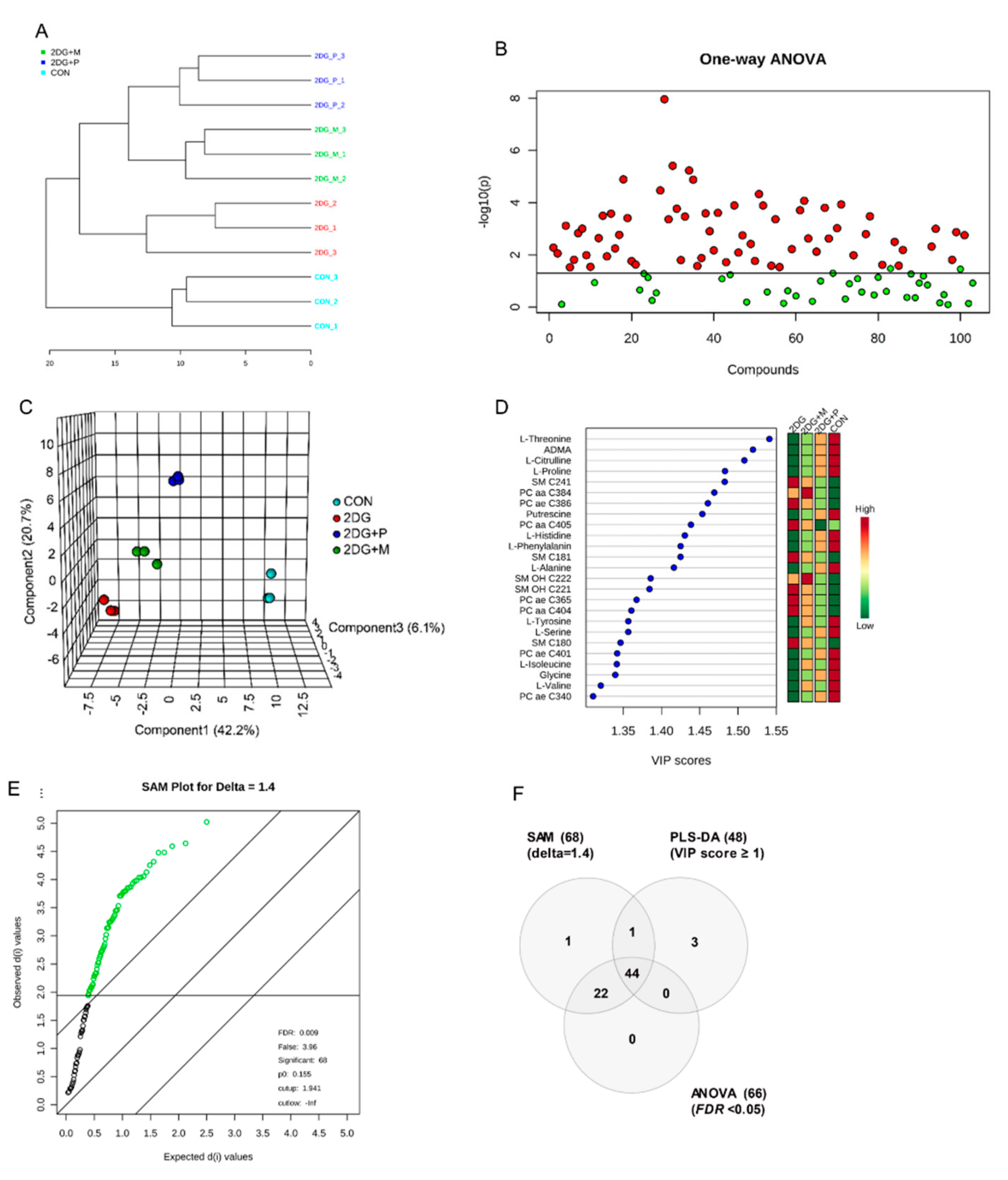
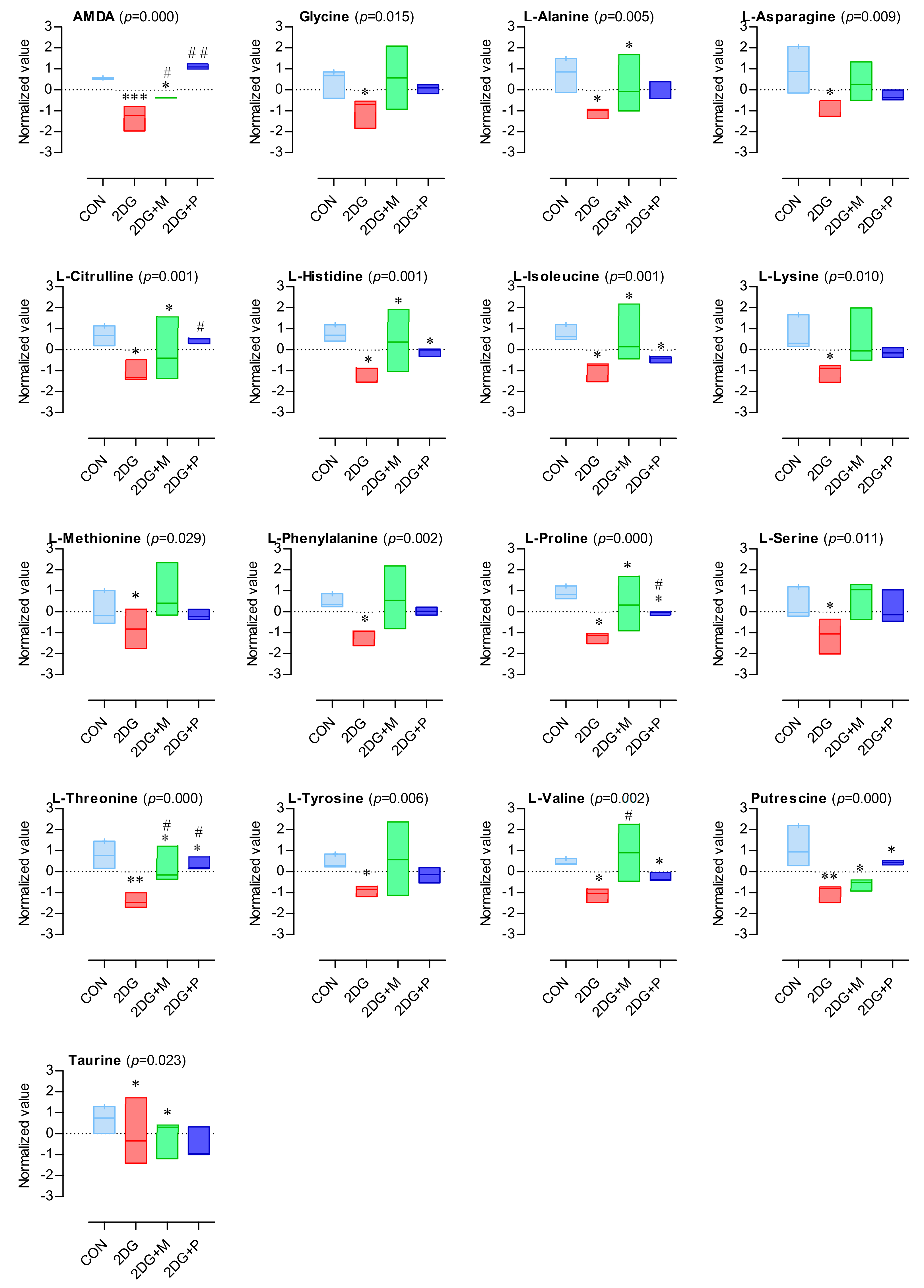
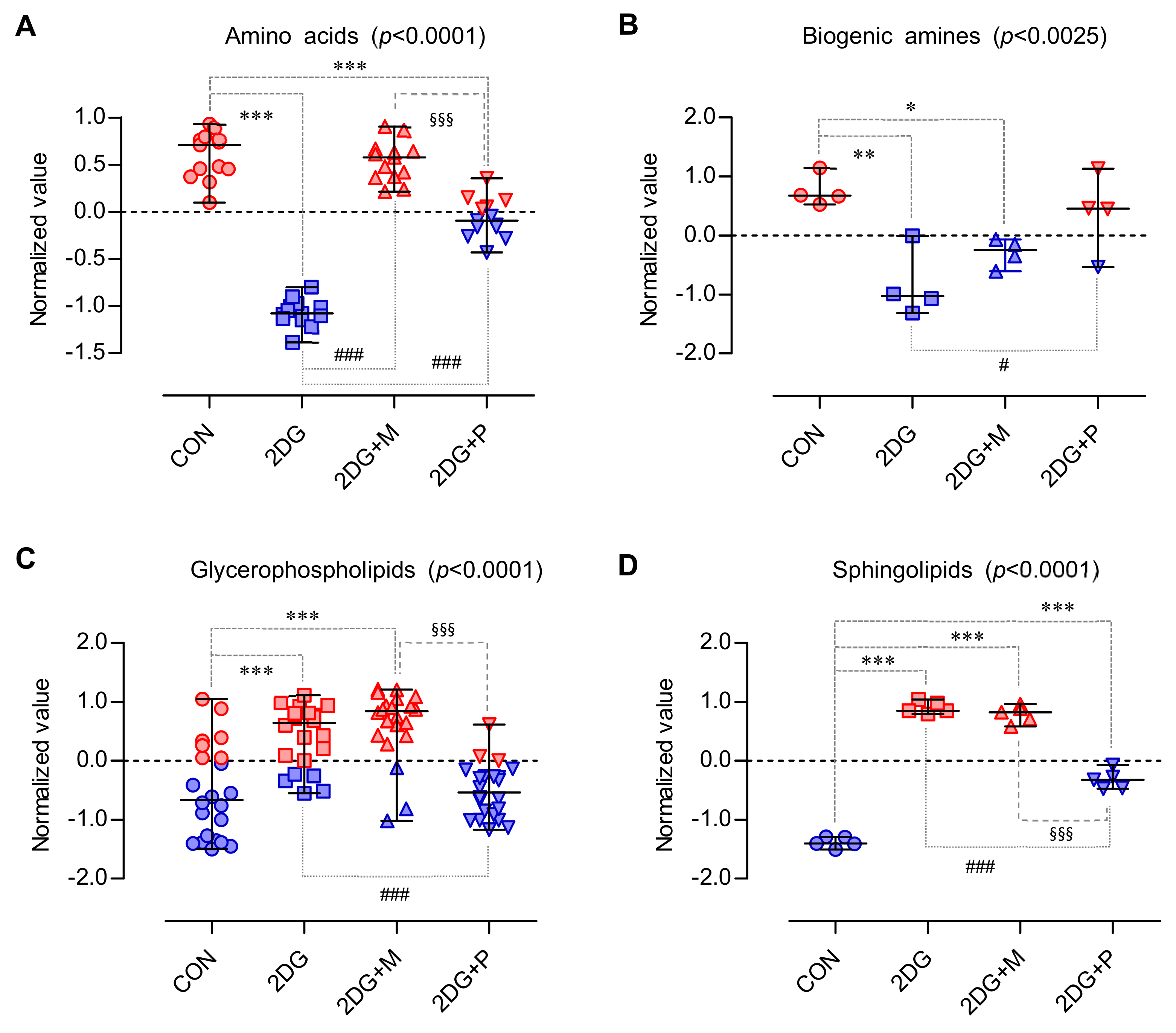
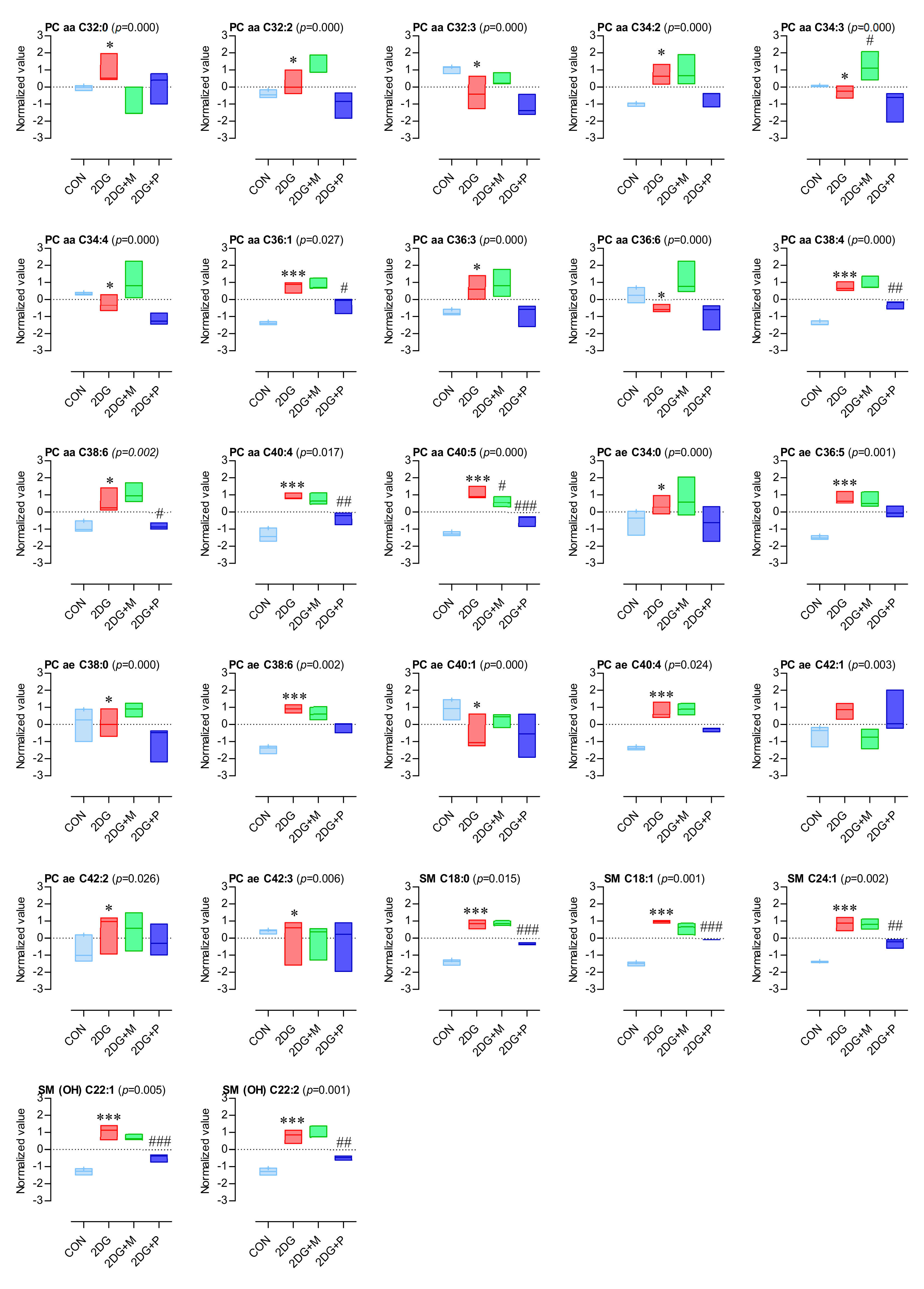
2.4. Metabolic Profiling by Targeted Metabolomics Analysis
2.5. Important Altered Metabolites by 2DG
3. Discussion
4. Materials and Methods
4.1. Chemicals and Reagents
4.2. Real-Time PCR
4.3. Phospho-RTK Array
4.4. Cell Lines and Cell Culture
4.5. Cell Viability Assay
4.6. Colony Formation Assay
4.7. Western Blot Analysis
4.8. RNA Interference
4.9. Measurement of Lactate Production and Glucose Consumption
4.10. Targeted Metabolomics Analysis
4.11. Data Processing and Statistical Analyses
Author Contributions
Funding
Conflicts of Interest
References
- Kroemer, G.; Pouyssegur, J. Tumor cell metabolism: cancer’s Achilles’ heel. Cancer Cell 2008, 13, 472–482. [Google Scholar] [CrossRef] [PubMed]
- Vander Heiden, M.G.; Cantley, L.C.; Thompson, C.B. Understanding the Warburg effect: The metabolic requirements of cell proliferation. Science 2009, 324, 1029–1033. [Google Scholar] [CrossRef] [PubMed]
- Warburg, O. On the origin of cancer cells. Science 1956, 123, 309–314. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Liu, H.; Liu, Z.; Ding, Y.; Ledoux, S.P.; Wilson, G.L.; Voellmy, R.; Lin, Y.; Lin, W.; Nahta, R.; et al. Overcoming trastuzumab resistance in breast cancer by targeting dysregulated glucose metabolism. Cancer Res. 2011, 71, 4585–4597. [Google Scholar] [CrossRef] [PubMed]
- Annibaldi, A.; Widmann, C. Glucose metabolism in cancer cells. Curr. Opin. Clin. Nutr. Metab. Care 2010, 13, 466–470. [Google Scholar] [CrossRef] [PubMed]
- Swietach, P.; Vaughan-Jones, R.D.; Harris, A.L. Regulation of tumor pH and the role of carbonic anhydrase 9. Cancer Metastasis Rev. 2007, 26, 299–310. [Google Scholar] [CrossRef] [PubMed]
- Porporato, P.E.; Dhup, S.; Dadhich, R.K.; Copetti, T.; Sonveaux, P. Anticancer targets in the glycolytic metabolism of tumors: A comprehensive review. Front. Pharmacol. 2011, 2, 49. [Google Scholar] [CrossRef]
- Robinson, G.L.; Dinsdale, D.; Macfarlane, M.; Cain, K. Switching from aerobic glycolysis to oxidative phosphorylation modulates the sensitivity of mantle cell lymphoma cells to TRAIL. Oncogene 2012, 31, 4996–5006. [Google Scholar] [CrossRef]
- Giammarioli, A.M.; Gambardella, L.; Barbati, C.; Pietraforte, D.; Tinari, A.; Alberton, M.; Gnessi, L.; Griffin, R.J.; Minetti, M.; Malorni, W. Differential effects of the glycolysis inhibitor 2-deoxy-d-glucose on the activity of pro-apoptotic agents in metastatic melanoma cells, and induction of a cytoprotective autophagic response. Int. J. Cancer 2012, 131, E337–E347. [Google Scholar] [CrossRef]
- Golding, J.P.; Wardhaugh, T.; Patrick, L.; Turner, M.; Phillips, J.B.; Bruce, J.I.; Kimani, S.G. Targeting tumour energy metabolism potentiates the cytotoxicity of 5-aminolevulinic acid photodynamic therapy. Br. J. Cancer 2013, 109, 976–982. [Google Scholar] [CrossRef] [Green Version]
- Zagorodna, O.; Martin, S.M.; Rutkowski, D.T.; Kuwana, T.; Spitz, D.R.; Knudson, C.M. 2-deoxyglucose-induced toxicity is regulated by Bcl-2 family members and is enhanced by antagonizing Bcl-2 in lymphoma cell lines. Oncogene 2012, 31, 2738–2749. [Google Scholar] [CrossRef]
- Aghaee, F.; Pirayesh Islamian, J.; Baradaran, B. Enhanced radiosensitivity and chemosensitivity of breast cancer cells by 2-deoxy-d-glucose in combination therapy. J. Breast Cancer 2012, 15, 141–147. [Google Scholar] [CrossRef]
- Maschek, G.; Savaraj, N.; Priebe, W.; Braunschweiger, P.; Hamilton, K.; Tidmarsh, G.F.; De Young, L.R.; Lampidis, T.J. 2-deoxy-d-glucose increases the efficacy of adriamycin and paclitaxel in human osteosarcoma and non-small cell lung cancers in vivo. Cancer Res. 2004, 64, 31–34. [Google Scholar] [CrossRef]
- Dwarakanath, B.S.; Singh, D.; Banerji, A.K.; Sarin, R.; Venkataramana, N.K.; Jalali, R.; Vishwanath, P.N.; Mohanti, B.K.; Tripathi, R.P.; Kalia, V.K.; et al. Clinical studies for improving radiotherapy with 2-deoxy-d-glucose: Present status and future prospects. J. Cancer Res. Ther. 2009, 5, 21–26. [Google Scholar] [CrossRef]
- Mohanti, B.K.; Rath, G.K.; Anantha, N.; Kannan, V.; Das, B.S.; Chandramouli, B.A.; Banerjee, A.K.; Das, S.; Jena, A.; Ravichandran, R.; et al. Improving cancer radiotherapy with 2-deoxy-d-glucose: Phase I/II clinical trials on human cerebral gliomas. Int. J. Radiat. Oncol. Biol. Phys. 1996, 35, 103–111. [Google Scholar] [CrossRef]
- Raez, L.E.; Papadopoulos, K.; Ricart, A.D.; Chiorean, E.G.; Dipaola, R.S.; Stein, M.N.; Rocha Lima, C.M.; Schlesselman, J.J.; Tolba, K.; Langmuir, V.K.; et al. A phase I dose-escalation trial of 2-deoxy-d-glucose alone or combined with docetaxel in patients with advanced solid tumors. Cancer Chemother. Pharmacol. 2013, 71, 523–530. [Google Scholar] [CrossRef]
- Singh, D.; Banerji, A.K.; Dwarakanath, B.S.; Tripathi, R.P.; Gupta, J.P.; Mathew, T.L.; Ravindranath, T.; Jain, V. Optimizing cancer radiotherapy with 2-deoxy-d-glucose dose escalation studies in patients with glioblastoma multiforme. Strahlenther. Onkol. 2005, 181, 507–514. [Google Scholar] [CrossRef]
- Stein, M.; Lin, H.; Jeyamohan, C.; Dvorzhinski, D.; Gounder, M.; Bray, K.; Eddy, S.; Goodin, S.; White, E.; Dipaola, R.S. Targeting tumor metabolism with 2-deoxyglucose in patients with castrate-resistant prostate cancer and advanced malignancies. Prostate 2010, 70, 1388–1394. [Google Scholar] [CrossRef] [Green Version]
- Boutrid, H.; Jockovich, M.E.; Murray, T.G.; Pina, Y.; Feuer, W.J.; Lampidis, T.J.; Cebulla, C.M. Targeting hypoxia, a novel treatment for advanced retinoblastoma. Investig. Ophthalmol. Vis. Sci. 2008, 49, 2799–2805. [Google Scholar] [CrossRef]
- Maher, J.C.; Krishan, A.; Lampidis, T.J. Greater cell cycle inhibition and cytotoxicity induced by 2-deoxy-d-glucose in tumor cells treated under hypoxic vs aerobic conditions. Cancer Chemother. Pharmacol. 2004, 53, 116–122. [Google Scholar] [CrossRef]
- Kurtoglu, M.; Gao, N.; Shang, J.; Maher, J.C.; Lehrman, M.A.; Wangpaichitr, M.; Savaraj, N.; Lane, A.N.; Lampidis, T.J. Under normoxia, 2-deoxy-d-glucose elicits cell death in select tumor types not by inhibition of glycolysis but by interfering with N-linked glycosylation. Mol. Cancer Ther. 2007, 6, 3049–3058. [Google Scholar] [CrossRef]
- Kovacs, K.; Decatur, C.; Toro, M.; Pham, D.G.; Liu, H.; Jing, Y.; Murray, T.G.; Lampidis, T.J.; Merchan, J.R. 2-Deoxy-Glucose Downregulates Endothelial AKT and ERK via Interference with N-Linked Glycosylation, Induction of Endoplasmic Reticulum Stress, and GSK3beta Activation. Mol. Cancer Ther. 2016, 15, 264–275. [Google Scholar] [CrossRef]
- Larrue, C.; Saland, E.; Vergez, F.; Serhan, N.; Delabesse, E.; Mansat-De Mas, V.; Hospital, M.A.; Tamburini, J.; Manenti, S.; Sarry, J.E.; et al. Antileukemic Activity of 2-Deoxy-d-Glucose through Inhibition of N-Linked Glycosylation in Acute Myeloid Leukemia with FLT3-ITD or c-KIT Mutations. Mol. Cancer Ther. 2015, 14, 2364–2373. [Google Scholar] [CrossRef]
- Manfioletti, G.; Brancolini, C.; Avanzi, G.; Schneider, C. The protein encoded by a growth arrest-specific gene (gas6) is a new member of the vitamin K-dependent proteins related to protein S, a negative coregulator in the blood coagulation cascade. Mol Cell Biol. 1993, 13, 4976–4985. [Google Scholar] [CrossRef]
- Linger, R.M.; Keating, A.K.; Earp, H.S.; Graham, D.K. TAM receptor tyrosine kinases: Biologic functions, signaling, and potential therapeutic targeting in human cancer. Adv Cancer Res. 2008, 100, 35–83. [Google Scholar] [CrossRef]
- Brand, T.M.; Iida, M.; Stein, A.P.; Corrigan, K.L.; Braverman, C.M.; Coan, J.P.; Pearson, H.E.; Bahrar, H.; Fowler, T.L.; Bednarz, B.P.; et al. AXL Is a Logical Molecular Target in Head and Neck Squamous Cell Carcinoma. Clin Cancer Res. 2015, 21, 2601–2612. [Google Scholar] [CrossRef]
- Uchida, D.; Kawamata, H.; Omotehara, F.; Nakashiro, K.; Kimura-Yanagawa, T.; Hino, S.; Begum, N.M.; Hoque, M.O.; Yoshida, H.; Sato, M.; et al. Role of HGF/c-met system in invasion and metastasis of oral squamous cell carcinoma cells in vitro and its clinical significance. Int. J. Cancer 2001, 93, 489–496. [Google Scholar] [CrossRef]
- Noguti, J.; De Moura, C.F.; De Jesus, G.P.; Da Silva, V.H.; Hossaka, T.A.; Oshima, C.T.; Ribeiro, D.A. Metastasis from oral cancer: An overview. Cancer Genom. Proteom. 2012, 9, 329–335. [Google Scholar]
- Dennis, J.W.; Granovsky, M.; Warren, C.E. Glycoprotein glycosylation and cancer progression. Biochim. Biophys. Acta 1999, 1473, 21–34. [Google Scholar] [CrossRef]
- Dwek, R.A. Glycobiology: Toward Understanding the Function of Sugars. Chem. Rev. 1996, 96, 683–720. [Google Scholar] [CrossRef]
- Contessa, J.N.; Bhojani, M.S.; Freeze, H.H.; Rehemtulla, A.; Lawrence, T.S. Inhibition of N-linked glycosylation disrupts receptor tyrosine kinase signaling in tumor cells. Cancer Res. 2008, 68, 3803–3809. [Google Scholar] [CrossRef]
- Li, J.; Jia, L.; Ma, Z.H.; Ma, Q.H.; Yang, X.H.; Zhao, Y.F. Axl glycosylation mediates tumor cell proliferation, invasion and lymphatic metastasis in murine hepatocellular carcinoma. World J. Gastroenterol. 2012, 18, 5369–5376. [Google Scholar] [CrossRef]
- Contessa, J.N.; Bhojani, M.S.; Freeze, H.H.; Ross, B.D.; Rehemtulla, A.; Lawrence, T.S. Molecular imaging of N-linked glycosylation suggests glycan biosynthesis is a novel target for cancer therapy. Clin. Cancer Res. 2010, 16, 3205–3214. [Google Scholar] [CrossRef]
- Wellen, K.E.; Lu, C.; Mancuso, A.; Lemons, J.M.; Ryczko, M.; Dennis, J.W.; Rabinowitz, J.D.; Coller, H.A.; Thompson, C.B. The hexosamine biosynthetic pathway couples growth factor-induced glutamine uptake to glucose metabolism. Genes Dev. 2010, 24, 2784–2799. [Google Scholar] [CrossRef] [Green Version]
- Cairns, R.A.; Harris, I.S.; Mak, T.W. Regulation of cancer cell metabolism. Nat. Rev. Cancer 2011, 11, 85–95. [Google Scholar] [CrossRef] [Green Version]
- Holmes, E.; Wilson, I.D.; Nicholson, J.K. Metabolic phenotyping in health and disease. Cell 2008, 134, 714–717. [Google Scholar] [CrossRef]
- Nicholson, J.K.; Connelly, J.; Lindon, J.C.; Holmes, E. Metabonomics: A platform for studying drug toxicity and gene function. Nat. Rev. Drug Discov. 2002, 1, 153–161. [Google Scholar] [CrossRef]
- Sborov, D.W.; Haverkos, B.M.; Harris, P.J. Investigational cancer drugs targeting cell metabolism in clinical development. Expert Opin. Investig. Drugs 2015, 24, 79–94. [Google Scholar] [CrossRef]
- Le Pogam, P.; Doue, M.; Le Page, Y.; Habauzit, D.; Zhadobov, M.; Sauleau, R.; Le Drean, Y.; Rondeau, D. Untargeted Metabolomics Reveal Lipid Alterations upon 2-Deoxyglucose Treatment in Human HaCaT Keratinocytes. J. Proteome Res. 2018, 17, 1146–1157. [Google Scholar] [CrossRef]
- Miwa, H.; Shikami, M.; Goto, M.; Mizuno, S.; Takahashi, M.; Tsunekawa-Imai, N.; Ishikawa, T.; Mizutani, M.; Horio, T.; Gotou, M.; et al. Leukemia cells demonstrate a different metabolic perturbation provoked by 2-deoxyglucose. Oncol. Rep. 2013, 29, 2053–2057. [Google Scholar] [CrossRef] [Green Version]
- Urakami, K.; Zangiacomi, V.; Yamaguchi, K.; Kusuhara, M. Impact of 2-deoxy-d-glucose on the target metabolome profile of a human endometrial cancer cell line. Biomed. Res. 2013, 34, 221–229. [Google Scholar] [CrossRef]
- Maddocks, O.D.; Berkers, C.R.; Mason, S.M.; Zheng, L.; Blyth, K.; Gottlieb, E.; Vousden, K.H. Serine starvation induces stress and p53-dependent metabolic remodelling in cancer cells. Nature 2013, 493, 542–546. [Google Scholar] [CrossRef]
- Possemato, R.; Marks, K.M.; Shaul, Y.D.; Pacold, M.E.; Kim, D.; Birsoy, K.; Sethumadhavan, S.; Woo, H.K.; Jang, H.G.; Jha, A.K.; et al. Functional genomics reveal that the serine synthesis pathway is essential in breast cancer. Nature 2011, 476, 346–350. [Google Scholar] [CrossRef] [Green Version]
- Locasale, J.W. Serine, glycine and one-carbon units: Cancer metabolism in full circle. Nat. Rev. Cancer 2013, 13, 572–583. [Google Scholar] [CrossRef]
- Coloff, J.L.; Murphy, J.P.; Braun, C.R.; Harris, I.S.; Shelton, L.M.; Kami, K.; Gygi, S.P.; Selfors, L.M.; Brugge, J.S. Differential Glutamate Metabolism in Proliferating and Quiescent Mammary Epithelial Cells. Cell Metab. 2016, 23, 867–880. [Google Scholar] [CrossRef]
- Loayza-Puch, F.; Rooijers, K.; Buil, L.C.; Zijlstra, J.; Oude Vrielink, J.F.; Lopes, R.; Ugalde, A.P.; van Breugel, P.; Hofland, I.; Wesseling, J.; et al. Tumour-specific proline vulnerability uncovered by differential ribosome codon reading. Nature 2016, 530, 490–494. [Google Scholar] [CrossRef]
- Kremer, J.C.; Prudner, B.C.; Lange, S.E.S.; Bean, G.R.; Schultze, M.B.; Brashears, C.B.; Radyk, M.D.; Redlich, N.; Tzeng, S.C.; Kami, K.; et al. Arginine Deprivation Inhibits the Warburg Effect and Upregulates Glutamine Anaplerosis and Serine Biosynthesis in ASS1-Deficient Cancers. Cell Rep. 2017, 18, 991–1004. [Google Scholar] [CrossRef] [Green Version]
- Knott, S.R.V.; Wagenblast, E.; Khan, S.; Kim, S.Y.; Soto, M.; Wagner, M.; Turgeon, M.O.; Fish, L.; Erard, N.; Gable, A.L.; et al. Asparagine bioavailability governs metastasis in a model of breast cancer. Nature 2018, 554, 378–381. [Google Scholar] [CrossRef]
- Krall, A.S.; Xu, S.; Graeber, T.G.; Braas, D.; Christofk, H.R. Asparagine promotes cancer cell proliferation through use as an amino acid exchange factor. Nat. Commun. 2016, 7, 11457. [Google Scholar] [CrossRef] [Green Version]
- Siminska, E.; Koba, M. Amino acid profiling as a method of discovering biomarkers for early diagnosis of cancer. Amino Acids 2016, 48, 1339–1345. [Google Scholar] [CrossRef]
- Maeda, J.; Higashiyama, M.; Imaizumi, A.; Nakayama, T.; Yamamoto, H.; Daimon, T.; Yamakado, M.; Imamura, F.; Kodama, K. Possibility of multivariate function composed of plasma amino acid profiles as a novel screening index for non-small cell lung cancer: A case control study. BMC Cancer 2010, 10, 690. [Google Scholar] [CrossRef]
- Miyagi, Y.; Higashiyama, M.; Gochi, A.; Akaike, M.; Ishikawa, T.; Miura, T.; Saruki, N.; Bando, E.; Kimura, H.; Imamura, F.; et al. Plasma free amino acid profiling of five types of cancer patients and its application for early detection. PLoS ONE 2011, 6, e24143. [Google Scholar] [CrossRef]
- Medzihradszky, K.F. Characterization of site-specific N-glycosylation. Methods Mol. Biol. 2008, 446, 293–316. [Google Scholar] [CrossRef]
- Selvi, R.; Angayarkanni, N.; Asma, B.; Seethalakshmi, T.; Vidhya, S. Amino acids influence the glucose uptake through GLUT4 in CHO-K1 cells under high glucose conditions. Mol. Cell. Biochem. 2010, 344, 43–53. [Google Scholar] [CrossRef]
- Torres-Zamorano, V.; Leibach, F.H.; Ganapathy, V. Sodium-dependent homo- and hetero-exchange of neutral amino acids mediated by the amino acid transporter ATB degree. Biochem. Biophys. Res. Commun. 1998, 245, 824–829. [Google Scholar] [CrossRef]
- Console, L.; Scalise, M.; Tarmakova, Z.; Coe, I.R.; Indiveri, C. N-linked glycosylation of human SLC1A5 (ASCT2) transporter is critical for trafficking to membrane. Biochim. Biophys. Acta 2015, 1853, 1636–1645. [Google Scholar] [CrossRef]
- Polet, F.; Martherus, R.; Corbet, C.; Pinto, A.; Feron, O. Inhibition of glucose metabolism prevents glycosylation of the glutamine transporter ASCT2 and promotes compensatory LAT1 upregulation in leukemia cells. Oncotarget 2016, 7, 46371–46383. [Google Scholar] [CrossRef] [Green Version]
- Lemonnier, L.A.; Dillehay, D.L.; Vespremi, M.J.; Abrams, J.; Brody, E.; Schmelz, E.M. Sphingomyelin in the suppression of colon tumors: Prevention versus intervention. Arch. Biochem. Biophys. 2003, 419, 129–138. [Google Scholar] [CrossRef]
- Modrak, D.E.; Lew, W.; Goldenberg, D.M.; Blumenthal, R. Sphingomyelin potentiates chemotherapy of human cancer xenografts. Biochem. Biophys. Res. Commun. 2000, 268, 603–606. [Google Scholar] [CrossRef]
- Lee, S.; Jang, W.J.; Choi, B.; Joo, S.H.; Jeong, C.H. Comparative metabolomic analysis of HPAC cells following the acquisition of erlotinib resistance. Oncol. Lett. 2017, 13, 3437–3444. [Google Scholar] [CrossRef] [Green Version]
| Class | Metabolite | KEGG ID | Normalized Value | p-Value | FDR | d-Value | VIP Score | |||
|---|---|---|---|---|---|---|---|---|---|---|
| CON | 2DG | 2DG+M | 2DG+P | |||||||
| Amino Acids and Biogenic Amines | ADMA | C03626 | 0.53 | −1.31 | −0.35 | 1.13 | 0.000 | 0.000 | 4.482 | 1.520 |
| Amino Acids and Biogenic Amines | Glycine | C00037 | 0.37 | −1.00 | 0.58 | 0.05 | 0.015 | 0.029 | 2.340 | 1.340 |
| Amino Acids and Biogenic Amines | l-Alanine | C00041 | 0.74 | −1.08 | 0.21 | 0.13 | 0.005 | 0.013 | 2.816 | 1.416 |
| Amino Acids and Biogenic Amines | l-Asparagine | C00152 | 0.93 | −1.01 | 0.36 | −0.28 | 0.009 | 0.019 | 2.595 | 1.303 |
| Amino Acids and Biogenic Amines | l-Citrulline | C00327 | 0.67 | −1.07 | −0.06 | 0.46 | 0.001 | 0.003 | 3.534 | 1.508 |
| Amino Acids and Biogenic Amines | l-Histidine | C00135 | 0.76 | -1.09 | 0.42 | −0.09 | 0.001 | 0.005 | 3.310 | 1.431 |
| Amino Acids and Biogenic Amines | l-Isoleucine | C00407 | 0.77 | −0.97 | 0.63 | −0.43 | 0.001 | 0.004 | 3.448 | 1.342 |
| Amino Acids and Biogenic Amines | l-Lysine | C00047 | 0.71 | −1.05 | 0.48 | −0.14 | 0.010 | 0.021 | 2.523 | 1.308 |
| Amino Acids and Biogenic Amines | l-Methionine | C00073 | 0.10 | −0.80 | 0.87 | −0.16 | 0.029 | 0.046 | 2.041 | 1.229 |
| Amino Acids and Biogenic Amines | l-Phenylalanine | C00079 | 0.48 | −1.15 | 0.64 | 0.03 | 0.002 | 0.007 | 3.149 | 1.425 |
| Amino Acids and Biogenic Amines | l-Proline | C00148 | 0.89 | −1.22 | 0.37 | −0.05 | 0.000 | 0.002 | 3.801 | 1.483 |
| Amino Acids and Biogenic Amines | l-Serine | C00065 | 0.32 | −1.13 | 0.67 | 0.15 | 0.011 | 0.023 | 2.478 | 1.357 |
| Amino Acids and Biogenic Amines | l-Threonine | C00188 | 0.79 | −1.39 | 0.24 | 0.35 | 0.000 | 0.002 | 3.850 | 1.541 |
| Amino Acids and Biogenic Amines | l-Tyrosine | C00082 | 0.45 | −0.90 | 0.61 | −0.16 | 0.006 | 0.014 | 2.784 | 1.357 |
| Amino Acids and Biogenic Amines | l-Valine | C00183 | 0.46 | −1.10 | 0.90 | −0.26 | 0.002 | 0.005 | 3.256 | 1.321 |
| Amino Acids and Biogenic Amines | Putrescine | C00134 | 1.14 | −0.99 | −0.60 | 0.45 | 0.000 | 0.002 | 3.741 | 1.454 |
| Amino Acids and Biogenic Amines | Taurine | C00245 | 0.69 | 0.00 | −0.15 | −0.53 | 0.023 | 0.041 | 2.139 | 1.067 |
| Glycerophospholipids | PC aa C32:0 | C00157 | −0.04 | 0.98 | −1.02 | 0.08 | 0.000 | 0.000 | 5.023 | 1.218 |
| Glycerophospholipids | PC aa C32:2 | C00157 | −0.41 | 0.21 | 1.21 | −1.01 | 0.000 | 0.000 | 4.643 | 1.208 |
| Glycerophospholipids | PC aa C32:3 | C00157 | 1.05 | −0.34 | 0.42 | −1.13 | 0.000 | 0.001 | 3.966 | 1.187 |
| Glycerophospholipids | PC aa C34:2 | C00157 | −1.00 | 0.72 | 0.92 | −0.64 | 0.000 | 0.002 | 3.779 | 1.125 |
| Glycerophospholipids | PC aa C34:3 | C00157 | 0.05 | −0.25 | 1.21 | −1.01 | 0.000 | 0.000 | 4.592 | 1.251 |
| Glycerophospholipids | PC aa C34:4 | C00157 | 0.34 | -0.23 | 1.05 | −1.17 | 0.000 | 0.000 | 4.478 | 1.196 |
| Glycerophospholipids | PC aa C36:1 | C00157 | −1.39 | 0.78 | 0.88 | −0.27 | 0.027 | 0.043 | 2.077 | 1.295 |
| Glycerophospholipids | PC aa C36:3 | C00157 | −0.76 | 0.68 | 0.93 | −0.85 | 0.000 | 0.002 | 3.858 | 1.013 |
| Glycerophospholipids | PC aa C36:6 | C00157 | 0.26 | −0.52 | 1.16 | −0.91 | 0.000 | 0.002 | 3.871 | 1.246 |
| Glycerophospholipids | PC aa C38:4 | C00157 | −1.40 | 0.75 | 0.93 | −0.28 | 0.000 | 0.001 | 4.037 | 1.469 |
| Glycerophospholipids | PC aa C38:6 | C00157 | -0.88 | 0.61 | 1.09 | −0.81 | 0.002 | 0.005 | 3.241 | 1.073 |
| Glycerophospholipids | PC aa C40:4 | C00157 | −1.36 | 0.93 | 0.75 | −0.32 | 0.017 | 0.031 | 2.290 | 1.361 |
| Glycerophospholipids | PC aa C40:5 | C00157 | −1.27 | 1.11 | 0.61 | −0.45 | 0.000 | 0.001 | 4.256 | 1.439 |
| Glycerophospholipids | PC ae C34:0 | - | −0.55 | 0.40 | 0.82 | −0.67 | 0.000 | 0.001 | 3.932 | 1.311 |
| Glycerophospholipids | PC ae C36:5 | - | −1.50 | 0.81 | 0.68 | 0.01 | 0.001 | 0.004 | 3.463 | 1.368 |
| Glycerophospholipids | PC ae C38:0 | - | 0.05 | 0.09 | 0.87 | −1.01 | 0.000 | 0.001 | 4.057 | 1.287 |
| Glycerophospholipids | PC ae C38:6 | - | −1.45 | 0.94 | 0.65 | −0.14 | 0.002 | 0.005 | 3.280 | 1.461 |
| Glycerophospholipids | PC ae C40:1 | - | 0.88 | −0.55 | 0.28 | −0.62 | 0.000 | 0.002 | 3.788 | 1.342 |
| Glycerophospholipids | PC ae C40:4 | - | −1.39 | 0.78 | 0.90 | −0.29 | 0.024 | 0.041 | 2.127 | 1.288 |
| Glycerophospholipids | PC ae C42:1 | - | −0.61 | 0.81 | −0.81 | 0.62 | 0.003 | 0.009 | 3.020 | 1.068 |
| Glycerophospholipids | PC ae C42:2 | - | −0.71 | 0.43 | 0.44 | −0.15 | 0.026 | 0.043 | 2.090 | 1.167 |
| Glycerophospholipids | PC ae C42:3 | - | 0.39 | 0.00 | −0.12 | −0.27 | 0.006 | 0.015 | 2.726 | 1.230 |
| Sphingolipids | SM C18:0 | C00550 | −1.41 | 0.85 | 0.88 | −0.32 | 0.015 | 0.029 | 2.336 | 1.347 |
| Sphingolipids | SM C18:1 | C00550 | −1.50 | 0.98 | 0.59 | −0.07 | 0.001 | 0.005 | 3.340 | 1.425 |
| Sphingolipids | SM C24:1 | C00550 | −1.40 | 0.85 | 0.83 | −0.28 | 0.002 | 0.005 | 3.246 | 1.483 |
| Sphingolipids | SM (OH) C22:1 | C00550 | −1.29 | 1.04 | 0.71 | −0.46 | 0.005 | 0.012 | 2.854 | 1.384 |
| Sphingolipids | SM (OH) C22:2 | C00550 | −1.29 | 0.80 | 0.96 | −0.47 | 0.001 | 0.004 | 3.445 | 1.386 |
| Target Gene | Primers | Nucleotide Sequence |
|---|---|---|
| LDHA | F | GTGGGTCCTTGGGGAACATGGAG |
| R | CAGGTTATCGGGTCCTACACATCGG | |
| LDHB | F | CCGTCAGCAAGAAGGGGAGAGTCG |
| R | GGTTAGTGCGCCACAAACCCATTAGG | |
| HIF-1α | F | ACCACCTATGACCTGCTTGGTGCTG |
| R | GTATAGGTCCGACACAGCTGACTCC | |
| GAPDH | F | TCGACAGTCAGCCGCATCT |
| R | CCGTTGACTCCGACCTTCA |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, N.; Jang, W.-J.; Seo, J.H.; Lee, S.; Jeong, C.-H. 2-Deoxy-d-Glucose-Induced Metabolic Alteration in Human Oral Squamous SCC15 Cells: Involvement of N-Glycosylation of Axl and Met. Metabolites 2019, 9, 188. https://doi.org/10.3390/metabo9090188
Lee N, Jang W-J, Seo JH, Lee S, Jeong C-H. 2-Deoxy-d-Glucose-Induced Metabolic Alteration in Human Oral Squamous SCC15 Cells: Involvement of N-Glycosylation of Axl and Met. Metabolites. 2019; 9(9):188. https://doi.org/10.3390/metabo9090188
Chicago/Turabian StyleLee, Naeun, Won-Jun Jang, Ji Hae Seo, Sooyeun Lee, and Chul-Ho Jeong. 2019. "2-Deoxy-d-Glucose-Induced Metabolic Alteration in Human Oral Squamous SCC15 Cells: Involvement of N-Glycosylation of Axl and Met" Metabolites 9, no. 9: 188. https://doi.org/10.3390/metabo9090188
APA StyleLee, N., Jang, W.-J., Seo, J. H., Lee, S., & Jeong, C.-H. (2019). 2-Deoxy-d-Glucose-Induced Metabolic Alteration in Human Oral Squamous SCC15 Cells: Involvement of N-Glycosylation of Axl and Met. Metabolites, 9(9), 188. https://doi.org/10.3390/metabo9090188





