Relationship between Vitamin D Level and Lipid Profile in Non-Obese Children
Abstract
1. Introduction
2. Experimental Section
2.1. Study Population
2.2. Anthropometric and Laboratory Assessment
2.3. Statistical Analysis
2.4. Ethical Considerations
3. Results
3.1. Subject Characteristics
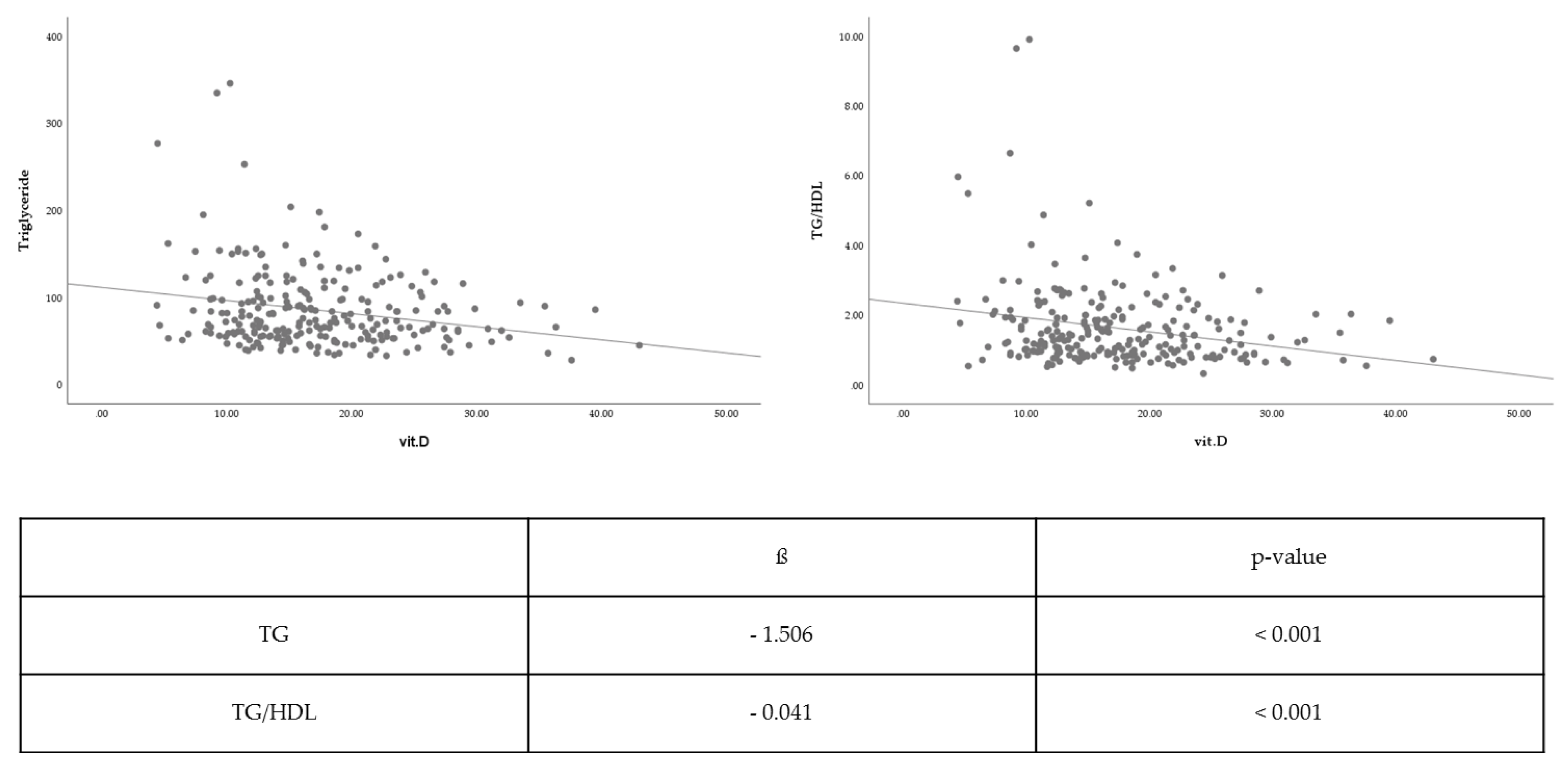
3.2. Association between Vitamin D Level and Lipid Profile
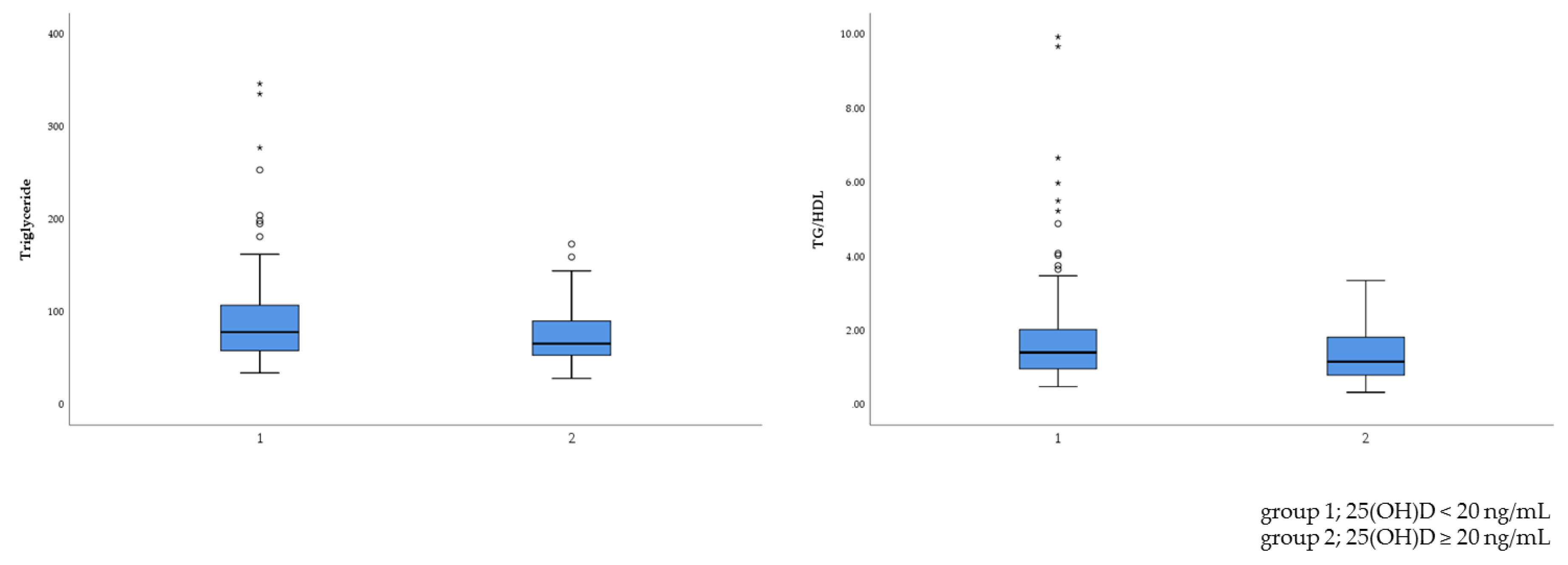
3.3. Comparison of Vitamin D Levels Between Two Groups
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Lee, J.H.; O’Keefe, J.H.; Bell, D.; Hensrud, D.D.; Holick, M.F. Vitamin D deficiency an important, common, and easily treatable cardiovascular risk factor? J. Am. Coll. Cardiol. 2008, 52, 1949–1956. [Google Scholar] [CrossRef] [PubMed]
- Tangpricha, V.; Pearce, E.N.; Chen, T.C.; Holick, M.F. Vitamin D insufficiency among free-living healthy young adults. Am. J. Med. 2002, 112, 659–662. [Google Scholar] [CrossRef]
- Johnson, M.D.; Nader, N.S.; Weaver, A.L.; Singh, R.; Kumar, S. Relationships between 25-hydroxyvitamin D levels and plasma glucose and lipid levels in pediatric outpatients. J. Pediatr. 2010, 156, 444–449. [Google Scholar] [CrossRef] [PubMed]
- Earthman, C.P.; Beckman, L.M.; Masodkar, K.; Sibley, S.D. The link between obesity and low circulating 25-hydroxyvitamin D concentrations: Considerations and implications. Int. J. Obes. (Lond.) 2012, 36, 387–396. [Google Scholar] [CrossRef] [PubMed]
- Lagunova, Z.; Porojnicu, A.C.; Vieth, R.; Lindberg, F.A.; Hexeberg, S.; Moan, J. Serum 25-hydroxyvitamin D is a predictor of serum 1,25-dihydroxyvitamin D in overweight and obese patients. J. Nutr. 2011, 141, 112–117. [Google Scholar] [CrossRef] [PubMed]
- Turer, C.B.; Lin, H.; Flores, G. Prevalence of vitamin D deficiency among overweight and obese US children. Pediatrics 2013, 131, e152–e161. [Google Scholar] [CrossRef] [PubMed]
- Wortsman, J.; Matsuoka, L.Y.; Chen, T.C.; Lu, Z.; Holick, M.F. Decreased bioavailability of vitamin D in obesity. Am. J. Clin. Nutr. 2000, 72, 690–693. [Google Scholar] [CrossRef] [PubMed]
- Dobnig, H.; Pilz, S.; Scharnagl, H.; Renner, W.; Seelhorst, U.; Wellnitz, B.; Kinkeldei, J.; Boehm, B.O.; Weihrauch, G.; Maerz, W. Independent association of low serum 25-hydroxyvitamin d and 1,25-dihydroxyvitamin d levels with all-cause and cardiovascular mortality. Arch. Intern. Med. 2008, 168, 1340–1349. [Google Scholar] [CrossRef] [PubMed]
- Martini, L.A.; Wood, R.J. Vitamin D status and the metabolic syndrome. Nutr. Rev. 2006, 64, 479–486. [Google Scholar] [CrossRef]
- Polkowska, A.; Głowińska-Olszewska, B.; Tobiaszewska, M.; Bossowski, A. Risk factors for cardiovascular disease in children with type 1 diabetes in 2000-2010 in Podlasie Province. Pediatr. Endocrinol. Diabetes Metab. 2014, 20, 47–54. [Google Scholar] [CrossRef]
- Potenza, M.V.; Mechanick, J.I. The metabolic syndrome: Definition, global impact, and pathophysiology. Nutr. Clin. Pract 2009, 24, 560–577. [Google Scholar] [CrossRef] [PubMed]
- Nilsson, G.; Hedberg, P.; Ohrvik, J. Inflammation and the metabolic syndrome: Clustering and impact on survival in a Swedish community-based cohort of 75 year olds. Metab. Syndr. Relat. Disord. 2013, 11, 92–101. [Google Scholar] [CrossRef] [PubMed]
- Lamberg-Allardt, C. Vitamin D in children and adolescents. Scand. J. Clin. Lab. Investig. Suppl. 2012, 243, 124–128. [Google Scholar] [CrossRef]
- Gupta, N.; Shah, P.; Nayyar, S.; Misra, A. Childhood obesity and the metabolic syndrome in developing countries. Indian J. Pediatr. 2013, 80 (Suppl. 1), S28–S37. [Google Scholar] [CrossRef]
- Kelishadi, R. Childhood overweight, obesity, and the metabolic syndrome in developing countries. Epidemiol. Rev. 2007, 29, 62–76. [Google Scholar] [CrossRef] [PubMed]
- Nam, G.E.; Kim, D.H.; Cho, K.H.; Park, Y.G.; Han, K.D.; Kim, S.M.; Lee, S.H.; Ko, B.J.; Kim, M.J. 25-Hydroxyvitamin D insufficiency is associated with cardiometabolic risk in Korean adolescents: The 2008–2009 Korea National Health and Nutrition Examination Survey (KNHANES). Public Health Nutr. 2014, 17, 186–194. [Google Scholar] [CrossRef] [PubMed]
- Williams, D.M.; Fraser, A.; Sayers, A.; Fraser, W.D.; Hingorani, A.; Deanfield, J.; Davey Smith, G.; Sattar, N.; Lawlor, D.A. Associations of 25-hydroxyvitamin D2 and D3 with cardiovascular risk factors in childhood: Cross-sectional findings from the Avon Longitudinal Study of Parents and Children. J. Clin. Endocrinol. Metab. 2012, 97, 1563–1571. [Google Scholar] [CrossRef]
- Hirschler, V.; Maccallini, G.; Molinari, C.; Aranda, C.; San Antonio de los Cobres Study Group. Low vitamin D concentrations among indigenous Argentinean children living at high altitudes. Pediatr. Diabetes 2013, 14, 203–210. [Google Scholar] [CrossRef]
- Mark, S.; Gray-Donald, K.; Delvin, E.E.; O’Loughlin, J.; Paradis, G.; Levy, E.; Lambert, M. Low vitamin D status in a representative sample of youth from Quebec, Canada. Clin. Chem. 2008, 54, 1283–1289. [Google Scholar] [CrossRef]
- Rusconi, R.E.; De Cosmi, V.; Gianluca, G.; Giavoli, C.; Agostoni, C. Vitamin D insufficiency in obese children and relation with lipid profile. Int. J. Food Sci. Nutr. 2015, 66, 132–134. [Google Scholar] [CrossRef]
- Hirschler, V.; Maccallini, G.; Sanchez, M.S.; Castano, L.; Molinari, C. Improvement in high-density lipoprotein cholesterol levels in argentine Indian school children after vitamin D supplementation. Horm. Res. Paediatr. 2013, 80, 335–342. [Google Scholar] [CrossRef] [PubMed]
- Misra, A.; Chowbey, P.; Makkar, B.M.; Vikram, N.K.; Wasir, J.S.; Chadha, D.; Joshi, S.R.; Sadikot, S.; Gupta, R.; Gulati, S.; et al. Consensus statement for diagnosis of obesity, abdominal obesity and the metabolic syndrome for Asian Indians and recommendations for physical activity, medical and surgical management. J. Assoc. Physicians India 2009, 57, 163–170. [Google Scholar] [PubMed]
- Banerji, M.A.; Faridi, N.; Atluri, R.; Chaiken, R.L.; Lebovitz, H.E. Body composition, visceral fat, leptin, and insulin resistance in Asian Indian men. J. Clin. Endocrinol. Metab. 1999, 84, 137–144. [Google Scholar] [CrossRef] [PubMed]
- Choi, H.S.; Oh, H.J.; Choi, H.; Choi, W.H.; Kim, J.G.; Kim, K.M.; Kim, K.J.; Rhee, Y.; Lim, S.K. Vitamin D insufficiency in Korea—A greater threat to younger generation: The Korea National Health and Nutrition Examination Survey (KNHANES) 2008. J. Clin. Endocrinol. Metab. 2011, 96, 643–651. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Si, S.; Liu, J.; Wang, Z.; Jia, H.; Feng, K.; Sun, L.; Song, S.J. The associations of serum lipids with vitamin D status. PLoS ONE 2016, 11, e0165157. [Google Scholar] [CrossRef] [PubMed]
- Chaudhuri, J.R.; Mridula, K.R.; Anamika, A.; Boddu, D.B.; Misra, P.K.; Lingaiah, A.; Balaraju, B.; Bandaru, V.S. Deficiency of 25-hydroxyvitamin D and dyslipidemia in Indian subjects. J. Lipids 2013, 2013, 623420. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Lee, S.H.; Kim, S.M.; Park, H.S.; Choi, K.M.; Cho, G.J.; Ko, B.J.; Kim, J.H. Serum 25-hydroxyvitamin D levels, obesity and the metabolic syndrome among Korean children. Nutr. Metab. Cardiovasc. Dis. 2013, 23, 785–791. [Google Scholar] [CrossRef] [PubMed]
- Vaskonen, T.; Mervaala, E.; Sumuvuori, V.; Seppänen-Laakso, T.; Karppanen, H. Effects of calcium and plant sterols on serum lipids in obese Zucker rats on a low-fat diet. Br. J. Nutr. 2007, 87, 239–245. [Google Scholar] [CrossRef]
- Song, S.J.; Si, S.; Liu, J.; Chen, X.; Zhou, L.; Jia, G.; Liu, G.; Niu, Y.; Wu, J.; Zhang, W.; et al. Vitamin D status in Chinese pregnant women and their newborns in Beijing and their relationships to birth size. Public Health Nutr. 2012, 16, 687–692. [Google Scholar] [CrossRef]
- Zittermann, A.; Frisch, S.; Berthold, H.K.; Gotting, C.; Kuhn, J.; Kleesiek, K.; Stehle, P.; Koertke, H.; Koerfer, R. Vitamin D supplementation enhances the beneficial effects of weight loss on cardiovascular disease risk markers. Am. J. Clin. Nutr. 2009, 89, 1321–1327. [Google Scholar] [CrossRef]
- Lee, J.H.; Jeong, S.J. What is the appropriate strategy for diagnosing NAFLD using ultrasonography in obese children? World J. Pediatr. 2017, 13, 248–254. [Google Scholar] [CrossRef] [PubMed]
- Challoumas, D. Vitamin D supplementation and lipid profile: What does the best available evidence show? Atherosclerosis 2014, 235, 130–139. [Google Scholar] [CrossRef] [PubMed]
- Ponda, M.P.; Huang, X.; Odeh, M.A.; Breslow, J.L.; Kaufman, H.W. Vitamin D may not improve lipid levels: A serial clinical laboratory data study. Circulation 2012, 126, 270–277. [Google Scholar] [CrossRef] [PubMed]
- Jorde, R.; Grimnes, G. Vitamin D and lipids: Do we really need more studies? Circulation 2012, 126, 252–254. [Google Scholar] [CrossRef] [PubMed][Green Version]
| Variables | Subjects (n = 243) |
|---|---|
| Mean (Range, SD) | |
| Age (years) | 11.26 (9–18, 1.91) |
| Anthropometric profile | |
| Height (cm) | 146.05 (120.8–182.1, 10.5) |
| Weight (kg) | 40.47 (22–68.8, 8.51) |
| BMI (kg/m2) | 18.82 (11.93–22.94, 2.38) |
| Laboratory profile | |
| 25(OH)D (ng/mL) | 17.27 (4.4–43, 6.89) |
| TC (mg/dL) | 169.16 (66.2–286, 30.43) |
| LDL-C (mg/dL) | 94.93 (23–177, 24.65) |
| HDL-C (mg/dL) | 57.38 (18.8–114.5, 13.01) |
| TG (mg/dL) | 85.54 (28–346, 45.13) |
| TG/HDL-C | 1.63 (0.32–9.91, 1.2) |
| Variables | 25(OH)D < 20ng/mL (n = 169) | 25(OH)D Normal (n = 74) | p-Value |
|---|---|---|---|
| Mean (Range, SD) | Mean (Range, SD) | ||
| Age (years) | 11.36 (9–18, 2.07) | 11.04 (9–16, 1.48) | - |
| Anthropometric profile | - | - | - |
| Height (cm) | 146.84 (121.1–178, 10.59) | 144.26 (120.8–182.1, 10.12) | - |
| Weight (kg) | 40.78 (22–65, 8.91) | 39.76 (25.8–68.8, 7.54) | - |
| BMI (kg/m2) | 18.73 (11.93–22.94, 2.43) | 19.01 (12.49–22.62, 2.27) | - |
| Laboratory profile | - | - | - |
| 25(OH)D (ng/mL) | 13.58 (4.4–19.9, 3.62) | 25.68 (20–43.03, 4.93) | - |
| TC (mg/dL) | 168.97 (66.2–286, 30.39) | 169.59 (77–235, 30.74) | 0.883 |
| LDL-C (mg/dL) | 95.04 (23–177, 24.71) | 94.66 (31–158, 24.68) | 0.911 |
| HDL-C (mg/dL) | 56.64 (18.8–98.4, 12.53) | 59.09 (32.5–114.5, 14) | 0.178 |
| TG (mg/dL) | 90.27 (34–346, 49.4) | 74.74 (28–173, 31.05) | 0.003 |
| TG/HDL-C | 1.75 (0.48–9.91, 1.35) | 1.36 (0.32–3.34, 0.7) | 0.003 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, M.R.; Jeong, S.J. Relationship between Vitamin D Level and Lipid Profile in Non-Obese Children. Metabolites 2019, 9, 125. https://doi.org/10.3390/metabo9070125
Kim MR, Jeong SJ. Relationship between Vitamin D Level and Lipid Profile in Non-Obese Children. Metabolites. 2019; 9(7):125. https://doi.org/10.3390/metabo9070125
Chicago/Turabian StyleKim, Mi Ra, and Su Jin Jeong. 2019. "Relationship between Vitamin D Level and Lipid Profile in Non-Obese Children" Metabolites 9, no. 7: 125. https://doi.org/10.3390/metabo9070125
APA StyleKim, M. R., & Jeong, S. J. (2019). Relationship between Vitamin D Level and Lipid Profile in Non-Obese Children. Metabolites, 9(7), 125. https://doi.org/10.3390/metabo9070125





