Effects of Long-Term Storage at −80 °C on the Human Plasma Metabolome
Abstract
1. Introduction
2. Results
2.1. Long-Term Storage Affects the Plasma Metabolome
2.2. Impact of Long-Term Storage on Selected Plasma Metabolites
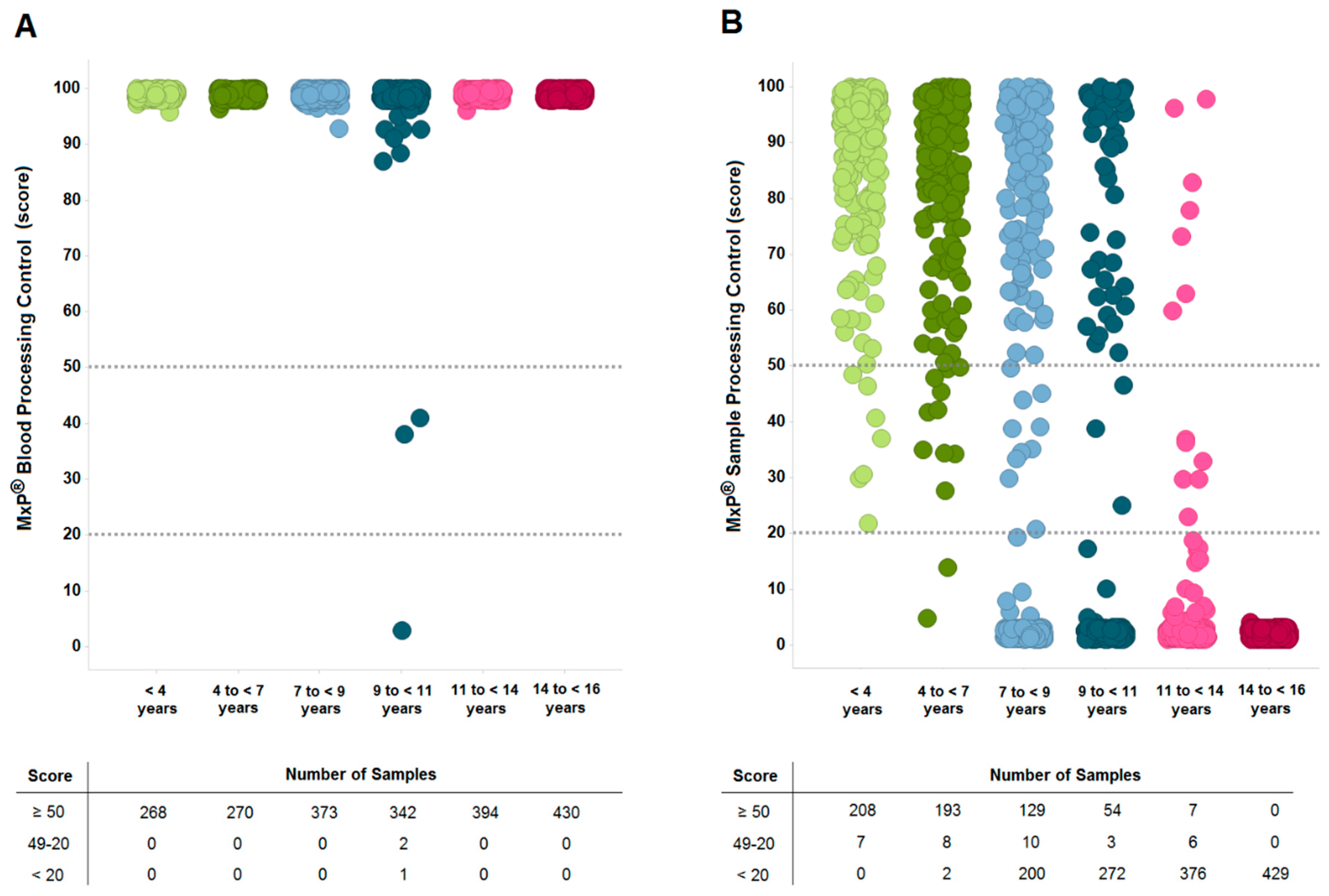
2.3. Sample Quality is Affected by Long-Term Storage
3. Discussion
4. Materials and Methods
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Armitage, E.G.; Barbas, C. Metabolomics in cancer biomarker discovery: Current trends and future perspectives. J. Pharm. Biomed. Anal. 2014, 87, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Patel, S.; Ahmed, S. Emerging field of metabolomics: Big promise for cancer biomarker identification and drug discovery. J. Pharm. Biomed. Anal. 2015, 107, 63–74. [Google Scholar] [CrossRef]
- Wang, T.J.; Larson, M.G.; Vasan, R.S.; Cheng, S.; Rhee, E.P.; McCabe, E.; Lewis, G.D.; Fox, C.S.; Jacques, P.F.; Fernandez, C.; et al. Metabolite profiles and the risk of developing diabetes. Nat. Med. 2011, 17, 448–453. [Google Scholar] [CrossRef]
- Roberts, L.D.; Koulman, A.; Griffin, J.L. Towards metabolic biomarkers of insulin resistance and type 2 diabetes: Progress from the metabolome. Lancet Diabetes Endocrinol. 2014, 2, 65–75. [Google Scholar] [CrossRef]
- Suhre, K. Metabolic profiling in diabetes. J. Endocrinol. 2014, 221, R75–R85. [Google Scholar] [CrossRef] [PubMed]
- Barderas, M.G.; Laborde, C.M.; Posada, M.; de La Cuesta, F.; Zubiri, I.; Vivanco, F.; Alvarez-Llamas, G. Metabolomic profiling for identification of novel potential biomarkers in cardiovascular diseases. J. Biomed. Biotechnol. 2011, 2011, 790132. [Google Scholar] [CrossRef]
- Rhee, E.P.; Gerszten, R.E. Metabolomics and cardiovascular biomarker discovery. Clin. Chem. 2012, 58, 139–147. [Google Scholar] [CrossRef] [PubMed]
- Ganna, A.; Salihovic, S.; Sundström, J.; Broeckling, C.D.; Hedman, A.K.; Magnusson, P.K.E.; Pedersen, N.L.; Larsson, A.; Siegbahn, A.; Zilmer, M.; et al. Large-scale metabolomic profiling identifies novel biomarkers for incident coronary heart disease. PLoS Genet. 2014, 10, e1004801. [Google Scholar] [CrossRef]
- Zivkovic, A.M.; Wiest, M.M.; Nguyen, U.T.; Davis, R.; Watkins, S.M.; German, J.B. Effects of sample handling and storage on quantitative lipid analysis in human serum. Metabolomics 2009, 5, 507–516. [Google Scholar] [CrossRef] [PubMed]
- Fliniaux, O.; Gaillard, G.; Lion, A.; Cailleu, D.; Mesnard, F.; Betsou, F. Influence of common preanalytical variations on the metabolic profile of serum samples in biobanks. J. Biomol. NMR 2011, 51, 457–465. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Chen, Y.; Xi, C.; Zhang, R.; Song, Y.; Zhan, Q.; Bi, X.; Abliz, Z. Liquid chromatography-tandem mass spectrometry-based plasma metabonomics delineate the effect of metabolites’ stability on reliability of potential biomarkers. Anal. Chem. 2013, 85, 2606–2610. [Google Scholar] [CrossRef] [PubMed]
- Yin, P.; Peter, A.; Franken, H.; Zhao, X.; Neukamm, S.S.; Rosenbaum, L.; Lucio, M.; Zell, A.; Häring, H.-U.; Xu, G.; et al. Preanalytical aspects and sample quality assessment in metabolomics studies of human blood. Clin. Chem. 2013, 59, 833–845. [Google Scholar] [CrossRef] [PubMed]
- Breier, M.; Wahl, S.; Prehn, C.; Fugmann, M.; Ferrari, U.; Weise, M.; Banning, F.; Seissler, J.; Grallert, H.; Adamski, J.; et al. Targeted metabolomics identifies reliable and stable metabolites in human serum and plasma samples. PLoS ONE 2014, 9, e89728. [Google Scholar] [CrossRef] [PubMed]
- Kamlage, B.; Maldonado, S.G.; Bethan, B.; Peter, E.; Schmitz, O.; Liebenberg, V.; Schatz, P. Quality markers addressing preanalytical variations of blood and plasma processing identified by broad and targeted metabolite profiling. Clin. Chem. 2014, 60, 399–412. [Google Scholar] [CrossRef] [PubMed]
- Anton, G.; Wilson, R.; Yu, Z.-H.; Prehn, C.; Zukunft, S.; Adamski, J.; Heier, M.; Meisinger, C.; Römisch-Margl, W.; Wang-Sattler, R.; et al. Pre-analytical sample quality: Metabolite ratios as an intrinsic marker for prolonged room temperature exposure of serum samples. PLoS ONE 2015, 10, e0121495. [Google Scholar] [CrossRef] [PubMed]
- Yin, P.; Lehmann, R.; Xu, G. Effects of pre-analytical processes on blood samples used in metabolomics studies. Anal. Bioanal. Chem. 2015, 407, 4879–4892. [Google Scholar] [CrossRef]
- Hirayama, A.; Sugimoto, M.; Suzuki, A.; Hatakeyama, Y.; Enomoto, A.; Harada, S.; Soga, T.; Tomita, M.; Takebayashi, T. Effects of processing and storage conditions on charged metabolomic profiles in blood. Electrophoresis 2015, 36, 2148–2155. [Google Scholar] [CrossRef] [PubMed]
- Kamlage, B.; Neuber, S.; Bethan, B.; González Maldonado, S.; Wagner-Golbs, A.; Peter, E.; Schmitz, O.; Schatz, P. Impact of Prolonged Blood Incubation and Extended Serum Storage at Room Temperature on the Human Serum Metabolome. Metabolites 2018, 8, 6. [Google Scholar] [CrossRef] [PubMed]
- Lippi, G.; Becan-McBride, K.; Behúlová, D.; Bowen, R.A.; Church, S.; Delanghe, J.; Grankvist, K.; Kitchen, S.; Nybo, M.; Nauck, M.; et al. Preanalytical quality improvement: In quality we trust. Clin. Chem. Lab. Med. 2013, 51, 229–241. [Google Scholar] [CrossRef] [PubMed]
- Diamandis, E.P. Cancer biomarkers: Can we turn recent failures into success? J. Natl. Cancer Inst. 2010, 102, 1462–1467. [Google Scholar] [CrossRef] [PubMed]
- Rai, A.J.; Vitzthum, F. Effects of preanalytical variables on peptide and protein measurements in human serum and plasma: Implications for clinical proteomics. Expert Rev. Proteom. 2006, 3, 409–426. [Google Scholar] [CrossRef] [PubMed]
- Aguilar-Mahecha, A.; Kuzyk, M.A.; Domanski, D.; Borchers, C.H.; Basik, M. The effect of pre-analytical variability on the measurement of MRM-MS-based mid- to high-abundance plasma protein biomarkers and a panel of cytokines. PLoS ONE 2012, 7, e38290. [Google Scholar] [CrossRef] [PubMed]
- Günther, K.; Malentacchi, F.; Verderio, P.; Pizzamiglio, S.; Ciniselli, C.M.; Tichopad, A.; Kubista, M.; Wyrich, R.; Pazzagli, M.; Gelmini, S. Implementation of a proficiency testing for the assessment of the preanalytical phase of blood samples used for RNA based analysis. Clin. Chim. Acta 2012, 413, 779–786. [Google Scholar] [CrossRef]
- Ellervik, C.; Vaught, J. Preanalytical variables affecting the integrity of human biospecimens in biobanking. Clin. Chem. 2015, 61, 914–934. [Google Scholar] [CrossRef] [PubMed]
- Dunn, W.B.; Broadhurst, D.; Begley, P.; Zelena, E.; Francis-McIntyre, S.; Anderson, N.; Brown, M.; Knowles, J.D.; Halsall, A.; Haselden, J.N.; et al. Procedures for large-scale metabolic profiling of serum and plasma using gas chromatography and liquid chromatography coupled to mass spectrometry. Nat. Protoc. 2011, 6, 1060–1083. [Google Scholar] [CrossRef] [PubMed]
- Abuja, P.M.; Ehrhart, F.; Schoen, U.; Schmidt, T.; Stracke, F.; Dallmann, G.; Friedrich, T.; Zimmermann, H.; Zatloukal, K. Alterations in Human Liver Metabolome during Prolonged Cryostorage. J. Proteome Res. 2015, 14, 2758–2768. [Google Scholar] [CrossRef]
- Jenab, M.; Bingham, S.; Ferrari, P.; Friesen, M.D.; Al-Delaimy, W.K.; Luben, R.; Wareham, N.; Khaw, K.-T.; Riboli, E. Long-term cryoconservation and stability of vitamin C in serum samples of the European prospective investigation into cancer and nutrition. Cancer Epidemiol. Prev. Biomark. 2005, 14, 1837–1840. [Google Scholar] [CrossRef] [PubMed]
- Bernini, P.; Bertini, I.; Luchinat, C.; Nincheri, P.; Staderini, S.; Turano, P. Standard operating procedures for pre-analytical handling of blood and urine for metabolomic studies and biobanks. J. Biomol. NMR 2011, 49, 231–243. [Google Scholar] [CrossRef] [PubMed]
- Shabihkhani, M.; Lucey, G.M.; Wei, B.; Mareninov, S.; Lou, J.J.; Vinters, H.V.; Singer, E.J.; Cloughesy, T.F.; Yong, W.H. The procurement, storage, and quality assurance of frozen blood and tissue biospecimens in pathology, biorepository, and biobank settings. Clin. Biochem. 2014, 47, 258–266. [Google Scholar] [CrossRef] [PubMed]
- Hubel, A.; Spindler, R.; Skubitz, A.P.N. Storage of human biospecimens: Selection of the optimal storage temperature. Biopreserv. Biobank. 2014, 12, 165–175. [Google Scholar] [CrossRef]
- Teahan, O.; Gamble, S.; Holmes, E.; Waxman, J.; Nicholson, J.K.; Bevan, C.; Keun, H.C. Impact of analytical bias in metabonomic studies of human blood serum and plasma. Anal. Chem. 2006, 78, 4307–4318. [Google Scholar] [CrossRef] [PubMed]
- Hebels, D.G.A.J.; Georgiadis, P.; Keun, H.C.; Athersuch, T.J.; Vineis, P.; Vermeulen, R.; Portengen, L.; Bergdahl, I.A.; Hallmans, G.; Palli, D.; et al. Performance in omics analyses of blood samples in long-term storage: Opportunities for the exploitation of existing biobanks in environmental health research. Environ. Health Perspect. 2013, 121, 480–487. [Google Scholar] [CrossRef] [PubMed]
- Moriya, T.; Satomi, Y.; Kobayashi, H. Intensive determination of storage condition effects on human plasma metabolomics. Metabolomics 2016, 12, 179. [Google Scholar] [CrossRef]
- Hustad, S.; Eussen, S.; Midttun, Ø.; Ulvik, A.; van de Kant, P.M.; Mørkrid, L.; Gislefoss, R.; Ueland, P.M. Kinetic modeling of storage effects on biomarkers related to B vitamin status and one-carbon metabolism. Clin. Chem. 2012, 58, 402–410. [Google Scholar] [CrossRef] [PubMed]
- Haid, M.; Muschet, C.; Wahl, S.; Römisch-Margl, W.; Prehn, C.; Möller, G.; Adamski, J. Long-Term Stability of Human Plasma Metabolites during Storage at −80 °C. J. Proteome Res. 2018, 17, 203–211. [Google Scholar] [CrossRef] [PubMed]
- Onsager, L.; Runnels, L.K. Diffusion and Relaxation Phenomena in Ice. J. Chem. Phys. 1969, 50, 1089–1103. [Google Scholar] [CrossRef]
- Eigen, M.; de Mayer, L. Self-dissociation and protonic charge transport in water and. Proc. R. Soc. Lond. A 1958, 247, 505–533. [Google Scholar] [CrossRef]
- Mullen, S.F.; Critser, J.K. The science of cryobiology. Cancer Treat. Res. 2007, 138, 83–109. [Google Scholar]
- Wright, H.T. Nonenzymatic deamidation of asparaginyl and glutaminyl residues in proteins. Crit. Rev. Biochem. Mol. Biol. 1991, 26, 1–52. [Google Scholar] [CrossRef]
- Liu, L.; Aa, J.; Wang, G.; Yan, B.; Zhang, Y.; Wang, X.; Zhao, C.; Cao, B.; Shi, J.; Li, M.; et al. Differences in metabolite profile between blood plasma and serum. Anal. Biochem. 2010, 406, 105–112. [Google Scholar] [CrossRef]
- Bauldry, S.A.; Wooten, R.E.; Bass, D.A. Activation of cytosolic phospholipase A2 in permeabilized human neutrophils. Biochim. Biophys. Acta 1996, 1299, 223–234. [Google Scholar] [CrossRef]
- Steinbrecher, U.P.; Pritchard, P.H. Hydrolysis of phosphatidylcholine during LDL oxidation is mediated by platelet-activating factor acetylhydrolase. J. Lipid Res. 1989, 30, 305–315. [Google Scholar] [PubMed]
- Derksen, A.; Cohen, P. Patterns of fatty acid release from endogenous substrates by human platelet homogenates and membranes. J. Biol. Chem. 1975, 250, 9342–9347. [Google Scholar] [PubMed]
- Kuehl, F.A.; Egan, R.W. Prostaglandins, arachidonic acid, and inflammation. Science 1980, 210, 978–984. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, Y.; Umeno, A.; Shichiri, M. Lipid peroxidation biomarkers for evaluating oxidative stress and assessing antioxidant capacity in vivo. J. Clin. Biochem. Nutr. 2013, 52, 9–16. [Google Scholar] [CrossRef] [PubMed]
- Lind, L.; Fors, N.; Hall, J.; Marttala, K.; Stenborg, A. A comparison of three different methods to evaluate endothelium-dependent vasodilation in the elderly: The Prospective Investigation of the Vasculature in Uppsala Seniors (PIVUS) study. Arterioscler. Thromb. Vasc. Biol. 2005, 25, 2368–2375. [Google Scholar] [CrossRef]
- Lind, L.; Salihovic, S.; Lampa, E.; Lind, P.M. Mixture effects of 30 environmental contaminants on incident metabolic syndrome-A prospective study. Environ. Int. 2017, 107, 8–15. [Google Scholar] [CrossRef] [PubMed]
- van Ravenzwaay, B.; Cunha, G.C.-P.; Leibold, E.; Looser, R.; Mellert, W.; Prokoudine, A.; Walk, T.; Wiemer, J. The use of metabolomics for the discovery of new biomarkers of effect. Toxicol. Lett. 2007, 172, 21–28. [Google Scholar] [CrossRef]
- Mutch, D.M.; Fuhrmann, J.C.; Rein, D.; Wiemer, J.C.; Bouillot, J.-L.; Poitou, C.; Clément, K. Metabolite profiling identifies candidate markers reflecting the clinical adaptations associated with Roux-en-Y gastric bypass surgery. PLoS ONE 2009, 4, e7905. [Google Scholar] [CrossRef] [PubMed]
- Walk, T.B.; Dostler, M. Mass Spectrometry Method for Analysing Mixtures of Substances. International Patent Application PCT/EP2003/001274, 10 February 2003. [Google Scholar]
- Meller, S.; Meyer, H.-A.; Bethan, B.; Dietrich, D.; Maldonado, S.G.; Lein, M.; Montani, M.; Reszka, R.; Schatz, P.; Peter, E.; et al. Integration of tissue metabolomics, transcriptomics and immunohistochemistry reveals ERG- and gleason score-specific metabolomic alterations in prostate cancer. Oncotarget 2016, 7, 1421–1438. [Google Scholar] [CrossRef] [PubMed]
- Kamlage, B.; Schmitz, O.; Bethan, B.; Schatz, P.; Peter, E. Means and Methods for Determination of Quality of Blood Samples Based on Metabolite Panel. International Patent Application PCT/IB2015/052243, 26 March 2014. [Google Scholar]
- Benjamini, Y.; Hochberg, Y. Controlling the False Discovery Rate: A Practical and Powerful Approach to Multiple Testing. J. R. Stat. Soc. Ser. B (Methodol.) 1995, 1995, 289–300. [Google Scholar] [CrossRef]
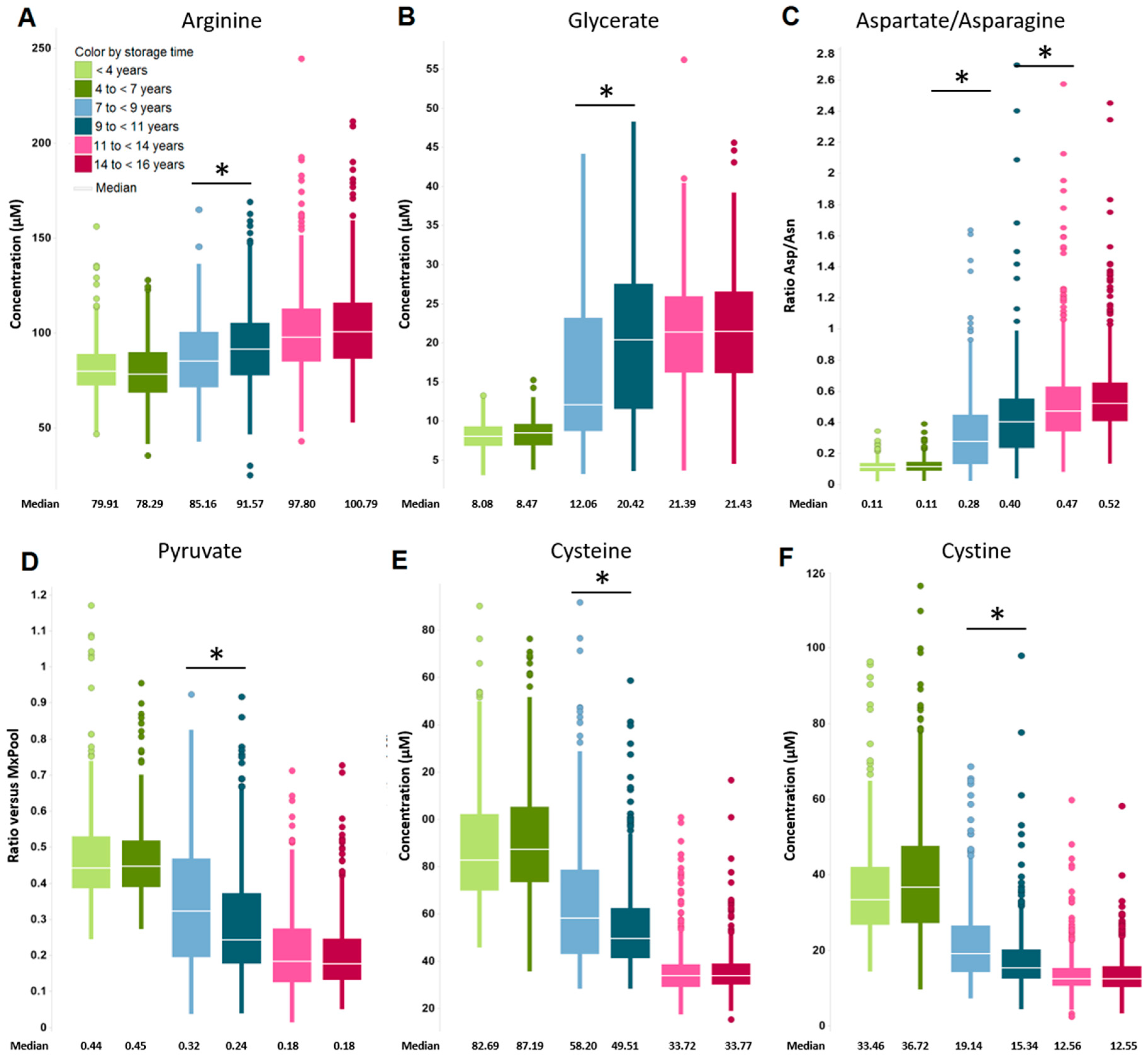

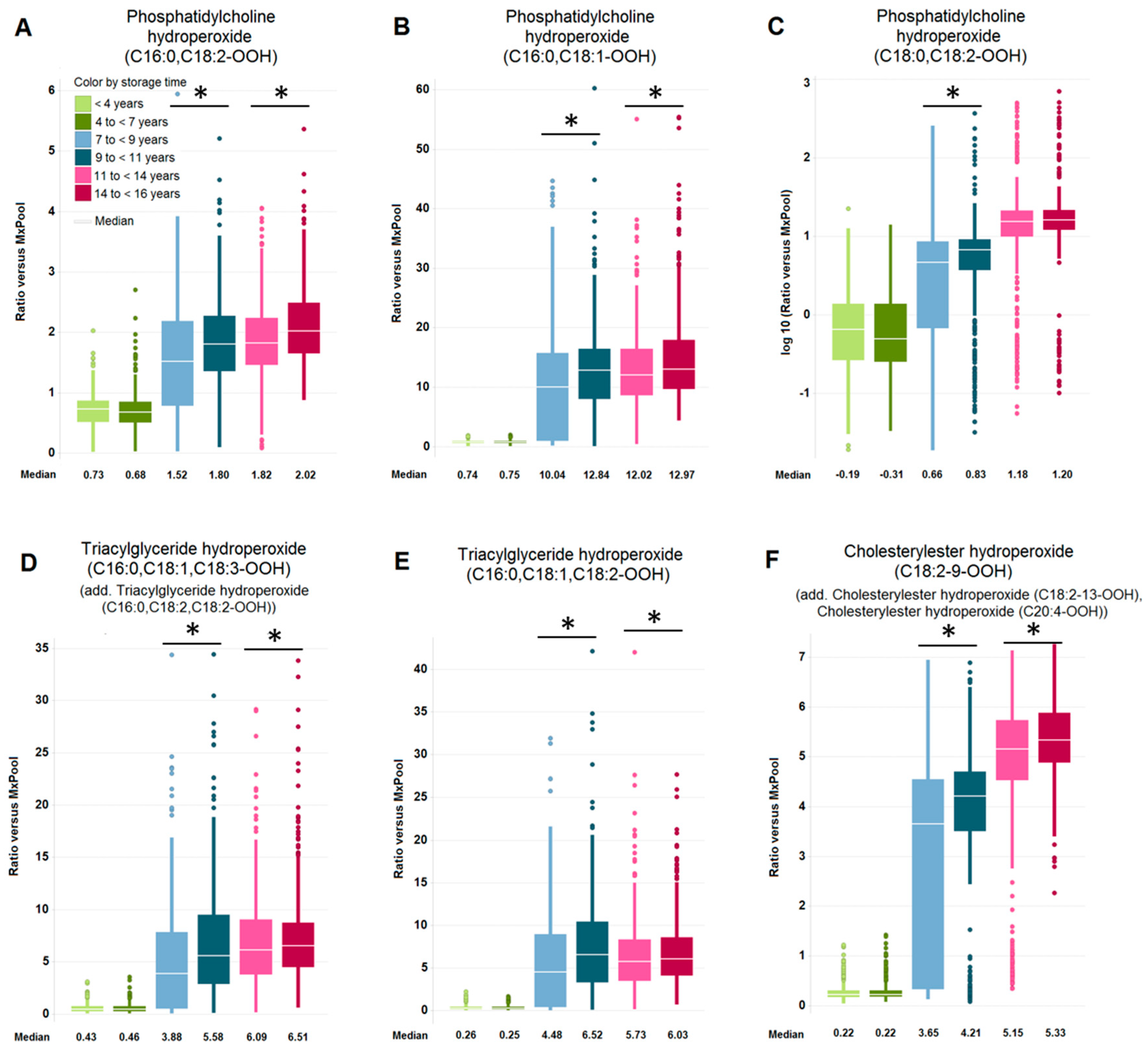
| Significantly Changed Metabolites Long Storage vs. Shorter Storage within Subject Age Groups (Increase/Decrease). Inconsistencies in Percentage Sums are Due to Rounding. | ||||||
|---|---|---|---|---|---|---|
| Storage for 4 to <7 years vs. <4 years (subject age 80 years) | Storage for 9 to <11 years vs. 7 to <9 years (subject age 75 years) | Storage for 14 to <16 years vs. 11 to <14 years (subject age 70 years) | ||||
| Metabolite Ontology Class (Number of Metabolites) | Number | Percent Change | Number | Percent Change | Number | Percent Change |
| All (231) | 5 (4/1) | 2 (2/0) | 59 (46/13) | 26 (20/6) | 27 (17/10) | 12 (7/4) |
| Amino acids (22) | 1 (1/0) | 5 (5/0) | 8 (4/4) | 36 (18/18) | 1 (1/0) | 5 (5/0) |
| Amino acids related (15) | 0 | 0 | 3 (2/1) | 20 (13/7) | 0 | 0 |
| Carbohydrates and related (17) | 0 | 0 | 2 (2/0) | 12 (12/0) | 2 (0/2) | 12 (0/12) |
| Complex lipids, fatty acids and related (101) | 0 | 0 | 27 (24/3) | 27 (24/3) | 15 (10/5) | 15 (10/5) |
| Energy metabolism and related (17) | 1 (1/0) | 6 (6/0) | 5 (3/2) | 29 (17/12) | 2 (1/1) | 12 (6/6) |
| Miscellaneous (8) | 1 (1/0) | 13 (13/0) | 2 (0/2) | 25 (0/25) | 0 | 0 |
| Nucleobases and related (5) | 0 | 0 | 0 | 0 | 1 (0/1) | 20 (0/20) |
| Vitamins, cofactors and related (6) | 1 (1/0) | 17 (17/0) | 1 (1/0) | 17 (17/0) | 0 | 0 |
| Unknowns (38) | 1 (0/1) | 3 (0/3) | 11 (10/1) | 29 (26/3) | 6 (5/1) | 16 (13/3) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wagner-Golbs, A.; Neuber, S.; Kamlage, B.; Christiansen, N.; Bethan, B.; Rennefahrt, U.; Schatz, P.; Lind, L. Effects of Long-Term Storage at −80 °C on the Human Plasma Metabolome. Metabolites 2019, 9, 99. https://doi.org/10.3390/metabo9050099
Wagner-Golbs A, Neuber S, Kamlage B, Christiansen N, Bethan B, Rennefahrt U, Schatz P, Lind L. Effects of Long-Term Storage at −80 °C on the Human Plasma Metabolome. Metabolites. 2019; 9(5):99. https://doi.org/10.3390/metabo9050099
Chicago/Turabian StyleWagner-Golbs, Antje, Sebastian Neuber, Beate Kamlage, Nicole Christiansen, Bianca Bethan, Ulrike Rennefahrt, Philipp Schatz, and Lars Lind. 2019. "Effects of Long-Term Storage at −80 °C on the Human Plasma Metabolome" Metabolites 9, no. 5: 99. https://doi.org/10.3390/metabo9050099
APA StyleWagner-Golbs, A., Neuber, S., Kamlage, B., Christiansen, N., Bethan, B., Rennefahrt, U., Schatz, P., & Lind, L. (2019). Effects of Long-Term Storage at −80 °C on the Human Plasma Metabolome. Metabolites, 9(5), 99. https://doi.org/10.3390/metabo9050099





