Metabolomics Identifies Novel Blood Biomarkers of Pulmonary Function and COPD in the General Population
Abstract
1. Introduction
2. Results
2.1. Study Characteristics
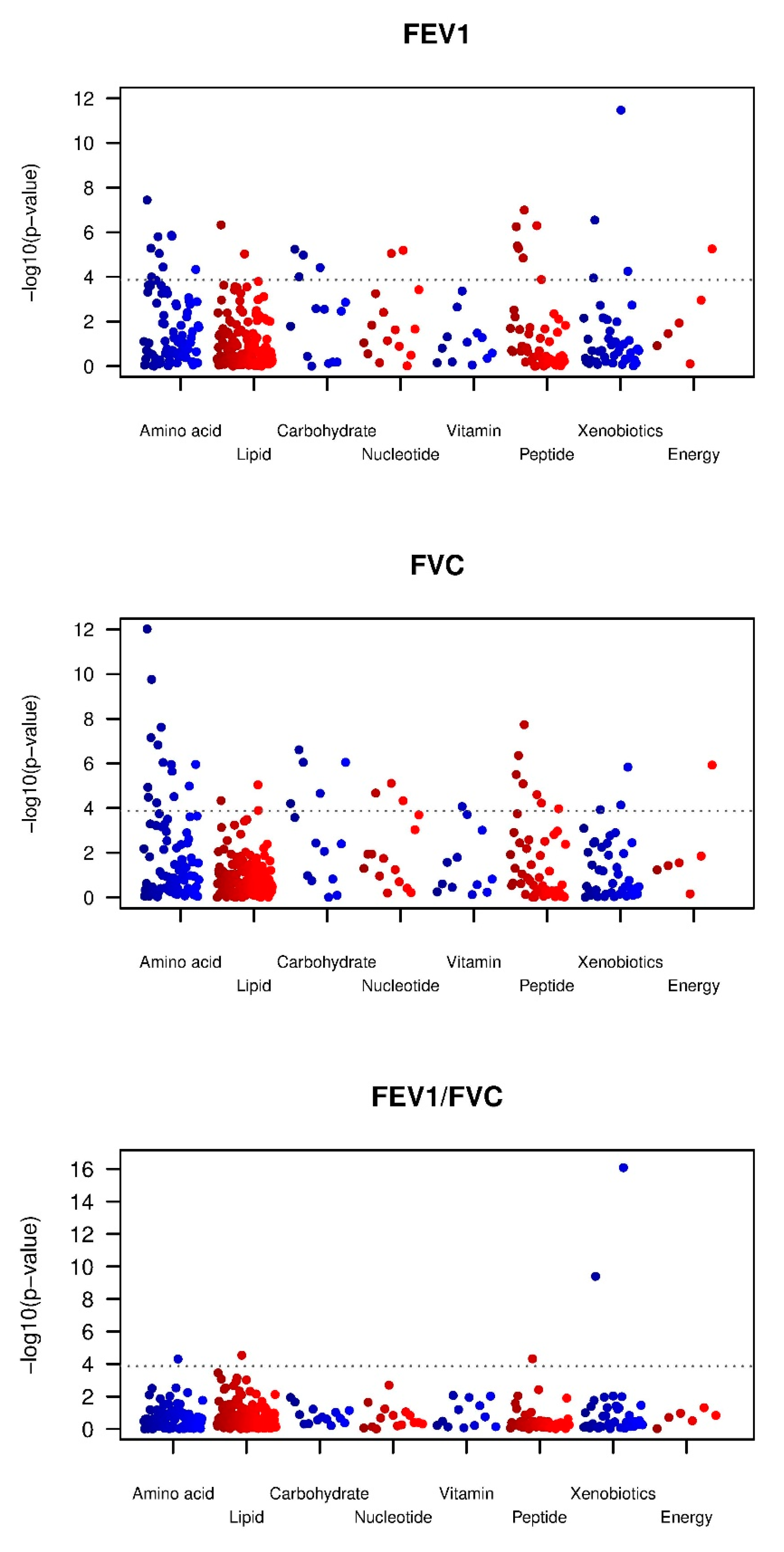
2.2. Metabolic Associations with Pulmonary Function Measures
2.3. Pathway Analyses
2.4. Metabolic Associations with COPD
2.5. Consistency of Associations
3. Discussion
4. Material and Methods
4.1. Study Populations
4.2. Metabolome Measurements
4.3. Assessment of Pulmonary Function and Covariates
4.4. Human Subjects
4.5. Statistical Analyses
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Data availability
References
- Shaaban, R.; Kony, S.; Driss, F.; Leynaert, B.; Soussan, D.; Pin, I.; Neukirch, F.; Zureik, M. Change in C-reactive protein levels and FEV1 decline: A longitudinal population-based study. Respir. Med. 2006, 100, 2112–2120. [Google Scholar] [CrossRef]
- Dahl, M.; Tybjaerg-Hansen, A.; Vestbo, J.; Lange, P.; Nordestgaard, B.G. Elevated plasma fibrinogen associated with reduced pulmonary function and increased risk of chronic obstructive pulmonary disease. Am. J. Respir. Crit. Care Med. 2001, 164, 1008–1011. [Google Scholar] [CrossRef]
- Chatterjee, R.; Brancati, F.L.; Shafi, T.; Edelman, D.; Pankow, J.S.; Mosley, T.H.; Selvin, E.; Yeh, H.C. Non-traditional risk factors are important contributors to the racial disparity in diabetes risk: The atherosclerosis risk in communities study. J. Gen. Intern. Med. 2014, 29, 290–297. [Google Scholar] [CrossRef] [PubMed]
- Sumida, K.; Kwak, L.; Grams, M.E.; Yamagata, K.; Punjabi, N.M.; Kovesdy, C.P.; Coresh, J.; Matsushita, K. Lung Function and Incident Kidney Disease: The Atherosclerosis Risk in Communities (ARIC) Study. Am. J. Kidney Dis. 2017, 70, 675–685. [Google Scholar] [CrossRef]
- Li, J.; Agarwal, S.K.; Alonso, A.; Blecker, S.; Chamberlain, A.M.; London, S.J.; Loehr, L.R.; McNeill, A.M.; Poole, C.; Soliman, E.Z.; et al. Airflow obstruction, lung function, and incidence of atrial fibrillation: The Atherosclerosis Risk in Communities (ARIC) study. Circulation 2014, 129, 971–980. [Google Scholar] [CrossRef]
- Schroeder, E.B.; Welch, V.L.; Couper, D.; Nieto, F.J.; Liao, D.; Rosamond, W.D.; Heiss, G. Lung function and incident coronary heart disease: The Atherosclerosis Risk in Communities Study. Am. J. Epidemiol. 2003, 158, 1171–1181. [Google Scholar] [CrossRef]
- Hancock, D.B.; Eijgelsheim, M.; Wilk, J.B.; Gharib, S.A.; Loehr, L.R.; Marciante, K.D.; Franceschini, N.; van Durme, Y.M.; Chen, T.H.; Barr, R.G.; et al. Meta-analyses of genome-wide association studies identify multiple loci associated with pulmonary function. Nat. Genet. 2010, 42, 45–52. [Google Scholar] [CrossRef] [PubMed]
- Loth, D.W.; Soler Artigas, M.; Gharib, S.A.; Wain, L.V.; Franceschini, N.; Koch, B.; Pottinger, T.D.; Smith, A.V.; Duan, Q.; Oldmeadow, C.; et al. Genome-wide association analysis identifies six new loci associated with forced vital capacity. Nat. Genet. 2014, 46, 669–677. [Google Scholar] [CrossRef] [PubMed]
- Collaborators, G.B.D.R.F. Global, regional, and national comparative risk assessment of 84 behavioural, environmental and occupational, and metabolic risks or clusters of risks, 1990-2016: A systematic analysis for the Global Burden of Disease Study 2016. Lancet 2017, 390, 1345–1422. [Google Scholar] [CrossRef]
- Burney, P.G.; Hooper, R. Forced vital capacity, airway obstruction and survival in a general population sample from the USA. Thorax 2011, 66, 49–54. [Google Scholar] [CrossRef]
- Young, R.P.; Hopkins, R.; Eaton, T.E. Forced expiratory volume in one second: Not just a lung function test but a marker of premature death from all causes. Eur. Respir. J. 2007, 30, 616–622. [Google Scholar] [CrossRef] [PubMed]
- Schunemann, H.J.; Dorn, J.; Grant, B.J.; Winkelstein, W., Jr.; Trevisan, M. Pulmonary function is a long-term predictor of mortality in the general population: 29-year follow-up of the Buffalo Health Study. Chest 2000, 118, 656–664. [Google Scholar] [CrossRef] [PubMed]
- Mannino, D.M.; Buist, A.S.; Petty, T.L.; Enright, P.L.; Redd, S.C. Lung function and mortality in the United States: Data from the First National Health and Nutrition Examination Survey follow up study. Thorax 2003, 58, 388–393. [Google Scholar] [CrossRef]
- Abbiss, H.; Maker, G.L.; Trengove, R.D. Metabolomics Approaches for the Diagnosis and Understanding of Kidney Diseases. Metabolites 2019, 9, 34. [Google Scholar] [CrossRef] [PubMed]
- Carulli, L.; Zanca, G.; Schepis, F.; Villa, E. The OMICs Window into Nonalcoholic Fatty Liver Disease (NAFLD). Metabolites 2019, 9, 25. [Google Scholar] [CrossRef] [PubMed]
- Zhai, G. Alteration of Metabolic Pathways in Osteoarthritis. Metabolites 2019, 9, 11. [Google Scholar] [CrossRef]
- Havelund, J.F.; Heegaard, N.H.H.; Faergeman, N.J.K.; Gramsbergen, J.B. Biomarker Research in Parkinson’s Disease Using Metabolite Profiling. Metabolites 2017, 7, 42. [Google Scholar] [CrossRef]
- Kilk, K.; Aug, A.; Ottas, A.; Soomets, U.; Altraja, S.; Altraja, A. Phenotyping of Chronic Obstructive Pulmonary Disease Based on the Integration of Metabolomes and Clinical Characteristics. Int. J. Mol. Sci. 2018, 19, 666. [Google Scholar] [CrossRef]
- Naz, S.; Kolmert, J.; Yang, M.; Reinke, S.N.; Kamleh, M.A.; Snowden, S.; Heyder, T.; Levanen, B.; Erle, D.J.; Skold, C.M.; et al. Metabolomics analysis identifies sex-associated metabotypes of oxidative stress and the autotaxin-lysoPA axis in COPD. Eur. Respir. J. 2017, 49. [Google Scholar] [CrossRef]
- Hodgson, S.; Griffin, T.J.; Reilly, C.; Harvey, S.; Witthuhn, B.A.; Sandri, B.J.; Kunisaki, K.M.; Wendt, C.H. Plasma sphingolipids in HIV-associated chronic obstructive pulmonary disease. BMJ Open Respir. Res. 2017, 4, e000180. [Google Scholar] [CrossRef]
- Chen, Q.; Deeb, R.S.; Ma, Y.; Staudt, M.R.; Crystal, R.G.; Gross, S.S. Serum Metabolite Biomarkers Discriminate Healthy Smokers from COPD Smokers. PLoS ONE 2015, 10, e0143937. [Google Scholar] [CrossRef]
- Zabek, A.; Stanimirova, I.; Deja, S.; Barg, W.; Kowal, A.; Korzeniewska, A.; Orczyk-Pawilowicz, M.; Baranowski, D.; Gdaniec, Z.; Jankowska, R.; et al. Fusion of the (1)H NMR data of serum, urine and exhaled breath condensate in order to discriminate chronic obstructive pulmonary disease and obstructive sleep apnea syndrome. Metabolomics 2015, 11, 1563–1574. [Google Scholar] [CrossRef] [PubMed]
- Adamko, D.J.; Nair, P.; Mayers, I.; Tsuyuki, R.T.; Regush, S.; Rowe, B.H. Metabolomic profiling of asthma and chronic obstructive pulmonary disease: A pilot study differentiating diseases. J. Allergy Clin. Immunol. 2015, 136, 571–580.e3. [Google Scholar] [CrossRef]
- Deja, S.; Porebska, I.; Kowal, A.; Zabek, A.; Barg, W.; Pawelczyk, K.; Stanimirova, I.; Daszykowski, M.; Korzeniewska, A.; Jankowska, R.; et al. Metabolomics provide new insights on lung cancer staging and discrimination from chronic obstructive pulmonary disease. J. Pharm. Biomed. Anal. 2014, 100, 369–380. [Google Scholar] [CrossRef]
- Ubhi, B.K.; Cheng, K.K.; Dong, J.; Janowitz, T.; Jodrell, D.; Tal-Singer, R.; MacNee, W.; Lomas, D.A.; Riley, J.H.; Griffin, J.L.; et al. Targeted metabolomics identifies perturbations in amino acid metabolism that sub-classify patients with COPD. Mol. Biosyst. 2012, 8, 3125–3133. [Google Scholar] [CrossRef]
- Paige, M.; Burdick, M.D.; Kim, S.; Xu, J.; Lee, J.K.; Shim, Y.M. Pilot analysis of the plasma metabolite profiles associated with emphysematous Chronic Obstructive Pulmonary Disease phenotype. Biochem. Biophys. Res. Commun. 2011, 413, 588–593. [Google Scholar] [CrossRef]
- McClay, J.L.; Adkins, D.E.; Isern, N.G.; O’Connell, T.M.; Wooten, J.B.; Zedler, B.K.; Dasika, M.S.; Webb, B.T.; Webb-Robertson, B.J.; Pounds, J.G.; et al. (1)H nuclear magnetic resonance metabolomics analysis identifies novel urinary biomarkers for lung function. J. Proteome Res. 2010, 9, 3083–3090. [Google Scholar] [CrossRef]
- Novotna, B.; Abdel-Hamid, M.; Koblizek, V.; Svoboda, M.; Hejduk, K.; Rehacek, V.; Bis, J.; Salajka, F. A pilot data analysis of a metabolomic HPLC-MS/MS study of patients with COPD. Adv. Clin. Exp. Med. 2018, 27, 531–539. [Google Scholar] [CrossRef] [PubMed]
- Bowler, R.P.; Jacobson, S.; Cruickshank, C.; Hughes, G.J.; Siska, C.; Ory, D.S.; Petrache, I.; Schaffer, J.E.; Reisdorph, N.; Kechris, K. Plasma sphingolipids associated with chronic obstructive pulmonary disease phenotypes. Am. J. Respir. Crit. Care Med. 2015, 191, 275–284. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Tang, Y.; Liu, S.; Mao, S.; Ling, Y.; Liu, D.; He, X.; Wang, X. Metabonomic profiling of serum and urine by (1)H NMR-based spectroscopy discriminates patients with chronic obstructive pulmonary disease and healthy individuals. PLoS ONE 2013, 8, e65675. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, N.; Dutta, M.; Singh, B.; Banerjee, R.; Bhattacharyya, P.; Chaudhury, K. Transcriptomics, proteomics and metabolomics driven biomarker discovery in COPD: An update. Expert Rev. Mol. Diagn. 2016, 16, 897–913. [Google Scholar] [CrossRef] [PubMed]
- Menni, C.; Metrustry, S.J.; Mohney, R.P.; Beevers, S.; Barratt, B.; Spector, T.D.; Kelly, F.J.; Valdes, A.M. Circulating levels of antioxidant vitamins correlate with better lung function and reduced exposure to ambient pollution. Am. J. Respir. Crit. Care Med. 2015, 191, 1203–1207. [Google Scholar] [CrossRef] [PubMed]
- Menni, C.; Kastenmuller, G.; Petersen, A.K.; Bell, J.T.; Psatha, M.; Tsai, P.C.; Gieger, C.; Schulz, H.; Erte, I.; John, S.; et al. Metabolomic markers reveal novel pathways of ageing and early development in human populations. Int. J. Epidemiol. 2013, 42, 1111–1119. [Google Scholar] [CrossRef]
- Sin, D.D.; Hollander, Z.; DeMarco, M.L.; McManus, B.M.; Ng, R.T. Biomarker Development for Chronic Obstructive Pulmonary Disease. From Discovery to Clinical Implementation. Am. J. Respir. Crit. Care Med. 2015, 192, 1162–1170. [Google Scholar] [CrossRef] [PubMed]
- Telenga, E.D.; Hoffmann, R.F.; Ruben, t.K.; Hoonhorst, S.J.; Willemse, B.W.; van Oosterhout, A.J.; Heijink, I.H.; van den Berge, M.; Jorge, L.; Sandra, P.; et al. Untargeted lipidomic analysis in chronic obstructive pulmonary disease. Uncovering sphingolipids. Am. J. Respir. Crit. Care Med. 2014, 190, 155–164. [Google Scholar] [CrossRef]
- Ubhi, B.K.; Riley, J.H.; Shaw, P.A.; Lomas, D.A.; Tal-Singer, R.; MacNee, W.; Griffin, J.L.; Connor, S.C. Metabolic profiling detects biomarkers of protein degradation in COPD patients. Eur. Respir. J. 2012, 40, 345–355. [Google Scholar] [CrossRef]
- White, E.; Kristal, A.R.; Shikany, J.M.; Wilson, A.C.; Chen, C.; Mares-Perlman, J.A.; Masaki, K.H.; Caan, B.J. Correlates of serum alpha- and gamma-tocopherol in the Women’s Health Initiative. Ann. Epidemiol. 2001, 11, 136–144. [Google Scholar] [CrossRef]
- Kohansal, R.; Martinez-Camblor, P.; Agusti, A.; Buist, A.S.; Mannino, D.M.; Soriano, J.B. The natural history of chronic airflow obstruction revisited: An analysis of the Framingham offspring cohort. Am. J. Respir. Crit. Care Med. 2009, 180, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Eisner, M.D.; Wang, Y.; Haight, T.J.; Balmes, J.; Hammond, S.K.; Tager, I.B. Secondhand smoke exposure, pulmonary function, and cardiovascular mortality. Ann. Epidemiol. 2007, 17, 364–373. [Google Scholar] [CrossRef] [PubMed]
- Newgard, C.B.; An, J.; Bain, J.R.; Muehlbauer, M.J.; Stevens, R.D.; Lien, L.F.; Haqq, A.M.; Shah, S.H.; Arlotto, M.; Slentz, C.A.; et al. A branched-chain amino acid-related metabolic signature that differentiates obese and lean humans and contributes to insulin resistance. Cell Metab. 2009, 9, 311–326. [Google Scholar] [CrossRef] [PubMed]
- Newgard, C.B. Metabolomics and Metabolic Diseases: Where Do We Stand? Cell Metab. 2017, 25, 43–56. [Google Scholar] [CrossRef]
- Palmer, N.D.; Stevens, R.D.; Antinozzi, P.A.; Anderson, A.; Bergman, R.N.; Wagenknecht, L.E.; Newgard, C.B.; Bowden, D.W. Metabolomic profile associated with insulin resistance and conversion to diabetes in the Insulin Resistance Atherosclerosis Study. J. Clin. Endocrinol. Metab. 2015, 100, E463–E468. [Google Scholar] [CrossRef]
- Shah, S.H.; Crosslin, D.R.; Haynes, C.S.; Nelson, S.; Turer, C.B.; Stevens, R.D.; Muehlbauer, M.J.; Wenner, B.R.; Bain, J.R.; Laferrere, B.; et al. Branched-chain amino acid levels are associated with improvement in insulin resistance with weight loss. Diabetologia 2012, 55, 321–330. [Google Scholar] [CrossRef]
- Wang, T.J.; Larson, M.G.; Vasan, R.S.; Cheng, S.; Rhee, E.P.; McCabe, E.; Lewis, G.D.; Fox, C.S.; Jacques, P.F.; Fernandez, C.; et al. Metabolite profiles and the risk of developing diabetes. Nat. Med. 2011, 17, 448–453. [Google Scholar] [CrossRef]
- Crowley, G.; Kwon, S.; Haider, S.H.; Caraher, E.J.; Lam, R.; St-Jules, D.E.; Liu, M.; Prezant, D.J.; Nolan, A. Metabolomics of World Trade Center-Lung Injury: A machine learning approach. BMJ Open Respir. Res. 2018, 5, e000274. [Google Scholar] [CrossRef]
- Zhao, H.; Dennery, P.A.; Yao, H. Metabolic reprogramming in the pathogenesis of chronic lung diseases, including BPD, COPD, and pulmonary fibrosis. Am. J. Physiol. Lung Cell Mol. Physiol. 2018, 314, L544–L554. [Google Scholar] [CrossRef]
- Yu, J.C.; Jiang, Z.M.; Li, D.M. Glutamine: A precursor of glutathione and its effect on liver. World J. Gastroenterol. 1999, 5, 143–146. [Google Scholar] [CrossRef]
- Oh, J.Y.; Lee, Y.S.; Min, K.H.; Hur, G.Y.; Lee, S.Y.; Kang, K.H.; Rhee, C.K.; Park, S.J.; Khan, A.; Na, J.; et al. Increased urinary l-histidine in patients with asthma-COPD overlap: A pilot study. Int. J. Chron. Obstruct. Pulmon. Dis. 2018, 13, 1809–1818. [Google Scholar] [CrossRef] [PubMed]
- Kelly, R.S.; Virkud, Y.; Giorgio, R.; Celedon, J.C.; Weiss, S.T.; Lasky-Su, J. Metabolomic profiling of lung function in Costa-Rican children with asthma. Biochim. Biophys. Acta 2017, 1863, 1590–1595. [Google Scholar] [CrossRef] [PubMed]
- Cruickshank-Quinn, C.I.; Jacobson, S.; Hughes, G.; Powell, R.L.; Petrache, I.; Kechris, K.; Bowler, R.; Reisdorph, N. Metabolomics and transcriptomics pathway approach reveals outcome-specific perturbations in COPD. Sci. Rep. 2018, 8, 17132. [Google Scholar] [CrossRef] [PubMed]
- Choi, G.G.; Han, Y.; Weston, B.; Ciftci, E.; Werner, T.J.; Torigian, D.; Salavati, A.; Alavi, A. Metabolic effects of pulmonary obstruction on myocardial functioning: A pilot study using multiple time-point 18F-FDG-PET imaging. Nucl. Med. Commun. 2015, 36, 78–83. [Google Scholar] [CrossRef] [PubMed]
- Conlon, T.M.; Bartel, J.; Ballweg, K.; Gunter, S.; Prehn, C.; Krumsiek, J.; Meiners, S.; Theis, F.J.; Adamski, J.; Eickelberg, O.; et al. Metabolomics screening identifies reduced L-carnitine to be associated with progressive emphysema. Clin. Sci. 2016, 130, 273–287. [Google Scholar] [CrossRef]
- Calverley, P.M.; Burge, P.S.; Spencer, S.; Anderson, J.A.; Jones, P.W. Bronchodilator reversibility testing in chronic obstructive pulmonary disease. Thorax 2003, 58, 659–664. [Google Scholar] [CrossRef] [PubMed]
- Lange, P.; Celli, B.; Agusti, A.; Boje Jensen, G.; Divo, M.; Faner, R.; Guerra, S.; Marott, J.L.; Martinez, F.D.; Martinez-Camblor, P.; et al. Lung-Function Trajectories Leading to Chronic Obstructive Pulmonary Disease. N. Engl. J. Med. 2015, 373, 111–122. [Google Scholar] [CrossRef] [PubMed]
- Hobbs, B.D.; de Jong, K.; Lamontagne, M.; Bosse, Y.; Shrine, N.; Artigas, M.S.; Wain, L.V.; Hall, I.P.; Jackson, V.E.; Wyss, A.B.; et al. Genetic loci associated with chronic obstructive pulmonary disease overlap with loci for lung function and pulmonary fibrosis. Nat. Genet. 2017, 49, 426–432. [Google Scholar] [CrossRef] [PubMed]
- The ARIC investigators. The Atherosclerosis Risk in Communities (ARIC) Study: Design and objectives. Am. J. Epidemiol. 1989, 129, 687–702. [Google Scholar]
- Holle, R.; Happich, M.; Lowel, H.; Wichmann, H.E.; Group, M.K.S. KORA—A research platform for population based health research. Gesundheitswesen 2005, 67 (Suppl. S1), S19–S25. [Google Scholar] [CrossRef]
- Lowel, H.; Doring, A.; Schneider, A.; Heier, M.; Thorand, B.; Meisinger, C.; Group, M.K.S. The MONICA Augsburg surveys—Basis for prospective cohort studies. Gesundheitswesen 2005, 67 (Suppl. S1), S13–S18. [Google Scholar] [CrossRef]
- Ohta, T.; Masutomi, N.; Tsutsui, N.; Sakairi, T.; Mitchell, M.; Milburn, M.V.; Ryals, J.A.; Beebe, K.D.; Guo, L. Untargeted metabolomic profiling as an evaluative tool of fenofibrate-induced toxicology in Fischer 344 male rats. Toxicol. Pathol. 2009, 37, 521–535. [Google Scholar] [CrossRef]
- Evans, A.M.; DeHaven, C.D.; Barrett, T.; Mitchell, M.; Milgram, E. Integrated, nontargeted ultrahigh performance liquid chromatography/electrospray ionization tandem mass spectrometry platform for the identification and relative quantification of the small-molecule complement of biological systems. Anal. Chem. 2009, 81, 6656–6667. [Google Scholar] [CrossRef] [PubMed]
- Suhre, K.; Shin, S.Y.; Petersen, A.K.; Mohney, R.P.; Meredith, D.; Wagele, B.; Altmaier, E.; CardioGram; Deloukas, P.; Erdmann, J.; et al. Human metabolic individuality in biomedical and pharmaceutical research. Nature 2011, 477, 54–60. [Google Scholar] [CrossRef]
- The National Heart and Blood Institute. Atherosclerosis Risk in Communities (ARIC) Study; Quality Assurance and Quality Control, Version 1.0; ARIC Coordinating Center, School of Public Health, University of North Carolina: Chapel Hill, NC, USA, 1989. Available online: https://www.ncbi.nlm.nih.gov/projects/gap/cgi-bin/GetPdf.cgi?id=phd000235.1 (accessed on 29 March 2019).
- Quanjer, P.H.; Stanojevic, S.; Cole, T.J.; Baur, X.; Hall, G.L.; Culver, B.H.; Enright, P.L.; Hankinson, J.L.; Ip, M.S.; Zheng, J.; et al. Multi-ethnic reference values for spirometry for the 3-95-yr age range: The global lung function 2012 equations. Eur. Respir. J. 2012, 40, 1324–1343. [Google Scholar] [CrossRef]
- Levey, A.S.; Stevens, L.A.; Schmid, C.H.; Zhang, Y.L.; Castro, A.F., 3rd; Feldman, H.I.; Kusek, J.W.; Eggers, P.; Van Lente, F.; Greene, T.; et al. A new equation to estimate glomerular filtration rate. Ann. Intern. Med. 2009, 150, 604–612. [Google Scholar] [CrossRef] [PubMed]
- Hankinson, J.L.; Odencrantz, J.R.; Fedan, K.B. Spirometric reference values from a sample of the general U.S. population. Am. J. Respir. Crit. Care Med. 1999, 159, 179–187. [Google Scholar] [CrossRef] [PubMed]
- Rice, K.; Higgins, J.; Lumley, T. A re-evaluation of fixed effect(s) meta-analysis. J. R. Stat. Soc. 2018, 181, 205–227. [Google Scholar] [CrossRef]
- R Core Team, R Foundation for Statistical Computing. R: A Language and Environment for Statistical Computing. Vienna, Austria, 2017. Available online: https://www.R-project.org/ (accessed on 27 March 2019).
- Xia, J.; Sinelnikov, I.V.; Han, B.; Wishart, D.S. MetaboAnalyst 3.0—Making metabolomics more meaningful. Nucleic Acids Res. 2015, 43, W251–W257. [Google Scholar] [CrossRef] [PubMed]
- Wishart, D.S.; Jewison, T.; Guo, A.C.; Wilson, M.; Knox, C.; Liu, Y.; Djoumbou, Y.; Mandal, R.; Aziat, F.; Dong, E.; et al. HMDB 3.0—The Human Metabolome Database in 2013. Nucleic Acids Res. 2013, 41, D801–D807. [Google Scholar] [CrossRef] [PubMed]
| ARIC African Ancestry | ARIC European Ancestry | KORA | |
|---|---|---|---|
| N | 2354 | 1529 | 859 |
| Female, n (%) | 1510 (64.1) | 826 (54.0) | 457 (53.2) |
| Age, years | 53.0 (5.7) | 54.6 (5.8) | 53.8 (4.4) |
| Weight, kg | 83.5 (17.0) | 77.3 (16.3) | 79.5 (17.0) |
| Height, cm | 167.9 (8.9) | 168.3 (9.6) | 169.2 (9.2) |
| Smoking status: Never, n (%) | 1172 (49.8) | 610 (39.9) | 319 (37.1) |
| Smoking status: Former, n (%) | 523 (22.2) | 527 (34.5) | 360 (41.9) |
| Smoking status: Current, n (%) | 659 (28.0) | 392 (25.6) | 180 (21.0) |
| Pack years, in ever smokers | 21.9 (20.5) | 29.2 (22.0) | 22.8 (22.5) |
| Cigarettes per day, in current smokers | 14.4 (9.6) | 22.5 (12.6) | 12.5 (9.3) |
| FEV1, mL | 2519 (651) | 2891 (777) | 3268 (790) |
| FVC, mL | 3286 (819) | 3931 (991) | 4220 (976) |
| % predicted FEV1 | 95.1 (17.1) | 92.9 (16.6) | 103.4 (16.1) |
| % predicted FVC | 99.0 (15.7) | 99.5 (14.5) | 105.3 (13.7) |
| FEV1/FVC, % | 76.8 (7.9) | 73.6 (7.7) | 77.5 (6.2) |
| eGFR, mL/min/1.73 m² | 104.0 (18.3) | 91.3 (14.5) | 86.8 (13.4) |
| COPD * cases, n (%) | 179 (7.6) | 179 (11.7) | 35 (4.1) |
| COPD severity classes *: Moderate | 152 (6.5) | 150 (9.8) | 33 (3.9) |
| COPD severity classes *: Severe | 25 (1.1) | 24 (1.6) | 2 (0.2) |
| COPD severity classes *: Very Severe | 2 (0.1) | 5 (0.3) | 0 (0) |
| Diabetes, n (%) | 413 (17.5) | 126 (8.2) | 36 (4.2) |
| Hypertension, n (%) | 1258 (53.4) | 469 (30.7) | 277 (32.3) |
| Meta-Analysis | ARIC African Ancestry (n = 2354) | ARIC European Ancestry (n = 1529) | KORA (n = 859) | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Metabolite | Super Pathway | Beta + | SE | P | Direction ** | Beta | SE | P | Beta | SE | P | Beta | SE | P |
| glycine | Amino Acid | 38.8 | 7.0 | 3.6 × 10−8 | +++ | 39.1 | 9.4 | 3.7 × 10−5 | 39.0 | 13.1 | 3.0 × 10−3 | 37.8 | 18.0 | 3.6 × 10−2 |
| 3-(4-hydroxyphenyl)lactate | Amino Acid | −37.2 | 7.7 | 1.4 × 10−6 | −−− | −32.9 | 10.0 | 9.9 × 10−4 | −26.9 | 15.2 | 7.6 × 10−2 | −73.5 | 20.3 | 3.1 × 10−4 |
| 3-phenylpropionate (hydrocinnamate) | Amino Acid | 32.7 | 6.8 | 1.5 × 10−6 | +++ | 20.9 | 9.5 | 2.8 × 10−2 | 42.0 | 11.9 | 4.2 × 10−4 | 51.2 | 16.9 | 2.5 × 10−3 |
| 2-methylbutyrylcarnitine (C5) | Amino Acid | −36.2 | 7.5 | 1.6 × 10−6 | −−− | −41.2 | 9.8 | 2.8 × 10−5 | −38.0 | 15.0 | 1.2 × 10−2 | −14.7 | 18.9 | 4.4 × 10−1 |
| asparagine | Amino Acid | 31.1 | 6.8 | 5.2 × 10−6 | ++− | 29.8 | 9.2 | 1.2 × 10−3 | 54.0 | 12.7 | 2.2 × 10−5 | −5.4 | 16.9 | 7.5 × 10−1 |
| alpha-hydroxyisovalerate | Amino Acid | −30.9 | 7.0 | 8.9 × 10−6 | −−− | −24.4 | 9.0 | 6.8 × 10−3 | −32.2 | 13.9 | 2.1 × 10−2 | −53.7 | 17.7 | 2.5 × 10−3 |
| glutamine | Amino Acid | 29.0 | 7.0 | 3.6 × 10−5 | +++ | 32.5 | 9.6 | 7.3 × 10−4 | 35.2 | 12.9 | 6.7 × 10−3 | 7.4 | 17.0 | 6.6 × 10−1 |
| isoleucine | Amino Acid | −28.9 | 7.4 | 1.0 × 10−4 | −−− | −28.9 | 9.4 | 2.2 × 10−3 | −34.8 | 14.5 | 1.7 × 10−2 | −15.3 | 21.8 | 4.8 × 10−1 |
| serotonin (5HT) | Amino Acid | 26.2 | 6.9 | 1.4 × 10−4 | +++ | 33.2 | 9.0 | 2.4 × 10−4 | 14.0 | 13.4 | 3.0 × 10−1 | 20.5 | 17.2 | 2.3 × 10−1 |
| glycerate | Carbohydrate | 31.7 | 7.0 | 5.8 × 10−6 | ++− | 39.2 | 9.3 | 2.8 × 10−5 | 43.8 | 13.5 | 1.2 × 10−3 | −11.0 | 16.7 | 5.1 × 10−1 |
| lactate | Carbohydrate | −30.4 | 6.9 | 1.1 × 10−5 | −−− | −27.7 | 9.3 | 2.9 × 10−3 | −36.4 | 12.7 | 4.1 × 10−3 | −28.4 | 17.7 | 1.1 × 10−1 |
| fructose | Carbohydrate | −27.6 | 6.7 | 3.8 × 10−5 | −−− | −28.6 | 8.5 | 8.2 × 10−4 | −39.2 | 14.3 | 6.3 × 10−3 | −8.2 | 16.7 | 6.2 × 10−1 |
| mannose | Carbohydrate | −27.4 | 7.0 | 9.9 × 10−5 | −−− | −33.1 | 8.9 | 2.0 × 10−4 | −21.2 | 14.9 | 1.5 × 10−1 | −12.5 | 18.1 | 4.9 × 10−1 |
| glycerol | Lipid | −36.4 | 7.2 | 4.7 × 10−7 | −−− | −36.5 | 9.5 | 1.3 × 10−4 | −43.0 | 14.0 | 2.1 × 10−3 | −24.8 | 18.2 | 1.7 × 10−1 |
| 7-alpha-hydroxy-3-oxo-4-cholestenoate (7-Hoca) | Lipid | −30.4 | 6.9 | 9.5 × 10−6 | −−− | −18.6 | 9.4 | 4.8 × 10−2 | −33.0 | 12.5 | 8.2 × 10−1 | −63.7 | 16.9 | 1.8 × 10−4 |
| N2,N2-dimethylguanosine | Nucleotide | −32.3 | 7.2 | 6.4 × 10−6 | −−− | −22.6 | 9.7 | 2.0 × 10−2 | −58.5 | 13.6 | 1.9 × 10−5 | −21.6 | 16.9 | 2.0 × 10−1 |
| pseudouridine | Nucleotide | −33.8 | 7.6 | 8.9 × 10−6 | −−− | −29.9 | 10.1 | 3.1 × 10−3 | −21.8 | 14.9 | 1.4 × 10−1 | −65.3 | 18.4 | 4.1 × 10−4 |
| gamma-glutamylthreonine | Peptide | 36.3 | 6.8 | 1.0 × 10−7 | +++ | 34.2 | 8.8 | 1.1 × 10−4 | 52.6 | 13.9 | 1.6 × 10−4 | 20.0 | 16.8 | 2.4 × 10−1 |
| gamma-glutamylleucine | Peptide | 34.8 | 7.0 | 5.6 × 10−7 | +++ | 34.4 | 8.8 | 9.6 × 10−5 | 40.6 | 13.6 | 2.9 × 10−3 | 23.7 | 20.5 | 2.5 × 10−1 |
| gamma-glutamylvaline | Peptide | 31.8 | 6.9 | 4.1 × 10−6 | +++ | 30.9 | 8.8 | 4.8 × 10−4 | 43.2 | 13.4 | 1.3 × 10−3 | 11.6 | 19.9 | 5.6 × 10−1 |
| Meta-Analysis | ARIC African-Ancestry (n = 2354) | ARIC European Ancestry (n = 1529) | KORA (n = 859) | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Metabolite | Super Pathway | Beta + | SE | P | Direction ** | Beta | SE | P | Beta | SE | P | Beta | SE | P |
| glycine | Amino Acid | 56.9 | 8.0 | 9.6 × 10−13 | +++ | 58.4 | 10.9 | 8.9 × 10−8 | 61.2 | 14.7 | 3.4 × 10−5 | 44.6 | 19.3 | 2.1 × 10−2 |
| isoleucine | Amino Acid | −53.8 | 8.4 | 1.7 × 10−10 | −−− | −47.0 | 10.9 | 1.6 × 10−5 | −69.7 | 16.3 | 2.0 × 10−5 | −52.0 | 23.2 | 2.5 × 10−2 |
| N-acetylglycine | Amino Acid | 44.3 | 7.9 | 2.4 × 10−8 | +++ | 43.8 | 11.1 | 8.5 × 10−5 | 49.8 | 14.3 | 5.0 × 10−4 | 36.4 | 18.8 | 5.3 × 10−2 |
| asparagine | Amino Acid | 41.6 | 7.7 | 7.0 × 10−8 | +++ | 42.7 | 10.6 | 6.0 × 10−5 | 62.6 | 14.3 | 1.3 × 10−5 | 4.4 | 18.1 | 8.1 × 10−1 |
| 2-methylbutyrylcarnitine (C5) | Amino Acid | −44.9 | 8.5 | 1.5 × 10−7 | −−− | −44.5 | 11.3 | 9.0 × 10−5 | −55.9 | 16.9 | 9.7 × 10−4 | −30.2 | 20.2 | 1.4 × 10−1 |
| glutamine | Amino Acid | 39.0 | 7.9 | 9.2 × 10−7 | +++ | 37.2 | 11.1 | 8.0 × 10−4 | 56.3 | 14.6 | 1.1 × 10−4 | 16.5 | 18.2 | 3.7 × 10−1 |
| 3-(4-hydroxyphenyl)lactate | Amino Acid | −42.6 | 8.7 | 1.1 × 10−6 | −−− | −35.2 | 11.5 | 2.3 × 10−3 | −32.9 | 17.1 | 5.4 × 10−2 | −84.3 | 21.7 | 1.1 × 10−4 |
| 3-phenylpropionate (hydrocinnamate) | Amino Acid | 36.3 | 7.7 | 2.3 × 10−6 | +++ | 24.1 | 11.0 | 2.8 × 10−2 | 42.0 | 13.4 | 1.8 × 10−3 | 59.2 | 18.1 | 1.1 × 10−3 |
| tyrosine | Amino Acid | −35.2 | 8.0 | 1.2 × 10−5 | −−− | −24.5 | 10.5 | 2.0 × 10−2 | −42.8 | 16.2 | 8.2 × 10−3 | −61.6 | 19.6 | 1.8 × 10−3 |
| valine | Amino Acid | −33.9 | 8.1 | 3.1 × 10−5 | −−− | −36.4 | 10.5 | 5.3 × 10−4 | −33.7 | 16.3 | 3.9 × 10−2 | −24.2 | 21.2 | 2.5 × 10−1 |
| phenylalanine | Amino Acid | −33.2 | 8.0 | 3.3 × 10−5 | −−− | −26.2 | 10.4 | 1.2 × 10−2 | −37.3 | 16.2 | 2.2 × 10−2 | −52.3 | 19.7 | 8.2 × 10−3 |
| 5-oxoproline | Amino Acid | 31.4 | 7.8 | 5.9 × 10−5 | ++− | 42.8 | 10.6 | 5.7 × 10−5 | 44.7 | 14.9 | 2.7 × 10−3 | −22.4 | 18.2 | 2.2 × 10−1 |
| mannose | Carbohydrate | −41.1 | 8.0 | 2.5 × 10−7 | −−− | −52.1 | 10.2 | 3.8 × 10−7 | −39.4 | 16.8 | 1.9 × 10−2 | −3.6 | 19.4 | 8.5 × 10−1 |
| lactate | Carbohydrate | −38.4 | 7.8 | 8.9 × 10−7 | −−− | −33.6 | 10.7 | 1.8 × 10−3 | −45.8 | 14.3 | 1.4 × 10−3 | −40.3 | 18.9 | 3.3 × 10−2 |
| fructose | Carbohydrate | −32.3 | 7.6 | 2.2 × 10−5 | −−− | −39.5 | 9.8 | 6.2 × 10−5 | −31.9 | 16.2 | 4.9 × 10−2 | −9.0 | 17.9 | 6.2 × 10−1 |
| glucose | Carbohydrate | −30.7 | 7.7 | 6.3 × 10−5 | −−− | −37.1 | 9.5 | 9.7 × 10−5 | −21.0 | 18.0 | 2.4 × 10−1 | −16.1 | 18.7 | 3.9 × 10−1 |
| biliverdin | Cofactors and Vitamins | 31.0 | 7.9 | 8.6 × 10−5 | +++ | 31.0 | 11.5 | 7.1 × 10−3 | 28.0 | 13.5 | 3.8 × 10−2 | 36.2 | 18.1 | 4.6 × 10−2 |
| glycerol | Lipid | −33.4 | 8.2 | 4.6 × 10−5 | −−− | −39.1 | 11.0 | 4.0 × 10−4 | −36.1 | 15.7 | 2.2 × 10−2 | −11.4 | 19.5 | 5.6 × 10−1 |
| pseudouridine | Nucleotide | −38.5 | 8.6 | 7.8 × 10−6 | −−− | −35.4 | 11.7 | 2.4 × 10−3 | −29.6 | 16.7 | 7.7 × 10−2 | −59.8 | 19.8 | 2.6 × 10−3 |
| urate | Nucleotide | −35.3 | 8.3 | 2.1× 10−5 | −−− | −38.2 | 11.1 | 5.9 × 10−4 | −26.2 | 14.8 | 7.7 × 10−2 | −45.3 | 23.5 | 5.4 × 10−2 |
| Meta-Analysis | ARIC African Ancestry (n = 2354) | ARIC European Ancestry (n = 1529) | KORA (n = 859) | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Metabolite | Super Pathway | Beta + | SE | P | Direction ** | Beta | SE | P | Beta | SE | P | Beta | SE | P |
| 3-methoxytyrosine | Amino Acid | −0.42 | 0.10 | 5.0 × 10−5 | −−− | −0.37 | 0.13 | 4.1 × 10−3 | −0.45 | 0.35 | 2.0 × 10−1 | −0.53 | 0.20 | 8.1 × 10−3 |
| androsterone sulfate | Lipid | 0.37 | 0.10 | 2.5 × 10−4 | +++ | 0.29 | 0.16 | 7.9 × 10−2 | 0.31 | 0.17 | 7.0 × 10−2 | 0.57 | 0.20 | 5.6 × 10−3 |
| glycerol | Lipid | −0.36 | 0.11 | 8.6 × 10−4 | −−− | −0.27 | 0.16 | 9.1 × 10−2 | −0.52 | 0.20 | 7.4 × 10−3 | −0.32 | 0.22 | 1.4 × 10−1 |
| dehydroisoandrosterone sulfate (DHEA-S) | Lipid | 0.35 | 0.11 | 1.2 × 10−3 | +++ | 0.39 | 0.17 | 2.1 × 10−2 | 0.37 | 0.19 | 5.1 × 10−2 | 0.27 | 0.22 | 2.2 × 10−1 |
| lathosterol | Lipid | 0.34 | 0.10 | 7.4 × 10−4 | +++ | 0.38 | 0.15 | 1.3 × 10−2 | 0.37 | 0.18 | 4.1 × 10−2 | 0.24 | 0.20 | 2.5 × 10−1 |
| oleoylcarnitine | Lipid | −0.42 | 0.10 | 2.9 × 10−5 | −−− | −0.46 | 0.15 | 2.1 × 10−3 | −0.50 | 0.18 | 4.8 × 10−3 | −0.23 | 0.20 | 2.7 × 10−1 |
| 7-alpha-hydroxy-3-oxo-4-cholestenoate (7-Hoca) | Lipid | −0.33 | 0.10 | 9.6 × 10−4 | −−− | −0.32 | 0.16 | 3.8 × 10−2 | −0.28 | 0.17 | 1.1 × 10−1 | −0.42 | 0.21 | 4.0 × 10−2 |
| theophylline | Xenobiotics | −0.62 | 0.10 | 4.0 × 10−10 | −−− | −0.72 | 0.14 | 4.8 × 10−7 | −0.73 | 0.18 | 7.5 × 10−5 | −0.26 | 0.21 | 2.1 × 10−1 |
| Trait | Pathway | Analytes in the Pathway, N | Analytes with FDR < 0.05, N | Analytes with FDR < 0.05 | P | FDR |
|---|---|---|---|---|---|---|
| FEV1 | Aminoacyl-tRNA biosynthesis | 75 | 8 | asparagine, phenylalanine, glutamine, cysteine, glycine, isoleucine, threonine, tyrosine | 3.3 × 10−4 | 0.02 |
| FEV1 | Phenylalanine metabolism | 45 | 6 | phenylalanine, hydrocinnamic acid, hippuric acid, succinic acid, n-acetyl-phenylalanine, tyrosine | 5.9 × 10−4 | 0.02 |
| FEV1 | Nitrogen metabolism | 39 | 5 | phenylalanine, tyrosine, asparagine, glutamine, glycine | 2.1× 10−3 | 0.05 |
| FEV1 | Alanine, aspartate and glutamate metabolism | 24 | 4 | asparagine, oxoglutaric acid, glutamine, succinic acid | 2.3 × 10−3 | 0.05 |
| FVC | Aminoacyl-tRNA biosynthesis | 75 | 9 | asparagine, phenylalanine, glutamine, glycine, valine, isoleucine, leucine, threonine, tyrosine. | 7.0 × 10−5 | 0.006 |
| FVC | Phenylalanine metabolism | 45 | 6 | phenylalanine, hydrocinnamic acid, hippuric acid, succinic acid, n-acetyl-phenylalanine, tyrosine | 7.1 × 10−4 | 0.03 |
| Meta-analysis | ARIC African Ancestry (n = 2354) | ARIC European Ancestry (n = 1529) | KORA (n = 859) | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Metabolite | Super Pathway | OR + (95% CI) | P | Direction ** | OR (95% CI) | P | OR (95% CI) | P | OR (95% CI) | P |
| 3-(4-hydroxyphenyl)lactate | Amino Acid | 1.28 (1.14, 1.44) | 3.6 × 10−5 | +++ | 1.28 (1.09, 1.51) | 3.2 × 10−3 | 1.13 (0.93, 1.38) | 2.3 × 10−1 | 1.76 (1.28, 2.42) | 4.6 × 10−4 |
| 3-methoxytyrosine | Amino Acid | 1.19 (1.1, 1.3) | 4.1 × 10−5 | +++ | 1.19 (1.08, 1.3) | 2.5 × 10−4 | 1.11 (0.79, 1.55) | 5.4 × 10−1 | 1.36 (1, 1.85) | 4.6 × 10−2 |
| homocitrulline | Amino Acid | 1.21 (1.08, 1.36) | 8.5 × 10−4 | ++− | 1.26 (1.09, 1.47) | 2.5 × 10−3 | 1.21 (1, 1.47) | 5.3 × 10−2 | 0.95 (0.66, 1.39) | 8.1 × 10−1 |
| serotonin (5HT) | Amino Acid | 0.83 (0.74, 0.93) | 1.7 × 10−3 | −−− | 0.8 (0.67, 0.94) | 7.7 × 10−3 | 0.87 (0.72, 1.05) | 1.4 × 10−1 | 0.82 (0.57, 1.19) | 3.0 × 10−1 |
| ornithine | Amino Acid | 1.2 (1.07, 1.34) | 1.9 × 10−3 | +++ | 1.06 (0.9, 1.24) | 5.0 × 10−1 | 1.4 (1.17, 1.68) | 2.7 × 10−4 | 1.21 (0.85, 1.73) | 2.8 × 10−1 |
| glycerate | Carbohydrate | 0.8 (0.71, 0.91) | 3.3 × 10−4 | −−+ | 0.77 (0.65, 0.92) | 3.9 × 10−3 | 0.77 (0.64, 0.92) | 5.2 × 10−3 | 1.12 (0.78, 1.61) | 5.4 × 10−1 |
| succinylcarnitine | Energy | 1.21 (1.08, 1.36) | 1.4 × 10−3 | +++ | 1.17 (1, 1.37) | 5.3 × 10−2 | 1.32 (1.08, 1.6) | 6.3 × 10−3 | 1.09 (0.74, 1.61) | 6.7 × 10−1 |
| oleoylcarnitine | Lipid | 1.22 (1.1, 1.35) | 1.6 × 10−4 | +++ | 1.28 (1.11, 1.48) | 6.1 × 10−4 | 1.14 (0.96, 1.35) | 1.3 × 10−1 | 1.2 (0.89, 1.62) | 2.3 × 10−1 |
| 5-dodecenoate (12:1n7) | Lipid | 1.23 (1.1, 1.37) | 1.9 × 10−4 | +++ | 1.27 (1.09, 1.48) | 1.7 × 10−3 | 1.14 (0.96, 1.36) | 1.5 × 10−1 | 1.33 (0.97, 1.81) | 7.7 × 10−2 |
| docosahexaenoate (DHA, 22:6n3) | Lipid | 0.79 (0.7, 0.9) | 2.6 × 10−4 | −−+ | 0.85 (0.72, 1) | 4.6 × 10−2 | 0.63 (0.51, 0.78) | 3.3 × 10−5 | 1.11 (0.78, 1.6) | 5.5 × 10−1 |
| androsterone sulfate | Lipid | 0.81 (0.71, 0.91) | 8.2 × 10−4 | −−− | 0.9 (0.75, 1.08) | 2.4 × 10−1 | 0.79 (0.66, 0.95) | 1.4 × 10−2 | 0.36 (0.21, 0.63) | 2.8 × 10−4 |
| 7-alpha-hydroxy-3-oxo-4-cholestenoate (7-Hoca) | Lipid | 1.19 (1.07, 1.33) | 1.0 × 10−3 | +++ | 1.15 (0.98, 1.35) | 9.3 × 10−2 | 1.22 (1.04, 1.43) | 1.3 × 10−2 | 1.29 (0.94, 1.76) | 1.2 × 10−1 |
| glycerol | Lipid | 1.21 (1.08, 1.36) | 1.0 × 10−3 | +++ | 1.21 (1.02, 1.42) | 2.4 × 10−2 | 1.2 (0.99, 1.45) | 6.4 × 10−2 | 1.28 (0.94, 1.75) | 1.2 × 10−1 |
| pseudouridine | Nucleotide | 1.23 (1.08, 1.39) | 1.2 × 10−3 | +++ | 1.23 (1.04, 1.44) | 1.5 × 10−2 | 1.22 (0.98, 1.53) | 7.7 × 10−2 | 1.27 (0.86, 1.86) | 2.3 × 10−1 |
| theophylline | Xenobiotics | 1.26 (1.17, 1.36) | 1.3 × 10−9 | +++ | 1.25 (1.14, 1.37) | 3.1 × 10−6 | 1.25 (1.09, 1.45) | 2.0 × 10−3 | 1.39 (1.07, 1.81) | 1.4 × 10−2 |
| 1-methylurate | Xenobiotics | 1.21 (1.1, 1.33) | 1.3 × 10−4 | +++ | 1.22 (1.07, 1.4) | 3.7 × 10−3 | 1.23 (1.06, 1.44) | 7.6 × 10−3 | 1.04 (0.75, 1.43) | 8.3 × 10−1 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yu, B.; Flexeder, C.; McGarrah, R.W., III; Wyss, A.; Morrison, A.C.; North, K.E.; Boerwinkle, E.; Kastenmüller, G.; Gieger, C.; Suhre, K.; et al. Metabolomics Identifies Novel Blood Biomarkers of Pulmonary Function and COPD in the General Population. Metabolites 2019, 9, 61. https://doi.org/10.3390/metabo9040061
Yu B, Flexeder C, McGarrah RW III, Wyss A, Morrison AC, North KE, Boerwinkle E, Kastenmüller G, Gieger C, Suhre K, et al. Metabolomics Identifies Novel Blood Biomarkers of Pulmonary Function and COPD in the General Population. Metabolites. 2019; 9(4):61. https://doi.org/10.3390/metabo9040061
Chicago/Turabian StyleYu, Bing, Claudia Flexeder, Robert W. McGarrah, III, Annah Wyss, Alanna C. Morrison, Kari E. North, Eric Boerwinkle, Gabi Kastenmüller, Christian Gieger, Karsten Suhre, and et al. 2019. "Metabolomics Identifies Novel Blood Biomarkers of Pulmonary Function and COPD in the General Population" Metabolites 9, no. 4: 61. https://doi.org/10.3390/metabo9040061
APA StyleYu, B., Flexeder, C., McGarrah, R. W., III, Wyss, A., Morrison, A. C., North, K. E., Boerwinkle, E., Kastenmüller, G., Gieger, C., Suhre, K., Karrasch, S., Peters, A., Wagner, G. R., Michelotti, G. A., Mohney, R. P., Schulz, H., & London, S. J. (2019). Metabolomics Identifies Novel Blood Biomarkers of Pulmonary Function and COPD in the General Population. Metabolites, 9(4), 61. https://doi.org/10.3390/metabo9040061






