Prospective Biomarkers from Plasma Metabolomics of Myalgic Encephalomyelitis/Chronic Fatigue Syndrome Implicate Redox Imbalance in Disease Symptomatology
Abstract
1. Introduction
2. Results
2.1. Cohort Statistics
2.2. Metabolite Data and Their Statistical Handling
2.3. The Metabolites Found to be Most Affected in ME/CFS Patients Belong to Four Classes
2.4. The MetaboAnalyst Statistical Analysis Unit Provides Additional Valuable Insight into the Metabolon® Dataset
2.5. The MetaboAnalyst Enrichment Analysis Unit Returns Compelling Disease-Associated Metabolite Sets in Plasma
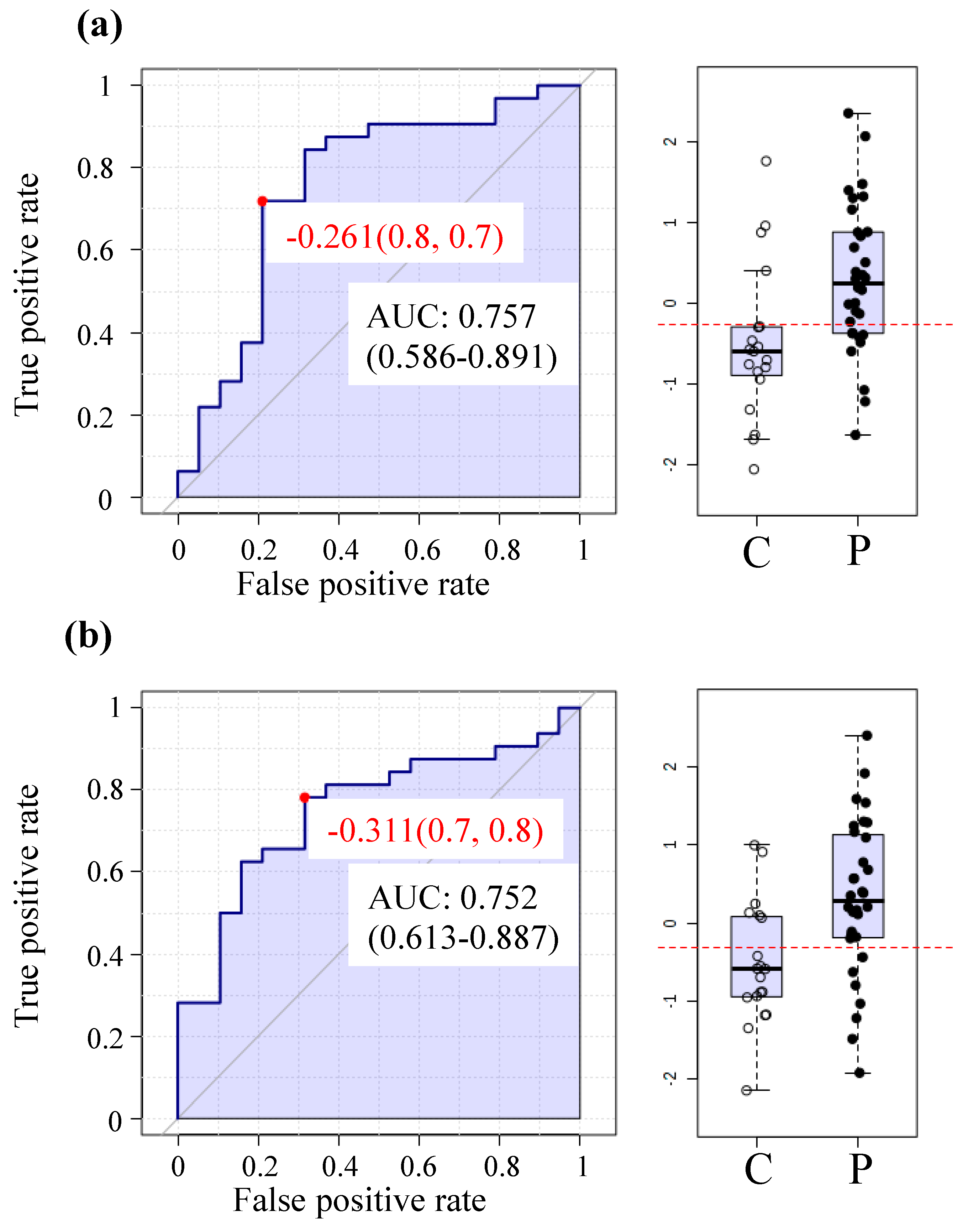
2.6. The MetaboAnalyst Biomarker Analysis Unit Yields Prospective Biomarkers for ME/CFS Using Plasma
2.7. Analysis Indicates that Close to 100% of the Cross-Identified Metabolites Exhibit Similar Statistical Behavior between Studies
2.8. Hierarchical Clustering Does not Yield any Definitive Subgroup Identification
3. Discussion
4. Materials and Methods
4.1. Sampling and Metabolomics Platform
4.2. Data Analysis through MetaboAnalyst
4.3. Data Analysis in-House
4.4. Data Availability
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Borody, T.; Nowak, A.; Finlayson, S. The GI microbiome and its role in chronic fatigue syndrome: A summary of bacteriotherapy. J. Nutr. Envron. Med. 2012, 31, 3–8. [Google Scholar]
- Fremont, M.; Coomans, D.; Massart, S.; De Meirleir, K. High-throughput 16S rRNA gene sequencing reveals alterations of intestinal microbiota in myalgic encephalomyelitis/chronic fatigue syndrome patients. Anaerobe 2013, 22, 50–56. [Google Scholar] [CrossRef] [PubMed]
- Shukla, S.K.; Cook, D.; Meyer, J.; Vernon, S.D.; Le, T.; Clevidence, D.; Robertson, C.E.; Schrodi, S.J.; Yale, S.; Frank, D.N. Changes in gut and plasma microbiome following exercise challenge in myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS). PLoS ONE 2015, 10, e0145453. [Google Scholar] [CrossRef]
- Giloteaux, L.; Goodrich, J.K.; Walters, W.A.; Levine, S.M.; Ley, R.E.; Hanson, M.R. Reduced diversity and altered composition of the gut microbiome in individuals with myalgic encephalomyelitis/chronic fatigue syndrome. Microbiome 2016, 4, 30. [Google Scholar] [CrossRef] [PubMed]
- Giloteaux, L.; Hanson, M.R.; Keller, B.A. A pair of identical twins discordant for myalgic encephalomyelitis/chronic fatigue syndrome differ in physiological parameters and gut microbiome composition. Am. J. Case Rep. 2016, 17, 720–729. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, C.W.; McGregor, N.R.; Lewis, D.P.; Butt, H.L.; Gooley, P.R. The association of fecal microbiota and fecal, blood serum and urine metabolites in myalgic encephalomyelitis/chronic fatigue syndrome. Metabolomics 2017, 13, 8. [Google Scholar] [CrossRef]
- Hanson, M.R.; Giloteaux, L. The gut microbiome in myalgic encephalomyelitis. Available online: http://www.biochemist.org/bio/03902/0010/039020010.pdf (accessed on 11 April 2017).
- Nagy-Szakal, D.; Williams, B.L.; Mishra, N.; Che, X.; Lee, B.; Bateman, L.; Klimas, N.G.; Komaroff, A.L.; Levine, S.; Montoya, J.G.; et al. Fecal metagenomic profiles in subgroups of patients with myalgic encephalomyelitis/chronic fatigue syndrome. Microbiome 2017, 5, 44. [Google Scholar] [CrossRef] [PubMed]
- Brenu, E.W.; van Driel, M.L.; Staines, D.R.; Ashton, K.J.; Ramos, S.B.; Keane, J.; Klimas, N.G.; Marshall-Gradisnik, S.M. Immunological abnormalities as potential biomarkers in chronic fatigue syndrome/myalgic encephalomyelitis. J. Transl. Med. 2011, 9, 81. [Google Scholar] [CrossRef] [PubMed]
- Morris, G.; Berk, M.; Galecki, P.; Maes, M. The emerging role of autoimmunity in myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS). Mol. Neurobiol. 2014, 49, 741–756. [Google Scholar] [CrossRef]
- Nijs, J.; Nees, A.; Paul, L.; De Kooning, M.; Ickmans, K.; Meeus, M.; Van Oosterwijck, J. Altered immune response to exercise in patients with chronic fatigue syndrome/myalgic encephalomyelitis: A systematic literature review. Exerc. Immunol. Rev. 2014, 20, 94–116. [Google Scholar]
- Hornig, M.; Montoya, J.G.; Klimas, N.G.; Levine, S.; Felsenstein, D.; Bateman, L.; Peterson, D.L.; Gottschalk, C.G.; Schultz, A.F.; Che, X.; et al. Distinct plasma immune signatures in ME/CFS are present early in the course of illness. Sci. Adv. 2015, 1, 1. [Google Scholar] [CrossRef] [PubMed]
- Boissoneault, J.; Letzen, J.; Lai, S.; O’Shea, A.; Craggs, J.; Robinson, M.E.; Staud, R. Abnormal resting state functional connectivity in patients with chronic fatigue syndrome: An arterial spin-labeling fMRI study. Magn. Reson. Imaging 2016, 34, 603–608. [Google Scholar] [CrossRef] [PubMed]
- Gay, C.W.; Robinson, M.E.; Lai, S.; O’Shea, A.; Craggs, J.G.; Price, D.D.; Staud, R. Abnormal resting-state functional connectivity in patients with chronic fatigue syndrome: Results of seed and data-driven analyses. Brain Connect 2016, 6, 48–56. [Google Scholar] [CrossRef] [PubMed]
- Keller, B.A.; Pryor, J.L.; Giloteaux, L. Inability of myalgic encephalomyelitis/chronic fatigue syndrome patients to reproduce VO(2)peak indicates functional impairment. J. Trans. Med. 2014, 12, 104. [Google Scholar] [CrossRef]
- Armstrong, C.W.; McGregor, N.R.; Sheedy, J.R.; Buttfield, I.; Butt, H.L.; Gooley, P.R. NMR metabolic profiling of serum identifies amino acid disturbances in chronic fatigue syndrome. Clin. Chim. Acta 2012, 413, 1525–1531. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, C.W.; McGregor, N.R.; Lewis, D.P.; Butt, H.L.; Gooley, P.R. Metabolic profiling reveals anomalous energy metabolism and oxidative stress pathways in chronic fatigue syndrome patients. Metabolomics 2015, 11, 1626–1639. [Google Scholar] [CrossRef]
- Fluge, O.; Mella, O.; Bruland, O.; Risa, K.; Dyrstad, S.E.; Alme, K.; Rekeland, I.G.; Sapkota, D.; Rosland, G.V.; Fossa, A.; et al. Metabolic profiling indicates impaired pyruvate dehydrogenase function in myalgic encephalopathy/chronic fatigue syndrome. JCI Insight 2016, 1, e89376. [Google Scholar] [CrossRef]
- Naviaux, R.K.; Naviaux, J.C.; Li, K.F.; Bright, A.T.; Alaynick, W.A.; Wang, L.; Baxter, A.; Nathan, N.; Anderson, W.; Gordon, E. Metabolic features of chronic fatigue syndrome. Proc. Nat. Acad. Sci. USA 2016, 113, E5472–E5480. [Google Scholar] [CrossRef]
- Yamano, E.; Sugimoto, M.; Hirayama, A.; Kume, S.; Yamato, M.; Jin, G.H.; Tajima, S.; Goda, N.; Iwai, K.; Fukuda, S.; et al. Index markers of chronic fatigue syndrome with dysfunction of TCA and urea cycles. Sci. Rep. 2016, 6, 34990. [Google Scholar] [CrossRef]
- Germain, A.; Ruppert, D.; Levine, S.M.; Hanson, M.R. Metabolic profiling of a myalgic encephalomyelitis/chronic fatigue syndrome discovery cohort reveals disturbances in fatty acid and lipid metabolism. Mol. Biosyst. 2017, 13, 371–379. [Google Scholar] [CrossRef]
- Nagy-Szakal, D.; Barupal, D.K.; Lee, B.; Che, X.; Williams, B.L.; Kahn, E.J.R.; Ukaigwe, J.E.; Bateman, L.; Klimas, N.G.; Komaroff, A.L.; et al. Insights into myalgic encephalomyelitis/chronic fatigue syndrome phenotypes through comprehensive metabolomics. Sci. Rep. 2018, 8, 10056. [Google Scholar] [CrossRef] [PubMed]
- Navaneetharaja, N.; Griffiths, V.; Wileman, T.; Carding, S.R. A role for the intestinal microbiota and virome in myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS)? J. Clin. Med. 2016, 5, 55. [Google Scholar] [CrossRef] [PubMed]
- Gerwyn, M.; Maes, M. Mechanisms explaining muscle fatigue and muscle pain in patients with myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS): A review of recent findings. Curr. Rheumatol. Rep. 2017, 19, 1. [Google Scholar] [CrossRef] [PubMed]
- Richards, R.S.; Roberts, T.K.; McGregor, N.; Dunstan, R.H.; Butt, H.L. Blood parameters indicative of oxidative stress are associated with symptom expression in chronic fatigue syndrome. Redox. Rep. 2000, 5, 35–41. [Google Scholar] [CrossRef] [PubMed]
- Maes, M.; Kubera, M.; Uytterhoeven, M.; Vrydags, N.; Bosmans, E. Increased plasma peroxides as a marker of oxidative stress in myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS). Med. Sci. Monitor. 2011, 17, Sc11–Sc15. [Google Scholar] [CrossRef]
- Arnett, S.V.; Clark, I.A. Inflammatory fatigue and sickness behaviour—lessons for the diagnosis and management of chronic fatigue syndrome. J. Affect Disord. 2012, 141, 130–142. [Google Scholar] [CrossRef]
- Jammes, Y.; Steinberg, J.G.; Delliaux, S. Chronic fatigue syndrome: acute infection and history of physical activity affect resting levels and response to exercise of plasma oxidant/antioxidant status and heat shock proteins. J. Intern. Med. 2012, 272, 74–84. [Google Scholar] [CrossRef]
- Morris, G.; Maes, M. Oxidative and nitrosative stress and immune-inflammatory pathways in patients with myalgic encephalomyelitis (ME)/chronic fatigue syndrome (CFS). Curr. Neuropharmacol. 2014, 12, 168–185. [Google Scholar] [CrossRef]
- Fenouillet, E.; Vigouroux, A.; Steinberg, J.G.; Chagvardieff, A.; Retornaz, F.; Guieu, R.; Jammes, Y. Association of biomarkers with health-related quality of life and history of stressors in myalgic encephalomyelitis/chronic fatigue syndrome patients. J. Transl. Med. 2016, 14, 251. [Google Scholar] [CrossRef]
- Fukuda, K.; Straus, S.E.; Hickie, I.; Sharpe, M.C.; Dobbins, J.G.; Komaroff, A. The chronic fatigue syndrome: A comprehensive approach to its definition and study. International Chronic Fatigue Syndrome Study Group. Ann. Intern. Med. 1994, 121, 953–959. [Google Scholar] [CrossRef]
- Benjamini, Y.; Hochberg, Y. Controlling the false discovery rate—a practical and powerful approach to multiple testing. J. Roy. Stat. Soc. B. Met. 1995, 57, 289–300. [Google Scholar]
- Schaer, D.J.; Buehler, P.W.; Alayash, A.I.; Belcher, J.D.; Vercellotti, G.M. Hemolysis and free hemoglobin revisited: exploring hemoglobin and hemin scavengers as a novel class of therapeutic proteins. Blood 2013, 121, 1276–1284. [Google Scholar] [CrossRef] [PubMed]
- Chiabrando, D.; Vinchi, F.; Fiorito, V.; Mercurio, S.; Tolosano, E. Heme in pathophysiology: A matter of scavenging, metabolism and trafficking across cell membranes. Front. Pharmacol. 2014, 5. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, M.O.S.; Rocha, L.C.; Reis, J.H.O.; Santos, T.D.; do Nascimento, V.M.L.; Carvalho, M.B.; Luz, N.F.; Borges, V.D.; Goncalves, M.S. Heme concentration as a biomarker of sickle cell disease severity: Its role in steady-state and in crisis patients. Blood 2015, 126, 975. [Google Scholar]
- Milburn, M.; Guo, L.; Wulff, J.E.; Lawton, K.A. Determining liver toxicity of an agent using metabolite biomarkers. U.S. Patent 8,658,351, 25 February 2014. [Google Scholar]
- Biswal, B.; Kunwar, P.; Natelson, B.H. Cerebral blood flow is reduced in chronic fatigue syndrome as assessed by arterial spin labeling. J. Neurol. Sci. 2011, 301, 9–11. [Google Scholar] [CrossRef] [PubMed]
- Staud, R.; Boissoneault, J.; Craggs, J.; Lai, S.; Robinson, M.E. Task related cerebral blood flow changes of patients with chronic fatigue syndrome: an arterial spin labeling study. Fatigue 2018, 6, 63–79. [Google Scholar] [CrossRef]
- Ninkovic, D.; Sarnavka, V.; Basnec, A.; Cuk, M.; Ramadza, D.P.; Fumic, K.; Kusec, V.; Santer, R.; Baric, I. Hyperinsulinism-hyperammonemia syndrome: A de novo mutation of the GLUD1 gene in twins and a review of the literature. J. Pediatr. Endocr. Met. 2016, 29, 1083–1088. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.S.; Aa, J.Y.; Wang, G.J.; Yan, B.; Zhang, Y.; Wang, X.W.; Zhao, C.Y.; Cao, B.; Shi, J.A.; Li, M.J.; et al. Differences in metabolite profile between blood plasma and serum. Anal. Biochem. 2010, 406, 105–112. [Google Scholar] [CrossRef]
- Maes, M.; Leunis, J.C.; Geffard, M.; Berk, M. Evidence for the existence of myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS) with and without abdominal discomfort (irritable bowel) syndrome. Neuroendocrinol. Lett. 2014, 35, 445–453. [Google Scholar]
- Guenther, S.; Loebel, M.; Mooslechner, A.A.; Knops, M.; Hanitsch, L.G.; Grabowski, P.; Wittke, K.; Meisel, C.; Unterwalder, N.; Volk, H.D.; et al. Frequent IgG subclass and mannose binding lectin deficiency in patients with chronic fatigue syndrome. Hum. Immunol. 2015, 76, 729–735. [Google Scholar] [CrossRef]
- Hornig, M.; Gottschalk, C.G.; Eddy, M.L.; Che, X.; Ukaigwe, J.E.; Peterson, D.L.; Lipkin, W.I. Immune network analysis of cerebrospinal fluid in myalgic encephalomyelitis/chronic fatigue syndrome with atypical and classical presentations. Transl. Psychiat. 2017, 7, e1080. [Google Scholar] [CrossRef] [PubMed]
- Scheibenbogen, C.; Loebel, M.; Freitag, H.; Krueger, A.; Bauer, S.; Antelmann, M.; Doehner, W.; Scherbakov, N.; Heidecke, H.; Reinke, P.; et al. Immunoadsorption to remove beta 2 adrenergic receptor antibodies in chronic fatigue syndrome CFS/ME. PLoS ONE 2018, 13, e0193672. [Google Scholar] [CrossRef] [PubMed]
- Comhaire, F. A novel nutriceutical treatment of myalgic encephalitis/chronic fatigue syndrome (ME/CFS): “What it is and what it is not”. Intern. Med. 2017, 7, 5. [Google Scholar] [CrossRef]
- Chen, W.Y.; Maghzal, G.J.; Ayer, A.; Suarna, C.; Dunn, L.L.; Stocker, R. Absence of the biliverdin reductase-a gene is associated with increased endogenous oxidative stress. Free Radical. Bio. Med. 2018, 115, 156–165. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.L.; Beecher, G.R.; Holden, J.M.; Haytowitz, D.B.; Gebhardt, S.E.; Prior, R.L. Lipophilic and hydrophilic antioxidant capacities of common foods in the United States. J. Agr. Food Chem. 2004, 52, 4026–4037. [Google Scholar] [CrossRef] [PubMed]
- Craig, C. Mitoprotective dietary approaches for myalgic encephalomyelitis/chronic fatigue syndrome: Caloric restriction, fasting, and ketogenic diets. Med. Hypotheses. 2015, 85, 690–693. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Rodriguez, M.M.; Molina, J.G.; Medina, I.M.F.; Sola, C.F.; Muelle, A.R. Patterns of food avoidance and eating behavior in women with fibromyalgia. Endocrinol. Diab. Nutr. 2017, 64, 480–490. [Google Scholar] [CrossRef]
- McCubrey, J.A.; Lertpiriyapong, K.; Steelman, L.S.; Abrams, S.L.; Yang, L.V.; Murata, R.M.; Rosalen, P.L.; Scalisi, A.; Neri, L.M.; Cocco, L.; et al. Effects of resveratrol, curcumin, berberine and other nutraceuticals on aging, cancer development, cancer stem cells and microRNAs. Aging 2017, 9, 1477–1536. [Google Scholar]
- Uhde, M.; Indart, A.C.; Yu, X.B.; Jang, S.S.; De Giorgio, R.; Green, P.H.R.; Volta, U.; Vernon, S.D.; Alaedini, A. Markers of non-coeliac wheat sensitivity in patients with myalgic encephalomyelitis/chronic fatigue syndrome. Gut 2018. In Press. [Google Scholar] [CrossRef]
- Campagnolo, N.; Johnston, S.; Collatz, A.; Staines, D.; Marshall-Gradisnik, S. Dietary and nutrition interventions for the therapeutic treatment of chronic fatigue syndrome/myalgic encephalomyelitis: A systematic review. J. Hum. Nutr. Diet. 2017, 30, 247–259. [Google Scholar] [CrossRef]
- Jones, K.; Probst, Y. Role of dietary modification in alleviating chronic fatigue syndrome symptoms: A systematic review. Nutrition 2017, 41, 338–344. [Google Scholar] [CrossRef] [PubMed]
- Fukao, T.; Sass, J.O.; Kursula, P.; Thimm, E.; Wendel, U.; Ficicioglu, C.; Monastiri, K.; Guffon, N.; Baric, I.; Zabot, M.T.; et al. Clinical and molecular characterization of five patients with succinyl-CoA:3-ketoacid CoA transferase (SCOT) deficiency. Bba-Mol. Basis Dis. 2011, 1812, 619–624. [Google Scholar] [CrossRef] [PubMed]
- Sigauke, E.; Rakheja, D.; Kitson, K.; Bennett, M.J. Carnitine palmitoyltransferase II deficiency: A clinical, biochemical, and molecular review. Lab. Invest. 2003, 83, 1543–1554. [Google Scholar] [CrossRef] [PubMed]
- Vermeulen, R.C.W.; Kurk, R.M.; Visser, F.C.; Sluiter, W.; Scholte, H.R. Patients with chronic fatigue syndrome performed worse than controls in a controlled repeated exercise study despite a normal oxidative phosphorylation capacity. J. Transl. Med. 2010, 8, 93. [Google Scholar] [CrossRef] [PubMed]
- Snell, C.R.; Stevens, S.R.; Davenport, T.E.; Van Ness, J.M. Discriminative validity of metabolic and workload measurements for identifying people with chronic fatigue syndrome. Phys. Ther. 2013, 93, 1484–1492. [Google Scholar] [CrossRef]
- Neary, J.P.; Roberts, A.D.W.; Leavins, N.; Harrison, M.F.; Croll, J.C.; Sexsmith, J.R. Prefrontal cortex oxygenation during incremental exercise in chronic fatigue syndrome. Clin. Physiol. Funct. I 2008, 28, 364–372. [Google Scholar] [CrossRef]
- Streeten, D.H.P.; Thomas, D.; Bell, D.S. The roles of orthostatic hypotension, orthostatic tachycardia, and subnormal erythrocyte volume in the pathogenesis of the chronic fatigue syndrome. Am. J. Med. Sci. 2000, 320, 1–8. [Google Scholar] [CrossRef]
- Streeten, D.H.P.; Bell, D.S. Circulating blood volume in chronic fatigue syndrome. Fatigue 2011, 4, 3–11. [Google Scholar] [CrossRef]
- Smith, K.A.; Waypa, G.B.; Schumacker, P.T. Redox signaling during hypoxia in mammalian cells. Redox. Biol. 2017, 13, 228–234. [Google Scholar] [CrossRef]
- Chong, J.; Soufan, O.; Li, C.; Caraus, I.; Li, S.; Bourque, G.; Wishart, D.S.; Xia, J. MetaboAnalyst 4.0: Towards more transparent and integrative metabolomics analysis. Nucleic Acids Res. 2018, 46, W486–W494. [Google Scholar] [CrossRef]
| Controls | Patients | ||
|---|---|---|---|
| Gender | Female | 19 | 32 |
| Age | Mean +/− SD | 49.2 +/− 10.5 | 48.5 +/− 13.7 |
| Median (range) | 50 (20–66) | 50 (19–71) | |
| BMI | Mean +/− SD | 26.8 +/− 5.9 | 25.5 +/− 5.7 |
| Median (range) | 26.1 (17–41) | 24.35 (16–41) | |
| Bell’s Disability Scale | 10–20 | ND | 7 |
| 30–40 | 13 | ||
| 50–60 | 5 | ||
| >60 | 3 | ||
| N/A | 4 | ||
| Onset of Illness | Gradual | ND | 17 |
| Sudden | 15 | ||
| Duration of Illness | <3 years | ND | 6 |
| >3 years | 26 | ||
| Intestinal Discomfort | Yes | ND | 23 |
| No | 9 |
| Super-Pathway | Metabolites | HMDB ID | Wilcoxon Rank-Sum Test | |
|---|---|---|---|---|
| p-Value | q-Value | |||
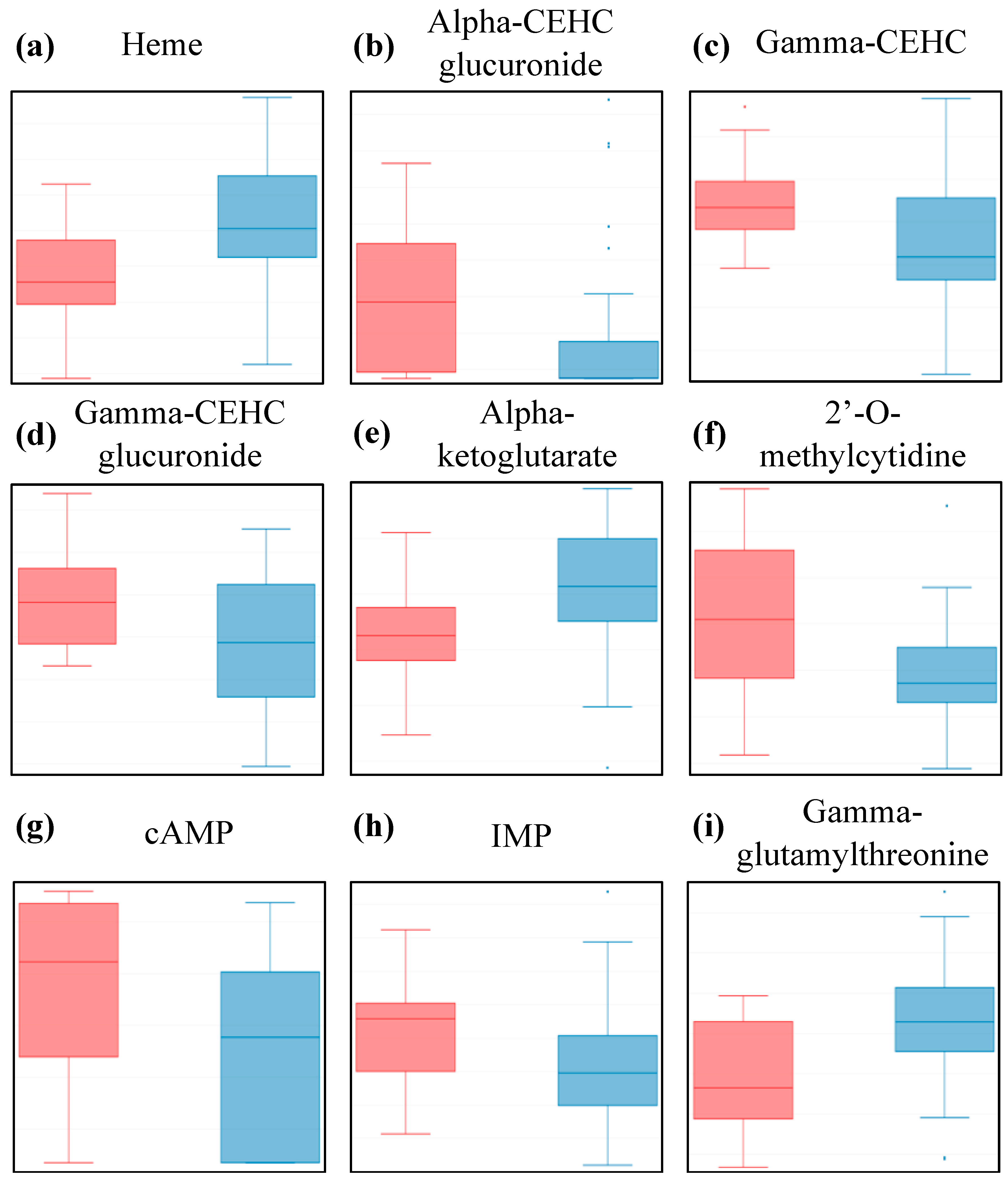
| Cofactors and Vitamins | Heme | HMDB03178 | 0.002 | 0.06 |
| Gamma-CEHC | HMDB01931 | 0.005 | 0.08 | |
| Alpha-CEHC glucuronide | HMDB62445 | 0.018 | 0.13 | |
| Gamma-CEHC glucuronide | N/A | 0.019 | 0.13 | |
| Energy | Alpha-ketoglutarate | HMDB00208 | 0.003 | 0.03 |
| Nucleotide | Inosine 5’-monophosphate (IMP) | HMDB00175 | 0.003 | 0.11 |
| 2’-O-methylcytidine | N/A | 0.009 | 0.13 | |
| Adenosine 3’-5’-cyclic monophosphate (cAMP) | HMDB00058 | 0.012 | 0.13 | |
| Peptide | Gamma-glutamylthreonine | HMDB29159 | 0.003 | 0.11 |
| Metabolite Set | Dataset | Total | Hits | p-Value | q-Value |
|---|---|---|---|---|---|
| Long-Chain-3-Hydroxyacyl-CoA Dehydrogenase Deficiency (LCHAD) | M® | 10 | 2 | 0.01 | 0.5 |
| [17] | 10 | 2 | 0.0005 | 0.004 | |
| [21] | - | - | - | - | |
| Anoxia | M® | 8 | 4 | 0.02 | 0.5 |
| [17] | - | - | - | - | |
| [21] | 8 | 6 | 0.23 | 0.33 | |
| Diabetic Ketoacidosis | M® | 2 | 1 | 0.02 | 0.5 |
| [17] | - | - | - | - | |
| [21] | - | - | - | - | |
| Obesity | M® | 2 | 1 | 0.02 | 0.5 |
| [17] | - | - | - | - | |
| [21] | - | - | - | - | |
| Persistent Hyperinsulinemic Hypoglycemia of Infancy (PHHI) | M® | 3 | 1 | 0.02 | 0.5 |
| [17] | 3 | 1 | 0.001 | 0.002 | |
| [21] | - | - | - | - | |
| Succinyl CoA:3-ketoacid CoA Transferase Deficiency | M® | 3 | 1 | 0.02 | 0.5 |
| [17] | - | - | - | - | |
| [21] | - | - | - | - | |
| Heart Failure | M® | 10 | 3 | 0.02 | 0.5 |
| [17] | - | - | - | - | |
| [21] | - | - | - | - | |
| Pyridoxamine 5’-Phosphate Oxidase Deficiency | M® | 3 | 2 | 0.04 | 0.6 |
| [17] | - | - | - | - | |
| [21] | 3 | 2 | 0.55 | 0.65 | |
| Carnitine Transporter Defect. Primary Systemic Carnitine Deficiency | M® | 4 | 1 | 0.05 | 0.6 |
| [17] | 4 | 1 | 0.001 | 0.002 | |
| [21] | - | - | - | - | |
| Carnitine Palmitoyl Transferase Deficiency II (CPT II) | M® | 8 | 4 | 0.05 | 0.6 |
| [17] | 8 | 1 | 0.001 | 0.002 | |
| [21] | 8 | 3 | 0.03 | 0.75 |
| Study 1 | Study 2 | q-Value < 0.15 |
|---|---|---|
| Metabolon® | Germain et al. [21] | None |
| Identical population (13 controls & 9 patients) | ||
| Metabolon® 19 controls & 32 patients | Germain et al. [21] 18 controls & 18 patients | 5,6-dihydrothymine, taurine, spermidine, N-acetylcarnosine |
| Metabolon® 19 controls & 32 patients | Naviaux et al. [19] 21 controls & 23 patients | adenosine, flavin adenine dinucleotide (FAD) |
| Metabolon® 19 controls & 32 patients | Armstrong et al. [17] 25 controls & 34 patients | glucose, glutamate, hypoxanthine, phenylalanine, alanine, proline, threonine, asparagine, lysine, serine, lactate, creatinine |
| Germain et al. [21] 18 controls & 18 patients | Armstrong et al. [17] 25 controls & 34 patients | glucose, glutamate, hypoxanthine |
| Germain et al. [21] 18 controls & 18 patients | Naviaux et al. [19] 21 controls & 23 patients | gluconate, S-1-pyrroline-5-carboxylate |
| Armstrong et al. [17] 25 controls & 34 patients | Naviaux et al. [19] 21 controls & 23 patients | hypoxanthine, lactate |
| Name | Patients | ||
|---|---|---|---|
| Median | Minimum | Maximum | |
| All Metabolites | 0.980 | 0.980 | 0.992 |
| Amino Acids | 0.992 | 0.988 | 1.000 |
| Carbohydrates | 0.976 | 0.968 | 0.988 |
| Cofactors and Vitamins | 0.500 | 0.432 | 0.552 |
| Energy | 0.216 | 0.208 | 0.264 |
| Lipids | 0.936 | 0.900 | 0.952 |
| Nucleotides | 0.624 | 0.596 | 0.632 |
| Peptides | 0.764 | 0.744 | 0.808 |
| Xenobiotics | 0.728 | 0.708 | 0.736 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Germain, A.; Ruppert, D.; Levine, S.M.; Hanson, M.R. Prospective Biomarkers from Plasma Metabolomics of Myalgic Encephalomyelitis/Chronic Fatigue Syndrome Implicate Redox Imbalance in Disease Symptomatology. Metabolites 2018, 8, 90. https://doi.org/10.3390/metabo8040090
Germain A, Ruppert D, Levine SM, Hanson MR. Prospective Biomarkers from Plasma Metabolomics of Myalgic Encephalomyelitis/Chronic Fatigue Syndrome Implicate Redox Imbalance in Disease Symptomatology. Metabolites. 2018; 8(4):90. https://doi.org/10.3390/metabo8040090
Chicago/Turabian StyleGermain, Arnaud, David Ruppert, Susan M. Levine, and Maureen R. Hanson. 2018. "Prospective Biomarkers from Plasma Metabolomics of Myalgic Encephalomyelitis/Chronic Fatigue Syndrome Implicate Redox Imbalance in Disease Symptomatology" Metabolites 8, no. 4: 90. https://doi.org/10.3390/metabo8040090
APA StyleGermain, A., Ruppert, D., Levine, S. M., & Hanson, M. R. (2018). Prospective Biomarkers from Plasma Metabolomics of Myalgic Encephalomyelitis/Chronic Fatigue Syndrome Implicate Redox Imbalance in Disease Symptomatology. Metabolites, 8(4), 90. https://doi.org/10.3390/metabo8040090






