Dynamics of Monoterpene Formation in Spike Lavender Plants
Abstract
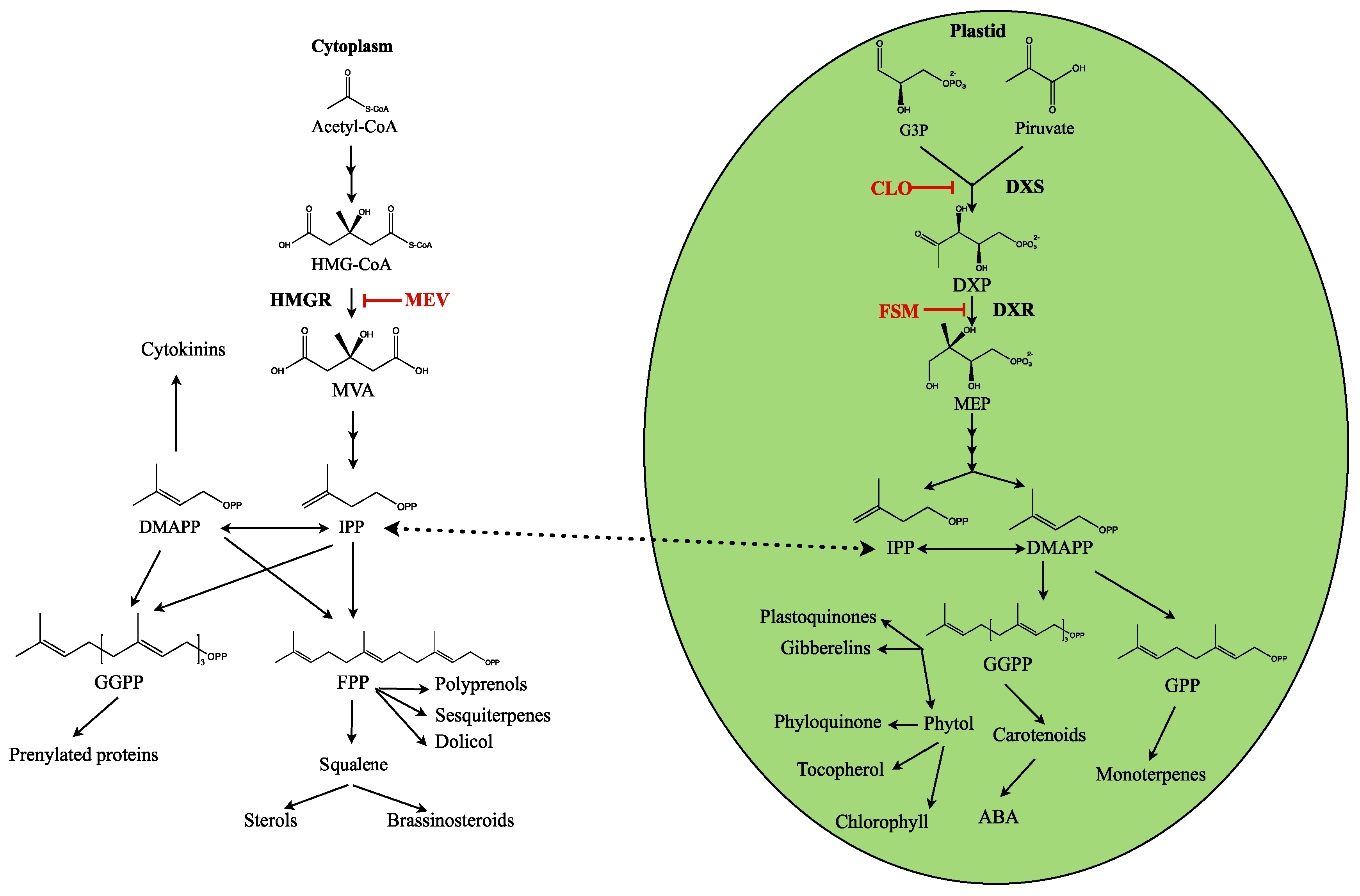
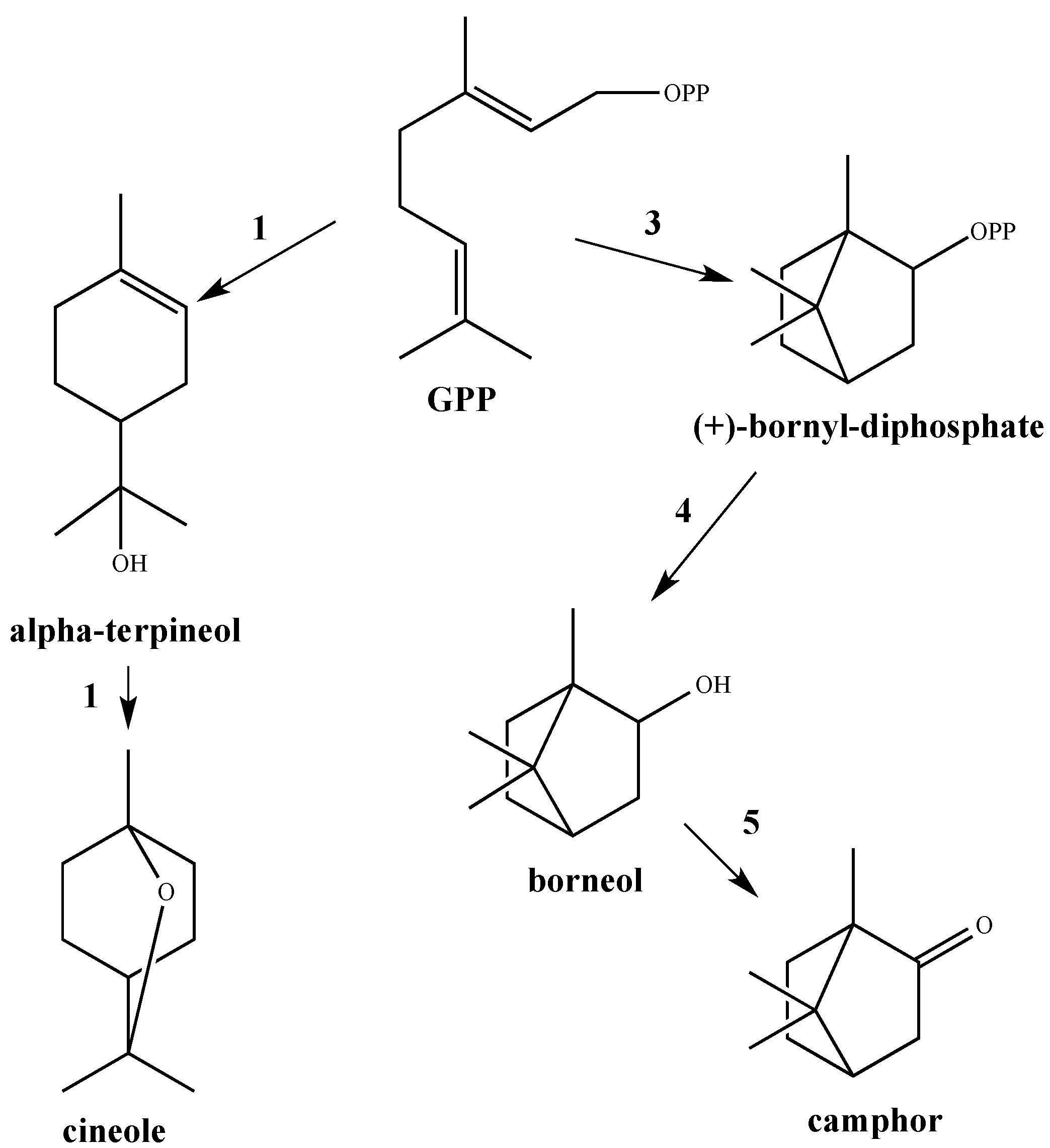
1. Introduction
2. Results
2.1. GC/MS Analysis
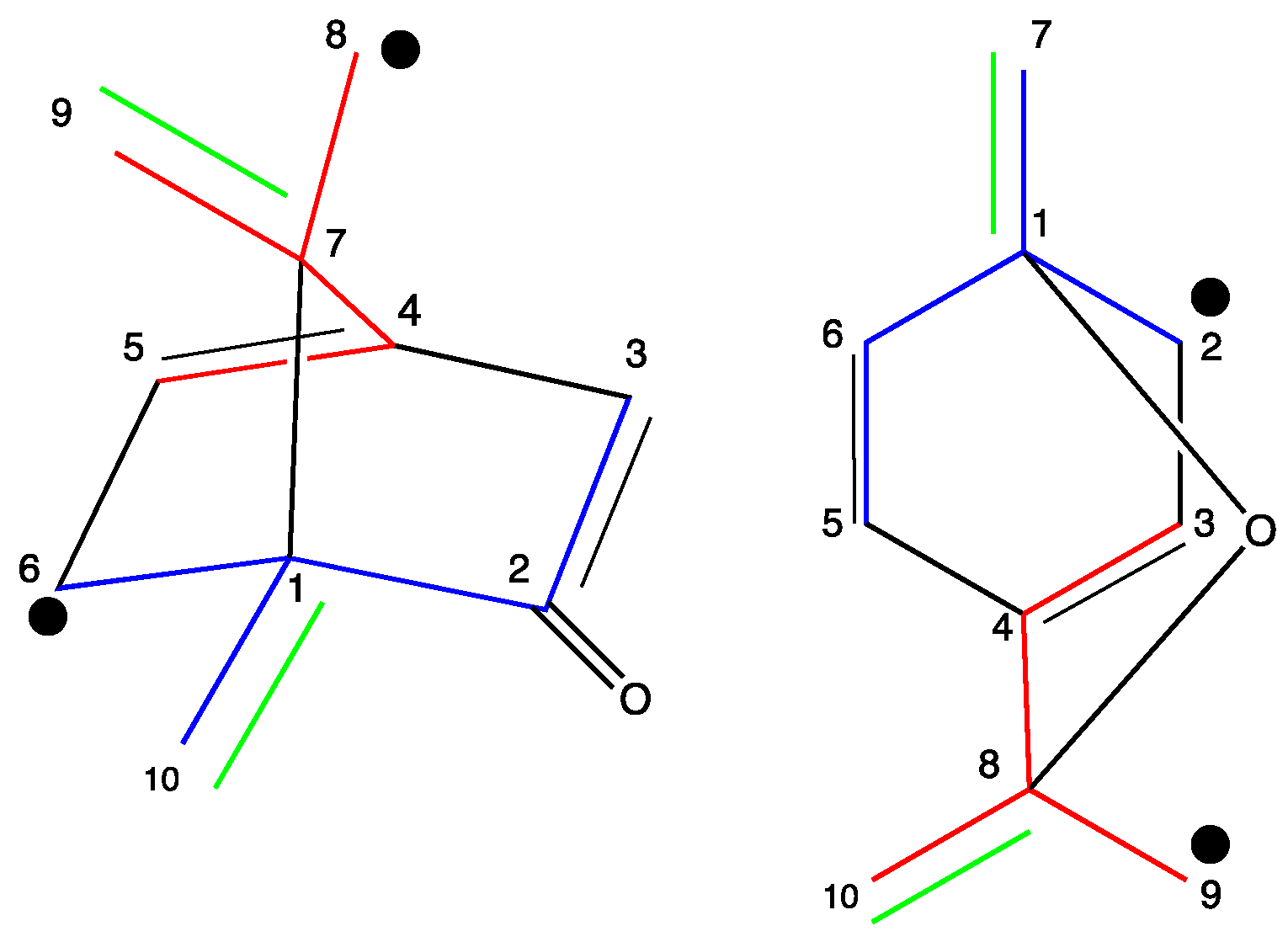
2.2. NMR Data
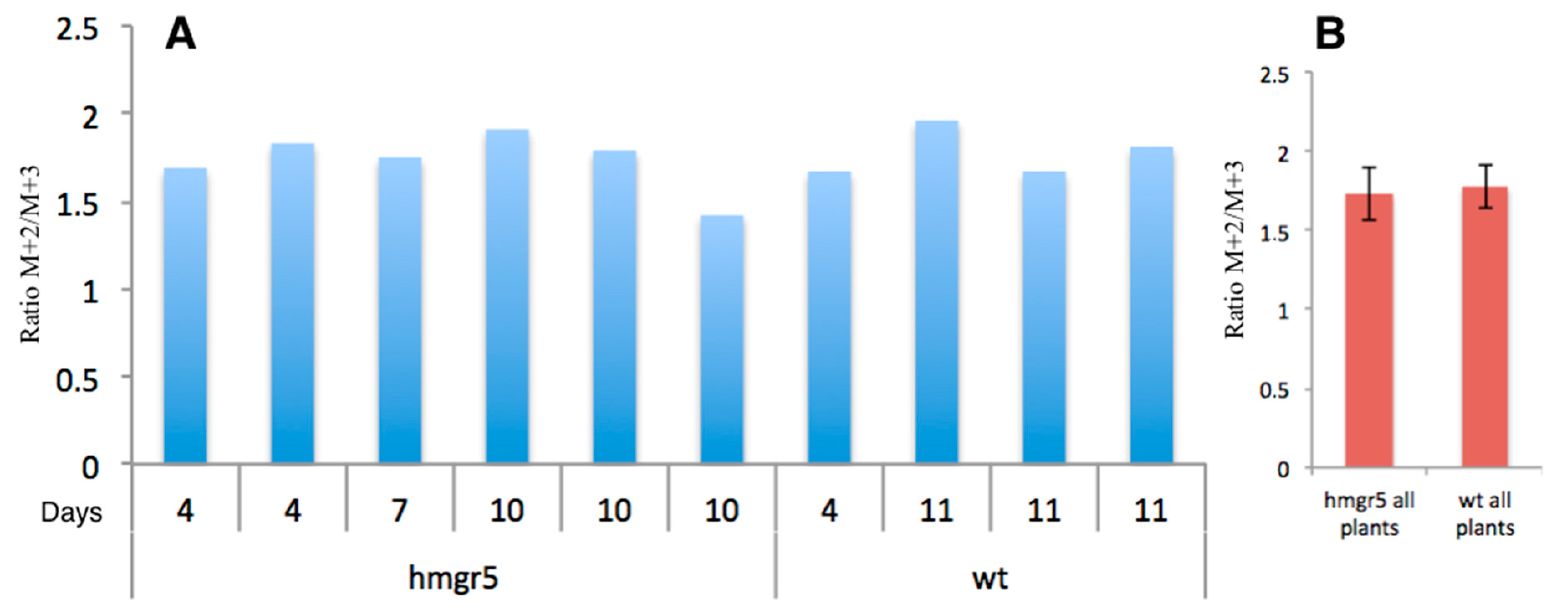
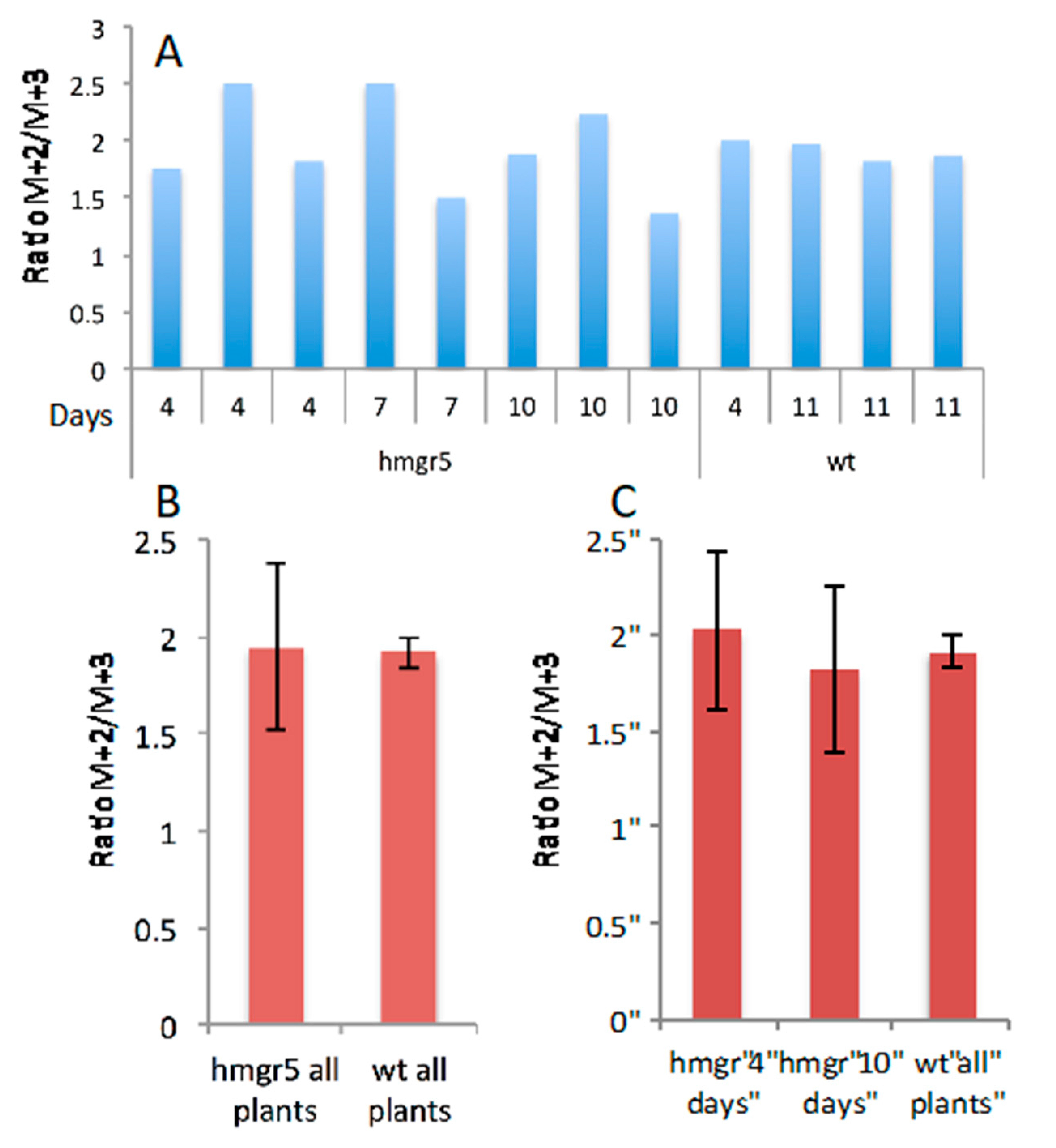
2.3. 13CO2 Labelling in Transgenic HMGR5 Plants
3. Discussion
3.1. GC/MS Analysis
3.2. NMR Data
3.3. 13CO2 Labelling in Transgenic HMGR5 Plants
4. Materials and Methods
4.1. Plant Material
4.2. 13CO2 Labelling Experiments
4.3. Essential Oil Extraction from Labelled Material
4.4. GC/MS Measurements
4.5. NMR Measurements
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Enfissi, E.M.A.; Fraser, P.D.; Lois, L.M.; Boronat, A.; Schuch, W.; Bramley, P.M. Metabolic engineering of the mevalonate and non-mevalonate isopentenyl diphosphate-forming pathways for the production of health-promoting isoprenoids in tomato. Plant Biotechnol. J. 2005, 3, 17–28. [Google Scholar] [CrossRef] [PubMed]
- Dudareva, N.; Pichersky, E.; Gershenzon, J. Biochemistry of plant volatiles. Plant Physiol. 2004, 135, 1893–1902. [Google Scholar] [CrossRef] [PubMed]
- Baldwin, I.T.; Halitschke, R.; Paschold, A.; von Dahl, C.C.; Preston, C.A. Volatile signalling in plant-plant interaction: “Talking trees” in the genomics era. Science 2006, 311, 812–815. [Google Scholar] [CrossRef] [PubMed]
- Tholl, D. Terpene synthases and the regulation, diversity and biological roles of terpene metabolism. Curr. Opin. Plant Biol. 2006, 9, 297–304. [Google Scholar] [CrossRef] [PubMed]
- Raut, J.S.; Karuppayil, S.M. A status review on the medicinal properties of Essential oils. Ind. Crops Prod. 2014, 62, 250–264. [Google Scholar] [CrossRef]
- Verlet, N. Comercial aspects of essencial oil production. In Volatile Oil Crops: Their Biology, Biochemistry and Production; Wiley: Weinheim, Germany, 1993; pp. 137–174. [Google Scholar]
- Aharoni, A.; Jongsma, M.A.; Bouwmeester, H.J. Volatile Science? Metabolic Engineering of Terpenoids in Plants. Trends Plant Sci. 2005, 10, 594–602. [Google Scholar] [CrossRef] [PubMed]
- Schwab, W.; Davidovich-Rikanati, R.; Lewinsohn, E. Biosynthesis of plant-derived flavor compounds. Plant J. 2008, 54, 712–732. [Google Scholar] [CrossRef] [PubMed]
- Dias, D.A.; Urban, S.; Roessner, U. A Historical Overview of Natural Products in Drug Discovery. Metabolites 2012, 2, 303–336. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Wang, H.; Ye, H.-C.; Li, G.-F. Advances in the plant isoprenoid biosíntesis pathway and its metabolic engineering. J. Integr. Plant Biol. 2005, 47, 769–782. [Google Scholar] [CrossRef]
- Rodríguez-Concepción, M.; Boronat, A. Breaking new ground in the regulation of the early steps of plant isoprenoid biosynthesis. Curr. Opin. Plant Biol. 2015, 25, 17–22. [Google Scholar] [CrossRef] [PubMed]
- Manzano, D.; Fernandez-Busquets, X.; Schaller, H.; González, V. The metabolic imbalance underlying lesion formation in Arabidopsis thaliana overexpressing farnesyl diphosphate synthase (isoform 1S) leads to oxidative stress and is triggered by the developmental decline of endogenous HMGR activity. Planta 2004, 219, 982–992. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Concepción, M. Early steps in isoprenoid biosynthesis: Multilevel regulation of the supply of common precursors in plant cells. Phytochem. Rev. 2006, 5, 1–15. [Google Scholar] [CrossRef]
- Leivar, P.; González, V.M.; Castel, S.; Trelease, R.N.; López-Iglesias, C.; Arró, M.; Boronat, A.; Campos, N.; Ferrer, A.; Fernàndez-Busquets, X. Sub-cellular localization of Arabidopsis 3-hydroxy-3-methylglutaryl coenzyme A reductase. Plant Physiol. 2005, 137, 57–69. [Google Scholar] [CrossRef] [PubMed]
- Carretero-Paulet, L.; Ahumanda, I.; Cunillera, N.; Rodríguez-Concepción, M.; Ferrer, A.; Boronat, A.; Campos, N. Expression and molecular analysis of the Arabidopsis DXR gene encoding 1-deoxy-d-xylulose 5-phosphate reductoisomerase, the first commit-ted enzyme of the 2-C-methyl-d-erythritol 4-phosphate pathway. Plant Physiol. 2002, 129, 1581–1591. [Google Scholar] [CrossRef] [PubMed]
- Cordoba, E.; Salmi, M.; León, P. Unravelling the regulatory mechanisms that modulatethe MEP pathway in higher plants. J. Exp. Bot. 2009, 60, 2933–2934. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Concepción, M.; Boronat, A. Elucidation of the methylerythritol phosphate pathway for isoprenoid biosynthesis in bacteria and plastids. A metabolic milestone achieved through genomics. Plant Physiol. 2002, 130, 1079–1089. [Google Scholar] [CrossRef] [PubMed]
- Skorupinska-Tudek, K.; Poznanski, J.; Wojcik, J.; Bienkowski, T.; Szostkiewicz, I.; Zelman-Femiak, M.; Bajda, A.; Chojnacki, T.; Olszowska, O.; Grunler, J.; et al. Contribution of the Mevalonate and Methylerythritol Phosphate Pathways to the Biosynthesis of Dolichols in Plants. J. Biol. Chem. 2008, 283, 21024–21035. [Google Scholar] [CrossRef] [PubMed]
- Schuhr, C.A.; Radykewicz, T.; Sagner, S.; Latzel, C.; Zenk, M.H.; Arigoni, D.; Bacher, A.; Rohdich, F.; Eisenreich, W. Quantitative assessment of crosstalk between the two isoprenoid biosynthesis pathways in plants by NMR spectroscopy. Phytochem. Rev. 2003, 2, 3–16. [Google Scholar] [CrossRef]
- Bouvier, F.; Rahier, A.; Camara, B. Biogenesis, molecular regulation and function of plant isoprenoids. Prog. Lipid Res. 2005, 44, 357–429. [Google Scholar] [CrossRef] [PubMed]
- Mahmoud, S.S.; Croteau, R.B. Metabolic engineering of essential oil yield and composition in mint by altering expression of deoxyxylulose phosphate reductoisomerase and menthofuran synthase. Proc. Natl. Acad. Sci. USA 2001, 98, 8915–8920. [Google Scholar] [CrossRef] [PubMed]
- Hallahan, D.L. Monoterpenoid biosynthesis in glandular trichomes of Labiate plants. Adv. Bot. Res. 2000, 31, 77–120. [Google Scholar]
- Muñoz-Bertomeu, J. Ingeniería Metabólica de la Biosíntesis de Monoterpenos en Lavandula latifolia. Ph.D. Thesis, University of Valencia, València, Spain, 2007. [Google Scholar]
- Peters, R.J.; Croteau, R.B. Alternative termination chemistries utilized by monoterpene cyclases: Chimeric analysis of bornyl diphosphate, 1,8-cineole, and sabinene synthases. Arch. Biochem. Biophys. 2003, 417, 203–211. [Google Scholar] [CrossRef]
- Dewick, P.M. Medicinal Natural Products, a Biosynthetic Approach, 2nd ed.; Wiley & Sons: West Sussex, UK, 2002. [Google Scholar]
- Harborne, J.B.; Williams, C.A. Phytochemistry of the genus Lavandula. In Lavender; Lis-Balchin, M., Ed.; Taylor & Francis Inc.: New York, NY, USA, 2002; pp. 86–99. [Google Scholar]
- Salido, S.; Altarejos, J.; Nogueras, M.; Sánchez, A.; Luque, P. Chemical composition and seasonal variations of spike lavender oil from Southern Spain. J. Essent. Oil Res. 2004, 16, 206–210. [Google Scholar] [CrossRef]
- Muñoz-Bertomeu, J.; Arrillaga, I.; Segura, J. Essential oil variation within and among natural populations of Lavandula latifolia and its relation to their ecological áreas. Biochem. Syst. Ecol. 2007, 35, 479–488. [Google Scholar] [CrossRef]
- Herraiz-Peñalver, D.; Cases, M.A.; Varela, F.; Navarrete, P.; Sánchez-Vioque, R.; Usano-Alemany, J. Chemical characterization of Lavandula latifolia Medik. Essential oil from Spanish wild populations. Biochem. Syst. Ecol. 2013, 46, 59–68. [Google Scholar] [CrossRef]
- Guillén, M.D.; Cabo, N.; Burillo, J. Characterisation of the essential oils of some cultivated aromatic plants of industrial interest. J. Sci. Food Agric. 1996, 70, 359–363. [Google Scholar] [CrossRef]
- Masotti, V.; Juteau, F.; Bessière, J.M.; Viano, J. Seasonal and phenological variations of the essential oil from the narrow endemic species Artemisia molinieri and its biological activities. J. Agric. Food Chem. 2003, 51, 7115–7121. [Google Scholar] [CrossRef] [PubMed]
- Angioni, A.; Barra, A.; Coroneo, V.; Dessi, S.; Cabras, P. Chemical composition, seasonal variability, and antifungal activity of Lavandula stoechas L. ssp. stoechas essential oils from stem/leaves and flowers. J. Agric. Food Chem. 2006, 54, 4364–4370. [Google Scholar] [PubMed]
- Figueiredo, A.C.; Barroso, J.G.; Pedro, L.G.; Scheffer, J.J.C. Factors affecting secondary metabolite production in plants: Volatile components and essential oils. Flavour Fragr. J. 2008, 23, 213–226. [Google Scholar] [CrossRef]
- Muñoz-Bertomeu, J.; Arrillaga, I.; Ros, R.; Segura, J. Up-regulation of 1-deoxy-d-xylulose-5-phosphate synthase enhances production of essential oils in transgenic spike lavender. Plant Physiol. 2006, 142, 890–900. [Google Scholar] [CrossRef] [PubMed]
- Mendoza-Poudereux, I.; Muñoz-Bertomeu, J.; Arrillaga, I.; Segura, J. Deoxyxylulose 5-phosphate reductoisomerase is not a rate-determining enzyme for essential oil production in spike lavender. J. Plant Physiol. 2014, 171, 1564–1570. [Google Scholar] [CrossRef] [PubMed]
- Liao, P.; Hemmerlin, A.; Bach, T.J.; Chye, M.L. The potential of the mevalonate pathway for enhanced isoprenoid production. Biotechnol. Adv. 2016, 34, 697–713. [Google Scholar] [CrossRef] [PubMed]
- Muñoz-Bertomeu, J.; Sales, E.; Ros, R.; Arrillaga, I.; Segura, J. Up-regulation of an N-terminal truncated 3-hydroxy-3-methylglutaryl CoA reductase enhances production of essential oils and sterols in transgenic Lavandula latifolia. Plant Biotechnol. J. 2007, 5, 746–758. [Google Scholar] [CrossRef] [PubMed]
- Eisenreich, W.; Bacher, A. Advances of highresolution NMR techniques in the structural and metabolic analysis of plant biochemistry. Phytochemistry 2007, 68, 2799–2815. [Google Scholar] [CrossRef] [PubMed]
- Mendoza-Poudereux, I.; Kutzner, E.; Huber, C.; Segura, J.; Eisenreich, W.; Arrillaga, I. Metabolic cross-talk between pathways of terpenoid backbone biosynthesis in spike lavender. Plant Physiol. Biochem. 2015, 95, 113–120. [Google Scholar] [CrossRef] [PubMed]
- Knispel, N.; Ostrozhenkova, E.; Schramek, N.; Huber, C.; Peña-Rodríguez, L.M.; Bonfill, M.; Palazón, J.; Wischmann, G.; Cusidó, R.M.; Eisenreich, W. Biosynthesis of Panaxynol and Panaxydol in Panax ginseng. Molecules 2013, 18, 7686–7698. [Google Scholar] [CrossRef] [PubMed]
- Schaefer, J.; Stejskal, E.O.; Beard, C.F. Carbon-13 Nuclear Magnetic Resonance Analysis of Metabolism in Soybean Labelled by 13CO2. Plant Physiol. 1975, 55, 1048–1053. [Google Scholar] [CrossRef] [PubMed]
- Eisenreich, W.; Huber, C.; Kutzner, E.; Knispel, N.; Schramek, N. Isotopologue Profiling—Towards a better understanding of metabolic pathways. In The Handbook of Plant Metabolomics; Wiley VCH: Weinheim, Germany, 2013. [Google Scholar]
- Wright, L.P.; Rohwer, J.M.; Ghirardo, A.; Hammerbacher, A.; Ortiz, M.; Raguschke, B.; Schnitzler, J.-P.; Gershenzon, J.; Phillips, M.P. Deoxyxylulose 5-phosphate synthase controls flux through the methylerythritol 4-phosphate pathway in Arabidopsis thaliana. Plant Physiol. 2014, 165, 1488–1504. [Google Scholar] [CrossRef] [PubMed]
- Schramek, N.; Wang, H.; Römisch-Margl, W.; Keil, B.; Radykewicz, T.; Winzenhörlein, B.; Beerhues, L.; Bacher, A.; Rohdich, F.; Gershenzon, J.; et al. Artemisinin biosynthesis in growing plants of Artemisia annua. A 13CO2 study. Phytochemistry 2010, 71, 179–187. [Google Scholar] [CrossRef] [PubMed]
- Römisch-Margl, W.; Schramek, N.; Radykewicz, T.; Ettenhuber, C.; Eylert, E.; Römisch-Margl, L.; Schwarz, C.; Dobner, M.; Demmel, N.; Winzenhörlein, B.; et al. 13CO2 as a universal metabolic tracer in isotopologue perturbation experiments. Phytochemistry 2007, 68, 2273–2289. [Google Scholar] [CrossRef] [PubMed]
- Ostrozhenkova, E.; Eylert, E.; Schramek, N.; Golan-Goldhirsh, A.; Bacher, A.; Eisenreich, W. Biosynthesis of the chromogen hermidin from Mercurialis annua L. Phytochemistry 2007, 68, 2816–2824. [Google Scholar] [CrossRef] [PubMed]
- Ghirardo, A.; Wright, L.P.; Bi, Z.; Rosenkranz, M.; Pulido, P.; Rodríguez-Concepción, M.; Niinemets, Ü.; Brüggemann, N.; Gershenzon, J.; Schnitzler, J.-P. Metabolic Flux Analysis of Plastidic Isoprenoid Biosynthesis in Poplar Leaves Emitting and Nonemitting Isoprene. Plant Physiol. 2014, 165, 37–51. [Google Scholar] [CrossRef] [PubMed]
- Woronuk, G.; Demissie, Z.; Rheault, M.; Mahmoud, S. Biosynthesis and therapeutic properties of Lavandula essential oil constituents. Planta Med. 2011, 77, 7–15. [Google Scholar] [CrossRef] [PubMed]
- Pascual, J.; Caballero, E.; Caballero, C.; Machin, G. Constituents of the essential oil of Lavandula latifolia. Phytochemistry 1983, 22, 1033–1034. [Google Scholar] [CrossRef]
- Dudareva, N.; Klempien, A.; Muhlemann, J.K.; Kaplan, I. Biosynthesis, function and metabolic engineering of plant volatile organic compounds. New Phytol. 2013, 198, 16–32. [Google Scholar] [CrossRef] [PubMed]
- Lange, B.M.; Wildung, M.R.; Stauber, E.J.; Sanchez, C.; Pouchnik, D.; Croteau, R. Probing essential oil biosynthesis and secretion by functional evaluation of expressed sequence tags from mint glandular trichomes. Proc. Natl. Acad. Sci. USA 2000, 97, 2934–2939. [Google Scholar] [CrossRef] [PubMed]
- Wildung, M.R.; Croteau, R.B. Genetic engineering of peppermint for improved essential oil composition and yield. Transgenic Res. 2005, 14, 365–372. [Google Scholar] [CrossRef] [PubMed]
- Peña-Rodríguez, L.M.; Yam-Puc, A.; Knispel, N.; Schramek, N.; Huber, C.; Graßberger, C.; Ramírez Torres, F.G.; Escalante-Erosa, F.; García-Sosa, K.; Hiebert-Giesbrecht, M.R.; et al. Isotopologue profiling of triterpene formation under physiological conditions. Biosynthesis of lupeol-3-(3′-R-hydroxy)-stearate in Pentalinon andrieuxii. J. Org. Chem. 2014, 79, 2864–2873. [Google Scholar] [CrossRef] [PubMed]
- Alam, P.; Abdin, M.Z. Over-expression of HMG-CoA reductase and amorpha-4,11-diene synthase genes in Artemisia annua L. and its influence on artemisinin content. Plant Cell Rep. 2011, 30, 1919–1928. [Google Scholar] [CrossRef] [PubMed]
- Shi, M.; Luo, X.; Ju, G.; Yu, X.; Hao, X.; Huang, Q.; Xiao, J.; Cui, L.; Kai, G. Increased accumulation of the cardio-cerebrovascular disease treatment drug tanshinone in Salvia miltiorrhiza hairy roots by the enzymes 3-hydroxy-3-methylglutaryl CoA reductase and 1-deoxy-d-xylulose 5-phosphate reductoisomerase. Funct. Integr. Genom. 2014, 14, 603–615. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.-J.; Lee, O.R.; Oh, J.Y.; Jang, M.-G.; Yang, D.-C. Functional Analysis of 3-Hydroxy-3-Methylglutaryl Coenzyme A Reductase Encoding Genes in Triterpene Saponin-Producing Ginseng. Plant Physiol. 2014, 165, 373–387. [Google Scholar] [CrossRef] [PubMed]
- Lipko, A.; Swiezewska, E. Isoprenoid generating systems in plants—A handy toolbox how to assess contribution of the mevalonate and methylerythritol phosphate pathways to the biosynthetic process. Prog. Lipid Res. 2016, 63, 70–92. [Google Scholar] [CrossRef] [PubMed]
- Courdavault, V.; Thiersault, M.; Courtois, M.; Gantet, P.; Oudin, A.; Doireau, P.; St-Pierre, B.; Giglioli-Guivarc’h, N. CaaX-prenyltransferases are essential for expression of genes involved in the early stages of monoterpenoid biosynthetic pathway in Catharanthus roseus cells. Plant Mol. Biol. 2005, 57, 855–870. [Google Scholar] [CrossRef] [PubMed]
- Calvo, M.C.; Segura, J. In vitro morphogenesis from explants of Lavandula latifolia and Lavandula stoeachas seedlings. Sci. Hortic. 1988, 36, 131–137. [Google Scholar] [CrossRef]
- Braumann, J.I. Least squares analysis and simplification of multi-isotope mass spectra. Anal. Chem. 1966, 38, 607–610. [Google Scholar] [CrossRef]
- Pickup, J.F.; McPherson, K. Theoretical considerations in stable isotope dilution mass spectrometry for organic analysis. Anal. Chem. 1976, 48, 1885–1890. [Google Scholar] [CrossRef]
- Korzekwa, K.; Howald, W.N.; Trager, W.F. The use of Brauman’s least squares approach for the quantification of deuterated chlorophenols. Biol. Mass Spectrom. 1990, 19, 211–217. [Google Scholar] [CrossRef]
- Lee, W.P.; Byerley, L.O.; Bergner, E.A.; Edmond, J. Mass isotopomer analysis: Theoretical and practical considerations. Biol. Mass Spectrom. 1991, 20, 451–458. [Google Scholar] [CrossRef] [PubMed]
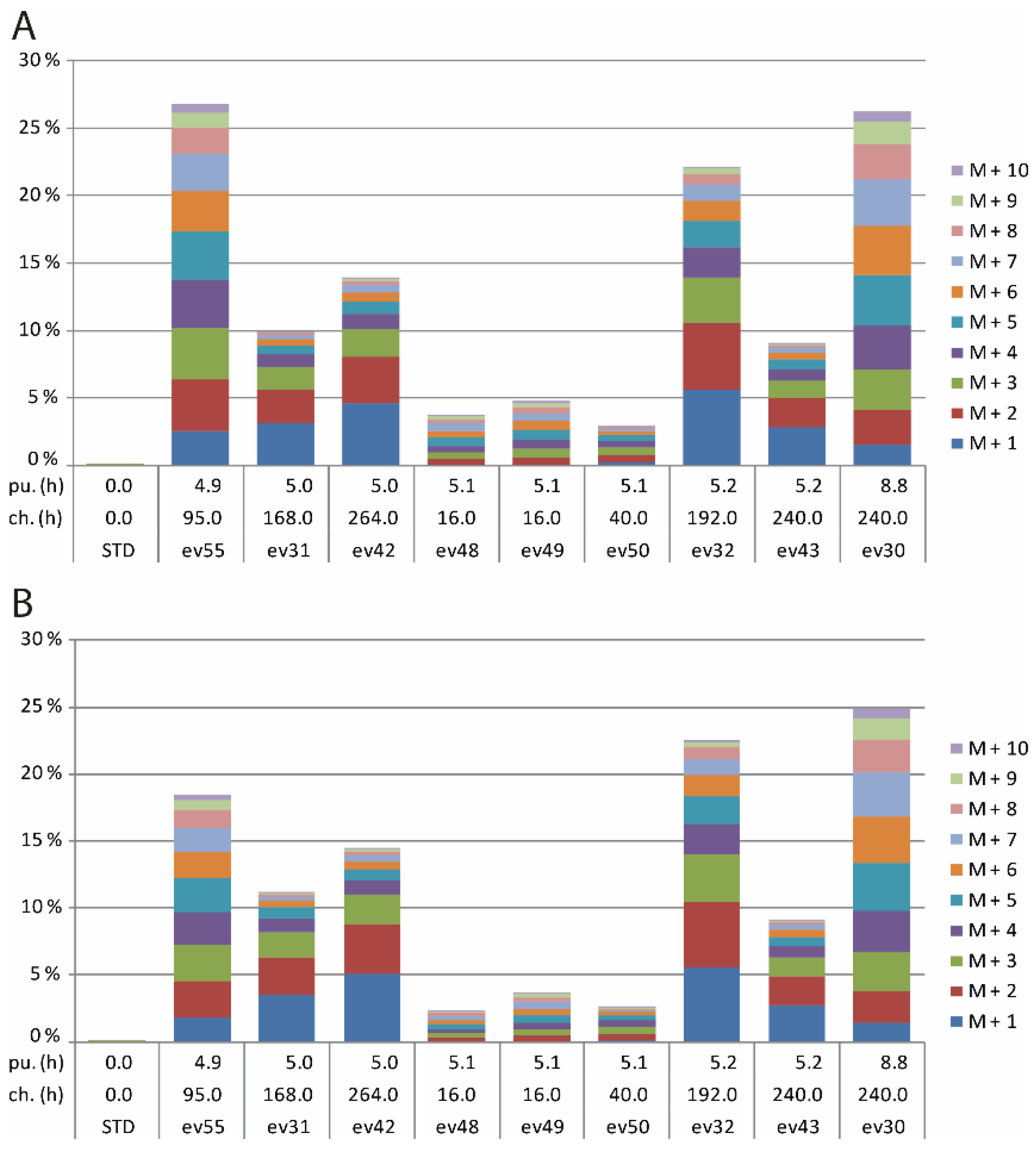


| Excess Values (%) | Ratio | ||||||
|---|---|---|---|---|---|---|---|
| Pulse time (h) | Chase time (h) | Sample | M+2 | M+3 | M+2/M+3 | M+2/(M+2 + M+3) | |
| Camphor | 4.9 a | 95 | 55 | 2.72 | 2.80 | 0.97 | 0.49 |
| 5.0 a | 168 | 31 | 2.78 | 1.91 | 1.45 | 0.59 | |
| 5.0 a | 264 | 42 | 3.65 | 2.25 | 1.63 | 0.62 | |
| 5.1 a | 16 | 48 | 0.29 | 0.34 | 0.86 | 0.46 | |
| 5.1 a | 16 | 49 | 0.44 | 0.48 | 0.90 | 0.47 | |
| 5.1 a | 40 | 50 | 0.54 | 0.54 | 1.00 | 0.50 | |
| 5.2 a | 192 | 32 | 4.86 | 3.53 | 1.38 | 0.58 | |
| 5.2 a | 240 | 43 | 2.10 | 1.40 | 1.50 | 0.60 | |
| 8.8 a | 240 | 30 | 2.40 | 2.86 | 0.84 | 0.46 | |
| - | - | - | - | Mean ± SD | 1.17 ± 0.31 | 0.53 ± 0.07 | |
| Cineole | 4.9 a | 95 | 55 | 3.81 | 3.87 | 0.98 | 0.50 |
| 5.0 a | 168 | 31 | 2.52 | 1.66 | 1.52 | 0.60 | |
| 5.0 a | 264 | 42 | 3.43 | 2.13 | 1.61 | 0.62 | |
| 5.1 a | 16 | 48 | 0.44 | 0.49 | 0.90 | 0.47 | |
| 5.1 a | 16 | 49 | 0.55 | 0.60 | 0.92 | 0.48 | |
| 5.1 a | 40 | 50 | 0.58 | 0.58 | 1.00 | 0.50 | |
| 5.2 a | 192 | 32 | 4.87 | 3.41 | 1.43 | 0.59 | |
| 5.2 a | 240 | 43 | 2.13 | 1.37 | 1.55 | 0.61 | |
| 8.8 a | 240 | 30 | 2.61 | 2.96 | 0.88 | 0.47 | |
| - | - | - | - | Mean ± SD | 1.20 ± 0.32 | 0.54 ± 0.07 | |
| 1,8-cineole Carbon Atom | Signal No. | Chemical Shift (ppm) | - | Multiplicity | Integral | Coupling Constant (Hz) | Correlation | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| - | 1H | 1H | 13C (HSQC, HMBC) | - | - | - | - | COSY | COSYph | TOCSY (long range) | HMBC | HSQC | NOESY |
| 3/5 | 1 | 2.05 | 22.80 | CH2 | Multiplet | 2 | - | 2, 3, 4 (w) | 2, 3, 4 | 2, 3, 5 | 1, 2, 3, 4, 5, 6 | 1, 3 | - |
| 2/6 | 2 | 1.69 | 31.48 | CH2 | Multiplet | 2 (2.4) | - | 1, 3 | 1, 3 | 1, 3, 4 | 1, 2, 3, 4, 6 (strong) | 2, 3 | - |
| - | 3 | 1.52 | 22.80 31.48 | CH2 | Multiplet | 4 | - | 1, 2 | 1, 2 | 1, 2, 4 | - | - | - |
| 4 | 4 | 1.43 | 32.91 | CH | Multiplet | 1 | - | 1 (weak) | 1 | 1, 2, 3 | 1, 2, 3, 5 (s) | 4 | - |
| 9/10 | 5 | 1.26 | 28.87 | CH3 | Singulet | 6 | - | - | - | - | 4, 5 (strong) | 5 | - |
| 7 | 6 | 1.07 | 27.55 | CH3 | Singulet | 3 | - | - | - | - | 2 (weak), 3 (weak) | 6 | - |
| 1 | - | - | 69.92 | - | - | - | - | - | - | - | 1, 2, 3 (weak), 6 (strong) | - | - |
| 8 | - | - | 73.76 | - | - | - | - | - | - | - | 1, 2 (weak), 3, 5 (strong), 6 (weak) | - | - |
| Camphor Carbon Atom | Signal No. | Chemical Shift (ppm) | - | Multiplicity | Integral | Coupling Constant (HZ) | Correlation | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| - | 1H | 1H | 13C (HSQC, HMBC) | - | - | - | - | COSY | COSYph | TOCSY (long range) | HMBC | HSQC | NOESY |
| 3 | 1 | 2.36 | 43.32 | CH2 | ddd | 1 | 18.0 4.5 3.3 | 2, 4 | 2, 4, 7 | 2, 3 | 1, 3, 4, 5+6+7 (weak), 8, 10 | 1, 2, 4 | - |
| 4 | 2 | 2.10 | 43.04 | CH | Triplet | 1 | 4.5 | 1 | 1 | 1, 3, 4, 5, 6, 7 | 1, 3, 4, 5+6+7 (weak), 8, 10 | 1, 2, 4 | - |
| 5 | 3 | 1.96 | 27.04 | CH2 | Multiplet | 1 | - | 5, 7 | 5, 7 | 1, 2, 5, 6, 7 | 1, 2 (weak), 4, 5, 6 | 3, 7 | - |
| 3 | 4 | 1.85 | 43.32 | CH2 | Doublet | 1 | 18.2 | 1 | 1 | 2 | 1, 3, 4, 5+6+7 (weak), 8, 10 | 1, 2, 4 | - |
| 6 | 5 | 1.69 | 29.91 | CH2 | dd | 1 | 13.1 4.1 | 3, 6 | 3, 6 | 3, 6, 7 | 2, 3, 7, 9 | 5, 6 | - |
| 6 | 6 | 1.41 | 29.91 | CH2 | Multiplet | 1 | - | 5, 7 | 5, 7 | 2, 3, 5, 7 | 2, 3, 7, 9 | 5, 6 | - |
| 5 | 7 | 1.35 | 27.04 | CH2 | Multiplet | 1 | - | 3, 6 | 3, 6 | 2, 3, 5, 6 | 1, 2 (weak), 4, 5, 6 | 3, 7 | - |
| 9 | 8 | 0.97 | 19.15 | CH3 | Singulet | 3 | - | - | - | - | 10 | 8 | 5, 3 |
| 10 | 9 | 0.92 | 9.27 | CH3 | Singulet | 3 | - | - | - | - | 5 | 9 | 5 |
| 8 | 10 | 0.85 | 19.80 | CH3 | Singulet | 3 | - | - | - | - | 2, 4, 8 | 10 | 1 |
| 2 | - | - | 220.06 | CH2 | ddd | 1 | - | - | - | - | 1, 2, 4, 5, 6, 9 | - | - |
| 1 | - | - | 57.76 | CH | Triplet | 1 | - | - | - | - | 2, 3+4 (weak), 5, 6, 7, 8, 9, 10 | - | - |
| 7 | - | - | 46.84 | CH2 | Multiplet | 1 | - | - | - | - | 4, 6, 7, 8, 9, 10 | - | - |
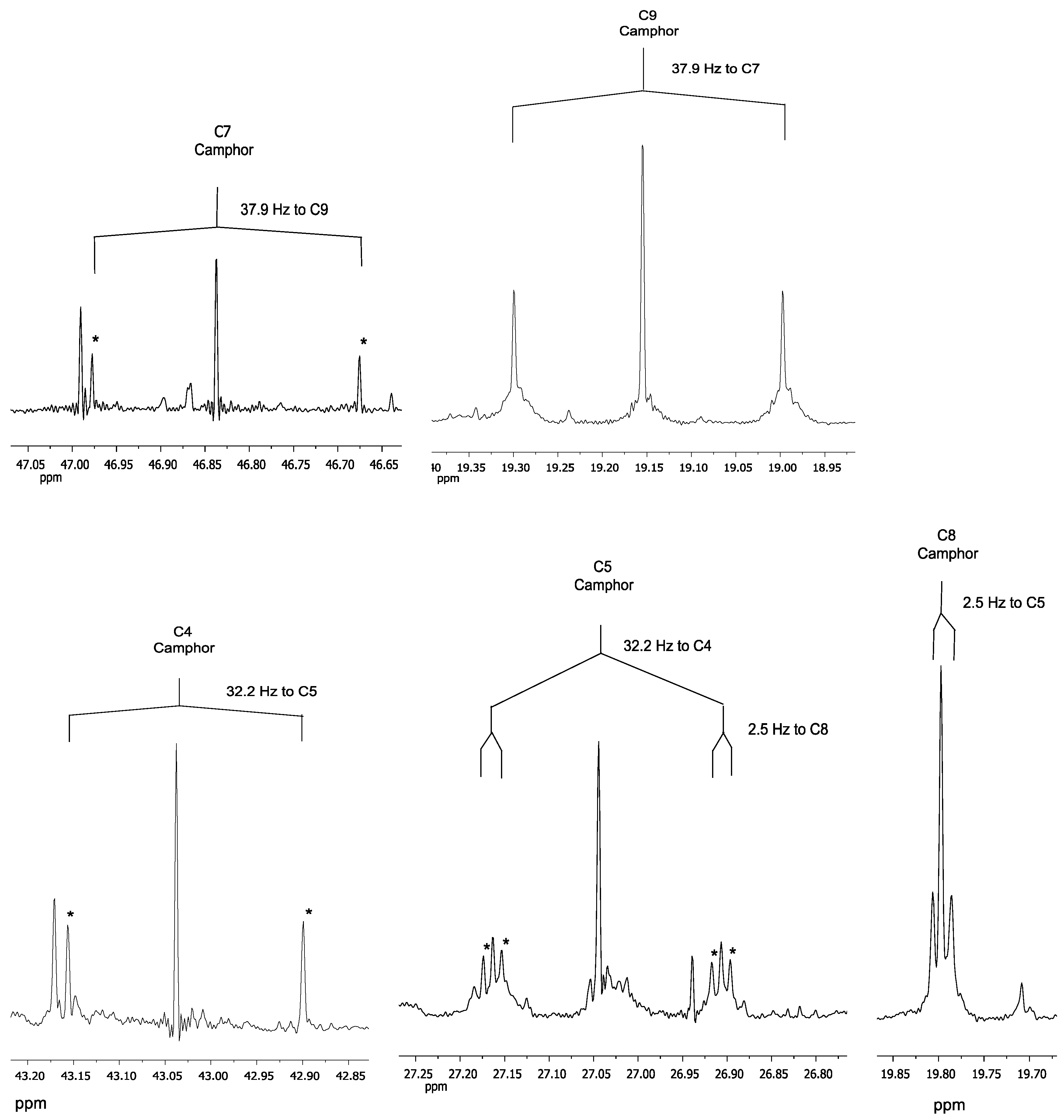
| Position Carbon Atoms | Chemical Shift [ppm] | 13C Coupling | 13C-13C Coupling Constant [Hz] |
|---|---|---|---|
| Camphor | |||
| 10 | 9.27 | to C1 | 41.1 |
| 9 | 19.15 | to C7 | 37.9 |
| 8 | 19.8 | to C5 | 2.5 |
| 5 | 27.04 | to C4 to C8 | 32.2 2.5 |
| 6 | 29.91 | to C2 | 2.3 |
| 4 | 43.04 | to C5 | 32.2 |
| 3 | 43.32 | to C2 | 34.3 |
| 7 | 46.84 | to C9 | 37.9 |
| 1 | 57.76 | to C10 | 41.1 |
| 2 | 220.06 | to C6 | 2.3 |
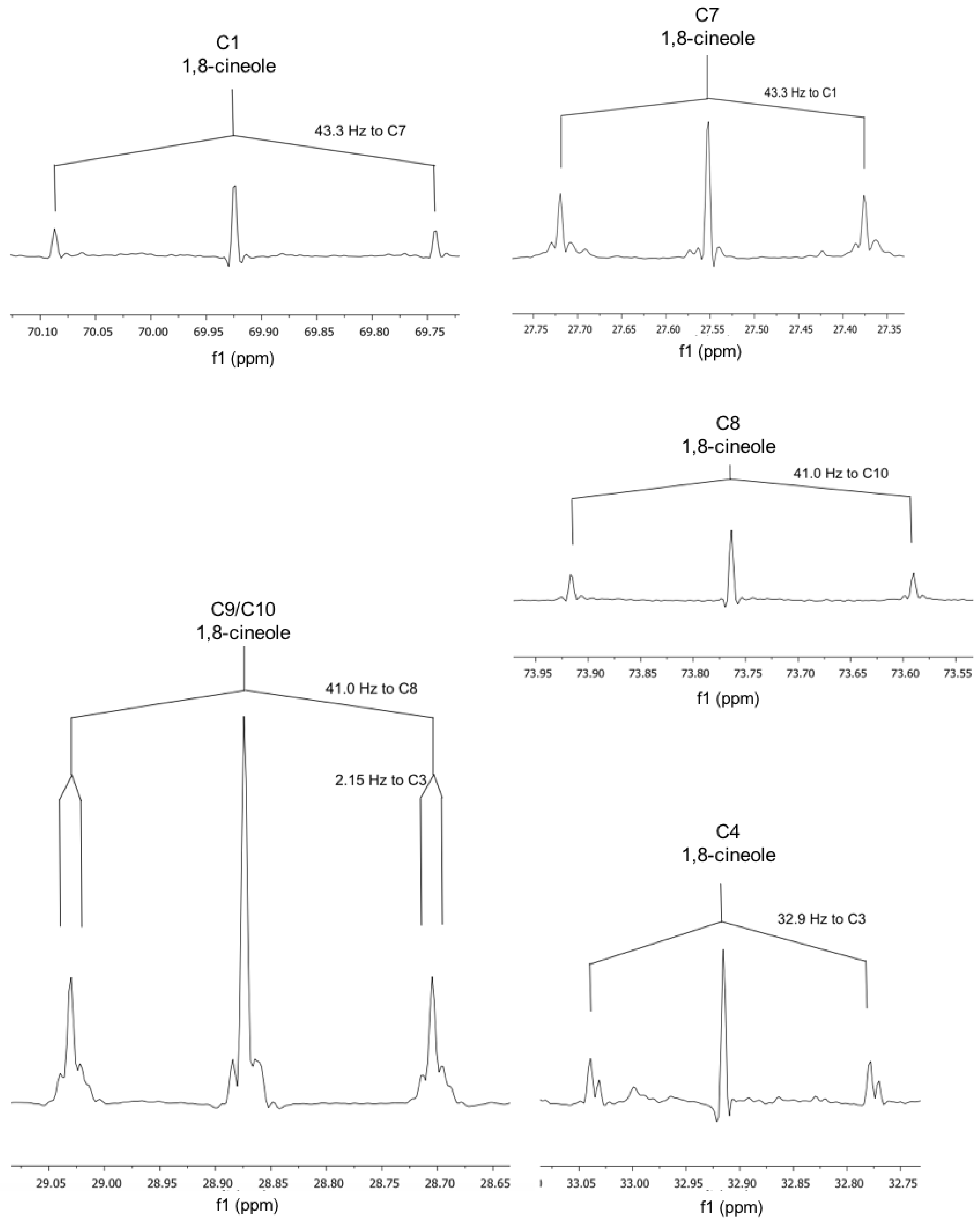
| 1,8-cineole | |||
| 3/5 | 22.8 | C3–C4 | 32.9 |
| 7 | 27.55 | to C1 | 43.3 |
| 9/10 | 28.87 | C10–C8 C9–C3/4 | 41.0 2.2 |
| 2/6 | 31.48 | - | ? |
| 4 | 32.91 | to C3 | 32.9 |
| 1 | 69.93 | to C7 | 43.3 |
| 8 | 73.76 | to C10 | 41 |
| Sample and Chase Period | α-Pinene rt: 5.9 | β-Pinene rt: 7.2 | Cineole rt: 9.2 | Limonene rt: 9.0 | Camphor rt: 14.3 |
|---|---|---|---|---|---|
| HMGR5 plants | - | - | - | - | - |
| 96 h | 8.15 | 6.77 | 53.78 | 2.09 | 16.83 |
| 96 h | 2.79 | 1.77 | 53.87 | 0.84 | 33.01 |
| 96 h | 5.33 | 3.42 | 49.91 | 1.79 | 30.49 |
| 168 h | 3.63 | 2.14 | 37.56 | 1.02 | 28.88 |
| 168 h | 2.37 | 1.58 | 52.94 | 0.65 | 31.96 |
| 240 h | 4.87 | 3.33 | 48.44 | 1.73 | 27.94 |
| 240 h | 5.02 | 2.98 | 50.99 | 1.27 | 30.42 |
| 240 h | 4.22 | 2.64 | 52.82 | 1.39 | 33.47 |
| Mean ± SD | 4.55 ± 1.80 | 3.08 ± 1.64 | 50.04 ± 5.40 | 1.35 ± 0.50 | 29.13 ± 5.32 |
| WT plants | - | - | - | - | - |
| 96 h | 4.8 | 3.05 | 49.83 | 1.54 | 29.23 |
| 264 h | 5.89 | 4.81 | 44.54 | 3.24 | 19.43 |
| 264 h | 5.54 | 4.5 | 42.8 | 3.84 | 12.13 |
| 264 h | 7.08 | 4.8 | 32.6 | 3.84 | 29.27 |
| Mean ± SD | 5.83 ± 0.95 | 4.29 ± 0.84 | 42.44 ± 7.21 | 3.12 ± 1.09 | 22.52 ± 8.33 |
| Sample and chase period | α-pinene rt: 5.9 | β-pinene rt: 7.2 | Cineole rt: 9.2 | Limonene rt: 9.0 | Camphor rt: 14.3 |
| Excess Values (%) | Ratio | ||||
|---|---|---|---|---|---|
| - | - | M+2 | M+3 | M+2/M+3 | M+2 |
| (M+2) + (M+3) | |||||
| Camphor | HMGR5 | - | - | - | - |
| - | 96 h | 0.30 | 0.17 | 1.76 | 0.64 |
| - | 96 h | 0.05 | 0.02 | 2.50 | 0.71 |
| - | 96 h | 0.20 | 0.11 | 1.82 | 0.65 |
| - | 168 h | 0.05 | 0.02 | 2.50 | 0.71 |
| - | 168 h | 0.03 | 0.02 | 1.50 | 0.60 |
| - | 240 h | 0.32 | 0.17 | 1.88 | 0.65 |
| - | 240 h | 0.29 | 0.13 | 2.23 | 0.69 |
| - | 240 h | 0.15 | 0.11 | 1.36 | 0.58 |
| - | WT | - | - | - | - |
| - | 96 h | 0.30 | 0.17 | 1.76 | 0.64 |
| - | 96 h | 0.05 | 0.02 | 2.50 | 0.71 |
| - | 96 h | 0.20 | 0.11 | 1.82 | 0.65 |
| - | 168 h | 0.05 | 0.02 | 2.50 | 0.71 |
| - | 168 h | 0.03 | 0.02 | 1.50 | 0.60 |
| Excess Values (%) | Ratio | ||||
|---|---|---|---|---|---|
| - | - | M+2 | M+3 | M+2/M+3 | M+2 |
| (M+2) + (M+3) | |||||
| 1,8-cineole | HMGR5 | - | - | - | - |
| - | 96 h | 0.27 | 0.16 | 1.69 | 0.63 |
| - | 96 h | 0.22 | 0.12 | 1.83 | 0.65 |
| - | 168 h | 0.07 | 0.04 | 1.75 | 0.64 |
| - | 240 h | 0.21 | 0.11 | 1.91 | 0.66 |
| - | 240 h | 0.32 | 0.14 | 1.79 | 0.70 |
| - | 240 h | 0.17 | 0.12 | 1.42 | 0.59 |
| - | WT | - | - | - | - |
| - | 96 h | 0.05 | 0.03 | 1.67 | 0.63 |
| - | 264 h | 0.53 | 0.27 | 1.96 | 0.66 |
| - | 264 h | 0.15 | 0.09 | 1.67 | 0.63 |
| 264 h | 0.87 | 0.48 | 1.81 | 0.64 | |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mendoza-Poudereux, I.; Kutzner, E.; Huber, C.; Segura, J.; Arrillaga, I.; Eisenreich, W. Dynamics of Monoterpene Formation in Spike Lavender Plants. Metabolites 2017, 7, 65. https://doi.org/10.3390/metabo7040065
Mendoza-Poudereux I, Kutzner E, Huber C, Segura J, Arrillaga I, Eisenreich W. Dynamics of Monoterpene Formation in Spike Lavender Plants. Metabolites. 2017; 7(4):65. https://doi.org/10.3390/metabo7040065
Chicago/Turabian StyleMendoza-Poudereux, Isabel, Erika Kutzner, Claudia Huber, Juan Segura, Isabel Arrillaga, and Wolfgang Eisenreich. 2017. "Dynamics of Monoterpene Formation in Spike Lavender Plants" Metabolites 7, no. 4: 65. https://doi.org/10.3390/metabo7040065
APA StyleMendoza-Poudereux, I., Kutzner, E., Huber, C., Segura, J., Arrillaga, I., & Eisenreich, W. (2017). Dynamics of Monoterpene Formation in Spike Lavender Plants. Metabolites, 7(4), 65. https://doi.org/10.3390/metabo7040065







