Metabolomics Analysis of Urine Samples from Children after Acetaminophen Overdose
Abstract
:1. Introduction
2. Results
3. Discussion
4. Materials and Methods
4.1. Ethical Approval
4.2. Subjects and Sample Collection
4.3. Metabolomics Methods
4.3.1. Chemicals
4.3.2. UPLC/QToF-MS Analysis
4.3.3. Nuclear Magnetic Resonance Spectroscopy Analysis
4.3.4. Pathway Analysis
4.3.5. Correlations Analysis
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Yoon, E.; Babar, A.; Choudhary, M.; Kutner, M.; Pyrsopoulos, N. Acetaminophen-induced hepatotoxicity: A comprehensive update. J. Clin. Transl. Hepatol. 2016, 4, 131–142. [Google Scholar] [PubMed]
- Larson, A.M. Acetaminophen hepatotoxicity. Clin. Liver Dis. 2007, 11, 525–548. [Google Scholar] [CrossRef] [PubMed]
- Bunchorntavakul, C.; Reddy, K.R. Acetaminophen-related hepatotoxicity. Clin. Liver Dis. 2013, 17, 587–607. [Google Scholar] [CrossRef] [PubMed]
- Alonso, E.M.; James, L.P.; Zhang, S.; Squires, R.H.; Pediatric acute liver failure study group. Acetaminophen adducts detected in serum of pediatric patients with acute liver failure. J. Pediatr. Gastroenterol. Nutr. 2015, 61, 102–107. [Google Scholar] [PubMed]
- Bhattacharyya, S.; Pence, L.; Beger, R.; Chaudhuri, S.; McCullough, S.; Yan, K.; Simpson, P.; Hennings, L.; Hinson, J.; James, L. Acylcarnitine profiles in acetaminophen toxicity in the mouse: Comparison to toxicity, metabolism and hepatocyte regeneration. Metabolites 2013, 3, 606–622. [Google Scholar] [CrossRef] [PubMed]
- Coen, M.; Lenz, E.M.; Nicholson, J.K.; Wilson, I.D.; Pognan, F.; Lindon, J.C. An integrated metabonomic investigation of acetaminophen toxicity in the mouse using NMR spectroscopy. Chem. Res. Toxicol. 2003, 16, 295–303. [Google Scholar] [CrossRef] [PubMed]
- Coen, M.; Ruepp, S.U.; Lindon, J.C.; Nicholson, J.K.; Pognan, F.; Lenz, E.M.; Wilson, I.D. Integrated application of transcriptomics and metabonomics yields new insight into the toxicity due to paracetamol in the mouse. J. Pharm. Biomed. Anal. 2004, 35, 93–105. [Google Scholar] [CrossRef] [PubMed]
- Fukuhara, K.; Ohno, A.; Ando, Y.; Yamoto, T.; Okuda, H. A 1H NMR-based metabolomics approach for mechanistic insight into acetaminophen-induced hepatotoxicity. Drug Metab. Pharmacokinet. 2011, 26, 399–406. [Google Scholar] [CrossRef] [PubMed]
- Ghauri, F.Y.; McLean, A.E.; Beales, D.; Wilson, I.D.; Nicholson, J.K. Induction of 5-oxoprolinuria in the rat following chronic feeding with N-acetyl 4-aminophenol (paracetamol). Biochem. Pharmacol. 1993, 46, 953–957. [Google Scholar] [CrossRef]
- James, L.; Yan, K.; Pence, L.; Simpson, P.; Bhattacharyya, S.; Gill, P.; Letzig, L.; Kearns, G.; Beger, R. Comparison of bile acids and acetaminophen protein adducts in children and adolescents with acetaminophen toxicity. PLoS ONE 2015, 10, e0131010. [Google Scholar] [CrossRef] [PubMed]
- Kumar, B.S.; Chung, B.C.; Kwon, O.S.; Jung, B.H. Discovery of common urinary biomarkers for hepatotoxicity induced by carbon tetrachloride, acetaminophen and methotrexate by mass spectrometry-based metabolomics. J. Appl. Toxicol. 2012, 32, 505–520. [Google Scholar] [CrossRef] [PubMed]
- Kyriakides, M.; Maitre, L.; Stamper, B.D.; Mohar, I.; Kavanagh, T.J.; Foster, J.; Wilson, I.D.; Holmes, E.; Nelson, S.D.; Coen, M. Comparative metabonomic analysis of hepatotoxicity induced by acetaminophen and its less toxic meta-isomer. Arch. Toxicol. 2016, 90, 3073–3085. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Ando, Y.; Ahlbory-Dieker, D.; Schnackenberg, L.K.; Yang, X.; Greenhaw, J.; Pence, L.; Qian, F.; Salminen, W.; Mendrick, D.L.; et al. Systems biology investigation to discover metabolic biomarkers of acetaminophen-induced hepatic injury using integrated transcriptomics and metabolomics. J. Mol. Biomark. Diagn. 2013. [Google Scholar] [CrossRef]
- Sun, J.; Schnackenberg, L.K.; Holland, R.D.; Schmitt, T.C.; Cantor, G.H.; Dragan, Y.P.; Beger, R.D. Metabonomics evaluation of urine from rats given acute and chronic doses of acetaminophen using NMR and UPLC/MS. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2008, 871, 328–340. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, T.; Tomizawa, K.; Fujikawa, M.; Sato, Y.; Yamada, H.; Horii, I. Evaluation of human hepatocyte chimeric mice as a model for toxicological investigation using panomic approaches—Effect of acetaminophen on the expression profiles of proteins and endogenous metabolites in liver, plasma and urine. J. Toxicol. Sci. 2007, 32, 205–215. [Google Scholar] [CrossRef] [PubMed]
- Yamazaki, M.; Miyake, M.; Sato, H.; Masutomi, N.; Tsutsui, N.; Adam, K.P.; Alexander, D.C.; Lawton, K.A.; Milburn, M.V.; Ryals, J.A.; et al. Perturbation of bile acid homeostasis is an early pathogenesis event of drug induced liver injury in rats. Toxicol. Appl. Pharmacol. 2013, 268, 79–89. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.W.; Ryu, S.H.; Kim, S.; Lee, H.W.; Lim, M.S.; Seong, S.J.; Kim, S.; Yoon, Y.R.; Kim, K.B. Pattern recognition analysis for hepatotoxicity induced by acetaminophen using plasma and urinary 1H NMR-based metabolomics in humans. Anal. Chem. 2013, 85, 11326–11334. [Google Scholar] [CrossRef] [PubMed]
- McGill, M.R.; Jaeschke, H. Mechanistic biomarkers in acetaminophen-induced hepatotoxicity and acute liver failure: From preclinical models to patients. Expert Opin. Drug Metab. Toxicol. 2014, 10, 1005–1017. [Google Scholar] [CrossRef] [PubMed]
- McGill, M.R.; Sharpe, M.R.; Williams, C.D.; Taha, M.; Curry, S.C.; Jaeschke, H. The mechanism underlying acetaminophen-induced hepatotoxicity in humans and mice involves mitochondrial damage and nuclear DNA fragmentation. J. Clin. Investig. 2012, 122, 1574–1583. [Google Scholar] [CrossRef] [PubMed]
- McGill, M.R.; Staggs, V.S.; Sharpe, M.R.; Lee, W.M.; Jaeschke, H.; Acute liver failure study group. Serum mitochondrial biomarkers and damage-associated molecular patterns are higher in acetaminophen overdose patients with poor outcome. Hepatology 2014, 60, 1336–1345. [Google Scholar] [CrossRef] [PubMed]
- Beger, R.D.; Bhattacharyya, S.; Yang, X.; Gill, P.S.; Schnackenberg, L.K.; Sun, J.; James, L.P. Translational biomarkers of acetaminophen-induced acute liver injury. Arch. Toxicol. 2015, 89, 1497–1522. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharyya, S.; Yan, K.; Pence, L.; Simpson, P.M.; Gill, P.; Letzig, L.G.; Beger, R.D.; Sullivan, J.E.; Kearns, G.L.; Reed, M.D.; et al. Targeted liquid chromatography-mass spectrometry analysis of serum acylcarnitines in acetaminophen toxicity in children. Biomark. Med. 2014, 8, 147–159. [Google Scholar] [CrossRef] [PubMed]
- Banfi, G.; Colombini, A.; Lombardi, G.; Lubkowska, A. Metabolic markers in sports medicine. Adv. Clin. Chem. 2012, 56, 1–54. [Google Scholar] [PubMed]
- Sookoian, S.; Pirola, C.J. Liver enzymes, metabolomics and genome-wide association studies: From systems biology to the personalized medicine. World J. Gastroenterol. 2015, 21, 711–725. [Google Scholar] [CrossRef] [PubMed]
- Zamora, S.; Adams, C.; Butzner, J.D.; Machida, H.; Scott, R.B. Elevated aminotransferase activity as an indication of muscular dystrophy: Case reports and review of the literature. Can. J. Gastroenterol. 1996, 10, 389–393. [Google Scholar] [CrossRef] [PubMed]
- Coen, M. Metabolic phenotyping applied to pre-clinical and clinical studies of acetaminophen metabolism and hepatotoxicity. Drug Metab. Rev. 2015, 47, 29–44. [Google Scholar] [CrossRef] [PubMed]
- Dabos, K.J.; Whalen, H.R.; Newsome, P.N.; Parkinson, J.A.; Henderson, N.C.; Sadler, I.H.; Hayes, P.C.; Plevris, J.N. Impaired gluconeogenesis in a porcine model of paracetamol induced acute liver failure. World J. Gastroenterol. 2011, 17, 1457–1461. [Google Scholar] [CrossRef] [PubMed]
- Jones, J.G.; Solomon, M.A.; Cole, S.M.; Sherry, A.D.; Malloy, C.R. An integrated 2H and 13C NMR study of gluconeogenesis and TCA cycle flux in humans. Am. J. Physiol. Endocrinol. Metab. 2001, 281, E848–E856. [Google Scholar] [PubMed]
- Winnike, J.H.; Li, Z.; Wright, F.A.; Macdonald, J.M.; O’Connell, T.M.; Watkins, P.B. Use of pharmaco-metabonomics for early prediction of acetaminophen-induced hepatotoxicity in humans. Clin. Pharmacol. Ther. 2010, 88, 45–51. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Krausz, K.W.; Shah, Y.M.; Idle, J.R.; Gonzalez, F.J. Serum metabolomics reveals irreversible inhibition of fatty acid beta-oxidation through the suppression of PPARalpha activation as a contributing mechanism of acetaminophen-induced hepatotoxicity. Chem. Res. Toxicol. 2009, 22, 699–707. [Google Scholar] [CrossRef] [PubMed]
- Duranti, G.; Boenzi, S.; Rizzo, C.; Rava, L.; Di Ciommo, V.; Carrozzo, R.; Meschini, M.C.; Johnson, D.W.; Dionisi-Vici, C. Urine acylcarnitine analysis by ESI-MS/MS: A new tool for the diagnosis of peroxisomal biogenesis disorders. Clin. Chim. Acta 2008, 398, 86–89. [Google Scholar] [CrossRef] [PubMed]
- Moder, M.; Kiessling, A.; Loster, H.; Bruggemann, L. The pattern of urinary acylcarnitines determined by electrospray mass spectrometry: A new tool in the diagnosis of diabetes mellitus. Anal. Bioanal. Chem. 2003, 375, 200–210. [Google Scholar] [CrossRef] [PubMed]
- Xia, J.; Wishart, D.S. Using MetaboAnalyst 3.0 for comprehensive metabolomics data analysis. Curr. Protoc. Bioinform. 2016, 55. [Google Scholar] [CrossRef]
- Xia, J.; Sinelnikov, I.; Han, B.; Wishart, D.S. MetaboAnalyst 3.0—Making metabolomics more meaningful. Nucl. Acids Res. 2015, 43, W251–W257. [Google Scholar] [CrossRef] [PubMed]
- Xia, J.; Wishart, D.S. Web-based inference of biological patterns, functions and pathways from metabolomic data using MetaboAnalyst. Nat. Protoc. 2011, 6, 743–760. [Google Scholar] [CrossRef] [PubMed]
- Xia, J.; Wishart, D.S. Metabolomic data processing, analysis, and interpretation using MetaboAnalyst. Curr. Protoc. Bioinform. 2011, 14.10.1–14.10.48. [Google Scholar]
- Xia, J.; Psychogios, N.; Young, N.; Wishart, D.S. MetaboAnalyst: A web server for metabolomic data analysis and interpretation. Nucl. Acids Res. 2009, 37, W652–W660. [Google Scholar] [CrossRef] [PubMed]
- Xia, J.; Broadhurst, D.; Wilson, M.; Wishart, D. Translational biomarker discovery in clinical metabolomics: An introductory tutorial. Metabolomics 2013, 9, 280–299. [Google Scholar] [CrossRef] [PubMed]
- Xia, J.; Wishart, D.S. MetPA: A web-based metabolomics tool for pathway analysis and visualization. Bioinformatics 2010, 26, 2342–2344. [Google Scholar] [CrossRef] [PubMed]
- Benjamini, Y.; Hochberg, Y. Controlling the false discovery rate: A practical and powerful approach to multiple testing. J. R. Stat. Soc. Ser. B 1995, 57, 289–300. [Google Scholar]
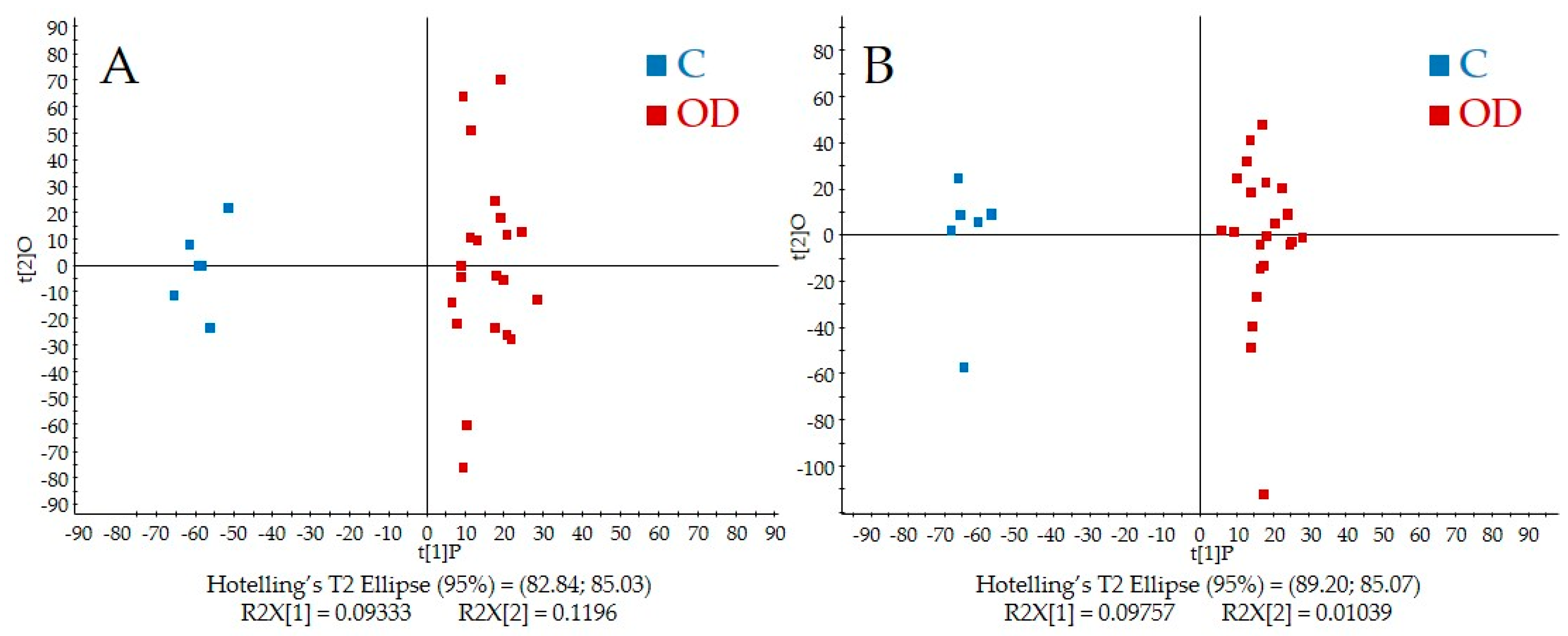
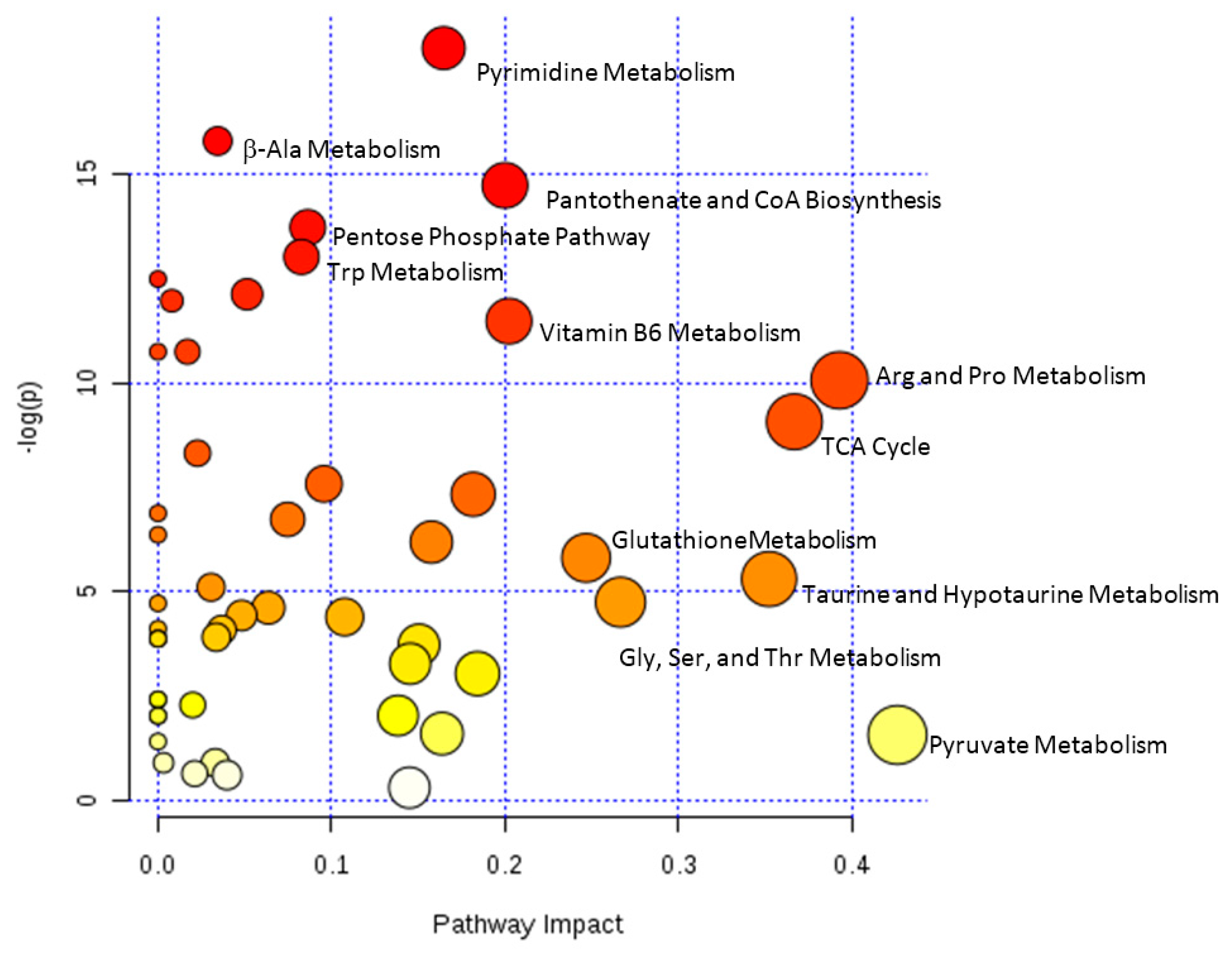
| Variables | Control N = 6 | Overdose N = 13 |
|---|---|---|
| Age (years) | 14.08 | 13.91 |
| PEAK ALT (IU/L) | NA | 2050 (106, 6072) |
| Peak Adduct (nmol/mL) | 0.00 (0.00, 0.00) | 1.48 (0.20, 6.69) |
| Metabolite | Platform | ALT | APAP-Protein Adducts |
|---|---|---|---|
| 2-Oxoarginine a | LCMS POS, 0.72 min, m/z 174.09 | 0.6192, p = 2.40 × 10−2 | 0.4318 |
| Ascorbic acid a | LCMS POS, 0.72 min, m/z 177.04 | 0.4361 | 0.8446, p = 2.81 × 10−4 |
| Ascorbic acid a | LCMS NEG, 0.72 min, m/z 175.02 | 0.3889 | 0.8159, p = 6.72 × 10−4 |
| Alanine | NMR, 1.47, 3.78 ppm | 0.2600 | 0.6792, p = 1.073 × 10−2 |
| Choline | NMR, 3.19, 3.51, 4.06 ppm | 0.1789 | 0.5993, p = 3.04 × 10−2 |
| Citrulline a | LCMS POS, 0.67 min, m/z 176.10 | −0.3810 | −0.6676, p = 1.27 × 10−2 |
| Cresol a | LCMS NEG, 3.77 min, m/z 107.05 | −0.5642, p = 4.46 × 10−2 | −0.4348 |
| Fructose | NMR, 3.55–4.11 ppm | 0.3900 | 0.8170, p = 6.51 × 10−4 |
| Glucose | NMR, 3.23–3.89, 4.64 ppm | 0.2778 | 0.6367, p = 1.93 × 10−2 |
| Hippurate a | LCMS NEG, 3.32 min, m/z 178.05 | 0.4858 | 0.8561, p = 1.88 × 10−4 |
| Hippurate | NMR, 3.96, 7.54, 7.63, 7.82 ppm | 0.4116 | 0.8350, p = 3.82 × 10−4 |
| Hydroxybutyrylcarnitine a | LCMS POS, 1.05 min, m/z 248.15 | −0.5674, p = 4.46 × 10−2 | −0.4900 |
| Indoxyl a | LCMS POS, 3.30 min, m/z 134.06 | 0.7062, p = 6.98 × 10−3 | 0.8230, p = 5.49 × 10 −4 |
| Lactate | NMR, 1.32, 4.11 ppm | - | 0.7906, p = 1.29 × 10−3 |
| Proline a | LCMS POS, 0.83 min, m/z 116.07 | −0.5733, p = 4.05 × 10−2 | −0.5376 |
| Propylene glycol | NMR, 1.13, 3.44, 3.54, 3.88 ppm | 0.4025 | 0.8263, p = 4.99 × 10−4 |
| Pyruvate | NMR, 2.36 ppm | 0.3680 | 0.7134, p = 6.18 × 10−3 |
| Taurocholic acid isomer b | LCMS NEG, 4.83 min, m/z 514.28 | 0.4259 | 0.6891, p = 9.18 × 10−3 |
| Trimethylamine N-oxide | NMR, 3.25 ppm | 0.5807, p = 3.74 × 10−2 | 0.3733 |
| Uracil a | LCMS POS, 0.78 min, m/z 113.03 | - | 0.6349, p = 1.97 × 10−2 |
| Uric acid a | LCMS NEG, 0.83 min, m/z 167.02 | - | 0.5636, p = 4.49 × 10−2 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schnackenberg, L.K.; Sun, J.; Bhattacharyya, S.; Gill, P.; James, L.P.; Beger, R.D. Metabolomics Analysis of Urine Samples from Children after Acetaminophen Overdose. Metabolites 2017, 7, 46. https://doi.org/10.3390/metabo7030046
Schnackenberg LK, Sun J, Bhattacharyya S, Gill P, James LP, Beger RD. Metabolomics Analysis of Urine Samples from Children after Acetaminophen Overdose. Metabolites. 2017; 7(3):46. https://doi.org/10.3390/metabo7030046
Chicago/Turabian StyleSchnackenberg, Laura K., Jinchun Sun, Sudeepa Bhattacharyya, Pritmohinder Gill, Laura P. James, and Richard D. Beger. 2017. "Metabolomics Analysis of Urine Samples from Children after Acetaminophen Overdose" Metabolites 7, no. 3: 46. https://doi.org/10.3390/metabo7030046
APA StyleSchnackenberg, L. K., Sun, J., Bhattacharyya, S., Gill, P., James, L. P., & Beger, R. D. (2017). Metabolomics Analysis of Urine Samples from Children after Acetaminophen Overdose. Metabolites, 7(3), 46. https://doi.org/10.3390/metabo7030046






