Micropreconcentrator in LTCC Technology with Mass Spectrometry for the Detection of Acetone in Healthy and Type-1 Diabetes Mellitus Patient Breath
Abstract
:1. Introduction
2. Experimental Section
2.1. Reference Gas and Breath Samples
2.1.1. Breath Measurements with Volunteers
2.1.2. Breath Measurements with Diabetes Patients
2.2. Apparatus
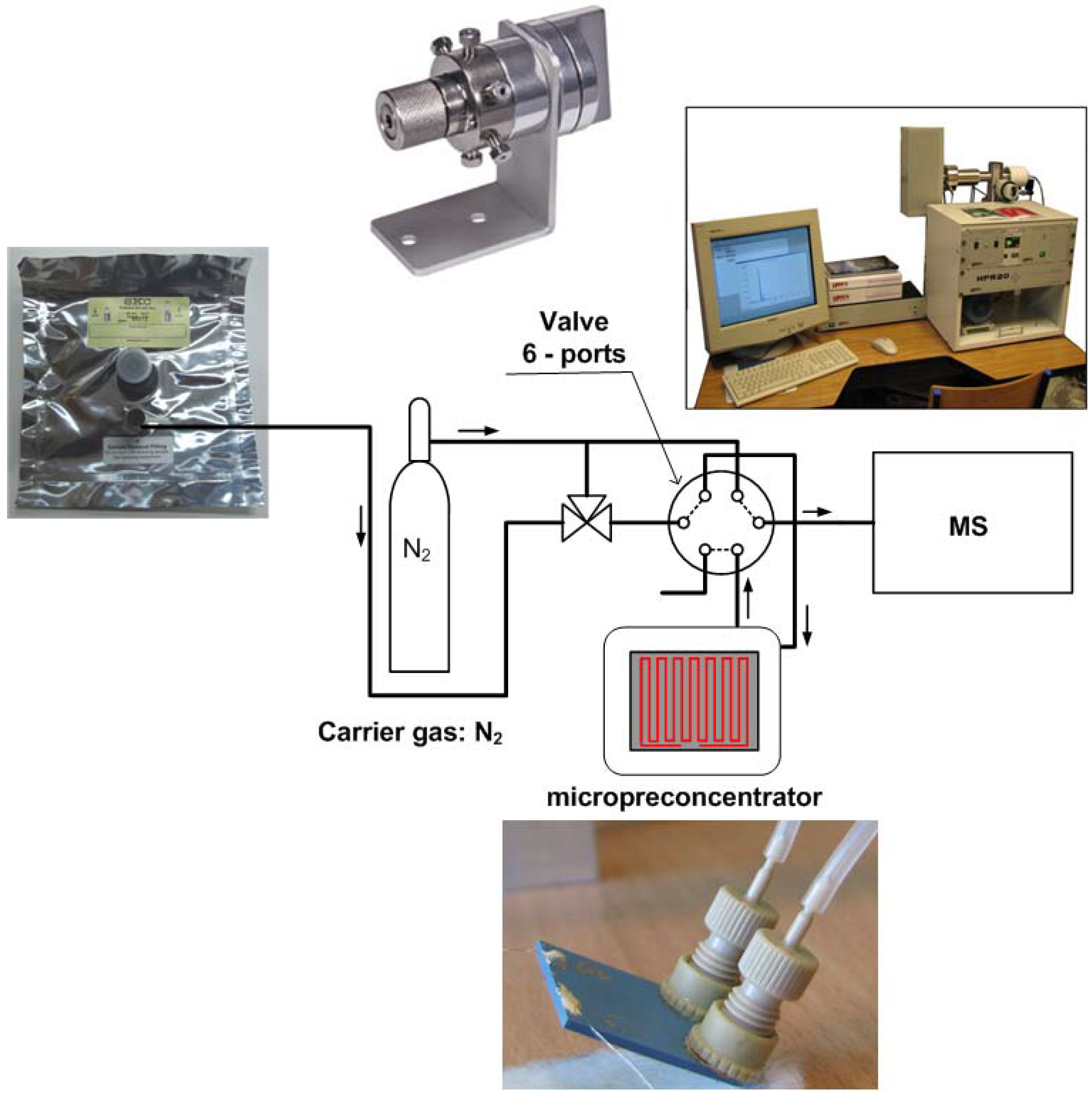
2.3. Experimental Methods and Conditions
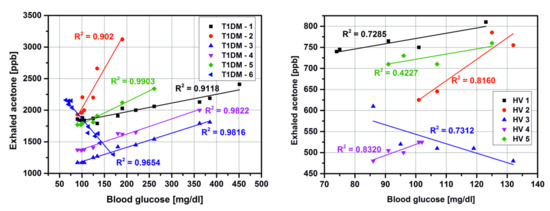
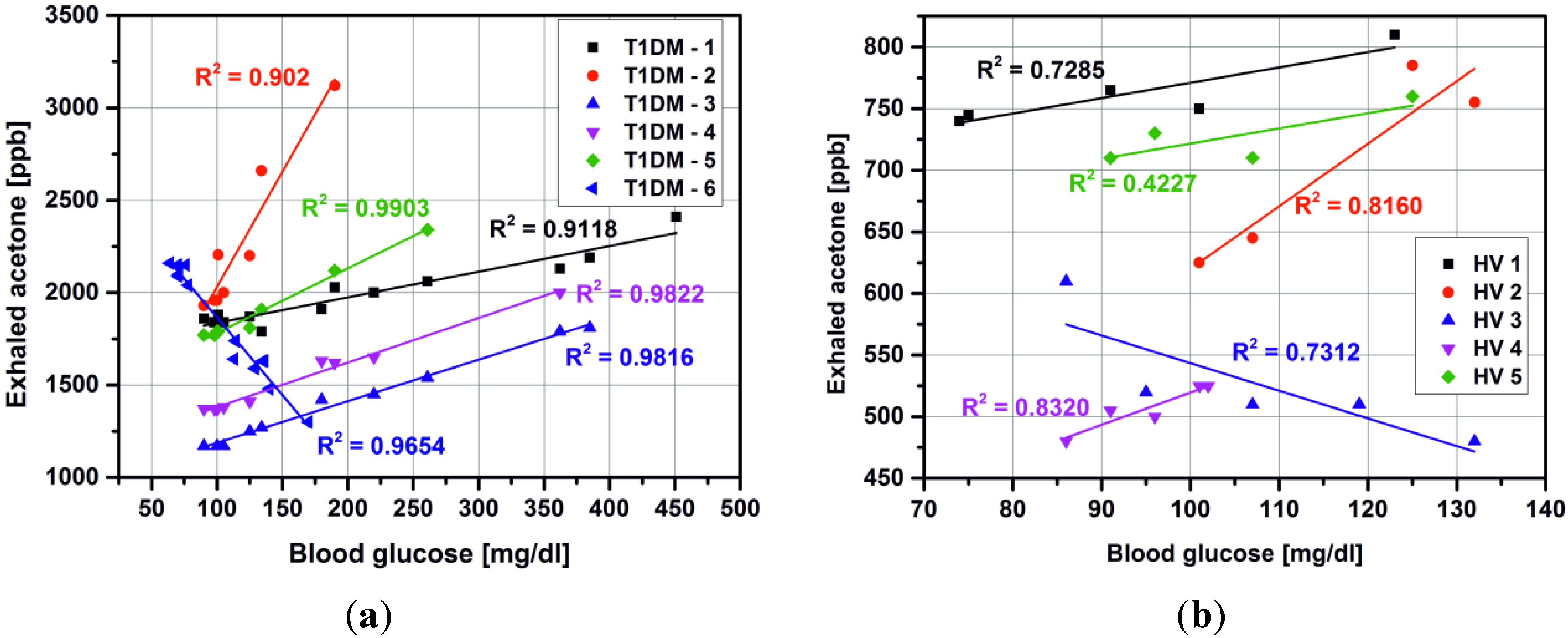
3. Results and Discussion
3.1. Detection of Acetone in Breath without Preconcentration
| Subject number | Group | Sex a | Age | Years of diabetes | Breath Acetone (ppmv) | Blood glucose (mg/dl) |
|---|---|---|---|---|---|---|
| 1 b | Healthy volunteer | M | 43 | X | 0.49 | 91 |
| 0.56 | 111 | |||||
| 2 c | F | 30 | 0.57 | 86 | ||
| 0.47 | 132 | |||||
| 3 | M | 28 | 0.63 | 101 | ||
| 0.83 | 137 | |||||
| 4 | M | 41 | 0.74 | 74 | ||
| 0.81 | 123 | |||||
| 5 | F | 32 | 0.65 | 103 | ||
| 0.73 | 150 | |||||
| 6 | M | 28 | 0.48 | 86 | ||
| 0.69 | 146 | |||||
| 7 | F | 30 | 0.59 | 84 | ||
| 0.71 | 136 | |||||
| 8 | M | 27 | 0.63 | 102 | ||
| 0.72 | 145 | |||||
| 9 | F | 39 | 0.45 | 86 | ||
| 0.80 | 135 | |||||
| 10 | F | 26 | 0.48 | 90 | ||
| 0.79 | 140 | |||||
| 11 | M | 29 | 0.45 | 86 | ||
| 0.73 | 151 | |||||
| 12 | Patients with diabetes type I | F | 33 | 15 | 1.52 | 60 |
| 1.70 | 168 | |||||
| 13 | M | 23 | 2 | 1.62 | 70 | |
| 3.12 | 196 | |||||
| 14 | M | 22 | 2 | 1.80 | 100 | |
| 2.75 | 231 | |||||
| 15 d | F | 29 | 15 | 2.16 | 64 | |
| 1.30 | 170 | |||||
| 16 | M | 23 | 13 | 1.51 | 174 | |
| 3.12 | 367 | |||||
| 17 | F | 25 | 4 | 1.11 | 65 | |
| 1.79 | 372 | |||||
| 18 | F | 21 | 5 | 1.55 | 82 | |
| 3.09 | 324 | |||||
| 19 | F | 40 | 10 | 1.63 | 70 | |
| 2.70 | 350 | |||||
| 20 | F | 23 | 3 | 1.70 | 75 | |
| 1.85 | 127 | |||||
| 21 | M | 20 | 1 | 1.87 | 181 | |
| 3.12 | 270 | |||||
| 22 | M | 24 | 8 | 1.78 | 63 | |
| 1.87 | 165 | |||||
| 23 | F | 22 | 2 | 1.86 | 90 | |
| 2.41 | 451 | |||||
| 24 | F | 55 | 15 | 1.61 | 70 | |
| 2.98 | 257 | |||||
| 25 | M | 35 | 21 | 1.65 | 62 | |
| 3.09 | 389 |
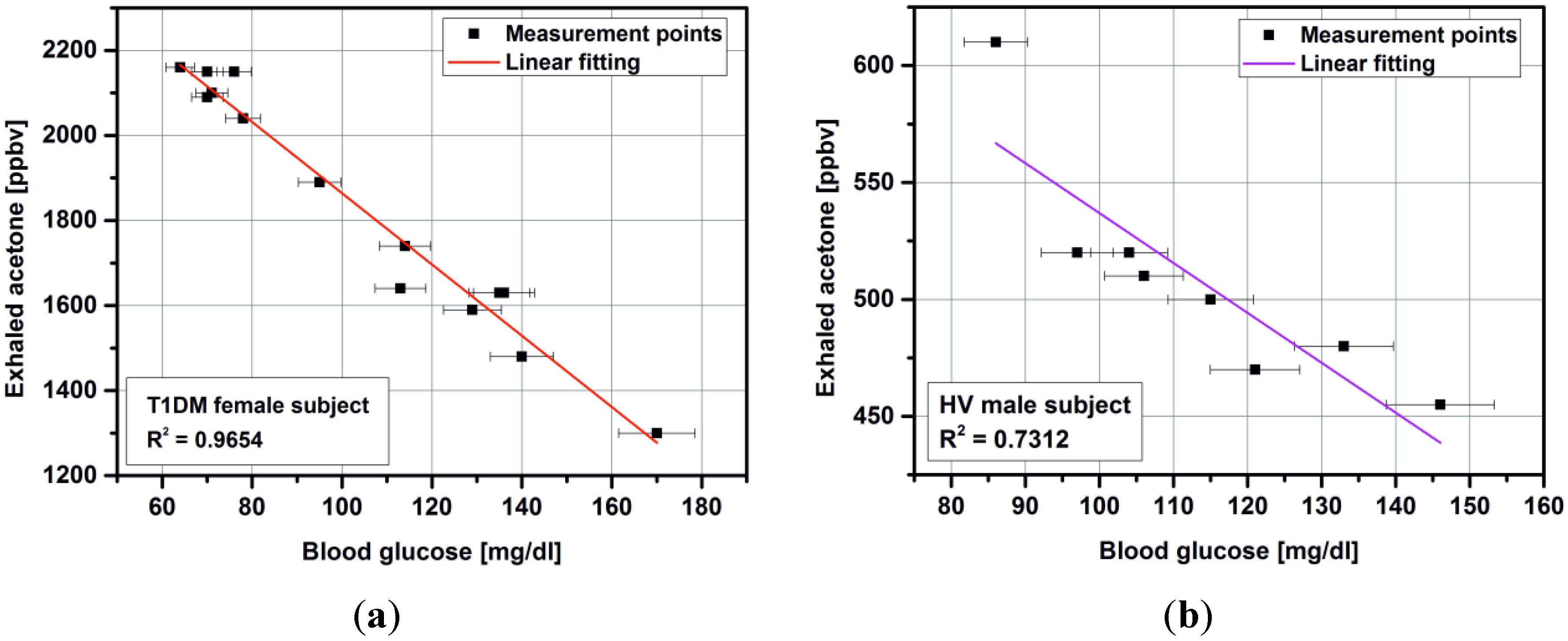
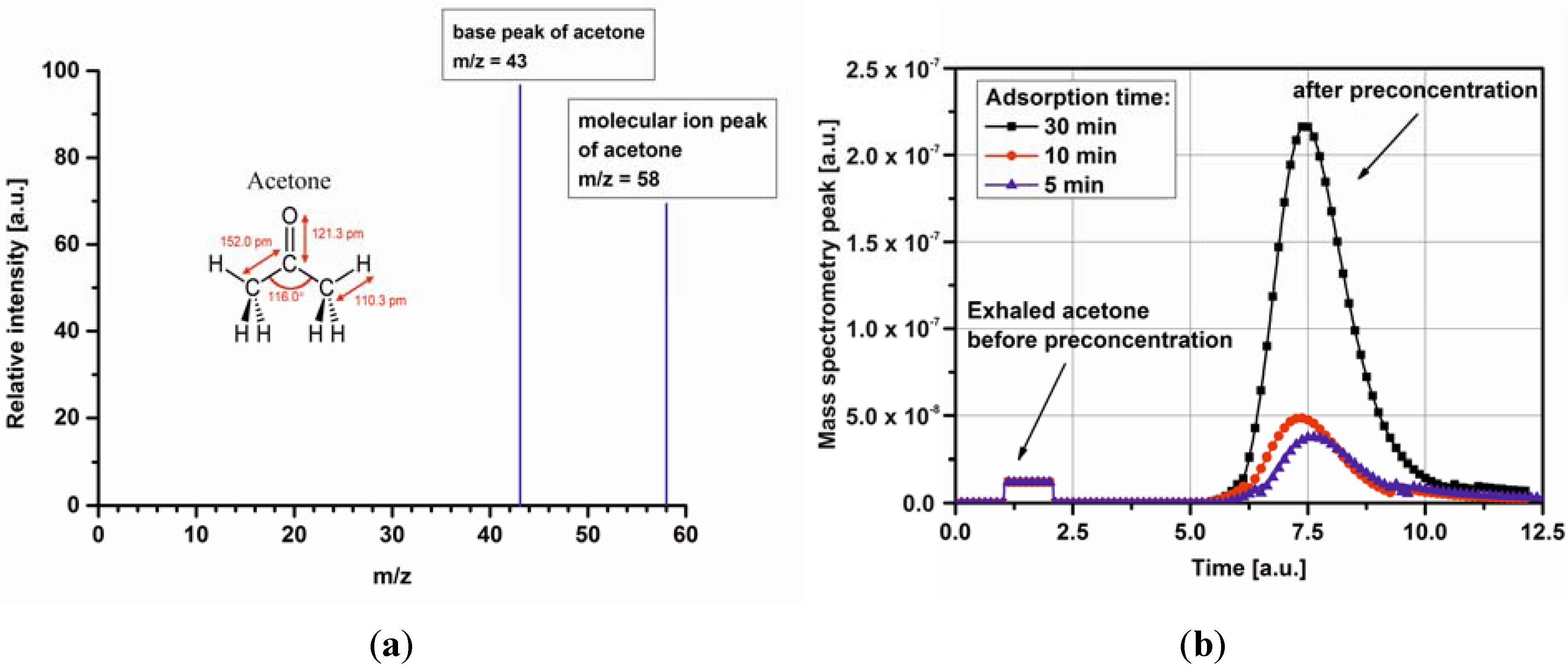
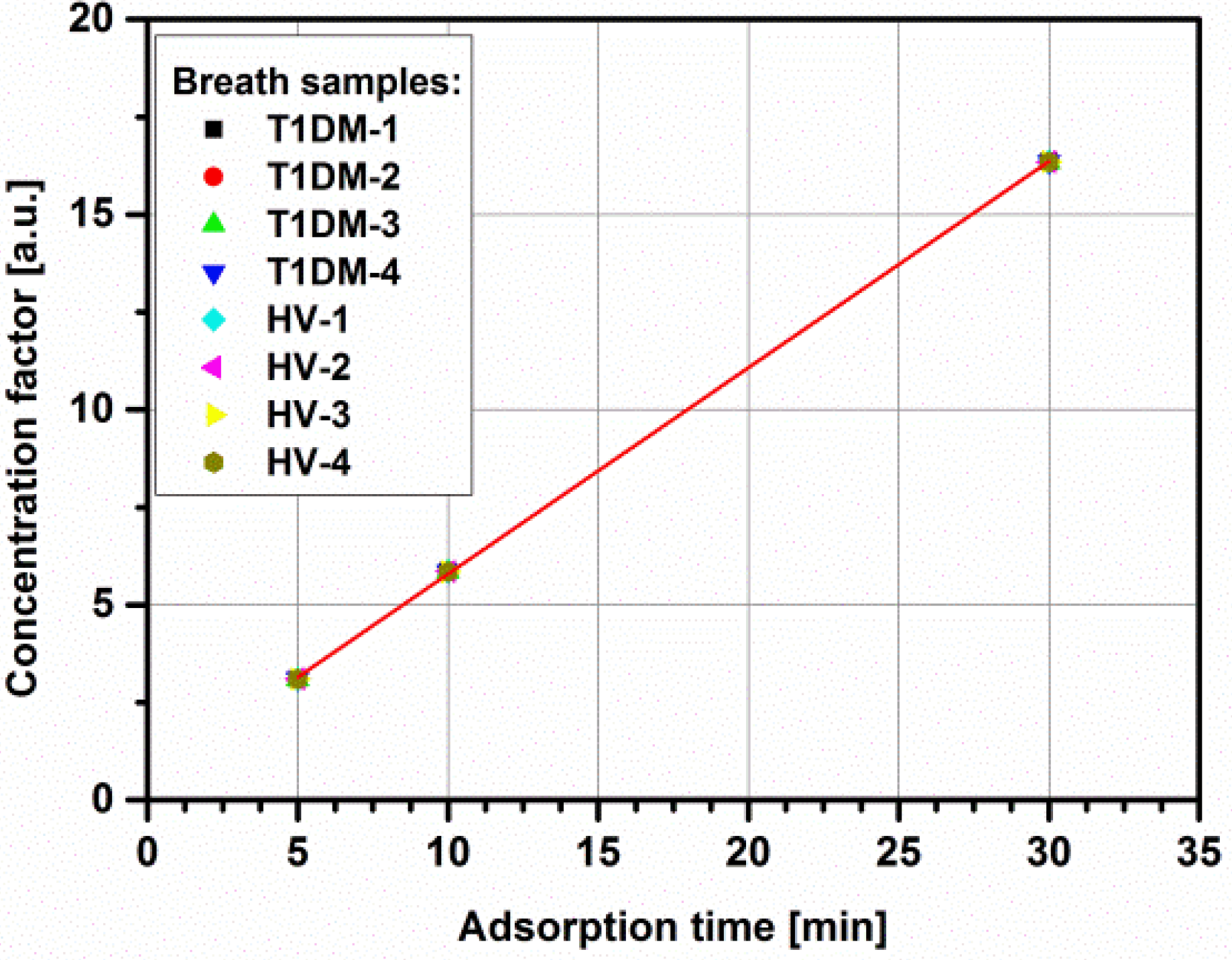
3.2. Impact of Micropreconcentrator on Breath Acetone Detection
4. Conclusions and Perspectives
Acknowledgments
Conflicts of Interest
References
- Tassopoulos, C.; Barnett, D.; Russell, F.T. Breath-acetone and blood-sugar measurements in diabetes. Lancet 1969, 239, 1282–1286. [Google Scholar] [CrossRef]
- Guo, D.; Zhang, D.; Zhang, L.; Lu, G. Non-invasive blood glucose monitoring for diabetics by means of breath signal analysis. Sens. Actuators 2012, 173, 106–113. [Google Scholar] [CrossRef]
- Miekisch, W.; Schubert, J.K.; Noeldge-Schomburg, G.F.E. Diagnostic potential of breath analysis: Focus on volatile organic compounds. Clin. Chim. Acta 2004, 347, 25–39. [Google Scholar] [CrossRef]
- Fleischer, M.; Simon, E.; Rumpel, E.; Ulmer, H.; Harbeck, M.; Wandel, M.; Fietzek, C.; Weimar, U.; Meixner, H. Detection of volatile compounds correlated to human diseases through breath analysis with chemical sensors. Sens. Actuators 2002, 83, 245–249. [Google Scholar] [CrossRef]
- Zhang, Q.; Wang, P.; Li, J.; Gao, X. Diagnosis of diabetes by image detection of breath using gas-sensitive laps. Biosens. Bioelectron. 2000, 15, 249–256. [Google Scholar] [CrossRef]
- Amann, A.; Smith, D. Breath Analysis for Clinical Diagnosis and Therapeutic Monitoring; World Scientific: Singapore, Singapore, 2005; pp. 305–316. [Google Scholar]
- Wang, C.J.; Mbi, A.; Shepherd, M. A study on breath acetone in diabetic patients using a cavity ringdown breath analyzer: Exploring correlations of breath acetone with blood glucose and glycohemoglobin A1C. IEEE Sens. J. 2010, 10, 43–63. [Google Scholar]
- Kinoyama, M.; Nitta, H.; Watanabe, A.; Ueda, H. Acetone and isoprene concentrations in exhaled breath in healthy subjects. J. Health Sci. 2008, 54, 471–477. [Google Scholar] [CrossRef]
- Worrall, A.D.; Bernstein, J.A.; Angelopoulos, A.P. Portable method of measuring gaseous acetone concentrations. Talanta 2013, 112, 26–30. [Google Scholar] [CrossRef]
- Righettoni, M.; Tricoli, A.; Gass, S.; Schmid, A.; Amann, A.; Pratsinis, S.E. Breath acetone monitoring by portable Si:WO3 gas sensors. Anal. Chim. Acta 2012, 738, 69–75. [Google Scholar] [CrossRef]
- Smith, D.; Spanel, P.; Fryer, A.A.; Hanna, F.; Ferns, G.A. Can volatile compounds in exhaled breath be used to monitor control in diabetes mellitus? J. Breath Res. 2001. [Google Scholar] [CrossRef]
- Minh, T.D.C.; Blake, D.R.; Galassetti, P.R. The clinical potential of exhaled breath analysis for diabetes mellitus. Diabetes Res. Clin. Pract. 2012, 97, 195–205. [Google Scholar] [CrossRef]
- Mohamed, E.; Linder, R.; Perriello, G.; di Daniele, N.; Poeppl, S.; de Lorenzo, A. Predicting type-2 diabetes using an electronic nose-based artificial neural network analysis. Diabetes Nutr. Metabol. 2002, 15, 215–221. [Google Scholar]
- Phillips, M.; Herrera, J.; Krishnan, S.; Zain, M.; Greenberg, J.; Cataneo, R.N. Variation in volatile organic compounds in the breath of normal humans. J. Chromatogr. B 1999, 729, 75–88. [Google Scholar] [CrossRef]
- Musa-veloso, K.; Likhodii, S.S.; Rarama, E.; Benoit, S.; Liu, Y.M.C.; Chartrand, D.; Curits, R.; Carmant, L.; Lortie, A.; Comeau, F.J.E.; et al. Breath acetone predicts plasma ketone bodies in children with epilepsy on a ketogenic diet. Nutrition 2006, 22, 1–8. [Google Scholar] [CrossRef]
- Massick, S.M.; Vakhtin, A.B. Breath acetone detection optical methods in the life science. Proc. SPIE 2006. [Google Scholar] [CrossRef]
- Moser, B.; Bodrogi, F.; Eibl, G.; Lechner, M.; Rieder, J.; Lirk, P. Mass spectrometric profile of exhaled breath-field study by PTR-MS. Respir. Physiol. Neurobiol. 2005, 145, 295–300. [Google Scholar] [CrossRef]
- Koyanagi, G.K.; Kapishon, V.; Blagojevic, V.; Boheme, D.K. Monitoring hydrogen sulfide in simulated breath f anesthetized subjects. Int. J. Mass Spectrom. 2013, 354–355, 139–143. [Google Scholar] [CrossRef]
- Fan, G.-T.; Yang, C.-L.; Lin, C.-H.; Chen, C.-C.; Shih, C.-H. Applications of Hadamard transform-gas chromatography/mass spectrometry to the detection of acetone in healthy human and diabetes mellitus patient breath. Talata 2014, 120, 386–390. [Google Scholar]
- Diskin, A.M.; Spanel, P.; Smith, D. Time variation of ammonia, acetone, isoprene and ethanol in breath: A quantitative SIFT-MS study over 30 days. Physiol. Meas. 2003, 24, 107–120. [Google Scholar] [CrossRef]
- Rydosz, A.; Maziarz, W.; Pisarkiewicz, T.; de Torres, H.B.; Mueller, J. A Micropreconcentrator Design Using Low Temperature Cofired Ceramics Technology for Acetone Detection Applications. IEEE Sens. J. 2013, 13, 1889–1896. [Google Scholar] [CrossRef]
- Rydosz, A.; Maziarz, W.; Pisakiewicz, T.; Domański, K.; Grabiec, P. A gas micropreconcentrator for low level acetone measurements. Microelectron. Reliability 2012, 52, 2640–2646. [Google Scholar] [CrossRef]
- Storer, M.; Dummer, J.; Lunt, H.; Scotter, J.; McCartin, F.; Cook, J.; Swanney, M.; Kendall, D.; Logan, F.; Epton, M. Measurement of breath acetone concentrations by selected ion flow tube mass spectrometry in type 2 Diebetes. J. Breath Res. 2011. [Google Scholar] [CrossRef]
- Kundu, S.K.; Bruzek, J.A.; Nair, R.; Judilla, A.M. Breath acetone analyzer: Diagnostic tool to monitor dietary fat loss. Clin. Chem. 1993, 39, 87–92. [Google Scholar]
- Ueta, I.; Saito, Y.; Hosoe, M.; Okamoto, M.; Ohkita, H.; Shirai, S.; Tamura, H.; Jinno, K. Breath acetone analysis with miniaturized sample preparation device: In-needle preconcentration and subsequent determination by gas chromatography-mass spectroscopy. J. Chromatogr. B 2009, 877, 2551–2556. [Google Scholar] [CrossRef]
- Groves, W.; Zellers, E.; Frye, G. Analyzing organic vapors in exhaled breath using a surface acoustic wave sensor array with preconcentration: Selection and characterization of the preconcentrator adsorbent. Anal. Chim. Acta 1998, 371, 131–143. [Google Scholar] [CrossRef]
- Dow, A.; Lang, W. Design and fabrication of a micropreconcentrator focuser for sensitivity enhancement of chemical sensing systems. IEEE Sens. J. 2012, 12, 2528–2534. [Google Scholar] [CrossRef]
- Ihira, S.-I.; Toda, K. Micro gas analyzers for environmental and medical applications. Anal. Chim. Acta 2008, 619, 143–156. [Google Scholar] [CrossRef]
- Figaro Product Information. TGS 822-for the detection of Organic Solvent Vapors. Available online: http://www.figarosensor.com/products/822pdf.pdf (accessed on 22 July 2014).
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rydosz, A. Micropreconcentrator in LTCC Technology with Mass Spectrometry for the Detection of Acetone in Healthy and Type-1 Diabetes Mellitus Patient Breath. Metabolites 2014, 4, 921-931. https://doi.org/10.3390/metabo4040921
Rydosz A. Micropreconcentrator in LTCC Technology with Mass Spectrometry for the Detection of Acetone in Healthy and Type-1 Diabetes Mellitus Patient Breath. Metabolites. 2014; 4(4):921-931. https://doi.org/10.3390/metabo4040921
Chicago/Turabian StyleRydosz, Artur. 2014. "Micropreconcentrator in LTCC Technology with Mass Spectrometry for the Detection of Acetone in Healthy and Type-1 Diabetes Mellitus Patient Breath" Metabolites 4, no. 4: 921-931. https://doi.org/10.3390/metabo4040921
APA StyleRydosz, A. (2014). Micropreconcentrator in LTCC Technology with Mass Spectrometry for the Detection of Acetone in Healthy and Type-1 Diabetes Mellitus Patient Breath. Metabolites, 4(4), 921-931. https://doi.org/10.3390/metabo4040921