Atlantinone A, a Meroterpenoid Produced by Penicillium ribeum and Several Cheese Associated Penicillium Species
Abstract
:1. Introduction
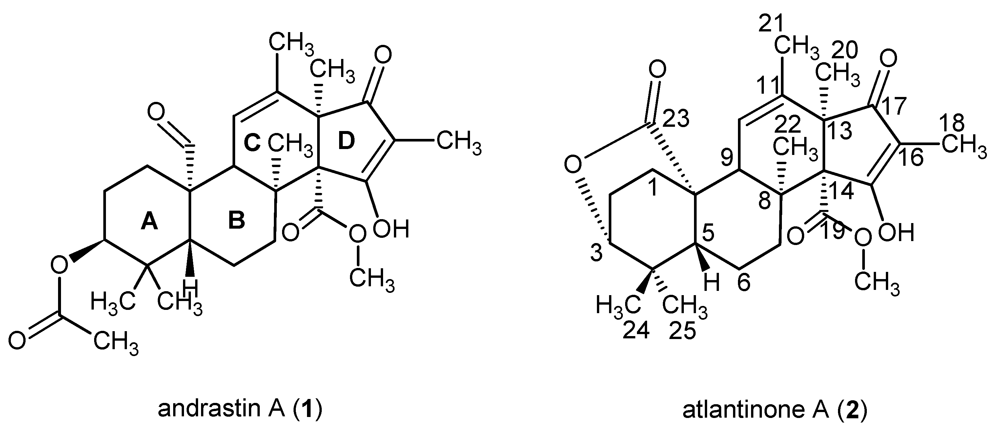
2. Results and Discussion
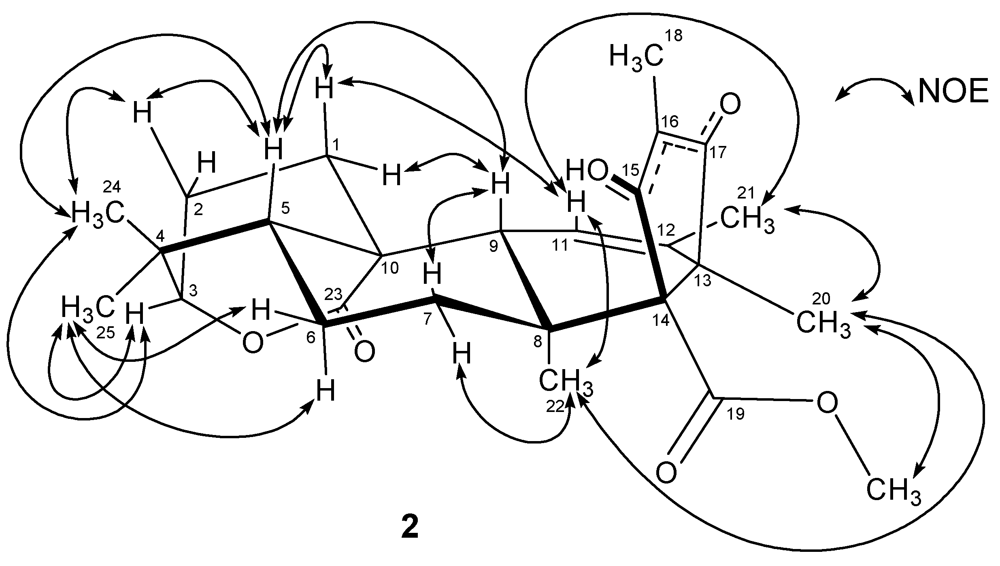
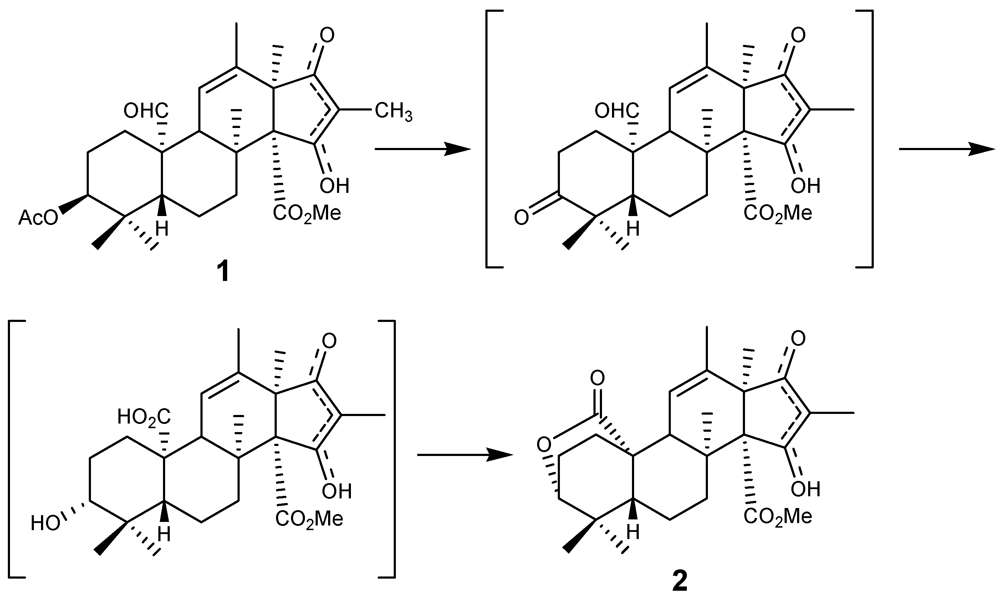
3. Experimental
3.1. Fungal Material and Fermentation
3.2. Extraction and Separation
3.3. Apparatus
3.4. Atlantinone A (2)
 −83.3° (c 0.42, MeOH). UV λmax (MeOH) nm (log ε): 260 (sh), 281 (4.56). 1H and 13C NMR see Table 1 (Supplementary Material). HREIMS m/z 442.2254 (calcd for C26H34O6: 442.2355).
−83.3° (c 0.42, MeOH). UV λmax (MeOH) nm (log ε): 260 (sh), 281 (4.56). 1H and 13C NMR see Table 1 (Supplementary Material). HREIMS m/z 442.2254 (calcd for C26H34O6: 442.2355).4. Conclusions
Acknowledgments
Conflict of Interest
References
- Uchida, R.; Shiomi, K.; Inokoshi, J.; Sunazuka, T.; Tanaka, H.; Iwai, Y.; Takayanagi, H.; Omura, S. Andrastins A–C, new protein farnesyltransferase inhibitors produced by Penicillium sp. FO-3929. II. Structure and biosynthesis. J. Antibiot. 1996, 49, 418–424. [Google Scholar] [CrossRef]
- Shiomi, K.; Tomoda, H.; Otoguro, K.; Omura, S. Meroterpenoids with various biological activities produced by fungi. Pure Appl. Chem. 1999, 71, 1059–1064. [Google Scholar] [CrossRef]
- Uchida, R.; Shiomi, K.; Inokoshi, J.; Tanaka, H.; Iwai, Y.; Omura, S. Andrastin D, novel protein farnesyltransferase inhibitor produced by Penicillium sp. FO-3929. J. Antibiot. 1996, 49, 1278–1280. [Google Scholar] [CrossRef]
- Kosemura, S. Meroterpenoids from Penicillium citreo-viride B. IFO 4692 and 6200 hybrid. Tetrahedron 2003, 59, 5055–5072. [Google Scholar] [CrossRef]
- Omura, S.; Inokoshi, J.; Uchida, R.; Shiomi, K.; Masuma, R.; Kawakubo, T.; Tanama, H.; Iwai, Y.; Kosemura, S.; Yamamura, S. Andrastins A–C, new protein farnesyltransferase inhibitors produced by Penicillium sp. FO-3929. I. Producing strain, fermentation, isolation, and biological activity. J. Antibiot. 1996, 49, 414–417. [Google Scholar] [CrossRef]
- Rho, M.-C.; Toyoshima, M.; Hayashi, M.; Uchida, R.; Shiomi, K.; Komiyama, K.; Omura, S. Enhancement of drug accumulation by andrastin A produced by Penicillium sp. FO-3929 in vincristine-resistant KB cells. J. Antibiot. 1998, 51, 68–72. [Google Scholar] [CrossRef]
- Pedretti, A.; Villa, L.; Vistoli, G. Modeling of binding modes and inhibition mechanism of some natural ligands of farnesyl transferase using molecular docking. J. Med. Chem. 2002, 45, 1460–1465. [Google Scholar] [CrossRef]
- Nielsen, K.F.; Dalsgaard, P.W.; Smedsgaard, J.; Larsen, T.O. Andrastins A-D, Penicillium roqueforti metabolites consistently produced in blue-mold-ripened cheese. J. Agric. Food Chem. 2005, 53, 2908–2913. [Google Scholar]
- Fernández-Bodega, M.A.; Mauriz, E.; Gómez, A.; Martin, J.F. Proteolytic activity, mycotoxins and andrastin A in Penicillium roqueforti strains isolated from Cabrales, Valdeón and Bejes-Tresviso local varieties of blue-weined cheeses. Int. J. Food Microbiol. 2009, 136, 18–25. [Google Scholar] [CrossRef]
- Overy, D.P.; Larsen, T.O.; Dalsgaard, P.W.; Frydenvang, K.; Phipps, R., Munro; Christophersen, C. Andrastin A and barceloneic acid metabolites, protein farnesyl transferase inhibitors from Penicillium albocoremium: Chemotaxonomic significance and pathological implications. Mycol. Res. 2005, 1243–1249. [Google Scholar]
- Sonjak, S.; Frisvad, J.C.; Gunde-Cimerman, N. Comparison of secondary metabolite production by Penicillium crustosum strains, isolated from Arctic and other various ecological niches. FEMS Microbiol. Ecol. 2005, 53, 51–60. [Google Scholar] [CrossRef]
- Frisvad, J.C.; Larsen, T.O; Dalsgaard, P.W.; Seifert, K.A.; Louis-Seize, G.; Lyhne, E.K.; Jarvis, B.B.; Fettinger, J.C.; Overy, D.P. Four psychrotolerant species with high chemical diversity consistently producing cycloaspeptide A, Penicillium jamesonlandense sp. nov., Penicillium ribeum sp. nov., Penicillium soppi, and Penicillium lanosum. Int. J. Syst. Evol. Microbiol. 2006, 56, 1427–1437. [Google Scholar] [CrossRef]
- Dalsgaard, P.W.; Larsen, T.O.; Frydenvang, K.; Christophersen, C. Psychrophilin A and cycloaspeptide D, novel cyclic peptides from the psychrotolerant fungus Penicillium ribeum. J. Nat. Prod. 2004, 67, 878–881. [Google Scholar] [CrossRef]
- Wang, X.; Sena Filho, J.G.; Hoover, A.R.; King, J.B.; Ellis, T.K.; Powell, D.R.; Cichewicz, R.H. Chemical epigenetics alters the secondary metabolite composition of guttate excreted by an atlantic-forest-soil-derived Penicillium citreonigrum. J. Nat. Prod. 2010, 73, 942–948. [Google Scholar] [CrossRef]
- Larsen, T.O.; Gareis, M.; Frisvad, J.C. Cell cytotoxicity and mycotoxin and secondary metabolite production by common penicillia on cheese agar. J. Agric. Food Chem. 2002, 50, 6148–6164. [Google Scholar]
- Schroeckh, V.; Scherlach, K.; Nützmann, H.W.; Shelest, E.; Schmidt-Heck, W.; Schuemann, J.; Martin, K.; Hertweck, C.; Brakhage, A.A. Intimate bacterial-fungal interaction triggers biosynthesis of archetypal polyketides in Aspergillus nidulans. Proc. Natl. Acad. Sci. USA 2009, 106, 14558–14563. [Google Scholar]
- Houbraken, J.; Samson, R.A. Phylogeny of Penicillium and the segregation of Trichocomaceae into three families. Stud. Mycol. 2011, 70, 1–51. [Google Scholar] [CrossRef]
- Rivera, K.G.; Seifert, K.A. A taxonomic and phylogenetic revision of the Penicillium sclerotiorum complex. Stud. Mycol. 2011, 70, 139–158. [Google Scholar] [CrossRef]
- Nielsen, M.L.; Nielsen, J.B.; Rank, C.; Klejnstrup, M.L.; Holm, D.K.; Brogaard, K.H.; Hansen, B.G.; Frisvad, J.C.; Larsen, T.O.; Mortensen, U.H. A genome-wide polyketide synthase deletion library uncovers novel genetic links to polyketides and meroterpenoids in Aspergillus nidulans. FEMS Microbiol. Lett. 2011, 321, 157–166. [Google Scholar] [CrossRef]
Supplementary Files
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Dalsgaard, P.W.; Petersen, B.O.; Duus, J.Ø.; Zidorn, C.; Frisvad, J.C.; Christophersen, C.; Larsen, T.O. Atlantinone A, a Meroterpenoid Produced by Penicillium ribeum and Several Cheese Associated Penicillium Species. Metabolites 2012, 2, 214-220. https://doi.org/10.3390/metabo2010214
Dalsgaard PW, Petersen BO, Duus JØ, Zidorn C, Frisvad JC, Christophersen C, Larsen TO. Atlantinone A, a Meroterpenoid Produced by Penicillium ribeum and Several Cheese Associated Penicillium Species. Metabolites. 2012; 2(1):214-220. https://doi.org/10.3390/metabo2010214
Chicago/Turabian StyleDalsgaard, Petur W., Bent O. Petersen, Jens Ø. Duus, Christian Zidorn, Jens C. Frisvad, Carsten Christophersen, and Thomas O. Larsen. 2012. "Atlantinone A, a Meroterpenoid Produced by Penicillium ribeum and Several Cheese Associated Penicillium Species" Metabolites 2, no. 1: 214-220. https://doi.org/10.3390/metabo2010214
APA StyleDalsgaard, P. W., Petersen, B. O., Duus, J. Ø., Zidorn, C., Frisvad, J. C., Christophersen, C., & Larsen, T. O. (2012). Atlantinone A, a Meroterpenoid Produced by Penicillium ribeum and Several Cheese Associated Penicillium Species. Metabolites, 2(1), 214-220. https://doi.org/10.3390/metabo2010214



