Differentiation of Therapeutic and Illicit Drug Use via Metabolite Profiling
Abstract
1. Introduction
2. Materials and Methods
3. Opioids
3.1. Morphine/Codeine/Diacethylmorphine (Heroin)
3.2. Oxycodone/Oxymorphone/Hydrocodone/Hydromorphone
3.3. Methadone
3.4. Fentanyl
4. Amphetamine-Type Stimulants
4.1. Screening and Cross-Reactivity in Clinical and Forensic Settings
4.2. Metabolic Overview of Amphetamine—Type Stimulants
4.3. Methamphetamine: Pharmacokinetics and Metabolism
4.4. MDMA and MDA: Parent-Metabolite Relationships
4.5. MDEA: Metabolic Pathways and Biomarkers
4.6. Synthetic Impurities and Adulterants of ATS
4.7. Synthetic Cathinones: Analytical and Toxicological Considerations
4.8. Chiral (Enantiomeric) Analysis for Source Attribution
5. Benzodiazepines and Z-Drugs
5.1. Toxicological Analysis of Benzodiazepines and Z-Drugs
5.2. Interpretation of Analytical Findings
5.2.1. Diazepam
5.2.2. Other Benzodiazepines
5.2.3. Z-Drugs
6. Cannabinoids
6.1. Therapeutic Use of Cannabinoids
6.2. Recreational Use of Cannabinoids
6.3. Toxicological Analysis of Cannabinoids
6.4. Cannabinoids Metabolism
6.5. Differentiating Therapeutic Epidyolex® Use from Recreational Cannabis Consumption
- CBD/THC ratio: Epidyolex usage shows high CBD with negligible THC, while recreational use presents THC dominance.
- Presence of 7-OH-CBD and 7-COOH-CBD: Indicative of pharmaceutical CBD metabolism.
- Absence of plant-derived impurities or minor cannabinoids in Epidyolex, versus their variable presence in unregulated cannabis.
6.6. Differentiating Therapeutic Dronabinol Use from Recreational Cannabis Consumption
6.7. Differentiating Therapeutic Nabilone Use from Recreational Cannabis Consumption
- LC–MS/MS or GC–MS: Required for specific detection of nabilone and its unique metabolites, which are not identifiable through routine THC screening assays.
- Absence of THC-COOH and plant-based cannabinoids (CBD, CBN, CBG): Strongly indicative of synthetic cannabinoid use.
- Negative detection for minor cannabinoids: Supports pharmaceutical origin.
- Immunoassays: Prone to false positives due to structural similarity of nabilone and THC; not reliable alone for differentiation.
6.8. Differentiating Therapeutic Nabiximols Use from Recreational Cannabis Consumption
7. Cocaine
7.1. Cocaine Pharmacokinetics
7.2. Toxicochemical Analysis and Metabolic Profiling of Cocaine
7.3. Biomarkers, Analytical Thresholds, and Interpretive Challenges in Cocaine Testing
8. Ketamine
8.1. Therapeutic Uses and Mechanisms of Action
8.2. Illicit Use and Misuse Patterns
8.3. Pharmacokinetics, Analytical Detection and Toxicochemical Profiling
8.4. Toxicology and Adverse Effects
9. Application of Metabolomics in Differentiating Legal Therapeutic Use from Illicit Drug Abuse
10. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| GC-MS | Gas Chromatography–Mass Spectrometry |
| LC-MS/MS | Liquid Chromatography–Tandem Mass Spectrometry |
| HRMS | High-Resolution Mass Spectrometry |
| EMA | European Medicines Agency |
| EUDA | European Union Drugs Agency |
| OUD | Opioid Use Disorder |
| WHO | World Health Organization |
| TOSU | Targeted Opioid Screen, Random, Urine |
| UHPLC | Ultra High Performance Liquid Chromatography |
| MS/MS | Tandem Mass Spectrometry |
| 6-MAM | 6-Monoacetylmorphine |
| EDDP | 2-ethylidene-1,5-dimethyl-3,3-diphenylpyrrolidine |
| CYP | Cytochrome P450 |
| M/C | Morphine/Codeine |
| 6-AC | 6-acetylcodeine |
| FDA | Food and Drug Administration |
| NSOs | Novel Synthetic Opioids |
| HPLC | High Performance Liquid Chromatography |
| Tmax | Time to Maximum Plasma Concentration |
| t½ (T1/2) | Elimination Half-Life |
| 4-MMC | 4-Methylmethcathinone (Mephedrone) |
| AM | Amphetamine |
| ATS | Amphetamine-Type Stimulants |
| bk-MDMA | β-Keto-3,4-Methylenedioxymethamphetamine (Methylone) |
| bk-MDEA | β-Keto-3,4-Methylenedioxyethylamphetamine (Ethylone) |
| DAT | Dopamine Transporter |
| DHEA | 3,4-Dihydroxyethylamphetamine—metabolite of MDEA |
| DFSA | Drug-Facilitated Sexual Assault |
| HMA | 4-Hydroxyamphetamine |
| HMMA | 4-Hydroxy-3-Methoxymethamphetamine—major metabolite of MDMA |
| HMEA | 4-Hydroxy-3-Methoxyethylamphetamine—metabolite of MDEA |
| HMMC | 4-Hydroxy-3-Methoxymethcathinone—metabolite related to methylone (bk-MDMA) |
| MA | Methamphetamine |
| MDA | 3,4-Methylenedioxyamphetamine |
| MDEA | 3,4-Methylenedioxyethylamphetamine (“Eve”) |
| MDMA | 3,4-Methylenedioxymethamphetamine (“Ecstasy”) |
| NET | Norepinephrine Transporter |
| PMA | Para-Methoxyamphetamine |
| PMMA | Para-Methoxymethamphetamine |
| SERT | Serotonin Transporter |
| VMAT2 | Vesicular Monoamine Transporter 2 |
| GABA | γ-aminobutyric acid |
| CNS | Central Nervous System |
| DBS | Dried Blood Spot |
| LOQ | Limit of Quantification |
| THC | Δ9-tetrahydrocannabinol |
| CBD | Cannabidiol |
| CUD | Cannabis Use Disorder |
| DALYs | Disability-Adjusted Life Years |
| CBN | Cannabinol |
| BE | Benzoylecgonine |
| EME | Ecgonine Methyl Ester |
| AEME | Anhydroecgonine Methyl Ester |
| NMDA | N-methyl-D-aspartate |
References
- Sun, E.C.; Dixit, A.; Humphreys, K.; Darnall, B.D.; Baker, L.C.; Mackey, S. Association between concurrent use of prescription opioids and benzodiazepines and overdose: Retrospective analysis. BMJ 2017, 356, j760. [Google Scholar] [CrossRef]
- United Nations Office on Drugs and Crime (UNODC). World Drug Report 2024; United Nations Publications: Vienna, Austria, 2024. [Google Scholar]
- Moody, M.T.; Diaz, S.; Shah, P.; Papsun, D.; Logan, B.K. Analysis of fentanyl analogs and novel synthetic opioids in blood, serum/plasma, and urine in forensic casework. Drug Test. Anal. 2018, 10, 1358–1367. [Google Scholar] [CrossRef]
- Langman, L.J.; Bechtel, L.K.; Holstege, C.P. Clinical toxicology. In Tietz Textbook of Laboratory Medicine, 7th ed.; Elsevier: Amsterdam, The Netherlands, 2022. [Google Scholar]
- Mogollón, N.G.S.; Quiroz-Moreno, C.D.; Prata, P.S.; de Almeida, J.R.; Cevallos, A.S.; Torres-Guiérrez, R.; Augusto, F. New Advances in Toxicological Forensic Analysis Using Mass Spectrometry Techniques. J. Anal. Methods Chem. 2018, 2018, 4142527. [Google Scholar] [CrossRef]
- Love, D.; Jones, N.S. Interpol Review of Drug Analysis 2019-2022. Forensic Sci. Int. Synerg. 2023, 6, 100299. [Google Scholar] [CrossRef]
- Cone, E.J.; Welch, P.; Mitchell, J.M.; Paul, B.D. Forensic Drug Testing for Opiates: I. Detection of 6-Acetylmorphine in Urine as an Indicator of Recent Heroin Exposure; Drug and Assay Considerations and Detection Times. J. Anal. Toxicol. 1991, 15, 1–7. [Google Scholar] [CrossRef]
- Qriouet, Z.; Qmichou, Z.; Bouchoutrouch, N.; Mahi, H.; Cherrah, Y.; Sefrioui, H. Analytical Methods Used for the Detection and Quantification of Benzodiazepines. J. Anal. Methods Chem. 2019, 2019, 2035492. [Google Scholar] [CrossRef] [PubMed]
- Rust, K.Y.; Baumgartner, M.R.; Meggiolaro, N.; Kraemer, T. Detection and validated quantification of 21 benzodiazepines and 3 “z-drugs” in human hair by LC-MS/MS. Forensic Sci. Int. 2012, 215, 64–72. [Google Scholar] [CrossRef] [PubMed]
- Losacker, M.; Kraemer, M.; Philipsen, A.; Duecker, K.; Dreimueller, N.; Engelmann, J.; Roehrich, J.; Hess, C. Enantioselective Quantification of Amphetamine and Metabolites in Serum Samples: Forensic Evaluation and Estimation of Consumption Time. Metabolites 2021, 11, 521. [Google Scholar] [CrossRef]
- Huestis, M.A.; Mazzoni, I.; Rabin, O. Cannabis in sport: Anti-doping perspective. Sports Med. 2011, 41, 949–966. [Google Scholar] [CrossRef] [PubMed]
- Dawidowska, J.; Krzyżanowska, M.; Markuszewski, M.J.; Kaliszan, M. The Application of Metabolomics in Forensic Science with Focus on Forensic Toxicology and Time-of-Death Estimation. Metabolites 2021, 11, 801. [Google Scholar] [CrossRef]
- Kruidering-Hall, M.; Katzung, B.G.; Tuan, R.; Vanderah, T.W. Opioid Analgesics & Antagonists. In Katzung’s Pharmacology Examination & Board Review, 14th ed.; McGraw Hill: New York, NY, USA, 2024; Available online: https://accessmedicine.mhmedical.com/content.aspx?bookid=3461§ionid=285595011 (accessed on 12 August 2025).
- Jutkiewicz, E.M.; Traynor, J.R. Opioid analgesics. In Goodman & Gilman’s: The Pharmacological Basis of Therapeutics, 14th ed.; Brunton, L.L., Knollmann, B.C., Eds.; McGraw-Hill Education: New York, NY, USA, 2023; Available online: https://accessmedicine.mhmedical.com/content.aspx?bookid=3191§ionid=269719481 (accessed on 12 August 2025).
- Gustafsson, M.; Silva, V.; Valeiro, C.; Joaquim, J.; van Hunsel, F.; Matos, C. Misuse, abuse and medication errors’ adverse events associated with opioids—A systematic review. Pharmaceuticals 2024, 17, 1009. [Google Scholar] [CrossRef]
- Ho, J.; Mitton, J.A.; Ristau, J. Opioids: Introduction. In Current Medical Diagnosis & Treatment 2025; Papadakis, M.A., Rabow, M.W., McQuaid, K.R., Gandhi, M., Eds.; McGraw-Hill Education: New York, NY, USA, 2025; Available online: https://accessmedicine.mhmedical.com/content.aspx?bookid=3495§ionid=288504802 (accessed on 12 August 2025).
- Rullo, L.; Morosini, C.; Lacorte, A.; Cristani, M.; Coluzzi, F.; Candeletti, S.; Romualdi, P. Opioid system and related ligands: From the past to future perspectives. J. Anesth. Analg. Crit. Care 2024, 4, 70. [Google Scholar] [CrossRef] [PubMed]
- American Psychiatric Association. DSM-5 Task Force. In Diagnostic and Statistical Manual of Mental Disorders: DSM-5™, 5th ed.; American Psychiatric Publishing, Inc.: Arlington, VA, USA, 2013. [Google Scholar] [CrossRef]
- Dydyk, A.M.; Jain, N.K.; Gupta, M. Opioid Use Disorder: Evaluation and Management. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2025. Available online: https://www.ncbi.nlm.nih.gov/books/NBK553166/ (accessed on 8 August 2025).
- National Institute on Drug Abuse (NIDA). Medications for Opioid Use Disorder. Available online: https://nida.nih.gov/research-topics/medications-opioid-use-disorder (accessed on 8 August 2025).
- Jenkins, R.A. The fourth wave of the US opioid epidemic and its implications for the rural US: A federal perspective. Prev. Med. 2021, 152 Pt 2, 106541. [Google Scholar] [CrossRef] [PubMed]
- Jenkins, R.A.; Whitney, B.M.; Nance, R.M.; Allen, T.M.; Cooper, H.L.F.; Feinberg, J.; Fredericksen, R.; Friedmann, P.D.; Go, V.F.; Jenkins, W.D.; et al. The Rural Opioid Initiative Consortium description: Providing evidence to understand the fourth wave of the opioid crisis. Addict. Sci. Clin. Pract. 2022, 17, 38. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Opioid Overdose. Available online: https://www.who.int/news-room/fact-sheets/detail/opioid-overdose (accessed on 8 August 2025).
- Health, T.L. Opioid overdose crisis: Time for a radical rethink. Lancet Public Health 2022, 7, e195. [Google Scholar] [CrossRef]
- Prevete, E.; Mason, N.L.; Kuypers, K.P.C.; Theunissen, E.L.; Mallaroni, P.; Pasquini, M.; Ramaekers, J.G. Use patterns of classic, novel, and herbal opioids. Emerg. Trends Drugs Addict. Health 2025, 5, 100166. [Google Scholar] [CrossRef]
- Pérez-Mañá, C.; Papaseit, E.; Fonseca, F.; Farré, A.; Torrens, M.; Farré, M. Drug interactions with new synthetic opioids. Front. Pharmacol. 2018, 9, 1145. [Google Scholar] [CrossRef]
- European Monitoring Centre for Drugs and Drug Addiction (EMCDDA). Heroin and Other Opioids. Available online: https://www.euda.europa.eu/publications/european-drug-report/2025/heroin-and-other-opioids_en (accessed on 8 August 2025).
- Holler, J.; Levine, B. Confirmation Methods for SAMHSA Drugs and Other Commonly Abused Drugs. In Critical Issues in Alcohol and Drugs of Abuse Testing, 2nd ed.; Langman, L.J., Dasgupta, A., Eds.; Academic Press: Cambridge, MA, USA, 2019; Chapter 15; pp. 189–206. [Google Scholar] [CrossRef]
- Langman, L.J.; Snozek, C.L.H. Introduction to Drugs of Abuse. In Critical Issues in Alcohol and Drugs of Abuse Testing, 2nd ed.; Langman, L.J., Dasgupta, A., Eds.; Academic Press: Cambridge, MA, USA, 2019; Chapter 6; pp. 71–78. [Google Scholar] [CrossRef]
- Dart, R.C.; Iwanicki, J.L.; Black, J.C.; Olsen, H.A.; Severtson, S.G. Measuring prescription opioid misuse and its consequences. Br. J. Clin. Pharmacol. 2021, 87, 1647–1653. [Google Scholar] [CrossRef] [PubMed]
- McCabe, S.E.; Cranford, J.A.; Boyd, C.J.; Teter, C.J. Motives, diversion and routes of administration associated with nonmedical use of prescription opioids. Addict. Behav. 2007, 32, 562–575. [Google Scholar] [CrossRef]
- Young, A.M.; Havens, J.R.; Leukefeld, C.G. Route of administration for illicit prescription opioids: A comparison of rural and urban drug users. Harm Reduct. J. 2010, 7, 24. [Google Scholar] [CrossRef]
- Schinas, A.; Nanji, S.; Vorobej, K.; Mills, C.; Govier, D.; Setnik, B. Key characteristics and habits of the recreational opioid user. J. Opioid Manag. 2019, 15, 507–520. [Google Scholar] [CrossRef]
- Dowell, D.; Brown, S.; Gyawali, S.; Hoenig, J.; Ko, J.; Mikosz, C.; Ussery, E.; Baldwin, G.; Jones, C.M.; Olsen, Y.; et al. Treatment for opioid use disorder: Population estimates-United States, 2022. Morb. Mortal. Wkly Rep. 2024, 73, 567–574. [Google Scholar] [CrossRef]
- United Nations Office on Drugs and Crime (UNODC). World Drug Report 2023; Booklet 1. Available online: https://www.unodc.org/unodc/en/data-and-analysis/world-drug-report-2023.html (accessed on 8 August 2025).
- Keary, C.J.; Wang, Y.; Moran, J.R.; Zayas, L.V.; Stern, T.A. Toxicologic testing for opiates: Understanding false-positive and false-negative test results. Prim. Care Companion CNS Disord. 2012, 14, PCC.12f01371. [Google Scholar] [CrossRef]
- Mayo Clinic Laboratories. New High-Resolution Targeted Opioid Screen: Superior Sensitivity and Specificity to Evaluate Adherence to Prescribed Opioid Therapy. Available online: https://news.mayocliniclabs.com/2020/07/13/new-high-resolution-targeted-opioid-screen-superior-sensitivity-and-specificity-to-evaluate-adherence-to-prescribed-opioid-therapy/ (accessed on 19 September 2025).
- Dasgupta, A. Analytical True Positives in Workplace Drugs Testings Due to Use of Prescription and OTC Medications. In Beating Drug Tests and Defending Positive Results; Humana Press: Totowa, NJ, USA, 2010; Chapter 10. [Google Scholar] [CrossRef]
- Reisfield, G.M.; Bertholf, R.L. Drug Testing in Pain Management. In Critical Issues in Alcohol and Drugs of Abuse Testing, 2nd ed.; Levine, B., Ed.; Elsevier: Cambridge, MA, USA, 2019; Chapter 25; pp. 343–358. [Google Scholar] [CrossRef]
- Danso, D.; Langman, L.J.; Jannetto, P.J. Targeted Opioid Screening Assay for Pain Management Using High-Resolution Mass Spectrometry. In Methods in Molecular Biology; Clifton, N.J., Ed.; Springer: Berlin/Heidelberg, Germany, 2019; Volume 1872, pp. 41–50. [Google Scholar] [CrossRef]
- Di Francesco, G.; Montesano, C.; Vincenti, F.; Bilel, S.; Corli, G.; Petrella, G.; Cicero, D.O.; Gregori, A.; Marti, M.; Sergi, M. Tackling new psychoactive substances through metabolomics: UHPLC-HRMS study on natural and synthetic opioids in male and female murine models. Sci. Rep. 2024, 14, 9432. [Google Scholar] [CrossRef]
- Milone, M.C. Laboratory Testing for Prescription Opioids. J. Med. Toxicol. 2012, 8, 408–416. [Google Scholar] [CrossRef] [PubMed]
- Krasowski, M.D.; Kwong, T.C. Analytical True Positive Drug Tests Due to Use of Prescription and Nonprescription Medications. In Critical Issues in Alcohol and Drugs of Abuse Testing, 2nd ed.; Academic Press: Cambridge, MA, USA, 2019; Chapter 30; pp. 441–448. [Google Scholar] [CrossRef]
- Milella, M.S.; D’Ottavio, G.; De Pirro, S.; Barra, M.; Caprioli, D.; Badiani, A. Heroin and Its Metabolites: Relevance to Heroin Use Disorder. Transl. Psychiatry 2023, 13, 120. [Google Scholar] [CrossRef]
- Dasgupta, A. Workplace Drug Testing: SAMHSA and Non-SAMHSA Drugs. In Beating Drug Tests and Defending Positive Results; Humana Press: Totowa, NJ, USA, 2010; pp. 25–40. [Google Scholar] [CrossRef]
- Moeller, K.E.; Kissack, J.C.; Atayee, R.S.; Lee, K.C. Clinical interpretation of urine drug tests: What clinicians need to know about urine drug screens. Mayo Clin. Proc. 2017, 92, 774–796. [Google Scholar] [CrossRef]
- Maas, A.; Madea, B.; Hess, C. Confirmation of Recent Heroin Abuse: Accepting the Challenge. Drug Test. Anal. 2018, 10, 54–71. [Google Scholar] [CrossRef] [PubMed]
- Ellis, A.D.; McGwin, G.; Davis, G.G.; Dye, D.W. Identifying Cases of Heroin Toxicity Where 6-Acetylmorphine (6-AM) Is Not Detected by Toxicological Analyses. Forensic Sci. Med. Pathol. 2016, 12, 243–247. [Google Scholar] [CrossRef]
- Seif-Barghi, T.; Moghadam, N.; Kobarfard, F. Morphine/Codeine Ratio, a Key in Investigating a Case of Doping. Asian J. Sports Med. 2015, 6, e28798. [Google Scholar] [CrossRef] [PubMed]
- Al-Amri, A.M.; Smith, R.M.; El-Haj, B.M. The GC-MS Detection and Characterization of Neopine Resulting from Opium Use and Codeine Metabolism and Its Potential as an Opiate-Product-Use Marker. Anal. Bioanal. Chem. 2005, 382, 830–835. [Google Scholar] [CrossRef] [PubMed]
- Karakasi, C.; Nikolaou, P.; Petropoulou, G.; Athanaselis, S.; Sakelliadis, E.; Dona, A.; Papoutsis, I. Determination of Indirect Heroin Biomarkers in Biological Samples of Heroin Users. Forensic Sci. Int. Rep. 2024, 10, 100383. [Google Scholar] [CrossRef]
- Medications for Opioid Use Disorder: For Healthcare and Addiction Professionals, Policymakers, Patients, and Families: Updated 2021 [Internet]. Rockville (MD): Substance Abuse and Mental Health Services Administration (US). 2018. (Treatment Improvement Protocol (TIP) Series, No. 63.) [Table, Urine Drug Testing Window of Detection]. Available online: https://www.ncbi.nlm.nih.gov/books/NBK574910/ (accessed on 4 September 2025).
- Mayo Clinic Laboratories. Specific Drug Groups. Available online: https://www.mayocliniclabs.com/test-catalog/drug-book/specific-drug-groups (accessed on 4 September 2025).
- Jones, A.W.; Holmgren, A.; Kugelberg, F.C. Driving Under the Influence of Opiates: Concentration Relationships Between Morphine, Codeine, 6-Acetyl Morphine, and Ethyl Morphine in Blood. J. Anal. Toxicol. 2008, 32, 265–272. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Akhgari, M.; Etemadi-Aleagha, A.; Jokar, F. Street Level Heroin, an Overview on Its Components and Adulterants. In Neuropathology of Drug Addictions and Substance Misuse; Preedy, V.R., Ed.; Elsevier: New York, NY, USA, 2016; Volume 3, pp. 863–877. [Google Scholar] [CrossRef]
- Demirkapu, M.J.; Yananli, H.R. Opium Alkaloids. In Bioactive Compounds in Nutraceutical and Functional Food for Good Human Health; Sharma, K., Mishra, K., Senapati, K.K., Danciu, C., Eds.; IntechOpen: London, UK, 2020. [Google Scholar] [CrossRef][Green Version]
- Kaboudin, B.; Sohrabi, M. Chemistry and Synthesis of Major Opium Alkaloids: A Comprehensive Review. J. Iran. Chem. Soc. 2021, 18, 3177–3218. [Google Scholar] [CrossRef]
- Morley, S.; Forrest, A.; Galloway, J. Validation of Meconin as a Marker for Illicit Opiate Use. J. Anal. Toxicol. 2007, 31, 105–108. [Google Scholar] [CrossRef][Green Version]
- Jones, J.T.; Jones, M.; Jones, B.; Sulaiman, K.; Plate, C.; Lewis, D. Detection of Codeine, Morphine, 6-Monoacetylmorphine, and Meconin in Human Umbilical Cord Tissue: Method Validation and Evidence of In Utero Heroin Exposure. Ther. Drug Monit. 2015, 37, 45–52. [Google Scholar] [CrossRef]
- Al-Amri, A.M. Marker Compounds in Urine for Opiate Product Use. Ph.D. Thesis, Department of Chemistry, Loughborough University, Loughborough, UK, 2004. Available online: https://repository.lboro.ac.uk/articles/thesis/Marker_compounds_in_urine_for_opiate_product_use/9398036 (accessed on 4 September 2025).
- Rahmanian-Devin, P.; Baradaran Rahimi, V.; Jaafari, M.R.; Golmohammadzadeh, S.; Sanei-Far, Z.; Askari, V.R. Noscapine, an Emerging Medication for Different Diseases: A Mechanistic Review. Evid.-Based Complement. Altern. Med. 2021, 2021, 8402517, Retraction in Evid.-Based Complement. Altern. Med. 2023, 2023, 9796340. https://doi.org/10.1155/2023/9796340. [Google Scholar] [CrossRef]
- El-Haj, B.M.; Al-Amri, A.M.; Ali, H.S. GC-MS Detection and Tentative Characterization of Two Noscapine Metabolites in Human Urine and Their Potential as Markers for Opium and Illicit Heroin Use. Forensic Toxicol. 2007, 25, 22–29. [Google Scholar] [CrossRef]
- Dasgupta, A. Defending Positive Opiate and Marijuana Test Results. In Beating Drug Tests and Defending Positive Results; Humana Press: Totowa, NJ, USA, 2010; pp. 75–90. [Google Scholar] [CrossRef]
- Brenneisen, R.; Hasler, F.; Wursch, D. Acetylcodeine as a Urinary Marker to Differentiate the Use of Street Heroin and Pharmaceutical Heroin. J. Anal. Toxicol. 2002, 26, 561–566. [Google Scholar] [CrossRef]
- Miller, P.M. Principles of Addiction: Comprehensive Addictive Behaviors and Disorders, Volume 1; Academic Press: Cambridge, MA, USA, 2013. [Google Scholar]
- Dasgupta, A. Analytical True Positive: Poppy Seed Products and Opiate Analysis. In Critical Issues in Alcohol and Drugs of Abuse Testing, 2nd ed.; Academic Press: Cambridge, MA, USA, 2019; pp. 449–462. [Google Scholar] [CrossRef]
- Reisfield, G.M.; Teitelbaum, S.A.; Jones, J.T. Poppy Seed Consumption May Be Associated with Codeine-Only Urine Drug Test Results. J. Anal. Toxicol. 2023, 47, 107–113. [Google Scholar] [CrossRef]
- Lee, S.; Park, Y.; Han, E.; Choi, H.; Chung, H.; Oh, S.M.; Chung, K.H. Thebaine in hair as a marker for chronic use of illegal opium poppy substances. Forensic Sci. Int. 2011, 204, 115–118. [Google Scholar] [CrossRef] [PubMed]
- Eisenreich, A.; Sachse, B.; Gürtler, R.; Dusemund, B.; Lindtner, O.; Schäfer, B. What do we know about health risks related to thebaine in food? Food Chem. 2020, 309, 125564. [Google Scholar] [CrossRef]
- U.S. Army. Department of Defense Changes Codeine Cutoff Rate. 2023. Available online: https://www.army.mil/article/277574/department_of_defense_changes_codeine_cutoff_rate (accessed on 19 September 2025).
- Maas-Gramlich, A.; Madea, B.; Krämer, M. Nachweis eines Heroinkonsums. Rechtsmed 2023, 33, 501–512. [Google Scholar] [CrossRef]
- Marton, J.; Cumming, P.; Rice, K.C.; Linders, J.T.M. Morphinan Alkaloids and Their Transformations: A Historical Perspective of a Century of Opioid Research in Hungary. Int. J. Mol. Sci. 2025, 26, 2736. [Google Scholar] [CrossRef]
- El-Haj, B.M.; Ali, H.S.; Hamoudi, N.M. Oripavine as a new marker of opiate product use. Forensic Toxicol. 2011, 29, 152–158. [Google Scholar] [CrossRef]
- Trafkowski, J.; Madea, B.; Musshoff, F. The Significance of Putative Urinary Markers of Illicit Heroin Use after Consumption of Poppy Seed Products. Ther. Drug Monit. 2006, 28, 552–558. [Google Scholar] [CrossRef]
- Al-Amri, A.M.; Smith, R.M.; El-Haj, B.M.; Juma’a, M.H. The GC-MS detection and characterization of reticuline as a marker of opium use. Forensic Sci. Int. 2004, 142, 61–69. [Google Scholar] [CrossRef]
- Al-Asmari, A.I.; Alharbi, H.; Zughaibi, T.A. Post-Mortem Analysis of Heroin Biomarkers, Morphine and Codeine in Stomach Wall Tissue in Heroin-Related Deaths. Toxics 2022, 10, 473. [Google Scholar] [CrossRef]
- Ciucă Anghel, D.M.; Ciobanu, A.M.; Guțu, C.M.; Stan, M.; Tudor, G.; Baconi, D.L. GC-MS Analysis of Methadone and EDDP in Addicted Patients under Methadone Substitution Treatment: Comparison of Urine and Plasma as Biological Samples. Molecules 2022, 27, 8360. [Google Scholar] [CrossRef] [PubMed]
- Himes, S.K.; Goodwin, R.S.; Rock, C.M.; Jones, H.E.; Johnson, R.E.; Wilkins, D.G.; Huestis, M.A. Methadone and Metabolites in Hair of Methadone-Assisted Pregnant Women and Their Infants. Ther. Drug Monit. 2012, 34, 337–344. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, M.K.K.; Johansen, S.S.; Linnet, K. Evaluation of Metabolite/Drug Ratios in Blood and Urine as a Tool for Confirmation of a Reduced Tolerance in Methadone-Related Deaths in Denmark. Drug Alcohol Depend. 2013, 133, 447–451. [Google Scholar] [CrossRef]
- Diong, S.H.; Yusoff, N.S.M.; Sim, M.S.; Aziddin, R.E.R.; Chik, Z.; Rajan, P.; Rashid, R.A.; Chemi, N.; Mohamed, Z. Quantitation of Methadone and Metabolite in Patients under Maintenance Treatment. J. Anal. Toxicol. 2014, 38, 660–666. [Google Scholar] [CrossRef]
- Collins, A.A.; Merritt, A.P.; Bourland, J.A. Cross-Reactivity of Tapentadol Specimens with DRI Methadone Enzyme Immunoassay. J. Anal. Toxicol. 2012, 36, 582–587. [Google Scholar] [CrossRef] [PubMed]
- Holm, K.M.D.; Linnet, K. Distribution of Enantiomers of Methadone and Its Main Metabolite EDDP in Human Tissues and Blood of Postmortem Cases. J. Forensic Sci. 2015, 60, 95–101. [Google Scholar] [CrossRef]
- Ribeiro, C.; Santos, C.; Gonçalves, V.; Ramos, A.; Afonso, C.; Tiritan, M.E. Chiral Drug Analysis in Forensic Chemistry: An Overview. Molecules 2018, 23, 262. [Google Scholar] [CrossRef] [PubMed]
- Mueller, F.; Losacco, G.L.; Nicoli, R.; Guillarme, D.; Thomas, A.; Grata, E. Enantiomeric Methadone Quantitation on Real Post-Mortem Dried Matrix Spots Samples: Comparison of Liquid Chromatography and Supercritical Fluid Chromatography Coupled to Mass Spectrometry. J. Chromatogr. B 2021, 1177, 122755. [Google Scholar] [CrossRef]
- Doriety, L.J.; Farrington, E.A. Urine Drug Screening: What Pediatric Clinicians Need to Know to Optimize Patient Care. Pharmacol. Contin. Educ. 2021, 35, 449–455. [Google Scholar] [CrossRef] [PubMed]
- Melanson, S.E.F. Assessing Medication Compliance in Palliative Care: What Methodology Should Be Utilized? In Toxicology Cases for the Clinical and Forensic Laboratory; Elsevier: Amsterdam, The Netherlands, 2020; pp. 455–458. [Google Scholar] [CrossRef]
- Galloway, F.R.; Bellet, N.F. Methadone conversion to EDDP during GCMS analysis of urine samples. J. Anal. Toxicol. 1999, 23, 615–619. [Google Scholar] [CrossRef][Green Version]
- Cheng, P.S.; Lee, C.H.; Liu, C.; Chen, C.S. Simultaneous determination of Ketamine, Tramadol, Methadone and their metabolites in urine by gas chromatography-mass spectrometry. J. Anal. Toxicol. 2008, 32, 253–259. [Google Scholar] [CrossRef]
- Stoeva-Grigorova, S.; Radeva-Ilieva, M.; Karkkeselyan, N.; Dragomanova, S.; Kehayova, G.; Dimitrova, S.; Petrova, M.; Zlateva, S.; Marinov, P. Historical Perspectives and Emerging Trends in Fentanyl Use: Part 1—Pharmacological Profile. Pharmacia 2025, 72, 1–9. [Google Scholar] [CrossRef]
- Stoeva-Grigorova, S.; Radeva-Ilieva, M.; Karkkeselyan, N.; Dragomanova, S.; Kehayova, G.; Dimitrova, S.; Petrova, M.; Zlateva, S.; Marinov, P. Historical Perspectives and Emerging Trends in Fentanyl Use: Part 2—Overdose and Misuse. Pharmacia 2025, 72, 1–13. [Google Scholar] [CrossRef]
- Angelini, D.J.; Biggs, T.D.; Maughan, M.N.; Feasel, M.G.; Sisco, E.; Sekowski, J.W. Evaluation of a lateral flow immunoassay for the detection of the synthetic opioid fentanyl. Forensic Sci. Int. 2019, 300, 75–81. [Google Scholar] [CrossRef]
- Green, T.C.; Park, J.N.; Gilbert, M.; McKenzie, M.; Struth, E.; Lucas, R.; Clarke, W.; Sherman, S.G. An Assessment of the Limits of Detection, Sensitivity and Specificity of Three Devices for Public Health-Based Drug Checking of Fentanyl in Street-Acquired Samples. Int. J. Drug Policy 2020, 77, 102661. [Google Scholar] [CrossRef] [PubMed]
- Crocombe, R.A.; Giuntini, G.; Schiering, D.W.; Profeta, L.T.M.; Hargreaves, M.D.; Leary, P.E.; Brown, C.D.; Chmura, J.W. Field-Portable Detection of Fentanyl and Its Analogs: A Review. J. Forensic Sci. 2023, 68, 1570–1600. [Google Scholar] [CrossRef]
- Wang, G.; Huynh, K.; Barhate, R.; Rodrigues, W.; Moore, C.; Coulter, C.; Vincent, M.; Soares, J. Development of a Homogeneous Immunoassay for the Detection of Fentanyl in Urine. Forensic Sci. Int. 2011, 206, 127–131. [Google Scholar] [CrossRef] [PubMed]
- Park, J.N.; Sherman, S.G.; Sigmund, V.; Breaud, A.; Martin, K.; Clarke, W.A. Validation of a Lateral Flow Chromatographic Immunoassay for the Detection of Fentanyl in Drug Samples. Drug Alcohol. Depend. 2022, 240, 109610. [Google Scholar] [CrossRef]
- Busardò, F.P.; Carlier, J.; Giorgetti, R.; Tagliabracci, A.; Pacifici, R.; Gottardi, M.; Pichini, S. Ultra-High-Performance Liquid Chromatography-Tandem Mass Spectrometry Assay for Quantifying Fentanyl and 22 Analogs and Metabolites in Whole Blood, Urine, and Hair. Front. Chem. 2019, 7, 184. [Google Scholar] [CrossRef] [PubMed]
- Clinton, H.A.; Thangada, S.; Gill, J.R.; Mirizzi, A.; Logan, S.B. Improvements in Toxicology Testing to Identify Fentanyl Analogs and Other Novel Synthetic Opioids in Fatal Drug Overdoses, Connecticut, January 2016–June 2019. Public Health Rep. 2021, 136, 80S–86S. [Google Scholar] [CrossRef]
- Omosule, C.; Roper, S.; Farnsworth, C. Utility of Detecting Fentanyl Analogs during LC-MS/MS Confirmation for Positive Fentanyl Urine Drug Screens. Am. J. Clin. Pathol. 2022, 158 (Suppl. S1), S18–S19. [Google Scholar] [CrossRef]
- Uljon, S. Advances in Fentanyl Testing. In Advances in Clinical Chemistry; Elsevier: Amsterdam, The Netherlands, 2023. [Google Scholar] [CrossRef]
- U.S. Pharmacist. Urine Drug Screening: Minimizing False-Positives and False-Negatives to Optimize Patient Care. Available online: https://www.uspharmacist.com/article/urine-drug-screening-minimizing-false-positives-and-false-negatives-to-optimize-patient-care?fbclid=IwAR1B60RNDxryUw-0VMpL5VWNX6nmEuvEr17DayLemqYBcxs2fxvsC-ABZdk (accessed on 19 September 2025).
- Rogers, S.C.; Pruitt, C.W.; Crouch, D.J.; Caravati, E.M. Rapid Urine Drug Screens: Diphenhydramine and Methadone Cross-Reactivity. Pediatr. Emerg. Care 2010, 26, 665–666. [Google Scholar] [CrossRef]
- Baden, L.R.; Horowitz, G.; Jacoby, H.; Eliopoulos, G.M. Quinolones and False-Positive Urine Screening for Opiates by Immunoassay Technology. JAMA 2001, 286, 3115–3119. [Google Scholar] [CrossRef]
- Levine, B.S.; Kerrigan, S. (Eds.) Principles of Forensic Toxicology, 5th ed.; Springer Publishing Company: New York, NY, USA, 2020. [Google Scholar]
- University of Illinois at Chicago. What Drugs Are Likely to Interfere with Urine Drug Screens; Drug Information Group, College of Pharmacy, University of Illinois at Chicago: Chicago, IL, USA, 2021; Available online: https://dig.pharmacy.uic.edu/faqs/2021-2/may-2021-faqs/what-drugs-are-likely-to-interfere-with-urine-drug-screens/ (accessed on 7 November 2025).
- U.S. Department of Transportation. DOT Rule 49 CFR Part 40 § 40.85: “What Are the Cutoff Concentrations for Urine Drug Tests?”; Office of Drug & Alcohol Policy & Compliance: Washington, DC, USA, 2023. Available online: https://www.transportation.gov/odapc/part40/40-85 (accessed on 7 November 2025).
- Paz-Ramos, M.I.; Cruz, S.L.; Violante-Soria, V. Amphetamine-type stimulants: Novel insights into their actions and use patterns. Rev. Investig. Clín. 2023, 75, 143–157. [Google Scholar] [CrossRef] [PubMed]
- United Nations Office on Drugs and Crime (UNODC). World Drug Report 2025—Key Findings (Booklet 1); UNODC: Vienna, Austria, 2025; Available online: https://www.unodc.org/documents/data-and-analysis/WDR_2025/WDR25_B1_Key_findings.pdf (accessed on 21 September 2025).
- Baumann, M.H.; Volkow, N.D. Abuse of new psychoactive substances: Threats and solutions. Neuropsychopharmacol. Off. Publ. Am. Coll. Neuropsychopharmacol. 2016, 41, 663–665. [Google Scholar] [CrossRef] [PubMed]
- Heal, D.J.; Smith, S.L.; Gosden, J.; Nutt, D.J. Amphetamine, past and present—A pharmacological and clinical perspective. J. Psychopharmacol. 2013, 27, 479–496. [Google Scholar] [CrossRef]
- U.S. Food and Drug Administration. Vyvanse (Lisdexamfetamine Dimesylate) Prescribing Information; FDA: Silver Spring, MD, USA, 2022. [Google Scholar]
- Sulzer, D.; Sonders, M.S.; Poulsen, N.W.; Galli, A. Mechanisms of neurotransmitter release by amphetamines: A review. Prog. Neurobiol. 2005, 75, 406–433. [Google Scholar] [CrossRef]
- Volkow, N.D.; Chang, L.; Wang, G.J.; Fowler, J.S.; Leonido-Yee, M.; Franceschi, D.; Sedler, M.J.; Gatley, S.J.; Hitzemann, R.; Ding, Y.S.; et al. Association of dopamine transporter reduction with psychomotor impairment in methamphetamine abusers. Am. J. Psychiatry 2001, 158, 377–382. [Google Scholar] [CrossRef] [PubMed]
- Darke, S.; Kaye, S.; McKetin, R.; Duflou, J. Major physical and psychological harms of methamphetamine use. Drug Alcohol. Rev. 2008, 27, 253–262. [Google Scholar] [CrossRef]
- Cruickshank, C.C.; Dyer, K.R. A review of the clinical pharmacology of methamphetamine. Addiction 2009, 104, 1085–1099. [Google Scholar] [CrossRef]
- McGregor, C.; Srisurapanont, M.; Jittiwutikarn, J.; Laobhripatr, S.; Wongtan, T.; White, J.M. The nature, time course and severity of methamphetamine withdrawal. Addiction 2005, 100, 1320–1329. [Google Scholar] [CrossRef]
- Scott-Ham, M.; Burton, F.C. Toxicological findings in cases of alleged drug-facilitated sexual assault in the United Kingdom over a 3-year period. J. Clin. Forensic Med. 2005, 12, 175–186. [Google Scholar] [CrossRef]
- Negrusz, A.; Gaensslen, R.E. Analytical developments in toxicological investigation of drug-facilitated sexual assault. Anal. Bioanal. Chem. 2003, 376, 1192–1197. [Google Scholar] [CrossRef] [PubMed]
- Siefried, K.J.; Acheson, L.S.; Lintzeris, N.; Ezard, N. Pharmacological Treatment of Methamphetamine/Amphetamine Dependence: A Systematic Review. CNS Drugs 2020, 34, 337–365. [Google Scholar] [CrossRef] [PubMed]
- Chan, B.; Freeman, M.; Kondo, K.; Ayers, C.; Montgomery, J.; Paynter, R.; Kansagara, D. Pharmacotherapy for methamphetamine/amphetamine use disorder—A systematic review and meta-analysis. Addiction 2019, 114, 2122–2136. [Google Scholar] [CrossRef] [PubMed]
- Degenhardt, L.; Hall, W. Extent of illicit drug use and dependence, and their contribution to the global burden of disease. Lancet 2012, 379, 55–70. [Google Scholar] [CrossRef]
- Kruithof, K.; Aldridge, J.; Décary-Hétu, D.; Sim, M.; Dujso, E.; Hoorens, S. Internet-Facilitated Drugs Trade: An Analysis of the Size, Scope and the Role of The Netherlands; RAND Corporation: Santa Monica, CA, USA, 2016. [Google Scholar]
- Compton, W.M.; Han, B.; Blanco, C.; Johnson, K.; Jones, C.M. Prevalence and correlates of prescription stimulant use, misuse, use disorders, and motivations for misuse among adults in the United States. Am. J. Psychiatry 2018, 175, 741–755. [Google Scholar] [CrossRef]
- Forrest, J.; Chen, W.; Jagadheesan, K. Misuse and diversion of stimulant medications prescribed for the treatment of ADHD: A systematic review. Front. Psychiatry 2025, 16, 1612785. [Google Scholar] [CrossRef]
- Thevis, M.; Geyer, H.; Thomas, A.; Schänzer, W. Trafficking of drug candidates relevant for sports drug testing: Detection of non-approved therapeutics categorized as anabolic and gene doping agents in products distributed via the Internet. Drug Test. Anal. 2011, 3, 331–336. [Google Scholar] [CrossRef]
- Cashman, C.M.; Ruotsalainen, J.H.; Greiner, B.A.; Beirne, P.V.; Verbeek, J.H. Alcohol and drug screening of occupational drivers for preventing injury. Cochrane Database Syst. Rev. 2009, 2, CD006566. [Google Scholar] [CrossRef] [PubMed]
- Musshoff, F.; Madea, B. New trends in hair analysis and scientific demands on validation and technical notes. Forensic Sci. Int. 2007, 165, 204–215. [Google Scholar] [CrossRef]
- Shin, I.; Choi, H.; Kang, S.; Kim, J.; Park, Y.; Yang, W. Detection of l-Methamphetamine and l-Amphetamine as Selegiline Metabolites. J. Anal. Toxicol. 2021, 45, 99–104. [Google Scholar] [CrossRef]
- van Nuijs, A.L.N.; Castiglioni, S.; Tarcomnicu, I.; Postigo, C.; de Alda, M.L.; Neels, H.; Zuccato, E.; Barcelo, D.; Covaci, A. Illicit drug consumption estimations derived from wastewater analysis: A critical review. Sci. Total Environ. 2011, 409, 3564–3577. [Google Scholar] [CrossRef] [PubMed]
- Ort, C.; van Nuijs, A.L.N.; Berset, J.D.; Bijlsma, L.; Castiglioni, S.; Covaci, A.; de Voogt, P.; Emke, E.; Fatta-Kassinos, D.; Griffiths, P.; et al. Spatial differences and temporal changes in illicit drug use in Europe quantified by wastewater analysis. Addiction 2014, 109, 1338–1352. [Google Scholar] [CrossRef]
- de la Torre, R.; Farré, M.; Navarro, M.; Pacifici, R.; Zuccaro, P.; Pichini, S. Clinical pharmacokinetics of amphetamine and related substances: Monitoring in conventional and non-conventional matrices. Clin. Pharmacokinet. 2004, 43, 157–185. [Google Scholar] [CrossRef]
- Fallon, J.K.; Kicman, A.T.; Henry, J.A.; Milligan, P.J.; Cowan, D.A.; Hutt, A.J. Stereospecific Analysis and Enantiomeric Disposition of 3,4-Methylenedioxymethamphetamine (Ecstasy) in Humans. Clin. Chem. 1999, 45, 1058–1069. [Google Scholar] [CrossRef]
- Substance Abuse and Mental Health Services Administration (SAMHSA). Mandatory Guidelines for Federal Workplace Drug Testing Programs—Authorized Testing Panels; Federal Register: Washington, DC, USA, 2025; Volume 90. Available online: https://www.federalregister.gov/documents/2025/01/16/2025-00425/mandatory-guidelines-for-federal-workplace-drug-testing-programs-authorized-testing-panels (accessed on 21 September 2025).
- Saitman, A.; Park, H.D.; Fitzgerald, R.L. False-positive interferences of common urine drug screen immunoassays: A review. J. Anal. Toxicol. 2014, 38, 387–396. [Google Scholar] [CrossRef]
- Sheng, W.; Sun, R.; Zhang, R.; Xu, P.; Wang, Y.; Xu, H.; Aa, J.; Wang, G.; Xie, Y. Identification of Biomarkers for Methamphetamine Exposure Time Prediction in Mice Using Metabolomics and Machine Learning Approaches. Metabolites 2022, 12, 1250. [Google Scholar] [CrossRef] [PubMed]
- EWDTS. European Guidelines for Workplace Drug Testing in Urine, Version 3.0; European Workplace Drug Testing Society: Huddinge, Sweden, 2022; Available online: https://www.scribd.com/document/658183463/European-Guidelines-for-Workplace-Drug-Testing-in-Urine-2022 (accessed on 21 September 2025).
- Marchei, E.; Pellegrini, M.; Pacifici, R.; Durgbanshi, A.; Zuccaro, P.; Pichini, S. Determination of atomoxetine and its metabolites in conventional and non-conventional biological matrices by liquid chromatography–tandem mass spectrometry. J. Pharm. Biomed. Anal. 2012, 60, 26–31. [Google Scholar] [CrossRef] [PubMed]
- Gilbert, R.B.; Peng, P.I.; Wong, D. A labetalol metabolite with analytical characteristics resembling amphetamines. J. Anal. Toxicol. 1995, 19, 84–86. [Google Scholar] [CrossRef]
- Kaddah, M.M.Y.; El Demellawy, M.A.; Talaat, W. Comprehensive analytical approaches: The use of LC-MS and IC-MS in modern pharmaceutical and biomedical sciences. Microchem. J. 2025, 216, 114780. [Google Scholar] [CrossRef]
- Cody, J.T. Precursor medications as a source of methamphetamine and/or amphetamine positive drug testing results. J. Occup. Environ. Med. 2002, 44, 435–450. [Google Scholar] [CrossRef]
- Meyer, M.R.; Maurer, H.H. Metabolism of designer drugs of abuse: An updated review. Curr. Drug Metab. 2010, 11, 468–482. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, M.; Carmo, H.; Costa, V.M.; Capela, J.P.; Pontes, H.; Remião, F.; Carvalho, F.; de Lourdes Bastos, M. Toxicity of amphetamines: An update. Arch. Toxicol. 2012, 86, 1167–1231. [Google Scholar] [CrossRef]
- Schwaninger, A.E.; Meyer, M.R.; Zapp, J.; Maurer, H.H. Investigations on the stereoselectivity of the phase II metabolism of the 3,4-methylenedioxyethylamphetamine (MDEA) metabolites 3,4-dihydroxyethylamphetamine (DHEA) and 4-hydroxy-3-methoxyethylamphetamine (HMEA). Toxicol. Lett. 2012, 212, 38–47. [Google Scholar] [CrossRef] [PubMed]
- Xiang, P.; Bu, J.; Qiao, Z.; Zhuo, X.Y.; Wu, H.J.; Shen, M. Identification of Methamphetamine Abuse and Selegiline Use: Chiral Analysis of Methamphetamine and Amphetamine in Urine. Fa Yi Xue Za Zhi 2017, 33, 599–603. [Google Scholar] [CrossRef]
- de la Torre, R.; Farré, M.; Roset, P.N.; Pizarro, N.; Abanades, S.; Segura, M.; Segura, J.; Camí, J. Human pharmacology of MDMA: Pharmacokinetics, metabolism, and disposition. Ther. Drug Monit. 2004, 26, 137–144. [Google Scholar] [CrossRef]
- Kalant, H. The pharmacology and toxicology of “ecstasy” (MDMA) and related drugs. CMAJ 2001, 165, 917–928. [Google Scholar]
- Pizarro, N.; Farré, M.; Pujadas, M.; Ma, A.; Roset, P.N.; Joglar, J.; de la Torre, R. Stereochemical analysis of 3,4-methylenedioxymethamphetamine and its main metabolites in human samples including the catechol-type metabolite (3,4-dihydroxymethamphetamine). Drug Metab. Dispos. 2004, 32, 1001–1007. [Google Scholar] [CrossRef]
- Ensslin, H.K.; Kovar, K.-A.; Maurer, H.H. Toxicological Detection of the Designer Drug 3,4-Methylenedioxyethylamphetamine (MDE, “Eve”) and Its Metabolites in Urine by GC–MS and Fluorescence Polarization Immunoassay. J. Chromatogr. B 1996, 683, 189–197. [Google Scholar] [CrossRef]
- Maurer, H.H.; Kraemer, T.; Springer, D.; Staack, R.F. Chemistry, pharmacology, toxicology, and hepatic metabolism of designer drugs of the amphetamine (ecstasy), piperazine, and pyrrolidinophenone types: A synopsis. Ther. Drug Monit. 2004, 26, 127–131. [Google Scholar] [CrossRef]
- Swortwood, M.J.; Hearn, W.L.; DeCaprio, A.P. Cross-reactivity of designer drugs, including cathinone derivatives, in commercial enzyme-linked immunosorbent assays. Drug Test. Anal. 2014, 6, 716–727. [Google Scholar] [CrossRef] [PubMed]
- Torrance, H.; Cooper, G. The detection of mephedrone (4-methylmethcathinone) in 4 fatalities in Scotland. Forensic Sci. Int. 2010, 202, e62–e63. [Google Scholar] [CrossRef]
- Kamata, H.T.; Shima, N.; Zaitsu, K.; Kamata, T.; Miki, A.; Nishikawa, M.; Katagi, M.; Tsuchihashi, H. Metabolism of the recently encountered designer drug, methylone, in humans and rats. Xenobiotica 2006, 36, 709–723. [Google Scholar] [CrossRef]
- Cheng, K.W.; Hsieh, C.M.; Chen, H.W.; Chi, P.C.; Yang, D.P.; Chan, S.H.; Chen, J.Y.; Hwa, H.L.; Fang, C.C.; Weng, T.I.; et al. Determination of Synthetic Cathinone α-Pyrrolidinovalero-phenone and Its Metabolite in Urine Using Solid-Phase Extraction and Gas Chromatography–Mass Spectrometry. Rapid Commun. Mass Spectrom. 2020, 34 (Suppl. S1), e8579. [Google Scholar] [CrossRef] [PubMed]
- Pascual-Caro, S.; Borrull, F.; Aguilar, C.; Calull, M. Determination of Synthetic Cathinones in Urine and Oral Fluid by Liquid Chromatography High-Resolution Mass Spectrometry and Low-Resolution Mass Spectrometry: A Method Comparison. Separations 2020, 7, 53. [Google Scholar] [CrossRef]
- Toennes, S.W.; Harder, S.; Schramm, M.; Niess, C.; Kauert, G.F. Pharmacokinetics of cathinone, cathine and norephedrine after the chewing of khat leaves. Br. J. Clin. Pharmacol. 2003, 56, 125–130. [Google Scholar] [CrossRef]
- Brenneisen, R.; Fisch, H.U.; Koelbing, U.; Geisshusler, S.; Kalix, P. Amphetamine-like effects in humans of the khat alkaloid cathinone. Br. J. Clin. Pharmacol. 1990, 30, 825–828. [Google Scholar] [CrossRef] [PubMed]
- Riss, J.; Cloyd, J.; Gates, J.; Collins, S. Benzodiazepines in epilepsy: Pharmacology and pharmacokinetics. Acta Neurol. Scand. 2008, 118, 69–86. [Google Scholar] [CrossRef]
- Griffin, C.E.; Kaye, A.M.; Bueno, F.R.; Kaye, A.D. Benzodiazepine pharmacology and central nervous system–mediated effects. Ochsner J. 2013, 13, 214–223. [Google Scholar]
- Longo, L.P.; Johnson, B. Addiction: Part I. Benzodiazepines—Side effects, abuse risk and alternatives. Am. Fam. Physician 2000, 61, 2121–2128. [Google Scholar]
- Greenblatt, D.J.; Divoll, M.; Abernethy, D.R.; Ochs, H.R.; Shader, R.I. Clinical pharmacokinetics of the newer benzodiazepines. Clin. Pharmacokinet. 1983, 8, 233–252. [Google Scholar] [CrossRef]
- Lader, M. Benzodiazepines revisited—Will we ever learn? Addiction 2011, 106, 2086–2109. [Google Scholar] [CrossRef] [PubMed]
- Soyka, M. Treatment of benzodiazepine dependence. N. Engl. J. Med. 2017, 376, 1147–1157. [Google Scholar] [CrossRef]
- Bachhuber, M.A.; Hennessy, S.; Cunningham, C.O.; Starrels, J.L. Increasing benzodiazepine prescriptions and overdose mortality in the United States, 1996–2013. Am. J. Public Health 2016, 106, 686–688. [Google Scholar] [CrossRef]
- Holbrook, A.M.; Crowther, R.; Lotter, A.; Cheng, C.; King, D. Meta-analysis of benzodiazepine use in the treatment of insomnia. CMAJ 2000, 162, 225–233. [Google Scholar]
- Glass, J.; Lanctôt, K.L.; Herrmann, N.; Sproule, B.A.; Busto, U.E. Sedative hypnotics in older people with insomnia: Meta-analysis of risks and benefits. BMJ 2005, 331, 1169. [Google Scholar] [CrossRef]
- Sateia, M.J. International Classification of Sleep Disorders-third edition: Highlights and modifications. Chest 2014, 146, 1387–1394. [Google Scholar] [CrossRef]
- Kripke, D.F. Risks of chronic hypnotic use. In Madame Curie Bioscience Database; Landes Bioscience: Austin, TX, USA, 2000. Available online: https://www.ncbi.nlm.nih.gov/books/NBK6630/ (accessed on 3 October 2025).
- Riess, S.; Muckensturm, A.; Eliot, E.; Gonçalves, R.; Roussel, O.; Cirimele, V.; Cirimele, V.; Cheze, M. Drug Facilitated Sexual Assault (DFSA) in a chemsex context: Challenges in determining imputability. Toxicol. Anal. Clin. 2025, 37, S43. [Google Scholar] [CrossRef]
- McHugh, R.K.; Votaw, V.R.; Trapani, E.W.; McCarthy, M.D. Prevalence and correlates of the misuse of Z-drugs and benzodiazepines in the National Survey on Drug Use and Health. Front. Psychiatry 2023, 14, 1129447. [Google Scholar] [CrossRef] [PubMed]
- European Union Drugs Agency (EUDA). EU Drug Market: New Psychoactive Substances—Distribution and Supply in Europe: Benzodiazepines. 27 June 2024. Available online: https://www.euda.europa.eu/publications/eu-drug-markets/new-psychoactive-substances/distribution-and-supply/benzodiazepines_en (accessed on 1 October 2025).
- European Union Drugs Agency (EUDA). European Drug Report 2023: Drug-Induced Deaths; EMCDDA: Lisbon, Portugal, 2023; Available online: https://euda.europa.eu/publications/european-drug-report/2023/drug-induced-deaths_en (accessed on 1 October 2025).
- Wang, L.L.; Ren, X.X.; He, Y.; Cui, G.F.; Liu, J.J.; Jia, J.; Cao, J.; Liu, Y.; Cong, B.; Wei, Z.W.; et al. Pharmacokinetics of Diazepam and Its Metabolites in Urine of Chinese Participants. Drugs RD 2022, 22, 43–50. [Google Scholar] [CrossRef] [PubMed]
- Khalid, S.; Rasool, M.F.; Imran, I.; Majeed, A.; Saeed, H.; Rehman, A.U.; Ashraf, W.; Ahmad, T.; Bin Jardan, Y.A.; Alqahtani, F. A Physiologically Based Pharmacokinetic Model for Predicting Diazepam Pharmacokinetics after Intravenous, Oral, Intranasal, and Rectal Applications. Pharmaceutics 2021, 13, 1480. [Google Scholar] [CrossRef] [PubMed]
- Luk, S.; Atayee, R.S.; Ma, J.D.; Best, B.M. Urinary diazepam metabolite distribution in a chronic pain population. J. Anal. Toxicol. 2014, 38, 135–142. [Google Scholar] [CrossRef] [PubMed]
- Burkat, P.M. Physiologically based pharmacokinetic and pharmacodynamic modelling of alprazolam: Implications for anxiety and addiction. Br. J. Clin. Pharmacol. 2023, 89, 2569–2581. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Liu, S.; Wolf, C.A.; Wolber, G.; Parr, M.K.; Bureik, M. Changes in Alprazolam Metabolism by CYP3A43 Mutants. Biomedicines 2022, 10, 3022. [Google Scholar] [CrossRef]
- Chèze, M.; Villain, M.; Pépin, G. Determination of bromazepam, clonazepam and metabolites after a single intake in urine and hair by LC-MS/MS. Application to forensic cases of drug facilitated crimes. Forensic Sci. Int. 2004, 145, 123–130. [Google Scholar] [CrossRef]
- Peter, J.-U.; Dieudonnè, P.; Zolk, O. Pharmacokinetics, Pharmacodynamics, and Side Effects of Midazolam: A Review and Case Example. Pharmaceuticals 2024, 17, 473. [Google Scholar] [CrossRef] [PubMed]
- Boulieu, R.; Lehmann, B.; Salord, F.; Fisher, C.; Morlet, D. Pharmacokinetics of midazolam and its main metabolite 1-hydroxymidazolam in intensive care patients. Eur. J. Drug Metab. Pharmacokinet. 1998, 23, 255–258. [Google Scholar] [CrossRef]
- Pan, R.N.; Hsiong, C.H.; Huang, P.W.; Tsai, W.S.; Li, W.Y.; Pao, L.H. Simultaneous quantification of triazolam and its metabolites in human urine by liquid chromatography-tandem mass spectrometry. J. Anal. Toxicol. 2011, 35, 170–175. [Google Scholar] [CrossRef][Green Version]
- Watanabe, S.; Vikingsson, S.; Astrand, A.; Auwarter, V.; Green, H.; Kronstrand, R. Metabolism of the benzodiazepines norflurazepam, flurazepam, fludiazepam and cinolazepam by human hepatocytes using high-resolution mass spectrometry and distinguishing their intake in authentic urine samples. Forensic Toxicol. 2020, 38, 79–94. [Google Scholar] [CrossRef]
- Nikfarjam, Z.; Doustkhah, E.; Zamani, F.; Brown, R.W. Pharmaceutical applications of 1,4-benzodiazepines. In Benzodiazepine-Based Drug Discovery; Elsevier: Amsterdam, The Netherlands, 2022; pp. 125–182. [Google Scholar] [CrossRef]
- Tóth, K.; Csukly, G.; Sirok, D.; Belic, A.; Kiss, Á.; Háfra, E.; Déri, M.; Menus, Á.; Bitter, I.; Monostory, K. Optimization of Clonazepam Therapy Adjusted to Patient’s CYP3A Status and NAT2 Genotype. Int. J. Neuropsychopharmacol. 2016, 19, pyw083. [Google Scholar] [CrossRef]
- DrugBank. Clonazepam (DB01068). DrugBank Online. Available online: https://go.drugbank.com/drugs/DB01068?utm_source=chatgpt.com (accessed on 1 October 2025).
- Kangas, L.; Breimer, D.D. Clinical Pharmacokinetics of Nitrazepam. Clin. Pharmacokinet. 1981, 6, 294–301. [Google Scholar] [CrossRef]
- Kangas, L. Urinary Elimination of Nitrazepam and its Main Metabolites. Acta Pharmacol. Toxicol. 1979, 45, 16–19. [Google Scholar] [CrossRef]
- Jullien, V.; Chhun, S.; Rey, E.; Dulac, O.; Tod, M.; Chiron, C.; Pons, G. Pharmacokinetics of Clobazam and N-desmethylclobazam in children in children with dravet syndrome receiving concomitant stiripentol and valproic acid. Clin. Pharmacokinet. 2015, 54, 105–118. [Google Scholar] [CrossRef] [PubMed]
- Jensen, H.S.; Nichol, K.; Lee, D.; Ebert, B. Clobazam and its active metabolite N-desmethylclobazam display significantly greater affinities for α2- versus α1-GABA(A)-receptor complexes. PLoS ONE 2014, 9, e88456. [Google Scholar] [CrossRef]
- Forsman, M.; Nyström, I.; Roman, M.; Berglund, L.; Ahlner, J.; Kronstrand, R. Urinary detection times and excretion patterns of flunitrazepam and its metabolites after a single oral dose. J. Anal. Toxicol. 2009, 33, 491–501. [Google Scholar] [CrossRef]
- Negrusz, A.; Moore, C.M.; Stockham, T.L.; Poiser, K.R.; Kern, J.L.; Palaparthy, R.; Le, N.L.; Janicak, P.G.; Levy, N.A. Elimination of 7-aminoflunitrazepam and flunitrazepam in urine after a single dose of Rohypnol. J. Forensic Sci. 2000, 45, 1031–1040. [Google Scholar] [CrossRef]
- Verstraete, A.G. Detection times of drugs of abuse in blood, urine, and oral fluid. Ther. Drug Monit. 2004, 26, 200–205. [Google Scholar] [CrossRef]
- Fluyau, D.; Ponnarasu, S.; Patel, P. Temazepam; Updated 2024 January 11. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2025. Available online: https://www.ncbi.nlm.nih.gov/books/NBK599496/?utm_source=chatgpt.com (accessed on 4 October 2025).
- Bardhi, K.; Coates, S.; Zhao, M.; Anyachebelu, M.C.; Lazarus, P. Identification of human hepatic UDP-glucuronosyltransferases involved in the glucuronidation of temazepam. Drug Metab. Dispos. 2025, 53, 100104. [Google Scholar] [CrossRef]
- Bouchette, D.; Akhondi, H.; Patel, P.; Quick, J. Zolpidem; Updated 2024 February 29. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2025. Available online: https://www.ncbi.nlm.nih.gov/books/NBK442008/?utm_source=chatgpt.com (accessed on 4 October 2025).
- Ebbens, M.M.; Verster, J.C. Clinical evaluation of zaleplon in the treatment of insomnia. Nat. Sci. Sleep 2010, 2, 115–126. [Google Scholar] [CrossRef][Green Version]
- European Medicines Agency (EMA). Summary of Product Characteristics. Available online: https://ec.europa.eu/health/documents/community-register/2015/20150527131872/anx_131872_en.pdf (accessed on 1 October 2025).
- Fernandez, C.; Martin, C.; Gimenez, F.; Farinotti, R. Clinical pharmacokinetics of zopiclone. Clin. Pharmacokinet. 1995, 29, 431–441. [Google Scholar] [CrossRef] [PubMed]
- Brielmaier, B.D. Eszopiclone (Lunesta): A new nonbenzodiazepine hypnotic agent. Proc. Bayl. Univ. Med. Cent. 2006, 19, 54–59. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Najib, J. Eszopiclone, a nonbenzodiazepine sedative-hypnotic agent for the treatment of transient and chronic insomnia. Clin. Ther. 2006, 28, 491–516. [Google Scholar] [CrossRef]
- Greenblatt, D.J.; Wright, C.E. Clinical Pharmacokinetics of Alprazolam: Therapeutic Implications. Clin. Pharmacokinet. 1993, 24, 453–471. [Google Scholar] [CrossRef]
- Crouch, D.J.; Rollins, D.E.; Canfield, D.V.; Andrenyak, D.M.; Schulties, J.E. Quantitation of alprazolam and alpha-hydroxyalprazolam in human plasma using liquid chromatography electrospray ionization MS-MS. J. Anal. Toxicol. 1999, 23, 479–485. [Google Scholar] [CrossRef]
- Gunja, N. The Clinical and Forensic Toxicology of Z-Drugs. J. Med. Toxicol. 2013, 9, 155–162. [Google Scholar] [CrossRef]
- Pires, B.; Catarro, G.; Soares, S.; Gonçalves, J.; Rosado, T.; Barroso, M.; Araujo, A.R.T.S.; Gallardo, E. Volumetric Absorptive Microsampling in Toxicology. Toxics 2025, 13, 25. [Google Scholar] [CrossRef] [PubMed]
- Fang, Z.; Zhang, H.; Guo, J.; Guo, J. Overview of therapeutic drug monitoring and clinical practice. Talanta 2024, 266 Pt 1, 124996. [Google Scholar] [CrossRef]
- Del Mar Ramírez Fernández, M.; Wille, S.M.; di Fazio, V.; Kummer, N.; Hill, V.; Samyn, N. Detection of Benzodiazepines and z-Drugs in Hair Using an UHPLC-MS/MS Validated Method: Application to Workplace Drug Testing. Ther. Drug Monit. 2015, 37, 600–608. [Google Scholar] [CrossRef] [PubMed]
- Xu, R.; Zhu, J. Unveiling the dark matter of the metabolome: A narrative review of bioinformatics tools for LC-HRMS-based compound annotation. Talanta 2025, 295, 128327. [Google Scholar] [CrossRef]
- Liu, Y.; Liu, X.Z.; Guo, Z.S.; Xiang, P.; Yan, H. Development and validation of a LC-MS/MS method for the detection of 38 benzodiazepines and 2 Z-drugs in blood. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 2025, 1266, 124772. [Google Scholar] [CrossRef]
- Marin, S.J.; Coles, R.; Merrell, M.; McMillin, G.A. Quantitation of benzodiazepines in urine, serum, plasma, and meconium by LC-MS-MS. J. Anal. Toxicol. 2008, 32, 491–498. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hadland, S.E.; Levy, S. Objective Testing: Urine and Other Drug Tests. Child Adolesc. Psychiatr. Clin. N. Am. 2016, 25, 549–565. [Google Scholar] [CrossRef] [PubMed]
- Kintz, P. Hair Analysis in Forensic Toxicology: An Updated Review with a Special Focus on Pitfalls. Curr. Pharm. Des. 2017, 23, 5480–5486. [Google Scholar] [CrossRef]
- Arantes, A.C.F.; da Cunha, K.F.; Cardoso, M.S.; Oliveira, K.D.; Costa, J.L. Development and validation of quantitative analytical method for 50 drugs of antidepressants, benzodiazepines and opioids in oral fluid samples by liquid chromatography–tandem mass spectrometry. Forensic Toxicol. 2021, 39, 179–197. [Google Scholar] [CrossRef]
- de Campos, E.G.; da Costa, B.R.B.; dos Santos, F.S.; Monedeiro, F.; Nogueira Rabelo Alves, M.; Ramos Santos, W.J., Jr.; Spinosa De Martinis, B. Alternative matrices in forensic toxicology: A critical review. Forensic Toxicol. 2021, 40, 1–18. [Google Scholar] [CrossRef]
- Mandrioli, R.; Mercolini, L.; Raggi, M.A. Benzodiazepine Metabolism: An Analytical Perspective. Curr. Drug Metab. 2008, 9, 827–844. [Google Scholar] [CrossRef]
- Øiestad, E.L.; Øiestad, Å.M.L.; Middelkoop, G.; Brochmann, G.W.; Thaulow, C.H.; Vindenes, V. Comparative Study of Postmortem Concentrations of Benzodiazepines and Z-Hypnotics in Several Different Matrices. J. Anal. Toxicol. 2023, 47, 287–298. [Google Scholar] [CrossRef]
- Maust, D.T.; Lin, L.A.; Blow, F.C. Benzodiazepine Use and Misuse Among Adults in the United States. Psychiatr. Serv. 2018, 70, 97–106. [Google Scholar] [CrossRef]
- Votaw, V.R.; Geyer, R.; Rieselbach, M.M.; McHugh, R.K. The Epidemiology of Benzodiazepine Misuse: A Systematic Review. Drug Alcohol. Depend. 2019, 200, 95–114. [Google Scholar] [CrossRef] [PubMed]
- Ho, T.T.; Noble, M.; Tran, B.A.; Sunjić, K.; Gupta, S.V.; Turgeon, J.; Crutchley, R.D. Clinical Impact of the CYP2C19 Gene on Diazepam for the Management of Alcohol Withdrawal Syndrome. J. Pers. Med. 2023, 13, 285. [Google Scholar] [CrossRef] [PubMed]
- Dhaliwal, J.S.; Rosani, A.; Saadabadi, A. Diazepam; Updated 2023 August 28. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2025. Available online: https://www.ncbi.nlm.nih.gov/books/NBK537022/ (accessed on 5 October 2025).
- Zubiaur, P.; Figueiredo-Tor, L.; Villapalos-García, G.; Soria-Chacartegui, P.; Navares-Gómez, M.; Novalbos, J.; Matas, M.; Calleja, S.; Mejía-Abril, G.; Román, M.; et al. Association between CYP2C19 and CYP2B6 phenotypes and the pharmacokinetics and safety of diazepam. Biomed. Pharmacother. 2022, 155, 113747. [Google Scholar] [CrossRef]
- Jones, A.W.; Holmgren, A. Concentrations of Diazepam and Nordiazepam in 1000 Blood Samples from Apprehended Drivers—Therapeutic Use or Abuse of Anxiolytics? J. Pharm. Pract. 2013, 26, 198–203. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.L.; Ren, X.X.; He, Y.; Cui, G.F.; Wei, Z.W.; Jia, J.; Cao, J.; Liu, Y.; Cong, B.; Niu, Y.; et al. Study on the Pharmacokinetics of Diazepam and Its Metabolites in Blood of Chinese People. Eur. J. Drug Metab. Pharmacokinet. 2020, 45, 477–485. [Google Scholar] [CrossRef] [PubMed]
- Thalib, H.I.; Damanhory, A.A.; Shaikh, A.H.; Haidar, S.; Khan, S.; Jamal, A.; Abdelghany, O.A. Forensic toxicology of benzodiazepines: Neuropharmacological effects, analytical challenges, and emerging detection strategies. Front. Toxicol. 2025, 7, 2025. [Google Scholar] [CrossRef]
- Kamal, H.; Gandhi, V.; Akil, L.; Al-Tannak, N.F.; Rattray, N.J.W.; Khadra, I. Development and Validation of an LC-MS/MS Method for the Simultaneous Determination of Alprazolam, Bromazepam, Clonazepam, Diazepam and Flunitrazpam in Human Urine and Its Application to Samples from Suspected Drug Abusers. Molecules 2025, 30, 3451. [Google Scholar] [CrossRef]
- Dinis-Oliveira, R.J. Metabolic Profile of Flunitrazepam: Clinical and Forensic Toxicological Aspects. Drug Metab. Lett. 2017, 11, 14–20. [Google Scholar] [CrossRef] [PubMed]
- Zhuo, Y.; Xiang, P.; Wu, J.; Wang, X. Segmental hair analysis for flunitrazepam and 7-aminoflunitrazepam in users: A comparison to existing literature. Forensic Sci. Res. 2020, 7, 299–307. [Google Scholar] [CrossRef]
- Strano Rossi, S.; Anzillotti, L.; Castrignanò, E.; Frison, G.; Zancanaro, F.; Chiarotti, M. UHPLC-MS/MS and UHPLC-HRMS identification of zolpidem and zopiclone main urinary metabolites and method development for their toxicological determination. Drug Test. Anal. 2014, 6, 226–233. [Google Scholar] [CrossRef]
- Miyaguchi, H.; Kuwayama, K. Enantioselective determination of (R)-zopiclone and (S)-zopiclone (eszopiclone) in human hair by micropulverized extraction and chiral liquid chromatography/high resolution mass spectrometry. J. Chromatogr. A 2017, 1519, 55–63. [Google Scholar] [CrossRef]
- Ohno-Shosaku, T.; Maejima, T.; Kano, M. Endogenous cannabinoids mediate retrograde signals from depolarized postsynaptic neurons to presynaptic terminals. Neuron 2001, 29, 729–738. [Google Scholar] [CrossRef]
- Lu, H.C.; Mackie, K. An Introduction to the Endogenous Cannabinoid System. Biol. Psychiatry 2016, 79, 516–525. [Google Scholar] [CrossRef]
- Blebea, N.M.; Pricopie, A.I.; Vlad, R.A.; Hancu, G. Phytocannabinoids: Exploring Pharmacological Profiles and Their Impact on Therapeutical Use. Int. J. Mol. Sci. 2024, 25, 4204. [Google Scholar] [CrossRef]
- Heal, D.J.; Gosden, J.; Smith, S.L. A critical assessment of the abuse, dependence and associated safety risks of naturally occurring and synthetic cannabinoids. Front. Psychiatry 2024, 15, 1322434. [Google Scholar] [CrossRef] [PubMed]
- U.S. Food & Drug Administration. FDA and Cannabis: Research and Drug Approval Process. Available online: https://www.fda.gov/news-events/public-health-focus/fda-and-cannabis-research-and-drug-approval-process (accessed on 30 September 2025).
- Lipnik-Štangelj, M.; Razinger, B. A regulatory take on cannabis and cannabinoids for medicinal use in the European Union. Arh. Hig. Rada Toksikol. 2020, 71, 12–18. [Google Scholar] [CrossRef] [PubMed]
- Le Foll, B.; Tang, V.M.; Rueda, S.; Trick, L.V.; Boileau, I. Cannabis use disorder: From neurobiology to treatment. J. Clin. Investig. 2024, 134, e172887. [Google Scholar] [CrossRef]
- Safi, K.; Sobieraj, J.; Błaszkiewicz, M.; Żyła, J.; Salata, B.; Dzierżanowski, T. Tetrahydrocannabinol and Cannabidiol for Pain Treatment-An Update on the Evidence. Biomedicines 2024, 12, 307. [Google Scholar] [CrossRef] [PubMed]
- Hansen, J.S.; Gustavsen, S.; Roshanisefat, H.; Kant, M.; Biering-Sørensen, F.; Andersen, C.; Olsson, A.; Chow, H.H.; Asgari, N.; Hansen, J.R.; et al. Cannabis-Based Medicine for Neuropathic Pain and Spasticity-A Multicenter, Randomized, Double-Blinded, Placebo-Controlled Trial. Pharmaceuticals 2023, 16, 1079. [Google Scholar] [CrossRef]
- Azadvari, M.; Pourshams, M.; Guitynavard, F.; Emami-Razavi, S.Z.; Taftian-Banadkouki, E.; Ghajarzade, M.; Rastkar, M. Cannabinoids for spasticity in patients with multiple sclerosis: A systematic review and meta-analysis. Mult. Scler. J. Exp. Transl. Clin. 2024, 10, 20552173241282379. [Google Scholar] [CrossRef]
- Hoch, E.; Volkow, N.D.; Friemel, C.M.; Lorenzetti, V.; Freeman, T.P.; Hall, W. Cannabis, cannabinoids and health: A review of evidence on risks and medical benefits. Eur. Arch. Psychiatry Clin. Neurosci. 2025, 275, 281–292. [Google Scholar] [CrossRef]
- Sic, A.; George, C.; Gonzalez, D.F.; Tseriotis, V.-S.; Knezevic, N.N. Cannabinoids in Chronic Pain: Clinical Outcomes, Adverse Effects and Legal Challenges. Neurol. Int. 2025, 17, 141. [Google Scholar] [CrossRef]
- Onaemo, V.N.; Fawehinmi, T.O.; D’Arcy, C. Comorbid Cannabis Use Disorder with Major Depression and Generalized Anxiety Disorder: A Systematic Review with Meta-analysis of Nationally Representative Epidemiological Surveys. J. Affect. Disord. 2021, 281, 467–475. [Google Scholar] [CrossRef]
- Hasin, D.S.; Sarvet, A.L.; Cerdá, M. US adult illicit cannabis use, cannabis use disorder, and medical marijuana laws: 1991–1992 to 2012–2013. JAMA Psychiatry 2017, 74, 579–588. [Google Scholar] [CrossRef]
- Feingold, D.; Livne, O.; Rehm, J.; Lev-Ran, S. Probability and correlates of transition from cannabis use to DSM-5 cannabis use disorder: Results from a large-scale nationally representative study. Drug. Alcohol. Rev. 2020, 39, 142–151. [Google Scholar] [CrossRef]
- Lev-Ran, S.; Roerecke, M.; Le Foll, B.; George, T.P.; McKenzie, K.; Rehm, J. The Association between Cannabis Use and Depression: A Systematic Review and Meta-Analysis of Longitudinal Studies. Psychol. Med. 2014, 44, 797–810. [Google Scholar] [CrossRef]
- Weinstock, L.M.; Gaudiano, B.A.; Wenze, S.J.; Epstein-Lubow, G.; Miller, I.W. Demographic and clinical characteristics associated with comorbid cannabis use disorders (CUDs) in hospitalized patients with bipolar I disorder. Compr. Psychiatry 2016, 65, 57–62. [Google Scholar] [CrossRef] [PubMed]
- GBD 2016 Risk Factors Collaborators. Global, regional, and national comparative risk assessment of 84 behavioural, environmental and occupational, and metabolic risks or clusters of risks, 1990–2016: A systematic analysis for the Global Burden of Disease Study 2016. Lancet 2017, 390, 1345–1422. [Google Scholar] [CrossRef]
- Hasin, D.S. US epidemiology of cannabis use and associated problems. Neuropsychopharmacol 2018, 43, 195–212. [Google Scholar] [CrossRef]
- The SEED Team. Cannabis use disorder among adults: A living systematic review. In Cannabis Evidence; The SEED Team: Shizuoka City, Japan, 2024; Available online: https://www.cannabisevidence.org/evidence-syntheses/adult-cannabis-use-disorder/ (accessed on 4 October 2025).
- Bahji, A.; Stephenson, C.; Tyo, R.; Hawken, E.R.; Seitz, D.P. Prevalence of Cannabis Withdrawal Symptoms Among People with Regular or Dependent Use of Cannabinoids: A Systematic Review and Meta-analysis. JAMA Netw. Open 2020, 3, e202370. [Google Scholar] [CrossRef]
- Choi, N.G.; Moore, J.; Choi, B.Y. Cannabis use disorder and substance use treatment among U.S. adults. J. Subst. Use Addict. Treat. 2024, 167, 209486. [Google Scholar] [CrossRef] [PubMed]
- Reber, J.D.; Karschner, E.L.; Seither, J.Z.; Knittel, J.L.; Walterscheid, J.P. Screening and confirmation methods for the qualitative identification of nine phytocannabinoids in urine by LC-MS/MS. Clin Biochem. 2021, 98, 54–62. [Google Scholar] [CrossRef] [PubMed]
- Gaunitz, F.; Kieliba, T.; Thevis, M.; Mercer-Chalmers-Bender, K. Solid-phase extraction-liquid chromatography-tandem mass spectrometry method for the qualitative analysis of 61 synthetic cannabinoid metabolites in urine. Drug Test. Anal. 2020, 12, 27–40. [Google Scholar] [CrossRef]
- Ballotari, M.; Truver, M.T.; Sojin, N.A.; Agliano, L.A.; Parimoo, R.; Hoyer, J.L.; Goodin, A.J.; Varma, D.S.; Chronister, C.W.; Roussos-Ross, K.; et al. Quantitative analysis of Δ8- and Δ9-tetrahydrocannabinol metabolites and isomers: A rapid assay in urine by LC-MS/MS. Anal. Bioanal. Chem. 2025, 417, 1507–1518. [Google Scholar] [CrossRef] [PubMed]
- Koch, C.D.; Xu, L.; Curtis, S.A.; Roberts, J.D.; Bunch, D.R.; El-Khoury, J.M. Urinary cannabinoid mass spectrometry profiles differentiate dronabinol from cannabis use. Clin. Chim. Acta. 2020, 510, 515–521. [Google Scholar] [CrossRef]
- Myoli, A.; Choene, M.; Kappo, A.P.; Madala, N.E.; van der Hooft, J.J.J.; Tugizimanaet, F. Charting the Cannabis plant chemical space with computational metabolomics. Metabolomics 2024, 20, 62. [Google Scholar] [CrossRef] [PubMed]
- Frei, P.; Frauchiger, S.; Scheurer, E.; Mercer-Chalmers-Bender, K. Quantitative determination of five cannabinoids in blood and urine by gas chromatography tandem mass spectrometry applying automated on-line solid phase extraction. Drug Test. Anal. 2022, 14, 1223–1233. [Google Scholar] [CrossRef]
- Sempio, C.; Almaraz-Quinones, N.; Jackson, M.; Zhao, W.; Wang, G.S.; Liu, Y.; Leehey, M.; Knupp, K.; Klawitter, J.; Christians, U.; et al. Simultaneous Quantification of 17 Cannabinoids by LC-MS-MS in Human Plasma. J. Anal. Toxicol. 2022, 46, 383–392. [Google Scholar] [CrossRef] [PubMed]
- Aizpurua-Olaizola, O.; Zarandona, I.; Ortiz, L.; Navarro, P.; Etxebarria, N.; Usobiaga, A. Simultaneous quantification of major cannabinoids and metabolites in human urine and plasma by HPLC-MS/MS and enzyme-alkaline hydrolysis. Drug Test. Anal. 2017, 9, 626–633. [Google Scholar] [CrossRef]
- Shimadzu Corporation. Analysis of THC and Its Metabolites in Urine Using Triple Quadrupole LCMS-8050 [Application Note]; Shimadzu Corporation: Kyoto, Japan, 2016; Available online: https://www.shimadzu.com/an/apl/14071/index.html (accessed on 4 October 2025).
- Wei, B.; Wang, L.; Blount, B.C. Analysis of Cannabinoids and Their Metabolites in Human Urine. Anal. Chem. 2015, 87, 10183–10187. [Google Scholar] [CrossRef]
- Nelson, K.M.; Bisson, J.; Singh, G.; Graham, J.G.; Chen, S.N.; Friesen, J.B.; Dahlin, J.L.; Niemitz, M.; Walters, M.A.; Pauli, G.F. The Essential Medicinal Chemistry of Cannabidiol (CBD). J. Med. Chem. 2020, 63, 12137–12155. [Google Scholar] [CrossRef]
- Mazur, A.; Lichti, C.F.; Prather, P.L.; Zielinska, A.K.; Bratton, S.M.; Gallus-Zawada, A.; Finel, M.; Miller, G.P.; Radomińska-Pandya, A.; Moran, J.H. Characterization of human hepatic and extrahepatic UDP-glucuronosyltransferase enzymes involved in the metabolism of classic cannabinoids. Drug Metab. Dispos. 2009, 37, 1496–1504. [Google Scholar] [CrossRef]
- Beers, J.L.; Authement, A.K.; Isoherranen, N.; Jackson, K.D. Cytosolic Enzymes Generate Cannabinoid Metabolites 7-Carboxycannabidiol and 11-Nor-9-carboxytetrahydrocannabinol. ACS Med. Chem. Lett. 2023, 14, 614–620. [Google Scholar] [CrossRef]
- Huestis, M.A. Pharmacokinetics and metabolism of the plant cannabinoids, delta9-tetrahydrocannabinol, cannabidiol and cannabinol. Handb. Exp. Pharmacol. 2005, 168, 657–690. [Google Scholar] [CrossRef]
- Huestis, M.A. Human cannabinoid pharmacokinetics. Chem. Biodivers. 2007, 4, 1770–1804. [Google Scholar] [CrossRef]
- Smith, R.T.; Gruber, S.A. Contemplating cannabis? The complex relationship between cannabinoids and hepatic metabolism resulting in the potential for drug-drug interactions. Front. Psychiatry 2023, 13, 1055481. [Google Scholar] [CrossRef]
- Watanabe, K.; Yamaori, S.; Funahashi, T.; Kimura, T.; Yamamoto, I. Cytochrome P450 enzymes involved in the metabolism of tetrahydrocannabinols and cannabinol by human hepatic microsomes. Life Sci. 2007, 80, 1415–1419. [Google Scholar] [CrossRef]
- Nasrin, S.; Watson, C.J.W.; Perez-Paramo, Y.X.; Lazarus, P. Cannabinoid Metabolites as Inhibitors of Major Hepatic CYP450 Enzymes, with Implications for Cannabis-Drug Interactions. Drug Metab. Dispos. 2021, 49, 1070–1080. [Google Scholar] [CrossRef]
- Nasrin, S.; Watson, C.J.W.; Bardhi, K.; Fort, G.; Chen, G.; Lazarus, P. Inhibition of UDP-Glucuronosyltransferase Enzymes by Major Cannabinoids and Their Metabolites. Drug Metab. Dispos. 2021, 49, 1081–1089. [Google Scholar] [CrossRef]
- Yabut, K.C.B.; Winnie Wen, Y.; Simon, K.T.; Isoherranen, N. CYP2C9, CYP3A and CYP2C19 metabolize Δ9-tetrahydrocannabinol to multiple metabolites but metabolism is affected by human liver fatty acid binding protein (FABP1). Biochem. Pharmacol. 2024, 228, 116191. [Google Scholar] [CrossRef] [PubMed]
- Qian, L.; Zhang, T.; Dinh, J.; Paine, M.F.; Zhou, Z. Physiologically Based Pharmacokinetic Modeling of Cannabidiol, Delta-9-Tetrahydrocannabinol, and Their Metabolites in Healthy Adults After Administration by Multiple Routes. Clin. Transl. Sci. 2025, 18, e70119. [Google Scholar] [CrossRef]
- Chen, S.; Li, Y.; Li, X.; Wu, Q.; Puig, M.; Moulin, F.; Gingrich, J.; Guo, L. Metabolism and liver toxicity of cannabidiol. J. Environ. Sci. Health C Toxicol. Carcinog. 2024, 42, 238–254. [Google Scholar] [CrossRef]
- Molnar, A.; Fu, S. Techniques and technologies for the bioanalysis of Sativex®, metabolites and related compounds. Bioanalysis 2016, 8, 829–845. [Google Scholar] [CrossRef] [PubMed]
- Thomsen, R.; Nielsen, L.M.; Holm, N.B.; Rasmussen, H.B.; Linnet, K.; INDICES Consortium. Synthetic cannabimimetic agents metabolized by carboxylesterases. Drug Test. Anal. 2015, 7, 565–576. [Google Scholar] [CrossRef] [PubMed]
- Bardhi, K.; Coates, S.; Watson, C.J.W.; Lazarus, P. Cannabinoids and drug metabolizing enzymes: Potential for drug-drug interactions and implications for drug safety and efficacy. Expert. Rev. Clin. Pharmacol. 2022, 15, 1443–1460. [Google Scholar] [CrossRef]
- Millar, S.A.; Stone, N.L.; Bellman, Z.D.; Yates, A.S.; England, T.J.; O’Sullivan, S.E. A systematic review of cannabidiol dosing in clinical populations. Br. J. Clin. Pharmacol. 2019, 85, 1888–1900. [Google Scholar] [CrossRef] [PubMed]
- Devinsky, O.; Patel, A.D.; Thiele, E.A.; Wong, M.H.; Appleton, R.; Harden, C.L.; Greenwood, S.; Morrison, G.; Sommerville, K. GWPCARE1 Part A Study Group. Randomized, dose-ranging safety trial of cannabidiol in Dravet syndrome. Neurology 2018, 90, e1204–e1211. [Google Scholar] [CrossRef] [PubMed]
- Scheidweiler, K.B.; Desrosiers, N.A.; Huestis, M.A. Simultaneous quantification of free and glucuronidated cannabinoids in human urine by liquid chromatography tandem mass spectrometry. Clin. Chim. Acta 2012, 413, 1839–1847. [Google Scholar] [CrossRef]
- Scheunemann, A.; Elsner, K.; Germerott, T.; Groppa, S.; Hess, C.; Miederer, I.; Poplawski, A.; Röhrich, J. Identification of Potential Distinguishing Markers for the Use of Cannabis-Based Medicines or Street Cannabis in Serum Samples. Metabolites 2021, 11, 316. [Google Scholar] [CrossRef]
- Karschner, E.L.; Darwin, W.D.; Goodwin, R.S.; Wright, S.; Huestis, M.A. Plasma cannabinoid pharmacokinetics following controlled oral delta9-tetrahydrocannabinol and oromucosal cannabis extract administration. Clin. Chem. 2011, 57, 66–75. [Google Scholar] [CrossRef]
- Rubin, A.; Lemberger, L.; Warrick, P.; Crabtree, R.E.; Sullivan, H.; Rowe, H.; Obermeyer, B.D. Physiologic disposition of nabilone, a cannabinol derivative, in man. Clin. Pharmacol. Ther. 1977, 22, 85–91. [Google Scholar] [CrossRef]
- Navarro, G.; Moreno, E.; Aymerich, M.; Marcellino, D.; McCormick, P.J.; Mallol, J.; Cortés, A.; Casadó, V.; Canela, E.I.; Ortiz, J.; et al. Direct involvement of σ-1 receptors in the dopamine D1 receptor-mediated effects of cocaine. Proc. Natl. Acad. Sci. USA 2010, 107, 18676–18681. [Google Scholar] [CrossRef]
- Narayanan, S.; Mesangeau, C.; Poupaert, J.H.; McCurdy, C.R. Sigma receptors and cocaine abuse. Curr. Top. Med. Chem. 2011, 11, 1128–1150. [Google Scholar] [CrossRef] [PubMed]
- Lutfallah, S.C.; Brown, E.; Spillers, N.J.; Tandon, A.; Kelkar, R.A.; Ahmadzadeh, S.; Viswanath, O.; Varrassi, G.; Shekoohi, S.; Kaye, A.D. Topical Cocaine Hydrochloride Nasal Solution: Anesthetic and Surgical Considerations. Cureus 2023, 15, e42804. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- UNODC. Cocaine—A Spectrum of Products, Cocaine Insights 2; UNODC: Vienna, Austria, 2021. [Google Scholar]
- Carbone, M.G.; Maremmani, I. Chronic Cocaine Use and Parkinson’s Disease: An Interpretative Model. Int. J. Environ. Res. Public Health 2024, 21, 1105. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Schwartz, E.K.C.; Wolkowicz, N.R.; De Aquino, J.P.; MacLean, R.R.; Sofuoglu, M. Cocaine Use Disorder (CUD): Current Clinical Perspectives. Subst. Abus. Rehabil. 2022, 13, 25–46. [Google Scholar] [CrossRef] [PubMed]
- UNODC. World Drug Report 2025. Available online: https://www.unodc.org/unodc/data-and-analysis/world-drug-report-2025.html (accessed on 15 October 2025).
- Chen, D.; Sun, Y.; Li, X.; Yin, Z. Global burden on drug use disorders from 1990 to 2021 and projections to 2046. Front. Public Health 2025, 13, 1550518. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Zhu, D.T.; Bajaj, S.S.; Sen, A. Methamphetamine and Cocaine Overdose Deaths in the United States, 1999–2023. Subst. Use Misuse 2025, 60, 1946–1949. [Google Scholar] [CrossRef] [PubMed]
- Spencer, M.R.; Miniño, A.M.; Garnett, M.F. Co-involvement of Opioids in Drug Overdose Deaths Involving Cocaine and Psychostimulants, 2011–2021. NCHS Data Brief. 2023, 474, 1–8. [Google Scholar] [PubMed]
- Traccis, F.; Minozzi, S.; Trogu, E.; Vacca, R.; Vecchi, S.; Pani, P.P.; Agabio, R. Disulfiram for the treatment of cocaine dependence. Cochrane Database Syst. Rev. 2024, 1, CD007024. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Martinotti, G.; Pettorruso, M.; Montemitro, C.; Spagnolo, P.A.; Acuti Martellucci, C.; Di Carlo, F.; Fanella, F.; di Giannantonio, M.; Brainswitch Study Group. Repetitive transcranial magnetic stimulation in treatment-seeking subjects with cocaine use disorder: A randomized, double-blind, sham-controlled trial. Prog. Neuropsychopharmacol. Biol. Psychiatry 2022, 116, 110513. [Google Scholar] [CrossRef] [PubMed]
- Moody, D.E.; Cheever, M.L. Evaluation of immunoassays for semiquantitative detection of cocaine and metabolites or heroin and metabolites in extracts of sweat patches. J. Anal. Toxicol. 2001, 25, 190–197. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Jeffcoat, A.R.; Perez-Reyes, M.; Hill, J.M.; Sadler, B.M.; Cook, C.E. Cocaine disposition in humans after intravenous injection, nasal insufflation (snorting), or smoking. Drug Metab. Dispos. 1989, 17, 153–159. [Google Scholar] [CrossRef] [PubMed]
- Cone, E.J. Pharmacokinetics and pharmacodynamics of cocaine. J. Anal. Toxicol. 1995, 19, 459–478. [Google Scholar] [CrossRef] [PubMed]
- Foltin, R.W.; Fischman, M.W. Smoked and intravenous cocaine in humans: Acute tolerance, cardiovascular and subjective effects. J. Pharmacol. Exp. Ther. 1991, 257, 247–261. [Google Scholar] [CrossRef] [PubMed]
- Barnett, G.; Hawks, R.; Resnick, R. Cocaine pharmacokinetics in humans. J. Ethnopharmacol. 1981, 3, 353–366. [Google Scholar] [CrossRef] [PubMed]
- Nayak, P.K.; Misra, A.L.; Mulé, S.J. Physiological disposition and biotransformation of (3H) cocaine in acutely and chronically treated rats. J. Pharmacol. Exp. Ther. 1976, 196, 556–569. [Google Scholar] [CrossRef] [PubMed]
- Javaid, J.I.; Fischman, M.W.; Schuster, C.R.; Dekirmenjian, H.; Davis, J.M. Cocaine plasma concentration: Relation to physiological and subjective effects in humans. Science 1978, 202, 227–228. [Google Scholar] [CrossRef] [PubMed]
- Glauser, J.; Queen, J.R. An overview of non-cardiac cocaine toxicity. J. Emerg. Med. 2007, 32, 181–186. [Google Scholar] [CrossRef] [PubMed]
- Dean, R.A.; Christian, C.D.; Sample, R.H.; Bosron, W.F. Human liver cocaine esterases: Ethanol-mediated formation of ethylcocaine. FASEB J. 1991, 5, 2735–2739. [Google Scholar] [CrossRef] [PubMed]
- Kalasinsky, K.S.; Bosy, T.Z.; Schmunk, G.A.; Ang, L.; Adams, V.; Gore, S.B.; Smialek, J.; Furukawa, Y.; Guttman, M.; Kish, S.J. Regional distribution of cocaine in postmortem brain of chronic human cocaine users. J. Forensic Sci. 2000, 45, 1041–1048. [Google Scholar] [CrossRef] [PubMed]
- Brzezinski, M.R.; Abraham, T.L.; Stone, C.L.; Dean, R.A.; Bosron, W.F. Purification and characterization of a human liver cocaine carboxylesterase that catalyzes the production of benzoylecgonine and the formation of cocaethylene from alcohol and cocaine. Biochem. Pharmacol. 1994, 48, 1747–1755. [Google Scholar] [CrossRef] [PubMed]
- Schindler, C.W.; Goldberg, S.R. Accelerating Cocaine Metabolism as an Approach to the Treatment of Cocaine Abuse and Toxicity. Future Med. Chem. 2012, 4, 163–175. [Google Scholar] [CrossRef]
- Hearn, W.L.; Flynn, D.D.; Hime, G.W.; Rose, S.; Cofino, J.C.; Mantero-Atienza, E.; Wetli, C.V.; Mash, D.C. Cocaethylene: A unique cocaine metabolite displays high affinity for the dopamine transporter. J. Neurochem. 1991, 56, 698–701. [Google Scholar] [CrossRef] [PubMed]
- Jatlow, P. Cocaethylene: Pharmacologic activity and clinical significance. Ther. Drug Monit. 1993, 15, 533–536. [Google Scholar] [CrossRef] [PubMed]
- National Institute on Drug Abuse (NIDA). Cocaine Research Report; National Institute on Drug Abuse (NIDA): North Bethesda, MD, USA, 2023. [Google Scholar]
- Carney, S.; Wolf, C.E.; Tarnai-Moak, L.; Poklis, A. Evaluation of two enzyme immunoassays for the detection of the cocaine metabolite benzoylecgonine in 1398 urine specimens. J. Clin. Lab. Anal. 2012, 26, 130–135. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Johansen, S.S.; Bhatia, H.M. Quantitative analysis of cocaine and its metabolites in whole blood and urine by high-performance liquid chromatography coupled with tandem mass spectrometry. J. Chrom B 2007, 852, 338–344. [Google Scholar] [CrossRef]
- Mulé, S.J.; Casella, G.A. Confirmation and quantitation of cocaine, benzoylecgonine, ecgonine methyl ester in human urine by GC/MS. J. Anal. Toxicol. 1988, 12, 153–155. [Google Scholar] [CrossRef] [PubMed]
- Makhdoom, H.S.; Afzal, S.; Sultana, K.; Shah, S.N.H.; Mujahid, M.; Hassan, Z.U.; Munir, F.; Jahan, F.; Abbas, Z.; Abid, A.I.; et al. SPE-UHPLC-ESI-MS/MS Analysis of Cocaine and Its Metabolites in Conventional and Alternative Biological Specimens: Application to Real Samples. ACS Omega 2024, 9, 23355–23363. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Romero-Noreña, A.; Contreras-Martinez, H.J.; Mendivil-Perez, M. Toxicology of anhydroecgonine methyl ester: A systematic review of a cocaine pyrolysis product. Toxicol. Rep. 2024, 13, 101690. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Pergolizzi, J.; Breve, F.; Magnusson, P.; LeQuang, J.A.K.; Varrassi, G. Cocaethylene: When Cocaine and Alcohol Are Taken Together. Cureus 2022, 14, e22498. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Mattes, C.E.; Lynch, T.J.; Singh, A.; Bradley, R.M.; Kellaris, P.A.; Brady, R.O.; Dretchen, K.L. Therapeutic use of butyrylcholinesterase for cocaine intoxication. Toxicol. Appl. Pharmacol. 1997, 145, 372–380. [Google Scholar] [CrossRef] [PubMed]
- Pragst, F.; Balikova, M.A. State of the art in hair analysis for detection of drug and alcohol abuse. Clin. Chim. Acta 2006, 370, 17–49. [Google Scholar] [CrossRef] [PubMed]
- Substance Abuse and Mental Health Services Administration (SAMHSA). Mandatory Guidelines for Federal Workplace Drug Testing Programs. Fed. Regist. 2017, 82, 7920–7970. Available online: https://www.federalregister.gov/documents/2023/10/12/2023-21734/mandatory-guidelines-for-federal-workplace-drug-testing-programs (accessed on 10 October 2025).
- Broussard, L.A. Chapter 15–Challenges in Confirmation Testing for Drugs of Abuse, Accurate Results in the Clinical Laboratory, 2nd ed.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 243–256. [Google Scholar] [CrossRef]
- Nickley, J.; Pesce, A.J.; Krock, K. A sensitive assay for urinary cocaine metabolite benzoylecgonine shows more positive results and longer half-lives than those using traditional cut-offs. Drug Test. Anal. 2017, 9, 1214–1216. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Kim, J.A.; Ptolemy, A.S.; Melanson, S.E.; Janfaza, D.R.; Ross, E.L. The clinical impact of a false-positive urine cocaine screening result on a patient’s pain management. Pain. Med. 2015, 16, 1073–1076. [Google Scholar] [CrossRef] [PubMed][Green Version]
- North Carolina Office of the Chief Medical Examiner. SOP-403: Cocaine and Metabolites by LC-MS/MS; North Carolina Office of the Chief Medical Examiner: Raleigh, NC, USA, 2019. [Google Scholar]
- Jones, A.W. Forensic Drug Profile: Cocaethylene. J. Anal. Toxicol. 2019, 43, 155–160. [Google Scholar] [CrossRef] [PubMed]
- Jacob, P., 3rd; Lewis, E.R.; Elias-Baker, B.A.; Jones, R.T. A pyrolysis product, anhydroecgonine methyl ester (methylecgonidine), is in the urine of cocaine smokers. J. Anal. Toxicol. 1990, 14, 353–357. [Google Scholar] [CrossRef] [PubMed]
- Koren, G.; Klein, J.; Forman, R.; Graham, K. Hair analysis of cocaine: Differentiation between systemic exposure and external contamination. J. Clin. Pharmacol. 1992, 32, 671–675. [Google Scholar] [CrossRef] [PubMed]
- Domino, E.F. Taming the ketamine tiger. 1965. Anesthesiology 2010, 113, 678–684. [Google Scholar] [CrossRef] [PubMed]
- Zanos, P.; Gould, T.D. Mechanisms of ketamine action as an antidepressant. Mol. Psychiatry 2018, 23, 801–811. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Berman, R.M.; Cappiello, A.; Anand, A.; Oren, D.A.; Heninger, G.R.; Charney, D.S.; Krystal, J.H. Antidepressant effects of ketamine in depressed patients. Biol. Psychiatry 2000, 47, 351–354. [Google Scholar] [CrossRef] [PubMed]
- Zarate, C.A., Jr.; Singh, J.B.; Carlson, P.J.; Brutsche, N.E.; Ameli, R.; Luckenbaugh, D.A.; Charney, D.S.; Manji, H.K. A randomized trial of an N-methyl-D-aspartate antagonist in treatment-resistant major depression. Arch. Gen. Psychiatry 2006, 63, 856–864. [Google Scholar] [CrossRef] [PubMed]
- Duman, R.S.; Aghajanian, G.K. Synaptic dysfunction in depression: Potential therapeutic targets. Science 2012, 338, 68–72. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Li, Y.; Shen, R.; Wen, G.; Ding, R.; Du, A.; Zhou, J.; Dong, Z.; Ren, X.; Yao, H.; Zhao, R.; et al. Effects of Ketamine on Levels of Inflammatory Cytokines IL-6, IL-1β, and TNF-α in the Hippocampus of Mice Following Acute or Chronic Administration. Front. Pharmacol. 2017, 8, 139. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Morgan, C.J.; Muetzelfeldt, L.; Curran, H.V. Consequences of chronic ketamine self-administration upon neurocognitive function and psychological wellbeing: A 1-year longitudinal study. Addiction 2010, 105, 121–133, Erratum in Addiction 2010, 105, 766. [Google Scholar] [CrossRef] [PubMed]
- Lankenau, S.E.; Sanders, B. Patterns of ketamine use among young injection drug users. J. Psychoact. Drugs. 2007, 39, 21–29. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Jansen, K.L.R. Ketamine: Dreams and Realities; Multidisciplinary Association for Psychedelic Studies: San Jose, CA, USA, 2000. [Google Scholar]
- UNODC. World Drug Report 2023; UNODC: Vienna, Austria, 2023. [Google Scholar]
- Clements, J.A.; Nimmo, W.S.; Grant, I.S. Bioavailability, pharmacokinetics, and analgesic activity of ketamine in humans. J. Pharm. Sci. 1982, 71, 539–542. [Google Scholar] [CrossRef] [PubMed]
- Peltoniemi, M.A.; Hagelberg, N.M.; Olkkola, K.T.; Saari, T.I. Ketamine: A Review of Clinical Pharmacokinetics and Pharmacodynamics in Anesthesia and Pain Therapy. Clin. Pharmacokinet. 2016, 55, 1059–1077. [Google Scholar] [CrossRef] [PubMed]
- Hijazi, Y.; Boulieu, R. Protein binding of ketamine and its active metabolites to human serum. Eur. J. Clin. Pharmacol. 2002, 58, 37–40. [Google Scholar] [CrossRef] [PubMed]
- Zanos, P.; Moaddel, R.; Morris, P.J.; Georgiou, P.; Fischell, J.; Elmer, G.I.; Alkondon, M.; Yuan, P.; Pribut, H.J.; Singh, N.S.; et al. NMDAR inhibition-independent antidepressant actions of ketamine metabolites. Nature 2016, 533, 481–486. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Chu, P.S.; Ma, W.K.; Wong, S.C.; Chu, R.W.; Cheng, C.H.; Wong, S.; Tse, J.M.; Lau, F.L.; Yiu, M.K.; Man, C.W. The destruction of the lower urinary tract by ketamine abuse: A new syndrome? BJU Int. 2008, 102, 1616–1622. [Google Scholar] [CrossRef] [PubMed]
- Olney, J.W.; Labruyere, J.; Price, M.T. Pathological changes induced in cerebrocortical neurons by phencyclidine and related drugs. Science 1989, 244, 1360–1362. [Google Scholar] [CrossRef] [PubMed]
- Harun, N.; Anderson, R.A.; Miller, E.I. Validation of an enzyme-linked immunosorbent assay screening method and a liquid chromatography-tandem mass spectrometry confirmation method for the identification and quantification of ketamine and norketamine in urine samples from Malaysia. J. Anal. Toxicol. 2009, 33, 310–321. [Google Scholar] [CrossRef] [PubMed]
- Thomann, J.; Kraus, S.; Erne, L.; Vogt, S.B.; Liechti, M.E.; Luethi, D. Development and Validation of a Rapid LC-MS/MS Method for Plasma Analysis of Ketamine, Norketamine, Dehydronorketamine, and Hydroxynorketamine. Biomed. Chromatogr. 2025, 39, e70171. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Porpiglia, N.; Musile, G.; Bortolotti, F.; De Palo, E.F.; Tagliaro, F. Chiral separation and determination of ketamine and norketamine in hair by capillary electrophoresis. Forensic Sci. Int. 2016, 266, 304–310. [Google Scholar] [CrossRef] [PubMed]
- Malinovsky, J.M.; Servin, F.; Cozian, A.; Lepage, J.Y.; Pinaud, M. Ketamine and norketamine plasma concentrations after i.v., nasal and rectal administration in children. Br. J. Anaesth. 1996, 77, 203–207. [Google Scholar] [CrossRef] [PubMed]
- Adamowicz, P.; Kala, M. Urinary Excretion Rates of Ketamine and Norketamine Following Therapeutic Ketamine Administration: Method and Detection Window Considerations. J. Anal. Toxicol. 2005, 29, 376–382. [Google Scholar] [CrossRef] [PubMed]
- Seibert, E.; Hubner, E.-M.; Schmid, M.G. Chiral Separation and Determination of Enantiomer Elution Order of Novel Ketamine Derivatives Using CE-UV and HPLC-UV-ORD. Separations 2025, 12, 44. [Google Scholar] [CrossRef]
- Zanos, P.; Moaddel, R.; Morris, P.J.; Riggs, L.M.; Highland, J.N.; Georgiou, P.; Pereira, E.F.R.; Albuquerque, E.X.; Thomas, C.J.; Zarate, C.A., Jr.; et al. Ketamine and Ketamine Metabolite Pharmacology: Insights into Therapeutic Mechanisms. Pharmacol. Rev. 2018, 70, 621–660, Erratum in Pharmacol Rev. 2018, 70, 879. https://doi.org/10.1124/pr.116.015198err. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Cuvelier, E.; Khazri, H.; Lecluse, C.; Hennart, B.; Amad, A.; Roche, J.; Tod, M.; Vaiva, G.; Cottencin, O.; Odou, P.; et al. Therapeutic Drug Monitoring and Pharmacogenetic Testing as Guides to Psychotropic Drug Dose Adjustment: An Observational Study. Pharmaceuticals 2024, 17, 21. [Google Scholar] [CrossRef]
- Morelato, M.; Beavis, A.; Kirkbride, P.; Roux, C. Forensic Applications of Desorption Electrospray Ionisation Mass Spectrometry (DESI-MS). Forensic Sci. Int. 2013, 226, 10–21. [Google Scholar] [CrossRef]
- Butler, K.E.; Baker, E.S. A High-Throughput Ion Mobility Spectrometry–Mass Spectrometry Screening Method for Opioid Profiling. J. Am. Soc. Mass Spectrom. 2022, 33, 1904–1913. [Google Scholar] [CrossRef]
- Guleria, A.; Kumar, A.; Kumar, U.; Raj, R.; Kumar, D. NMR-Based Metabolomics: An Exquisite and Facile Method for Evaluating Therapeutic Efficacy and Screening Drug Toxicity. Curr. Top. Med. Chem. 2018, 18, 1827–1849. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Pasin, D.; Skinnider, M.A.; Liigand, J.; Kleis, J.N.; Brown, D.; Oler, E.; Sajed, T.; Gautam, V.; Harrison, S.; et al. Deep Learning-Enabled MS/MS Spectrum Prediction Facilitates Automated Identification of Novel Psychoactive Substances. Anal. Chem. 2023, 95, 18326–18334. [Google Scholar] [CrossRef]
- Vincenti, F.; Montesano, C.; Di Ottavio, F.; Gregori, A.; Compagnone, D.; Sergi, M.; Dorrestein, P.C. Molecular Networking: A Useful Tool for the Identification of New Psychoactive Substances in Seizures by LC-HRMS. Front. Chem. 2020, 8, 572952. [Google Scholar] [CrossRef] [PubMed]
- Szeremeta, M.; Pietrowska, K.; Niemcunowicz-Janica, A.; Kretowski, A.; Ciborowski, M. Applications of Metabolomics in Forensic Toxicology and Forensic Medicine. Int. J. Mol. Sci. 2021, 22, 3010. [Google Scholar] [CrossRef]
- Ghanbari, R.; Sumner, S. Using Metabolomics to Investigate Biomarkers of Drug Addiction. Trends Mol. Med. 2018, 24, 197–205. [Google Scholar] [CrossRef]
- Ghanbari, R.; Li, Y.; Pathmasiri, W.; McRitchie, S.; Etemadi, A.; Pollock, J.D.; Poustchi, H.; Rahimi-Movaghar, A.; Amin-Esmaeili, M.; Roshandel, G.; et al. Metabolomics Reveals Biomarkers of Opioid Use Disorder. Transl. Psychiatry 2021, 11, 103. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.; Jang, W.J.; Shakya, R.; Choi, B.; Jeong, C.H.; Lee, S. Current Understanding of Methamphetamine-Associated Metabolic Changes Revealed by the Metabolomics Approach. Metabolites 2019, 9, 195. [Google Scholar] [CrossRef]
- Jie, Z.; Qin, S.; Zhang, W.; Wang, J.; Lu, J.; Qin, G.; Hou, X.; Xu, P. Metabolic Profile Analysis of Designer Benzodiazepine Etizolam in Zebrafish and Human Liver Microsomes. Metabolites 2023, 13, 699. [Google Scholar] [CrossRef]





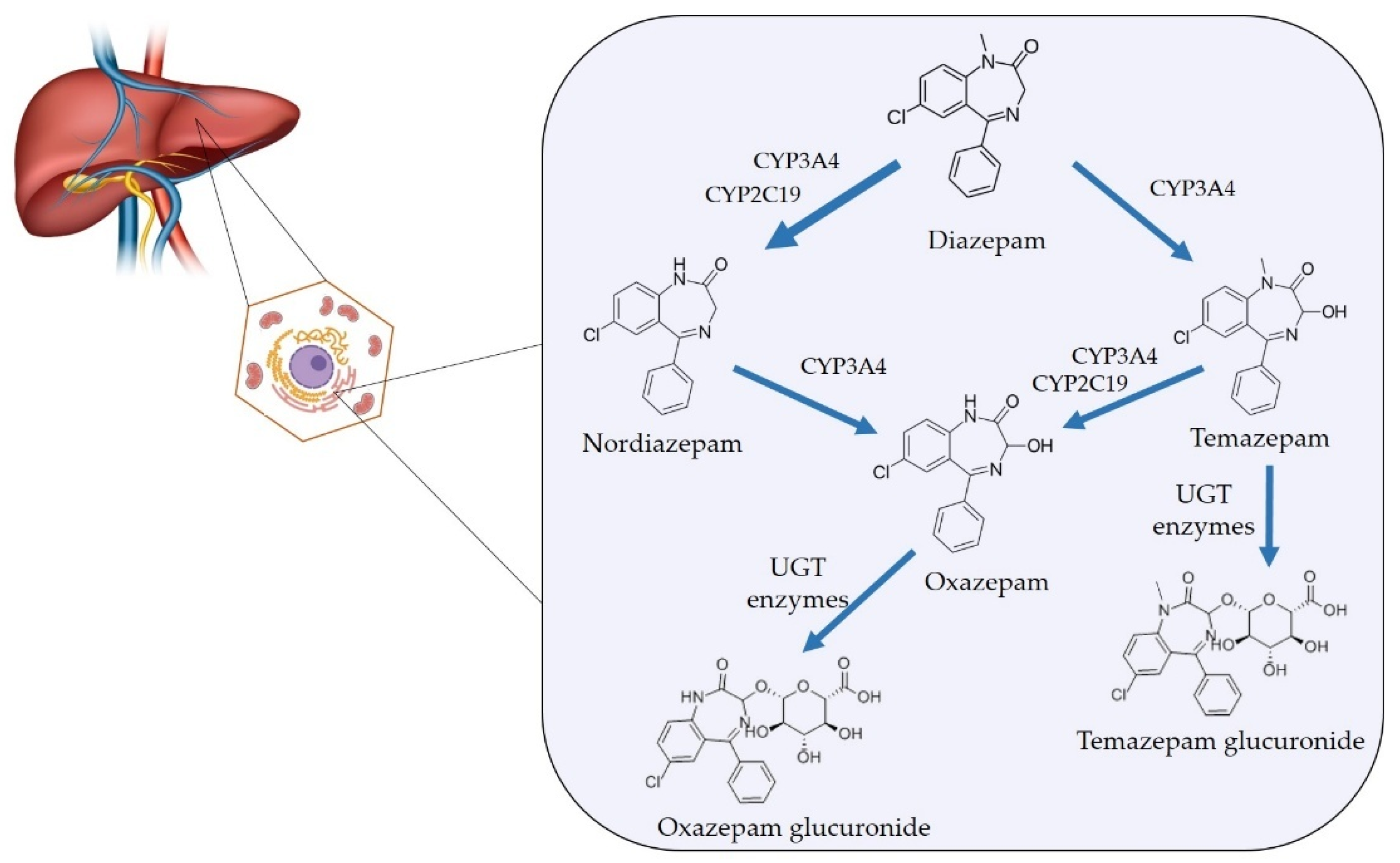
| Primary Molecule | Metabolite |
|---|---|
| Codeine | Codeine-6-β-glucuronide |
| Morphine | Morphine-6-β-glucuronide |
| - | 6-Monoacetylmorphine (6-MAM) † |
| Hydrocodone | Norhydrocodone |
| Dihydrocodeine | – |
| Hydromorphone | Hydromorphone-3-β-glucuronide |
| Oxycodone | Noroxycodone |
| Oxymorphone | Oxymorphone-3-β-glucuronide, Noroxymorphone |
| Meperidine | Normeperidine |
| Methadone | 2-ethylidene-1,5-dimethyl-3,3-diphenylpyrrolidine (EDDP) |
| Propoxyphene | Norpropoxyphene |
| Tramadol | O-Desmethyltramadol |
| Tapentadol | Tapentadol-β-glucuronide, N-Desmethyltapentadol |
| Buprenorphine | Norbuprenorphine, Norbuprenorphine glucuronide |
| Naloxone | Naloxone glucuronide |
| Fentanyl | Norfentanyl |
| Opioid | Pharmacokinetics | Biomarkers of Use/Recommended Matrices and Analytical Methods | Urine Cut-Off Limits (Screening/Confirmatory, ng/mL) | Major Analytical Challenges |
|---|---|---|---|---|
| Morphine |
|
| Initial: 2000 ng/mL (group opiate screen) Confirmatory: 4000 ng/mL (morphine) |
|
| Codeine |
|
| Initial: 2000 ng/mL (group opiate screen) Confirmatory: 2000 ng/mL (codeine) |
|
| Heroin |
|
| Initial/Confirmatory: 10 ng/mL (6-MAM) |
|
| Oxycodone/Oxymorphone/Hydrocodone/Hydromorphone |
|
| Oxycodone/Oxymorphon: Initial: 100 ng/mL; Confirmatory: 100 ng/mL Hydrocodone/Hydromorphone: Initial: 300 ng/mL; Confirmatory: 100 ng/mL |
|
| Methadone |
|
| Initial: 300 ng/mL (typical immunoassay screening) Confirmatory: 300 ng/mL (methadone and/or EDDP; program-dependent). Some EDDP assays use 100 ng/mL as qualitative threshold |
|
| Context/Program | Initial Screen Cut-Off (ng/mL) | Confirmatory Cut-Off (ng/mL) | Analytes Covered by Screen | Common Cross-Reactants/Interferents | Differentiation |
|---|---|---|---|---|---|
| Clinical/workplace (typical) | 500–1000 (as amphetamine equivalents) | By LC-MS/MS, laboratory-defined (commonly 150–250) | Amphetamine, methamphetamine (variable cross-reactivity to MDMA/MDEA) | Pseudoephedrine/ephedrine, bupropion, atomoxetine, phentermine, metoprolol, labetalol metabolite (1-methyl-3-phenylpropylamine), ofloxacin, moxifloxacin | False positives most frequent with bupropion and labetalol metabolite; false negatives possible for ring-substituted ATS (e.g., MDMA) [134,137]. |
| US Federal programs (SAMHSA) | 500 (amphetamine class) | 250 (amphetamine class) | Targets amphetamine class; MDMA/MDA often on separate panels | Pseudoephedrine/ephedrine; phenethylamines; DMAA (reported); selegiline → L-methamphetamine | Lower confirmatory cut-off reduces spurious positives; enantiomeric analysis helps distinguish L-methamphetamine from therapeutic selegiline vs. illicit D-methamphetamine [132,139]. |
| Pediatric/adolescent screens | Assay-dependent (commonly 500) | Per laboratory | Amphetamine class | Aripiprazole; trazodone; promethazine; chlorpromazine (assay-dependent) | False positives with aripiprazole in youth; confirmation by GC-MS or LC-MS/MS is mandatory [136]. |
| MDMA/MDEA (ring-substituted ATS) | Often poor cross-reactivity on amphetamine-calibrated screens | Targeted LC-MS/MS confirmation | MDMA/MDA/MDEA (on dedicated assays) | — | Generic amphetamine immunoassays may miss MDMA/MDEA; targeted panels required for detection [46]. |
| Drug | Pharmacokinetic Properties | Major Metabolic Pathways and Key Metabolites | Notes |
|---|---|---|---|
| Amphetamine | Rapid oral absorption; Tmax ≈ 3 h; t1/2 ≈ 9–14 h; renal clearance strongly pH-dependent (acidic urine ↑ excretion) | Oxidative deamination and p-hydroxylation mainly via CYP2D6; conjugation with sulfate and glucuronic acid | D-isomer is more potent on CNS than L-isomer; urinary acidification increases clearance; detection of high AM/MA ratio suggests primary amphetamine use |
| Methamphetamine | Rapid absorption (oral, nasal, intravenous); Tmax ≈ 2–3 h; t1/2 ≈ 10–12 h (influenced by urinary pH) | N-demethylation (CYP2D6) → amphetamine; para-hydroxylation → 4-hydroxymethamphetamine and 4-hydroxyamphetamine → conjugation | MA/AM ratio > 2–3 indicates methamphetamine intake; alkaline urine prolongs elimination; chiral analysis distinguishes therapeutic L-methamphetamine (selegiline/decongestants) from illicit D-isomer |
| Lisdexamfetamine | Prodrug; enzymatic hydrolysis in blood; Tmax ≈ 1 h for parent, 3.5 h for released d-amphetamine; t1/2 ≈ 8–13 h (as d-amphetamine) | Enzymatic hydrolysis to d-amphetamine and L-lysine; non-CYP-mediated metabolism | Lower abuse potential; absence of L-isomer confirms therapeutic origin; metabolite profile identical to d-amphetamine |
| MDMA (3,4-Methylenedioxymethamphetamine) | Oral Tmax ≈ 2 h; t1/2 ≈ 6–10 h; extensive first-pass metabolism | O-demethylenation (CYP2D6) → MDA; O-methylation → HMMA, HMA; subsequent conjugation | MDMA/MDA ratio > 1 = recent use; CYP2D6 polymorphism or SSRI co-therapy delays MDA formation |
| MDA (3,4-Methylenedioxyamphetamine) | Oral Tmax ≈ 1.5–2 h; t1/2 ≈ 8–12 h | O-demethylenation, hydroxylation, and conjugation → HMA, HMAA | Active metabolite of MDMA; predominance of MDA without MDMA indicates direct use |
| MDEA (3,4-Methylenedioxyethylamphetamine) | Oral Tmax ≈ 1.5–2 h; t1/2 ≈ 6–9 h | O-demethylenation → DHEA; O-methylation → HMEA; minor N-deethylation → MDA | Detection of HMEA confirms MDEA intake; concurrent presence of MDEA and MDMA indicates polydrug use |
| Mephedrone (4-MMC, 4-methylmethcathinone) | Rapid absorption (oral/nasal); Tmax ≈ 1–2 h; t1/2 ≈ 6–8 h | N-demethylation → nor-mephedrone; reduction → dihydromephedrone; para-hydroxylation | Parent compound unstable; detection indicates illicit use; no approved therapeutic application; detection = illicit exposure |
| Methylone (bk-MDMA) | Oral Tmax ≈ 1–2 h; t1/2 ≈ 6–9 h | N-demethylation → 3,4-methylenedioxycathinone; O-methylation → HMMC | Overlaps with MDMA metabolites; requires HRMS for differentiation |
| Methcathinone (ephedrone) | Oral Tmax ≈ 1–2 h; t1/2 ≈ 4–6 h | N-demethylation, reduction, and hydroxylation; conjugation to glucuronides | Structurally related to cathinone; rapid metabolism limits detection; illicitly synthesized from ephedrine |
| Cathinone (natural from Catha edulis) | Rapid absorption; short t1/2 ≈ 1.5 h | Reduction → cathine (norpseudoephedrine) and norephedrine | Naturally occurring stimulant; detection of cathine/norephedrine profile distinguishes natural vs. synthetic cathinones |
| Analyte(s) | Typical Parent/Metabolite Ratio | Influence of Urine pH | Forensic Notes |
|---|---|---|---|
| Methamphetamine (MA); Amphetamine (AM) | MA:AM usually >2–3:1 after methamphetamine intake | Acidic urine → faster excretion, lower ratio; Alkaline urine → slower excretion, higher ratio | Detection of AM may result from MA metabolism; High MA with some AM = methamphetamine use; High AM with little/no MA = primary amphetamine use [27,37]. |
| Amphetamine (AM) (parent drug) | AM >> MA (if any MA detected at all) | Urine pH affects excretion rate of AM (faster in acidic, slower in alkaline urine), but no conversion to MA | Predominant AM with minimal/no MA indicates direct amphetamine intake [46]. |
| Hydroxylated metabolites (e.g., 4-hydroxyamphetamine, 4-hydroxymethamphetamine) | Generally low compared to parent drug(s) | Also pH-dependent excretion | Supportive markers of metabolism but less critical for distinguishing AM vs. MA intake [140]. |
| Chiral analysis | - | - | Consider chiral analysis when L-methamphetamine exposure (e.g., from selegiline therapy or decongestants) is plausible [143]. |
| Compound | Main Origin/Metabolism | Interpretive Significance |
|---|---|---|
| MDMA (3,4-Methylenedioxymethamphetamine, “Ecstasy”) | Parent drug; metabolized to MDA, HMMA | High MDMA with little metabolite → acute/recent use [42] |
| MDA (3,4-Methylenedioxyamphetamine) | Active metabolite of MDMA; also a parent drug | Presence with MDMA → expected metabolite. Predominant MDA may suggest direct MDA ingestion [43] |
| HMMA (4-Hydroxy-3-methoxymethamphetamine) | Major MDMA metabolite (O-demethylenation + O-methylation) | Typically higher in urine at later time points → late/ongoing excretion; supportive for MDMA intake [44] |
| MDEA (3,4-Methylenedioxyethylamphetamine, “Eve”) | Structurally related drug; metabolized to MDA | Detection indicates separate ingestion (polydrug or substitution) [45] |
| DHEA (3,4-Dihydroxyethylamphetamine) | Hydroxylated metabolite of MDEA | Confirms MDEA metabolism; interpret cautiously due to instability [46] |
| HMEA (4-Hydroxy-3-methoxyethylamphetamine) | O-methylated metabolite of MDEA | Supportive marker of MDEA use; usually detected later in urine [46] |
| Parent Cathinone | Major Metabolites | Analytical Challenges | Differentiation |
|---|---|---|---|
| Mephedrone (4-MMC, 4-methylmethcathinone) | Nor-mephedrone (N-demethylated), 4-hydroxytolylmephedrone, dihydromephedrone | Structural similarity to other cathinones; extensive metabolism reduces parent detectability; instability in stored samples. | No therapeutic use; detection implies illicit exposure. Risk of misclassification with other cathinones without targeted LC-MS/MS [140,150]. |
| Methylone (bk-MDMA, 3,4-methylenedioxy-N-methylcathinone) | 3,4-methylenedioxycathinone (N-demethylated), HMMC (4-hydroxy-3-methoxymethcathinone) | Overlap with MDMA metabolites complicates interpretation; rapid metabolism; need for high-resolution MS. | Misattribution as MDMA/MDEA intake possible if only metabolite profile is assessed. Requires HRMS or targeted transitions [140,151]. |
| Ethylone (bk-MDEA, 3,4-methylenedioxy-N-ethylcathinone) | MDEC (3,4-methylenedioxycathinone, N-deethylated), HMEEC (hydroxy-methoxyethylcathinone) | Difficult differentiation from methylone and MDEA use; low stability in biological matrices. | No approved therapeutic analogue; may be misinterpreted as MDEA ingestion without confirmatory HRMS [140,151]. |
| α-PVP (alpha-pyrrolidinovalerophenone, “Flakka”) | Hydroxylated α-PVP, keto-reduced metabolites | Strong structural overlap with other pyrrolidinophenones; poor chromatographic separation; need for LC-HRMS/MS confirmation. | Lacks therapeutic indication; detection is highly specific for illicit/designer use [152,153]. |
| Cathinone (natural, from khat) | Cathine (norpseudoephedrine), norephedrine | Parent unstable; low concentrations in biological specimens; co-use with synthetic cathinones complicates interpretation. | Only naturally occurring cathinone with traditional use; differentiation from synthetic analogues requires precise metabolite profiling [154,155]. |
| Finding | Interpretive Significance |
|---|---|
| Predominance of D-methamphetamine | Indicative of illicit or prescription D-methamphetamine use [139]. |
| Predominance of L-methamphetamine | Supports exposure from selegiline metabolism or intranasal decongestants [46,127]. |
| Detection of D-amphetamine only (e.g., from lisdexamfetamine) | Consistent with therapeutic adherence [46,127]. |
| Enantiomeric profiles interpreted with MA/AM ratio, urinary pH, and time since intake | Provides contextual confirmation; prevents misattribution [130]. |
| Drug | Pharmacokinetic Properties | Major Metabolic Pathways and Key Metabolites |
|---|---|---|
| BENZODIAZEPINES | ||
| Diazepam | Tmax ≈ 1–2 h Long T1/2 for parent drug ≈ 30–60 h; | CYP-mediated (CYP3A4, CYP2C19) N-demethylation to desmethyldiazepam (nordiazepam) (active) → oxazepam (active) → glucuronides; CYP-mediated N-hydroxylation to temazepam (active) → oxazepam → glucuronides |
| Alprazolam | Tmax ≈ 1–2 h T1/2 ≈ 9–16 h (prolonged in elderly and in hepatic impairment) | CYP3A4 oxidation to α-hydroxyalprazolam (active, low levels), 4-hydroxyalprazolam and minor inactive metabolites |
| Bromazepam | Tmax ≈ 1–3 h (oral) T1/2 ≈ 10–20 h (variable; prolonged in elderly) | Oxidation to 3-hydroxybromazepam and other metabolites and subsequent conjugation; urinary detection of bromazepam and its major metabolite is conducted after hydrolysis of the glucuronide conjugates |
| Midazolam | Tmax ≈ 0.5–1.5 h (oral) T1/2 ≈ 1.5–6 h (single dose; prolonged in ICU/renal/hepatic impairment) | CYP3A4 hydroxylation to 1-hydroxymidazolam (major metabolite) and 4-hydroxymidazolam and subsequent biotransformation to glucuronides that are often measured in the urine |
| Triazolam | Tmax ≈ 0.5–2 h T1/2 ≈ 1.5–5.5 h | Hydroxylation by CYP3A to α-hydroxytriazolam (active) and 4-hydroxytriazolam and conjugation to inactive metabolites |
| Cinolazepam | Tmax ≈ 1–2 h (oral); variable T1/2 (limited data) | Metabolized to N-(hydroxyethyl) cinolazepam and glucuronides |
| Lorazepam | Tmax ≈ 1–2 h T1/2 ≈ 10–20 h (variable) | Direct glucuronidation to lorazepam-glucuronide (inactive). |
| Clonazepam | Tmax ≈ 1–4 h (varies with formulation); T1/2 ≈ 30–60 h (wide intersubject variability) | Nitro reduction to 7-aminoclonazepam and subsequent N-acetylation to 7-acetamidoclonazepam; minor hydroxylation products |
| Nitrazepam | Tmax ≈ 1–2 h; good oral absorption T1/2 ≈ 16–48 h | Nitro reduction to 7-aminonitrazepam and subsequent N-acetylation to 7-acetamidonitrazepam (major urinary metabolites); no clinically significant active metabolites |
| Clobazam | Tmax ≈ 1–2 h T1/2 ≈ 36–42 h (parent drug); T1/2 ≈ 59–74 h (active metabolite) | Oxidative N-demethylation by CYP450 enzymes (CYP3A4, CYP2C19) to N-desmethylclobazam (active); hydroxylation and glucuronidation |
| Flunitrazepam | Tmax ≈ 1–2 h T1/2 ≈ 18–26 h (parent drug); Active metabolite is detectable in urine for days even weeks | Nitro-reduction to 7-aminoflunitrazepam and related desmethyl metabolites; conjugation → urinary metabolites |
| Temazepam | Tmax ≈ 1–2 h T1/2 ≈ 8–20 h | CYP3A4 hydroxylation to oxazepam (active); glucuronidation |
| Z-DRUGS | ||
| Zolpidem | Tmax ≈ 1–2 h; T1/2 ≈ 1.5–3 h (↑ in elderly and in hepatic impairment) | Hydroxylation mainly via CYP3A4 (also CYP2C9, CYP1A2) to inactive metabolites; <1% is excreted unchanged in the urine |
| Zaleplon | Tmax ≈ 0.7–1.4 h; T1/2 ≈ ~1 h (ultra-short) | Oxidation by aldehyde oxidase to 5-oxo-zaleplon (inactive) that is further dealkylated or N-dealkylation by CYP3A4 to N-desethyl-zaleplon that is further oxidated; glucuronidation |
| Zopiclone/Eszopiclone | Tmax ≈ 1–2 h; T1/2 ≈ 5–7 h (longer than zolpidem) | N-demethylation to N-desmethylzopiclone (inactive) and zopiclone N-oxide (active); eszopiclone is oxidized and demethylated by CYP3A4 and CYP2E1 to inactive metabolites |
| Drug; Primary Biomarker(s)/Key Metabolites | Recommended Matrix | Detection Method/Detection Window | Typical Confirmatory Cut-Off or LOQ (Reported Range) | Notes |
|---|---|---|---|---|
| BENZODIAZEPINES | ||||
| Diazepam Biomarkers: Diazepam; nordiazepam; oxazepam; temazepam | Plasma/serum; urine; hair | LC–MS/MS (often with GC–MS backup); Detection window Plasma: hours to days Urine: days to weeks | ~5–50 ng/mL (urine) LOQ/confirmatory cut-off varies across methods | Long-acting active metabolites prolong the detection window; metabolite/parent ratios are used to help estimate time since dosing |
| Alprazolam Biomarkers: Alprazolam; α-hydroxyalprazolam; 4-hydroxyalprazolam | Plasma; DBS; urine | LC–MS/MS (including DBS); Detection window Plasma: hours Urine: ~1–3 days | ~0.05–5 ng/mL (LOQ (DBS/plasma); higher LOQ in urine) | Parent drug is largely responsible for the effect; CYP3A4 inhibitors markedly increase exposure so metabolite ratios are typically low |
| Bromazepam Biomarkers: Bromazepam; 3-hydroxybromazepam; minor desmethyl forms | Urine; plasma; hair | LC–MS/MS Detection window Urine: several days for 3-OH metabolite and conjugates | ~5–20 ng/mL (urine) | 3-hydroxybromazepam and conjugates are used as urinary markers; it is reported for them to be detectable for days depending on dose and method |
| Midazolam Biomarkers: Midazolam; 1′-hydroxymidazolam (active) and glucuronide | Plasma; urine | LC–MS/MS HPLC-UV Detection window Plasma: minutes to hours Urine: ~1–2 days | ~1–10 ng/mL (urine) | 1′-hydroxymidazolam and its glucuronide are the major analytes in plasma and urine; their concentrations and metabolite/parent ratios are strongly influenced by the duration of administration and organ function |
| Lorazepam Biomarkers: Lorazepam; lorazepam-3-O-glucuronide | Urine (after hydrolysis); plasma; hair | LC–MS/MS (in urine after hydrolysis) Detection window Plasma: hours to days Urine: days | ~5–50 ng/mL (urine; glucuronide measurement may require hydrolysis) | Lack of active metabolites makes lorazepam less likely to accumulate; lorazepam and its glucuronide metabolites are detected in urine |
| Clonazepam Biomarkers: Clonazepam; 7-amnoclonazepam | Plasma; urine; hair | LC–MS/MS Detection window Urine: days to weeks for metabolite | ~1–20 ng/mL (urine) | 7-aminoclonazepam is a major urinary metabolite used in forensic/toxicology screens |
| Nitrazepam Biomarkers: Nitrazepam; 7-aminonitrazepam | Urine; hair; plasma | LC–MS/MS Detection window Urine: days to weeks for 7-aminonitrazepam | ~1–20 ng/mL (urine) | 7-aminonitrazepam is the primary forensic marker; acetylated derivatives are commonly detected after hydrolysis |
| Flunitrazepam Biomarkers: Flunitrazepam; 7-aminoflunitrazepam | Urine; hair; plasma | LC–MS/MS Detection window Urine: days to weeks for 7-aminoflunitrazepam | ~1–20 ng/mL (urine LOQ); hair: pg/mg—low ng/mg depending on the method | 7-aminoflunitrazepam is the key forensic marker (often detectable long after parent drug); used in “date-rape” forensic investigations—metabolite ratios may help estimate time since ingestion |
| Z-DRUGS | ||||
| Zolpidem Biomarkers: Zolpidem; oxidative metabolites (phenyl-4-carboxylic acid) | Plasma; urine; hair | LC–MS/MS and immunoassay screening methods Detection window Plasma: hours Urine: ~1–2 days (metabolites) | ~0.5–10 ng/mL (urine) | Zolpidem has short detection window; metabolites are generally inactive; parent drug concentration reflects recent intake (useful to differentiate recent therapeutic dosing) |
| Zaleplon Biomarkers: Zaleplon; 5-oxo-zaleplon (aldehyde oxidase product); | Plasma; urine | HPLC/LC–MS Detection window Plasma: hours Urine: <24h (typical metabolites) | ~1–10 ng/mL (method dependent; short detection window) | Very short detection window; metabolites are inactive; parent drug detection indicates very recent dosing |
| Zopiclone/Eszopiclone Biomarkers: Zopiclone/eszopiclone; N-oxide; N-desmethyl zopiclone; 2-amino-5-chloropyridine | Plasma; urine; hair | LC–MS/MS/HRMS Detection window Plasma: hours Urine: ~1–3 days (metabolites) | ~0.5–10 ng/mL (LOQ range in modern LC–MS/MS methods) | Moderate detection window; presence of N-oxide metabolite may be informative in forensic screens |
| Product Name | Active Ingredient(s) | Regulatory Agency | Approved Indications |
|---|---|---|---|
| Epidyolex® | CBD | FDA EMA | Treatment of seizures associated with Lennox-Gastaut syndrome and Dravet syndrome in patients ≥ 2 years old. EMA also approves it for Tuberous Sclerosis Complex, in combination with other antiepileptics |
| Dronabinol (Marinol®, Syndros®) | Synthetic Δ9-THC | FDA | Appetite stimulation in patients with AIDS-related anorexia. Nausea and vomiting associated with chemotherapy in patients unresponsive to conventional antiemetics |
| Nabilone (Cesamet®) | Synthetic cannabinoid (THC analog) | FDA | Chemotherapy-induced nausea and vomiting in patients unresponsive to conventional antiemetics |
| Nabiximols (Sativex®) | THC + CBD (1:1, plant-derived) | EMA | Treatment of moderate to severe spasticity in multiple sclerosis, in patients who have not responded to other antispastic medications |
| Cannabinoid Metabolite | Matrix (Blood, Urine) | Detection Method(s) | Typical LOD/LOQ (or Range) | Notes/Comments |
|---|---|---|---|---|
| Δ9-THC | Blood, Urine | GC–MS/MS with on-line SPE | LOD~0.15 ng/mL; LOQ~0.3 ng/mL | Validated for both blood and urine |
| 11-OH-THC | Blood, Urine | GC–MS/MS with SPE | LOD~0.15 ng/mL; LOQ~0.3 ng/mL | Major active metabolite of THC |
| THC-COOH | Blood, Urine | GC–MS/MS with SPE | LOD~1.0 ng/mL; LOQ~3.0 ng/mL | Often measured as glucuronide conjugate in urine |
| CBD | Blood, Urine | GC–MS/MS with SPE | LOD~0.15 ng/mL; LOQ~0.3 ng/mL | Used to support interpretation of intake of CBD-rich products |
| CBN | Blood, Urine | GC–MS/MS with SPE | LOD~0.10 ng/mL; LOQ~0.2 ng/mL | Minor cannabinoid, sometimes marker of degradation or aging of product |
| Δ9-THC, 11-OH-THC, THC-COOH, CBD, CBN, etc. (9 analytes) | Urine | LC–MS/MS with SPE (minimal sample prep) | LOD~1 µg/L for non-carboxylated analytes and 5 µg/L for carboxylated analytes | Simultaneous detection without hydrolysis; rapid protocol |
| Synthetic cannabinoid metabolites (up to 61 analytes) | Urine | LC–MS/MS with SPE | LOD between 0.025 and 0.5 ng/mL | Validated panel for synthetic cannabinoids; high-throughput forensic tool |
| Δ9-THC, 11-OH-THC, THC-COOH, CBD, CBN, etc. (broad panel) | Plasma | HPLC-MS/MS LC–MS/MS (online extraction) | LOQ range~0.78–7.8 ng/mL | Method for 17 cannabinoids and metabolites |
| THC, 11-OH-THC, THC-COOH, CBD, CBG, THCV, CBN | Urine, Plasma | HPLC-MS/MS with hydrolysis | LOD < 1 ng/mL for many analytes | Method includes glucuronide hydrolysis for total analytes |
| Δ9-THC, 11-OH-THC, THC-COOH, Δ8-THC, etc. | Urine | LC–MS/MS (chromatographic separation of isomers) | LLOQ = 10 ng/mL for many analytes | Method distinguishes Δ8 vs. Δ9 isomers |
| THC, 11-OH-THC, THC-COOH, THCAA, CBN, CBG, THCV, THC-glucuronide, THCCOOH-glucuronide | Urine | LC–MS/MS with WAX-S pipette extraction | Linear ranges: 0.5–100 µg/L | Comprehensive urinary panel (11 analytes) |
| THC, 11-OH-THC, THC-COOH, CBD, CBN, THC-Gluc, THCCOOH-Gluc, etc. (11-analyte panel) | Urine | LC–MS/MS | LOQ~0.3–1.0 ng/mL (free and conjugated forms) | Differentiates dronabinol from cannabis use via metabolite profiles; detects minor cannabinoids |
| THC metabolites (urine) | Urine | LC–MS triple quadrupole (SLE extraction) | LOQ~5 ng/mL (for some THC metabolites) | According to Shimadzu application note |
| THC, CBD, CBN, 11-OH-THC, THC-COOH (ultra-trace) | Urine | UHPLC–MS/MS | LOD~0.002–0.008 ng/mL (free forms); ~0.005–0.017 ng/mL (total) | Very sensitive method for low exposure or secondhand smoke |
| Cannabinoid | Pharmacokinetic Properties | Major Metabolic Pathways and Key Metabolites | Notes |
|---|---|---|---|
| Δ9-THC Dronabinol | Tmax: Inhalation ≈ 0–0.25 h (minutes); Oral ≈ 1–3 h T½: Multi-phasic; initial distribution 1–4 h; terminal elimination ~25–36 h (highly variable) Bioavailability: Inhalation 10–35%; Oral ≈ 6–20% (low, subject to first-pass) Vd: Large (highly lipophilic, extensive tissue distribution) | Major: 11-OH-THC (active) → 11-COOH-THC (inactive, major urinary metabolite) Enzymes: Primarily CYP2C9, CYP2C19 and CYP3A4 (hydroxylation and subsequent oxidation); conjugation to glucuronides | 11-COOH-THC is the primary urinary marker used in forensic/toxicology screening; 11-OH-THC is pharmacologically active and important after oral dosing; long detection windows in chronic users due to lipophilic storage and slow release; metabolite/parent ratios and route of administration can be informative. |
| Δ8-THC | Tmax: Inhalation—minutes; Oral ≈ 1–3 h T½: Likely similar to Δ9-THC (multi-phasic; terminal elimination in the order of days), but data are limited Bioavailability: Expected similar to Δ9-THC (low oral bioavailability) | Major: 11-OH-Δ8-THC (reported), further oxidation to corresponding carboxy metabolite Enzymes: Similar CYP-mediated hydroxylation pathways as Δ9-THC (CYP2C9/2C19/3A4) | Pharmacology and metabolism are similar to Δ9-THC but clinical/pharmacokinetic data are less well characterized; forensic assays for Δ9-THC metabolites may cross-react. |
| CBD | Tmax: Oral ≈ 1–4 h (variable with formulation and fed state) T½: Reported ~18–32 h (single dose; can be longer with chronic dosing) Bioavailability: Oral ≈ 6–19% (formulation-dependent); extensive first-pass metabolism Vd: Large, highly tissue-distributed | Major: 7-OH-CBD, 7-COOH-CBD; conjugated metabolites (glucuronides) Enzymes: CYP2C19, CYP3A4 (oxidation) and UGT enzymes (glucuronidation) | CBD shows large interindividual variability; major metabolites (7-COOH) are abundant in plasma/urine; CBD can inhibit and be affected by CYP enzymes and UGTs (drug–drug interactions). Therapeutic formulations (e.g., Epidiolex) have well-characterized PK. |
| Nabiximols (THC + CBD; oromucosal spray) | Tmax: Oromucosal/oral absorption—typically 1–4 h (variable; partial buccal absorption produces faster onset than oral alone) T½: Parent compounds show multi-phasic kinetics; effective half-lives in the range of ~1–2 days for combined exposure (variable) Bioavailability: Higher than plain oral for oromucosal administration but variable between users | Major: 11-OH-THC (from THC), 7-OH-CBD (from CBD) and subsequent carboxy metabolites and glucuronides Enzymes: CYP2C9, CYP2C19, CYP3A4, CYP2D6 (minor), and UGTs for glucuronidation | Licensed pharmaceutical product; oromucosal route reduces but does not eliminate first-pass metabolism; predictable composition (standardized THC:CBD ratio) facilitates therapeutic monitoring. |
| AB-PINACA | Tmax: Smoking/inhalation—rapid (minutes); Oral Tmax may be 0.5–2 h depending on formulation T½: Reported short to moderate plasma half-lives (often a few hours for parent compound); terminal metabolites can persist longer Bioavailability: Variable; parent compound often rapidly metabolized | Major: Carboxylated and hydroxylated metabolites (e.g., AB-PINACA-COOH and multiple hydroxylated products) Enzymes/Pathways: Hydrolysis (CES1 reported) and oxidative metabolism (hydroxylation) on the alkyl/amino side chains; phase II conjugation | Parent drug is frequently absent or at low levels in urine; detection relies on specific metabolites (carboxy and hydroxylated derivatives). Novel synthetic cannabinoids show structural variability that complicates routine screening. |
| AB-FUBINACA | Tmax: Rapid after inhalation; Oral Tmax variable (often <2 h) T½: Parent compound short (hours); metabolites may be detectable for longer periods Bioavailability: Variable; rapid metabolism reduces parent exposure | Major: AB-FUBINACA-COOH and multiple hydroxylated metabolites; phase II conjugates Enzymes/Pathways: Hydrolysis by CES1 and oxidative metabolism on the indazole/alkyl side chains; conjugation for renal excretion | Forensic/toxicology screens target the carboxylated and hydroxylated metabolites rather than parent drug; potency and toxic effects can be considerably higher than phytocannabinoids. |
| Substance | Pharmacokinetic Features | Biomarkers of Use/Recommended Matrices and Analytical Methods | Urine Cut-Off Limits (Screening/Confirmatory, ng/mL) | Major Analytical Challenges |
|---|---|---|---|---|
| Cocaine |
|
| Screen: 300 ng/mL (cocaine metabolites) Confirmatory: 100–150 ng/mL (cocaine metabolites) |
|
| Primary Molecule | Metabolite(s) or Related Analytes | Diagnostic Relevance |
|---|---|---|
| Cocaine | Benzoylecgonine (BE) | Primary urinary biomarker; persists longest |
| Ecgonine methyl ester (EME) | Marker of enzymatic hydrolysis by plasma cholinesterases | |
| Norcocaine | Active metabolite indicating hepatic oxidation | |
| Anhydroecgonine methyl ester (AEME) | Biomarker of crack cocaine smoking | |
| Cocaethylene | Marker of concurrent ethanol consumption | |
| Ecgonine | Secondary hydrolysis product, less specific |
| Analyte | Relevance |
|---|---|
| Ketamine | Parent compound; short detection window |
| Norketamine (NK) | Primary active metabolite; extends detection window |
| Hydroxynorketamine (HNK) | Antidepressant biomarker; low psychotomimetic activity |
| Dehydronorketamine | Minor metabolite; limited clinical significance |
| Glucuronide conjugates | Indicate metabolic clearance |
| Substance | Pharmacokinetic Features [333,334,335] | Biomarkers of Use/Recommended Matrices and Analytical Methods | Urine Cut-Off Limits (Screening/Confirmatory, ng/mL) | Major Analytical Challenges |
|---|---|---|---|---|
| Ketamine |
|
| Screen: 50–100 ng/mL Confirmatory: 50 ng/mL |
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stoeva-Grigorova, S.; Hvarchanova, N.; Gancheva, S.; Eftimov, M.; Georgiev, K.D.; Radeva-Ilieva, M. Differentiation of Therapeutic and Illicit Drug Use via Metabolite Profiling. Metabolites 2025, 15, 745. https://doi.org/10.3390/metabo15110745
Stoeva-Grigorova S, Hvarchanova N, Gancheva S, Eftimov M, Georgiev KD, Radeva-Ilieva M. Differentiation of Therapeutic and Illicit Drug Use via Metabolite Profiling. Metabolites. 2025; 15(11):745. https://doi.org/10.3390/metabo15110745
Chicago/Turabian StyleStoeva-Grigorova, Stanila, Nadezhda Hvarchanova, Silvia Gancheva, Miroslav Eftimov, Kaloyan D. Georgiev, and Maya Radeva-Ilieva. 2025. "Differentiation of Therapeutic and Illicit Drug Use via Metabolite Profiling" Metabolites 15, no. 11: 745. https://doi.org/10.3390/metabo15110745
APA StyleStoeva-Grigorova, S., Hvarchanova, N., Gancheva, S., Eftimov, M., Georgiev, K. D., & Radeva-Ilieva, M. (2025). Differentiation of Therapeutic and Illicit Drug Use via Metabolite Profiling. Metabolites, 15(11), 745. https://doi.org/10.3390/metabo15110745









