Reliability of Time-Series Plasma Metabolome Data over 6 Years in a Large-Scale Cohort Study
Abstract
1. Introduction
2. Materials and Methods
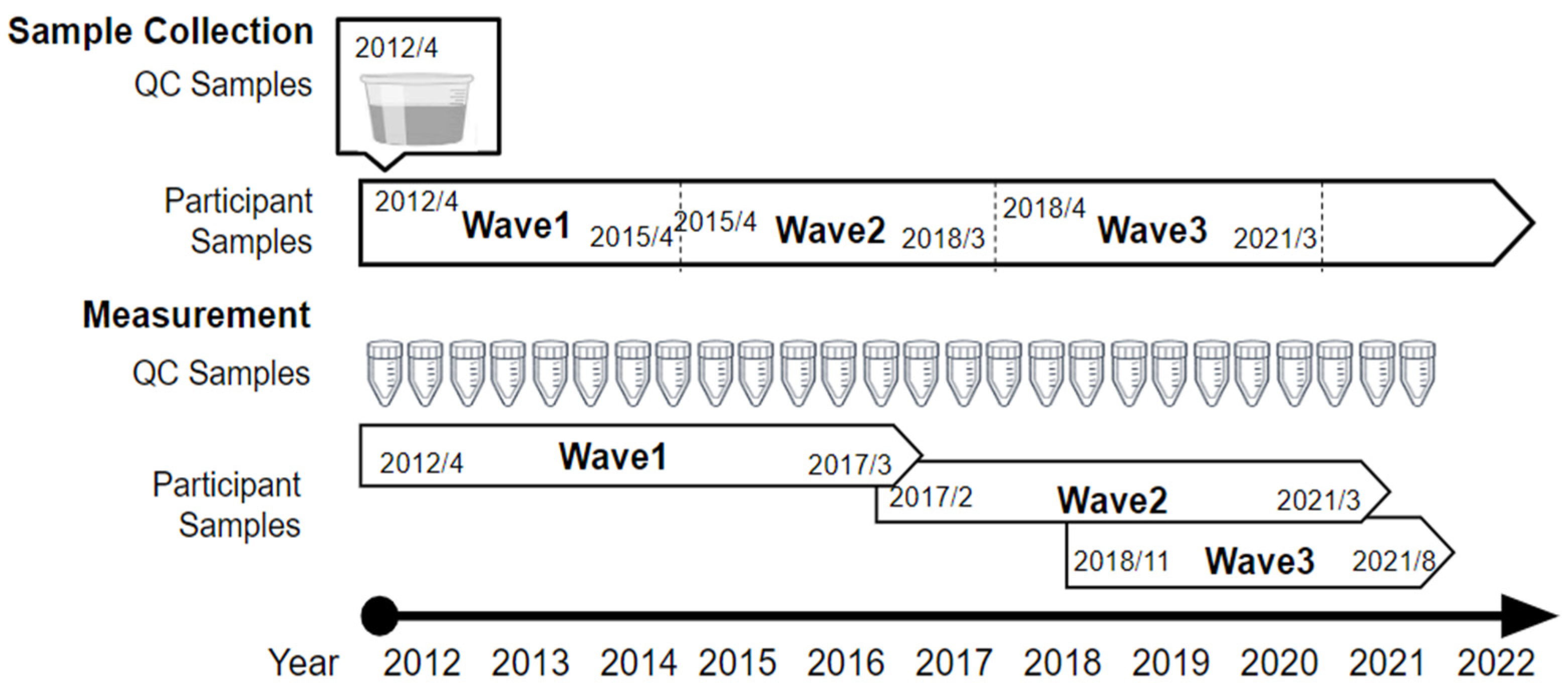
2.1. Tsuruoka Metabolomics Cohort Study
2.2. Study Subjects and Sample Collection
2.3. Metabolomics Measurements and Quality Control Samples
2.4. Variables Definition
2.5. Statistical Analyses
3. Results
3.1. CV of QC Samples, ICC, and Change Rate of QC Samples
3.2. Characteristics of the Study Participants and Sample Collection/Measurement Process
3.3. Intra-Individual Changes in Metabolites over Time
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Nicholson, G.; Rantalainen, M.; Maheret, A.D.; Li, J.V.; Malmodin, D.; Ahmadi, K.R.; Faber, J.H.; Hallgrimsdottir, I.B.; Barrett, A.; Toft, H.; et al. Human metabolic profiles are stably controlled by genetic and environmental variation. Mol. Syst. Biol. 2011, 7, 525. [Google Scholar] [CrossRef] [PubMed]
- Sekula, P.; Goek, O.N.; Quaye, L.; Barrios, C.; Levey, A.S.; Margl, W.R.; Menni, C.; Yet, I.; Gieger, C.; Inker, L.A.; et al. A metabolome-wide association study of kidney function and disease in the general population. J. Am. Soc. Nephrol. 2016, 27, 1175–1188. [Google Scholar] [CrossRef]
- Wang, T.J.; Larson, M.G.; Vasan, R.S.; Cheng, S.; Rhee, E.P.; McCabe, E.; Lewis, G.D.; Fox, C.S.; Jacques, P.F.; Fernandez, C.; et al. Metabolite profiles and the risk of developing diabetes. Nat. Med. 2011, 17, 448–454. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Wurtz, P.; Auro, K.; Makinen, V.P.; Kangas, A.J.; Soininen, P.; Tiainen, M.; Tynkkynen, T.; Jokelainen, J.; Santalahti, K.; et al. Metabolic profiling of pregnancy: Cross-sectional and longitudinal evidence. BMC Med. 2016, 14, 205. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Ferreira, D.L.S.; Nelson, S.M.; Sattar, N.; Korpela, M.A.; Lawlor, D.A. Metabolic characterization of menopause: Cross-sectional and longitudinal evidence. BMC Med. 2018, 16, 17. [Google Scholar] [CrossRef] [PubMed]
- Makinen, V.P.; Karsikas, M.; Kettunen, J.; Lehtimaki, T.; Kahonen, M.; Viikari, J.; Perola, M.; Salomaa, V.; Jarvelin, M.R.; Raitatari, O.T.; et al. Longitudinal profiling of metabolic ageing trends in two population cohorts of young adults. Int. J. Epidemiol. 2022, 51, 1970–1983. [Google Scholar] [CrossRef] [PubMed]
- Han, W.; Li, L. Evaluating and minimizing batch effects in metabolomics. Mass Spectrom. Rev. 2022, 41, 421–442. [Google Scholar] [CrossRef] [PubMed]
- Townsend, M.K.; Clish, C.B.; Kraft, P.; Wu, C.; Souza, A.L.; Deik, A.A.; Tworoger, S.S.; Wolpin, B.M. Reproducibility of metabolomic profiles among men and women in two large cohort studies. Clin. Chem. 2013, 59, 1657–1667. [Google Scholar] [CrossRef]
- Ramautar, R.; Somsen, G.W.; De Jong, G.J. CE-MS for metabolomics: Developments and applications in the period 2014–2016. Electrophoresis 2017, 38, 190–202. [Google Scholar] [CrossRef] [PubMed]
- Harada, S.; Hirayama, A.; Chan, Q.; Kurihara, A.; Fukai, K.; Iida, M.; Kato, S.; Sugiyama, D.; Kuwabara, K.; Takeuchi, A.; et al. Reliability of plasma polar metabolite concentrations in a large-scale cohort study using capillary electrophoresis-mass spectrometry. PLoS ONE 2018, 13, e0191230. [Google Scholar] [CrossRef]
- Harada, S.; Iida, M.; Miyagawa, N.; Hirata, A.; Kuwabara, K.; Matsumoto, M.; Okamura, T.; Edagawa, S.; Kawada, Y.; Miyake, A.; et al. Study profile of the Tsuruoka Metabolomics Cohort Study (TMCS). J. Epidemiol. 2023. advance online publication. Available online: https://www.jstage.jst.go.jp/article/jea/advpub/0/advpub_JE20230192/_article/-char/en (accessed on 15 January 2024).
- Hirayama, A.; Ishikawa, T.; Takahashi, H.; Yamanaka, S.; Ikeda, S.; Hirata, A.; Harada, S.; Sugimoto, M.; Soga, T.; Tomita, M.; et al. Quality control of targeted plasma lipids in a large-scale cohort study using liquid chromatography-tandem mass spectrometry. Metabolites 2023, 13, 558. [Google Scholar] [CrossRef] [PubMed]
- Hirayama, A.; Nakashima, E.; Sugimoto, M.; Akiyama, S.; Sato, W.; Maruyama, S.; Matsuno, S.; Tomita, M.; Yuzawa, T.; Soga, T. Metabolic profiling reveals new serum biomarkers for differentiating diabetic nephropathy. Anal. Bioanal. Chem. 2012, 404, 3101–3109. [Google Scholar] [CrossRef] [PubMed]
- Ramautar, R.; Somsen, G.W.; De Jong, G.J. CE-MS in metabolomics. Electrophoresis 2009, 30, 276–291. [Google Scholar] [CrossRef]
- Hornung, R.W.; Reed, L.D. Estimation of average concentration in the presence of non detectable values. Appl. Occup. Environ. Hyg. 1990, 5, 46–51. [Google Scholar] [CrossRef]
- Rosner, B. Fundamentals of Biostatistics, 6th ed.; Duxbury Press: Belmont, CA, USA, 2006; pp. 611–617. [Google Scholar]
- Benjamini, Y.; Yekutieli, D. The control of the false discovery rate in multiple testing under dependency. Ann. Stat. 2001, 29, 1165–1188. [Google Scholar] [CrossRef]
- Narum, S.R. Beyond Bonferroni: Less conservative analyses for conservation genetics. Conserv. Genet. 2006, 7, 783–787. [Google Scholar] [CrossRef]
- Ministry of Health, Labour and Welfare. The National Health and Nutrition Survey. (In Japanese). Available online: https://www.mhlw.go.jp/bunya/kenkou/eiyou/h24-houkoku.html (accessed on 14 December 2023).
- KEGG PATHWAY Database. Available online: https://www.genome.jp/kegg/pathway.html#nucleotide (accessed on 2 December 2023).
- Darst, B.F.; Koscik, R.L.; Hogan, K.J.; Johnson, S.C.; Engelman, C.D. Longitudinal plasma metabolomics of aging and sex. Aging 2019, 11, 1262–1282. [Google Scholar] [CrossRef] [PubMed]
- White, E. Measurement error in biomarkers: Sources, assessment, and impact on studies. IARC Sci. Publ. 2011, 163, 143–161. [Google Scholar]
- Yin, X.; Prendiville, O.; McNamara, A.E.; Brennan, L. Targeted metabolomic approach to assess the reproducibility of plasma metabolites over a four month period in a free-living population. J. Proteome Res. 2022, 21, 683–690. [Google Scholar] [CrossRef]
- Lamont, L.S.; McCullough, A.J.; Kalhan, S.C. Gender differences in leucine, but not lysine, kinetics. J. Appl. Physiol. 2001, 91, 357–362. [Google Scholar] [CrossRef]
- Gu, F.; Derkach, A.; Freedman, N.D.; Landi, M.T.; Albanes, D.; Weinstein, S.J.; Mondul, A.M.; Matthews, C.E.; Guertin, K.A.; Xiao, Q.; et al. Cigarette smoking behaviour and blood metabolomics. Int. J. Epidemiol. 2016, 45, 1421–1432. [Google Scholar] [CrossRef] [PubMed]
- Jin, Q.; Ma, R.C.W. Metabolomics in diabetes and diabetic complications: Insights from epidemiological studies. Cells 2021, 11, 2832. [Google Scholar] [CrossRef] [PubMed]


| All (n = 2999) | Men (n = 1317) | Women (n = 1682) | |
|---|---|---|---|
| Age (years) | 54.8 ± 10.5 | 55.1 ± 10.4 | 54.6 ± 10.6 |
| Body mass index (kg/m2) | 23.0 ± 3.3 | 23.8 ± 3.0 | 22.4 ± 3.4 |
| Any current alcohol intake | 1544 (51.5%) | 1022 (77.7%) | 522 (31.1%) |
| Current smoker | 507 (16.9%) | 425 (32.4%) | 82 (4.9%) |
| Hypertension | 1079 (36.1%) | 547 (41.6%) | 532 (31.7%) |
| Diabetes mellitus | 206 (6.9%) | 132 (10.1%) | 74 (4.4%) |
| Dyslipidemia | 1367 (45.6%) | 676 (51.3%) | 691 (41.1%) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Miyake, A.; Harada, S.; Sugiyama, D.; Matsumoto, M.; Hirata, A.; Miyagawa, N.; Toki, R.; Edagawa, S.; Kuwabara, K.; Okamura, T.; et al. Reliability of Time-Series Plasma Metabolome Data over 6 Years in a Large-Scale Cohort Study. Metabolites 2024, 14, 77. https://doi.org/10.3390/metabo14010077
Miyake A, Harada S, Sugiyama D, Matsumoto M, Hirata A, Miyagawa N, Toki R, Edagawa S, Kuwabara K, Okamura T, et al. Reliability of Time-Series Plasma Metabolome Data over 6 Years in a Large-Scale Cohort Study. Metabolites. 2024; 14(1):77. https://doi.org/10.3390/metabo14010077
Chicago/Turabian StyleMiyake, Atsuko, Sei Harada, Daisuke Sugiyama, Minako Matsumoto, Aya Hirata, Naoko Miyagawa, Ryota Toki, Shun Edagawa, Kazuyo Kuwabara, Tomonori Okamura, and et al. 2024. "Reliability of Time-Series Plasma Metabolome Data over 6 Years in a Large-Scale Cohort Study" Metabolites 14, no. 1: 77. https://doi.org/10.3390/metabo14010077
APA StyleMiyake, A., Harada, S., Sugiyama, D., Matsumoto, M., Hirata, A., Miyagawa, N., Toki, R., Edagawa, S., Kuwabara, K., Okamura, T., Sato, A., Amano, K., Hirayama, A., Sugimoto, M., Soga, T., Tomita, M., Arakawa, K., Takebayashi, T., & Iida, M. (2024). Reliability of Time-Series Plasma Metabolome Data over 6 Years in a Large-Scale Cohort Study. Metabolites, 14(1), 77. https://doi.org/10.3390/metabo14010077










