A Systematic Review on the Association between Obesity and Mood Disorders and the Role of Gut Microbiota
Abstract
1. Introduction
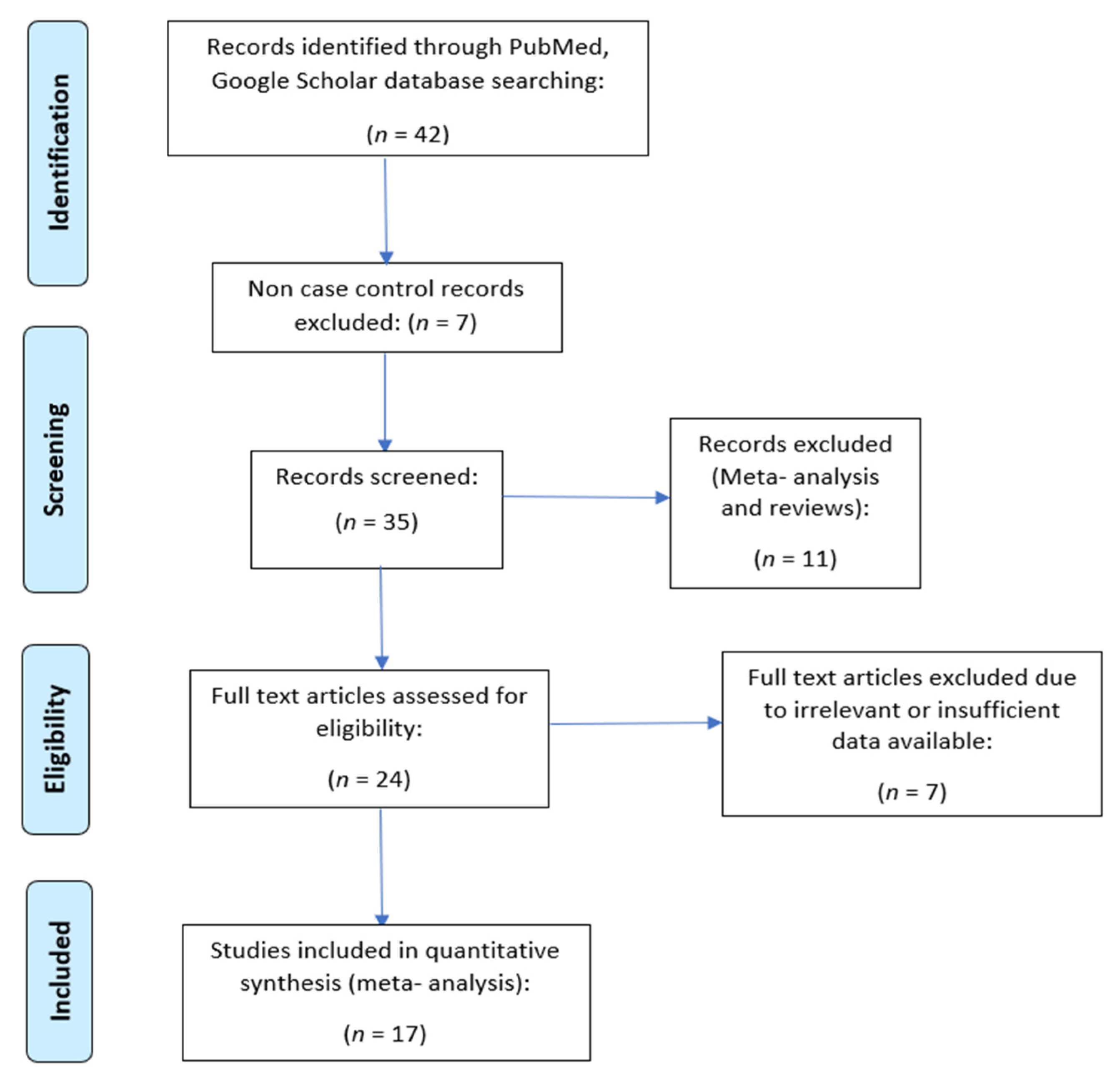
2. Materials and Methods
2.1. Literature Survey
2.2. Selection Criteria
2.3. Data Extraction
2.4. Statistical Analysis
3. Results
3.1. Obesity and Mood Disorders
3.2. Microbiota and Obesity
3.3. Microbiota and Mood Disorders
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Huang, P.L. A comprehensive definition for metabolic syndrome. Dis. Model. Mech. 2009, 2, 231–237. [Google Scholar] [CrossRef]
- Grundy, S.M. Metabolic syndrome update. Trends Cardiovasc. Med. 2015, 26, 364–373. [Google Scholar] [CrossRef]
- Han, T.S.; Lean, M.E. A clinical perspective of obesity, metabolic syndrome and cardiovascular disease. JRSM Cardiovasc. Dis. 2016, 5, 2048004016633371. [Google Scholar] [CrossRef] [PubMed]
- Abdelaal, M.; le Roux, C.W.; Docherty, N.G. Morbidity and mortality associated with obesity. Ann. Transl. Med. 2017, 5, 161. [Google Scholar] [CrossRef] [PubMed]
- Sarwer, D.B.; Polonsky, H.M. The Psychosocial Burden of Obesity. Endocrinol. Metab. Clin. N. Am. 2016, 45, 677–688. [Google Scholar] [CrossRef]
- Dong, C.; Li, W.-D.; Li, D.; Price, R.A. Extreme obesity is associated with attempted suicides: Results from a family study. Int. J. Obes. 2005, 30, 388–390. [Google Scholar] [CrossRef] [PubMed]
- Baxter, A.J.; Scott, K.M.; Ferrari, A.J.; Norman, R.E.; Vos, T.; Whiteford, H.A. Challenging the myth of an “epidemic” of common mental disorders: Trends in the global prevalence of anxiety and depression between 1990 and 2010. Depress. Anxiety 2014, 31, 506–516. [Google Scholar] [CrossRef]
- Müller, A. Mental health disorders: Prevalent but widely ignored in academia? J. Physiol. 2020, 598, 1279–1281. [Google Scholar] [CrossRef]
- Scott, K.M.; McGee, M.A.; Wells, J.E.; Browne, M.A.O. Obesity and mental disorders in the adult general population. J. Psychosom. Res. 2008, 64, 97–105. [Google Scholar] [CrossRef]
- Goodman, E.; Whitaker, R.C. A Prospective Study of the Role of Depression in the Development and Persistence of Adolescent Obesity. Pediatrics 2002, 110, 497–504. [Google Scholar] [CrossRef]
- Villares, J.M.M. Stigma Experienced by Children and Adolescents with Obesity. Acta Pediatr. Esp. 2018, 76, 63. [Google Scholar] [CrossRef]
- Li, Y.; Cheng, Y.; Zhou, Y.; Du, H.; Zhang, C.; Zhao, Z.; Chen, Y.; Zhou, Z.; Mei, J.; Wu, W. High fat diet-induced obesity leads to depressive and anxiety-like behaviors in mice via AMPK/mTOR-mediated autophagy. Exp. Neurol. 2022, 348, 113949. [Google Scholar] [CrossRef]
- Dallman, M.F. Stress-induced obesity and the emotional nervous system. Trends Endocrinol. Metab. 2010, 21, 159–165. [Google Scholar] [CrossRef]
- McMillan, F.D. Stress-induced and emotional eating in animals: A review of the experimental evidence and implications for companion animal obesity. J. Vet. Behav. 2013, 8, 376–385. [Google Scholar] [CrossRef]
- Halfon, N.; Larson, K.; Slusser, W. Associations Between Obesity and Comorbid Mental Health, Developmental, and Physical Health Conditions in a Nationally Representative Sample of US Children Aged 10 to 17. Acad. Pediatr. 2013, 13, 6–13. [Google Scholar] [CrossRef] [PubMed]
- Swallen, K.C.; Reither, E.N.; Haas, S.A.; Meier, A.M. Overweight, Obesity, and Health-Related Quality of Life Among Adolescents: The National Longitudinal Study of Adolescent Health. Pediatrics 2005, 115, 340–347. [Google Scholar] [CrossRef]
- Wardle, J.; Cooke, L. The impact of obesity on psychological well-being. Best Pract. Res. Clin. Endocrinol. Metab. 2005, 19, 421–440. [Google Scholar] [CrossRef] [PubMed]
- Whitehead, R.; Berg, C.; Cosma, A.; Gobina, I.; Keane, E.; Neville, F.; Ojala, K.; Kelly, C. Trends in Adolescent Overweight Perception and Its Association with Psychosomatic Health 2002–2014: Evidence From 33 Countries. J. Adolesc. Health 2016, 60, 204–211. [Google Scholar] [CrossRef]
- Nigatu, Y.T.; Reijneveld, S.A.; de Jonge, P.; van Rossum, E.; Bültmann, U. The Combined Effects of Obesity, Abdominal Obesity and Major Depression/Anxiety on Health-Related Quality of Life: The LifeLines Cohort Study. PLoS ONE 2016, 11, e0148871. [Google Scholar] [CrossRef] [PubMed]
- García-García, I.; Michaud, A.; Dadar, M.; Zeighami, Y.; Neseliler, S.; Collins, D.L.; Evans, A.C.; Dagher, A. Neuroanatomical differences in obesity: Meta-analytic findings and their validation in an independent dataset. Int. J. Obes. 2018, 43, 943–951. [Google Scholar] [CrossRef]
- Hamer, M.; Batty, G.D. Association of body mass index and waist-to-hip ratio with brain structure: UK Biobank study. Neurology 2019, 92, e594–e600. [Google Scholar] [CrossRef] [PubMed]
- Raji, C.A.; Ho, A.J.; Parikshak, N.; Becker, J.; Lopez, O.L.; Kuller, L.H.; Hua, X.; Leow, A.D.; Toga, A.W.; Thompson, P.M. Brain structure and obesity. Hum. Brain Mapp. 2009, 31, 353–364. [Google Scholar] [CrossRef] [PubMed]
- Repple, J.; Opel, N.; Meinert, S.; Redlich, R.; Hahn, T.; Winter, N.R.; Kaehler, C.; Emden, D.; Leenings, R.; Grotegerd, D. Elevated body-mass index is associated with reduced white matter integrity in two large independent cohorts. Psychoneuroendocrinology 2018, 91, 179–185. [Google Scholar] [CrossRef]
- Bliss, E.S.; Whiteside, E. The Gut-Brain Axis, the Human Gut Microbiota and Their Integration in the Development of Obesity. Front. Physiol. 2018, 9, 900. [Google Scholar] [CrossRef]
- Yi, C.-X.; Tschöp, M.H. Brain–gut–adipose-tissue communication pathways at a glance. Dis. Model. Mech. 2012, 5, 583–587. [Google Scholar] [CrossRef]
- Yu, Z.; Wang, Y.; Yu, Z.; Lu, M.; Xu, B. Crosstalk between adipose tissue and the microbiota-gut-brain axis in metabolic diseases. Int. J. Biol. Sci. 2022, 18, 1706–1723. [Google Scholar] [CrossRef]
- Rooks, M.; Garrett, W. Gut microbiota, metabolites and host immunity. Nat. Rev. Immunol. 2016, 16, 341–352. [Google Scholar] [CrossRef] [PubMed]
- Ma, Q.; Xing, C.; Long, W.; Wang, H.Y.; Liu, Q.; Wang, R.-F. Impact of microbiota on central nervous system and neurological diseases: The gut-brain axis. J. Neuroinflamm. 2019, 16, 53. [Google Scholar] [CrossRef] [PubMed]
- Martin, A.M.; Sun, E.W.; Rogers, G.B.; Keating, D.J. The Influence of the Gut Microbiome on Host Metabolism through the Regulation of Gut Hormone Release. Front. Physiol. 2019, 10, 428. [Google Scholar] [CrossRef]
- Mirzaei, R.; Bouzari, B.; Hosseini-Fard, S.R.; Mazaheri, M.; Ahmadyousefi, Y.; Abdi, M.; Jalalifar, S.; Karimitabar, Z.; Teimoori, A.; Keyvani, H. Role of microbiota-derived short-chain fatty acids in nervous system disorders. Biomed. Pharmacother. 2021, 139, 111661. [Google Scholar] [CrossRef]
- Silva, Y.P.; Bernardi, A.; Frozza, R.L. The Role of Short-Chain Fatty Acids from Gut Microbiota in Gut-Brain Communication. Front. Endocrinol. 2020, 11, 25. [Google Scholar] [CrossRef] [PubMed]
- Brown, K.; DeCoffe, D.; Molcan, E.; Gibson, D.L. Diet-Induced Dysbiosis of the Intestinal Microbiota and the Effects on Immunity and Disease. Nutrients 2012, 4, 1095–1119. [Google Scholar] [CrossRef] [PubMed]
- Xiao, H.; Kang, S. The Role of the Gut Microbiome in Energy Balance with a Focus on the Gut-Adipose Tissue Axis. Front. Genet. 2020, 11, 297. [Google Scholar] [CrossRef]
- Berglund, P. Cost-benefit analysis for sustainable stormwater management. Nature 2018, 444, 234–270. [Google Scholar] [CrossRef]
- Murphy, E.F.; Cotter, P.D.; Healy, S.; Marques, T.M.; O’Sullivan, O.; Fouhy, F.; Clarke, S.F.; O’Toole, P.W.; Quigley, E.M.; Stanton, C. Composition and energy harvesting capacity of the gut microbiota: Relationship to diet, obesity and time in mouse models. Gut 2010, 59, 1635–1642. [Google Scholar] [CrossRef]
- Turnbaugh, P.J.; Bäckhed, F.; Fulton, L.; Gordon, J.I. Diet-Induced Obesity Is Linked to Marked but Reversible Alterations in the Mouse Distal Gut Microbiome. Cell Host Microbe 2008, 3, 213–223. [Google Scholar] [CrossRef]
- Kabouridis, P.S.; Pachnis, V. Emerging roles of gut microbiota and the immune system in the development of the enteric nervous system. J. Clin. Investig. 2015, 125, 956–964. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.-X.; Wang, Y.-P. Gut Microbiota-brain Axis. Chin. Med. J. 2016, 129, 2373–2380. [Google Scholar] [CrossRef]
- Huang, Y.; Shi, X.; Li, Z.; Shen, Y.; Shi, X.; Wang, L.; Li, G.; Yuan, Y.; Wang, J.; Zhang, Y. Possible association of Firmicutes in the gut microbiota of patients with major depressive disorder. Neuropsychiatr. Dis. Treat. 2018, 14, 3329–3337. [Google Scholar] [CrossRef]
- Järbrink-Sehgal, E.; Andreasson, A. The gut microbiota and mental health in adults. Curr. Opin. Neurobiol. 2020, 62, 102–114. [Google Scholar] [CrossRef]
- Guida, F.; Turco, F.; Iannotta, M.; De Gregorio, D.; Palumbo, I.; Sarnelli, G.; Furiano, A.; Napolitano, F.; Boccella, S.; Luongo, L. Antibiotic-induced microbiota perturbation causes gut endocannabinoidome changes, hippocampal neuroglial reorganization and depression in mice. Brain Behav. Immun. 2018, 67, 230–245. [Google Scholar] [CrossRef] [PubMed]
- Katz-Barber, M.W.; Hollins, S.L.; Cuskelly, A.; Leong, A.J.; Dunn, A.; Harms, L.; Hodgson, D.M. Investigating the gut-brain axis in a neurodevelopmental rodent model of schizophrenia. Brain Behav. Immun.-Health 2020, 3, 100048. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Yun, Y.; An, H.; Zhao, W.; Ma, T.; Wang, Z.; Yang, F. Gut Microbiome Composition Associated with Major Depressive Disorder and Sleep Quality. Front. Psychiatry 2021, 12, 551. [Google Scholar] [CrossRef] [PubMed]
- Hadi, S.; Momenan, M.; Cheraghpour, K.; Hafizi, N.; Pourjavidi, N.; Malekahmadi, M.; Foroughi, M.; Alipour, M. Abdominal volume index: A predictive measure in relationship between depression/anxiety and obesity. Afr. Health Sci. 2020, 20, 257–265. [Google Scholar] [CrossRef]
- Baumeister, H.; Härter, M. Mental disorders in patients with obesity in comparison with healthy probands. Int. J. Obes. 2007, 31, 1155–1164. [Google Scholar] [CrossRef]
- Bodenlos, J.S.; Lemon, S.C.; Schneider, K.L.; August, M.A.; Pagoto, S.L. Associations of mood and anxiety disorders with obesity: Comparisons by ethnicity. J. Psychosom. Res. 2011, 71, 319–324. [Google Scholar] [CrossRef]
- Brumpton, B.; Langhammer, A.; Romundstad, P.; Chen, Y.; Mai, X.-M. The associations of anxiety and depression symptoms with weight change and incident obesity: The HUNT Study. Int. J. Obes. 2012, 37, 1268–1274. [Google Scholar] [CrossRef]
- Chen, Y.; Jiang, Y.; Mao, Y.; Shlay, J.C.; Zolot, L.; Bell, D.; Maravi, M.E.; Urbina, C.; Conroy, K.; Rosenthal, S.L. Association between Obesity and Depression in Canadians. J. Women’s Health 2009, 18, 1687–1692. [Google Scholar] [CrossRef]
- Heo, M.; Pietrobelli, A.; Fontaine, K.R.; Sirey, J.A.; Faith, M.S. Depressive mood and obesity in US adults: Comparison and moderation by sex, age, and race. Int. J. Obes. 2005, 30, 513–519. [Google Scholar] [CrossRef]
- Lindberg, L.; Hagman, E.; Danielsson, P.; Marcus, C.; Persson, M. Anxiety and depression in children and adolescents with obesity: A nationwide study in Sweden. BMC Med. 2020, 18, 30. [Google Scholar] [CrossRef]
- Mather, A.A.; Cox, B.J.; Enns, M.W.; Sareen, J. Associations of obesity with psychiatric disorders and suicidal behaviors in a nationally representative sample. J. Psychosom. Res. 2009, 66, 277–285. [Google Scholar] [CrossRef] [PubMed]
- Roberts, R.E.; Deleger, S.; Strawbridge, W.J.; Kaplan, G.A. Prospective association between obesity and depression: Evidence from the Alameda County Study. Int. J. Obes. 2003, 27, 514–521. [Google Scholar] [CrossRef] [PubMed]
- Scott, K.M.; Bruffaerts, R.; Simon, G.E.; Alonso, J.; Angermeyer, M.; de Girolamo, G.; Demyttenaere, K.; Gasquet, I.; Haro, J.M.; Karam, E. Obesity and mental disorders in the general population: Results from the world mental health surveys. Int. J. Obes. 2007, 32, 192–200. [Google Scholar] [CrossRef] [PubMed]
- Simon, G.E.; Von Korff, M.; Saunders, K.; Miglioretti, D.L.; Crane, P.K.; van Belle, G.; Kessler, R.C. Association Between Obesity and Psychiatric Disorders in the US Adult Population. Arch. Gen. Psychiatry 2006, 63, 824–830. [Google Scholar] [CrossRef] [PubMed]
- Zhao, G.; Ford, E.S.; Dhingra, S.; Li, C.; Strine, T.W.; Mokdad, A.H. Depression and anxiety among US adults: Associations with body mass index. Int. J. Obes. 2009, 33, 257–266. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, A.; Yang, W.; Chen, G.; Shafiq, M.; Javed, S.; Zaidi, S.S.A.; Shahid, R.; Liu, C.; Bokhari, H. Analysis of gut microbiota of obese individuals with type 2 diabetes and healthy individuals. PLoS ONE 2019, 14, e0226372. [Google Scholar] [CrossRef]
- Duan, M.; Wang, Y.; Zhang, Q.; Zou, R.; Guo, M.; Zheng, H. Characteristics of gut microbiota in people with obesity. PLoS ONE 2021, 16, e0255446. [Google Scholar] [CrossRef]
- Huttenhower, C.; Gevers, D.; Knight, R.; Abubucker, S.; Badger, J.H.; Chinwalla, A.T.; Creasy, H.H.; Earl, A.M.; FitzGerald, M.G.; Fulton, R.S. Structure, function and diversity of the healthy human microbiome. Nature 2012, 486, 207–214. [Google Scholar] [CrossRef]
- Kasai, C.; Sugimoto, K.; Moritani, I.; Tanaka, J.; Oya, Y.; Inoue, H.; Tameda, M.; Shiraki, K.; Ito, M.; Takei, Y. Comparison of the gut microbiota composition between obese and non-obese individuals in a Japanese population, as analyzed by terminal restriction fragment length polymorphism and next-generation sequencing. BMC Gastroenterol. 2015, 15, 100. [Google Scholar] [CrossRef]
- Koliada, A.; Syzenko, G.; Moseiko, V.; Budovska, L.; Puchkov, K.; Perederiy, V.; Gavalko, Y.; Dorofeyev, A.; Romanenko, M.; Tkach, S. Association between body mass index and Firmicutes/Bacteroidetes ratio in an adult Ukrainian population. BMC Microbiol. 2017, 17, 120. [Google Scholar] [CrossRef]
- Shin, S.; Cho, K.Y. Altered Gut Microbiota and Shift in Bacteroidetes between Young Obese and Normal-Weight Korean Children: A Cross-Sectional Observational Study. BioMed Res. Int. 2020, 2020, 6587136. [Google Scholar] [CrossRef] [PubMed]
- Turnbaugh, P.J.; Hamady, M.; Yatsunenko, T.; Cantarel, B.L.; Duncan, A.; Ley, R.E.; Sogin, M.L.; Jones, W.J.; Roe, B.A.; Affourtit, J.P. A core gut microbiome in obese and lean twins. Nature 2009, 457, 480–484. [Google Scholar] [CrossRef] [PubMed]
- Verdam, F.J.; Fuentes, S.; de Jonge, C.; Zoetendal, E.G.; Erbil, R.; Greve, J.W.; Buurman, W.A.; de Vos, W.M.; Rensen, S.S. Human intestinal microbiota composition is associated with local and systemic inflammation in obesity. Obesity 2013, 21, E607–E615. [Google Scholar] [CrossRef]
- Wu, G.D.; Chen, J.; Hoffmann, C.; Bittinger, K.; Chen, Y.Y.; Keilbaugh, S.A.; Lewis, J.D. Linking Long-Term Dietary Patterns with Gut Microbial Enterotypes. Science 2011, 334, 105–108. [Google Scholar] [CrossRef] [PubMed]
- Yatsunenko, T.; Rey, F.E.; Manary, M.J.; Trehan, I.; Dominguez-Bello, M.G.; Contreras, M.; Magris, M.; Hidalgo, G.; Baldassano, R.N.; Anokhin, A.P. Human gut microbiome viewed across age and geography. Nature 2012, 486, 222–227. [Google Scholar] [CrossRef] [PubMed]
- Zupancic, M.L.; Cantarel, B.L.; Liu, Z.; Drabek, E.F.; Ryan, K.A.; Cirimotich, S.; Jones, C.; Knight, R.; Walters, W.A.; Knights, D. Analysis of the Gut Microbiota in the Old Order Amish and Its Relation to the Metabolic Syndrome. PLoS ONE 2012, 7, e43052. [Google Scholar] [CrossRef]
- Chen, J.-J.; Zheng, P.; Liu, Y.-Y.; Zhong, X.-G.; Wang, H.-Y.; Guo, Y.-J.; Xie, P. Sex differences in gut microbiota in patients with major depressive disorder. Neuropsychiatr. Dis. Treat. 2018, 14, 647–655. [Google Scholar] [CrossRef]
- Chung, Y.-C.E.; Chen, H.-C.; Chou, H.-C.L.; Chen, I.-M.; Lee, M.-S.; Chuang, L.-C.; Liu, Y.-W.; Lu, M.-L.; Chen, C.-H.; Wu, C.-S. Exploration of microbiota targets for major depressive disorder and mood related traits. J. Psychiatr. Res. 2019, 111, 74–82. [Google Scholar] [CrossRef] [PubMed]
- Guo, Q.; Lin, H.; Chen, P.; Tan, S.; Wen, Z.; Lin, L.; He, J.; Wen, J.; Lu, S. Dynamic changes of intestinal flora in patients with irritable bowel syndrome combined with anxiety and depression after oral administration of enterobacteria capsules. Bioengineered 2021, 12, 11885–11897. [Google Scholar] [CrossRef]
- Hu, S.; Li, A.; Huang, T.; Lai, J.; Li, J.; Sublette, M.E.; Lu, H.; Lu, Q.; Du, Y.; Hu, Z. Gut Microbiota Changes in Patients with Bipolar Depression. Adv. Sci. 2019, 6, 1900752. [Google Scholar] [CrossRef]
- Jiang, H.; Ling, Z.; Zhang, Y.; Mao, H.; Ma, Z.; Yin, Y.; Wang, W.; Tang, W.; Tan, Z.; Shi, J. Altered fecal microbiota composition in patients with major depressive disorder. Brain Behav. Immun. 2015, 48, 186–194. [Google Scholar] [CrossRef]
- Jiang, H.-Y.; Zhang, X.; Yu, Z.-H.; Zhang, Z.; Deng, M.; Zhao, J.-H.; Ruan, B. Altered gut microbiota profile in patients with generalized anxiety disorder. J. Psychiatr. Res. 2018, 104, 130–136. [Google Scholar] [CrossRef] [PubMed]
- Kim, C.-S.; Shin, G.-E.; Cheong, Y.; Shin, J.; Shin, D.-M.; Chun, W.Y. Experiencing social exclusion changes gut microbiota composition. Transl. Psychiatry 2022, 12, 254. [Google Scholar] [CrossRef] [PubMed]
- Lin, P.; Ding, B.; Feng, C.; Yin, S.; Zhang, T.; Qi, X.; Lv, H.; Guo, X.; Dong, K.; Zhu, Y. Prevotella and Klebsiella proportions in fecal microbial communities are potential characteristic parameters for patients with major depressive disorder. J. Affect. Disord. 2017, 207, 300–304. [Google Scholar] [CrossRef]
- Ling, Y.; Gu, Q.; Zhang, J.; Gong, T.; Weng, X.; Liu, J.; Sun, J. Structural Change of Gut Microbiota in Patients with Post-Stroke Comorbid Cognitive Impairment and Depression and Its Correlation with Clinical Features. J. Alzheimer’s Dis. 2020, 77, 1595–1608. [Google Scholar] [CrossRef]
- Liu, Y.; Zhang, L.; Wang, X.; Wang, F.; Zhang, J.; Jiang, R.; Wang, X.; Wang, K.; Liu, Z.; Xia, Z. Similar Fecal Microbiota Signatures in Patients with Diarrhea-Predominant Irritable Bowel Syndrome and Patients with Depression. Clin. Gastroenterol. Hepatol. 2016, 14, 1602–1611.e5. [Google Scholar] [CrossRef] [PubMed]
- Rong, H.; Xie, X.-H.; Zhao, J.; Lai, W.-T.; Wang, M.-B.; Xu, D.; Liu, Y.-H.; Guo, Y.-Y.; Xu, S.-X.; Deng, W.-F. Similarly in depression, nuances of gut microbiota: Evidences from a shotgun metagenomics sequencing study on major depressive disorder versus bipolar disorder with current major depressive episode patients. J. Psychiatr. Res. 2019, 113, 90–99. [Google Scholar] [CrossRef]
- Shen, Y.; Yang, X.; Li, G.; Gao, J.; Liang, Y. The change of gut microbiota in MDD patients under SSRIs treatment. Sci. Rep. 2021, 11, 14918. [Google Scholar] [CrossRef]
- Ye, X.; Wang, D.; Zhu, H.; Wang, D.; Li, J.; Tang, Y.; Wu, J. Gut Microbiota Changes in Patients with Major Depressive Disorder Treated with Vortioxetine. Front. Psychiatry 2021, 12, 641491. [Google Scholar] [CrossRef] [PubMed]
- Tan, C.C.; Chow, C.M. Stress and emotional eating: The mediating role of eating dysregulation. Pers. Individ. Differ. 2014, 66, 1–4. [Google Scholar] [CrossRef]
- van Strien, T.; Ouwens, M.A. Effects of distress, alexithymia and impulsivity on eating. Eat. Behav. 2007, 8, 251–257. [Google Scholar] [CrossRef] [PubMed]
- Laitinen, J.; Ek, E.; Sovio, U. Stress-Related Eating and Drinking Behavior and Body Mass Index and Predictors of This Behavior. Prev. Med. 2002, 34, 29–39. [Google Scholar] [CrossRef] [PubMed]
- Amiri, S.; Behnezhad, S. Obesity and anxiety symptoms: A systematic review and meta-analysis. Neuropsychiatrie 2019, 33, 72–89. [Google Scholar] [CrossRef]
- Macht, M.; Simons, G. Emotions and eating in everyday life. Appetite 2000, 35, 65–71. [Google Scholar] [CrossRef]
- Blair, A.; Lewis, V.; Booth, D. Does emotional eating interfere with success in attempts at weight control? Appetite 1990, 15, 151–157. [Google Scholar] [CrossRef] [PubMed]
- Geliebter, A.; Aversa, A. Emotional eating in overweight, normal weight, and underweight individuals. Eat. Behav. 2002, 3, 341–347. [Google Scholar] [CrossRef]
- Dallman, M.F.; Pecoraro, N.C.; La Fleur, S.E. Chronic stress and comfort foods: Self-medication and abdominal obesity. Brain Behav. Immun. 2005, 19, 275–280. [Google Scholar] [CrossRef]
- Gariepy, G.; Nitka, D.; Schmitz, N. The association between obesity and anxiety disorders in the population: A systematic review and meta-analysis. Int. J. Obes. 2009, 34, 407–419. [Google Scholar] [CrossRef]
- Torres, S.J.; Nowson, C.A. Relationship between stress, eating behavior, and obesity. Nutrition 2007, 23, 887–894. [Google Scholar] [CrossRef]
- Klaczynski, P.A.; Goold, K.W.; Mudry, J.J. Culture, Obesity Stereotypes, Self-Esteem, and the “Thin Ideal”: A Social Identity Perspective. J. Youth Adolesc. 2004, 33, 307–317. [Google Scholar] [CrossRef]
- Harriger, J.A.; Calogero, R.M.; Witherington, D.C.; Smith, J.E. Body Size Stereotyping and Internalization of the Thin Ideal in Preschool Girls. Sex Roles 2010, 63, 609–620. [Google Scholar] [CrossRef]
- Walley, A.J.; Asher, J.E.; Froguel, P. The genetic contribution to non-syndromic human obesity. Nat. Rev. Genet. 2009, 10, 431–442. [Google Scholar] [CrossRef] [PubMed]
- Hettema, J.M.; Neale, M.C.; Kendler, K.S. Reviews and overviews A review and meta-analysis of the genetic epidemiology of anxiety disorders. Psychiatry Interpers. Biol. Process. 2001, 158, 1568–1578. [Google Scholar]
- Elobeid, M.A.; Allison, D.B. Putative environmental-endocrine disruptors and obesity: A review. Curr. Opin. Endocrinol. Diabetes 2008, 15, 403–408. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Gower, B.A.; Shelton, R.C.; Wu, X. Gender-Specific Relationship between Obesity and Major Depression. Front. Endocrinol. 2017, 8, 292. [Google Scholar] [CrossRef]
- Kalra, S.; Kapoor, N.; Arora, S. Gender disparities in people living with obesity—An unchartered territory. J. Mid-Life Health 2021, 12, 103–107. [Google Scholar] [CrossRef]
- Dao, M.C.; Clément, K. Gut microbiota and obesity: Concepts relevant to clinical care. Eur. J. Intern. Med. 2018, 48, 18–24. [Google Scholar] [CrossRef]
- Million, M.; Raoult, D. The Role of the Manipulation of the Gut Microbiota in Obesity. Curr. Infect. Dis. Rep. 2012, 15, 25–30. [Google Scholar] [CrossRef]
- Liu, B.-N.; Liu, X.-T.; Liang, Z.-H.; Wang, J.-H. Gut microbiota in obesity. World J. Gastroenterol. 2021, 27, 3837–3850. [Google Scholar] [CrossRef]
- Rinninella, E.; Raoul, P.; Cintoni, M.; Franceschi, F.; Miggiano, G.A.D.; Gasbarrini, A.; Mele, M.C. What Is the Healthy Gut Microbiota Composition? A Changing Ecosystem across Age, Environment, Diet, and Diseases. Microorganisms 2019, 7, 14. [Google Scholar] [CrossRef]
- Ottman, N.; Smidt, H.; De Vos, W.M.; Belzer, C. The function of our microbiota: Who is out there and what do they do? Front. Cell. Infect. Microbiol. 2012, 2, 104. [Google Scholar] [CrossRef] [PubMed]
- Stojanov, S.; Berlec, A.; Štrukelj, B. The Influence of Probiotics on the Firmicutes/Bacteroidetes Ratio in the Treatment of Obesity and Inflammatory Bowel disease. Microorganisms 2020, 8, 1715. [Google Scholar] [CrossRef]
- Barlow, G.M.; Yu, A.; Mathur, R. Role of the Gut Microbiome in Obesity and Diabetes Mellitus. Nutr. Clin. Pract. 2015, 30, 787–797. [Google Scholar] [CrossRef] [PubMed]
- Sweeney, T.; Morton, J.M. The Human Gut Microbiome: A review of the effect of obesity and surgically induced weight loss. JAMA Surg. 2013, 148, 563–569. [Google Scholar] [CrossRef]
- Schwiertz, A.; Taras, D.; Schäfer, K.; Beijer, S.; Bos, N.A.; Donus, C.; Hardt, P.D. Microbiota and SCFA in Lean and Overweight Healthy Subjects. Obesity 2010, 18, 190–195. [Google Scholar] [CrossRef] [PubMed]
- Magne, F.; Gotteland, M.; Gauthier, L.; Zazueta, A.; Pesoa, S.; Navarrete, P.; Balamurugan, R. The Firmicutes/Bacteroidetes Ratio: A Relevant Marker of Gut Dysbiosis in Obese Patients? Nutrients 2020, 12, 1474. [Google Scholar] [CrossRef]
- Houtman, T.A.; Eckermann, H.A.; Smidt, H.; de Weerth, C. Gut microbiota and BMI throughout childhood: The role of firmicutes, bacteroidetes, and short-chain fatty acid producers. Sci. Rep. 2022, 12, 3140. [Google Scholar] [CrossRef] [PubMed]
- Lin, K.; Zhu, L.; Yang, L. Gut and obesity/metabolic disease: Focus on microbiota metabolites. Medcomm 2022, 3, e171. [Google Scholar] [CrossRef] [PubMed]
- Nobs, S.P.; Tuganbaev, T.; Elinav, E. Microbiome diurnal rhythmicity and its impact on host physiology and disease risk. EMBO Rep. 2019, 20, e47129. [Google Scholar] [CrossRef]
- Sanada, K.; Nakajima, S.; Kurokawa, S.; Barceló-Soler, A.; Ikuse, D.; Hirata, A.; Yoshizawa, A.; Tomizawa, Y.; Salas-Valero, M.; Noda, Y. Gut microbiota and major depressive disorder: A systematic review and meta-analysis. J. Affect. Disord. 2020, 266, 1–13. [Google Scholar] [CrossRef]
- Barandouzi, Z.A.; Starkweather, A.R.; Henderson, W.A.; Gyamfi, A.; Cong, X.S. Altered Composition of Gut Microbiota in Depression: A Systematic Review. Front. Psychiatry 2020, 11, 541. [Google Scholar] [CrossRef] [PubMed]
- Dinan, T.G.; Stilling, R.M.; Stanton, C.; Cryan, J.F. Collective unconscious: How gut microbes shape human behavior. J. Psychiatr. Res. 2015, 63, 1–9. [Google Scholar] [CrossRef]
- Kunugi, H. Gut Microbiota and Pathophysiology of Depressive Disorder. Ann. Nutr. Metab. 2021, 77 (Suppl. 2), 11–20. [Google Scholar] [CrossRef] [PubMed]
- Qin, J.; Li, R.; Raes, J.; Arumugam, M.; Burgdorf, K.S.; Manichanh, C.; Nielsen, T.; Pons, N.; Levenez, F.; Yamada, T. A human gut microbial gene catalogue established by metagenomic sequencing. Nature 2010, 464, 59–65. [Google Scholar] [CrossRef] [PubMed]
- Bai, S.; Bai, H.; Li, D.; Zhong, Q.; Xie, J.; Chen, J.-J. Gut Microbiota-Related Inflammation Factors as a Potential Biomarker for Diagnosing Major Depressive Disorder. Front. Cell. Infect. Microbiol. 2022, 12, 47. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Li, J.; Gui, S.; Zhou, C.; Chen, J.-J.; Yang, C.; Hu, Z.; Wang, H.; Zhong, X.; Zeng, L. Comparative metaproteomics analysis shows altered fecal microbiota signatures in patients with major depressive disorder. Neuroreport 2018, 29, 417–425. [Google Scholar] [CrossRef]
- Młynarska, E.; Gadzinowska, J.; Tokarek, J.; Forycka, J.; Szuman, A.; Franczyk, B.; Rysz, J. The Role of the Microbiome-Brain-Gut Axis in the Pathogenesis of Depressive Disorder. Nutrients 2022, 14, 1921. [Google Scholar] [CrossRef]
- Naseribafrouei, A.; Hestad, K.; Avershina, E.; Sekelja, M.; Linløkken, A.; Wilson, R.; Rudi, K. Correlation between the human fecal microbiota and depression. Neurogastroenterol. Motil. 2014, 26, 1155–1162. [Google Scholar] [CrossRef]
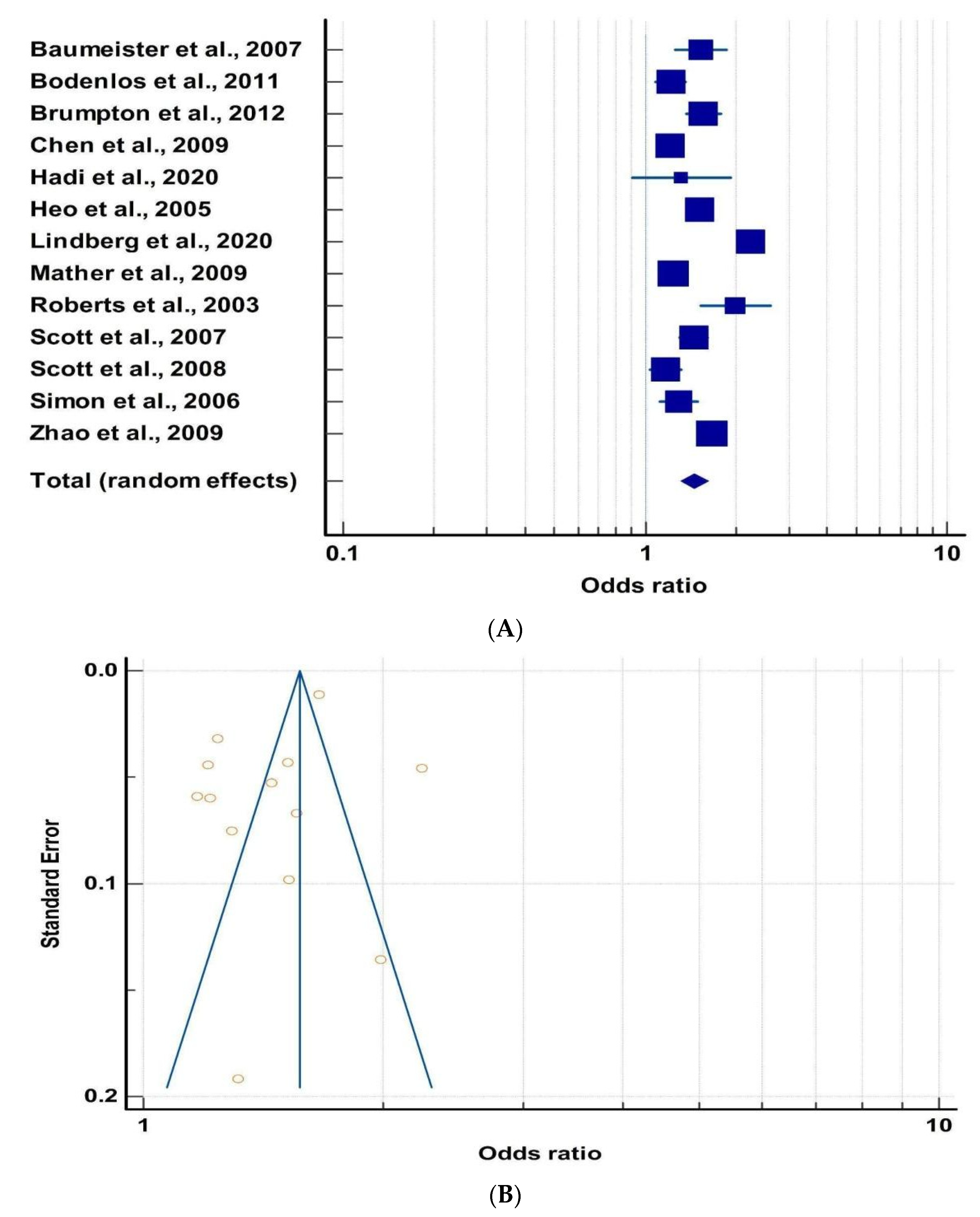
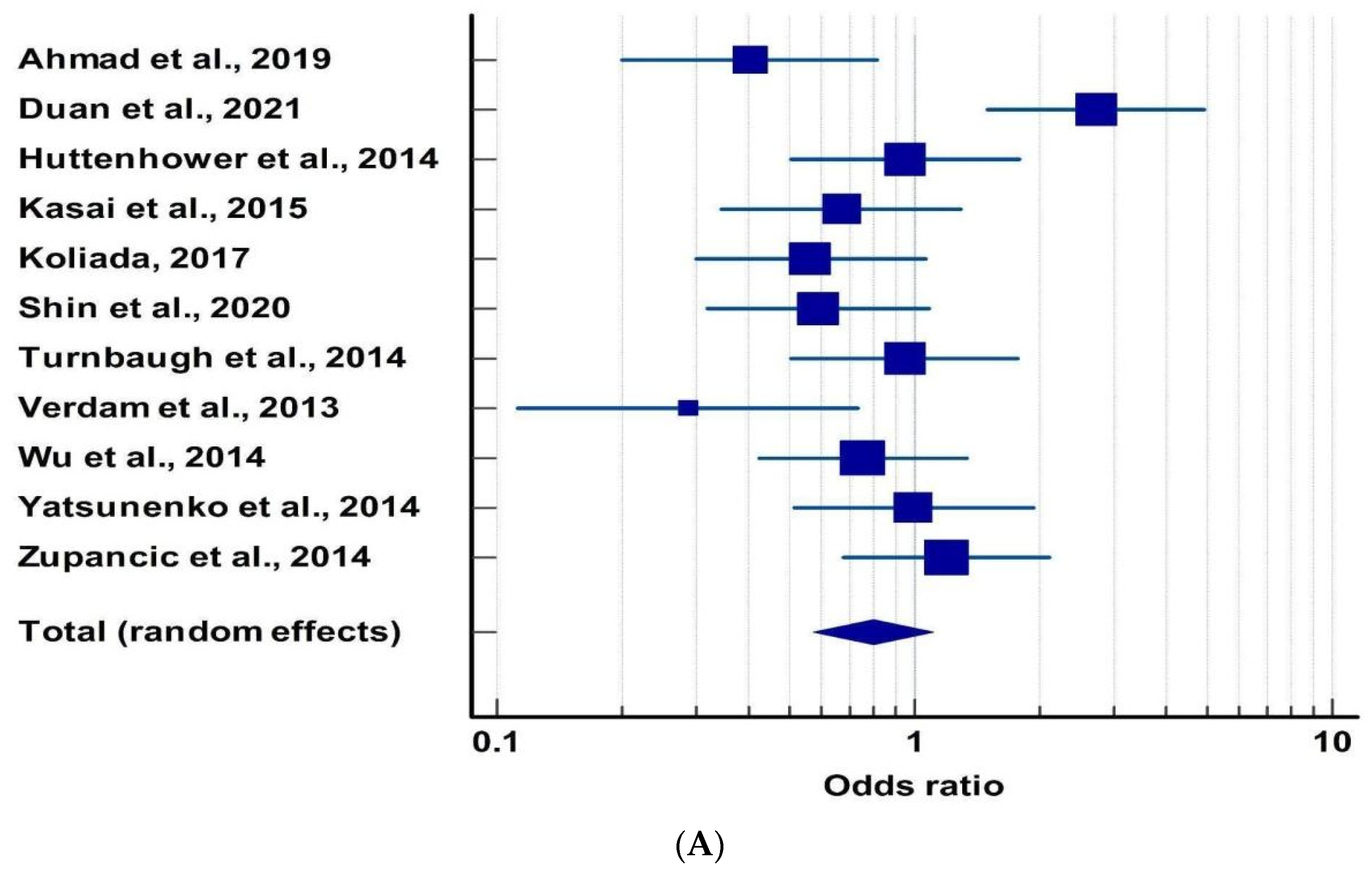
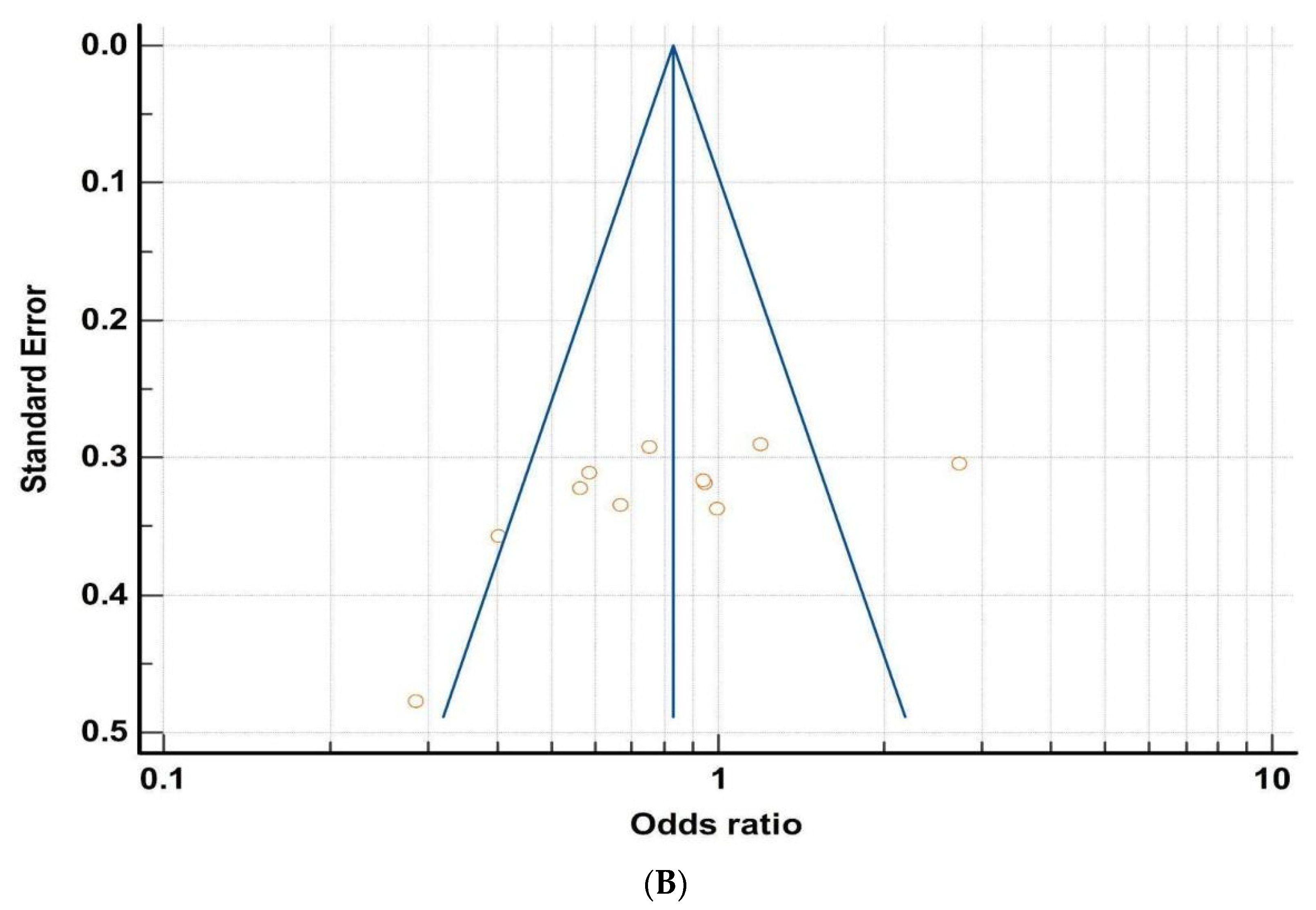
| Publication | Reference | Criteria for MD Evaluation | No. of Subjects (N) | OR | 95% CI | |
|---|---|---|---|---|---|---|
| Lower CI | Upper CI | |||||
| Baumeister et al., 2007 | [45] | Munich composite | 2955 | 1.53 | 1.26 | 1.85 |
| Bodenlos et al., 2011 | [46] | CIDI | 10,899 | 1.22 | 1.08 | 1.37 |
| Brumpton et al., 2012 | [47] | Hospital Anxiety and Depression scale | 44,076 | 1.56 | 1.37 | 1.78 |
| Chen et al., 2009 | [48] | CIDI | 59,652 | 1.21 | 1.1 | 1.31 |
| Hadi et. al., 2020 | [44] | HAMD | 614 | 1.32 | 0.9 | 1.91 |
| Heo et al., 2005 | [49] | DSM | 34,702 | 1.52 | 1.4 | 1.66 |
| Lindberg et al., 2020 | [50] | ICD-10, ATC, etc. | 72,570 | 2.24 | 2.05 | 2.45 |
| Mather et al., 2009 | [51] | DSM | 34,900 | 1.24 | 1.16 | 1.32 |
| Roberts et al., 2003 | [52] | DSM | 3772 | 1.99 | 1.52 | 2.59 |
| Scott et al., 2008 | [53] | DSM | 12,738 | 1.45 | 1.31 | 1.61 |
| Scott et al., 2008 | [9] | CIDI 3.0 | 73,135 | 1.17 | 1.04 | 1.31 |
| Simon et al., 2006 | [54] | WHO Composite | 9125 | 1.29 | 1.12 | 1.5 |
| Zhao et al., 2009 | [55] | PHQ-8 | 177,047 | 1.67 | 1.63 | 1.7 |
| Cumulative Odds: 1.46 | ||||||
| Publication | Reference | BMI Range (kg/m2) | No. of Subjects (N) | OR | 95% CI | ||
|---|---|---|---|---|---|---|---|
| Obese | Normal | Lower CI | Upper CI | ||||
| Ahmad et al., 2019 | [56] | 32.4 + −3.6 | 22.08 + −3.1 | 60 | 0.40 | 0.19 | 0.81 |
| Duan et al., 2021 | [57] | >30 | <25 | 42 | 2.72 | 1.49 | 4.94 |
| Huttenhower et al., 2014 | [58] | >30 | <25 | N/A | 0.95 | 0.51 | 1.77 |
| Kasai et al., 2015 | [59] | ≥25 | 18.5–25 | 56 | 0.67 | 0.35 | 1.28 |
| Koliada et al., 2017 | [60] | >30 | 18.5–24.9 | 38 | 0.56 | 0.29 | 1.06 |
| Shin et al., 2020 | [61] | 18.5–25 | ≥25 | 46 | 0.58 | 0.32 | 1.08 |
| Turnbaugh et al., 2014 | [62] | >30 | <25 | 154 | 0.94 | 0.51 | 1.76 |
| Verdam et al., 2013 | [63] | >30 | <25 | 28 | 0.29 | 0.11 | 0.73 |
| Wu et al., 2014 | [64] | >30 | <25 | N/A | 0.75 | 0.42 | 1.33 |
| Yatsunenko et al., 2014 | [65] | >30 | <25 | N/A | 0.99 | 0.51 | 1.93 |
| Zupancic et al., 2014 | [66] | >30 | <25 | 310 | 1.19 | 0.68 | 2.10 |
| Cumulative Odds: 0.797 | |||||||
| Publication | Reference | Criteria for MDs Evaluation | No. of Subjects (N) | OR | 95% CI | |
|---|---|---|---|---|---|---|
| Lower CI | Upper CI | |||||
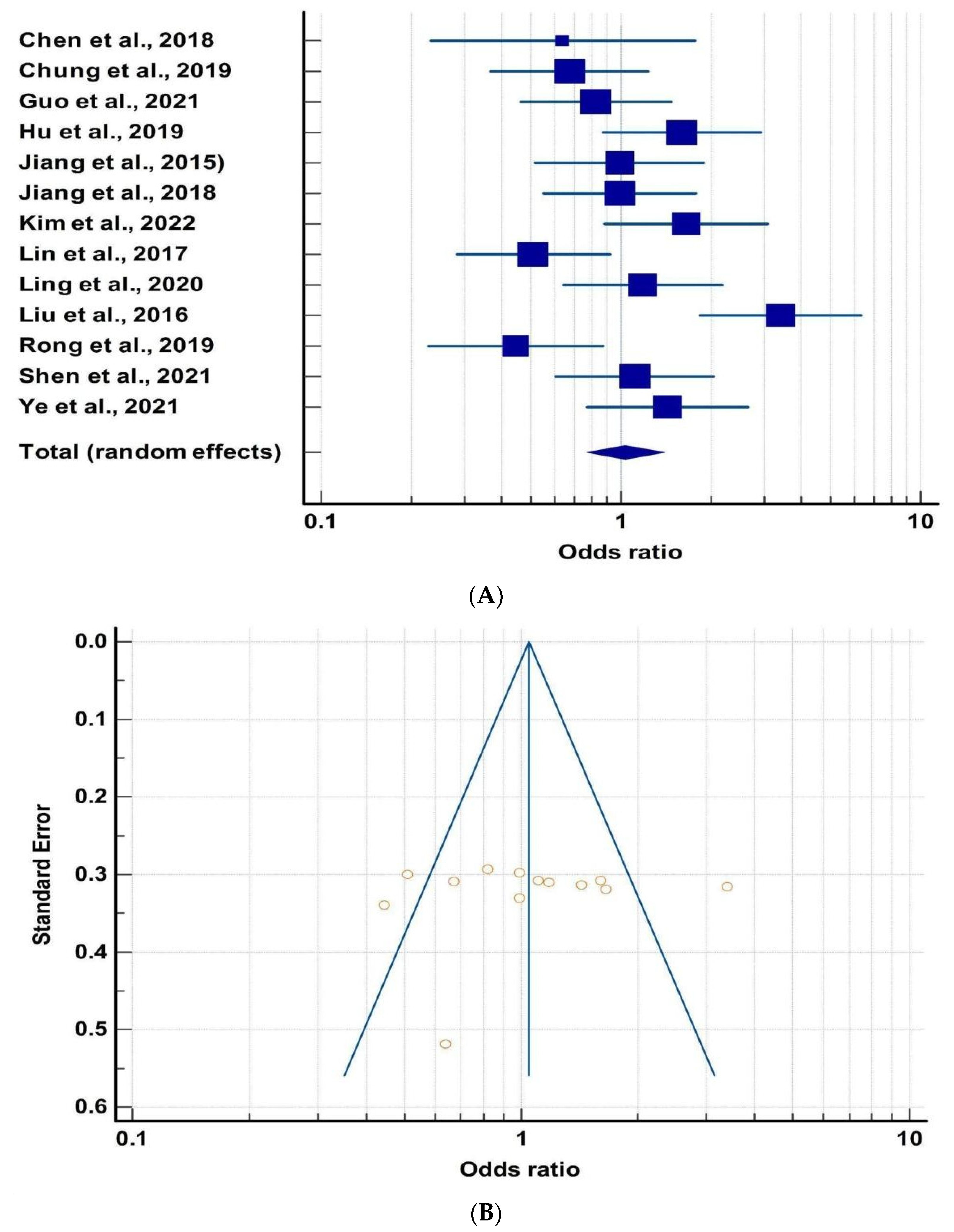
| Chen et al., 2018 | [67] | DSM, HAMD | 48 | 0.64 | 0.23 | 1.76 |
| Chung et al., 2019 | [68] | DSM, BAI, BDI | 73 | 0.67 | 0.36 | 1.23 |
| Guo et al., 2021 | [69] | HAMD, HAMA | 18 | 0.82 | 0.46 | 1.46 |
| Hu et al., 2019 | [70] | MINI, DSM, HAMD, MADRS | 97 | 1.6 | 0.87 | 2.92 |
| Jiang et al., 2015 | [71] | HAMD, MADRS | 59 | 0.98 | 0.51 | 1.89 |
| Jiang et al., 2018 | [72] | MINI, HAMA | 76 | 0.99 | 0.55 | 2.77 |
| Kim et al., 2022 | [73] | BAI, BDI | 39 | 1.65 | 0.88 | 3.09 |
| Lin et al., 2017 | [74] | DSM, HAMD | 20 | 0.51 | 0.28 | 0.91 |
| Ling et al., 2020 | [75] | HAMD, MoCA | 66 | 1.18 | 0.64 | 2.16 |
| Liu et al., 2017 | [76] | MINI, DSM | 35 | 3.4 | 1.83 | 6.31 |
| Rong et al., 2019 | [77] | DSM, HAMD | 61 | 0.44 | 0.22 | 0.86 |
| Shen et al., 2021 | [78] | MINI, HAMD | 60 | 1.11 | 0.6 | 2.03 |
| Ye et al., 2021 | [79] | DSM, HAMD | 54 | 1.42 | 0.77 | 2.64 |
| Cumulative Odds: 1.03 | ||||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Panda, S.S.; Nayak, A.; Shah, S.; Aich, P. A Systematic Review on the Association between Obesity and Mood Disorders and the Role of Gut Microbiota. Metabolites 2023, 13, 488. https://doi.org/10.3390/metabo13040488
Panda SS, Nayak A, Shah S, Aich P. A Systematic Review on the Association between Obesity and Mood Disorders and the Role of Gut Microbiota. Metabolites. 2023; 13(4):488. https://doi.org/10.3390/metabo13040488
Chicago/Turabian StylePanda, Swati Sagarika, Akankshya Nayak, Srishti Shah, and Palok Aich. 2023. "A Systematic Review on the Association between Obesity and Mood Disorders and the Role of Gut Microbiota" Metabolites 13, no. 4: 488. https://doi.org/10.3390/metabo13040488
APA StylePanda, S. S., Nayak, A., Shah, S., & Aich, P. (2023). A Systematic Review on the Association between Obesity and Mood Disorders and the Role of Gut Microbiota. Metabolites, 13(4), 488. https://doi.org/10.3390/metabo13040488







