Branched-Chain and Aromatic Amino Acids in Relation to Fat Mass and Fat-Free Mass Changes among Adolescents: A School-Based Intervention
Abstract
:1. Introduction
2. Results
2.1. Overall Weight Changes
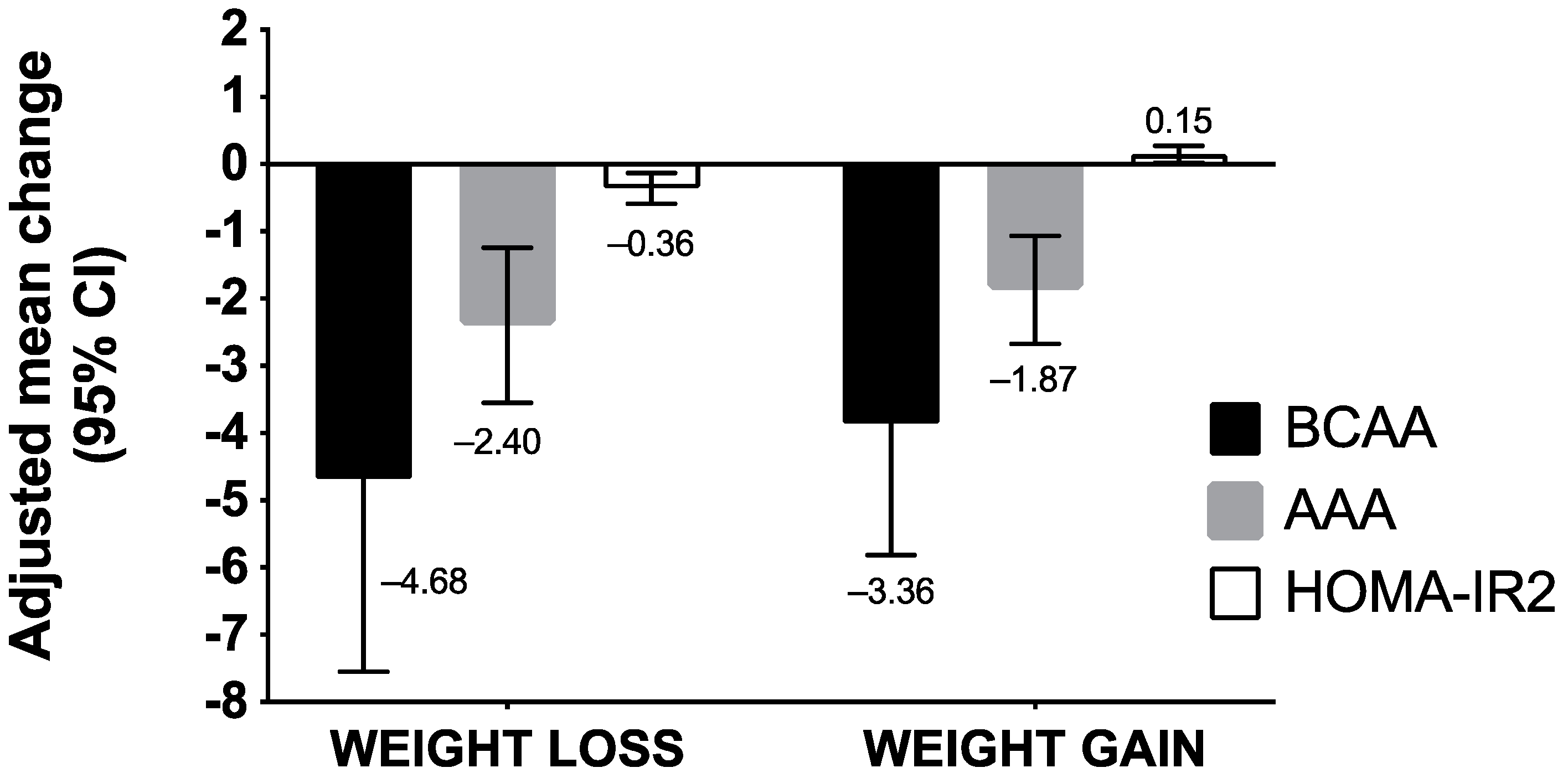
2.2. Changes in BCAA/AAA Concentrations and HOMA-IR2 According to Weight Status at Baseline
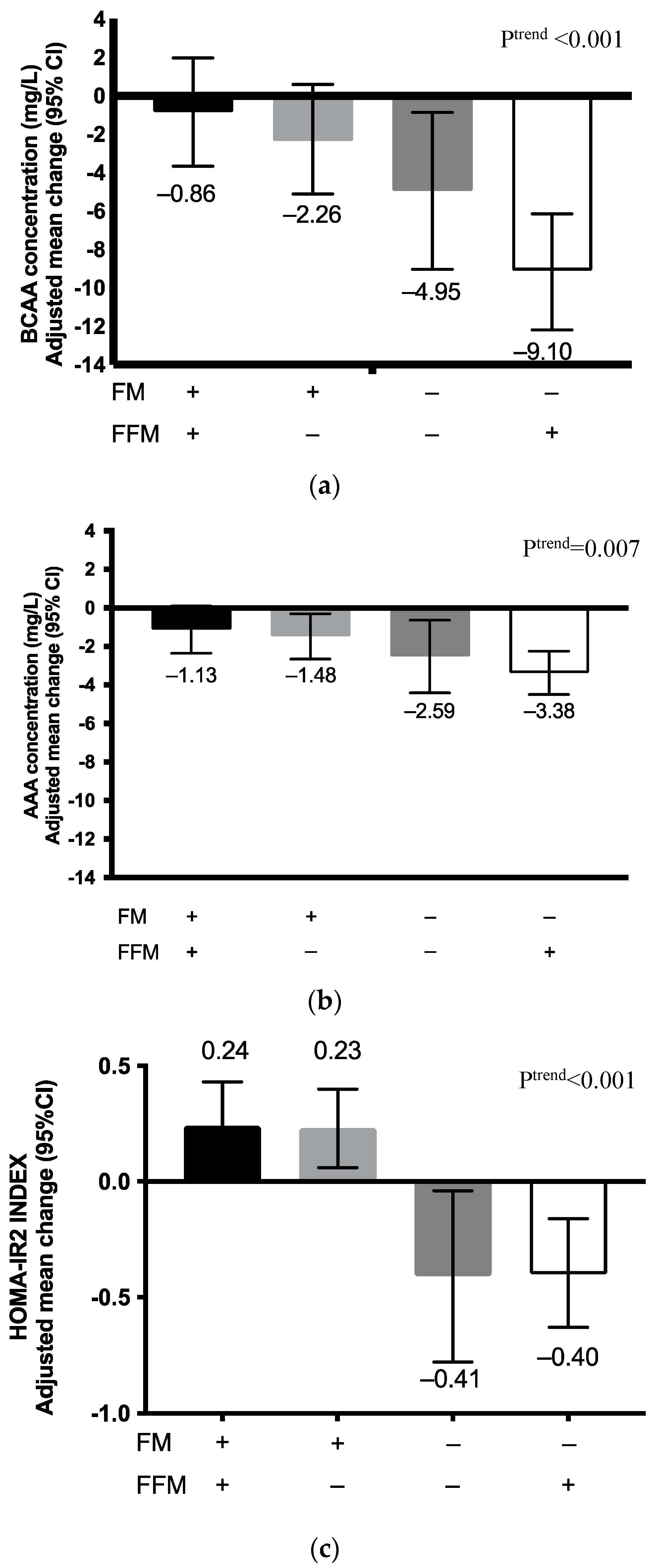
2.3. Changes in BCAA and AAA Concentrations and HOMA-IR2 According to Fat Mass and Fat-Free Mass Changes
3. Discussion
4. Materials and Methods
4.1. Ethical Statement
4.2. Study Design and Participants
4.3. Intervention
4.4. Outcomes
4.4.1. Plasma Amino Acids
4.4.2. Homeostasis Model Assessment of Insulin Resistance
4.4.3. Body Weight and Body Composition
4.4.4. Triglyceride and HDL-Cholesterol
4.5. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Taylor, S.A.; Borzutzky, C.; Jasik, C.B.; Mihalopoulos, N.L.; Smith-Barron, K.; Woolford, S.J.; Garber, A.; McPherson, M.; AlBuhairan, F.S.; Kohn, M.; et al. Society for Adolescent Health and Medicine Preventing and Treating Adolescent Obesity: A Position Paper of the Society for Adolescent Health and Medicine. J. Adolesc. Health 2016, 59, 602–606. [Google Scholar] [CrossRef]
- GBD 2015 Obesity Collaborators; Afshin, A.; Forouzanfar, M.H.; Reitsma, M.B.; Sur, P.; Estep, K.; Lee, A.; Marczak, L.; Mokdad, A.H.; Moradi-Lakeh, M.; et al. Health Effects of Overweight and Obesity in 195 Countries over 25 Years. N. Engl. J. Med. 2017, 377, 13–27. [Google Scholar] [CrossRef] [PubMed]
- Gatti, C.; Lucas, M.; Dewailly, É.; Suhas, E. Ressources Alimentaires et Santé aux Australes (Rapport Final); Institut Louis Malardé: Papeete, French Polynesia, 2013. (In French) [Google Scholar]
- Sievert, K.; Lawrence, M.; Naika, A.; Baker, P. Processed Foods and Nutrition Transition in the Pacific: Regional Trends, Patterns and Food System Drivers. Nutrients 2019, 11, 1328. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Counil, E.; Brindel, P.; Château-Degat, M. La Transition Alimentaire et Sanitaire en Polynésie Françaised: Faits Saillants des Résultats de L’étude Menée en 2007 à Papeete et Dans les Îles Australes; Université Laval, Institut Louis Malardé: Papeete, French Polynesia, 2009. [Google Scholar]
- Sahoo, K.; Sahoo, B.; Choudhury, A.K.; Sofi, N.Y.; Kumar, R.; Bhadoria, A.S. Childhood Obesity: Causes and Consequences. J. Fam. Med. Prim. Care 2015, 4, 187–192. [Google Scholar] [CrossRef]
- Ward, Z.J.; Long, M.W.; Resch, S.C.; Giles, C.M.; Cradock, A.L.; Gortmaker, S.L. Simulation of Growth Trajectories of Childhood Obesity into Adulthood. N. Engl. J. Med. 2017, 377, 2145–2153. [Google Scholar] [CrossRef]
- Hawley, N.L.; McGarvey, S.T. Obesity and Diabetes in Pacific Islanders: The Current Burden and the Need for Urgent Action. Curr. Diabetes Rep. 2015, 15, 29. [Google Scholar] [CrossRef]
- Atallah, A.; Atallah, V.; Daigre, J.; Boissin, J.; Kangambega, P.; Larifla, L.; Inamo, J. High Blood Pressure and Obesity: Disparities among Four French Overseas Territories. J. Hypertens. 2014, 63, 155–162. [Google Scholar]
- Dobbins, M.; Husson, H.; DeCorby, K.; LaRocca, R.L. School-based Physical Activity Programs for Promoting Physical Activity and Fitness in Children and Adolescents Aged 6 to 18. Cochrane Database Syst. Rev. 2013, 1, CD007651. [Google Scholar] [CrossRef]
- Shackleton, N.; Jamal, F.; Viner, R.M.; Dickson, K.; Patton, G.; Bonell, C. School-Based Interventions Going beyond Health Education to Promote Adolescent Health: Systematic Review of Reviews. J. Adolesc. Health 2016, 58, 382–396. [Google Scholar] [CrossRef] [Green Version]
- Millstein, R.A. Measuring Outcomes in Adult Weight Loss Studies That Include Diet and Physical Activity: A Systematic Review. J. Nutr. Metab. 2014, 2014, 421423. [Google Scholar] [CrossRef]
- Prentice, A.M.; Jebb, S.A. Beyond Body Mass Index. Obes. Rev. 2001, 2, 141–147. [Google Scholar] [CrossRef] [PubMed]
- Suhre, K. Metabolic Profiling in Diabetes. J. Endocrinol. 2014, 221, R75–R85. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, T.J.; Larson, M.G.; Vasan, R.S.; Cheng, S.; Rhee, E.P.; McCabe, E.; Lewis, G.D.; Fox, C.S.; Jacques, P.F.; Fernandez, C. Metabolite Profiles and the Risk of Developing Diabetes. Nat. Med. 2011, 17, 448–453. [Google Scholar] [CrossRef]
- Würtz, P.; Soininen, P.; Kangas, A.J.; Rönnemaa, T.; Lehtimäki, T.; Kähönen, M.; Viikari, J.S.; Raitakari, O.T.; Ala-Korpela, M. Branched-Chain and Aromatic Amino Acids Are Predictors of Insulin Resistance in Young Adults. Diabetes Care 2013, 36, 648–655. [Google Scholar] [CrossRef] [Green Version]
- Gaggini, M.; Carli, F.; Rosso, C.; Buzzigoli, E.; Marietti, M.; Della Latta, V.; Ciociaro, D.; Abate, M.L.; Gambino, R.; Cassader, M.; et al. Altered Amino Acid Concentrations in NAFLD: Impact of Obesity and Insulin Resistance. Hepatology 2018, 67, 145–158. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gatti, C.; Suhas, E.; Côté, S.; Laouan-Sidi, E.A.; Dewailly, É.; Lucas, M. Obesity and Metabolic Parameters in Adolescents: A School-Based Intervention Program in French Polynesia. J. Adolesc. Health 2015, 56, 174–180. [Google Scholar] [CrossRef]
- Herman, M.A.; She, P.; Peroni, O.D.; Lynch, C.J.; Kahn, B.B. Adipose Tissue Branched Chain Amino Acid (BCAA) Metabolism Modulates Circulating BCAA Levels. J. Biol. Chem. 2010, 285, 11348–11356. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Reinehr, T.; Wolters, B.; Knop, C.; Lass, N.; Hellmuth, C.; Harder, U.; Peissner, W.; Wahl, S.; Grallert, H.; Adamski, J. Changes in the Serum Metabolite Profile in Obese Children with Weight Loss. Eur. J. Nutr. 2015, 54, 173–181. [Google Scholar] [CrossRef]
- Grulich-Henn, J.; Lichtenstein, S.; Hörster, F.; Hoffmann, G.; Nawroth, P.; Hamann, A. Moderate Weight Reduction in an Outpatient Obesity Intervention Program Significantly Reduces Insulin Resistance and Risk Factors for Cardiovascular Disease in Severely Obese Adolescents. Int. J. Endocrinol. 2011, 2011, 541021. [Google Scholar] [CrossRef] [Green Version]
- Marks, B.L.; Rippe, J.M. The Importance of Fat Free Mass Maintenance in Weight Loss Programmes. Sports Med. 1996, 22, 273–281. [Google Scholar] [CrossRef]
- Stiegler, P.; Cunliffe, A. The Role of Diet and Exercise for the Maintenance of Fat-Free Mass and Resting Metabolic Rate during Weight Loss. Sports Med. 2006, 36, 239–262. [Google Scholar] [CrossRef]
- Fukushima, Y.; Kurose, S.; Shinno, H.; Thu, H.C.; Takao, N.; Tsutsumi, H.; Kimura, Y. Importance of Lean Muscle Maintenance to Improve Insulin Resistance by Body Weight Reduction in Female Patients with Obesity. Diabetes Metab. J. 2016, 40, 147–153. [Google Scholar] [CrossRef] [PubMed]
- Petersen, K.F.; Befroy, D.; Dufour, S.; Dziura, J.; Ariyan, C.; Rothman, D.L.; DiPietro, L.; Cline, G.W.; Shulman, G.I. Mitochondrial Dysfunction in the Elderly: Possible Role in Insulin Resistance. Science 2003, 300, 1140–1142. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huang, J.H.; Hood, D.A. Age-Associated Mitochondrial Dysfunction in Skeletal Muscle: Contributing Factors and Suggestions for Long-Term Interventions. IUBMB Life 2009, 61, 201–214. [Google Scholar] [CrossRef] [PubMed]
- Tabák, A.G.; Jokela, M.; Akbaraly, T.N.; Brunner, E.J.; Kivimäki, M.; Witte, D.R. Trajectories of Glycaemia, Insulin Sensitivity, and Insulin Secretion before Diagnosis of Type 2 Diabetes: An Analysis from the Whitehall II Study. Lancet 2009, 373, 2215–2221. [Google Scholar] [CrossRef] [Green Version]
- Achamrah, N.; Colange, G.; Delay, J.; Rimbert, A.; Folope, V.; Petit, A.; Grigioni, S.; Déchelotte, P.; Coëffier, M. Comparison of Body Composition Assessment by DXA and BIA According to the Body Mass Index: A Retrospective Study on 3655 Measures. PLoS ONE 2018, 13, e0200465. [Google Scholar] [CrossRef] [Green Version]
- Institut National D’excellence en Santé et en Services Sociaux. Traitement de L’OBÉSITÉ des Enfants et des Adolescents en 1re et 2e Ligne: Guide de Pratique Clinique; INESSS: Montreal, QC, Canada, 2012; p. 62. [Google Scholar]
- Roy, C.; Tremblay, P.-Y.; Bienvenu, J.-F.; Ayotte, P. Quantitative Analysis of Amino Acids and Acylcarnitines Combined with Untargeted Metabolomics Using Ultra-High Performance Liquid Chromatography and Quadrupole Time-of-Flight Mass Spectrometry. J. Chromatogr. B 2016, 1027, 40–49. [Google Scholar] [CrossRef]
- University of Oxford. Homa2 Calculator. Available online: https://www.dtu.ox.ac.uk/homacalculator/ (accessed on 19 January 2017).
- Cole, T.J.; Bellizzi, M.C.; Flegal, K.M.; Dietz, W.H. Establishing a Standard Definition for Child Overweight and Obesity Worldwide: International Survey. BMJ 2000, 320, 1240. [Google Scholar] [CrossRef] [Green Version]
| Characteristics Mean (SD) | TOTAL (n = 226) | Attendance Status | p-Value † | |
|---|---|---|---|---|
| Residents (n = 69) | Externs/ Half-Residents (n = 157) | |||
| Age, years | 13.5 (1.6) | 14.3 (1.5) | 13.2 (1.6) | <0.001 |
| (min., max.) | (10, 18) | (11, 18) | (10, 17) | |
| Girls, n (%) * | 108 (47.79) | 37 (53.62) | 71 (45.22) | 0.24 |
| Weight, kg | 69.6 (19.1) | 72.7 (17.1) | 68.2 (19.9) | 0.10 |
| Height, cm | 167 (9.46) | 168 (8.96) | 16 (9.67) | 0.15 |
| BMI, kg/m2 | 24.7 (5.5) | 25.5 (4.7) | 24.4 (5.8) | 0.18 |
| Fat-free mass, kg | 51.7 (12.4) | 53.4 (12.1) | 50.9 (12.5) | 0.17 |
| Fat mass, kg | 17.7 (11.2) | 18.6 (10.3) | 17.3 (11.6) | 0.42 |
| Fat mass, % | 24.9 (10.2) | 24.6 (10.5) | 23.7 (10.1) | 0.52 |
| Fasting insulin, pmol/L | 108 (92.8) | 92.4 (49.1) | 115 (112) | 0.11 |
| Fasting glucose, pmol/L | 4.90 (0.38) | 4.76 (0.33) | 4.95 (0.40) | 0.001 |
| TG, mmol/L | 0.96 (0.62) | 0.87 (0.82) | 1.00 (0.54) | 0.09 |
| HDL, mmol/L | 1.23 (0.27) | 1.26 (0.23) | 1.23 (0.29) | 0.44 |
| TG/HDL | 0.87 (0.66) | 0.74 (0.48) | 0.93 (0.76) | 0.06 |
| Weight status, N (%) *‡ | 0.51 | |||
| Normal | 93 (41.15) | 25 (36.23) | 68 (43.31) | |
| Overweight | 56 (24.78) | 17 (24.64) | 39 (24.84) | |
| Obese | 77 (34.07) | 27 (39.13) | 50 (31.85) | |
| Analysis § | ||||||
|---|---|---|---|---|---|---|
| IOTF Weight and Attendance Status | Baseline † | Post-Intervention † | Change ‡ Mean (95% CI) | Group Effect | Time Effect | Group × Time Interaction |
| p Value | ||||||
| All (n = 226)¶ | ||||||
| BCAA | 0.95 | <0.001 | 0.48 | |||
| Residents | 59.3 (10.8) | 56.5 (9.2) | −3.25 (−6.43 to −0.07) | |||
| Half-Residents/Ext. | 61.6 (10.7) | 57.3 (8.9) | −4.53 (−6.39 to −2.68) | |||
| AAA | 0.71 | <0.001 | 0.08 | |||
| Residents | 26.1 (4.7) | 25.1 (3.1) | −1.16 (−2.16 to −0.15) | |||
| Half-Residents/Ext. | 27.7 (4.6) | 25.3 (3.1) | −2.44 (−3.27 to −1.61) | |||
| Normal weight (n = 93) | ||||||
| BCAA | 0.57 | 0.11 | 0.84 | |||
| Residents | 59.8 (11.7) | 56.2 (7.4) | −2.68 (−8.04 to 2.67) | |||
| Half-Residents/Ext. | 57.5 (9.5) | 54.6 (9.2) | −2.09 (−4.92 to 0.74) | |||
| AAA | 0.30 | 0.59 | 0.35 | |||
| Residents | 25.0 (3.2) | 25.1 (2.6) | 0.24 (−1.30 to 1.78) | |||
| Half-Residents/Ext. | 25.6 (4.5) | 24.4 (3.1) | −0.87 (−2.15 to 0.40) | |||
| Overweight (n = 56) | ||||||
| BCAA | 0.52 | <0.001 | 0.26 | |||
| Residents | 64.9 (11.9) | 57.9 (7.5) | −8.87 (−13.2 to −4.52) | |||
| Half-Residents/Ext. | 61.4 (9.5) | 57.0 (9.3) | −5.73 (−8.71 to −2.75) | |||
| AAA | 0.28 | <0.001 | 0.73 | |||
| Residents | 26.9 (3.2) | 24.2 (2.6) | −3.08 (−4.46 to −1.69) | |||
| Half-Residents/Ext. | 27.2 (4.6) | 25.1 (3.1) | −2.68 (−4.04 to −1.32) | |||
| Obese (n = 77) | ||||||
| BCAA | 0.62 | 0.03 | 0.04 | |||
| Residents | 58.5 (11.8) | 57.2 (7.4) | −0.25 (−5.70 to 5.20) | |||
| Half-Residents/Ext. | 65.2 (9.6) | 59.8 (9.4) | −6.92 (−10.5 to −3.38) | |||
| AAA | 0.96 | <0.001 | 0.01 | |||
| Residents | 27.2 (3.2) | 25.8 (2.6) | −1.24 (−3.02 to 0.54) | |||
| Half-Residents/Ext. | 30.6 (4.6) | 26.8 (3.5) | −4.39 (−5.83 to −2.95) | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pana, M.P.; Ayotte, P.; Anassour-Laouan-Sidi, E.; Suhas, E.; Gatti, C.M.I.; Lucas, M. Branched-Chain and Aromatic Amino Acids in Relation to Fat Mass and Fat-Free Mass Changes among Adolescents: A School-Based Intervention. Metabolites 2022, 12, 589. https://doi.org/10.3390/metabo12070589
Pana MP, Ayotte P, Anassour-Laouan-Sidi E, Suhas E, Gatti CMI, Lucas M. Branched-Chain and Aromatic Amino Acids in Relation to Fat Mass and Fat-Free Mass Changes among Adolescents: A School-Based Intervention. Metabolites. 2022; 12(7):589. https://doi.org/10.3390/metabo12070589
Chicago/Turabian StylePana, Magnoudewa Priscille, Pierre Ayotte, Elhadji Anassour-Laouan-Sidi, Edouard Suhas, Clémence Mahana Iti Gatti, and Michel Lucas. 2022. "Branched-Chain and Aromatic Amino Acids in Relation to Fat Mass and Fat-Free Mass Changes among Adolescents: A School-Based Intervention" Metabolites 12, no. 7: 589. https://doi.org/10.3390/metabo12070589
APA StylePana, M. P., Ayotte, P., Anassour-Laouan-Sidi, E., Suhas, E., Gatti, C. M. I., & Lucas, M. (2022). Branched-Chain and Aromatic Amino Acids in Relation to Fat Mass and Fat-Free Mass Changes among Adolescents: A School-Based Intervention. Metabolites, 12(7), 589. https://doi.org/10.3390/metabo12070589








