Integration of Liver Glycogen and Triglyceride NMR Isotopomer Analyses Provides a Comprehensive Coverage of Hepatic Glucose and Fructose Metabolism
Abstract
1. Introduction
1.1. Background
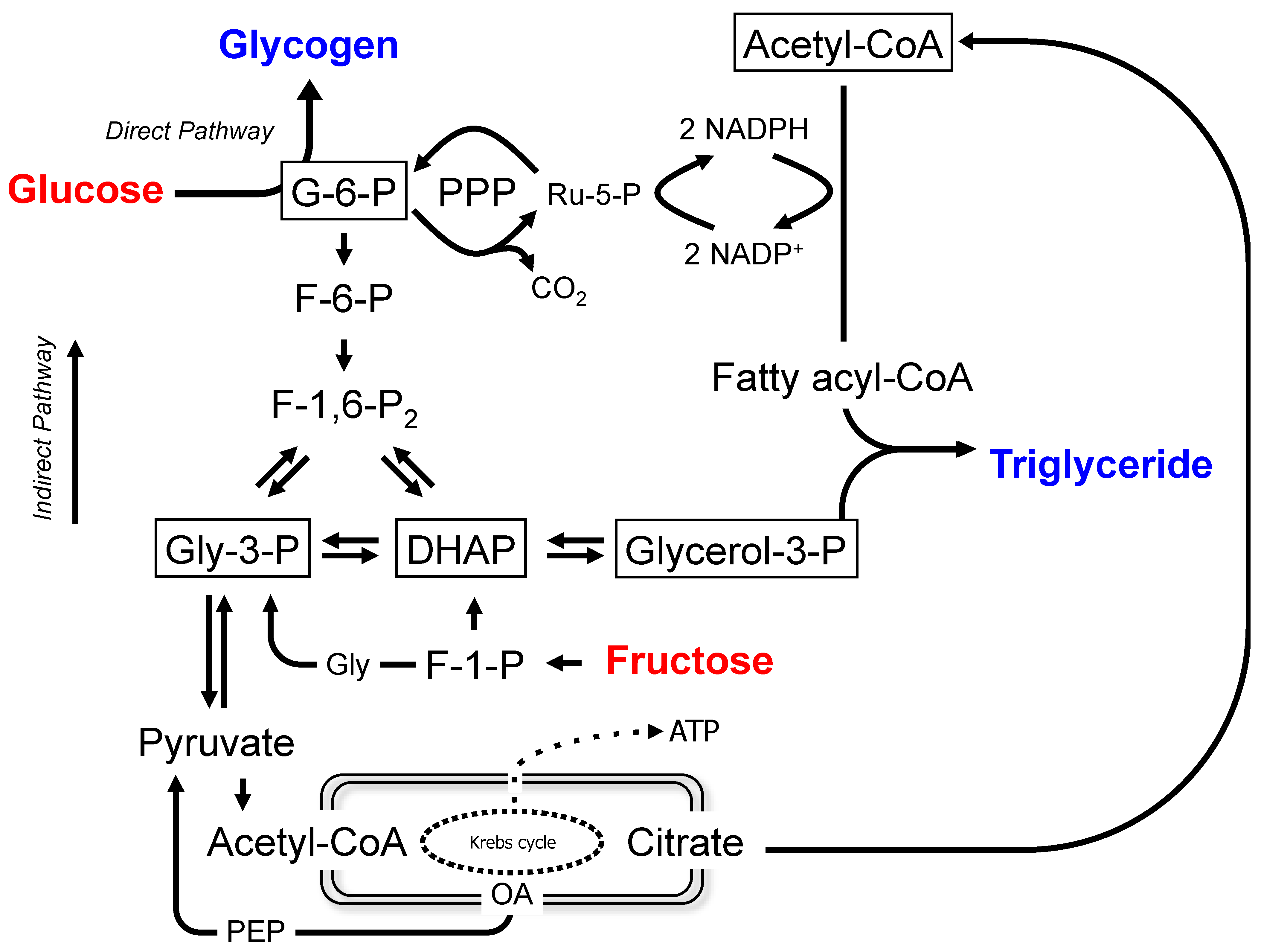
1.2. Metabolic Model
2. Methods
2.1. Materials
2.2. Animal Studies
2.3. Analysis of Glycogen and Triglyceride Isotopic Enrichments by NMR
2.3.1. NMR Analysis of Glycogen 2H and 13C-Enrichments
2.3.2. NMR Analysis of Triglyceride 2H and 13C Enrichments
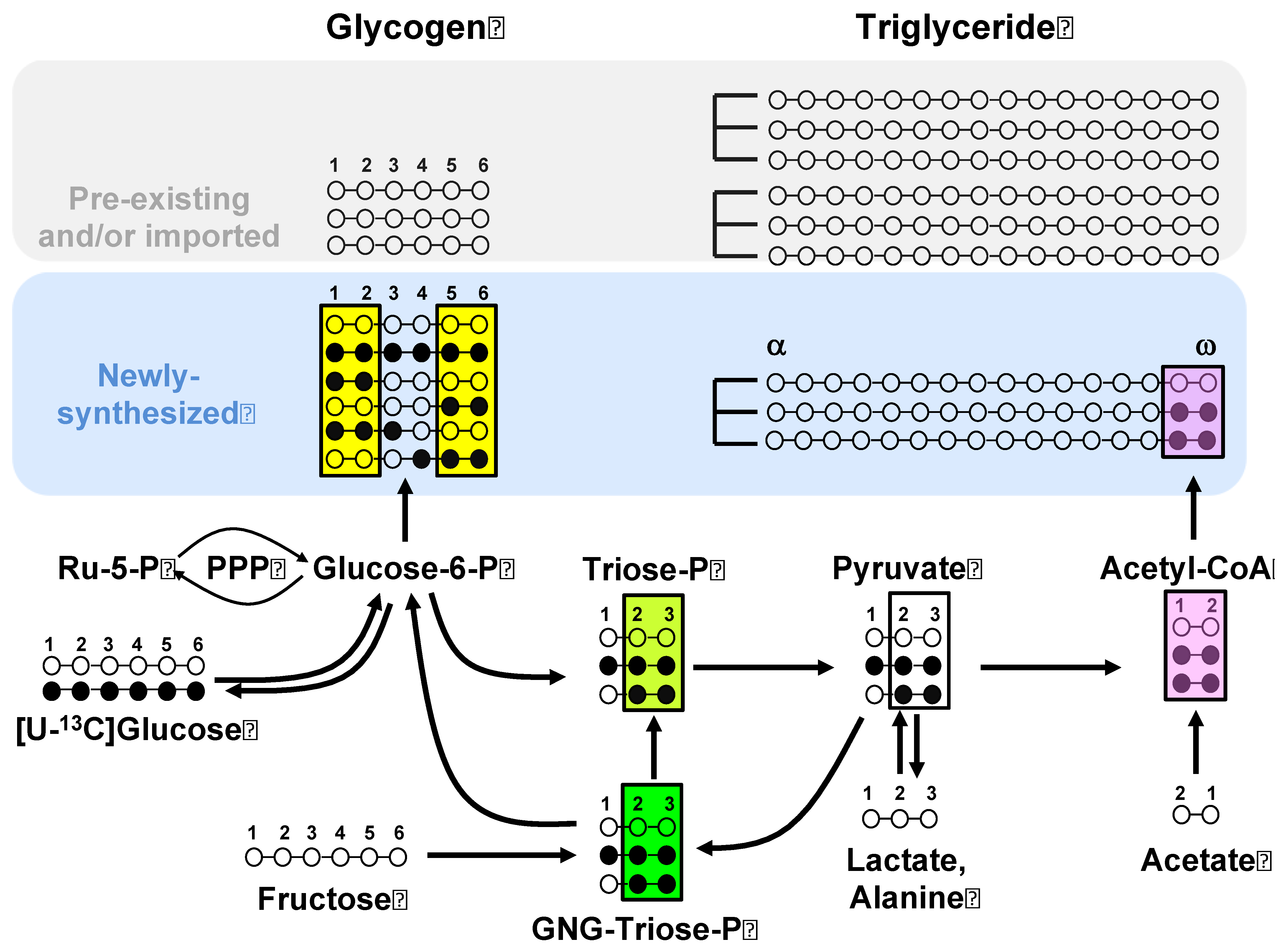
2.4. Estimation of Substrate Contributions to Lipogenesis from Analysis of Newly Synthesized Glycogen and Triglyceride 13C Isotopomers
2.5. Estimation of the Fraction of Glucose-6-P Metabolized by the PPP
2.6. Statistical Analyses
3. Results
3.1. Enrichment of Hepatic Metabolic Pools from [U-13C]Glucose and [U-13C]Fructose
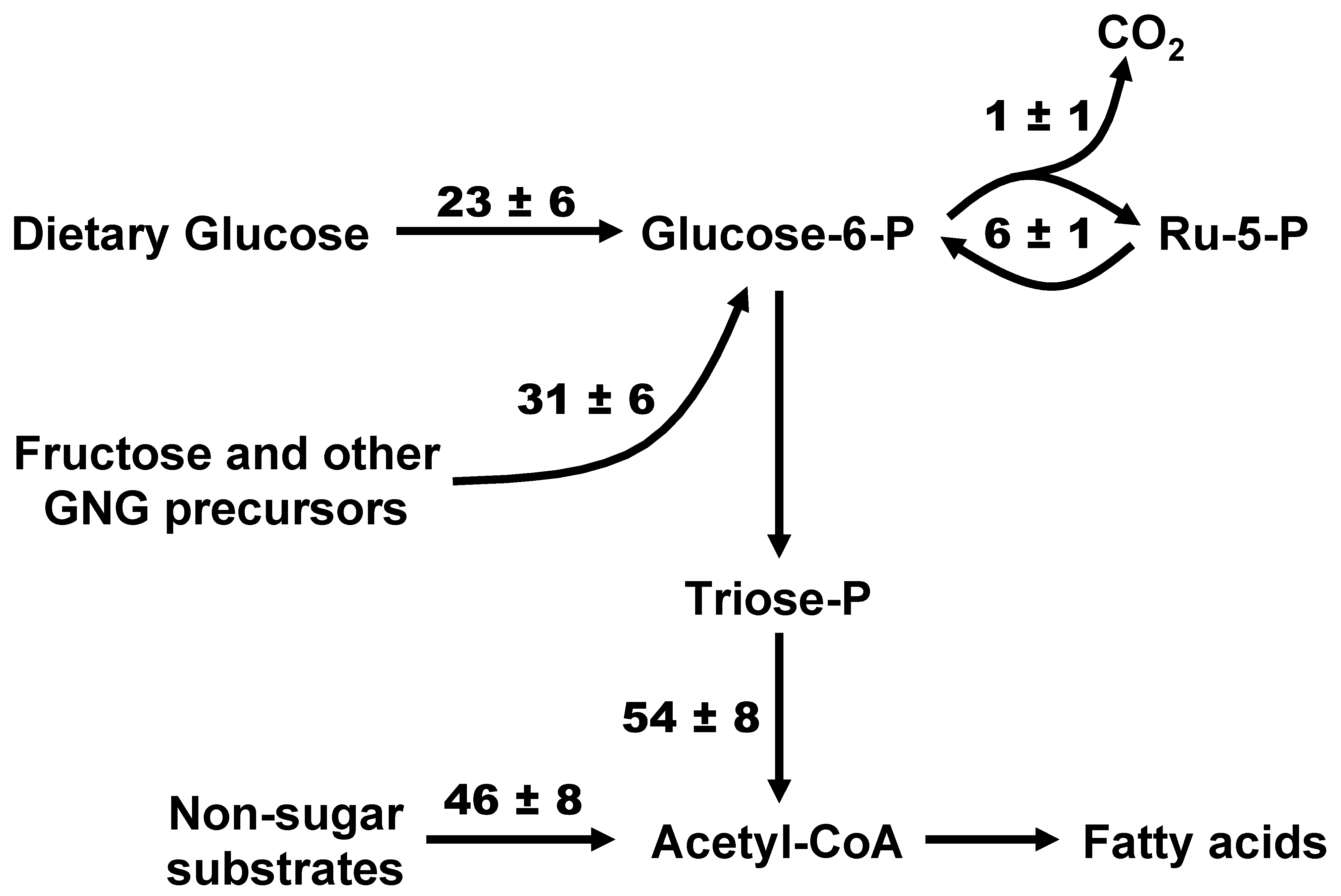
3.2. Sourcing of Lipogenic Acetyl-CoA Carbons Reported by [U-13C]Glucose and [U-13C]Fructose and PPP Activity
4. Discussion
4.1. General Overview
4.2. Hepatic Versus Extrahepatic Fructose Metabolism
4.3. PPP Flux in Relation to De Novo Lipogenesis
4.4. Limitations of the Approach
4.5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sclafani, A. Gut-brain nutrient signaling. Appetition vs. satiation. Appetite 2013, 71, 454–458. [Google Scholar] [CrossRef] [PubMed]
- Xiao, W.S.; Wang, R.S.; Handy, D.E.; Loscalzo, J. NAD(H) and NADP(H) Redox Couples and Cellular Energy Metabolism. Antioxid. Redox Signal. 2018, 28, 251–272. [Google Scholar] [CrossRef]
- DiNunzio, G.; Belew, G.D.; Torres, A.N.; Silva, J.G.; Silva, L.P.; Barosa, C.; Tavares, L.; Jones, J.G. Determining the contribution of a high-fructose corn syrup formulation to hepatic glycogen synthesis during ad-libitum feeding in mice. Sci. Rep. 2020, 10, 12852. [Google Scholar] [CrossRef] [PubMed]
- Belew, G.D.; Di Nunzio, G.; Tavares, L.; Silva, J.G.; Torres, A.N.; Jones, J.G. Estimating pentose phosphate pathway activity from the analysis of hepatic glycogen C-13-isotopomers derived from U-C-13 fructose and U-C-13 glucose. Magn. Reson. Med. 2020, 84, 2765–2771. [Google Scholar] [CrossRef] [PubMed]
- Silva, J.C.P.; Marques, C.; Martins, F.O.; Viegas, I.; Tavares, L.; Macedo, M.P.; Jones, J.G. Determining contributions of exogenous glucose and fructose to de novo fatty acid and glycerol synthesis in liver and adipose tissue. Metab. Eng. 2019, 56, 69–76. [Google Scholar] [CrossRef] [PubMed]
- Jensen, T.; Abdelmalek, M.F.; Sullivan, S.; Nadeau, K.J.; Green, M.; Roncal, C.; Nakagawa, T.; Kuwabara, M.; Sato, Y.; Kang, D.-H.; et al. Fructose and sugar: A major mediator of non-alcoholic fatty liver disease. J. Hepatol. 2018, 68, 1063–1075. [Google Scholar] [CrossRef] [PubMed]
- Chiu, S.; Mulligan, K.; Schwarz, J.-M. Dietary carbohydrates and fatty liver disease: De novo lipogenesis. Curr. Opin. Clin. Nutr. Metab. Care 2018, 21, 277–282. [Google Scholar] [CrossRef]
- Hodson, L.; Gunn, P.J. The regulation of hepatic fatty acid synthesis and partitioning: The effect of nutritional state. Nat. Rev. Endocrinol. 2019, 15, 689–700. [Google Scholar] [CrossRef] [PubMed]
- Jarak, I.; Barosa, C.; Martins, F.O.; Silva, J.C.P.; Santos, C.; Belew, G.D.; Rito, J.; Viegas, I.; Teixeira, J.; Oliveira, P.J.; et al. Sources of hepatic glycogen synthesis in mice fed with glucose or fructose as the sole dietary carbohydrate. Magn. Reson. Med. 2019, 81, 639–644. [Google Scholar] [CrossRef] [PubMed]
- Soares, A.F.; Carvalho, R.A.; Veiga, F.J.; Alves, M.G.; Martins, F.O.; Viegas, I.; Gonzalez, J.D.; Meton, I.; Baanante, I.V.; Jones, J.G. Restoration of direct pathway glycogen synthesis flux in the STZ-diabetes rat model by insulin administration. Am. J. Physiol.-Endocrinol. Metab. 2012, 303, E875–E885. [Google Scholar] [CrossRef]
- Perdigoto, R.; Rodrigues, T.B.; Furtado, A.L.; Porto, A.; Geraldes, C.; Jones, J.G. Integration of U-C-13 glucose and (H2O)-H-2 for quantification of hepatic glucose production and gluconeogenesis. Nmr Biomed. 2003, 16, 189–198. [Google Scholar] [CrossRef] [PubMed]
- Newgard, C.B.; Hirsch, L.J.; Foster, D.W.; McGarry, J.D. Studies on the Mechanism by Which Exogenous Glucose Is Converted into Liver-Glycogen in the Rat—A Direct or an Indirect Pathway. J. Biol. Chem. 1983, 258, 8046–8052. [Google Scholar] [CrossRef]
- Belew, G.D.; Silva, J.; Rito, J.; Tavares, L.; Viegas, I.; Teixeira, J.; Oliveira, P.J.; Macedo, M.P.; Jones, J.G. Transfer of glucose hydrogens via acetyl-CoA, malonyl-CoA, and NADPH to fatty acids during de novo lipogenesis S. J. Lipid Res. 2019, 60, 2050–2056. [Google Scholar] [CrossRef] [PubMed]
- Jones, J.G.; Barosa, C.; Gomes, F.; Mendes, A.C.; Delgado, T.C.; Diogo, L.; Garcia, P.; Bastos, M.; Barros, L.; Fagulha, A.; et al. NMR derivatives for quantification of H-2 and C-13-enrichment of human glucuronide from metabolic tracers. J. Carbohydr. Chem. 2006, 25, 203–217. [Google Scholar] [CrossRef]
- Jones, J.G.; Merritt, M.; Malloy, C. Quantifying tracer levels of 2H2O enrichment from microliter amounts of plasma and urine by 2H NMR. Magn. Res. Med. 2001, 45, 156–158. [Google Scholar] [CrossRef]
- Jin, E.S.; Sherry, A.D.; Malloy, C.R. Interaction between the Pentose Phosphate Pathway and Gluconeogenesis from Glycerol in the Liver. J. Biol. Chem. 2014, 289, 32593–32603. [Google Scholar] [CrossRef] [PubMed]
- Jang, C.; Hui, S.; Lu, W.; Cowan, A.J.; Morscher, R.J.; Lee, G.; Liu, W.; Tesz, G.J.; Birnbaum, M.J.; Rabinowitz, J.D. The Small Intestine Converts Dietary Fructose into Glucose and Organic Acids. Cell Metab. 2018, 27, 351–361. [Google Scholar] [CrossRef] [PubMed]
- Soares, A.F.; Carvalho, R.A.; Veiga, F.J.; Jones, J.G. Effects of galactose on direct and indirect pathway estimates of hepatic glycogen synthesis. Metab. Eng. 2010, 12, 552–560. [Google Scholar] [CrossRef] [PubMed]
- Rother, K.I.; Schwenk, W.F. Glucose-Production in Glycogen-Storage-Disease-I Is Not Associated with Increased Cycling through Hepatic Glycogen. Am. J. Physiol.-Endocrinol. Metab. 1995, 32, E774–E778. [Google Scholar] [CrossRef] [PubMed]
- Jin, E.S.; Sherry, A.D.; Malloy, C.R. An Oral Load of C-13(3) Glycerol and Blood NMR Analysis Detect Fatty Acid Esterification, Pentose Phosphate Pathway, and Glycerol Metabolism through the Tricarboxylic Acid Cycle in Human Liver. J. Biol. Chem. 2016, 291, 19031–19041. [Google Scholar] [CrossRef]
- Jones, J.G.; Garcia, P.; Barosa, C.; Delgado, T.C.; Diogo, L. Hepatic anaplerotic outflow fluxes are redirected from gluconeogenesis to lactate synthesis in patients with Type 1a glycogen storage disease. Metab. Eng. 2009, 11, 155–162. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.F.; Grosfeld, A.; Williams, E.; Vasiliauskas, D.; Barretto, S.; Smith, L.; Mariadassou, M.; Philippe, C.; Devime, F.; Melchior, C.; et al. Fructose malabsorption induces cholecystokinin expression in the ileum and cecum by changing microbiota composition and metabolism. Faseb J. 2019, 33, 7126–7142. [Google Scholar] [CrossRef] [PubMed]
- Zhao, S.; Jang, C.; Liu, J.; Uehara, K.; Gilbert, M.; Izzo, L.; Zeng, X.; Trefely, S.; Fernandez, S.; Carrer, A.; et al. Dietary fructose feeds hepatic lipogenesis via microbiota-derived acetate. Nature 2020, 579, 586–591. [Google Scholar] [CrossRef] [PubMed]
- Bachmanov, A.A.; Reed, D.R.; Beauchamp, G.D.; Tordoff, M.G. Food intake, water intake, and drinking spout side preference of 28 mouse strains. Behav. Genet. 2002, 32, 435–443. [Google Scholar] [CrossRef] [PubMed]
- Yoo, H.; Stephanopoulos, G.; Kelleher, J.K. Quantifying carbon sources for de novo lipogenesis in wild-type and IRS-1 knockout brown adipocytes. J. Lipid Res. 2004, 45, 1324–1332. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Shah, S.; Fan, J.; Park, J.O.; Wellen, K.E.; Rabinowitz, J.D. Malic enzyme tracers reveal hypoxia-induced switch in adipocyte NADPH pathway usage. Nat. Chem. Biol. 2016, 12, 345–352. [Google Scholar] [CrossRef]
- Minehira, K.; Bettschart, V.; Vidal, H.; Vega, N.; Di Vetta, V.; Rey, V.; Schneiter, P.; Tappy, L. Effect of carbohydrate overfeeding on whole body and adipose tissue metabolism in humans. Obes. Res. 2003, 11, 1096–1103. [Google Scholar] [CrossRef]
- Zhang, Z.; TeSlaa, T.; Xu, X.; Zeng, X.; Yang, L.; Xing, G.; Tesz, G.J.; Clasquin, M.F.; Rabinowitz, J.D. Serine catabolism generates liver NADPH and supports hepatic lipogenesis. Nat. Metab. 2021, 3, 1608–1620. [Google Scholar] [CrossRef]
- Wu, W.C.; Zhao, W.; Li, S. Small intestinal bacteria overgrowth decreases small intestinal motility in the NASH rats. World J. Gastroenterol. 2008, 14, 313–317. [Google Scholar] [CrossRef]
- Batt, R.M.; Hall, E.J.; McLean, L.; Simpson, K.W. Small intestinal bacterial overgrowth and enhanced intestinal permeability in healthy beagles. Am. J. Vet. Res. 1992, 53, 1935–1940. [Google Scholar]
- O’Brien, P.; Han, G.; Ganpathy, P.; Pitre, S.; Zhang, Y.; Ryan, J.; Sim, P.Y.; Harding, S.V.; Gray, R.; Preedy, V.R.; et al. Chronic Effects of a High Sucrose Diet on Murine Gastrointestinal Nutrient Sensor Gene and Protein Expression Levels and Lipid Metabolism. Int. J. Mol. Sci. 2021, 22, 137. [Google Scholar] [CrossRef] [PubMed]
| 13CIUA (Equation) | |||||
|---|---|---|---|---|---|
| Experiment | Hepatic Glucose-6-P (Equation (1)) | Dietary Glucose (Equation (2)) | GNG precursors (Equation (3a,b)) | Triose-P (Equation (4)) | Acetyl-CoA (Equation (5)) |
| [U-13C]Glucose and unlabeled fructose (n = 4) | 3.78 ± 0.74 | 5.47 ± 1.25 | 1.30 ± 0.15 | 2.50 ± 0.66 | 1.53 ± 0.43 |
| Unlabeled glucose and [U-13C]fructose (n = 5) | 5.06 ± 0.34 | N.D. | 14.99 ± 1.24 | 8.99 ± 1.05 | 4.32 ± 0.50 |
| Pathway Component | [U-13C]Glucose | [U-13C]Fructose | p Value | |
|---|---|---|---|---|
| Acetyl-CoA → Fatty acids | 100 | 100 | N.D. | |
| Non-Triose-P → Acetyl-CoA | Equation (7) | 40 ± 4 | 51 ± 8 | 0.08 |
| Triose-P → Acetyl-CoA | Equation (6) | 60 ± 4 | 49 ± 8 | 0.08 |
| Dietary glucose → Triose-P | Equations (8) and (11) | 28 ± 4 | 21 ± 9 | 0.32 |
| [U-13C]glucose → Dietary glucose | Equation (12) | 8 ± 4 | N.D. | N.D. |
| Other dietary glucose sources → Dietary glucose | Equation (13) | 19 ± 4 | N.D. | N.D. |
| GNG precursors → Triose-P | Equations (9) and (10) | 34 ± 8 | 29 ± 4 | 0.38 |
| [U-13C]fructose → GNG | Equation (14) | N.D. | 22 ± 6 | N.D. |
| Other precursors → GNG | Equation (15) | N.D. | 7 ± 4 | N.D. |
| Glucose-6-P → PPP → Glucose-6-P | 7 ± 1 | 5 ± 1 | 0.13 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Viegas, I.; Di Nunzio, G.; Belew, G.D.; Torres, A.N.; Silva, J.G.; Perpétuo, L.; Barosa, C.; Tavares, L.C.; Jones, J.G. Integration of Liver Glycogen and Triglyceride NMR Isotopomer Analyses Provides a Comprehensive Coverage of Hepatic Glucose and Fructose Metabolism. Metabolites 2022, 12, 1142. https://doi.org/10.3390/metabo12111142
Viegas I, Di Nunzio G, Belew GD, Torres AN, Silva JG, Perpétuo L, Barosa C, Tavares LC, Jones JG. Integration of Liver Glycogen and Triglyceride NMR Isotopomer Analyses Provides a Comprehensive Coverage of Hepatic Glucose and Fructose Metabolism. Metabolites. 2022; 12(11):1142. https://doi.org/10.3390/metabo12111142
Chicago/Turabian StyleViegas, Ivan, Giada Di Nunzio, Getachew D. Belew, Alejandra N. Torres, João G. Silva, Luis Perpétuo, Cristina Barosa, Ludgero C. Tavares, and John G. Jones. 2022. "Integration of Liver Glycogen and Triglyceride NMR Isotopomer Analyses Provides a Comprehensive Coverage of Hepatic Glucose and Fructose Metabolism" Metabolites 12, no. 11: 1142. https://doi.org/10.3390/metabo12111142
APA StyleViegas, I., Di Nunzio, G., Belew, G. D., Torres, A. N., Silva, J. G., Perpétuo, L., Barosa, C., Tavares, L. C., & Jones, J. G. (2022). Integration of Liver Glycogen and Triglyceride NMR Isotopomer Analyses Provides a Comprehensive Coverage of Hepatic Glucose and Fructose Metabolism. Metabolites, 12(11), 1142. https://doi.org/10.3390/metabo12111142










