Role of microRNA in Endocrine Disruptor-Induced Immunomodulation of Metabolic Health
Abstract
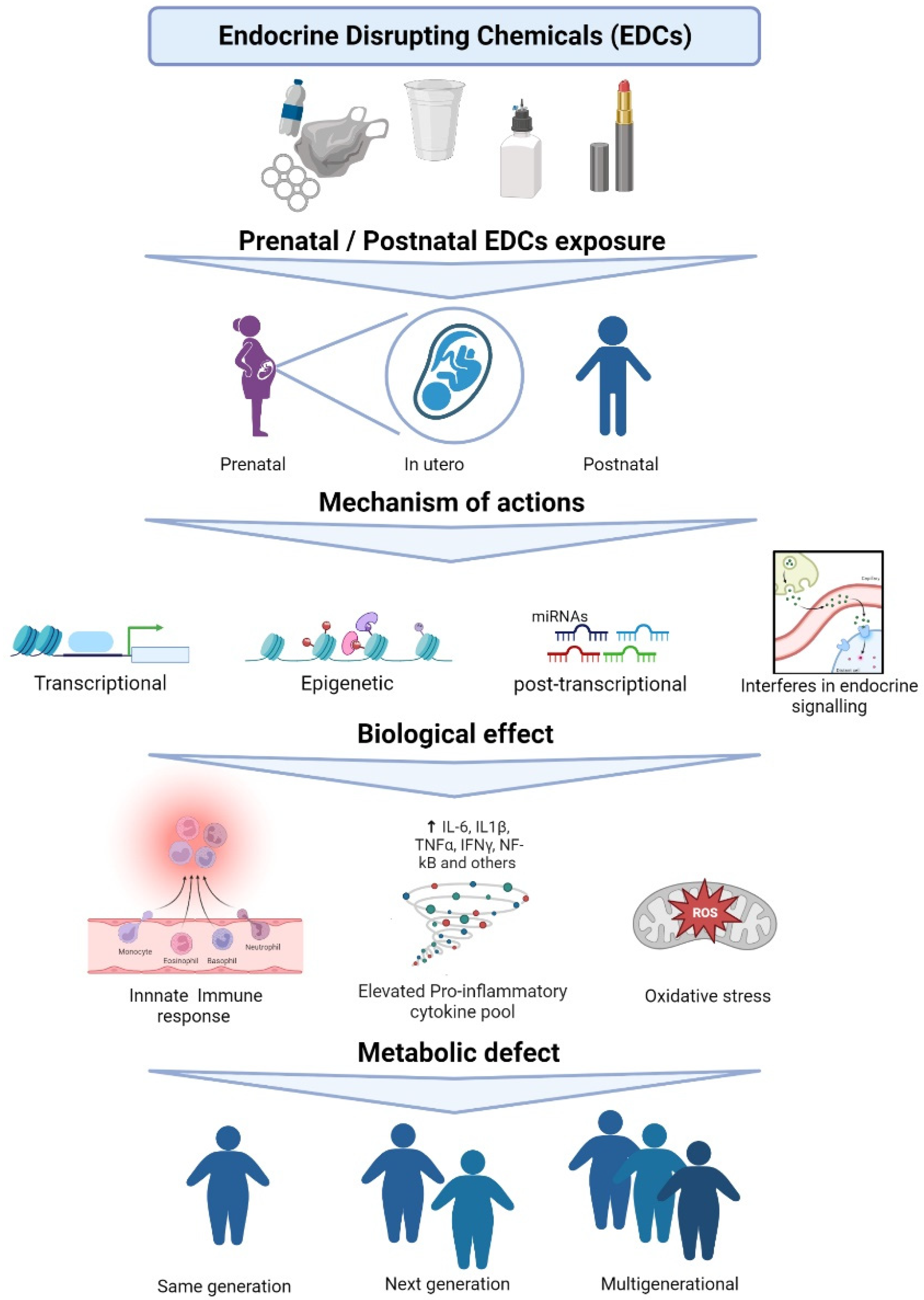
1. Introduction
2. EDCs Exposure and the Immune System
3. EDCs, MiR, and Innate Immunity
3.1. Impact of EDC Exposure on MiR Regulation
3.2. MiR in Innate Immunity
4. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- Paczkowska-Abdulsalam, M.; Kretowski, A. Obesity, Metabolic Health and Omics: Current Status and Future Directions. World J. Diabetes 2021, 12, 420–436. [Google Scholar] [CrossRef]
- Blaszczak, A.M.; Jalilvand, A.; Hsueh, W.A. Adipocytes, Innate Immunity and Obesity: A Mini-Review. Front. Immunol. 2021, 12, 650768. [Google Scholar] [CrossRef]
- The Lancet Diabetes Endocrinol. Metabolic Health: A Priority for the Post-Pandemic Era. Lancet Diabetes Endocrinol. 2021, 9, 189. [Google Scholar] [CrossRef]
- Liang, S.; Ji, L.; Kang, L.; Hu, X. Metabolic Regulation of Innate Immunity. Adv. Immunol. 2020, 145, 129–157. [Google Scholar] [PubMed]
- Paragh, G.; Seres, I.; Harangi, M.; Fülöp, P. Dynamic Interplay between Metabolic Syndrome and Immunity. Adv. Exp. Med. Biol. 2014, 824, 171–190. [Google Scholar] [PubMed]
- Zamora, A.N.; Jansen, E.C.; Tamayo-Ortiz, M.; Goodrich, J.M.; Sánchez, B.N.; Watkins, D.J.; Tamayo-Orozco, J.A.; Téllez-Rojo, M.M.; Mercado-García, A.; Baylin, A.; et al. Exposure to Phenols, Phthalates, and Parabens and Development of Metabolic Syndrome among Mexican Women in Midlife. Front. Public Health 2021, 9, 620769. [Google Scholar] [CrossRef] [PubMed]
- Heindel, J.J.; Blumberg, B.; Cave, M.; Machtinger, R.; Mantovani, A.; Mendez, M.A.; Nadal, A.; Palanza, P.; Panzica, G.; Sargis, R.; et al. Metabolism Disrupting Chemicals and Metabolic Disorders. Rep. Toxicol. 2017, 68, 3–33. [Google Scholar] [CrossRef]
- Nowak, K.; Jabłońska, E.; Ratajczak-Wrona, W. Immunomodulatory Effects of Synthetic Endocrine Disrupting Chemicals on the Development and Functions of Human Immune Cells. Environ. Int. 2019, 125, 350–364. [Google Scholar] [CrossRef]
- Diamanti-Kandarakis, E.; Bourguignon, J.-P.; Giudice, L.C.; Hauser, R.; Prins, G.S.; Soto, A.M.; Zoeller, R.T.; Gore, A.C. Endocrine-Disrupting Chemicals: An Endocrine Society Scientific Statement. Endocr. Rev. 2009, 30, 293–342. [Google Scholar] [CrossRef]
- Kumar, M.; Sarma, D.K.; Shubham, S.; Kumawat, M.; Verma, V.; Prakash, A.; Tiwari, R. Environmental Endocrine-Disrupting Chemical Exposure: Role in Non-Communicable Diseases. Front. Public Health 2020, 8, 553850. [Google Scholar] [CrossRef] [PubMed]
- Bowman, J.D.; Choudhury, M. Phthalates in Neonatal Health: Friend or Foe? J. Dev. Orig. Health Dis. 2016, 7, 652–664. [Google Scholar] [CrossRef] [PubMed]
- Schjenken, J.E.; Green, E.S.; Overduin, T.S.; Mah, C.Y.; Russell, D.L.; Robertson, S.A. Endocrine Disruptor Compounds-a Cause of Impaired Immune Tolerance Driving Inflammatory Disorders of Pregnancy? Front. Endocrinol. 2021, 12, 607539. [Google Scholar] [CrossRef]
- La Merrill, M.A.; Vandenberg, L.N.; Smith, M.T.; Goodson, W.; Browne, P.; Patisaul, H.B.; Guyton, K.Z.; Kortenkamp, A.; Cogliano, V.J.; Woodruff, T.J.; et al. Consensus on the Key Characteristics of Endocrine-Disrupting Chemicals as a Basis for Hazard Identification. Nat. Rev. Endocrinol. 2020, 16, 45–57. [Google Scholar] [CrossRef]
- Kim, J. Regulation of Immune Cell Functions by Metabolic Reprogramming. J. Immunol. Res. 2018, 2018, 8605471. [Google Scholar] [CrossRef]
- Exley, M.A.; Hand, L.; O’shea, D.; Lynch, L. Interplay between the Immune System and Adipose Tissue in Obesity. J. Endocrinol. 2014, 223, R41–R48. [Google Scholar] [CrossRef] [PubMed]
- Huang, N.; Perl, A. Metabolism as a Target for Modulation in Autoimmune Diseases. Trends Immunol. 2018, 39, 562–576. [Google Scholar] [CrossRef]
- Luebke, R.W.; Chen, D.H.; Dietert, R.; Yang, Y.; King, M.; Luster, M.I. The Comparative Immunotoxicity of Five Selected Compounds Following Developmental or Adult Exposure. J. Toxicol. Environ. Health B Crit. Rev. 2006, 9, 1–26. [Google Scholar] [CrossRef]
- Lavastre, V.; Girard, D. Tributyltin Induces Human Neutrophil Apoptosis and Selective Degradation of Cytoskeletal Proteins by Caspases. J. Toxicol. Environ. Health A 2002, 65, 1013–1024. [Google Scholar] [CrossRef] [PubMed]
- Gostner, J.M.; Raggl, E.; Becker, K.; Überall, F.; Schennach, H.; Pease, J.E.; Fuchs, D. Bisphenol a Suppresses Th1-Type Immune Response in Human Peripheral Blood Mononuclear Cells in Vitro. Immunol. Lett. 2015, 168, 285–292. [Google Scholar] [CrossRef]
- Brown, S.; Whalen, M. Tributyltin Alters Secretion of Interleukin 1 Beta from Human Immune Cells. J. Appl. Toxicol. 2015, 35, 895–908. [Google Scholar] [CrossRef]
- Yan, S.; Chen, Y.; Dong, M.; Song, W.; Belcher, S.M.; Wang, H.S. Bisphenol a and 17β-Estradiol Promote Arrhythmia in the Female Heart Via Alteration of Calcium Handling. PLoS ONE 2011, 6, e25455. [Google Scholar] [CrossRef] [PubMed]
- Soriano, S.; Alonso-Magdalena, P.; García-Arévalo, M.; Novials, A.; Muhammed, S.J.; Salehi, A.; Gustafsson, J.-A.; Quesada, I.; Nadal, A. Rapid Insulinotropic Action of Low Doses of Bisphenol-a on Mouse and Human Islets of Langerhans: Role of Estrogen Receptor B. PLoS ONE 2012, 7, e31109. [Google Scholar] [CrossRef] [PubMed]
- O’shaughnessy, K.L.; Fischer, F.; Zenclussen, A.C. Perinatal Exposure to Endocrine Disrupting Chemicals and Neurodevelopment: How Articles of Daily Use Influence the Development of Our Children. Best Pr. Res. Clin. Endocrinol. Metab. 2021, 35, 101568. [Google Scholar] [CrossRef] [PubMed]
- Barker, D.J. The Fetal and Infant Origins of Adult Disease. BMJ 1990, 301, 1111. [Google Scholar] [CrossRef]
- Dover, G.J. The Barker Hypothesis: How Pediatricans Will Diagnose and Prevent Common Adult-Onset Diseases. Trans. Am. Clin. Climatol. Assoc. 2009, 120, 199–207. [Google Scholar]
- Kang, S.C.; Lee, B.M. DNA Methylation of Estrogen Receptor Alpha Gene by Phthalates. J. Toxicol. Environ. Health A 2005, 68, 1995–2003. [Google Scholar] [CrossRef] [PubMed]
- Meruvu, S.; Zhang, J.; Bedi, Y.S.; Choudhury, M. Mono-(2-Ethylhexyl) Phthalate Induces Apoptosis through Mir-16 in Human First Trimester Placental Cell Line Htr-8/Svneo. Toxicol. Vitr. Int. J. Publ. Assoc. BIBRA 2016, 31, 35–42. [Google Scholar] [CrossRef] [PubMed]
- Meruvu, S.; Zhang, J.; Choudhury, M. Mono-(2-Ethylhexyl) Phthalate Increases Oxidative Stress Responsive Mirnas in First Trimester Placental Cell Line Htr8/Svneo. Chem. Res. Toxicol. 2016, 29, 430–435. [Google Scholar] [CrossRef] [PubMed]
- Park, M.H.; Jeong, E.; Choudhury, M. Mono-(2-Ethylhexyl)Phthalate Regulates Cholesterol Efflux Via Micrornas Regulated M(6)a Rna Methylation. Chem. Res. Toxicol. 2020, 33, 461–469. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Ali, H.I.; Bedi, Y.S.; Choudhury, M. The Plasticizer Bbp Selectively Inhibits Epigenetic Regulator Sirtuins. Toxicology 2015, 338, 130–141. [Google Scholar] [CrossRef]
- Zhang, J.; Choudhury, M. The Plasticizer Bbp Selectively Inhibits Epigenetic Regulator Sirtuin During Differentiation of C3h10t1/2 Stem Cell Line. Toxicol. Vitr. Int. J. Publ. Assoc. BIBRA 2017, 39, 75–83. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Choudhury, M. Benzyl Butyl Phthalate Induced Early Lncrna H19 Regulation in C3h10t1/2 Stem Cell Line. Chem. Res. Toxicol. 2021, 34, 54–62. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Powell, C.; Meruvu, S.; Sonkar, R.; Choudhury, M. Pyrroloquinoline Quinone Attenuated Benzyl Butyl Phthalate Induced Metabolic Aberration and a Hepatic Metabolomic Analysis. Biochem. Pharm. 2021, 197, 114883. [Google Scholar] [CrossRef]
- Howard, S.G. Developmental Exposure to Endocrine Disrupting Chemicals and Type 1 Diabetes Mellitus. Front. Endocrinol. 2018, 9, 513. [Google Scholar] [CrossRef]
- Iwasaki, A.; Medzhitov, R. Control of Adaptive Immunity by the Innate Immune System. Nat. Immunol. 2015, 16, 343–353. [Google Scholar] [CrossRef]
- Bennasroune, A.; Rojas, L.; Foucaud, L.; Goulaouic, S.; Laval-Gilly, P.; Fickova, M.; Couleau, N.; Durandet, C.; Henry, S.; Falla, J. Effects of 4-Nonylphenol and/or Diisononylphthalate on Thp-1 Cells: Impact of Endocrine Disruptors on Human Immune System Parameters. Int. J. Immunopathol. Pharm. 2012, 25, 365–376. [Google Scholar] [CrossRef]
- Lee, J.W.; Han, H.K.; Park, S.; Moon, E.Y. Nonylphenol Increases Tumor Formation and Growth by Suppressing Gender-Independent Lymphocyte Proliferation and Macrophage Activation. Environ. Toxicol. 2017, 32, 1679–1687. [Google Scholar] [CrossRef]
- Couleau, N.; Falla, J.; Beillerot, A.; Battaglia, E.; Innocenzo, M.D.; Plancon, S.; Laval-Gilly, P.; Bennasroune, A. Effects of Endocrine Disruptor Compounds, Alone or in Combination, on Human Macrophage-Like Thp-1 Cell Response. PLoS ONE 2015, 10, e0131428. [Google Scholar] [CrossRef]
- Hansen, J.F.; Bendtzen, K.; Boas, M.; Frederiksen, H.; Nielsen, C.H.; Rasmussen, K.; Feldt-Rasmussen, U. Influence of Phthalates on Cytokine Production in Monocytes and Macrophages: A Systematic Review of Experimental Trials. PLoS ONE 2015, 10, e0120083. [Google Scholar] [CrossRef]
- Kuan, Y.H.; Li, Y.C.; Huang, F.M.; Chang, Y.C. The Upregulation of Tumour Necrosis Factor-A and Surface Antigens Expression on Macrophages by Bisphenol a-Glycidyl-Methacrylate. Int. Endod. J. 2012, 45, 619–626. [Google Scholar] [CrossRef]
- Liu, Z.; Lu, Y.; Zhong, K.; Wang, C.; Xu, X. The Associations between Endocrine Disrupting Chemicals and Markers of Inflammation and Immune Responses: A Systematic Review and Meta-Analysis. Ecotoxicol. Environ. Saf. 2022, 234, 113382. [Google Scholar] [CrossRef] [PubMed]
- Ochiai, S.; Roediger, B.; Abtin, A.; Shklovskaya, E.; De St Groth, B.F.; Yamane, H.; Weninger, W.; Le Gros, G.; Ronchese, F. Cd326(Lo)Cd103(Lo)Cd11b(Lo) Dermal Dendritic Cells Are Activated by Thymic Stromal Lymphopoietin During Contact Sensitization in Mice. J. Immunol. 2014, 193, 2504–2511. [Google Scholar] [CrossRef] [PubMed]
- Makki, K.; Froguel, P.; Wolowczuk, I. Adipose Tissue in Obesity-Related Inflammation and Insulin Resistance: Cells, Cytokines, and Chemokines. ISRN Inflamm. 2013, 2013, 139239. [Google Scholar] [CrossRef] [PubMed]
- Bornehag, C.G.; Nanberg, E. Phthalate Exposure and Asthma in Children. Int. J. Androl. 2010, 33, 333–345. [Google Scholar] [CrossRef] [PubMed]
- Weatherly, L.M.; Nelson, A.J.; Shim, J.; Riitano, A.M.; Gerson, E.D.; Hart, A.J.; de Juan-Sanz, J.; Ryan, T.A.; Sher, R.; Hess, S.T.; et al. Antimicrobial Agent Triclosan Disrupts Mitochondrial Structure, Revealed by Super-Resolution Microscopy, and Inhibits Mast Cell Signaling Via Calcium Modulation. Toxicol. Appl. Pharmacol. 2018, 349, 39–54. [Google Scholar] [CrossRef] [PubMed]
- O’brien, E.; Dolinoy, D.C.; Mancuso, P. Bisphenol a at Concentrations Relevant to Human Exposure Enhances Histamine and Cysteinyl Leukotriene Release from Bone Marrow-Derived Mast Cells. J. Immunotoxicol. 2014, 11, 84–89. [Google Scholar] [CrossRef]
- Vetrano, A.M.; Laskin, D.L.; Archer, F.; Syed, K.; Gray, J.P.; Laskin, J.D.; Nwebube, N.; Weinberger, B. Inflammatory Effects of Phthalates in Neonatal Neutrophils. Pediatr. Res. 2010, 68, 134–139. [Google Scholar] [CrossRef]
- Balistrieri, A.; Hobohm, L.; Srivastava, T.; Meier, A.; Corriden, R. Alterations in Human Neutrophil Function Caused by Bisphenol, A. Am. J. Physiol. Cell. Physiol. 2018, 315, C636–C642. [Google Scholar] [CrossRef]
- Zhang, Y.; Mi, K.; Xue, W.; Wei, W.; Yang, H. Acute Bpa Exposure-Induced Oxidative Stress, Depressed Immune Genes Expression and Damage of Hepatopancreas in Red Swamp Crayfish Procambarus Clarkii. Fish. Shellfish. Immunol. 2020, 103, 95–102. [Google Scholar] [CrossRef]
- Thomas, L.D.; Shah, H.; Green, S.A.; Bankhurst, A.D.; Whalen, M.M. Tributyltin Exposure Causes Decreased Granzyme B and Perforin Levels in Human Natural Killer Cells. Toxicology 2004, 200, 221–233. [Google Scholar] [CrossRef]
- Brown, S.; Wilburn, W.; Martin, T.; Whalen, M. Butyltin Compounds Alter Secretion of Interleukin 6 from Human Immune Cells. J. Appl. Toxicol. 2018, 38, 201–218. [Google Scholar] [CrossRef] [PubMed]
- Rolfo, A.; Nuzzo, A.M.; De Amicis, R.; Moretti, L.; Bertoli, S.; Leone, A. Fetal-Maternal Exposure to Endocrine Disruptors: Correlation with Diet Intake and Pregnancy Outcomes. Nutrients 2020, 12, 1744. [Google Scholar] [CrossRef]
- Varshavsky, J.; Smith, A.; Wang, A.; Hom, E.; Izano, M.; Huang, H.; Padula, A.; Woodruff, T.J. Heightened Susceptibility: A Review of How Pregnancy and Chemical Exposures Influence Maternal Health. Rep. Toxicol. 2020, 92, 14–56. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Park, H.; Lee, J.; Cho, G.; Choi, S.; Choi, G.; Kim, S.Y.; Eun, S.-H.; Suh, E.; Kim, S.K.; et al. Association of Diethylhexyl Phthalate with Obesity-Related Markers and Body Mass Change from Birth to 3 Months of Age. J. Epidemiol. Community. Health 2016, 70, 466–472. [Google Scholar] [CrossRef]
- Buckley, J.P.; Engel, S.M.; Braun, J.M.; Whyatt, R.M.; Daniels, J.L.; Mendez, M.A.; Richardson, D.B.; Xu, Y.; Calafat, A.M.; Wolff, M.S.; et al. Prenatal Phthalate Exposures and Body Mass Index among 4- to 7-Year-Old Children: A Pooled Analysis. Epidemiology 2016, 27, 449–458. [Google Scholar] [CrossRef]
- Liu, J.; Liao, M.; Huang, R.; You, Y.; Lin, X.; Yang, H.; Fan, L.; Zhong, Y.; Li, X.; Li, J.; et al. Perinatal Combinational Exposure to Bisphenol a and a High-Fat Diet Contributes to Transgenerational Dysregulation of Cardiovascular and Metabolic Systems in Mice. Front Cell. Dev. Biol. 2022, 10, 834346. [Google Scholar] [CrossRef] [PubMed]
- Bansal, A.; Henao-Mejia, J.; Simmons, R.A. Immune System: An Emerging Player in Mediating Effects of Endocrine Disruptors on Metabolic Health. Endocrinology 2018, 159, 32–45. [Google Scholar] [CrossRef]
- Ni, J.; Zhang, Z.; Luo, X.; Xiao, L.; Wang, N. Plasticizer Dbp Activates Nlrp3 Inflammasome through the P2x7 Receptor in Hepg2 and L02 Cells. J. Biochem. Mol. Toxicol. 2016, 30, 178–185. [Google Scholar] [CrossRef] [PubMed]
- Kadowaki, T.; Yamauchi, T.; Kubota, N.; Hara, K.; Ueki, K.; Tobe, K. Adiponectin and Adiponectin Receptors in Insulin Resistance, Diabetes, and the Metabolic Syndrome. J. Clin. Invest. 2006, 116, 1784–1792. [Google Scholar] [CrossRef]
- Valentino, R.; D’Esposito, V.; Passaretti, F.; Liotti, A.; Cabaro, S.; Longo, M.; Perruolo, G.; Oriente, F.; Beguinot, F.; Formisano, P. Bisphenol-a Impairs Insulin Action and up-Regulates Inflammatory Pathways in Human Subcutaneous Adipocytes and 3t3-L1 Cells. PLoS ONE 2013, 8, e82099. [Google Scholar] [CrossRef]
- Bateman, M.E.; Strong, A.L.; Mclachlan, J.A.; Burow, M.E.; Bunnell, B.A. The Effects of Endocrine Disruptors on Adipogenesis and Osteogenesis in Mesenchymal Stem Cells: A Review. Front. Endocrinol. 2016, 7, 171. [Google Scholar] [CrossRef]
- Pourgholaminejad, A.; Aghdami, N.; Baharvand, H.; Moazzeni, S.M. The Effect of Pro-Inflammatory Cytokines on Immunophenotype, Differentiation Capacity and Immunomodulatory Functions of Human Mesenchymal Stem Cells. Cytokine 2016, 85, 51–60. [Google Scholar] [CrossRef] [PubMed]
- Szabó, E.; Fajka-Boja, R.; Kriston-Pál; Hornung, A.; Makra, I.; Kudlik, G.; Uher, F.; Katona, R.L.; Monostori, E.; Czibula, A. Licensing by Inflammatory Cytokines Abolishes Heterogeneity of Immunosuppressive Function of Mesenchymal Stem Cell Population. Stem. Cells. Dev. 2015, 24, 2171–2180. [Google Scholar] [CrossRef] [PubMed]
- Crop, M.J.; Baan, C.C.; Korevaar, S.S.; Ijzermans, J.N.M.; Pescatori, M.; Stubbs, A.P.; Van Ijcken, W.F.J.; Dahlke, M.H.; Eggenhofer, E.; Weimar, W.; et al. Inflammatory Conditions Affect Gene Expression and Function of Human Adipose Tissue-Derived Mesenchymal Stem Cells. Clin. Exp. Immunol. 2010, 162, 474–486. [Google Scholar] [CrossRef] [PubMed]
- Mercer, T.R.; Dinger, M.E.; Mattick, J.S. Long Non-Coding Rnas: Insights into Functions. Nat. Rev. Genet. 2009, 10, 155–159. [Google Scholar] [CrossRef] [PubMed]
- Ambros, V.; Bartel, B.; Bartel, D.P.; Burge, C.B.; Carrington, J.C.; Chen, X.; Dreyfuss, G.; Eddy, S.R.; Griffiths-Jones, S.; Marshall, M.; et al. A Uniform System for Microrna Annotation. Rna 2003, 9, 277–279. [Google Scholar] [CrossRef]
- Ling, H.; Fabbri, M.; Calin, G.A. Micrornas and Other Non-Coding Rnas as Targets for Anticancer Drug Development. Nat. Rev. Drug. Discov. 2013, 12, 847–865. [Google Scholar] [CrossRef]
- Huntzinger, E.; Izaurralde, E. Gene Silencing by Micrornas: Contributions of Translational Repression and Mrna Decay. Nat. Rev. Genet. 2011, 12, 99–110. [Google Scholar] [CrossRef]
- Gangaraju, V.K.; Lin, H. Micrornas: Key Regulators of Stem Cells. Nat. Rev. Mol. Cell. Biol. 2009, 10, 116–125. [Google Scholar] [CrossRef]
- Ye, J.; Zou, M.; Li, P.; Liu, H. Microrna Regulation of Energy Metabolism to Induce Chemoresistance in Cancers. Technol. Cancer Res. Treat. 2018, 17, 1533033818805997. [Google Scholar] [CrossRef]
- Vienberg, S.; Geiger, J.; Madsen, S.; Dalgaard, L.T. Micrornas in Metabolism. Acta. Physiol. 2017, 219, 346–361. [Google Scholar] [CrossRef] [PubMed]
- Rassoulzadegan, M.; Grandjean, V.; Gounon, P.; Cuzin, F. Epigenetic Heredity in Mice: Involvement of Rna and Mirnas. J. De La Soc. De Biol. 2007, 201, 397–399. [Google Scholar] [CrossRef] [PubMed]
- Hussain, N. Epigenetic Influences That Modulate Infant Growth, Development, and Disease. Antioxid Redox Signal 2012, 17, 224–236. [Google Scholar] [CrossRef]
- Gillet, V.; Hunting, D.J.; Takser, L. Turing Revisited: Decoding the Microrna Messages in Brain Extracellular Vesicles for Early Detection of Neurodevelopmental Disorders. Curr. Environ. Health Rep. 2016, 3, 188–201. [Google Scholar] [CrossRef] [PubMed]
- Price, T.M.; Murphy, S.K.; Younglai, E.V. Perspectives: The Possible Influence of Assisted Reproductive Technologies on Transgenerational Reproductive Effects of Environmental Endocrine Disruptors. Toxicol. Sci. 2007, 96, 218–226. [Google Scholar] [CrossRef]
- Gluckman, P.D.; Hanson, M.A.; Cooper, C.; Thornburg, K.L. Effect of in Utero and Early-Life Conditions on Adult Health and Disease. N. Engl. J. Med. 2008, 359, 61–73. [Google Scholar] [CrossRef]
- Thornburg, K.L.; Shannon, J.; Thuillier, P.; Turker, M.S. In Utero Life and Epigenetic Predisposition for Disease. Adv. Genet. 2010, 71, 57–78. [Google Scholar]
- Basak, S.; Das, M.K.; Duttaroy, A.K. Plastics Derived Endocrine-Disrupting Compounds and Their Effects on Early Development. Birth. Defects. Res. 2020, 112, 1308–1325. [Google Scholar] [CrossRef]
- Voglova, K.; Bezakova, J.; Herichova, I. Progress in Micro Rna Focused Research in Endocrinology. Endocr. Regul. 2016, 50, 83–105. [Google Scholar] [CrossRef]
- Petri, R.; Malmevik, J.; Fasching, L.; Åkerblom, M.; Jakobsson, J. Mirnas in Brain Development. Exp. Cell Res. 2014, 321, 84–89. [Google Scholar] [CrossRef]
- Rajasekhar, M.; Schmitz, U.; Flamant, S.; Wong, J.J.-L.; Bailey, C.G.; Ritchie, W.; Holst, J.; Rasko, J.E.J. Identifying Microrna Determinants of Human Myelopoiesis. Sci. Rep. 2018, 8, 7264. [Google Scholar] [CrossRef] [PubMed]
- Lontchi-Yimagou, E.; Sobngwi, E.; Matsha, T.E.; Kengne, A.P. Diabetes Mellitus and Inflammation. Curr. Diab. Rep. 2013, 13, 435–444. [Google Scholar] [PubMed]
- Kawai, T.; Autieri, M.V.; Scalia, R. Adipose Tissue Inflammation and Metabolic Dysfunction in Obesity. Am. J. Physiol. Cell. Physiol. 2021, 320, C375–C391. [Google Scholar] [CrossRef]
- Rahman, S.M.; Janssen, R.C.; Choudhury, M.; Baquero, K.C.; Aikens, R.M.; de la Houssaye, B.A.; Friedman, J.E. Ccaat/Enhancer-Binding Protein B (C/Ebpβ) Expression Regulates Dietary-Induced Inflammation in Macrophages and Adipose Tissue in Mice. J. Biol. Chem. 2012, 287, 34349–34360. [Google Scholar] [CrossRef] [PubMed]
- Grün, F.; Blumberg, B. Environmental Obesogens: Organotins and Endocrine Disruption Via Nuclear Receptor Signaling. Endocrinology 2006, 147 (Suppl. 6), S50–S55. [Google Scholar] [CrossRef]
- Rönn, M.; Lind, L.; Örberg, J.; Kullberg, J.; Söderberg, S.; Larsson, A.; Johansson, L.; Ahlström, H.; Lind, P.M. Bisphenol a Is Related to Circulating Levels of Adiponectin, Leptin and Ghrelin, but Not to Fat Mass or Fat Distribution in Humans. Chemosphere 2014, 112, 42–48. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Nichols, R.G.; Correll, J.; Murray, I.A.; Tanaka, N.; Smith, P.B.; Hubbard, T.D.; Sebastian, A.; Albert, I.; Hatzakis, E.; et al. Persistent Organic Pollutants Modify Gut Microbiota-Host Metabolic Homeostasis in Mice through Aryl Hydrocarbon Receptor Activation. Environ. Health Perspect. 2015, 123, 679–688. [Google Scholar] [CrossRef] [PubMed]
- Vidal, O.S.; Deepika, D.; Schuhmacher, M.; Kumar, V. Edc-Induced Mechanisms of Immunotoxicity: A Systematic Review. Crit. Rev. Toxicol. 2021, 51, 634–652. [Google Scholar] [CrossRef] [PubMed]
- Avissar-Whiting, M.; Veiga, K.R.; Uhl, K.M.; Maccani, M.A.; Gagne, L.A.; Moen, E.L.; Marsit, C.J. Bisphenol a Exposure Leads to Specific Microrna Alterations in Placental Cells. Reprod. Toxicol. 2010, 29, 401–406. [Google Scholar] [CrossRef]
- Perry, M.M.; Moschos, S.A.; Williams, A.E.; Shepherd, N.J.; Larner-Svensson, H.M.; Lindsay, M.A. Rapid Changes in Microrna-146a Expression Negatively Regulate the Il-1beta-Induced Inflammatory Response in Human Lung Alveolar Epithelial Cells. J. Immunol. 2008, 180, 5689–5698. [Google Scholar] [CrossRef]
- He, Y.; Sun, X.; Huang, C.; Long, X.-R.; Lin, X.; Zhang, L.; Lv, X.-W.; Li, J. Mir-146a Regulates Il-6 Production in Lipopolysaccharide-Induced Raw264.7 Macrophage Cells by Inhibiting Notch1. Inflammation 2014, 37, 71–82. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Feng, B.; Thomas, A.A.; Chakrabarti, S. Mir-146a Regulates Glucose Induced Upregulation of Inflammatory Cytokines Extracellular Matrix Proteins in the Retina and Kidney in Diabetes. PLoS ONE 2017, 12, e0173918. [Google Scholar] [CrossRef] [PubMed]
- Wallet, M.A.; Calderon, N.L.; Alonso, T.R.; Choe, C.S.; Catalfamo, D.L.; Lalane, C.J.; Neiva, K.G.; Panagakos, F.; Wallet, S.M. Triclosan Alters Antimicrobial and Inflammatory Responses of Epithelial Cells. Oral. Dis. 2013, 19, 296–302. [Google Scholar] [CrossRef]
- Wu, D.; Xi, Q.-Y.; Cheng, X.; Dong, T.; Zhu, X.-T.; Shu, G.; Wang, L.-N.; Jiang, Q.-Y.; Zhang, Y.-L. Mir-146a-5p Inhibits Tnf-A-Induced Adipogenesis Via Targeting Insulin Receptor in Primary Porcine Adipocytes. J. Lipid Res. 2016, 57, 1360–1372. [Google Scholar] [CrossRef] [PubMed]
- He, Q.; Song, J.; Cui, C.; Wang, J.; Hu, H.; Guo, X.; Yang, M.; Wang, L.; Yan, F.; Liang, K.; et al. Mesenchymal Stem Cell-Derived Exosomal Mir-146a Reverses Diabetic B-Cell Dedifferentiation. Stem. Cell. Res. Ther. 2021, 12, 449. [Google Scholar] [CrossRef] [PubMed]
- Li, K.; Zhao, B.; Wei, D.; Wang, W.; Cui, Y.; Qian, L.; Liu, G. Mir-146a Improves Hepatic Lipid and Glucose Metabolism by Targeting Med1. Int. J. Mol. Med. 2020, 45, 543–555. [Google Scholar] [CrossRef] [PubMed]
- Tilghman, S.L.; Bratton, M.R.; Segar, H.C.; Martin, E.C.; Rhodes, L.V.; Li, M.; McLachlan, J.A.; Wiese, T.E.; Nephew, K.P.; Burow, M.E. Endocrine Disruptor Regulation of Microrna Expression in Breast Carcinoma Cells. PLoS ONE 2012, 7, e32754. [Google Scholar] [CrossRef]
- Al-Badrani, M.; Seth, R.K.; Sarkar, S.; Kimono, D.; Mondal, A.; Bose, D.; Porter, D.E.; Scott, G.I.; Brooks, B.; Raychoudhury, S.; et al. Exogenous Pp2a Inhibitor Exacerbates the Progression of Nonalcoholic Fatty Liver Disease Via Nox2-Dependent Activation of Mir21. Am. J. Physiol. Gastrointest Liver. Physiol. 2019, 317, G408–G428. [Google Scholar] [CrossRef]
- Lai, C.Y.; Yeh, K.Y.; Lin, C.Y.; Hsieh, Y.W.; Lai, H.H.; Chen, J.R.; Hsu, C.C.; Her, G.G. Microrna-21 Plays Multiple Oncometabolic Roles in the Process of Nafld-Related Hepatocellular Carcinoma Via Pi3k/Akt, Tgf-B, and Stat3 Signaling. Cancers 2021, 13, 940. [Google Scholar] [CrossRef]
- Sun, C.; Huang, F.; Liu, X.; Xiao, X.; Yang, M.; Hu, G.; Liu, H.; Liao, L. Mir-21 Regulates Triglyceride and Cholesterol Metabolism in Non-Alcoholic Fatty Liver Disease by Targeting Hmgcr. Int. J. Mol. Med. 2015, 35, 847–853. [Google Scholar] [CrossRef]
- Lhamyani, S.; Gentile, A.-M.; Giráldez-Pérez, R.M.; Feijóo-Cuaresma, M.; Romero-Zerbo, S.Y.; Clemente-Postigo, M.; Zayed, H.; Oliva-Olivera, W.; Bermúdez-Silva, F.J.; Salas, J.; et al. Mir-21 Mimic Blocks Obesity in Mice: A Novel Therapeutic Option. Mol. Ther. Nucleic. Acids. 2021, 26, 401–416. [Google Scholar] [CrossRef] [PubMed]
- Dong, W.; Gao, W.; Cui, J.; Xu, T.; Sun, Y. Microrna-148 Is Involved in Nf-Κb Signaling Pathway Regulation after Lps Stimulation by Targeting Il-1β in Miiuy Croaker. Fish Shellfish. Immunol. 2021, 118, 66–71. [Google Scholar] [CrossRef] [PubMed]
- Yin, Q.; Wang, P.; Wu, X. Microrna -148 Alleviates Cardiac Dysfunction, Immune Disorders and Myocardial Apoptosis in Myocardial Ischemia-Reperfusion (Mi/R) Injury by Targeting Pyruvate Dehydrogenase Kinase (Pdk4). Bioengineered 2021, 12, 5552–5565. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Cheng, H.; Yue, Y.; Li, S.; Zhang, D.; He, R. H19 Knockdown Suppresses Proliferation and Induces Apoptosis by Regulating Mir-148b/Wnt/B-Catenin in Ox-Ldl -Stimulated Vascular Smooth Muscle Cells. J. Biomed. Sci. 2018, 25, 11. [Google Scholar] [CrossRef] [PubMed]
- Melkman-Zehavi, T.; Oren, R.; Kredo-Russo, S.; Shapira, T.; Mandelbaum, A.D.; Rivkin, N.; Nir, T.; Lennox, K.A.; Behlke, M.A.; Dor, Y.; et al. miRNAs Control Insulin Content in Pancreatic B-Cells Via Downregulation of Transcriptional Repressors. Embo. J. 2011, 30, 835–845. [Google Scholar] [CrossRef]
- Hanieh, H.; Alzahrani, A. Microrna-132 Suppresses Autoimmune Encephalomyelitis by Inducing Cholinergic Anti-Inflammation: A New Ahr-Based Exploration. Eur. J. Immunol. 2013, 43, 2771–2782. [Google Scholar]
- Liu, F.; Li, Y.; Jiang, R.; Nie, C.; Zeng, Z.; Zhao, N.; Huang, C.; Shao, Q.; Ding, C.; Qing, C.; et al. Mir-132 Inhibits Lipopolysaccharide-Induced Inflammation in Alveolar Macrophages by the Cholinergic Anti-Inflammatory Pathway. Exp. Lung. Res. 2015, 41, 261–269. [Google Scholar] [CrossRef]
- Mziaut, H.; Henniger, G.; Ganss, K.; Hempel, S.; Wolk, S.; McChord, J.; Chowdhury, K.; Ravassard, P.; Knoch, K.-P.; Krautz, C.; et al. Mir-132 Controls Pancreatic Beta Cell Proliferation and Survival through Pten/Akt/Foxo3 Signaling. Mol. Metab. 2020, 31, 150–162. [Google Scholar] [CrossRef]
- Smith, P.Y.; Hernandez-Rapp, J.; Jolivette, F.; Lecours, C.; Bisht, K.; Goupil, C.; Dorval, V.; Parsi, S.; Morin, F.; Planel, E.; et al. Mir-132/212 Deficiency Impairs Tau Metabolism and Promotes Pathological Aggregation in Vivo. Hum. Mol. Genet. 2015, 24, 6721–6735. [Google Scholar] [CrossRef]
- Heo, J.S.; Kim, S. Human Adipose Mesenchymal Stem Cells Modulate Inflammation and Angiogenesis through Exosomes. Sci. Rep. 2022, 12, 2776. [Google Scholar] [CrossRef]
- Liu, H.; Zhang, L.; Li, M.; Zhao, F.; Lu, F.; Zhang, F.; Chen, S.; Guo, J.; Zhang, R.; Yin, H. Bone Mesenchymal Stem Cell-Derived Extracellular Vesicles Inhibit Dapk1-Mediated Inflammation by Delivering Mir-191 to Macrophages. Biochem. Biophys. Res. Commun. 2022, 598, 32–39. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Zou, Z.; Liu, Y.; Wang, Q.; Sun, B.; Zeng, Q.; Liu, Q.; Zhang, A. Mir-191 Is Involved in Renal Dysfunction in Arsenic-Exposed Populations by Regulating Inflammatory Response Caused by Arsenic from Burning Arsenic-Contaminated Coal. Hum. Exp. Toxicol. 2020, 39, 37–46. [Google Scholar] [CrossRef] [PubMed]
- Guida, M.; Marra, M.; Zullo, F.; Guida, M.; Trifuoggi, M.; Biffali, E.; Borra, M.; De Mieri, G.; D’Alessandro, R.; De Felice, B. Association between Exposure to Dioxin-Like Polychlorinated Biphenyls and Mir-191 Expression in Human Peripheral Blood Mononuclear Cells. Mutat. Res. 2013, 753, 36–41. [Google Scholar] [CrossRef] [PubMed]
- Du, K.; Zhao, C.; Wang, L.; Wang, Y.; Zhang, K.-Z.; Shen, X.-Y.; Sun, H.-X.; Gao, W.; Lu, X. Mir-191 Inhibit Angiogenesis after Acute Ischemic Stroke Targeting Vezf1. Aging 2019, 11, 2762–2786. [Google Scholar] [CrossRef]
- Gu, Y.; Ampofo, E.; Menger, M.D.; Laschke, M.W. Mir-191 Suppresses Angiogenesis by Activation of Nf-Κb Signaling. Faseb. J. 2017, 31, 3321–3333. [Google Scholar] [CrossRef]
- Xu, X.; Zhou, X.; Zhang, J.; Li, H.; Cao, Y.; Tan, X.; Zhu, X.; Yang, J. Microrna-191 Modulates Cisplatin-Induced DNA Damage Response by Targeting Rcc2. Faseb. J. 2020, 34, 13573–13585. [Google Scholar] [CrossRef]
- Li, W.; Wu, L.; Sun, Q.; Yang, Q.; Xue, J.; Shi, M.; Tang, H.; Zhang, J.; Liu, Q. Microrna-191 Blocking the Translocation of Glut4 Is Involved in Arsenite-Induced Hepatic Insulin Resistance through Inhibiting the Irs1/Akt Pathway. Ecotoxicol. Environ. Saf. 2021, 215, 112130. [Google Scholar] [CrossRef]
- Badry, A.; Jaspers, V.L.B.; Waugh, C.A. Environmental Pollutants Modulate Rna and DNA Virus-Activated Mirna-155 Expression and Innate Immune System Responses: Insights into New Immunomodulative Mechanisms. J. Immunotoxicol. 2020, 17, 86–93. [Google Scholar] [CrossRef]
- Waugh, C.A.; Arukwe, A.; Jaspers, V.L.B. Deregulation of Microrna-155 and Its Transcription Factor Nf-Kb by Polychlorinated Biphenyls During Viral Infections. Apmis 2018, 126, 234–240. [Google Scholar] [CrossRef]
- Yang, X.; Wan, J.; Li, N.; He, C.; Zhang, Y.; Ren, Y.; Li, X.; Zhu, Y.; Liu, F.; Xia, L.; et al. Mir155 Disrupts the Intestinal Barrier by Inducing Intestinal Inflammation and Altering the Intestinal Microecology in Severe Acute Pancreatitis. Dig. Dis. Sci. 2021, 67, 2209–2219. [Google Scholar] [CrossRef]
- Wang, Y.-Z.; Feng, X.-G.; Shi, Q.-G.; Hao, Y.-L.; Yang, Y.; Zhang, A.-M.; Kong, Q.-X. Silencing of Mir155 Promotes the Production of Inflammatory Mediators in Guillain-Barré Syndrome in Vitro. Inflammation 2013, 36, 337–345. [Google Scholar] [CrossRef] [PubMed]
- Bala, S.; Csak, T.; Saha, B.; Zatsiorsky, J.; Kodys, K.; Catalano, D.; Satishchandran, A.; Szabo, G. The Pro-Inflammatory Effects of Mir-155 Promote Liver Fibrosis and Alcohol-Induced Steatohepatitis. J. Hepatol. 2016, 64, 1378–1387. [Google Scholar] [CrossRef] [PubMed]
- Du, F.; Yu, F.; Wang, Y.; Hui, Y.; Carnevale, K.; Fu, M.; Lu, H.; Fan, D. Microrna-155 Deficiency Results in Decreased Macrophage Inflammation and Attenuated Atherogenesis in Apolipoprotein E-Deficient Mice. Arter. Thromb. Vasc. Biol. 2014, 34, 759–767. [Google Scholar] [CrossRef] [PubMed]
- Zheng, C.; Zhang, J.; Chen, X.; Zhang, J.; Ding, X.; You, X.; Fan, L.; Chen, C.; Zhou, Y. Microrna-155 Mediates Obesity-Induced Renal Inflammation and Dysfunction. Inflammation 2019, 42, 994–1003. [Google Scholar] [CrossRef]
- Wu, N.; Ye, C.; Zheng, F.; Wan, G.-W.; Wu, L.-L.; Chen, Q.; Li, Y.-H.; Kang, Y.-M.; Zhu, G.-Q. Mir155-5p Inhibits Cell Migration and Oxidative Stress in Vascular Smooth Muscle Cells of Spontaneously Hypertensive Rats. Antioxidants 2020, 9, 204. [Google Scholar] [CrossRef]
- Zhang, X.; Hua, F.; Yang, Z.; Chen, Y.; Teng, X.; Huang, H.; Zhao, Y.; Shen, Z. Enhancement of Immunoregulatory Function of Modified Bone Marrow Mesenchymal Stem Cells by Targeting Socs1. Biomed. Res. Int. 2018, 2018, 3530647. [Google Scholar] [CrossRef]
- Velázquez, K.T.; Enos, R.T.; Carson, M.S.; Cranford, T.L.; Bader, J.E.; Sougiannis, A.T.; Pritchett, C.; Fan, D.; Carson, J.A.; Murphy, E.A. Mir155 Deficiency Aggravates High-Fat Diet-Induced Adipose Tissue Fibrosis in Male Mice. Physiol. Rep. 2017, 5, e13412. [Google Scholar] [CrossRef]
- Zhang, C.; Yu, S.; Zheng, B.; Liu, D.; Wan, F.; Ma, Y.; Wang, J.; Gao, Z.; Shan, Z. Mir-30c-5p Reduces Renal Ischemia-Reperfusion Involving Macrophage. Med. Sci. Monit. 2019, 25, 4362–4369. [Google Scholar] [CrossRef]
- Demolli, S.; Doebele, C.; Doddaballapur, A.; Lang, V.; Fisslthaler, B.; Chavakis, E.; Vinciguerra, M.; Sciacca, S.; Henschler, R.; Hecker, M.; et al. Microrna-30 Mediates Anti-Inflammatory Effects of Shear Stress and Klf2 Via Repression of Angiopoietin 2. J. Mol. Cell. Cardiol. 2015, 88, 111–119. [Google Scholar] [CrossRef]
- Lin, X.; Yu, S.; Ren, P.; Sun, X.; Jin, M. Human Microrna-30 Inhibits Influenza Virus Infection by Suppressing the Expression of Socs1, Socs3, and Nedd4. Cell. Microbiol. 2020, 22, e13150. [Google Scholar] [CrossRef]
- Wu, Y.; Sun, Q.; Dai, L. Immune Regulation of Mir-30 on the Mycobacterium Tuberculosis-Induced Tlr/Myd88 Signaling Pathway in Thp-1 Cells. Exp. Ther. Med. 2017, 14, 3299–3303. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Duan, Z.Q.; Shi, J.D.; Wu, M.N.; Hu, N.Z.; Hu, Y.Z. Influence of Mir-30b Regulating Humoral Immune Response by Genetic Difference. Immunol. Res. 2016, 64, 181–190. [Google Scholar] [CrossRef] [PubMed]
- Koh, E.-H.; Chernis, N.; Saha, P.K.; Xiao, L.; Bader, D.A.; Zhu, B.; Rajapakshe, K.; Hamilton, M.P.; Liu, X.; Perera, D.; et al. Mir-30a Remodels Subcutaneous Adipose Tissue Inflammation to Improve Insulin Sensitivity in Obesity. Diabetes 2018, 67, 2541–2553. [Google Scholar] [CrossRef] [PubMed]
- Mahjoor, M.; Afkhami, H.; Mollaei, M.; Nasr, A.; Shahriary, S.; Khorrami, S. Microrna-30c Delivered by Bone Marrow-Mesenchymal Stem Cells Induced Apoptosis and Diminished Cell Invasion in U-251 Glioblastoma Cell Line. Life Sci. 2021, 279, 119643. [Google Scholar] [CrossRef]
- Li, W.F.; Dai, H.; Ou, Q.; Zuo, G.Q.; Liu, C.A. Overexpression of Microrna-30a-5p Inhibits Liver Cancer Cell Proliferation and Induces Apoptosis by Targeting Mtdh/Pten/Akt Pathway. Tumour. Biol. 2016, 37, 5885–5895. [Google Scholar] [CrossRef]
- Saha, P.K.; Hamilton, M.P.; Rajapakshe, K.; Putluri, V.; Felix, J.B.; Masschelin, P.; Cox, A.R.; Bajaj, M.; Putluri, N.; Coarfa, C.; et al. Mir-30a Targets Gene Networks That Promote Browning of Human and Mouse Adipocytes. Am. J. Physiol. Endocrinol. Metab. 2020, 319, E667–E677. [Google Scholar] [CrossRef]
- Miranda, K.; Yang, X.; Bam, M.; Murphy, E.A.; Nagarkatti, P.S.; Nagarkatti, M. Microrna-30 Modulates Metabolic Inflammation by Regulating Notch Signaling in Adipose Tissue Macrophages. Int. J. Obes. 2018, 42, 1140–1150. [Google Scholar] [CrossRef]
- Zhang, Q.; Ma, X.-F.; Dong, M.-Z.; Tan, J.; Zhang, J.; Zhuang, L.-K.; Liu, S.-S.; Xin, Y.-N. Mir-30b-5p Regulates the Lipid Metabolism by Targeting Ppargc1a in Huh-7 Cell Line. Lipids. Health Dis. 2020, 19, 76. [Google Scholar] [CrossRef]
- Singh, N.P.; Miranda, K.; Singh, U.P.; Nagarkatti, P.; Nagarkatti, M. Diethylstilbestrol (Des) Induces Autophagy in Thymocytes by Regulating Beclin-1 Expression through Epigenetic Modulation. Toxicology 2018, 410, 49–58. [Google Scholar] [CrossRef]
- Zhong, J.; Baccarelli, A.A.; Mansur, A.; Adir, M.; Nahum, R.; Hauser, R.; Bollati, V.; Racowsky, C.; Machtinger, R. Maternal Phthalate and Personal Care Products Exposure Alters Extracellular Placental Mirna Profile in Twin Pregnancies. Reprod. Sci. 2019, 26, 289–294. [Google Scholar] [CrossRef]
- Waterhouse, N.J.; Sedelies, K.A.; Browne, K.A.; Wowk, M.E.; Newbold, A.; Sutton, V.R.; Clarke, C.J.P.; Oliaro, J.; Lindemann, R.K.; Bird, P.; et al. A Central Role for Bid in Granzyme B-Induced Apoptosis. J. Biol. Chem. 2005, 280, 4476–4482. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Zhong, B.; Zhang, C.; Luo, C.; Zhan, Y. Mir-373 Regulates Inflammatory Cytokine-Mediated Chondrocyte Proliferation in Osteoarthritis by Targeting the P2x7 Receptor. FEBS Open Bio. 2018, 8, 325–331. [Google Scholar] [CrossRef] [PubMed]
- Yin, Q.H.; Zhou, Y.; Li, Z.H.Y. Mir-373 Suppresses Cell Proliferation and Apoptosis Via Regulation of Sirt1/Pgc-1α/Nrf2 Axis in Pancreatic Cancer. Cell J. 2021, 23, 199–210. [Google Scholar] [PubMed]
- Peng, T.; Wang, T.; Liu, G.; Zhou, L. Effects of Mir-373 Inhibition on Glioblastoma Growth by Reducing Limk1 in Vitro. J Immunol Res. 2020, 2020, 7671502. [Google Scholar] [CrossRef]
- Lv, P.; Luo, Y.; Zhou, W.; Liu, B.; Zhou, Z.; Shi, Y.; Huang, R.; Peng, C.; He, Z.; Wang, J.; et al. Mir-373 Inhibits Autophagy and Further Promotes Apoptosis of Cholangiocarcinoma Cells by Targeting Ulk1. Kaohsiung J. Med. Sci. 2020, 36, 429–440. [Google Scholar] [CrossRef] [PubMed]
- Li, C.H.; Tang, S.C.; Wong, C.H.; Wang, Y.; Jiang, J.D.; Chen, Y. Berberine Induces Mir-373 Expression in Hepatocytes to Inactivate Hepatic Steatosis Associated Akt-S6 Kinase Pathway. Eur. J. Pharmacol. 2018, 825, 107–118. [Google Scholar] [CrossRef] [PubMed]
- Hart, M.; Walch-Rückheim, B.; Friedmann, K.S.; Rheinheimer, S.; Tänzer, T.; Glombitza, B.; Sester, M.; Lenhof, H.; Hoth, M.; Schwarz, E.C.; et al. Mir-34a: A New Player in the Regulation of T Cell Function by Modulation of Nf-Κb Signaling. Cell. Death. Dis. 2019, 10, 46. [Google Scholar] [CrossRef]
- Wu, J.; Li, X.; Li, D.; Ren, X.; Li, Y.; Herter, E.K.; Qian, M.; Toma, M.-A.; Wintler, A.-M.; Sérézal, I.G.; et al. Microrna-34 Family Enhances Wound Inflammation by Targeting Lgr4. J. Invest. Derm. 2020, 140, 465–476.e11. [Google Scholar] [CrossRef]
- Dehghan, R.; Najafi, R.; Jalilian, F.A.; Saidijam, M.; Radaei, Z.; Zamani, A.; Ezati, R.; Asna-Ashari, F.; Amini, R. A Promising Effect of Zerumbone with Improved Anti-Tumor-Promoting Inflammation Activity of Mir-34a in Colorectal Cancer Cell Lines. Mol. Biol. Rep. 2021, 48, 203–218. [Google Scholar] [CrossRef]
- Ostrycharz, E.; Wasik, U.; Kempinska-Podhorodecka, A.; Banales, J.M.; Milkiewicz, P.; Milkiewicz, M. Melatonin Protects Cholangiocytes from Oxidative Stress-Induced Proapoptotic and Proinflammatory Stimuli Via Mir-132 and Mir-34. Int. J. Mol. Sci. 2020, 21, 9667. [Google Scholar] [CrossRef]
- Meruvu, S.; Zhang, J.; Choudhury, M. Butyl Benzyl Phthalate Promotes Adipogenesis in 3t3-L1 Cells Via the Mirna-34a-5p Signaling Pathway in the Absence of Exogenous Adipogenic Stimuli. Chem. Res. Toxicol. 2021, 34, 2251–2260. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.R.; Roe, J.S.; Lee, J.E.; Cho, E.J.; Youn, H.D. P53 Regulates Glucose Metabolism by Mir-34a. Biochem. Biophys. Res. Commun 2013, 437, 225–231. [Google Scholar] [CrossRef] [PubMed]
- Kaller, M.; Liffers, S.-T.; Oeljeklaus, S.; Kuhlmann, K.; Roeh, S.; Hoffmann, R.; Warscheid, B.; Hermeking, H. Genome-Wide Characterization of Mir-34a Induced Changes in Protein and Mrna Expression by a Combined Pulsed Silac and Microarray Analysis. Mol. Cell. Proteom. 2011, 10, M111.010462. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.; Lu, Z.; Gu, J.; Liu, J.; Huang, E.; Liu, X.; Wang, L.; Yang, J.; Deng, Y.; Qian, J.; et al. Micrornas 15a and 16-1 Activate Signaling Pathways That Mediate Chemotaxis of Immune Regulatory B Cells To colorectal Tumors. Gastroenterology 2018, 154, 637–651.e7. [Google Scholar] [CrossRef]
- Tian, Y.; Cui, L.; Lin, C.; Wang, Y.; Liu, Z.; Miao, X. Lncrna Cdkn2b-As1 Relieved Inflammation of Ulcerative Colitis Via Sponging Mir-16 and Mir-195. Int. Immunopharmacol. 2020, 88, 106970. [Google Scholar] [CrossRef]
- Li, X.; Chu, Q.; Wang, H. Microrna-16 Regulates Lipopolysaccharide-Induced Inflammatory Factor Expression by Targeting Tlr4 in Normal Human Bronchial Epithelial Cells. Exp. Ther. Med. 2021, 22, 982. [Google Scholar] [CrossRef]
- Wang, M.; Li, J.; Cai, J.; Cheng, L.; Wang, X.; Xu, P.; Li, G.; Liang, X. Overexpression of Microrna-16 Alleviates Atherosclerosis by Inhibition of Inflammatory Pathways. Biomed Res. Int. 2020, 2020, 8504238. [Google Scholar] [CrossRef]
- Du, K.; He, X.; Deng, J. Microrna-16 Inhibits the Lipopolysaccharide-Induced Inflammatory Response in Nucleus Pulposus Cells of the Intervertebral Disc by Targeting Tab3. Arch. Med. Sci. 2021, 17, 500–513. [Google Scholar] [CrossRef]
- Liu, L. The Anti-Inflammatory Effect of Mir-16 through Targeting C- Reactive Protein Is Regulated by Hur in Vascular Smooth Muscle Cells. Biochem. Biophys. Res. Commun. 2020, 528, 636–643. [Google Scholar] [CrossRef]
- Wang, D.W.; Wang, Y.Q.; Shu, H.S. Mir-16 Inhibits Pituitary Adenoma Cell Proliferation Via the Suppression of Erk/Mapk Signal Pathway. Eur. Rev. Med. Pharmacol. Sci. 2020, 24, 9236. [Google Scholar]
- Wu, R.; Li, X.; Li, S.; Tang, G.; Zhang, S.; Zhu, Y.; Zhang, X.; Deng, M.; Tan, S.; Luo, S.; et al. Decreased Microrna-126 Expression in Psoriatic Cd4(+) T Cells Promotes T-Helper 17 Cell Differentiation and the Formation of Dermatitis in Imiquimod-Induced Psoriasis-Like Mice. J. Dermatol. 2021, 49, 432–440. [Google Scholar] [CrossRef] [PubMed]
- Chu, F.; Hu, Y.; Zhou, Y.; Guo, M.; Lu, J.; Zheng, W.; Xu, H.; Zhao, J.; Xu, L. Microrna-126 Deficiency Enhanced the Activation and Function of Cd4(+) T Cells by Elevating Irs-1 Pathway. Clin. Exp. Immunol. 2018, 191, 166–179. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Wei, E.; Wei, J.; Zhou, W.; Webster, K.A.; Zhang, B.; Li, D.; Zhang, G.; Wei, Y.; Long, Y.; et al. Mir-126-Hmgb1-Hif-1 Axis Regulates Endothelial Cell Inflammation During Exposure to Hypoxia-Acidosis. Dis. Markers 2021, 2021, 4933194. [Google Scholar] [CrossRef] [PubMed]
- Tomasetti, M.; Monaco, F.; Manzella, N.; Rohlena, J.; Rohlenova, K.; Staffolani, S.; Gaetani, S.; Ciarapica, V.; Amati, M.; Bracci, M.; et al. Microrna-126 Induces Autophagy by Altering Cell Metabolism in Malignant Mesothelioma. Oncotarget 2016, 7, 36338–36352. [Google Scholar] [CrossRef]
- Shi, C.C.; Pan, L.Y.; Peng, Z.Y.; Li, J.G. Mir-126 Regulated Myocardial Autophagy on Myocardial Infarction. Eur. Rev. Med. Pharmacol. Sci. 2020, 24, 6971–6979. [Google Scholar]
- Najm, A.; Masson, F.; Preuss, P.; Georges, S.; Ory, B.; Quillard, T.; Sood, S.; Goodyear, C.S.; Veale, D.J.; Fearon, U.; et al. Microrna-17-5p Reduces Inflammation and Bone Erosions in Mice with Collagen-Induced Arthritis and Directly Targets the Jak/Stat Pathway in Rheumatoid Arthritis Fibroblast-Like Synoviocytes. Arthritis Rheumatol. 2020, 72, 2030–2039. [Google Scholar] [CrossRef]
- Liu, Y.; Lou, G.; Li, A.; Zhang, T.; Qi, J.; Ye, D.; Zheng, M.; Chen, Z. Amsc-Derived Exosomes Alleviate Lipopolysaccharide/D-Galactosamine-Induced Acute Liver Failure by Mir-17-Mediated Reduction of Txnip/Nlrp3 Inflammasome Activation in Macrophages. EBioMedicine 2018, 36, 140–150. [Google Scholar] [CrossRef]
- Zhang, X.; Liu, J.; Wu, L.; Hu, X. Micrornas of the Mir-17~9 Family Maintain Adipose Tissue Macrophage Homeostasis by Sustaining Il-10 Expression. eLife 2020, 9, 55676. [Google Scholar] [CrossRef]
- Benhamou, D.; Labi, V.; Novak, R.; Dai, I.; Shafir-Alon, S.; Weiss, A.; Gaujoux, R.; Arnold, R.; Shen-Orr, S.S.; Rajewsky, K.; et al. A C-Myc/Mir17-92/Pten Axis Controls Pi3k-Mediated Positive and Negative Selection in B Cell Development and Reconstitutes Cd19 Deficiency. Cell. Rep. 2016, 16, 419–431. [Google Scholar] [CrossRef]
- Benhamou, D.; Labi, V.; Getahun, A.; Benchetrit, E.; Dowery, R.; Rajewsky, K.; Cambier, J.C.; Melamed, D. The C-Myc/Mir17-92/Pten Axis Tunes Pi3k Activity to Control Expression of Recombination Activating Genes in Early B Cell Development. Front. Immunol. 2018, 9, 2715. [Google Scholar] [CrossRef]
- Huang, Y.; Zhang, H.; Dong, M.; Zhang, L.; Lin, J.; Ye, R.; Zhou, H.; Liu, X.; Jin, W. Mir17-92 Cluster Drives White Adipose Tissue Browning. J. Mol. Endocrinol. 2020, 65, 97–107. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Qian, D.; Zhao, H.; Lv, N.; Yu, P.; Sun, Z. Mir17 Improves Insulin Sensitivity through Inhibiting Expression of Ask1 and Anti-Inflammation of Macrophages. Biomed. Pharm. 2018, 100, 448–454. [Google Scholar] [CrossRef] [PubMed]
- Chiba, T.; Cerqueira, D.M.; Li, Y.; Bodnar, A.J.; Mukherjee, E.; Pfister, K.; Phua, Y.L.; Shaikh, K.; Sanders, B.T.; Hemker, S.L.; et al. Endothelial-Derived Mir-17∼92 Promotes Angiogenesis to Protect against Renal Ischemia-Reperfusion Injury. J. Am. Soc. Nephrol. 2021, 32, 553–562. [Google Scholar] [CrossRef]
- Akkouch, A.; Zhu, M.; Romero-Bustillos, M.; Eliason, S.; Qian, F.; Salem, A.K.; Amendt, B.A.; Hong, L. Microrna-200c Attenuates Periodontitis by Modulating Proinflammatory and Osteoclastogenic Mediators. Stem. Cells. Dev. 2019, 28, 1026–1036. [Google Scholar] [CrossRef] [PubMed]
- Reddy, M.A.; Jin, W.; Villeneuve, L.; Wang, M.; Lanting, L.; Todorov, I.; Kato, M.; Natarajan, R. Pro-Inflammatory Role of Microrna-200 in Vascular Smooth Muscle Cells from Diabetic Mice. Arter. Thromb. Vasc. Biol. 2012, 32, 721–729. [Google Scholar] [CrossRef]
- Chen, L.; Gibbons, D.L.; Goswami, S.; Cortez, M.A.; Ahn, Y.H.; Byers, L.A.; Zhang, X.; Yi, X.; Dwyer, D.; Lin, W.; et al. Metastasis Is Regulated Via Microrna-200/Zeb1 Axis Control of Tumour Cell Pd-L1 Expression and Intratumoral Immunosuppression. Nat. Commun. 2014, 5, 5241. [Google Scholar] [CrossRef]
- Wang, Y.; Wen, Y.; Xiao, P.; Sun, J.; Chen, M.; Gu, C.; Kong, Y.; Gu, A.; Zhang, J.; Wang, Y. Di-N-Butyl Phthalate Promotes Lipid Accumulation Via the Mir200c-5p-Abca1 Pathway in Thp-1 Macrophages. Environ. Pollut. 2020, 264, 114723. [Google Scholar] [CrossRef]
- Tao, C.; Ren, H.; Xu, P.; Cheng, J.; Huang, S.; Zhou, R.; Mu, Y.; Yang, S.; Qi, D.; Wang, Y.; et al. Adipocyte Mir-200b/a/429 Ablation in Mice Leads to High-Fat-Diet-Induced Obesity. Oncotarget 2016, 7, 67796–67807. [Google Scholar] [CrossRef]
- Jones, R.; Watson, K.; Bruce, A.; Nersesian, S.; Kitz, J.; Moorehead, R. Re-Expression of Mir-200c Suppresses Proliferation, Colony Formation and in Vivo Tumor Growth of Murine Claudin-Low Mammary Tumor Cells. Oncotarget 2017, 8, 23727–23749. [Google Scholar] [CrossRef]
- Zhao, C.; Xie, R.; Qian, Q.; Yan, J.; Wang, H.; Wang, X. Triclosan Induced Zebrafish Immunotoxicity by Targeting Mir-19a and Its Gene Socs3b to Activate Il-6/Stat3 Signaling Pathway. Sci. Total. Environ. 2022, 815, 152916. [Google Scholar] [CrossRef]
- Mu, N.; Gu, J.; Huang, T.; Zhang, C.; Shu, Z.; Li, M.; Hao, Q.; Li, W.; Zhang, W.; Zhao, J.; et al. A Novel Nf-Κb/Yy1/Microrna-10a Regulatory Circuit in Fibroblast-Like Synoviocytes Regulates Inflammation in Rheumatoid Arthritis. Sci. Rep. 2016, 6, 20059. [Google Scholar] [CrossRef]
- Wang, W.; Zhan, R.; Zhou, J.; Wang, J.; Chen, S. Mir-10 Targets Ngr to Modulate the Proliferation of Microglial Cells and the Secretion of Inflammatory Cytokines. Exp. Mol. Pathol. 2018, 105, 357–363. [Google Scholar] [CrossRef] [PubMed]
- Cho, Y.K.; Son, Y.; Kim, S.-N.; Song, H.-D.; Kim, M.; Park, J.-H.; Jung, Y.-S.; Ahn, S.-Y.; Saha, A.; Granneman, J.G.; et al. Microrna-10a-5p Regulates Macrophage Polarization and Promotes Therapeutic Adipose Tissue Remodeling. Mol. Metab. 2019, 29, 86–98. [Google Scholar] [CrossRef] [PubMed]
- Xu, D.; Li, W.; Zhang, T.; Wang, G. Mir-10a Overexpression Aggravates Renal Ischemia-Reperfusion Injury Associated with Decreased Pik3ca Expression. BMC Nephrol. 2020, 21, 248. [Google Scholar] [CrossRef] [PubMed]
- Jiajie, T.; Yanzhou, Y.; Hoi-Hung, A.C.; Zi-Jiang, C.; Wai-Yee, C. Conserved Mir-10 Family Represses Proliferation and Induces Apoptosis in Ovarian Granulosa Cells. Sci. Rep. 2017, 7, 41304. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.; Ha, S.E.; Wei, L.; Jin, B.; Zogg, H.; Poudrier, S.M.; Jorgensen, B.G.; Park, C.; Ronkon, C.F.; Bartlett, A.; et al. Mir-10b-5p Rescues Diabetes and Gastrointestinal Dysmotility. Gastroenterology 2021, 160, 1662–1678.e18. [Google Scholar] [CrossRef]
- Gouesse, R.J.; Plante, I. Environmental Exposure to Bfrs: Unraveling Endocrine and Mammary Gland Effects That May Increase Disease Risk. Toxicol. Sci. 2022, 186, 190–207. [Google Scholar] [CrossRef]
- Ehrlich, S.; Lambers, D.; Baccarelli, A.; Khoury, J.; Macaluso, M.; Ho, S.M. Endocrine Disruptors: A Potential Risk Factor for Gestational Diabetes Mellitus. Am. J. Perinatol. 2016, 33, 1313–1318. [Google Scholar]
- Wolstenholme, J.T.; Rissman, E.F.; Connelly, J.J. The Role of Bisphenol a in Shaping the Brain, Epigenome and Behavior. Horm. Behav. 2011, 59, 296–305. [Google Scholar] [CrossRef]
- Hafezi, S.A.; Abdel-Rahman, W.M. The Endocrine Disruptor Bisphenol a (Bpa) Exerts a Wide Range of Effects in Carcinogenesis and Response to Therapy. Curr. Mol. Pharmacol. 2019, 12, 230–238. [Google Scholar] [CrossRef]
- Miguel, V.; Cui, J.Y.; Daimiel-Ruiz, L.; Espinosa-Díez, C.; Fernández-Hernando, C.; Kavanagh, T.J.; Lamas, S. The Role of Micrornas in Environmental Risk Factors, Noise-Induced Hearing Loss, and Mental Stress. Antioxid Redox. Signal. 2018, 28, 773–796. [Google Scholar] [CrossRef] [PubMed]
- McAllister, J.M.; Han, A.X.; Modi, B.; Teves, M.E.; Mavodza, G.R.; Anderson, Z.L.; Shen, T.; Christenson, L.K.; Archer, K.J.; Strauss, J.F. Mirna Profiling Reveals Mirna-130b-3p Mediates Dennd1a Variant 2 Expression and Androgen Biosynthesis. Endocrinology 2019, 160, 1964–1981. [Google Scholar] [CrossRef]
- Dong, H.; Curran, I.; Williams, A.; Bondy, G.; Yauk, C.L.; Wade, M.G. Hepatic Mirna Profiles and Thyroid Hormone Homeostasis in Rats Exposed to Dietary Potassium Perfluorooctanesulfonate (Pfos). Environ. Toxicol. Pharmacol. 2016, 41, 201–210. [Google Scholar] [CrossRef]
- Wang, X.; Yang, L.; Wang, H.; Shao, F.; Yu, J.; Jiang, H.; Han, Y.; Gong, D.; Gu, Z. Growth Hormone-Regulated Mrnas and Mirnas in Chicken Hepatocytes. PLoS ONE 2014, 9, e112896. [Google Scholar] [CrossRef] [PubMed]
- Aliberti, P.; Sethi, R.; Belgorosky, A.; Chandran, U.R.; Plant, T.M.; Walker, W.H. Gonadotrophin-Mediated Mirna Expression in Testis at Onset of Puberty in Rhesus Monkey: Predictions on Regulation of Thyroid Hormone Activity and Dlk1-Dio3 Locus. Mol. Hum. Reprod. 2019, 25, 124–136. [Google Scholar] [CrossRef] [PubMed]
- Lo, W.-Y.; Peng, C.-T.; Wang, H.-J. Microrna-146a-5p Mediates High Glucose-Induced Endothelial Inflammation Via Targeting Interleukin-1 Receptor-Associated Kinase 1 Expression. Front Physiol. 2017, 8, 551. [Google Scholar] [CrossRef]
- Gao, G.-Z.; Zhao, Y.; Li, H.-X.; Li, W. Bisphenol a-Elicited Mir-146a-5p Impairs Murine Testicular Steroidogenesis through Negative Regulation of Mta3 Signaling. Biochem. Biophys. Res. Commun. 2018, 501, 478–485. [Google Scholar] [CrossRef]
- Larocca, J.; Binder, A.M.; Mcelrath, T.F.; Michels, K.B. First-Trimester Urine Concentrations of Phthalate Metabolites and Phenols and Placenta Mirna Expression in a Cohort of Us Women. Environ. Health Perspect. 2016, 124, 380–387. [Google Scholar] [CrossRef]
- Park, M.H.; Gutiérrez-García, A.K.; Choudhury, M. Mono-(2-Ethylhexyl) Phthalate Aggravates Inflammatory Response Via Sirtuin Regulation and Inflammasome Activation in Raw 264.7 Cells. Chem. Res. Toxicol. 2019, 32, 935–942. [Google Scholar] [CrossRef]
- Li, G.; Chang, H.; Xia, W.; Mao, Z.; Li, Y.; Xu, S. F0 Maternal Bpa Exposure Induced Glucose Intolerance of F2 Generation through DNA Methylation Change in Gck. Toxicol. Lett. 2014, 228, 192–199. [Google Scholar] [CrossRef]
- Skinner, M.K.; Manikkam, M.; Tracey, R.; Guerrero-Bosagna, C.; Haque, M.; Nilsson, E.E. Ancestral Dichlorodiphenyltrichloroethane (Ddt) Exposure Promotes Epigenetic Transgenerational Inheritance of Obesity. BMC Med. 2013, 11, 228. [Google Scholar] [CrossRef] [PubMed]
- Crews, D.; Mclachlan, J.A. Epigenetics, Evolution, Endocrine Disruption, Health, and Disease. Endocrinology 2006, 147 (Suppl. 6), S4–S10. [Google Scholar] [CrossRef]
- Kaur, S.; Kinkade, J.A.; Green, M.T.; Martin, R.E.; Willemse, T.E.; Bivens, N.J.; Schenk, A.K.; Helferich, W.G.; Trainor, B.C.; Fass, J.; et al. Disruption of Global Hypothalamic Microrna (Mir) Profiles and Associated Behavioral Changes in California Mice (Peromyscus Californicus) Developmentally Exposed to Endocrine Disrupting Chemicals. Horm. Behav. 2021, 128, 104890. [Google Scholar] [CrossRef]
- Brevik, A.; Lindeman, B.; Brunborg, G.; Duale, N. Paternal Benzo[a]Pyrene Exposure Modulates Microrna Expression Patterns in the Developing Mouse Embryo. Int. J. Cell Biol. 2012, 2012, 407431. [Google Scholar] [CrossRef] [PubMed]
- Veiga-Lopez, A.; Luense, L.J.; Christenson, L.K.; Padmanabhan, V. Developmental Programming: Gestational Bisphenol-a Treatment Alters Trajectory of Fetal Ovarian Gene Expression. Endocrinology 2013, 154, 1873–1884. [Google Scholar] [CrossRef]
- Brieño-Enríquez, M.A.; García-López, J.; Cárdenas, D.B.; Guibert, S.; Cleroux, E.; Děd, L.; Hourcade, J.D.D.; Pěknicová, J.; Weber, M.; del Mazo, J. Exposure to Endocrine Disruptor Induces Transgenerational Epigenetic Deregulation of Micrornas in Primordial Germ Cells. PLoS ONE 2015, 10, e0124296. [Google Scholar] [CrossRef]
- Fullston, T.; Teague, E.M.C.O.; Palmer, N.O.; DeBlasio, M.J.; Mitchell, M.; Corbett, M.; Print, C.G.; Owens, J.A.; Lane, M. Paternal Obesity Initiates Metabolic Disturbances in Two Generations of Mice with Incomplete Penetrance to the F2 Generation and Alters the Transcriptional Profile of Testis and Sperm Microrna Content. Faseb. J. 2013, 27, 4226–4243. [Google Scholar] [CrossRef] [PubMed]
- Ho, S.-M.; Cheong, A.; Adgent, M.A.; Veevers, J.; Suen, A.A.; Tam, N.N.C.; Leung, Y.-K.; Jefferson, W.N.; Williams, C.J. Environmental Factors, Epigenetics, and Developmental Origin of Reproductive Disorders. Reprod. Toxicol. 2017, 68, 85–104. [Google Scholar] [CrossRef] [PubMed]
- Kelly, T.L.; Trasler, J.M. Reproductive Epigenetics. Clin. Genet. 2004, 65, 247–260. [Google Scholar] [CrossRef] [PubMed]
- Bredfeldt, T.G.; Greathouse, K.L.; Safe, S.H.; Hung, M.C.; Bedford, M.T.; Walker, C.L. Xenoestrogen-Induced Regulation of Ezh2 and Histone Methylation Via Estrogen Receptor Signaling to Pi3k/Akt. Mol Endocrinol. 2010, 24, 993–1006. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.-Y.; Li, M.; Ge, Z.-J.; Luo, Y.; Ou, X.-H.; Song, S.; Tian, D.; Yang, J.; Zhang, B.; Ou-Yang, Y.-C.; et al. Whole Transcriptome Analysis of the Effects of Type I Diabetes on Mouse Oocytes. PLoS ONE 2012, 7, e41981. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Song, G.; Lim, W. A Mechanism for the Effect of Endocrine Disrupting Chemicals on Placentation. Chemosphere 2019, 231, 326–336. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.Y.; Chen, C.P.; Lin, K.H. Biological Functions of Thyroid Hormone in Placenta. Int. J. Mol. Sci. 2015, 16, 4161–4179. [Google Scholar] [CrossRef]
- Zoeller, R.T. Endocrine Disrupting Chemicals and Thyroid Hormone Action. Adv. Pharmacol. 2021, 92, 401–417. [Google Scholar] [PubMed]
- Aranda, A. Micrornas and Thyroid Hormone Action. Mol. Cell Endocrinol. 2021, 525, 111175. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.-H.; Wu, M.-H.; Liao, C.-J.; Huang, Y.-H.; Chi, H.-C.; Wu, S.-M.; Chen, C.-Y.; Tseng, Y.-H.; Tsai, C.-Y.; Chung, I.-H.; et al. Repression of Microrna-130b by Thyroid Hormone Enhances Cell Motility. J. Hepatol. 2015, 62, 1328–1340. [Google Scholar] [CrossRef]
- Jazdzewski, K.; Boguslawska, J.; Jendrzejewski, J.; Liyanarachchi, S.; Pachucki, J.; Wardyn, K.A.; Nauman, A.; de la Chapelle, A. Thyroid Hormone Receptor B (Thrb) Is a Major Target Gene for Micrornas Deregulated in Papillary Thyroid Carcinoma (Ptc). J. Clin. Endocrinol. Metab. 2011, 96, E546–E553. [Google Scholar] [CrossRef]
- Yu, Z.; Han, Y.; Shen, R.; Huang, K.; Xu, Y.-Y.; Wang, Q.-N.; Zhou, S.-S.; Xu, D.-X.; Tao, F.-B. Gestational Di-(2-Ethylhexyl) Phthalate Exposure Causes Fetal Intrauterine Growth Restriction through Disturbing Placental Thyroid Hormone Receptor Signaling. Toxicol. Lett. 2018, 294, 1–10. [Google Scholar] [CrossRef]
- Berlin, M.; Barchel, D.; Brik, A.; Kohn, E.; Livne, A.; Keidar, R.; Tovbin, J.; Betser, M.; Moskovich, M.; Mandel, D.; et al. Maternal and Newborn Thyroid Hormone, and the Association with Polychlorinated Biphenyls (Pcbs) Burden: The Ehf (Environmental Health Fund) Birth Cohort. Front Pediatr. 2021, 9, 705395. [Google Scholar] [CrossRef]
- Wang, X.K.; Agarwal, M.; Parobchak, N.; Rosen, A.; Vetrano, A.M.; Srinivasan, A.; Wang, B.; Rosen, T. Mono-(2-Ethylhexyl) Phthalate Promotes Pro-Labor Gene Expression in the Human Placenta. PLoS ONE 2016, 11, e0147013. [Google Scholar] [CrossRef]
- Shoaito, H.; Petit, J.; Chissey, A.; Auzeil, N.; Guibourdenche, J.; Gil, S.; Laprévote, O.; Fournier, T.; Degrelle, S.A. The Role of Peroxisome Proliferator–Activated Receptor Gamma (Pparγ) in Mono(2-Ethylhexyl) Phthalate (Mehp)-Mediated Cytotrophoblast Differentiation. Environ. Health Perspect. 2019, 127, 27003. [Google Scholar] [CrossRef] [PubMed]
- Petit, J.; Wakx, A.; Gil, S.; Fournier, T.; Auzeil, N.; Rat, P.; Laprévote, O. Lipidome-Wide Disturbances of Human Placental Jeg-3 Cells by the Presence of Mehp. Biochimie 2018, 149, 1–8. [Google Scholar] [CrossRef] [PubMed]
- De Felice, B.; Manfellotto, F.; Palumbo, A.R.; Troisi, J.; Zullo, F.; Di Carlo, C.; Sardo, A.D.S.; De Stefano, N.; Ferbo, U.; Guida, M.; et al. Genome-Wide Microrna Expression Profiling in Placentas from Pregnant Women Exposed to Bpa. BMC Med. Genom. 2015, 8, 56. [Google Scholar] [CrossRef]
- Vizcaino, E.; Grimalt, J.O.; Fernández-Somoano, A.; Tardon, A. Transport of Persistent Organic Pollutants across the Human Placenta. Environ. Int. 2014, 65, 107–115. [Google Scholar] [CrossRef] [PubMed]
- Cariou, R.; Veyrand, B.; Yamada, A.; Berrebi, A.; Zalko, D.; Durand, S.; Pollono, C.; Marchand, P.; Leblanc, J.-C.; Antignac, J.-P.; et al. Perfluoroalkyl Acid (Pfaa) Levels and Profiles in Breast Milk, Maternal and Cord Serum of French Women and Their Newborns. Environ. Int. 2015, 84, 71–81. [Google Scholar] [CrossRef]
- Mørck, T.J.; Sorda, G.; Bechi, N.; Rasmussen, B.S.; Nielsen, J.B.; Ietta, F.; Rytting, E.; Mathiesen, L.; Paulesu, L.; Knudsen, L.E. Placental Transport and in Vitro Effects of Bisphenol A. Reprod. Toxicol. 2010, 30, 131–137. [Google Scholar] [CrossRef]
- Stefanidou, M.; Maravelias, C.; Spiliopoulou, C. Human Exposure to Endocrine Disruptors and Breast Milk. Endocr. Metab. Immune Disord Drug Targets 2009, 9, 269–276. [Google Scholar] [CrossRef]
- Chang, G.; Mouillet, J.-F.; Mishima, T.; Chu, T.; Sadovsky, E.; Coyne, C.B.; Parks, W.T.; Surti, U.; Sadovsky, Y. Expression and Trafficking of Placental Micrornas at the Feto-Maternal Interface. FASEB J. 2017, 31, 2760–2770. [Google Scholar] [CrossRef]
- Tsitsiou, E.; Lindsay, M.A. Micrornas and the Immune Response. Curr. Opin Pharm. 2009, 9, 514–520. [Google Scholar] [CrossRef]
- Lorente-Cebrián, S.; Mejhert, N.; Kulyté, A.; Laurencikiene, J.; Åström, G.; Hedén, P.; Ryden, M.; Arner, P. Micrornas Regulate Human Adipocyte Lipolysis: Effects of Mir-145 Are Linked to Tnf-A. PLoS ONE 2014, 9, e86800. [Google Scholar] [CrossRef]
- Tili, E.; Michaille, J.J.; Cimino, A.; Costinean, S.; Dumitru, C.D.; Adair, B.; Fabbri, M.; Alder, H.; Liu, C.G.; Calin, G.A.; et al. Modulation of Mir-155 and Mir-125b Levels Following Lipopolysaccharide/Tnf-Alpha Stimulation and Their Possible Roles in Regulating the Response to Endotoxin Shock. J. Immunol. 2007, 179, 5082–5089. [Google Scholar] [CrossRef] [PubMed]
- Imaizumi, T.; Tanaka, H.; Tajima, A.; Yokono, Y.; Matsumiya, T.; Yoshida, H.; Tsuruga, K.; Aizawa-Yashiro, T.; Hayakari, R.; Inoue, I.; et al. Ifn-Γ and Tnf-A Synergistically Induce Microrna-155 Which Regulates Tab2/Ip-10 Expression in Human Mesangial Cells. Am. J. Nephrol. 2010, 32, 462–468. [Google Scholar] [CrossRef]
- Xie, Y.F.; Shu, R.; Jiang, S.Y.; Liu, D.L.; Ni, J.; Zhang, X.L. Microrna-146 Inhibits Pro-Inflammatory Cytokine Secretion through Il-1 Receptor-Associated Kinase 1 in Human Gingival Fibroblasts. J. Inflamm. 2013, 10, 20. [Google Scholar] [CrossRef] [PubMed]
- Doberstein, K.; Steinmeyer, N.; Hartmetz, A.-K.; Eberhardt, W.; Mittelbronn, M.; Harter, P.N.; Juengel, E.; Blaheta, R.; Pfeilschifter, J.; Gutwein, P. Microrna-145 Targets the Metalloprotease Adam17 and Is Suppressed in Renal Cell Carcinoma Patients. Neoplasia 2013, 15, 218–230. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.C.; Zou, X.B.; Chai, Y.F.; Yao, Y.M. Macrophage Polarization in Inflammatory Diseases. Int. J. Biol. Sci. 2014, 10, 520–529. [Google Scholar] [CrossRef] [PubMed]
- Essandoh, K.; Li, Y.; Huo, J.; Fan, G.C. Mirna-Mediated Macrophage Polarization and Its Potential Role in the Regulation of Inflammatory Response. Shock 2016, 46, 122–131. [Google Scholar] [CrossRef]
- Curtale, G.; Mirolo, M.; Renzi, T.A.; Rossato, M.; Bazzoni, F.; Locati, M. Negative Regulation of Toll-Like Receptor 4 Signaling by Il-10-Dependent Microrna-146b. Proc. Natl. Acad. Sci. USA 2013, 110, 11499–11504. [Google Scholar] [CrossRef]
- Ban, E.; Jeong, S.; Park, M.; Kwon, H.; Park, J.; Song, E.J.; Kim, A. Accelerated Wound Healing in Diabetic Mice by Mirna-497 and Its Anti-Inflammatory Activity. Biomed. Pharm. 2020, 121, 109613. [Google Scholar] [CrossRef]
- Gordon, S.; Taylor, P.R. Monocyte and Macrophage Heterogeneity. Nat. Rev. Immunol. 2005, 5, 953–964. [Google Scholar] [CrossRef]
- Mantovani, A.; Biswas, S.K.; Galdiero, M.R.; Sica, A.; Locati, M. Macrophage Plasticity and Polarization in Tissue Repair and Remodelling. J. Pathol. 2013, 229, 176–185. [Google Scholar] [CrossRef]
- Ginhoux, F.; Greter, M.; Leboeuf, M.; Nandi, S.; See, P.; Gokhan, S.; Mehler, M.F.; Conway, S.J.; Ng, L.G.; Stanley, E.R.; et al. Fate Mapping Analysis Reveals That Adult Microglia Derive from Primitive Macrophages. Science 2010, 330, 841–845. [Google Scholar] [CrossRef] [PubMed]
- Hoeffel, G.; Wang, Y.; Greter, M.; See, P.; Teo, P.; Malleret, B.; Leboeuf, M.; Low, D.; Oller, G.; Almeida, F.; et al. Adult Langerhans Cells Derive Predominantly from Embryonic Fetal Liver Monocytes with a Minor Contribution of Yolk Sac–Derived Macrophages. J. Exp. Med. 2012, 209, 1167–1181. [Google Scholar] [CrossRef] [PubMed]
- Luinenburg, D.G.; De Haan, G. Micrornas in Hematopoietic Stem Cell Aging. Mech. Ageing Dev. 2020, 189, 111281. [Google Scholar] [CrossRef] [PubMed]
- Lazare, S.S.; Wojtowicz, E.E.; Bystrykh, L.V.; De Haan, G. Micrornas in Hematopoiesis. Exp. Cell. Res. 2014, 329, 234–238. [Google Scholar] [CrossRef] [PubMed]
- Gerrits, A.; Walasek, M.A.; Olthof, S.; Weersing, E.; Ritsema, M.; Zwart, E.; Van Os, R.; Bystrykh, L.V.; de Haan, G. Genetic Screen Identifies Microrna Cluster 99b/Let-7e/125a as a Regulator of Primitive Hematopoietic Cells. Blood 2012, 119, 377–387. [Google Scholar] [CrossRef]
- Lechman, E.R.; Gentner, B.; van Galen, P.; Giustacchini, A.; Saini, M.; Boccalatte, F.E.; Hiramatsu, H.; Restuccia, U.; Bachi, A.; Voisin, V.; et al. Attenuation of Mir-126 Activity Expands Hsc in Vivo without Exhaustion. Cell. Stem. Cell. 2012, 11, 799–811. [Google Scholar] [CrossRef]
- Lechman, E.R.; Gentner, B.; Ng, S.W.; Schoof, E.M.; van Galen, P.; Kennedy, J.A.; Nucera, S.; Ciceri, F.; Kaufmann, K.B.; Takayama, N.; et al. Mir-126 Regulates Distinct Self-Renewal Outcomes in Normal and Malignant Hematopoietic Stem Cells. Cancer Cell 2016, 29, 214–228. [Google Scholar] [CrossRef]
- Starczynowski, D.T.; Kuchenbauer, F.; Argiropoulos, B.; Sung, S.; Morin, R.; Muranyi, A.; Hirst, M.; Hogge, D.; Marra, M.; Wells, R.A.; et al. Identification of Mir-145 and Mir-146a as Mediators of the 5q- Syndrome Phenotype. Nat. Med. 2010, 16, 49–58. [Google Scholar] [CrossRef]
- Guo, S.; Lu, J.; Schlanger, R.; Zhang, H.; Wang, J.Y.; Fox, M.C.; Purton, L.E.; Fleming, H.H.; Cobb, B.; Merkenschlager, M.; et al. Microrna Mir-125a Controls Hematopoietic Stem Cell Number. Proc. Natl. Acad. Sci. USA 2010, 107, 14229–14234. [Google Scholar] [CrossRef]
- Bowers, E.; Singer, K. Obesity-Induced Inflammation: The Impact of the Hematopoietic Stem Cell Niche. JCI Insight. 2021, 6, e145295. [Google Scholar] [CrossRef]
- Vinci, M.C.; Gambini, E.; Bassetti, B.; Genovese, S.; Pompilio, G. When Good Guys Turn Bad: Bone Marrow’s and Hematopoietic Stem Cells’ Role in the Pathobiology of Diabetic Complications. Int. J. Mol. Sci. 2020, 21, 3864. [Google Scholar] [CrossRef] [PubMed]
- Pospisil, V.; Vargova, K.S.; Kokavec, J.; Rybarova, J.; Savvulidi, F.G.; Jonasova, A.; Nečas, E.; Zavadil, J.; Laslo, P.; Stopka, T. Epigenetic Silencing of the Oncogenic Mir-17-92 Cluster During Pu.1-Directed Macrophage Differentiation. Embo. J. 2011, 30, 4450–4464. [Google Scholar] [CrossRef] [PubMed]
- Berg, S.M.; Seijkens, T.T.P.; Kusters, P.J.H.; Beckers, L.; Toom, M.; Smeets, E.; Levels, J.; Winther, M.P.J.; Lutgens, E. Diet-Induced Obesity in Mice Diminishes Hematopoietic Stem and Progenitor Cells in the Bone Marrow. Faseb. J. 2016, 30, 1779–1788. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.-M.; Govindarajah, V.; Goddard, B.; Hinge, A.; Muench, D.E.; Filippi, M.-D.; Aronow, B.; Cancelas, J.A.; Salomonis, N.; Grimes, H.L.; et al. Obesity Alters the Long-Term Fitness of the Hematopoietic Stem Cell Compartment through Modulation of Gfi1 Expression. J. Exp. Med. 2018, 215, 627–644. [Google Scholar] [CrossRef] [PubMed]
- Wynn, T.A.; Chawla, A.; Pollard, J.W. Macrophage Biology in Development, Homeostasis and Disease. Nature 2013, 496, 445–455. [Google Scholar] [CrossRef]
- Caescu, C.I.; Guo, X.; Tesfa, L.; Bhagat, T.D.; Verma, A.; Zheng, D.; Stanley, E.R. Colony Stimulating Factor-1 Receptor Signaling Networks Inhibit Mouse Macrophage Inflammatory Responses by Induction of Microrna-21. Blood 2015, 125, e1–e13. [Google Scholar] [CrossRef]
- Lafontan, M. Fat Cells: Afferent and Efferent Messages Define New Approaches to Treat Obesity. Annu. Rev. Pharmacol. Toxicol. 2005, 45, 119–146. [Google Scholar] [CrossRef]
- Longo, M.; Zatterale, F.; Naderi, J.; Parrillo, L.; Formisano, P.; Raciti, G.A.; Beguinot, F.; Miele, C. Adipose Tissue Dysfunction as Determinant of Obesity-Associated Metabolic Complications. Int. J. Mol. Sci. 2019, 20, 2538. [Google Scholar] [CrossRef]
- Yan, X.; Huang, Y.; Zhao, J.-X.; Rogers, C.J.; Zhu, M.-J.; Ford, S.P.; Nathanielsz, P.W.; Du, M. Maternal Obesity Downregulates Microrna Let-7g Expression, a Possible Mechanism for Enhanced Adipogenesis During Ovine Fetal Skeletal Muscle Development. Int. J. Obes. 2013, 37, 568–575. [Google Scholar] [CrossRef]
- Yasmin, F.; Sahito, A.M.; Mir, S.L.; Khatri, G.; Shaikh, S.; Gul, A.; Hassan, S.A.; Koritala, T.; Surani, S. Electrical Neuromodulation Therapy for Inflammatory Bowel Disease. World J. Gastrointest Pathophysiol. 2022, 13, 128–142. [Google Scholar] [CrossRef]
- Wang, J.; Long, R.; Han, Y. The Role of Exosomes in the Tumour Microenvironment on Macrophage Polarisation. Biochim. Biophys Acta. Rev. Cancer. 2022, 1877, 188811. [Google Scholar] [CrossRef] [PubMed]
- Ghafouri-Fard, S.; Niazi, V.; Taheri, M. Role of Mirnas and Lncrnas in Hematopoietic Stem Cell Differentiation. Noncoding RNA Res. 2021, 6, 8–14. [Google Scholar] [CrossRef] [PubMed]
- Chu, A.J.; Williams, J.M. Astrocytic Microrna in Ageing, Inflammation, and Neurodegenerative Disease. Front. Physiol. 2021, 12, 826697. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Ren, C.; Qu, X.; Wu, X.; Dong, F.; Chand, Y.K.; Fan, H.; Yao, R.; Geng, D. Mir-219 Attenuates Demyelination in Cuprizone-Induced Demyelinated Mice by Regulating Monocarboxylate Transporter 1. Eur. J. Neurosci. 2017, 45, 249–259. [Google Scholar] [CrossRef] [PubMed]
- Krug, A.; Wöhr, M.; Seffer, D.; Rippberger, H.; Sungur, A.; Dietsche, B.; Stein, F.; Sivalingam, S.; Forstner, A.J.; Witt, S.H.; et al. Advanced Paternal Age as a Risk Factor for Neurodevelopmental Disorders: A Translational Study. Mol. Autism. 2020, 11, 54. [Google Scholar] [CrossRef]
- Rodriguez-Gomez, A.; Filice, F.; Gotti, S.; Panzica, G. Perinatal Exposure to Genistein Affects the Normal Development of Anxiety and Aggressive Behaviors and Nitric Oxide System in Cd1 Male Mice. Physiol. Behav. 2014, 133, 107–114. [Google Scholar] [CrossRef]
- Marraudino, M.; Ponti, G.; Moussu, C.; Farinetti, A.; Macchi, E.; Accornero, P.; Gotti, S.; Collado, P.; Keller, M.; Panzica, G. Early Postnatal Genistein Administration Affects Mice Metabolism and Reproduction in a Sexually Dimorphic Way. Metabolites 2021, 11, 449. [Google Scholar] [CrossRef]
- Zhou, P.; Wu, S.; Huang, D.; Wang, K.; Su, X.; Yang, R.; Shao, C.; Wu, J. Oral Exposure to Dehp May Stimulate Prostatic Hyperplasia Associated with Upregulation of Cox-2 and L-Pgds Expressions in Male Adult Rats. Reprod. Toxicol. 2022, 112, 160–170. [Google Scholar] [CrossRef]
- Singh, V.; Cortes-Ramirez, J.; Toms, L.M.; Sooriyagoda, T.; Karatela, S. Effects of Polybrominated Diphenyl Ethers on Hormonal and Reproductive Health in E-Waste-Exposed Population: A Systematic Review. Int. J. Environ. Res. Public Health 2022, 19, 7820. [Google Scholar] [CrossRef]
- Chen, S.-D.; Sun, X.-Y.; Niu, W.; Kong, L.-M.; He, M.-J.; Fan, H.-M.; Li, W.-S.; Zhong, A.-F.; Zhang, L.-Y.; Lu, J. Correlation between the Level of Microrna Expression in Peripheral Blood Mononuclear Cells and Symptomatology in Patients with Generalized Anxiety Disorder. Compr. Psychiatry 2016, 69, 216–224. [Google Scholar] [CrossRef]
| EDCs | Primary Sources | MiRs | Role in Immunity | Role in Metabolic Health |
|---|---|---|---|---|
| Bisphenol-A | Industrial plasticizer | miR-146a | Inflammatory response and cell growth [89]; Inhibit inflammatory cytokines expression [90,91,92]; Inhibit inflammatory response through TLR-signaling [93] | Inhibit adipogenesis [94]; Protect β cell function [95]; Improve glucose metabolism and inhibit hepatic steatosis [96] |
| miR-21 | Tumor growth [97]; Promote liver inflammation [98,99] | Promote hepatic steatosis [99]; Inhibit triglyceride and cholesterol synthesis [100]; Promote adipose tissue browning and thermogenesis [101] | ||
| miR-148 | Inhibit IL-1β expression [102]; Improve immune dysfunction [103]; | Inhibit proliferation and promote apoptosis [104]; Promote insulin expression [105] | ||
| Dioxin | Combustion | miR-132 | Decrease IL-17, IFN-γ expression and suppress T-cell proliferation [106]; Enhance cholinergic anti-inflammatory reaction [107]; Upregulate NF-κB and STAT3 activity [107] | Promote β cell proliferation [108]; Inhibit neural tau expression [109]; Promote angiogenesis and endothelial proliferation [110] |
| Polychlorinated biphenyls | Dietary intake | miR-191 | Inhibit inflammation by targeting DAPK1 [111]; Suppress inflammatory response [112] | Tumor growth [113]; Inhibit angiogenesis [114,115]; Induce DNA damage [116]; Block GLUT4 translocation and induce insulin resistance [117] |
| miR-155 | Regulate innate immunity [118,119]; Induce intestinal inflammation [120]; Inhibit the production of Th1-type cytokines [121]; Promote macrophage Inflammatory response [122,123,124]; | Reduce oxidative stress and cell migration [125,126]; Promote adipocyte lipid accumulation [127]; Promote liver fibrosis [122] | ||
| Diethylstilbestrol | Birth control pills | miR-30 | Promote M2 polarization [128]; Inhibit inflammatory response [129,130,131] Suppress humoral immune response induced by lipopolysaccharide [132] | Peripheral insulin sensitivity [133]; Promote apoptosis [134,135]; Promote adipocyte browning [136]; Inhibit adipocyte inflammation [137]; Regulate the intracellular lipid metabolism [138]; Regulate autophagy in thymocytes [139]; |
| Methylparaben | Personal care products | miR-373 | Targets the tumor-suppressor LATS2 and neutralize p53-mediated CDK inhibition [140,141]; Promote inflammatory response [142] | Inhibit proliferation and induce apoptosis [142,143,144]; Inhibit autophagy [145]; Inhibit liver steatosis [146] |
| Phthalate | Food sources | miR-34a | Regulate T cell function [147]; Promote inflammatory response [148,149,150] | Alter NAD levels and sirtuins activity Promote adipogenesis [151]; Inhibit glycolysis [152,153]; Promote mitochondrial respiration [152] |
| miR-16 | Inhibit B cell chemotaxis [154]; Promote inflammatory response [155]; Inhibit inflammatory response [156,157,158,159] | Promote apoptosis [27,160]; | ||
| miR-126 | Inhibit T cell differentiation [161,162]; Promote inflammatory response [163] | Promote oxidative stress [28] Promote autophagy [164,165]; Promote lipid accumulation [164]; | ||
| miR-17 | Inhibit inflammatory response [166,167,168]; Regulate B cell development [169,170] | Promote adipocyte browning [171]; Enhance insulin sensitivity [172]; Promote angiogenesis [173] | ||
| miR-200c | Inhibit inflammatory response [174]; Promote inflammatory response [175,176] | Promote lipid accumulation [177]; Promote lipolysis [178]; Inhibit proliferation [179]; Enhance insulin sensitivity [178] | ||
| Triclosan | Personal care products, Cosmetics | miR-10a | Promote inflammatory response [180,181]; Inhibit inflammatory cytokine secretion [182]; Suppress M1 polarization [183] | Inhibit proliferation [184,185]; Promote adipocyte browning [183]; Prevent β cell degeneration [186] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shree, N.; Ding, Z.; Flaws, J.; Choudhury, M. Role of microRNA in Endocrine Disruptor-Induced Immunomodulation of Metabolic Health. Metabolites 2022, 12, 1034. https://doi.org/10.3390/metabo12111034
Shree N, Ding Z, Flaws J, Choudhury M. Role of microRNA in Endocrine Disruptor-Induced Immunomodulation of Metabolic Health. Metabolites. 2022; 12(11):1034. https://doi.org/10.3390/metabo12111034
Chicago/Turabian StyleShree, Nitya, Zehuan Ding, Jodi Flaws, and Mahua Choudhury. 2022. "Role of microRNA in Endocrine Disruptor-Induced Immunomodulation of Metabolic Health" Metabolites 12, no. 11: 1034. https://doi.org/10.3390/metabo12111034
APA StyleShree, N., Ding, Z., Flaws, J., & Choudhury, M. (2022). Role of microRNA in Endocrine Disruptor-Induced Immunomodulation of Metabolic Health. Metabolites, 12(11), 1034. https://doi.org/10.3390/metabo12111034







