Metabolic Profile Characterization of Different Thyroid Nodules Using FTIR Spectroscopy: A Review
Abstract
1. Introduction
2. Thyroid Gland
3. Nodular Thyroid Pathology
3.1. Epidemiology and Etiology
3.2. Management and Prognosis
3.3. Diagnosis
4. New Diagnostic Approach—Metabolomics Techniques
FTIR Spectroscopy and Thyroid Cancer
5. Conclusions
Funding
Conflicts of Interest
References
- Nguyen, Q.T.; Lee, E.J.; Huang, M.G.; Park, Y.I.; Khullar, A.; Plodkowski, R.A. Diagnosis and treatment of patients with thyroid cancer. Am. Health Drug Benefits 2015, 8, 30–40. [Google Scholar]
- Bailey, S.; Wallwork, B. Differentiating between benign and malignant thyroid nodules. Aust. J. Gen. Pract. 2018, 47, 770–774. [Google Scholar] [CrossRef]
- Chen, D.; Hu, J.; Zhu, M.; Tang, N.; Yang, Y.; Feng, Y. Diagnosis of thyroid nodules for ultrasonographic characteristics indicative of malignancy using random forest. BioData Min. 2020, 13, 1–21. [Google Scholar] [CrossRef]
- Institute for Quality and Efficiency in Health Care, How Does the Thyroid Gland Work? Institute for Quality and Efficiency in Health Care (IQWiG): Köln, Germany, 2018.
- Gardner, D.G.; Shoback, D. Greenspan’s Basic & Clinical Endocrinology, 9th ed.; McGraw Hill Education: New York, NY, USA, 2011. [Google Scholar]
- Geetha, K.; Baboo, C.S.S. An Empirical Model for Thyroid Disease Classification using Evolutionary Multivariate Bayseian Prediction Method. Glob. J. Comput. Sci. Technol. E Netw. Web Secur. 2016, 16, 1–10. [Google Scholar]
- Mounika, B.; Brahmaiah, B.; Ramesh, R.; Bravaneswari, K.; Lakshmi, T.A.; Nama, S. Review on thyroid disorders. Int. J. Pharm. Res. Bio-Sci. 2013, 2, 197–214. [Google Scholar]
- Cooper, D.S.; Doherty, G.M.; Haugen, B.R.; Kloos, R.T.; Lee, S.L.; Mandel, S.J.; Mazzaferri, E.L.; McIver, B.; Pacini, F.; Schlumberger, M.; et al. Revised American thyroid association management guidelines for patients with thyroid nodules and differentiated thyroid cancer. Thyroid 2009, 19, 1167–1214. [Google Scholar] [CrossRef] [PubMed]
- Durante, C.; Costante, G.; Lucisano, G.; Bruno, R.; Meringolo, D.; Paciaroni, A.; Puxeddu, E.; Torlontano, M.; Tumino, S.; Attard, M.; et al. The natural history of benign thyroid nodules. JAMA–J. Am. Med. Assoc. 2015, 313, 926–935. [Google Scholar] [CrossRef]
- Kwong, N.; Medici, M.; Angell, T.E.; Liu, X.; Marqusee, E.; Cibas, E.S.; Krane, J.F.; Barletta, J.A.; Kim, M.I.; Larsen, P.R.; et al. The influence of patient age on thyroid nodule formation, multinodularity, and thyroid cancer risk. J. Clin. Endocrinol. Metab. 2015, 100, 4434–4440. [Google Scholar] [CrossRef]
- Guth, S.; Theune, U.; Aberle, J.; Galach, A.; Bamberger, C.M. Very high prevalence of thyroid nodules detected by high frequency (13 MHz) ultrasound examination. Eur. J. Clin. Investg. 2009, 39, 699–706. [Google Scholar] [CrossRef]
- Durante, C.; Grani, G.; Lamartina, L.; Filetti, S.; Mandel, S.J.; Cooper, D.S. The diagnosis and management of thyroid nodules a review. JAMA–J. Am. Med. Assoc. 2018, 319, 919–924. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.S.; Nam, C.M.; Kim, E.K.; Moon, H.J.; Han, K.H.; Kwak, J.Y. Evaluation of serum thyroid-stimulating hormone as indicator for fine-needle aspiration in patients with thyroid nodules. Head Neck 2015, 37, 498–504. [Google Scholar] [CrossRef]
- Xu, W.; Huo, L.; Chen, Z.; Huang, Y.; Jin, X.; Deng, J.; Zhu, S.; Yu, Y. The relationship of TPOab and TGAb with risk of thyroid nodules: A large epidemiological study. Int. J. Environ. Res. Public Health 2017, 14, 723. [Google Scholar] [CrossRef]
- Panagiotou, G.; Komninou, D.; Anagnostis, P.; Linardos, G.; Karoglou, E.; Somali, M.; Duntas, L.; Kita, M.; Tziomalos, K.; Pazaitou-Panayiotou, K. Association between lifestyle and anthropometric parameters and thyroid nodule features. Endocrine 2017, 56, 560–567. [Google Scholar] [CrossRef]
- Shin, J.; Kim, M.H.; Yoon, K.H.; Kang, M.-I.; Cha, B.Y.; Lim, D.J. Relationship between metabolic syndrome and thyroid nodules in healthy Koreans. Korean J. Intern. Med. 2016, 31, 98–105. [Google Scholar] [CrossRef]
- Haugen, B.R.; Alexander, E.K.; Bible, K.C.; Doherty, G.M.; Mandel, S.J.; Nikiforov, Y.E.; Pacini, F.; Randolph, G.W.; Sawka, A.M.; Schlumberger, M.; et al. 2015 American Thyroid Association Management Guidelines for Adult Patients with Thyroid Nodules and Differentiated Thyroid Cancer: The American Thyroid Association Guidelines Task Force on Thyroid Nodules and Differentiated Thyroid Cancer. Thyroid 2016, 26, 1–133. [Google Scholar] [CrossRef] [PubMed]
- Kumar, V.; Abbas, A.K.; Aster, J.C. Robbins Basic Pathology, 10th ed.; Elsevier–Health Sciences Division, Ed.; Elsevier: Amsterdam, The Netherlands, 2017. [Google Scholar]
- Popoveniuc, G.; Jonklaas, J. Thyroid Nodules. Med. Clin. North Am. 2012, 96, 329–349. [Google Scholar] [CrossRef]
- Erdogan, M.F.; Gursoy, A.; Erdogan, G. Natural course of benign thyroid nodules in a moderately iodine-deficient area. Clin. Endocrinol. 2006, 65, 767–771. [Google Scholar] [CrossRef]
- Bible, K.C.; Kebebew, E.; Brierley, J.; Brito, J.P.; Cabanillas, M.E.; Clark, T.J.; Di Cristofano, A.; Foote, R.; Giordano, T.; Kasperbauer, J.; et al. 2021 American Thyroid Association Guidelines for Management of Patients with Anaplastic Thyroid Cancer. Thyroid 2021, 31, 337–386. [Google Scholar] [CrossRef] [PubMed]
- Cabanillas, M.E.; McFadden, D.G.; Durante, C. Thyroid cancer. Lancet 2016, 388, 2783–2795. [Google Scholar] [CrossRef]
- Papaleontiou, M.; Hughes, D.T.; Guo, C.; Banerjee, M.; Haymart, M.R. Population-based assessment of complications following surgery for thyroid cancer. J. Clin. Endocrinol. Metab. 2017, 102, 2543–2551. [Google Scholar] [CrossRef] [PubMed]
- Filho, J.G.; Kowalski, L.P. Postoperative complications of thyroidectomy for differentiated thyroid carcinoma. Am. J. Otolaryngol.–Head Neck Med. Surg. 2004, 25, 225–230. [Google Scholar] [CrossRef] [PubMed]
- Verloop, H.; Louwerens, M.; Schoones, J.W.; Kievit, J.; Smit, J.W.A.; Dekkers, O.M. Risk of hypothyroidism following hemithyroidectomy: Systematic review and meta-analysis of prognostic studies. J. Clin. Endocrinol. Metab. 2012, 97, 2243–2255. [Google Scholar] [CrossRef]
- McCaffrey, T.V.; Bergstralh, E.J.; Hay, I.D. Locally invasive papillary thyroid carcinoma: 1940–1990. Head Neck 1994, 16, 165–172. [Google Scholar] [CrossRef] [PubMed]
- Adair, C. Non-Neoplastic Lesions of the Thyroid Gland. In Head and Neck Pathology; Thompson, L.D.R., Goldblum, J.R., Eds.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 498–545. [Google Scholar]
- Lloyd, R.V.; Osamura, Y.R.; Kloppel, G.; Rosai, J. WHO Classification of Tumours of Endocrine Organs, 4th ed.; Lloyd, R.V., Osamura, R.Y., Klöppel, G., Rosai, J., Eds.; International Agency for Research on Cancer (IARC): Lyon, France, 2017. [Google Scholar]
- Benbassat, C.A.; Mechlis-Frish, S.; Hirsch, D. Clinicopathological characteristics and long-term outcome in patients with distant metastases from differentiated thyroid cancer. World J. Surg. 2006, 30, 1088–1095. [Google Scholar] [CrossRef]
- National Cancer Institute. Thyroid Cancer–Cancer Stat Facts. Available online: https://seer.cancer.gov/statfacts/html/thyro.html?fbclid=IwAR0dkaKUSnRAXHiJSiZV6lzmVO3nijA1Y1pSjYDeMzqLjFkGoqBhZEu9QaM (accessed on 25 November 2021).
- Davies, L.; Welch, H.G. Current thyroid cancer trends in the United States. JAMA Otolaryngol.—Head Neck Surg. 2014, 140, 317–322. [Google Scholar] [CrossRef] [PubMed]
- Davies, L.; Welch, H.G. Increasing incidence of thyroid cancer in the United States, 1973–2002. J. Am. Med. Assoc. 2006, 295, 2164–2167. [Google Scholar] [CrossRef]
- Colonna, M.; Guizard, A.V.; Schvartz, C.; Velten, M.; Raverdy, N.; Molinie, F.; Delafosse, P.; Franc, B.; Grosclaude, P. A time trend analysis of papillary and follicular cancers as a function of tumour size: A study of data from six cancer registries in France (1983–2000). Eur. J. Cancer 2007, 43, 891–900. [Google Scholar] [CrossRef]
- Roti, E.; Rossi, R.; Trasforini, G.; Bertelli, F.; Ambrosio, M.R.; Busutti, L.; Pearce, E.N.; Braverman, L.E.; Degli Uberti, E.C. Clinical and histological characteristics of papillary thyroid microcarcinoma: Results of a retrospective study in 243 patients. J. Clin. Endocrinol. Metab. 2006, 91, 2171–2178. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Maso, L.D.; Vaccarella, S. Global trends in thyroid cancer incidence and the impact of overdiagnosis. Lancet Diabetes Endocrinol. 2020, 8, 468–470. [Google Scholar] [CrossRef]
- Hedman, C.; Strang, P.; Djärv, T.; Widberg, I.; Lundgren, C.I. Anxiety and Fear of Recurrence Despite a Good Prognosis: An Interview Study with Differentiated Thyroid Cancer Patients. Thyroid 2017, 27, 1417–1423. [Google Scholar] [CrossRef] [PubMed]
- Tamhane, S.; Gharib, H. Thyroid nodule update on diagnosis and management. Clin. Diabetes Endocrinol. 2016, 2, 1–10. [Google Scholar] [CrossRef]
- Russ, G.; Bonnema, S.J.; Erdogan, M.F.; Durante, C.; Ngu, R.; Leenhardt, L. European Thyroid Association Guidelines for Ultrasound Malignancy Risk Stratification of Thyroid Nodules in Adults: The EU-TIRADS. Eur. Thyroid J. 2017, 6, 225–237. [Google Scholar] [CrossRef] [PubMed]
- Tessler, F.N.; Middleton, W.D.; Grant, E.G.; Hoang, J.K.; Berland, L.L.; Teefey, S.A.; Cronan, J.J.; Beland, M.D.; Desser, T.S.; Frates, M.C.; et al. ACR Thyroid Imaging, Reporting and Data System (TI-RADS): White Paper of the ACR TI-RADS Committee. J. Am. Coll. Radiol. 2017, 14, 587–595. [Google Scholar] [CrossRef] [PubMed]
- Paschke, R.; Cantara, S.; Crescenzi, A.; Jarzab, B.; Musholt, T.J.; Sobrinho Simoes, M. European Thyroid Association Guidelines regarding Thyroid Nodule Molecular Fine-Needle Aspiration Cytology Diagnostics. Eur. Thyroid J. 2017, 6, 115–129. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.C.C.; Friedman, L.; Kennedy, G.C.; Wang, H.; Kebebew, E.; Steward, D.L.; Zeiger, M.A.; Westra, W.H.; Wang, Y.; Khanafshar, E.; et al. A large multicenter correlation study of thyroid nodule cytopathology and histopathology. Thyroid 2011, 21, 243–251. [Google Scholar] [CrossRef]
- Navas-Carrillo, D.; Rodriguez, J.M.; Montoro-García, S.; Orenes-Piñero, E. High-resolution proteomics and metabolomics in thyroid cancer: Deciphering novel biomarkers. Crit. Rev. Clin. Lab. Sci. 2017, 54, 446–457. [Google Scholar] [CrossRef]
- Wojakowska, A.; Chekan, M.; Widlak, P.; Pietrowska, M. Application of metabolomics in thyroid cancer research. Int. J. Endocrinol. 2015, 2015, 1–13. [Google Scholar] [CrossRef]
- Abooshahab, R.; Gholami, M.; Sanoie, M.; Azizi, F.; Hedayati, M. Advances in metabolomics of thyroid cancer diagnosis and metabolic regulation. Endocrine 2019, 65, 1–14. [Google Scholar] [CrossRef]
- Sussulini, A. Erratum to: Chapters 1 and 11 of Metabolomics: From Fundamentals to Clinical Applications. Adv. Exp. Med. Biol. 2017, 965, E1–E2. [Google Scholar] [CrossRef]
- Rocha, A.; Magalhães, S.; Nunes, A. Cell Culture Studies: A Promising Approach to the Metabolomic Study of Human Aging. Curr. Metab. Syst. Biol. 2021, 8, 1–26. [Google Scholar] [CrossRef]
- Ellis, D.I.; Dunn, W.B.; Griffin, J.L.; Allwood, J.W.; Goodacre, R. Metabolic fingerprinting as a diagnostic tool. Pharmacogenomics 2007, 8, 1243–1266. [Google Scholar] [CrossRef]
- Singh, A.; Ham, J.; Po, J.W.; Niles, N.; Roberts, T.; Lee, C.S. The Genomic Landscape of Thyroid Cancer Tumourigenesis and Implications for Immunotherapy. Cells 2021, 10, 1082. [Google Scholar] [CrossRef]
- Yoo, S.K.; Song, Y.S.; Lee, E.K.; Hwang, J.; Kim, H.H.; Jung, G.; Kim, Y.A.; Kim, S.J.; Cho, S.W.; Won, J.K.; et al. Integrative analysis of genomic and transcriptomic characteristics associated with progression of aggressive thyroid cancer. Nat. Commun. 2019, 10, 1–12. [Google Scholar] [CrossRef] [PubMed]
- García-Vence, M.; Chantada-Vázquez, M.D.P.; Cameselle-Teijeiro, J.M.; Bravo, S.B.; Núñez, C. A Novel Nanoproteomic Approach for the Identification of Molecular Targets Associated with Thyroid Tumors. Nanomaterials 2020, 10, 2370. [Google Scholar] [CrossRef] [PubMed]
- Dai, J.; Yu, X.; Han, Y.; Chai, L.; Liao, Y.; Zhong, P.; Xie, R.; Sun, X.; Huang, Q.; Wang, J.; et al. TMT-labeling proteomics of papillary thyroid carcinoma reveal invasive biomarkers. J. Cancer 2020, 11, 6122–6132. [Google Scholar] [CrossRef]
- Pu, W.; Shi, X.; Yu, P.; Zhang, M.; Liu, Z.; Tan, L.; Han, P.; Wang, Y.; Ji, D.; Gan, H.; et al. Single-cell transcriptomic analysis of the tumor ecosystems underlying initiation and progression of papillary thyroid carcinoma. Nat. Commun. 2021, 12, 1–18. [Google Scholar] [CrossRef]
- Coelho, M.; Raposo, L.; Goodfellow, B.J.; Atzori, L.; Jones, J.; Manadas, B. The potential of metabolomics in the diagnosis of thyroid cancer. Int. J. Mol. Sci. 2020, 21, 5272. [Google Scholar] [CrossRef] [PubMed]
- Du, Y.; Fan, P.; Zou, L.; Jiang, Y.; Gu, X.; Yu, J.; Zhang, C. Serum Metabolomics Study of Papillary Thyroid Carcinoma Based on HPLC-Q-TOF-MS/MS. Front. Cell Dev. Biol. 2021, 9, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Abooshahab, R.; Hooshmand, K.; Razavi, S.A.; Gholami, M.; Sanoie, M.; Hedayati, M. Plasma Metabolic Profiling of Human Thyroid Nodules by Gas Chromatography-Mass Spectrometry (GC-MS)-Based Untargeted Metabolomics. Front. Cell Dev. Biol. 2020, 8, 1–13. [Google Scholar] [CrossRef]
- Rezig, L.; Servadio, A.; Torregrossa, L.; Miccoli, P.; Basolo, F.; Shintu, L.; Caldarelli, S. Diagnosis of post-surgical fine-needle aspiration biopsies of thyroid lesions with indeterminate cytology using HRMAS NMR-based metabolomics. Metabolomics 2018, 14, 1–10. [Google Scholar] [CrossRef]
- Wang, T.; Sun, Z.; Wang, Y.; Li, F.; Zhou, X.; Tian, X.; Wang, S. Diagnosis of papillary thyroid carcinoma by 1H NMR spectroscopy-based metabolomic analysis of whole blood. Drug Discov. Ther. 2020, 14, 187–196. [Google Scholar] [CrossRef]
- Sbroscia, M.; Di Gioacchino, M.; Ascenzi, P.; Crucitti, P.; di Masi, A.; Giovannoni, I.; Longo, F.; Mariotti, D.; Naciu, A.M.; Palermo, A.; et al. Thyroid cancer diagnosis by Raman spectroscopy. Sci. Rep. 2020, 10, 1–10. [Google Scholar] [CrossRef]
- Liang, X.; Miao, X.; Xiao, W.; Ye, Q.; Wang, S.; Lin, J.; Li, C.; Huang, Z. Filter-membrane-based ultrafiltration coupled with surface-enhanced raman spectroscopy for potential differentiation of benign and malignant thyroid tumors from blood plasma. Int. J. Nanomed. 2020, 15, 2303–2314. [Google Scholar] [CrossRef]
- O’Dea, D.; Bongiovanni, M.; Sykiotis, G.P.; Ziros, P.G.; Meade, A.D.; Lyng, F.M.; Malkin, A. Raman spectroscopy for the preoperative diagnosis of thyroid cancer and its subtypes: An in vitro proof-of-concept study. Cytopathology 2019, 30, 51–60. [Google Scholar] [CrossRef] [PubMed]
- Depciuch, J.; Stanek-Widera, A.; Skrzypiec, D.; Lange, D.; Biskup-Frużyńska, M.; Kiper, K.; Stanek-Tarkowska, J.; Kula, M.; Cebulski, J. Spectroscopic identification of benign (follicular adenoma) and cancerous lesions (follicular thyroid carcinoma) in thyroid tissues. J. Pharm. Biomed. Anal. 2019, 170, 321–326. [Google Scholar] [CrossRef] [PubMed]
- Liu, K.Z.; Schultz, C.P.; Salamon, E.A.; Man, A.; Mantsch, H.H. Infrared spectroscopic diagnosis of thyroid tumors. J. Mol. Struct. 2003, 661–662, 397–404. [Google Scholar] [CrossRef]
- Zhang, W.; Tian, P.; Zhu, Q.; Zhang, Y.; Cui, L.; Xu, Z. Noninvasive surface detection of papillary thyroid carcinoma by Fourier transform infrared spectroscopy. Chem. Res. Chin.Univ. 2015, 31, 198–202. [Google Scholar] [CrossRef]
- Liu, Y.-Q.; Xu, Y.-Z.; Sun, Q.-G.; Zhang, X.-Q.; Xu, Z.; Zhang, Y.-F.; Wu, J.-Q.; Zhou, X.-S.; Ling, X.-F. [Surface detection of thyroid cancer by Fourier transform infrared spectroscopy]. Zhonghua Zhong Liu Za Zhi 2009, 31, 908–910. [Google Scholar]
- Ling, X.F.; Xu, Z.; Xu, Y.Z.; Zhou, S.; Zhang, N.W.; Wang, L.X.; Hou, C.S.; Zhang, Y.F.; Zhou, X.S.; Wu, J.G. Nodular goiter surface detection by FTIR spectroscopy. Guang Pu Xue Yu Guang Pu Fen Xi 2005, 25, 1955–1958. [Google Scholar]
- Liu, Y.Q.; Gao, M.J.; Xu, Y.Z.; Sun, Q.G.; Zhang, X.Q.; Xu, Z.; Zhang, Y.F.; Wu, J.G.; Zhou, X.S.; Ling, X.F. Investigation on fourier transform infrared spectrum of metastatic lymph node in thyroid cancer. Guang Pu Xue Yu Guang Pu Fen Xi 2009, 29, 2917–2921. [Google Scholar] [CrossRef]
- Liu, Y.; Xu, Y.; Liu, Y.; Zhang, Y.; Wang, D.; Xiu, D.; Xu, Z.; Zhou, X.; Wu, J.; Ling, X. Detection of cervical metastatic lymph nodes in papillary thyroid carcinoma by Fourier transform infrared spectroscopy. Br. J. Surg. 2011, 98, 380–384. [Google Scholar] [CrossRef]
- Zhang, X.; Xu, Y.; Zhang, Y.; Wang, L.; Hou, C.; Zhou, X.; Ling, X.; Xu, Z. Intraoperative detection of thyroid carcinoma by fourier transform infrared spectrometry. J. Surg. Res. 2011, 171, 650–656. [Google Scholar] [CrossRef]
- da Silva, R.M.; Pupin, B.; Bhattacharjee, T.T.; Vamondes Kulcsar, M.A.; Uno, M.; Chammas, R.; de Azevedo Canevari, R. ATR-FTIR spectroscopy and CDKN1C gene expression in the prediction of lymph nodes metastases in papillary thyroid carcinoma. Spectrochim. Acta. A Mol. Biomol. Spectrosc. 2020, 228, 1–22. [Google Scholar] [CrossRef]
- Santillan, A.; Tomas, R.C.; Bangaoil, R.; Lopez, R.; Gomez, M.H.; Fellizar, A.; Lim, A.; Abanilla, L.; Ramos, M.C.; Guevarra, L.; et al. Discrimination of malignant from benign thyroid lesions through neural networks using FTIR signals obtained from tissues. Anal. Bioanal. Chem. 2021, 413, 2163–2180. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.; Zhang, W.T.; Tian, P.R.; Ling, X.F.; Xu, Z. Characteristics analysis of the thyroid gland surface of normal human with Fourier transform infrared (FTIR) spectra. Guang Pu Xue Yu Guang Pu Fen Xi 2016, 36, 3092–3096. [Google Scholar] [CrossRef]
- Albero, F.G. Diagnóstico de Lesões da Tireóide Pela Espectroscopia de Absorção no Infravermelho por Transformada de Fourier-FTIR; Biblioteca Digital de Teses e Dissertações da Universidade de São Paulo: São Paulo, Brazil, 2009. [Google Scholar]
- Pereira, T.M.; Zezell, D.M.; Bird, B.; Miljković, M.; Diem, M. The characterization of normal thyroid tissue by micro-FTIR spectroscopy. Analyst 2013, 138, 7094–7100. [Google Scholar] [CrossRef]
- Zeng, X.T.; Xu, Y.Z.; Zhang, X.Q.; Xu, Z.; Zhang, Y.F.; Wu, J.G.; Zhou, X.S.; Ling, X.F. FTIR spectroscopic explorations of freshly resected thyroid malignant tissues. Guang Pu Xue Yu Guang Pu Fen Xi 2007, 27, 2422–2426. [Google Scholar] [PubMed]
- Schultz, C.P.; Liu, K.Z.; Salamon, E.A.; Riese, K.T.; Mantsch, H.H. Application of FT-IR microspectroscopy in diagnosing thyroid neoplasms. J. Mol. Struct. 1999, 480–481, 369–377. [Google Scholar] [CrossRef]
- Martinez-Marin, D.; Sreedhar, H.; Varma, V.K.; Eloy, C.; Sobrinho-Simões, M.; Kajdacsy-Balla, A.; Walsh, M.J. Accounting for tissue heterogeneity in infrared spectroscopic imaging for accurate diagnosis of thyroid carcinoma subtypes. Vib. Spectrosc. 2017, 91, 77–82. [Google Scholar] [CrossRef]
- Wu, M.; Zhang, W.; Tian, P.; Ling, X.; Xu, Z. Intraoperative diagnosis of thyroid diseases by fourier transform infrared spectroscopy based on support vector machine. Int. J. Clin. Exp. Med. 2016, 9, 2351–2358. [Google Scholar]
- Villela, L.F.d.A.; de Freitas, L.C.C.; Brandão, M.; Bachmann, L.; Iwakura, R. Characterization of thyroid tissue using infrared spectroscopy. Arch. Head Neck Surg. 2018, 47, 1–10. [Google Scholar] [CrossRef]
- Depciuch, J.; Stanek-Widera, A.; Lange, D.; Biskup-Frużyńska, M.; Stanek-Tarkowska, J.; Czarny, W.; Cebulski, J. Spectroscopic analysis of normal and neoplastic (WI-FTC) thyroid tissue. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2018, 204, 18–24. [Google Scholar] [CrossRef] [PubMed]
- Minuto, M.N.; Shintu, L.; Caldarelli, S. Proteomics, and Metabolomics: Magnetic Resonance Spectroscopy for the Presurgical Screening of Thyroid Nodules. Curr. Genom. 2014, 15, 178–183. [Google Scholar] [CrossRef] [PubMed]
- Gupta, N.; Goswami, B.; Chowdhury, V.; RaviShankar, L.; Kakar, A. Evaluation of the role of magnetic resonance spectroscopy in the diagnosis of follicular malignancies of thyroid. Arch. Surg. 2011, 146, 179–182. [Google Scholar] [CrossRef] [PubMed]
- Kaplan, M.M. Clinical perspectives in the diagnosis of thyroid disease. Clin. Chem. 1999, 45, 1377–1383. [Google Scholar] [CrossRef] [PubMed]
- Nunes, A. FTIR Spectroscopy–A Potential Tool to Identify Metabolic Changes in Dementia Patients. HSOA J. Alzheimers Neurodegener. Dis. 2016, 2, 1–9. [Google Scholar] [CrossRef]
- Bellisola, G.; Sorio, C. Infrared spectroscopy and microscopy in cancer research and diagnosis. Am. J. Cancer Res. 2012, 2, 1–21. [Google Scholar]
- Kaznowska, E.; Depciuch, J.; Łach, K.; Kołodziej, M.; Koziorowska, A.; Vongsvivut, J.; Zawlik, I.; Cholewa, M.; Cebulski, J. The classification of lung cancers and their degree of malignancy by FTIR, PCA-LDA analysis, and a physics-based computational model. Talanta 2018, 186, 337–345. [Google Scholar] [CrossRef]
- Lu, Y.; Zhao, Y.; Zhu, Y.; Xu, X.; Yin, J. In situ research and diagnosis of breast cancer by using HOF-ATR-FTIR spectroscopy. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2020, 235, 1–17. [Google Scholar] [CrossRef]
- Felgueiras, J.; Vieira Silva, J.; Nunes, A.; Patrício, A.; Pelech, S.; Fardilha, M. Understanding prostate cancer biology using metabolomics and proteomics approaches: Potentials in the improvement of the diagnosis, prognosis and identification of new therapeutic targets. Eur. J. Cancer 2017, 72, S192. [Google Scholar] [CrossRef]
- Santos, F.; Magalhães, S.; Henriques, M.C.; Silva, B.; Valença, I.; Ribeiro, D.; Fardilha, M.; Nunes, A. Understanding Prostate Cancer Cells Metabolome: A Spectroscopic Approach. Curr. Metab. 2018, 6, 218–224. [Google Scholar] [CrossRef]
- Thumanu, K.; Sangrajrang, S.; Khuhaprema, T.; Kalalak, A.; Tanthanuch, W.; Pongpiachan, S.; Heraud, P. Diagnosis of liver cancer from blood sera using FTIR microspectroscopy: A preliminary study. J. Biophotonics 2014, 7, 222–231. [Google Scholar] [CrossRef] [PubMed]
- Zelig, U.; Barlev, E.; Bar, O.; Gross, I.; Flomen, F.; Mordechai, S.; Kapelushnik, J.; Nathan, I.; Kashtan, H.; Wasserberg, N.; et al. Early detection of breast cancer using total biochemical analysis of peripheral blood components: A preliminary study. BMC Cancer 2015, 15, 1–10. [Google Scholar] [CrossRef]
- Su, K.Y.; Lee, W.L. Fourier transform infrared spectroscopy as a cancer screening and diagnostic tool: A review and prospects. Cancers 2020, 12, 115. [Google Scholar] [CrossRef] [PubMed]
- Stuart, B.H. Infrared Spectroscopy of Biological Applications: An Overview. In Encyclopedia of Analytical Chemistry; John Wiley & Sons, Ltd.: Chichester, UK, 2012; pp. 1–17. [Google Scholar]
- Santos, F.; Magalhaes, S.; Henriques, M.C.; Fardilha, M.; Nunes, A. Spectroscopic Features of Cancer Cells: FTIR Spectroscopy as a Tool for Early Diagnosis. Curr. Metab. 2018, 6, 103–111. [Google Scholar] [CrossRef]
- Magalhães, S.; Goodfellow, B.J.; Nunes, A. FTIR spectroscopy in biomedical research: How to get the most out of its potential. Appl. Spectrosc. Rev. 2021, 56, 869–907. [Google Scholar] [CrossRef]
- Morais, C.L.M.; Lima, K.M.G.; Singh, M.; Martin, F.L. Tutorial: Multivariate classification for vibrational spectroscopy in biological samples. Nat. Protoc. 2020, 15, 2143–2162. [Google Scholar] [CrossRef]
- Rudd, T.R.; Mauri, L.; Marinozzi, M.; Stancanelli, E.; Yates, E.A.; Naggi, A.; Guerrini, M. Multivariate analysis applied to complex biological medicines. Faraday Discuss. 2019, 218, 303–316. [Google Scholar] [CrossRef]
| Sample Type | Acquisition Conditions | Analysis Type | Main Findings | References |
|---|---|---|---|---|
| FNA citology (original aspirate; cell-free supernant; cell pellet) | µ-FTIR Scans: 256 Resolution: 4 cm −1 Spectral region: 1200−900 cm−1 | Unsupervised Cluster analysis (Ward’s minimum variance algorithm and Euclidian distances) and Bivariate statistical analysis; |
| [75] |
| Tissue from thyroid gland | µ-FTIR Scans: 16 | |||
| n = 89 FNA citology samples of 89 thyroid nodules (original aspirate; cell pellet) Thyroid powder | Transmission Resolution: 4 cm−1 Spectral region: 1800−900 cm−1 | Baseline correction, area normalization and second derivatives using the Savitzky-Golay algorithm; Unsupervised Cluster analysis (Ward’s minimum variance algorithm and Euclidian distances) and Supervised Linear discriminant analysis; |
| [62] |
| n = 184 Cervical lymph nodes (61 metastatic; 123 non-metastatic) from 22 PTC patients | ATR Scans: 32 Resolution: 8 cm−1 Spectral region: 4000−1000 cm−1 | Intensity measurement of spectroscopic signals with relative intensity ratios; Wilks’ lambda linear discriminant analysis; |
| [67] |
| n = 60 60 Thyroid gland tissue samples (43 of NG; 17 of PTC) | ATR Scans: 32 Resolution: 4 cm−1 Spectral region: 4000−800 cm−1 | Baseline correction, straight-line generated and smoothed; Measured the wave intensity ratios and the peak positions; canonical discriminant analysis |
| [68] |
| n = 80 80 Thyroid gland tissue samples T3 and T4 Thyroid hormones | µ-FTIR Resolution: 4 cm−1 Spectral region: 4000−750 cm−1 | Vector normalization, converted to second derivatives using the Savitzky–Golay algorithm, noise-filtered and phase corrected; Hierarchical cluster analysis (Ward’s minimum variance algorithm and Euclidian distances); |
| [73] |
| n = 161 161 subjects (25 males; 136 females)—111 patients undergoing thyroid surgery; 50 healthy volunteers | ATR using optical fiber trough skin surface Scans: 32 Resolution: 8 cm−1 Spectral region: 3100−1000 cm−1 | Baseline correction and a smoothing with a 9-point moving average; Intensity measurement of spectroscopic signals with relative intensity ratios; Wilks’ lambda stepwise discriminant analysis |
| [63] |
| n = 20 20 Thyroid gland tissue samples (10 of FvPTC; 10 of FTC) | µ-FTIR Scans: 8 Resolution: 4 cm−1 Spectral region: 3850−900 cm−1 | Baseline correction; Normalization of average spectra to the absorbance of 1651 cm−1; Principle component analysis and Linear discriminant analysis; |
| [76] |
| n = 112 112 Thyroid gland tissue samples (67 of NG; 10 of thyroid carcinomas) | ATR Scans: 32 Resolution: 8 cm−1 Spectral region: 1900−1050 cm−1 | Intensity measurement of spectroscopic signals with relative intensity ratios; Tests of normal distribution and variance of homogeneity and Student’s t test |
| [77] |
| n = 44 44 Thyroid gland tissue samples (16 of NG; 3 of FA; 4 of thyroiditis; 19 of PTC; 2 of FTC) | Not indicated | Normalization of the spectra to the absorbance of 1651 cm−1; Second derivative; Determination of bands areas; Calculation of relative areas ratio; one-way ANOVA and the Tukey and Fisher tests |
| [78] |
| n = 15 Thyroid gland tissue samples of 15 WI-FTC patients | ATR Scans: 64 Resolution: 2 cm−1 Spectral region: 4000−400 cm−1 | Baseline correction (concave rubberband method), normalization (min.-max. method) and second derivative; Measurement of area under the curve; Principle component analysis and Linear discriminant analysis; |
| [79] |
| n = 32 32 Thyroid gland tissue samples (15 FTC patients and 17 FA patients) | ATR Scans: 32 Resolution: 2 cm−1 Spectral region: 1800−800 cm−1 | Baseline correction and normalization of average spectra to the intensity sum of all peaks; Principle component analysis and Linear discriminant analysis; |
| [61] |
| n = 14 14 Thyroid gland tissue samples (3 of metastatic lymph nodes PTC (PTC+); 6 of non-metastatic lymph nodes PTC (PTC-); 5 of normal tissue) | ATR Scans: 48 Resolution: 4 cm−1 Spectral region: 4000−600 cm−1 | Baseline correction, vector normalization, first deivative and smoothing; Principle component analysis and Linear discriminant analysis; Leave one out cross validation; |
| [69] |
| n = 164 164 Thyroid gland tissue samples (76 malignant; 88 benign) | ATR Scans: 64 Resolution: 8 cm−1 Spectral region: 4000−1000 cm−1 | Normalization (z-score normalization) and baseline correction (rubberband method); Principle component analysis and Linear discriminant analysis; Shapiro–Wilk test, tests of variance of homogeneity and Mann–Whitney U test; |
| [70] |
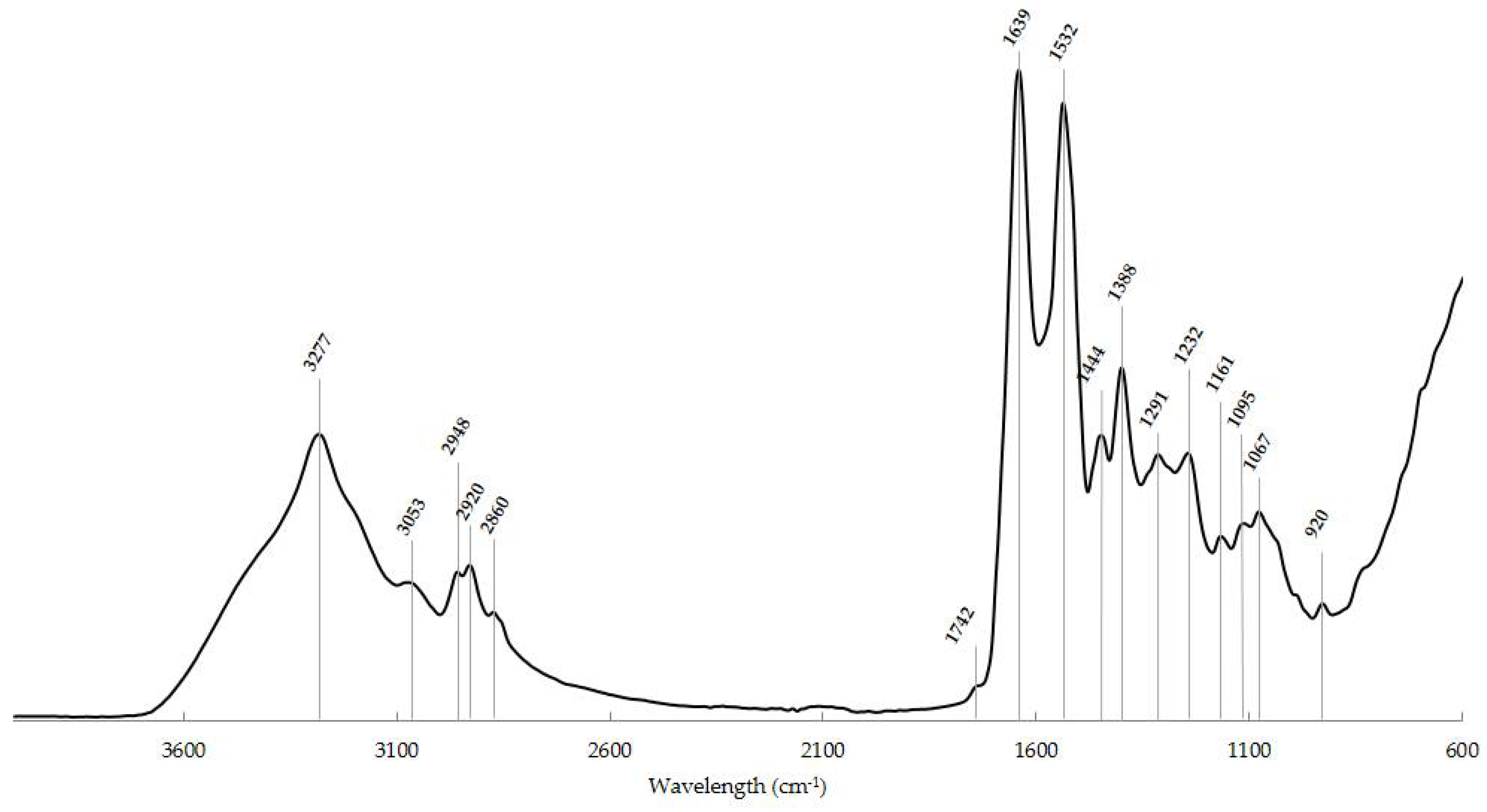
| Wavenumber (cm−1) | Assigned Biochemical Component | Vibrational Mode |
|---|---|---|
| 3277 | Amide A: peptide, protein | N-H stretching |
| 3053 | Amide B: peptide, protein | N-H stretching |
| 2948 | Lipids | CH3 asymmetric stretching |
| 2920 | CH2 asymmetric stretching | |
| 2860 | CH2 symmetric stretching | |
| 1742 | Phospholipid esters | C = O stretching |
| 1639 | Amide I: parallel β-sheets | C = O stretching, C-N stretching, in-plane N-H bending |
| 1532 | Amide II | N-H stretching, C-N stretching, C-C stretching |
| 1444 | Membrane lipids and proteins | CH3 and CH2 deformation |
| 1388 | Phospholipid, fatty acid, triglyceride | CH3 symmetric wagging |
| 1291 | Amide III | N-H bending, C-N stretching, C = O stretching, C-C stretching, CH3 stretching |
| 1232 | Nucleic acids | PO2—symmetric stretching |
| 1161 | Carbohydrates | C-O stretching |
| 1093 and 1067 | DNA, RNA, phospholipid, phosphorylated protein | PO2—symmetric stretching |
| 920 | Dianionic phosphate monoesters of phosphorylated proteins and nucleic acids | PO3 2− symmetric stretching |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Neto, V.; Esteves-Ferreira, S.; Inácio, I.; Alves, M.; Dantas, R.; Almeida, I.; Guimarães, J.; Azevedo, T.; Nunes, A. Metabolic Profile Characterization of Different Thyroid Nodules Using FTIR Spectroscopy: A Review. Metabolites 2022, 12, 53. https://doi.org/10.3390/metabo12010053
Neto V, Esteves-Ferreira S, Inácio I, Alves M, Dantas R, Almeida I, Guimarães J, Azevedo T, Nunes A. Metabolic Profile Characterization of Different Thyroid Nodules Using FTIR Spectroscopy: A Review. Metabolites. 2022; 12(1):53. https://doi.org/10.3390/metabo12010053
Chicago/Turabian StyleNeto, Vanessa, Sara Esteves-Ferreira, Isabel Inácio, Márcia Alves, Rosa Dantas, Idália Almeida, Joana Guimarães, Teresa Azevedo, and Alexandra Nunes. 2022. "Metabolic Profile Characterization of Different Thyroid Nodules Using FTIR Spectroscopy: A Review" Metabolites 12, no. 1: 53. https://doi.org/10.3390/metabo12010053
APA StyleNeto, V., Esteves-Ferreira, S., Inácio, I., Alves, M., Dantas, R., Almeida, I., Guimarães, J., Azevedo, T., & Nunes, A. (2022). Metabolic Profile Characterization of Different Thyroid Nodules Using FTIR Spectroscopy: A Review. Metabolites, 12(1), 53. https://doi.org/10.3390/metabo12010053








