Lipid Metabolite Biomarkers in Cardiovascular Disease: Discovery and Biomechanism Translation from Human Studies
Abstract
1. Introduction
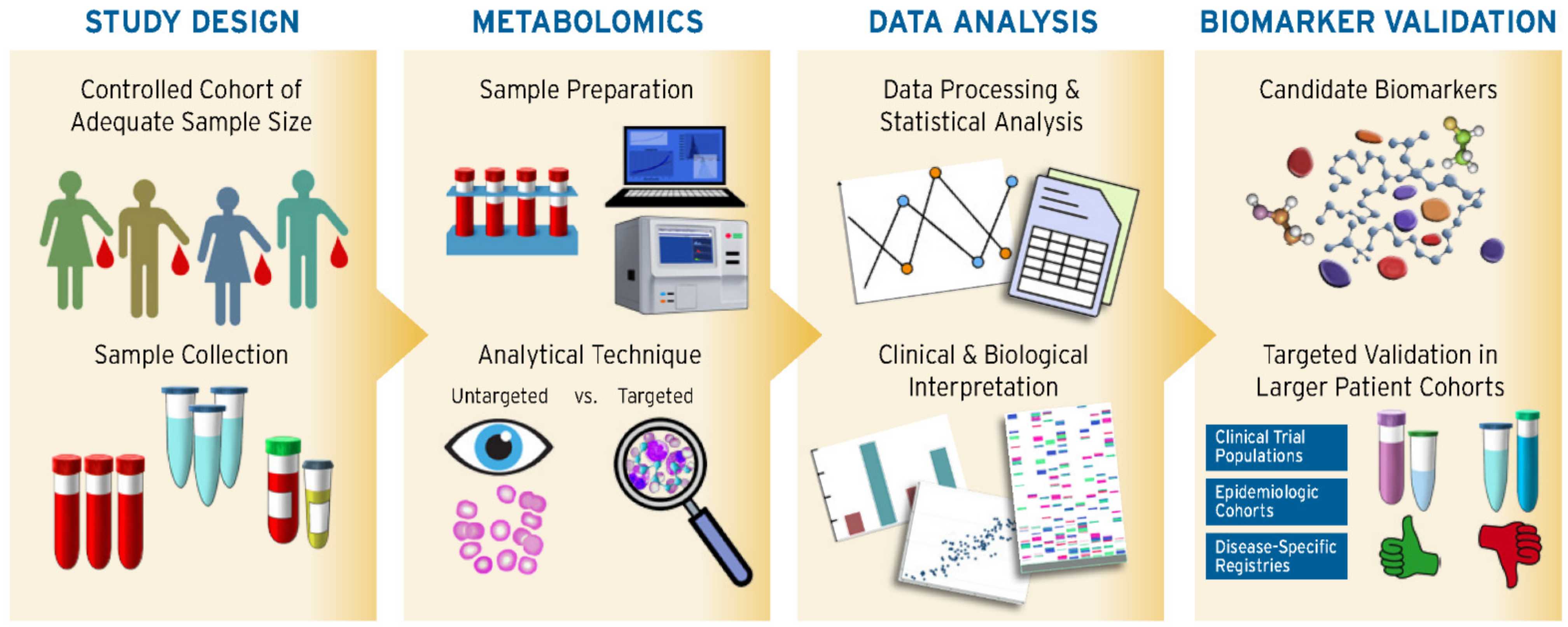
2. Lipidomic Biomarker Discovery Approach and Summarized Findings
- Metabolomics studies using MS or NMR approaches
- Lipid or lipid-related metabolites selected in final model
- Human blood samples
- CVD outcomes
- Exclusion criteria:
- Exclusively proteomic or other non-metabolomic studies which did not incorporate MS or NMR approaches
- Non-lipid or lipid-related metabolites selected in the final model
- Meta-analyses or literature reviews
- Animal studies
- in vitro studies
- Non-CVD outcomes
2.1. Sample Selection
2.2. Untargeted and Targeted Approaches
2.3. Analytical Platforms
2.4. Data Processing and Analysis
2.5. Diagnostic and Prognostic Value
3. Lipid Metabolism Translation from Human CVD Studies
3.1. Summary of Lipidomic Findings and Potential Pathomechanisms
3.2. Acylcarnitines and Fatty Acids
3.3. Phospholipids
3.4. Glycolipids
3.5. Cholesterol Esters
3.6. Sphingolipids/Ceramides
4. Future Directions
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Virani, S.S.; Alonso, A.; Benjamin, E.J.; Bittencourt, M.S.; Callaway, C.W.; Carson, A.P.; Chamberlain, A.M.; Chang, A.R.; Cheng, S.; Delling, F.N.; et al. Heart Disease and Stroke Statistics-2020 Update: A Report From the American Heart Association. Circulation 2020, 141, e139–e596. [Google Scholar] [CrossRef] [PubMed]
- Neubauer, S. The failing heart—An engine out of fuel. N. Engl. J. Med. 2007, 356, 1140–1151. [Google Scholar] [CrossRef]
- Karwi, Q.G.; Uddin, G.M.; Ho, K.L.; Lopaschuk, G.D. Loss of Metabolic Flexibility in the Failing Heart. Front. Cardiovasc. Med. 2018, 5, 68. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, T.; Kelly, J.P.; McGarrah, R.W.; Hellkamp, A.S.; Fiuzat, M.; Testani, J.M.; Wang, T.S.; Verma, A.; Samsky, M.D.; Donahue, M.P.; et al. Prognostic Implications of Long-Chain Acylcarnitines in Heart Failure and Reversibility With Mechanical Circulatory Support. J. Am. Coll. Cardiol. 2016, 67, 291–299. [Google Scholar] [CrossRef]
- Alshehry, Z.H.; Mundra, P.A.; Barlow, C.K.; Mellett, N.A.; Wong, G.; McConville, M.J.; Simes, J.; Tonkin, A.M.; Sullivan, D.R.; Barnes, E.H.; et al. Plasma Lipidomic Profiles Improve on Traditional Risk Factors for the Prediction of Cardiovascular Events in Type 2 Diabetes Mellitus. Circulation 2016, 134, 1637–1650. [Google Scholar] [CrossRef]
- Andersson, C.; Liu, C.; Cheng, S.; Wang, T.J.; Gerszten, R.E.; Larson, M.G.; Vasan, R.S. Metabolomic signatures of cardiac remodelling and heart failure risk in the community. ESC Heart Fail. 2020, 7, 3707–3715. [Google Scholar] [CrossRef]
- Anroedh, S.; Hilvo, M.; Akkerhuis, K.M.; Kauhanen, D.; Koistinen, K.; Oemrawsingh, R.; Serruys, P.; van Geuns, R.J.; Boersma, E.; Laaksonen, R.; et al. Plasma concentrations of molecular lipid species predict long-term clinical outcome in coronary artery disease patients. J. Lipid Res. 2018, 59, 1729–1737. [Google Scholar] [CrossRef] [PubMed]
- Cavus, E.; Karakas, M.; Ojeda, F.M.; Kontto, J.; Veronesi, G.; Ferrario, M.M.; Linneberg, A.; Jorgensen, T.; Meisinger, C.; Thorand, B.; et al. Association of Circulating Metabolites With Risk of Coronary Heart Disease in a European Population: Results From the Biomarkers for Cardiovascular Risk Assessment in Europe (BiomarCaRE) Consortium. JAMA Cardiol. 2019, 4, 1270–1279. [Google Scholar] [CrossRef] [PubMed]
- Cheng, M.L.; Wang, C.H.; Shiao, M.S.; Liu, M.H.; Huang, Y.Y.; Huang, C.Y.; Mao, C.T.; Lin, J.F.; Ho, H.Y.; Yang, N.I. Metabolic disturbances identified in plasma are associated with outcomes in patients with heart failure: Diagnostic and prognostic value of metabolomics. J. Am. Coll. Cardiol. 2015, 65, 1509–1520. [Google Scholar] [CrossRef]
- Cheng, J.M.; Suoniemi, M.; Kardys, I.; Vihervaara, T.; de Boer, S.P.; Akkerhuis, K.M.; Sysi-Aho, M.; Ekroos, K.; Garcia-Garcia, H.M.; Oemrawsingh, R.M.; et al. Plasma concentrations of molecular lipid species in relation to coronary plaque characteristics and cardiovascular outcome: Results of the ATHEROREMO-IVUS study. Atherosclerosis 2015, 243, 560–566. [Google Scholar] [CrossRef]
- Delles, C.; Rankin, N.J.; Boachie, C.; McConnachie, A.; Ford, I.; Kangas, A.; Soininen, P.; Trompet, S.; Mooijaart, S.P.; Jukema, J.W.; et al. Nuclear magnetic resonance-based metabolomics identifies phenylalanine as a novel predictor of incident heart failure hospitalisation: Results from PROSPER and FINRISK 1997. Eur. J. Heart Fail. 2018, 20, 663–673. [Google Scholar] [CrossRef] [PubMed]
- Fernandez, C.; Sandin, M.; Sampaio, J.L.; Almgren, P.; Narkiewicz, K.; Hoffmann, M.; Hedner, T.; Wahlstrand, B.; Simons, K.; Shevchenko, A.; et al. Plasma lipid composition and risk of developing cardiovascular disease. PLoS ONE 2013, 8, e71846. [Google Scholar]
- Floegel, A.; Kühn, T.; Sookthai, D.; Johnson, T.; Prehn, C.; Rolle-Kampczyk, U.; Otto, W.; Weikert, C.; Illig, T.; von Bergen, M.; et al. Serum metabolites and risk of myocardial infarction and ischemic stroke: A targeted metabolomic approach in two German prospective cohorts. Eur. J. Epidemiol. 2018, 33, 55–66. [Google Scholar] [CrossRef]
- Ganna, A.; Salihovic, S.; Sundstrom, J.; Broeckling, C.D.; Hedman, A.K.; Magnusson, P.K.; Pedersen, N.L.; Larsson, A.; Siegbahn, A.; Zilmer, M.; et al. Large-scale metabolomic profiling identifies novel biomarkers for incident coronary heart disease. PLoS Genet. 2014, 10, e1004801. [Google Scholar] [CrossRef]
- Gao, X.; Ke, C.; Liu, H.; Liu, W.; Li, K.; Yu, B.; Sun, M. Large-scale Metabolomic Analysis Reveals Potential Biomarkers for Early Stage Coronary Atherosclerosis. Sci. Rep. 2017, 7, 11817. [Google Scholar] [CrossRef] [PubMed]
- Havulinna, A.S.; Sysi-Aho, M.; Hilvo, M.; Kauhanen, D.; Hurme, R.; Ekroos, K.; Salomaa, V. Circulating Ceramides Predict Cardiovascular Outcomes in the Population-Based FINRISK 2002 Cohort. Arterioscler. Thromb. Vasc. Biol. 2016, 36, 2424–2430. [Google Scholar] [CrossRef] [PubMed]
- Hilvo, M.; Meikle, P.J.; Pedersen, E.R.; Tell, G.S.; Dhar, I.; Brenner, H.; Schottker, B.; Laaperi, M.; Kauhanen, D.; Koistinen, K.M.; et al. Development and validation of a ceramide- and phospholipid-based cardiovascular risk estimation score for coronary artery disease patients. Eur. Heart J. 2020, 41, 371–380. [Google Scholar] [CrossRef] [PubMed]
- Holmes, M.V.; Millwood, I.Y.; Kartsonaki, C.; Hill, M.R.; Bennett, D.A.; Boxall, R.; Guo, Y.; Xu, X.; Bian, Z.; Hu, R.; et al. Lipids, Lipoproteins, and Metabolites and Risk of Myocardial Infarction and Stroke. J. Am. Coll. Cardiol. 2018, 71, 620–632. [Google Scholar] [CrossRef]
- Jadoon, A.; Mathew, A.V.; Byun, J.; Gadegbeku, C.A.; Gipson, D.S.; Afshinnia, F.; Pennathur, S. Gut Microbial Product Predicts Cardiovascular Risk in Chronic Kidney Disease Patients. Am. J. Nephrol. 2018, 48, 269–277. [Google Scholar] [CrossRef]
- Ji, R.; Akashi, H.; Drosatos, K.; Liao, X.; Jiang, H.; Kennel, P.J.; Brunjes, D.L.; Castillero, E.; Zhang, X.; Deng, L.Y.; et al. Increased de novo ceramide synthesis and accumulation in failing myocardium. JCI Insight 2017, 2, e82922. [Google Scholar] [CrossRef]
- Kalim, S.; Clish, C.B.; Wenger, J.; Elmariah, S.; Yeh, R.W.; Deferio, J.J.; Pierce, K.; Deik, A.; Gerszten, R.E.; Thadhani, R.; et al. A plasma long-chain acylcarnitine predicts cardiovascular mortality in incident dialysis patients. J. Am. Heart Assoc. 2013, 2, e000542. [Google Scholar] [CrossRef] [PubMed]
- Laaksonen, R.; Ekroos, K.; Sysi-Aho, M.; Hilvo, M.; Vihervaara, T.; Kauhanen, D.; Suoniemi, M.; Hurme, R.; Marz, W.; Scharnagl, H.; et al. Plasma ceramides predict cardiovascular death in patients with stable coronary artery disease and acute coronary syndromes beyond LDL-cholesterol. Eur. Heart J. 2016, 37, 1967–1976. [Google Scholar] [CrossRef]
- Lemaitre, R.N.; Jensen, P.N.; Hoofnagle, A.; McKnight, B.; Fretts, A.M.; King, I.B.; Siscovick, D.S.; Psaty, B.M.; Heckbert, S.R.; Mozaffarian, D.; et al. Plasma Ceramides and Sphingomyelins in Relation to Heart Failure Risk. Circ. Heart Fail. 2019, 12, e005708. [Google Scholar] [CrossRef]
- Lu, J.; Chen, B.; Chen, T.; Guo, S.; Xue, X.; Chen, Q.; Zhao, M.; Xia, L.; Zhu, Z.; Zheng, L.; et al. Comprehensive metabolomics identified lipid peroxidation as a prominent feature in human plasma of patients with coronary heart diseases. Redox Biol. 2017, 12, 899–907. [Google Scholar] [CrossRef]
- Mayerhofer, C.C.K.; Kummen, M.; Holm, K.; Broch, K.; Awoyemi, A.; Vestad, B.; Storm-Larsen, C.; Seljeflot, I.; Ueland, T.; Bohov, P.; et al. Low fibre intake is associated with gut microbiota alterations in chronic heart failure. ESC Heart Fail. 2020, 7, 456–466. [Google Scholar] [CrossRef]
- McGranaghan, P.; Dungen, H.D.; Saxena, A.; Rubens, M.; Salami, J.; Radenkovic, J.; Bach, D.; Apostolovic, S.; Loncar, G. Incremental prognostic value of a novel metabolite-based biomarker score in congestive heart failure patients. ESC Heart Fail. 2020, 7, 3029–3039. [Google Scholar] [CrossRef]
- McGranaghan, P.; Saxena, A.; Dungen, H.D.; Rubens, M.; Appunni, S.; Salami, J.; Veledar, E.; Lacour, P.; Blaschke, F.; Obradovic, D.; et al. Performance of a cardiac lipid panel compared to four prognostic scores in chronic heart failure. Sci. Rep. 2021, 11, 8164. [Google Scholar] [CrossRef] [PubMed]
- Meikle, P.J.; Wong, G.; Tsorotes, D.; Barlow, C.K.; Weir, J.M.; Christopher, M.J.; MacIntosh, G.L.; Goudey, B.; Stern, L.; Kowalczyk, A.; et al. Plasma lipidomic analysis of stable and unstable coronary artery disease. Arterioscler. Thromb. Vasc. Biol. 2011, 31, 2723–2732. [Google Scholar] [CrossRef] [PubMed]
- Miller, C.D.; Thomas, M.J.; Hiestand, B.; Samuel, M.P.; Wilson, M.D.; Sawyer, J.; Rudel, L.L. Cholesteryl esters associated with acyl-CoA:cholesterol acyltransferase predict coronary artery disease in patients with symptoms of acute coronary syndrome. Acad. Emerg. Med. 2012, 19, 673–682. [Google Scholar] [CrossRef]
- Mueller-Hennessen, M.; Dungen, H.D.; Lutz, M.; Trippel, T.D.; Kreuter, M.; Sigl, J.; Muller, O.J.; Tahirovic, E.; Witt, H.; Ternes, P.; et al. A Novel Lipid Biomarker Panel for the Detection of Heart Failure with Reduced Ejection Fraction. Clin. Chem. 2017, 63, 267–277. [Google Scholar] [CrossRef]
- Mueller-Hennessen, M.; Sigl, J.; Fuhrmann, J.C.; Witt, H.; Reszka, R.; Schmitz, O.; Kastler, J.; Fischer, J.J.; Müller, O.J.; Giannitsis, E.; et al. Metabolic profiles in heart failure due to non-ischemic cardiomyopathy at rest and under exercise. ESC Heart Fail. 2017, 4, 178–189. [Google Scholar] [CrossRef]
- Mundra, P.A.; Barlow, C.K.; Nestel, P.J.; Barnes, E.H.; Kirby, A.; Thompson, P.; Sullivan, D.R.; Alshehry, Z.H.; Mellett, N.A.; Huynh, K.; et al. Large-scale plasma lipidomic profiling identifies lipids that predict cardiovascular events in secondary prevention. JCI Insight 2018, 3, e121326. [Google Scholar] [CrossRef]
- Nwabuo, C.C.; Duncan, M.; Xanthakis, V.; Peterson, L.R.; Mitchell, G.F.; McManus, D.; Cheng, S.; Vasan, R.S. Association of Circulating Ceramides With Cardiac Structure and Function in the Community: The Framingham Heart Study. J. Am. Heart Assoc. 2019, 8, e013050. [Google Scholar] [CrossRef]
- Ottosson, F.; Emami Khoonsari, P.; Gerl, M.J.; Simons, K.; Melander, O.; Fernandez, C. A plasma lipid signature predicts incident coronary artery disease. Int. J. Cardiol. 2021, 331, 249–254. [Google Scholar] [CrossRef] [PubMed]
- Paapstel, K.; Kals, J.; Eha, J.; Tootsi, K.; Ottas, A.; Piir, A.; Jakobson, M.; Lieberg, J. Inverse relations of serum phosphatidylcholines and lysophosphatidylcholines with vascular damage and heart rate in patients with atherosclerosis. Nutr. Metab. Cardiovasc. Dis. 2018, 28, 44–52. [Google Scholar] [CrossRef] [PubMed]
- Paynter, N.P.; Balasubramanian, R.; Giulianini, F.; Wang, D.D.; Tinker, L.F.; Gopal, S.; Deik, A.A.; Bullock, K.; Pierce, K.A.; Scott, J.; et al. Metabolic Predictors of Incident Coronary Heart Disease in Women. Circulation 2018, 137, 841–853. [Google Scholar] [CrossRef]
- Peterson, L.R.; Xanthakis, V.; Duncan, M.S.; Gross, S.; Friedrich, N.; Volzke, H.; Felix, S.B.; Jiang, H.; Sidhu, R.; Nauck, M.; et al. Ceramide Remodeling and Risk of Cardiovascular Events and Mortality. J. Am. Heart Assoc. 2018, 7, e007931. [Google Scholar] [CrossRef]
- Poss, A.M.; Maschek, J.A.; Cox, J.E.; Hauner, B.J.; Hopkins, P.N.; Hunt, S.C.; Holland, W.L.; Summers, S.A. Machine learning reveals serum sphingolipids as cholesterol-independent biomarkers of coronary artery disease. J. Clin. Investig. 2020, 130, 1363–1376. [Google Scholar] [CrossRef] [PubMed]
- Razquin, C.; Liang, L.; Toledo, E.; Clish, C.B.; Ruiz-Canela, M.; Zheng, Y.; Wang, D.D.; Corella, D.; Castaner, O.; Ros, E.; et al. Plasma lipidome patterns associated with cardiovascular risk in the, P.R.EDIMED trial: A case-cohort study. Int. J. Cardiol. 2018, 253, 126–132. [Google Scholar] [CrossRef] [PubMed]
- Rizza, S.; Copetti, M.; Rossi, C.; Cianfarani, M.A.; Zucchelli, M.; Luzi, A.; Pecchioli, C.; Porzio, O.; Di Cola, G.; Urbani, A.; et al. Metabolomics signature improves the prediction of cardiovascular events in elderly subjects. Atherosclerosis 2014, 232, 260–264. [Google Scholar] [CrossRef]
- Seah, J.Y.H.; Chew, W.S.; Torta, F.; Khoo, C.M.; Wenk, M.R.; Herr, D.R.; Choi, H.; Tai, E.S.; van Dam, R.M. Plasma sphingolipids and risk of cardiovascular diseases: A large-scale lipidomic analysis. Metabolomics 2020, 16, 89. [Google Scholar] [CrossRef]
- Shah, S.H.; Bain, J.R.; Muehlbauer, M.J.; Stevens, R.D.; Crosslin, D.R.; Haynes, C.; Dungan, J.; Newby, L.K.; Hauser, E.R.; Ginsburg, G.S.; et al. Association of a peripheral blood metabolic profile with coronary artery disease and risk of subsequent cardiovascular events. Circ. Cardiovasc. Genet. 2010, 3, 207–214. [Google Scholar] [CrossRef] [PubMed]
- Shah, S.H.; Sun, J.L.; Stevens, R.D.; Bain, J.R.; Muehlbauer, M.J.; Pieper, K.S.; Haynes, C.; Hauser, E.R.; Kraus, W.E.; Granger, C.B.; et al. Baseline metabolomic profiles predict cardiovascular events in patients at risk for coronary artery disease. Am. Heart J. 2012, 163, 844–850 e1. [Google Scholar] [CrossRef] [PubMed]
- Sigruener, A.; Kleber, M.E.; Heimerl, S.; Liebisch, G.; Schmitz, G.; Maerz, W. Glycerophospholipid and sphingolipid species and mortality: The Ludwigshafen Risk and Cardiovascular Health (LURIC) study. PLoS ONE 2014, 9, e85724. [Google Scholar] [CrossRef] [PubMed]
- Sigruener, A.; Kleber, M.E.; Heimerl, S.; Liebisch, G.; Schmitz, G.; Maerz, W. Comparative lipidomics profiling of human atherosclerotic plaques. Circ. Cardiovasc. Genet. 2011, 4, 232–242. [Google Scholar]
- Stegemann, C.; Pechlaner, R.; Willeit, P.; Langley, S.R.; Mangino, M.; Mayr, U.; Menni, C.; Moayyeri, A.; Santer, P.; Rungger, G.; et al. Lipidomics profiling and risk of cardiovascular disease in the prospective population-based Bruneck study. Circulation 2014, 129, 1821–1831. [Google Scholar] [CrossRef]
- Stenemo, M.; Ganna, A.; Salihovic, S.; Nowak, C.; Sundström, J.; Giedraitis, V.; Broeckling, C.D.; Prenni, J.E.; Svensson, P.; Magnusson, P.K.E.; et al. The metabolites urobilin and sphingomyelin (30:1) are associated with incident heart failure in the general population. ESC Heart Fail. 2019, 6, 764–773. [Google Scholar] [CrossRef]
- Sun, Y.; Koh, H.W.; Choi, H.; Koh, W.P.; Yuan, J.M.; Newman, J.W.; Su, J.; Fang, J.; Ong, C.N.; van Dam, R.M. Plasma fatty acids, oxylipins, and risk of myocardial infarction: The Singapore Chinese Health Study. J. Lipid Res. 2016, 57, 1300–1307. [Google Scholar] [CrossRef]
- Syme, C.; Czajkowski, S.; Shin, J.; Abrahamowicz, M.; Leonard, G.; Perron, M.; Richer, L.; Veillette, S.; Gaudet, D.; Strug, L.; et al. Glycerophosphocholine Metabolites and Cardiovascular Disease Risk Factors in Adolescents: A Cohort Study. Circulation 2016, 134, 1629–1636. [Google Scholar] [CrossRef]
- Tang, W.H.; Wang, Z.; Levison, B.S.; Koeth, R.A.; Britt, E.B.; Fu, X.; Wu, Y. Intestinal microbial metabolism of phosphatidylcholine and cardiovascular risk. N. Engl. J. Med. 2013, 368, 1575–1584. [Google Scholar] [CrossRef]
- Tang, W.H.; Wang, Z.; Fan, Y.; Levison, B.; Hazen, J.E.; Donahue, L.M.; Wu, Y.; Hazen, S.L. Prognostic value of elevated levels of intestinal microbe-generated metabolite trimethylamine-N-oxide in patients with heart failure: Refining the gut hypothesis. J. Am. Coll. Cardiol. 2014, 64, 1908–1914. [Google Scholar] [CrossRef]
- Tarasov, K.; Ekroos, K.; Suoniemi, M.; Kauhanen, D.; Sylvänne, T.; Hurme, R. Molecular lipids identify cardiovascular risk and are efficiently lowered by simvastatin and PCSK9 deficiency. J. Clin. Endocrinol. Metab. 2014, 99, E45–E52. [Google Scholar] [CrossRef] [PubMed]
- Tzoulaki, I.; Castagne, R.; Boulange, C.L.; Karaman, I.; Chekmeneva, E.; Evangelou, E.; Ebbels, T.M.D.; Kaluarachchi, M.R.; Chadeau-Hyam, M.; Mosen, D.; et al. Serum metabolic signatures of coronary and carotid atherosclerosis and subsequent cardiovascular disease. Eur. Heart J. 2019, 40, 2883–2896. [Google Scholar] [CrossRef] [PubMed]
- Vaarhorst, A.A.; Verhoeven, A.; Weller, C.M.; Bohringer, S.; Goraler, S.; Meissner, A.; Deelder, A.M.; Henneman, P.; Gorgels, A.P.; van den Brandt, P.A.; et al. A metabolomic profile is associated with the risk of incident coronary heart disease. Am. Heart J. 2014, 168, 45–52 e7. [Google Scholar] [CrossRef]
- Vorkas, P.A.; Isaac, G.; Holmgren, A.; Want, E.J.; Shockcor, J.P.; Holmes, E.; Henein, M.Y. Perturbations in fatty acid metabolism and apoptosis are manifested in calcific coronary artery disease: An exploratory lipidomic study. Int. J. Cardiol. 2015, 197, 192–199. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.D.; Zheng, Y.; Toledo, E.; Razquin, C.; Ruiz-Canela, M.; Guasch-Ferre, M.; Yu, E.; Corella, D.; Gomez-Gracia, E.; Fiol, M.; et al. Lipid metabolic networks, Mediterranean diet and cardiovascular disease in the PREDIMED trial. Int. J. Epidemiol. 2018, 47, 1830–1845. [Google Scholar] [CrossRef]
- Wang, Z.; Klipfell, E.; Bennett, B.J.; Koeth, R.; Levison, B.S.; Dugar, B.; Feldstein, A.E.; Britt, E.B.; Fu, X.; Chung, Y.M.; et al. Gut flora metabolism of phosphatidylcholine promotes cardiovascular disease. Nature 2011, 472, 57–63. [Google Scholar] [CrossRef]
- Wang, D.D.; Toledo, E.; Hruby, A.; Rosner, B.A.; Willett, W.C.; Sun, Q.; Razquin, C.; Zheng, Y.; Ruiz-Canela, M.; Guasch-Ferre, M.; et al. Plasma Ceramides, Mediterranean Diet, and Incident Cardiovascular Disease in the PREDIMED Trial (Prevencion con Dieta Mediterranea). Circulation 2017, 135, 2028–2040. [Google Scholar] [CrossRef]
- Wittenbecher, C.; Eichelmann, F.; Toledo, E.; Guasch-Ferre, M.; Ruiz-Canela, M.; Li, J.; Aros, F.; Lee, C.H.; Liang, L.; Salas-Salvado, J.; et al. Lipid Profiles and Heart Failure Risk: Results From Two Prospective Studies. Circ. Res. 2021, 128, 309–320. [Google Scholar] [CrossRef]
- Wurtz, P.; Havulinna, A.S.; Soininen, P.; Tynkkynen, T.; Prieto-Merino, D.; Tillin, T.; Ghorbani, A.; Artati, A.; Wang, Q.; Tiainen, M.; et al. Metabolite profiling and cardiovascular event risk: A prospective study of 3 population-based cohorts. Circulation 2015, 131, 774–785. [Google Scholar] [CrossRef]
- Zordoky, B.N.; Sung, M.M.; Ezekowitz, J.; Mandal, R.; Han, B.; Bjorndahl, T.C.; Bouatra, S.; Anderson, T.; Oudit, G.Y.; Wishart, D.S.; et al. Metabolomic fingerprint of heart failure with preserved ejection fraction. PLoS ONE 2015, 10, e0124844. [Google Scholar] [CrossRef]
- Bowling, F.G.; Thomas, M. Analyzing the metabolome. Methods Mol. Biol. 2014, 1168, 31–45. [Google Scholar] [PubMed]
- Karu, N.; Deng, L.; Slae, M.; Guo, A.C.; Sajed, T.; Huynh, H.; Wine, E.; Wishart, D.S. A review on human fecal metabolomics: Methods, applications and the human fecal metabolome database. Anal. Chim. Acta 2018, 1030, 1–24. [Google Scholar] [CrossRef] [PubMed]
- Lind, M.V.; Savolainen, O.I.; Ross, A.B. The use of mass spectrometry for analysing metabolite biomarkers in epidemiology: Methodological and statistical considerations for application to large numbers of biological samples. Eur. J. Epidemiol. 2016, 31, 717–733. [Google Scholar] [CrossRef] [PubMed]
- Yu, Z.; Kastenmüller, G.; He, Y.; Belcredi, P.; Möller, G.; Prehn, C.; Mendes, J.; Wahl, S.; Roemisch-Margl, W.; Ceglarek, U.; et al. Differences between human plasma and serum metabolite profiles. PLoS ONE 2011, 6, e21230. [Google Scholar] [CrossRef]
- Marchand, C.R.; Farshidfar, F.; Rattner, J.; Bathe, O.F. A Framework for Development of Useful Metabolomic Biomarkers and Their Effective Knowledge Translation. Metabolites 2018, 8, 59. [Google Scholar] [CrossRef] [PubMed]
- Yin, P.; Peter, A.; Franken, H.; Zhao, X.; Neukamm, S.S.; Rosenbaum, L.; Lucio, M.; Zell, A.; Häring, H.U.; Xu, G.; et al. Preanalytical aspects and sample quality assessment in metabolomics studies of human blood. Clin. Chem. 2013, 59, 833–845. [Google Scholar] [CrossRef]
- Kamlage, B.; Neuber, S.; Bethan, B.; González Maldonado, S.; Wagner-Golbs, A.; Peter, E.; Schmitz, O.; Schatz, P. Impact of Prolonged Blood Incubation and Extended Serum Storage at Room Temperature on the Human Serum Metabolome. Metabolites 2018, 8, 6. [Google Scholar] [CrossRef]
- Wishart, D.S.; Tzur, D.; Knox, C.; Eisner, R.; Guo, A.C.; Young, N.; Cheng, D.; Jewell, K.; Arndt, D.; Sawhney, S.; et al. HMDB: The Human Metabolome Database. Nucleic Acids Res. 2007, 35, D521–D526. [Google Scholar] [CrossRef]
- Smith, C.A.; O’Maille, G.; Want, E.J.; Qin, C.; Trauger, S.A.; Brandon, T.R.; Custodio, D.E.; Abagyan, R.; Siuzdak, G. METLIN: A metabolite mass spectral database. Ther. Drug Monit. 2005, 27, 747–751. [Google Scholar] [CrossRef]
- Wishart, D.S.; Jewison, T.; Guo, A.C.; Wilson, M.; Knox, C.; Liu, Y.; Djoumbou, Y.; Mandal, R.; Aziat, F.; Dong, E.; et al. HMDB 3.0—The Human Metabolome Database in 2013. Nucleic Acids Res. 2013, 41, D801–D807. [Google Scholar] [CrossRef]
- Horai, H.; Arita, M.; Kanaya, S.; Nihei, Y.; Ikeda, T.; Suwa, K.; Ojima, Y.; Tanaka, K.; Tanaka, S.; Aoshima, K.; et al. MassBank: A public repository for sharing mass spectral data for life sciences. J. Mass Spectrom. 2010, 45, 703–714. [Google Scholar] [CrossRef]
- Kind, T.; Liu, K.H.; Lee, D.Y.; DeFelice, B.; Meissen, J.K.; Fiehn, O. LipidBlast in silico tandem mass spectrometry database for lipid identification. Nat. Methods 2013, 10, 755–758. [Google Scholar] [CrossRef]
- Witting, M.; Ruttkies, C.; Neumann, S.; Schmitt-Kopplin, P. LipidFrag: Improving reliability of in silico fragmentation of lipids and application to the Caenorhabditis elegans lipidome. PLoS ONE 2017, 12, e0172311. [Google Scholar] [CrossRef] [PubMed]
- Hutchins, P.D.; Russell, J.D.; Coon, J.J. LipiDex: An Integrated Software Package for High-Confidence Lipid Identification. Cell Syst. 2018, 6, 621–625.e5. [Google Scholar] [CrossRef] [PubMed]
- Koelmel, J.P.; Kroeger, N.M.; Ulmer, C.Z.; Bowden, J.A.; Patterson, R.E.; Cochran, J.A.; Beecher, C.W.W.; Garrett, T.J.; Yost, R.A. LipidMatch: An automated workflow for rule-based lipid identification using untargeted high-resolution tandem mass spectrometry data. BMC Bioinform. 2017, 18, 331. [Google Scholar] [CrossRef]
- Xu, F.; Zou, L.; Ong, C.N. Multiorigination of chromatographic peaks in derivatized, G.C./MS metabolomics: A confounder that influences metabolic pathway interpretation. J. Proteome Res. 2009, 8, 5657–5665. [Google Scholar] [CrossRef]
- Xu, F.; Zou, L.; Ong, C.N. Experiment-originated variations, and multi-peak and multi-origination phenomena in derivatization-based, G.C.-MS metabolomics. TrAC Trends Anal. Chem. 2010, 29, 269–280. [Google Scholar] [CrossRef]
- Kanani, H.; Chrysanthopoulos, P.K.; Klapa, M.I. Standardizing GC-MS metabolomics. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 2008, 871, 191–201. [Google Scholar] [CrossRef]
- Misra, B.B. New software tools, databases, and resources in metabolomics: Updates from 2020. Metabolomics 2021, 17, 49. [Google Scholar] [CrossRef]
- Hsu, F.F. Mass spectrometry-based shotgun lipidomics—A critical review from the technical point of view. Anal. Bioanal. Chem. 2018, 410, 6387–6409. [Google Scholar] [CrossRef]
- Annesley, T.M. Ion suppression in mass spectrometry. Clin. Chem. 2003, 49, 1041–1044. [Google Scholar] [CrossRef] [PubMed]
- Mei, H.; Hsieh, Y.; Nardo, C.; Xu, X.; Wang, S.; Ng, K.; Korfmacher, W.A. Investigation of matrix effects in bioanalytical high-performance liquid chromatography/tandem mass spectrometric assays: Application to drug discovery. Rapid Commun. Mass Spectrom. 2003, 17, 97–103. [Google Scholar] [CrossRef]
- Trufelli, H.; Palma, P.; Famiglini, G.; Cappiello, A. An overview of matrix effects in liquid chromatography-mass spectrometry. Mass Spectrom. Rev. 2011, 30, 491–509. [Google Scholar] [CrossRef] [PubMed]
- Hu, C.; Duan, Q.; Han, X. Strategies to Improve/Eliminate the Limitations in Shotgun Lipidomics. Proteomics 2020, 20, e1900070. [Google Scholar] [CrossRef]
- Ryan, E.; Reid, G.E. Chemical Derivatization and Ultrahigh Resolution and Accurate Mass Spectrometry Strategies for “Shotgun” Lipidome Analysis. Acc. Chem. Res. 2016, 49, 1596–1604. [Google Scholar] [CrossRef] [PubMed]
- Han, X.; Yang, K.; Cheng, H.; Fikes, K.N.; Gross, R.W. Shotgun lipidomics of phosphoethanolamine-containing lipids in biological samples after one-step in situ derivatization. J. Lipid Res. 2005, 46, 1548–1560. [Google Scholar] [CrossRef] [PubMed]
- Thompson, J.W.; Adams, K.J.; Adamski, J.; Asad, Y.; Borts, D.; Bowden, J.A.; Byram, G.; Dang, V.; Dunn, W.B.; Fernandez, F.; et al. International Ring Trial of a High Resolution Targeted Metabolomics and Lipidomics Platform for Serum and Plasma Analysis. Anal. Chem. 2019, 91, 14407–14416. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Vosegaard, T.; Guo, Z. Applications of nuclear magnetic resonance in lipid analyses: An emerging powerful tool for lipidomics studies. Prog. Lipid Res. 2017, 68, 37–56. [Google Scholar] [CrossRef]
- Kirschenlohr, H.L.; Griffin, J.L.; Clarke, S.C.; Rhydwen, R.; Grace, A.A.; Schofield, P.M.; Brindle, K.M.; Metcalfe, J.C. Proton NMR analysis of plasma is a weak predictor of coronary artery disease. Nat. Med. 2006, 12, 705–710. [Google Scholar] [CrossRef]
- Hansen, A.L.; Kupče, E.R.; Li, D.W.; Bruschweiler-Li, L.; Wang, C.; Brüschweiler, R. 2D NMR-Based Metabolomics with HSQC/TOCSY NOAH Supersequences. Anal. Chem. 2021, 93, 6112–6119. [Google Scholar] [CrossRef]
- Mahrous, E.A.; Lee, R.B.; Lee, R.E. Lipid profiling using two-dimensional heteronuclear single quantum coherence NMR. Methods Mol. Biol. 2009, 579, 89–102. [Google Scholar] [PubMed]
- Sachleben, J.R.; Yi, R.; Volden, P.A.; Conzen, S.D. Aliphatic chain length by isotropic mixing (ALCHIM): Determining composition of complex lipid samples by ¹H NMR spectroscopy. J. Biomol. NMR 2014, 59, 161–173. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Tokunaga, T.; Okamoto, M. Recent progress in LC-NMR. Sumitomo Kagaku 2010, 2010, 40–48. [Google Scholar]
- Akira, K.; Mitome, H.; Imachi, M.; Shida, Y.; Miyaoka, H.; Hashimoto, T. LC-NMR identification of a novel taurine-related metabolite observed in (1)H NMR-based metabonomics of genetically hypertensive rats. J. Pharm. Biomed. Anal. 2010, 51, 1091–1096. [Google Scholar] [CrossRef] [PubMed]
- Cheng, S.; Shah, S.H.; Corwin, E.J.; Fiehn, O.; Fitzgerald, R.L.; Gerszten, R.E.; Illig, T.; Rhee, E.P.; Srinivas, P.R.; Wang, T.J.; et al. Potential Impact and Study Considerations of Metabolomics in Cardiovascular Health and Disease: A Scientific Statement From the American Heart Association. Circ. Cardiovasc. Genet. 2017, 10, e000032. [Google Scholar] [CrossRef]
- McGarrah, R.W.; Crown, S.B.; Zhang, G.F.; Shah, S.H.; Newgard, C.B. Cardiovascular Metabolomics. Circ. Res. 2018, 122, 1238–1258. [Google Scholar] [CrossRef]
- Dunn, W.B.; Broadhurst, D.; Begley, P.; Zelena, E.; Francis-McIntyre, S.; Anderson, N.; Brown, M.; Knowles, J.D.; Halsall, A.; Haselden, J.N.; et al. Procedures for large-scale metabolic profiling of serum and plasma using gas chromatography and liquid chromatography coupled to mass spectrometry. Nat. Protoc. 2011, 6, 1060–1083. [Google Scholar] [CrossRef]
- Di Guida, R.; Engel, J.; Allwood, J.W.; Weber, R.J.; Jones, M.R.; Sommer, U.; Viant, M.R.; Dunn, W.B. Non-targeted UHPLC-MS metabolomic data processing methods: A comparative investigation of normalisation, missing value imputation, transformation and scaling. Metabolomics 2016, 12, 93. [Google Scholar] [CrossRef]
- MacLean, B.; Tomazela, D.M.; Shulman, N.; Chambers, M.; Finney, G.L.; Frewen, B.; Kern, R.; Tabb, D.L.; Liebler, D.C.; MacCoss, M.J. Skyline: An open source document editor for creating and analyzing targeted proteomics experiments. Bioinformatics 2010, 26, 966–968. [Google Scholar] [CrossRef]
- Smith, C.A.; Want, E.J.; O’Maille, G.; Abagyan, R.; Siuzdak, G. XCMS: Processing mass spectrometry data for metabolite profiling using nonlinear peak alignment, matching, and identification. Anal. Chem. 2006, 78, 779–787. [Google Scholar] [CrossRef]
- Katajamaa, M.; Miettinen, J.; Oresic, M. MZmine: Toolbox for processing and visualization of mass spectrometry based molecular profile data. Bioinformatics 2006, 22, 634–636. [Google Scholar] [CrossRef] [PubMed]
- Xia, J.; Psychogios, N.; Young, N.; Wishart, D.S. MetaboAnalyst: A web server for metabolomic data analysis and interpretation. Nucleic Acids Res. 2009, 37, W652–W660. [Google Scholar] [CrossRef]
- Jacob, D.; Deborde, C.; Lefebvre, M.; Maucourt, M.; Moing, A. NMRProcFlow: A graphical and interactive tool dedicated to 1D spectra processing for NMR-based metabolomics. Metabolomics 2017, 13, 36. [Google Scholar] [CrossRef] [PubMed]
- Cuperlovic-Culf, M. Machine Learning Methods for Analysis of Metabolic Data and Metabolic Pathway Modeling. Metabolites 2018, 8, 4. [Google Scholar] [CrossRef]
- Harrell, F.E.; Califf, R.M.; Pryor, D.B.; Lee, K.L.; Rosati, R.A. Evaluating the yield of medical tests. JAMA 1982, 247, 2543–2546. [Google Scholar] [CrossRef]
- Pencina, M.J.; D’Agostino, R.B.; Steyerberg, E.W. Extensions of net reclassification improvement calculations to measure usefulness of new biomarkers. Stat. Med. 2011, 30, 11–21. [Google Scholar] [CrossRef] [PubMed]
- Düngen, H.D.; Tscholl, V.; Obradovic, D.; Radenovic, S.; Matic, D.; Musial Bright, L.; Tahirovic, E.; Marx, A.; Inkrot, S.; Hashemi, D.; et al. Prognostic performance of serial in-hospital measurements of copeptin and multiple novel biomarkers among patients with worsening heart failure: Results from the MOLITOR study. ESC Heart Fail. 2018, 5, 288–296. [Google Scholar] [CrossRef]
- Krackhardt, F.; Düngen, H.D.; Trippel, T.D.; Inkrot, S.; Tscholl, V.; Schlattmann, P.; Kehrt, K.; Haverkamp, W. N-terminal pro-B-type natriuretic peptide and long-term mortality in non-ischaemic cardiomyopathy. Wien. Klin. Wochenschr. 2011, 123, 738–742. [Google Scholar] [CrossRef] [PubMed]
- Loncar, G.; Bozic, B.; Neskovic, A.N.; Cvetinovic, N.; Lainscak, M.; Prodanovic, N.; Dungen, H.D.; von Haehling, S.; Radojicic, Z.; Trippel, T.; et al. Androgen status in non-diabetic elderly men with heart failure. Aging Male 2017, 20, 215–224. [Google Scholar] [CrossRef]
- Kato, T.; Niizuma, S.; Inuzuka, Y.; Kawashima, T.; Okuda, J.; Tamaki, Y.; Iwanaga, Y.; Narazaki, M.; Matsuda, T.; Soga, T.; et al. Analysis of metabolic remodeling in compensated left ventricular hypertrophy and heart failure. Circ. Heart Fail. 2010, 3, 420–430. [Google Scholar] [CrossRef] [PubMed]
- McGranaghan, P.; Saxena, A.; Rubens, M.; Radenkovic, J.; Bach, D.; Schleussner, L.; Pieske, B.; Edelmann, F.; Trippel, T.D. Predictive value of metabolomic biomarkers for cardiovascular disease risk: A systematic review and meta-analysis. Biomarkers 2020, 25, 101–111. [Google Scholar] [CrossRef]
- Ruiz-Canela, M.; Hruby, A.; Clish, C.B.; Liang, L.; Martinez-Gonzalez, M.A.; Hu, F.B. Comprehensive Metabolomic Profiling and Incident Cardiovascular Disease: A Systematic Review. J. Am. Heart Assoc. 2017, 6, e005705. [Google Scholar] [CrossRef] [PubMed]
- Grösch, S.; Schiffmann, S.; Geisslinger, G. Chain length-specific properties of ceramides. Prog. Lipid Res. 2012, 51, 50–62. [Google Scholar] [CrossRef]
- Bremer, J. Carnitine–metabolism and functions. Physiol. Rev. 1983, 63, 1420–1480. [Google Scholar] [CrossRef]
- Nickel, A.; Löffler, J.; Maack, C. Myocardial energetics in heart failure. Basic Res. Cardiol. 2013, 108, 358. [Google Scholar] [CrossRef]
- Carley, A.N.; Maurya, S.K.; Fasano, M.; Wang, Y.; Selzman, C.H.; Drakos, S.G.; Lewandowski, E.D. Short-Chain Fatty Acids Outpace Ketone Oxidation in the Failing Heart. Circulation 2021, 143, 1797–1808. [Google Scholar] [CrossRef]
- Trøseid, M.; Andersen, G.; Broch, K.; Hov, J.R. The gut microbiome in coronary artery disease and heart failure: Current knowledge and future directions. EBioMedicine 2020, 52, 102649. [Google Scholar] [CrossRef]
- Lopaschuk, G.D.; Ussher, J.R.; Folmes, C.D.; Jaswal, J.S.; Stanley, W.C. Myocardial fatty acid metabolism in health and disease. Physiol. Rev. 2010, 90, 207–258. [Google Scholar] [CrossRef]
- Thies, F.; Garry, J.M.; Yaqoob, P.; Rerkasem, K.; Williams, J.; Shearman, C.P.; Gallagher, P.J.; Calder, P.C.; Grimble, R.F. Association of n-3 polyunsaturated fatty acids with stability of atherosclerotic plaques: A randomised controlled trial. Lancet 2003, 361, 477–485. [Google Scholar] [CrossRef]
- Renier, G.; Skamene, E.; DeSanctis, J.; Radzioch, D. Dietary n-3 polyunsaturated fatty acids prevent the development of atherosclerotic lesions in mice. Modulation of macrophage secretory activities. Arterioscler. Thromb. 1993, 13, 1515–1524. [Google Scholar] [CrossRef] [PubMed]
- von Schacky, C.; Angerer, P.; Kothny, W.; Theisen, K.; Mudra, H. The effect of dietary omega-3 fatty acids on coronary atherosclerosis. A randomized, double-blind, placebo-controlled trial. Ann. Intern. Med. 1999, 130, 554–562. [Google Scholar] [CrossRef] [PubMed]
- Kotwal, S.; Jun, M.; Sullivan, D.; Perkovic, V.; Neal, B. Omega 3 Fatty acids and cardiovascular outcomes: Systematic review and meta-analysis. Circ. Cardiovasc. Qual. Outcomes 2012, 5, 808–818. [Google Scholar] [CrossRef] [PubMed]
- Jakobsen, M.U.; O’Reilly, E.J.; Heitmann, B.L.; Pereira, M.A.; Bälter, K.; Fraser, G.E. Major types of dietary fat and risk of coronary heart disease: A pooled analysis of 11 cohort studies. Am. J. Clin. Nutr. 2009, 89, 1425–1432. [Google Scholar] [CrossRef] [PubMed]
- Mozaffarian, D.; Micha, R.; Wallace, S. Effects on coronary heart disease of increasing polyunsaturated fat in place of saturated fat: A systematic review and meta-analysis of randomized controlled trials. PLoS Med. 2010, 7, e1000252. [Google Scholar] [CrossRef] [PubMed]
- Roncaglioni, M.C.; Tombesi, M.; Avanzini, F.; Barlera, S.; Caimi, V.; Longoni, P.; Marzona, I.; Milani, V.; Silletta, M.G.; Tognoni, G.; et al. n-3 fatty acids in patients with multiple cardiovascular risk factors. N. Engl. J. Med. 2013, 368, 1800–1808. [Google Scholar]
- Rizos, E.C.; Ntzani, E.E.; Bika, E.; Kostapanos, M.S.; Elisaf, M.S. Association between omega-3 fatty acid supplementation and risk of major cardiovascular disease events: A systematic review and meta-analysis. JAMA 2012, 308, 1024–1033. [Google Scholar] [CrossRef]
- Trippel, T.D.; Anker, S.D.; von Haehling, S. The role of micronutrients and macronutrients in patients hospitalized for heart failure. Heart Fail. Clin. 2013, 9, 345–357. [Google Scholar] [CrossRef]
- Tallman, K.A.; Kim, H.Y.; Ji, J.X.; Szapacs, M.E.; Yin, H.; McIntosh, T.J.; Liebler, D.C.; Porter, N.A. Phospholipid-protein adducts of lipid peroxidation: Synthesis and study of new biotinylated phosphatidylcholines. Chem. Res. Toxicol. 2007, 20, 227–234. [Google Scholar] [CrossRef]
- Reis, A.; Spickett, C.M. Chemistry of phospholipid oxidation. Biochim. Biophys. Acta 2012, 1818, 2374–2387. [Google Scholar] [CrossRef]
- Gianazza, E.; Brioschi, M.; Fernandez, A.M.; Banfi, C. Lipoxidation in cardiovascular diseases. Redox Biol. 2019, 23, 101119. [Google Scholar] [CrossRef]
- Berliner, J.A.; Watson, A.D. A role for oxidized phospholipids in atherosclerosis. N. Engl. J. Med. 2005, 353, 9–11. [Google Scholar] [CrossRef] [PubMed]
- Camont, L.; Lhomme, M.; Rached, F.; Le Goff, W.; Nègre-Salvayre, A.; Salvayre, R.; Calzada, C.; Lagarde, M. Small, dense high-density lipoprotein-3 particles are enriched in negatively charged phospholipids: Relevance to cellular cholesterol efflux, antioxidative, antithrombotic, anti-inflammatory, and antiapoptotic functionalities. Arterioscler. Thromb. Vasc. Biol. 2013, 33, 2715–2723. [Google Scholar] [CrossRef] [PubMed]
- Ding, M.; Rexrode, K.M. A Review of Lipidomics of Cardiovascular Disease Highlights the Importance of Isolating Lipoproteins. Metabolites 2020, 10, 163. [Google Scholar] [CrossRef] [PubMed]
- Lessig, J.; Fuchs, B. Plasmalogens in biological systems: Their role in oxidative processes in biological membranes, their contribution to pathological processes and aging and plasmalogen analysis. Curr. Med. Chem. 2009, 16, 2021–2041. [Google Scholar] [CrossRef]
- Holzer, M.; Birner-Gruenberger, R.; Stojakovic, T.; El-Gamal, D.; Binder, V.; Wadsack, C.; Heinemann, A.; Marsche, G. Uremia alters HDL composition and function. J. Am. Soc. Nephrol. 2011, 22, 1631–1641. [Google Scholar] [CrossRef]
- Janeiro, M.H.; Ramírez, M.J.; Milagro, F.I.; Martínez, J.A.; Solas, M. Implication of Trimethylamine N-Oxide (TMAO) in Disease: Potential Biomarker or New Therapeutic Target. Nutrients 2018, 10, 1398. [Google Scholar] [CrossRef] [PubMed]
- Zhu, W.; Wang, Z.; Tang, W.H.W.; Hazen, S.L. Gut Microbe-Generated Trimethylamine. Circulation 2017, 135, 1671–1673. [Google Scholar] [CrossRef]
- Senthong, V.; Wang, Z.; Li, X.S.; Fan, Y.; Wu, Y.; Tang, W.H.; Hazen, S.L. Intestinal Microbiota-Generated Metabolite Trimethylamine-N-Oxide and 5-Year Mortality Risk in Stable Coronary Artery Disease: The Contributory Role of Intestinal Microbiota in a COURAGE-Like Patient Cohort. J. Am. Heart Assoc. 2016, 5, e002816. [Google Scholar] [CrossRef]
- Tang, W.H.; Wang, Z.; Shrestha, K.; Borowski, A.G.; Wu, Y.; Troughton, R.W.; Klein, A.L.; Hazen, S.L. Intestinal microbiota-dependent phosphatidylcholine metabolites, diastolic dysfunction, and adverse clinical outcomes in chronic systolic heart failure. J. Card. Fail. 2015, 21, 91–96. [Google Scholar] [CrossRef]
- Organ, C.L.; Otsuka, H.; Bhushan, S.; Wang, Z.; Bradley, J.; Trivedi, R.; Polhemus, D.J.; Tang, W.H.; Wu, Y.; Hazen, S.L.; et al. Choline Diet and Its Gut Microbe-Derived Metabolite, Trimethylamine N-Oxide, Exacerbate Pressure Overload-Induced Heart Failure. Circ. Heart Fail. 2016, 9, e002314. [Google Scholar] [CrossRef] [PubMed]
- Senthong, V.; Wang, Z.; Fan, Y.; Wu, Y.; Hazen, S.L.; Tang, W.H. Trimethylamine N-Oxide and Mortality Risk in Patients with Peripheral Artery Disease. J. Am. Heart Assoc. 2016, 5, e004237. [Google Scholar] [CrossRef] [PubMed]
- Undurti, A.; Huang, Y.; Lupica, J.A.; Smith, J.D.; DiDonato, J.A.; Hazen, S.L. Modification of high density lipoprotein by myeloperoxidase generates a pro-inflammatory particle. J. Biol. Chem. 2009, 284, 30825–30835. [Google Scholar] [CrossRef] [PubMed]
- Coleman, R.A.; Lee, D.P. Enzymes of triacylglycerol synthesis and their regulation. Prog. Lipid Res. 2004, 43, 134–176. [Google Scholar] [CrossRef]
- Prentki, M.; Madiraju, S.R. Glycerolipid metabolism and signaling in health and disease. Endocr. Rev. 2008, 29, 647–676. [Google Scholar] [CrossRef]
- Tremblay, A.J.; Després, J.P.; Piché, M.E.; Nadeau, A.; Bergeron, J.; Alméras, N.; Tremblay, A.; Lemieux, S. Associations between the fatty acid content of triglyceride, visceral adipose tissue accumulation, and components of the insulin resistance syndrome. Metabolism 2004, 53, 310–317. [Google Scholar] [CrossRef]
- Kotronen, A.; Velagapudi, V.R.; Yetukuri, L.; Westerbacka, J.; Bergholm, R.; Ekroos, K.; Makkonen, J.; Taskinen, M.R.; Oresic, M.; Yki-Järvinen, H. Serum saturated fatty acids containing triacylglycerols are better markers of insulin resistance than total serum triacylglycerol concentrations. Diabetologia 2009, 52, 684–690. [Google Scholar] [CrossRef]
- Zhang, L.; Ussher, J.R.; Oka, T.; Cadete, V.J.; Wagg, C.; Lopaschuk, G.D. Cardiac diacylglycerol accumulation in high fat-fed mice is associated with impaired insulin-stimulated glucose oxidation. Cardiovasc. Res. 2011, 89, 148–156. [Google Scholar] [CrossRef]
- Koves, T.R.; Ussher, J.R.; Noland, R.C.; Slentz, D.; Mosedale, M.; Ilkayeva, O.; Bain, J.; Stevens, R.; Dyck, J.R.; Newgard, C.B.; et al. Mitochondrial overload and incomplete fatty acid oxidation contribute to skeletal muscle insulin resistance. Cell Metab. 2008, 7, 45–56. [Google Scholar] [CrossRef]
- Do, R.; Willer, C.J.; Schmidt, E.M.; Sengupta, S.; Gao, C.; Peloso, G.M.; Gustafsson, S.; Kanoni, S.; Ganna, A.; Chen, J.; et al. Common variants associated with plasma triglycerides and risk for coronary artery disease. Nat. Genet. 2013, 45, 1345–1352. [Google Scholar] [CrossRef] [PubMed]
- Jerome, W.G. Lysosomes, cholesterol and atherosclerosis. Clin. Lipidol. 2010, 5, 853–865. [Google Scholar] [CrossRef]
- Suarna, C.; Dean, R.T.; May, J.; Stocker, R. Human atherosclerotic plaque contains both oxidized lipids and relatively large amounts of alpha-tocopherol and ascorbate. Arterioscler. Thromb. Vasc. Biol. 1995, 15, 1616–1624. [Google Scholar] [CrossRef] [PubMed]
- Rudel, L.L.; Shelness, G.S. Cholesterol esters and atherosclerosis-a game of ACAT and mouse. Nat. Med. 2000, 6, 1313–1314. [Google Scholar] [CrossRef]
- Mallat, Z.; Nakamura, T.; Ohan, J.; Lesèche, G.; Tedgui, A.; Maclouf, J.; Murphy, R.C. The relationship of hydroxyeicosatetraenoic acids and F2-isoprostanes to plaque instability in human carotid atherosclerosis. J. Clin. Investig. 1999, 103, 421–427. [Google Scholar] [CrossRef] [PubMed]
- Degirolamo, C.; Shelness, G.S.; Rudel, L.L. LDL cholesteryl oleate as a predictor for atherosclerosis: Evidence from human and animal studies on dietary fat. J. Lipid Res. 2009, 50, S434–S439. [Google Scholar] [CrossRef]
- Gerl, M.J.; Vaz, W.L.C.; Domingues, N.; Klose, C.; Surma, M.A.; Sampaio, J.L.; Almeida, M.S.; Rodrigues, G.; Araújo-Gonçalves, P.; Ferreira, J.; et al. Cholesterol is Inefficiently Converted to Cholesteryl Esters in the Blood of Cardiovascular Disease Patients. Sci. Rep. 2018, 8, 14764. [Google Scholar] [CrossRef] [PubMed]
- Maceyka, M.; Spiegel, S. Sphingolipid metabolites in inflammatory disease. Nature 2014, 510, 58–67. [Google Scholar] [CrossRef]
- Edsfeldt, A.; Dunér, P.; Ståhlman, M.; Mollet, I.G.; Asciutto, G.; Grufman, H.; Nitulescu, M.; Persson, A.F.; Fisher, R.M.; Melander, O.; et al. Sphingolipids Contribute to Human Atherosclerotic Plaque Inflammation. Arterioscler. Thromb. Vasc. Biol. 2016, 36, 1132–1140. [Google Scholar] [CrossRef]
- Tirodkar, T.S.; Voelkel-Johnson, C. Sphingolipids in apoptosis. Exp. Oncol. 2012, 34, 231–242. [Google Scholar]
- van Heerebeek, L.; Borbély, A.; Niessen, H.W.; Bronzwaer, J.G.; van der Velden, J.; Stienen, G.J.; Linke, W.A.; Laarman, G.J.; Paulus, W.J. Myocardial structure and function differ in systolic and diastolic heart failure. Circulation 2006, 113, 1966–1973. [Google Scholar] [CrossRef] [PubMed]
- Li, X.M.; Ma, Y.T.; Yang, Y.N.; Liu, F.; Chen, B.D.; Han, W.; Zhang, J.F.; Gao, X.M. Downregulation of survival signalling pathways and increased apoptosis in the transition of pressure overload-induced cardiac hypertrophy to heart failure. Clin. Exp. Pharmacol. Physiol. 2009, 36, 1054–1061. [Google Scholar] [CrossRef] [PubMed]
- Sabbah, H.N.; Sharov, V.G.; Goldstein, S. Cell death, tissue hypoxia and the progression of heart failure. Heart Fail. Rev. 2000, 5, 131–138. [Google Scholar] [CrossRef] [PubMed]
- Schissel, S.L.; Tweedie-Hardman, J.; Rapp, J.H.; Graham, G.; Williams, K.J.; Tabas, I. Rabbit aorta and human atherosclerotic lesions hydrolyze the sphingomyelin of retained low-density lipoprotein. Proposed role for arterial-wall sphingomyelinase in subendothelial retention and aggregation of atherogenic lipoproteins. J. Clin. Investig. 1996, 98, 1455–1464. [Google Scholar] [CrossRef] [PubMed]
- Kauhanen, D.; Sysi-Aho, M.; Koistinen, K.M.; Laaksonen, R.; Sinisalo, J.; Ekroos, K. Development and validation of a high-throughput, L.C.-MS/MS assay for routine measurement of molecular ceramides. Anal. Bioanal. Chem. 2016, 408, 3475–3483. [Google Scholar] [CrossRef]
- Wishart, D.S. Emerging applications of metabolomics in drug discovery and precision medicine. Nat. Rev. Drug Discov. 2016, 15, 473–484. [Google Scholar] [CrossRef] [PubMed]
- Dang, V.T.; Huang, A.; Werstuck, G.H. Untargeted Metabolomics in the Discovery of Novel Biomarkers and Therapeutic Targets for Atherosclerotic Cardiovascular Diseases. Cardiovasc. Hematol. Disord. Drug Targets. 2018, 18, 166–175. [Google Scholar] [CrossRef]
- Karlstädt, A.; Fliegner, D.; Kararigas, G.; Ruderisch, H.S.; Regitz-Zagrosek, V.; Holzhütter, H.G. CardioNet: A human metabolic network suited for the study of cardiomyocyte metabolism. BMC Syst. Biol. 2012, 6, 114. [Google Scholar] [CrossRef] [PubMed]
| First Author, Year | Study Design | Sample Matrix | Platform | Targeted vs. Untargeted | Outcome | Cohort Characteristics | Candidate Lipid or Lipid-Related Biomarkers |
|---|---|---|---|---|---|---|---|
| Ahmad, 2016 [4] | Case-cohort | Plasma | FIA-MS/MS | Not Specified | CHF Death/Event | CHF patients; 29% female; 64 mean age; 67% white | Long chain acylcarnitines |
| Alshehry, 2016 [5] | Case-cohort | Plasma | LC-MS/MS | Targeted | Incident CVD in T2DM | 2 cohorts of T2DM patients; 39% female; 67 mean age; 20 countries from Asia, Australasia, Europe, and North America | PC(O-36:1), CE(18:0), PE(O-36:4), PC(28:0), LPC(20:0), PC(35:4), LPC(18:2), DG(16:0_22:5), SM (34:1), PC (O-36:5) |
| Andersson, 2020 [6] | Cohort, prospective | Plasma | LC-MS/MS | Targeted | Incident HF | Community-based cohort; 53% female; 55 mean age; MA, USA cohort | PC 36:4, LPC 18:2 |
| Anroedh, 2018 [7] | Case-cohort | Plasma | LC-MS/MS, FIA-MS/MS | Targeted | CVD event/death | Patients who underwent diagnostic CAG or PCI for ACS or stable angina pectoris; 25% female; 62 mean age; Netherlands medical center | Cer(d18:1/16:0), Cer(d18:1/20:0), Cer(d18:1/24:1), Cer(d18:1/24:0) |
| Cavus, 2019 [8] | Case-cohort | Serum | LC-MS/MS, FIA-MS/MS | Targeted | Incident CHD | Population-based cohort; 39% female; 57 mean age; 6 European cohorts: Finland, 2 Italy cohorts, Germany, Denmark, Scotland | acyl-alkyl-PC C40:6, diacyl-PC C40:6, acyl-alkyl-PC C38:6, diacyl-PC C38:6, and diacyl-PC |
| Cheng, 2015 [9] | Case-control | Plasma | LC-MS/MS, FIA-MS/MS | Untargeted and Targeted | CHF Diagnosis | CHF patients; 27% female; 61 mean age; Taiwan medical center | PC C34:4 |
| Cheng-Laaksonen, 2015 [10] | Case-cohort | Plasma | LC-MS/MS | Targeted | CVD event/death | Patients who underwent diagnostic CAG or PCI for ACS or stable angina pectoris; 25% female; 62 mean age; Netherlands medical center | Cer-d18:1/16:0 |
| Delles, 2018 [11] | Case-cohort | Serum | NMR | Targeted | Incident HF hospitalization | Elderly individuals at high risk of CVD; 52% female; 77 mean age; 1 Scotland, 1 Ireland, 1 Netherlands cohort | SCFA (acetate), phenylalanine |
| Fernandez, 2013 [12] | Case-control | Plasma | FIA-MS/MS | Targeted | Incident CVD | Population-based cohort; 47% female; 60 mean age; Swedish cohort | LPC16:0, LPC20:4, SM 38:2, TG48:1, TG48:2, TG48:3, TG50:3, TG50:4 |
| Floegel, 2018 [13] | Cohort, prospective | Plasma | LC-MS/MS, FIA-MS/MS | Targeted | Incident MI | 2 Population-based cohorts; 61% female; 49 mean age; 2 German cohorts | Acylalkyl-PC (C36:3), diacyl-PC (C38:3 and C40:4) |
| Ganna, 2014 [14] | Cohort, prospective | Plasma | LC-MS/MS | Untargeted | Incident CVD | 3 Population-based cohorts; 37% female; 69 mean age; Northern European | LPC-18:1, LPC-18:2, MG (18:2), and SM-28:1 |
| Gao, 2017 [15] | Case-control | Plasma | LC-MS/MS | Untargeted | Incident CAD | Patients undergoing diagnostic CAG; 49% female; 59 mean age; Chinese medical center | LPC (20:4), LPC (16:0), PG(18:0/0:0), elaidic acid, MG (0:0/18:2(9Z,12Z)/0:0), DG (20:2(11Z,14Z)/18:3(9Z,12Z,15Z)/0:0) |
| Havulinna, 2016 [16] | Cohort, prospective | Serum | LC-MS/MS | Targeted | Incident CVD | Population-based cohort; 53% female; 49 mean age; Finnish cohort | Cer-d18:1/18:0 |
| Hilvo, 2020 [17] | Cohort, prospective | Plasma and Serum | LC-MS/MS | Targeted | CVD event/death | 3 CHD cohorts; 21% female; 65 mean age; 1 Norwegian, 1 German, 1 Australian cohort | Cer(d18:1/16:0), Cer(d18:1/18:0), Cer(d18:1/24:1), Cer(d18:1/24:0), PC(16:0/16:0), PC(16:0/22:5), PC(14:0/22:6) |
| Holmes, 2018 [18] | Nested case-control | Plasma | NMR | Targeted | Incident CVD | Population-based cohort; 52% female; 45 mean age; Chinese cohort | Total FA, omega-6 FA, linoleic acid, PUFA |
| Jadoon, 2018 [19] | Case-cohort | Serum | LC-MS/MS | Targeted | CKD + Incident CVD | CKD patients; 49% female; 62 mean age; 70% white | SCFA (valerate) |
| Ji, 2018 [20] | Case-control | Serum | LC-MS/MS | Targeted | CHF progression | CHF patients; 20% female; 57 mean age; NY, USA medical center | Cer16, Cer18, Cer20:1, Cer20, Cer22:1, and Cer24:1 |
| Kalim, 2013 [21] | Nested case-control | Plasma | LC-MS/MS | Targeted | CVD death | Hemodialysis patients; 47% female; 70 mean age; 69% white | Oleoylcarnitine (C18:1) |
| Laaksonen, 2016 [22] | Case-cohort | Plasma | LC-MS/MS | Targeted | CVD death | Patients undergoing CAG; 31% female; 69 mean age; Finnish, Norwegian, and Swiss cohorts | Cer(d18:1/16:0), Cer(d18:1/24:1), Cer(d18:1/16:0)/Cer(d18:1/24:0), Cer(d18:1/18:0)/Cer(d18:1/24:0), Cer(d18:1/24:1)/Cer(d18:1/24:0) |
| Lemaitre, 2019 [23] | Cohort, prospective | Plasma | LC-MS/MS | Targeted | Incident HF | Population-based cohort; 60% female; 76 mean age; 16% black from 4 US communities NC, CA, MD, PA | Cer-16, SM-16, Cer-22, SM-20, SM-22, and SM-24 |
| Lu, 2017 [24] | Case-control | Plasma | LC-MS | Untargeted and Targeted | MI | MI and stable angina patients; 75% female; 59 mean age; China medical center | 9 oxyphospholipids (HODA-PC, KDdiA-PC, D2/E2-IsoP-PC, PEIPC, HETE-PC, IsoF-PC, PECPC, F2-IsoP-PC, HODE-PC), 9 hydrolyzed FA (20-HETE, 11,12 DHET, 13-HODE, 5-HETE, D2/E2-IsoP, 14,15-DHET, 5,6-DHET, 14(15)-EET, 9-HODE) |
| Mayerhofer, 2020 [25] | Case-control | Plasma | LC-MS/MS, GC-MS | Targeted | All-cause mortality or listing for heart transplant | CHF patients; 59% female; 59 median age; Norway cohort | TMAO, SCFA (butyrate) |
| McGranaghan, 2020, 2021 [26,27] | Case-cohort | Serum | LC-MS/MS, GC-MS | Untargeted and Targeted | CHF Death | CHF patients; 26% female; 72 mean age; German medical center | SM d18:1/23:1, SM d18:2/23:0, SM d17:1/24:1, TG 18:1/18:0/18:0, PC 16:0/18:2 |
| Meikle, 2011 [28] | Cross-sectional | Plasma | LC-MS/MS | Targeted | unstable CAD/stable CAD | de Novo CAD patients; 22% female; 66 mean age; Australian cohort | 10 species of PE(O) |
| Miller, 2012 [29] | Cohort, prospective | Plasma | LC-MS/MS | Not Specified | Incident CAD | Chest pain or angina patients; 38% female; 48 mean age; 72% white | CE 16:1, CE 18:1 |
| Mueller-Hennessen, 2017 [30] | Cohort, prospective | Plasma | LC-MS/MS, GC-MS | Untargeted and Targeted | Incident HF | CHF patients; 30% female; 59 mean age; 3 German medical centers | SM d18:1/23:1, SM d18:2/23:0, SM d17:1/24:1, TG 18:1/18:0/18:0, PC 16:0/18:2 |
| Mueller-Hennessen, 2017 [31] | Case-control | Plasma | LC-MS/MS, GC-MS | Untargeted and Targeted | CHF Diagnosis | CHF patients; 0% female; 50 mean age; Germany medical center | Cholesterol, Behenic acid (C22:0), Lignoceric acid (C24:0), Linoleic acid (C18:cis [9,12] 2), Tricosanoic acid (C23:0), LPC (C17:0), LPC (C18:0), LPC (C18:1), LPC (C18:2), PC (C16:1, C18:2), 5-O-Methylsphingosine, erythro-Sphingosine, Phytosphingosine |
| Mundra, 2018 [32] | Case-cohort | Plasma | LC-MS/MS | Targeted | CVD event/death | Patients with MI or unstable angina; 18% female; 63 median age; Australia and New Zealand medical centers | PC (O-34:2), PC (38:5), PI (38:3), PC (O-36:1), GM3(d18:1/16:0), PI (18:2/0:0), PE (38:6) |
| Nwabuo, 2019 [33] | Cross-sectional | Plasma | LC-MS/MS | Targeted | Echo measures correlation | Community-based cohort; 65% female; 66 mean age; MA, USA community | Cer16:0/Cer24:0 |
| Ottosson, 2021 [34] | Case-control | Plasma | FIA-MS/MS | Untargeted | Incident CAD | Population-based cohort; 60% female; 58 mean age; Swedish cohort | PC 15:0;0_18:2;0, PC 17:0;0_20:3;0, PC 16:0;0_20:1;0, PC O 16:2;0_18:0;0, SM 34:1;2, DAG 18:1;0_18:3;0, PI 16:0;0_20:4;0; CE 18:0;0 |
| Paapstel, 2017 [35] | Case-control | Serum | LC-MS/MS, FIA-MS/MS | Targeted | Atherosclerosis | PAD and CAD patients; 0% female; 63 mean age; Estonia medical center | PC-diacyl-28:1, PC-diacyl-30:0, PC-diacyl-32:2, PC-acyl-alkyl-30:0, PC-acyl-alkyl-34:2, LPC-acyl-18:2 |
| Paynter, 2018 [36] | Case-control | Plasma | LC-MS, LC-MS/MS | Untargeted | Incident CVD | Post-menopausal women cohort; 100% female; 67 mean age; 77% white | Hydroxy-PC (C34:2) |
| Peterson, 2018 [37] | Case-control | Plasma | LC-MS/MS | Targeted | Incident CVD; HF | 2 Community-based cohorts; 53% female; 60 mean age; 2 US communities MO and MA | C24:0/C16:0 |
| Poss, 2020 [38] | Case-control | Serum | LC-MS/MS | Targeted | Incident CAD | CAD patients; 34% female; 55 mean age; UT, USA medical center | dihydro-cer(d18:0/18:0), cer(d18:1/18:0), cer(d18:1/22:0), cer(d18:1/24:0), dihydro-SM(d18:0/24:1), SM(d18:1/24:0), SM(d18:1/18:0), and sphingosine |
| Razquin, 2017 [39] | Case-cohort | Plasma | LC-MS | Untargeted | Incident CVD | Population-based cohort; 57% female; 67 mean age; Spanish cohort | Polyunsaturated PCs, LPCs, PC-plasmalogens, CEs, long TGs, short TGs (saturated/monounsaturated), hPCs and, MGs, DGs and PEs |
| Rizza, 2014 [40] | Cohort, prospective | Serum | LC-MS/MS, FIA-MS/MS | Targeted | CVD event/death | Geriatric ambulatory patients; 43% female; 77 mean age; Italian medical center | medium-long-chain acylcarnitines (acetyl carnitine C2, C6, C8, C10, C10:1, C12, C12:1, C14, C14:1, C14:2, C16, C16:1, C18:1, C18:2) |
| Seah, 2020 [41] | Cohort, prospective | Plasma | LC-MS/MS | Targeted | CVD event/death | Population-based cohort; 53% female; 49 mean age; Singapore Chinese cohort | total monohexoylceramides, total long-chain sphingolipids (C16–C18), and total 18:1 sphingolipids |
| Shah, 2010 [42] | Cohort, prospective repository | Plasma | LC-MS/MS | Targeted | CVD event/death | Cardiac catheterization patients; 24% female; 46 mean age; 67% white | Short-chain dicarboxylacylcarnitines; medium-chain acylcarnitines |
| Shah, 2012 [43] | Cohort, prospective | Plasma | LC-MS/MS | Targeted | All-cause mortality or MI | Cardiac catheterization patients; 38% female; 62 median age; 73% white | Short-chain dicarboxylacylcarnitines, Long-chain dicarboxylacylcarnitines, Fatty acids |
| Sigruener, 2014 [44] | Cohort, prospective | Plasma | FIA-MS/MS | Targeted | Mortality | Hospitalized coronary angiography patients; 30% female; 63 mean age; 100% white | PC-32:0, SM-16:0, SM-24:1 and CM-24:1 |
| Stegemann, 2011 [45] | Case-control | Plaque; Plasma | FIA-MS/MS | Targeted | Atherosclerosis | Endarterectomy patients; 29% female; 69 mean age; British cohort | 10 CEs, 9 SMs, 8 LPCs, and 31 PCs |
| Stegemann, 2014 [46] | Cohort, prospective | Plasma | FIA-MS/MS | Targeted | Incident CVD | Population-based cohort; 52% female; 66 mean age; 100% white | TG-54:2, CE-16:1, and PC-36:5 |
| Stenemo, 2019 [47] | Cohort, observational | Plasma and Serum | LC-MS/MS | Untargeted | Incident HF | 3 Community-based cohorts; 33% female; 70 mean age; 3 Sweden cohorts | SM (30:1) |
| Sun, 2016 [48] | Nested case-control, prospective | Plasma | GC-MS/MS | Targeted | Incident MI | Population-based cohort; 35% female; 66 mean age; Singapore Chinese cohort | Long-chain n-3 fatty acids, stearic acid, and arachidonic acid |
| Syme, 2016 [49] | Cohort, observational | Serum | LC-MS/MS | Untargeted | Incident CVD | Population-based cohort; 52% female; 15 median age; Canadian Cohort | PC-16:0/2:0, PC-14:1/0:0 |
| Tang, 2013 [50] | Cohort, prospective | Plasma | LC-MS/MS | Targeted | CVD event/death | Cardiac catheterization patients; 36% female; 63 mean age; Cleveland, Ohio USA Medical Center | TMAO |
| Tang, 2014 [51] | Cohort, prospective | Plasma | LC-MS/MS | Targeted | All-cause mortality IN CHF | Patients who underwent diagnostic CAG; 41% female; 66 mean age; Cleveland, Ohio USA Medical Center | TMAO |
| Tarasov, 2014 [52] | Case-control | Serum | LC-MS/MS, FIA-MS/MS | Targeted | CVD Death | CAD patients; 0% female; 66 mean age; German medical center | Cer(d18:1/16:0)/Cer(d18:1/24:0), Cer(d18:1/20:0)/Cer(d18:1/24:0), Cer(d18:1/24:0)/Cer(d18:1/24:1) |
| Tzoulaki, 2019 [53] | Cohort, prospective | Serum | NMR | Untargeted | Atherosclerosis/Incident CVD | 3 Population-based cohorts; 47% female; 63 mean age; 53% white | Triglycerides, Phospholipids, CE |
| Vaarhorst, 2014 [54] | Case-cohort, prospective | Plasma | NMR | Untargeted | Incident CVD | Population-based cohort; 51% female; 49 mean age; Netherlands cohort | TMAO, an unsaturated lipid structure |
| Vorkas, 2015 [55] | Cross-sectional | Serum | LC-MS/MS | Untargeted | Calcific CAD | Exertional angina patients; 59% female; 65 mean age; Sweden medical center | PC(16:0/20:4), lysoPC(20:4), PI(18:2/18:0), SM(d17:1/16:0), SM(d18:1/16:0), SM(d17:1/22:0), SM(d18:1/23:0), SM(d18:2/16:0), SM(d18:2/22:0), SM(d18:2/24:1), TG(16:0/18:1/22:5), TG(18:1/18:1/20:4), TG(16:0/18:1/18:1) |
| Wang-Dong, 2018 [56] | Case-cohort | Plasma | LC-MS | Untargeted | Incident CVD | Population-based cohort; 53% female; 69 mean age; Spanish cohort | hPC, DG, MG, highly unsaturated phospholipids, and CE |
| Wang-Hazen, 2011 [57] | Case-control | Plasma | LC-MS, LC-MS/MS, GC-MS, NMR | Targeted | Incident CVD | Stable non-symptomatic subjects undergoing elective cardiac evaluations; 51% female; 64 mean age; Cleveland, Ohio USA Medical Center | TMAO, choline, betaine |
| Wang-Hu, 2017 [58] | Case-cohort, prospective | Plasma | LC-MS/MS | Targeted | Incident CVD | Population-based cohort; 57% female; 67 mean age; Spanish cohort | Cer(16:0), Cer(22:0), Cer(24:0), Cer(24:1) |
| Wittenbecher, 2021 [59] | Nested case-control, prospective | Plasma | LC-MS, FIA-IM-MS/MS | Untargeted and Targeted | Incident HF | 2 Population-based cohorts; 56% female; 72 mean age; 1 German and 1 Spanish cohort | PC C16:0/C16:0 and CerC16:0 |
| Würtz, 2015 [60] | Cohort, prospective | Serum | LC-MS/MS, GC-MS, NMR | Untargeted and Targeted | Incident CVD | 3 Population-based cohorts; 57% female; 56 mean age; 1 Finnish and 2 UK cohorts | MUFA, omega-6 fatty acid, docosahexaenoic acids |
| Zordoky, 2015 [61] | Case-control | Plasma | LC-MS/MS, FIA-MS/MS, NMR | Untargeted and Targeted | HFrEF vs HFpEF | CHF patients; 39% female; 65 mean age; Canadian cohort | 2-hydroxybutyrate, octadecenoylcarnitine (C18:1), hydroxyprionylcarnitine (C3-OH), SM(C24:1), octanoylcarnitine, and SM(C20:2) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
McGranaghan, P.; Kirwan, J.A.; Garcia-Rivera, M.A.; Pieske, B.; Edelmann, F.; Blaschke, F.; Appunni, S.; Saxena, A.; Rubens, M.; Veledar, E.; et al. Lipid Metabolite Biomarkers in Cardiovascular Disease: Discovery and Biomechanism Translation from Human Studies. Metabolites 2021, 11, 621. https://doi.org/10.3390/metabo11090621
McGranaghan P, Kirwan JA, Garcia-Rivera MA, Pieske B, Edelmann F, Blaschke F, Appunni S, Saxena A, Rubens M, Veledar E, et al. Lipid Metabolite Biomarkers in Cardiovascular Disease: Discovery and Biomechanism Translation from Human Studies. Metabolites. 2021; 11(9):621. https://doi.org/10.3390/metabo11090621
Chicago/Turabian StyleMcGranaghan, Peter, Jennifer A. Kirwan, Mariel A. Garcia-Rivera, Burkert Pieske, Frank Edelmann, Florian Blaschke, Sandeep Appunni, Anshul Saxena, Muni Rubens, Emir Veledar, and et al. 2021. "Lipid Metabolite Biomarkers in Cardiovascular Disease: Discovery and Biomechanism Translation from Human Studies" Metabolites 11, no. 9: 621. https://doi.org/10.3390/metabo11090621
APA StyleMcGranaghan, P., Kirwan, J. A., Garcia-Rivera, M. A., Pieske, B., Edelmann, F., Blaschke, F., Appunni, S., Saxena, A., Rubens, M., Veledar, E., & Trippel, T. D. (2021). Lipid Metabolite Biomarkers in Cardiovascular Disease: Discovery and Biomechanism Translation from Human Studies. Metabolites, 11(9), 621. https://doi.org/10.3390/metabo11090621







