Will Casuarina glauca Stress Resilience Be Maintained in the Face of Climate Change?
Abstract
:1. Introduction
2. Results
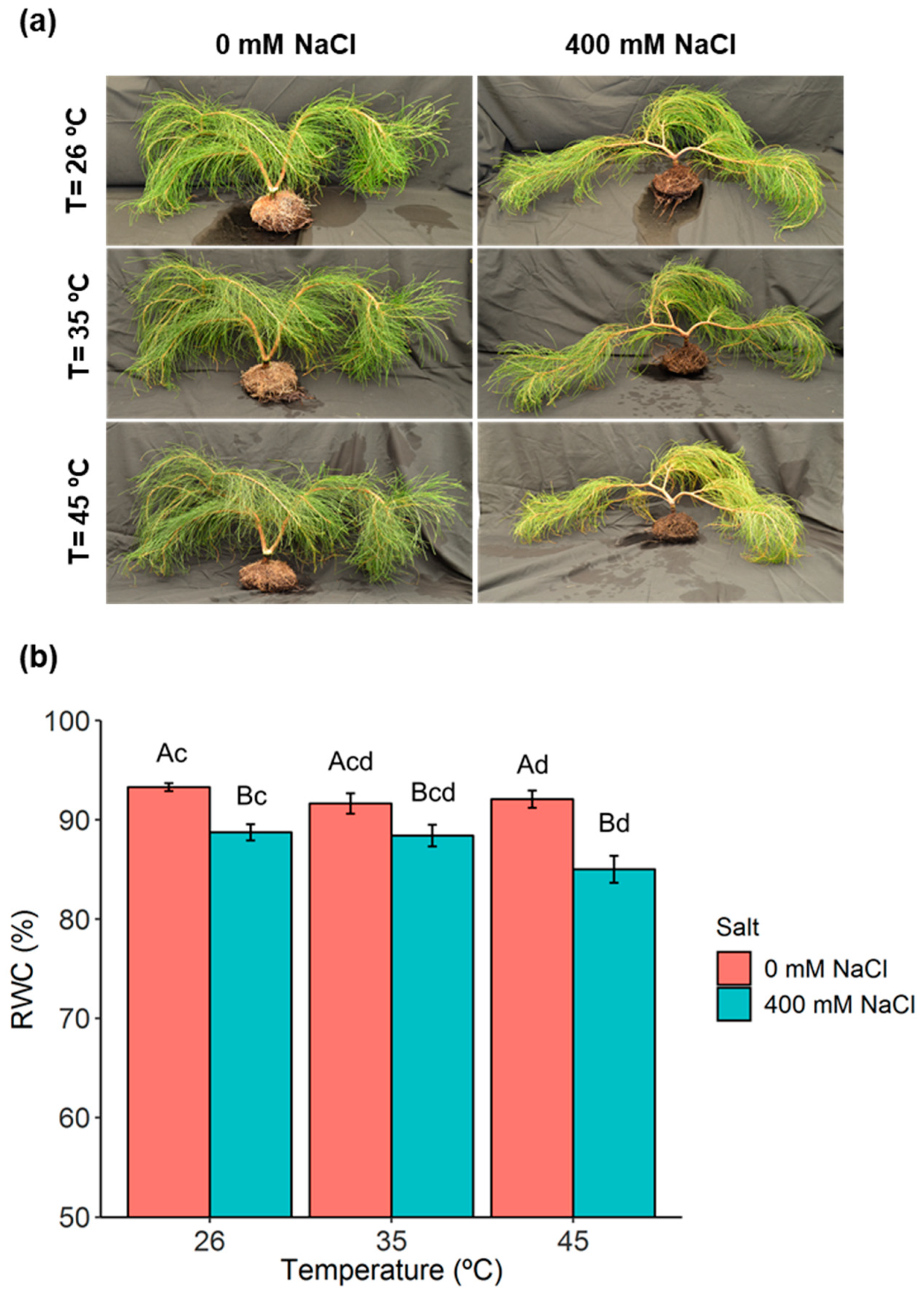
2.1. Phenotypic Analysis and Plant Water Relations
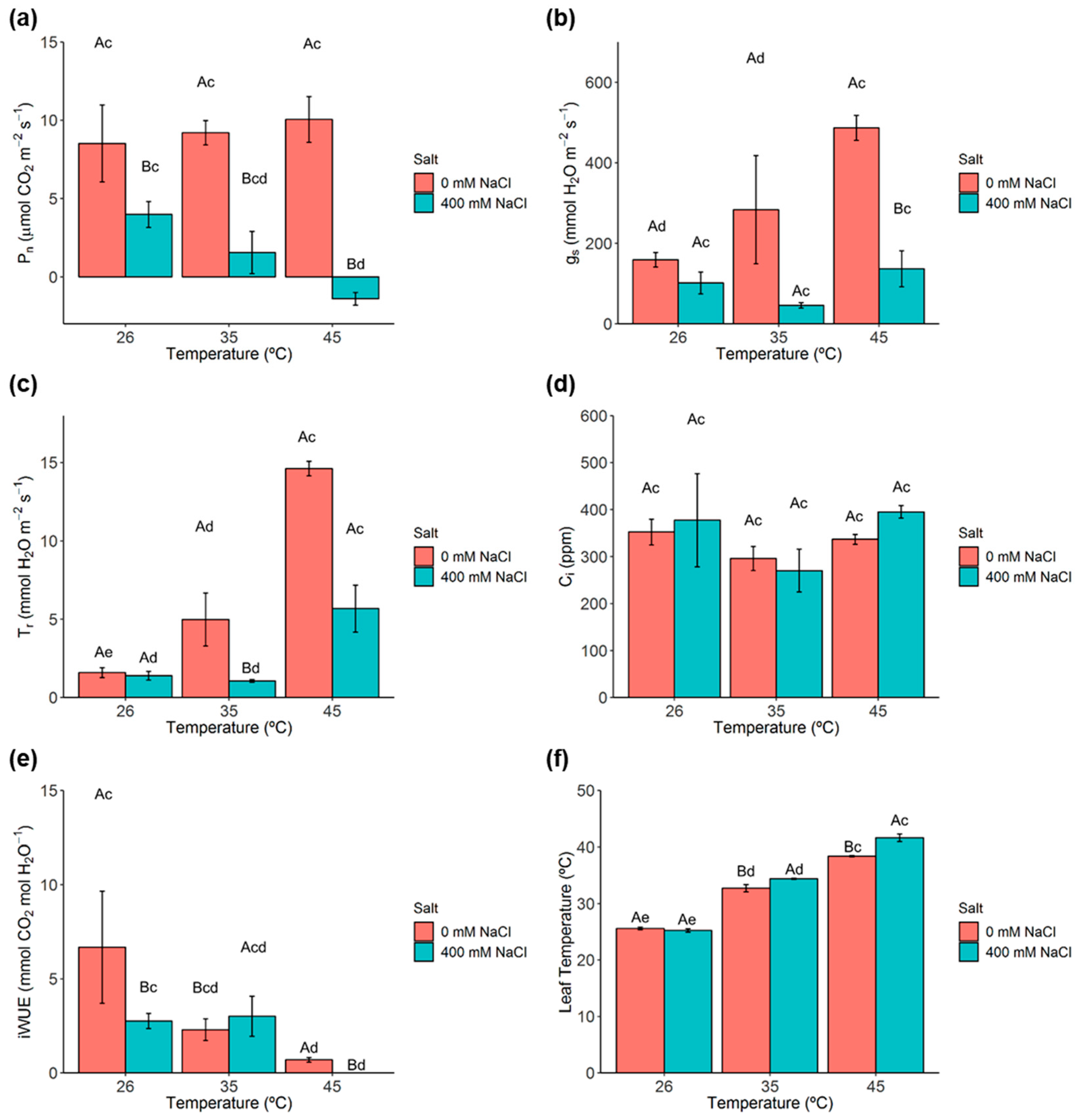
2.2. Photosynthetic Gas Exchanges
2.3. Chlorophyll a Fluorescence
2.4. Photosynthetic Pigments
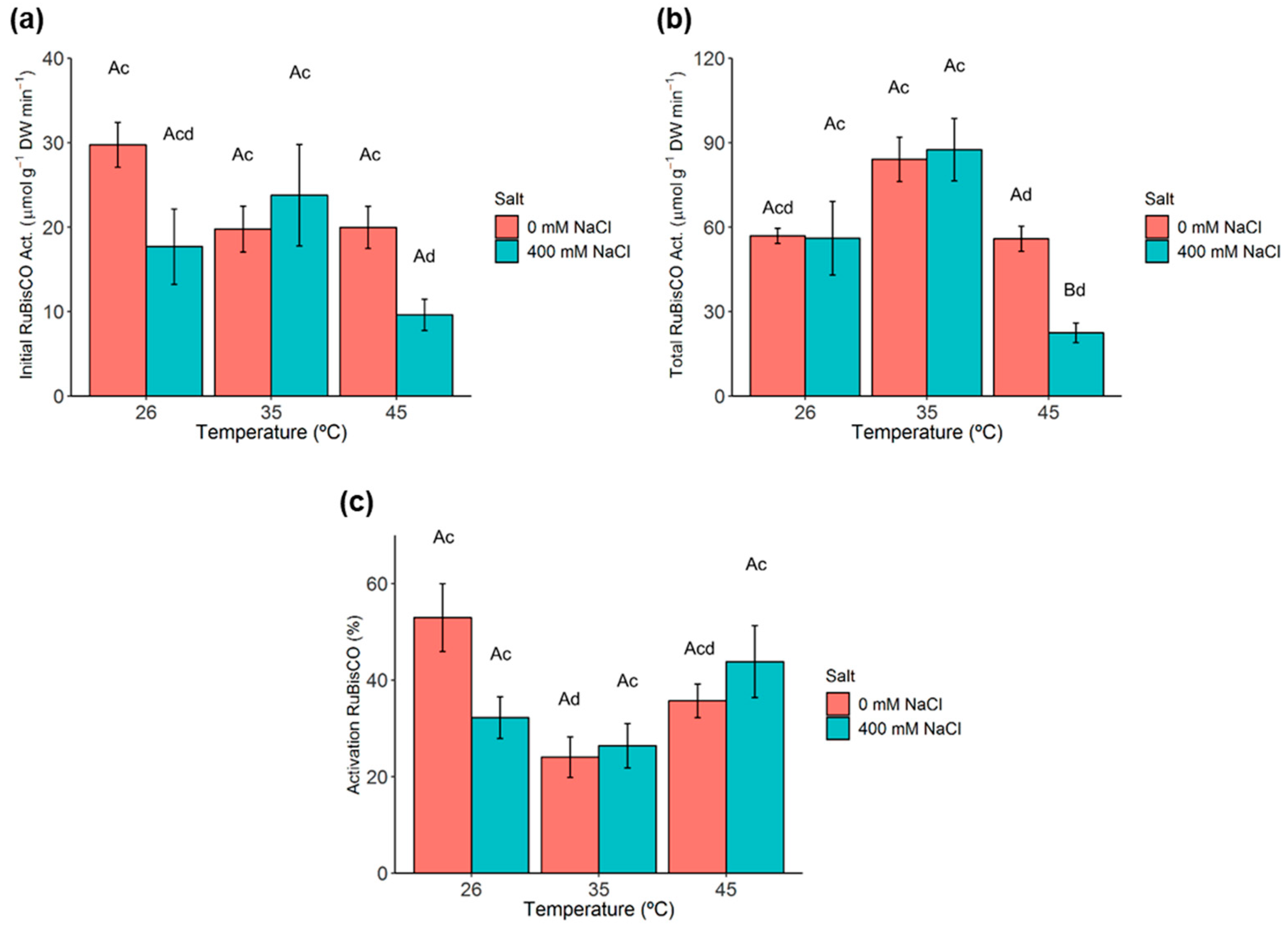
2.5. Ribulose-1,5-Biphosphate Carboxylase/Oxygenase (RuBisCO) Activity and Activation State
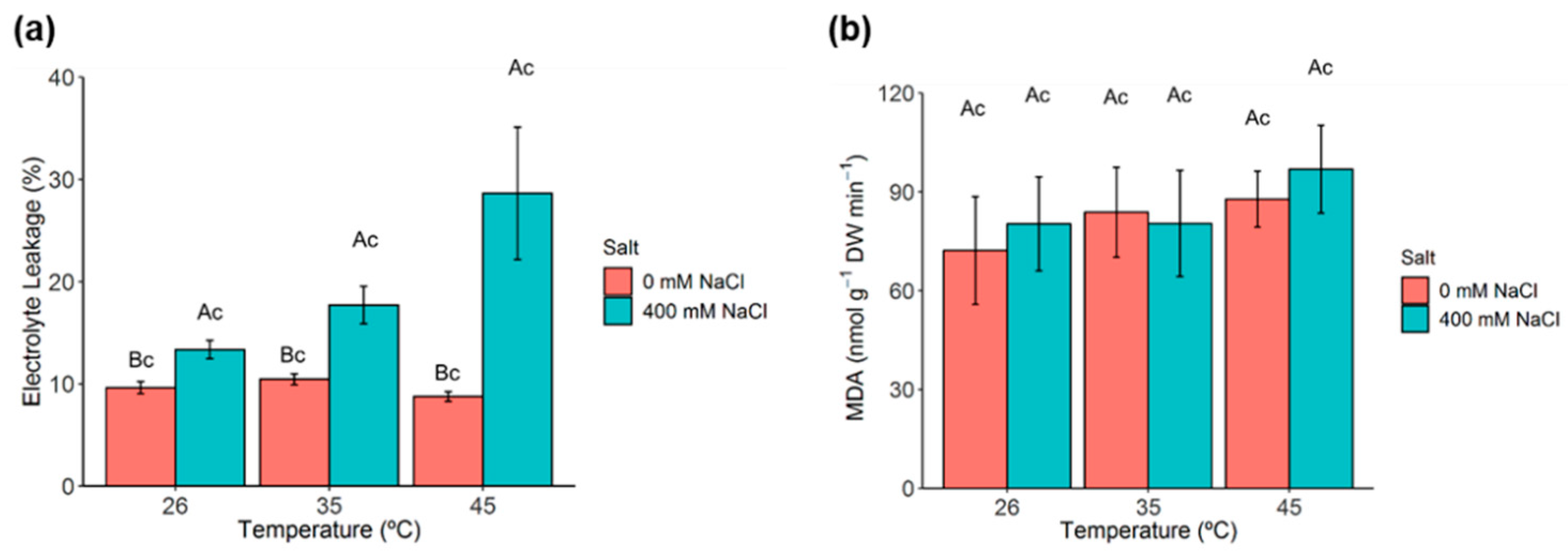
2.6. Membrane Permeability and Lipid Peroxidation
2.7. Lipid Analyses
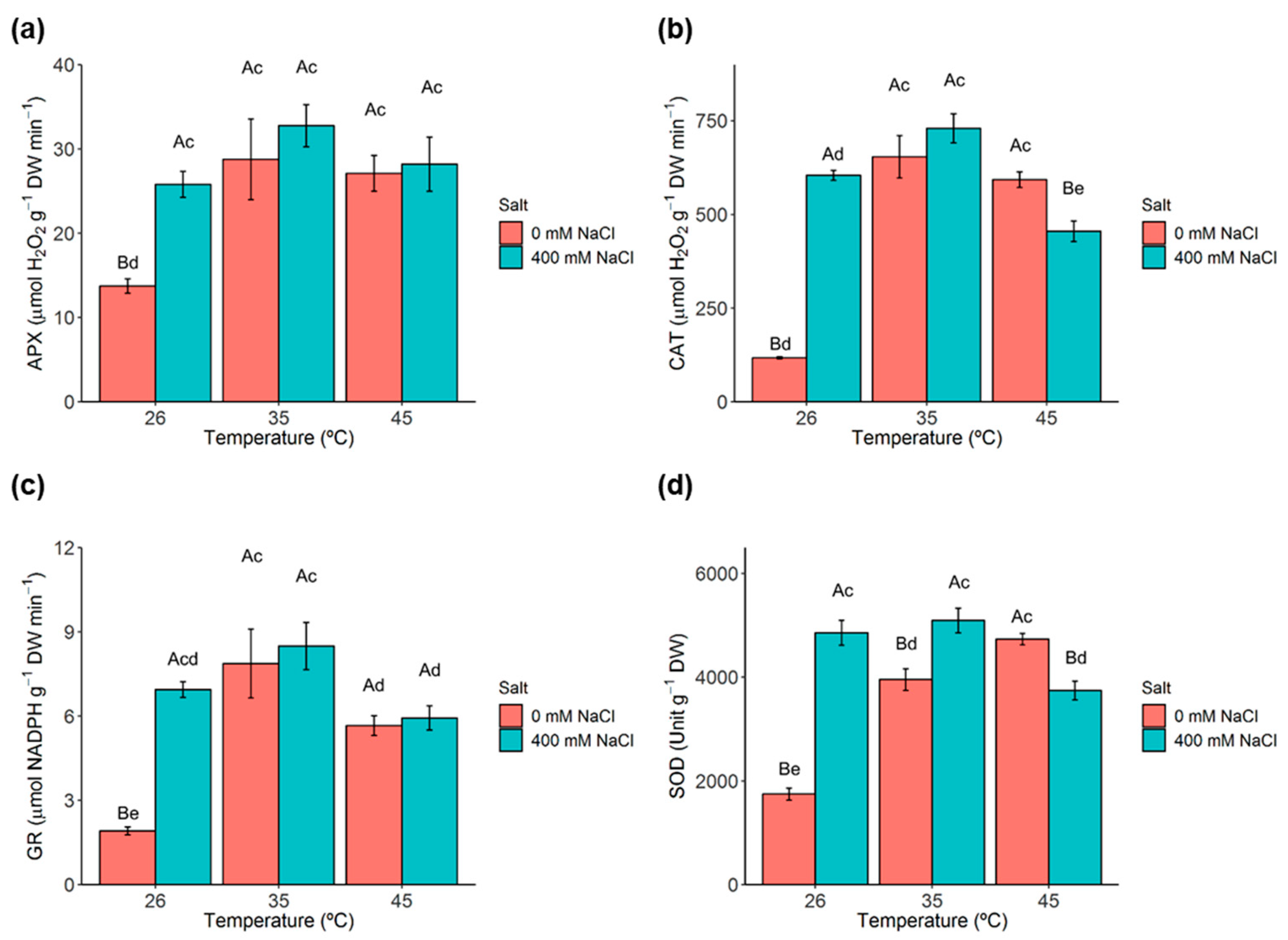
2.8. Antioxidant Enzymes
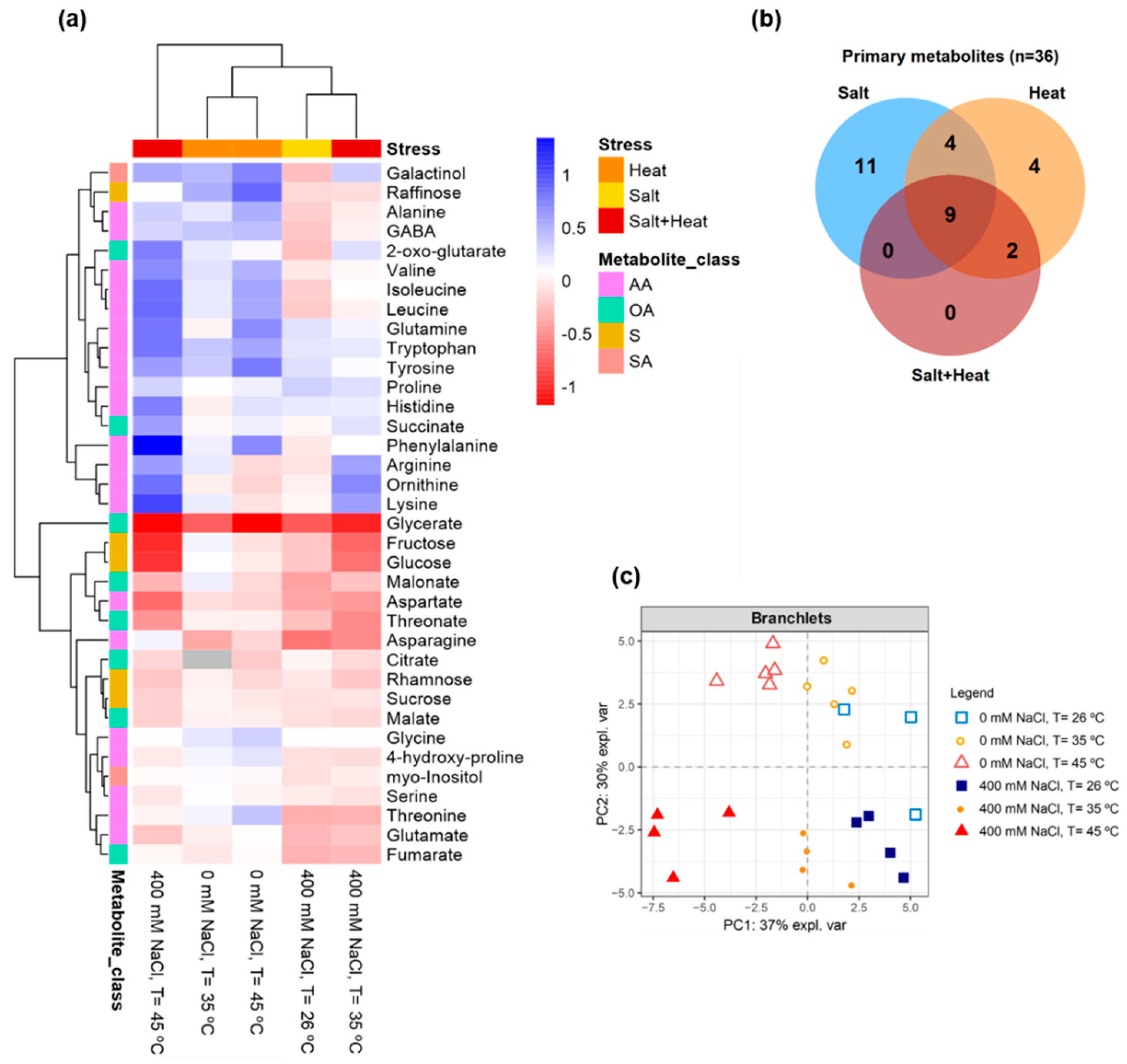
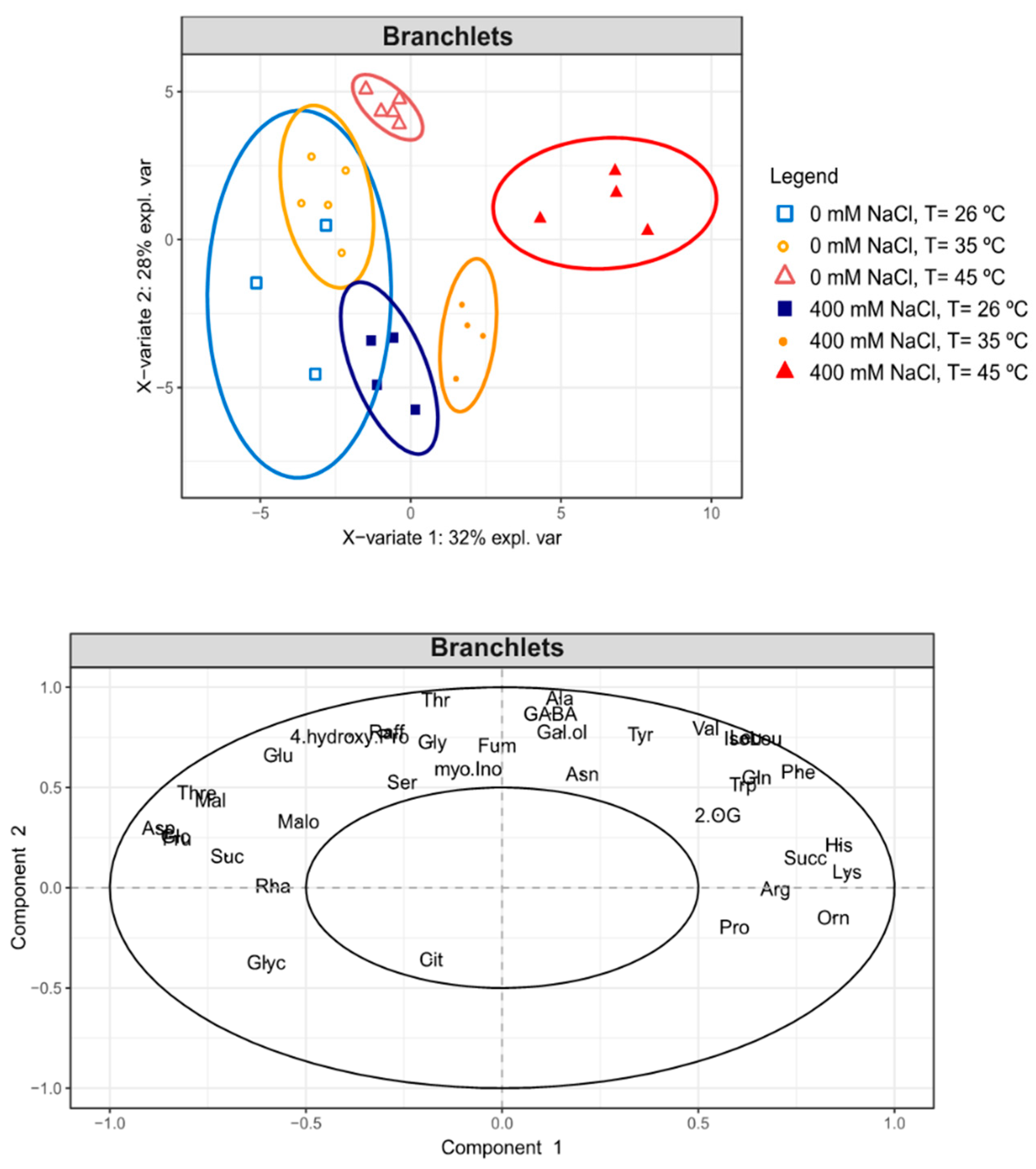
2.9. GC-TOF-MS Primary Metabolite Profiling Analysis
2.10. LC-HRMS/MS Target Secondary Metabolite Analysis
3. Discussion
4. Materials and Methods
4.1. Plant Growth Conditions and Stress Treatments
4.2. Harvest and Storage of Casuarina Glauca Branchlets
4.3. Plant Water Relations
4.4. Gas Exchange Measurements and Chlorophyll a Fluorescence
4.5. Photosynthetic Pigment Measurements
4.6. Ribulose-1,5-Bisphosphate Carboxylase/Oxygenase Activity
4.7. Electrolyte Leakage
4.8. Lipid Analyses
4.9. Antioxidant Enzyme Activities
4.10. GC-TOF-MS Primary Metabolite Profiling Analysis
4.11. LC-HRMS/MS Target Secondary Metabolite Analysis
4.12. Statistical Data Analyses
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- IPCC Core Writing Team. Climate Change 2014: Synthesis Report. Contribution of Working Groups I, II and III to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; Intergovernmental Panel on Climate Change: Geneva, Switzerland, 2014. [Google Scholar]
- Hirayama, T.; Shinozaki, K. Research on plant abiotic stress responses in the post-genome era: Past, present and future. Plant J. 2010, 61, 1041–1052. [Google Scholar] [CrossRef]
- Pereira, A. Plant abiotic stress challenges from the changing environment. Front. Plant Sci. 2011, 7, 1123. [Google Scholar] [CrossRef] [Green Version]
- Mittler, R. Abiotic stress, the field environment and stress combination. Trends Plant Sci. 2006, 11, 15–19. [Google Scholar] [CrossRef]
- Prasch, C.M.; Sonnewald, U. Simultaneous application of heat, drought, and virus to Arabidopsis plants reveals significant shifts in signalling networks. Plant Physiol. 2013, 162, 1849–1866. [Google Scholar] [CrossRef]
- Rasmussen, S.; Barah, P.; Suarez-Rodriguez, M.C.; Bressendorff, S.; Friis, P.; Costantino, P.; Bones, A.M.; Nielsen, H.B.; Mundy, J. Transcriptome responses to combinations of stresses in Arabidopsis. Plant Physiol. 2013, 161, 1783–1794. [Google Scholar] [CrossRef] [Green Version]
- Obata, T.; Witt, S.; Lisec, J.; Palacios-Rojas, N.; Florez-Sarasa, I.; Yousfi, S.; Araus, J.L.; Cairns, J.E.; Fernie, A.R. Metabolite profiles of maize leaves in drought, heat and combined stress field trials reveal the relationship between metabolism and grain yield. Plant Physiol. 2015, 69, 2665–2683. [Google Scholar] [CrossRef] [Green Version]
- Pandey, P.; Ramegowda, V.; Senthil-Kumar, M. Shared and unique responses of plants to multiple individual stresses and stress combinations: Physiological and molecular mechanisms. Front. Plant Sci. 2015, 6, 723. [Google Scholar] [CrossRef] [Green Version]
- Shaar-Moshe, L.; Blumwald, E.; Peleg, Z. Unique physiological and transcriptional shifts under combinations of salinity, drought, and heat. Plant Physiol. 2017, 174, 421–434. [Google Scholar] [CrossRef] [Green Version]
- Suzuki, N.; Rivero, R.M.; Shulaev, V.; Blumwald, E.; Mittler, R. Abiotic and biotic stress combinations. New Phytol. 2014, 203, 32–43. [Google Scholar] [CrossRef]
- Jorge, T.F.; António, C. Plant metabolomics in a changing world: Metabolite responses to abiotic stress combinations, plant, abiotic stress and responses to climate change. In Plant, Abiotic Stress and Responses to Climate Change; Andjelkovic, V., Ed.; InTechOpen Publishing: London, UK, 2017; pp. 111–132. [Google Scholar] [CrossRef] [Green Version]
- Zandalinas, S.I.; Mittler, R.; Balfagón, D.; Arbona, V.; Gómez-Cadenas, A. Plant adaptations to the combination of drought and high temperatures. Physiol. Plant 2018, 162, 2–12. [Google Scholar] [CrossRef] [Green Version]
- Keles, Y.; Oncel, I. Response of antioxidative defence system to temperature and water stress combinations in wheat seedlings. Plant Sci. 2002, 163, 783–790. [Google Scholar] [CrossRef]
- Rivero, R.M.; Mestre, T.C.; Mittler, R.; Rubio, F.; Garcia-Sanchez, F.; Martinez, V. The combined effect of salinity and heat reveals a specific physiological, biochemical and molecular responses in tomato plants. Plant Cell Environ. 2014, 37, 1059–1073. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Sonnewald, U. Differences and commonalities of plant responses to single and combined stresses. Plant J. 2017, 90, 839–855. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Munns, R.; Tester, M. Mechanisms of salinity tolerance. Annu. Rev. Plant Biol. 2008, 59, 651–681. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, Y.; Guo, Y. Elucidating the molecular mechanisms mediating plant salt-stress responses. New Phytol. 2018, 217, 523–539. [Google Scholar] [CrossRef] [Green Version]
- Wahid, A.; Gelani, S.; Ashraf, M.; Foolad, M.R. Heat tolerance in plants: An overview. Environ. Exp. Bot. 2007, 61, 199–223. [Google Scholar] [CrossRef]
- Mittler, R.; Finka, A.; Goloubinoff, P. How do plants feel the heat? Trends Biochem. Sci. 2012, 37, 118–125. [Google Scholar] [CrossRef]
- Slama, I.; Abdelly, C.; Bouchereau, A.; Flowers, T.; Savouré, A. Diversity, distribution and roles of osmoprotective compounds accumulated in halophytes under abiotic stress. Ann. Bot. 2015, 115, 433–447. [Google Scholar] [CrossRef] [Green Version]
- Potgieter, L.J.; Richardson, D.M.; Wilson, J.R.U. Casuarina: Biogeography and ecology of an important tree genus in a changing world. Biol. Invasions 2014, 16, 609–633. [Google Scholar] [CrossRef]
- Li, H.L.; Wang, W.; Mortimer, P.E.; Li, R.Q.; Li, D.Z.; Hyde, K.D.; Xu, J.C.; Soltis, D.E.; Chen, Z.D. Large-scale phylogenetic analyses reveal multiple gains of actinorhizal nitrogen-fixing symbioses in angiosperms associated with climate change. Sci. Rep. 2015, 5, 14023. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Diagne, N.; Arumugam, K.; Ngom, M.; Nambiar-Veetil, M.; Franche, C.; Narayanan, K.K.; Laplaze, L. Use of Frankia and actinorhizal plants for degraded lands reclamation. Biomed. Res. Int. 2013, 2013, 948258. [Google Scholar] [CrossRef] [Green Version]
- Zhong, C.; Mansour, S.; Nambiar-Veetil, M.; Bogusz, D.; Franche, C. Casuarina glauca: A model tree for basic research in actinorhizal symbiosis. J. Biosci. 2013, 38, 815–823. [Google Scholar] [CrossRef] [PubMed]
- Sayed, W.F. Improving Casuarina growth and symbiosis with Frankia under different soil and environmental conditions—Review. Folia Microbiol. 2011, 56, 1–9. [Google Scholar] [CrossRef]
- Huang, J.; Ji, M.; Xie, Y.; Wang, S.; He, Y.; Ran, J. Global semi-arid climate change over last 60 years. Clim. Dyn. 2016, 46, 1131–1150. [Google Scholar] [CrossRef] [Green Version]
- Lionello, P.; Scarascia, L. The relation between climate change in the Mediterranean region and global warming. Reg. Environ. Chang. 2018, 18, 1481–1493. [Google Scholar] [CrossRef]
- Batista-Santos, P.; Duro, N.; Rodrigues, A.P.; Semedo, J.N.; Alves, P.; da Costa, M.; Graça, I.; Pais, I.P.; Scotti-Campos, P.; Lidon, F.C.; et al. Is salt stress tolerance in Casuarina glauca Sieb. ex Spreng. associated with its nitrogen-fixing root-nodule symbiosis? An analysis at the photosynthetic level. Plant Physiol. Biochem. 2015, 96, 97–109. [Google Scholar] [CrossRef] [PubMed]
- Da Costa, M.; Duro, N.; Batista-Santos, P.; Ramalho, J.C.; Ribeiro-Barros, A.I. Validation of candidate reference genes for qRT-CR studies in symbiotic and non-symbiotic Casuarina glauca Sieb. ex Spreng. under salinity conditions. Symbiosis 2015, 66, 21–35. [Google Scholar] [CrossRef]
- Duro, N.; Batista-Santos, P.; da Costa, M.; Maia, R.; Castro, I.V.; Ramos, M.; Ramalho, J.C.; Pawlowski, K.; Máguas, C.; Ribeiro-Barros, A. The impact of salinity on the symbiosis between Casuarina glauca Sieb. ex Spreng. and N2-fixing Frankia bacteria based on the analysis of Nitrogen and Carbon metabolism. Plant Soil 2016, 398, 327–337. [Google Scholar] [CrossRef]
- Ribeiro-Barros, A.I.; da Costa, M.; Duro, N.; Graça, I.; Batista-Santos, P.; Jorge, T.F.; Lidon, F.J.C.; Pawlowski, K.; António, C.; Ramalho, J.C. An integrated approach to understand the mechanisms underlying salt stress tolerance in Casuarina glauca and its relation with nitrogen-fixing Frankia Thr. Symbiosis 2016, 70, 111–116. [Google Scholar] [CrossRef]
- Scotti-Campos, P.; Duro, N.; da Costa, M.; Pais, I.P.; Rodrigues, A.P.; Batista-Santos, P.; Semedo, J.N.; Leitão, A.E.; Lidon, F.C.; Pawlowski, K.; et al. Antioxidative ability 9 integrity in salt-induced responses of Casuarina glauca Sieber ex Spreng. in symbiosis with N2-fixing Frankia Thr or supplemented with mineral nitrogen. J. Plant Physiol. 2016, 196, 60–69. [Google Scholar] [CrossRef]
- Jorge, T.F.; Duro, N.; da Costa, M.; Florian, A.; Ramalho, J.C.; Ribeiro-Barros, A.I.; Fernie, A.R.; António, C. GC-TOF-MS analysis reveals salt stress-responsive primary metabolites in Casuarina glauca tissues. Metabolomics 2017, 13, 95. [Google Scholar] [CrossRef]
- Jorge, T.F.; Florêncio, M.H.; Ribeiro-Barros, A.I.; António, C. Quantification and structural characterization of raffinose family oligosaccharides in Casuarina glauca plant tissues by porous graphitic carbon electrospray quadrupole ion trap mass spectrometry. Int. J. Mass Spectrom. 2017, 413, 127–134. [Google Scholar] [CrossRef]
- Jorge, T.F.; Tohge, T.; Wendenburg, R.; Ramalho, J.C.; Lidon, F.C.; Ribeiro-Barros, A.I.; Fernie, A.R.; António, C. Salt-stress secondary metabolite signatures involved in the ability of Casuarina glauca to mitigate oxidative stress. Environ. Exp. Bot. 2019, 166, 103808. [Google Scholar] [CrossRef]
- Graça, I.; Mendes, V.M.; Marques, I.; Duro, N.; da Costa, M.; Ramalho, J.C.; Pawlowski, K.; Manadas, B.; Ricardo, C.P.P.; Ribeiro-Barros, A.I. Comparative proteomic analysis of nodulated and non-nodulated Casuarina glauca Sieb. ex Spreng. Grown under salinity conditions using sequential window acquisition of all theoretical mass spectra (SWATH-MS). Int. J. Mol. Sci. 2020, 21, 78. [Google Scholar] [CrossRef] [Green Version]
- Chaves, M.M.; Flexas, J.; Pinheiro, C. Photosynthesis under drought and salt stress: Regulation mechanisms from whole plant to cell. Ann. Bot. 2009, 103, 551–560. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Negrão, S.; Schmöckel, S.M.; Tester, M. Evaluating physiological responses of plants to salinity stress. Ann. Bot. 2017, 119, 13–26. [Google Scholar] [CrossRef] [Green Version]
- Medeira, C.; Quartin, V.; Maia, I.; Diniz, I.; Matos, M.C.; Semedo, J.N.; Scotti-Campos, P.; Ramalho, J.C.; Pais, I.P.; Ramos, P.; et al. Cryptogein and capsicein promote defence responses in Quercus suber against Phytophthora cinnamomi Infection. Eur. J. Plant Pathol. 2012, 134, 145–159. [Google Scholar] [CrossRef]
- Baker, N.R. Chlorophyll fluorescence: A probe of photosynthesis in vivo. Annu. Rev. Plant Biol. 2008, 59, 89–113. [Google Scholar] [CrossRef] [Green Version]
- Kramer, D.M.; Johnson, G.; Kiirats, O.; Edwards, G.E. New fluorescence parameters for the determination of QA redox state and excitation energy fluxes. Photosynth. Res. 2004, 79, 209–218. [Google Scholar] [CrossRef]
- Busch, F.; Hunter, N.P.A.; Ensminger, I. Biochemical constrains limit the potential of the photochemical reflectance index as a predictor of effective quantum efficiency of photosynthesis during the winter spring transition in Jack pine seedlings. Funct. Plant Biol. 2009, 36, 1016–1026. [Google Scholar] [CrossRef]
- Huang, W.; Zhang, S.B.; Cao, K.F. Cyclic electron flow plays an important role in photoprotection of tropical trees illuminated at temporal chilling temperature. Plant Cell Physiol. 2011, 52, 297–305. [Google Scholar] [CrossRef]
- Rodrigues, W.P.; Martins, M.Q.; Fortunato, A.S.; Rodrigues, A.P.; Semedo, J.N.; Simões-Costa, M.C.; Pais, I.P.; Leitão, A.E.; Colwell, F.; Goulao, L.; et al. Long-term elevated air [CO2] strengthens photosynthetic functioning and mitigates the impact of supra-optimal temperatures in tropical Coffea arabica and Coffea canephora species. Glob. Chang. Biol. 2016, 22, 415–431. [Google Scholar] [CrossRef]
- Maxwell, K.; Johnson, G.N. Chlorophyll fluorescence—A practical guide. J. Exp. Bot. 2000, 345, 659–668. [Google Scholar] [CrossRef]
- Fortunato, A.; Lidon, F.C.; Batista-Santos, P.; Leitão, A.E.; Pais, I.P.; Ribeiro, A.I.; Ramalho, J.C. Biochemical and molecular characterization of the antioxidative system of Coffea sp. under cold conditions in genotypes with contrasting tolerance. J. Plant Physiol. 2010, 167, 333–342. [Google Scholar] [CrossRef] [PubMed]
- Ashraf, M.; Harris, P.J.C. Photosynthesis under stressful environments: An overview. Photosynthetica 2013, 51, 163–190. [Google Scholar] [CrossRef]
- Martins, M.Q.; Rodrigues, W.P.; Fortunato, A.S.; Leitão, A.E.; Rodrigues, A.P.; Pais, I.P.; Martins, L.D.; Silva, M.J.; Reboredo, F.H.; Partelli, F.L.; et al. Protective response mechanisms to heat stress in interaction with high [CO2] conditions in Coffea spp. Front. Plant Sci. 2016, 7, 947. [Google Scholar] [CrossRef] [Green Version]
- Mansour, M.M.F. Plasma membrane permeability as an indicator of salt tolerance in plants. Biol. Plant 2013, 57, 1–10. [Google Scholar] [CrossRef]
- Hasanuzzaman, M.; Nahar, K.; Alam, M.M.; Roychowdhury, R.; Fujita, M. Physiological, biochemical, and molecular mechanisms of heat stress tolerance in plants. Int. J. Mol. Sci. 2013, 14, 9643–9684. [Google Scholar] [CrossRef]
- Murakami, Y.; Tsuyama, M.; Kobayashi, Y.; Kodama, H.; Iba, K. Trienoic fatty acids and plant tolerance of high temperature. Science 2000, 287, 476–479. [Google Scholar] [CrossRef]
- Sakamoto, T.; Murata, N. Regulation of the desaturation of fatty acids and its role in tolerance to cold and salt stress. Curr. Opin. Microbiol. 2002, 5, 206–210. [Google Scholar] [CrossRef]
- Das, K.; Roychoudhury, A. Reactive oxygen species (ROS) and response of antioxidants as ROS-scavengers during environmental stress in plants. Front. Environ. Sci. 2014, 2, 53. [Google Scholar] [CrossRef] [Green Version]
- Yaeno, T.; Matsuda, O.; Iba, K. Role of chloroplast trienoic fatty acids in plant disease defense responses. Plant J. 2004, 40, 931–941. [Google Scholar] [CrossRef] [PubMed]
- Upchurch, R.G. Fatty acid unsaturation, mobilization, and regulation in the response of plants to stress. Biotechnol. Lett. 2008, 30, 967–977. [Google Scholar] [CrossRef]
- ElSayed, A.I.; Rafudeen, M.S.; Golldack, D. Physiological aspects of raffinose family oligosaccharides in plants: Protection against abiotic stress. Plant Biol. 2014, 6, 1–8. [Google Scholar] [CrossRef]
- Sengupta, S.; Mukherjee, S.; Basak, P.; Majumder, A.L. Significance of galactinol and raffinose family oligosaccharide synthesis in plants. Front. Plant Sci. 2015, 6, 656. [Google Scholar] [CrossRef] [Green Version]
- Bauwe, H.; Hagemann, M.; Fernie, A.R. Photorespiration: Players, partners and origin. Trends Plant Sci. 2010, 15, 330–336. [Google Scholar] [CrossRef]
- Timm, S.; Florian, A.; Wittmiß, M.; Jahnke, K.; Hagemann, M.; Fernie, A.R.; Bauwe, H. Serine acts as a metabolic signal for the transcriptional control of photorespiration-related genes in Arabidopsis. Plant Physiol. 2013, 162, 379–389. [Google Scholar] [CrossRef] [Green Version]
- Eisenhut, M.; Bräutigam, A.; Timm, S.; Florian, A.; Tohge, T.; Fernie, A.R.; Bauwe, H.; Weber, A.P.M. Photorespiration is crucial for dynamic response of photosynthetic metabolism and stomatal movement to altered CO2 availability. Mol. Plant 2017, 10, 47–61. [Google Scholar] [CrossRef] [Green Version]
- Tohge, T.; Watanabe, M.; Hoefgen, R.; Fernie, A.R. Shikimate and phenylalanine biosynthesis in the green lineage. Front. Plant Sci. 2013, 4, 62. [Google Scholar] [CrossRef] [Green Version]
- Hildebrandt, T.M. Synthesis versus degradation: Directions of amino acid metabolism during Arabidopsis abiotic stress response. Plant Mol. Biol. 2018, 98, 121–135. [Google Scholar] [CrossRef]
- Szecowka, M.; Heise, R.; Tohge, T.; Nunes-Nesi, A.; Vosloh, D.; Huege, J.; Feil, R.; Lunn, J.; Nikoloski, Z.; Stitt, M.; et al. Metabolic fluxes in an illuminated Arabidopsis rosette. Plant Cell 2013, 25, 694–714. [Google Scholar] [CrossRef] [Green Version]
- Arrivault, S.; Obata, T.; Szecówka, M.; Mengin, V.; Guenther, M.; Hoehne, M.; Fernie, A.R.; Stitt, M. Metabolite pools and carbon flow during C4 photosynthesis in maize: 13CO2 labeling kinetics and cell type fractionation. J. Exp. Bot. 2017, 68, 283–298. [Google Scholar] [CrossRef] [Green Version]
- Obata, T.; Fernie, A.R. The use of metabolomics to dissect plant responses to abiotic stresses. Cell. Mol. Life Sci. 2012, 69, 3225–3243. [Google Scholar] [CrossRef] [Green Version]
- Mata, A.T.; Jorge, T.F.; Pires, M.V.; António, C. Drought stress tolerance in plants: Insights from metabolomics. In Drought Stress Tolerance in Plants, Molecular and Genetic Perspectives; Hossain, M.A., Wani, S.H., Bhattachajee, S., Burritt, D.J., Phan, L.S., Eds.; Springer International Publishing: Cham, Switzerland, 2016; Volume 2, pp. 187–216. [Google Scholar] [CrossRef]
- Li, Z.; Yu, J.; Peng, Y.; Huang, B. Metabolic pathways regulated by γ-aminobutyric acid (GABA) contributing to heat tolerance in creeping bentgrass (Agrostis stolonifera). Sci. Rep. 2016, 6, 30338. [Google Scholar] [CrossRef] [Green Version]
- Fait, A.; Fromm, H.; Walter, D.; Galili, G.; Fernie, A.R. Highway or byway: The metabolic role of the GABA shunt in plants. Trends Plant Sci. 2008, 13, 14–19. [Google Scholar] [CrossRef]
- António, C.; Päpke, C.; Rocha, M.; Diab, H.; Limami, A.M.; Obata, T.; Fernie, A.R.; van Dongen, J.T. Regulation of primary metabolism in response to low oxygen availability as revealed by carbon and nitrogen isotope redistribution. Plant Physiol. 2016, 170, 43–56. [Google Scholar] [CrossRef] [PubMed]
- Szabados, L.; Savoure, A. Proline: A multifunctional amino acid. Trends Plant Sci. 2010, 15, 89–97. [Google Scholar] [CrossRef]
- Liang, X.; Zhang, L.; Natarajan, S.K.; Becker, D.F. Proline mechanisms of stress survival. Antioxid. Redox Signal. 2013, 19, 998–1011. [Google Scholar] [CrossRef] [Green Version]
- Kaur, G.; Asthir, B. Proline: A key player in plant abiotic stress tolerance. Biol. Plant 2015, 59, 609–619. [Google Scholar] [CrossRef]
- Per, T.S.; Khan, N.A.; Reddy, P.S.; Masood, A.; Hasanuzzaman, M.; Khan, M.I.R.; Anjum, N.A. Approaches in modulating proline metabolism in plants for salt and drought stress tolerance: Phytohormones, mineral nutrients and transgenics. Plant Physiol. Biochem. 2017, 115, 126–140. [Google Scholar] [CrossRef]
- Sellstedt, A.; Atkins, C.A. Composition of amino compounds transported in xylem of Casuarina sp. J. Exp. Bot. 1991, 42, 1493–1497. [Google Scholar] [CrossRef]
- Galili, G. The aspartate-family pathway of plants: Linking production of essential amino acids with energy and stress regulation. Plant Signal. Behav. 2011, 6, 192–195. [Google Scholar] [CrossRef] [Green Version]
- Bailey-Serres, J.; Mittler, R. The roles of reactive oxygen species in plant cells. Plant Physiol. 2006, 141, 311. [Google Scholar] [CrossRef] [Green Version]
- Blokhina, O.; Vironlainen, E.; Fagerstedt, K.V. Antioxidants, oxidative damage and oxygen deprivation stress: A review. Ann. Bot. 2003, 91, 179–194. [Google Scholar] [CrossRef] [Green Version]
- Foyer, C.H.; Shigeoka, S. Understanding oxidative stress and antioxidant functions to enhance photosynthesis. Plant Physiol. 2011, 155, 93–100. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Agati, G.; Azzarello, E.; Pollastri, S.; Tattini, M. Flavonoids as antioxidants in plants: Location and functional significance. Plant Sci. 2012, 196, 67–76. [Google Scholar] [CrossRef] [PubMed]
- Tromas, A.; Parizot, B.; Diagne, N.; Champion, A.; Hocher, V.; Cissoko, M.; Crabos, A.; Prodjinoto, H.; Lahouze, B.; Bogusz, D.; et al. Heart of endosymbioses: Transcriptomics reveals a conserved genetic program among arbuscular mycorrhizal, actinorhizal and legume-rhizobial symbioses. PLoS ONE 2012, 7, e44742. [Google Scholar] [CrossRef]
- Zhong, C.; Zhang, Y.; Chen, Y.; Jiang, Q.; Chen, Z.; Liang, J.; Pinyopusarerk, K.; Franche, C.; Bogusz, D. Casuarina research and applications in China. Symbiosis 2010, 50, 107–114. [Google Scholar] [CrossRef]
- Ramalho, J.C.; Rodrigues, A.P.; Lidon, F.; Marques, L.M.C.; Leitão, A.E.; Fortunato, A.S.; Pais, I.P.; Silva, M.J.; Scotti-Campos, P.; Lopes, A.; et al. Stress cross-response of the antioxidative system promoted by superimposed drought and cold conditions in Coffea spp. PLoS ONE 2018, 13, e0198694. [Google Scholar] [CrossRef]
- Schreiber, U. Pulse-amplitude-modulation (PAM) fluorometry and saturation pulse method: An overview. In Chlorophyll a Fluorescence: A Signature of Photosynthesis; Papageorgiou, G.C., Ed.; Springer: Dordrecht, The Netherlands, 2004; pp. 279–319. [Google Scholar] [CrossRef]
- Klughammer, C.; Schreiber, U. Complementary PS II quantum yields calculated from simple fluorescence parameters measured by PAM fluorometry and the saturation pulse method. PAM Appl. Notes 2008, 1, 27–35. [Google Scholar]
- Ramalho, J.C.; Pons, T.L.; Groeneveld, H.W.; Nunes, M.A. Photosynthetic responses of Coffea arabica leaves to a short-term high light exposure in relation to N availability. Physiol. Plant 1997, 101, 229–239. [Google Scholar] [CrossRef]
- Ramalho, J.C.; Rodrigues, A.P.; Semedo, J.N.; Pais, I.P.; Martins, L.D.; Simões-Costa, M.C.; Leitão, A.E.; Fortunato, A.S.; Batista-Santos, P.; Palos, I.M.; et al. Sustained photosynthetic performance of Coffea spp. under long-term enhanced [CO2]. PLoS ONE 2013, 8, e82712. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Matos, M.C.; Campos, P.S.; Passarinho, J.A.; Semedo, J.N.; Marques, N.M.; Ramalho, J.C.; Ricardo, C.P. Drought effect on photosynthetic activity, osmolyte accumulation and membrane integrity of two Cicer arietinum genotypes. Photosynthetica 2010, 48, 303–312. [Google Scholar] [CrossRef]
- Campos, P.S.; Quartin, V.L.; Ramalho, J.C.; Nunes, M.A. Electrolyte leakage and lipid degradation account for cold sensitivity in leaves of Coffea sp. plants. J. Plant Physiol. 2003, 160, 283–292. [Google Scholar] [CrossRef] [Green Version]
- Beauchamp, C.; Fridovich, I. Superoxide dismutase: Improved assay and an assay applicable to acrylamide gels. Anal. Biochem. 1971, 44, 276–287. [Google Scholar] [CrossRef]
- Esterbauer, H.; Grill, D. Seasonal variation of glutathione and glutathione reductase in needles of Picea abies. Plant Physiol. 1978, 61, 119–121. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kato, M.; Shimizu, S. Chlorophyll metabolism in higher plants. VII. Chlorophyll degradation in senescing tobacco leaves; phenolic-dependent peroxidative degradation. Can. J. Bot. 1987, 65, 729–735. [Google Scholar] [CrossRef]
- Ramalho, J.C.; Campos, P.S.; Teixeira, M.; Nunes, M.A. Nitrogen dependent changes in antioxidant systems and in fatty acid composition of chloroplast membranes from Coffea arabica L. plants submitted to high irradiance. Plant Sci. 1998, 135, 115–124. [Google Scholar] [CrossRef]
- Lisec, J.; Schauer, N.; Kopka, J.; Willmitzer, L.; Fernie, A.R. Gas chromatography mass spectrometry-based metabolite profiling in plants. Nat. Protoc. 2006, 1, 387–396. [Google Scholar] [CrossRef]
- Luedemann, A.; Strassburg, K.; Erban, A.; Kopka, J. TagFinder for the quantitative analysis of gas chromatography-mass spectrometry (GC-MS)-based metabolite profiling experiments. Bioinformatics 2008, 24, 732–737. [Google Scholar] [CrossRef]
- Kopka, J.; Schauer, N.; Krueger, S.; Birkemeyer, C.; Usadel, B.; Bergmüller, E.; Dörmann, P.; Weckwerth, W.; Gibon, Y.; Stitt, M.; et al. Gmd@csb.Db: The Golm Metabolome Database. Bioinformatics 2005, 21, 1635–1638. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alseekh, A.; Aharoni, A.; Brotman, Y.; Contrepois, K.; D’Auria, J.; Ewald, J.; Ewald, J.C.; Fraser, P.D.; Giavalisco, P.; Hall, R.D.; et al. Mass spectrometry-based metabolomics: A guide for annotation, quantification and best reporting practices. Nat. Methods 2021, 18, 747–756. [Google Scholar] [CrossRef]
- Tohge, T.; Fernie, A.R. Combining genetic diversity, informatics and metabolomics to facilitate annotation of plant gene function. Nat. Protoc. 2010, 5, 1210–1227. [Google Scholar] [CrossRef]
- Guijas, C.; Montenegro-Burke, J.R.; Domingo-Almenara, X.; Palermo, A.; Warth, B.; Hermann, G.; Koellensperger, G.; Huan, T.; Uritboonthai, W.; Aisporna, A.E.; et al. METLIN: A technology platform for identifying knowns and unknowns. Anal. Chem. 2018, 90, 3156–3164. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- R Core Team. R: A Language and Environment for Statistical Computing; R Package v.3.4.3; R Foundation for Statistical Computing: Vienna, Austria, 2018; Available online: https://www.R-project.org/ (accessed on 12 December 2019).
- Raivo, K. Pheatmap: Pretty Heatmaps. R Package v.1.0.8. 2015. Available online: https://CRAN.R-project.org/package=pheatmap (accessed on 12 December 2019).
- Rohart, F.; Gautier, B.; Singh, A.; Cao, K.A.L. mixOmics: An R package for ‘omics feature selection and multiple data integration. PLoS Comput. Biol. 2017, 13, e1005752. [Google Scholar] [CrossRef] [Green Version]
- Chen, H.; Boutros, P.C. VennDiagram: A package for the generation of highly-customizable Venn and Euler diagrams in R. BMC Bioinformat. 2011, 26, 12–35. [Google Scholar] [CrossRef] [Green Version]
- Werner, C.; Correia, O.; Beyschlag, W. Characteristic patterns of chronic and dynamic photoinhibition of different functional groups in a Mediterranean ecosystem. Funct. Plant Biol. 2002, 29, 999–1011. [Google Scholar] [CrossRef] [PubMed]
- Fiehn, O.; Sumner, L.W.; Rhee, S.Y.; Ward, J.; Dickerson, J.; Lange, B.M.; Lane, G.; Roessner, U.; Last, R.; Nikolau, B. Minimum reporting standards for plant biology context in metabolomics studies. Metabolomics 2007, 3, 195–201. [Google Scholar] [CrossRef] [Green Version]
- Sumner, L.W.; Amberg, A.; Barrett, D.; Beale, M.H.; Beger, R.; Daykin, C.A.; Fan, T.W.M.; Fiehn, O.; Goodacre, R.; Griffin, J.L.; et al. Proposed minimum reporting standards for chemical analysis. Metabolomics 2007, 3, 211–221. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fernie, A.R.; Aharoni, A.; Willmitzer, L.; Stitt, M.; Tohge, T.; Kopka, J.; Carroll, A.J.; Saito, K.; Fraser, P.D.; De Luca, V. Recommendations for reporting metabolite data. Plant Cell 2011, 2, 2477–2482. [Google Scholar] [CrossRef] [Green Version]
| Parameter | [NaCl] | Temperature | ||
|---|---|---|---|---|
| 26 °C | 35 °C | 45 °C | ||
| Fo | 0 mM | 0.216 ± 0.010 Ac | 0.164 ± 0.003 Ae | 0.176 ± 0.005 Ad |
| 400 mM | 0.201 ± 0.005 Bc | 0.170 ± 0.004 Ad | 0.201 ± 0.010 Ad | |
| Fv/Fm | 0 mM | 0.772 ± 0.008 Ac | 0.818 ± 0.004 Ac | 0.786 ± 0.007 Ac |
| 400 mM | 0.750 ± 0.017 Ac | 0.783 ± 0.009 Ac | 0.489 ± 0.075 Bd | |
| Y(II) = Øe | 0 mM | 0.533 ± 0.029 Ac | 0.456 ± 0.022 Ac | 0.365 ± 0.018 Ad |
| 400 mM | 0.414 ± 0.034 Bc | 0.376 ± 0.033 Bc | 0.159 ± 0.043 Bd | |
| Y(NPQ) | 0 mM | 0.108 ± 0.010 Bd | 0.190 ± 0.019 Ad | 0.254 ± 0.013 Ac |
| 400 mM | 0.252 ± 0.016 Ac | 0.285 ± 0.040 Ac | 0.217 ± 0.067 Ac | |
| Y(NO) | 0 mM | 0.359 ± 0.027 Ac | 0.370 ± 0.026 Ac | 0.381 ± 0.016 Bc |
| 400 mM | 0.334 ± 0.022 Ad | 0.339 ± 0.012 Ad | 0.623 ± 0.106 Ac | |
| NPQ | 0 mM | 0.322 ± 0.035 Bd | 0.543 ± 0.061 Acd | 0.670 ± 0.049 Ac |
| 400 mM | 0.768 ± 0.047 Ac | 0.853 ± 0.137 Ac | 0.439 ± 0.181 Ad | |
| qN | 0 mM | 0.329 ± 0.018 Bd | 0.490 ± 0.031 Ac | 0.529 ± 0.018 Ac |
| 400 mM | 0.556 ± 0.018 Ac | 0.597 ± 0.044 Ac | 0.534 ± 0.069 Ac | |
| qP | 0 mM | 0.778 ± 0.027 Ac | 0.714 ± 0.017 Ac | 0.590 ± 0.024 Ad |
| 400 mM | 0.686 ± 0.047 Bc | 0.656 ± 0.035 Bc | 0.477 ± 0.045 Bd | |
| qL | 0 mM | 0.549 ± 0.032 Ac | 0.475 ± 0.019 Acd | 0.356 ± 0.021 Ad |
| 400 mM | 0.500 ± 0.045 Ac | 0.451 ± 0.030 Acd | 0.380 ± 0.029 Ad | |
| ETR | 0 mM | 114.2 ± 6.3 Ac | 97.7 ± 4.7 Ac | 78.1 ± 3.9 Ac |
| 400 mM | 88.7 ± 7.3 Bc | 80.6 ± 7.1 Ac | 34.1 ± 9.1 Ad | |
| Fv’/Fm’ | 0 mM | 0.679 ± 0.016 Ac | 0.638 ± 0.021 Ac | 0.618 ± 0.008 Ac |
| 400 mM | 0.591 ± 0.019 Bc | 0.571 ± 0.025 Ac | 0.319 ± 0.065 Bd | |
| Fs/Fm’ | 0 mM | 0.467 ± 0.029 Bc | 0.544 ± 0.022 Ac | 0.635 ± 0.018 Ac |
| 400 mM | 0.586 ± 0.034 Ad | 0.624 ± 0.033 Acd | 0.841 ± 0.043 Ac | |
| PIChr | 0 mM | 6.92 ± 0.77 Ac | 1.55 ± 0.59 Ac | 5.38 ± 0.84 Bc |
| 400 mM | 8.91 ± 1.34 Ad | 5.76 ± 1.44 Ad | 41.2 ± 13.8 Ac | |
| PIDyn | 0 mM | 11.4 ± 1.3 Bd | 21.7 ± 2.6 Bc | 20.3 ± 1.4 Bcd |
| 400 mM | 20.0 ± 1.4 Ad | 25.5 ± 1.8 Ac | 20.4 ± 7.4 Acd | |
| Total PI | 0 mM | 18.3 ± 1.9 Bc | 23.3 ± 2.5 Ac | 25.7 ± 0.9 Bc |
| 400 mM | 28.9 ± 2.3 Ad | 31.3 ± 3.0 Ad | 61.6 ± 7.8 Ac | |
| Pigments | [NaCl] | Temperature | ||
|---|---|---|---|---|
| (mg g−1 DW) | 26 °C | 35 °C | 45 °C | |
| Neoxanthin (Neo) | 0 mM | 0.101 ± 0.006 Ac | 0.121 ± 0.014 Ac | 0.112 ± 0.003 Ac |
| 400 mM | 0.058 ± 0.004 Bc | 0.061 ± 0.006 Bc | 0.069 ± 0.007 Bc | |
| Violaxanthin (Viol) | 0 mM | 0.116 ± 0.008 Ac | 0.120 ± 0.014 Ac | 0.105 ± 0.005 Ac |
| 400 mM | 0.067 ± 0.003 Bc | 0.051 ± 0.009 Bc | 0.042 ± 0.004 Bc | |
| Anteraxanthin (Ant) | 0 mM | 0.004 ± 0.001 Ad | 0.005 ± 0.000 Ad | 0.013 ± 0.001 Bc |
| 400 mM | 0.006 ± 0.000 Ad | 0.009 ± 0.001 Ad | 0.022 ± 0.002 Ac | |
| Zeaxanthin (Zea) | 0 mM | 0.000 ± 0.000 Bd | 0.002 ± 0.001 Bd | 0.011 ± 0.003 Bc |
| 400 mM | 0.003 ± 0.000 Ad | 0.015 ± 0.005 Ad | 0.035 ± 0.007 Ac | |
| Viol+Ant+Zea | 0 mM | 0.120 ± 0.008 Ac | 0.126 ± 0.012 Ac | 0.129 ± 0.002 Ac |
| 400 mM | 0.075 ± 0.003 Bc | 0.075 ± 0.004 Bc | 0.099 ± 0.011 Bc | |
| DEPS | 0 mM | 0.018 ± 0.002 Ac | 0.046 ± 0.021 Bc | 0.139 ± 0.023 Bc |
| 400 mM | 0.070 ± 0.005 Ad | 0.282 ± 0.077 Ad | 0.442 ± 0.034 Ac | |
| Lutein | 0 mM | 0.257 ± 0.018 Ad | 0.338 ± 0.037 Acd | 0.367 ± 0.008 Ac |
| 400 mM | 0.174 ± 0.011 Bd | 0.196 ± 0.015 Bcd | 0.245 ± 0.023 Bc | |
| α-Carotene | 0 mM | 0.019 ± 0.002 Ad | 0.054 ± 0.001 Ac | 0.050 ± 0.004 Ac |
| 400 mM | 0.032 ± 0.005 Ad | 0.050 ± 0.010 Ac | 0.055 ± 0.005 Ac | |
| β-Carotene | 0 mM | 0.007 ± 0.001 Ad | 0.020 ± 0.002 Ac | 0.017 ± 0.001 Ac |
| 400 mM | 0.007 ± 0.001 Ac | 0.011 ± 0.002 Bc | 0.013 ± 0.002 Ac | |
| Parameter | [NaCl] | Temperature (°C) | ||
|---|---|---|---|---|
| 26 °C | 35 °C | 45 °C | ||
| TFA (mg g−1 DW) | 0 mM | 13.65 ± 1.50 Ac | 12.80 ± 1.17 Ac | 12.26 ± 0.34 Ac |
| 400 mM | 8.10 ± 1.40 Bc | 7.94 ± 0.53 Bc | 6.80 ± 0.65 Bc | |
| C18:3 (mol%) | 0 mM | 41.52 ± 0.27 Ac | 35.75 ± 1.90 Ac | 37.05 ± 0.41 Ac |
| 400 mM | 33.82 ± 2.44 Bc | 34.72 ± 1.01 Ac | 24.53 ± 2.43 Bd | |
| C18:2 (mol%) | 0 mM | 19.51 ± 0.33 Bd | 22.50 ± 1.03 Ac | 21.93 ± 0.60 Bcd |
| 400 mM | 22.94 ± 0.55 Ac | 24.15 ± 0.44 Ac | 23.87 ± 1.35 Ac | |
| C18:1 (mol%) | 0 mM | 5.73 ± 0.08 Ad | 7.35 ± 0.26 Acd | 7.66 ± 0.24 Bc |
| 400 mM | 6.06 ± 0.76 Ad | 6.28 ± 0.83 Acd | 8.16 ± 0.56 Ac | |
| C18:0 (mol%) | 0 mM | 2.65 ± 0.21 Ac | 2.64 ± 0.24 Ac | 2.00 ± 0.19 Ac |
| 400 mM | 2.92 ± 0.27 Ac | 2.89 ± 0.30 Ac | 3.25 ± 0.42 Ac | |
| C16:1 (mol%) | 0 mM | 3.68 ± 0.23 Ac | 3.39 ± 0.40 Ac | 3.92 ± 0.12 Ac |
| 400 mM | 3.76 ± 0.23 Ac | 3.17 ± 0.03 Ac | 3.27 ± 0.28 Ac | |
| C16:0 (mol%) | 0 mM | 25.67 ± 0.68 Ac | 27.12 ± 2.35 Ac | 26.30 ± 0.80 Bc |
| 400 mM | 28.36 ± 1.60 Acd | 26.88 ± 1.18 Ad | 34.67 ± 2.90 Ac | |
| Less Rep (mol%) | 0 mM | 1.13 ± 0.07 Ac | 1.06 ± 0.14 Ac | 0.95 ± 0.04 Ac |
| 400 mM | 1.13 ± 0.18 Ac | 1.02 ± 0.07 Ac | 1.01 ± 0.14 Ac | |
| DBI | 0 mM | 6.47 ± 0.22 Ac | 5.94 ± 0.76 Ac | 6.10 ± 0.25 Ac |
| 400 mM | 5.25 ± 0.48 Ac | 5.67 ± 0.31 Ac | 3.77 ± 0.65 Bd | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jorge, T.F.; Ramalho, J.C.; Alseekh, S.; Pais, I.P.; Leitão, A.E.; Rodrigues, A.P.; Scotti-Campos, P.; Ribeiro-Barros, A.I.; Fernie, A.R.; António, C. Will Casuarina glauca Stress Resilience Be Maintained in the Face of Climate Change? Metabolites 2021, 11, 593. https://doi.org/10.3390/metabo11090593
Jorge TF, Ramalho JC, Alseekh S, Pais IP, Leitão AE, Rodrigues AP, Scotti-Campos P, Ribeiro-Barros AI, Fernie AR, António C. Will Casuarina glauca Stress Resilience Be Maintained in the Face of Climate Change? Metabolites. 2021; 11(9):593. https://doi.org/10.3390/metabo11090593
Chicago/Turabian StyleJorge, Tiago F., José C. Ramalho, Saleh Alseekh, Isabel P. Pais, António E. Leitão, Ana P. Rodrigues, Paula Scotti-Campos, Ana I. Ribeiro-Barros, Alisdair R. Fernie, and Carla António. 2021. "Will Casuarina glauca Stress Resilience Be Maintained in the Face of Climate Change?" Metabolites 11, no. 9: 593. https://doi.org/10.3390/metabo11090593
APA StyleJorge, T. F., Ramalho, J. C., Alseekh, S., Pais, I. P., Leitão, A. E., Rodrigues, A. P., Scotti-Campos, P., Ribeiro-Barros, A. I., Fernie, A. R., & António, C. (2021). Will Casuarina glauca Stress Resilience Be Maintained in the Face of Climate Change? Metabolites, 11(9), 593. https://doi.org/10.3390/metabo11090593