Circulating Metabolites Associated with Body Fat and Lean Mass in Adults with Overweight/Obesity
Abstract
1. Introduction
2. Results
2.1. Circulating Metabolites Associated with Body Composition Measures
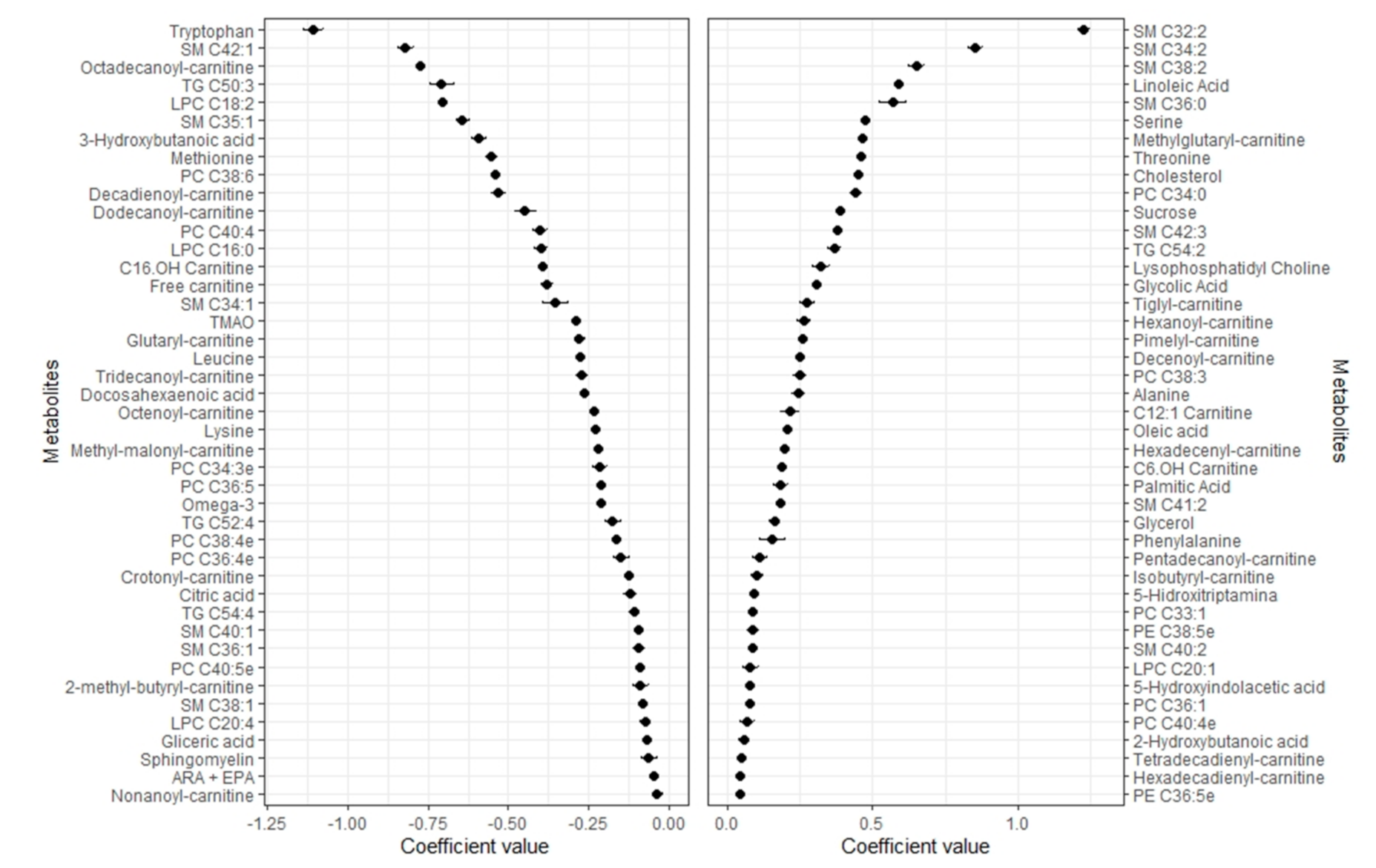
2.2. Metabolomic Profile of Body Fat
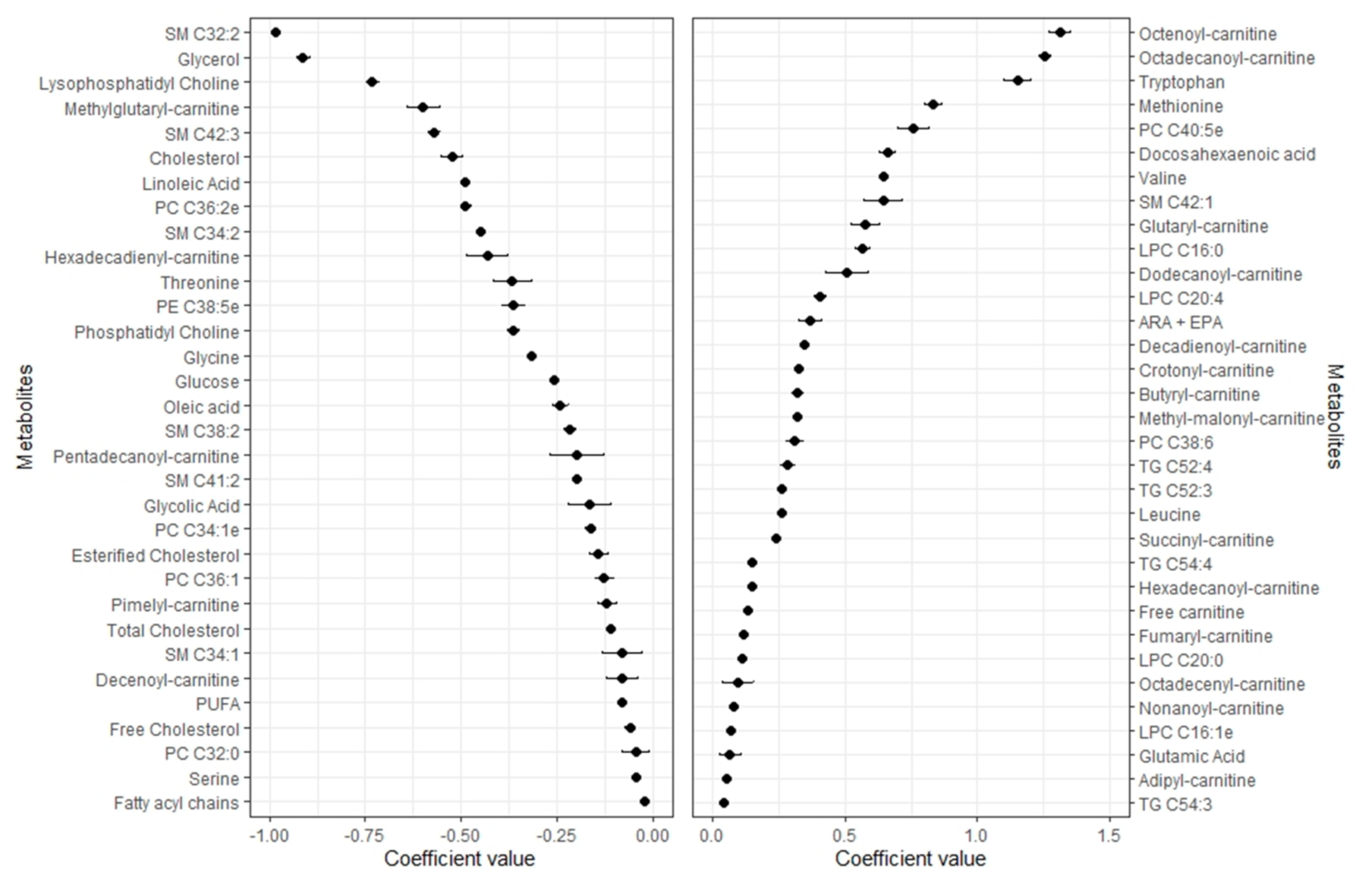
2.3. Metabolomic Profile of Lean Mass
2.4. Pearson Correlations between Metabolomic Profiles and Body Compartments
3. Discussion
4. Materials and Methods
4.1. Study Design and Participants
4.2. Anthropometry and Routine Biochemical Measurements
4.3. Body Composition Assessment
4.4. Multiplatform Targeted Metabolomics
4.5. Automated Plasma Sample Extraction
4.5.1. 1H-NMR
4.5.2. LC-HRMS
4.5.3. GC-HRMS
4.6. Statistical Analyses
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Aslibekyan, S.; Timothy Garvey, W. Obesity and cardiometabolic disease-more than meets the eye. Nat. Rev. Endocrinol. 2017, 13, 566–568. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.Y.; Fang, W.H.; Wang, C.C.; Kao, T.W.; Chang, Y.W.; Yang, H.F.; Wu, C.J.; Sun, Y.S.; Chen, W.L. Cardiometabolic Risks in Different Combination of Anthropometric Parameters and Percentage Body Fat. Fat. Sci. Rep. 2019, 9, 14104. [Google Scholar] [CrossRef] [PubMed]
- Khazem, S.; Itani, L.; Kreidieh, D.; El Masri, D.; Tannir, H.; Citarella, R.; El Ghoch, M. Reduced lean body mass and cardiometabolic diseases in adult males with overweight and obesity: A pilot study. Int. J. Environ. Res. Public Health 2018, 15, 2754. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.H.; Keum, N.N.; Hu, F.B.; Orav, E.J.; Rimm, E.B.; Willett, W.C.; Giovannucci, E.L. Predicted lean body mass, fat mass, and all cause and cause specific mortality in men: Prospective US cohort study. BMJ 2018, 362, k2575. [Google Scholar] [CrossRef] [PubMed]
- Vasan, S.K.; Osmond, C.; Canoy, D.; Christodoulides, C.; Neville, M.J.; Di Gravio, C.; Fall, C.H.D.; Karpe, F. Comparison of regional fat measurements by dual-energy X-ray absorptiometry and conventional anthropometry and their association with markers of diabetes and cardiovascular disease risk. Int. J. Obes. 2018, 42, 850–857. [Google Scholar] [CrossRef] [PubMed]
- Lustgarten, M.S.; Lyn Price, L.; Phillips, E.M.; Fielding, R.A. Serum glycine is associated with regional body fat and insulin resistance in functionally-limited older adults. PLoS ONE 2013, 8. [Google Scholar] [CrossRef] [PubMed]
- Murphy, R.A.; Moore, S.C.; Playdon, M.; Meirelles, O.; Newman, A.B.; Milijkovic, I.; Kritchevsky, S.B.; Schwartz, A.; Goodpaster, B.H.; Sampson, J.; et al. Metabolites Associated with Lean Mass and Adiposity in Older Black Men. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2017, 72, 1352–1359. [Google Scholar] [CrossRef] [PubMed]
- Mai, M.; Tönjes, A.; Kovacs, P.; Stumvoll, M.; Fiedler, G.M.; Leichtle, A.B. Serum levels of acylcarnitines are altered in prediabetic conditions. PLoS ONE 2013, 8. [Google Scholar] [CrossRef] [PubMed]
- Jourdan, C.; Petersen, A.K.; Gieger, C.; Döring, A.; Illig, T.; Wang-Sattler, R.; Meisinger, C.; Peters, A.; Adamski, J.; Prehn, C.; et al. Body fat free mass is associated with the serum metabolite profile in a population-based study. PLoS ONE 2012, 7. [Google Scholar] [CrossRef]
- Lustgarten, M.S.; Price, L.L.; Phillips, E.M.; Kirn, D.R.; Mills, J.; Fielding, R.A. Serum Predictors of Percent Lean Mass in Young Adults. J. Strength Cond. Res. 2016, 30, 2194–2201. [Google Scholar] [CrossRef]
- Lustgarten, M.S.; Price, L.L.; Chale, A.; Phillips, E.M.; Fielding, R.A. Branched Chain Amino acids are associated with muscle mass in functionally limited older adults. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2014, 69, 717–724. [Google Scholar] [CrossRef]
- Cheng, S.; Rhee, E.P.; Larson, M.G.; Lewis, G.D.; McCabe, E.L.; Shen, D.; Palma, M.J.; Roberts, L.D.; Dejam, A.; Souza, A.L.; et al. Metabolite profiling identifies pathways associated with metabolic risk in humans. Circulation 2012, 125, 2222–2231. [Google Scholar] [CrossRef]
- Miller, M.; Stone, N.J.; Ballantyne, C.; Bittner, V.; Criqui, M.H.; Ginsberg, H.N.; Goldberg, A.C.; Howard, W.J.; Jacobson, M.S.; Kris-Etherton, P.M.; et al. Triglycerides and cardiovascular disease: A scientific statement from the American Heart Association. Circulation 2011, 123, 2292–2333. [Google Scholar] [CrossRef]
- Franks, P.W.; Atabaki-Pasdar, N. Causal inference in obesity research. J. Intern. Med. 2017, 281, 222–232. [Google Scholar] [CrossRef]
- Heimerl, S.; Fischer, M.; Baessler, A.; Liebisch, G.; Sigruener, A.; Wallner, S.; Schmitz, G. Alterations of plasma lysophosphatidylcholine species in obesity and weight loss. PLoS ONE 2014, 9. [Google Scholar] [CrossRef]
- Rauschert, S.; Uhl, O.; Koletzko, B.; Kirchberg, F.; Mori, T.A.; Huang, R.C.; Beilin, L.J.; Hellmuth, C.; Oddy, W.H. Lipidomics reveals associations of phospholipids with obesity and insulin resistance in young adults. J. Clin. Endocrinol. Metab. 2016, 101, 871–879. [Google Scholar] [CrossRef]
- Weir, J.M.; Wong, G.; Barlow, C.K.; Greeve, M.A.; Kowalczyk, A.; Almasy, L.; Comuzzie, A.G.; Mahaney, M.C.; Jowett, J.B.M.; Shaw, J.; et al. Plasma lipid profiling in a large population-based cohort. J. Lipid Res. 2013, 54, 2898–2908. [Google Scholar] [CrossRef]
- Chaurasia, B.; Kaddai, V.A.; Lancaster, G.I.; Henstridge, D.C.; Sriram, S.; Galam, D.L.A.; Gopalan, V.; Prakash, K.N.B.; Velan, S.S.; Bulchand, S.; et al. Adipocyte Ceramides Regulate Subcutaneous Adipose Browning, Inflammation, and Metabolism. Cell Metab. 2016, 24, 820–834. [Google Scholar] [CrossRef]
- Chatterjee, S. Sphingolipids in atherosclerosis and vascular biology. Arterioscler. Thromb. Vasc. Biol. 1998, 18, 1523–1533. [Google Scholar] [CrossRef]
- Barber, M.N.; Risis, S.; Yang, C.; Meikle, P.J.; Staples, M.; Febbraio, M.A.; Bruce, C.R. Plasma lysophosphatidylcholine levels are reduced in obesity and type 2 diabetes. PLoS ONE 2012, 7. [Google Scholar] [CrossRef]
- Mak, I.L.; Cohen, T.R.; Vanstone, C.A.; Weiler, H.A. Increased adiposity in children with obesity is associated with low red blood cell omega-3 fatty acid status and inadequate polyunsaturated fatty acid dietary intake. Pediatr. Obes. 2020, 15. [Google Scholar] [CrossRef]
- Alvheim, A.R.; Malde, M.K.; Osei-Hyiaman, D.; Hong Lin, Y.; Pawlosky, R.J.; Madsen, L.; Kristiansen, K.; Frøyland, L.; Hibbeln, J.R. Dietary linoleic acid elevates endogenous 2-AG and anandamide and induces obesity. Obesity 2012, 20, 1984–1994. [Google Scholar] [CrossRef]
- Malodobra-Mazur, M.; Cierzniak, A.; Dobosz, T. Oleic acid influences the adipogenesis of 3T3-L1 cells via DNA Methylation and may predispose to obesity and obesity-related disorders. Lipids Health Dis. 2019, 18. [Google Scholar] [CrossRef]
- Takashina, C.; Tsujino, I.; Watanabe, T.; Sakaue, S.; Ikeda, D.; Yamada, A.; Sato, T.; Ohira, H.; Otsuka, Y.; Oyama-Manabe, N.; et al. Associations among the plasma amino acid profile, obesity, and glucose metabolism in Japanese adults with normal glucose tolerance. Nutr. Metab. 2016, 13. [Google Scholar] [CrossRef]
- Yamakado, M.; Tanaka, T.; Nagao, K.; Ishizaka, Y.; Mitushima, T.; Tani, M.; Toda, A.; Toda, E.; Okada, M.; Miyano, H.; et al. Plasma amino acid profile is associated with visceral fat accumulation in obese Japanese subjects. Clin. Obes. 2012, 2, 29–40. [Google Scholar] [CrossRef]
- Johnson, D.J.; Colpoys, M.; Smith, R.J. Branched chain amino acid uptake and muscle free amino acid concentrations predict postoperative muscle nitrogen balance. Ann. Surg. 1986, 204, 513–523. [Google Scholar] [CrossRef]
- She, P.; Van Horn, C.; Reid, T.; Hutson, S.M.; Cooney, R.N.; Lynch, C.J. Obesity-related elevations in plasma leucine are associated with alterations in enzymes involved in branched-chain amino acid metabolism. Am. J. Physiol. Endocrinol. Metab. 2007, 293. [Google Scholar] [CrossRef]
- Hansen, T.T.; Mead, B.R.; García-Gavilán, J.F.; Korndal, S.K.; Harrold, J.A.; Camacho-Barcía, L.; Ritz, C.; Christiansen, P.; Salas-Salvadó, J.; Hjorth, M.F.; et al. Is reduction in appetite beneficial for body weight management in the context of overweight and obesity? Yes, according to the SATIN (Satiety Innovation) study. J. Nutr. Sci. 2019, 8, e39. [Google Scholar] [CrossRef] [PubMed]
- Hansen, T.T.; Hjorth, M.F.; Sandby, K.; Andersen, S.V.; Astrup, A.; Ritz, C.; Bulló, M.; Camacho-Barcía, M.L.; García-Gavilán, J.F.; Salas-Salvadó, J.; et al. Predictors of successful weight loss with relative maintenance of fat-free mass in individuals with overweight and obesity on an 8-week low-energy diet. Br. J. Nutr. 2019, 122. [Google Scholar] [CrossRef] [PubMed]
- Murphy, R.A.; Ip, E.H.; Zhang, Q.; Boudreau, R.M.; Cawthon, P.M.; Newman, A.B.; Tylavsky, F.A.; Visser, M.; Goodpaster, B.H.; Harris, T.B. Transition to sarcopenia and determinants of transitions in older adults: A population-based study. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2014, 69, 751–758. [Google Scholar] [CrossRef] [PubMed]
- Dam, R.M.V.; Spiegelman, D.; Heymsfield, S.B.; Willett, W.C.; Hu, F.B.; Sun, Q. Comparison of dual-energy x-ray absorptiometric and anthropometric measures of adiposity in relation to adiposity-related biologic factors. Am. J. Epidemiol. 2010, 172, 1442–1454. [Google Scholar] [CrossRef]
- Hernández-Alonso, P.; Giardina, S.; Cañueto, D.; Salas-Salvadó, J.; Cañellas, N.; Bulló, M. Changes in Plasma Metabolite Concentrations after a Low-Glycemic Index Diet Intervention. Mol. Nutr. Food Res. 2019, 63. [Google Scholar] [CrossRef]
- Watanabe, R.; Sugai, C.; Yamazaki, T.; Matsushima, R.; Uchida, H.; Matsumiya, M.; Takatsu, A.; Suzuki, T. Quantitative nuclear magnetic resonance spectroscopy based on PULCON methodology: Application to quantification of invaluable marine toxin, okadaic acid. Toxins 2016, 8, 294. [Google Scholar] [CrossRef]
- Hernández-Alonso, P.; García-Gavilán, J.; Camacho-Barcia, L.; Sjödin, A.; Hansen, T.T.; Harrold, J.; Salas-Salvadó, J.; Halford, J.C.G.; Canudas, S.; Bulló, M. Plasma metabolites associated with homeostatic model assessment of insulin resistance: Metabolite-model design and external validation. Sci. Rep. 2019, 9, 13895. [Google Scholar] [CrossRef]
- Friedman, J.; Hastie, T.; Tibshirani, R. Regularization paths for generalized linear models via coordinate descent. J. Stat. Softw. 2010, 33, 1–22. [Google Scholar] [CrossRef]
| Characteristics | (n = 236) |
|---|---|
| Age, years | 46.4 ± 10.7 |
| Women sex, N (%) | 184 (78) |
| Weight, kg | 87.5 ± 11.2 |
| BMI, kg/m2 | 31.1 ± 2.2 |
| Body fat, % | 42.0 ± 5.6 |
| Lean mass, kg | 47.2 ± 9.2 |
| Glucose, mg/dL | 93.3 ± 11.0 |
| Total cholesterol, mg/dL | 196.0 ± 34.9 |
| HDL-C, mg/dL | 55.7 ± 15.3 |
| LDL-C, mg/dL | 119.9 ± 30.5 |
| Triglycerides, mg/dL | 102.3 ± 48.9 |
| % Body Fat | p-Value | Lean Mass | p-Value | |
|---|---|---|---|---|
| Pearson’s correlation coefficient (95%CI) | 0.80 (0.75, 0.84) | <0.001 | 0.78 (0.72, 0.83) | <0.001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Papandreou, C.; García-Gavilán, J.; Camacho-Barcia, L.; Hansen, T.T.; Sjödin, A.; Harrold, J.A.; Halford, J.C.G.; Bulló, M. Circulating Metabolites Associated with Body Fat and Lean Mass in Adults with Overweight/Obesity. Metabolites 2021, 11, 317. https://doi.org/10.3390/metabo11050317
Papandreou C, García-Gavilán J, Camacho-Barcia L, Hansen TT, Sjödin A, Harrold JA, Halford JCG, Bulló M. Circulating Metabolites Associated with Body Fat and Lean Mass in Adults with Overweight/Obesity. Metabolites. 2021; 11(5):317. https://doi.org/10.3390/metabo11050317
Chicago/Turabian StylePapandreou, Christopher, Jesús García-Gavilán, Lucía Camacho-Barcia, Thea T. Hansen, Anders Sjödin, Joanne A. Harrold, Jason C. G. Halford, and Mònica Bulló. 2021. "Circulating Metabolites Associated with Body Fat and Lean Mass in Adults with Overweight/Obesity" Metabolites 11, no. 5: 317. https://doi.org/10.3390/metabo11050317
APA StylePapandreou, C., García-Gavilán, J., Camacho-Barcia, L., Hansen, T. T., Sjödin, A., Harrold, J. A., Halford, J. C. G., & Bulló, M. (2021). Circulating Metabolites Associated with Body Fat and Lean Mass in Adults with Overweight/Obesity. Metabolites, 11(5), 317. https://doi.org/10.3390/metabo11050317








