The Effect of Kefir Supplementation on Improving Human Endurance Exercise Performance and Antifatigue
Abstract
1. Introduction
2. Results
2.1. Subject’s Basic Biochemical Parameters and Hematology
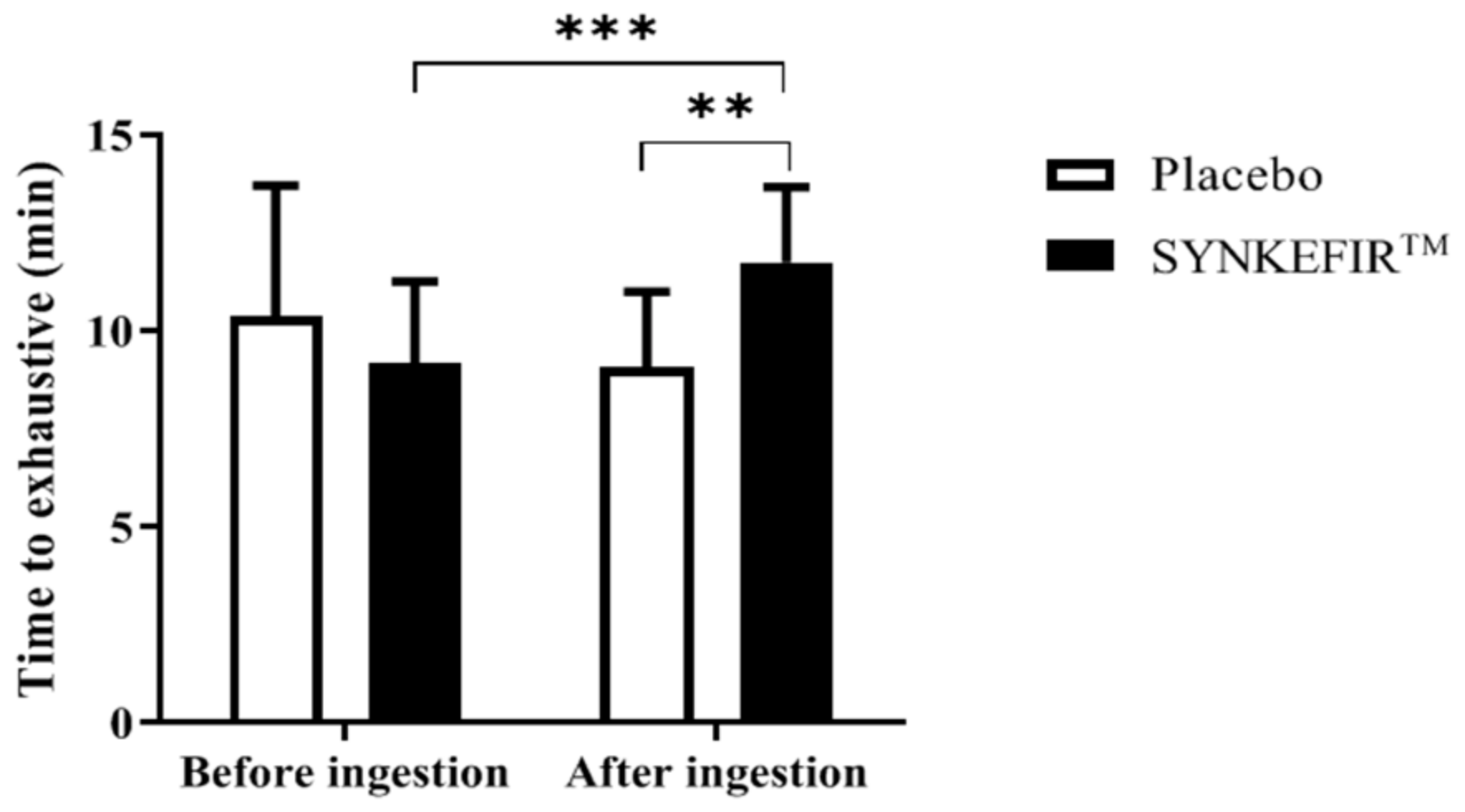
2.2. Effects of SYNKEFIR™ Supplementation on Endurance Performance
2.3. Effect of SYNKEFIR™ Supplementation on Physiological Adaptation and Biochemical Indices
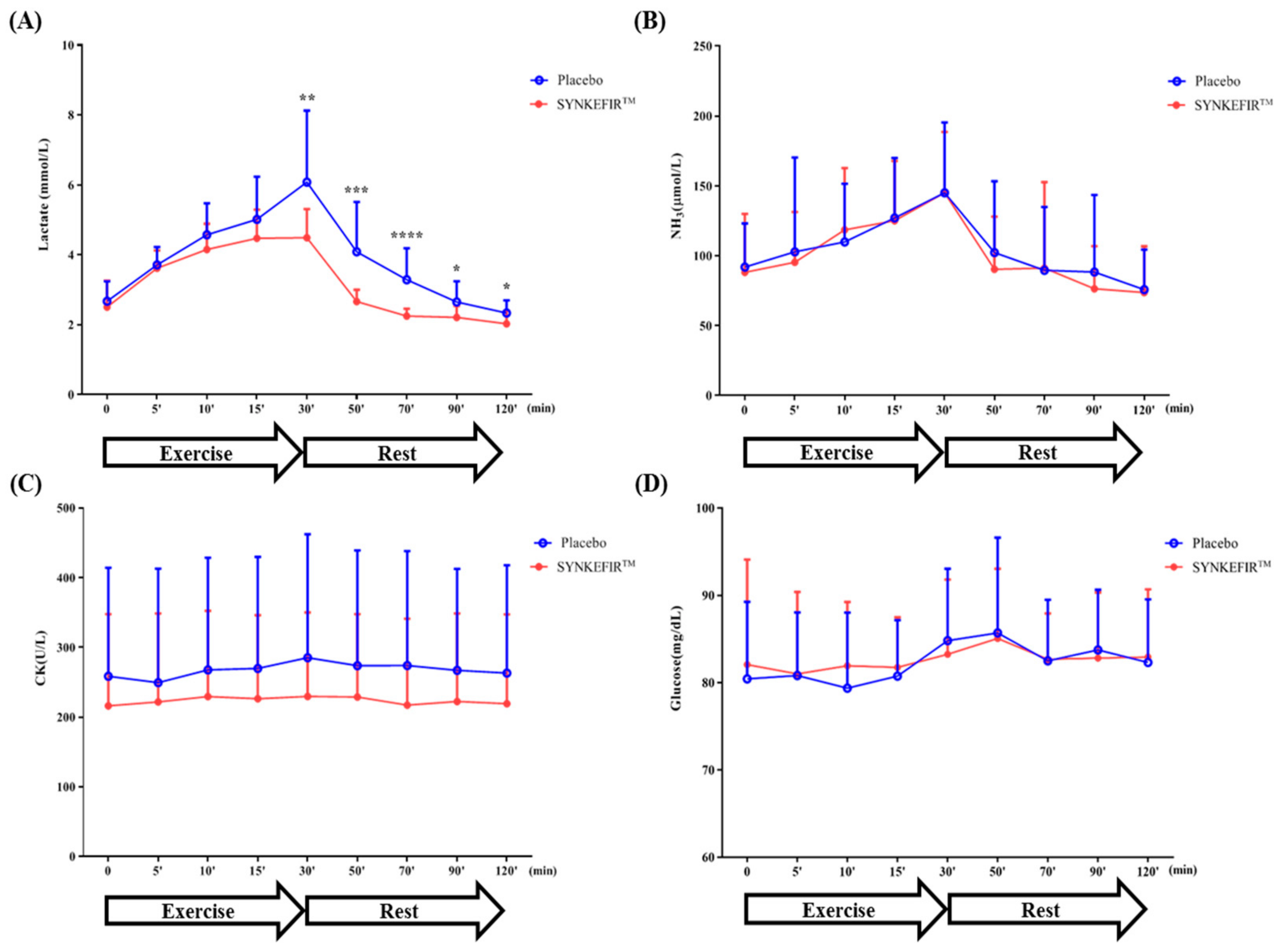
2.4. Effect of SYNKEFIR™ Supplementation on Exercise/Rest Biochemical Parameters and Hematology
2.5. Effect of SYNKEFIR™ Supplementation on Body Composition
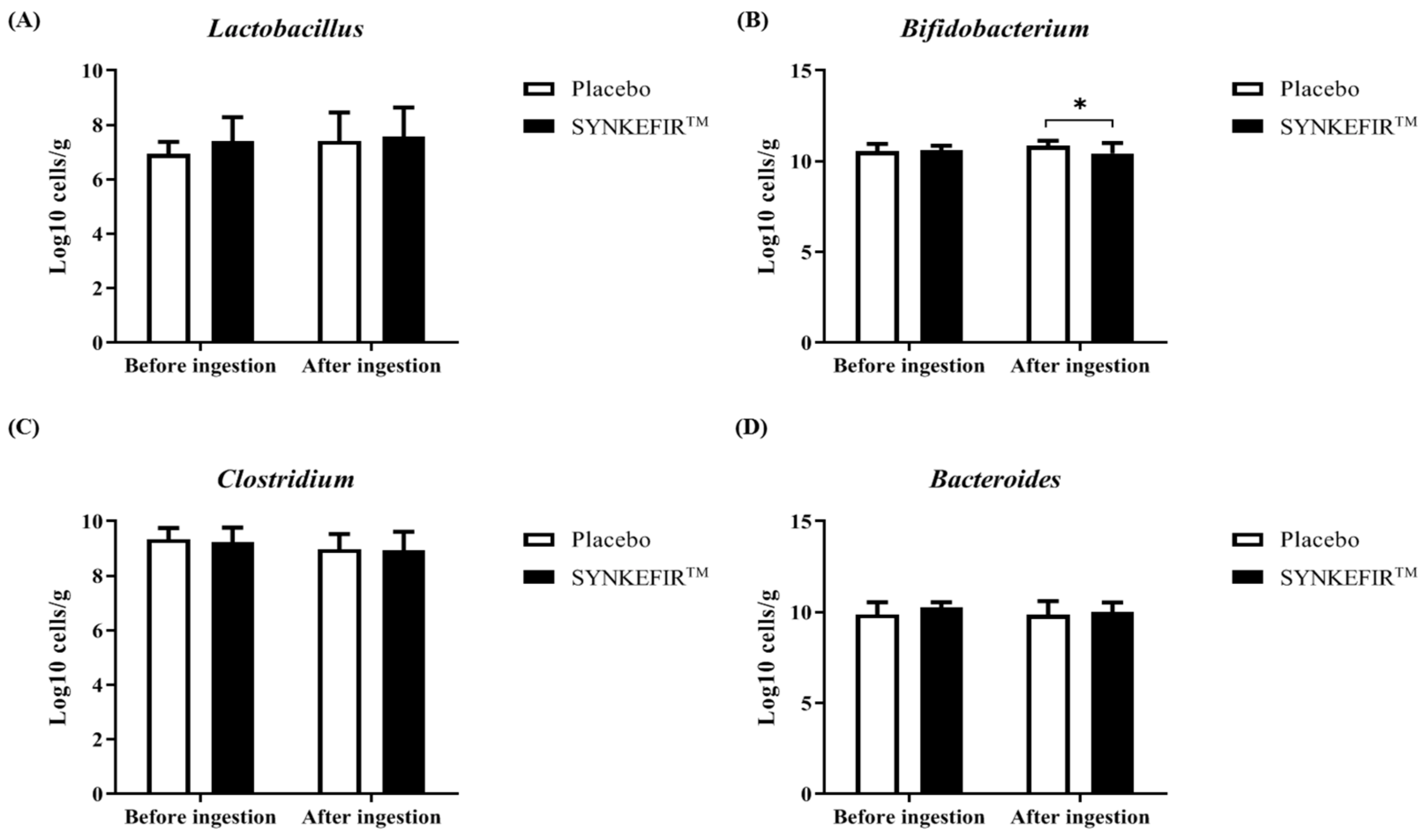
2.6. Effect of SYNKEFIR™ Supplementation on the Gut Microbiota
3. Discussion
4. Materials and Methods
4.1. SYNKEFIR™ and Placebo Preparation
4.2. Subjects
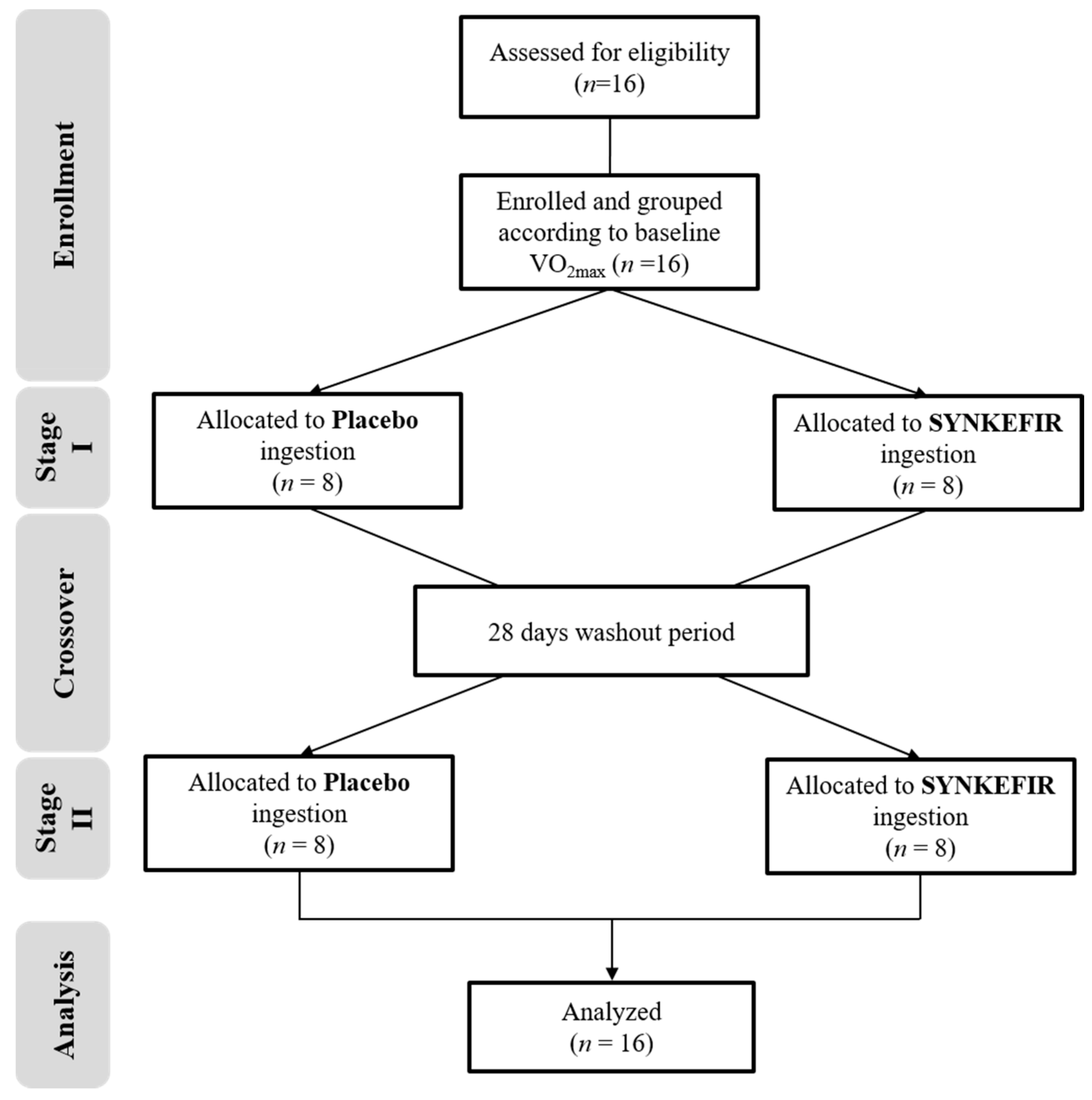
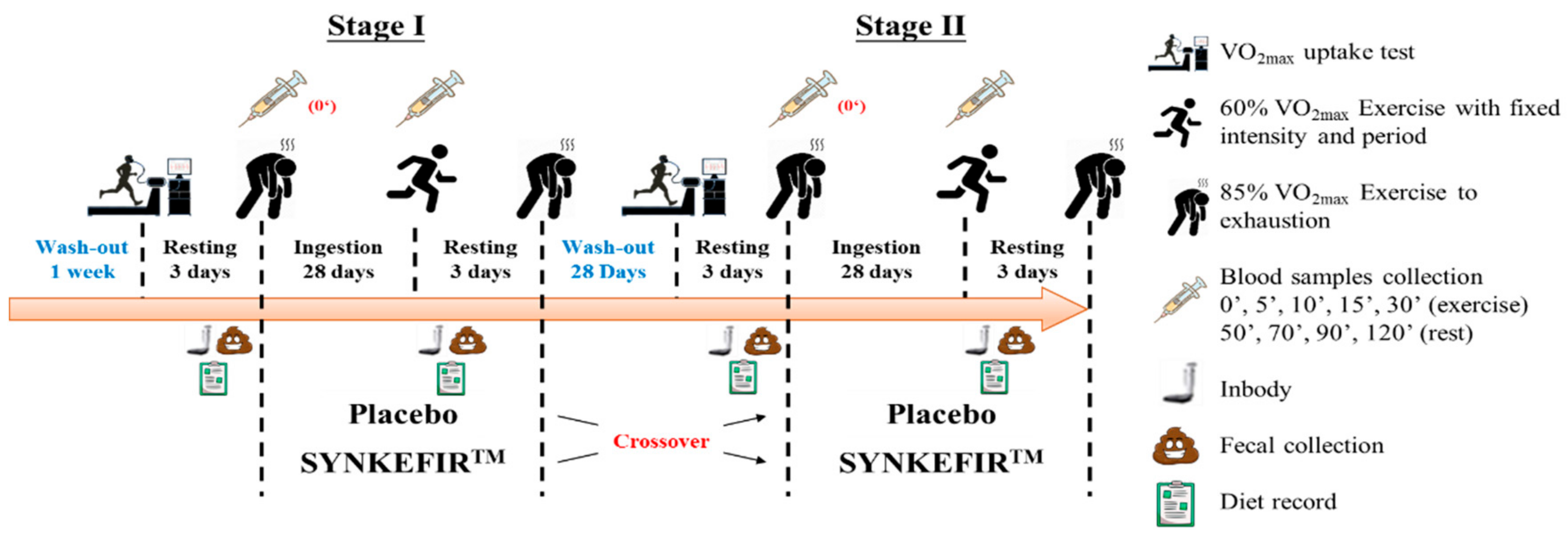
4.3. Experimental Design
4.4. VO2max and Endurance Performance Test
4.5. Body Composition
4.6. Clinical Biochemistry and Hematology Analysis
4.7. Detection of Lactobacillus spp., Bifidobacterium spp., Clostridium spp., and Bacteroides spp. by Quantitative PCR
4.8. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Wan, J.J.; Qin, Z.; Wang, P.Y.; Sun, Y.; Liu, X. Muscle fatigue: General understanding and treatment. Exp. Mol. Med. 2017, 49, e384. [Google Scholar] [CrossRef]
- Finsterer, J. Biomarkers of peripheral muscle fatigue during exercise. BMC Musculoskelet. Disord. 2012, 13, 218. [Google Scholar] [CrossRef] [PubMed]
- Chi, A.; Li, H.; Kang, C.; Guo, H.; Wang, Y.; Guo, F.; Tang, L. Anti-fatigue activity of a novel polysaccharide conjugates from Ziyang green tea. Int. J. Biol. Macromol. 2015, 80, 566–572. [Google Scholar] [CrossRef] [PubMed]
- Rivera-Brown, A.M.; Frontera, W.R. Principles of exercise physiology: Responses to acute exercise and long-term adaptations to training. Am. Acad. Phys. Med. Rehabil. 2012, 4, 797–804. [Google Scholar] [CrossRef]
- Proia, P.; Di Liegro, C.M.; Schiera, G.; Fricano, A.; Di Liegro, I. Lactate as a metabolite and a regulator in the central nervous system. Int. J. Mol. Sci. 2016, 17, 1450. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.C.; Huang, W.C.; Chiu, C.C.; Chang, Y.K.; Huang, C.C. Whey protein improves exercise performance and biochemical profiles in trained mice. Med. Sci. Sports Exerc. 2014, 46, 1517–1524. [Google Scholar] [CrossRef] [PubMed]
- Allen, J.M.; Mailing, L.J.; Niemiro, G.M.; Moore, R.; Cook, M.D.; White, B.A.; Holscher, H.D.; Woods, J.A. Exercise alters gut microbiota composition and function in lean and obese humans. Med. Sci. Sports Exerc. 2018, 50, 747–757. [Google Scholar] [CrossRef]
- Clarke, S.F.; Murphy, E.F.; O’Sullivan, O.; Lucey, A.J.; Humphreys, M.; Hogan, A.; Hayes, P.; O’Reilly, M.; Jeffery, I.B.; Wood-Martin, R.; et al. Exercise and associated dietary extremes impact on gut microbial diversity. Gut 2014, 63, 1913–1920. [Google Scholar] [CrossRef]
- Mohr, A.E.; Jäger, R.; Carpenter, K.C.; Kerksick, C.M.; Purpura, M.; Townsend, J.R.; West, N.P.; Black, K.; Gleeson, M.; Pyne, D.B.; et al. The athletic gut microbiota. J. Int. Soc. Sports Nutr. 2020, 17, 24. [Google Scholar] [CrossRef] [PubMed]
- Jung, Y.P.; Earnest, C.P.; Koozehchian, M.; Cho, M.; Barringer, N.; Walker, D.; Rasmussen, C.; Greenwood, M.; Murano, P.S.; Kreider, R.B. Effects of ingesting a pre-workout dietary supplement with and without synephrine for 8 weeks on training adaptations in resistance-trained males. J. Int. Soc. Sports Nutr. 2017, 14, 1. [Google Scholar] [CrossRef]
- LeBlanc, J.G.; Chain, F.; Martín, R.; Bermúdez-Humarán, L.G.; Courau, S.; Langella, P. Beneficial effects on host energy metabolism of short-chain fatty acids and vitamins produced by commensal and probiotic bacteria. Microb. Cell Fact. 2017, 16, 79. [Google Scholar] [CrossRef]
- Mach, N.; Fuster-Botella, D. Endurance exercise and gut microbiota: A review. J. Sport Health Sci. 2017, 6, 179–197. [Google Scholar] [CrossRef]
- Bensmira, M.; Nsabimana, C.; Jiang, B. Effects of fermentation conditions and homogenization pressure on the rheological properties of Kefir. Food Sci. Technol. 2010, 43, 1180–1184. [Google Scholar] [CrossRef]
- Serafini, F.; Turroni, F.; Ruas-Madiedo, P.; Lugli, G.A.; Milani, C.; Duranti, S.; Zamboni, N.; Bottacini, F.; van Sinderen, D.; Margolles, A.; et al. Kefir fermented milk and kefiran promote growth of Bifidobacterium bifidum PRL2010 and modulate its gene expression. Int. J. Food Microbiol. 2014, 178, 50–59. [Google Scholar] [CrossRef]
- Wszolek, M.; Tamime, A.; Muir, D.; Barclay, M. Properties of kefir made in Scotland and Poland using bovine, caprine and ovine milk with different starter cultures. LWT Food Sci. Technol. 2001, 34, 251–261. [Google Scholar] [CrossRef]
- Altay, F.; Karbancıoglu-Güler, F.; Daskaya-Dikmen, C.; Heperkan, D. A review on traditional Turkish fermented non-alcoholic beverages: Microbiota, fermentation process and quality characteristics. Int. J. Food Microbiol. 2013, 167, 44–56. [Google Scholar] [CrossRef] [PubMed]
- Slattery, C.; Cotter, P.D.; O’Toole, P.W. Analysis of health benefits conferred by Lactobacillus species from kefir. Nutrients 2019, 11, 1252. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, K.V.; Stewart, L.K.; Forney, L.A.; Aryana, K.J.; Prinyawiwatkul, W.; Boeneke, C.A. The effects of postexercise consumption of a kefir beverage on performance and recovery during intensive endurance training. J. Dairy Sci. 2015, 98, 7446–7449. [Google Scholar] [CrossRef]
- Hsu, Y.J.; Huang, W.C.; Lin, J.S.; Chen, Y.M.; Ho, S.T.; Huang, C.C.; Tung, Y.T. Kefir supplementation modifies gut microbiota composition, reduces physical fatigue, and improves exercise performance in mice. Nutrients 2018, 10, 862. [Google Scholar] [CrossRef] [PubMed]
- Barton, W.; Penney, N.C.; Cronin, O.; Garcia-Perez, I.; Molloy, M.G.; Holmes, E.; Shanahan, F.; Cotter, P.D.; O’Sullivan, O. The microbiome of professional athletes differs from that of more sedentary subjects in composition and particularly at the functional metabolic level. Gut 2018, 67, 625–633. [Google Scholar] [CrossRef]
- Samuel, B.S.; Shaito, A.; Motoike, T.; Rey, F.E.; Backhed, F.; Manchester, J.K.; Hammer, R.E.; Williams, S.C.; Crowley, J.; Yanagisawa, M.; et al. Effects of the gut microbiota on host adiposity are modulated by the short-chain fatty-acid binding G protein-coupled receptor, Gpr41. Proc. Natl. Acad. Sci. USA 2008, 105, 16767–16772. [Google Scholar] [CrossRef]
- Clark, A.; Mach, N. The crosstalk between the gut microbiota and mitochondria during exercise. Front. Physiol. 2017, 8, 319. [Google Scholar] [CrossRef]
- Leite, G.S.F.; Resende Master Student, A.S.; West, N.P.; Lancha, A.H., Jr. Probiotics and sports: A new magic bullet? Nutrition 2019, 60, 152–160. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.L.; Hsu, Y.J.; Ho, H.H.; Chang, Y.C.; Kuo, Y.W.; Yeh, Y.T.; Tsai, S.Y.; Chen, C.W.; Chen, J.F.; Huang, C.C.; et al. Bifidobacterium longum subsp longum OLP-01 supplementation during endurance running training improves exercise performance in middle- and long-distance runners: A double-blind controlled trial. Nutrients 2020, 12, 1972. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.M.; Wei, L.; Chiu, Y.S.; Hsu, Y.J.; Tsai, T.Y.; Wang, M.F.; Huang, C.C. Lactobacillus plantarum TWK10 Supplementation improves exercise performance and increases muscle mass in mice. Nutrients 2016, 8, 205. [Google Scholar] [CrossRef]
- Huang, W.C.; Lee, M.C.; Lee, C.C.; Ng, K.S.; Hsu, Y.J.; Tsai, T.Y.; Young, S.L.; Lin, J.S.; Huang, C.C. Effect of Lactobacillus plantarum TWK10 on exercise physiological adaptation, performance, and body composition in healthy humans. Nutrients 2019, 11, 2836. [Google Scholar] [CrossRef] [PubMed]
- Salarkia, N.; Ghadamli, L.; Zaeri, F.; Sabaghian Rad, L. Effects of probiotic yogurt on performance, respiratory and digestive systems of young adult female endurance swimmers: A randomized controlled trial. Med. J. Islam. Repub. Iran. 2013, 27, 141–146. [Google Scholar] [PubMed]
- Osnes, J.B.; Hermansen, L. Acid-base balance after maximal exercise of short duration. J. Appl. Physiol. 1972, 32, 59–63. [Google Scholar] [CrossRef] [PubMed]
- Hall, M.M.; Rajasekaran, S.; Thomsen, T.W.; Peterson, A.R. Lactate: Friend or foe. Am. Acad. Phys. Med. Rehabil. 2016, 8, S8–S15. [Google Scholar] [CrossRef]
- Hermansen, L.; Maehlum, S.; Pruett, E.D.R.; Vaage, O.; Waldum, H.; Wessel-Aas, T. Lactate removal at rest and during exercise. In Metabolic Adaptation to Prolonged Physical Exercise; Birkhäuser: Basel, Switzerland, 1975; pp. 101–105. [Google Scholar]
- Chatham, J.C. Lactate—The forgotten fuel! J. Physiol. 2002, 542, 333. [Google Scholar] [CrossRef]
- Coqueiro, A.Y.; de Oliveira Garcia, A.B.; Rogero, M.M.; Tirapegui, J. Probiotic supplementation in sports and physical exercise: Does it present any ergogenic effect? Nutr. Health 2017, 23, 239–249. [Google Scholar] [CrossRef] [PubMed]
- Lisko, D.J.; Johnston, G.P.; Johnston, C.G. Effects of dietary yogurt on the healthy human gastrointestinal (GI) microbiome. Microorganisms 2017, 5, 6. [Google Scholar] [CrossRef]
- Mazani, M.; Nemati, A.; Amani, M.; Haedari, K.; Mogadam, R.A.; Baghi, A.N. The effect of probiotic yoghurt consumption on oxidative stress and inflammatory factors in young females after exhaustive exercise. J. Pak. Med. Assoc. 2018, 68, 1748–1754. [Google Scholar]
- Bruce, R.A.; Kusumi, F.; Hosmer, D. Maximal oxygen intake and nomographic assessment of functional aerobic impairment in cardiovascular disease. Am. Heart J. 1973, 85, 546–562. [Google Scholar] [CrossRef]
- Matsuki, T.; Watanabe, K.; Fujimoto, J.; Kado, Y.; Takada, T.; Matsumoto, K.; Tanaka, R. Quantitative PCR with 16S rRNA-gene-targeted species-specific primers for analysis of human intestinal bifidobacteria. Appl. Environ. Microbiol. 2004, 70, 167–173. [Google Scholar] [CrossRef] [PubMed]
- Kook, S.Y.; Kim, Y.; Kang, B.; Choe, Y.H.; Kim, Y.H.; Kim, S. Characterization of the fecal microbiota differs between age groups in Koreans. Intest. Res. 2018, 16, 246–254. [Google Scholar] [CrossRef]
- Layton, A.; McKay, L.; Williams, D.; Garrett, V.; Gentry, R.; Sayler, G. Development of bacteroides 16S rRNA gene TaqMan-based real-time PCR assays for estimation of total, human, and bovine fecal pollution in water. Appl. Environ. Microbiol. 2006, 72, 4214–4224. [Google Scholar] [CrossRef] [PubMed]
| Baseline | Placebo (n = 16) | SYNKEFIR™ (n = 16) |
|---|---|---|
| Lactate (mmol/L) | 2.23 ± 0.49 | 2.14 ± 0.67 |
| NH3 (μmol/L) | 72 ± 32 | 94 ± 35 |
| CPK (U/L) | 148 ± 45 | 200 ± 124 |
| Glucose (mg/dL) | 91 ± 11 | 89 ± 9 |
| FFA (mmol/L) | 0.50 ± 0.26 | 0.45 ± 0.21 |
| AST (U/L) | 19 ± 4 | 18 ± 8 |
| ALT (U/L) | 23 ± 12 | 22 ± 10 |
| TC (mg/dL) | 211 ± 32 | 191 ± 22 * |
| TG (mg/dL) | 81 ± 24 | 83 ± 26 |
| HDL (mg/dL) | 60 ± 6 | 57 ± 6 |
| LDL (mg/dL) | 112 ± 21 | 106 ± 14 |
| BUN (mg/dL) | 14.3 ± 2.9 | 12.9 ± 1.9 |
| CREA (mg/dL) | 1.16 ± 0.09 | 1.13 ± 0.11 |
| UA (mg/dL) | 5.64 ± 0.99 | 5.63 ± 1.17 |
| TP (g/dL) | 7.48 ± 0.43 | 7.36 ± 0.33 |
| ALB (g/dL) | 5.10 ± 0.33 | 5.11 ± 0.24 |
| Baseline (CBC) | Placebo (n = 16) | SYNKEFIR™ (n = 16) |
|---|---|---|
| WBC (cells/mcL) | 7309 ± 1800 | 6824 ± 1543 |
| Neutrophils (%) | 64.0 ± 12.4 | 59.1 ± 6.8 |
| Lymphocytes (%) | 28.7 ± 10.8 | 31.5 ± 6.4 |
| Monocytes (%) | 5.0 ± 1.5 | 5.9 ± 1.3 |
| Eosinophil (%) | 1.7 ± 1.5 | 2.8 ± 1.9 |
| Basophil (%) | 0.5 ± 0.2 | 0.6 ± 0.3 |
| Platelet (103/mcL) | 297 ± 53 | 289 ± 59 |
| NLR | 3.21 ± 3.24 | 2.00 ± 0.66 |
| PLR | 174.8 ± 99.7 | 140.5 ± 31.8 |
| Placebo (n = 16) | SYNKEFIR™ (n = 16) | |||||
|---|---|---|---|---|---|---|
| Time Point | 0′ | E30′ | R90′ | 0′ | E30′ | R90′ |
| FFA (mmol/L) | 0.63 ± 0.26 | 0.73 ± 0.30 | 0.90 ± 0.22 | 0.55 ± 0.18 | 0.58 ± 0.23 | 0.82 ± 0.20 |
| AST (U/L) | 26 ± 8 | 25 ± 7 | 22 ± 5 | 27 ± 11 | 27 ± 12 | 26 ± 11 |
| ALT (U/L) | 28 ± 14 | 28 ± 15 | 24 ± 9 | 32 ± 22 | 34 ± 20 | 31 ± 21 |
| TC (mg/dL) | 209 ± 33 | 224 ± 39 | 207 ± 30 | 204 ± 33 | 207 ± 23 | 195 ± 23 |
| TG (mg/dL) | 87 ± 36 | 103 ± 36 | 75 ± 29 | 84 ± 19 | 95 ± 21 | 76 ± 22 |
| HDL (mg/dL) | 60 ± 9 | 63 ± 11 | 59 ± 5 | 63 ± 16 | 64 ± 10 | 62 ± 8 |
| LDL (mg/dL) | 111 ± 15 | 117 ± 26 | 110 ± 16 | 110 ± 18 | 112 ± 20 | 105 ± 14 |
| BUN (mg/dL) | 13.9 ± 2.4 | 14.2 ± 2.8 | 14.6 ± 3.1 | 13.7 ± 3.0 | 13.9 ± 2.9 | 14.2 ± 3.3 |
| CREA (mg/dL) | 1.19 ± 0.13 | 1.29 ± 0.13 | 1.24 ± 0.13 | 1.16 ± 0.12 | 1.23 ± 0.12 | 1.20 ± 0.10 |
| UA (mg/dL) | 5.74 ± 1.35 | 6.06 ± 1.33 | 6.10 ± 1.36 | 5.66 ± 1.17 | 6.01 ± 1.15 | 5.95 ± 1.14 |
| TP (g/dL) | 7.58 ± 0.65 | 7.93 ± 0.87 | 7.41 ± 0.48 | 7.83 ± 0.75 | 8.16 ± 1.00 | 7.55 ± 0.58 |
| ALB (g/dL) | 5.1 ± 0.4 | 5.3 ± 0.5 | 5.0 ± 0.4 | 5.3 ± 0.5 | 5.4 ± 0.6 | 5.1 ± 0.5 |
| CBC | Placebo (n = 16) | SYNKEFIR™ (n = 16) | ||||
|---|---|---|---|---|---|---|
| Time Point | 0′ | E30′ | R90′ | 0′ | E30′ | R90′ |
| WBC (cells/mcL) | 6817 ± 1556 | 9144 ± 1693 | 8188 ± 2012 | 7307 ± 2066 | 8797 ± 2295 | 7812 ± 2136 |
| Neutrophils (%) | 59.36 ± 8.17 | 55.60 ± 10.66 | 64.29 ± 7.86 | 59.92 ± 7.32 | 57.68 ± 8.07 | 64.49 ± 8.11 |
| Lymphocytes (%) | 31.27 ± 7.23 | 35.88 ± 10.16 | 27.49 ± 7.24 | 30.61 ± 6.95 | 33.51 ± 7.31 | 27.33 ± 7.43 |
| Monocytes (%) | 5.44 ± 1.70 | 5.67 ± 1.88 | 5.51 ± 1.33 | 5.94 ± 1.92 | 5.72 ± 1.65 | 5.69 ± 1.44 |
| Eosinophil (%) | 3.36 ± 0.84 | 2.26 ± 1.34 | 2.17 ± 1.22 | 2.92 ± 1.09 | 2.54 ± 1.20 | 2.01 ± 0.91 |
| Basophil (%) | 0.6 ± 0.2 | 0.6 ± 0.2 | 0.5 ± 0.2 | 0.6 ± 0.3 | 0.6 ± 0.2 | 0.5 ± 0.1 |
| Platelet (103/mcL) | 281 ± 66 | 320 ± 69 | 284 ± 62 | 286 ± 52 | 308 ± 50 | 285 ± 51 |
| NLR | 2.06 ± 0.78 | 1.76 ± 0.86 | 2.60 ± 1.08 | 2.14 ± 0.88 | 1.88 ± 1.09 | 2.67 ± 1.30 |
| PLR | 140.0 ± 38.3 | 109.6 ± 42.0 | 144.2 ± 59.0 | 137.9 ± 34.0 | 112.9 ± 31.2 | 147.0 ± 42.7 |
| Body Composition | Placebo (n = 16) | SYNKEFIR™ (n = 16) | ||||
|---|---|---|---|---|---|---|
| Before Ingestion | After Ingestion | Change | Before Ingestion | After Ingestion | Change | |
| Body Weight (g) | 70.69 ± 9.52 | 70.75 ± 9.69 | 0.06 ± 1.15 | 72.38 ± 10.84 | 72.33 ± 10.68 | −0.04 ± 1.07 |
| BMI (kg/m2) | 23.66 ± 2.83 | 23.67 ± 2.84 | 0.01 ± 0.41 | 24.00 ± 2.95 | 24.56 ± 3.92 | 0.56 ± 2.43 |
| Fat Mass (%) | 20.14 ± 6.86 | 19.91 ± 7.03 | −0.23 ± 1.28 | 21.01 ± 7.07 | 20.31 ± 6.26 | −0.69 ± 1.40 |
| Muscle Mass (kg) | 31.76 ± 3.21 | 32.02 ± 3.39 | 0.26 ± 0.79 | 32.14 ± 3.38 | 32.48 ± 3.64 | 0.33 ± 0.75 |
| Group | Placebo in Stage I | SYNKEFIR™ in Stage I |
|---|---|---|
| Subjects | n = 8 | n = 8 |
| Age | 25.6 ± 4.1 | 24.6 ± 2.8 |
| Height (cm) | 172.5 ± 5.0 | 174.6 ± 5.2 |
| VO2max (mL/kg/min) | 46.6 ± 8.2 | 47.3 ± 6.5 |
| Target Bacterial Group | Primer Sequence (5′ → 3′) | Predicted Product Size (bp) |
|---|---|---|
| Lactobacillus genus [37] | F: AGCAGTAGGGAATCTTCCA R: CACCGCTACACATGGAG | 341 |
| Bifidobacterium genus [37] | F: GGGTGGTAATGCCGGATG | 442 |
| R: TAAGCGATGGACTTTCACACC | ||
| Clostridium genus [37] | F: CGGTACCTGACTAAGAAGC | 429 |
| R: AGTTTYATTCTTGCGAACG | ||
| Bacteroides genus [38] | F: GGGTTTAAAGGGAGCGTAGG | 116 |
| R: CTACACCACGAATTCCGCCT |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, M.-C.; Jhang, W.-L.; Lee, C.-C.; Kan, N.-W.; Hsu, Y.-J.; Ho, C.-S.; Chang, C.-H.; Cheng, Y.-C.; Lin, J.-S.; Huang, C.-C. The Effect of Kefir Supplementation on Improving Human Endurance Exercise Performance and Antifatigue. Metabolites 2021, 11, 136. https://doi.org/10.3390/metabo11030136
Lee M-C, Jhang W-L, Lee C-C, Kan N-W, Hsu Y-J, Ho C-S, Chang C-H, Cheng Y-C, Lin J-S, Huang C-C. The Effect of Kefir Supplementation on Improving Human Endurance Exercise Performance and Antifatigue. Metabolites. 2021; 11(3):136. https://doi.org/10.3390/metabo11030136
Chicago/Turabian StyleLee, Mon-Chien, Wei-Lun Jhang, Chia-Chia Lee, Nai-Wen Kan, Yi-Ju Hsu, Chin-Shan Ho, Chun-Hao Chang, Yi-Chen Cheng, Jin-Seng Lin, and Chi-Chang Huang. 2021. "The Effect of Kefir Supplementation on Improving Human Endurance Exercise Performance and Antifatigue" Metabolites 11, no. 3: 136. https://doi.org/10.3390/metabo11030136
APA StyleLee, M.-C., Jhang, W.-L., Lee, C.-C., Kan, N.-W., Hsu, Y.-J., Ho, C.-S., Chang, C.-H., Cheng, Y.-C., Lin, J.-S., & Huang, C.-C. (2021). The Effect of Kefir Supplementation on Improving Human Endurance Exercise Performance and Antifatigue. Metabolites, 11(3), 136. https://doi.org/10.3390/metabo11030136








