Plasma Acylcarnitines during Pregnancy and Neonatal Anthropometry: A Longitudinal Study in a Multiracial Cohort
Abstract
:1. Introduction
2. Results
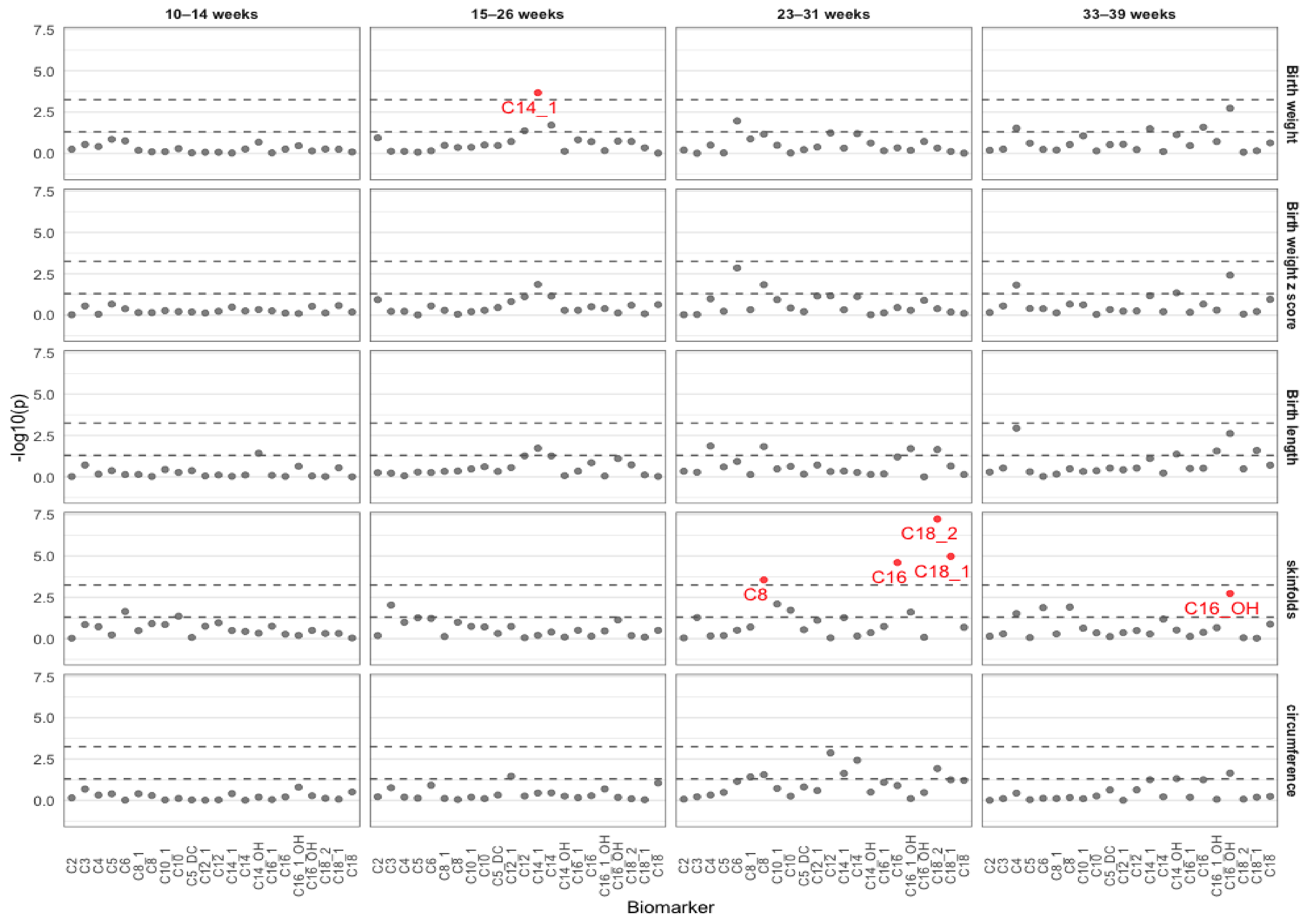
2.1. Associations between Acylcarnitines and Neonatal Biometry in Each Visit
2.2. Longitudinal Associations between Individual Acylcarnitine Trajectories and Neonatal Biometry
2.3. Associations of Joint Acylcarnitine Trajectories and Neonatal Biometry
2.4. Lack of Significant Effect Modifications by Fasting Status, GDM, and Infant Sex
3. Discussion
4. Materials and Methods
4.1. Study Design and Population
4.2. Assessment of Acylcarnitine Profiling
4.3. Neonatal Anthropometry
4.4. Assessment of Covariates
4.5. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hadden, D.R.; McLaughlin, C. Normal and abnormal maternal metabolism during pregnancy. Semin. Fetal Neonatal Med. 2009, 14, 66–71. [Google Scholar] [CrossRef]
- Lassance, L.; Haghiac, M.; Minium, J.; Catalano, P.; Hauguel-de Mouzon, S. Obesity-induced down-regulation of the mitochondrial translocator protein (TSPO) impairs placental steroid production. J. Clin. Endocrinol. Metab. 2015, 100, E11–E18. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mele, J.; Muralimanoharan, S.; Maloyan, A.; Myatt, L. Impaired mitochondrial function in human placenta with increased maternal adiposity. Am. J. Physiol. Endocrinol. Metab. 2014, 307, E419–E425. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Calabuig-Navarro, V.; Haghiac, M.; Minium, J.; Glazebrook, P.; Ranasinghe, G.C.; Hoppel, C.; Hauguel de-Mouzon, S.; Catalano, P.; O’Tierney-Ginn, P. Effect of maternal obesity on placental lipid metabolism. Endocrinology 2017, 158, 2543–2555. [Google Scholar] [CrossRef]
- Adams, S.H.; Hoppel, C.L.; Lok, K.H.; Zhao, L.; Wong, S.W.; Minkler, P.E.; Hwang, D.H.; Newman, J.W.; Garvey, W.T. Plasma acylcarnitine profiles suggest incomplete long-chain fatty acid beta-oxidation and altered tricarboxylic acid cycle activity in type 2 diabetic African-American women. J. Nutr. 2009, 139, 1073–1081. [Google Scholar] [CrossRef] [PubMed]
- Reuter, S.E.; Evans, A.M. Carnitine and acylcarnitines: Pharmacokinetic, pharmacological and clinical aspects. Clin. Pharmacokinet. 2012, 51, 553–572. [Google Scholar] [CrossRef]
- Schooneman, M.G.; Vaz, F.M.; Houten, S.M.; Soeters, M.R. Acylcarnitines: Reflecting or inflicting insulin resistance? Diabetes 2013, 62, 1–8. [Google Scholar] [CrossRef] [Green Version]
- Muoio, D.M. Metabolic inflexibility: When mitochondrial indecision leads to metabolic gridlock. Cell 2014, 159, 1253–1262. [Google Scholar] [CrossRef] [Green Version]
- Rinaldo, P.; Cowan, T.M.; Matern, D. Acylcarnitine profile analysis. Genet. Med. 2008, 10, 151–156. [Google Scholar] [CrossRef] [Green Version]
- Smith, E.H.; Matern, D. Acylcarnitine analysis by tandem mass spectrometry. Curr. Protoc. Hum. Genet. 2010, 64, 17–18. [Google Scholar] [CrossRef]
- Koves, T.R.; Ussher, J.R.; Noland, R.C.; Slentz, D.; Mosedale, M.; Ilkayeva, O.; Bain, J.; Stevens, R.; Dyck, J.R.; Newgard, C.B.; et al. Mitochondrial overload and incomplete fatty acid oxidation contribute to skeletal muscle insulin resistance. Cell Metab. 2008, 7, 45–56. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aichler, M.; Borgmann, D.; Krumsiek, J.; Buck, A.; MacDonald, P.E.; Fox, J.E.M.; Lyon, J.; Light, P.E.; Keipert, S.; Jastroch, M.; et al. N-acyl taurines and acylcarnitines cause an imbalance in insulin synthesis and secretion provoking beta cell dysfunction in type 2 diabetes. Cell Metab. 2017, 25, 1334–1347.e4. [Google Scholar] [CrossRef] [Green Version]
- Mai, M.; Tonjes, A.; Kovacs, P.; Stumvoll, M.; Fiedler, G.M.; Leichtle, A.B. Serum levels of acylcarnitines are altered in prediabetic conditions. PLoS ONE 2013, 8, e82459. [Google Scholar] [CrossRef] [Green Version]
- Ryckman, K.K.; Donovan, B.M.; Fleener, D.K.; Bedell, B.; Borowski, K.S. Pregnancy-related changes of amino acid and acylcarnitine concentrations: The impact of obesity. AJP Rep. 2016, 6, e329–e336. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sandler, V.; Reisetter, A.C.; Bain, J.R.; Muehlbauer, M.J.; Nodzenski, M.; Stevens, R.D.; Ilkayeva, O.; Lowe, L.P.; Metzger, B.E.; Newgard, C.B.; et al. Associations of maternal BMI and insulin resistance with the maternal metabolome and newborn outcomes. Diabetologia 2017, 60, 518–530. [Google Scholar] [CrossRef]
- Jacob, S.; Nodzenski, M.; Reisetter, A.C.; Bain, J.R.; Muehlbauer, M.J.; Stevens, R.D.; Ilkayeva, O.R.; Lowe, L.P.; Metzger, B.E.; Newgard, C.B.; et al. Targeted metabolomics demonstrates distinct and overlapping maternal metabolites associated with BMI, glucose, and insulin sensitivity during pregnancy across four ancestry groups. Diabetes Care 2017, 40, 911–919. [Google Scholar] [CrossRef] [Green Version]
- Batchuluun, B.; Al Rijjal, D.; Prentice, K.J.; Eversley, J.A.; Burdett, E.; Mohan, H.; Bhattacharjee, A.; Gunderson, E.P.; Liu, Y.; Wheeler, M.B. Elevated Medium-chain acylcarnitines are associated with gestational diabetes mellitus and early progression to type 2 diabetes and induce pancreatic beta-cell dysfunction. Diabetes 2018, 67, 885–897. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kadakia, R.; Nodzenski, M.; Talbot, O.; Kuang, A.; Bain, J.R.; Muehlbauer, M.J.; Stevens, R.D.; Ilkayeva, O.R.; O’Neal, S.K.; Lowe, L.P.; et al. Maternal metabolites during pregnancy are associated with newborn outcomes and hyperinsulinaemia across ancestries. Diabetologia 2019, 62, 473–484. [Google Scholar] [CrossRef] [Green Version]
- Jansson, T.; Powell, T.L. Role of the placenta in fetal programming: Underlying mechanisms and potential interventional approaches. Clin. Sci. 2007, 113, 1–13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rutkowsky, J.M.; Knotts, T.A.; Ono-Moore, K.D.; McCoin, C.S.; Huang, S.; Schneider, D.; Singh, S.; Adams, S.H.; Hwang, D.H. Acylcarnitines activate proinflammatory signaling pathways. Am. J. Physiol. Endocrinol. Metab. 2014, 306, E1378–E1387. [Google Scholar] [CrossRef] [Green Version]
- Schoderbeck, M.; Auer, B.; Legenstein, E.; Genger, H.; Sevelda, P.; Salzer, H.; Marz, R.; Lohninger, A. Pregnancy-related changes of carnitine and acylcarnitine concentrations of plasma and erythrocytes. J. Perinat. Med. 1995, 23, 477–485. [Google Scholar] [CrossRef] [PubMed]
- Marchlewicz, E.H.; Dolinoy, D.C.; Tang, L.; Milewski, S.; Jones, T.R.; Goodrich, J.M.; Soni, T.; Domino, S.E.; Song, P.X.; Burant, C.F.; et al. Lipid metabolism is associated with developmental epigenetic programming. Sci. Rep. 2016, 6, 34857. [Google Scholar] [CrossRef]
- Talian, G.C.; Komlosi, K.; Decsi, T.; Koletzko, B.; Melegh, B. Determination of carnitine ester patterns during the second half of pregnancy, at delivery, and in neonatal cord blood by tandem mass spectrometry: Complex and dynamic involvement of carnitine in the intermediary metabolism. Pediatr. Res. 2007, 62, 88–92. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Robinson, O.; Keski-Rahkonen, P.; Chatzi, L.; Kogevinas, M.; Nawrot, T.; Pizzi, C.; Plusquin, M.; Richiardi, L.; Robinot, N.; Sunyer, J.; et al. Cord blood metabolic signatures of birth weight: A population-based study. J. Proteome Res. 2018, 17, 1235–1247. [Google Scholar] [CrossRef] [Green Version]
- Lowe, W.L., Jr.; Bain, J.R.; Nodzenski, M.; Reisetter, A.C.; Muehlbauer, M.J.; Stevens, R.D.; Ilkayeva, O.R.; Lowe, L.P.; Metzger, B.E.; Newgard, C.B.; et al. Maternal BMI and glycemia impact the fetal metabolome. Diabetes Care 2017, 40, 902–910. [Google Scholar] [CrossRef] [Green Version]
- Hellmuth, C.; Lindsay, K.L.; Uhl, O.; Buss, C.; Wadhwa, P.D.; Koletzko, B.; Entringer, S. Maternal metabolomic profile and fetal programming of offspring adiposity: Identification of potentially protective lipid metabolites. Mol. Nutr. Food Res. 2019, 63, e1700889. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lu, Y.P.; Reichetzeder, C.; Prehn, C.; Yin, L.H.; Yun, C.; Zeng, S.; Chu, C.; Adamski, J.; Hocher, B. Cord blood lysophosphatidylcholine 16: 1 is positively associated with birth weight. Cell Physiol. Biochem. 2018, 45, 614–624. [Google Scholar] [CrossRef]
- Mitro, S.D.; Wu, J.; Rahman, M.L.; Cao, Y.; Zhu, Y.; Chen, Z.; Chen, L.; Li, M.; Hinkle, S.N.; Bremer, A.A.; et al. Longitudinal plasma metabolomics profile in pregnancy-A study in an ethnically diverse U.S. pregnancy cohort. Nutrients 2021, 13, 3080. [Google Scholar] [CrossRef]
- Buck Louis, G.M.; Grewal, J.; Albert, P.S.; Sciscione, A.; Wing, D.A.; Grobman, W.A.; Newman, R.B.; Wapner, R.; D’Alton, M.E.; Skupski, D.; et al. Racial/ethnic standards for fetal growth: The NICHD Fetal Growth Studies. Am. J. Obstet. Gynecol. 2015, 213, e441–e449. [Google Scholar] [CrossRef]
- Grewal, J.; Grantz, K.L.; Zhang, C.; Sciscione, A.; Wing, D.A.; Grobman, W.A.; Newman, R.B.; Wapner, R.; D’Alton, M.E.; Skupski, D.; et al. Cohort profile: NICHD Fetal Growth Studies-Singletons and Twins. Int. J. Epidemiol. 2018, 47, 25–25l. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xia, J.; Song, Y.; Rawal, S.; Wu, J.; Hinkle, S.N.; Tsai, M.Y.; Zhang, C. Vitamin D status during pregnancy and the risk of gestational diabetes mellitus: A longitudinal study in a multiracial cohort. Diabetes Obes. Metab. 2019, 21, 1895–1905. [Google Scholar] [CrossRef]
- Zhu, Y.; Mendola, P.; Albert, P.S.; Bao, W.; Hinkle, S.N.; Tsai, M.Y.; Zhang, C. Insulin-like growth factor axis and gestational diabetes mellitus: A longitudinal study in a multiracial cohort. Diabetes 2016, 65, 3495–3504. [Google Scholar] [CrossRef] [Green Version]
- Hinkle, S.N.; Rawal, S.; Zhu, Y.; Grewal, J.; Albert, P.S.; Zhang, C. Validation of self-reported diagnosis of gestational diabetes at 6-weeks postpartum. Epidemiology 2017, 28, 747–752. [Google Scholar] [CrossRef] [PubMed]
- Hinkle, S.N.; Rawal, S.; Liu, D.; Chen, J.; Tsai, M.Y.; Zhang, C. Maternal adipokines longitudinally measured across pregnancy and their associations with neonatal size, length, and adiposity. Int. J. Obes. 2019, 43, 1422–1434. [Google Scholar] [CrossRef] [PubMed]
- Oken, E.; Kleinman, K.P.; Rich-Edwards, J.; Gillman, M.W. A nearly continuous measure of birth weight for gestational age using a United States national reference. BMC Pediatr. 2003, 3, 6. [Google Scholar] [CrossRef]
- De Onis, M.; Onyango, A.W.; Van den Broeck, J.; Chumlea, W.C.; Martorell, R. Measurement and standardization protocols for anthropometry used in the construction of a new international growth reference. Food Nutr. Bull. 2004, 25, S27–S36. [Google Scholar] [CrossRef]
- Johnson, T.S.; Engstrom, J.L.; Gelhar, D.K. Intra- and interexaminer reliability of anthropometric measurements of term infants. J. Pediatr. Gastroenterol. Nutr. 1997, 24, 497–505. [Google Scholar] [CrossRef] [PubMed]
- Ulijaszek, S.J.; Kerr, D.A. Anthropometric measurement error and the assessment of nutritional status. Br. J. Nutr. 1999, 82, 165–177. [Google Scholar] [CrossRef] [Green Version]
- Schmelzle, H.R.; Fusch, C. Body fat in neonates and young infants: Validation of skinfold thickness versus dual-energy X-ray absorptiometry. Am. J. Clin. Nutr. 2002, 76, 1096–1100. [Google Scholar] [CrossRef] [Green Version]
- Weile, B.; Bach-Mortensen, N.; Peitersen, B. Caliper skinfold measurements in newborns: Analysis of a method. Biol. Neonatol. 1986, 50, 192–199. [Google Scholar] [CrossRef]
- Smarr, M.M.; Louis, G.M.B.; Albert, P.S.; Kim, S.; Fuchs, K.M.; Grewal, J.; D’Alton, M.E.; Grantz, K.L. Characterization of thermal and mechanical indices from serial ultrasound exams and associations with neonatal anthropometry: The NICHD Fetal Growth Studies. Am. J. Perinatol. 2018, 35, 632–642. [Google Scholar] [CrossRef]
- Francis, E.C.; Hinkle, S.N.; Song, Y.; Rawal, S.; Donnelly, S.R.; Zhu, Y.; Chen, L.; Zhang, C. Longitudinal maternal vitamin D status during pregnancy is associated with neonatal anthropometric measures. Nutrients 2018, 10, 1631. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Benjamini, Y.; Drai, D.; Elmer, G.; Kafkafi, N.; Golani, I. Controlling the false discovery rate in behavior genetics research. Behav. Brain Res. 2001, 125, 279–284. [Google Scholar] [CrossRef] [Green Version]
- Nagin, D.S.; Tremblay, R.E. Analyzing developmental trajectories of distinct but related behaviors: A group-based method. Psychol. Methods 2001, 6, 18–34. [Google Scholar] [CrossRef] [PubMed]
- Calinski, T.; Karabasz, J. A dendrite method for cluster analysis. Commun. Stat. Theory Methods 1974, 3, 1–27. [Google Scholar] [CrossRef]
| Characteristics | Participants (n = 321) a,b |
|---|---|
| Age, years | 28.0 (24.0, 32.0) |
| Race-ethnicity | |
| Non-Hispanic White | 75 (30.9) |
| Non-Hispanic Black | 45 (23.3) |
| Hispanic | 123 (27.2) |
| Asian and Pacific Islander | 78 (18.5) |
| Education | |
| High school or less | 81 (25.1) |
| Some college/associate degree | 117 (35.2) |
| Four-year college degree or higher | 123 (39.8) |
| Nulliparous | 143 (51.1) |
| Smoked | 5 (0.7) |
| Pre-pregnancy BMI, kg/m2 (self-reported) | 24.6 (22.0, 27.5) |
| 19.0–24.9 | 156 (51.73) |
| 25.0–29.9 | 99 (33.05) |
| 30.0–45.0 | 66 (13.22) |
| Gestational diabetes | 107 (3.9) |
| Infant sex | |
| Male | 166 (52.04) |
| Female | 153 (47.96) |
| Maternal Acylcarnitines | Neonatal Outcome, Adjusted β (95% CI) | ||||
|---|---|---|---|---|---|
| Birthweight, g | Birthweight, z Score | Length, cm | Sum of Skinfolds, mm | Sum of Circumference, cm | |
| C2 | |||||
| Group 3 | −59.3 (−521, 403) | 0.11 (−0.41, 0.64) | 0.57 (−1.61, 2.74) | 2.01 (−2.58, 6.60) | −0.46 (−6.11, 5.19) |
| Group 2 | 85.6 (−157, 329) | 0.11 (−0.25, 0.47) | 0.32 (−0.77, 1.41) | 0.84 (−1.88, 3.55) | 0.14 (−4.27, 4.55) |
| Group 1 | 0.0 (ref) | 0.0 (ref) | 0.0 (ref) | 0.0 (ref) | 0.0 (ref) |
| C3 | |||||
| Group 3 | 188 (−113, 490) | 0.17 (−0.29, 0.62) | 1.13 (−0.44, 2.70) | −0.92 (−4.03, 2.20) | 2.92 (−2.01, 7.85) |
| Group 2 | 115 (−55.5, 286) | 0.26 (−0.08, 0.60) | 0.30 (−0.90, 1.50) | −1.12 (−3.74, 1.51) | 2.24 (−1.65, 6.14) |
| Group 1 | 0.0 (ref) | 0.0 (ref) | 0.0 (ref) | 0.0 (ref) | 0.0 (ref) |
| C4 | |||||
| Group 3 | 44.2 (−187, 275) | −0.13 (−0.51, 0.25) | −0.42 (−2.08, 1.24) | −0.47 (−1.89, 0.94) | 1.77 (−2.60, 6.14) |
| Group 2 | 80.9 (−107, 269) | −0.03 (−0.29, 0.22) | 0.29 (−0.68, 1.26) | 0.46 (−1.24, 2.15) | 0.10 (−2.77, 2.97) |
| Group 1 | 0.0 (ref) | 0.0 (ref) | 0.0 (ref) | 0.0 (ref) | 0.0 (ref) |
| C5 | |||||
| Group 2 | −86.3 (−324, 151) | −0.25 (−0.57, 0.07) | −0.10 (−1.56, 1.37) | 0.17 (−1.89, 2.24) | −1.34 (−5.84, 3.16) |
| Group 1 | 0.0 (ref) | 0.0 (ref) | 0.0 (ref) | 0.0 (ref) | 0.0 (ref) |
| C8:1 | |||||
| Group 2 | −159 (−414, 95.3) | −0.17 (−0.47, 0.14) | −0.12 (−1.39, 1.15) | −1.37 (−3.64, 0.90) | −2.01 (−5.58, 1.55) |
| Group 1 | 0.0 (ref) | 0.0 (ref) | 0.0 (ref) | 0.0 (ref) | 0.0 (ref) |
| C10:1 | |||||
| Group 2 | −127 (−508, 253) | −0.05 (−0.39, 0.30) | −0.26 (−1.95, 1.43) | 3.23 (0.19, 6.27) | −0.95 (−4.54, 2.65) |
| Group 1 | 0.0 (ref) | 0.0 (ref) | 0.0 (ref) | 0.0 (ref) | 0.0 (ref) |
| C10 | |||||
| Group 2 | 31.4 (−165, 227) | −0.08 (−0.45, 0.29) | −0.28 (−1.33, 0.77) | 4.91 (0.85, 8.98) | −2.64 (−8.39, 3.11) |
| Group 1 | 0.0 (ref) | 0.0 (ref) | 0.0 (ref) | 0.0 (ref) | 0.0 (ref) |
| C5:DC | |||||
| Group 2 | −500 (−1104, 104) | −0.34 (−0.82, 0.14) | −1.31 (−2.99, 0.36) | −1.50 (−3.84, 0.84) | −1.61 (−6.94, 3.72) |
| Group 1 | 0.0 (ref) | 0.0 (ref) | 0.0 (ref) | 0.0 (ref) | 0.0 (ref) |
| C12 | |||||
| Group 2 | −475 (−942, −6.79) | −0.39 (−0.71, −0.06) | −1.38 (−2.49, −0.27) | −2.00 (−5.06, 1.07) | −1.90 (−6.69, 2.88) |
| Group 1 | 0.0 (ref) | 0.0 (ref) | 0.0 (ref) | 0.0 (ref) | 0.0 (ref) |
| Groups | Acylcarnitines | Gestational Weeks | |||
|---|---|---|---|---|---|
| 10–14 | 15–26 | 23–31 | 33–39 | ||
| Short-chain | C2 (Acetylcarnitine) | 3.15 (2.59, 3.99) | 2.94 (2.28, 3.73) | 2.56 (1.95, 3.12) | 2.60 (2.01, 3.05) |
| C3 (Propionylcarnitine) | 0.21 (0.15, 0.25) | 0.16 (0.13, 0.19) | 0.17 (0.13, 0.21) | 0.16 (0.12, 0.20) | |
| C4 (Butyrylcarnitine) | 0.15 (0.12, 0.20) | 0.12 (0.09, 0.15) | 0.13 (0.09, 0.17) | 0.13 (0.09,0.15) | |
| C5 (Valerylcarnitine) | 0.06 (0.04, 0.08) | 0.05 (0.04, 0.06) | 0.05 (0.04, 0.07) | 0.06 (0.04, 0.07) | |
| C6 (Hexanoylcarnitine) | 0.02 (0.01, 0.03) | 0.02 (0.01, 0.03) | 0.01 (0.01, 0.02) | 0.01 (0.01, 0.02) | |
| Medium-chain | C8 (Octanoylcarnitine) | 0.08 (0.05, 0.11) | 0.10 (0.08, 0.12) | 0.07 (0.05, 0.09) | 0.08 (0.05, 0.10) |
| C8:1 (Octenoylcarnitine) | 0.07 (0.06, 0.11) | 0.08 (0.05, 0.11) | 0.07 (0.04, 0.10) | 0.07 (0.04, 0.09) | |
| C10 (Decanoylcarnitine) | 0.06 (0.03, 0.10) | 0.08 (0.05, 0.11) | 0.04 (0.02, 0.06) | 0.05 (0.03, 0.07) | |
| C10:1 (Decenoylcarnitine) | 0.05 (0.03, 0.08) | 0.07 (0.05, 0.08) | 0.04 (0.03, 0.06) | 0.04 (0.03, 0.07) | |
| C12 (Dodecanoylcarnitine) | 0.03 (0.02, 0.04) | 0.03 (0.02, 0.04) | 0.02 (0.02, 0.03) | 0.02 (0.02, 0.03) | |
| C12:1 (Dodecenoylcarnitine) | 0.02 (0.01, 0.03) | 0.02 (0.01, 0.03) | 0.01 (0.01, 0.02) | 0.02 (0.01, 0.02) | |
| C14 (Tetradecanoylcarnitine) | 0.01 (0.01, 0.02) | 0.01 (0.01, 0.02) | 0.01 (0.01, 0.02) | 0.01 (0.01, 0.02) | |
| C14:1 (Tetradecenoylcarnitine) | 0.02 (0.01, 0.03) | 0.02 (0.01, 0.03) | 0.01 (0.01, 0.02) | 0.01 (0.01, 0.02) | |
| Long-chain | C16 (Hexadecanoylcarnitine) | 0.05 (0.04, 0.06) | 0.05 (0.04, 0.06) | 0.04 (0.04, 0.05) | 0.04 (0.03, 0.06) |
| C16:1 (Hexadecenoylcarnitine) | 0.01 (0.01, 0.01) | 0.01 (0.01, 0.01) | 0.01 (0.007, 0.01) | 0.01 (0.01, 0.01) | |
| C18 (Octadecanoylcarnitine); | 0.02 (0.02, 0.03) | 0.02 (0.02, 0.03) | 0.02 (0.02, 0.03) | 0.02 (0.02, 0.03) | |
| C18:1 (Octadecenoylcarnitine) | 0.05 (0.04, 0.08) | 0.05 (0.04, 0.06) | 0.04 (0.03, 0.06) | 0.04 (0.03, 0.06) | |
| C18:2 (Octadecadienylcarnitine) | 0.03 (0.02, 0.03) | 0.02 (0.02, 0.03) | 0.02 (0.01, 0.02) | 0.02 (0.01, 0.02) | |
| Carnitine esters derived from dicarboxylic acids | C5-DC (Glutarylcarnitine) | 0.02 (0.01, 0.02) | 0.02 (0.01, 0.02) | 0.01 (0.01, 0.02) | 0.01 (0.01, 0.02) |
| Carnitine esters derived from hydroxylated acids | C14-OH (3-OH-Tetradecanolycarnitine) | 0.000 (0.000, 0.02) | 0.000 (0.000, 0.01) | 0.000 (0.000, 0.02) | 0.000 (0.000, 0.01) |
| C16:1-OH (3-OH-Hexadecenoylcarnitine) | 0.000 (0.000, 0.04) | 0.000 (0.000, 0.01) | 0.000 (0.000, 0.03) | 0.000 (0.000, 0.01) | |
| C16-OH (3-OH-Hexadecanoylcarnitine) | 0.000 (0.000, 0.01) | 0.000 (0.000, 0.02) | 0.000 (0.000, 0.03) | 0.000 (0.000, 0.02) | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Song, Y.; Lyu, C.; Li, M.; Rahman, M.L.; Chen, Z.; Zhu, Y.; Hinkle, S.N.; Chen, L.; Mitro, S.D.; Li, L.-J.; et al. Plasma Acylcarnitines during Pregnancy and Neonatal Anthropometry: A Longitudinal Study in a Multiracial Cohort. Metabolites 2021, 11, 885. https://doi.org/10.3390/metabo11120885
Song Y, Lyu C, Li M, Rahman ML, Chen Z, Zhu Y, Hinkle SN, Chen L, Mitro SD, Li L-J, et al. Plasma Acylcarnitines during Pregnancy and Neonatal Anthropometry: A Longitudinal Study in a Multiracial Cohort. Metabolites. 2021; 11(12):885. https://doi.org/10.3390/metabo11120885
Chicago/Turabian StyleSong, Yiqing, Chen Lyu, Ming Li, Mohammad L. Rahman, Zhen Chen, Yeyi Zhu, Stefanie N. Hinkle, Liwei Chen, Susanna D. Mitro, Ling-Jun Li, and et al. 2021. "Plasma Acylcarnitines during Pregnancy and Neonatal Anthropometry: A Longitudinal Study in a Multiracial Cohort" Metabolites 11, no. 12: 885. https://doi.org/10.3390/metabo11120885
APA StyleSong, Y., Lyu, C., Li, M., Rahman, M. L., Chen, Z., Zhu, Y., Hinkle, S. N., Chen, L., Mitro, S. D., Li, L.-J., Weir, N. L., Tsai, M. Y., & Zhang, C. (2021). Plasma Acylcarnitines during Pregnancy and Neonatal Anthropometry: A Longitudinal Study in a Multiracial Cohort. Metabolites, 11(12), 885. https://doi.org/10.3390/metabo11120885










