Associations of BMI and Body Fat with Urine Metabolome in Adolescents Are Sex-Specific: A Cross-Sectional Study
Abstract
1. Introduction
2. Results
2.1. Basic Characteristics
2.2. Linear Regression Models
2.2.1. Summarizing Metabolites into Groups Using Independent Component Analysis
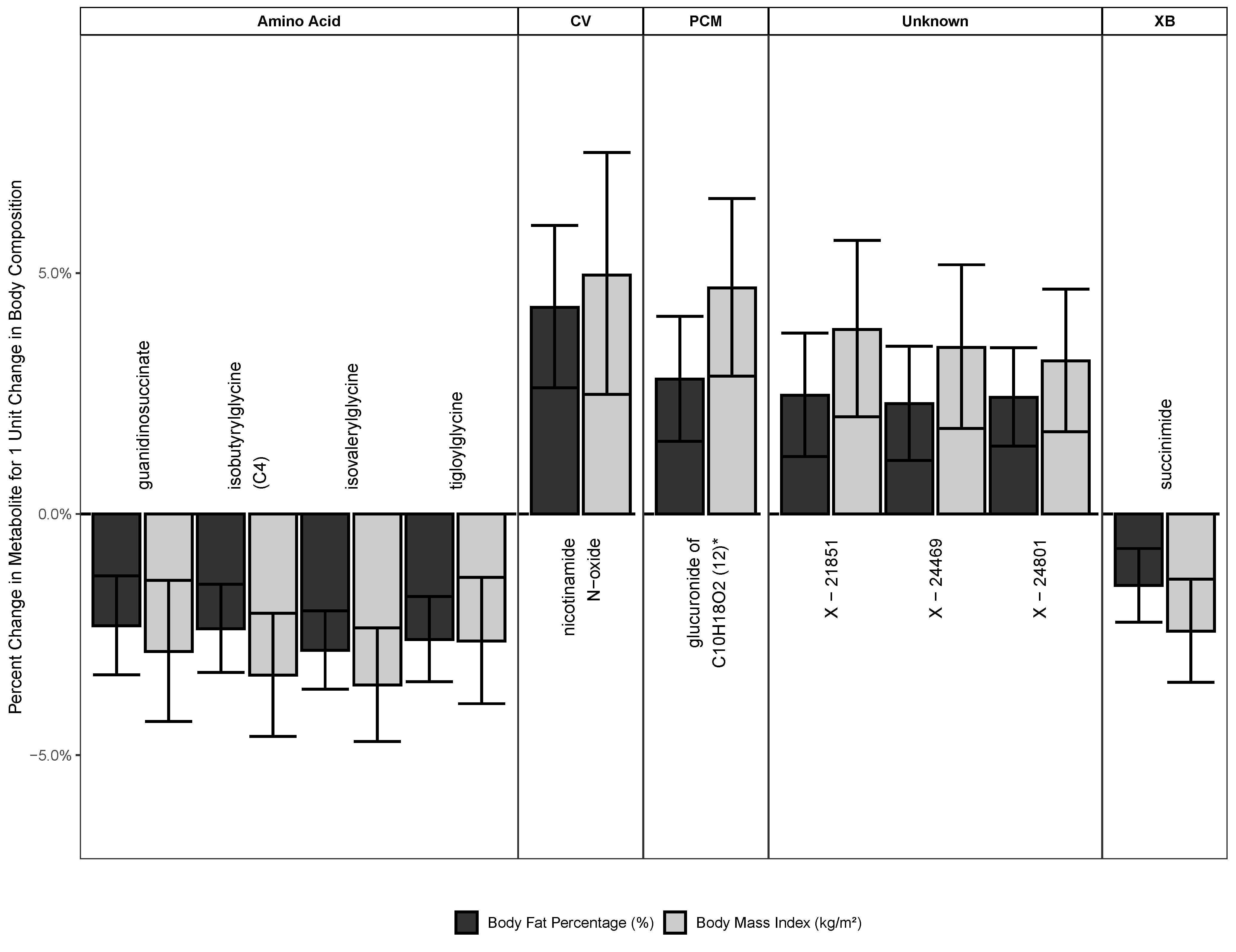
2.2.2. Metabolites Associated with Both BMI and BF
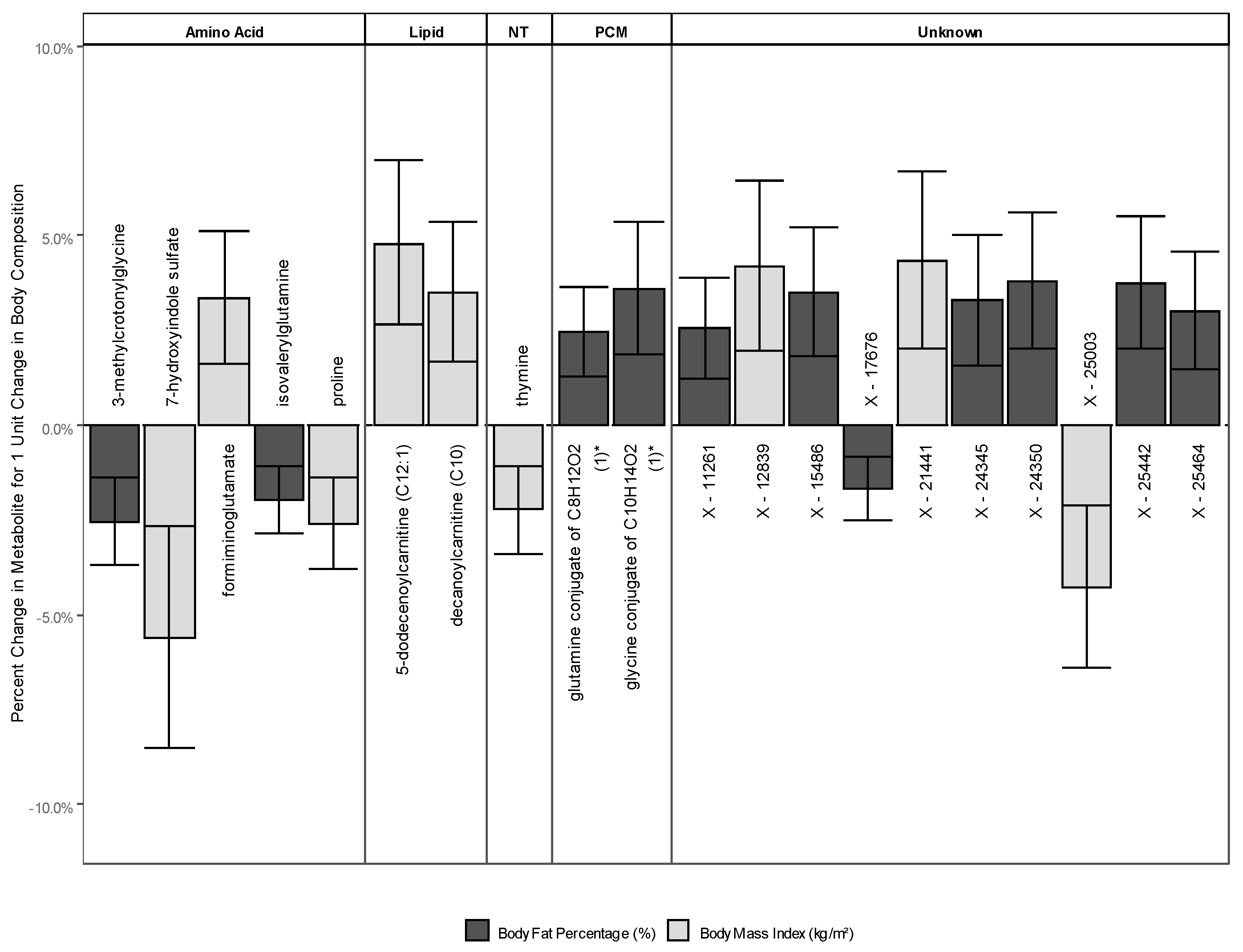
2.2.3. Metabolites Associated with Either BMI or BF
2.3. Metabolites Associated with BMI
2.4. Metabolites Associated with BF
3. Discussion
4. Materials and Methods
4.1. Study Design
4.2. Study Participants
4.3. Variable Assessment
4.3.1. Outcome: Untargeted Metabolomic Profiling of the Urine Metabolome
4.3.2. Exposure: Body Composition Measures
4.3.3. Covariates
4.4. Statistical Analysis
4.4.1. Data Pre-Treatment
4.4.2. Imputation of Missing Values
4.4.3. Summarizing Metabolites into Groups Using the Independent Component Analysis
4.4.4. Linear Regression Model
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A: Additional Information on Independent Components
| IC | Constructed by |
|---|---|
| Sex: female | |
| IC1 | Amino acid (10), unknown (5), lipid (2), nucleotide (2), and xenobiotics (1) |
| IC2 | Amino acid (6), xenobiotics (5), unknown (5), partially characterized molecules (3), and peptide (1) |
| IC3 | Unknown (10), xenobiotics (4), amino acid (3), peptide (2), and lipid (1) |
| IC4 | Unknown (9), xenobiotics (5), amino acid (3), and lipid (3) |
| IC5 | Amino acid (6), lipid (5), unknown (5), partially characterized molecules (2), nucleotide (1), and xenobiotics (1) |
| IC6 | Unknown (12), amino acid (3), xenobiotics (2), lipid (1), partially characterized molecules (1), and peptide (1) |
| IC7 | Amino acid (5), xenobiotics (5), unknown (5), lipid (2), nucleotide (1), partially characterized molecules (1), and peptide (1) |
| Sex: male | |
| IC1 | Unknown (9), xenobiotics (4), amino acid (3), nucleotide (2), energy (1), and lipid (1) |
| IC2 | Amino acid (5), lipid (5), nucleotide (3), xenobiotics (2), unknown (2), carbohydrate (1), partially characterized molecules (1), and peptide (1) |
| IC3 | Unknown (8), amino acid (5), xenobiotics (3), lipid (2), carbohydrate (1), and energy (1) |
| IC4 | Xenobiotics (13), unknown (6), and lipid (1) |
| IC5 | Xenobiotics (7), unknown (6), partially characterized molecules (4), amino acid (1), lipid (1), and nucleotide (1) |
| IC6 | Unknown (10), lipid (4), partially characterized molecules (4), amino acid (1), and xenobiotics (1) |
| IC7 | Xenobiotics (8), unknown (6), carbohydrate (4), lipid (1), and partially characterized molecules (1) |
| IC | β Body Mass Index | 95% CI | p (FDR) | β Body Fat Percent | 95% CI | p (FDR) |
|---|---|---|---|---|---|---|
| Sex: Female | ||||||
| IC1 | 1.025 | 0.890 to 1.180 | 0.991 | 0.996 | 0.914 to 1.086 | 0.991 |
| IC2 | 0.974 | 0.832 to 1.141 | 0.991 | 1.030 | 0.943 to 1.125 | 0.991 |
| IC3 | 0.979 | 0.838 to 1.143 | 0.991 | 1.024 | 0.946 to 1.109 | 0.991 |
| IC4 | 0.945 | 0.829 to 1.076 | 0.991 | 1.014 | 0.942 to 1.093 | 0.991 |
| IC5 | 0.885 | 0.733 to 1.070 | 0.991 | 1.054 | 0.942 to 1.179 | 0.991 |
| IC6 | 0.930 | 0.814 to 1.064 | 0.991 | 1.057 | 0.979 to 1.141 | 0.991 |
| IC7 | 0.928 | 0.799 to 1.078 | 0.991 | 1.034 | 0.949 to 1.126 | 0.991 |
| Sex: Male | ||||||
| IC1 | 1.012 | 0.903 to 1.135 | 1.000 | 0.986 | 0.893 to 1.088 | 1.000 |
| IC2 | 0.969 | 0.880 to 1.067 | 1.000 | 1.001 | 0.923 to 1.086 | 1.000 |
| IC3 | 1.045 | 0.940 to 1.162 | 1.000 | 0.974 | 0.891 to 1.064 | 1.000 |
| IC4 | 0.980 | 0.877 to 1.095 | 1.000 | 1.053 | 0.958 to 1.157 | 1.000 |
| IC5 | 0.993 | 0.896 to 1.100 | 1.000 | 1.039 | 0.959 to 1.127 | 1.000 |
| IC6 | 0.956 | 0.872 to 1.048 | 1.000 | 1.015 | 0.943 to 1.093 | 1.000 |
| IC7 | 0.991 | 0.889 to 1.104 | 1.000 | 0.952 | 0.870 to 1.041 | 1.000 |
Appendix B: Regression Coefficient Tables
| Biochemical | Sub Pathway | β Body Mass Index | 95% CI | p (FDR) | β Body Fat Percent | 95% CI | p (FDR) |
|---|---|---|---|---|---|---|---|
| Super-pathway: Amino Acid | |||||||
| guanidinosuccinate | Guanidino and acetamido metabolism | 0.971 | 0.957 to 0.986 | 0.046 | 0.977 | 0.967 to 0.987 | 0.014 |
| isobutyrylglycine (C4) | Leucine, isoleucine, and valine metabolism | 0.967 | 0.954 to 0.979 | 0.002 | 0.976 | 0.967 to 0.985 | 0.001 |
| isovalerylglycine | Leucine, isoleucine, and valine metabolism | 0.965 | 0.953 to 0.976 | 0.000 | 0.972 | 0.964 to 0.980 | 0.000 |
| tigloylglycine | Leucine, isoleucine, and valine metabolism | 0.974 | 0.961 to 0.987 | 0.036 | 0.974 | 0.965 to 0.983 | 0.000 |
| Super-pathway: CV | |||||||
| Nicotinamide N-oxide | Nicotinate and nicotinamide metabolism | 1.050 | 1.025 to 1.075 | 0.030 | 1.043 | 1.026 to 1.060 | 0.001 |
| Super-pathway: PCM | |||||||
| Glucuronide of C10H18O2 (12) * | Partially characterized molecules | 1.047 | 1.029 to 1.066 | 0.001 | 1.028 | 1.015 to 1.041 | 0.016 |
| Super-pathway: Unknown | |||||||
| X-21851 | 1.038 | 1.020 to 1.057 | 0.021 | 1.025 | 1.012 to 1.038 | 0.044 | |
| X-24469 | 1.035 | 1.018 to 1.052 | 0.025 | 1.023 | 1.011 to 1.035 | 0.044 | |
| X-24801 | 1.032 | 1.017 to 1.047 | 0.016 | 1.024 | 1.014 to 1.034 | 0.004 | |
| Super-pathway: XB | |||||||
| Succinimide | Chemical | 0.976 | 0.965 to 0.986 | 0.011 | 0.985 | 0.978 to 0.993 | 0.046 |
| Biochemical | Sub-Pathway | Body Mass Index | 95% CI | p (FDR) | Body Fat Percent | 95% CI | p (FDR) |
|---|---|---|---|---|---|---|---|
| Super-Pathway: Amino Acid | |||||||
| Formiminoglutamate | Histidine metabolism | 1.033 | 1.016 to 1.051 | 0.041 | 1.019 | 1.007 to 1.031 | 0.162 |
| 3-methylcrotonylglycine | Leucine, isoleucine, and valine metabolism | 0.971 | 0.954 to 0.988 | 0.091 | 0.975 | 0.963 to 0.986 | 0.021 |
| Isovalerylglutamine | Leucine, isoleucine, and valine metabolism | 0.978 | 0.966 to 0.991 | 0.120 | 0.980 | 0.972 to 0.989 | 0.016 |
| 7-hydroxyindole sulfate | Tryptophan metabolism | 0.944 | 0.915 to 0.973 | 0.050 | 0.961 | 0.940 to 0.982 | 0.060 |
| proline | Urea cycle; arginine and proline metabolism | 0.974 | 0.962 to 0.986 | 0.023 | 0.988 | 0.979 to 0.997 | 0.281 |
| Super-Pathway: Lipid | |||||||
| decanoylcarnitine (C10) | Fatty acid metabolism (acyl carnitine, medium chain) | 1.035 | 1.017 to 1.054 | 0.046 | 1.023 | 1.010 to 1.036 | 0.083 |
| 5-dodecenoylcarnitine (C12:1) | Fatty acid metabolism (acyl carnitine, monounsaturated) | 1.048 | 1.027 to 1.070 | 0.009 | 1.025 | 1.010 to 1.041 | 0.110 |
| Super-Pathway: Nucleotide | |||||||
| Thymine | Pyrimidine metabolism, thymine containing | 0.978 | 0.966 to 0.989 | 0.046 | 0.987 | 0.979 to 0.995 | 0.169 |
| Super-Pathway: PCM | |||||||
| Glutamine conjugate of C8H12O2 (1) * | Partially characterized molecules | 1.030 | 1.013 to 1.047 | 0.065 | 1.025 | 1.013 to 1.036 | 0.021 |
| Glycine conjugate of C10H14O2 (1) * | Partially characterized molecules | 1.044 | 1.019 to 1.069 | 0.071 | 1.036 | 1.019 to 1.054 | 0.023 |
| Super-Pathway: Unknown | |||||||
| X-11261 | 1.032 | 1.013 to 1.051 | 0.111 | 1.025 | 1.012 to 1.039 | 0.045 | |
| X-12839 | 1.042 | 1.020 to 1.065 | 0.048 | 1.024 | 1.009 to 1.040 | 0.175 | |
| X-15486 | 1.039 | 1.015 to 1.064 | 0.118 | 1.035 | 1.018 to 1.052 | 0.021 | |
| X-17676 | 0.981 | 0.969 to 0.993 | 0.142 | 0.983 | 0.975 to 0.991 | 0.034 | |
| X-21441 | 1.043 | 1.020 to 1.067 | 0.047 | 1.029 | 1.013 to 1.046 | 0.078 | |
| X-24345 | 1.040 | 1.015 to 1.065 | 0.123 | 1.033 | 1.016 to 1.050 | 0.044 | |
| X-24350 | 1.040 | 1.014 to 1.067 | 0.156 | 1.038 | 1.020 to 1.056 | 0.020 | |
| X-25003 | 0.957 | 0.936 to 0.979 | 0.044 | 0.976 | 0.960 to 0.992 | 0.192 | |
| X-25442 | 1.041 | 1.016 to 1.067 | 0.120 | 1.038 | 1.020 to 1.055 | 0.015 | |
| X-25464 | 1.039 | 1.017 to 1.062 | 0.076 | 1.030 | 1.015 to 1.046 | 0.037 | |
Appendix C: Additional Discussion for Metabolites Associated with Either BMI or BF
| Biochemical | Sub-Pathway | Sex | Body Composition | Discussion |
|---|---|---|---|---|
| Super-Pathway: Amino Acid | ||||
| 7-hydroxyindole sulfate | Tryptophan metabolism | male | BMI | Part of the serotonin-related pathway of tryptophan [18]. A relationship to mood and depression, which has been documented to be influenced by weight and the perception thereof in adolescents [40], is a possible explanation for this association. |
| Formiminoglutamate | Histidine metabolism | male | BMI | Measurements in urine after oral application of histidine are used to determine folate deficiency [18]. Higher levels of this metabolite in the urine of individuals with higher adiposity might point to an increased need for folate. In fact, overweightness was previously shown to be associated with decreased levels of folate [41]. |
| Proline | Urea cycle; arginine and proline metabolism | male | BMI | Proline was inversely associated with adiposity in our study. This is in agreement with findings in children, in which lower levels of the metabolite have been observed in overweight children [42], but is in contrast to findings in adults [43,44]. This suggests that the relationship of adiposity with proline varies with the developmental stage of life. |
| 3-methylcrotonylglycine | Leucine, isoleucine, and valine metabolism | male | BF | A catabolite of leucine. Elevated levels of this metabolite in urine are usually found in patients with a deficiency of 3-methylcrotonyl-CoA carboxylase, an inborn error of the metabolism [18]. Decreased levels in our sample could be explained by hyperactivation of 3-methylcrotonyl-CoA carboxylase or disruption of the leucine metabolism. |
| Isovalerylglutamine | Leucine, isoleucine and valine metabolism | male | BF | No information |
| Super Pathway: Lipid | ||||
| 5-dodecenoylcarnitine (C12:1) | Fatty acid metabolism (acyl carnitine, monounsaturated) | male | BMI | Medium-chain acylcarnitines (MCACs), see decanoylcarnitine (C10) |
| Decanoylcarnitine (C10) | Fatty acid metabolism (acyl carnitine, medium chain) | male | BMI | Decanoylcarnitine (C10) is a medium-chain fatty acid acylcarnitine that was significantly associated with higher measures of body composition. In fact, urine decanoylcarnitine has been shown to differentiate young men with normal weight from those with obesity [45], and differentiates individuals with metabolically healthy obesity from those with metabolically abnormal obesity [46]. Additionally, it is among a group of acylcarnitines that is positively related to fat oxidation [47]. It was suggested previously that high levels of medium-chain acylcarnitines (MCACs) reflect distal β- oxidation for energy use. C6 and C10 in particular are used as markers for MCAC flux [48]. Higher levels of MCAC have also been related to a disrupted branched-chain amino acid (BCAA) metabolism [49,50]. Additionally, increased levels of MCAC were suggested as markers for insulin resistance in overweight and obese individuals [51]. Increased levels of C10 in our sample are in line with the findings of previous studies in adults and children, reporting either higher levels of closely related acylcarnitines or C10 exactly [19]. However, most of these were using different tissues (e.g., blood or muscle fiber) as their biospeciminen [19]. |
| Super Pathway: Nucleotide | ||||
| Thymine | Pyrimidine metabolism, thymine containing | male | BMI | Change within increasing adiposity is in line with cytosine per thymine change present in a single-nucleotide polymorphism (SNP) that is associated with BMI and BF [52]. This supports evidence that adiposity has a genetic component. Future studies should explore the relation between this SNP and adiposity |
| Super-Pathway: PCM | ||||
| Glutamine conjugate of C8H12O2 (1) * | Partially characterized molecules | male | BF | No information |
| Glycine conjugate of C10H14O2 (1) * | Partially characterized molecules | male | BF | No information |
| Super-Pathway: Unknown | ||||
| X-12839 | male | BMI | No information | |
| X-21441 | male | BMI | No information | |
| X-25003 | male | BMI | No information | |
| X-11261 | male | BF | No information | |
| X-15486 | male | BF | No information | |
| X-17676 | male | BF | No information | |
| X-24345 | male | BF | No information | |
| X-24350 | male | BF | No information | |
| X-25442 | male | BF | No information | |
| X-25464 | male | BF | No information | |
References
- Liu, M.; Li, L.; Chu, J.; Zhu, B.; Zhang, Q.; Yin, X.; Jiang, W.; Dai, G.; Ju, W.; Wang, Z.; et al. Serum N(1)-Methylnicotinamide Is Associated With Obesity and Diabetes in Chinese. J. Clin. Endocrinol. Metab. 2015, 100, 3112–3117. [Google Scholar] [CrossRef]
- Thévenot, E.A.; Roux, A.; Xu, Y.; Ezan, E.; Junot, C. Analysis of the Human Adult Urine metabolome Variations with Age, Body Mass Index, and Gender by Implementing a Comprehensive Workflow for Univariate and OPLS Statistical Analyses. J. Proteome Res. 2015, 14, 3322–3335. [Google Scholar] [CrossRef] [PubMed]
- Won, E.-Y.; Yoon, M.-K.; Kim, S.-W.; Jung, Y.; Bae, H.-W.; Lee, D.; Park, S.G.; Lee, C.-H.; Hwang, G.-S.; Chi, S.-W. Gender-Specific Metabolomic Profiling of Obesity in Leptin-Deficient ob/ob Mice by 1H NMR Spectroscopy. PLoS ONE 2013, 8, e75998. [Google Scholar] [CrossRef] [PubMed]
- Xie, G.; Ma, X.; Zhao, A.; Wang, C.; Zhang, Y.; Nieman, D.; Nicholson, J.K.; Jia, W.; Bao, Y.; Jia, W. The Metabolite Profiles of the Obese Population Are Gender-Dependent. J. Proteome Res. 2014, 13, 4062–4073. [Google Scholar] [CrossRef] [PubMed]
- Raven, P.W.; Taylor, N.F. Sex differences in the human metabolism of cortisol. Endocr. Res. 1996, 22, 751–755. [Google Scholar] [CrossRef]
- Saner, C.; Harcourt, B.E.; Pandey, A.; Ellul, S.; McCallum, Z.; Kao, K.-T.; Twindyakirana, C.; Pons, A.; Alexander, E.J.; Saffery, R.; et al. Sex and puberty-related differences in metabolomic profiles associated with adiposity measures in youth with obesity. Metabolomics 2019, 15, 75. [Google Scholar] [CrossRef]
- Oluwagbemigun, K.; Anesi, A.; Ulaszewska, M.M.; Clarke, G.; Alexy, U.; Schmid, M.; Roden, M.; Herder, C.; Mattivi, F.; Nöthlings, U. Longitudinal relationship of amino acids and indole metabolites with long-term body mass index and cardiometabolic risk markers in young individuals. Sci. Rep. 2020, 10, 1–13. [Google Scholar] [CrossRef]
- Kim, C.; Dabelea, D.; Kalyani, R.R.; Christophi, C.A.; Bray, G.A.; Pi-Sunyer, X.; Darwin, C.H.; Yalamanchi, S.; Barrett-Connor, E.; Golden, S.H.; et al. Changes in Visceral Adiposity, Subcutaneous Adiposity, and Sex Hormones in the Diabetes Prevention Program. J. Clin. Endocrinol. Metab. 2017, 102, 3381–3389. [Google Scholar] [CrossRef]
- Rauschert, S.; Uhl, O.; Koletzko, B.; Mori, T.A.; Beilin, L.J.; Oddy, W.H.; Hellmuth, C. Sex differences in the association of phospholipids with components of the metabolic syndrome in young adults. Biol. Sex Differ. 2017, 8, 10. [Google Scholar] [CrossRef]
- Brown, L.M.; Gent, L.; Davis, K.; Clegg, D.J. Metabolic impact of sex hormones on obesity. Brain Res. 2010, 1350, 77–85. [Google Scholar] [CrossRef]
- Mongraw-Chaffin, M.L.; Anderson, C.A.M.; Allison, M.A.; Ouyang, P.; Szklo, M.; Vaidya, D.; Woodward, M.; Golden, S.H. Association between sex hormones and adiposity: Qualitative differences in women and men in the multi-ethnic study of atherosclerosis. J. Clin. Endocrinol. Metab. 2015, 100, E596–E600. [Google Scholar] [CrossRef] [PubMed]
- Clegg, D.J.; Mauvais-Jarvis, F. An integrated view of sex differences in metabolic physiology and disease. Mol. Metab. 2018, 15, 1–2. [Google Scholar] [CrossRef] [PubMed]
- Darst, B.F.; Koscik, R.L.; Hogan, K.J.; Johnson, S.C.; Engelman, C.D. Longitudinal plasma metabolomics of aging and sex. Aging (Albany Ny) 2019, 11, 1262–1282. [Google Scholar] [CrossRef] [PubMed]
- Rist, M.J.; Roth, A.; Frommherz, L.; Weinert, C.H.; Krüger, R.; Merz, B.; Bunzel, D.; Mack, C.; Egert, B.; Bub, A.; et al. Metabolite patterns predicting sex and age in participants of the Karlsruhe Metabolomics and Nutrition (KarMeN) study. PLoS ONE 2017, 12, e0183228. [Google Scholar] [CrossRef]
- Kalyani, R.R.; Franco, M.; Dobs, A.S.; Ouyang, P.; Vaidya, D.; Bertoni, A.G.; Gapstur, S.M.; Golden, S.H. The association of endogenous sex hormones, adiposity, and insulin resistance with incident diabetes in postmenopausal women. J. Clin. Endocrinol. Metab. 2009, 94, 4127–4135. [Google Scholar] [CrossRef]
- Shen, M.; Shi, H. Sex Hormones and Their Receptors Regulate Liver Energy Homeostasis. Int. J. Endocrinol. 2015, 2015, 294278. [Google Scholar] [CrossRef]
- Wang, X.; Magkos, F.; Mittendorfer, B. Sex differences in lipid and lipoprotein metabolism: It’s not just about sex hormones. J. Clin. Endocrinol. Metab. 2011, 96, 885–893. [Google Scholar] [CrossRef]
- Wishart, D.S.; Feunang, Y.D.; Marcu, A.; Guo, A.C.; Liang, K.; Vázquez-Fresno, R.; Sajed, T.; Johnson, D.; Li, C.; Karu, N.; et al. HMDB 4.0: The human metabolome database for 2018. Nucleic Acids Res. 2018, 46, D608–D617. [Google Scholar] [CrossRef]
- Rangel-Huerta, O.D.; Pastor-Villaescusa, B.; Gil, A. Are we close to defining a metabolomic signature of human obesity? A systematic review of metabolomics studies. Metabolomics 2019, 15, 93. [Google Scholar] [CrossRef]
- Oglesbee, D.; He, M.; Majumder, N.; Vockley, J.; Ahmad, A.; Angle, B.; Burton, B.; Charrow, J.; Ensenauer, R.; Ficicioglu, C.; et al. Development of a newborn screening follow-up algorithm for the diagnosis of isobutyryl-CoA dehydrogenase deficiency. Genet. Med. 2007, 9, 108–116. [Google Scholar] [CrossRef]
- Zampieri, T.T.; Torres-Leal, F.L.; Campaña, A.B.; Lima, F.B.; Donato, J. l-Leucine Supplementation Worsens the Adiposity of Already Obese Rats by Promoting a Hypothalamic Pattern of Gene Expression that Favors Fat Accumulation. Nutrients 2014, 6, 1364–1373. [Google Scholar] [CrossRef] [PubMed]
- Hjerpsted, J.B.; Ritz, C.; Schou, S.S.; Tholstrup, T.; Dragsted, L.O. Effect of cheese and butter intake on metabolites in urine using an untargeted metabolomics approach. Metabolomics 2014, 10, 1176–1185. [Google Scholar] [CrossRef]
- Elliott, P.; Posma, J.M.; Chan, Q.; Garcia-Perez, I.; Wijeyesekera, A.; Bictash, M.; Ebbels, T.M.D.; Ueshima, H.; Zhao, L.; Van Horn, L.; et al. Urinary metabolic signatures of human adiposity. Sci. Transl. Med. 2015, 7, 285ra62. [Google Scholar] [CrossRef] [PubMed]
- Holeček, M. Branched-chain amino acids in health and disease: Metabolism, alterations in blood plasma, and as supplements. Nutr. Metab. 2018, 15, 33. [Google Scholar] [CrossRef]
- Menon, R.M.; González, M.A.; Adams, M.H.; Tolbert, D.S.; Leu, J.H.; Cefali, E.A. Effect of the rate of niacin administration on the plasma and urine pharmacokinetics of niacin and its metabolites. J. Clin. Pharmacol. 2007, 47, 681–688. [Google Scholar] [CrossRef]
- Jung, J.-Y.; Kim, I.Y.; Na Kim, Y.; Kim, J.-S.; Shin, J.H.; Jang, Z.-H.; Lee, H.-S.; Hwang, G.-S.; Seong, J.K. 1H NMR-based metabolite profiling of diet-induced obesity in a mouse mode. Bmb Rep. 2012, 45, 419–424. [Google Scholar] [CrossRef]
- Pickrell, W.O.; Lacey, A.S.; Thomas, R.H.; Smith, P.E.M.; Rees, M.I. Weight change associated with antiepileptic drugs. J. Neurol. Neurosurg. Psychiatry 2013, 84, 796–799. [Google Scholar] [CrossRef]
- Kroke, A.; Manz, F.; Kersting, M.; Remer, T.; Sichert-Hellert, W.; Alexy, U.; Lentze, M.J. The DONALD Study. History, current status and future perspectives. Eur. J. Nutr. 2004, 43, 45–54. [Google Scholar] [CrossRef]
- Oluwagbemigun, K.; Buyken, A.E.; Alexy, U.; Schmid, M.; Herder, C.; Nöthlings, U. Developmental trajectories of body mass index from childhood into late adolescence and subsequent late adolescence-young adulthood cardiometabolic risk markers. Cardiovasc. Diabetol. 2019, 18, 9. [Google Scholar] [CrossRef]
- Evans, A.M.; Bridgewater, B.R.; Liu, Q.; Mitchell, M.W.; Robinson, R.J.; Dai, H.; Stewart, S.J.; DeHaven, C.D.; Miller, L.A.D. High Resolution Mass Spectrometry Improves Data Quantity and Quality as Compared to Unit Mass Resolution Mass Spectrometry in High-Throughput Profiling Metabolomics. Metabolomics 2014, 4, 1–7. [Google Scholar] [CrossRef]
- Deurenberg, P.; Pieters, J.J.L.; Hautvast, J.G.A.J. The assessment of the body fat percentage by skinfold thickness measurements in childhood and young adolescence. Br. J. Nutr. 1990, 63, 293. [Google Scholar] [CrossRef] [PubMed]
- Sichert-Hellert, W.; Kersting, M.; Chahda, C.; Schäfer, R.; Kroke, A. German food composition database for dietary evaluations in children and adolescents. J. Food Compos. Anal. 2007, 20, 63–70. [Google Scholar] [CrossRef]
- Schofield, W.N. Predicting basal metabolic rate, new standards and review of previous work. Hum Nutr. Clin. Nutr. 1985, 39, 5–41. [Google Scholar] [PubMed]
- Booth, M.L.; Okely, A.D.; Chey, T.N.; Bauman, A. The reliability and validity of the Adolescent Physical Activity Recall Questionnaire. Med. Sci. Sports Exerc. 2002, 34, 1986–1995. [Google Scholar] [CrossRef]
- R Development Core Team. R: A Language and Environment for Statistical Computing; R Core Team: Vienna, Austria, 2019. [Google Scholar]
- van Buuren, S.; Groothuis-Oudshoorn, K. Mice: Multivariate Imputation by Chained Equations in R. J. Stat. Soft. 2011, 1–68. [Google Scholar] [CrossRef]
- van Buuren, S. Flexible Imputation of Missing Data, Second edition; Chapman and Hall/CRC: Boca Raton, FL, USA; London, UK; New York, NY, USA, 2018. [Google Scholar]
- Independent Component Analysis. 1.0-2; R Package; Helwig, N.E. 2018.
- Benjamini, Y.; Hochberg, Y. Controlling the False Discovery Rate: A Practical and Powerful Approach to Multiple Testing. J. R. Stat. Soc. Ser. B (Methodol.) 1995, 57, 289–300. [Google Scholar] [CrossRef]
- Martyn-Nemeth, P.; Penckofer, S.; Gulanick, M.; Velsor-Friedrich, B.; Bryant, F.B. The relationships among self-esteem, stress, coping, eating behavior, and depressive mood in adolescents. Res. Nurs. Health 2009, 32, 96–109. [Google Scholar] [CrossRef]
- Bird, J.K.; Ronnenberg, A.G.; Choi, S.-W.; Du, F.; Mason, J.B.; Liu, Z. Obesity is associated with increased red blood cell folate despite lower dietary intakes and serum concentrations. J. Nutr. 2015, 145, 79–86. [Google Scholar] [CrossRef]
- Guevara-Cruz, M.; Vargas-Morales, J.M.; Méndez-García, A.L.; López-Barradas, A.M.; Granados-Portillo, O.; Ordaz-Nava, G.; Rocha-Viggiano, A.K.; Gutierrez-Leyte, C.A.; Medina-Cerda, E.; Rosado, J.L.; et al. Amino acid profiles of young adults differ by sex, body mass index and insulin resistance. Nutr. Metab. Cardiovasc. Dis. 2018, 28, 393–401. [Google Scholar] [CrossRef]
- Bagheri, M.; Djazayery, A.; Farzadfar, F.; Qi, L.; Yekaninejad, M.S.; Aslibekyan, S.; Chamari, M.; Hassani, H.; Koletzko, B.; Uhl, O. Plasma metabolomic profiling of amino acids and polar lipids in Iranian obese adults. Lipids Health Dis. 2019, 18, 1–9. [Google Scholar] [CrossRef]
- Wahl, S.; Yu, Z.; Kleber, M.; Singmann, P.; Holzapfel, C.; He, Y.; Mittelstrass, K.; Polonikov, A.; Prehn, C.; Römisch-Margl, W.; et al. Childhood obesity is associated with changes in the serum metabolite profile. Obes. Facts 2012, 5, 660–670. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Feng, R.; Sun, D.; Li, Y.; Bi, X.; Sun, C. Metabolic profiling of urine in young obese men using ultra performance liquid chromatography and Q-TOF mass spectrometry (UPLC/Q-TOF MS). J. Chromatogr. B 2011, 879, 2871–2876. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.-H.; Tseng, Y.J.; Wang, S.-Y.; Tsai, Y.-S.; Chang, C.-S.; Kuo, T.-C.; Yao, W.-J.; Shieh, C.-C.; Wu, C.-H.; Kuo, P.-H. The metabolome profiling and pathway analysis in metabolic healthy and abnormal obesity. Int. J. Obes. 2015, 39, 1241–1248. [Google Scholar] [CrossRef]
- Butte, N.F.; Liu, Y.; Zakeri, I.F.; Mohney, R.P.; Mehta, N.; Voruganti, V.S.; Goring, H.; Cole, S.A.; Comuzzie, A.G. Global metabolomic profiling targeting childhood obesity in the Hispanic population. Am. J. Clin. Nutr. 2015, 102, 256–267. [Google Scholar] [CrossRef] [PubMed]
- Baker, P.R.; Boyle, K.E.; Koves, T.R.; Ilkayeva, O.R.; Muoio, D.M.; Houmard, J.A.; Friedman, J.E. Metabolomic analysis reveals altered skeletal muscle amino acid and fatty acid handling in obese humans. Obesity 2015, 23, 981–988. [Google Scholar] [CrossRef]
- Newgard, C.B.; An, J.; Bain, J.R.; Muehlbauer, M.J.; Stevens, R.D.; Lien, L.F.; Haqq, A.M.; Shah, S.H.; Arlotto, M.; Slentz, C.A.; et al. A Branched-Chain Amino Acid-Related Metabolic Signature that Differentiates Obese and Lean Humans and Contributes to Insulin Resistance. Cell Metab. 2009, 9, 311–326. [Google Scholar] [CrossRef]
- Schooneman, M.G.; Vaz, F.M.; Houten, S.M.; Soeters, M.R. Acylcarnitines: Reflecting or inflicting insulin resistance? Diabetes 2013, 62, 1–8. [Google Scholar] [CrossRef]
- Makrecka-Kuka, M.; Sevostjanovs, E.; Vilks, K.; Volska, K.; Antone, U.; Kuka, J.; Makarova, E.; Pugovics, O.; Dambrova, M.; Liepinsh, E. Plasma acylcarnitine concentrations reflect the acylcarnitine profile in cardiac tissues. Sci. Rep. 2017, 7, 17528. [Google Scholar] [CrossRef]
- Moleres, A.; Martinez, J.A.; Marti, A. Genetics of Obesity. Curr Obes Rep. 2013, 2, 23–31. [Google Scholar] [CrossRef]
| Variable | n | Total | Male | Female |
|---|---|---|---|---|
| n = 369 | n = 189 | n = 180 | ||
| Age (years) | 369 | 17.3 (1.0) | 17.2 (1.0) | 17.4 (1.0) |
| Body Fat Percent | 368 | 21.8 (8.0) | 16.6 (5.6) | 27.3 (6.4) |
| BMI (kg/m2) | 369 | 22.2 (3.7) | 22.5 (4.0) | 21.9 (3.2) |
| Overweight (BMI ≥ 25): Yes | 369 | 62 (16.8%) | 39 (20.6%) | 23 (12.8%) |
| Metabolic Equivalent of Task-Hours (met-h/week) | 207 | 41.3 (37.0) | 45.9 (43.3) | 36.6 (28.8) |
| Calories (kcal) | 364 | 2189.2 (616.7) | 2545.8 (565.4) | 1816.5 (415.4) |
| Protein (%kcal) | 364 | 13.9 (2.8) | 14.1 (2.7) | 13.7 (2.8) |
| Fat (%kcal) | 364 | 33.7 (6.6) | 33.9 (7.0) | 33.6 (6.1) |
| Carbohydrates (%kcal) | 364 | 50.5 (6.9) | 49.8 (7.4) | 51.2 (6.3) |
| Smoking Status | 118 | |||
| Never | 56 (15.2%) | 28 (14.8%) | 28 (15.6%) | |
| Former | 35 (9.5%) | 13 (6.9%) | 22 (12.2%) | |
| Current | 27 (7.3%) | 12 (6.3%) | 15 (8.3%) | |
| Alcohol Status | 155 | |||
| Never | 8 (2.2%) | 5 (2.6%) | 3 (1.7%) | |
| Former | 11 (3%) | 5 (2.6%) | 6 (3.3%) | |
| Current | 136 (36.9%) | 65 (34.4%) | 71 (39.4%) | |
| Maternal Occupation: Working (full or part-time) | 364 | 222 (60.2%) | 122 (64.6%) | 100 (55.6%) |
| Maternal Education: >12 Years of Education | 365 | 190 (51.5%) | 101 (53.4%) | 89 (49.4%) |
| Breastfeeding Duration (weeks) | 363 | 25.0 (18.3) | 24.3 (18.9) | 25.7 (17.7) |
| Maternal Gestational Weight Gain (kg) | 348 | 12.8 (4.1) | 12.7 (4.2) | 13.0 (4.1) |
| Maternal BMI (kg/m2) (kg/m2) | 358 | 23.7 (3.7) | 23.8 (3.5) | 23.7 (3.9) |
| Smoking Household: Yes | 265 | 86 (23.3%) | 43 (22.8%) | 43 (23.9%) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Brachem, C.; Langenau, J.; Weinhold, L.; Schmid, M.; Nöthlings, U.; Oluwagbemigun, K. Associations of BMI and Body Fat with Urine Metabolome in Adolescents Are Sex-Specific: A Cross-Sectional Study. Metabolites 2020, 10, 330. https://doi.org/10.3390/metabo10080330
Brachem C, Langenau J, Weinhold L, Schmid M, Nöthlings U, Oluwagbemigun K. Associations of BMI and Body Fat with Urine Metabolome in Adolescents Are Sex-Specific: A Cross-Sectional Study. Metabolites. 2020; 10(8):330. https://doi.org/10.3390/metabo10080330
Chicago/Turabian StyleBrachem, Christian, Julia Langenau, Leonie Weinhold, Matthias Schmid, Ute Nöthlings, and Kolade Oluwagbemigun. 2020. "Associations of BMI and Body Fat with Urine Metabolome in Adolescents Are Sex-Specific: A Cross-Sectional Study" Metabolites 10, no. 8: 330. https://doi.org/10.3390/metabo10080330
APA StyleBrachem, C., Langenau, J., Weinhold, L., Schmid, M., Nöthlings, U., & Oluwagbemigun, K. (2020). Associations of BMI and Body Fat with Urine Metabolome in Adolescents Are Sex-Specific: A Cross-Sectional Study. Metabolites, 10(8), 330. https://doi.org/10.3390/metabo10080330





