Caulerpa lentillifera (Sea Grapes) Improves Cardiovascular and Metabolic Health of Rats with Diet-Induced Metabolic Syndrome
Abstract
1. Introduction
2. Results
2.1. Caulerpa lentillifera Composition
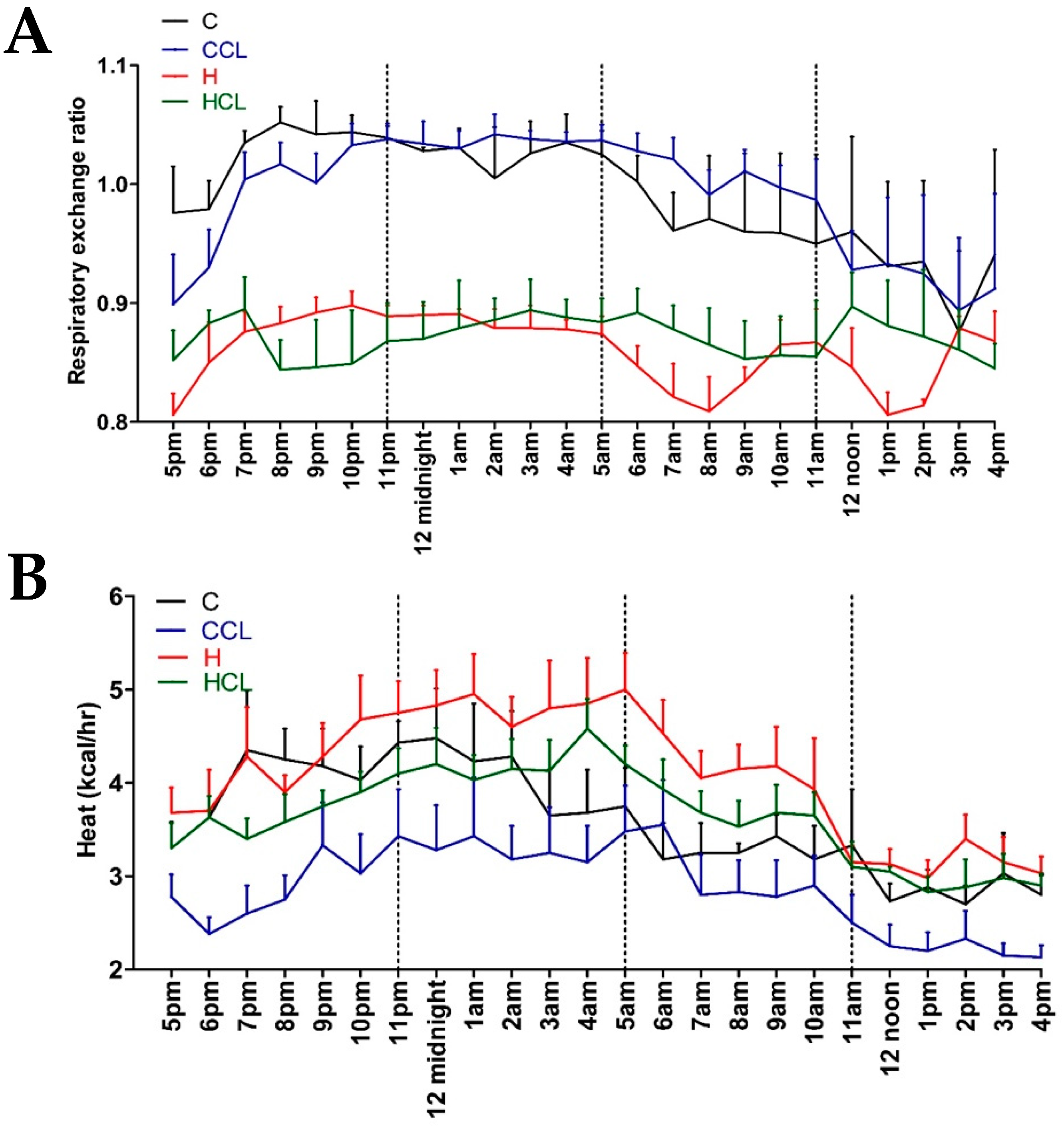
2.2. Physiological Variables
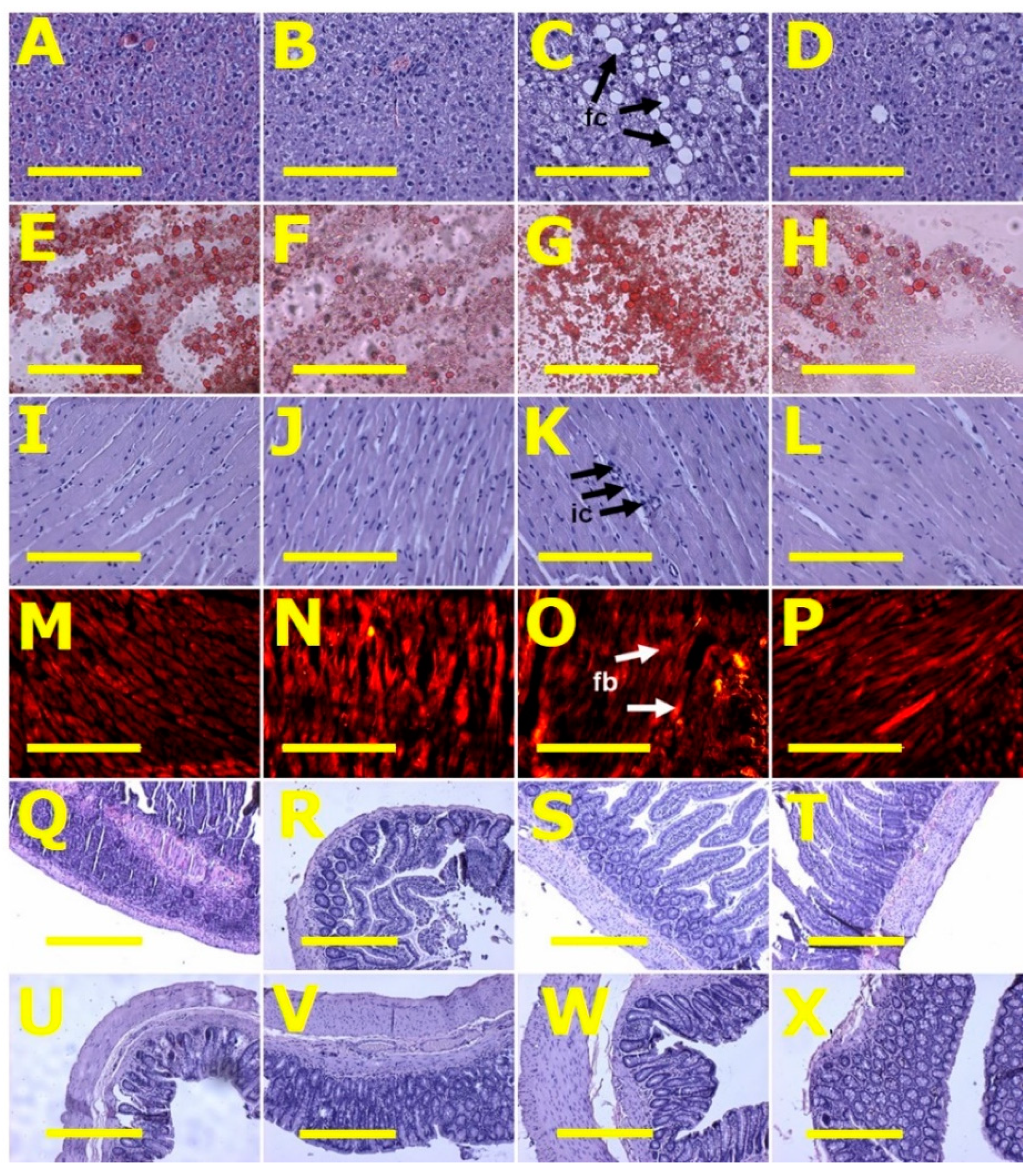
2.3. Gut Structure and Microbiota
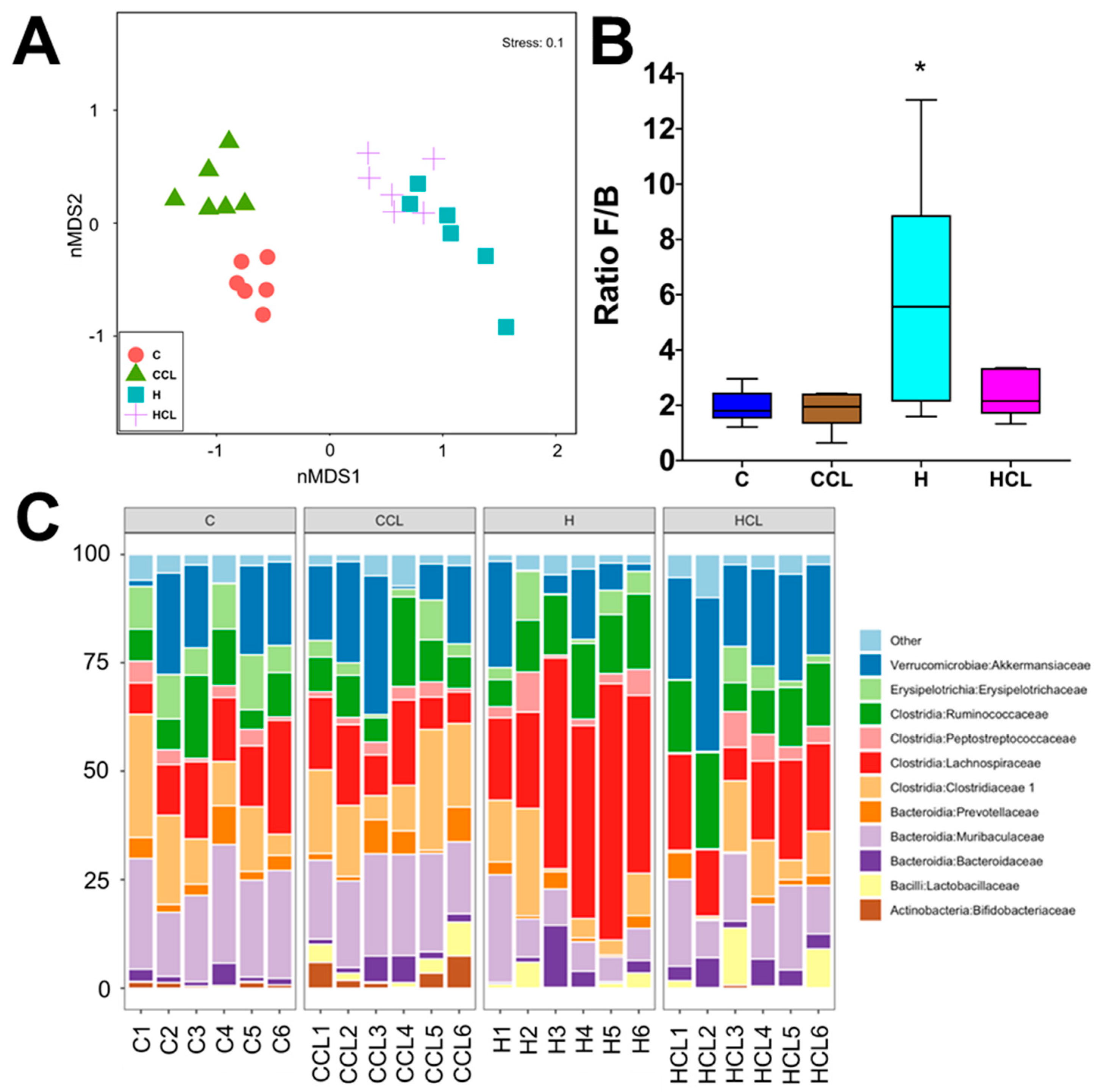
2.4. Taxonomic Structure of the Bacterial Communities
2.5. Multivariate Analysis of Physiological and Microbiota Data
3. Discussion
4. Materials and Methods
4.1. Caulerpa lentillifera Source and Elemental Composition Analysis
4.2. Rats and Diets
4.3. Rat Measurements
4.4. Gut Microbiota Analysis
4.5. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Cherry, P.; O’Hara, C.; Magee, P.J.; McSorley, E.M.; Allsopp, P.J. Risks and benefits of consuming edible seaweeds. Nutr. Rev. 2019, 77, 307–329. [Google Scholar] [CrossRef] [PubMed]
- Wells, M.L.; Potin, P.; Craigie, J.S.; Raven, J.A.; Merchant, S.S.; Helliwell, K.E.; Smith, A.G.; Camire, M.E.; Brawley, S.H. Algae as nutritional and functional food sources: Revisiting our understanding. J. Appl. Phycol. 2017, 29, 949–982. [Google Scholar] [CrossRef] [PubMed]
- Famà, P.; Wysor, B.; Kooistra, W.H.; Zuccarello, G.C. Molecular phylogeny of the genus Caulerpa (Caulerpales, Chlorophyta) inferred from chloroplast tufA gene1. J. Phycol. 2002, 38, 1040–1050. [Google Scholar] [CrossRef]
- Pierucci, A.; De La Fuente, G.; Cannas, R.; Chiantore, M. A new record of the invasive seaweed Caulerpa cylindracea Sonder in the South Adriatic Sea. Heliyon 2019, 5, e02449. [Google Scholar] [CrossRef]
- Paul, N.A.; Dworjanyn, S.A.; de Nys, R. “Green Caviar” and “Sea Grapes”: Targeted Cultivation of High-Value Seaweeds from the Genus Caulerpa; Australian Flora Foundation: Willoughby, NSW, Australia, 2009; Available online: http://aff.org.au/wp-content/uploads/Paul_edible_seaweed_final.pdf (accessed on 30 June 2019).
- Chen, X.; Sun, Y.; Liu, H.; Liu, S.; Qin, Y.; Li, P. Advances in cultivation, wastewater treatment application, bioactive components of Caulerpa lentillifera and their biotechnological applications. PeerJ 2019, 7, e6118. [Google Scholar] [CrossRef]
- Brown, L.; Poudyal, H.; Panchal, S.K. Functional foods as potential therapeutic options for metabolic syndrome. Obes. Rev. 2015, 16, 914–941. [Google Scholar] [CrossRef]
- O’Neill, S.; O’Driscoll, L. Metabolic syndrome: A closer look at the growing epidemic and its associated pathologies. Obes. Rev. 2015, 16, 1–12. [Google Scholar] [CrossRef]
- Matanjun, P.; Mohamed, S.; Muhammad, K.; Mustapha, N.M. Comparison of cardiovascular protective effects of tropical seaweeds, Kappaphycus alvarezii, Caulerpa lentillifera, and Sargassum polycystum, on high-cholesterol/high-fat diet in rats. J. Med. Food 2010, 13, 792–800. [Google Scholar] [CrossRef]
- Sharma, B.R.; Kim, H.J.; Rhyu, D.Y. Caulerpa lentillifera extract ameliorates insulin resistance and regulates glucose metabolism in C57BL/KsJ-db/db mice via PI3K/AKT signaling pathway in myocytes. J. Transl. Med. 2015, 13, 62. [Google Scholar] [CrossRef]
- Sun, Y.; Gong, G.; Guo, Y.; Wang, Z.; Song, S.; Zhu, B.; Zhao, L.; Jiang, J. Purification, structural features and immunostimulatory activity of novel polysaccharides from Caulerpa lentillifera. Int. J. Biol. Macromol. 2018, 108, 314–323. [Google Scholar] [CrossRef]
- Sun, Y.; Liu, Y.; Ai, C.; Song, S.; Chen, X. Caulerpa lentillifera polysaccharides enhance the immunostimulatory activity in immunosuppressed mice in correlation with modulating gut microbiota. Food Funct. 2019, 10, 4315–4329. [Google Scholar] [CrossRef]
- Paul, N.A.; Neveux, N.; Magnusson, M.; de Nys, R. Comparative production and nutritional value of “sea grapes”—the tropical green seaweeds Caulerpa lentillifera and C. racemosa. J. Appl. Phycol. 2014, 26, 1833–1844. [Google Scholar] [CrossRef]
- Panchal, S.K.; Poudyal, H.; Iyer, A.; Nazer, R.; Alam, A.; Diwan, V.; Kauter, K.; Sernia, C.; Campbell, F.; Ward, L.; et al. High-carbohydrate high-fat diet-induced metabolic syndrome and cardiovascular remodeling in rats. J. Cardiovasc. Pharmacol. 2011, 57, 611–624. [Google Scholar] [CrossRef] [PubMed]
- Radulovich, R.; Umanzor, S.; Cabrera, R.; Mata, R. Tropical seaweeds for human food, their cultivation and its effect on biodiversity enrichment. Aquaculture 2015, 436, 40–46. [Google Scholar] [CrossRef]
- de Gaillande, C.; Payri, C.; Remoissenet, G.; Zubia, M. Caulerpa consumption, nutritional value and farming in the Indo-Pacific region. J. Appl. Phycol. 2017, 29, 2249–2266. [Google Scholar] [CrossRef]
- Morris, C.; Bala, S.; South, G.R.; Lako, J.; Lober, M.; Simos, T. Supply chain and marketing of sea grapes, Caulerpa racemosa (Forsskål) J. Agardh (Chlorophyta: Caulerpaceae) in Fiji, Samoa and Tonga. J. Appl. Phycol. 2014, 26, 783–789. [Google Scholar] [CrossRef][Green Version]
- Misheer, N.; Kindness, A.; Jonnalagadda, S.B. Seaweeds along KwaZulu-Natal Coast of South Africa--4: Elemental uptake by edible seaweed Caulerpa racemosa (sea grapes) and the arsenic speciation. J. Environ. Sci. Health A Tox. Hazard Subst. Environ. Eng. 2006, 41, 1217–1233. [Google Scholar] [CrossRef]
- Yap, W.F.; Tay, V.; Tan, S.H.; Yow, Y.Y.; Chew, J. Decoding antioxidant and antibacterial potentials of Malaysian green seaweeds: Caulerpa racemosa and Caulerpa lentillifera. Antibiotics 2019, 8, 152. [Google Scholar] [CrossRef]
- Shevchenko, N.; Burtseva, Y.V.; Zvyagintseva, T.; Makar, T.; Sergeeva, O.; Zakharenko, A.; Isakov, V.; Linh, N.T.; Hoa, N.X.; Ly, B.M. Polysaccharides and sterols from green algae Caulerpa lentillifera and C. sertularioides. Chem. Nat. Compd. 2009, 45, 1–5. [Google Scholar] [CrossRef]
- Kumar, M.; Gupta, V.; Kumari, P.; Reddy, C.R.K.; Jha, B. Assessment of nutrient composition and antioxidant potential of Caulerpaceae seaweeds. Subtrop. Plant Sci. 2011, 24, 270–278. [Google Scholar] [CrossRef]
- Meyer, K.D.; Paul, V.J. Intraplant variation in secondary metabolite concentration in three species of Caulerpa (Chlorophyta: Caulerpales) and its effects on herbivorous fishes. Mar. Ecol. Prog. Ser. 1992, 82, 249–257. [Google Scholar] [CrossRef]
- Zheng, J.; Li, Z.; Manabe, Y.; Kim, M.; Goto, T.; Kawada, T.; Sugawara, T. Siphonaxanthin, a carotenoid from green algae, inhibits lipogenesis in hepatocytes via the suppression of liver X receptor a activity. Lipids 2018, 53, 41–52. [Google Scholar] [CrossRef]
- Manabe, Y.; Takii, Y.; Sugawara, T. Siphonaxanthin, a carotenoid from green algae, suppresses advanced glycation end product-induced inflammatory responses. J. Nat. Med. 2020, 74, 127–134. [Google Scholar] [CrossRef]
- Lunagariya, J.; Bhadja, P.; Zhong, S.; Vekariya, R.; Xu, S. Marine natural product bis-indole alkaloid caulerpin: Chemistry and biology. Mini. Rev. Med. Chem. 2019, 19, 751–761. [Google Scholar] [CrossRef] [PubMed]
- Mao, S.; Guo, Y.; Shen, X. Two novel aromatic valerenane-type sesquiterpenes from the Chinese green alga Caulerpa taxifolia. Bioorg. Med. Chem. Lett. 2006, 16, 2947–2950. [Google Scholar] [CrossRef] [PubMed]
- de Souza, E.T.; de Lira, D.P.; de Queiroz, A.C.; da Silva, D.J.; de Aquino, A.B.; Mella, E.A.; Lorenzo, V.P.; de Miranda, G.E.; de Araujo-Junior, J.X.; Chaves, M.C.; et al. The antinociceptive and anti-inflammatory activities of caulerpin, a bisindole alkaloid isolated from seaweeds of the genus Caulerpa. Mar. Drugs 2009, 7, 689–704. [Google Scholar] [CrossRef] [PubMed]
- Holdt, S.L.; Kraan, S. Bioactive compounds in seaweed: Functional food applications and legislation. J. Appl. Phycol. 2011, 23, 543–597. [Google Scholar] [CrossRef]
- Chin, Y.X.; Mi, Y.; Cao, W.X.; Lim, P.E.; Xue, C.H.; Tang, Q.J. A pilot study on anti-obesity mechanisms of Kappaphycus Alvarezii: The role of native k-carrageenan and the leftover sans-carrageenan fraction. Nutrients 2019, 11, 1133. [Google Scholar] [CrossRef]
- Wanyonyi, S.; du Preez, R.; Brown, L.; Paul, N.A.; Panchal, S.K. Kappaphycus alvarezii as a food supplement prevents diet-induced metabolic syndrome in rats. Nutrients 2017, 9, 1261. [Google Scholar] [CrossRef]
- du Preez, R.; Paul, N.; Mouatt, P.; Majzoub, M.E.; Thomas, T.; Panchal, S.K.; Brown, L. Carrageenans from the red seaweed Sarconema filiforme attenuate symptoms of diet-induced metabolic syndrome in rats. Mar. Drugs 2020, 18, 97. [Google Scholar] [CrossRef]
- Rodrigues, J.A.G.; Quinderé, A.L.G.; de Queiroz, I.N.L.; Coura, C.O.; Benevides, N.M.B. Comparative study of sulfated polysaccharides from Caulerpa spp. (Chlorophyceae). Biotechnological tool for species identification? Acta Sci. Biol. Sci. 2012, 34, 381–389. [Google Scholar] [CrossRef]
- Zaporozhets, T.S.; Besednova, N.N.; Kuznetsova, T.A.; Zvyagintseva, T.N.; Makarenkova, I.D.; Kryzhanovsky, S.P.; Melnikov, V.G. The prebiotic potential of polysaccharides and extracts of seaweeds. Russ. J. Mar. Biol. 2014, 40, 1–9. [Google Scholar] [CrossRef]
- Gibson, G.R.; Scott, K.P.; Rastall, R.A.; Tuohy, K.M.; Hotchkiss, A.; Dubert-Ferrandon, A.; Gareau, M.; Murphy, E.F.; Saulnier, D.; Loh, G. Dietary prebiotics: Current status and new definition. Food Sci. Technol. Bull. Funct. Foods 2010, 7, 1–19. [Google Scholar] [CrossRef]
- Katiyar, R.; Gurjar, B.; Biswas, S.; Pruthi, V.; Kumar, N.; Kumar, P. Microalgae: An emerging source of energy based bio-products and a solution for environmental issues. Renew. Sust. Energ. Rev. 2017, 72, 1083–1093. [Google Scholar] [CrossRef]
- Lattimer, J.M.; Haub, M.D. Effects of dietary fiber and its components on metabolic health. Nutrients 2010, 2, 1266–1289. [Google Scholar] [CrossRef]
- Kumar, S.A.; Ward, L.C.; Brown, L. Inulin oligofructose attenuates metabolic syndrome in high-carbohydrate, high-fat diet-fed rats. Br. J. Nutr. 2016, 116, 1502–1511. [Google Scholar] [CrossRef]
- Granado-Serrano, A.B.; Martin-Gari, M.; Sanchez, V.; Riart Solans, M.; Berdun, R.; Ludwig, I.A.; Rubio, L.; Vilaprinyo, E.; Portero-Otin, M.; Serrano, J.C.E. Faecal bacterial and short-chain fatty acids signature in hypercholesterolemia. Sci. Rep. 2019, 9, 1772. [Google Scholar] [CrossRef]
- Dhingra, D.; Michael, M.; Rajput, H.; Patil, R.T. Dietary fibre in foods: A review. J. Food Sci. Technol. 2012, 49, 255–266. [Google Scholar] [CrossRef]
- Hotamisligil, G.S. Inflammation and metabolic disorders. Nature 2006, 444, 860–867. [Google Scholar] [CrossRef]
- Saetang, J.; Sangkhathat, S. Role of innate lymphoid cells in obesity and metabolic disease. Mol. Med. Rep. 2018, 17, 1403–1412. [Google Scholar] [CrossRef]
- McLaughlin, T.; Ackerman, S.E.; Shen, L.; Engleman, E. Role of innate and adaptive immunity in obesity-associated metabolic disease. J. Clin. Investig. 2017, 127, 5–13. [Google Scholar] [CrossRef] [PubMed]
- Everaere, L.; Ait Yahia, S.; Boute, M.; Audousset, C.; Chenivesse, C.; Tsicopoulos, A. Innate lymphoid cells at the interface between obesity and asthma. Immunology 2018, 153, 21–30. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, T.; Moro, K.; Kubota, T.; Kubota, N.; Kato, T.; Ohno, H.; Nakae, S.; Saito, H.; Koyasu, S. Innate lymphoid cells in the induction of obesity. Cell Rep. 2019, 28, 202–217. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Zhao, M.; Qing, Y.; Luo, Y.; Xia, G.; Li, Y. Study on immunostimulatory activity and extraction process optimization of polysaccharides from Caulerpa lentillifera. Int. J. Biol. Macromol. 2020, 143, 677–684. [Google Scholar] [CrossRef] [PubMed]
- Chambers, E.S.; Preston, T.; Frost, G.; Morrison, D.J. Role of gut microbiota-generated short-chain fatty acids in metabolic and cardiovascular health. Curr. Nutr. Rep. 2018, 7, 198–206. [Google Scholar] [CrossRef] [PubMed]
- Cardoso, C.; Ripol, A.; Afonso, C.; Freire, M.; Varela, J.; Quental-Ferreira, H.; Pousao-Ferreira, P.; Bandarra, N. Fatty acid profiles of the main lipid classes of green seaweeds from fish pond aquaculture. Food Sci. Nutr. 2017, 5, 1186–1194. [Google Scholar] [CrossRef]
- Koh, A.; De Vadder, F.; Kovatcheva-Datchary, P.; Backhed, F. From dietary fiber to host physiology: Short-chain fatty acids as key bacterial metabolites. Cell 2016, 165, 1332–1345. [Google Scholar] [CrossRef]
- de Jesus Raposo, M.F.; de Morais, A.M.; de Morais, R.M. Emergent sources of prebiotics: Seaweeds and microalgae. Mar. Drugs 2016, 14, 27. [Google Scholar] [CrossRef]
- Lordan, C.; Thapa, D.; Ross, R.P.; Cotter, P.D. Potential for enriching next-generation health-promoting gut bacteria through prebiotics and other dietary components. Gut Microbes 2020, 11, 1–20. [Google Scholar] [CrossRef]
- O’Sullivan, L.; Murphy, B.; McLoughlin, P.; Duggan, P.; Lawlor, P.G.; Hughes, H.; Gardiner, G.E. Prebiotics from marine macroalgae for human and animal health applications. Mar. Drugs 2010, 8, 2038–2064. [Google Scholar] [CrossRef]
- Matsumura, Y. Nutrition trends in Japan. Asia Pac. J. Clin. Nutr. 2001, 10, S40–S47. [Google Scholar] [CrossRef] [PubMed]
- Reagan-Shaw, S.; Nihal, M.; Ahmad, N. Dose translation from animal to human studies revisited. FASEB J. 2008, 22, 659–661. [Google Scholar] [CrossRef] [PubMed]
- Zheng, H.; You, Y.; Hua, M.; Wu, P.; Liu, Y.; Chen, Z.; Zhang, L.; Wei, H.; Li, Y.; Luo, M.; et al. Chlorophyllin modulates gut microbiota and inhibits intestinal inflammation to ameliorate hepatic fibrosis in mice. Front. Physiol. 2018, 9, 1671. [Google Scholar] [CrossRef] [PubMed]
- Sekar, S.; Shafie, S.R.; Prasadam, I.; Crawford, R.; Panchal, S.K.; Brown, L.; Xiao, Y. Saturated fatty acids induce development of both metabolic syndrome and osteoarthritis in rats. Sci. Rep. 2017, 7, 46457. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Bruns, M.A.; Tiedje, J.M. DNA recovery from soils of diverse composition. Appl. Environ. Microbiol. 1996, 62, 316–322. [Google Scholar] [CrossRef]
- Illumina. MiSeq System User Guide. In Illumina Inc.; FU Berlin: Berlin, Germany; Available online: https://sapac.support.illumina.com/sequencing/sequencing_instruments/miseq/documentation.html (accessed on 30 June 2019).
- Bolger, A.M.; Lohse, M.; Usadel, B. Trimmomatic: A flexible trimmer for Illumina sequence data. Bioinformatics 2014, 30, 2114–2120. [Google Scholar] [CrossRef] [PubMed]
- Edgar, R.C. Search and clustering orders of magnitude faster than BLAST. Bioinformatics 2010, 26, 2460–2461. [Google Scholar] [CrossRef]
- Wemheuer, B.; Wemheuer, F. Assessing bacterial and fungal diversity in the plant endosphere. Methods Mol. Biol. 2017, 1539, 75–84. [Google Scholar] [CrossRef]
- Ozkan, J.; Willcox, M.; Wemheuer, B.; Wilcsek, G.; Coroneo, M.; Thomas, T. Biogeography of the human ocular microbiota. Ocul. Surf. 2019, 17, 111–118. [Google Scholar] [CrossRef]
- Edgar, R.C.; Haas, B.J.; Clemente, J.C.; Quince, C.; Knight, R. UCHIME improves sensitivity and speed of chimera detection. Bioinformatics 2011, 27, 2194–2200. [Google Scholar] [CrossRef]
- Quast, C.; Pruesse, E.; Yilmaz, P.; Gerken, J.; Schweer, T.; Yarza, P.; Peplies, J.; Glockner, F.O. The SILVA ribosomal RNA gene database project: Improved data processing and web-based tools. Nucleic Acids Res. 2013, 41, D590–D596. [Google Scholar] [CrossRef] [PubMed]
- Camacho, C.; Coulouris, G.; Avagyan, V.; Ma, N.; Papadopoulos, J.; Bealer, K.; Madden, T.L. BLAST+: Architecture and applications. BMC Bioinform. 2009, 10, 421. [Google Scholar] [CrossRef] [PubMed]
- Oksanen, J.; Blanchet, F.G.; Kindt, R.; Legendre, P.; Minchin, P.R.; O’Hara, R.B.; Simpson, G.L.; Solymos, P.; Stevens, M.H.H.; Szoecs, E.; et al. Vegan: Community Ecology Package, R Package Version 2.5-2; 2018. Available online: https://CRAN.R-project.org/package=vegan (accessed on 30 March 2019).
- Clarke, K.; Gorley, R. PRIMER v6: User Manual/Tutorial: Plymouth; Primer-E, Ltd.: Plymouth, UK, 2006. [Google Scholar]
- Anderson, M. A new method for non-parametric multivariate analysis of variance. Austral Ecol. 2001, 26, 32–46. [Google Scholar] [CrossRef]
- Bikker, P.; van Krimpen, M.M.; van Wikselaar, P.; Houweling-Tan, B.; Scaccia, N.; van Hal, J.W.; Huijgen, W.J.; Cone, J.W.; Lopez-Contreras, A.M. Biorefinery of the green seaweed Ulva lactuca to produce animal feed, chemicals and biofuels. J. Appl. Phycol. 2016, 28, 3511–3525. [Google Scholar] [CrossRef] [PubMed]
| Element | Concentration (mg/kg) |
|---|---|
| Aluminium (Al) | 744 ± 8 |
| Arsenic (As) | ≤1 |
| Barium (Ba) | 4.75 ± 0.19 |
| Boron (B) | 21.7 ± 0.5 |
| Cadmium (Cd) | 1.14 ± 0.03 |
| Calcium (Ca) | 8137 ± 73 |
| Chromium (Cr) | 3.3 ± 0.5 |
| Cobalt (Co) | 1.35 ± 0.05 |
| Copper (Cu) | 2.74 ± 0.3 |
| Iron (Fe) | 595 ± 5 |
| Lead (Pb) | 2.22 ± 0.08 |
| Magnesium (Mg) | 10,663 ± 52 |
| Manganese (Mn) | 425 ± 14 |
| Mercury (Hg) | ≤1 |
| Molybdenum (Mo) | 1.32 ± 0.14 |
| Nickel (Ni) | 1.88 ± 0.19 |
| Phosphorus (P) | 1073 ± 17 |
| Potassium (K) | 1066 ± 546 |
| Selenium (Se) | ≤1 |
| Sodium (Na) | 130,794 ± 763 |
| Strontium (Sr) | 104 ± 3 |
| Sulphur (S) | 6733 ± 72 |
| Vanadium (V) | 2.46 ± 0.08 |
| Zinc (Zn) | 15.2 ± 0.3 |
| Variables | C | CCL | H | HCL | p Value | ||
|---|---|---|---|---|---|---|---|
| Diet | Treatment | Interaction | |||||
| Physiological Variables | |||||||
| 0 week body weight, g | 337 ± 1 | 339 ± 1 | 338 ± 1 | 337 ± 1 | 0.6196 | 0.6196 | 0.1408 |
| 8 week body weight, g | 364 ± 12 b | 358 ± 6 b | 439 ± 9 a | 440 ± 8 a | <0.0001 | 0.7828 | 0.6997 |
| 16 week body weight, g | 411 ± 14 c | 389 ± 6 c | 573 ± 10 a | 468 ± 10 b | <0.0001 | <0.0001 | 0.0002 |
| 16 week lean mass, g | 316 ± 7 | 314 ± 5 | 303 ± 14 | 303 ± 9 | 0.2069 | 0.9155 | 0.9155 |
| 16 week fat mass, g | 80 ± 9 c | 57 ± 4 d | 258 ± 28 a | 148 ± 14 b | <0.0001 | 0.0002 | 0.0111 |
| 8 week lean/fat mass proportion | 5.9 ± 0.7 b | 10.7 ± 2.6 a | 2.6 ± 0.2 c | 3.2 ± 1.2 c | 0.0007 | 0.0744 | 0.1622 |
| 16 week lean/fat mass proportion | 4.2 ± 0.5 b | 6.0 ± 0.7 a | 1.3 ± 0.2 d | 2.3 ± 0.4 c | <0.0001 | 0.0060 | 0.4137 |
| 16 week bone mineral content, g | 11.9 ± 0.4 c | 11.4 ± 0.2 c | 17.7 ± 1.1 a | 14.3 ± 0.5 b | <0.0001 | 0.0014 | 0.0139 |
| 16 week bone mineral density, g/cm2 | 0.184 ± 0.004 b | 0.171 ± 0.003 c | 0.191 ± 0.005 a | 0.183 ± 0.004 b | 0.0309 | 0.0179 | 0.5565 |
| Food intake 0–8 weeks, g/day | 37.5 ± 1.4 a | 37.6 ± 0.4 a | 27.8 ± 1.7 b | 25.9 ± 0.7 b | <0.0001 | 0.4469 | 0.3984 |
| Food intake 9–16 weeks, g/day | 39.7 ± 2.2 a | 37.5 ± 0.9 a | 29.3 ± 2.5 b | 19.3 ± 1.0 c | <0.0001 | 0.0015 | 0.0353 |
| Water intake 0–8 weeks, g/day | 33.4 ± 2.8 | 30.2 ± 1.7 | 31.4 ± 3.1 | 29.1 ± 0.7 | 0.5005 | 0.2346 | 0.8446 |
| Water intake 9–16 weeks, g/day | 25.9 ± 3.3 b | 52.6 ± 2.0 a | 29.8 ± 2.1 b | 50.8 ± 1.2 a | 0.6470 | <0.0001 | 0.2173 |
| Energy intake 0–8 weeks, kJ/day | 421 ± 16 b | 422 ± 4 b | 603 ± 34 a | 598 ± 25 a | <0.0001 | 0.9301 | 0.8953 |
| Energy intake 9–16 weeks, kJ/day | 447 ± 24 b | 416 ± 10 b | 620 ± 46 a | 551 ± 21 a | <0.0001 | 0.0856 | 0.5074 |
| 16 week abdominal circumference, cm | 19.0 ± 0.4 c | 18.4 ± 0.2 c | 23.4 ± 0.4 a | 21.1 ± 0.3 b | <0.0001 | <0.0001 | 0.0149 |
| Body mass index, g/cm2 | 0.67 ± 0.02 b | 0.63 ± 0.01 b | 0.77 ± 0.02 a | 0.69 ± 0.01 b | <0.0001 | 0.0004 | 0.2126 |
| Retroperitoneal fat, mg/mm | 231 ± 15 c | 178 ± 13 d | 628 ± 68 a | 398 ± 37 b | <0.0001 | 0.0010 | 0.0320 |
| Epididymal fat, mg/mm | 69 ± 6 c | 41 ± 4 d | 182 ± 32 a | 134 ± 16 b | <0.0001 | 0.0432 | 0.5865 |
| Omental fat, mg/mm | 149 ± 14 c | 134 ± 7 c | 325 ± 39 a | 229 ± 31 b | <0.0001 | 0.0392 | 0.1280 |
| Total abdominal fat, mg/mm | 450 ± 27 c | 353 ± 21 d | 1136 ± 135 a | 761 ± 77 b | <0.0001 | 0.0049 | 0.0876 |
| Visceral adiposity, % | 5.1 ± 0.2 c | 4.3 ± 0.2 d | 9.8 ± 0.8 a | 7.7 ± 0.7 b | <0.0001 | 0.0115 | 0.2436 |
| Liver wet weight, mg/mm | 235 ± 11 b | 250 ± 6 b | 403 ± 18 a | 379 ± 13 a | <0.0001 | 0.7258 | 0.1332 |
| Cardiovascular Variables | |||||||
| 8 week systolic blood pressure, mmHg | 118 ± 6 | 120 ± 2 | 131 ± 2 | 126 ± 3 | 0.0083 | 0.6597 | 0.3075 |
| 16 week systolic blood pressure, mmHg | 119 ± 3 b | 117 ± 4 b | 135 ± 3 a | 118 ± 3 b | 0.0356 | 0.0198 | 0.0617 |
| Left ventricle + septum wet weight, mg/mm | 21.5 ± 1.5 b | 21.4 ± 1.1 b | 25.8 ± 2.0 a | 22.5 ± 0.4 b | 0.0314 | 0.1661 | 0.1917 |
| Right ventricle, mg/mm | 4.2 ± 0.7 | 4.7 ± 0.3 | 4.5 ± 0.2 | 4.7 ± 0.3 | 0.7057 | 0.3805 | 0.7057 |
| Left ventricular diastolic stiffness (κ) | 21.1 ± 1.5 c | 22.1 ± 1.2 c | 30.2 ± 0.6 a | 27.1 ± 0.8 b | <0.0001 | 0.3375 | 0.0649 |
| Left ventricle collagen area, % | 7.8 ± 0.7 c | 9.1 ± 1.2 c | 29.4 ± 2.2 a | 22.1 ± 2.8 b | <0.0001 | 0.2748 | 0.1482 |
| Metabolic Variables | |||||||
| Plasma total cholesterol, mmol/L | 1.59 ± 0.06 b | 1.54 ± 0.05 b | 1.73 ± 0.09 a | 1.56 ± 0.06 b | 0.2368 | 0.1063 | 0.3733 |
| HDL-cholesterol, mmol/L | 0.93 ± 0.14 a | 1.11 ± 0.07 a | 0.95 ± 0.09 a | 0.70 ± 0.05 b | 0.0268 | <0.0001 | <0.0001 |
| LDL-cholesterol, mmol/L | 0.55 ± 0.14 | 0.80 ± 0.32 | 0.92 ± 0.08 | 0.61 ± 0.07 | 0.7120 | 0.9020 | 0.2551 |
| Plasma triglycerides, mmol/L | 0.50 ± 0.05 b | 0.64 ± 0.10 b | 1.15 ± 0.13 a | 1.12 ± 0.18 a | <0.0001 | 0.6603 | 0.4977 |
| Plasma non-esterified fatty acids, mmol/L | 0.68 ± 0.12 c | 0.47 ± 0.04 d | 2.71 ± 0.29 a | 1.61 ± 0.49 b | <0.0001 | 0.0298 | 0.1342 |
| Alanine transaminase, U/L | 39 ± 5 | 39 ± 6 | 50 ± 11 | 62 ± 12 | 0.0663 | 0.5098 | 0.5098 |
| Aspartate transaminase, U/L | 138 ± 20 | 163 ± 14 | 174 ± 17 | 220 ± 45 | 0.0917 | 0.1949 | 0.6989 |
| Liver inflammatory cells, cells/200µm2 | 12 ± 2 | 13 ± 2 | 28 ± 2 | 21 ± 3 | 0.0001 | 0.2859 | 0.1576 |
| Liver fat vacuoles area, µm2 | 11.2 ± 1.9 c | 9.2 ± 1.4 c | 84.5 ± 2.6 a | 45.5 ± 3.1 b | <0.0001 | <0.0001 | <0.0001 |
| Oral Glucose Tolerance Test | |||||||
| 0 week basal blood glucose, mmol/L | 2.8 ± 0.1 | 2.8 ± 0.1 | 2.9 ± 0.1 | 2.8 ± 0.1 | 0.6196 | 0.6196 | 0.6196 |
| 0 week area under the curve, mmol/L × min | 648 ± 31 | 641 ± 19 | 620 ± 44 | 633 ± 19 | 0.5526 | 0.9210 | 0.7411 |
| 8 week basal blood glucose, mmol/L | 2.5 ± 0.2 | 2.5 ± 0.1 | 3.2 ± 0.1 | 3.4 ± 0.2 | <0.0001 | 0.5304 | 0.5304 |
| 8 week 120 min blood glucose, mmol/L | 4.0 ± 0.1 | 3.7 ± 0.1 | 4.8 ± 0.4 | 5.3 ± 0.1 | <0.0001 | 0.6486 | 0.0732 |
| 8 week area under the curve, mmol/L × min | 526 ± 24 | 566 ± 15 | 613 ± 23 | 688 ± 14 | <0.0001 | 0.0052 | 0.3751 |
| 16 week basal blood glucose, mmol/L | 2.9 ± 0.2 | 2.4 ± 0.1 | 2.8 ± 0.2 | 2.8 ± 0.1 | 0.3480 | 0.1210 | 0.1210 |
| 16 week 120 min blood glucose, mmol/L | 3.4 ± 0.1 | 3.3 ± 0.1 | 4.7 ± 0.3 | 4.5 ± 0.1 | <0.0001 | 0.3912 | 0.7742 |
| 16 week area under the curve, mmol/L × mins | 478 ± 17 | 472 ± 11 | 563 ± 16 | 571 ± 9 | <0.0001 | 0.9505 | 0.6643 |
| Insulin Tolerance Test | |||||||
| 8 week 120 min blood glucose, mmol/L | 3.3 ± 0.9 | 2.7 ± 0.5 | 3.0 ± 0.4 | 4.3 ± 0.2 | 0.2531 | 0.5361 | 0.0976 |
| 8 week area under the curve, mmol/L × min | 147 ± 45 | 144 ± 22 | 353 ± 40 | 441 ± 20 | <0.0001 | 0.2122 | 0.1823 |
| 16 week 120 min blood glucose, mmol/L | 1.1 ± 0.4 | 2.3 ± 0.5 | 3.6 ± 0.3 | 3.4 ± 0.3 | <0.0001 | 0.1997 | 0.0752 |
| 16 week area under the curve, mmol/L × min | 111 ± 11 | 207 ± 28 | 356 ± 36 | 332 ± 28 | <0.0001 | 0.1944 | 0.0334 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
du Preez, R.; Majzoub, M.E.; Thomas, T.; Panchal, S.K.; Brown, L. Caulerpa lentillifera (Sea Grapes) Improves Cardiovascular and Metabolic Health of Rats with Diet-Induced Metabolic Syndrome. Metabolites 2020, 10, 500. https://doi.org/10.3390/metabo10120500
du Preez R, Majzoub ME, Thomas T, Panchal SK, Brown L. Caulerpa lentillifera (Sea Grapes) Improves Cardiovascular and Metabolic Health of Rats with Diet-Induced Metabolic Syndrome. Metabolites. 2020; 10(12):500. https://doi.org/10.3390/metabo10120500
Chicago/Turabian Styledu Preez, Ryan, Marwan E. Majzoub, Torsten Thomas, Sunil K. Panchal, and Lindsay Brown. 2020. "Caulerpa lentillifera (Sea Grapes) Improves Cardiovascular and Metabolic Health of Rats with Diet-Induced Metabolic Syndrome" Metabolites 10, no. 12: 500. https://doi.org/10.3390/metabo10120500
APA Styledu Preez, R., Majzoub, M. E., Thomas, T., Panchal, S. K., & Brown, L. (2020). Caulerpa lentillifera (Sea Grapes) Improves Cardiovascular and Metabolic Health of Rats with Diet-Induced Metabolic Syndrome. Metabolites, 10(12), 500. https://doi.org/10.3390/metabo10120500







