Novel Biomarkers to Distinguish between Type 3c and Type 2 Diabetes Mellitus by Untargeted Metabolomics
Abstract
1. Introduction
2. Results
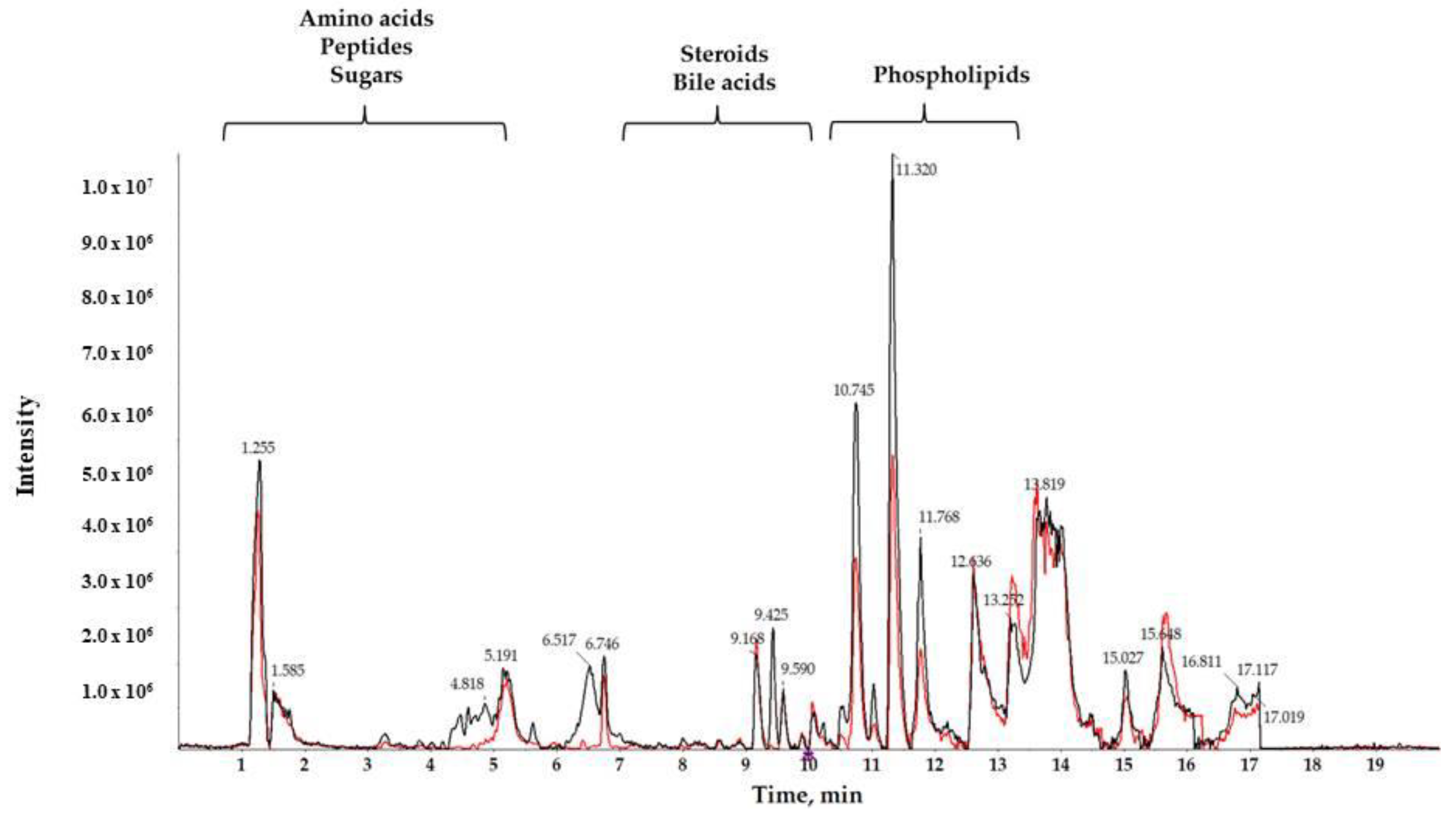
2.1. LC-HRMS Analysis
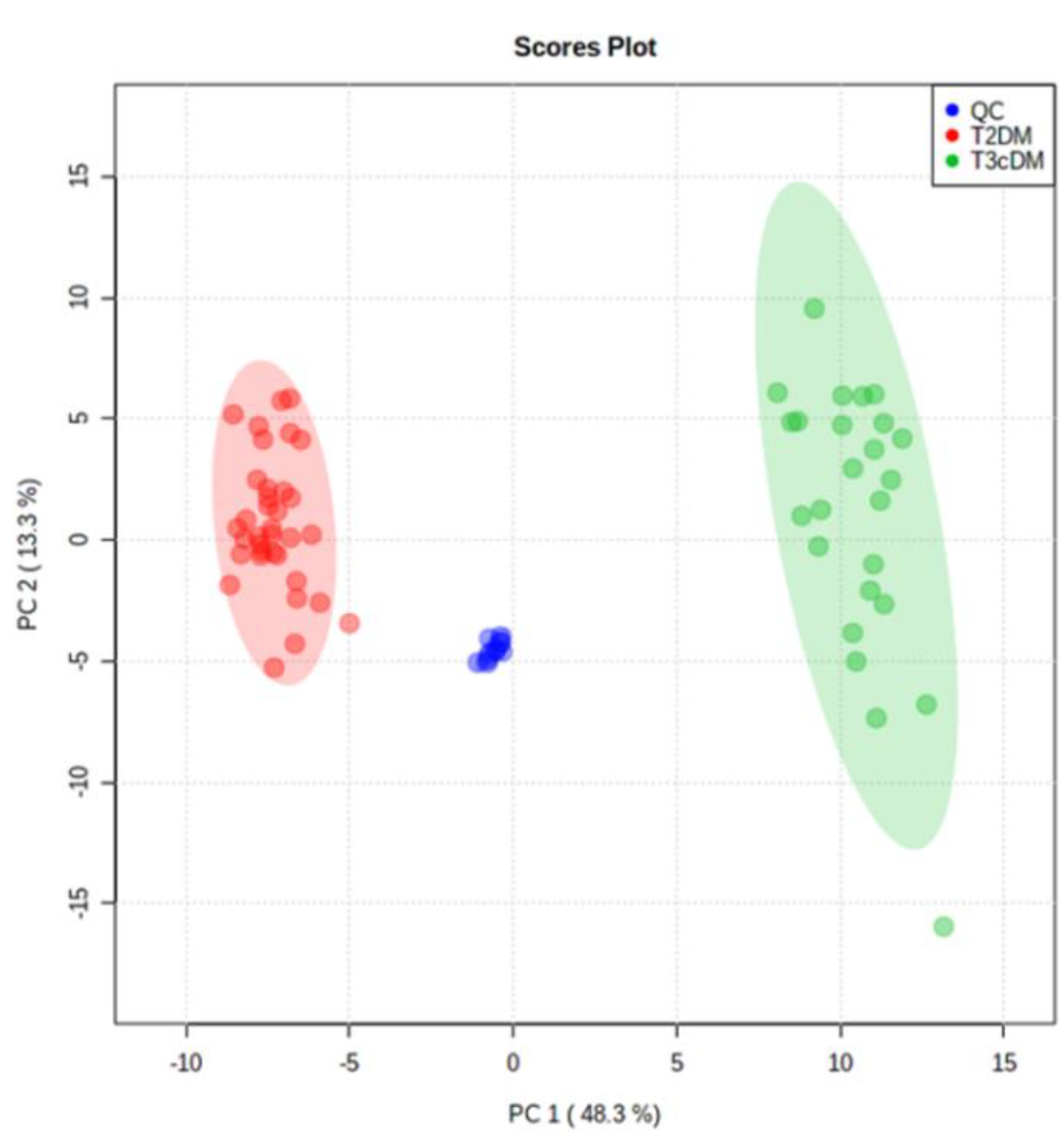
2.2. Chemometric Analysis
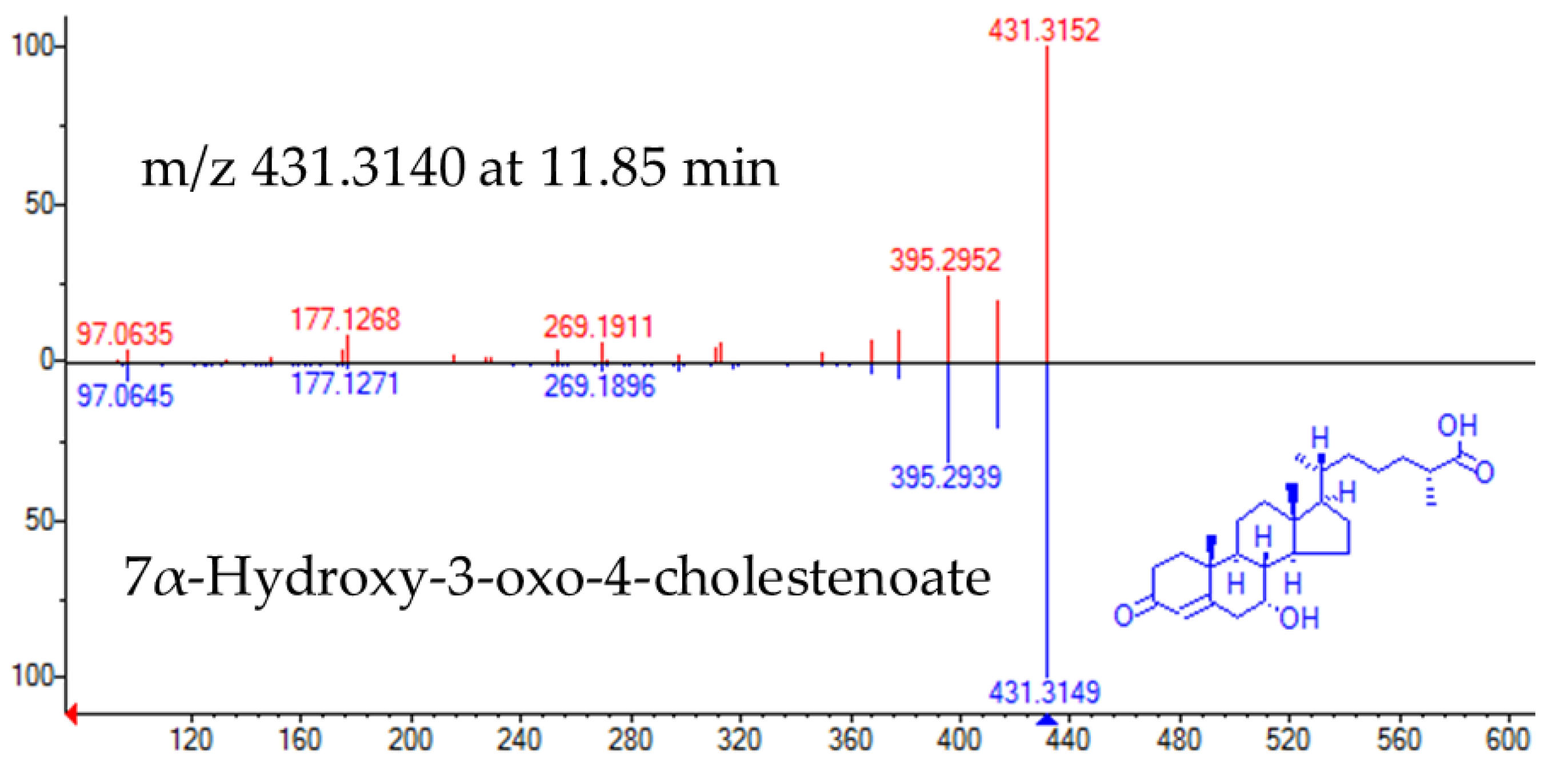
2.3. Identification of Potential Biomarkers
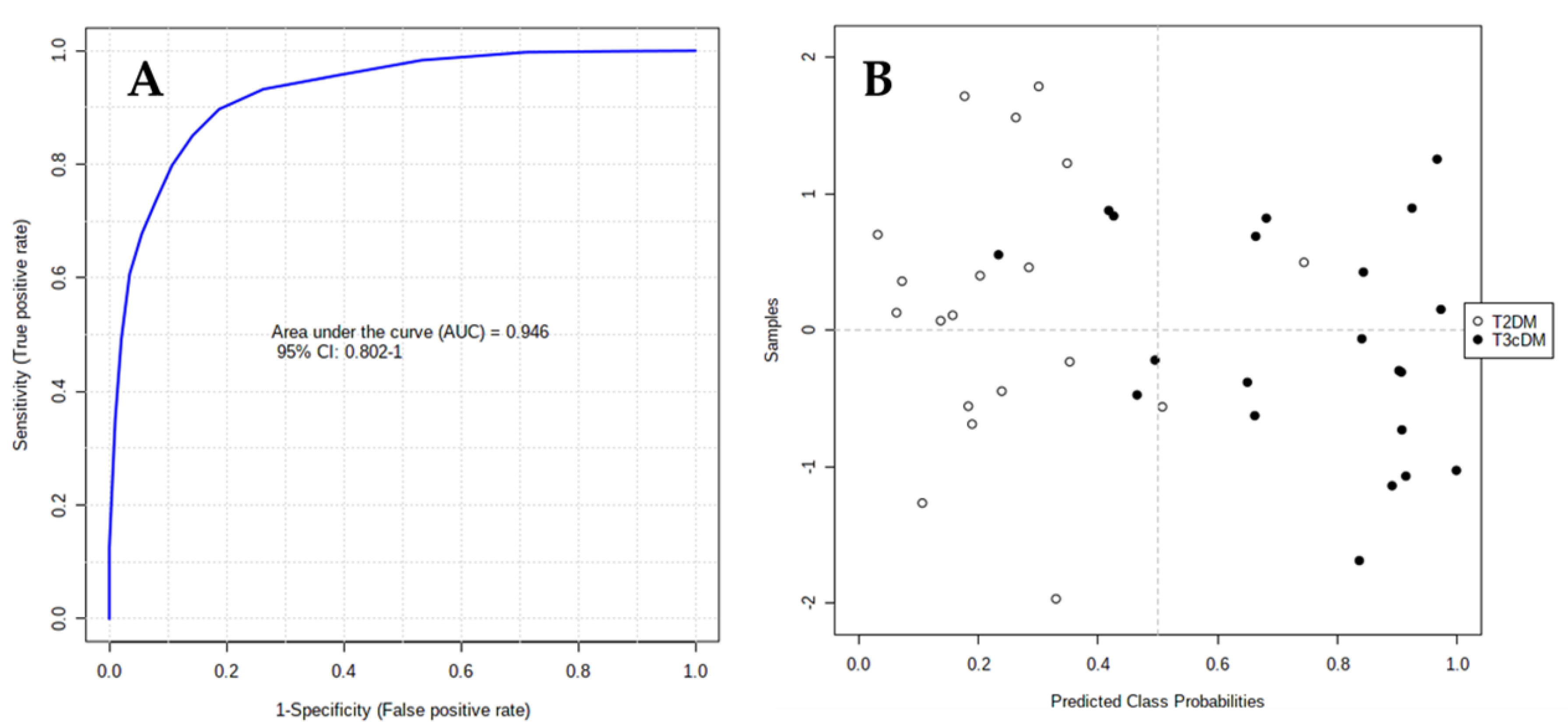
2.4. Biomarker Evaluation
3. Discussion
4. Materials and Methods
4.1. Sample Collection
4.2. Metabolite Extraction
4.3. LC-HRMS Analysis
4.4. Data Set Creation
4.5. Data Pre-Treatment
4.6. Analytical Validation and Outlier Detection
4.7. Statistical Analysis
4.8. Biomarker Identification
4.9. Metabolite Evaluation
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Skyler, J.S.; Bakris, G.L.; Bonifacio, E.; Darsow, T.; Eckel, R.H.; Groop, L.; Groop, P.H.; Handelsman, Y.; Insel, R.A.; Mathieu, C.; et al. Differentiation of diabetes by pathophysiology, natural history, and prognosis. Diabetes 2017, 66, 241–255. [Google Scholar] [CrossRef]
- Goodarzi, M.O.; Nagpal, T.; Greer, P.; Cui, J.; Chen, Y.I.; Guo, X.; Pankow, J.S.; Rotter, J.I.; Alkaade, S.; Amann, S.T.; et al. Genetic Risk Score in Diabetes Associated With Chronic Pancreatitis Versus Type 2 Diabetes Mellitus. Clin. Transl. Gastroenterol. 2019, 10, e00057. [Google Scholar] [CrossRef] [PubMed]
- Kleeff, J.; Whitcomb, D.C.; Shimosegawa, T.; Esposito, I.; Lerch, M.M.; Gress, T.; Mayerle, J.; Drewes, A.M.; Rebours, V.; Akisik, F.; et al. Chronic pancreatitis. Nat. Rev. Dis. Primers 2017, 3, 17060. [Google Scholar] [CrossRef] [PubMed]
- Petrov, M.S.; Yadav, D. Global epidemiology and holistic prevention of pancreatitis. Nat. Rev. Gastroenterol. Hepatol. 2019, 16, 175–184. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.; Liu, D.; Wei, Q.; Lin, H.; Zhi, M.; Chen, Y.; Qi, L.; Waldron, R.T.; Lugea, A.; Pandol, S.J.; et al. New-Onset Diabetes Mellitus After Chronic Pancreatitis Diagnosis: A Systematic Review and Meta-analysis. Pancreas 2019, 48, 868–875. [Google Scholar] [CrossRef] [PubMed]
- Bhattamisra, S.K.; Siang, T.C.; Rong, C.Y.; Annan, N.C.; Sean, E.H.Y.; Xi, L.W.; Lyn, O.S.; Shan, L.H.; Choudhury, H.; Pandey, M.; et al. Type-3c Diabetes Mellitus, Diabetes of Exocrine Pancreas—An Update. Curr. Diabetes Rev. 2019, 15, 382–394. [Google Scholar] [CrossRef]
- Woodmansey, C.; McGovern, A.P.; McCullough, K.A.; Whyte, M.B.; Munro, N.M.; Correa, A.C.; Gatenby, P.A.C.; Jones, S.A.; de Lusignan, S. Incidence, Demographics, and Clinical Characteristics of Diabetes of the Exocrine Pancreas (Type 3c): A Retrospective Cohort Study. Diabetes Care 2017, 40, 1486–1493. [Google Scholar] [CrossRef]
- Rawla, P.; Sunkara, T.; Gaduputi, V. Epidemiology of Pancreatic Cancer: Global Trends, Etiology and Risk Factors. World J. Oncol. 2019, 10, 10–27. [Google Scholar] [CrossRef]
- Kirkegård, J.; Mortensen, F.V.; Cronin-Fenton, D. Chronic Pancreatitis and Pancreatic Cancer Risk: A Systematic Review and Meta-analysis. Am. J. Gastroenterol. 2017, 112, 1366–1372. [Google Scholar] [CrossRef]
- Hart, P.A.; Bellin, M.D.; Andersen, D.K.; Bradley, D.; Cruz-Monserrate, Z.; Forsmark, C.E.; Goodarzi, M.O.; Habtezion, A.; Korc, M.; Kudva, Y.C. Type 3c (pancreatogenic) diabetes mellitus secondary to chronic pancreatitis and pancreatic cancer. Lancet Gastroenterol. Hepatol. 2016, 1, 226–237. [Google Scholar] [CrossRef]
- Zhang, A.; Sun, H.; Wang, P.; Han, Y.; Wang, X. Modern analytical techniques in metabolomics analysis. Analyst 2012, 137, 293–300. [Google Scholar] [CrossRef] [PubMed]
- Cajka, T.; Fiehn, O. Toward Merging Untargeted and Targeted Methods in Mass Spectrometry-Based Metabolomics and Lipidomics. Anal Chem. 2016, 88, 524–545. [Google Scholar] [CrossRef] [PubMed]
- González-Domínguez, R.; Sayago, A.; Fernández-Recamales, A. High-Throughput Direct Mass Spectrometry-Based Metabolomics to Characterize Metabolite Fingerprints Associated with Alzheimer’s Disease Pathogenesis. Metabolites 2018, 8, 52. [Google Scholar] [CrossRef] [PubMed]
- Ríos Peces, S.; Díaz Navarro, C.; Márquez López, C.; Caba, O.; Jiménez-Luna, C.; Melguizo, C.; Prados, J.C.; Genilloud, O.; Vicente Pérez, F.; Pérez Del Palacio, J. Untargeted LC-HRMS-Based Metabolomics for Searching New Biomarkers of Pancreatic Ductal Adenocarcinoma: A Pilot Study. SLAS Discov. 2017, 22, 348–359. [Google Scholar] [CrossRef] [PubMed]
- Xu, F.; Tavintharan, S.; Sum, C.F.; Woon, K.; Lim, S.C.; Ong, C.N. Metabolic signature shift in type 2 diabetes mellitus revealed by mass spectrometry-based metabolomics. J. Clin. Endocrinol. Metab. 2013, 98, E1060–E1065. [Google Scholar] [CrossRef] [PubMed]
- Ahonen, L.; Jäntti, S.; Suvitaival, T.; Theilade, S.; Risz, C.; Kostiainen, R.; Rossing, P.; Orešič, M.; Hyötyläinen, T. Targeted Clinical Metabolite Profiling Platform for the Stratification of Diabetic Patients. Metabolites 2019, 9, 184. [Google Scholar] [CrossRef]
- Johnston, P.C.; Thompson, J.; Mckee, A.; Hamill, C.; Wallace, I. Diabetes and Chronic Pancreatitis: Considerations in the Holistic Management of an Often Neglected Disease. J. Diabetes Res. 2019, 2019, 2487804. [Google Scholar] [CrossRef]
- Ewald, N.; Hardt, P.D. Diagnosis and treatment of diabetes mellitus in chronic pancreatitis. World J. Gastroenterol. 2013, 19, 7276–7281. [Google Scholar] [CrossRef]
- Duggan, S.N.; Ewald, N.; Kelleher, L.; Griffin, O.; Gibney, J.; Conlon, K.C. The nutritional management of type 3c (pancreatogenic) diabetes in chronic pancreatitis. Eur. J. Clin. Nutr. 2017, 71, 3–8. [Google Scholar] [CrossRef]
- Makuc, J. Management of pancreatogenic diabetes: Challenges and solutions. Diabetes Metab. Syndr. Obes. 2016, 9, 311–315. [Google Scholar] [CrossRef]
- Gaul, D.A.; Mezencev, R.; Long, T.Q.; Jones, C.M.; Benigno, B.B.; Gray, A.; Fernández, F.M.; McDonald, J.F. Highly-accurate metabolomic detection of early-stage ovarian cancer. Sci. Rep. 2015, 5, 16351. [Google Scholar] [CrossRef]
- Ressom, H.W.; Xiao, J.F.; Tuli, L.; Varghese, R.S.; Zhou, B.; Tsai, T.H.; Ranjbar, M.R.; Zhao, Y.; Wang, J.; Di Poto, C.; et al. Utilization of metabolomics to identify serum biomarkers for hepatocellular carcinoma in patients with liver cirrhosis. Anal. Chim. Acta 2012, 743, 90–100. [Google Scholar] [CrossRef]
- Ha, C.Y.; Kim, J.Y.; Paik, J.K.; Kim, O.Y.; Paik, Y.H.; Lee, E.J.; Lee, J.H. The association of specific metabolites of lipid metabolism with markers of oxidative stress, inflammation and arterial stiffness in men with newly diagnosed type 2 diabetes. Clin. Endocrinol. 2012, 76, 674–682. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.J.; Larson, M.G.; Vasan, R.S.; Cheng, S.; Rhee, E.P.; McCabe, E.; Lewis, G.D.; Fox, C.S.; Jacques, P.F.; Fernandez, C.; et al. Metabolite Profiles and the Risk of Developing Diabetes. Nat. Med. 2011, 17, 448–453. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.; Zheng, X.; Ma, X.; Bao, Y.; Ni, Y.; Hu, C.; Rajani, C.; Huang, F.; Zhao, A.; Jia, W.; et al. Tryptophan Predicts the Risk for Future Type 2 Diabetes. PLoS ONE 2016, 11, e0162192. [Google Scholar] [CrossRef]
- Yu, E.; Papandreou, C.; Ruiz-Canela, M.; Guasch-Ferre, M.; Clish, C.B.; Dennis, C.; Liang, L.; Corella, D.; Fitó, M.; Razquin, C.; et al. Association of Tryptophan Metabolites With Incident Type 2 Diabetes in the PREDIMED Trial: A Case-Cohort Study. Clin. Chem. 2018, 64, 1211–1220. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Zhang, C.; Chen, L.; Han, X.; Ji, L. Human Serum Acylcarnitine Profiles in Different Glucose Tolerance States. Diabetes Res. Clin. Pract. 2014, 104, 376–382. [Google Scholar] [CrossRef]
- Sun, L.; Liang, L.; Gao, X.; Zhang, H.; Yao, P.; Hu, Y.; Ma, Y.; Wang, F.; Jin, Q.; Li, H.; et al. Early Prediction of Developing Type 2 Diabetes by Plasma Acylcarnitines: A Population-Based Study. Diabetes Care 2016, 39, 1563–1570. [Google Scholar] [CrossRef]
- Strand, E.; Rebnord, E.W.; Flygel, M.R.; Lysne, V.; Svingen, G.F.T.; Tell, G.S.; Løland, K.H.; Berge, R.K.; Svardal, A.; Nygård, O.; et al. Serum Carnitine Metabolites and Incident Type 2 Diabetes Mellitus in Patients With Suspected Stable Angina Pectoris. J. Clin. Endocrinol. Metab. 2018, 103, 1033–1041. [Google Scholar] [CrossRef]
- Bouchouirab, F.Z.; Fortin, M.; Noll, C.; Dubé, J.; Carpentier, A.C. Plasma Palmitoyl-Carnitine (AC16:0) Is a Marker of Increased Postprandial Nonesterified Incomplete Fatty Acid Oxidation Rate in Adults with Type 2 Diabetes. Can. J. Diabetes 2018, 42, 382–388. [Google Scholar] [CrossRef]
- Sun, Y.; Gao, H.Y.; Fan, Z.Y.; He, Y.; Yan, Y.X. Metabolomics Signatures in Type 2 Diabetes: A Systematic Review and Integrative Analysis. J. Clin. Endocrinol. Metab. 2020, 105. [Google Scholar] [CrossRef]
- Gordon, D.M.; Adeosun, S.O.; Ngwudike, S.I.; Anderson, C.D.; Hall, J.E.; Hinds, T.D., Jr.; Stec, D.E. CRISPR Cas9-mediated Deletion of Biliverdin Reductase A (BVRA) in Mouse Liver Cells Induces Oxidative Stress and Lipid Accumulation. Arch. Biochem. Biophys. 2019, 672, 108072. [Google Scholar] [CrossRef]
- Takei, R.; Inoue, T.; Sonoda, N.; Kohjima, M.; Okamoto, M.; Sakamoto, R.; Inoguchi, T.; Ogawa, Y. Bilirubin Reduces Visceral Obesity and Insulin Resistance by Suppression of Inflammatory Cytokines. PLoS ONE 2019, 14, e0223302. [Google Scholar] [CrossRef]
- Inoguchi, T.; Sasaki, S.; Kobayashi, K.; Takayanagi, R.; Yamada, T. Relationship between Gilbert Syndrome and Prevalence of Vascular Complications in Patients with Diabetes. JAMA 2007, 298, 1398–1400. [Google Scholar] [CrossRef]
- Nuhn, P.; Mitkus, T.; Ceyhan, G.O.; Künzli, B.M.; Bergmann, F.; Fischer, L.; Giese, N.; Friess, H.; Berberat, P.O. Heme Oxygenase 1-generated Carbon Monoxide and Biliverdin Attenuate the Course of Experimental Necrotizing Pancreatitis. Pancreas 2013, 42, 265–271. [Google Scholar] [CrossRef] [PubMed]
- Chang, M.; Xue, J.; Sharma, V.; Habtezion, A. Protective role of Hemeoxygenase-1 in Gastrointestinal Diseases. Cell. Mol. Life Sci. 2015, 72, 1161–1173. [Google Scholar] [CrossRef]
- Meaney, S.; Babiker, A.; Lütjohann, D.; Diczfalusy, U.; Axelson, M.; Björkhem, I. On the origin of the cholestenoic acids in human circulation. Steroids 2003, 68, 595–601. [Google Scholar] [CrossRef]
- Saeed, A.A.; Edström, E.; Pikuleva, I.; Eggertsen, G.; Björkhem, I. On the importance of albumin binding for the flux of 7α-hydroxy-3-oxo-4-cholestenoic acid in the brain. J. Lipid Res. 2017, 58, 455–459. [Google Scholar] [CrossRef]
- Saeed, A.; Floris, F.; Andersson, U.; Pikuleva, I.; Lövgren-Sandblom, A.; Bjerke, M.; Paucar, M.; Wallin, A.; Svenningsson, P.; Björkhem, I. 7α-hydroxy-3-oxo-4-cholestenoic acid in cerebrospinal fluid reflects the integrity of the blood-brain barrier. J. Lipid Res. 2014, 55, 313–318. [Google Scholar] [CrossRef]
- Hoffmeister, T.; Kaiser, J.; Lüdtke, S.; Drews, G.; Düfer, M. Interactions between Atorvastatin and the Farnesoid X Receptor Impair Insulinotropic Effects of Bile Acids and Modulate Diabetogenic Risk. Mol. Pharmacol. 2020, 97, 202–211. [Google Scholar] [CrossRef]
| m/z | RT | Adduct | MF | ppm | FDR | FC | AUC | Tentative Identification |
|---|---|---|---|---|---|---|---|---|
| 188.0716 | 3.3 | [M+H-NH3]+ | C11H9NO2 | −2 | 7.86 × 10−3 | 2.9319 | 0.93 | L-Tryptophan |
| 400.3408 | 9.9 | [M+H]+ | C23H45NO4 | 3 | 2.75 × 10−2 | 2.62 | 0.81 | Palmitoylcarnitine |
| 431.3140 | 11.85 | [M+H+ | C27H42O4 | 4 | 1.39 × 10−2 | 3.5449 | 0.80 | 7-HOCA |
| 526.2931 | 10.6 | [M+H]+ | C27H44NO7P | 1 | 7.86 × 10−3 | 2.3703 | 0.71 | LysoPE(22:6) |
| 583.255 | 8.2 | [M+H]+ | C33H34N4O6 | 0 | 2.75 × 10−2 | 2.9319 | 0.82 | Biliverdin |
| T3cDM | T2DM | |
|---|---|---|
| n | 21 | 19 |
| Age, mean years (±SD) | 58.42 (±9.57) | 55.96 (±6.16) |
| Sex | ||
| Male | 21 | 19 |
| Female | 0 | 0 |
| Caucasian | 21 | 19 |
| Stage | ||
| A | 0 | - |
| B | 0 | - |
| C | 21 | - |
| Disease duration, years | 5.36 | 7.14 |
| HbA1c, % | 7.82 | 7.69 |
| BMI, mean (±SD) | 21.4 (±6.61) | 24.3 (±3.54) |
| Fasting glucose, mg/dL | 144.36 | 138.42 |
| Insulin treatment | 21 | 19 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jimenez-Luna, C.; Martin-Blazquez, A.; Dieguez-Castillo, C.; Diaz, C.; Martin-Ruiz, J.L.; Genilloud, O.; Vicente, F.; del Palacio, J.P.; Prados, J.; Caba, O. Novel Biomarkers to Distinguish between Type 3c and Type 2 Diabetes Mellitus by Untargeted Metabolomics. Metabolites 2020, 10, 423. https://doi.org/10.3390/metabo10110423
Jimenez-Luna C, Martin-Blazquez A, Dieguez-Castillo C, Diaz C, Martin-Ruiz JL, Genilloud O, Vicente F, del Palacio JP, Prados J, Caba O. Novel Biomarkers to Distinguish between Type 3c and Type 2 Diabetes Mellitus by Untargeted Metabolomics. Metabolites. 2020; 10(11):423. https://doi.org/10.3390/metabo10110423
Chicago/Turabian StyleJimenez-Luna, Cristina, Ariadna Martin-Blazquez, Carmelo Dieguez-Castillo, Caridad Diaz, Jose Luis Martin-Ruiz, Olga Genilloud, Francisca Vicente, Jose Perez del Palacio, Jose Prados, and Octavio Caba. 2020. "Novel Biomarkers to Distinguish between Type 3c and Type 2 Diabetes Mellitus by Untargeted Metabolomics" Metabolites 10, no. 11: 423. https://doi.org/10.3390/metabo10110423
APA StyleJimenez-Luna, C., Martin-Blazquez, A., Dieguez-Castillo, C., Diaz, C., Martin-Ruiz, J. L., Genilloud, O., Vicente, F., del Palacio, J. P., Prados, J., & Caba, O. (2020). Novel Biomarkers to Distinguish between Type 3c and Type 2 Diabetes Mellitus by Untargeted Metabolomics. Metabolites, 10(11), 423. https://doi.org/10.3390/metabo10110423





