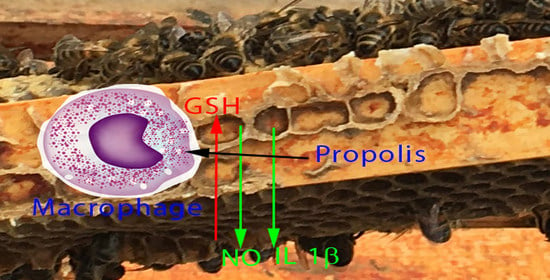
Temperate Propolis Has Anti-Inflammatory Effects and Is a Potent Inhibitor of Nitric Oxide Formation in Macrophages
Abstract
:1. Introduction
2. Results
2.1. The Effect of Propolis on NO Production and Cytokine Production by Macrophages
2.2. Propolis Does Not Affect Cell Viability
2.3. The Effect of Propolis on the Metabolomic Profile of Macrophages
2.3.1. Propolis Treatment Inhibits NO Formation
2.3.2. Propolis Treatment May Promote Energy Metabolism and Stimulate the Formation of High Energy Phosphates
2.3.3. Propolis Treatment Stimulates Formation of Amino Sugars
2.3.4. Propolis Treatment Lowers Citrate and Increases Itaconate
2.4. Propolis Treatment Increases GSH Levels
2.5. Propolis Treatment Increases Fatty Acid Metabolism
3. Discussion
4. Materials and Methods
4.1. Chemicals and Reagents
4.2. Preparation of Propolis Extracts
4.3. Generation of Bone Marrow-Derived Macrophages (BMMs)
4.4. Flow Cytometry
4.5. Effect of Propolis Treatment on LPS-Activated Macrophages
4.6. Measurement of Cell Viability
4.7. Measurement of NO Production in BMMs
4.8. Cytokine Assays
4.9. Liquid Chromatography/Mass Spectroscopy (LC/MS)
4.10. Metabolomic Data Analysis
4.11. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Bankova, V.; Popova, M.; Trusheva, B. The phytochemistry of the honeybee. Phytochemistry 2018, 155, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Wilson, M.B.; Spivak, M.; Hegeman, A.D.; Rendahl, A.; Cohen, J.D. Metabolomics reveals the origins of antimicrobial plant resins collected by honey bees. PLoS ONE 2013, 8, e77512. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bueno-Silva, B.; Kawamoto, D.; Ando-Suguimoto, E.S.; Alencar, S.M.; Rosalen, P.L.; Mayer, M.P. Brazilian red propolis attenuates inflammatory signaling cascade in LPS-activated macrophages. PLoS ONE 2015, 10, e0144954. [Google Scholar] [CrossRef] [PubMed]
- Bueno-Silva, B.; Kawamoto, D.; Ando-Suguimoto, E.S.; Casarin, R.C.; Alencar, S.M.; Rosalen, P.L.; Mayer, M.P. Brazilian red propolis effects on peritoneal macrophage activity: Nitric oxide, cell viability, pro-inflammatory cytokines and gene expression. J. Ethnopharmacol. 2017, 207, 100–107. [Google Scholar] [CrossRef]
- Comalada, M.; Ballester, I.; Bailon, E.; Sierra, S.; Xaus, J.; Gálvez, J.; de Medina, F.S.; Zarzuelo, A. Inhibition of pro-inflammatory markers in primary bone marrow-derived mouse macrophages by naturally occurring flavonoids: Analysis of the structure–activity relationship. Biochem. Pharmacol. 2006, 72, 1010–1021. [Google Scholar] [CrossRef]
- Touri, K.; Belguendouz, H.; Medjeber, O.; Djeraba, Z.; Lahmar, K.; Touil-Boukoffa, C. Propolis modulates NOS2/arginase-1 pathway in tropomyosin-induced experimental autoimmune uveitis. Inflammopharmacology 2018, 26, 1293–1303. [Google Scholar] [CrossRef]
- Siheri, W.; Alenezi, S.; Tusiimire, J.; Watson, D.G. The chemical and biological properties of propolis. In Bee Products-Chemical and Biological Properties; Springer: Berlin/Heidelberg, Germany, 2017; pp. 137–178. [Google Scholar]
- Alqarni, A.M.; Niwasabutra, K.; Sahlan, M.; Fearnley, H.; Fearnley, J.; Ferro, V.A.; Watson, D.G. Propolis exerts an anti-inflammatory effect on pma-differentiated thp-1 cells via inhibition of purine nucleoside phosphorylase. Metabolites 2019, 9, 75. [Google Scholar] [CrossRef] [Green Version]
- Zakerkish, M.; Jenabi, M.; Zaeemzadeh, N.; Hemmati, A.A.; Neisi, N. The effect of Iranian propolis on glucose metabolism, lipid profile, insulin resistance, renal function and inflammatory biomarkers in patients with type 2 diabetes mellitus: A randomized double-blind clinical trial. Sci. Rep. 2019, 9, 7289. [Google Scholar] [CrossRef]
- Marchisio, P.; Esposito, S.; Bianchini, S.; Desantis, C.; Galeone, C.; Nazzari, E.; Pignataro, L.; Principi, N. Effectiveness of a propolis and zinc solution in preventing acute otitis media in children with a history of recurrent acute otitis media. Int. J. Immunopathol. Pharmacol. 2010, 23, 567–575. [Google Scholar] [CrossRef]
- Cohen, H.A.; Varsano, I.; Kahan, E.; Sarrell, E.M.; Uziel, Y. Effectiveness of an herbal preparation containing echinacea, propolis, and vitamin C in preventing respiratory tract infections in children: A randomized, double-blind, placebo-controlled, multicenter study. Arch. Pediatrics Adolesc. Med. 2004, 158, 217–221. [Google Scholar] [CrossRef] [Green Version]
- Zonouz, A.T.; Mehdipour, M.; Abadi, R.T.A.; Shokri, J.; Rajaee, M.; Aghazadeh, M. Effect of the use of propolis on serum levels of interleukin-17 and clinical symptoms and signs in patients with ulcerative oral lichen planus. Adv. Nat. Appl. Sci. 2015, 9, 39–45. [Google Scholar]
- Brätter, C.; Tregel, M.; Liebenthal, C.; Volk, H. Prophylactic effectiveness of propolis for immunostimulation: A clinical pilot study. Forsch. Komplement. 1999, 6, 256–260. [Google Scholar]
- Mills, E.L.; Kelly, B.; Logan, A.; Costa, A.S.; Varma, M.; Bryant, C.E.; Tourlomousis, P.; Däbritz, J.H.M.; Gottlieb, E.; Latorre, I. Succinate dehydrogenase supports metabolic repurposing of mitochondria to drive inflammatory macrophages. Cell 2016, 167, 457–470. e13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mills, E.L.; O’Neill, L.A. Reprogramming mitochondrial metabolism in macrophages as an anti-inflammatory signal. Eur. J. Immunol. 2016, 46, 13–21. [Google Scholar] [CrossRef] [PubMed]
- O’Neill, L.A. A broken Krebs cycle in macrophages. Immunity 2015, 42, 393–394. [Google Scholar] [CrossRef] [Green Version]
- O’Neill, L.A.; Pearce, E.J. Immunometabolism governs dendritic cell and macrophage function. J. Exp. Med. 2016, 213, 15–23. [Google Scholar] [CrossRef] [PubMed]
- Palsson-McDermott, E.M.; Curtis, A.M.; Goel, G.; Lauterbach, M.A.; Sheedy, F.J.; Gleeson, L.E.; van den Bosch, M.W.; Quinn, S.R.; Domingo-Fernandez, R.; Johnston, D.G. Pyruvate kinase M2 regulates Hif-1α activity and IL-1β induction and is a critical determinant of the Warburg effect in LPS-activated macrophages. Cell Metab. 2015, 21, 65–80. [Google Scholar] [CrossRef] [Green Version]
- Van den Bossche, J.; O’Neill, L.A.; Menon, D. Macrophage immunometabolism: Where are we (going)? Trends Immunol. 2017, 38, 395–406. [Google Scholar] [CrossRef]
- Viola, A.; Munari, F.; Sánchez-Rodríguez, R.; Scolaro, T.; Castegna, A. The metabolic signature of macrophage responses. Front. Immunol. 2019, 10, 1462. [Google Scholar] [CrossRef] [Green Version]
- Szabó, C.; Ischiropoulos, H.; Radi, R. Peroxynitrite: Biochemistry, pathophysiology and development of therapeutics. Nat. Rev. Drug Discov. 2007, 6, 662–680. [Google Scholar] [CrossRef]
- He, C.; Carter, A.B. The Metabolic Prospective and Redox Regulation of Macrophage Polarization. J. Clin. Cell. Immunol. 2015, 6, 371. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- O’Neill, L.A.J.; Artyomov, M.N. Itaconate: The poster child of metabolic reprogramming in macrophage function. Nat. Rev. Immunol. 2019, 19, 273–281. [Google Scholar] [CrossRef] [PubMed]
- Saleh, K.; Zhang, T.; Fearnley, J.; Watson, D.G. A Comparison of the Constituents of Propolis from Different Regions of the United Kingdom by Liquid Chromatography-high Resolution Mass Spectrometry Using a Metabolomics Approach. Curr. Metab. 2015, 3, 42–53. [Google Scholar] [CrossRef]
- Liang, Y.-C.; Tsai, S.-H.; Tsai, D.-C.; Lin-Shiau, S.-Y.; Lin, J.-K. Suppression of inducible cyclooxygenase and nitric oxide synthase through activation of peroxisome proliferator-activated receptor-γ by flavonoids in mouse macrophages. FEBS Lett. 2001, 496, 12–18. [Google Scholar] [CrossRef] [Green Version]
- Change, E.Y.; Guo, B.; Doyle, S.E.; Cheng, G. Cutting Edge: Involvement of the Type I IFN Production and Signaling Pathway in Lipopolysaccharide-Induced IL-10 Production. J. Immunol. 2007, 178, 6705–6709. [Google Scholar] [CrossRef] [Green Version]
- Beisswenger, P.J.; Howell, S.K.; Smith, K.; Szwergold, B.S. Glyceraldehyde-3-phosphate dehydrogenase activity as an independent modifier of methylglyoxal levels in diabetes. Biochim. Biophys. Acta Mol. Basis Dis. 2003, 1637, 98–106. [Google Scholar] [CrossRef] [Green Version]
- Allaman, I.; Bélanger, M.; Magistretti, P.J. Methylglyoxal, the dark side of glycolysis. Front. Neurosci. 2015, 9, 23. [Google Scholar] [CrossRef] [Green Version]
- Lai, Y.-L.; Aoyama, S.; Nagai, R.; Miyoshi, N.; Ohshima, H. Inhibition of L-arginine metabolizing enzymes by L-arginine-derived advanced glycation end products. J. Clin. Biochem. Nutr. 2010, 46, 177–185. [Google Scholar] [CrossRef] [Green Version]
- Palmieri, E.M.; Spera, I.; Menga, A.; Infantino, V.; Porcelli, V.; Iacobazzi, V.; Pierri, C.L.; Hooper, D.C.; Palmieri, F.; Castegna, A. Acetylation of human mitochondrial citrate carrier modulates mitochondrial citrate/malate exchange activity to sustain NADPH production during macrophage activation. Biochim. Biophys. Acta Bioenergy 2015, 187, 729–738. [Google Scholar] [CrossRef] [Green Version]
- Williard, D.E.; Kaduce, T.L.; Harmon, S.D.; Spector, A.A. Conversion of eicosapentaenoic acid to chain-shortened omega–3 fatty acid metabolites by peroxisomal oxidation. J. Lipid Res. 1998, 39, 978–986. [Google Scholar]
- Ricote, M.; Li, A.C.; Willson, T.M.; Kelly, C.J.; Glass, C.K. The peroxisome proliferator-activated receptor-γ is a negative regulator of macrophage activation. Nature 1998, 391, 79–82. [Google Scholar] [CrossRef] [PubMed]
- Mueller, M.; Lukas, B.; Novak, J.; Simoncini, T.; Genazzani, A.R.; Jungbauer, A. Oregano: A source for peroxisome proliferator-activated receptor γ antagonists. J. Agric. Food Chem. 2008, 56, 11621–11630. [Google Scholar] [CrossRef] [PubMed]
- Odegaard, J.I.; Ricardo-Gonzalez, R.R.; Goforth, M.H.; Morel, C.R.; Subramanian, V.; Mukundan, L.; Eagle, A.R.; Vats, D.; Brombacher, F.; Ferrante, A.W. Macrophage-specific PPARγ controls alternative activation and improves insulin resistance. Nature 2007, 447, 1116–1120. [Google Scholar] [CrossRef] [Green Version]
- Nomura, M.; Liu, J.; Rovira, I.I.; Gonzalez-Hurtado, E.; Lee, J.; Wolfgang, M.J.; Finkel, T. Fatty acid oxidation in macrophage polarization. Nat. Immunol. 2016, 17, 216–217. [Google Scholar] [CrossRef] [PubMed]
- De Groot, A.C.; Popova, M.P.; Bankova, V.S. An Update on the Constituents of Poplar-Type Propolis; Acdegroot Publishing: Wapserveen, The Netherlands, 2014. [Google Scholar]
- Hendriks, J.J.; de Vries, H.E.; van der Pol, S.M.; van den Berg, T.K.; van Tol, E.A.; Dijkstra, C.D. Flavonoids inhibit myelin phagocytosis by macrophages; A structure-activity relationship study. Biochem. Pharmacol. 2003, 65, 877–885. [Google Scholar] [CrossRef]
- Kim, H.K.; Cheon, B.S.; Kim, Y.H.; Kim, S.Y.; Kim, H.P. Effects of naturally occurring flavonoids on nitric oxide production in the macrophage cell line RAW 264.7 and their structure-activity relationships. Biochem. Pharmacol. 1999, 58, 759–765. [Google Scholar] [CrossRef]
- Matsuda, H.; Morikawa, T.; Ando, S.; Toguchida, I.; Yoshikawa, M. Structural requirements of flavonoids for nitric oxide production inhibitory activity and mechanism of action. Bioorg. Med. Chem. 2003, 11, 1995–2000. [Google Scholar] [CrossRef]
- Al-Riyami, L.; Pineda, M.A.; Rzepecka, J.; Huggan, J.K.; Khalaf, A.I.; Suckling, C.J.; Scott, F.J.; Rodgers, D.T.; Harnett, M.M.; Harnett, W. Designing anti-inflammatory drugs from parasitic worms: A synthetic small molecule analogue of the Acanthocheilonema viteae product ES-62 prevents development of collagen-induced arthritis. J. Med. Chem. 2013, 56, 9982–10002. [Google Scholar] [CrossRef]
- Griess, P. Griess reagent: A solution of sulphanilic acid and α-naphthylamine in acetic acid which gives a pink colour on reaction with the solution obtained after decomposition of nitrosyl complexes. Chem. Ber. 1879, 12, 427. [Google Scholar]
- Pluskal, T.; Castillo, S.; Villar-Briones, A.; Oresic, M. MZmine 2: Modular framework for processing, visualizing, and analyzing mass spectrometry-based molecular profile data. BMC Bioinform. 2010, 11, 395. [Google Scholar] [CrossRef] [Green Version]
- Howe, C.C.; Alshehri, A.; Muggeridge, D.; Mullen, A.B.; Boyd, M.; Spendiff, O.; Moir, H.J.; Watson, D.G. Untargeted metabolomics profiling of an 80.5 km simulated treadmill ultramarathon. Metabolites 2018, 8, 14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sumner, L.W.; Amberg, A.; Barrett, D.; Beale, M.H.; Beger, R.; Daykin, C.A.; Fan, T.W.-M.; Fiehn, O.; Goodacre, R.; Griffin, J.L. Proposed minimum reporting standards for chemical analysis. Metabolomics 2007, 3, 211–221. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chong, J.; Wishart, D.S.; Xia, J. Using Metaboanalyst 4.0 for Comprehensive Integrative Metabolomics Data. Curr. Protoc. Bioinform. 2019, 68, e86. [Google Scholar] [CrossRef] [PubMed]
- Saelao, P.; Borba, R.S.; Ricigliano, V.; Spivak, M.; Simone-Finstrom, M. Honeybee microbiome is stabilized in the presence of propolis. Biol. Lett. 2020, 16, 20200003. [Google Scholar] [CrossRef] [PubMed]
- Alotaibi, A.; Ebiloma, G.U.; Williams, R.; Alenezi, S.; Donachie, A.-M.; Guillaume, S.; Igoli, J.O.; Fearnley, J.; De Koning, H.P.; Watson, D.G. European propolis is highly active against trypanosomatids including crithidia fasciculata. Sci. Rep. 2019, 9, 11364. [Google Scholar] [CrossRef] [Green Version]
- Al Rofaidi, M.; Alotaibi, A.; Aqarni, A.; Alghamdi, A.; Fearnley, J.; Watson, D.G. A Preliminary study of the absorption of and metabolism of temperate propolis by human subjects. J. Food Nutr. Metab. 2020, 3, 1–6. [Google Scholar]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |












| Anova Tukey’s HSD | m/z | Rt min | Metabolite | P LM | L/M | P224L | 224/L | P225L | 225/L |
|---|---|---|---|---|---|---|---|---|---|
| Nitric oxide biosynthesis | |||||||||
| 4.8 × 10−14 ¥ | 176.103 | 16.0 | l-Citrulline * | 0.001 | 31.765 | 0.002 | 0.199 | 0.009 | 0.432 |
| 1.8 × 10−18 ¥ | 191.114 | 22.5 | N-(Omega)-Hydroxyarginine | 0.001 | 18030.784 | 0.002 | 0.056 | 0.002 | 0.157 |
| 1.0 × 10−14 ¥ | 247.140 | 14.3 | N2-(d-1-Carboxyethyl)-l-Arginine | 0.002 | 4.929 | <0.001 | 6.081 | <0.001 | 10.833 |
| 3.3 × 10−15 ¥ | 291.129 | 16.8 | N-(l-Arginino) Succinate | 0.002 | 24.687 | 0.002 | 3.091 | 0.001 | 5.223 |
| Glycolysis/TCA cycle | |||||||||
| 1.9 × 10−8 ¥ | 129.018 | 14.9 | Itaconic Acid * | 0.000 | 125.289 | 0.002 | 4.069 | 0.001 | 5.428 |
| 8.0 × 10−8 ¥ | 168.990 | 16.0 | Glyceraldehyde 3-Phosphate * | 0.003 | 150.353 | 0.001 | 10.092 | 0.001 | 12.626 |
| 1.3 × 10−7 ¥ | 173.021 | 15.7 | Sn-Glycerol 3-Phosphate * | 0.002 | 12.756 | 0.005 | 0.289 | 0.006 | 0.331 |
| 1.4 × 10−6 ¥ | 179.0552 | 14.7 | d-Glucose * | <0.001 | 0.429 | 0.002 | 0.268 | <0.001 | 0.094 |
| 3.0 × 10−16 ¥ | 191.0189 | 18.1 | Citrate * | <0.001 | >1000 | <0.001 | 0.024 | <0.001 | 0.017 |
| 3.7 × 10−9 ¥ | 666.131 | 13.3 | NADH * | 0.009 | 2.221 | 0.001 | 2.477 | 0.001 | 3.351 |
| 6.1 × 10−7 # | 810.131 | 12.3 | Acetyl-CoA | 0.004 | 4.363 | 0.480 | 1.213 | <0.001 | 2.257 |
| Oxidative stress/glutathione metabolism | |||||||||
| 1.3 × 10−13 ¥ | 76.040 | 15.9 | Glycine * | 0.005 | 2.367 | <0.001 | 7.222 | <0.001 | 8.452 |
| 6.5 × 10−12 ¥ | 122.027 | 14.1 | l-Cysteine * | 0.027 | 20.174 | 0.001 | 14.423 | <0.001 | 26.437 |
| 4.8 × 10−9 ¥ | 241.031 | 16.1 | l-Cystine * | 0.009 | 0.492 | 0.005 | 0.108 | 0.005 | 0.159 |
| 6.1 × 10−17 ¥ | 251.069 | 14.1 | Gamma-l-Glutamyl-l-Cysteine | 0.001 | 14.204 | <0.001 | 8.063 | 0.001 | 14.927 |
| 6.4 × 10−11 ¥ | 148.060 | 14.6 | l-Glutamate * | 0.012 | 0.729 | <0.001 | 3.126 | <0.001 | 4.074 |
| 1.8 × 10−16 ¥ | 308.090 | 14.2 | Glutathione * | 0.000 | 3.903 | 0.001 | 6.268 | <0.001 | 8.205 |
| 1.6 × 10−11 ¥ | 336.087 | 14.6 | S-Formylglutathione | <0.001 | 4.273 | <0.001 | 0.536 | <0.001 | 0.518 |
| 5.3 × 10−15 ¥ | 338.0488 | 17.3 | S-Sulfanylglutathione | 0.080 | >1000 | 0.006 | 326.271 | 0.005 | 320.140 |
| 1.8 × 10−22 ¥ | 380.111 | 12.1 | S-Lactoylglutathione * | 0.001 | >1000 | <0.001 | 7.408 | <0.001 | 10.455 |
| 2.4 × 10−18 ¥ | 613.159 | 17.4 | Glutathione Disulphide * | 0.001 | 5.952 | <0.001 | 24.600 | <0.001 | 20.853 |
| High-energy phosphate metabolism | |||||||||
| 1.1 × 10−10 ¥ | 212.043 | 15.1 | Phosphocreatine * | 0.001 | 2.157 | 0.006 | 1.768 | 0.002 | 2.739 |
| 4.5 × 10−10 ¥ | 348.070 | 16.6 | AMP * | 0.035 | 4.217 | 0.007 | 3.455 | 0.007 | 5.228 |
| 1.8 × 10−10 ¥ | 322.0446 | 16.0 | CMP * | 0.002 | 1.795 | 0.001 | 2.107 | <0.001 | 2.095 |
| 1.9 × 10−11 ¥ | 489.114 | 15.3 | CDP–Choline * | 0.001 | 22.575 | 0.001 | 0.142 | 0.001 | 0.191 |
| 9.6 × 10−13 ¥ | 484.975 | 17.9 | UTP * | 0.001 | 0.534 | <0.001 | 7.534 | <0.001 | 11.550 |
| 8.2 × 10−11 ¥ | 508.002 | 16.6 | ATP * | 0.002 | 1.987 | 0.001 | 2.006 | 0.001 | 2.416 |
| 5.7 × 10−10 ¥ | 523.997 | 19.3 | GTP * | <0.001 | 7.584 | 0.060 | 1.485 | 0.002 | 2.214 |
| 4.8 × 10−10 § | 565.0477 | 16.1 | UDP–Glucose * | 0.897 | 1.058 | 0.002 | 24.657 | 0.007 | 50.760 |
| 8.4 × 10−10 ¥ | 606.0744 | 15.0 | UDP–N-Acetyl-d-glucosamine * | 0.107 | 3.312 | 0.008 | 33.266 | 0.016 | 46.857 |
| Fatty acid metabolism | |||||||||
| 3.5 × 10−9 § | 185.117 | 3.9 | 10-Oxodecanoate | 0.318 | 0.702 | <0.001 | 100.192 | 0.001 | 90.767 |
| 3.0 × 10−9 ¥ | 204.123 | 11.1 | O-Acetylcarnitine * | <0.001 | 4.658 | <0.001 | 0.002 | 0.490 | 0.002 |
| 1.7 × 10−7 € | 218.138 | 9.9 | O-Propanoylcarnitine | <0.001 | 0.321 | <0.001 | 2.871 | <0.001 | 2.967 |
| 1.9 × 10−9 § | 227.201 | 4.2 | Tetradecanoic Acid | <0.001 | 2.432 | 0.004 | 1.413 | <0.001 | 1.464 |
| 9.2 × 10−10 § | 225.186 | 4.2 | Tetradecenoic Acid | 0.126 | 2.130 | 0.025 | 2.131 | <0.001 | 5.490 |
| 3.5 × 10−14 § | 269.212 | 4.2 | Oxo-Hexadecanoic Acid | 0.323 | 1.305 | 0.003 | 38.013 | <0.001 | 257.413 |
| 3.4 × 10−10 § | 271.228 | 3.8 | Hydroxypalmitate | 0.193 | 0.610 | <0.001 | 129.447 | <0.001 | 132.527 |
| 1.7 × 10−9 § | 279.231 | 4.0 | Linoleate | 0.350 | 0.340 | <0.001 | 20.934 | <0.001 | 22.371 |
| 4.1 × 10−8 § | 283.264 | 3.8 | Octadecanoic Acid | <0.001 | 1.779 | <0.001 | 0.589 | <0.001 | 0.537 |
| 1.7 × 10−9 § | 295.227 | 3.8 | Hydroxyoctadecadienoic Acid | 0.293 | 1.227 | 0.008 | 41.194 | <0.001 | 102.576 |
| 1.4 × 10−7 § | 299.259 | 3.9 | Hydroxyoctadecanoic Acid | 0.829 | 0.937 | 0.020 | 29.732 | <0.001 | 68.922 |
| 1.7 × 10−6 § | 301.217 | 4.0 | Eicosapentaenoic Acid | 0.001 | 1.822 | 0.001 | 0.585 | 0.001 | 0.583 |
| 1.7 × 10−10 § | 303.233 | 4.0 | Eicosatetraenoic Acid | 0.267 | 0.938 | <0.001 | 0.511 | <0.001 | 0.509 |
| 3.8 × 10−8 § | 309.280 | 3.7 | Eicosenoic Acid | <0.001 | 3.417 | 0.006 | 0.699 | 0.001 | 0.660 |
| 1.2 × 10−9 § | 327.233 | 3.7 | Docosahexaenoic Acid | 0.001 | 1.905 | <0.001 | 0.336 | <0.001 | 0.395 |
| 7.8 × 10−9 § | 333.280 | 3.7 | Docosatrienoic Acid | <0.001 | 2.118 | <0.001 | 0.485 | <0.001 | 0.468 |
| 2.2 × 10−8 # | 386.289 | 4.9 | Hydroxytetradecenoylcarnitine | 0.002 | 9.282 | 0.107 | 1.459 | <0.001 | 6.815 |
| 7.1 × 10−9 § | 414.320 | 4.9 | Hydroxyhexadecenoylcarnitine | 0.002 | 29.177 | 0.014 | 2.715 | <0.001 | 4.851 |
| 4.0 × 10−10 § | 428.373 | 4.6 | Stearoylcarnitine | 0.005 | 1.898 | 0.001 | 2.351 | 0.003 | 1.699 |
| 4.3 × 10−22 § | 442.352 | 7.6 | Hydroxyoctadecenoylcarnitine | 0.001 | >1000 | <0.001 | 13.029 | <0.001 | 11.175 |
| Phospholipid biosynthesis | |||||||||
| 1.4 × 10−3 € | 104.107 | 14.5 | Choline * | <0.001 | 4.826 | <0.001 | 0.407 | <0.001 | 0.379 |
| 8.8 × 10−13 ¥ | 184.073 | 15.0 | Choline Phosphate * | 0.001 | 2.530 | <0.001 | 2.827 | 0.001 | 2.582 |
| 2.0 × 10−9 ¥ | 258.110 | 14.5 | Sn-Glycero-3-Phosphocholine * | <0.001 | 5.060 | 0.001 | 0.427 | 0.001 | 0.400 |
| 1.9 × 10−11 ¥ | 568.339 | 4.6 | LPC 22:6 | 0.009 | 2.501 | <0.001 | 3.014 | <0.001 | 2.920 |
| 5.0 × 10−8 € | 810.526 | 3.8 | PS 38:5 | <0.001 | 3.432 | <0.001 | 0.186 | <0.001 | 0.291 |
| 6.0 × 10−5 ¥ | 820.619 | 4.1 | PC40:5 | 0.034 | 1.269 | 0.002 | 0.618 | 0.001 | 0.572 |
| Purines and pyrimidines | |||||||||
| 1.1 × 10−13 § | 136.062 | 9.7 | Adenine * | <0.001 | 4.250 | <0.001 | 1.708 | <0.001 | 2.160 |
| 1.5 × 10−10 § | 137.046 | 8.3 | Hypoxanthine * | 0.002 | 2.386 | 0.002 | 4.181 | <0.001 | 6.198 |
| 3.4 × 10−8 ¥ | 243.062 | 12.0 | Uridine * | 0.624 | 1.141 | <0.001 | 3.804 | <0.001 | 4.458 |
| 8.6 × 10−11 § | 251.0784 | 9.6 | Deoxyinosine | 0.072 | 2.383 | 0.007 | 21.680 | <0.001 | 35.912 |
| 5.3 × 10−10 § | 284.098 | 12.7 | Guanosine * | 0.046 | 13.235 | 0.001 | 23.082 | <0.001 | 29.869 |
| 8.3 × 10−09 ¥ | 384.114 | 14.4 | Succinyladenosine | 0.050 | >1000 | 0.006 | 3.782 | 0.006 | 2.794 |
| Aminosugars | |||||||||
| 1.5 × 10−7 € | 180.086 | 18.1 | d-Glucosamine * | <0.001 | 6.336 | <0.001 | 0.210 | <0.001 | 0.148 |
| 1.2 × 10−9 ¥ | 222.098 | 12.7 | N-Acetyl-d-Glucosamine * | 0.018 | 21.617 | <0.001 | 5.012 | 0.003 | 6.840 |
| 4.6 × 10−14 § | 266.0895 | 11.2 | Neuraminic Acid | 0.164 | 2.320 | <0.001 | 62.644 | <0.001 | 97.751 |
| 8.8 × 10−15 ¥ | 300.0489 | 15.0 | N-Acetyl-d-Glucosamine 6-Phosphate * | 0.003 | 0.428 | <0.001 | 27.336 | <0.001 | 26.704 |
| 3.2 × 10−6 § | 310.113 | 13.2 | N-Acetylneuraminate | 0.013 | 2.197 | 0.002 | 0.170 | 0.004 | 0.297 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alanazi, S.; Alenzi, N.; Fearnley, J.; Harnett, W.; Watson, D.G. Temperate Propolis Has Anti-Inflammatory Effects and Is a Potent Inhibitor of Nitric Oxide Formation in Macrophages. Metabolites 2020, 10, 413. https://doi.org/10.3390/metabo10100413
Alanazi S, Alenzi N, Fearnley J, Harnett W, Watson DG. Temperate Propolis Has Anti-Inflammatory Effects and Is a Potent Inhibitor of Nitric Oxide Formation in Macrophages. Metabolites. 2020; 10(10):413. https://doi.org/10.3390/metabo10100413
Chicago/Turabian StyleAlanazi, Samyah, Naif Alenzi, James Fearnley, William Harnett, and David G. Watson. 2020. "Temperate Propolis Has Anti-Inflammatory Effects and Is a Potent Inhibitor of Nitric Oxide Formation in Macrophages" Metabolites 10, no. 10: 413. https://doi.org/10.3390/metabo10100413
APA StyleAlanazi, S., Alenzi, N., Fearnley, J., Harnett, W., & Watson, D. G. (2020). Temperate Propolis Has Anti-Inflammatory Effects and Is a Potent Inhibitor of Nitric Oxide Formation in Macrophages. Metabolites, 10(10), 413. https://doi.org/10.3390/metabo10100413