Identification and Reproducibility of Plasma Metabolomic Biomarkers of Habitual Food Intake in a US Diet Validation Study
Abstract
1. Introduction
2. Results
2.1. Participant Characteristics
2.2. Fasting Plasma Metabolites Correlated with Habitual Food Intake Assessed by FFQ and 24-h Diet Recalls (24HRs)
2.2.1. Fruits
2.2.2. Vegetables
2.2.3. Grains
2.2.4. Proteins
2.2.5. Dairy/Dairy Alternatives
2.2.6. Fats and Oils
2.2.7. Alcohol
2.2.8. Beverages
2.2.9. Miscellaneous
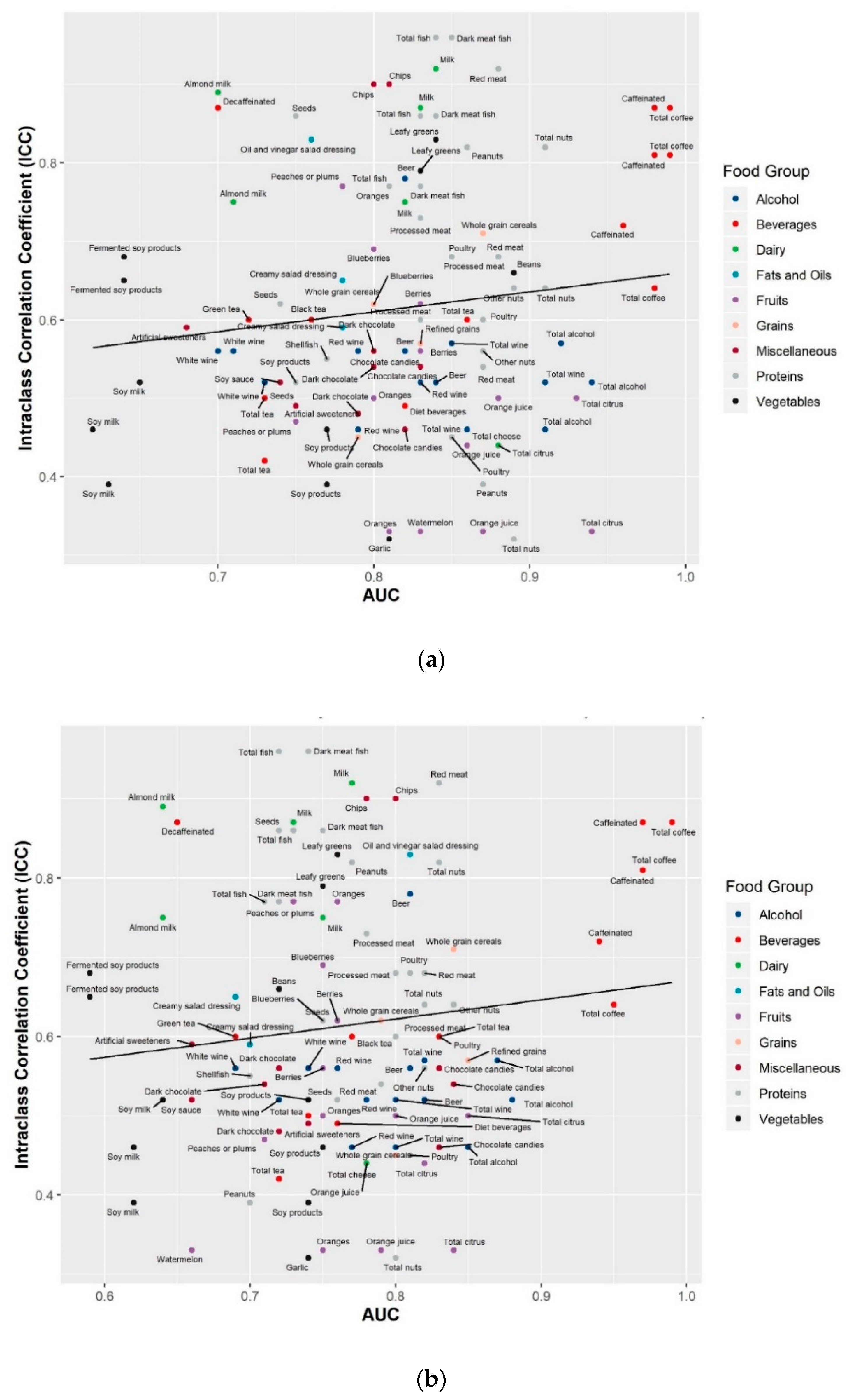
2.3. Reproducibility of the Identified Food Metabolites
3. Discussion
4. Materials and Methods
4.1. Study Population
4.2. Diet Assessment
4.3. Blood Collection and Processing
4.4. Metabolomics Analysis
4.5. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Brennan, L.; Hu, F.B. Metabolomics-Based Dietary Biomarkers in Nutritional Epidemiology-Current Status and Future Opportunities. Mol. Nutr. Food Res. 2019, 63, e1701064. [Google Scholar] [CrossRef] [PubMed]
- Bingham, S.A. Urine nitrogen as a biomarker for the validation of dietary protein intake. J. Nutr. 2003, 133, 921S–924S. [Google Scholar] [CrossRef] [PubMed]
- Tasevska, N.; Runswick, S.A.; Bingham, S.A. Urinary potassium is as reliable as urinary nitrogen for use as a recovery biomarker in dietary studies of free living individuals. J. Nutr. 2006, 136, 1334–1340. [Google Scholar] [CrossRef] [PubMed]
- Clark, A.J.; Mossholder, S. Sodium and potassium intake measurements: Dietary methodology problems. Am. J. Clin. Nutr. 1986, 43, 470–476. [Google Scholar] [CrossRef]
- Campbell, D.R.; Gross, M.D.; Martini, M.C.; Grandits, G.A.; Slavin, J.L.; Potter, J.D. Plasma carotenoids as biomarkers of vegetable and fruit intake. Cancer Epidemiol. Biomark. Prev. 1994, 3, 493–500. [Google Scholar]
- Tasevska, N.; Midthune, D.; Potischman, N.; Subar, A.F.; Cross, A.J.; Bingham, S.A.; Schatzkin, A.; Kipnis, V. Use of the predictive sugars biomarker to evaluate self-reported total sugars intake in the Observing Protein and Energy Nutrition (OPEN) study. Cancer Epidemiol. Biomark. Prev. 2011, 20, 490–500. [Google Scholar] [CrossRef]
- Scalbert, A.; Brennan, L.; Manach, C.; Andres-Lacueva, C.; Dragsted, L.O.; Draper, J.; Rappaport, S.M.; Van der Hooft, J.J.; Wishart, D.S. The food metabolome: A window over dietary exposure. Am. J. Clin. Nutr. 2014, 99, 1286–1308. [Google Scholar] [CrossRef]
- Wang, Y.; Gapstur, S.M.; Carter, B.D.; Hartman, T.J.; Stevens, V.L.; Gaudet, M.M.; McCullough, M.L. Untargeted Metabolomics Identifies Novel Potential Biomarkers of Habitual Food Intake in a Cross-Sectional Study of Postmenopausal Women. J. Nutr. 2018, 148, 932–943. [Google Scholar] [CrossRef]
- Pallister, T.; Jennings, A.; Mohney, R.P.; Yarand, D.; Mangino, M.; Cassidy, A.; MacGregor, A.; Spector, T.D.; Menni, C. Characterizing Blood Metabolomics Profiles Associated with Self-Reported Food Intakes in Female Twins. PLoS ONE 2016, 11, e0158568. [Google Scholar] [CrossRef]
- Guertin, K.A.; Moore, S.C.; Sampson, J.N.; Huang, W.Y.; Xiao, Q.; Stolzenberg-Solomon, R.Z.; Sinha, R.; Cross, A.J. Metabolomics in nutritional epidemiology: Identifying metabolites associated with diet and quantifying their potential to uncover diet-disease relations in populations. Am. J. Clin. Nutr. 2014, 100, 208–217. [Google Scholar] [CrossRef]
- Andersen, M.B.; Kristensen, M.; Manach, C.; Pujos-Guillot, E.; Poulsen, S.K.; Larsen, T.M.; Astrup, A.; Dragsted, L. Discovery and validation of urinary exposure markers for different plant foods by untargeted metabolomics. Anal. Bioanal. Chem. 2014, 406, 1829–1844. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.; Yu, B.; Alexander, D.; Steffen, L.M.; Boerwinkle, E. Human metabolome associates with dietary intake habits among African Americans in the atherosclerosis risk in communities study. Am. J. Epidemiol. 2014, 179, 1424–1433. [Google Scholar] [CrossRef] [PubMed]
- Playdon, M.C.; Sampson, J.N.; Cross, A.J.; Sinha, R.; Guertin, K.A.; Moy, K.A.; Rothman, N.; Irwin, M.L.; Mayne, S.T.; Stolzenberg-Solomon, R.; et al. Comparing metabolite profiles of habitual diet in serum and urine. Am. J. Clin. Nutr. 2016, 104, 776–789. [Google Scholar] [CrossRef] [PubMed]
- Edmands, W.M.; Ferrari, P.; Rothwell, J.A.; Rinaldi, S.; Slimani, N.; Barupal, D.K.; Biessy, C.; Jenab, M.; Clavel-Chapelon, F.; Fagherazzi, G.; et al. Polyphenol metabolome in human urine and its association with intake of polyphenol-rich foods across European countries. Am. J. Clin. Nutr. 2015, 102, 905–913. [Google Scholar] [CrossRef] [PubMed]
- Playdon, M.C.; Moore, S.C.; Derkach, A.; Reedy, J.; Subar, A.F.; Sampson, J.N.; Albanes, D.; Gu, F.; Kontto, J.; Lassale, C.; et al. Identifying biomarkers of dietary patterns by using metabolomics. Am. J. Clin. Nutr. 2017, 105, 450–465. [Google Scholar] [CrossRef] [PubMed]
- McCullough, M.L.; Maliniak, M.L.; Stevens, V.L.; Carter, B.D.; Hodge, R.A.; Wang, Y. Metabolomic markers of healthy dietary patterns in US postmenopausal women. Am. J. Clin. Nutr. 2019, 109, 1439–1451. [Google Scholar] [CrossRef]
- Sampson, J.N.; Boca, S.M.; Shu, X.O.; Stolzenberg-Solomon, R.Z.; Matthews, C.E.; Hsing, A.W.; Tan, Y.T.; Ji, B.T.; Chow, W.H.; Cai, Q.; et al. Metabolomics in epidemiology: Sources of variability in metabolite measurements and implications. Cancer Epidemiol. Biomark. Prev. 2013, 22, 631–640. [Google Scholar] [CrossRef]
- Atkinson, W.; Downer, P.; Lever, M.; Chambers, S.T.; George, P.M. Effects of orange juice and proline betaine on glycine betaine and homocysteine in healthy male subjects. Eur. J. Nutr. 2007, 46, 446–452. [Google Scholar] [CrossRef]
- Lloyd, A.J.; Beckmann, M.; Fave, G.; Mathers, J.C.; Draper, J. Proline betaine and its biotransformation products in fasting urine samples are potential biomarkers of habitual citrus fruit consumption. Br. J. Nutr. 2011, 106, 812–824. [Google Scholar] [CrossRef]
- Heinzmann, S.S.; Brown, I.J.; Chan, Q.; Bictash, M.; Dumas, M.E.; Kochhar, S.; Stamler, J.; Holmes, E.; Elliott, P.; Nicholson, J.K. Metabolic profiling strategy for discovery of nutritional biomarkers: Proline betaine as a marker of citrus consumption. Am. J. Clin. Nutr. 2010, 92, 436–443. [Google Scholar] [CrossRef]
- Pujos-Guillot, E.; Hubert, J.; Martin, J.F.; Lyan, B.; Quintana, M.; Claude, S.; Chabanas, B.; Rothwell, J.A.; Bennetau-Pelissero, C.; Scalbert, A.; et al. Mass spectrometry-based metabolomics for the discovery of biomarkers of fruit and vegetable intake: Citrus fruit as a case study. J. Proteome Res. 2013, 12, 1645–1659. [Google Scholar] [CrossRef] [PubMed]
- Ulaszewska, M.; Vazquez-Manjarrez, N.; Garcia-Aloy, M.; Llorach, R.; Mattivi, F.; Dragsted, L.O.; Pratico, G.; Manach, C. Food intake biomarkers for apple, pear, and stone fruit. Genes Nutr. 2018, 13, 29. [Google Scholar] [CrossRef] [PubMed]
- Nieman, D.C.; Gillitt, N.D.; Sha, W.; Meaney, M.P.; John, C.; Pappan, K.L.; Kinchen, J.M. Metabolomics-Based Analysis of Banana and Pear Ingestion on Exercise Performance and Recovery. J. Proteome Res. 2015, 14, 5367–5377. [Google Scholar] [CrossRef] [PubMed]
- Playdon, M.C.; Ziegler, R.G.; Sampson, J.N.; Stolzenberg-Solomon, R.; Thompson, H.J.; Irwin, M.L.; Mayne, S.T.; Hoover, R.N.; Moore, S.C. Nutritional metabolomics and breast cancer risk in a prospective study. Am. J. Clin. Nutr. 2017, 106, 637–649. [Google Scholar] [CrossRef] [PubMed]
- Perera, T.; Young, M.R.; Zhang, Z.; Murphy, G.; Colburn, N.H.; Lanza, E.; Hartman, T.J.; Cross, A.J.; Bobe, G. Identification and monitoring of metabolite markers of dry bean consumption in parallel human and mouse studies. Mol. Nutr. Food Res. 2015, 59, 795–806. [Google Scholar] [CrossRef]
- Johansson-Persson, A.; Barri, T.; Ulmius, M.; Onning, G.; Dragsted, L.O. LC-QTOF/MS metabolomic profiles in human plasma after a 5-week high dietary fiber intake. Anal. Bioanal. Chem. 2013, 405, 4799–4809. [Google Scholar] [CrossRef]
- Zhu, Y.; Wang, P.; Sha, W.; Sang, S. Urinary Biomarkers of Whole Grain Wheat Intake Identified by Non-targeted and Targeted Metabolomics Approaches. Sci. Rep. 2016, 6, 36278. [Google Scholar] [CrossRef]
- Foti, R.S.; Fisher, M.B. Assessment of UDP-glucuronosyltransferase catalyzed formation of ethyl glucuronide in human liver microsomes and recombinant UGTs. Forensic Sci. Int. 2005, 153, 109–116. [Google Scholar] [CrossRef]
- Bogaki, T.; Mitani, K.; Oura, Y.; Ozeki, K. Effects of ethyl-alpha-d-glucoside on human dermal fibroblasts. Biosci. Biotechnol. Biochem. 2017, 81, 1706–1711. [Google Scholar] [CrossRef]
- Generoso, W.C.; Brinek, M.; Dietz, H.; Oreb, M.; Boles, E. Secretion of 2, 3-dihydroxyisovalerate as a limiting factor for isobutanol production in Saccharomyces cerevisiae. FEMS Yeast Res. 2017, 17. [Google Scholar] [CrossRef]
- Wang, G.F.; Shi, L.P.; Ren, Y.D.; Liu, Q.F.; Liu, H.F.; Zhang, R.J.; Li, Z.; Zhu, F.H.; He, P.L.; Tang, W.; et al. Anti-hepatitis B virus activity of chlorogenic acid, quinic acid and caffeic acid in vivo and in vitro. Antivir. Res. 2009, 83, 186–190. [Google Scholar] [CrossRef] [PubMed]
- Matsunaga, K.; Katayama, M.; Sakata, K.; Kuno, T.; Yoshida, K.; Yamada, Y.; Hirose, Y.; Yoshimi, N.; Mori, H. Inhibitory Effects of Chlorogenic Acid on Azoxymethane-induced Colon Carcinogenesis in Male F344 Rats. Asian Pac. J. Cancer Prev. 2002, 3, 163–166. [Google Scholar] [PubMed]
- Tanaka, T.; Nishikawa, A.; Shima, H.; Sugie, S.; Shinoda, T.; Yoshimi, N.; Iwata, H.; Mori, H. Inhibitory effects of chlorogenic acid, reserpine, polyprenoic acid (E-5166), or coffee on hepatocarcinogenesis in rats and hamsters. Basic Life Sci. 1990, 52, 429–440. [Google Scholar] [PubMed]
- Tinker, L.F.; Sarto, G.E.; Howard, B.V.; Huang, Y.; Neuhouser, M.L.; Mossavar-Rahmani, Y.; Beasley, J.M.; Margolis, K.L.; Eaton, C.B.; Phillips, L.S.; et al. Biomarker-calibrated dietary energy and protein intake associations with diabetes risk among postmenopausal women from the Women’s Health Initiative. Am. J. Clin. Nutr. 2011, 94, 1600–1606. [Google Scholar] [CrossRef] [PubMed]
- Lampe, J.W.; Huang, Y.; Neuhouser, M.L.; Tinker, L.F.; Song, X.; Schoeller, D.A.; Kim, S.; Raftery, D.; Di, C.; Zheng, C.; et al. Dietary biomarker evaluation in a controlled feeding study in women from the Women’s Health Initiative cohort. Am. J. Clin. Nutr. 2017, 105, 466–475. [Google Scholar] [CrossRef]
- Floegel, A.; Drogan, D.; Wang-Sattler, R.; Prehn, C.; Illig, T.; Adamski, J.; Joost, H.G.; Boeing, H.; Pischon, T. Reliability of serum metabolite concentrations over a 4-month period using a targeted metabolomic approach. PLoS ONE 2011, 6, e21103. [Google Scholar] [CrossRef]
- Carayol, M.; Licaj, I.; Achaintre, D.; Sacerdote, C.; Vineis, P.; Key, T.J.; Onland Moret, N.C.; Scalbert, A.; Rinaldi, S.; Ferrari, P. Reliability of Serum Metabolites over a Two-Year Period: A Targeted Metabolomic Approach in Fasting and Non-Fasting Samples from EPIC. PLoS ONE 2015, 10, e0135437. [Google Scholar] [CrossRef]
- Patel, A.V.; Jacobs, E.J.; Dudas, D.M.; Briggs, P.J.; Lichtman, C.J.; Bain, E.B.; Stevens, V.L.; McCullough, M.L.; Teras, L.R.; Campbell, P.T.; et al. The American Cancer Society’s Cancer Prevention Study 3 (CPS-3): Recruitment, study design, and baseline characteristics. Cancer 2017, 123, 2014–2024. [Google Scholar] [CrossRef]
- Troeschel, A.N.; Hartman, T.J.; Flanders, W.D.; Wang, Y.; Hodge, R.A.; McCullough, L.E.; Mitchell, D.C.; Sampson, L.; Patel, A.V.; McCullough, M.L. The American Cancer Society Cancer Prevention Study-3 FFQ Has Reasonable Validity and Reproducibility for Food Groups and a Diet Quality Score. J. Nutr. 2020, 150, 1566–1578. [Google Scholar] [CrossRef]
- Rimm, E.B.; Giovannucci, E.L.; Stampfer, M.J.; Colditz, G.A.; Litin, L.B.; Willett, W.C. Reproducibility and validity of an expanded self-administered semiquantitative food frequency questionnaire among male health professionals. Am. J. Epidemiol. 1992, 135, 1114–1126. [Google Scholar] [CrossRef]
- Feskanich, D.; Rimm, E.B.; Giovannucci, E.L.; Colditz, G.A.; Stampfer, M.J.; Litin, L.B.; Willett, W.C. Reproducibility and validity of food intake measurements from a semiquantitative food frequency questionnaire. J. Am. Diet. Assoc. 1993, 93, 790–796. [Google Scholar] [CrossRef]
- Evans, A.M.; DeHaven, C.D.; Barrett, T.; Mitchell, M.; Milgram, E. Integrated, nontargeted ultrahigh performance liquid chromatography/electrospray ionization tandem mass spectrometry platform for the identification and relative quantification of the small-molecule complement of biological systems. Anal. Chem. 2009, 81, 6656–6667. [Google Scholar] [CrossRef]
- Huber, W.; Von Heydebreck, A.; Sültmann, H.; Poustka, A.; Vingron, M. Variance stabilization applied to microarray data calibration and to the quantification of differential expression. Bioinformatics 2002, 18, S96–S104. [Google Scholar] [CrossRef] [PubMed]
- Robin, X.; Turck, N.; Hainard, A.; Tiberti, N.; Lisacek, F.; Sanchez, J.-C.; Müller, M. pROC: An open-source package for R and S+ to analyze and compare ROC curves. BMC Bioinform. 2011, 12, 77. [Google Scholar] [CrossRef] [PubMed]
| Characteristics | Men (n = 234) | Women (n = 437) |
|---|---|---|
| Age (year) | 52.4 ± 10.0 | 52.2 ± 9.2 |
| Race/ethnicity | ||
| White | 147 (62.8) | 256 (58.6) |
| Black | 42 (17.9) | 124 (28.4) |
| Hispanic | 45 (19.2) | 57 (13.0) |
| BMI at pre-FFQ (kg/m2) | 27.5 ± 5.4 | 27.7 ± 6.6 |
| Education | ||
| <College | 40 (17.1) | 108 (24.7) |
| College | 82 (35.0) | 144 (33.0) |
| ≥Graduate school | 103 (44.0) | 170 (38.9) |
| Unknown | 9 (3.8) | 15 (3.4) |
| Smoking status | ||
| Never | 181 (77.4) | 347 (79.4) |
| Former | 53 (22.6) | 90 (20.6) |
| Recreational physical activity (MET-h/wk) | ||
| 0–<5 | 44 (18.8) | 124 (28.4) |
| 5–<10 2 | 74 (31.6) | 147 (33.6) |
| 10–<15 | 50 (21.4) | 78 (17.8) |
| ≥15 | 66 (28.2) | 88 (20.1) |
| Ethanol intake (g/d) | 10.3 ± 13.9 | 7.0 ± 11.5 |
| Energy from post-FFQ (kcal/d) | 2136 ± 690 | 2007 ± 609 |
| Average energy intake from 24HRs (kcal/d) | 2214 ± 583 | 1730 ± 414 |
| Food Group/Items | Biochemical Name 2 | Super Pathway | Post-FFQ | Average Dietary Recalls | ICC 3 | ||||
|---|---|---|---|---|---|---|---|---|---|
| R | p Value | AUC | R | p Value | AUC | ||||
| FRUITS | |||||||||
| Avocado | X-11315 | 0.22 | 2.43 × 10−8 | 0.82 | 0.18 | 6.24 × 10−6 | 0.74 | 0.64 (0.59, 0.68) | |
| X-24475 | 0.21 | 9.21 × 10−8 | 0.82 | 0.13 | 1.07 × 10−3 | 0.73 | 0.56 (0.51, 0.61) | ||
| X-11858 | 0.24 | 2.51 × 10−10 | 0.82 | 0.15 | 1.64 × 10−4 | 0.73 | 0.52 (0.47, 0.58) | ||
| Apples or pears | 4-allylphenol sulfate | Xenobiotics | 0.20 | 1.63 × 10−7 | 0.79 | 0.44 (0.38, 0.50) | |||
| Apples 4 | 4-allylphenol sulfate | Xenobiotics | 0.21 | 3.04 × 10−8 | 0.70 | 0.44 (0.38, 0.50) | |||
| β-cryptoxanthin | Cofactors and Vitamins | 0.20 | 1.58 × 10−7 | 0.70 | 0.77 (0.74, 0.80) | ||||
| Total citrus fruits and juices | 3-hydroxystachydrine * | Xenobiotics | 0.50 | 1.37 × 10−43 | 0.94 | 0.47 | 4.27 × 10−38 | 0.84 | 0.33 (0.27, 0.40) |
| stachydrine | Xenobiotics | 0.50 | 1.31 × 10−43 | 0.93 | 0.46 | 3.27 × 10−36 | 0.85 | 0.50 (0.44, 0.55) | |
| N-methylproline | Amino Acid | 0.38 | 7.98 × 10−24 | 0.88 | 0.39 | 3.38 × 10−25 | 0.82 | 0.44 (0.38, 0.51) | |
| Oranges | β-cryptoxanthin | Cofactors and Vitamins | 0.30 | 3.05 × 10−15 | 0.81 | 0.33 | 1.07 × 10−17 | 0.76 | 0.77 (0.74, 0.80) |
| 3-hydroxystachydrine * | Xenobiotics | 0.31 | 1.13 × 10−15 | 0.81 | 0.27 | 1.38 × 10−12 | 0.75 | 0.33 (0.27, 0.40) | |
| stachydrine | Xenobiotics | 0.30 | 7.26 × 10−15 | 0.80 | 0.25 | 3.91 × 10−11 | 0.75 | 0.50 (0.44, 0.55) | |
| Orange juice | stachydrine | Xenobiotics | 0.35 | 6.46 × 10−21 | 0.88 | 0.34 | 1.28 × 10−19 | 0.80 | 0.50 (0.44, 0.55) |
| 3-hydroxystachydrine * | Xenobiotics | 0.35 | 7.40 × 10−21 | 0.87 | 0.33 | 1.19 × 10−18 | 0.79 | 0.33 (0.27, 0.40) | |
| N-methylproline | Amino Acid | 0.30 | 3.41 × 10−15 | 0.86 | 0.31 | 3.86 × 10−16 | 0.78 | 0.44 (0.38, 0.51) | |
| Grapefruit | stachydrine | Xenobiotics | 0.26 | 2.86 × 10−11 | 0.73 | 0.19 | 1.63 × 10−6 | 0.62 | 0.50 (0.44, 0.55) |
| 3-hydroxystachydrine * | Xenobiotics | 0.22 | 1.34 × 10−8 | 0.70 | 0.18 | 3.73 × 10−6 | 0.62 | 0.33 (0.27, 0.40) | |
| Watermelon | X-25271 | 0.37 | 2.65 × 10−22 | 0.83 | 0.26 | 1.47 × 10−11 | 0.66 | 0.33 (0.27, 0.40) | |
| Cantaloupe | X-25271 | 0.30 | 6.08 × 10−15 | 0.76 | 0.19 | 1.39 × 10−6 | 0.65 | 0.33 (0.27, 0.40) | |
| Berries | methyl glucopyranoside (α + β) | Xenobiotics | 0.17 | 2.00 × 10−5 | 0.83 | 0.23 | 4.29 × 10−9 | 0.76 | 0.62 (0.57, 0.67) |
| X-24475 | 0.21 | 7.82 × 10−8 | 0.83 | 0.21 | 7.16 × 10−8 | 0.75 | 0.56 (0.51, 0.61) | ||
| X-17354 | 0.16 | 2.32 × 10−5 | 0.83 | 0.20 | 2.18 × 10−7 | 0.73 | 0.62 (0.57, 0.67) | ||
| Blueberries | 4-allylphenol sulfate | Xenobiotics | 0.22 | 1.24 × 10−8 | 0.82 | 0.15 | 1.75 × 10−4 | 0.75 | 0.44 (0.38, 0.50) |
| γ-tocopherol/β-tocopherol | Cofactors and Vitamins | −0.15 | 1.72 × 10−4 | 0.80 | −0.22 | 1.62 × 10−8 | 0.75 | 0.69 (0.65, 0.73) | |
| methyl glucopyranoside (α + β) | Xenobiotics | 0.12 | 1.47 × 10−3 | 0.80 | 0.23 | 2.21 × 10−9 | 0.75 | 0.62 (0.57, 0.67) | |
| Raspberries | methyl glucopyranoside (α + β) | Xenobiotics | 0.16 | 4.89 × 10−5 | 0.77 | 0.20 | 1.58 × 10−7 | 0.65 | 0.62 (0.57, 0.67) |
| Peaches or plums | β-cryptoxanthin | Cofactors and Vitamins | 0.18 | 3.25 × 10−6 | 0.78 | 0.23 | 4.64 × 10−9 | 0.73 | 0.77 (0.74, 0.80) |
| X-12306 | 0.09 | 2.73 × 10−2 | 0.75 | 0.21 | 8.44 × 10−8 | 0.71 | 0.47 (0.41, 0.53) | ||
| VEGETABLES | |||||||||
| Tomatoes | 4-hydroxychlorothalonil | Xenobiotics | 0.21 | 6.64 × 10−8 | 0.82 | 0.15 | 8.00 × 10−5 | 0.72 | 0.85 (0.83, 0.87) |
| Asparagus | ergothioneine | Xenobiotics | 0.23 | 1.31 × 10−9 | 0.77 | 0.12 | 1.67 × 10−3 | 0.62 | 0.86 (0.84, 0.88) |
| X-11849 | 0.20 | 1.63 × 10−7 | 0.75 | 0.06 | 1.20 × 10−1 | 0.61 | 0.66 (0.62, 0.70) | ||
| X-11847 | 0.22 | 1.33 × 10−8 | 0.75 | 0.07 | 9.24 × 10−2 | 0.60 | 0.58 (0.52, 0.63) | ||
| Beans | S-methylcysteine | Amino Acid | 0.21 | 3.85 × 10−8 | 0.90 | 0.18 | 2.53 × 10−6 | 0.71 | 0.36 (0.30, 0.43) |
| pipecolate | Amino Acid | 0.21 | 5.06 × 10−8 | 0.89 | 0.19 | 1.68 × 10−6 | 0.72 | 0.32 (0.26, 0.39) | |
| X-11849 | 0.08 | 3.98 × 10−2 | 0.89 | 0.21 | 5.75 × 10−8 | 0.72 | 0.66 (0.62, 0.70) | ||
| Soy products | X-16649 | 0.33 | 1.41 × 10−18 | 0.77 | 0.37 | 6.46 × 10−23 | 0.75 | 0.46 (0.40, 0.52) | |
| X-24637 | 0.33 | 3.68 × 10−18 | 0.77 | 0.36 | 8.49 × 10−22 | 0.74 | 0.39 (0.33, 0.46) | ||
| 4-ethylphenyl sulfate | Xenobiotics | 0.30 | 4.86 × 10−15 | 0.75 | 0.35 | 1.37 × 10−20 | 0.74 | 0.52 (0.47, 0.58) | |
| Fermented soy products | X-11381 | −0.21 | 6.92 × 10−8 | 0.66 | −0.18 | 3.67 × 10−6 | 0.58 | 0.92 (0.91, 0.93) | |
| X-14939 | −0.01 | 8.53 × 10−1 | 0.64 | −0.21 | 6.91 × 10−8 | 0.59 | 0.68 (0.63, 0.72) | ||
| X-11261 | −0.07 | 7.02 × 10−2 | 0.64 | −0.22 | 6.49 × 10−9 | 0.59 | 0.65 (0.60, 0.69) | ||
| Soymilk | 4-ethylphenyl sulfate | Xenobiotics | 0.28 | 2.10 × 10−13 | 0.65 | 0.34 | 6.36 × 10−19 | 0.64 | 0.52 (0.47, 0.58) |
| X-24637 | 0.26 | 5.13 × 10−12 | 0.63 | 0.33 | 1.23 × 10−18 | 0.62 | 0.39 (0.33, 0.46) | ||
| X-16649 | 0.29 | 1.67 × 10−14 | 0.62 | 0.29 | 5.38 × 10−14 | 0.62 | 0.46 (0.40, 0.52) | ||
| Soy protein powder | X-16649 | 0.21 | 8.91 × 10−8 | 0.63 | 0.13 | 1.06 × 10−3 | 0.60 | 0.46 (0.40, 0.52) | |
| Cruciferous vegetables | S-methylcysteine | Amino Acid | 0.26 | 1.95 × 10−11 | 0.85 | 0.14 | 3.61 × 10−4 | 0.74 | 0.36 (0.30, 0.43) |
| carotene diol (2) | Cofactors and Vitamins | 0.23 | 1.78 × 10−9 | 0.83 | 0.20 | 1.35 × 10−7 | 0.74 | 0.79 (0.75, 0.81) | |
| X-13866 | 0.26 | 2.71 × 10−11 | 0.83 | 0.12 | 2.27 × 10−3 | 0.71 | 0.52 (0.47, 0.58) | ||
| Leafy greens | carotene diol (1) | Cofactors and Vitamins | 0.23 | 1.56 × 10−9 | 0.84 | 0.23 | 1.28 × 10−9 | 0.76 | 0.83 (0.80, 0.85) |
| carotene diol (2) | Cofactors and Vitamins | 0.22 | 2.02 × 10−8 | 0.83 | 0.21 | 3.22 × 10−8 | 0.75 | 0.79 (0.75, 0.81) | |
| docosahexaenoate (DHA; 22:6 n3) | Lipid | 0.20 | 2.24 × 10−7 | 0.81 | 0.12 | 2.02 × 10−3 | 0.69 | 0.55 (0.50, 0.60) | |
| Iceberg or head lettuce | pentose acid * | Partially Characterized Molecules | −0.23 | 1.09 × 10−9 | 0.71 | −0.03 | 4.61 × 10−1 | 0.57 | 0.56 (0.50, 0.61) |
| Peppers | X-23780 | 0.29 | 3.19 × 10−14 | 0.81 | 0.18 | 4.96 × 10−6 | 0.75 | 0.39 (0.33, 0.46) | |
| Mushrooms 4 | ergothioneine | Xenobiotics | 0.26 | 2.57 × 10−11 | 0.70 | 0.86 (0.84, 0.88) | |||
| X-11847 | 0.24 | 6.54 × 10−10 | 0.69 | 0.58 (0.52, 0.63) | |||||
| X-11858 | 0.22 | 1.34 × 10−8 | 0.69 | 0.52 (0.47, 0.58) | |||||
| Allium vegetables | N-methyltaurine | Amino Acid | 0.27 | 3.08 × 10−12 | 0.81 | 0.20 | 4.41 × 10−7 | 0.73 | 0.32 (0.25, 0.39) |
| ergothioneine | Xenobiotics | 0.22 | 1.18 × 10−8 | 0.80 | 0.10 | 7.38 × 10−3 | 0.71 | 0.86 (0.84, 0.88) | |
| N-acetylalliin | Xenobiotics | 0.22 | 1.19 × 10−8 | 0.79 | 0.06 | 1.08 × 10−1 | 0.70 | 0.29 (0.22, 0.36) | |
| Onion | N-methyltaurine | Amino Acid | 0.26 | 5.25 × 10−12 | 0.82 | 0.19 | 1.04 × 10−6 | 0.72 | 0.32 (0.25, 0.39) |
| ergothioneine | Xenobiotics | 0.21 | 4.36 × 10−8 | 0.79 | 0.10 | 1.08 × 10−2 | 0.70 | 0.86 (0.84, 0.88) | |
| N-acetylalliin | Xenobiotics | 0.21 | 1.07 × 10−7 | 0.79 | 0.06 | 1.57 × 10−1 | 0.69 | 0.29 (0.22, 0.36) | |
| Garlic | N-methyltaurine | Amino Acid | 0.25 | 8.60 × 10−11 | 0.81 | 0.24 | 8.70 × 10−10 | 0.74 | 0.32 (0.25, 0.39) |
| δ-CEHC | Cofactors and Vitamins | −0.23 | 3.48 × 10−9 | 0.81 | −0.14 | 4.34 × 10−4 | 0.69 | 0.48 (0.42, 0.54) | |
| N-acetylalliin | Xenobiotics | 0.29 | 3.06 × 10−14 | 0.81 | 0.12 | 2.72 × 10−3 | 0.67 | 0.29 (0.22, 0.36) | |
| Garlic powder | S-allylcysteine | Xenobiotics | 0.22 | 1.25 × 10−8 | 0.74 | 0.08 | 5.13 × 10−2 | 0.68 | 0.31 (0.24, 0.38) |
| GRAINS | |||||||||
| Whole grains | X-21752 | 0.31 | 8.54 × 10−16 | 0.89 | 0.19 | 1.10 × 10−6 | 0.80 | 0.71 (0.67, 0.75) | |
| 2,6-dihydroxybenzoic acid | Xenobiotics | 0.23 | 1.22 × 10−9 | 0.88 | 0.18 | 3.38 × 10−6 | 0.79 | 0.62 (0.57, 0.67) | |
| 4-methoxyphenol sulfate | Amino Acid | 0.21 | 9.90 × 10−8 | 0.87 | 0.17 | 8.86 × 10−6 | 0.77 | 0.34 (0.28, 0.41) | |
| Whole grain bread | 2-aminophenol sulfate | Xenobiotics | 0.22 | 7.79 × 10−9 | 0.80 | 0.20 | 4.50 × 10−7 | 0.71 | 0.45 (0.39, 0.51) |
| Whole grain cereals | X-21752 | 0.42 | 7.24 × 10−29 | 0.87 | 0.38 | 2.86 × 10−24 | 0.84 | 0.71 (0.67, 0.75) | |
| 2,6-dihydroxybenzoic acid | Xenobiotics | 0.27 | 1.50 × 10−12 | 0.80 | 0.22 | 1.69 × 10−8 | 0.79 | 0.62 (0.57, 0.67) | |
| 2-aminophenol sulfate | Xenobiotics | 0.30 | 6.86 × 10−15 | 0.79 | 0.25 | 5.65 × 10−11 | 0.80 | 0.45 (0.39, 0.51) | |
| Corn products | X-24545 | 0.23 | 2.55 × 10−9 | 0.83 | 0.08 | 4.03 × 10−2 | 0.71 | 0.72 (0.68, 0.75) | |
| X-16935 | 0.21 | 5.49 × 10−8 | 0.83 | 0.15 | 7.12 × 10−5 | 0.71 | 0.89 (0.87, 0.90) | ||
| γ-tocopherol/β-tocopherol | Cofactors and Vitamins | 0.20 | 1.84 × 10−7 | 0.83 | 0.02 | 5.57 × 10−1 | 0.71 | 0.69 (0.65, 0.73) | |
| Refined grains | γ-tocopherol/β-tocopherol | Cofactors and Vitamins | 0.24 | 4.12 × 10−10 | 0.84 | 0.12 | 1.86 × 10−3 | 0.85 | 0.69 (0.65, 0.73) |
| X-24475 | −0.20 | 1.29 × 10−7 | 0.84 | −0.17 | 1.19 × 10−5 | 0.84 | 0.56 (0.51, 0.61) | ||
| X-23680 | 0.13 | 9.26 × 10−4 | 0.83 | 0.21 | 4.76 × 10−8 | 0.85 | 0.57 (0.52, 0.62) | ||
| PROTEINS | |||||||||
| Eggs | PE (p-18:0/20:4) * | Lipid | 0.25 | 5.58 × 10−11 | 0.79 | 0.20 | 3.60 × 10−7 | 0.75 | 0.68 (0.63, 0.72) |
| PE (p-16:0/20:4) * | Lipid | 0.21 | 8.46 × 10−8 | 0.78 | 0.18 | 2.67 × 10−6 | 0.73 | 0.60 (0.55, 0.65) | |
| Red meat | X-11381 | 0.40 | 2.62 × 10−26 | 0.88 | 0.37 | 1.28 × 10−22 | 0.83 | 0.92 (0.91, 0.93) | |
| PE (p-18:0/20:4) * | Lipid | 0.40 | 4.29 × 10−26 | 0.88 | 0.37 | 1.73 × 10−22 | 0.82 | 0.68 (0.63, 0.72) | |
| PE (p-18:0/18:1) | Lipid | 0.30 | 1.61 × 10−15 | 0.87 | 0.26 | 3.22 × 10−11 | 0.79 | 0.54 (0.49, 0.59) | |
| Processed meat | PE (p-18:0/20:4) * | Lipid | 0.38 | 2.27 × 10−23 | 0.85 | 0.31 | 6.65 × 10−16 | 0.80 | 0.68 (0.63, 0.72) |
| PE (p-16:0/20:4) * | Lipid | 0.31 | 1.03 × 10−15 | 0.83 | 0.30 | 5.97 × 10−15 | 0.80 | 0.60 (0.55, 0.65) | |
| PC (p-16:0/20:4) * | Lipid | 0.31 | 8.70 × 10−16 | 0.83 | 0.24 | 8.37 × 10−10 | 0.78 | 0.73 (0.70, 0.77) | |
| Poultry | PE (p-16:0/20:4) * | Lipid | 0.47 | 3.24 × 10−37 | 0.87 | 0.42 | 6.64 × 10−30 | 0.83 | 0.60 (0.55, 0.65) |
| PE (p-18:0/20:4) * | Lipid | 0.45 | 2.97 × 10−34 | 0.85 | 0.40 | 4.40 × 10−27 | 0.81 | 0.68 (0.63, 0.72) | |
| 3-methylhistidine | Amino Acid | 0.54 | 5.73 × 10−51 | 0.85 | 0.40 | 8.50 × 10−27 | 0.81 | 0.45 (0.39, 0.51) | |
| Total fish | hydroxy-CMPF * | Lipid | 0.43 | 1.37 × 10−31 | 0.84 | 0.27 | 8.43 × 10−13 | 0.72 | 0.96 (0.95, 0.96) |
| CMPF | Lipid | 0.43 | 1.94 × 10−30 | 0.83 | 0.30 | 1.31 × 10−15 | 0.73 | 0.86 (0.84, 0.88) | |
| PC (16:0/22:6) | Lipid | 0.30 | 1.52 × 10−15 | 0.81 | 0.27 | 3.03 × 10−12 | 0.71 | 0.77 (0.74, 0.80) | |
| Dark meat fish | hydroxy-CMPF * | Lipid | 0.44 | 3.03 × 10−32 | 0.85 | 0.27 | 2.07 × 10−12 | 0.74 | 0.96 (0.95, 0.96) |
| CMPF | Lipid | 0.43 | 2.93 × 10−31 | 0.84 | 0.28 | 1.58 × 10−13 | 0.75 | 0.86 (0.84, 0.88) | |
| PC (16:0/22:6) | Lipid | 0.35 | 4.59 × 10−20 | 0.83 | 0.24 | 7.08 × 10−10 | 0.72 | 0.77 (0.74, 0.80) | |
| Shellfish | X-25810 | 0.35 | 2.61 × 10−20 | 0.77 | 0.24 | 3.13 × 10−10 | 0.70 | 0.55 (0.50, 0.60) | |
| CMPF | Lipid | 0.27 | 1.28 × 10−12 | 0.74 | 0.17 | 7.56 × 10−6 | 0.70 | 0.86 (0.84, 0.88) | |
| X-25419 | 0.36 | 9.16 × 10−22 | 0.73 | 0.20 | 2.06 × 10−7 | 0.69 | 0.64 (0.60, 0.69) | ||
| Total nuts | tryptophan betaine | Amino Acid | 0.43 | 8.29 × 10−31 | 0.91 | 0.30 | 4.82 × 10−15 | 0.83 | 0.82 (0.80, 0.85) |
| X-11315 | 0.27 | 1.22 × 10−12 | 0.91 | 0.26 | 1.62 × 10−11 | 0.82 | 0.64 (0.59, 0.68) | ||
| X-23644 | 0.31 | 5.97 × 10−16 | 0.89 | 0.26 | 1.11 × 10−11 | 0.80 | 0.32 (0.26, 0.39) | ||
| Peanuts | 4-vinylphenol sulfate | Xenobiotics | 0.39 | 1.27 × 10−25 | 0.87 | 0.23 | 4.54 × 10−9 | 0.70 | 0.39 (0.32, 0.46) |
| tryptophan betaine | Amino Acid | 0.39 | 7.63 × 10−26 | 0.86 | 0.33 | 1.14 × 10−17 | 0.77 | 0.82 (0.80, 0.85) | |
| behenoylcarnitine (C22) * | Lipid | 0.33 | 2.54 × 10−18 | 0.85 | 0.20 | 1.76 × 10−7 | 0.69 | 0.45 (0.39, 0.51) | |
| Other nuts | X-11315 | 0.29 | 1.26 × 10−14 | 0.89 | 0.32 | 3.85 × 10−17 | 0.84 | 0.64 (0.59, 0.68) | |
| X-24475 | 0.30 | 2.21 × 10−15 | 0.87 | 0.30 | 7.54 × 10−15 | 0.82 | 0.56 (0.51, 0.61) | ||
| tryptophan betaine | Amino Acid | 0.25 | 9.08 × 10−11 | 0.85 | 0.19 | 7.64 × 10−7 | 0.78 | 0.82 (0.80, 0.85) | |
| Seeds | X-11858 | 0.17 | 1.89 × 10−5 | 0.75 | 0.27 | 4.08 × 10−12 | 0.76 | 0.52 (0.47, 0.58) | |
| ergothioneine | Xenobiotics | 0.23 | 3.15 × 10−9 | 0.75 | 0.21 | 5.22 × 10−8 | 0.72 | 0.86 (0.84, 0.88) | |
| X-17354 | 0.21 | 6.02 × 10−8 | 0.74 | 0.26 | 1.57 × 10−11 | 0.75 | 0.62 (0.57, 0.67) | ||
| DAIRY/DAIRY ALTERNATIVES | |||||||||
| Milk | X-11381 | 0.33 | 3.73 × 10−18 | 0.84 | 0.27 | 3.03 × 10−12 | 0.77 | 0.92 (0.91, 0.93) | |
| N,N,N-trimethyl-5-aminovalerate | Amino Acid | 0.27 | 2.10 × 10−12 | 0.83 | 0.23 | 4.07 × 10−9 | 0.73 | 0.87 (0.85, 0.89) | |
| 3-bromo-5-chloro-2,6-dihydroxybenzoic acid * | Xenobiotics | 0.28 | 3.04 × 10−13 | 0.82 | 0.23 | 1.36 × 10−9 | 0.75 | 0.75 (0.72, 0.79) | |
| Almond or rice milk | X-24475 | 0.24 | 4.75 × 10−10 | 0.72 | 0.19 | 1.59 × 10−6 | 0.65 | 0.56 (0.51, 0.61) | |
| 3-bromo-5-chloro-2,6-dihydroxybenzoic acid * | Xenobiotics | −0.18 | 3.89 × 10−6 | 0.71 | −0.21 | 3.93 × 10−8 | 0.64 | 0.75 (0.72, 0.79) | |
| 3,5-dichloro-2,6-dihydroxybenzoic acid | Xenobiotics | −0.19 | 1.39 × 10−6 | 0.70 | −0.21 | 1.05 × 10−7 | 0.64 | 0.89 (0.88, 0.91) | |
| Total cheese | heptenedioate (C7:1-DC) * | Lipid | 0.30 | 1.58 × 10−15 | 0.88 | 0.23 | 1.28 × 10−9 | 0.78 | 0.44 (0.38, 0.50) |
| SM (d17:2/16:0, d18:2/15:0) * | Lipid | 0.24 | 2.57 × 10−10 | 0.88 | 0.19 | 1.80 × 10−6 | 0.78 | 0.65 (0.60, 0.69) | |
| margaroylcarnitine (C17) * | Lipid | 0.25 | 1.24 × 10−10 | 0.88 | 0.20 | 1.21 × 10−7 | 0.78 | 0.37 (0.31, 0.44) | |
| Cream | X-21442 | 0.36 | 2.77 × 10−21 | 0.80 | 0.12 | 3.11 × 10−3 | 0.71 | 0.87 (0.85, 0.88) | |
| quinate | Xenobiotics | 0.34 | 1.82 × 10−19 | 0.80 | 0.12 | 2.97 × 10−3 | 0.70 | 0.81 (0.79, 0.84) | |
| X-12816 | 0.26 | 2.67 × 10−11 | 0.75 | 0.11 | 4.69 × 10−3 | 0.70 | 0.87 (0.85, 0.89) | ||
| FATS AND OILS | |||||||||
| Creamy salad dressing | X-16944 | 0.27 | 1.69 × 10−12 | 0.78 | 0.25 | 7.40 × 10−11 | 0.70 | 0.59 (0.54, 0.64) | |
| X-11261 | 0.28 | 2.55 × 10−13 | 0.78 | 0.22 | 1.92 × 10−8 | 0.69 | 0.65 (0.60, 0.69) | ||
| X-15486 | 0.27 | 1.40 × 10−12 | 0.78 | 0.20 | 1.22 × 10−7 | 0.68 | 0.55 (0.49, 0.60) | ||
| Oil and vinegar salad dressing | carotene diol (1) | Cofactors and Vitamins | 0.18 | 4.41 × 10−6 | 0.76 | 0.22 | 1.25 × 10−8 | 0.81 | 0.83 (0.80, 0.85) |
| X-24475 | 0.22 | 1.50 × 10−8 | 0.76 | 0.09 | 2.84 × 10−2 | 0.78 | 0.56 (0.51, 0.61) | ||
| Olive oil | X-25419 | 0.22 | 1.57 × 10−8 | 0.78 | 0.15 | 8.26 × 10−5 | 0.74 | 0.64 (0.60, 0.69) | |
| δ-CEHC | Cofactors and Vitamins | −0.24 | 2.84 × 10−10 | 0.78 | −0.15 | 1.87 × 10−4 | 0.74 | 0.48 (0.42, 0.54) | |
| MISCELLANEOUS | |||||||||
| French fries | γ-tocopherol/β-tocopherol | Cofactors and Vitamins | 0.21 | 4.60 × 10−8 | 0.85 | 0.10 | 1.48 × 10−2 | 0.72 | 0.69 (0.65, 0.73) |
| pentose acid * | Partially Characterized Molecules | −0.24 | 4.27 × 10−10 | 0.85 | −0.07 | 6.36 × 10−2 | 0.71 | 0.56 (0.50, 0.61) | |
| X-07765 | 0.23 | 4.00 × 10−9 | 0.84 | 0.07 | 7.28 × 10−2 | 0.71 | 0.47 (0.41, 0.53) | ||
| Ice cream | X-07765 | 0.20 | 1.43 × 10−7 | 0.82 | 0.09 | 1.95 × 10−2 | 0.68 | 0.47 (0.41, 0.53) | |
| tridecenedioate (C13:1-DC) * | Lipid | 0.21 | 7.05 × 10−8 | 0.80 | 0.07 | 6.05 × 10−2 | 0.68 | 0.58 (0.53, 0.63) | |
| margaroylcarnitine (C17) * | Lipid | 0.21 | 8.68 × 10−8 | 0.80 | 0.10 | 1.19 × 10−2 | 0.68 | 0.37 (0.31, 0.44) | |
| Chips | X-21339 | 0.31 | 1.57 × 10−16 | 0.81 | 0.26 | 1.38 × 10−11 | 0.78 | 0.90 (0.89, 0.92) | |
| X-11880 | 0.30 | 1.04 × 10−14 | 0.80 | 0.28 | 1.35 × 10−13 | 0.80 | 0.90 (0.89, 0.91) | ||
| X-11308 | 0.25 | 1.29 × 10−10 | 0.79 | 0.19 | 1.65 × 10−6 | 0.76 | 0.95 (0.95, 0.96) | ||
| Chocolate candies | X-13728 | 0.32 | 1.62 × 10−17 | 0.83 | 0.32 | 1.11 × 10−16 | 0.84 | 0.54 (0.48, 0.59) | |
| theobromine | Xenobiotics | 0.29 | 1.39 × 10−14 | 0.82 | 0.29 | 5.24 × 10−14 | 0.83 | 0.56 (0.51, 0.62) | |
| 3,7-dimethylurate | Xenobiotics | 0.29 | 1.46 × 10−14 | 0.82 | 0.29 | 1.82 × 10−14 | 0.83 | 0.46 (0.40, 0.52) | |
| Dark chocolate | theobromine | Xenobiotics | 0.26 | 7.78 × 10−12 | 0.80 | 0.22 | 1.64 × 10−8 | 0.72 | 0.56 (0.51, 0.62) |
| X-13728 | 0.30 | 1.70 × 10−15 | 0.80 | 0.22 | 5.69 × 10−9 | 0.71 | 0.54 (0.48, 0.59) | ||
| 7-methylxanthine | Xenobiotics | 0.29 | 3.17 × 10−14 | 0.79 | 0.23 | 3.73 × 10−9 | 0.72 | 0.48 (0.42, 0.54) | |
| Energy/protein Bars | X-16649 | 0.20 | 1.40 × 10−7 | 0.80 | 0.19 | 1.79 × 10−6 | 0.71 | 0.46 (0.40, 0.52) | |
| Soy sauce | X-11858 | 0.20 | 2.42 × 10−7 | 0.74 | 0.21 | 1.09 × 10−7 | 0.66 | 0.52 (0.47, 0.58) | |
| X-11849 | 0.20 | 2.33 × 10−7 | 0.74 | 0.20 | 1.53 × 10−7 | 0.66 | 0.66 (0.62, 0.70) | ||
| X-11847 | 0.20 | 1.59 × 10−7 | 0.74 | 0.20 | 1.47 × 10−7 | 0.66 | 0.58 (0.52, 0.63) | ||
| Artificial sweeteners | acesulfame | Xenobiotics | 0.24 | 3.28 × 10−10 | 0.75 | 0.23 | 2.16 × 10−9 | 0.74 | 0.49 (0.43, 0.55) |
| saccharin | Xenobiotics | 0.21 | 9.48 × 10−8 | 0.68 | 0.21 | 3.09 × 10−8 | 0.66 | 0.59 (0.54, 0.64) | |
| erythritol | Xenobiotics | 0.19 | 1.07 × 10−6 | 0.66 | 0.20 | 1.28 × 10−7 | 0.63 | 0.48 (0.42, 0.54) | |
| ALCOHOL | |||||||||
| Total alcohol | ethyl α-glucopyranoside | Xenobiotics | 0.52 | 9.89 × 10−47 | 0.94 | 0.46 | 2.91 × 10−36 | 0.88 | 0.52 (0.47, 0.58) |
| ethyl glucuronide | Xenobiotics | 0.43 | 1.11 × 10−31 | 0.92 | 0.40 | 3.66 × 10−27 | 0.87 | 0.57 (0.52, 0.62) | |
| 2,3-dihydroxyisovalerate | Xenobiotics | 0.37 | 2.51 × 10−23 | 0.91 | 0.40 | 1.57 × 10−26 | 0.85 | 0.46 (0.40, 0.52) | |
| Beer | ethyl α-glucopyranoside | Xenobiotics | 0.38 | 1.23 × 10−23 | 0.84 | 0.33 | 7.63 × 10−18 | 0.82 | 0.52 (0.47, 0.58) |
| theophylline | Xenobiotics | 0.29 | 1.18 × 10−14 | 0.82 | 0.23 | 2.04 × 10−9 | 0.81 | 0.78 (0.74, 0.81) | |
| X-11795 | 0.25 | 3.27 × 10−11 | 0.82 | 0.23 | 1.13 × 10−9 | 0.81 | 0.56 (0.51, 0.62) | ||
| Total wine | ethyl α-glucopyranoside | Xenobiotics | 0.49 | 6.42 × 10−41 | 0.91 | 0.41 | 8.57 × 10−28 | 0.80 | 0.52 (0.47, 0.58) |
| 2,3-dihydroxyisovalerate | Xenobiotics | 0.44 | 1.69 × 10−32 | 0.86 | 0.42 | 7.57 × 10−30 | 0.80 | 0.46 (0.40, 0.52) | |
| ethyl glucuronide | Xenobiotics | 0.45 | 6.20 × 10−34 | 0.85 | 0.40 | 1.30 × 10−26 | 0.82 | 0.57 (0.52, 0.62) | |
| Red wine | ethyl α-glucopyranoside | Xenobiotics | 0.45 | 1.76 × 10−33 | 0.83 | 0.35 | 1.05 × 10−20 | 0.78 | 0.52 (0.47, 0.58) |
| 2,3-dihydroxyisovalerate | Xenobiotics | 0.40 | 2.37 × 10−27 | 0.79 | 0.36 | 2.34 × 10−21 | 0.77 | 0.46 (0.40, 0.52) | |
| pentose acid * | Partially Characterized Molecules | 0.32 | 3.25 × 10−17 | 0.79 | 0.31 | 1.17 × 10−15 | 0.76 | 0.56 (0.50, 0.61) | |
| White wine | ethyl α-glucopyranoside | Xenobiotics | 0.33 | 1.17 × 10−18 | 0.73 | 0.32 | 2.76 × 10−17 | 0.72 | 0.52 (0.47, 0.58) |
| pentose acid * | Partially Characterized Molecules | 0.24 | 5.40 × 10−10 | 0.71 | 0.29 | 2.21 × 10−14 | 0.74 | 0.56 (0.50, 0.61) | |
| X-11795 | 0.25 | 1.17 × 10−10 | 0.70 | 0.25 | 1.27 × 10−10 | 0.69 | 0.56 (0.51, 0.62) | ||
| Liquor | ethyl α-glucopyranoside | Xenobiotics | 0.34 | 6.55 × 10−19 | 0.75 | 0.16 | 2.35 × 10−5 | 0.68 | 0.52 (0.47, 0.58) |
| 2-hydroxyphytanate * | Lipid | 0.20 | 1.25 × 10−7 | 0.72 | 0.14 | 4.17 × 10−4 | 0.67 | 0.60 (0.55, 0.65) | |
| ethyl glucuronide | Xenobiotics | 0.30 | 3.51 × 10−15 | 0.72 | 0.15 | 1.80 × 10−4 | 0.66 | 0.57 (0.52, 0.62) | |
| BEVERAGES | |||||||||
| Total coffee | X-21442 | 0.81 | 0.35 × 10−153 | 0.99 | 0.80 | 0.81 × 10−148 | 0.99 | 0.87 (0.85, 0.88) | |
| quinate | Xenobiotics | 0.77 | 0.38 × 10−129 | 0.99 | 0.74 | 0.13 × 10−113 | 0.97 | 0.81 (0.79, 0.84) | |
| X-23655 | 0.56 | 4.11 × 10−56 | 0.98 | 0.52 | 3.17 × 10−47 | 0.95 | 0.64 (0.59, 0.68) | ||
| Decaffeinated | X-21442 | 0.27 | 1.09 × 10−12 | 0.70 | 0.23 | 1.20 × 10−9 | 0.65 | 0.87 (0.85, 0.88) | |
| quinate | Xenobiotics | 0.21 | 1.08 × 10−7 | 0.69 | 0.15 | 1.43 × 10−4 | 0.65 | 0.81 (0.79,0.84) | |
| Caffeinated | X-21442 | 0.75 | 0.21 × 10−120 | 0.98 | 0.75 | 0.57 × 10−118 | 0.97 | 0.87 (0.85, 0.88) | |
| quinate | Xenobiotics | 0.71 | 0.96 × 10−101 | 0.98 | 0.71 | 0.81 × 10−100 | 0.97 | 0.81 (0.79, 0.84) | |
| 3-hydroxypyridine sulfate | Xenobiotics | 0.59 | 3.37 × 10−63 | 0.96 | 0.57 | 5.09 × 10−57 | 0.94 | 0.72 (0.69, 0.76) | |
| Total tea | theanine | Xenobiotics | 0.40 | 8.58 × 10−27 | 0.86 | 0.39 | 1.53 × 10−25 | 0.83 | 0.60 (0.55, 0.65) |
| X-17685 | 0.20 | 2.32 × 10−7 | 0.73 | 0.24 | 5.72 × 10−10 | 0.74 | 0.50 (0.44, 0.55) | ||
| 3-methoxycatechol sulfate (1) | Xenobiotics | 0.20 | 2.49 × 10−7 | 0.73 | 0.22 | 1.46 × 10−8 | 0.72 | 0.42 (0.36, 0.49) | |
| Green tea | theanine | Xenobiotics | 0.25 | 5.18 × 10−11 | 0.72 | 0.28 | 1.44 × 10−13 | 0.69 | 0.60 (0.55, 0.65) |
| Black tea | theanine | Xenobiotics | 0.34 | 1.03 × 10−19 | 0.76 | 0.35 | 8.60 × 10−21 | 0.77 | 0.60 (0.55, 0.65) |
| X-17685 | 0.23 | 2.89 × 10−9 | 0.69 | 0.20 | 1.66 × 10−7 | 0.68 | 0.50 (0.44, 0.55) | ||
| 3-methoxycatechol sulfate (1) | Xenobiotics | 0.20 | 1.68 × 10−7 | 0.68 | 0.19 | 1.74 × 10−6 | 0.66 | 0.42 (0.36, 0.49) | |
| Herbal tea | X-18901 | 0.22 | 9.41 × 10−9 | 0.74 | 0.20 | 2.60 × 10−7 | 0.65 | 0.68 (0.63, 0.72) | |
| X-12306 | 0.20 | 1.50 × 10−7 | 0.71 | 0.18 | 4.49 × 10−6 | 0.63 | 0.47 (0.41, 0.53) | ||
| Diet beverages | acesulfame | Xenobiotics | 0.42 | 1.15 × 10−29 | 0.82 | 0.29 | 4.97 × 10−14 | 0.76 | 0.49 (0.43, 0.55) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, Y.; Hodge, R.A.; Stevens, V.L.; Hartman, T.J.; McCullough, M.L. Identification and Reproducibility of Plasma Metabolomic Biomarkers of Habitual Food Intake in a US Diet Validation Study. Metabolites 2020, 10, 382. https://doi.org/10.3390/metabo10100382
Wang Y, Hodge RA, Stevens VL, Hartman TJ, McCullough ML. Identification and Reproducibility of Plasma Metabolomic Biomarkers of Habitual Food Intake in a US Diet Validation Study. Metabolites. 2020; 10(10):382. https://doi.org/10.3390/metabo10100382
Chicago/Turabian StyleWang, Ying, Rebecca A. Hodge, Victoria L. Stevens, Terryl J. Hartman, and Marjorie L. McCullough. 2020. "Identification and Reproducibility of Plasma Metabolomic Biomarkers of Habitual Food Intake in a US Diet Validation Study" Metabolites 10, no. 10: 382. https://doi.org/10.3390/metabo10100382
APA StyleWang, Y., Hodge, R. A., Stevens, V. L., Hartman, T. J., & McCullough, M. L. (2020). Identification and Reproducibility of Plasma Metabolomic Biomarkers of Habitual Food Intake in a US Diet Validation Study. Metabolites, 10(10), 382. https://doi.org/10.3390/metabo10100382






