Physicochemical Characteristics of Cardiological Drugs and Practical Recommendations for Intravenous Administration: A Systematic Review
Abstract
1. Introduction
2. Materials and Methods
- In vitro and/or animal studies.
- Papers not written in English.
- Papers not about cardiological agents.
- Papers about non-IV cardiological agents.
- Papers discussing cardiological drugs that have been withdrawn due to adverse events or removed from the market.
- Papers on cardiological agents are currently under investigation in phases II and III or awaiting approval.
3. Results
- Antiarrhythmics
- Vasoconstrictor and Inotropic (V&I) Drugs
- Thrombolytics
- Antiaggregants
- Antihypertensives
- Anticoagulants
- Diuretics
- Other drugs
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Dychter, S.S.; Gold, D.A.; Carson, D.; Haller, M. Intravenous Therapy: A review of complications and economic considerations of peripheral access. J. Infus. Nurs. 2012, 35, 84–91. [Google Scholar] [CrossRef] [PubMed]
- Pinelli, F.; Pittiruti, M.; Annetta, M.G.; Barbani, F.; Bertoglio, S.; Biasucci, D.G.; Bolis, D.; Brescia, F.; Capozzoli, G.; D’arrigo, S.; et al. A GAVeCeLT consensus on the indication, insertion, and management of central venous access devices in the critically ill. J. Vasc. Access 2024. [Google Scholar] [CrossRef] [PubMed]
- Helm, R.E.; Klausner, J.D.; Klemperer, J.D.; Flint, L.M.; Huang, E. Accepted but Unacceptable: Peripheral IV catheter failure. J. Infus. Nurs. 2015, 38, 189–203. [Google Scholar] [CrossRef] [PubMed]
- Foor, J.S.; Moureau, N.L.; Gibbons, D.; Gibson, S.M. Investigative study of hemodilution ratio: 4Vs for vein diameter, valve, velocity, and volumetric blood flow as factors for optimal forearm vein selection for intravenous infusion. J. Vasc. Access 2022, 25, 140–148. [Google Scholar] [CrossRef]
- Nickel, B.A.-C.; Gorski, L.M.; Kleidon, T.P.; Kyes, A.M.; DeVries, M.M.; Keogh, S.P.; Meyer, B.P.; Sarver, M.J.M.; Crickman, R.D.; Ong, J.; et al. Infusion Therapy Standards of Practice, 9th Edition. J. Infus. Nurs. 2024, 47, S1–S2. [Google Scholar] [CrossRef]
- Annetta, M.G.; Elli, S.; Marche, B.; Pinelli, F.; Pittiruti, M. Femoral venous access: State of the art and future perspectives. J. Vasc. Access 2023. [Google Scholar] [CrossRef]
- Pittiruti, M.; Van Boxtel, T.; Scoppettuolo, G.; Carr, P.; Konstantinou, E.; Miluy, G.O.; Lamperti, M.; Goossens, G.A.; Simcock, L.; Dupont, C.; et al. European recommendations on the proper indication and use of peripheral venous access devices (the ERPIUP consensus): A WoCoVA project. J. Vasc. Access 2023, 24, 165–182. [Google Scholar] [CrossRef]
- Teja, B.; Bosch, N.A.; Diep, C.; Pereira, T.V.; Mauricio, P.; Sklar, M.C.; Sankar, A.; Wijeysundera, H.C.; Saskin, R.; Walkey, A.; et al. Complication Rates of Central Venous Catheters: A Systematic Review and Meta-Analysis. JAMA Intern. Med. 2024, 184, 474–482. [Google Scholar] [CrossRef]
- Ullman, A.J.; Marsh, N.; Mihala, G.; Cooke, M.; Rickard, C.M. Complications of Central Venous Access Devices: A Systematic Review. Pediatrics 2015, 136, e1331–e1344. [Google Scholar] [CrossRef]
- Gorski, L.A.M.; Ong, J.; Van Gerpen, R.M.; Nickel, B.A.-C.; Kokotis, K.R.; Hadaway, L.M. Development of an Evidence-Based List of Non-Antineoplastic Vesicants. J. Infus. Nurs. 2024, 47, 290–323. [Google Scholar] [CrossRef]
- Manrique-Rodríguez, S.; Heras-Hidalgo, I.; Pernia-López, M.S.; Herranz-Alonso, A.; Pisabarro, M.C.d.R.; Suárez-Mier, M.B.; Cubero-Pérez, M.A.; Viera-Rodríguez, V.; Cortés-Rey, N.; Lafuente-Cabrero, E.; et al. Standardization and Chemical Characterization of Intravenous Therapy in Adult Patients: A Step Further in Medication Safety. Drugs RD 2020, 21, 39–64. [Google Scholar] [CrossRef] [PubMed]
- Cardenas-Garcia, J.; Schaub, K.F.; Belchikov, Y.G.; Narasimhan, M.; Koenig, S.J.; Mayo, P.H. Safety of peripheral intravenous administration of vasoactive medication. J. Hosp. Med. 2015, 10, 581–585. [Google Scholar] [CrossRef] [PubMed]
- Loubani, O.M.; Green, R.S. A systematic review of extravasation and local tissue injury from administration of vasopressors through peripheral intravenous catheters and central venous catheters. J. Crit. Care 2015, 30, 653.e9–653.e17. [Google Scholar] [CrossRef]
- Borgonovo, F.; Quici, M.; Gidaro, A.; Giustivi, D.; Cattaneo, D.; Gervasoni, C.; Calloni, M.; Martini, E.; La Cava, L.; Antinori, S.; et al. Physicochemical Characteristics of Antimicrobials and Practical Recommendations for Intravenous Administration: A Systematic Review. Antibiotics 2023, 12, 1338. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef]
- Pittiruti, M.; Scoppettuolo, G. Manuale GAVeCeLT dei PICC e dei Midline. Indicazioni, Impianto, Gestione; EDRA: Milan Italy, 2022. [Google Scholar]
- Ross, A.M.; Gibbons, R.J.; Stone, G.W.; Kloner, R.A.; Alexander, R.W. A Randomized, double-blinded, placebo-controlled multicenter trial of adenosine as an adjunct to reperfusion in the treatment of acute myocardial infarction (AMISTAD-II). J. Am. Coll. Cardiol. 2005, 45, 1775–1780. [Google Scholar] [CrossRef]
- Yavuz, T.; Bertolet, B.; Bebooul, Y.; Tunerir, B.; Aslan, R.; Ocal, A.; Ybribim, E.; Kutsal, A. Role of endogenous adenosine in atrial fibrillation after coronary artery bypass graft. Clin. Cardiol. 2004, 27, 343–346. [Google Scholar] [CrossRef]
- Wasir, H.; Bhan, A.; Choudhary, S.K.; Sharma, R.; Chauhan, S.; Venugopal, P. Pretreatment of human myocardium with adenosine. Eur. J. Cardio-Thorac. Surg. 2001, 19, 41–46. [Google Scholar] [CrossRef]
- Thomas, G.S.; Prill, N.V.; Majmundar, H.; Fabrizi, R.R.; Thomas, J.J.; Hayashida, C.; Kothapalli, S.; Payne, J.L.; Payne, M.M.; Miyamoto, M.I. Treadmill exercise during adenosine infusion is safe, results in fewer adverse reactions, and improves myocardial perfusion image quality. J. Nucl. Cardiol. 2000, 7, 439–446. [Google Scholar] [CrossRef]
- Tawa, C.; Baker, W.; Kleiman, N.; Trakhtenbroit, A.; Desir, R.; Zoghbi, W. Comparison of adenosine echocardiography, with and without isometric handgrip, to exercise echocardiography in the detection of ischemia in patients with coronary artery disease. J. Am. Soc. Echocardiogr. 1996, 9, 33–43. [Google Scholar] [CrossRef]
- Meijer, P.; Oyen, W.J.; Dekker, D.; Broek, P.H.v.D.; Wouters, C.W.; Boerman, O.C.; Scheffer, G.J.; Smits, P.; Rongen, G.A. Rosuvastatin increases extracellular adenosine formation in humans in vivo: A new perspective on cardiovascular protection. Arter. Thromb. Vasc. Biol. 2009, 29, 963–968. [Google Scholar] [CrossRef] [PubMed]
- Som, A.; Sen, C.; Goswami, A. Prophylactic Amiodarone vs Dronedarone for Prevention of Perioperative Arrhythmias in off-pump Coronary Artery Bypass Grafting: A Pilot Randomized Controlled Trial. J. Perioper. Pract. 2017, 27, 9–15. [Google Scholar] [CrossRef] [PubMed]
- Tsanaxidis, N.; Aidonidis, I.; Hatziefthimiou, A.; Daskalopoulou, S.S.; Giamouzis, G.; Triposkiadis, F.; Skoularigis, I. Ranolazine Added to Amiodarone Facilitates Earlier Conversion of Atrial Fibrillation Compared to Amiodarone-Only Therapy. Pacing Clin. Electrophysiol. 2017, 40, 372–378. [Google Scholar] [CrossRef] [PubMed]
- Simopoulos, V.; Tagarakis, G.I.; Daskalopoulou, S.S.; Daskalopoulos, M.E.; Lenos, A.; Chryssagis, K.; Skoularingis, I.; Molyvdas, P.-A.; Tsilimingas, N.B.; Aidonidis, I. Ranolazine Enhances the Antiarrhythmic Activity of Amiodarone by Accelerating Conversion of New-Onset Atrial Fibrillation After Cardiac Surgery. Angiology 2014, 65, 294–297. [Google Scholar] [CrossRef]
- Cushing, D.J.; Adams, M.P.; Cooper, W.D.; Kowey, P.R.; Lipicky, R.J. Bioequivalence of 2 Intravenous Amiodarone Formulations in Healthy Participants. J. Clin. Pharmacol. 2009, 49, 407–415. [Google Scholar] [CrossRef]
- Kiran, U.; Selvaraj, T.; Das, S.; Chauhan, S.; Sahu, B.; Gharde, P. Effect of single intraoperative dose of amiodarone in patients with rheumatic valvular heart disease and atrial fibrillation undergoing valve replacement surgery. Ann. Card. Anaesth. 2009, 12, 10–16. [Google Scholar] [CrossRef]
- Turk, T.; Ata, Y.; Vural, H.; Ozkan, H.; Yavuz, S.; Ozyazicioglu, A. Intravenous and Oral Amiodarone for the Prevention of Postoperative Atrial Fibrillation in Patients Undergoing Off-Pump Coronary Artery Bypass Surgery. Heart Surg. Forum 2007, 10, 201–205. [Google Scholar] [CrossRef]
- Thomas, S.P.; Guy, D.; Wallace, E.; Crampton, R.; Kijvanit, P.; Eipper, V.; Ross, D.L.; Cooper, M.J. Rapid loading of sotalol or amiodarone for management of recent onset symptomatic atrial fibrillation: A randomized, digoxin-controlled trial. Am. Heart J. 2004, 147, E3. [Google Scholar] [CrossRef]
- Somberg, J.C.; Bailin, S.J.; Haffajee, C.I.; Paladino, W.P.; Kerin, N.Z.; Bridges, D.; Timar, S.; Molnar, J. Intravenous lidocaine versus intravenous amiodarone (in a new aqueous formulation) for incessant ventricular tachycardia. Am. J. Cardiol. 2002, 90, 853–859. [Google Scholar] [CrossRef]
- Rousseau, M.F.; Massart, P.-E.; van Eyll, C.; Etienne, J.; Ahn, S.; Ghadanfar, M.; Friedrich, T.; Pouleur, H. Cardiac and hemodynamic effects of intravenous dofetilide in patients with heart failure. Am. J. Cardiol. 2001, 87, 1250–1254. [Google Scholar] [CrossRef]
- Dörge, H.; Schoendube, F.A.; Schoberer, M.; Stellbrink, C.; Voss, M.; Messmer, B.J. Intraoperative amiodarone as prophylaxis against atrial fibrillation after coronary operations. Ann. Thorac. Surg. 2000, 69, 1358–1362. [Google Scholar] [CrossRef] [PubMed]
- Joseph, A.P.; Ward, M.R. A prospective, randomized controlled trial comparing the efficacy and safety of Sotalol, Amiodarone, and Digoxin for the reversion of new-onset atrial fibrillation. Ann. Emerg. Med. 2000, 36, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Levine, J.H.; Massumi, A.; Scheinman, M.M.; Winkle, R.A.; Platia, E.V.; Chilson, D.A.; Gomes, J.A.; Woosley, R.L. Intravenous amiodarone for recurrent sustained hypotensive ventricular tachyarrhythmias. Am. J. Cardiol. 1996, 27, 67–75. [Google Scholar] [CrossRef]
- Capucci, A.; Lenzi, T.; Boriani, G.; Trisolino, G.; Binetti, N.; Cavazza, M.; Fontana, G.; Magnani, B. Effectiveness of loading oral flecainide for converting recent-onset atrial fibrillation to sinus rhythm in patients without organic heart disease or with only systemic hypertension. Am. J. Cardiol. 1992, 70, 69–72. [Google Scholar] [CrossRef]
- McAlister, H.F.; Luke, R.A.; Whitlock, R.M.; Smith, W.M. Intravenous amiodarone bolus versus oral quinidine for atrial flutter and fibrillation after cardiac operations. J. Thorac. Cardiovasc. Surg. 1990, 99, 911–918. [Google Scholar] [CrossRef]
- Balik, M.; Maly, M.; Brozek, T.; Rulisek, J.; Porizka, M.; Sachl, R.; Otahal, M.; Brestovansky, P.; Svobodova, E.; Flaksa, M.; et al. Propafenone versus amiodarone for supraventricular arrhythmias in septic shock: A randomised controlled trial. Intensiv. Care Med. 2023, 49, 1283–1292. [Google Scholar] [CrossRef]
- Amar, D.; Zhang, H.; Chung, M.K.; Tan, K.S.; Desiderio, D.; Park, B.J.; Pedoto, A.; Roistacher, N.; Isbell, J.M.; Molena, D.; et al. Amiodarone with or without N-Acetylcysteine for the Prevention of Atrial Fibrillation after Thoracic Surgery: A Double-blind, Randomized Trial. Anesthesiology 2022, 136, 916–926. [Google Scholar] [CrossRef]
- Arvind, B.; Kothari, S.S.; Juneja, R.; Saxena, A.; Ramakrishnan, S.; Gupta, S.K.; Chowdhury, U.K.; Devagourou, V.; Talwar, S.; Hote, M.P.; et al. Ivabradine Versus Amiodarone in the Management of Postoperative Junctional Ectopic Tachycardia: A Randomized, Open-Label, Noninferiority Study. JACC Clin. Electrophysiol. 2021, 7, 1052–1060. [Google Scholar] [CrossRef]
- Yagdi, T.; Nalbantgil, S.; Ayik, F.; Apaydin, A.; Islamoglu, F.; Posacioglu, H.; Calkavur, T.; Atay, Y.; Buket, S. Amiodarone reduces the incidence of atrial fibrillation after coronary artery bypass grafting. J. Thorac. Cardiovasc. Surg. 2003, 125, 1420–1425. [Google Scholar] [CrossRef]
- Goldsmith, S.R.; Simon, A.B.; Miller, E. Effect of digitalis on norepinephrine kinetics in congestive heart failure. J. Am. Cardiol. 1992, 20, 858–863. [Google Scholar] [CrossRef]
- Simpson, C.S.; Ghali, W.A.; Sanfilippo, A.J.; Moritz, S.; Abdollah, H. Clinical assessment of clonidine in the treatment of new-onset rapid atrial fibrillation: A prospective, randomized clinical trial. Am. Heart J. 2001, 142, 19A–23A. [Google Scholar] [CrossRef] [PubMed]
- Amar, D.; Roistacher, N.; Burt, M.E.; Rusch, V.W.; Bains, M.S.; Leung, D.H.Y.; Downey, R.J.; Ginsberg, R.J. Effects of diltiazem versus digoxin on dysrhythmias and cardiac function after pneumonectomy. Ann. Thorac. Surg. 1997, 63, 1374–1381. [Google Scholar] [CrossRef]
- Schreck, D.M.; Rivera, A.R.; Tricarico, V.J. Emergency Management of Atrial Fibrillation and Flutter: Intravenous Diltiazem Versus Intravenous Digoxin. Ann. Emerg. Med. 1997, 29, 135–140. [Google Scholar] [CrossRef] [PubMed]
- Amar, D.; Roistacher, N.; Rusch, V.W.; Leung, D.H.; Ginsburg, I.; Zhang, H.; Bains, M.S.; Downey, R.J.; Korst, R.J.; Ginsberg, R.J. Effects of diltiazem prophylaxis on the incidence and clinical outcome of atrial arrhythmias after thoracic surgery. J. Thorac. Cardiovasc. Surg. 2000, 120, 790–798. [Google Scholar] [CrossRef] [PubMed]
- Malhotra, R.; Mishra, M.; Singh Kler, T.; Mohan Kohli, V.; Mehta, Y.; Trehan, N. Cardioprotective effects of diltiazem infusion in the perioperative period. Eur. J. Cardio-Thorac. Surg. 1997, 12, 420–427. [Google Scholar] [CrossRef]
- Goldenberg, I.F.; Lewis, W.R.; Dias, V.C.; Heywood, J.; Pedersen, W.R. Intravenous diltiazem for the treatment of patients with atrial fibrillation or flutter and moderate to severe congestive heart failure. Am. J. Cardiol. 1994, 74, 884–889. [Google Scholar] [CrossRef]
- Bonde, J.; Pedersen, L.E.; Angelo, H.R.; Svendsen, T.L.; Trap-Jensen, J.; Graudal, N.A.; Kampmann, J.P. Haemodynamic Effects of Disopyramide Using a New Model of Intravenous Administration. Pharmacol. Toxicol. 1987, 60, 179–183. [Google Scholar] [CrossRef]
- Maman, S.R.; Vargas, A.F.; Ahmad, T.A.; Miller, A.J.; Gao, Z.; Leuenberger, U.A.; Proctor, D.N.; Muller, M.D.; Vermeulen, T.D.; Boulet, L.M.; et al. Beta-1 vs. beta-2 adrenergic control of coronary blood flow during isometric handgrip exercise in humans. J. Appl. Physiol. 2017, 123, 337–343. [Google Scholar] [CrossRef]
- Muller, M.D.; Ahmad, T.A.; Pelaez, A.F.V.; Proctor, D.N.; Bonavia, A.S.; Luck, J.C.; Maman, S.R.; Ross, A.J.; Leuenberger, U.A.; McQuillan, P.M. Esmolol infusion versus propranolol infusion: Effects on heart rate and blood pressure in healthy volunteers. J. Appl. Physiol. 2017, 122, 511. [Google Scholar] [CrossRef]
- Hassan, S.; Ahmad, S.; Kamalakannan, D.; Khoury, R.; Kakish, E.; Maria, V.; Ahmed, S.; Pires, L.A.; Kronick, S.L.; Oral, H.; et al. Conversion of Atrial Fibrillation to Sinus Rhythm During Treatment with Intravenous Esmolol or Diltiazem: A Prospective, Randomized Comparison. J. Cardiovasc. Pharmacol. Ther. 2007, 12, 227–231. [Google Scholar] [CrossRef]
- Williams, M.J.A. Randomised comparison of the effects of nicardipine and esmolol on coronary artery wall stress: Implications for the risk of plaque rupture. Heart 2000, 84, 377–382. [Google Scholar] [CrossRef] [PubMed]
- Singh, P.P.; Dimich, I.; Sampson, I.; Sonnenklar, N. A comparison of esmolol and labetalol for the treatment of perioperative hypertension in geriatric ambulatory surgical patients. Can. J. Anaesth. 1992, 39, 559–562. [Google Scholar] [CrossRef] [PubMed]
- Gray, R.J.; Bateman, T.M.; Czer, L.S.; Conklin, C.; Matloff, J.M. Comparison of esmolol and nitroprusside for acute post-cardiac surgical hypertension. Am. J. Cardiol. 1987, 59, 887–891. [Google Scholar] [CrossRef]
- Sheppard, D.; DiStefano, S.; Byrd, R.C.; Eschenbacher, W.L.; Bell, V.; Steck, J.; Laddu, A. Effects of Esmolol on Airway Function in Patients with Asthma. J. Clin. Pharmacol. 1986, 26, 169–174. [Google Scholar] [CrossRef]
- Iskandrian, A.S.; Hakki, A.-H.; Laddu, A.; Steck, J.; Saunders, R.; Kane-Marsch, S.; Morganroth, J. Effects of intravenous infusion of esmolol and propranolol on biventricular performance at rest and during exercise as assessed by quantitative radionuclide angiography. Am. J. Cardiol. 1985, 55, 1287–1292. [Google Scholar] [CrossRef]
- Krumpl, G.; Ulč, I.; Trebs, M.; Kadlecová, P.; Hodisch, J.; Maurer, G.; Husch, B. Pharmacokinetics and Pharmacodynamics of Low-, Intermediate-, and High-Dose Landiolol and Esmolol During Long-Term Infusion in Healthy Whites. J. Cardiovasc. Pharmacol. 2018, 71, 137–146. [Google Scholar] [CrossRef]
- Kannankeril, P.J.; Moore, J.P.; Cerrone, M.; Priori, S.G.; Kertesz, N.J.; Ro, P.S.; Batra, A.S.; Kaufman, E.S.; Fairbrother, D.L.; Saarel, E.V.; et al. Efficacy of Flecainide in the Treatment of Catecholaminergic Polymorphic Ventricular Tachycardia: A Randomized Clinical Trial. JAMA Cardiol. 2017, 2, 759–766. [Google Scholar] [CrossRef]
- Singh, S.M.; D’Avila, A.; Kim, Y.-H.; Aryana, A.; Mangrum, J.M.; Michaud, G.F.; Dukkipati, S.R.; Barrett, C.D.; Heist, E.K.; Parides, M.K.; et al. The modified stepwise ablation guided by low-dose ibutilide in chronic atrial fibrillation trial (The MAGIC-AF Study). Eur. Heart J. 2016, 37, 1614–1621. [Google Scholar] [CrossRef]
- Sun, J.-L.; Guo, J.-H.; Zhang, N.; Zhang, H.-C.; Zhang, P. Clinical Comparison of Ibutilide and Propafenone for Converting Atrial Flutter. Cardiovasc. Drugs Ther. 2005, 19, 57–64. [Google Scholar] [CrossRef]
- Vos, M.A.; Golitsyn, S.R.; Stangl, K.; Ruda, M.Y.; Van Wijk, L.; Harry, J.D.; Perry, K.T.; Touboul, P.; Steinbeck, G.; Wellens, H.J.J. Superiority of ibutilide (a new class III agent) over DL-sotalol in converting atrial flutter and atrial fibrillation. Heart 1998, 79, 568–575. [Google Scholar] [CrossRef]
- Guo, G.B.-F.; Ellenbogen, K.A.; Wood, M.A.; Stambler, B.S. Conversion of atrial flutter by ibutilide is associated with increased atrial cycle length variability. J. Am. Cardiol. 1996, 27, 1083–1089. [Google Scholar] [CrossRef]
- Sharma, V.K.; Tan, B.Y.; Sim, M.Y.; Kulkarni, A.; Seow, P.A.; Hong, C.S.; Du, Z.; Wong, L.Y.; Chen, J.; Chee, E.Y.; et al. Rationale and design of a randomized trial of early intensive blood pressure lowering on cerebral perfusion parameters in thrombolysed acute ischemic stroke patients. Medicine 2018, 97, e12721. [Google Scholar] [CrossRef] [PubMed]
- Shi, D.-D.; Yang, F.-Z.; Zhou, L.; Wang, N. Oral nifedipine vs. intravenous labetalol for treatment of pregnancy-induced severe pre-eclampsia. J. Clin. Pharm. Ther. 2016, 41, 657–661. [Google Scholar] [CrossRef] [PubMed]
- Chung, K.S.; Sinatra, R.S.; Chung, J.H. The effect of an intermediate dose of labetalol on heart rate and blood pressure responses to laryngoscopy and intubation. J. Clin. Anesth. 1992, 4, 11–15. [Google Scholar] [CrossRef]
- Maharaj, R.J.; Thompson, M.; Brock-Utne, J.G.; Williamson, R.; Downing, J.W. Treatment of hypertension following endotracheal intubation. A study comparing the efficacy of labetalol, practolol and placebo. S. Afr. Med. J. 1983, 63, 691–694. Available online: https://pubmed.ncbi.nlm.nih.gov/6342168/ (accessed on 7 November 2023).
- Ferraris, A.; Jacquet-Lagrèze, M.; Cazenave, L.; Fornier, W.; Jalalzai, W.; Rousseau-Saine, N.; Pozzi, M.; Fellahi, J.-L. Effects of Landiolol on Macrocirculatory Parameters and Left and Right Ventricular Performances Following Cardiac Surgery: A Randomized Controlled Trial. J. Cardiothorac. Vasc. Anesth. 2022, 36, 2864–2869. [Google Scholar] [CrossRef]
- Kakihana, Y.; Nishida, O.; Taniguchi, T.; Okajima, M.; Morimatsu, H.; Ogura, H.; Yamada, Y.; Nagano, T.; Morishima, E.; Matsuda, N. Efficacy and safety of landiolol, an ultra-short-acting β1-selective antagonist, for treatment of sepsis-related tachyarrhythmia (J-Land 3S): A multicentre, open-label, randomised controlled trial. Lancet Respir. Med. 2020, 8, 863–872. [Google Scholar] [CrossRef]
- Krumpl, G.; Ulč, I.; Trebs, M.; Kadlecová, P.; Hodisch, J.; Maurer, G.; Husch, B. Pharmacodynamic and -kinetic Behavior of Low-, Intermediate-, and High-Dose Landiolol During Long-Term Infusion in Whites. J. Cardiovasc. Pharmacol. 2017, 70, 42–51. [Google Scholar] [CrossRef]
- Horikoshi, Y.; Goyagi, T.; Kudo, R.; Kodama, S.; Horiguchi, T.; Nishikawa, T. The suppressive effects of landiolol administration on the occurrence of postoperative atrial fibrillation and tachycardia, and plasma IL-6 elevation in patients undergoing esophageal surgery: A randomized controlled clinical trial. J. Clin. Anesth. 2017, 38, 111–116. [Google Scholar] [CrossRef]
- Pharand, C.; Kluger, J.; O’Rangers, E.; Ujhelyi, M.; Fisher, J.; Chow, M. Lidocaine prophylaxis for fatal ventricular arrhythmias after acute myocardial infarction. Clin. Pharmacol. Ther. 1995, 57, 471–478. [Google Scholar] [CrossRef]
- Manolis, A.S.; Smith, E.; Payne, D.; Rastegar, H.; Cleveland, R.; Estes, N.A.M. Randomized double-blind study of intravenous tocainide versus lidocaine for suppression of ventricular arrhythmias after cardiac surgery. Clin. Cardiol. 1990, 13, 177–181. [Google Scholar] [CrossRef] [PubMed]
- Paula-Garcia, W.N.; Oliveira-Paula, G.H.; de Boer, H.D.; Garcia, L.V. Lidocaine combined with magnesium sulfate preserved hemodynamic stability during general anesthesia without prolonging neuromuscular blockade: A randomized, double-blind, controlled trial. BMC Anesthesiol. 2021, 21, 1–8. [Google Scholar] [CrossRef]
- Singh, S.; Chouhan, R.S.; Bindra, A.; Radhakrishna, N. Comparison of effect of dexmedetomidine and lidocaine on intracranial and systemic hemodynamic response to chest physiotherapy and tracheal suctioning in patients with severe traumatic brain injury. J. Anesth. 2018, 32, 518–523. [Google Scholar] [CrossRef] [PubMed]
- Firouzi, A.; Maadani, M.; Kiani, R.; Shakerian, F.; Sanati, H.R.; Zahedmehr, A.; Nabavi, S.; Heidarali, M. Intravenous magnesium sulfate: New method in prevention of contrast-induced nephropathy in primary percutaneous coronary intervention. Int. Urol. Nephrol. 2015, 47, 521–525. [Google Scholar] [CrossRef]
- Clemente-Moragón, A.; Martínez-Milla, J.; Oliver, E.; Santos, A.; Flandes, J.; Fernández, I.; Rodríguez-González, L.; del Castillo, C.S.; Ioan, A.-M.; López-Álvarez, M.; et al. Metoprolol in Critically Ill Patients With COVID-19. J. Am. Cardiol. 2021, 78, 1001–1011. [Google Scholar] [CrossRef]
- Díaz-Munoz, R.; Valle-Caballero, M.J.; Sanchez-Gonzalez, J.; Pizarro, G.; García-Rubira, J.C.; Escalera, N.; Fuster, V.; Fernández-Jiménez, R.; Ibanez, B. Intravenous metoprolol during ongoing STEMI ameliorates markers of ischemic injury: A METOCARD-CNIC trial electrocardiographic study. Basic Res. Cardiol. 2021, 116, 1–15. [Google Scholar] [CrossRef]
- Fabris, E.; Hermanides, R.; Roolvink, V.; Ibanez, B.; Ottervanger, J.P.; Pizarro, G.; van Royen, N.; Mateos-Rodriguez, A.; Dambrink, J.H.; Albarran, A.; et al. Original research: Beta-blocker effect on ST-segment: A prespecified analysis of the EARLY-BAMI randomised trial. Open Heart 2020, 7, e001316. [Google Scholar] [CrossRef]
- Podlesnikar, T.; Pizarro, G.; Fernández-Jiménez, R.; Montero-Cabezas, J.M.; Sánchez-González, J.; Bucciarelli-Ducci, C.; Marsan, N.A.; Fras, Z.; Bax, J.J.; Fuster, V.; et al. Five-Year Outcomes and Prognostic Value of Feature-Tracking Cardiovascular Magnetic Resonance in Patients Receiving Early Prereperfusion Metoprolol in Acute Myocardial Infarction. Am. J. Cardiol. 2020, 133, 39–47. [Google Scholar] [CrossRef]
- Podlesnikar, T.; Pizarro, G.; Fernández-Jiménez, R.; Montero-Cabezas, J.M.; Greif, N.; Sánchez-González, J.; Bucciarelli-Ducci, C.; Marsan, N.A.; Fras, Z.; Bax, J.J.; et al. Left ventricular functional recovery of infarcted and remote myocardium after ST-segment elevation myocardial infarction (METOCARD-CNIC randomized clinical trial substudy). J. Cardiovasc. Magn. Reson. 2020, 22, 44. [Google Scholar] [CrossRef]
- García-Ruiz, J.M.; Fernández-Jiménez, R.; García-Alvarez, A.; Pizarro, G.; Galán-Arriola, C.; Fernández-Friera, L.; Mateos, A.; Nuno-Ayala, M.; Aguero, J.; Sánchez-González, J.; et al. Impact of the Timing of Metoprolol Administration During STEMI on Infarct Size and Ventricular Function. J. Am. Cardiol. 2016, 67, 2093–2104. [Google Scholar] [CrossRef]
- Roolvink, V.; Ibáñez, B.; Ottervanger, J.P.; Pizarro, G.; van Royen, N.; Mateos, A.; Dambrink, J.-H.E.; Escalera, N.; Lipsic, E.; Albarran, A.; et al. Early Intravenous Beta-Blockers in Patients With ST-Segment Elevation Myocardial Infarction Before Primary Percutaneous Coronary Intervention. J. Am. Cardiol. 2016, 67, 2705–2715. [Google Scholar] [CrossRef] [PubMed]
- Roolvink, V.; Rasoul, S.; Ottervanger, J.P.; Dambrink, J.-H.E.; Lipsic, E.; van der Horst, I.C.; de Smet, B.; Kedhi, E.; Gosselink, A.M.; Piek, J.J.; et al. Rationale and design of a double-blind, multicenter, randomized, placebo-controlled clinical trial of early administration of intravenous β-blockers in patients with ST-elevation myocardial infarction before primary percutaneous coronary intervention: EARLY β-blocker Administration before primary PCI in patients with ST-elevation Myocardial Infarction trial. Am. Heart J. 2014, 168, 661–666. [Google Scholar] [CrossRef] [PubMed]
- Pizarro, G.; Fernández-Friera, L.; Fuster, V.; Fernández-Jiménez, R.; García-Ruiz, J.M.; García-Álvarez, A.; Mateos, A.; Barreiro, M.V.; Escalera, N.; Rodriguez, M.D.; et al. Long-Term Benefit of Early Pre-Reperfusion Metoprolol Administration in Patients With Acute Myocardial Infarction: Results From the METOCARD-CNIC Trial (Effect of Metoprolol in Cardioprotection During an Acute Myocardial Infarction). J. Am. Cardiol. 2014, 63, 2356–2362. [Google Scholar] [CrossRef] [PubMed]
- Ibanez, B.; Macaya, C.; Sánchez-Brunete, V.; Pizarro, G.; Fernández-Friera, L.; Mateos, A.; Fernández-Ortiz, A.; García-Álvarez, A.; Iñiguez, A.; Jiménez-Borreguero, J.; et al. Effect of Early Metoprolol on Infarct Size in ST-Segment–Elevation Myocardial Infarction Patients Undergoing Primary Percutaneous Coronary Intervention. Circulation 2013, 128, 1495–1503. [Google Scholar] [CrossRef]
- Arsura, E.; Lefkin, A.S.; Scher, D.L.; Solar, M.; Tessler, S. A randomized, double-blind, placebo-controlled study of verapamil and metoprolol in treatment of multifocal atrial tachycardia. Am. J. Med. 1988, 85, 519–524. [Google Scholar] [CrossRef]
- Löfdahl, C.-G.; Marlin, G.E.; Svedmyr, N. Pafenolol, a highly selective β1-adrenoceptor–antagonist, in asthmatic patients: Interaction with terbutaline. Clin. Pharmacol. Ther. 1983, 33, 1–9. [Google Scholar] [CrossRef]
- Karlsson, E.; Kinman, A.; Sonnhag, C. Comparative evaluation of intravenous phenytoin, procainamide and practolol in the acute treatment of ventricular arrhythmias. Eur. J. Clin. Pharmacol. 1977, 11, 1–6. [Google Scholar] [CrossRef]
- Bigelow, C.A.; Pan, S.; Overbey, J.R.; Stone, J. Propranolol for Induction of Labor in Nulliparas trial a double-blind, randomized, placebo-controlled trial. Am. J. Obstet. Gynecol. MFM 2021, 3, 100301. [Google Scholar] [CrossRef]
- Joubert, P.H.; Venter, C.P.; Wellstein, A. Ethnic differences in response to beta-blockade: Fact or artefact? A study with bisoprolol and propranolol. Eur. J. Clin. Pharmacol. 1988, 34, 363–368. [Google Scholar] [CrossRef]
- Vicente, J.; Johannesen, L.; Mason, J.W.; Crumb, W.J.; Pueyo, E.; Stockbridge, N.; Strauss, D.G. Comprehensive T wave Morphology Assessment in a Randomized Clinical Study of Dofetilide, Quinidine, Ranolazine, and Verapamil. J. Am. Heart Assoc. 2015, 4, e001615. [Google Scholar] [CrossRef]
- Lloyd, E.; Charles, R.; Gordon, G.; Adams, C.; Mabin, T.; Commerford, P.; Opie, L. Beta-blockade by sotalol in early myocardial-infarction decreases ventricular arrhythmias without increasing left-ventricular volume. S. Afr. Med. J. 1988, 74, 5–10. [Google Scholar] [PubMed]
- Tullo, N.G.; Landau, S.; Goldman, I.; Coutinho, N.; Somberg, J. A randomized comparative study of the electrophysiological and electrocardiographic effects of isradipine vs verapamil. Acta Anaesthesiol. Scand. 1993, 37, 43–47. [Google Scholar] [CrossRef]
- Xia, F.; Wang, S.; Chen, M.; Wang, X.; Feng, X.; Dong, J. Protective effect of Verapamil on hepatic ischemia–reperfusion injury during hepatectomy in the cirrhotic patients with hepatocellular carcinoma. Langenbeck’s Arch. Surg. 2009, 394, 1041–1046. [Google Scholar] [CrossRef]
- Popović, J.; Mitić, R.; Sabo, A.; Mikov, M.; Jakovljević, V.; Daković-Švajcer, K. Spline functions in convolutional modeling of verapamil bioavailability and bioequivalence. II: Study in healthy volunteers. Eur. J. Drug Metab. Pharmacokinet. 2006, 31, 87–96. [Google Scholar] [CrossRef]
- Dilger, K.; Eckhardt, K.; Hofmann, U.; Kucher, K.; Mikus, G.; Eichelbaum, M. Chronopharmacology of intravenous and oral modified release verapamil. Br. J. Clin. Pharmacol. 1999, 47, 413–419. [Google Scholar] [CrossRef]
- Weinstein, G.S.; Rao, P.S.; Tyras, D.H. Reduction of myocardial injury with verapamil before aortic cross-clamping. Ann. Thorac. Surg. 1990, 49, 419–423. [Google Scholar] [CrossRef]
- Roy, D.; Rowe, B.H.; Stiell, I.G.; Coutu, B.; Ip, J.H.; Phaneuf, D.; Lee, J.; Vidaillet, H.; Dickinson, G.; Grant, S.; et al. A randomized, controlled trial of RSD1235, a novel anti-arrhythmic agent, in the treatment of recent onset atrial fibrillation. J. Am. Cardiol. 2004, 44, 2355–2361. [Google Scholar] [CrossRef]
- Mader, T.J.; Smithline, H.A.; Durkin, L.; Scriver, G. A Randomized Controlled Trial of Intravenous Aminophylline for Atropine-resistant Out-of-hospital Asystolic Cardiac Arrest. Acad. Emerg. Med. 2003, 10, 192–197. [Google Scholar] [CrossRef]
- Pacher, R.; Globits, S.; Wutte, M.; Rodler, S.; Heinz, G.; Kreiner, G.; Radosztics, S.; Berger, R.; Presch, I.; Weber, H. Beneficial hemodynamic effects of prostaglandin Ex infusion in catecholamine-dependent heart failure: Results of a prospective, randomized, controlled study. Crit. Care Med. 1994, 22, 1084–1090. [Google Scholar] [CrossRef]
- Rich, M.W.; Woods, W.L.; Davila-Roman, V.G.; Morello, P.J.; Kurz, H.; Barbarash, R.; Spinner, L.; Sperry, J.; Beckham, V.; Coulter, L.; et al. A Randomized Comparison of Intravenous Amrinone Versus Dobutamine in Older Patients with Decompensated Congestive Heart Failure. J. Am. Geriatr. Soc. 1995, 43, 271–274. [Google Scholar] [CrossRef]
- de Franchis, R. Randomized controlled trial of desmopressin plus terlipressin vs. terlipressin alone for the treatment of acute variceal hemorrhage in cirrhotic patients: A multicenter, double-blind study. Hepatology 1993, 18, 1102–1107. [Google Scholar] [CrossRef] [PubMed]
- Teng, R.; Ma, P.D.M.; Butler, K. The effect of desmopressin on bleeding time and platelet aggregation in healthy volunteers administered ticagrelor. J. Clin. Pharm. Ther. 2014, 39, 186–191. [Google Scholar] [CrossRef] [PubMed]
- Yoo, H.; Iirola, T.; Vilo, S.; Manner, T.; Aantaa, R.; Lahtinen, M.; Scheinin, M.; Olkkola, K.T.; Jusko, W.J. Mechanism-based population pharmacokinetic and pharmacodynamic modeling of intravenous and intranasal dexmedetomidine in healthy subjects. Eur. J. Clin. Pharmacol. 2015, 71, 1197–1207. [Google Scholar] [CrossRef] [PubMed]
- Stanek, B.; Sturm, B.; Frey, B.; Hülsmann, M.; Bojic, A.; Berger, R.; Rödler, S.; Locker, G.; Grimm, M.; Laufer, G.; et al. Bridging to heart transplantation: Prostaglandin E1 versus prostacyclin versus dobutamine. J. Heart Lung Transplant. 1999, 18, 358–366. [Google Scholar] [CrossRef]
- ARISE Investigators; ANZICS Clinical Trials Group; Peake, S.L.; Delaney, A.; Bailey, M.; Bellomo, R.; Cameron, P.A.; Cooper, D.J.; Higgins, A.M.; Holdgate, A.; et al. Goal-Directed Resuscitation for Patients with Early Septic Shock. N. Engl. J. Med. 2014, 371, 1496–1506. [Google Scholar] [CrossRef]
- Hashim, T.; Sanam, K.; Revilla-Martinez, M.; Morgan, C.J.; Tallaj, J.A.; Pamboukian, S.V.; Loyaga-Rendon, R.Y.; George, J.F.; Acharya, D. Clinical Characteristics and Outcomes of Intravenous Inotropic Therapy in Advanced Heart Failure. J. Am. Cardiol. Heart Fail. 2015, 8, 880–886. [Google Scholar] [CrossRef]
- Yilmaz, M.B.; Yalta, K.; Yontar, C.; Karadas, F.; Erdem, A.; Turgut, O.O.; Yilmaz, A.; Tandogan, I. Levosimendan Improves Renal Function in Patients with Acute Decompensated Heart Failure: Comparison with Dobutamine. Cardiovasc. Drugs Ther. 2007, 21, 431–435. [Google Scholar] [CrossRef]
- Dominguez-Rodriguez, A.; Samimi-Fard, S.; Garcia-Gonzalez, M.J.; Abreu-Gonzalez, P. Effects of levosimendan versus dobutamine on left ventricular diastolic function in patients with cardiogenic shock after primary angioplasty. Int. J. Cardiol. 2008, 128, 214–217. [Google Scholar] [CrossRef]
- García-González, M.J.; Domínguez-Rodríguez, A.; Ferrer-Hita, J.J.; Abreu-González, P.; Muñoz, M.B. Cardiogenic shock after primary percutaneous coronary intervention: Effects of levosimendan compared with dobutamine on haemodynamics. Eur. J. Heart Fail. 2006, 8, 723–728. [Google Scholar] [CrossRef]
- Avgeropoulou, C.; Andreadou, I.; Markantonis-Kyroudis, S.; Demopoulou, M.; Missovoulos, P.; Androulakis, A.; Kallikazaros, I. The Ca2+-sensitizer levosimendan improves oxidative damage, BNP and pro-inflammatory cytokine levels in patients with advanced decompensated heart failure in comparison to dobutamine. Eur. J. Heart Fail. 2005, 7, 882–887. [Google Scholar] [CrossRef]
- Nanas, J.N.; Tsagalou, E.P.; Kanakakis, J.; Nanas, S.N.; Terrovitis, J.V.; Moon, T.; Anastasiou-Nana, M.I. Long-term Intermittent Dobutamine Infusion, Combined with Oral Amiodarone for End-Stage Heart Failure: A randomized double-blind study. Chest 2004, 125, 1198–1204. [Google Scholar] [CrossRef] [PubMed]
- Follath, F. Levosimendan in patients with low-output heart failure: Lessons from the LIDO trial. Ital. Heart J. 2003, 4, 34S–38S. [Google Scholar] [PubMed]
- López-Candales, A.; Vora, T.; Gibbons, W.; Carron, C.; Simmons, P.; Schwartz, J. Symptomatic improvement in patients treated with intermittent infusion of inotropes: A double-blind placebo controled pilot study. J. Med. 2002, 33, 129–146. [Google Scholar] [PubMed]
- Silver, M.A.; Horton, D.P.; Ghali, J.K.; Elkayam, U. Effect of nesiritide versus dobutamine on short-term outcomes in the treatment of patients with acutely decompensated heart failure. J. Am. Cardiol. 2002, 39, 798–803. [Google Scholar] [CrossRef]
- Oliva, F.; Latini, R.; Politi, A.; Staszewsky, L.; Maggioni, A.P.; Nicolis, E.; Mauri, F. Intermittent 6-month low-dose dobutamine infusion in severe heart failure: DICE Multicenter Trial. Am. Heart J. 1999, 138, 247–253. [Google Scholar] [CrossRef]
- Elis, A.; Bental, T.; Kimchi, O.; Ravid, M.; Lishner, M. Intermittent dobutamine treatment in patients with chronic refractory congestive heart failure: A randomized, double-blind, placebo-controlled study. Clin. Pharmacol. Ther. 1998, 63, 682–685. [Google Scholar] [CrossRef]
- Miyagi, Y.; Sasayama, S.; Nakajima, H.; Fujita, M.; Asanoi, H. Comparative Hemodynamic Effects of Intravenous Dobutamine and Dibutyryl Cyclic AMP, a New Inotropic Agent, in Severe Congestive Heart Failure. J. Cardiovasc. Pharmacol. 1990, 15, 138–143. [Google Scholar] [CrossRef]
- Eichhorn, E.J.; Konstam, M.A.; Weiland, D.S.; Roberts, D.J.; Martin, T.T.; Stransky, N.B.; Salem, D.N. Differential effects of milrinone and dobutamine on right ventricular preload, afterload and systolic performance in congestive heart failure secondary to ischemic or idiopathic dilated cardiomyopathy. Am. J. Cardiol. 1987, 60, 1329–1333. [Google Scholar] [CrossRef]
- Biddle, T.L.; Benotti, J.R.; Creager, M.A.; Faxon, D.P.; Firth, B.G.; Fitzpatrick, P.G.; Konstam, M.A.; Krebs, C.; Walton, L.; Kershner, R.P.; et al. Comparison of intravenous milrinone and dobutamine for congestive heart failure secondary to either ischemic or dilated cardiomyopathy. Am. J. Cardiol. 1987, 59, 1345–1350. [Google Scholar] [CrossRef]
- Bain, A.R.; Dujic, Z.; Hoiland, R.L.; Barak, O.F.; Madden, D.; Drvis, I.; Stembridge, M.; MacLeod, D.B.; MacLeod, D.M.; Ainslie, P.N.; et al. Peripheral chemoreflex inhibition with low-dose dopamine: New insight into mechanisms of extreme apnea. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2015, 309, R1162–R1171. [Google Scholar] [CrossRef]
- Arino, M.; Barrington, J.P.; Morrison, A.L.; Gillies, D. Management of the changeover of inotrope infusions in children. Intensiv. Crit. Care Nurs. 2004, 20, 275–280. [Google Scholar] [CrossRef] [PubMed]
- Bellomo, R.; Chapman, M.; Finfer, S.; Hickling, K.; Myburgh, J. Low-dose dopamine in patients with early renal dysfunction: A placebo-controlled randomised trial. Australian and New Zealand Intensive Care Society (ANZICS) Clinical Trials Group. Lancet 2000, 356, 2139–2143. [Google Scholar] [CrossRef] [PubMed]
- Varriale, P.; Mossavi, A. The benefit of low-dose dopamine during vigorous diuresis for congestive heart failure associated with renal insufficiency: Does it protect renal function? Clin. Cardiol. 1997, 20, 627–630. [Google Scholar] [CrossRef] [PubMed]
- Weisberg, L.S.; Kurnik, P.B.; Kurnik, B.R. Risk of radiocontrast nephropathy in patients with and without diabetes mellitus. Kidney Int. 1994, 45, 259–265. [Google Scholar] [CrossRef]
- Baumgart, D.; Buck, T.; Leischik, R.; Oelert, H.; Farahati, J.; Reiners, C.; Erbel, R. Enoximon-echocardiography. A new diagnostic approach for the detection of viable myocardium comparison to dobutamin-echocardiography. Herz 1994, 19, 227–234. [Google Scholar]
- Smith, N.A.; Kates, R.E.; Lebsack, C.; Ruder, M.A.; Mead, R.H.; Bekele, T.; Okerholm, R.A.; Rubin, G.M.; Winkle, R.A. Clinical pharmacology of intravenous enoximone: Pharmacodynamics and pharmacokinetics in patients with heart failure. Am. Heart J. 1991, 122, 755–763. [Google Scholar] [CrossRef]
- Vedung, T.; Werner, M.; Ljung, B.-O.; Jorfeldt, L.; Henriksson, J. Blood Flow to the Extensor Carpi Radialis Brevis Muscle Following Adrenaline Infusion in Patients with Lateral Epicondylitis. J. Hand Surg. 2011, 36, 1974–1980. [Google Scholar] [CrossRef]
- van der Poll, T.; Coyle, S.M.; Barbosa, K.; Braxton, C.C.; Lowry, S.F. Epinephrine inhibits tumor necrosis factor-alpha and potentiates interleukin 10 production during human endotoxemia. J. Clin. Investig. 1996, 97, 713–719. [Google Scholar] [CrossRef]
- Sharrock, N.E.; Mineo, R.; Go, G. The effect of cardiac output on intraoperative blood loss during total hip arthroplasty. Reg. Anesth. Pain Med. 1993, 18, 24–29. [Google Scholar]
- Sharrock, N.E.; Go, G.; Mineo, R.; Harpel, P.C. The Hemodynamic and Fibrinolytic Response to Low Dose Epinephrine and Phenylephrine Infusions During Total Hip Replacement Under Epidural Anesthesia. Thromb. Haemost. 1992, 68, 436–441. [Google Scholar] [CrossRef]
- Udy, A.A.; Finnis, M.; Jones, D.; Delaney, A.; Macdonald, S.; Bellomo, R.; Peake, S. Incidence, Patient Characteristics, Mode of Drug Delivery, and Outcomes of Septic Shock Patients Treated with Vasopressors in the Arise Trial. Shock 2019, 52, 400–407. [Google Scholar] [CrossRef] [PubMed]
- Simonneau, G.; Rubin, L.J.; Galiè, N.; Barst, R.J.; Fleming, T.R.; Frost, A.; Engel, P.; Kramer, M.R.; Serdarevic-Pehar, M.; Layton, G.R.; et al. Long-term sildenafil added to intravenous epoprostenol in patients with pulmonary arterial hypertension. J. Heart Lung Transplant. 2014, 33, 689–697. [Google Scholar] [CrossRef] [PubMed]
- Sitbon, O.; Delcroix, M.; Bergot, E.; Boonstra, A.B.; Granton, J.; Langleben, D.; Subías, P.E.; Galiè, N.; Pfister, T.; Lemarié, J.-C.; et al. EPITOME-2: An open-label study assessing the transition to a new formulation of intravenous epoprostenol in patients with pulmonary arterial hypertension. Am. Heart J. 2014, 167, 210–217. [Google Scholar] [CrossRef]
- Badesch, D.B.; Tapson, V.F.; McGoon, M.D.; Brundage, B.H.; Rubin, L.J.; Wigley, F.M.; Rich, S.; Barst, R.J.; Barrett, P.S.; Kral, K.M.; et al. Continuous Intravenous Epoprostenol for Pulmonary Hypertension Due to the Scleroderma Spectrum of Disease: A randomized, controlled trial. Ann. Intern. Med. 2000, 132, 425–434. [Google Scholar] [CrossRef]
- Barst, R.J.; Rubin, L.J.; Long, W.A.; McGoon, M.D.; Rich, S.; Badesch, D.B.; Groves, B.M.; Tapson, V.F.; Bourge, R.C.; Brundage, B.H.; et al. A Comparison of Continuous Intravenous Epoprostenol (Prostacyclin) with Conventional Therapy for Primary Pulmonary Hypertension. N. Engl. J. Med. 1996, 51, 993. [Google Scholar] [CrossRef]
- Sueta, C.A.; Gheorghiade, M.; Adams, K.F.; Bourge, R.C.; Murali, S.; Uretsky, B.F.; Pritzker, M.R.; McGoon, M.D.; Butman, S.M.; Grossman, S.H.; et al. Safety and efficacy of epoprostenol in patients with severe congestive heart failure. Am. J. Cardiol. 1995, 75, 34A–43A. [Google Scholar] [CrossRef]
- Lichstein, E.; Mendizabal, R.; Theroux, P.; Muller, O.; Willerson, J.T.; Rapaport, E.; Molk, B.L.; Linet, O.I. Epoprostenol (Prostacyclin) in Unstable Angina. J. Clin. Pharmacol. 1988, 28, 300–305. [Google Scholar] [CrossRef]
- Donadoni, M.; Calloni, M.; Romano, M.E.; Mutti, A.; Bartoli, A.; La Cava, L.; Celano, R.; Urso, F.; Janu, V.P.; Foschi, A.; et al. Long peripheral catheters for intravenous infusions of iloprost or alprostadil therapy in rheumatologic outpatients. J. Vasc. Access 2024, 11297298241252896. [Google Scholar] [CrossRef]
- Chang, P.C.; van Veen, S.; van der Krogt, J.A.; Vermeij, P.; van Brummelen, P. β1Adrenoceptor Selectivity of Single Oral Doses of Bisoprolol and Atenolol. J. Cardiovasc. Pharmacol. 1988, 12, 317–322. [Google Scholar] [CrossRef]
- Marlin, G.; Turner, P. Comparison of the beta2-adrenoceptor selectivity of rimiterol, salbutamol and isoprenaline by the intravenous route in man. Br. J. Clin. Pharmacol. 1975, 2, 41–48. [Google Scholar] [CrossRef]
- Antila, S.; Lehtonen, L.; Sandell, E.-P.; Eha, J.; Heinpalu, M.; Loogna, I.; Mesikepp, A. Haemodynamic interactions of a new calcium sensitizing drug levosimendan and captopril. Eur. J. Clin. Pharmacol. 1996, 49, 451–458. [Google Scholar] [CrossRef] [PubMed]
- García-González, M.J.; Perona, A.A.; Padron, A.L.; Rull, J.L.M.; Martínez-Sellés, M.; Martin, M.d.M.; Díaz, J.L.; Fernandez, S.L.; Oficialdegui, P.O.; Sosa, A.J. Efficacy and safety of intermittent repeated levosimendan infusions in advanced heart failure patients: The LAICA study. ESC Heart Fail. 2021, 8, 4820–4831. [Google Scholar] [CrossRef] [PubMed]
- Burkhoff, D.; Borlaug, B.A.; Shah, S.J.; Zolty, R.; Tedford, R.J.; Thenappan, T.; Zamanian, R.T.; Mazurek, J.A.; Rich, J.D.; Simon, M.A.; et al. Levosimendan Improves Hemodynamics and Exercise Tolerance in PH-HFpEF: Results of the Randomized Placebo-Controlled HELP Trial. Heart Fail. 2021, 9, 360–370. [Google Scholar] [CrossRef]
- Abril-Molina, A.; Gómez-Luque, J.M.; Perin, F.; Esteban-Molina, M.; Ferreiro-Marzal, A.; Fernandez-Guerrero, C.; Ocete-Hita, E. Effect of Preoperative Infusion of Levosimendan on Biomarkers of Myocardial Injury and Haemodynamics After Paediatric Cardiac Surgery: A Randomised Controlled Trial. Drugs RD 2021, 21, 79–89. [Google Scholar] [CrossRef]
- Bharathi, K.; Kundra, T.S.; Nagaraja, P.; Kaur, P.; Manjunatha, N. Inhaled levosimendan versus intravenous levosimendan in patients with pulmonary hypertension undergoing mitral valve replacement. Ann. Card. Anaesth. 2018, 21, 328–332. [Google Scholar] [CrossRef]
- Najjar, E.; Stålhberg, M.; Hage, C.; Ottenblad, E.; Manouras, A.; Löfman, I.H.; Lund, L.H. Haemodynamic effects of levosimendan in advanced but stable chronic heart failure. ESC Heart Fail. 2018, 5, 302–308. [Google Scholar] [CrossRef]
- Mehta, R.H.; Leimberger, J.D.; van Diepen, S.; Meza, J.; Wang, A.; Jankowich, R.; Hay, D.; Fremes, S.; Duncan, A.; Soltesz, E.G.; et al. Levosimendan in Patients with Left Ventricular Dysfunction Undergoing Cardiac Surgery. N. Engl. J. Med. 2017, 376, 2032–2042. [Google Scholar] [CrossRef]
- Mehta, R.H.; Van Diepen, S.; Meza, J.; Bokesch, P.; Leimberger, J.D.; Tourt-Uhlig, S.; Swartz, M.; Parrotta, J.; Jankowich, R.; Hay, D.; et al. Levosimendan in patients with left ventricular systolic dysfunction undergoing cardiac surgery on cardiopulmonary bypass: Rationale and study design of the Levosimendan in Patients with Left Ventricular Systolic Dysfunction Undergoing Cardiac Surgery Requiring Cardiopulmonary Bypass (LEVO-CTS) trial. Am. Heart J. 2016, 182, 62–71. [Google Scholar] [CrossRef]
- Packer, M.; Colucci, W.; Fisher, L.; Massie, B.M.; Teerlink, J.R.; Young, J.; Padley, R.J.; Thakkar, R.; Delgado-Herrera, L.; Salon, J.; et al. Effect of Levosimendan on the Short-Term Clinical Course of Patients with Acutely Decompensated Heart Failure. JACC Heart Fail. 2013, 1, 103–111. [Google Scholar] [CrossRef]
- Mavrogeni, S.; Giamouzis, G.; Papadopoulou, E.; Thomopoulou, S.; Dritsas, A.; Athanasopoulos, G.; Adreanides, E.; Vassiliadis, I.; Spargias, K.; Panagiotakos, D.; et al. A 6-Month Follow-up of Intermittent Levosimendan Administration Effect on Systolic Function, Specific Activity Questionnaire, and Arrhythmia in Advanced Heart Failure. J. Card. Fail. 2007, 13, 556–559. [Google Scholar] [CrossRef]
- Lilleberg, J.; Laine, M.; Palkama, T.; Kivikko, M.; Pohjanjousi, P.; Kupari, M. Duration of the haemodynamic action of a 24-h infusion of levosimendan in patients with congestive heart failure. Eur. J. Heart Fail. 2007, 9, 75–82. [Google Scholar] [CrossRef] [PubMed]
- Morelli, A.; Teboul, J.-L.; Maggiore, S.M.; Vieillard-Baron, A.; Rocco, M.; Conti, G.; De Gaetano, A.; Picchini, U.; Orecchioni, A.; Carbone, I.; et al. Effects of levosimendan on right ventricular afterload in patients with acute respiratory distress syndrome: A pilot study. Crit. Care Med. 2006, 34, 2287–2293. [Google Scholar] [CrossRef] [PubMed]
- De Luca, L.; Proietti, P.; Celotto, A.; Bucciarelli-Ducci, C.; Benedetti, G.; Di Roma, A.; Sardella, G.; Genuini, I.; Fedele, F. Levosimendan improves hemodynamics and coronary flow reserve after percutaneous coronary intervention in patients with acute myocardial infarction and left ventricular dysfunction. Am. Heart J. 2005, 150, 563–568. [Google Scholar] [CrossRef]
- Ukkonen, H.; Saraste, M.; Akkila, J.; Knuuti, J.; Karanko, M.; Iida, H.; Lehikoinen, P.; Någren, K.; Lehtonen, L.; Voipio-Pulkki, L. Myocardial efficiency during levosimendan infusion in congestive heart failure. Clin. Pharmacol. Ther. 2000, 68, 522–531. [Google Scholar] [CrossRef]
- Nieminen, M.S.; Akkila, J.; Hasenfuss, G.; Kleber, F.X.; Lehtonen, L.A.; Mitrovic, V.; Nyquist, O.; Remme, W.J. Hemodynamic and neurohumoral effects of continuous infusion of levosimendan in patients with congestive heart failure. J. Am. Cardiol. 2000, 36, 1903–1912. [Google Scholar] [CrossRef]
- Slawsky, M.T.; Colucci, W.S.; Gottlieb, S.S.; Greenberg, B.H.; Haeusslein, E.; Hare, J.; Hutchins, S.; Leier, C.V.; LeJemtel, T.H.; Loh, E.; et al. Acute Hemodynamic and Clinical Effects of Levosimendan in Patients with Severe Heart Failure. Circulation 2000, 102, 2222–2227. [Google Scholar] [CrossRef]
- Gomes, U.C.; Cleland, J.G. Heart failure update. Eur. J. Heart Fail. 1999, 1, 301–302. [Google Scholar] [CrossRef]
- Antila, S.; Järvinen, A.; Akkila, J.; Honkanen, T.; Karlsson, M.; Lehtonen, L. Studies on psychomotoric effects and pharmacokinetic interactions of the new calcium sensitizing drug levosimendan and ethanol. Arzneim.-Forsch. 1997, 47, 816–820. [Google Scholar]
- Lilleberg, J.; Sundberg, S.; Nieminen, M.S. Dose-range study of a new calcium sensitizer, levosimendan, in patients with left ventricular dysfunction. J. Cardiovasc. Pharmacol. 1995, 26 (Suppl. S1), 63–69. [Google Scholar] [CrossRef]
- Jones, S.J.; Scott, D.A.; Watson, R.; Morrison, W.A. Milrinone Does Not Improve Free Flap Survival in Microvascular Surgery. Anaesth. Intensiv. Care 2007, 35, 720–725. [Google Scholar] [CrossRef]
- Felker, G.; Leimberger, J.D.; Califf, R.M.; Cuffe, M.S.; Massie, B.M.; Adams, K.F.; Gheorghiade, M.; O’Connor, C.M. Risk stratification after hospitalization for decompensated heart failure. J. Card. Fail. 2004, 10, 460–466. [Google Scholar] [CrossRef]
- Benza, R.L.; Tallaj, J.A.; Felker, G.; Zabel, K.; Kao, W.; Bourge, R.C.; Pearce, D.; Leimberger, J.D.; Borzak, S.; O’Connor, C.M.; et al. The impact of arrhythmias in acute heart failure. J. Card. Fail. 2004, 10, 279–284. [Google Scholar] [CrossRef] [PubMed]
- Möllhoff, T.; Schmidt, C.; Van Aken, H.; Berendes, E.; Buerkle, H.; Marmann, P.; Reinbold, T.; Prenger-Berninghoff, R.; Tjan, T.D.T.; Scheld, H.H.; et al. Myocardial ischaemia in patients with impaired left ventricular function undergoing coronary artery bypass grafting—Milrinone versus nifedipine. Eur. J. Anaesthesiol. 2002, 19, 796–802. [Google Scholar] [CrossRef] [PubMed]
- Cuffe, M.S.; Califf, R.M.; Adams, J.K.F.; Benza, R.; Bourge, R.; Colucci, W.S.; Massie, B.M.; O’Connor, C.M.; Pina, I.; Quigg, R.; et al. Short-term Intravenous Milrinone for Acute Exacerbation of Chronic Heart FailureA Randomized Controlled Trial. JAMA 2002, 287, 1541–1547. [Google Scholar] [CrossRef] [PubMed]
- El Adly, D.H.; Bazan, N.S.; El Borolossy, R.M.; Anan, I.F.; Fakher, M.A.; El Wakeel, L.M. Midodrine improves clinical and economic outcomes in patients with septic shock: A randomized controlled clinical trial. Ir. J. Med. Sci. 2022, 191, 2785–2795. [Google Scholar] [CrossRef]
- Aykanat, V.M.; Myles, P.S.; Weinberg, L.; Burrell, A.; Bellomo, R. Low-Concentration Norepinephrine Infusion for Major Surgery: A Safety and Feasibility Pilot Randomized Controlled Trial. Anesth. Analg. 2021, 134, 410–418. [Google Scholar] [CrossRef]
- LaPortaDr, R.F.; Arthur, G.R.; Datta, S. Phenylephrine in treating maternal hypotension due to spinal anaesthesia for caesarean delivery: Effects on neonatal catecholamine concentrations, acid base status and Apgar scores. Acta Anaesthesiol. Scand. 1995, 39, 901–905. [Google Scholar] [CrossRef]
- Snowdon, V.K.; Lachlan, N.J.; Hoy, A.M.; Hadoke, P.W.F.; Semple, S.I.; Patel, D.; Mungall, W.; Kendall, T.J.; Thomson, A.; Lennen, R.J.; et al. Serelaxin as a potential treatment for renal dysfunction in cirrhosis: Preclinical evaluation and results of a randomized phase 2 trial. PLoS Med. 2017, 14, e1002248. [Google Scholar] [CrossRef]
- Argenziano, M.; Choudhri, A.F.; Oz, M.C.; Rose, E.A.; Smith, C.R.; Landry, D.W. A prospective randomized trial of arginine vasopressin in the treatment of vasodilatory shock after left ventricular assist device placement. Circulation 1997, 96, II-286. [Google Scholar]
- Bleich, S.D.; Nichols, T.C.; Schumacher, R.R.; Cooke, D.H.; Tate, D.A.; Teichman, S.L. Effect of heparin on coronary arterial patency after thrombolysis with tissue plasminogen activator in acute myocardial infarction. Am. J. Cardiol. 1990, 66, 1412–1417. [Google Scholar] [CrossRef]
- Wang, X.; Song, L.; Yang, J.; Sun, L.; Moullaali, T.J.; Sandset, E.C.; Delcourt, C.; Lindley, R.I.; Robinson, T.G.; Minhas, J.S.; et al. Interaction of Blood Pressure Lowering and Alteplase Dose in Acute Ischemic Stroke: Results of the Enhanced Control of Hypertension and Thrombolysis Stroke Study. Cerebrovasc. Dis. 2020, 48, 207–216. [Google Scholar] [CrossRef] [PubMed]
- Song, L.; Wang, X.; Robinson, T.; Lindley, R.I.; Arima, H.; Lavados, P.M.; Chen, X.; Chalmers, J.; Anderson, C.S. Characteristics, management and response to alteplase in China versus non-China participants of the ENCHANTED trial. Stroke Vasc. Neurol. 2017, 2, 53–58. [Google Scholar] [CrossRef] [PubMed]
- Fischer, U.; Kaesmacher, J.; Strbian, D.; Eker, O.; Cognard, C.; Plattner, P.S.; Bütikofer, L.; Mordasini, P.; Deppeler, S.; Pereira, V.M.; et al. Thrombectomy alone versus intravenous alteplase plus thrombectomy in patients with stroke: An open-label, blinded-outcome, randomised non-inferiority trial. Lancet 2022, 400, 104–115. [Google Scholar] [CrossRef]
- Lyden, P.; Pryor, K.E.; Coffey, C.S.; Cudkowicz, M.M.; Conwit, R.; Jadhav, A.; Sawyer, R.N., Jr.; Claassen, J.; Adeoye, O.; Song, S.; et al. Final Results of the RHAPSODY Trial: A Multi-Center, Phase 2 Trial Using a Continual Reassessment Method to Determine the Safety and Tolerability of 3K3A-APC, A Recombinant Variant of Human Activated Protein C, in Combination with Tissue Plasminogen Activator, Mechanical Thrombectomy or both in Moderate to Severe Acute Ischemic Stroke. Ann. Neurol. 2019, 85, 125–136. [Google Scholar] [CrossRef]
- Ohman, E.M.; Kleiman, N.S.; Gacioch, G.; Worley, S.J.; Navetta, F.I.; Talley, J.D.; Anderson, H.V.; Ellis, S.G.; Cohen, M.D.; Spriggs, D.; et al. Combined Accelerated Tissue-Plasminogen Activator and Platelet Glycoprotein IIb/IIIa Integrin Receptor Blockade with Integrilin in Acute Myocardial Infarction: Results of a randomized, placebo-controlled, dose-ranging trial. IMPACT-AMI Investigators. Circulation 1997, 95, 846–854. [Google Scholar] [CrossRef]
- The GUSTO Investigators. An International Randomized Trial Comparing Four Thrombolytic Strategies for Acute Myocardial Infarction. N. Engl. J. Med. 1993, 329, 673–682. [Google Scholar] [CrossRef]
- Van de Werf, F. Lessons from the European Cooperative recombinant tissue-type plasminogen activator (rt-PA) versus placebo trial. J. Am. Cardiol. 1988, 12, A14–A19. [Google Scholar] [CrossRef]
- Rawles, J. Magnitude of benefit from earlier thrombolytic treatment in acute myocardial infarction: New evidence from Grampian region early anistreplase trial (GREAT). BMJ 1996, 312, 212–215. [Google Scholar] [CrossRef]
- Reynolds, H.R.; Farkouh, M.E.; Lincoff, A.M.; Hsu, A.; Swahn, E.; Sadowski, Z.P.; White, J.A.; Topol, E.J.; Hochman, J.S. Impact of Female Sex on Death and Bleeding After Fibrinolytic Treatment of Myocardial Infarction in GUSTO V. Arch. Intern. Med. 2007, 167, 2054–2060. [Google Scholar] [CrossRef]
- Qureshi, A.I.; Harris-Lane, P.; Kirmani, J.F.; Janjua, N.; Divani, A.A.; Mohammad, Y.M.; Suarez, J.I.; Montgomery, M.O. Intra-arterial Reteplase and Intravenous Abciximab in Patients with Acute Ischemic Stroke: An Open-label, Dose-ranging, Phase I Study. Neurosurgery 2006, 59, 789–797. [Google Scholar] [CrossRef]
- White, H.; Hirulog and Early Reperfusion or Occlusion (HERO)-2 Trial Investigators. Thrombin-specific anticoagulation with bivalirudin versus heparin in patients receiving fibrinolytic therapy for acute myocardial infarction: The HERO-2 randomised trial. Lancet 2001, 358, 1855–1863. [Google Scholar] [CrossRef] [PubMed]
- Van de Werf, F.; Topol, E.J.; Lee, K.L.; Woodlief, L.H.; Granger, C.B.; Armstrong, P.W.; Barbash, G.I.; Hampton, J.R.; Guerci, A.; Simes, R.J.; et al. Variations in patient management and outcomes for acute myocardial infarction in the United States and other countries. Results from the GUSTO trial. Global Utilization of Streptokinase and Tissue Plasminogen Activator for Occluded Coronary Arteries. JAMA 1995, 273, 1586–1591. [Google Scholar] [CrossRef] [PubMed]
- Sinnaeve, P.R.; Alexander, J.H.; Bogaerts, K.; Belmans, A.; Wallentin, L.; Armstrong, P.; Adgey, J.A.; Tendera, M.; Diaz, R.; Soares-Piegas, L.; et al. Efficacy of tenecteplase in combination with enoxaparin, abciximab, or unfractionated heparin: One-year follow-up results of the assessment of the safety of a new thrombolytic-3 (ASSENT-3) randomized trial in acute myocardial infarction. Am. Heart J. 2004, 147, 993–998. [Google Scholar] [CrossRef] [PubMed]
- Wallentin, L.; Goldstein, P.; Armstrong, P.; Granger, C.; Adgey, A.; Arntz, H.; Bogaerts, K.; Danays, T.; Lindahl, B.; Mäkijärvi, M.; et al. Efficacy and Safety of Tenecteplase in Combination With the Low-Molecular-Weight Heparin Enoxaparin or Unfractionated Heparin in the Prehospital Setting: The Assessment of the Safety and Efficacy of a New Thrombolytic Regimen (ASSENT)-3 PLUS randomized trial in acute myocardial infarction. Circulation 2003, 108, 135–142. [Google Scholar] [CrossRef]
- Schreiber, T.L.; Macina, G.; McNulty, A.; Bunnell, P.; Kikel, M.; Miller, D.H.; Devereux, R.B.; Tenney, R.; Cowley, M.; Zola, B. Urokinase plus heparin versus aspirin in unstable angina and non-Q-wave myocardial infarction. Am. J. Cardiol. 1989, 64, 840–844. [Google Scholar] [CrossRef]
- Dizon, J.M.; Brener, S.J.; Maehara, A.; Witzenbichler, B.; Biviano, A.; Godlewski, J.; Parise, H.; Dambrink, J.-H.; Mehran, R.; Gibson, C.M.; et al. Relationship between ST-segment resolution and anterior infarct size after primary percutaneous coronary intervention: Analysis from the INFUSE-AMI trial. Eur. Heart J. Acute Cardiovasc. Care 2014, 3, 78–83. [Google Scholar] [CrossRef]
- Singh, H.S.; Dangas, G.D.; Guagliumi, G.; Yu, J.; Witzenbichler, B.; Kornowski, R.; Grines, C.; Gersh, B.; Dudek, D.; Mehran, R.; et al. Comparison of Abciximab Versus Eptifibatide During Percutaneous Coronary Intervention in ST-Segment Elevation Myocardial Infarction (from the HORIZONS-AMI Trial). Am. J. Cardiol. 2012, 110, 940–947. [Google Scholar] [CrossRef]
- Thiele, H.; Wöhrle, J.; Hambrecht, R.; Rittger, H.; Birkemeyer, R.; Lauer, B.; Neuhaus, P.; Brosteanu, O.; Sick, P.; Wiemer, M.; et al. Intracoronary versus intravenous bolus abciximab during primary percutaneous coronary intervention in patients with acute ST-elevation myocardial infarction: A randomised trial. Lancet 2012, 379, 923–931. [Google Scholar] [CrossRef]
- Eitel, I.; Desch, S.; Schindler, K.; Fuernau, G.; Schuler, G.; Thiele, H. Aborted myocardial infarction in intracoronary compared with standard intravenous abciximab administration in patients undergoing primary percutaneous coronary intervention for ST-elevation myocardial infarction. Int. J. Cardiol. 2011, 153, 21–25. [Google Scholar] [CrossRef]
- Thiele, H.; Wöhrle, J.; Neuhaus, P.; Brosteanu, O.; Sick, P.; Prondzinsky, R.; Birkemeyer, R.; Wiemer, M.; Kerber, S.; Schuehlen, H.; et al. Intracoronary compared with intravenous bolus abciximab application during primary percutaneous coronary intervention: Design and rationale of the Abciximab Intracoronary versus intravenously Drug Application in ST-Elevation Myocardial Infarction (AIDA STEMI) trial. Am. Heart J. 2010, 159, 547–554. [Google Scholar] [CrossRef]
- Valgimigli, M.; Campo, G.; Percoco, G.; Bolognese, L.; Vassanelli, C.; Colangelo, S.; de Cesare, N.; Rodriguez, A.E.; Ferrario, M.; Moreno, R.; et al. Comparison of Angioplasty With Infusion of Tirofiban or Abciximab and With Implantation of Sirolimus-Eluting or Uncoated Stents for Acute Myocardial InfarctionThe MULTISTRATEGY Randomized Trial. JAMA 2008, 299, 1788–1799. [Google Scholar] [CrossRef] [PubMed]
- Kereiakes, D.J.; Runyon, J.P.; Kleiman, N.S.; Higby, N.A.; Anderson, L.C.; Hantsbarger, G.; McDonald, S.; Anders, R.J. Differential Dose-Response to Oral Xemilofiban After Antecedent Intravenous Abciximab: Administration for Complex Coronary Intervention. Circulation 1996, 94, 906–910. [Google Scholar] [CrossRef] [PubMed]
- Kario, K.; Kodama, K.; Koide, M.; Matsuo, T. Thrombin inhibition in the acute phase of ischaemic stroke using argatroban. Blood Coagul. Fibrinolysis 1995, 6, 423–427. [Google Scholar] [CrossRef]
- Linke, A.; Mbbs, J.C.; Sartori, S.; Lefevre, T.; van Belle, E.; Schaefer, U.; Tchetche, D.; Sardella, G.; Webb, J.; Colombo, A.; et al. Effect of valve design and anticoagulation strategy on 30-day clinical outcomes in transcatheter aortic valve replacement: Results from the BRAVO 3 randomized trial. Catheter. Cardiovasc. Interv. 2017, 90, 1016–1026. [Google Scholar] [CrossRef]
- White, H.D.; Bhatt, D.L.; Gibson, C.M.; Hamm, C.W.; Mahaffey, K.W.; Price, M.J.; Steg, P.G.; Stone, G.W.; Cortese, B.; Wilensky, M.; et al. Outcomes With Cangrelor Versus Clopidogrel on a Background of Bivalirudin: Insights from the CHAMPION PHOENIX (A Clinical Trial Comparing Cangrelor to Clopidogrel Standard Therapy in Subjects Who Require Percutaneous Coronary Intervention [PCI]). JACC Cardiovasc. Interv. 2015, 8, 424–433. [Google Scholar] [CrossRef]
- Cortese, B.; Picchi, A.; Micheli, A.; Ebert, A.G.; Parri, F.; Severi, S.; Limbruno, U. Comparison of Prolonged Bivalirudin Infusion Versus Intraprocedural in Preventing Myocardial Damage After Percutaneous Coronary Intervention in Patients with Angina Pectoris. Am. J. Cardiol. 2009, 104, 1063–1068. [Google Scholar] [CrossRef]
- Mehran, R.; Lansky, A.J.; Witzenbichler, B.; Guagliumi, G.; Peruga, J.Z.; Brodie, B.R.; Dudek, D.; Kornowski, R.; Hartmann, F.; Gersh, B.J.; et al. Bivalirudin in patients undergoing primary angioplasty for acute myocardial infarction (HORIZONS-AMI): 1-year results of a randomised controlled trial. Lancet 2009, 374, 1149–1159. [Google Scholar] [CrossRef]
- Doll, J.A.; Nikolsky, E.; Stone, G.W.; Mehran, R.; Lincoff, A.M.; Caixeta, A.; Mclaurin, B.; Cristea, E.; Fahy, M.; Kesanakurthy, V.; et al. Outcomes of Patients with Coronary Artery Perforation Complicating Percutaneous Coronary Intervention and Correlations with the Type of Adjunctive Antithrombotic Therapy: Pooled Analysis from REPLACE-2, ACUITY, and HORIZONS-AMI Trials. J. Interv. Cardiol. 2009, 22, 453–459. [Google Scholar] [CrossRef]
- Edmond, J.; French, J.; Aylward, P.; Wong, C.; Stewart, R.; Williams, B.; De Pasquale, C.; O’Connell, R.; Berg, K.V.D.; Van de Werf, F.; et al. Variations in the use of emergency PCI for the treatment of re-infarction following intravenous fibrinolytic therapy: Impact on outcomes in HERO-2. Eur. Heart J. 2007, 28, 1418–1424. [Google Scholar] [CrossRef]
- White, H.D. Direct thrombin inhibition and thrombolytic therapy: Rationale for the hirulog and early reperfusion/occlusion (HERO-2) trial. Am. J. Cardiol. 1998, 82, 57P–62P. [Google Scholar] [CrossRef]
- Cannon, C.P.; Maraganore, J.M.; Loscalzo, J.; McAllister, A.; Eddings, K.; George, D.; Selwyn, A.P.; Adelman, B.; Fox, I.; Braunwald, E.; et al. Anticoagulant effects of hirulog, ∗∗Hirulog™ brand direct thrombin inhibitor. a novel thrombin inhibitor, in patients with coronary artery disease. Am. J. Cardiol. 1993, 71, 778–782. [Google Scholar] [CrossRef] [PubMed]
- Green, C.L.; Whellan, D.J.; Lambe, L.; Bellibas, S.E.; Wijngaard, P.; Prats, J.; Krucoff, M.W. Electrocardiographic Safety of Cangrelor, a New Intravenous Antiplatelet Agent: A randomized, double-blind, placebo- and moxifloxacin-controlled thorough QT study. J. Cardiovasc. Pharmacol. 2013, 62, 466–478. [Google Scholar] [CrossRef]
- Steg, P.G.; Mehta, S.R.; Pollack, C.V.; Bode, C.; Gaudin, C.; Fanouillere, K.; Moryusef, A.; Wiviott, S.D.; Sabatine, M.S. Design and rationale of the Treatment of Acute Coronary Syndromes with Otamixaban trial: A double-blind triple-dummy 2-stage randomized trial comparing otamixaban to unfractionated heparin and eptifibatide in non–ST-segment elevation acute coronary syndromes with a planned early invasive strategy. Am. Heart J. 2012, 164, 817–824.e13. [Google Scholar] [CrossRef] [PubMed]
- Harrington, R.A.; Kleiman, N.S.; Kottke-Marchant, K.; Lincoff, A.; Tcheng, J.E.; Sigmon, K.N.; Joseph, D.; Rios, G.; Trainor, K.; Rose, D.; et al. Immediate and reversible platelet inhibition after intravenous administration of a peptide glycoprotein IIb/IIIa inhibitor during percutaneous coronary intervention. Am. J. Cardiol. 1995, 76, 1222–1227. [Google Scholar] [CrossRef] [PubMed]
- Cohen, M.; Gensini, G.F.; Maritz, F.; Gurfinkel, E.P.; Huber, K.; Timerman, A.; Krzeminska-Pakula, M.; Danchin, N.; White, H.D.; Santopinto, J.; et al. The safety and efficacy of subcutaneous enoxaparin versus intravenous unfractionated heparin and tirofiban versus placebo in the treatment of acute ST-segment elevation myocardial infarction patients ineligible for reperfusion (TETAMI): A randomized trial. J. Am. Cardiol. 2003, 42, 1348–1356. [Google Scholar] [CrossRef]
- Kereiakes, J.; Kleiman, N.S.; Ambrose, J.; Cohen, M.; Rodriguez, S.; Palabrica, T.; Herrmann, H.C.; Sutton, J.M.; Weaver, W.; McKee, D.B.; et al. Randomized, double-blind, placebo-controlled dose-ranging study of tirofiban (MK-383) platelet IIb/IIIa blockade in high risk patients undergoing coronary angioplasty. J. Am. Cardiol. 1996, 27, 536–542. [Google Scholar] [CrossRef]
- Peacock, W.F.; Chandra, A.; Char, D.; Collins, S.; Der Sahakian, G.; Ding, L.; Dunbar, L.; Fermann, G.; Fonarow, G.C.; Garrison, N.; et al. Clevidipine in acute heart failure: Results of the A Study of Blood Pressure Control in Acute Heart Failure—A Pilot Study (PRONTO). Am. Heart J. 2014, 167, 529–536. [Google Scholar] [CrossRef]
- Nielsen, B.N.; Anderson, B.J.; Falcon, L.; Henneberg, S.W.; Lauritsen, T.; Lomstein, E.; Ydemann, M.; Afshari, A. Pharmacokinetics of an intravenous bolus dose of clonidine in children undergoing surgery. Pediatr. Anesth. 2020, 30, 607–613. [Google Scholar] [CrossRef]
- Gupta, N.; Kumar, A.; Jain, A.; Hayaran, N.; Mishra, P. A Randomized Controlled Trial to Compare the Efficacy of Intravenous Dexmedetomidine and Clonidine as Adjuvants to Low Dose Opioid in Attenuation of Hemodynamic Response to Laryngoscopy and Tracheal Intubation. Mymensingh. Med. J. 2018, 27, 389–396. [Google Scholar]
- Perera, P.M.S.; Jayamanna, S.F.; Hettiarachchi, R.; Abeysinghe, C.; Karunatilake, H.; Dawson, A.H.; Buckley, N.A. A phase II clinical trial to assess the safety of clonidine in acute organophosphorus pesticide poisoning. Trials 2009, 10, 73. [Google Scholar] [CrossRef]
- Koppert, W.; Sittl, R.; Scheuber, K.; Alsheimer, M.; Schmelz, M.; Schüttler, J. Differential Modulation of Remifentanil-induced Analgesia and Postinfusion Hyperalgesia by S-Ketamine and Clonidine in Humans. Anesthesiology 2003, 99, 152–159. [Google Scholar] [CrossRef]
- Kumari, V.; Cotter, P.; Corr, P.J.; Gray, J.A.; Checkley, S.A. Effect of clonidine on the human acoustic startle reflex. Psychopharmacology 1996, 123, 353–360. [Google Scholar] [CrossRef] [PubMed]
- Warren, J.B.; Dollery, C.T.; Sciberras, D.; Goldberg, M.R. Assessment of MK-467, a peripheral α2-adrenergic receptor antagonist, with intravenous clonidine. Clin. Pharmacol. Ther. 1991, 50, 71–77. [Google Scholar] [CrossRef] [PubMed]
- Kallio, A.; Saraste, M.; Scheinin, M.; Hartiala, J.; Scheinin, H. Acute Hemodynamic Effects of Medetomidine and Clonidine in Healthy Volunteers: A Noninvasive Echocardiographic Study. J. Cardiovasc. Pharmacol. 1990, 16, 28–33. [Google Scholar] [CrossRef]
- Warren, J.B.; Dollery, C.T.; Fuller, R.W.; Williams, V.C.; Gertz, B.J. Assessment of MK-912, an α2-adrenoceptor antagonist, with use of intravenous clonidine. Clin. Pharmacol. Ther. 1989, 46, 103–109. [Google Scholar] [CrossRef]
- Späh, F.; Grosser, K.D. Treatment of Hypertensive Urgencies and Emergencies with Nitrendipine, Nifedipine, and Clonidine: Effect on blood pressure and heart rate. J. Cardiovasc. Pharmacol. 1988, 12, S154–S156. [Google Scholar] [CrossRef]
- Annane, D.; Bellissant, E.; Pussard, E.; Asmar, R.; Lacombe, F.; Lanata, E.; Madonna, O.; Safar, M.; Giudicelli, J.-F.; Gajdos, P. Placebo-Controlled, Randomized, Double-Blind Study of Intravenous Enalaprilat Efficacy and Safety in Acute Cardiogenic Pulmonary Edema. Circulation 1996, 94, 1316–1324. [Google Scholar] [CrossRef]
- Kiowski, W.; Beermann, J.; Rickenbacher, P.; Haemmerli, R.; Thomas, M.; Burkart, F.; Meinertz, T. Angiotensinergic versus nonangiotensinergic hemodynamic effects of converting enzyme inhibition in patients with chronic heart failure. Assessment by acute renin and converting enzyme inhibition. Circulation 1994, 90, 2748–2756. [Google Scholar] [CrossRef]
- Swedberg, K.; Held, P.; Kjekshus, J.; Rasmussen, K.; Rydén, L.; Wedel, H. Effects of the Early Administration of Enalapril on Mortality in Patients with Acute Myocardial Infarction — Results of the Cooperative New Scandinavian Enalapril Survival Study II (Consensus II). N. Engl. J. Med. 1992, 327, 678–684. [Google Scholar] [CrossRef]
- Konstam, M.A.; Kronenberg, M.W.; Udelson, J.E.; Metherall, J.; Dolan, N.; Edens, T.; Howe, D.; Yusuf, S.; Youngblood, M.; Toltsis, H. Effect of acute angiotensin converting enzyme inhibition on left ventricular filling in patients with congestive heart failure: Relation to right ventricular volume. Circulation 1990, 15, A210. [Google Scholar] [CrossRef]
- Stone, G.W.; McCullough, P.A.; Tumlin, J.A.; Lepor, N.E.; Madyoon, H.; Murray, P.; Wang, A.; Chu, A.A.; Schaer, G.L.; Stevens, M.; et al. Fenoldopam Mesylate for the Prevention of Contrast-Induced Nephropathy: A Randomized Controlled Trial. JAMA 2003, 290, 2284–2291. [Google Scholar] [CrossRef] [PubMed]
- Tumlin, J.A.; Dunbar, L.M.; Oparil, S.; Buckalew, V.; Ram, C.V.; Mathur, V.; Ellis, D.; McGuire, D.; Fellmann, J.; Luther, R.R.; et al. Fenoldopam, a Dopamine Agonist, for Hypertensive Emergency: A Multicenter Randomized Trial. Acad. Emerg. Med. 2000, 7, 653–662. [Google Scholar] [CrossRef]
- Reisin, E.; Huth, M.M.; Nguyen, B.P.; Weed, S.G.; Gonzalez, F.M. Intravenous fenoldopam versus sodium nitroprusside in patients with severe hypertension. Hypertension 1990, 15, I59–I62. [Google Scholar] [CrossRef] [PubMed]
- Aali, B.S.; Nejad, S.S. Nifedipine or hydralazine as a first-line agent to control hypertension in severe preeclampsia. Acta Obstet. Gynecol. Scand. 2002, 81, 25–30. [Google Scholar] [CrossRef] [PubMed]
- Duggan, P.M.; McCowan, L.M.E.; Stewart, A.W. Antihypertensive Drug Effects on Placental Flow Velocity Waveforms in Pregnant Women with Severe Hypertension. Aust. N. Z. J. Obstet. Gynaecol. 1992, 32, 335–338. [Google Scholar] [CrossRef]
- Adnot, S.; Defouilloy, C.; Brunbuisson, C.; Abrouk, F.; Piquet, J.; Lemaire, F. Hemodynamic Effects of Urapidil in Patients with Pulmonary Hypertension A Comparative Study with Hydralazine. Am. Rev. Respir. Dis. 1987, 135, 288–293. [Google Scholar] [CrossRef]
- De Ponti, C.; Bardi, U.; Marchetti, M. A comparative study of the hemodynamic effects of single intravenous doses of 3-hydrazino-6-[N,N-bis-(2-hydroxyethyl)-amino]-pyridazine-dihydrochloride and hydralazine. Arzneimittelforschung 1976, 26, 2089–2092. [Google Scholar]
- Li, Q.; Warren, A.D.; Qureshi, A.I.; Morotti, A.; Falcone, G.J.; Sheth, K.N.; Shoamanesh, A.; Dowlatshahi, D.; Viswanathan, A.; Goldstein, J.N. Ultra-Early Blood Pressure Reduction Attenuates Hematoma Growth and Improves Outcome in Intracerebral Hemorrhage. Ann. Neurol. 2020, 88, 388–395. [Google Scholar] [CrossRef]
- Halpern, N.A.; Alicea, M.; Krakoff, L.R.; Greenstein, R. Postoperative hypertension: A prospective, placebo-controlled, randomized, double-blind trial, with intravenous nicardipine hydrochloride. Angiology 1990, 41 Pt 2, 992–1004. [Google Scholar]
- Goldberg, M.E.; Clark, S.; Joseph, J.; Moritz, H.; Maguire, D.; Seltzer, J.L.; Turlapaty, P. Nicardipine versus placebo for the treatment of postoperative hypertension. Am. Heart J. 1990, 119, 446–450. [Google Scholar] [CrossRef]
- Wallin, J.D. Intravenous nicardipine for the treatment of severe hypertension. A double-blind, placebo-controlled multicenter trial. Arch. Intern. Med. 1989, 149, 2662–2669. [Google Scholar] [CrossRef] [PubMed]
- Bellet, M.; Sassano, P.; Guyenne, T.; Corvol, P.; Menard, J. Converting-enzyme inhibition buffers the counter-regulatory response to acute administration of nicardipine. Br. J. Clin. Pharmacol. 1987, 24, 465–472. [Google Scholar] [CrossRef] [PubMed]
- Tabone, X.; Funck-Brentano, C.; Billon, N.; Jaillon, P. Comparison of tolerance to intravenous nitroglycerin during nicorandil and intermittent nitroglycerin patch in healthy volunteers. Clin. Pharmacol. Ther. 1994, 56, 672–679. [Google Scholar] [CrossRef]
- Brown, H.E.; Freudenreich, O.; Fan, X.; Heard, S.O.; Goff, D.; Petrides, G.; Harrington, A.L.; Kane, J.M.; Judge, H.; Hoeppner, B.; et al. Efficacy and Tolerability of Adjunctive Intravenous Sodium Nitroprusside Treatment for Outpatients with Schizophrenia: A Randomized Clinical Trial. JAMA Psychiatry 2019, 76, 691–699. [Google Scholar] [CrossRef]
- Tran, D.; Lowy, A.; Howes, J.B.; Howes, L.G. Effects of cerivastatin on forearm vascular responses, blood pressure responsiveness and ambulatory blood pressure in type 2 diabetic men. Diabetes Obes. Metab. 2005, 7, 273–281. [Google Scholar] [CrossRef]
- Polak, K.; Dorner, G.; Kiss, B.; Polska, E.; Findl, O.; Rainer, G.; Eichler, H.-G.; Schmetterer, L. Evaluation of the Zeiss retinal vessel analyser. Br. J. Ophthalmol. 2000, 84, 1285–1290. [Google Scholar] [CrossRef]
- Hirschl, M.M.; Binder, M.; Bur, A.; Herkner, H.; Müllner, M.; Woisetschläger, C.; Laggner, A.N. Safety and efficacy of urapidil and sodium nitroprusside in the treatment of hypertensive emergencies. Intensive Care Med. 1997, 23, 885–888. [Google Scholar] [CrossRef]
- Scrivani, S.J.; Chaudry, A.; Maciewicz, R.J.; Keith, D.A. Chronic neurogenic facial pain: Lack of response to intravenous phentolamine. J. Oral Facial Pain Headache 1999, 13, 89–96. [Google Scholar]
- Wacker, J.; Werner, P.; Walter-Sack, I.; Bastert, G. Treatment of hypertension in patients with pre-eclampsia: A prospective parallel-group study comparing dihydralazine with urapidil. Nephrol. Dial. Transplant. 1998, 13, 318–325. [Google Scholar] [CrossRef]
- de Leeuw, P.W.; Birkenhäger, W.H. Acute responses to urapidil in hypertensive persons. Am. J. Cardiol. 1989, 64, D22–D24. [Google Scholar] [CrossRef]
- de Boer, A.; Stiekema, J.C.J.; Danhof, M.; Moolenaar, A.J.; Breimer, D.D. Interaction of ORG 10172, a low molecular weight heparinoid, and digoxin in healthy volunteers. Eur. J. Clin. Pharmacol. 1991, 41, 245–250. [Google Scholar] [CrossRef] [PubMed]
- Stiekema, J.; de Boer, A.; Danhof, M.; Kroon, C.; Broekmans, A.; van Dinther, T.; Voerman, J.; Breimer, D. Interaction of the Combined Medication with the New Low-Molecular-Weight Heparinoid Lomoparan® (Org 10172) and Acenocoumarol. Pathophysiol. Haemost. Thromb. 1990, 20, 136–146. [Google Scholar] [CrossRef] [PubMed]
- Albada, J.; Nieuwenhuis, H.K.; Sixma, J.J. Treatment of acute venous thromboembolism with low molecular weight heparin (Fragmin). Results of a double-blind randomized study. Circulation 1989, 80, 935–940. [Google Scholar] [CrossRef] [PubMed]
- Yip, H.-K.; Chang, H.-W.; Wu, C.-J.; Chen, M.-C.; Hang, C.-L.; Fang, C.-Y.; Hsieh, K.Y.-K. A Safe and Effective Regimen without Heparin Therapy after Successful Primary Coronary Stenting in Patients with Acute Myocardial Infarction. Jpn. Heart J. 2000, 41, 697–711. [Google Scholar] [CrossRef]
- Veiga, F.; Escriba, A.; Maluenda, M.P.; Rubio, M.L.; Margalet, I.; Lezana, A.; Gallego, J.; Ribera, J.M. Low molecular weight heparin (enoxaparin) versus oral anticoagulant therapy (acenocoumarol) in the long-term treatment of deep venous thrombosis in the elderly: A randomized trial. Thromb. Haemost. 2000, 84, 559–564. [Google Scholar]
- Hull, R.D. Low-Molecular-Weight Heparin vs. Heparin in the Treatment of Patients with Pulmonary Embolism. Arch. Intern. Med. 2000, 160, 229–236. [Google Scholar] [CrossRef]
- Braga, J.C.; Esteves, F.P.; Esteves, J.P.; Latado, A.L.; Godinho, A.G.; Azevedo, A.; Brito, J.C.; Silva, P.R.; Teixeira, M.S.; Souza, V.P.; et al. Confirmation that heparin is an alternative means of promoting early reperfusion. Coron. Artery Dis. 1998, 9, 335–338. [Google Scholar] [CrossRef]
- Saxena, A.; Gupta, R.; Kumar, R.K.; Kothari, S.S.; Wasir, H.S. Predictors of arterial thrombosis after diagnostic cardiac catheterization in infants and children randomized to two heparin dosages. Catheter. Cardiovasc. Diagn. 1997, 41, 400–403. [Google Scholar] [CrossRef]
- Schiele, F.; Lindgaerde, F.; Eriksson, H.; Bassand, J.-P.; Wallmark, A.; Hansson, P.-O.; Grollier, G.; Sjo, M.; Moia, M.; Camez, A.; et al. Subcutaneous Recombinant Hirudin (HBW 023) Versus Intravenous Sodium Heparin in Treatment of Established Acute Deep Vein Thrombosis of the Legs: A Multicentre Prospective Dose-ranging Randomized Trial. Thromb. Haemost. 1997, 77, 0834–0838. [Google Scholar] [CrossRef]
- Metz, B.; Topol, E. Heparin as an adjuvant to thrombolytic therapy in acute myocardial infarction. Biomed. Pharmacother. 1996, 50, 243–253. [Google Scholar] [CrossRef]
- Sellevold, O.F.M.; Berg, T.M.; Rein, K.A.; Levang, O.W.; Iversen, O.; Bergh, K. Heparin–coated circuit during cardiopulmonary bypass Aclinical study using closed circuit, centrifugal pump and reduced heparinization. Acta Anaesthesiol. Scand. 1994, 38, 372–379. [Google Scholar] [CrossRef] [PubMed]
- Raschke, R.A.; Reilly, B.M.; Guidry, J.R.; Fontana, J.R.; Srinivas, S. The Weight-based Heparin Dosing Nomogram Compared with a Standard Care Nomogram. A randomized controlled trial. Ann. Intern. Med. 1993, 119, 874–881. [Google Scholar] [CrossRef] [PubMed]
- Schreiber, T.L.; Rizik, D.; White, C.; Sharma, G.V.; Cowley, M.; Macina, G.; Reddy, P.S.; Kantounis, L.; Timmis, G.C.; Margulis, A. Randomized trial of thrombolysis versus heparin in unstable angina. Circulation 1992, 86, 1407–1414. [Google Scholar] [CrossRef] [PubMed]
- Huo, M.H.; Salvati, E.A.; Sharrock, N.E.; Brien, W.W.; Sculco, T.P.; Pellicci, P.M.; Paul, M.; Mineo, R.; Go, G. Intraoperative heparin thromboembolic prophylaxis in primary total hip arthroplasty. A prospective, randomized, controlled, clinical trial. Clin. Orthop. Relat. Res. 1992, 274, 35–46. [Google Scholar] [CrossRef]
- Sharrock, N.E.; Brien, W.W.; Salvati, E.A.; Mineo, R.; Garvin, K.; Sculco, T.P. The effect of intravenous fixed-dose heparin during total hip arthroplasty on the incidence of deep-vein thrombosis. A randomized, double-blind trial in patients operated on with epidural anesthesia and controlled hypotension. JBJS 1990, 72, 1456–1461. [Google Scholar] [CrossRef]
- Francis, C.W.; Pellegrini, V.D.; Marder, V.J.; Harris, C.M.; Totterman, S.; Gabriel, K.R.; Baughman, D.J.; Roemer, S.; Burke, J.; Goodman, T.L. Prevention of venous thrombosis after total hip arthroplasty. Antithrombin III and low-dose heparin compared with dextran 40. J. Bone Jt. Surg. 1989, 71, 327–335. [Google Scholar] [CrossRef]
- Kirchmaier, C.M.; Wolf, H.; Schäfer, H.; Ehlers, B.; Breddin, H.K. Efficacy of a low molecular weight heparin administered intravenously or subcutaneously in comparison with intravenous unfractionated heparin in the treatment of deep venous thrombosis. Int. Angiol. 1998, 17, 135–145. [Google Scholar]
- Büller, H.R.; Davidson, B.L.; Decousus, H.; Gallus, A.; Gent, M.; Piovella, F.; Prins, M.H.; Raskob, G.; Berg-Segers, A.E.M.V.D.; Cariou, R.; et al. Subcutaneous Fondaparinux versus Intravenous Unfractionated Heparin in the Initial Treatment of Pulmonary Embolism. N. Engl. J. Med. 2003, 349, 1695–1702. [Google Scholar] [CrossRef]
- Sharrock, N.E.; Go, G.; Sculco, T.P.; Salvati, E.A.; Westrich, G.H.; Harpel, P.C. Dose Response of Intravenous Heparin on Markers of Thrombosis during Primary Total Hip Replacement. Anesthesiology 1999, 90, 981–987. [Google Scholar] [CrossRef]
- Nishijima, A.; Yamamoto, N.; Gosho, M.; Yanagibayashi, S.; Yoshida, R.; Takikawa, M.; Hayasaka, R.; Nagano, H.; Maruyama, E.; Sekido, M. Appropriate Use of Intravenous Unfractionated Heparin after Digital Replantation: A Randomized Controlled Trial Involving Three Groups. Plast. Reconstr. Surg. 2019, 143, 1224e–1232e. [Google Scholar] [CrossRef]
- Kurtkoti, J.; Bose, B.; Hiremagalur, B.; Sun, J.; Cochrane, T. Arterial line versus venous line administration of low molecular weight heparin, enoxaparin for prevention of thrombosis in the extracorporeal blood circuit of patients on haemodialysis or haemodiafiltration: A randomized cross-over trial. Nephrology 2016, 21, 663–668. [Google Scholar] [CrossRef] [PubMed]
- Upadhyay, A.; Verma, K.K.; Lal, P.; Chawla, D.; Sreenivas, V. Heparin for prolonging peripheral intravenous catheter use in neonates: A randomized controlled trial. J. Perinatol. 2015, 35, 274–277. [Google Scholar] [CrossRef] [PubMed]
- Warkentin, T.E.; Sheppard, J.-A.I.; Heels-Ansdell, D.; Marshall, J.C.; McIntyre, L.; Rocha, M.G.; Mehta, S.; Davies, A.R.; Bersten, A.D.; Crozier, T.M.; et al. Heparin-Induced Thrombocytopenia in Medical Surgical Critical Illness. Chest 2013, 144, 848–858. [Google Scholar] [CrossRef] [PubMed]
- Montalescot, G.; Zeymer, U.; Silvain, J.; Boulanger, B.; Cohen, M.; Goldstein, P.; Ecollan, P.; Combes, X.; Huber, K.; Pollack, C.; et al. Intravenous enoxaparin or unfractionated heparin in primary percutaneous coronary intervention for ST-elevation myocardial infarction: The international randomised open-label ATOLL trial. Lancet 2011, 378, 693–703. [Google Scholar] [CrossRef]
- Siu, C.-W.; Jim, M.-H.; Lau, C.-P.; Tse, H.-F. Low molecular weight heparin versus unfractionated heparin for thromboprophylaxis in patients with acute atrial fibrillation: A randomized control trial. Acute Card. Care 2011, 13, 196–198. [Google Scholar] [CrossRef]
- Birch, P.; Ogden, S.; Hewson, M. A randomised, controlled trial of heparin in total parenteral nutrition to prevent sepsis associated with neonatal long lines: The Heparin in Long Line Total Parenteral Nutrition (HILLTOP) trial. Arch. Dis. Child. Fetal Neonatal Ed. 2010, 95, F252–F257. [Google Scholar] [CrossRef]
- Rao, S.V.; Melloni, C.; Myles-DiMauro, S.; Broderick, S.; Kosinski, A.S.; Kleiman, N.S.; Dzavík, V.; Tanguay, J.F.; Chandna, H.; Gammon, R.; et al. Evaluation of a New Heparin Agent in Percutaneous Coronary Intervention: Results of the phase 2 evaluation of M118 IN pErcutaNeous Coronary intErvention (EMINENCE) Trial. Circulation 2010, 121, 1713–1721. [Google Scholar] [CrossRef]
- Montalescot, G.; Gallo, R.; White, H.D.; Cohen, M.; Steg, P.G.; Aylward, P.E.; Bode, C.; Chiariello, M.; King, S.B.; Harrington, R.A.; et al. Enoxaparin Versus Unfractionated Heparin in Elective Percutaneous Coronary Intervention 1-year results from the STEEPLE (SafeTy and efficacy of enoxaparin in percutaneous coronary intervention patients, an international randomized evaluation) trial. JACC Cardiovasc. Interv. 2009, 2, 1083–1091. [Google Scholar] [CrossRef]
- Broom, J.K.; O’Shea, S.; Govindarajulu, S.; Playford, E.G.; Hawley, C.M.; Isbel, N.M.; Campbell, S.B.; Mudge, D.W.; Carpenter, S.; Johnson, B.C.; et al. Rationale and design of the HEALTHY-CATH trial: A randomised controlled trial of Heparin versus EthAnol Lock THerapY for the prevention of Catheter Associated infecTion in Haemodialysis patients. BMC Nephrol. 2009, 10, 23. [Google Scholar] [CrossRef]
- White, H.D.; Gallo, R.; Cohen, M.; Steg, P.G.; Aylward, P.E.; Bode, C.; Steinhubl, S.; Montalescot, G. The use of intravenous enoxaparin in elective percutaneous coronary intervention in patients with renal impairment: Results from the SafeTy and Efficacy of Enoxaparin in PCI patients, an internationaL randomized Evaluation (STEEPLE) trial. Am. Heart J. 2009, 157, 125–131. [Google Scholar] [CrossRef]
- D’Ayala, M.; Smith, R.M.; Martone, C.; Briggs, W.; Deitch, J.S.; Wise, L. The Effect of Systemic Anticoagulation in Patients Undergoing Angioaccess Surgery. Ann. Vasc. Surg. 2008, 22, 11–15. [Google Scholar] [CrossRef] [PubMed]
- Mousa, S.A.; Zhang, F.; Aljada, A.; Chaturvedi, S.; Takieddin, M.; Zhang, H.; Chi, L.; Castelli, M.C.; Friedman, K.; Goldberg, M.M.; et al. Pharmacokinetics and Pharmacodynamics of Oral Heparin Solid Dosage Form in Healthy Human Subjects. J. Clin. Pharmacol. 2007, 47, 1508–1520. [Google Scholar] [CrossRef] [PubMed]
- Kearon, C.; Ginsberg, J.S.; Julian, J.A.; Douketis, J.; Solymoss, S.; Ockelford, P.; Jackson, S.; Turpie, A.G.; MacKinnon, B.; Hirsh, J.; et al. Comparison of Fixed-Dose Weight-Adjusted Unfractionated Heparin and Low-Molecular-Weight Heparin for Acute Treatment of Venous Thromboembolism. JAMA 2006, 296, 935–942. [Google Scholar] [CrossRef] [PubMed]
- Maejima, Y.; Yasu, T.; Ueba, H.; Kobayashi, N.; Hashimoto, S.; Kubo, N.; Kakehashi, A.; Isobe, M.; Kawakami, M.; Saito, M. Exercise After Heparin Administration: New therapeutic program for patients with-option arteriosclerosis oblitrans. J. Am. Cardiol. J. 2005, 69, 1099–1104. [Google Scholar] [CrossRef]
- Chong, B.H.; Brighton, T.A.; Baker, R.I.; Thurlow, P.; Lee, C.H.; ASTH DVT Study Group. Once-Daily Enoxaparin in The Outpatient Setting Versus Unfractionated Heparin in Hospital for the Treatment of Symptomatic Deep-Vein Thrombosis. J. Thromb. Thrombolysis 2005, 19, 173–181. [Google Scholar] [CrossRef]
- Westrich, G.H.; Salvati, E.A.; Sharrock, N.; Potter, H.G.; Sánchez, P.M.; Sculco, T.P. The effect of intraoperative heparin administered during total hip arthroplasty on the incidence of proximal deep vein thrombosis assessed by magnetic resonance venography. J. Arthroplast. 2005, 20, 42–50. [Google Scholar] [CrossRef]
- Ferguson, J.J.; Califf, R.M.; Antman, E.M.; Cohen, M.; Grines, C.L.; Goodman, S.; Kereiakes, D.J.; Langer, A.; Mahaffey, K.W.; Nessel, C.C.; et al. Enoxaparin vs Unfractionated Heparin in High-Risk Patients with Non–ST-Segment Elevation Acute Coronary Syndromes Managed with an Intended Early Invasive Strategy: Primary results of the SYNERGY randomized trial. JAMA 2004, 292, 45–54. [Google Scholar] [CrossRef]
- Noveck, R.J.; Hubbard, R.C. Parecoxib Sodium, an Injectable COX-2-Specific Inhibitor, Does Not Affect Unfractionated Heparin-Regulated Blood Coagulation Parameters. J. Clin. Pharmacol. 2004, 44, 474–480. [Google Scholar] [CrossRef]
- Demers, C. ESSENCE trial results: Breaking new ground. Efficacy and Safety of Subcutaneous Enoxaparin in Non-Q wave Coronary Events. Can. J. Cardiol. 1998, 14, 15E–19E. [Google Scholar]
- McMullen, A.; Fioravanti, I.D.; Pollack, V.; Rideout, K.; Sciera, M. Heparinized saline or normal saline as a flush solution in intermittent intravenous lines in infants and children. MCN Am. J. Matern. Child. Nurs. 1993, 18, 78–85. [Google Scholar] [CrossRef]
- Abbo, R.; Gruber, A.; Volis, I.; Aronson, D.; Girerd, N.; Kristensen, S.L.; Zukermann, R.; Alberkant, N.; Sitnitsky, E.; Kruger, A.; et al. Diuresis Efficacy in Ambulatory Congested Heart Failure Patients: Intrapatient Comparison of 3 Diuretic Regimens (DEA-HF). JACC Heart Fail. 2024, 12, 1396–1405. [Google Scholar] [CrossRef] [PubMed]
- Ferguson, J.A.; Sundblad, K.J.; Becker, P.K.; Gorski, J.C.; Rudy, D.W.; Brater, D.C. Role of duration of diuretic effect in preventing sodium retention. Clin. Pharmacol. Ther. 1997, 62, 203–208. [Google Scholar] [CrossRef] [PubMed]
- Ricci, Z.; Haiberger, R.; Pezzella, C.; Garisto, C.; Favia, I.; Cogo, P. Furosemide versus ethacrynic acid in pediatric patients undergoing cardiac surgery: A randomized controlled trial. Crit. Care 2015, 19, 2. [Google Scholar] [CrossRef] [PubMed]
- Cox, Z.L.; Hung, R.; Lenihan, D.J.; Testani, J.M. Diuretic Strategies for Loop Diuretic Resistance in Acute Heart Failure: The 3T Trial. JACC Heart Fail. 2020, 8, 157–168. [Google Scholar] [CrossRef]
- Reed, S.; Greene, P.; Ryan, T.; Cerimele, B.; Schwertschlag, U.; Weinberger, M.; Voelker, J. The renin angiotensin aldosterone system and frusemide response in congestive heart failure. Br. J. Clin. Pharmacol. 1995, 39, 51–57. [Google Scholar] [CrossRef]
- Hariman, R.J.; Bremner, S.; Louie, E.K.; Rogers, W.J.; Kostis, J.B.; Nocero, M.A.; Jones, J.P. Dose-response study of intravenous torsemide in congestive heart failure. Am. Heart J. 1994, 128, 352–357. [Google Scholar] [CrossRef]
- McNAIR, A.; Krogsgaard, A.R.; Hilden, T.; Nielsen, P.E. Severe Hypertension with Cerebral Symptoms Treated with Furosemide, Fractionated Diazoxide or Dihydralazine. Acta Medica Scand. 1986, 220, 15–23. [Google Scholar] [CrossRef]
- Stroobandt, R.; Dodion, L.; Kesteloot, H. Clinical efficacy of torasemide, a new diuretic agent, in patients with acute heart failure: A double blind comparison with furosemide. Arch. Int. Pharmacodyn. Ther. 1982, 260, 151–158. [Google Scholar]
- Hamo, C.E.; Abdelmoneim, S.S.; Han, S.Y.; Chandy, E.; Muntean, C.; Khan, S.A.; Sunkesula, P.; Meykler, M.; Ramachandran, V.; Rosenberg, E.; et al. OUTpatient intravenous LASix Trial in reducing hospitalization for acute decompensated heart failure (OUTLAST). PLoS ONE 2021, 16, e0253014. [Google Scholar] [CrossRef]
- Paterna, S.; Di Gaudio, F.; La Rocca, V.; Balistreri, F.; Greco, M.; Torres, D.; Lupo, U.; Rizzo, G.; di Pasquale, P.; Indelicato, S.; et al. Hypertonic Saline in Conjunction with High-Dose Furosemide Improves Dose–Response Curves in Worsening Refractory Congestive Heart Failure. Adv. Ther. 2015, 32, 971–982. [Google Scholar] [CrossRef]
- Mentz, R.J.; Stevens, S.R.; DeVore, A.D.; Lala, A.; Vader, J.M.; AbouEzzeddine, O.F.; Khazanie, P.; Redfield, M.M.; Stevenson, L.W.; O’connor, C.M.; et al. Decongestion Strategies and Renin-Angiotensin-Aldosterone System Activation in Acute Heart Failure. JACC Heart Fail. 2014, 3, 97–107. [Google Scholar] [CrossRef] [PubMed]
- Palazzuoli, A.; Testani, J.M.; Ruocco, G.; Pellegrini, M.; Ronco, C.; Nuti, R. Different diuretic dose and response in acute decompensated heart failure: Clinical characteristics and prognostic significance. Int. J. Cardiol. 2016, 224, 213–219. [Google Scholar] [CrossRef] [PubMed]
- Galiwango, P.J.; McReynolds, A.; Ivanov, J.; Chan, C.T.; Floras, J.S. Activity with Ambulation Attenuates Diuretic Responsiveness in Chronic Heart Failure. J. Card. Fail. 2011, 17, 797–803. [Google Scholar] [CrossRef] [PubMed]
- Paterna, S.; Fasullo, S.; Cannizzaro, S.; Vitrano, G.; Terrazzino, G.; Maringhini, G.; Ganci, F.; Scalzo, S.; Di Pasquale, P.; Parrinello, G.; et al. Short-Term Effects of Hypertonic Saline Solution in Acute Heart Failure and Long-Term Effects of a Moderate Sodium Restriction in Patients With Compensated Heart Failure With New York Heart Association Class III (Class C) (SMAC-HF Study). Am. J. Med. Sci. 2011, 342, 27–37. [Google Scholar] [CrossRef]
- Mitrovic, V.; Seferovic, P.; Dodic, S.; Krotin, M.; Neskovic, A.; Dickstein, K.; de Voogd, H.; Böcker, C.; Ziegler, D.; Godes, M.; et al. Cardio-Renal Effects of the A1 Adenosine Receptor Antagonist SLV320 in Patients with Heart Failure. J. Am. Cardiol. Heart Fail. 2009, 2, 523–531. [Google Scholar] [CrossRef]
- Dharmaraj, R.; Hari, P.; Bagga, A. Randomized cross-over trial comparing albumin and frusemide infusions in nephrotic syndrome. Pediatr. Nephrol. 2009, 24, 775–782. [Google Scholar] [CrossRef]
- Licata, G.; Di Pasquale, P.; Parrinello, G.; Cardinale, A.; Scandurra, A.; Follone, G.; Argano, C.; Tuttolomondo, A.; Paterna, S. Effects of high-dose furosemide and small-volume hypertonic saline solution infusion in comparison with a high dose of furosemide as bolus in refractory congestive heart failure: Long-term effects. Am. Heart J. 2003, 145, 459–466. [Google Scholar] [CrossRef]
- Agostoni, P.; Marenzi, G.; Lauri, G.; Perego, G.; Schianni, M.; Sganzerla, P.; Guazzi, M.D. Sustained improvement in functional capacity after removal of body fluid with isolated ultrafiltration in chronic cardiac insufficiency: Failure of furosemide to provide the same result. Am. J. Med. 1994, 96, 191–199. [Google Scholar] [CrossRef]
- Acharya, S.K.; Balwinder, S.; Padhee, A.K.; Nijhawan, S.; Tandon, B.N. Large Volume Paracentesis and Intravenous Dextran to Treat Tense Ascites. J. Clin. Gastroenterol. 1992, 14, 31–35. [Google Scholar] [CrossRef]
- Riva, E.; Farina, P.; Tognoni, G.; Bottino, S.; Orrico, C.; Pardi, G. Pharmacokinetics of furosemide in gestosis of pregnancy. Eur. J. Clin. Pharmacol. 1978, 14, 361–366. [Google Scholar] [CrossRef]
- Spaliviero, M.; Power, N.E.; Murray, K.S.; Sjoberg, D.D.; Benfante, N.E.; Bernstein, M.L.; Wren, J.; Russo, P.; Coleman, J.A. Intravenous Mannitol Versus Placebo During Partial Nephrectomy in Patients with Normal Kidney Function: A Double-blind, Clinically-integrated, Randomized Trial. Eur. Urol. 2018, 73, 53–59. [Google Scholar] [CrossRef] [PubMed]
- Kurnik, B.; Weisberg, L.; Cuttler, I.; Kurnik, P. Effects of atrial-natriuretic-peptide versus mannitol on renal blood-flow during radiocontrast infusion in chronic-renal-failure. J. Lab. Clin. Med. 1990, 116, 27–35. [Google Scholar] [PubMed]
- Beygui, F.; Cayla, G.; Roule, V.; Roubille, F.; Delarche, N.; Silvain, J.; Van Belle, E.; Belle, L.; Galinier, M.; Motreff, P.; et al. Early Aldosterone Blockade in Acute Myocardial Infarction: The ALBATROSS Randomized Clinical Trial. J. Am. Cardiol. 2016, 67, 1917–1927. [Google Scholar] [CrossRef]
- Beygui, F.; Vicaut, E.; Ecollan, P.; Machecourt, J.; Van Belle, E.; Zannad, F.; Montalescot, G. Rationale for an early aldosterone blockade in acute myocardial infarction and design of the ALBATROSS trial. Am. Heart J. 2010, 160, 642–648.e1. [Google Scholar] [CrossRef]
- Elkenany, S.; Alseoudy, M.; Elshehawi, M.; Bakrey, S.; Aboelela, M. Effect of coadministration of 10 mg/kg calcium chloride and neostigmine on extubation time: A randomized controlled trial. Rev. Esp. Anestesiol. Reanim. 2024, 71, 438–444. [Google Scholar] [CrossRef]
- Agarwal, S.; Kumar, M.; Gupta, A.; Rautela, R.; Jain, N. Effect of Intravenous Calcium Gluconate on Prevention of Post Spinal Hypotension during Spinal Anaesthesia for Caesarean Section: A Randomized Double-Blind Controlled Study. J. Nepal Health Res. Counc. 2022, 20, 89–95. [Google Scholar] [CrossRef]
- Udelson, J.E.; Smith, W.B.; Hendrix, G.H.; Painchaud, C.A.; Ghazzi, M.; Thomas, I.; Ghali, J.K.; Selaru, P.; Chanoine, F.; Pressler, M.L.; et al. Acute Hemodynamic Effects of Conivaptan, a Dual V 1A and V 2 Vasopressin Receptor Antagonist, in Patients With Advanced Heart Failure. Circulation 2001, 104, 2417–2423. [Google Scholar] [CrossRef]
- Verma, A.; Jaiswal, S.; Reid, C.; Borah, P.; Lal, M.; Gupta, S.; Khanna, P. Efficacy of 10%, 25% and 50% dextrose in the treatment of hypoglycemia in the emergency department—A randomized controlled study. Am. J. Emerg. Med. 2024, 82, 101–104. [Google Scholar] [CrossRef]
- Ponikowski, P.; Van Veldhuisen, D.J.; Comin-Colet, J.; Ertl, G.; Komajda, M.; Mareev, V.; McDonagh, T.; Parkhomenko, A.; Tavazzi, L.; Levesque, V.; et al. Beneficial effects of long-term intravenous iron therapy with ferric carboxymaltose in patients with symptomatic heart failure and iron deficiency. Eur. Heart J. 2015, 36, 657–668. [Google Scholar] [CrossRef]
- Charles-Edwards, G.; Amaral, N.; Sleigh, A.; Ayis, S.; Catibog, N.; McDonagh, T.; Monaghan, M.; Amin-Youssef, G.; Kemp, G.J.; Shah, A.M.; et al. Effect of Iron Isomaltoside on Skeletal Muscle Energetics in Patients with Chronic Heart Failure and Iron Deficiency: FERRIC-HF II Randomized Mechanistic Trial: FERRIC-HF II Randomized Mechanistic Trial. Circulation 2019, 139, 2386–2398. [Google Scholar] [CrossRef]
- Osawa, E.A.; Cutuli, S.L.; Cioccari, L.; Bitker, L.; Peck, L.; Young, H.; Hessels, L.; Yanase, F.; Fukushima, J.T.; Hajjar, L.A.; et al. Continuous Magnesium Infusion to Prevent Atrial Fibrillation After Cardiac Surgery: A Sequential Matched Case-Controlled Pilot Study. J. Cardiothorac. Vasc. Anesth. 2020, 34, 2940–2947. [Google Scholar] [CrossRef] [PubMed]
- Easterling, T.; Hebert, M.; Bracken, H.; Darwish, E.; Ramadan, M.C.; Shaarawy, S.; Charles, D.; Abdel-Aziz, T.; Nasr, A.S.; Safwal, S.M.; et al. A randomized trial comparing the pharmacology of magnesium sulfate when used to treat severe preeclampsia with serial intravenous boluses versus a continuous intravenous infusion. BMC Pregnancy Childbirth 2018, 18, 290. [Google Scholar] [CrossRef] [PubMed]
- Lai, Y.; Rouse, D.J.; Spong, C.Y.; Leveno, K.J.; Varner, M.W.; Mercer, B.M.; Iams, J.D.; Wapner, R.J.; Sorokin, Y.; Thorp, J.M.; et al. Effect of Magnesium Sulfate Administration for Neuroprotection on Latency in Women with Preterm Premature Rupture of Membranes. Am. J. Perinatol. 2015, 32, 387–392. [Google Scholar] [CrossRef]
- Carrió, M.L.; Ventura, J.L.; Javierre, C.; Rodríguez-Castro, D.; Farrero, E.; Torrado, H.; Badia, M.B.; Granados, J. Does post-cardiac surgery magnesium supplementation improve outcome? Magnes. Res. 2012, 25, 159–167. [Google Scholar] [CrossRef] [PubMed]
- Wong, G.K.C.; Chan, M.T.V.; Gin, T.; Poon, W.S. Intravenous magnesium sulfate after aneurysmal subarachnoid hemorrhage: Current status. Acta Neurochir. Suppl. 2011, 110, 169–173. [Google Scholar] [CrossRef]
- Abbade, J.F.; Costa, R.A.A.; Martins, A.M.V.C.; Borges, V.T.M.; Rudge, M.V.C.; Peraçoli, J.C. Zuspan’s Scheme Versus an Alternative Magnesium Sulfate Scheme: Randomized Clinical Trial of Magnesium Serum Concentrations. Hypertens. Pregnancy 2010, 29, 82–92. [Google Scholar] [CrossRef]
- Chu, K.; Evans, R.; Emerson, G.; Greenslade, J.; Brown, A. Magnesium Sulfate versus Placebo for Paroxysmal Atrial Fibrillation: A Randomized Clinical Trial. Acad. Emerg. Med. 2009, 16, 295–300. [Google Scholar] [CrossRef]
- Chiu, C.; Heyer, E.J.; Rampersad, A.D.; Zurica, J.; Ornstein, E.; Sahlein, D.H.; Sciacca, R.R.; Connolly, E.S. High Dose Magnesium Infusions Are Not Associated with Increased Pressor Requirements after Carotid Endarterectomy. Neurosurgery 2006, 58, 71–77. [Google Scholar] [CrossRef]
- Bergh, W.M.v.D. Magnesium Sulfate in Aneurysmal Subarachnoid Hemorrhage: A randomized controlled trial. Stroke 2005, 36, 1011–1015. [Google Scholar] [CrossRef]
- Domanski, M.; Antman, E.M.; McKinlay, S.; Varshavsky, S.; Platonov, P.; Assmann, S.F.; Norman, J. Geographic variability in patient characteristics, treatment and outcome in an international trial of magnesium in acute myocardial infarction. Control. Clin. Trials 2004, 25, 553–562. [Google Scholar] [CrossRef]
- Livingston, J.C.; Livingston, L.W.; Ramsey, R.; Mabie, B.C.; Sibai, B.M. Magnesium sulfate in women with mild preeclampsia: A randomized controlled trial. Obstet. Gynecol. 2003, 101, 217–220. [Google Scholar] [CrossRef] [PubMed]
- MAGIC Steering Committee Rationale and design of the magnesium in coronaries (MAGIC) study: A clinical trial to reevaluate the efficacy of early administration of magnesium in acute myocardial infarction. Am. Heart J. 2000, 139, 10–14. [CrossRef]
- Jin, X.-Q.; Shen, Y.-H.; Fu, F.; Yu, J.; Xiao, F.; Huang, X.-D. Oxytocin infusion for maintenance of uterine tone under prophylactic phenylephrine infusion for prevention of post-spinal hypotension in cesarean delivery: A prospective randomised double-blinded dose-finding study. BMC Pregnancy Childbirth 2023, 23, 1–9. [Google Scholar] [CrossRef]
- Saratzis, A.; Chiocchia, V.; Jiffry, A.; Hassanali, N.; Singh, S.; Imray, C.H.; Bown, M.J.; Mahmood, A. HYDration and Bicarbonate to Prevent Acute Renal Injury After Endovascular Aneurysm Repair with Suprarenal Fixation: Pilot/Feasibility Randomised Controlled Study (HYDRA Pilot Trial). Eur. J. Vasc. Endovasc. Surg. 2018, 55, 648–656. [Google Scholar] [CrossRef]
- Nicholson, T.A.; Kirsch, J.M.; Churchill, R.; Lazarus, M.D.; Abboud, J.A.; Namdari, S. The effect of tranexamic acid for visualization on pump pressure and visualization during arthroscopic rotator cuff repair: An anonymized, randomized controlled trial. J. Shoulder Elb. Surg. 2022, 31, 2211–2216. [Google Scholar] [CrossRef]
- Meretoja, A.; Yassi, N.; Wu, T.Y.; Churilov, L.; Sibolt, G.; Jeng, J.-S.; Kleinig, T.; Spratt, N.J.; Thijs, V.; Wijeratne, T.; et al. Tranexamic acid in patients with intracerebral haemorrhage (STOP-AUST): A multicentre, randomised, placebo-controlled, phase 2 trial. Lancet Neurol. 2020, 19, 980–987. [Google Scholar] [CrossRef]
- Marcucci, M.; Painter, T.W.; Conen, D.; Leslie, K.; Lomivorotov, V.V.; Sessler, D.; Chan, M.T.V.; Borges, F.K.; Zapata, M.J.M.; Wang, C.Y.; et al. Rationale and design of the PeriOperative ISchemic Evaluation-3 (POISE-3): A randomized controlled trial evaluating tranexamic acid and a strategy to minimize hypotension in noncardiac surgery. Trials 2022, 23, 1–12. [Google Scholar] [CrossRef]
- Tazmini, K.; Fraz, M.S.A.; Nymo, S.H.; Stokke, M.K.; Louch, W.E.; Øie, E. Potassium infusion increases the likelihood of conversion of recent-onset atrial fibrillation—A single-blinded, randomized clinical trial. Am. Heart J. 2020, 221, 114–124. [Google Scholar] [CrossRef]
- Lin, C.; Kaper, H.J.; Li, W.; Splinter, R.; Sharma, P.K. Role of endothelial glycocalyx in sliding friction at the catheter-blood vessel interface. Sci Rep. 2020, 10, 1–10. [Google Scholar] [CrossRef]
- Scarano, M.; D’arrigo, S.; De Letteriis, S.; Grasso, S.; Pittiruti, M.; Scoppettuolo, G. Risk of thrombophlebitis associated with continuous peripheral infusion of vancomycin: The effect of dilution. J. Vasc. Access 2024, 25, 107–112. [Google Scholar] [CrossRef]
- Owen, V.S.; Rosgen, B.K.; Cherak, S.J.; Ferland, A.; Stelfox, H.T.; Fiest, K.M.; Niven, D.J. Adverse events associated with administration of vasopressor medications through a peripheral intravenous catheter: A systematic review and meta-analysis. Crit. Care 2021, 25, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Shibata, Y.; Taogoshi, T.; Matsuo, H. Extravasation of Noncytotoxic Agents: Skin Injury and Risk Classification. Biol. Pharm. Bull. 2023, 46, 746–755. [Google Scholar] [CrossRef] [PubMed]
- Marsh, N.; Larsen, E.N.; Takashima, M.; Kleidon, T.; Keogh, S.; Ullman, A.J.; Mihala, G.; Chopra, V.; Rickard, C.M. Peripheral intravenous catheter failure: A secondary analysis of risks from 11,830 catheters. Int. J. Nurs. Stud. 2021, 124, 104095. [Google Scholar] [CrossRef] [PubMed]
- NHS Resolution. Did You Know? Extravasation. Published Online 1 March 2022. Available online: https://resolution.nhs.uk/wp-content/uploads/2023/05/Did-You-Know-Extravasation.pdf (accessed on 9 October 2024).
- On behalf of the AMOR-VENUS Study Group; Yasuda, H.; Yamamoto, R.; Hayashi, Y.; Kotani, Y.; Kishihara, Y.; Kondo, N.; Sekine, K.; Shime, N.; Morikane, K.; et al. Occurrence and incidence rate of peripheral intravascular catheter-related phlebitis and complications in critically ill patients: A prospective cohort study (AMOR-VENUS study). J. Intensiv. Care 2021, 9, 1–12. [Google Scholar] [CrossRef]
- Lv, L.; Zhang, J. The incidence and risk of infusion phlebitis with peripheral intravenous catheters: A meta-analysis. J. Vasc. Access 2019, 21, 342–349. [Google Scholar] [CrossRef]
- Gershengorn, H.B.; Basu, T.; Horowitz, J.K.; McLaughlin, E.; Munroe, E.; O’malley, M.; Hsaiky, L.; Flanders, S.A.; Bernstein, S.J.; Paje, D.; et al. The Association of Vasopressor Administration through a Midline Catheter with Catheter-related Complications. Ann. Am. Thorac. Soc. 2023, 20, 1003–1011. [Google Scholar] [CrossRef] [PubMed]
- Holohan, J.A.; Hatola, C.; Mitchell, C.; Epping, G.; Hooker, E.; Kwon, J.; Mordente, L.; Sladek, A. The Use of Midline Catheters in Place of Central Venous Catheters for Vasopressor Administration to Reduce Central Line-Associated Bacteremia. Am. J. Infect. Control. 2024, 52, S75–S76. [Google Scholar] [CrossRef]
- Prasanna, N.; Yamane, D.; Haridasa, N.; Davison, D.; Sparks, A.; Hawkins, K. Safety and efficacy of vasopressor administration through midline catheters. J. Crit. Care 2021, 61, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Hadaway, L.M.; Gorski, L.A.M. Infiltration and Extravasation Risk with Midline Catheters: A Narrative Literature Review. J. Infus. Nurs. 2024, 47, 324–346. [Google Scholar] [CrossRef] [PubMed]
- Marsh, N.; Webster, J.; Ullman, A.J.; Mihala, G.; Cooke, M.; Chopra, V.; Rickard, C.M. Peripheral intravenous catheter non-infectious complications in adults: A systematic review and meta-analysis. J. Adv. Nurs. 2020, 76, 3346–3362. [Google Scholar] [CrossRef]
- Ballesteros-Peña, S.; Fernández-Aedo, I.; Vallejo-De la Hoz, G.; Tønnesen, J.; Miguelez, C. Identification of potentially irritating intravenous medications. Enfermería Intensiv. 2022, 33, 132–140. [Google Scholar] [CrossRef] [PubMed]
- Brørs, G.; Gjeilo, K.H.; Lund, T.; Skevik, K.; Aa, E.; Høvik, L.H.; Skarsvaag, T.; Mjølstad, O.C. Amiodarone-induced phlebitis: Incidence and adherence to a clinical practice guideline. Eur. J. Cardiovasc. Nurs. 2023, 22, 824–831. [Google Scholar] [CrossRef] [PubMed]
- Bahl, A.; Johnson, S.; Mielke, N.; Karabon, P. Early recognition of peripheral intravenous catheter failure using serial ultrasonographic assessments. PLoS ONE 2021, 16, e0253243. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Mielke, N.; Johnson, S.; Karabon, P.; Bahl, A. A prospective sonographic evaluation of peripheral intravenous catheter associated thrombophlebitis. J. Vasc. Access 2022, 23, 754–763. [Google Scholar] [CrossRef] [PubMed]
- Giustivi, D.; Celano, R.; Cattalani, M.; Camilli, C.; Trombetta, L.; Facchinetti, P.; Bartoli, A.; Bizzi, E.; Urso, F.; Donadoni, M.; et al. Ultrasound assessment of short peripheral catheter failure. J. Vasc. Access 2024. [Google Scholar] [CrossRef] [PubMed]
- Yabunaka, K.; Murayama, R.; Tanabe, H.; Takahashi, T.; Oe, M.; Oya, M.; Fujioka, M.; Sanada, H. Ultrasonographic Classification of Subcutaneous Edema Caused by Infusion via Peripheral Intravenous Catheter. J. Med. Ultrasound 2016, 24, 60–65. [Google Scholar] [CrossRef]
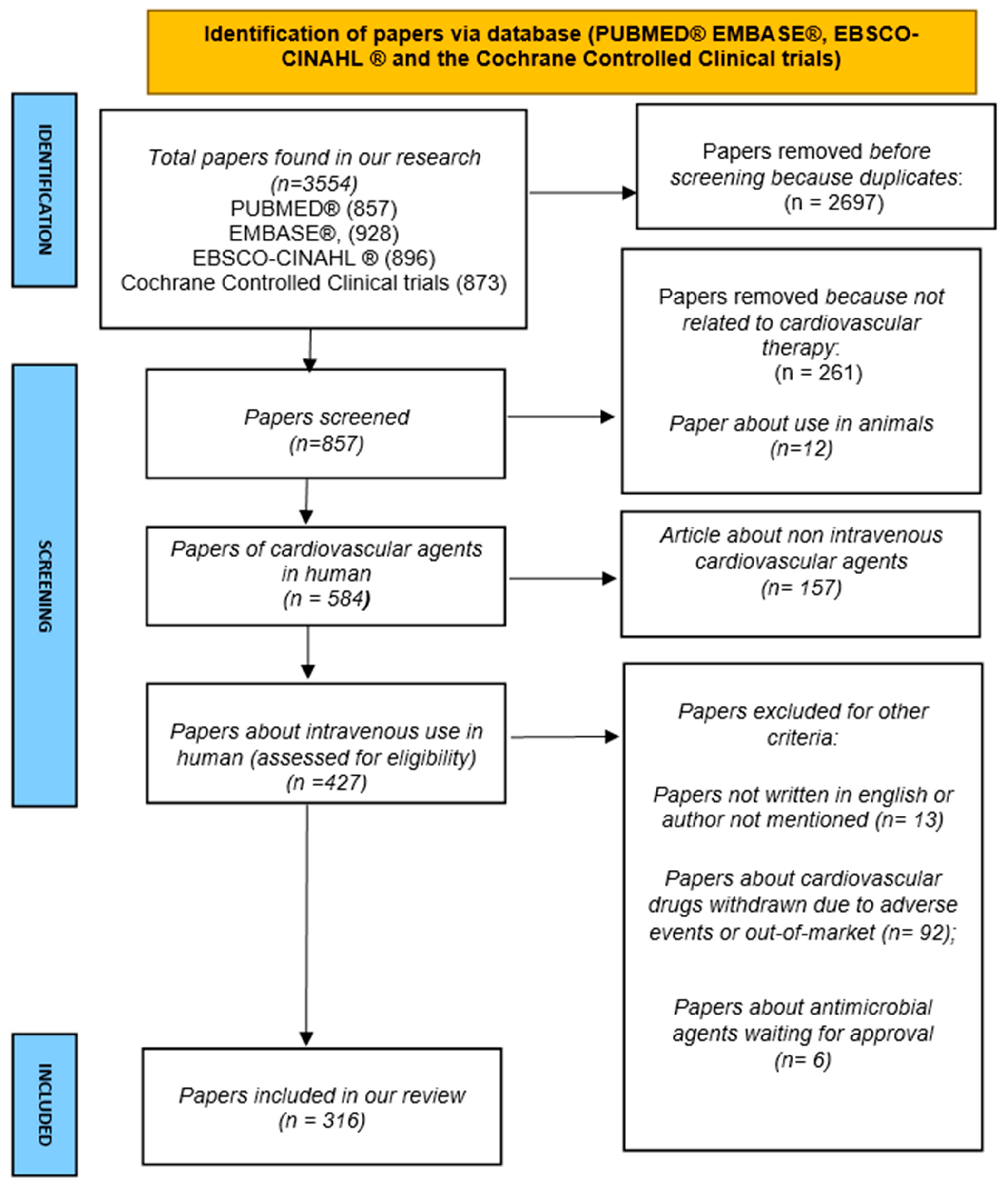
| Cardiologic Drugs and Line of Infusion | |||||||
|---|---|---|---|---|---|---|---|
| Name | Red Flag and/or Vesicant Properties | pH | mOsm/Kg | Phlebitis (Incidence ≥ 5%) | Diluent | Loading Dose | Maintenance Dose |
| Antiarrhythmics | |||||||
| Adenosine [17,18,19,20,21,22] | 6–7.5 | 261–319 | no | D5W/NS | 6 mg | NA | |
| Amiodarone [23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40] | vesicant | 3.7–4.3 | 304 | yes | D5W | 5 mg/Kg | 10–20 mg/Kg |
| Deslanoside [41] | 5–7 | NA | no | NS | NA | NA | |
| Digoxin [33,42,43,44] | vesicant | 6.8–7.2 | 455 | yes | D5W | 8–12 mcg/Kg | ¼ loading dose |
| vesicant | 6.8–7.2 | 421 | yes | NS | 8–12 mcg/Kg | ¼ loading dose | |
| Diltiazem [43,44,45,46,47] | 3.7–4.1 | NA | no | D5W/NS | 0.25 mg/Kg | 10 mg/h | |
| Disopyramide [48] | 4–5 | NA | no | NS | NA | NA | |
| Esmolol [49,50,51,52,53,54,55,56,57] | vesicant | 4.5–5.5 | 300 | no | NS | 0.5 mg/Kg | 0.05–0.2 mg/Kg/min |
| Flecainide [58] | 5.8 | 300 | no | D5W/NS | 2 mg/Kg | 1.5 mg/Kg/ for 1 h then 0.1–0.25 mg/Kg/h | |
| Ibutilide [59,60,61,62] | 4.6 | NA | no | D5W/NS | NA | 0.1 mg/Kg (max 1 mg) | |
| Labetalol [53,63,64,65,66] | 3–4.5 | 249 | no | D5W | 20 mg | 1–2 mg/min | |
| 3–4.5 | 229 | no | NS | 20 mg | 1–2 mg/min | ||
| Landiolol [57,67,68,69,70] | 6.3 | 1896 | no | D5W/NS | 100 mcg/Kg/min | 10–40 mcg/Kg/min | |
| Lidocaine [30,71,72,73,74,75] | 6.5 | 280–305 | no | D5W | 1–1.5 mg/Kg | NA | |
| Metoprolole [76,77,78,79,80,81,82,83,84,85,86,87] | 6–7 | NA | no | NS | 5–15 mg | NA | |
| Phenytoin [88] | 12 | 312 | yes | NS | 3.5–5 mg/Kg | < 50 mg/min | |
| Procainamide [88] | 4–6 | NA | no | D5W/NS | 20 mg/min | 2–6 mg/min | |
| Propafenone [37,60] | 6.5 | NA | no | NS | 1 mg/Kg | 0.5–1 mg/min for 3 h | |
| Propranolol [51,55,56,89,90] | 3.9–6.4 | NA | NA | D5W/NS | 1–3 mg at 1 mg/min | Not recommended | |
| Quinidine gluconate [91] | 5.5–7 | NA | no | NS | NA | Max 10 mg/Kg at 0.25 mg/Kg/min | |
| Sotatol [29,33,61,92] | 6–7 | NA | no | D5W/NS | 0.5–1.5 mg/Kg in 30 min | 1 mg/Kg in 4–6 h | |
| Verapamil [86,93,94,95,96,97] | 5–6 | NA | no | NS | 2.5–10 mg | NA | |
| Vernakalant [98] | 5.5 | 270–320 | no | D5W/NS | NA | 3 mg/Kg over 10 min | |
| Vasoconstrictor and Inotropic | |||||||
| Aminophylline [99] | vesicant | 8.6–9 | 124 | yes | D5W/NS | 25 mg/min | 0.25–0.9 mg/Kg/h |
| Alprostadil [100] | 4–8 | NA | no | D5W/NS | NO | 10 mcg/day in 1–2 h upgradable to 20 mcg | |
| Amrinone [101] | 3.2 | 300 | yes | D5W | 0.75 mcg/Kg | 5–10 mcg/Kg/min | |
| Desmopressin [102,103] | 3.5–6 | NA | No | NS | NA | 1–2 mcg x 2/day | |
| Dexmedetomidine [74,104] | 4.5–7 | NA | no | D5W/NS | 1 mcg/kg over 10 min | 0.2–0.7 mcg/Kg/h | |
| Dobutamine [100,105,106,107,108,109,110,111,112,113,114,115,116,117,118,119,120] | vesicant | 2.5–4 | 269 | yes | D5W | NA | 2–20 mcg/Kg/min |
| vesicant | 2.5–4 | 251 | yes | NS | NA | 2–20 mcg/Kg/min | |
| Dopamine [100,121,122,123,124,125] | vesicant | 2.5–5 | 560 | yes | D5W/NS | Variable | Variable |
| Enoximone [126,127] | 12 | NA | no | NS | 0.5 mg/Kg | 5–20 mcg/Kg/min | |
| Epinephrine [122,128,129,130,131,132] | vesicant | 2.5–3.5 | NA | yes | D5W/NS | NA | 0.05–2 mcg/Kg/min |
| Epoprosterenol [133,134,135,136,137,138] | 10.3–10.8 | NA | no | NS | 2 ng/Kg/min | titrate 1–2 ng/Kg/min until target | |
| Iloprost [139] | 7.7–8.7 | 430–490 | no | 0.5 ng/Kg/min | increase 0.5 ng/Kg/min (dose max: 2 ng/Kg/min) | ||
| Isoproterenol [140,141] | 2.5–4.5 | NA | no | D5W/NS | 0.02–0.06 mg | 2–20 mcg/min | |
| Levosimendan [108,109,110,111,113,142,143,144,145,146,147,148,149,150,151,152,153,154,155,156,157,158,159,160] | 5–7 | 450–550 | no | NS | 6–12 mcg/Kg | 0.05–0.2 mcg/Kg/min | |
| Milrinone [107,119,120,161,162,163,164,165] | 3.2–4 | 261–319 | no | D5W/NS | 50 mcg/Kg | 0.375–0.75 mcg/Kg/min | |
| Norepinephrine [122,132,166,167] | vesicant | 3–4.5 | 280–300 | yes | D5W | NA | 0.01–3.3 mcg/Kg/min |
| Phenilephrine [130,131,168] | vesicant | 4.7–5.3 | 270–300 | no | D5W/NS | 50–100 mcg | 0.5–6 mcg/Kg/min |
| Terlipressine [102,169] | vesicant | 3.7–4.2 | 290–360 | no | NS | NA | 1–2 mg × 3–4/day |
| Vasopressin [132,170] | vesicant | 3.5 | NA | no | NS | NA | 0.01–0.03 UI/min |
| Thrombolytics | |||||||
| Alteplase [171,172,173,174,175,176,177,178] | 6.7–7.8 | 215 | no | D5W/NS | Variable | Variable | |
| Anistreplase [179] | 4–5 | NA | no | NS | 30 mg in 5 min | NA | |
| Reteplase [180,181] | 6 | NA | no | NS | NA | 10 UI over 2 min | |
| Streptokinase [177,182,183] | 7.3 | NA | no | NS | 250,000 UI | 100,000 UI/h | |
| Tenecteplase [184,185] | 7.3 | 290 | no | NS | based on weight | based on weight | |
| Urokinase [186] | 6–7.5 | NA | no | NS | 3300–4400 UI/kg | 3300–4400 UI/kg/h | |
| Antiaggregants | |||||||
| Abciximab [180,181,184,187,188,189,190,191,192,193] | 7.2 | NA | no | D5W/NS | 0.25 mg/Kg | 0.125 mcg/min | |
| Argatroban [194] | 3.2–7.5 | NA | no | NS | 350 mcg/Kg | 25 mcg/Kg/min (targeted on ACT) | |
| Bivalirudin [182,195,196,197,198,199,200,201,202] | 4.6–6 | 250–450 | no | D5W/NS | 0.75 mg/Kg | 1.75 mg/Kg/h | |
| Cangrelor [196,203] | 8–9.5 | 287–290 | no | D5W/NS | 30 mcg/Kg | 4 mcg/Kg/min | |
| Eptifibatide [176,188,204,205] | 5.35 | NA | yes | NS | 180 mcg/Kg | 2 mcg/Kg/min | |
| Tirofiban [192,206,207] | 5.5–6.5 | NA | no | NS | 25 mcg/Kg | 4 mcg/Kg/min | |
| Antihypertensives | |||||||
| Clevidipine [208] | 6–8 | 341 | no | not to diluite | 1–2 mg/h | 4–6 mg/h | |
| Clonidine [209,210,211,212,213,214,215,216,217] | 4.5–5.5 | 300 | no | NS | NA | 0.2 mcg/Kg/min | |
| Enalaprilat [218,219,220,221] | 6.5–7.5 | NA | no | D5W/NS | 1.25 mg | 1–5 mg/6 h in bolus | |
| Fenoldopam [222,223,224] | 7.2 | NA | no | D5W/NS | NA | 0.01–1.6 mcg/Kg/min | |
| Hydralazine [225,226,227,228] | 3.4–4.4 | NA | no | D5W | NA | 20–40 mg repeatable (Max dose 300 mg) | |
| Nicardipine [52,229,230,231,232,233] | 3.5 | 300 | no | D5W/NS | NA | 5–15 mg/h | |
| Nitroglicerin [234] | 4 | 428–465 | no | D5W/NS | NA | 5–20 mcg/min | |
| Nitroprusside [22,54,85,224,235,236,237,238] | 3.2–6.5 | 305 | no | D5W | NA | 0.3–10 mcg/Kg/min | |
| Phentolamine [239] | 4.5–6.5 | NA | no | NS | 1–5 mg in bolus | NA | |
| Urapidil [238,240,241] | 5.6–6.6 | 815 | no | D5W | 0.3 mg/Kg | 4–30 mg/h | |
| 5.6–6.6 | 785 | no | NS | 0.3 mg/Kg | 4–30 mg/h | ||
| Anticoagulants | |||||||
| Danaparoid [242,243] | 7 | NA | no | NS | 1200–1500 UI | 400 UI/h for 4 h | |
| Deltaparine [244] | 5–7.5 | NA | no | not to diluite | Variable | Variable | |
| Enoxaparine [171,176,177,182,183,186,201,245,246,247,248,249,250,251,252,253,254,255,256,257,258] | 5.5–7.5 | NA | no | not to diluite | Variable | Variable | |
| Unfractionated Heparin (UFH) [184,185,195,199,200,206,258,259,260,261,262,263,264,265,266,267,268,269,270,271,272,273,274,275,276,277,278,279,280,281] | 5–6 | 298 | no | D5W | 80 UI/Kg | 18 UI/Kg/min | |
| 6–8 | 322 | no | NS | 80 UI/Kg | 18 UI/Kg/min | ||
| Diuretics | |||||||
| Acetazolamide [282] | vesicant | 9.6 | NA | Yes | NS | NA | 250–375 (5 mg/Kg/die) |
| Bumetanide [124,283] | 6.8–7.8 | NA | no | D5W/NS | 1 mg | 0.5–2 mg/h | |
| Ethacrynic acid [284] | 7 | NA | no | NS | 0.5 mg/Kg | 0.5–1 mg/Kg in C.I. | |
| Furosemide [285,286,287,288,289,290,291,292,293,294,295,296,297,298,299,300,301] | 8–9.3 | 286–300 | no | NS | NA | 20–1000 mg/die | |
| Mannitol [125,302,303] | vesicant | 6.3 | 1335 | yes | not diluite | NA | 1.5–2 g/Kg over 30–60 min |
| Potassium canreonate [304,305] | 10 | NA | no | D5W/NS | NA | Up to 800 mg/die | |
| Other Drugs | |||||||
| Calcium chloride [306] | vesicant | 4.3 | 400 | yes | D5W | 0.5–2 g | NA |
| vesicant | 4.5 | 361 | yes | NS | 0.5–2 g | NA | |
| Calcium Gluconate [307] | vesicant | 6–8.2 | 316 | yes | D5W | 1.5–3 g | NA |
| vesicant | 6–8.2 | 298 | yes | NS | 1.5–3 g | NA | |
| Conivaptan [308] | 3.4–3.8 | NA | yes | D5W | 20 mg over 30 min | 20 mg over 24 h | |
| Dextrose ≥ 10% [309] | vesicant | 3.5–6.5 | 505 | yes | D10W | Variable | Variable |
| Ferric carboxymaltose [310] | 5–7 | NA | no | NS | NA | 500–1000 mg | |
| Ferric derisomaltose [311] | 5–7 | NA | no | NS | NA | 20 mg/Kg (max 1000 mg) | |
| Glucagon [309] | 1.8–2.2 | NA | yes | NS | NA | 0.5–1 mg | |
| Magnesium Sulfate [312,313,314,315,316,317,318,319,320,321,322,323] | vesicant | 5.5–7 | 400–500 | no | NS | 1–2 g | 0.5–1 g/h |
| Oxytocin [324] | 3.5 | NA | no | NS | 0.5–1 mU/min | increment of 1–2 mU/min | |
| Salbutamol [141] | 3.5 | NA | no | NS | 4 mcg/Kg | 3–20 mcg/min | |
| Sodium bicarbonate ≥ 8.4% [325] | vesicant | 8.34 | 2000 | yes | NA | Variable | Variable |
| Tranexamic Acid [326,327,328] | 6.5–8 | 316 | no | NS | 10 mg/Kg | NA | |
| 6.5–8 | 346 | no | D5W | 10 mg/Kg | NA | ||
| Potassium chloride (10.1–29.9 mEq/100 mL) [329] | vesicant | 4.2–6 | 202–598 | yes | D5W/NS | Variable | Variable |
| Potassium chloride (30 mEq/100 mL) [329] | vesicant | 4.2–6 | 763 | yes | D5W/NS | Variable | Variable |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Published by MDPI on behalf of the Österreichische Pharmazeutische Gesellschaft. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Quici, M.; Martini, E.; Giustivi, D.; Calloni, M.; Cogliati, C.; Taino, A.; Foschi, A.; Gori, A.; Zappa, P.; Casella, F.; et al. Physicochemical Characteristics of Cardiological Drugs and Practical Recommendations for Intravenous Administration: A Systematic Review. Sci. Pharm. 2025, 93, 13. https://doi.org/10.3390/scipharm93010013
Quici M, Martini E, Giustivi D, Calloni M, Cogliati C, Taino A, Foschi A, Gori A, Zappa P, Casella F, et al. Physicochemical Characteristics of Cardiological Drugs and Practical Recommendations for Intravenous Administration: A Systematic Review. Scientia Pharmaceutica. 2025; 93(1):13. https://doi.org/10.3390/scipharm93010013
Chicago/Turabian StyleQuici, Massimiliano, Elena Martini, Davide Giustivi, Maria Calloni, Chiara Cogliati, Alba Taino, Antonella Foschi, Andrea Gori, Paolo Zappa, Francesco Casella, and et al. 2025. "Physicochemical Characteristics of Cardiological Drugs and Practical Recommendations for Intravenous Administration: A Systematic Review" Scientia Pharmaceutica 93, no. 1: 13. https://doi.org/10.3390/scipharm93010013
APA StyleQuici, M., Martini, E., Giustivi, D., Calloni, M., Cogliati, C., Taino, A., Foschi, A., Gori, A., Zappa, P., Casella, F., Bartoli, A., La Cava, L., Meschia, A., Celano, R., Urso, F., Cattaneo, D., & Gidaro, A. (2025). Physicochemical Characteristics of Cardiological Drugs and Practical Recommendations for Intravenous Administration: A Systematic Review. Scientia Pharmaceutica, 93(1), 13. https://doi.org/10.3390/scipharm93010013





