The Potential of Incorporating a Pharmacist-Only Medicine Category in Poland
Abstract
1. Introduction
- ➢
- Supported by a pharmacist’s involvement in their sale to ascertain the therapeutic need and proper use of the medication;
- ➢
- Stored out of public access;
- ➢
2. Material and Methods
Statistical Analysis
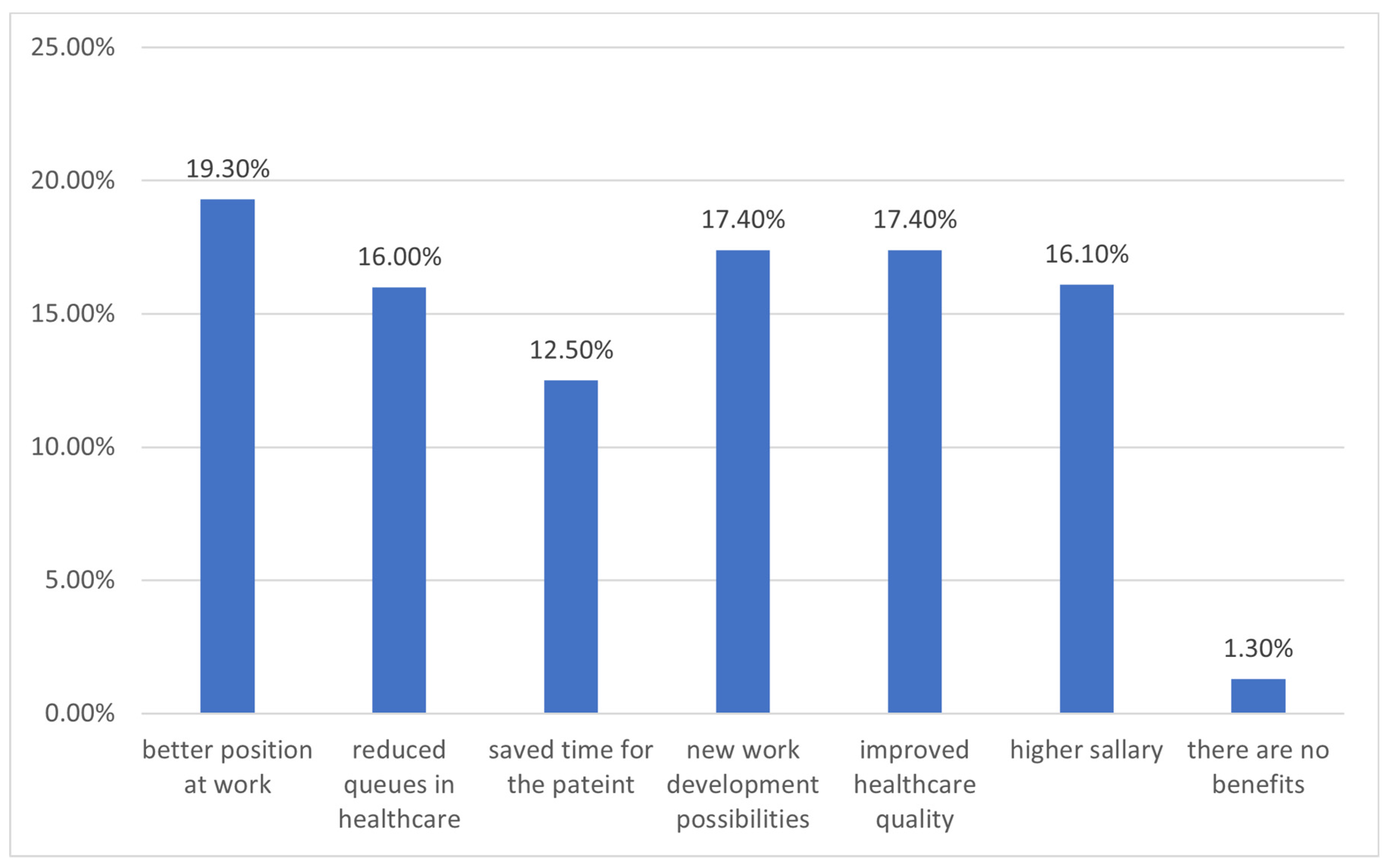
3. Results
4. Discussion
5. Limitations
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- McKnight, A.G.; Thomason, A.R. Pharmacists’ advancing roles in drug and disease management: A review of states’ legislation. J. Am. Pharm. Assoc. 2009, 49, 554–558. [Google Scholar] [CrossRef] [PubMed]
- Merks, P.; Jakubowska, M.; Drelich, E.; Świeczkowski, D.; Bogusz, J.; Bilmin, K.; Sola, K.F.; May, A.; Majchrowska, A.; Koziol, M.; et al. The legal extension of the role of pharmacists in light of the COVID-19 global pandemic. Res. Soc. Adm. Pharm. 2021, 17, 1807–1812. [Google Scholar] [CrossRef] [PubMed]
- Dzionek, J. Pharmaceutical Care in Ontario. J. Med. Sci. 2020, 89, e458. [Google Scholar] [CrossRef]
- Bochniarz, M.; Inglot-Brzęk, E.; Lewandowska, A.; Podgórska, J. Directions of Changes in the Profession of Hospital Pharmacist in Poland. Int. J. Environ. Res. Public Health 2022, 19, 14522. [Google Scholar] [CrossRef] [PubMed]
- Wiśniewski, M.; Religioni, U.; Merks, P. Community Pharmacies in Poland—The Journey from a Deregulated to a Strictly Regulated Market. Int. J. Environ. Res. Public Health 2020, 17, 8751. [Google Scholar] [CrossRef] [PubMed]
- Rada Ministrów Przyjęła Dokument “Polityka Lekowa Państwa 2018–2022” Ministerstwo Zdrowia—Portal Gov.pl. Ministerstwo Zdrowia. Available online: https://www.gov.pl/web/zdrowie/rada-ministrow-przyjela-dokument-polityka-lekowa-panstwa-20182022 (accessed on 10 March 2023).
- Steltenpohl, E.A.; Barry, B.K.; Coley, K.C.; McGivney, M.S.; Olenak, J.L.; Berenbrok, L.A. Point-of-Care Testing in Community Pharmacies: Keys to Success from Pennsylvania Pharmacists. J. Pharm. Pract. 2018, 31, 629–635. [Google Scholar] [CrossRef]
- Lowres, N.; Krass, I.; Neubeck, L.; Redfern, J.; McLachlan, A.J.; Bennett, A.A.; Freedman, S.B. Atrial fibrillation screening in pharmacies using an iPhone ECG: A qualitative review of implementation. Int. J. Clin. Pharm. 2015, 37, 1111–1120. [Google Scholar] [CrossRef]
- Zaprutko, T.; Hromovyk, B.; Lesyk, R.; Lesyk, L.; Kremin, Y.; Kus, K.; Kopciuch, D.; Ratajczak, P.; Paczkowska, A.; Nowakowska, E. Pharmacies for the Pharmacists—Ukrainian Fears and Polish Experiences. Sci. Pharm. 2020, 88, 7. [Google Scholar] [CrossRef]
- Blouin, R.A.; Adams, M.L. The Role of the Pharmacist in Health Care Expanding and Evolving. North Carol. Med. J. 2017, 78, 165–167. [Google Scholar] [CrossRef]
- Świeczkowski, D.; Merks, P.; Cwalina, N.; Jaguszewski, M.J. Development of Pharmacy Practice in European Countries—The Polish Perspective. Pharmacy 2017, 5, 43. [Google Scholar] [CrossRef]
- De Weerdt, E.; Simoens, S.; Hombroeckx, L.; Casteels, M.; Huys, I. Causes of drug shortages in the legal pharmaceutical framework. Regul. Toxicol. Pharmacol. 2015, 71, 251–258. [Google Scholar] [CrossRef]
- De Weerdt, E.; Simoens, S.; Casteels, M.; Huys, I. Toward a European definition for a drug shortage: A qualitative study. Front. Pharmacol. 2015, 6, 253. [Google Scholar] [CrossRef] [PubMed]
- Duong, M.H.; Moles, R.J.; Chaar, B.; Chen, T.F. Stakeholder perspectives on the challenges surrounding management and supply of essential medicines. Int. J. Clin. Pharm. 2019, 41, 1210–1219. [Google Scholar] [CrossRef] [PubMed]
- Acosta, A.; Vanegas, E.P.; Rovira, J.; Godman, B.; Bochenek, T. Medicine Shortages: Gaps between Countries and Global Perspectives. Front. Pharmacol. 2019, 10, 763. [Google Scholar] [CrossRef] [PubMed]
- Pauwels, K.; Huys, I.; Casteels, M.; Simoens, S. Drug shortages in European countries: A trade-off between market attractiveness and cost containment? BMC Health Serv. Res. 2014, 14, 438. [Google Scholar] [CrossRef] [PubMed]
- Deutsch, J. Europe Comes Up Short on Drug Supplies. POLITICO. 2019. Available online: https://www.politico.eu/article/europe-still-coming-up-short-on-drug-supplies/ (accessed on 2 September 2019).
- Shukar, S.; Zahoor, F.; Hayat, K.; Saeed, A.; Gillani, A.H.; Omer, S.; Hu, S.; Babar, Z.-U.-D.; Fang, Y.; Yang, C. Drug Shortage: Causes, Impact, and Mitigation Strategies. Front. Pharmacol. 2021, 12, 693426. [Google Scholar] [CrossRef] [PubMed]
- National Academies of Sciences, Engineering, and Medicine; Health and Medicine Division; Board on Health Sciences Policy; Committee on Security of America’s Medical Product Supply Chain; Shore, C.; Brown, L.; Hopp, W.J. Causes and Consequences of Medical Product Supply Chain Failures. In Building Resilience into the Nation’s Medical Product Supply Chains; National Academies Press: Washington, DC, USA, 2022. [Google Scholar]
- Why Are There Medicine Shortages and What Is the Solution? World Economic Forum. 2023. Available online: https://www.weforum.org/agenda/2023/02/why-is-world-experiencing-medicine-shortages-and-how-can-the-generics-industry-address-supply-challenges/ (accessed on 23 November 2023).
- Emmerton, L. The “third class” of medications: Sales and purchasing behavior are associated with pharmacist only and pharmacy medicine classifications in Australia. J. Am. Pharm. Assoc. 2009, 49, 31–37. [Google Scholar] [CrossRef] [PubMed]
- Medicines: Reclassify Your Product. GOVUK; 2023. Available online: https://www.gov.uk/guidance/medicines-reclassify-your-product (accessed on 13 November 2023).
- Gruchala, K.; Zimmermann, A.; Kawczak, P. Rx-to-Otc Switch and Double Registration Occurrence in Poland—An Illuminative Case Study. Acta Pol. Pharm. 2016, 73, 247–254. [Google Scholar] [PubMed]
- Zaprutko, T.; Kopciuch, D.; Ratajczak, P.; Paczkowska, A.; Adamczak, O.; Kus, K.; Nowakowska, E. The Prescription to Over-the-Counter Switches and Double Registration of Medicines—The Perspective of Pharmacists from the Greater Poland. Acta Pol. Pharm.-Drug Res. 2019, 76, 907–912. [Google Scholar] [CrossRef]
- Aronson, J.K. From prescription-only to over-the-counter medicines (‘PoM to P’): Time for an intermediate category. Br. Med. Bull. 2009, 90, 63–69. [Google Scholar] [CrossRef]
- Zaprutko, T.; Kopciuch, D.; Paczkowska, A.; Sprawka, J.; Cynar, J.; Pogodzińska, M.; Niewczas, K.; Stolecka, A.; Sygit, M.; Michalak, M.; et al. Facebook as a source of access to medicines. PLoS ONE 2022, 17, e0275272. [Google Scholar] [CrossRef] [PubMed]
- Bessell, T.L.; Hiller, J.E.; Sansom, L.N. “Pharmacist only” medicines. Aust. N. Z. J. Public Health 1999, 23, 661–662. [Google Scholar] [CrossRef]
- Casati, A.; Sedefov, R.; Pfeiffer-Gerschel, T. Misuse of medicines in the European Union: A systematic review of the literature. Eur. Addict. Res. 2012, 18, 228–245. [Google Scholar] [CrossRef] [PubMed]
- Emma Hammett Online Drug Addiction & Misuse: New Rules to Combat Misuse. Online First Aid. 2019. Available online: https://onlinefirstaid.com/online-drug-addiction/ (accessed on 9 July 2020).
- Chiappini, S.; Guirguis, A.; Corkery, J.M.; Chiappini, F.S. Misuse of prescription and over-the-counter drugs to obtain illicit highs: How pharmacists can prevent abuse. Pharm. J. 2020, 305, 7943. Available online: https://pharmaceutical-journal.com/article/research/misuse-of-prescription-and-over-the-counter-drugs-to-obtain-illicit-highs-how-pharmacists-can-prevent-abuse (accessed on 15 April 2021).
- Zaprutko, T.; Koligat, D.; Michalak, M.; Wieczorek, M.; Józiak, M.; Ratajczak, M.; Szydłowska, K.; Miazek, J.; Kus, K.; Nowakowska, E. Misuse of OTC drugs in Poland. Health Policy 2016, 120, 875–881. [Google Scholar] [CrossRef] [PubMed]
- Dulaney, K.; Hohmeier, K.; Fisher, C.; Cardosi, L.; Wasson, M. Exploring pharmacists’ perceptions regarding influenza and streptococcal testing within a chain pharmacy. J. Am. Pharm. Assoc. 2018, 58, 438–441. [Google Scholar] [CrossRef]
- Merks, P.; Religioni, U.; Waszyk-Nowaczyk, M.; Kaźmierczak, J.; Białoszewski, A.; Blicharska, E.; Kowalczuk, A.; Neumann-Podczaska, A. Assessment of Pharmacists’ Willingness to Conduct Medication Use Reviews in Poland. Int. J. Environ. Res. Public Health 2022, 19, 1867. [Google Scholar] [CrossRef]
- Rajiah, K.; Sivarasa, S.; Maharajan, M.K. Impact of Pharmacists’ Interventions and Patients’ Decision on Health Outcomes in Terms of Medication Adherence and Quality Use of Medicines among Patients Attending Community Pharmacies: A Systematic Review. Int. J. Environ. Res. Public Health 2021, 18, 4392. [Google Scholar] [CrossRef]
- Kirkdale, C.L.; Nebout, G.; Megerlin, F.; Thornley, T. Benefits of pharmacist-led flu vaccination services in community pharmacy. Ann. Pharm. Fr. 2017, 75, 3–8. [Google Scholar] [CrossRef]
- Gallimore, C.E.; Porter, A.L.; Barnett, S.G.; Portillo, E.; Zorek, J.A. A state-level needs analysis of community pharmacy point-of-care testing. J. Am. Pharm. Assoc. 2021, 61, e93–e98. [Google Scholar] [CrossRef]
- Hohmeier, K.C.; Loomis, B.; Gatwood, J. Consumer perceptions of and willingness-to-pay for point-of-care testing services in the community pharmacy. Res. Social. Adm. Pharm. 2018, 14, 360–366. [Google Scholar] [CrossRef] [PubMed]
- Brewer, A.; Hanna, C.; Eckmann, L.; Schadler, A.; Divine, H. Patient awareness, willingness, and barriers to point-of-care hepatitis C screening in community pharmacy. J. Am. Pharm. Assoc. 2018, 58, S69–S72. [Google Scholar] [CrossRef] [PubMed]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zaprutko, T.; Sprawka, J.; Maciuszek-Bartkowska, B.; Ratajczak, P.; Kopciuch, D.; Paczkowska, A.; Kus, K. The Potential of Incorporating a Pharmacist-Only Medicine Category in Poland. Sci. Pharm. 2024, 92, 11. https://doi.org/10.3390/scipharm92010011
Zaprutko T, Sprawka J, Maciuszek-Bartkowska B, Ratajczak P, Kopciuch D, Paczkowska A, Kus K. The Potential of Incorporating a Pharmacist-Only Medicine Category in Poland. Scientia Pharmaceutica. 2024; 92(1):11. https://doi.org/10.3390/scipharm92010011
Chicago/Turabian StyleZaprutko, Tomasz, Józefina Sprawka, Barbara Maciuszek-Bartkowska, Piotr Ratajczak, Dorota Kopciuch, Anna Paczkowska, and Krzysztof Kus. 2024. "The Potential of Incorporating a Pharmacist-Only Medicine Category in Poland" Scientia Pharmaceutica 92, no. 1: 11. https://doi.org/10.3390/scipharm92010011
APA StyleZaprutko, T., Sprawka, J., Maciuszek-Bartkowska, B., Ratajczak, P., Kopciuch, D., Paczkowska, A., & Kus, K. (2024). The Potential of Incorporating a Pharmacist-Only Medicine Category in Poland. Scientia Pharmaceutica, 92(1), 11. https://doi.org/10.3390/scipharm92010011






