Abstract
Microneedles are gaining popularity as a new paradigm in the area of transdermal drug delivery for biomedical and healthcare applications. Efficient drug delivery with minimal invasion is the prime advantage of microneedles. The concept of the microneedle array provides an extensive surface area for efficient drug delivery. Various types of inorganics (silicon, ceramic, metal, etc.) and polymeric materials are used for the fabrication of microneedles. The polymeric microneedles have various advantages over other microneedles fabricated using inorganic material, such as biocompatibility, biodegradation, and non-toxicity. The wide variety of polymers used in microneedle fabrication can provide a broad scope for drug delivery and other biomedical applications. Multiple metallic and polymeric microneedles can be functionalized by polymer coatings for various biomedical applications. The fabrication of polymeric microneedles is shifting from conventional to advanced 3D and 4D printing technology. The multifaceted biomedical applications of polymeric microneedles include drug delivery, vaccine delivery, biosensing, and diagnostic applications. Here, we provide the overview of the current and advanced information on polymers used for fabrication, the selection criteria for polymers, biomedical applications, and the regulatory perspective of polymer-based and polymer-coated microneedles, along with a patent scenario.
1. Introduction
Microneedle-based drug delivery is the most widely used technique for obtaining systemic efficacy of medications through the transdermal (TD) route. The oral route is far more straightforward: it is easier to obtain patient compliance, and it requires no special expertise and is less expensive than alternative delivery systems. However, it has some drawbacks, including: low drug bioavailability at the target site, hepatic biotransformation, diverse types of enzymatic reactions at the gastric pH, and inappropriate absorption due to the present clinical state [1].
It has been discovered that the TD route of drug administration can directly access the first layer of skin, while avoiding the difficulties associated with other drug delivery systems. To address the problems with conventional approaches, microneedle-based drug delivery via the TD route is recently becoming popular. Several projections on the microneedle can penetrate into the epidermis. MNs can be easily fabricated with materials such as metal, polymer, hydrogel, silicon, ceramic, etc. Microneedles (MNs) penetrate the stratum corneum and reach the upper dermis without contacting nerve fibers or blood vessels, which results in painless administration [2,3,4].
Microneedles have a length typically ranging from 150 to 1500 μm [5,6,7]. Designing of MNs mainly depends on the fabrication method, the material quality, the stability of material, and the shape/geometry of them [8].
Protein and peptide drug delivery through MNs have great advantages, such as improved bioavailability, improved patient compliance, and stability. MNs are classified into several types, such as solid, hollow, coated, dissolving, and hydrogel-forming. The main objective of this MN technology is to puncture the stratum corneum in order to allow the entry of larger molecules through skin and, hence, increase permeability [9].
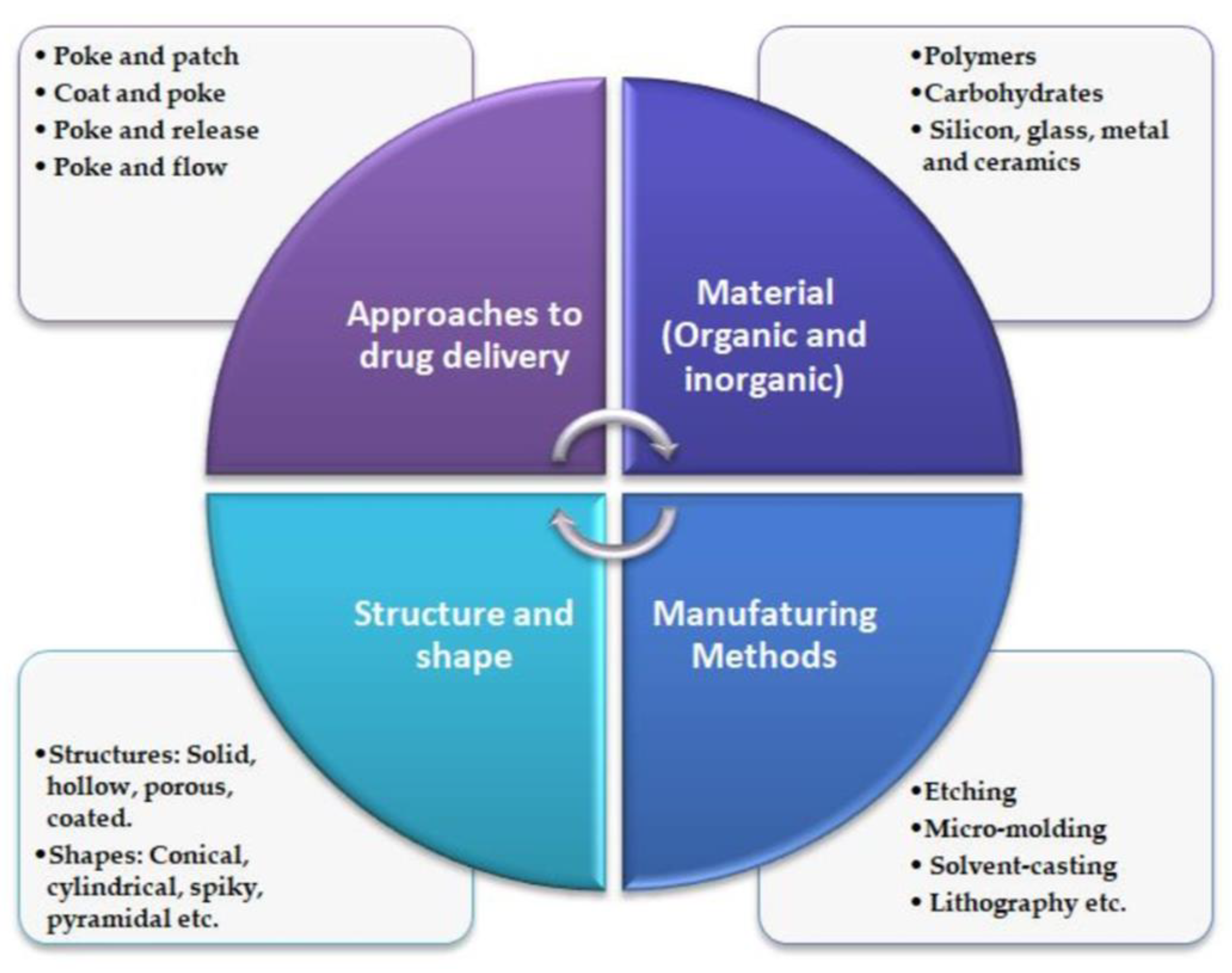
The literature shows that numerous fabrication techniques, materials, and approaches are employed for MN manufacturing (Figure 1). Its size, shape, radius of the tip, needle height, base width, overall design, and geometry are crucial for improving its mechanical performance and strength [10].
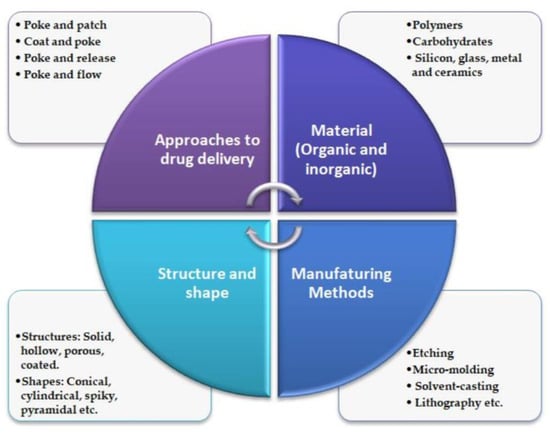
Figure 1.
General characteristics of MN technology. Adapted from Ref. [11].
2. Types of Microneedles
2.1. Solid Microneedles
Solid MNs are basically fabricated by micromachining techniques, such as photolithography and the etching method. The delivery of drugs occurs through the “poke-and-patch” mechanisms through the development of microchannels. Drug release can occur through passive diffusion. Solid MNs are typically made of non-biodegradable or degradable materials. They can be fabricated using microelectromechanical systems (MEMS) [12].
Solid MNs can be used for the administration of proteins, peptides, growth hormone, insulin, and vaccines. They can also be used to deliver drugs in semisolid dosage forms. Solid MNs make the skin more permeable without damaging the stratum corneum. Compared to hollow MNs, solid MNs are easier to formulate, have sharper tips, and have greater mechanical strength. Solid MNs are characterized by their physical, chemical, and mechanical properties. They can also be used to detect biomarkers in biosensing. This is because the needles can be coated with a material that can sense a particular molecule, such as glucose or lactate. The physical properties include the dimensions, various forms, and surface morphology. The dimensions include a length of 50–800 µm and diameter of 10–200 µm. The surface morphology of solid MNs can be studied using atomic force microscopy [13].
2.2. Hollow Microneedle
The stability and functionality of hollow MNs (HMNs) depend on the catheter aperture, height, and needle tip. Drugs can be administered using the “poke-and-flow” mechanism. HMNs allow the administration of larger doses of medication. They are typically made up of silicon, metal, glass, and ceramic materials. The main advantage of hollow microneedles is that they are painless. The size of a hollow needle is in the micron range. The flow of drugs through HMNs can be influenced by multiple factors, including tip dimension, length, pressure, inner diameter, insertion, and retraction of depth. One drawback of this technology is the occasional blockage of the MN and the crucial manufacturing process [14]. HMNs are small in length, typically around 1 mm. The diameter and length can vary from a few microns to several hundred microns. Fabrication methods such as photolithography, laser ablation, and micromolding techniques can be used to manufacture hollow MNs. Because of their small size, it can be challenging to fabricate hollow MNs, as it is difficult to maintain the integrity of the hollow bore. A hollow MN is used for body fluid sampling, while a syringe and a micropump are used to control the flow of medicine. Using HMNs, regulated drug administration is achievable; it makes a simple drug concentration–time profile. The basic design for an HMN includes a microfluidic chip, a micropump, and a heater [15].
2.3. Coated Microneedles
The “coat-and-poke” mechanism is used to deliver drugs in this method. Titanium and stainless steel are used to make it. The drug coating is applied to the MNs before they are inserted under the skin in a one-step process. The skin can absorb the drug coating from the MNs. The MNs are then dissolved and removed. The tip and shaft of MNs can only be coated with a certain amount of medication. The drug dosage is affected by the size of the needle and the thickness of the coating solution. The coating can improve the functionality and target specificity of the MN. It also improves the mechanical properties of the MN, making it more durable and resistant to breakage. Coating materials such as ceramics, metals, polymers, and hydrogels are used to achieve target-oriented medication delivery. Metal coatings increase MN stability and release-rate kinetics. Polymer coatings offer a barrier of protection and promote tissue regeneration. Coatings can be applied by a variety of mechanisms, such as electroplating and physical vapor deposition for metal coatings. Dip coating and spray coating are generally used in the polymer-coating method. Stainless sheet is used in the fabrication of MNs [9].
Other methods used in MN coating include gas-jet drying and electrohydrodynamic atomization (EHDA) technology [16]. Coated MNs can be used to administer drugs, including proteins and peptides, in a minimally invasive manner. Self-administration is possible with coated MNs, which does not require hospital visits, specifically trained staff, or their associated cost. One barrier that must be addressed is the development of biocompatible coated MNs that do not have any side effects. The second difficulty is that the coating process can sometimes be difficult to scale up due to its expensive and time-consuming manufacturing procedure. With continuous research and development, coated MNs have the potential to transform the healthcare system and improve quality of life for millions of people around the world [17].
2.4. Dissolving Microneedles
Dissolving MNs (DMNs) are extremely small, with lengths of a few millimeters. This technique involves encapsulating drugs within MNs. Once inserted, the MNs dissolve and cannot be removed from the skin after insertion. DMNs are typically composed of dextrin, polyglycolic acid, chondroitin sulphate, and polyvinylpyrrolidone. Its safest drug delivery method has no negative consequences after diffusion into the skin. The efficiency of dissolving MNs is influenced by their mechanical strength, flexibility, and dissolution time. The effectiveness of a dissolving MN depends on its ability to load drugs, its drug release profile, and its pharmacokinetic characteristics [18].
The two most important aspects to take into account when choosing a dissolving MN are the appropriate polymer, and its release kinetics. The most popular methods are solvent casting, droplet-born air blowing, laser machining, hot embossing, microinjection molding, and ultrasonic welding. The performance of MNs is affected by their hygroscopic nature. If a DMN dissolves too quickly, the body cannot absorb the drug. If it dissolves too slowly, skin irritation and discomfort may occur. Scientists are working to develop MNs that dissolve at the optimum rate of delivery. Another challenge in developing DMNs is its degradation. Biodegradable materials are susceptible to degradation by pathogens, which can compromise the stability of the MN. DMNs are frequently used because of their benefits for patient compliance, decreased dose frequency, and enhanced tissue bioavailability [10].
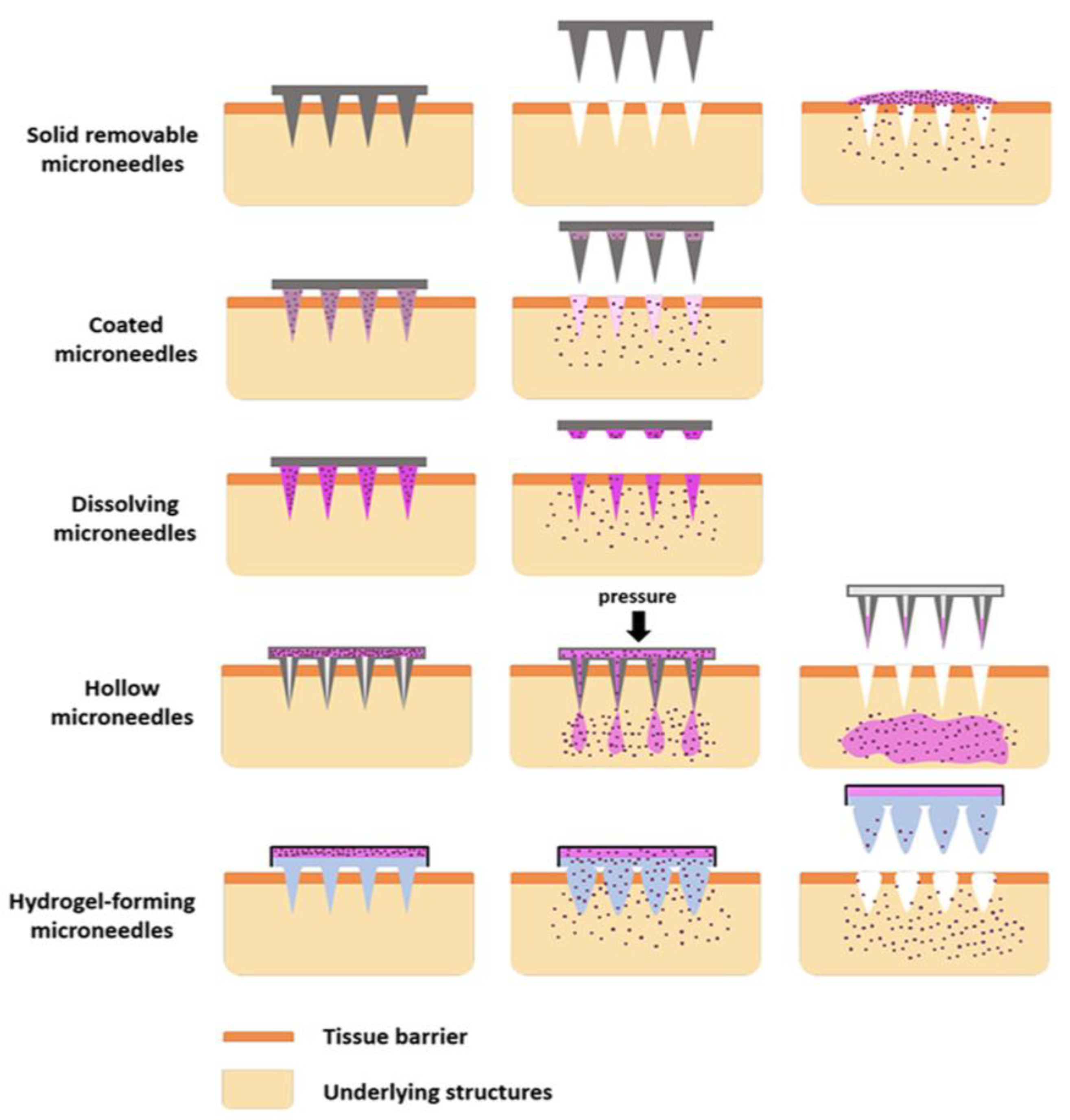
Figure 2 illustrate the types and drug delivery mechanisms of MNs.
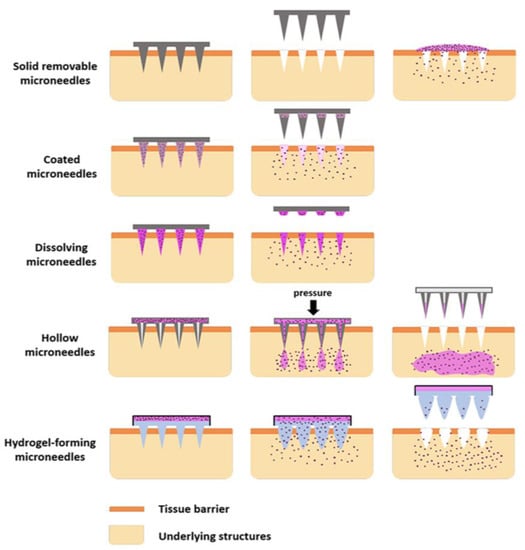
Figure 2.
Types of microneedles with drug delivery mechanisms. Adapted with permission from Ref. [19]. Copyright 2018 Elsevier.
2.5. Hydrogel Microneedles
Hydrogel-forming MNs is a novel and promising technique that overcomes the challenges of traditional drug delivery. Hydrogels and swellable polymer are crosslinked to create hydrogel MNs. When this sort of MN comes into contact with skin, the hydrogel causes them to expand, and water ingestion occurs. Drugs can be delivered in a minimally invasive way. With the help of hydrogel MNs, TD administration of proteins and vaccines is possible. Many prefer this convenient alternative method because of its painless medicine delivery. Controlled and prolonged drug release delivery is achievable with hydrogel MN. This type of MN has a greater ability to load drugs and an increased drug release kinetics [20].
Hydrogel-forming MNs are fabricated by various methods, such as micro-modeling, a potential method; polymer casting; laser drilling, etc. A controlled manner drug administration is possible with this type of MN. The typical dimensions are a height of the needle around 500–600 µm, and base width around 350 µm. The shape of the needle is conical in nature and it includes around 121 needles (11 × 11). They are made up of a complete aqueous blend. The surface morphology of the MN is often examined using scanning electron microscopy to characterize hydrogel MNs. The swelling index of the MN can be assessed based on the change in volume and weight when it comes into contact with liquid. Compression and blending techniques can be used to assess the hydrogel MNs. The drug release profile can be estimated with the aid of UV and HPLC equipment. The characteristics of hydrogel MNs demonstrate their safe and efficient application in the delivery of drugs [21].
3. Polymeric Microneedle Technology
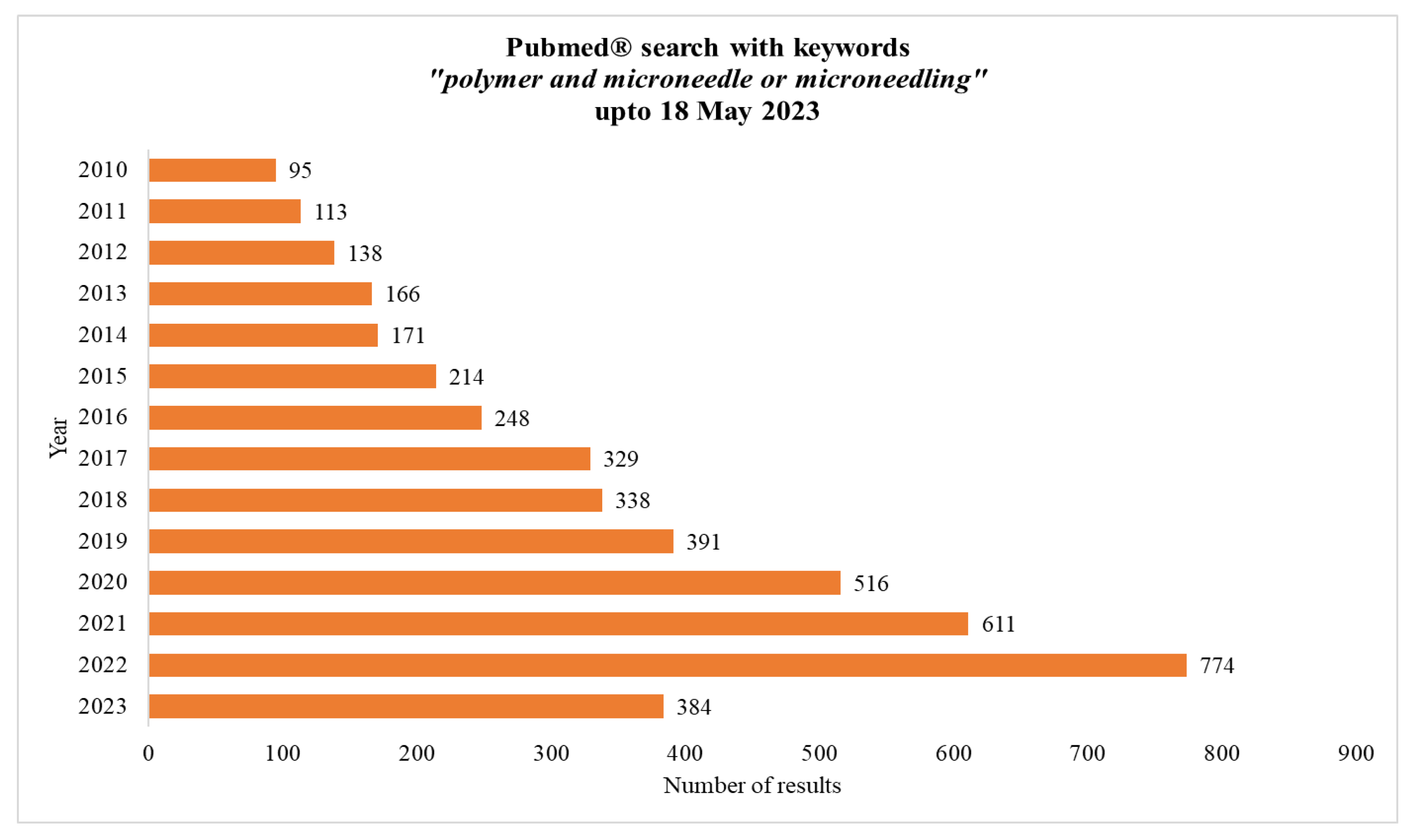
The applications of polymeric MNs are diverse and include drug delivery, skin analysis, and TD sensing. The use of polymers in MN fabrication offers a promising approach for drug delivery and other applications, providing a safe, efficient, and customizable way to deliver drugs, vaccines, and other substances through the skin. The increasing interest of the scientific community is evident from the increasing number of publications on the topic in Pubmed®, as depicted in Figure 3, showing results up to 18 May 2023.
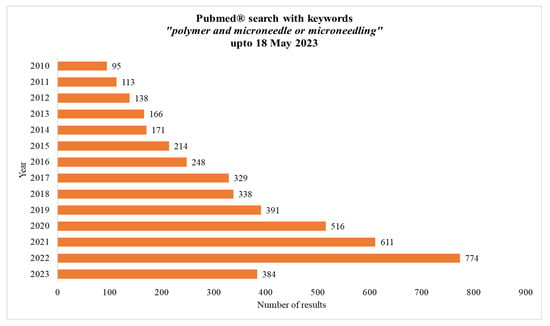
Figure 3.
Result of search with keywords “polymer and microneedle or microneedling” in Pubmed®.
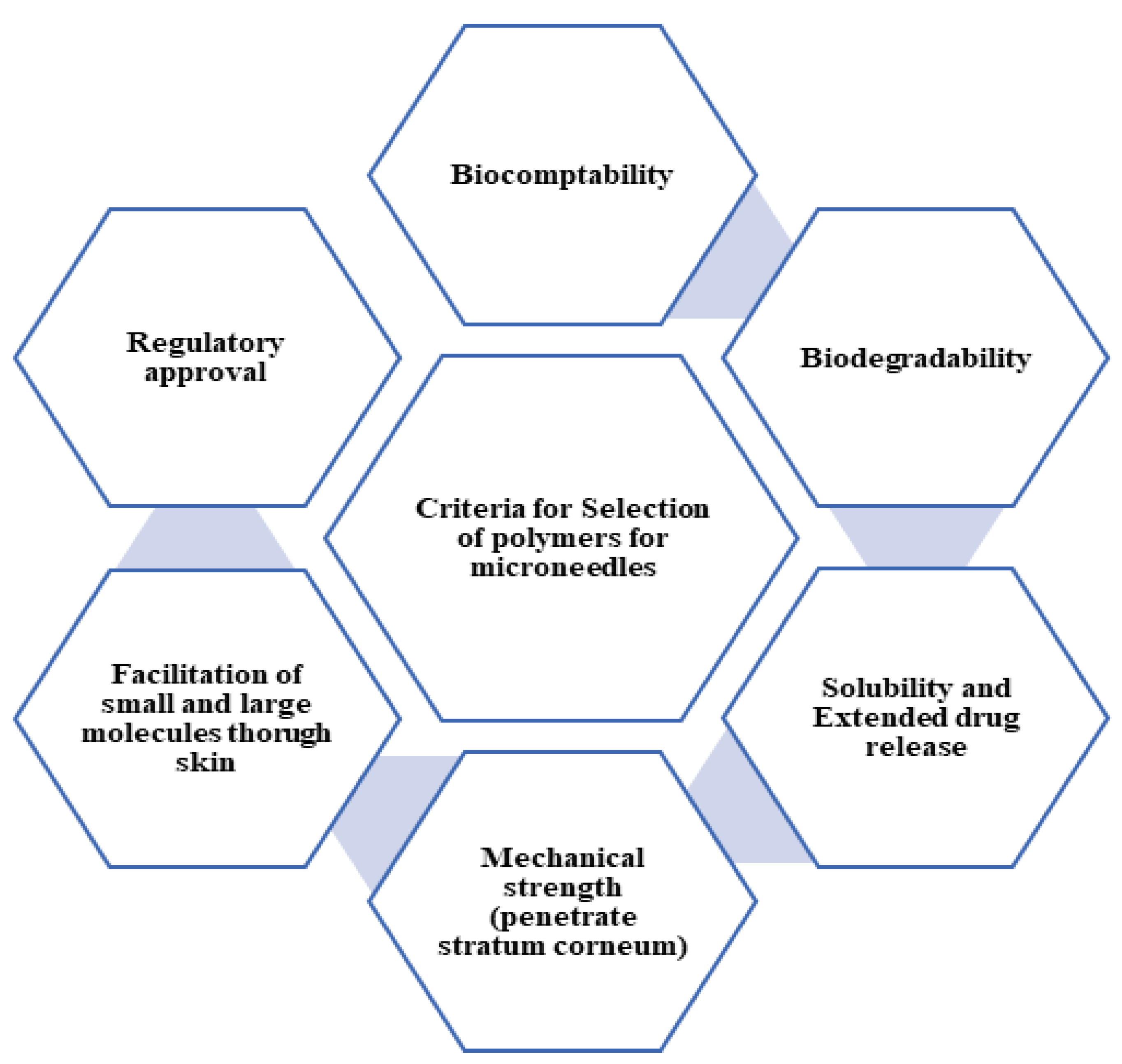
The major challenges with the development of MNs includes preclinical testing and clinical trials to establish safety and efficacy, a stable manufacturing process to achieve constant standards required for the desired application, regulatory approvals of the developed products, ease of use, and patient compliance. Polymers are commonly used for the fabrication of MNs as polymers of a diverse nature are available for their development. Polymers are considered as the choice of raw material for MN preparation as they do not show immune reactions and their functionality can be modulated needed by physicochemical modifications [22]. Polymers can be used to develop drug delivery systems that are capable of targeting specific tissues or cells, increasing drug efficacy while reducing drug toxicity [23]. The design of the polymeric system enables the release of drugs in response to external factors such as pH or temperature, ensuring accurate and controlled drug delivery at the intended moment [24]. These polymer characteristics can be beneficial to the development of biosensing devices which allow theranostic use. When choosing a polymeric matrix for MN formulation, factors such as biocompatibility (BC), biodegradability (BD), solubility, the facilitation of small and large molecules, extended drug release characteristics, and mechanical properties should be taken into consideration [25,26], as illustrated in Figure 4.
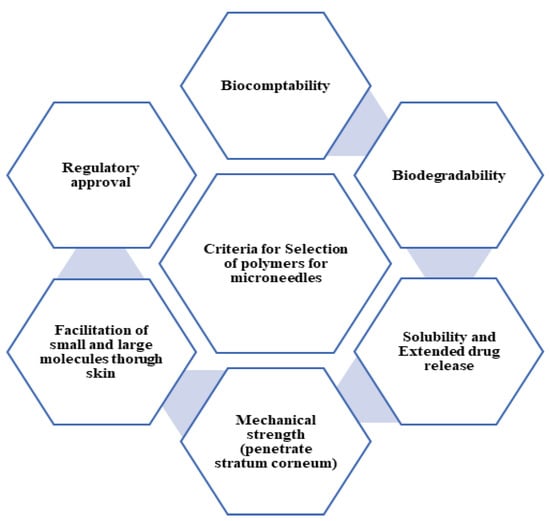
Figure 4.
Criteria for selection of polymers for MNs.
MNs are categorized as either dissolving or swellable depending on both the fabrication method and the polymer’s type. This may be based on the polymer dissolution kinetics, and drug’s response to external stimuli such as pH, temperature, and humidity, as well as the controlled release profile [27].
Several moulding techniques, such as master preparation, mould fabrication, and plasticization of thermoplastic polymers, are typically used to produce polymeric MNs. For the production of MNs, both natural and synthetic polymers are used [28].
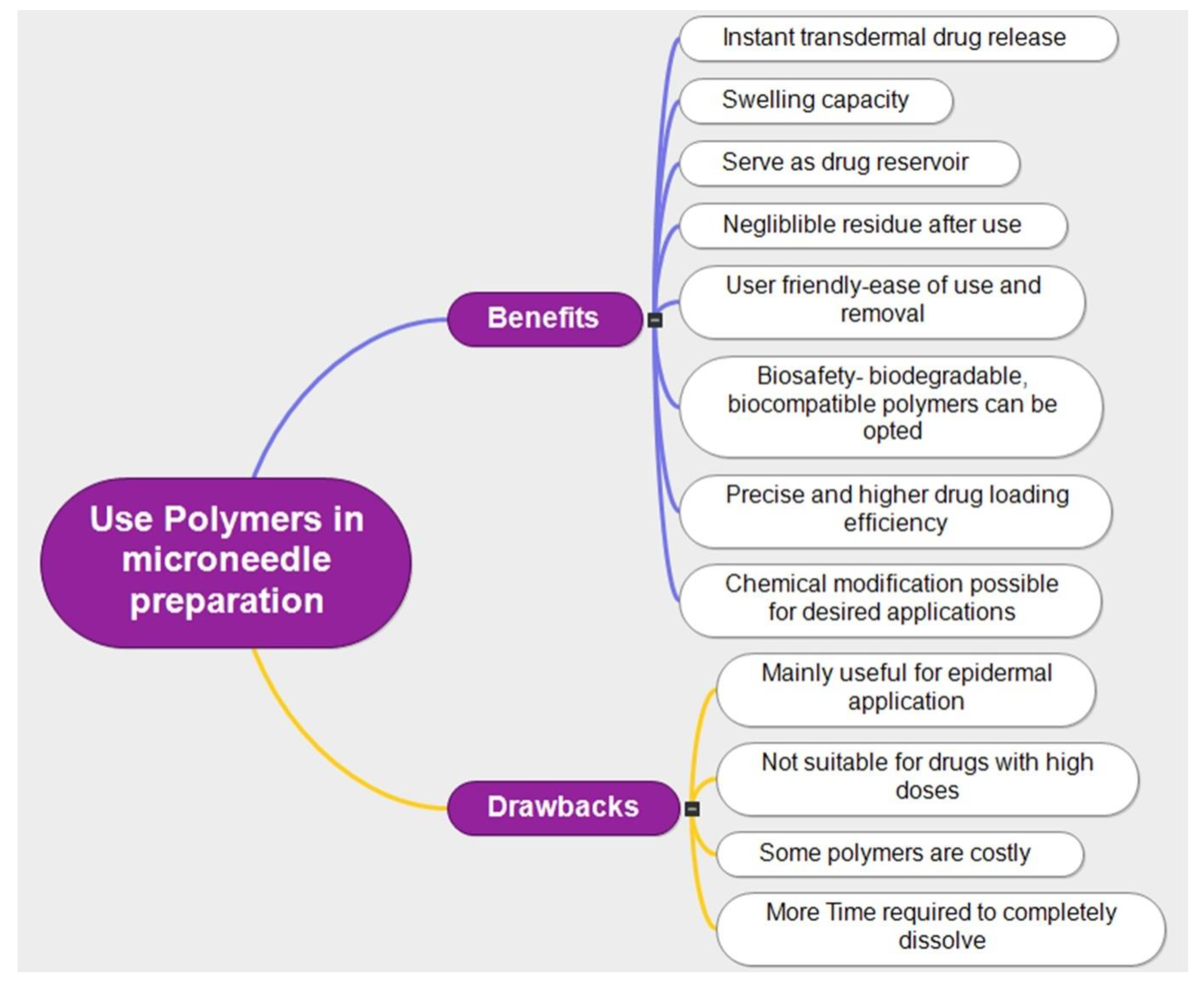
Polymers including poly(lactic-co-glycolic acid) (PLGA), polyvinyl alcohol (PVA), and polyethylene glycol are frequently used in the production of MNs. Polymeric MNs provide several benefits over more traditional medication delivery methods, including intravenous or oral administration. Polymeric MNs can skip first-pass metabolism in the gastrointestinal tract, improve bioavailability, and lower systemic toxicity [29,30,31]. Moreover, they provide a simple and practical means of drug delivery, making them especially beneficial for patient populations including children and the elderly. In preclinical and clinical trials, polymeric MNs have demonstrated promising outcomes for the delivery of insulin, vaccinations, and other medications, in order to improve their design, effectiveness, and safety for widespread clinical usage [32]. The typical benefits and drawbacks of using polymers in MN technology are illustrated in Figure 5.
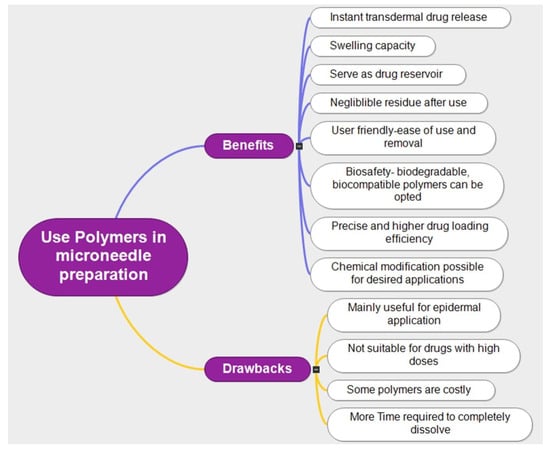
Figure 5.
Benefits and drawbacks of using polymers for MN preparation.
3.1. Classification of Polymer Used for Microneedle Fabrication
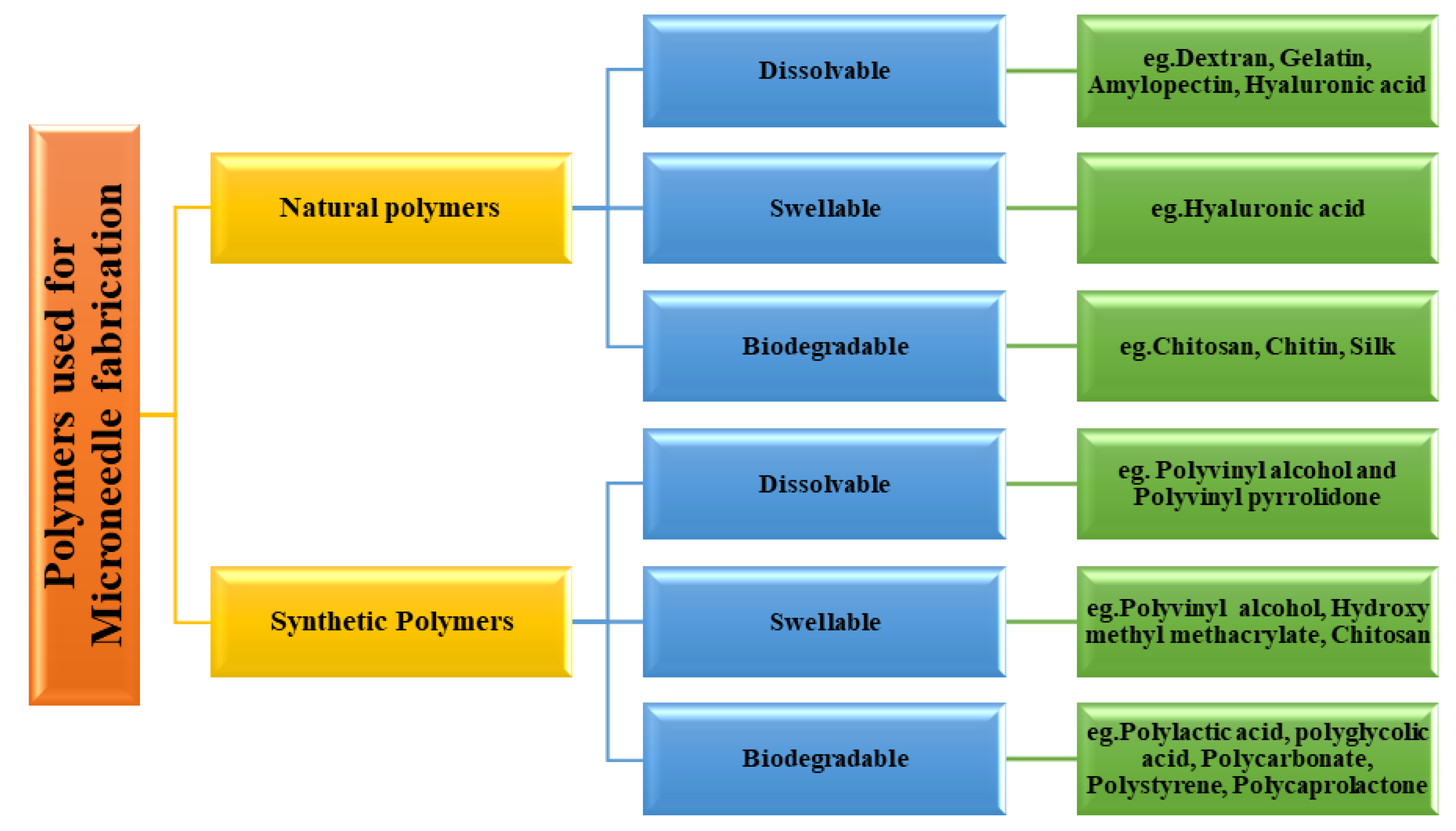
Polymers used for MN fabrication can be classified as natural and synthetic polymers [27]. Natural polymers used for this purpose include dextran, gelatin, amylopectin, hyaluronic acid, chitosan, chitin, and alginate. Synthetic polymers used for this purpose include hydroxypropylmethylcellulose, hydroxypropylcellulose, polyvinyl alcohol, polylactic acid, and polystyrene [28].
The natural polymers are eco-friendly materials which can be used in their natural form or with further physical or chemical modification per the requirement of the task. Synthetic polymers can be prepared by chemical or biochemical processes. These synthetic processes permit tailor-made functionalization. The synthetic processes allow the modulation of desired properties of the polymers such as mechanical strength, degradation characteristics, solubility, drug release behaviour, etc. [33]. The classification of polymers used in the fabrication of the MN is depicted in Figure 6.
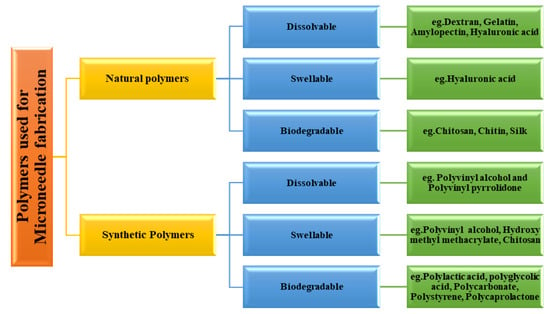
Figure 6.
Polymers used for microneedle fabrication.
3.1.1. Dextran
Dextran is a polysaccharide formed by glucose units associated through α-1,6 and α-1,3 bonds. It is widely used in drug delivery as a carrier due to its BC, BD, low immunogenicity, and potential to form hydrogels, nanoparticles, and an injectable polymer matrix [34].
Dextran methacrylate-based MN cargos containing the drugs Adriamycin and Oral MEK inhibitor GSK1120212 concurrently for a constant TD release were reported by Huang S. et al. for subcutaneous drug release. The hydrogel MNs were synthesized by a two-step process involving the reaction of photocrosslinkable dextran methacrylate with a crosslinking agent and the subsequent loading of the drugs. The prepared hydrogel MNs were found to be safe and effective for the continuous TD administration of the drugs, to allow synergistic drug delivery at a depth of at least 600 µm in the skin, to attain synergic delivery of both drugs [34]. Dextran methacrylate hydrogel MNs were also used recently for the delivery of Tofacitinib and α-melanocyte-stimulating hormone for the treatment of a skin condition called vitiligo. It was revealed that it improved the skin and hair pigmentation [35].
Non-degradable hydrogel photon-activated MNs to suppress protein aggregation for TD drug delivery were reported. To create the MNs, a dextran derivative was utilized, and a photolithography technique was employed to fabricate four-pointed star MNs. The hydrogel network was formed by combining sulfobetaine (SPB) monomer with dextran-glycidyl methacrylate/acrylic acid. Integrating poly-SPB into the MN system ensures that proteins are safeguarded during delivery and the system possesses the required strength for dermal penetration. The microneedling system offers a better drug loading capacity and effective drug release rate. Furthermore, the protein loading capacity of the hydrogel microneedling system was evaluated for the first time, and the results show that lysozyme and insulin could be incorporated into the dry microneedling system at practically a comparable capacity. The potential of these microneedling systems for protein delivery opens up the possibility of administering therapeutic proteinaceous materials to treat disorders where conventional drugs fail. The results show that MNs could be an effective option for administering therapeutic drugs [36].
3.1.2. Hyaluronic Acid
Hyaluronic acid is a natural constituent of the skin which is biocompatible, allows the tuning of viscoelastic characteristics, and is hygroscopic in nature. It is formed by repeating pieces of D-glucuronic acid with N-acetylglucosamine, which are connected by a β-(1–3) bonds. It is a vital biological material involved in multiple functions including angiogenesis, cell growth, immunity, and antioxidants in the body [37].
Acrylate-modified hyaluronic acid (MeHA) has been reported by Chen S.X. et al. The ultraviolet photocrosslinking MeHA improves the mechanical strength, showing a failure force of about 18N in a compression test. It allows skin insertion up to a depth of 300 µm in animal studies. Nanoparticle-loaded MNs exhibit swelling and about 80% drug release within 10 min in a phosphate buffer at pH 7.4, and simulated body fluid at pH 6.5 and 7.4 [38]. Further, the use of ultrasonic stimulation and electrical stimulation was found to facilitate drug penetration through the skin from hyaluronic acid MN patches. Acoustic pressure in ultrasonic stimulation and electrostatic force in electric stimulation increase the drug diffusion across the skin. The correlation of concentration of hyaluronic acid with parameters viz. solid height and tip angle was established. Increasing concentration from 1 to 4 wt% increased solid height while reducing the tip angle, indicating that the concentration of the polymer is a vital parameter in the design of MNs [39].
Y.H. Feng et al. reported the mechanism of delivery of insulin through MNs prepared using polyvinyl alcohol and hyaluronic acid. The study discusses the diffusion properties of insulin and gives an idea about the role of polymers in controlling protein release. The study provides a scientific basis for the development of an insulin-loaded dissolvable microneedling system. The results indicate that hyaluronic acid solution is a better material for the development of an insulin microneedling system compared to other polymers under study. The interaction between insulin and hyaluronic acid is stronger than that between insulin and polyvinyl alcohol. The study shows that these polymers do not affect the nature and expression of insulin in biological systems [40].
3.1.3. Chitin and Chitosan
Chitin and chitosan are widely used in the development of TD patches. Chitosan alkaline polysaccharide is derived from chitin. It possesses highly desired characteristics such as BD, safety, BC, antimicrobial properties, and hemostasis. A smart temperature-responsive MN array patch was reported by Chi J. et al. The system is effective in wound-healing promotion and has antibacterial properties against S. aureus and E. coli, which may be further useful in wound healing. It was reported that a chitosan-based microneedling system works through a variety of mechanisms such as controlling inflammation, collagen deposition, forming newer blood vessels, and promoting granulation cell formation for wound healing [41].
Bletilla striata polysaccharide/chitosan composite bilayer dissolvable MNs containing asiaticoside that achieved scar-free wound healing in rats were reported by Jinying Lv et al. The MNs have a better antimicrobial property and also control fibroblast proliferation. It was demonstrated that Bletilla striata had a substantial anti-inflammatory and curative effect in the early stage. Asiaticoside shows a strong inhibition of scar fibroblasts in the later phases. The synergistic treatments of Bletilla striata and asiaticoside healed the wounds swiftly, and are beneficial for practical clinical use. Additionally, overexpression of TGF-β1 and type-I collagen in scarred skin were reduced by the MN treatment, which could prevent scar formation. The study provides a new strategy for effective scarless wound dressings [42].
3.1.4. Alginate
Alginate has already demonstrated its usefulness in drug delivery owing to its BD and BC. Modified alginates provide extensive applications in MN fabrication. MN fabricated using alginate also showed better mechanical strength [3,43]. Tiraton et al. demonstrated the fabrication of a sodium alginate–gelatin MN for the TD delivery of clindamycin. Clindamycin has limited permeation through the stratum corneum which can be overcome with MN technology. The resulting MNs were found to be nontoxic in the study performed using normal human dermal fibroblast (NHDF) cells and also showed the significant inhibition of Cutibacterium acnes [44]. Yu et al. also reported the fabrication of polymeric MNs using alginate and hyaluronate for the TD delivery of insulin. The resulting MN showed adequate mechanical strength for insertion and better biodegradation to release the insulin [45]. A similar kind of study is also demonstrated by Zhang et al. by fabricating calcium-crosslinked maltose–alginate MN for insulin delivery. The in vivo study showed the better insertion and degradation-mediated release of insulin in diabetic rats [46].
3.1.5. Gelatin
Gelatin is a protein derived from collagen that is commonly used in food, pharmaceuticals, and cosmetics. It is a biodegradable and biocompatible material that has been explored for use in drug delivery systems, including MNs [47]. It is GRAS-listed substance commonly found in pharmaceutical formulations and food products with no significant adverse effects.
Gelatin can be used as a drug carrier or excipient to enhance drug delivery efficiency, reduce the toxicity of the drug, and provide a controlled release of the drug. It is explored for use in the delivery of a variety of drugs, proteins, peptides, vaccines, and cancer therapeutics [48].
Demir et al. demonstrated that MNs made from crosslinking gelatin methacrylate and polyethyleneglycol diacrylate and incorporating molybdenum sulfide (MoS2) nanosheets as a photothermal constituent have the potential to deliver high molecular drugs. It is swelling-type polymer. TD administration of insulin loaded into MoS2-MN TD patches decreases blood sugar in an animal study and is reported to provide equivalent results in subcutaneous injectable insulin preparation. The developed MN works using near-infrared light. It may be necessary to make a compact device for clinical use. However, technology transfer and scale-up issues need to be overcome for the developed MN to be used clinically in the future [49].
The MNs fabricated with a conjugate of gelatinmethacryloyl (GelMa) and β-cyclodextrin (β-CD) were reported by X. Zhou et al. They improved the stability and solubility of curcumin by forming a drug-inclusion complex. The resulting complex could be incorporated into a microneedling system which possesses adequate mechanical strength for dermal penetration and a tailor-made drug release. The in vivo studies showed that the developed curcumin microneedling array shows superior efficacy and provides better penetration than a control non-TD patch. The results demonstrated the BC and degradation characteristics of the polymeric MN array and suggested that hydrogel-based MN arrays are a potential alternative for the delivery of poorly water-soluble drugs [50]. In a recent study, GelMA MNs containing amoxicillin (AMX) were successfully developed by a 3D-printing technique. The 3D-printed AMX-loaded GelMA MNs displayed good mechanical and swelling properties, and demonstrated efficient drug release and antibacterial activity against both S. aureus as well as E. coli. It was concluded that 3D-printed drug-loaded GelMA MNs have a great potential for use as an advanced device for TD therapeutic delivery and could be useful for managing bacterial infections. The developed microneedling system can contain a higher amount of the drug and therefore can be used for a longer duration. The authors claim that the details reported the use of a novel method in the study, making this study a valuable contribution to the field [49].
3.1.6. Gantrez
Gantraz is a copolymer of methyl vinyl ether and maleic anhydride. One of the main benefits of using Gantrez in TD drug delivery is the bioavailability of certain drugs. Gantrez® S-97 forms a super-swellable MN system. The use of Gantrez can also help improve the stability and shelf-life of MN-based drug delivery systems [51].
Two MN formulations of Gantrez were reported by Donnelly et al. The first one consists of hydrolyzed gel of poly (methylvinylether-co-maleic acid) while another one was prepared by crosslinking with poly (ethylene glycol). The crosslinked system was used for MN preparation and it was reported that iontophoresis could enhance the permeation of large molecules such as insulin through the skin, but was not promising for small molecules such as ibuprofen [52]. It swells in the presence of water and possesses sufficient mechanical strength to work out microprotrusions to break the stratum corneum barrier for the drug delivery with MNs. The polymer (Gantrez)-to-crosslinker (polyhydric alcohol) ratio is important for the formation of a microprotrusion array [53].
Another study aimed to investigate the effectiveness of a poorly permeable acyclovir-loaded dissolving polymeric microneedling array system for the treatment of herpes labialis (cold sores), prepared with Gantrez®S-97. Results showed that the microneedling array system improved the local delivery of acyclovir, with in vitro permeation studies showing a 45-times-higher percentage of total acyclovir loading compared to the Liposore® cream formulation. It delivered acyclovir to epidermal layers at a five-times-higher concentration than required for HSV infection treatment, showing the potency of the medication. In vivo studies showed the successful intradermal delivery of acyclovir with the microneedling arrays over a period of 48 h, with a superior dermal drug concentration compared to the topical cream formulation [54].
3.1.7. Hydroxypropyl Methylcellulose (HPMC)
HPMC is a promising material for MN-based drug delivery due to its excellent BC, ease of formulation, and ability to form stable MNs that can efficiently penetrate the skin and release drugs for targeted delivery. First, HPMC has good mechanical strength and can form stable, long-lasting MNs that can penetrate the skin without breaking or deforming. Second, HPMC has a high swelling capacity, which can facilitate drug release and improve skin permeation. Third, HPMC can be easily dissolved in water, making it easy to prepare MN formulations. Finally, HPMC can be easily modified to tailor its properties, such as solubility, swelling behaviour, and mechanical strength, to suit the specific requirements of different drug delivery applications [55].
A recent study explored use of self-dissolving HPMC and HPMC-PVP MNs to improve the bioavailability of acyclovir, a commonly prescribed antiviral medication for the herpes virus. The research aimed to use a dissolvable microneedling system for the topical and systemic transport of acyclovir. A topical freeze-dried drug wafer was placed on MN-treated skin. It showed sufficient mechanical strength, and the wafer demonstrated an adequate porous nature required for faster hydration. The self-dissolving MN-assisted topical wafer gives about 10 times greater skin concentration than the ID99 with reduced lag time. The study showed that this delivery system could be effective against the herpes virus infection for topical as well as systemic action. It has the potential to reduce dosing frequency and improve drug bioavailability [56].
3.1.8. Polycigycidyl Methacrylate
Polyglycidyl methacrylate (PGMA) is a polymer that has been investigated for its potential in drug delivery applications. PGMA is known to have hydrophilic properties, which makes it useful for solubilizing and delivering hydrophobic drugs. Additionally, the epoxide functional groups available in PGMA offer polymer-drug conjugation, enabling a controlled drug release [57]. It can be chemically modified. It is biocompatible and has low toxicity, making it an attractive candidate for biomedical applications. PGMA-based drug delivery systems have the potential to enhance drug solubility, stability, and targeted delivery, leading to improved therapeutic efficacy and reduced side effects [58].
Porous MNs (PMNs) for minimally invasive TD drug delivery were reported by D. Terutsuki et al. The stratum corneum allows the passage of only low-molecular-weight drugs through the skin. While MNs have been used to enhance TD permeation, they are usually limited to small molecules. The authors investigated a truncated cone-shaped PMN made of PGMA for TD drug delivery. It was reported that a non-penetrable frustoconical PMN expanded the skin, reduced the TD resistance, and improved the penetration of larger molecules when combined with electro-osmotic flow (EOF) via charge-immobilized frustoconical PMN. This can provide a patient-acceptable method for TD drug delivery for molecules such as dextran (~10 kDa), but it was reported that the ovalbumin (45 kDa) molecule could not penetrate the skin with the developed PMN [28].
3.1.9. Polyvinyl Alcohol (PVA)
PVA is a promising material for use in drug delivery systems, particularly in the fabrication of MNs. It easily forms aqueous solutions. PVA MNs have been found to be superior to other dissolving polymers used for microneedling systems in terms of penetrating into skin layers [59]. PVA MNs have shown a potential for delivering a range of drugs, including vaccines, proteins, and small molecules. PVA MNs have been found to be effective in delivering drugs through the skin, with a high degree of reproducibility and consistency. Additionally, PVA MNs have been shown to be safe and well-tolerated by patients [60]. Recently, dissolving PVA MNs loaded with P. aeruginosa phages to combat antibiotic-resistant bacteria in biofilm-related skin infections were reported. The study found that the PVA MNs loaded with phages reduces P. aeruginosa PAO1 biofilms significantly more than the treatment with free phages. It is useful for the treatment of dermal infections. The research validates the use of a phage-loaded microneedling system for biofilm treatment. It may be used in the treatment of other resistant topical biofilm reduction, and it is especially helpful in wound healing [61].
3.1.10. Polystyrene-Block-Poly-(Acrylic Acid) (PSPAA)
PSPAA is obtained by hydrolysis of polystyrene-block-poly-(tert-butyl acrylate) polymer in solvent methylene chloride using catalyst trifluoracetic acid. PSPAA was used in the swellable tip while the core was made up of non-swellable polystyrene. Interestingly, it swells to about eight times its original volume. Further, swelling and deswelling do not affect the dimensions of MNs. The concentration of polymer can be varied to control the swellable tips’ height, and it forms highly porous structures [62]. Ferric oxide nanoparticles loaded in PSPAA-based MNs were reported by R.Z. Seeni for MN optical coherence tomography. It was revealed that polymer swells in 20 s in agarose gel, while, on fresh skin tissues, this takes about 30 to 35 s. The addition of ferric oxide nanoparticles does not have a significant effect on the mechanical strength and swelling behaviour of PSPAA polystyrene polymer, and enhances its imaging characteristics [63].
3.1.11. Polylactic Acid (PLA)
Polylactic acid is A biodegradable and biocompatible, FDA-approved polymer used for the preparation of MNs. PLA MNs coated with A drug could maintain mechanical strength in physiologically humid environments. Hence, coated PLA MNs are preferred for drug delivery over dissolvable MNs, although both can puncture the porcine skin [64]. It is reported that the anisotropic nature of PLA can lead to detachment from the substrate during the etching process due to weaker bonding forces. Localized heating with laser could solve the issue but caution is required to avoid hole formation and breakage induced by the laser. The optimization of PLA MNs is elaboratively discussed by the L. Wu et al. study. Controlling the temperature of the process is crucial for the 3D-printing process of PLA MNs. For the PLA filament, the glass transition and melting points were 60 °C and 170 °C, consequently. Above 220 °C, it becomes decomposed [65].
Polylactic acid was used for the development of uncoated MNs for the dermal delivery of Hepatitis B Surface Antigen (HbSAg). The antigen was dissolved in carboxy methyl cellulose and loaded on the polylactic acid MNs. It was revealed that these MNs produce immunogenicity comparable to that of an injectable preparation of the antigen, releasing about 74% of the antigen within the first 30 min. Further, the HbsAg-loaded polylactic acid MNs did not show any significant acute toxicity in the animal study [66].
3.1.12. Polymethyl Methacrylate (PMMA)
PMMA is a biocompatible, cheaper alternative with sufficient mechanical strength for the development of the polymeric MNs. It does not easily scratch the dermal layers and has higher light transmittance. It was explored for glucose measurement directly from the interstitial fluids within a few minutes using Raman spectroscopy-based biosensors. It maintains structural stability, is found to be safe, and does not swell during dermal application [67]. A recent comparative study of different polymeric needles revealed that PMMA MNs are superior in the decline of tissue fibrosis and the expression of proinflammatory factors viz. smooth muscle actin (α-SMA)+myobroblasts, Ki-67 protein, and transforming growth factor-β1 mRNA, when compared with polycarbonates, polylactic-co-glycolic acid, polypropylene, polyurethane, liquid crystal polymers, and stainless steel [68]. Passivation techniques improve the performance of the MN-based biosensors. PMMA coating is reported to improve the manufacturing process and performance of the painless biosensor MNs [69].
Gemcitabine-coated PMMA MN arrays were reported by Z. Kanaki et al. The drug was coated by a laser-induced forward transfer process. Gemcitabine-coated PMMA MN arrays offer an advantage in metronomic chemotherapy, allowing a sustained drug release in animal studies, although exploration in clinical studies is further required [70].
3.1.13. Polystyrene
Polystyrene is biocompatible material offering ease of MN fabrication. Painless MN can be developed using polystyrene and it does not generate biohazardous waste. It is a non-swellable material with sufficient mechanical strength required for MN preparation [51]. It was demonstrated that polystyrene MNs can withstand up to 4.7 N/needle, exhibiting its usefulness for clinical purposes. It smoothly penetrates mice skin without deformation. It was demonstrated that the addition of surfactants such as Tween 20 is useful during the loading of polystyrene microspheres in the MNs [69]. The optimal concentration of polystyrene is 50% for MN preparation, below which it causes brittleness in the needle structure. A carbon-polystyrene composite system was developed for the determination of TD pH [71].
3.1.14. Polycaprolactone
Polycaprolactone is a biodegradable, hydrophobic material with a lower melting point. It allows solvent-free MN synthesis at a temperature of around 65 °C by a thermal melting process. Capsaicin-loaded polycaprolactone MNs could provide drug release up to 15 days [72]. A pH-triggered electrolyte-based coating on the polycaprolactone microneedling patches was reported in the literature. It consists of dimethylmaleicanhydride-modified polylysine and a gene-containing layer of p53 expression plasmid/polyethyleneimine. In vivo studies of mice show polyelectrolyte-coated polycaprolactone MN patches exhibit tumor inhibition efficacy of 90.1% compared to the 46.4% efficacy with a nonpolyelectrolyte coating [73].
4. Fabrication Techniques of Microneedles
In the past several years, researchers have employed multiple techniques to create a diverse range of MNs. When engineering MNs, the functionalities are addressed first, which include the type of drug, its concentration, desirable BD, as well as the targets and attributes of the material utilized for MNs. The fundamental purpose in the production of MNs is to achieve needle geometrical consistency and predictability at a micron-scale resolution to permit efficient needle piercing in the skin. The literature review reveals that there are various methods which are used for the fabrication of MNs, which are discussed below. The number of fabrication techniques are evolving with reference to the payload and type of application. Here, the basic ten methods of fabrication are discussed; for the rest of the methods, readers can refer to recently published review articles [14].
The fabrication of MNs is influenced by various factors, including the material properties, the desired needle geometry, the fabrication method, and the intended application. The key practical parameters and practical considerations are given in Table 1.

Table 1.
Factors affecting fabrication of MNs.
4.1. Micromolding
It is a fabrication technique that involves creating structures or parts at a microscopic scale using a molded MN of submicron length, and it has applications in drug delivery, vaccination, and other medical purposes. The micromolding technique for MNs involves creating a mold with the desired needle shape and size. The mold is typically made using photolithography, a process that uses light to transfer a pattern onto a photosensitive material. The mold is then filled with a polymer material, such as polycarbonate or polydimethylsiloxane (PDMS), which is then cured to form the MNs. Once the MNs have been formed, they can be loaded with a drug or vaccine and then applied to the skin using a patch or applicator device. They are less painful and invasive, and they can be designed to dissolve or release their payload over time [82]. Various fabrication methods of MNs are comparatively presented in Table 2.

Table 2.
Comparison of microneedle fabrication methods.
4.2. Micromilling
Micro-milling is a precision fabrication technique used to create small parts with high accuracy and precision. It is a type of milling process that uses micro cutting tools to remove material from a workpiece. It is commonly used in the fabrication of microelectromechanical systems (MEMS), microfluidic devices, and other small-scale components for use in medical devices and electronics. This process typically involves a workpiece, which can be made from materials such as polymers, metal, or ceramic, and a cutting tool, which is typically a small end mill with the required diameter. The workpiece is typically held in place by a fixture or vise, and the cutting tool is moved along the surface of the workpiece in a precise pattern to remove material [83]. Micro-milling machines use computer numerical control (CNC) to control the movement of the cutting tool. This allows for precise control of the shape and size of the part being created.
Micro-milling machines are designed to be highly precise, with tight tolerances and positioning accuracy. They are typically equipped with advanced software and control systems to ensure that the cutting tool follows the programmed path accurately. It has several advantages over other manufacturing techniques, including high precision, excellent surface finish, the ability to create complex geometries, and flexibility in the selection of materials. It can be used with a wide range of materials, including polymers, metals, and ceramics [84].
4.3. Atomized Spraying to Fill Molds
Atomized spraying is a process of filling molds with liquid material by atomizing the liquid into tiny droplets. The mist is directed into the mold cavity, where it cools and solidifies to form the final product. Atomized spraying has several advantages over other methods of filling molds, including the ability to create more uniform and consistent products, improved surface finish, and reduced waste of material. The size of the droplets can be controlled by adjusting the pressure and flow rate of the liquid. It can also be used to create complex shapes and structures that would be difficult or impossible to achieve with other techniques. It is a powerful fabrication technique that can be used to create high-quality, complex products with a high degree of accuracy and precision. It is widely used in a variety of industries and can help to improve efficiency, reduce waste, and lower manufacturing costs [85].
4.4. 3D and 4D Printing
3D printing is a manufacturing technique that involves creating three-dimensional objects by layering material on top of each other. In recent years, 3D-printing technology has advanced significantly, allowing for the production of increasingly complex and precise structures. In addition, 3D printing has been used to create MNs, which are small needles that can be used for drug delivery, vaccination, and other medical applications; 3D printing allows for the creation of MNs with precise geometries, which can be customized for specific applications and patients. The 3D-printing process for MNs typically involves designing the MNs using specialized software, and then printing them using a 3D printer. The printer deposits layers of material, such as biocompatible polymers or metals, to create the MN structure [86].
The advantage of 3D printing for MNs is the ability to create complex structures that would be difficult or impossible to achieve with other manufacturing techniques; 3D printing can be used to create MNs with multiple branches or with varying needle lengths. Another advantage of 3D printing for MNs is the ability to create MNs on demand. This means that healthcare providers can quickly and easily produce MNs as needed, rather than relying on pre-manufactured needles. The development of MNs customized for specific applications and patients is possible. It offers a high degree of customization and precision, and it has the potential to revolutionize drug delivery and other medical applications [87].
A new emerging technique for the advanced manufacturing of MN is 4D printing. Four-dimensional printing or additive manufacturing has a broad use across a number of domains, including the fabrication of complex merchandise out of an assortment of materials and processes with the capacity to modify the resilience of materials throughout the course of manufacturing. The building blocks employed in 4D printing are additionally referred to as responsive materials because they acquire diverse dimensions, traits, and amenities over time in accordance with stressors such as heat, water, light, and pH [88,89]. Table 3 illustrates various 3D- and 4D-printing additive fabrication methods for MNs.

Table 3.
Various 3D- and 4D-printing additive fabrication methods for microneedles.
4.5. Laser Ablation
Laser ablation is an MN fabrication technique that involves using a laser to selectively ablate material from a substrate, creating MN structures. The process involves focusing a laser beam onto the surface of a substrate, typically a polymer or metal film, and moving the beam to create the desired MN pattern. The laser ablation process allows for the high precision and customization of MN geometry, and can be used to create arrays of MNs with varying shapes and sizes. The technique is also capable of creating MNs with highly sharp tips, which can aid in skin penetration. However, laser ablation may be limited by the type of material used; for example, CO2 lasers are typically used to ablate polymers, while excimer lasers are typically used to ablate metals. Other factors which can limit its use are the cost and complexity of the laser system required [96].
4.6. Photolithography
It is an MN fabrication technique that involves using a photoresist material to create an MN pattern on a substrate. The process involves coating a substrate, typically a silicon wafer or glass slide, with a layer of photoresist material. The photoresist is then exposed to UV light through a mask that contains the desired MN pattern. The exposed areas of the photoresist material are then chemically removed, leaving behind an MN pattern on the substrate. The substrate can then be coated with a material such as metal or polymer to create the final MNs. Photolithography allows for the high precision and reproducibility of MN structures, but can be complex and require specialized equipment. Soft lithography, nanoimprinting, and nano-molding are advanced variations of the techniques developed for better quality control of MNs [97].
4.7. Printing Techniques
Techniques such as inkjet printing and screen printing can be used for MN fabrication. In inkjet printing, MN patterns are printed onto a substrate using inkjet printing technology, typically with a polymer or hydrogel ink. The ink is deposited onto the substrate in droplets, which then solidify to form the MN structures. Inkjet printing allows for the high resolution and customization of MN patterns, and can be used to print MNs with multiple layers or materials. Screen printing involves using a mesh screen to transfer an MN pattern onto a substrate. The MN pattern is transferred onto the screen, which is then pressed onto the substrate to transfer the pattern. Screen printing can be used to create MNs with a wide range of sizes and shapes, and can be used to print multiple MN layers. Both inkjet printing and screen printing can be used for high-throughput MN fabrication, and can be compatible with a variety of materials [98].
4.8. Etching
Etching has two basic types: dry and wet etching. The first one uses inert or reactive gases, while the second uses a chemical etchant for the process. This method involves using a combination of chemical and mechanical processes to create needles with a diameter of less than 100 µm. The etching process begins by creating a template onto which the MNs are designed. This can be carried out through a variety of methods, including photolithography, wax or plastic molding, and laser cutting. Once the template is in place, a chemical etchant, such as hydrofluoric acid or potassium hydroxide, is used to cut the MNs into the template. This process is typically performed using a spray-etching technique, which is a highly precise and controllable method [13].
The etching process produces needles of varying lengths and shapes depending on the desired application. Commonly used shapes include hollow or solid cylinders, triangular tips, and conical tips. The etchant also treats the surface of the needles, creating a surface that is suitable for various applications [99]. Once the etching process is complete, the needles are typically cleaned and sterilized before being packaged and shipped to the customer. The needles can be used for a variety of applications, such as drug delivery, skin testing, and TD drug delivery. The etching method is a cost-effective and efficient way to create MNs. This process can be used to create needles of various shapes and sizes, and the needles can be used for a variety of applications [100].
4.9. Electrospinning
Electrospinning is an emerging method for the fabrication of MNs, which involves using electricity to spin polymer solutions into nanofibers. This method is used to create thin, uniform, and continuous nanofibers which have the potential to be used for various medical applications. The process of electrospinning begins with the preparation of a polymer solution, which is then placed in a syringe and connected to a high voltage source. As the voltage is increased, a thin stream of the solution is drawn from the syringe and extruded into an electrical field, which causes the solution to form nanofibers. These nanofibers are then collected on a grounded collector, which is usually made of metal, glass, or polymeric materials. The diameter of the nanofibers produced through electrospinning is usually between 100 nm to 10 μm, and the length of the nanofibers can range from a few millimeters to several meters [101]. The nanofibers created through electrospinning can be of single or multiple polymers, allowing for a wide range of properties to be designed. One of the major advantages of electrospinning is that it can be used to fabricate MNs with a wide range of sizes and shapes. The size and shape of the nanofibers can be tailored to meet the desired application, such as drug delivery or tissue engineering. Furthermore, the process is relatively inexpensive and easy to control, which allows for a wide range of applications to be developed. The electrospun nanofibers can be used to create MNs of various shapes and sizes, including MNs with hollow cores which can be filled with drugs or other materials. The MNs created through electrospinning can also be coated with various materials, such as biocompatible polymers, to further enhance their properties. The process is relatively inexpensive and easy to control, and the nanofibers produced can be tailored to meet the desired application, making it a versatile technique for creating MNs [102].
4.10. Co-Extrusion
The co-extrusion process begins with the selection of two or more materials, each with its own distinct properties. This technique is used to create MNs with a wide range of properties, including shape, size, and chemical composition. These materials are then combined in a single extruder, where they are simultaneously extruded into a single product. The co-extruded product is then cut into individual MNs of varying shapes and sizes. The advantages of this method are improved biocompatibility, mechanical strength, and drug release profiles. In addition, the integration of multiple materials in the same MN can help to enhance the drug delivery process. It can form hollow MNs, which are used for the delivery of drugs and other materials, as well as solid MNs, which can be used for the delivery of DNA, proteins, and other therapeutics. The co-extrusion process is also highly efficient, as it can produce large quantities of MNs in a relatively short amount of time. This makes it an ideal choice for the mass production of MNs. Co-extrusion is a versatile and efficient method of fabricating MNs, and is used to create MNs with a wide range of properties and shapes. This technique is an excellent choice for the mass production of MNs, and is becoming increasingly popular for the delivery of drugs and other materials [103].
5. Biomedical Applications of Polymeric Microneedles
MNs are safe and efficient alternatives to hypodermic needles with less invasiveness and better drug delivery. MNs are a promising and emerging paradigm in the area of biomedical science. The availability of biocompatible and biodegradable polymers provides a special advantage for MN fabrication and its coating. There are various therapeutic, diagnostic, and drug delivery applications of polymer-based MNs [104].
5.1. Therapeutic Applications
The polymeric MN has emerged as a promising drug delivery platform for achieving the simplified and patient-friendly delivery of medicines, immunization, and some biological uses. The utilization of polymers in MNs underlines extraordinary applications in TD medication conveyance, the examination of skin tissues, the sampling of body fluids, and biotherapeutics complexation, biosensing, etc. Drug delivery through polymeric MN can be biocompatible, biodegradable, non-immunogenic, and having great mechanical strength. Subsequently, this gives an extraordinary application in protein drug delivery through a TD path. Drug delivery the through stratum corneum is an effortless and insignificantly obtrusive way through polymeric MNs [28]. Polymeric MNs used for therapies of various diseases are illustrated below.
5.1.1. Rheumatoid Arthritis
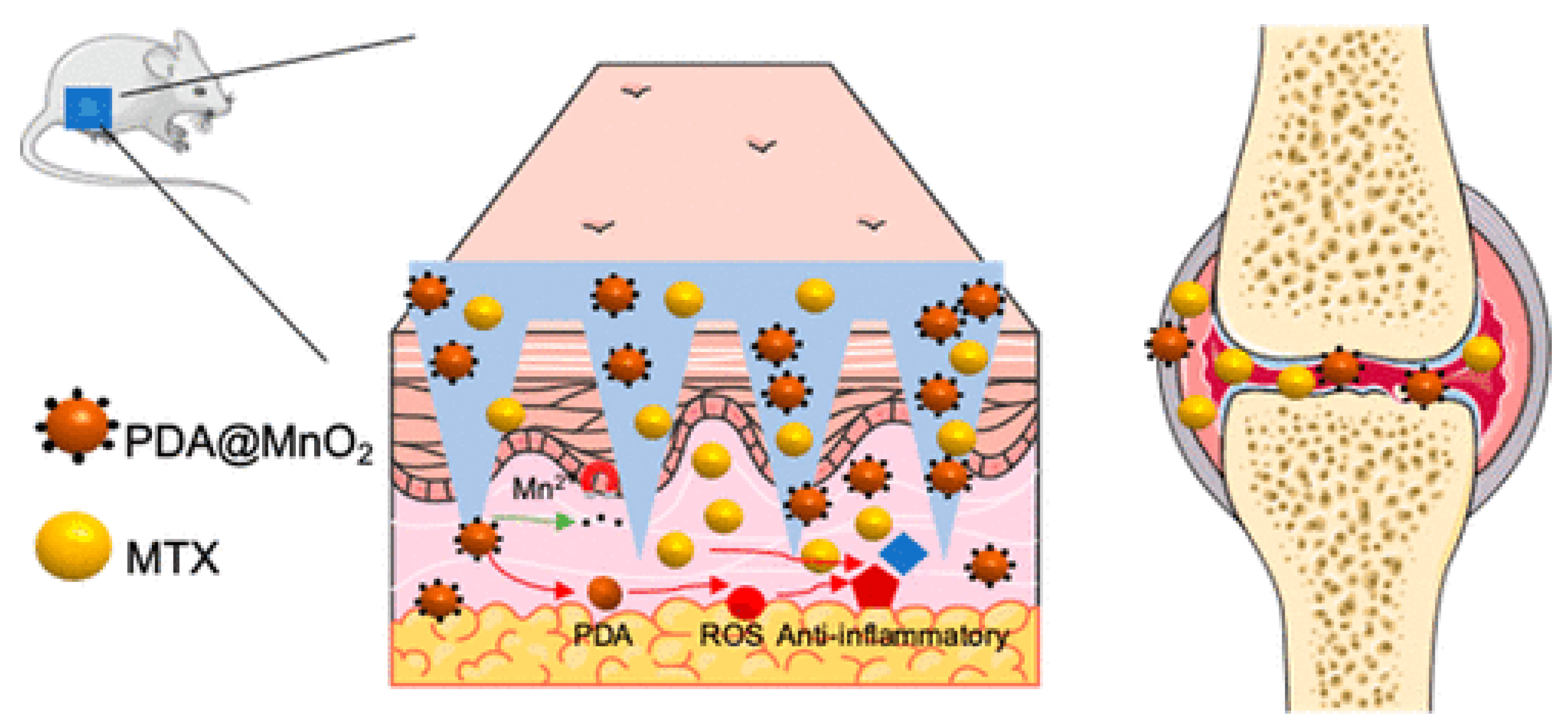
Localized drug delivery using MNs provides therapeutic efficiency in the treatment of rheumatoid arthritis (RA). Biodegradable polymeric MNs are highly preferred for this purpose. Wu et al. reported the fabrication of programmable MNs for the treatment of RA (Figure 7). The MNs were fabricated using polyvinylpyrrolidone (PVP). The polydopamine/manganese dioxide (PDA@MnO2) and methotrexate were incorporated for antioxidants and chemotherapy, respectively. The in vivo degradation of MNs in a synovial microenvironment released the incorporated content. The prepared MNs were found to be useful in removing reactive oxygenated species (ROS) and reducing inflammation in RA [105].
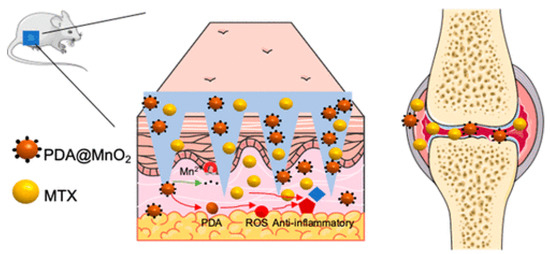
Figure 7.
Programmable polymeric MN for RA therapy. Adapted with permission from Ref. [105]. Copyright 2021 American Chemical Society.
Yao et al. demonstrated the fabrication of dissolving MNs for neurotoxin delivery for the treatment of RA. MNs were fabricated using polyvinyl pyrrolidone (PVP) and chondritin sulfate (CS). The prepared MNs showed less toxicity and BC when tested on chondrocyte cells. An ex vivo skin penetration study showed the efficiency in penetration depth of MNs and associated delivery of neurotoxin. An in vivo animal study on rats demonstrated the efficacy of fabricated dissolving MNs in RA therapy, which was confirmed with decreased toe swelling and suppressed levels of IL-1β and TNF-α [106].
Methotrexate is preferably used in the treatment of RA, but oral and subcutaneous drug delivery is limiting, with poor bioavailability and painful administration, respectively. MN-based drug delivery can overcome the aforementioned problems. Tekko et al. developed hydrogel-forming MN arrays loaded with a methotrexate reservoir. The MN array was fabricated using PVP and polyvinyl alcohol (PVA), and citric acid was used as crosslinking agent. The ex vivo study showed an efficient steady state flux of 506.8 ± 136.9 µg·cm2/h. The in vivo study in rats showed a plasma concentration of 7.6 ± 2.0 nM after 1 h and peak plasma concentration of 35.1 ± 5.1 nM after 24 h. The prepared polymeric-MN-based drug delivery system is promising for the treatment of RA and juvenile idiopathic arthritis (JIA) [107].
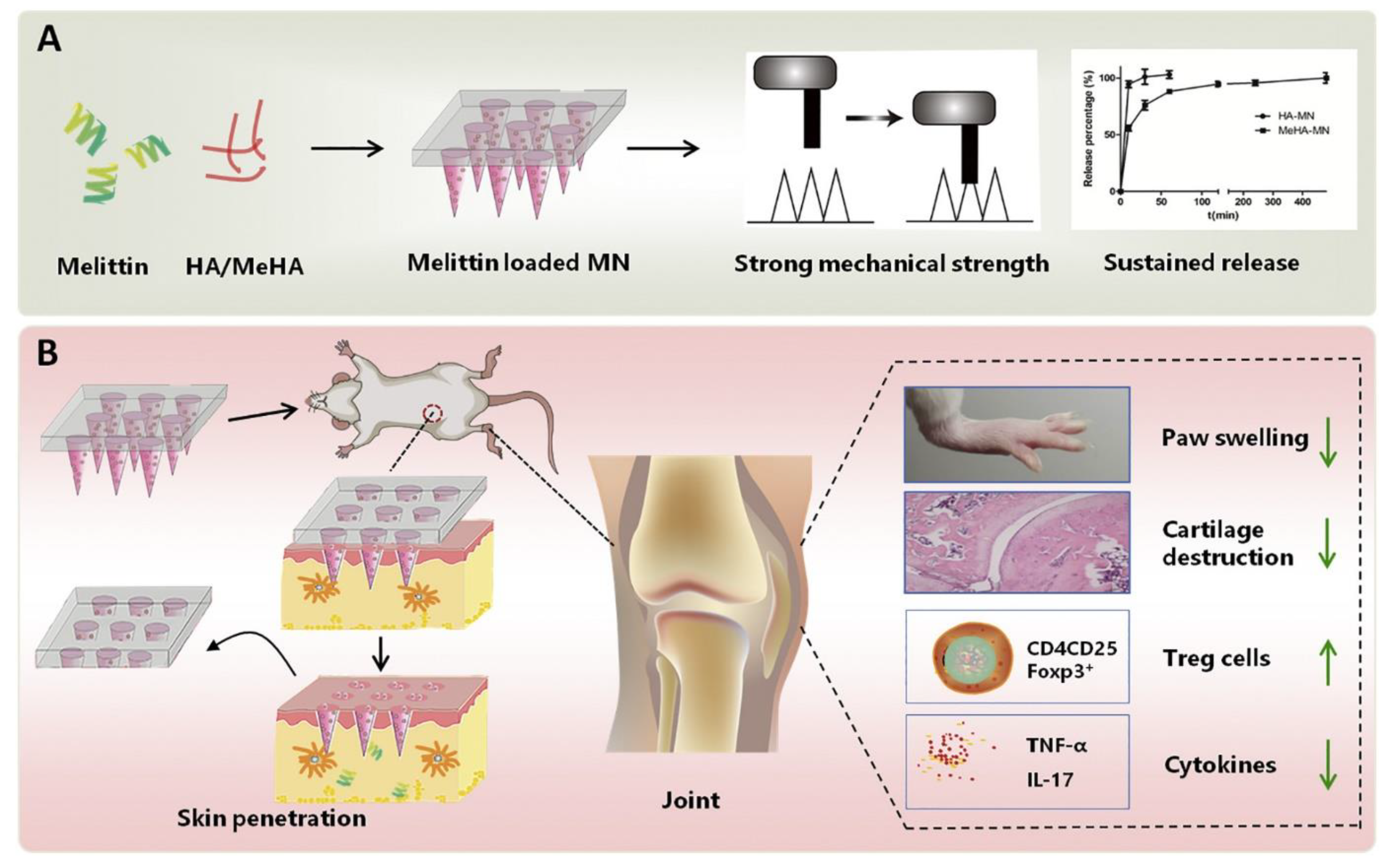
Du et al. fabricated the melittin-loaded hyaluronic-acid-based MNs for RA therapy (Figure 8). The crosslinking of MN by UV radiation resulted in a sustained release of melittin. The prepared MNs showed significant inhibition of RA, observed under an in vivo study in rodents and murines. An analysis of various parameters such as arthritis score, paw swelling, and histopathology revealed the significant inhibition of RA by the fabricated polymeric MNs.
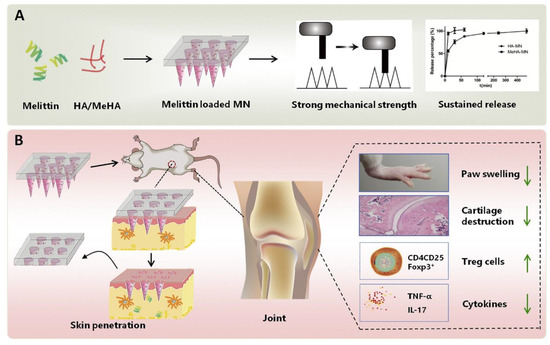
Figure 8.
(A,B) Fabrication and evaluation of melittin-loaded hyaluronic-acid-based MNs for RA therapy. Adapted with permission from Ref. [108]. Copyright 2021 Elsevier.
The suppression in the levels of inflammatory mediators such as TNF-α, IL-17, and cytokines also demonstrated the efficiency of prepared MNs in RA therapy [108].
The organic solvent-based drug delivery system for lipophilic drugs may pose safety concerns. To overcome this limitation, Dangol and coworkers developed innovative capsicin-polymeric-system-based MNs for the treatment of RA. The in vivo study in a mouse model revealed the efficiency of fabricated MNs in RA therapy [109].
5.1.2. Skin Diseases
TD drug delivery through MNs facilitates the treatment of skin diseases with enhanced efficiency. The skin diseases such as psoriasis, melasma, and melanoma can be efficiently treated using MNs. Polymers provide the scope in the fabrication of dissolving and biodegradable microneedles with the advantages of efficient drug delivery.
Melasma
Melasma is the symmetric hyper-pigmentation condition which occurs due to various reasons such as UV exposure or hormonal imbalance. A histopathology of melasma demonstrates the enlarged melanocytes and increased dermal pigmentation. The traditional therapy for melasma includes the use of hydroquinone or corticosteroid creams but this has limits, with poor TD permeation. Microneedling has emerged as one of the useful techniques for melasma therapy due to better TD penetration and efficient drug delivery [110]. Machekposhti et al. reported the fabrication of polymeric MNs for melasma therapy. Polymeric MNs were fabricated using PVP and methacrylic acid with the loading of tranexamic acid. Fabricated MNs were found to be biocompatible and nontoxic in acute toxicity testing. The complete release of MNs in skin was observed within 7 h. The efficiency of drug delivery was observed by a simulated determination of the diffusion coefficient in interstitial fluid. The fabricated polymeric MNs were found to be the promising for the treatment of melasma by melanin inhibition [111].
He and coworkers also demonstrated the application of polymer-based MNs in the therapy of melasma (hyperpigmented skin). Researchers fabricated a polymer MN roller (PMR) to deliver the hydroquinone cream (HQC). The direct application of HQC was compared with the PMR-pretreated application of HQC; the results of an in vivo study in a UV-mediated melasma-induced rat model revealed that the PMR-pretreated HQC has better therapeutic efficiency in the treatment of melasma. Histopathological observations and the level of malondialdehyde in the liver and skin demonstrate the therapeutic efficiency of fabricated MNs in the treatment of melasma [112].
Psoriasis
Psoriasis is an immunity and gene-mediated skin disease. The growth of skin cells in psoriasis is 10 times more than normal. The symptoms of psoriasis include lesions, burning, itching, and swelling. Drug delivery in psoriasis is also limited, with deficient penetration and permeability. Many research studies reported successful drug delivery in psoriasis using polymer-based MNs [113,114,115]. Tekko et al. demonstrated the fabrication of polymeric MNs for the delivery of nanocrystals in psoriasis. Nanocrystals of methotrexate sodium were prepared and incorporated in the shaft of polymeric MNs made up of PVP and PVA. An in vivo study in rats revealed that the retention of the drug in skin was comparatively improved when the drug is administered through polymeric MNs. The drug retained after 24 h of administration through polymeric MN was 322-fold (312.70 ± 161.95 µg/g) that of oral administration (0.942 ± 0.59 µg/g). The results of the study showed that the prepared polymeric-MN-based delivery of methotrexate nanocrystals could be a promising approach in psoriasis therapy [116].
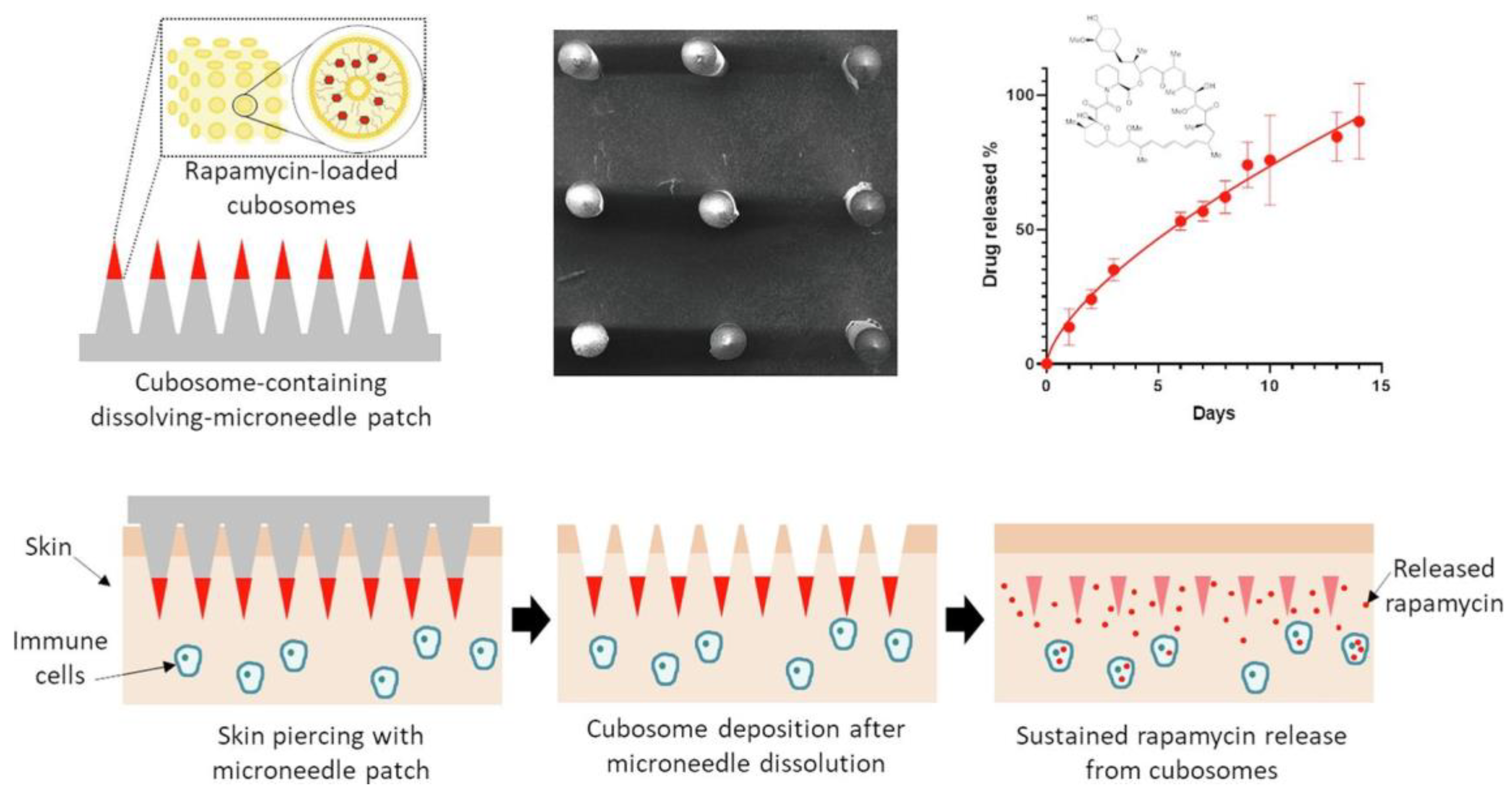
Ramalheiro et al. fabricated polymer-based dissolving MNs to deliver cubosomes such as liquid crystalline nanoparticles of rapamycin for the treatment of psoriasis (Figure 9). The polymeric MNs were fabricated from PVP and PVA, in which rapamycin nanoparticles were loaded. The efficient piercing and deposition of nanoparticles in skin demonstrated the successful application of the fabricated polymeric MN patch. The in vitro analysis showed the efficient antiproliferation of natural killer cells. The study demonstrated the application of the polymeric MN system in psoriasis therapy [117].
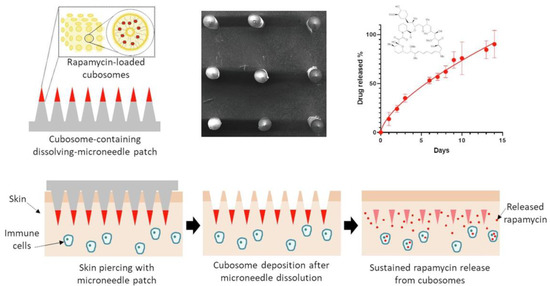
Figure 9.
Fabrication and evaluation of polymeric MNs for delivery of rapamycin nanoparticles for the treatment of psoriasis. Adapted with permission from Ref. [117]. Copyright 2021 Elsevier.
5.1.3. Cancer
Cancer is one of the leading causes of death across the globe. Targeted and localized drug delivery with better biodistribution is the intent behind the delivery of anticancer agents. MN-based drug delivery in cancer is becoming one of the prime areas of research. Melanoma is one of the common types of skin cancer. MNs can provide the scope for the efficient delivery of anticancer agents in melanoma therapy.
Demartis et al. demonstrated the fabrication of dissolving MNs for the delivery of Rose-Bengal-loaded transferosomes for melanoma therapy. The polymeric (dissolving) MNs were fabricated from PVA and PlasdoneTM. The dermatokinetic study demonstrated that the fabricated tranferosome-loaded MN system showed efficient drug delivery in melanoma management [118].
Polymer-coated MNs also demonstrated advanced medical applications. Matadh et al. reported the successful delivery of 5-Fluorouracil (5-FU) through a polymeric MN system. Polymeric MNs were fabricated by the mold casting method using PVP K-30. The coating of the MNs was carried out using Eudragit E-100. The polymer coating prolonged the drug release by 3 h. Then, 5-FU was loaded in the core of the MNs and further coated with polymer to prolong the drug release. This polymeric-MN-based controlled delivery of 5-FU in the sub-stratum corneum is useful in the treatment of melanoma [119].
Along with melanoma, MN-based drug delivery was found useful in the treatment of breast cancer. Alafnan et al. developed the polymeric-MN-based drug delivery system of Gemcetabine for breast cancer therapy. Polymeric MNs were fabricated using polyethylene glycol diacrylate (PEGDA) by 3D technology. The permeation study using the Franz diffusion cell demonstrated an efficient drug release. The prepared polymeric MN system could be a promising approach in the chemotherapy of breast cancer [120].
Bhatnagar and coworkers also developed polymeric MNs for THE co-delivery of doxorubicin and docetaxel for breast cancer therapy (Figure 10). Polymeric MNs were fabricated using PVP and PVA. Both the drugs were loaded very well in the MN patch. An in vitro permeation study on the excised murin skin showed the dissolution within 1 h. An in vivo study in a 4T1 breast-cancer-cell-xenografted athymic Balb/c mouse model demonstrated that the MN based drug delivery is efficient, safe, and non-toxic compared to intra-tumoral injection. DNA fragmentation and tumour volume analysis indicated the efficient delivery of anticancer drugs through the polymeric MN patch to counteract the breast cancer [121].
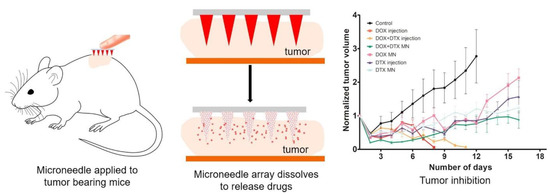
Figure 10.
Polymeric MNs for co-delivery of doxorubicin and docetaxel for breast cancer therapy Adapted with permission from Ref. [121]. Copyright 2019 Elsevier.
Fabricated MN-based patches of Dox@MicroN showed efficient cytotoxic effects against cervical cancer cells [122].
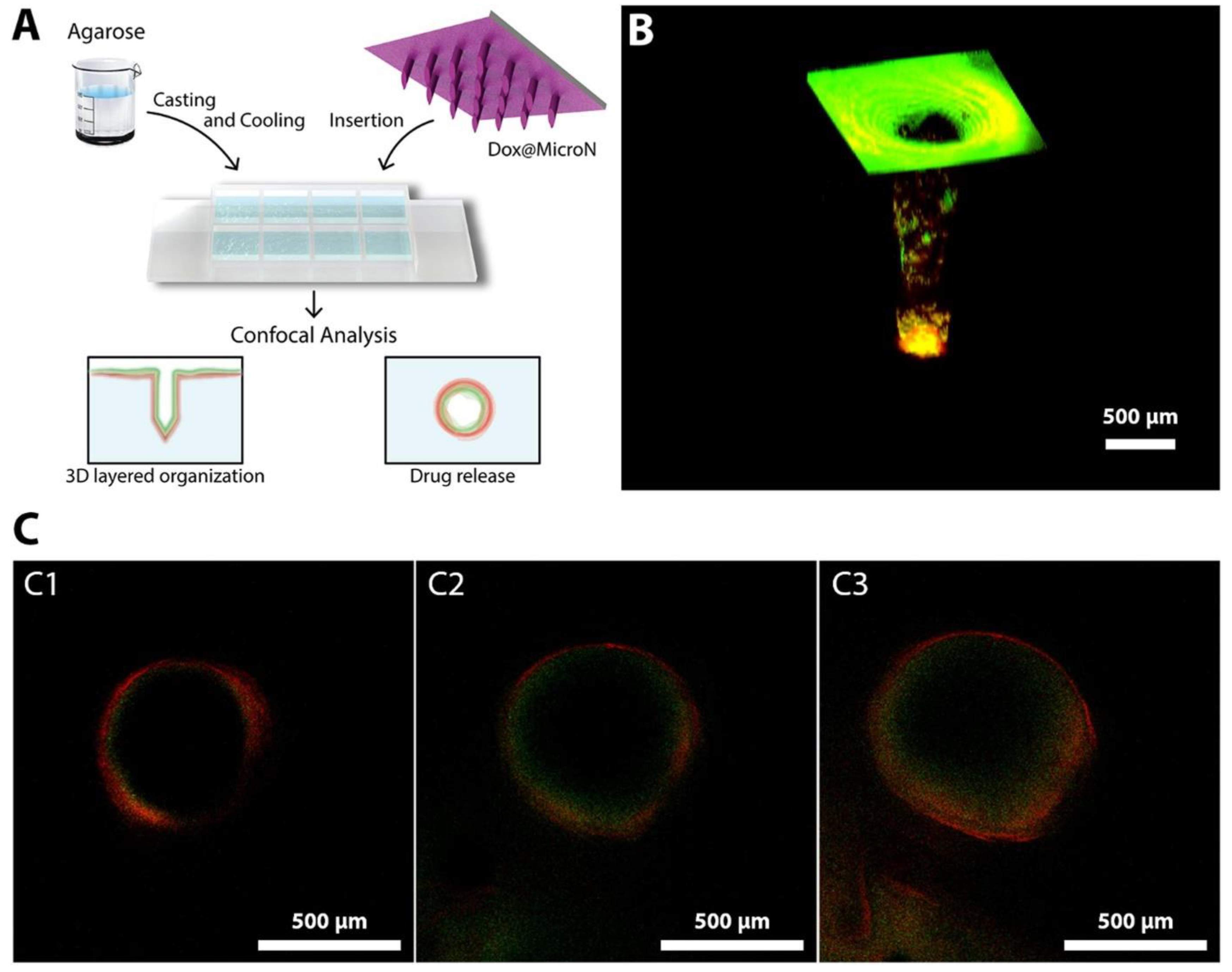
Moreira et al. demonstrated the study of polymer-coated polymeric MNs for photothermal therapy and chemotherapy in cancer (Figure 11). MNs were fabricated from PVP and coated with PVA and chitosan by micromoulding and electrospraying. Polymeric MN showed the efficient delivery of doxorubicin and AuMSS nanorods (Dox@MicroN). Chitosan showed the pH-responsive release of Dox@MicroN which further demonstrated the IR-responsive photothermal effect. Tumor-mimicking agarose gel was used to study the efficient drug delivery through MNs.
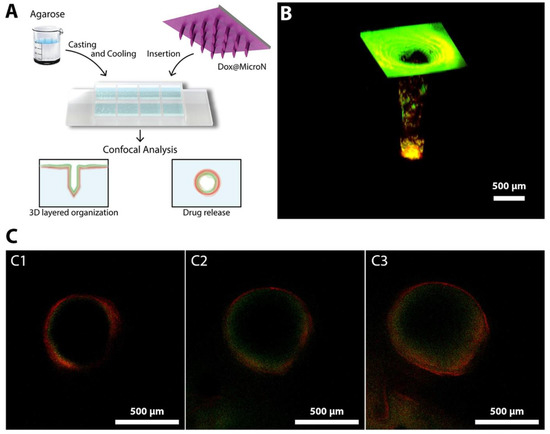
Figure 11.
Efficient drug delivery through MNs using tumor-mimicking agarose gel: (A) Dox@MicroN penetration through agarose gel; (B) 3D Dox@MicroN MNs; and (C) release from Dox@MicroN after 10, 20, and 30 min (C1, C2, and C3, respectively). Adapted with permission from Ref. [122]. Copyright 2020 Elsevier.
5.1.4. Diabetes and Obesity
The delivery of anti-obesity and antidiabetic agents through MNs is an efficient and promising approach. Polymeric MNs provide the advantage of BC and BD so drugs can be delivered through dissolving MNs.
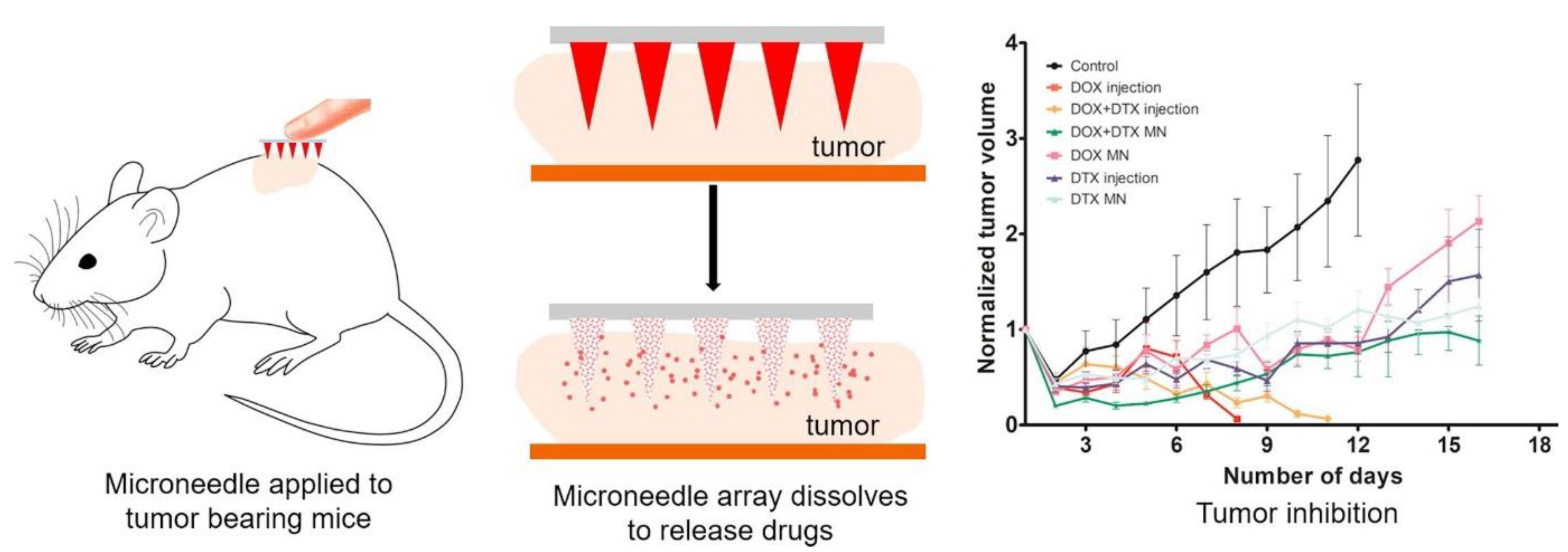
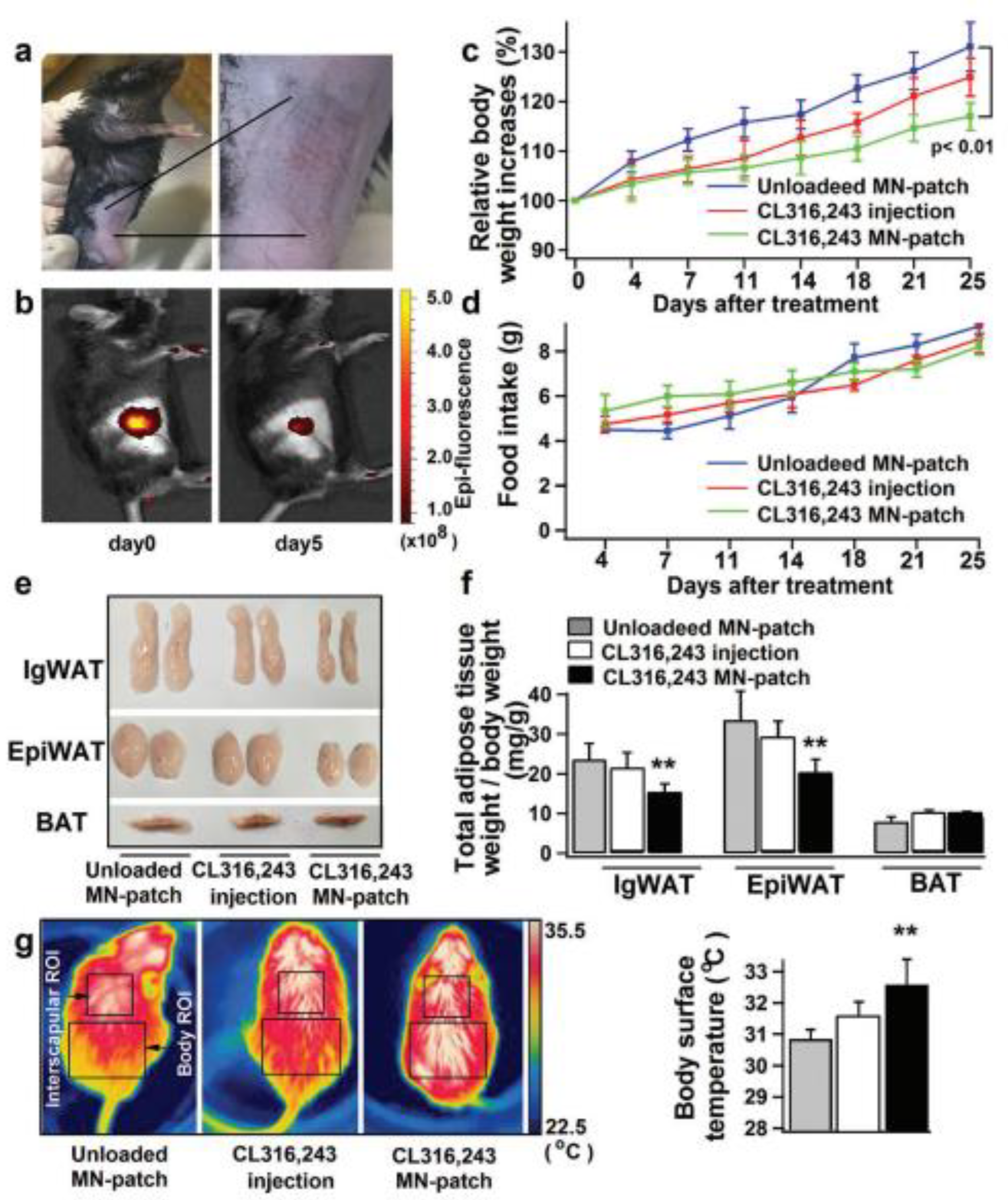
Obesity is the result of an increase in the level of white adipose tissue. The conversion of white adipose tissue to brown fat decreases obesity. There are challenges in treating obesity with the conventional delivery of anti-obesity drugs. To overcome this limitation, Than et al. fabricated TD patches with detachable polymeric MNs. MNs were fabricated using poly(dimethylsiloxane) (PDMS) micromolds. PLGA and HA are the polymers used for the fabrication of polymeric MNs. MNs were loaded with anti-obesity agents such CL316243 and Cyanine5. These polymeric dissolving MNs were analyzed for in vivo TD drug delivery using an obesity-induced mouse model. The polymeric-MN-based system showed the efficient delivery of an anti-obesity agent, which carries out the conversion of white fat to brown fat [123] (Figure 12).
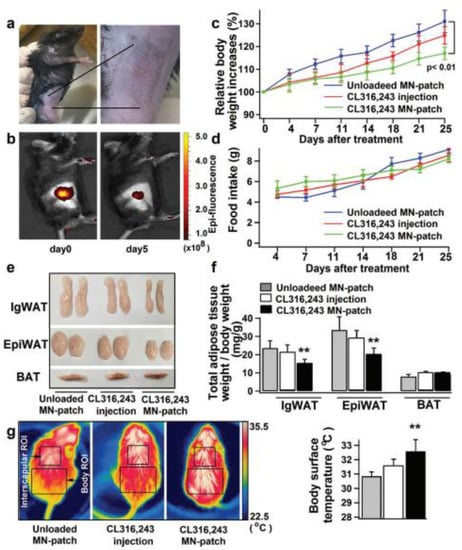
Figure 12.
(a–g) Comparative effect of anti-obesity agents (CL316,243 and Cyanine5) in injection and MN systems and associated conversion of white adipose tissue and brown adipose tissue. Student’s t-test: ** p < 0.01 versus control. Adapted with permission from Ref. [123]. Copyright 2017 John Wiley and Sons.
Rabiei et al. demonstrated the fabrication of polymeric MNs consist of PVP and PVA for the treatment of type 2 diabetes and obesity. The polymeric MNs were loaded with PLGA-based Liraglutide nanoparticles. The formulated polymeric-MN-based drug delivery system showed better skin penetration of Liraglutide and it was confirmed through a study performed using rat skin. The fabricated MN system was found to be effective in the treatment of type 2 diabetes and obesity and it eliminates the need of conventional hypodermic-needle-based injection of Liraglutide [124].
Xie and co-workers also demonstrated the fabrication of polymeric MNs for the delivery of an anti-obesity agent. The MNs were fabricated from PLA and PLGA using PDMS molds. The prepared polymeric MNs were loaded with CL316243 (anti-obesity agent). MNs were found to be efficiently delivering the anti-obesity drug with minimum invasion and painless skin penetration. The in vivo study in obese mice (induced with fatty diet) revealed the efficient treatment of obesity [125].
Dangol et al. reported the fabrication of a caffeine-loaded polymeric MN patch for the treatment of obesity. Caffeine has promising anti-obesity potential and can be efficiently used to treat obesity through MN-based drug delivery. MNs were fabricated using HA and loaded with caffeine. A pharmacodynamic study on obese mice (induced with fatty diet) revealed the significant anti-obesity activity of the prepared polymeric MN patch. Improved lipolysis and decreased level of triglycerides and LDL-C showed the promising application of the prepared MN patch in the treatment of obesity [126].
5.2. Drug Delivery
Efficient drug delivery with minimum invasion and minimum pain was the prime motive behind the development of MN technology. Polymeric MNs have the scope for the successful delivery of various types of drugs due to the availability of multiple polymers for fabrication. TD and intraocular drug delivery are prominent and preferred routes for the execution of MN-mediated drug delivery.
5.2.1. Transdermal
TD delivery of various drugs through MN technology was found to be useful in the treatment of various skin diseases, RA, melanoma, etc. MNs provide better TD penetration and better in vivo availability of a drug, resulting in improved therapeutics. Multiple researchers reported the application of polymeric MNs in TD drug delivery.
Chen et al. fabricated dissolvable polymeric MNs reinforced with graphene oxide (GO) for efficient TD drug delivery. Polymeric MNs were fabricated using PVP, HA, and CMC, and further reinforced with GO. The fabricated MNs showed better mechanical strength due to the incorporation of GO. The depth of insertion was found to be improved by six-fold due to GO. An in vitro skin penetration study demonstrated the efficiency of polymeric MNs in drug (GO) delivery in response to NIR light for antibacterial and antifungal activity [127].
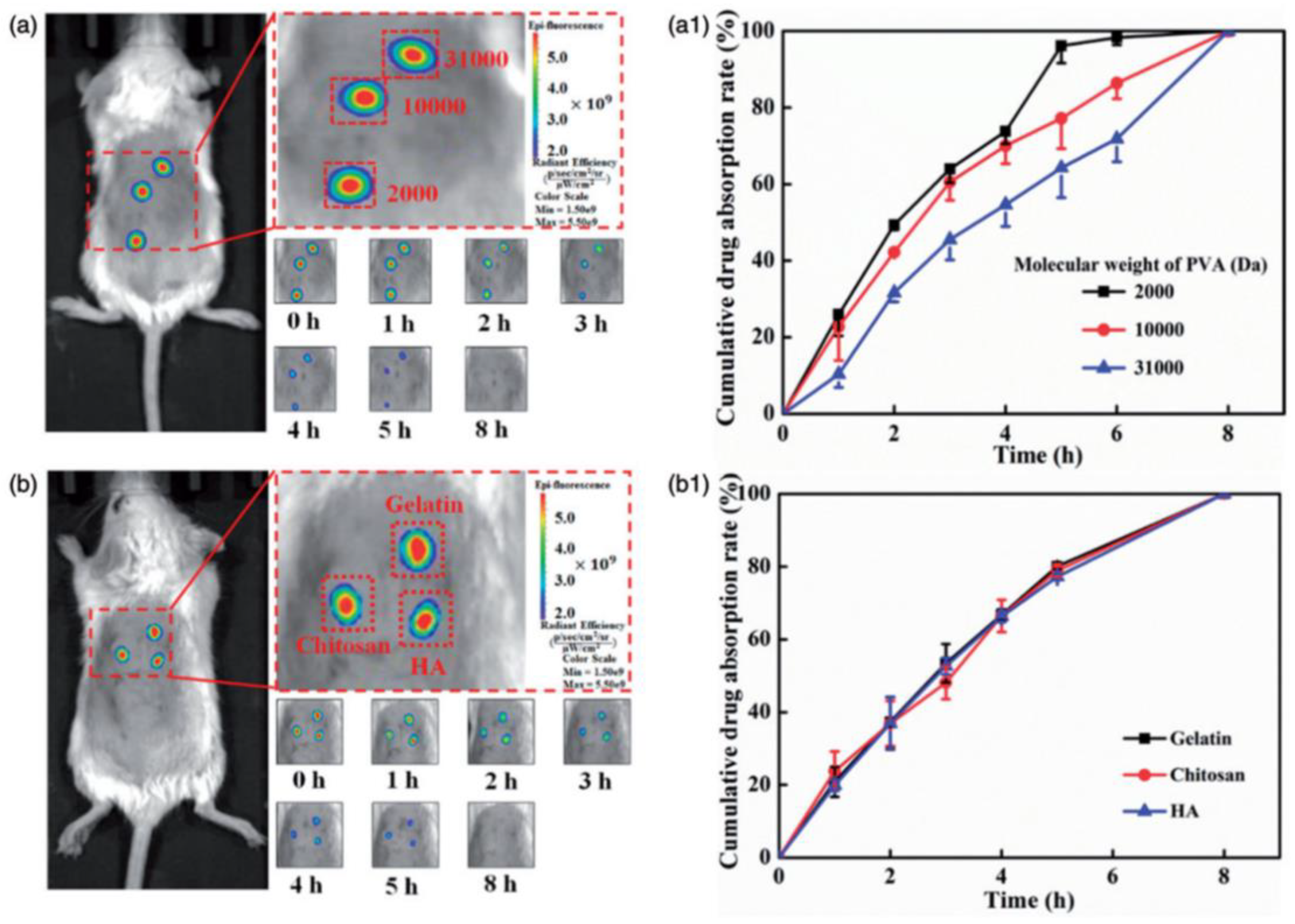
Chen et al. reported the fabrication of polymeric MNs for efficient TD drug delivery. MNs were mainly fabricated using PVA, HA, chitosan, and gelatin, and loaded with fluorescein 5(6)-isothiocyanate (FITC). The MN patch was subjected to a skin perforation study using porcine cadaver skin, and showed effective perforation and efficient subcutaneous drug delivery. The in vitro and in vivo drug release studies revealed that the drug release can be altered by altering the polymers on the basis of molecular weights, and this was proven by performing the polymeric-MN-based delivery of FITC in mice.
Figure 13 clearly indicates the effect of the change in the polymer or molecular weight of polymer (used for MN fabrication) on drug absorption. In the case of PVA, the increased molecular weight decreases the drug absorption, and this was found to be insignificant with gelatin, chitosan, and HA. Changing the polymers can change the drug release behavior and provide the advantage of controlled drug delivery [128].
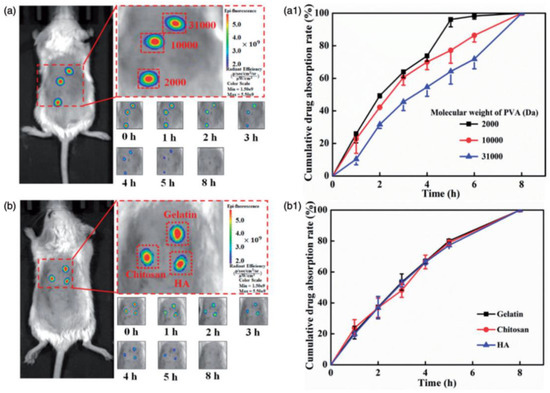
Figure 13.
Effect of change in polymer (used for MN fabrication) on in vivo drug absorption: (a,a1) fluorescence analysis of mice after drug administration through MNs made up from different molecular weight PVA and graphical representation of comparative drug release; and (b,b1) fluorescence analysis of mice after drug administration through MNs made up from gelatin, chitosan, and HA, and graphical representation of comparative drug release. Adapted with permission from Ref. [128]. Copyright 2018 Taylor and Francis.
Garland et al. fabricated the polymeric MNs for electrically assisted drug delivery. The authors reported the hybrid technology for delivery involving polymeric MNs and iontophoresis. The polymeric MNs were fabricated using poly vinyl methyl ether (PVMA) and malleic acid (MA). The dimensions of MNs play an important role in the efficient TD delivery of drugs. The increase in height and density of MNs increases the extent of TD drug delivery. The penetration study performed using neonatal porcine skin revealed that the developed hybrid drug delivery system delivers both small and large molecules efficiently. The delivery of large molecules such as protein becomes more efficient after combining iontophoresis with microneedling [129].
5.2.2. Intraocular
MNs exhibit potential applications in the treatment of various eye diseases by providing efficient localized drug delivery. MNs can bitterly target the various tissues in the eyes such as sclera, cornea, suprachoroidal space, etc. Various small and macromolecules are efficiently delivered in eyes using MNs with minimum invasion. Polymeric MNs have the BC and BD advantage in intraocular drug delivery [130].
Thakur et al. demonstrated the fabrication of polymeric MNs for the intraocular delivery of macromolecules (fluorescein sodium and fluorescein isothiocyanate–dextrans). MNs were fabricated using PVP of various molecular weights. Polymeric MNs showed the optimum force of insertion and depth of penetration. The PVP with high molecular weight can withstand higher forces. The PVP MNs showed better dissolution based on molecular weight. The efficient delivery of macromolecules was observed in corneal and scleral tissues [131].
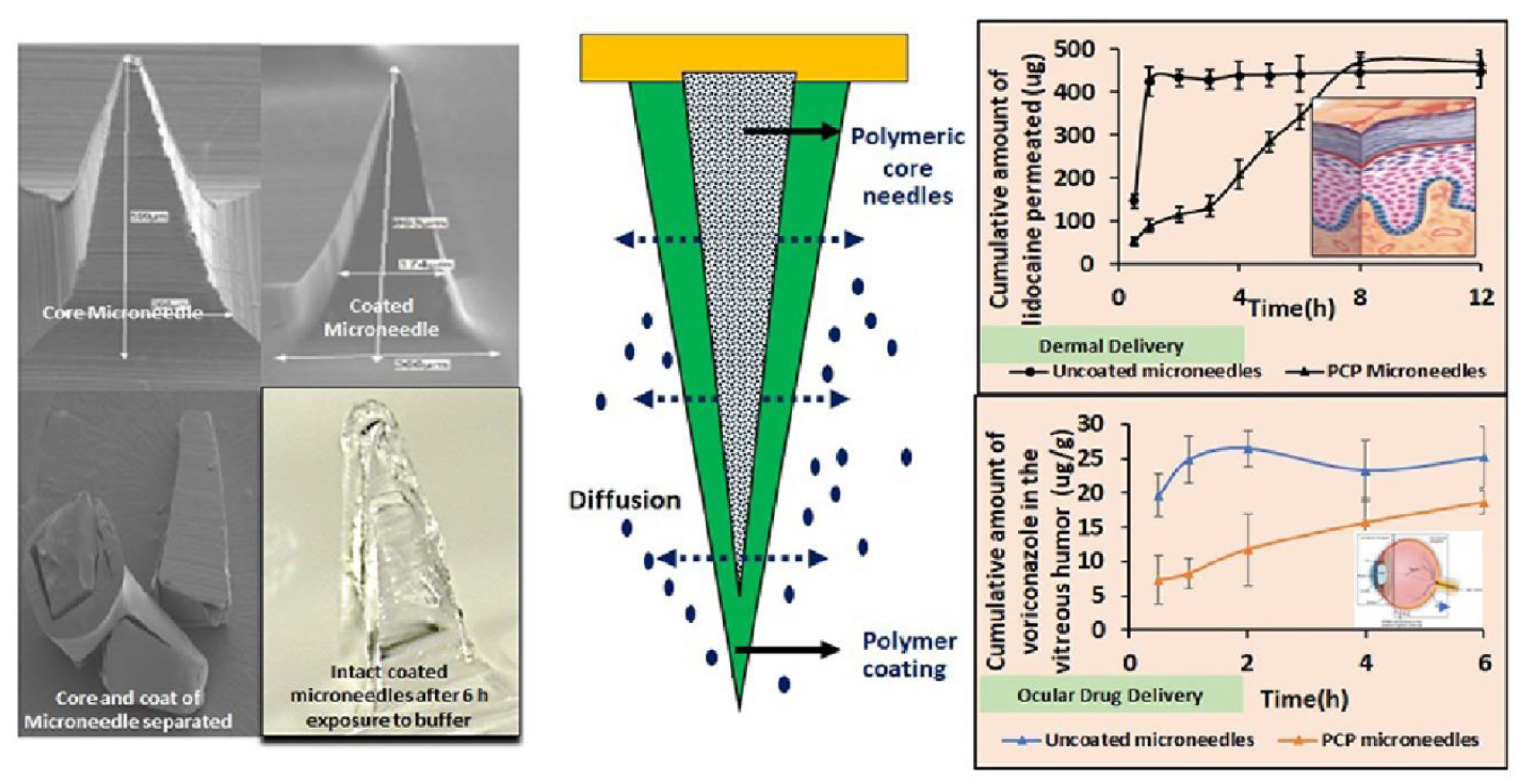
Jakka and coworkers developed a polymer-coated polymeric MN system for intravitreal drug delivery. MNs were fabricated using PVP-K30 as a core polymer. The drug release study revealed that the time required for the release of 50% dextran was 15 min; when PVP-K30 used, the time required was 20%. This drug release rate was extended to 90 min using 50% PVP-K30. Controlled intravitreal drug delivery can be achieved with polymer-coated polymeric MNs.
The fabricated polymer-coated polymeric MN system was found to be useful in delivering lidocaine hydrochloride for 9 h through the skin and voriconazole for 6 h intravitreally [132] (Figure 14).
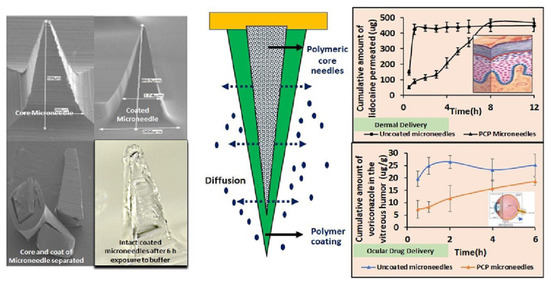
Figure 14.
Polymer-coated polymeric MNs for efficient delivery of idocaine hydrochloride in the skin tissue and voriconazole intravitreally. Adapted with permission from Ref. [132]. Copyright 2022 Elsevier.
5.3. Diagnostic and Biosensing Applications
Polymeric MNs are used for biosensing and diagnostic applications. Various former researchers have reported these applications of polymeric MNs. Barett et al. fabricated the gold-coated polymeric MNs for sensing the blood glucose level. Optical and electron microscopy were used for characterization. The detection of glucose was based on the principle of voltammetry. The ferrocene monocarboxylic acid acted as an oxidizing mediator in the presence of glucose oxidase. The prepared polymeric gold-coated MN device showed high sensitivity towards glucose with negligible selectivity towards other oxidizable substances such as ascorbic acid, uric acid, fructose, and drugs (paracetamol, aspirin, etc.). This fabricated MN device was demonstrated as a promising approach for glucose detection. A simple change in enzyme can make these MNs useful in the detection of cholesterol and lactate [133].
Polymeric micronnedles are also used to determine the plasma concentration of potent drugs such as opioids to avoid toxicity. Joshi and coworkers reported the fabrication of polymeric MNs to sense fentanyl in biofluid. A pyramidal array of polymeric MNs was integrated with silver and platinum wires with a microcavity. The microneedle sesnsor was functionalized with graphene ink and 4 (3-Butyl-1-imidazolio)-1-butanesulfonate) liquid. The voltametric-method-based detection of fentanyl was performed to detect fentanyl up to 27.8 μM. The detection was based on the oxidation of fentanyl in liquid. The fabricated biosensing device showed the selective and interference-free detection of fentanyl with high sensitivity [134]. Calio et al. demonstrated the fabrication of polymeric MNs for the biosensing of glucose and lactic acid. The MNs were fabricated from poly(ethylene glycol) diacrylate (PEGDA) by photolithography. The MNs were coated by gold solution and functionalized with enzymes (glucose and lactate oxidase) and redox mediator (vinylferrocene). The fabricated MN-based device showed the selective and sensitive detection of glucose and lactic acid in the mM range. This device can be useful in diabetic patients and athletes for monitoring the levels of glucose and lactic acid, respectively [135].
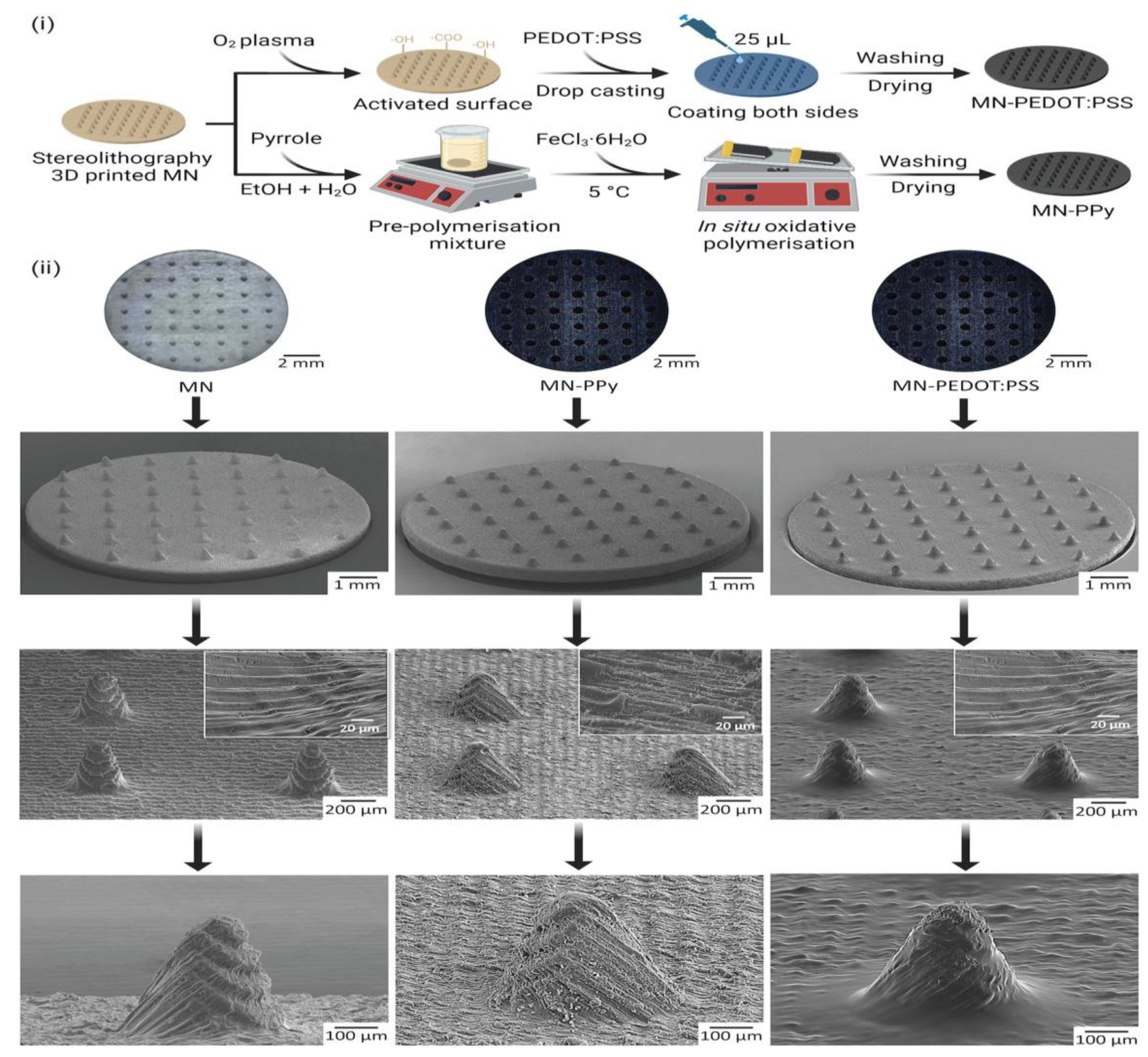
One of the emerging approaches used for the fabrication of MNs is 3D printing. Keirouz et al. reported the 3D-printing-based fabrication of MNs followed by coating with a conductive polymer for biosensing applications (Figure 15).
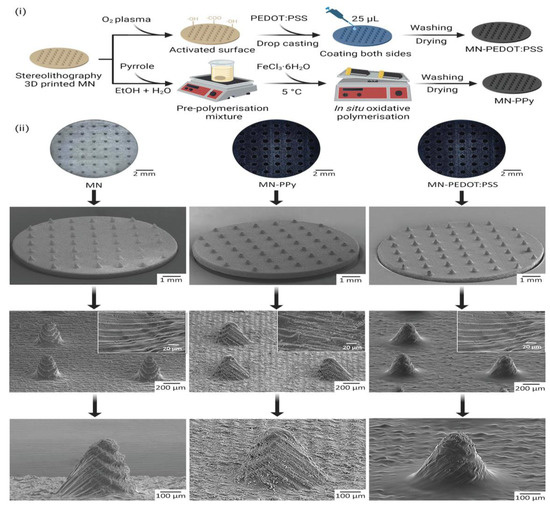
Figure 15.
Conductive-polymer-coated 3D-printing-based fabrication of MNs for biosensing applications. (i) Distinct coating approaches (ii) Micrograph and SEM images of coated and uncoated microneedles. Adapted from Ref. [136].
Polypyrrole (PPy) and poly(3,4-ethylenedioxythiophene)-poly(styrenesulfonate) were used to produce a conductive-polymer-based surface. The polymer-based MNs acted as conductive electrodes. These MNs were found to be non-cytotoxic when tested against human fibroblasts. The fabricated polymeric MN arrays were found to be a promising device for biosensing [136].
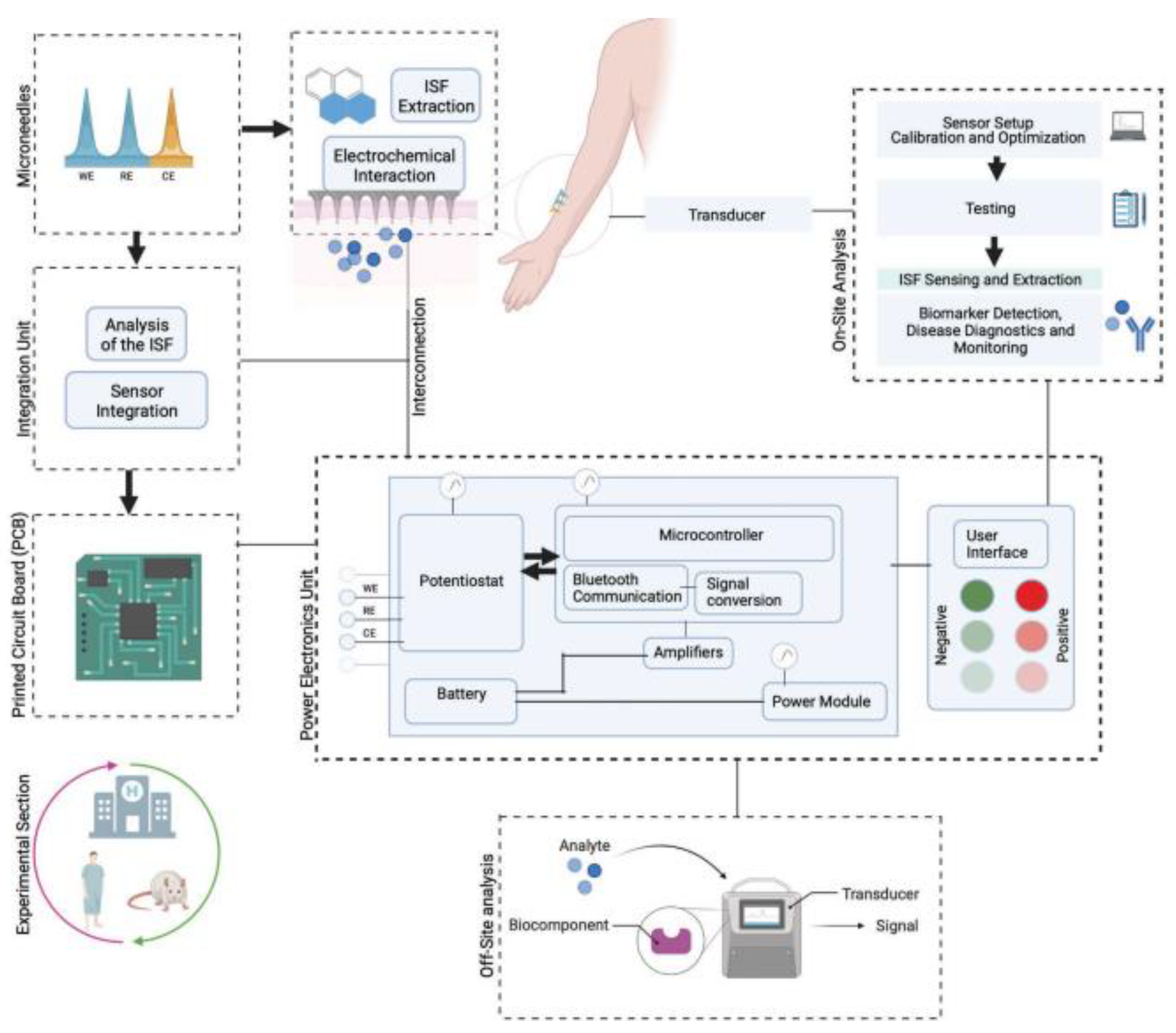
Interstitial fluid (ISF) has an important role in biosensing as it is one of the important body fluids containing various biomarkers. The extraction/sampling is challenging with conventional sampling techniques. Techniques such as microdialysis and suction blisters need expert monitoring. The MN-based sampling of ISF is an emerging area of research and useful tool with which to overcome the challenges of conventional techniques [137]. Figure 16 illustrates the MN-based sampling of ISF.
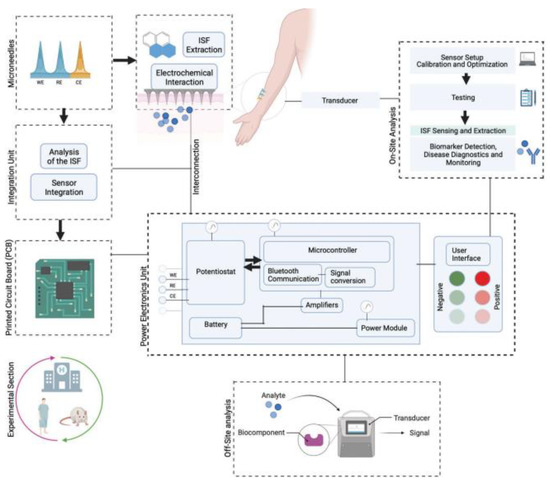
Figure 16.
MN-based interstitial fluid (ISF) sampling and biosensing device. Adapted from Ref. [137].
The MN-based sampling of ISF was found to be minimally invasive, economic, and less time-consuming compared to other techniques [137].
MNs improve the biosensing ability due to efficient TD permeation. The MN-based biosensing device can be useful to detect various analytes from interstitial fluid and demonstrate its usefulness in disease diagnosis [138,139,140].
5.4. Vaccination
As mentioned earlier, MNs are useful in delivering a wide variety of drug molecules. Multiple vaccines are delivered through the polymer-based MN system.
Ali et al. reported the application of polymeric MNs for the delivery of the DNA vaccine, useful against cervical cancer (Figure 17). The vaccine consists of cationic nanoparticles encapsulating the peptide RALA and condensed DNA (E6/E7). Polymeric MNs fabricated using PVP are loaded with these nanoparticles and subjected to a cutaneous delivery of the vaccine. The mice vaccinated with the proposed vaccine using polymeric MNs showed higher levels of E6/E7-specific IgGs with a high T-lymphocyte count and TC-1 cell-mediated cytotoxicity, and showed high levels of IFN-γ compared to the mice who received an intramuscular injection of vaccine-containing nanoparticles. The prepared MN-based vaccine delivery system can be a promising approach in cervical cancer therapy [141].
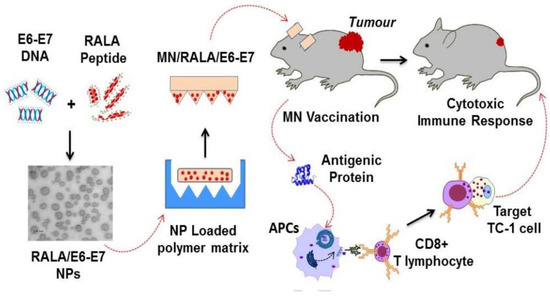
Figure 17.
Fabrication and therapeutic evaluation of DNA vaccine for cervical cancer. Adapted with permission from Ref. [141]. Copyright 2017 Elsevier.
Sullivan et al. reported the fabrication of polymeric MNs for the delivery of the influenza vaccine. The MNs were fabricated using PVP and encapsulated with the influenza vaccine. The penetration and dissolution study using porcine cadaver skin revealed that the MNs have an optimum insertion depth of 200 μm, and 89 ± 3% dissolution was observed within 15 min. Similar promising results were observed with an in vivo study in mice. The fabricated polymeric MNs demonstrated the successful delivery of the influenza vaccine [142].
Along with DNA and influenza vaccine delivery through polymeric MNs, Bacillus Calmette–Guérin (BCG) vaccine delivery was also successfully demonstrated using a polymeric MN system. MNs were fabricated using sodium alginate (10% w/v) and trehalose (20%). The delivery of BCG was compared with intradermal injection, and it was observed that, with minimum invasion, the polymeric MNs showed efficient delivery, and this was confirmed by estimating the count of granulocytes, lymphocytes, IgGs, etc. The levels of granulocytes, lymphocytes, and IgGs were found to be increased and this was the indication of efficient vaccination using the BCG-coated polymeric MNs [143].
Table 4 illustrates the biomedical applications of polymeric MNs along with the polymer and methods used for fabrication.

Table 4.
Polymeric MNs for biomedical applications.
6. Regulatory Considerations and Patent Scenario
The United States Food and Drug Administration (USFDA) has issued a guidance document on ‘Regulatory Considerations for Microneedling Products.’ The USFDA considers microneedling products to be instruments with an array of needles, “microprotrusion” tips, or pins of varying lengths that are incorporated into the body of an instrument. These needles can be blunt or sharp and are designed to be rolled or stamped across or into the skin. Microneedling products may be used for skin exfoliation, improving the appearance of skin, treating scars, wrinkles, and other skin conditions, and may be used for single or multiple users. Microneedling products may also be promoted with topical preparations such as semisolid creams, ointments and gels, vitamin solutions, other drugs, or blood related products. The FDA has also provided the definitions of stratum corneum, exfoliation, dermabrasion, and living layers of skin [168].
FDA has considered microneedling products under two categories: (a) medical devices and (b) products which are not devices. As per section 201(h) of the Federal Food, Drug, and Cosmetic Act (FDC Act), a medical device is “any instrument, machine, contrivance, implant, in vitro reagent that’s intended to treat, cure, prevent, mitigate, diagnose disease in man.” Microneedling products that are devices are subject to FDA regulations under the Federal Food, Drug, and Cosmetic Act. Another category of microneedling products are those used to enhance the appearance of the face affected by acne scars, wrinkles, and to enhance the appearance of abdominal scars in patients of 22 years or older. The products are reviewed and approved by the FDA, considering the safety and effectiveness of these devices for use as DeNovos (novel medical devices), which may require premarket notification under 510k. These medical products may be referred to such products which may not require the details from the clinical trials. Information about these products is available on the FDA’s website [169]. Table 5 illustrates the clinical trial data associated with polymeric MNs.

Table 5.
Clinical trial data of MNs [170].
Considering the FDA’s views on the authorization of microneedling products, the World Trade Organization has noted “Microneedling device for aesthetic use” under “Medical equipment” (ICS 11.040) to promote its innovation for the protection of human health or safety [179]. The Medicines & Healthcare Products Regulatory Agency (MHRA) conducted a consultation on the UK medical device regulations where respondents were positive on including microneedling products under the scope of medical devices [180]. The patenting scenario of MNs is illustrated in Table 6.

Table 6.
Patents on polymeric MN technology [170].
7. Conclusions and Future Perspective
Polymeric MNs are a promising new technology in biomedical science for TD drug delivery. It is effective in the delivery of a wide variety of drugs, including vaccines, hormone, and theranostic agents. The variety of polymers provides the scope for the fabrication of MNs with diversified biomedical applications. Painless penetration through the stratum corneum is the major advantage of drug delivery through MNs. The limitations of the oral, parenteral, and other routes can be overcome using MN technology. BC and BD are important criteria for the selection of polymers for MN fabrication. The availability of various natural and synthetic polymers provides multiple options for the fabrication of compatible MNs for the efficient delivery of specific drugs. The BD of polymeric MN after insertion is important for the TD delivery of various drug molecules. MN technology is demonstrating its broad-spectrum applications in the delivery of small as well as large molecules. Multiple polymer-based MN technologies are patented and many are under clinical trials. Novel approaches such as 3D-printing technology for the fabrication of high-quality MNs is creating the scope of future work in this area. Researchers are working on the improvement of mechanical strength, drug loading capacity, and biodegradability of MNs. However, MNs could be explored with regard to the diagnosis and treatment of orphan diseases, delivery of newer biologicals and theranostic agents, improving the safety and effectiveness of treatment, as well as making it economic for the wider public use. This technology has the potential to improve the lives of millions of people around the world.
Author Contributions
Conceptualization, D.K. and D.G.; methodology, N.C.; software, S.S.; validation, R.A., A.A. and S.P.; formal analysis, M.A.; investigation, R.B. (Rupali Badgule); resources, R.B. (Radhika Bansode); data curation, B.B.; writing—original draft preparation, D.K.; writing—review and editing, D.K.; visualization, D.G.; supervision, D.K.; project administration, D.G. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Acknowledgments
The authors are thankful to the Srinath College of Pharmacy, Aurangabad and DSEU, New Delhi for providing facilities for the literature survey.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Guillot, A.J.; Cordeiro, A.S.; Donnelly, R.F.; Montesinos, M.C.; Garrigues, T.M.; Melero, A. Microneedle-based delivery: An overview of current applications and trends. Pharmaceutics 2020, 12, 569. [Google Scholar] [CrossRef]
- Dugam, S.; Tade, R.; Dhole, R.; Nangare, S. Emerging era of microneedle array for pharmaceutical and biomedical applications: Recent advances and toxicological perspectives. Future J. Pharm. Sci. 2021, 7, 19. [Google Scholar] [CrossRef]
- Dawud, H.; Abu Ammar, A. Rapidly Dissolving Microneedles for the Delivery of Steroid-Loaded Nanoparticles Intended for the Treatment of Inflammatory Skin Diseases. Pharmaceutics 2023, 15, 526. [Google Scholar] [CrossRef] [PubMed]
- Avcil, M.; Çelik, A. Microneedles in Drug Delivery: Progress and Challenges. Micromachines 2021, 12, 1321. [Google Scholar] [CrossRef] [PubMed]
- Sharma, M. Chapter 18-Transdermal and Intravenous Nano Drug Delivery Systems: Present and Future. In Applications of Targeted Nano Drugs and Delivery Systems; Mohapatra, S.S., Ranjan, S., Dasgupta, N., Mishra, R.K., Thomas, S., Eds.; Micro and Nano Technologies; Elsevier: Amsterdam, The Netherlands, 2019; pp. 499–550. ISBN 978-0-12-814029-1. [Google Scholar]
- Gowda, B.H.J.; Ahmed, M.G.; Sahebkar, A.; Riadi, Y.; Shukla, R.; Kesharwani, P. Stimuli-Responsive Microneedles as a Transdermal Drug Delivery System: A Demand-Supply Strategy. Biomacromolecules 2022, 23, 1519–1544. [Google Scholar] [CrossRef]
- Waghule, T.; Singhvi, G.; Dubey, S.K.; Pandey, M.M.; Gupta, G.; Singh, M.; Dua, K. Microneedles: A smart approach and increasing potential for transdermal drug delivery system. Biomed. Pharmacother. 2019, 109, 1249–1258. [Google Scholar] [CrossRef] [PubMed]
- Rad, Z.F.; Prewett, P.D.; Davies, G.J. An overview of microneedle applications, materials, and fabrication methods. Beilstein J. Nanotechnol. 2021, 12, 1034–1046. [Google Scholar]
- Gera, A.K.; Burra, R.K. The Rise of Polymeric Microneedles: Recent Developments, Advances, Challenges, and Applications with Regard to Transdermal Drug Delivery. J. Funct. Biomater. 2022, 13, 81. [Google Scholar] [CrossRef]
- Karim, Z.; Karwa, P.; Hiremath, S.R.R. Polymeric Microneedles for Transdermal Drug Delivery—A Review of Recent Studies. J. Drug Deliv. Sci. Technol. 2022, 77, 103760. [Google Scholar] [CrossRef]
- Seetharam, A.A.; Choudhry, H.; Bakhrebah, M.A.; Abdulaal, W.H.; Gupta, M.S.; Rizvi, S.M.D.; Alam, Q.; Siddaramaiah; Gowda, D.V.; Moin, A. Microneedles Drug Delivery Systems for Treatment of Cancer: A Recent Update. Pharmaceutics 2020, 12, 1101. [Google Scholar] [CrossRef]
- Mahato, R. Microneedles in drug delivery. In Emerging Nanotechnologies for Diagnostics, Drug Delivery and Medical Devices; Elsevier: Amsterdam, The Netherlands, 2017; pp. 331–353. [Google Scholar]
- Aldawood, F.K.; Andar, A.; Desai, S. A comprehensive review of microneedles: Types, materials, processes, characterizations and applications. Polymers 2021, 13, 2815. [Google Scholar] [CrossRef] [PubMed]
- Kulkarni, D.; Damiri, F.; Rojekar, S.; Zehravi, M.; Ramproshad, S.; Dhoke, D.; Musale, S.; Mulani, A.A.; Modak, P.; Paradhi, R. Recent advancements in microneedle technology for multifaceted biomedical applications. Pharmaceutics 2022, 14, 1097. [Google Scholar] [CrossRef] [PubMed]
- Ita, K. Ceramic microneedles and hollow microneedles for transdermal drug delivery: Two decades of research. J. Drug Deliv. Sci. Technol. 2018, 44, 314–322. [Google Scholar] [CrossRef]
- Tucak, A.; Sirbubalo, M.; Hindija, L.; Rahić, O.; Hadžiabdić, J.; Muhamedagić, K.; Čekić, A.; Vranić, E. Microneedles: Characteristics, materials, production methods and commercial development. Micromachines 2020, 11, 961. [Google Scholar] [CrossRef]
- Haj-Ahmad, R.; Khan, H.; Arshad, M.S.; Rasekh, M.; Hussain, A.; Walsh, S.; Li, X.; Chang, M.-W.; Ahmad, Z. Microneedle coating techniques for transdermal drug delivery. Pharmaceutics 2015, 7, 486–502. [Google Scholar] [CrossRef] [PubMed]
- Ita, K. Dissolving Microneedles for Transdermal Drug Delivery: Advances and Challenges. Biomed. Pharmacother. 2017, 93, 1116–1127. [Google Scholar] [CrossRef]
- Rzhevskiy, A.S.; Singh, T.R.R.; Donnelly, R.F.; Anissimov, Y.G. Microneedles as the Technique of Drug Delivery Enhancement in Diverse Organs and Tissues. J. Control. Release 2018, 270, 184–202. [Google Scholar] [CrossRef]
- Turner, J.G.; White, L.R.; Estrela, P.; Leese, H.S. Hydrogel-forming microneedles: Current advancements and future trends. Macromol. Biosci. 2021, 21, 2000307. [Google Scholar] [CrossRef]
- Courtenay, A.J.; McAlister, E.; McCrudden, M.T.C.; Vora, L.; Steiner, L.; Levin, G.; Levy-Nissenbaum, E.; Shterman, N.; Kearney, M.-C.; McCarthy, H.O.; et al. Hydrogel-forming microneedle arrays as a therapeutic option for transdermal esketamine delivery. J. Control. Release 2020, 322, 177–186. [Google Scholar] [CrossRef]
- Lhernould, M.S.; Deleers, M.; Delchambre, A. Hollow polymer microneedles array resistance and insertion tests. Int. J. Pharm. 2015, 480, 152–157. [Google Scholar] [CrossRef]
- Tewabe, A.; Abate, A.; Tamrie, M.; Seyfu, A.; Abdela Siraj, E. Targeted Drug Delivery—From Magic Bullet to Nanomedicine: Principles, Challenges, and Future Perspectives. J. Multidiscip. Healthc. 2021, 14, 1711–1724. [Google Scholar] [CrossRef] [PubMed]
- Wells, C.M.; Harris, M.; Choi, L.; Murali, V.P.; Guerra, F.D.; Jennings, J.A. Stimuli-Responsive Drug Release from Smart Polymers. J. Funct. Biomater. 2019, 10, 34. [Google Scholar] [CrossRef]
- Begines, B.; Ortiz, T.; Pérez-Aranda, M.; Martínez, G.; Merinero, M.; Argüelles-Arias, F.; Alcudia, A. Polymeric Nanoparticles for Drug Delivery: Recent Developments and Future Prospects. Nanomaterials 2020, 10, 1403. [Google Scholar] [CrossRef] [PubMed]
- Ye, Y.; Yu, J.; Wen, D.; Kahkoska, A.R.; Gu, Z. Polymeric microneedles for transdermal protein delivery. Adv. Drug Deliv. Rev. 2018, 127, 106–118. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Hu, L.; Xu, C. Recent Advances in the Design of Polymeric Microneedles for Transdermal Drug Delivery and Biosensing. Lab Chip 2017, 17, 1373–1387. [Google Scholar] [CrossRef]
- Al-Japairai, K.A.S.; Mahmood, S.; Almurisi, S.H.; Venugopal, J.R.; Hilles, A.R.; Azmana, M.; Raman, S. Current trends in polymer microneedle for transdermal drug delivery. Int. J. Pharm. 2020, 587, 119673. [Google Scholar] [CrossRef]
- Zhang, R.; Miao, Q.; Deng, D.; Wu, J.; Miao, Y.; Li, Y. Research progress of advanced microneedle drug delivery system and its application in biomedicine. Colloids Surf. B Biointerfaces 2023, 226, 113302. [Google Scholar] [CrossRef]
- Abu-Much, A.; Darawshi, R.; Dawud, H.; Kasem, H.; Ammar, A.A. Preparation and characterization of flexible furosemide-loaded biodegradable microneedles for intradermal drug delivery. Biomater. Sci. 2022, 10, 6486–6499. [Google Scholar] [CrossRef]
- Chevala, N.T.; Jitta, S.R.; Marques, S.M.; Vaz, V.M.; Kumar, L. Polymeric Microneedles for Transdermal Delivery of Nanoparticles: Frontiers of Formulation, Sterility and Stability Aspects. J. Drug Deliv. Sci. Technol. 2021, 65, 102711. [Google Scholar] [CrossRef]
- Yang, L.; Yang, Y.; Chen, H.; Mei, L.; Zeng, X. Polymeric microneedle-mediated sustained release systems: Design strategies and promising applications for drug delivery. Asian J. Pharm. Sci. 2022, 17, 70–86. [Google Scholar] [CrossRef]
- Han, J.; Zhao, D.; Li, D.; Wang, X.; Jin, Z.; Zhao, K. Polymer-Based Nanomaterials and Applications for Vaccines and Drugs. Polymers 2018, 10, 31. [Google Scholar] [CrossRef]
- Ishak, R.A.H.; Osman, R.; Awad, G.A.S. Dextran-based Nanocarriers for Delivery of Bioactives. Curr. Pharm. Des. 2016, 22, 3411–3428. [Google Scholar] [CrossRef] [PubMed]
- Liang, J.; Yu, Y.; Li, C.; Li, Q.; Chen, P.; Li, W.; Liu, W.; Li, Z.; Liu, Y.; Zhang, S. Tofacitinib combined with melanocyte protector α-MSH to treat vitiligo through dextran based hydrogel microneedles. Carbohydr. Polym. 2023, 305, 120549. [Google Scholar] [CrossRef] [PubMed]
- Pitakjakpipop, H.; Rajan, R.; Tantisantisom, K.; Opaprakasit, P.; Nguyen, D.D.; Ho, V.A.; Matsumura, K.; Khanchaitit, P. Facile photolithographic fabrication of zwitterionic polymer microneedles with protein aggregation inhibition for transdermal drug delivery. Biomacromolecules 2021, 23, 365–376. [Google Scholar] [CrossRef] [PubMed]
- How, K.N.; Yap, W.H.; Lim, C.L.H.; Goh, B.H.; Lai, Z.W. Hyaluronic acid-mediated drug delivery system targeting for inflammatory skin diseases: A mini review. Front. Pharmacol. 2020, 11, 1105. [Google Scholar] [CrossRef]
- Chen, S.-X.; Ma, M.; Xue, F.; Shen, S.; Chen, Q.; Kuang, Y.; Liang, K.; Wang, X.; Chen, H. Construction of microneedle-assisted co-delivery platform and its combining photodynamic/immunotherapy. J. Control. Release 2020, 324, 218–227. [Google Scholar] [CrossRef]
- Pitt, W.G.; Husseini, G.A.; Staples, B.J. Ultrasonic drug delivery–a general review. Expert Opin. Drug Deliv. 2004, 1, 37–56. [Google Scholar] [CrossRef]
- Feng, Y.H.; Zhang, X.P.; Li, W.X.; Guo, X.D. Stability and diffusion properties of insulin in dissolvable microneedles: A multiscale simulation study. Langmuir 2021, 37, 9244–9252. [Google Scholar] [CrossRef]
- Chi, J.; Zhang, X.; Chen, C.; Shao, C.; Zhao, Y.; Wang, Y. Antibacterial and angiogenic chitosan microneedle array patch for promoting wound healing. Bioact. Mater. 2020, 5, 253–259. [Google Scholar] [CrossRef]
- Lv, J.; Ma, H.; Ye, G.; Jia, S.; He, J.; Jiaduo, W.; Ma, J.; Qu, Y.; Gou, K.; Zeng, R. Bilayer microneedles based on Bletilla striata polysaccharide containing asiaticoside effectively promote scarless wound healing. Mater. Des. 2023, 226, 111655. [Google Scholar] [CrossRef]
- Abourehab, M.A.S.; Rajendran, R.R.; Singh, A.; Pramanik, S.; Shrivastav, P.; Ansari, M.J.; Manne, R.; Amaral, L.S.; Deepak, A. Alginate as a Promising Biopolymer in Drug Delivery and Wound Healing: A Review of the State-of-the-Art. Int. J. Mol. Sci. 2022, 23, 9035. [Google Scholar] [CrossRef] [PubMed]
- Tiraton, T.; Suwantong, O.; Chuysinuan, P.; Ekabutr, P.; Niamlang, P.; Khampieng, T.; Supaphol, P. Biodegradable microneedle fabricated from sodium alginate-gelatin for transdermal delivery of clindamycin. Mater. Today Commun. 2022, 32, 104158. [Google Scholar] [CrossRef]
- Yu, W.; Jiang, G.; Zhang, Y.; Liu, D.; Xu, B.; Zhou, J. Polymer microneedles fabricated from alginate and hyaluronate for transdermal delivery of insulin. Mater. Sci. Eng. C Mater. Biol. Appl. 2017, 80, 187–196. [Google Scholar] [CrossRef]
- Zhang, Y.; Jiang, G.; Yu, W.; Liu, D.; Xu, B. Microneedles fabricated from alginate and maltose for transdermal delivery of insulin on diabetic rats. Mater. Sci. Eng. C 2018, 85, 18–26. [Google Scholar] [CrossRef] [PubMed]
- Al-Nimry, S.; Dayah, A.A.; Hasan, I.; Daghmash, R. Cosmetic, biomedical and pharmaceutical applications of fish gelatin/hydrolysates. Mar. Drugs 2021, 19, 145. [Google Scholar] [CrossRef]
- Wang, M.; Li, X.; Du, W.; Sun, M.; Ling, G.; Zhang, P. Microneedle-mediated treatment for superficial tumors by combining multiple strategies. Drug Deliv. Transl. Res. 2023, 13, 1600–1620. [Google Scholar] [CrossRef]
- Demir, B.; Rosselle, L.; Voronova, A.; Pagneux, Q.; Quenon, A.; Gmyr, V.; Jary, D.; Hennuyer, N.; Staels, B.; Hubert, T. Innovative transdermal delivery of insulin using gelatin methacrylate-based microneedle patches in mice and mini-pigs. Nanoscale Horiz. 2022, 7, 174–184. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Luo, Z.; Baidya, A.; Kim, H.; Wang, C.; Jiang, X.; Qu, M.; Zhu, J.; Ren, L.; Vajhadin, F. Biodegradable β-cyclodextrin conjugated gelatin methacryloyl microneedle for delivery of water-insoluble drug. Adv. Healthc. Mater. 2020, 9, 2000527. [Google Scholar] [CrossRef]
- Volpe-Zanutto, F.; Vora, L.K.; Tekko, I.A.; McKenna, P.E.; Permana, A.D.; Sabri, A.H.; Anjani, Q.K.; McCarthy, H.O.; Paredes, A.J.; Donnelly, R.F. Hydrogel-forming microarray patches with cyclodextrin drug reservoirs for long-acting delivery of poorly soluble cabotegravir sodium for HIV Pre-Exposure Prophylaxis. J. Control. Release 2022, 348, 771–785. [Google Scholar] [CrossRef]
- Donnelly, R.F.; Garland, M.J.; Alkilani, A.Z. Microneedle-iontophoresis combinations for enhanced transdermal drug delivery. Drug Deliv. Syst. 2014, 141, 121–132. [Google Scholar]
- Woolfson, A.D.; Morrow, D.I.J.; Morrissey, A.; Donnelly, R.F.; McCarron, P.A. Delivery Device and Method 2017. U.S. Patent 9549746B2, 24 January 2017. Available online: https://patents.google.com/patent/US9549746B2/en (accessed on 11 March 2023).
- Pamornpathomkul, B.; Ngawhirunpat, T.; Tekko, I.A.; Vora, L.; McCarthy, H.O.; Donnelly, R.F. Dissolving polymeric microneedle arrays for enhanced site-specific acyclovir delivery. Eur. J. Pharm. Sci. 2018, 121, 200–209. [Google Scholar] [CrossRef] [PubMed]
- Khiste, R.; Bhapkar, N.; Kulkarni, N. A Review on Applications of Hydroxy Propyl Methyl Cellulose and Natural polymers for the development of modified release drug delivery systems. Res. J. Pharm. Technol. 2021, 14, 1163–1170. [Google Scholar] [CrossRef]
- Nagra, U.; Barkat, K.; Ashraf, M.U.; Shabbir, M. Feasibility of Enhancing Skin Permeability of Acyclovir through Sterile Topical Lyophilized Wafer on Self-Dissolving Microneedle-Treated Skin. Dose-Response 2022, 20, 15593258221097594. [Google Scholar] [CrossRef]
- Poddar, D.; Singh, A.; Bansal, S.; Thakur, S.; Jain, P. Direct synthesis of Poly (Ԑ-Caprolactone)-block-poly (glycidyl methacrylate) copolymer and its usage as a potential nano micelles carrier for hydrophobic drugs. J. Indian Chem. Soc. 2022, 99, 100537. [Google Scholar] [CrossRef]
- Li, L.; Tian, H.; He, J.; Zhang, M.; Li, Z.; Ni, P. Fabrication of aminated poly (glycidyl methacrylate)-based polymers for co-delivery of anticancer drugs and the p53 gene. J. Mater. Chem. B 2020, 8, 9555–9565. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, H.X.; Bozorg, B.D.; Kim, Y.; Wieber, A.; Birk, G.; Lubda, D.; Banga, A.K. Poly (vinyl alcohol) microneedles: Fabrication, characterization, and application for transdermal drug delivery of doxorubicin. Eur. J. Pharm. Biopharm. 2018, 129, 88–103. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.P.; Wang, B.B.; Li, W.X.; Fei, W.M.; Cui, Y.; Guo, X.D. In vivo safety assessment, biodistribution and toxicology of polyvinyl alcohol microneedles with 160-day uninterruptedly applications in mice. Eur. J. Pharm. Biopharm. 2021, 160, 1–8. [Google Scholar] [CrossRef]
- Sillankorva, S.; Pires, L.; Pastrana, L.M.; Bañobre-López, M. Antibiofilm Efficacy of the Pseudomonas aeruginosa Pbunavirus vB_PaeM-SMS29 Loaded onto Dissolving Polyvinyl Alcohol Microneedles. Viruses 2022, 14, 964. [Google Scholar] [CrossRef]
- Seong, K.-Y.; Seo, M.-S.; Hwang, D.Y.; O’Cearbhaill, E.D.; Sreenan, S.; Karp, J.M.; Yang, S.Y. A self-adherent, bullet-shaped microneedle patch for controlled transdermal delivery of insulin. J. Control. Release 2017, 265, 48–56. [Google Scholar] [CrossRef]
- Seeni, R.Z.; Yu, X.; Chang, H.; Chen, P.; Liu, L.; Xu, C. Iron oxide nanoparticle-powered micro-optical coherence tomography for in situ imaging the penetration and swelling of polymeric microneedles in the skin. ACS Appl. Mater. Interfaces 2017, 9, 20340–20347. [Google Scholar] [CrossRef]
- Wu, L.; Shrestha, P.; Iapichino, M.; Cai, Y.; Kim, B.; Stoeber, B. Characterization method for calculating diffusion coefficient of drug from polylactic acid (PLA) microneedles into the skin. J. Drug Deliv. Sci. Technol. 2021, 61, 102192. [Google Scholar] [CrossRef]
- Wu, L.; Park, J.; Kamaki, Y.; Kim, B. Optimization of the fused deposition modeling-based fabrication process for polylactic acid microneedles. Microsyst. Nanoeng. 2021, 7, 58. [Google Scholar] [CrossRef] [PubMed]
- Na, Y.-G.; Kim, M.; Han, M.; Huh, H.W.; Kim, J.-S.; Kim, J.C.; Park, J.-H.; Lee, H.-K.; Cho, C.-W. Characterization of hepatitis B surface antigen loaded polylactic acid-based microneedle and its dermal safety profile. Pharmaceutics 2020, 12, 531. [Google Scholar] [CrossRef]
- Ju, J.; Hsieh, C.-M.; Tian, Y.; Kang, J.; Chia, R.; Chang, H.; Bai, Y.; Xu, C.; Wang, X.; Liu, Q. Surface enhanced Raman spectroscopy based biosensor with a microneedle array for minimally invasive in vivo glucose measurements. ACS Sens. 2020, 5, 1777–1785. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Luo, Y.; Chen, H.; Xu, S.; Zhang, D.; Sang, H.; Xu, C.; Zhang, M. Comparison of the efficacy of seven types of microneedles for treating rabbit hypertrophic scar model. Nanoscale Adv. 2023, 5, 927–933. [Google Scholar] [CrossRef]
- Bocchino, A.; Teixeira, S.R.; Iadanza, S.; Melnik, E.; Kurzhals, S.; Mutinati, G.C.; O’Mahony, C. Development and Characterization of Passivation Methods for Microneedle-based Biosensors. In Proceedings of the 2022 44th Annual International Conference of the IEEE Engineering in Medicine & Biology Society (EMBC), Glasgow, Scotland, UK, 11–15 July 2022; pp. 1275–1278. [Google Scholar]
- Kanaki, Z.; Chandrinou, C.; Orfanou, I.-M.; Kryou, C.; Ziesmer, J.; Sotiriou, G.A.; Klinakis, A.; Tamvakopoulos, C.; Zergioti, I. Laser-Induced Forward Transfer Printing on Microneedles for Transdermal Delivery of Gemcitabine. Int. J. Bioprint. 2022, 8, 554. [Google Scholar] [CrossRef]
- Hegarty, C.; McConville, A.; McGlynn, R.J.; Mariotti, D.; Davis, J. Design of composite microneedle sensor systems for the measurement of transdermal pH. Mater. Chem. Phys. 2019, 227, 340–346. [Google Scholar] [CrossRef]
- Eum, J.; Kim, Y.; Um, D.J.; Shin, J.; Yang, H.; Jung, H. Solvent-free polycaprolactone dissolving microneedles generated via the thermal melting method for the sustained release of capsaicin. Micromachines 2021, 12, 167. [Google Scholar] [CrossRef]
- Liu, W.; Speranza, G. Functionalization of carbon nanomaterials for biomedical applications. C 2019, 5, 72. [Google Scholar] [CrossRef]
- Ma, G.; Wu, C. Microneedle, bio-microneedle and bio-inspired microneedle: A review. J. Control. Release 2017, 251, 11–23. [Google Scholar] [CrossRef]
- Eskens, O.; Amin, S. Challenges and effective routes for formulating and delivery of epidermal growth factors in skin care. Int. J. Cosmet. Sci. 2021, 43, 123–130. [Google Scholar] [CrossRef] [PubMed]
- Damiri, F.; Kommineni, N.; Ebhodaghe, S.O.; Bulusu, R.; Jyothi, V.G.S.; Sayed, A.A.; Awaji, A.A.; Germoush, M.O.; Al-Malky, H.S.; Nasrullah, M.Z. Microneedle-Based Natural Polysaccharide for Drug Delivery Systems (DDS): Progress and Challenges. Pharmaceuticals 2022, 15, 190. [Google Scholar] [CrossRef] [PubMed]
- Vora, L.K.; Moffatt, K.; Tekko, I.A.; Paredes, A.J.; Volpe-Zanutto, F.; Mishra, D.; Peng, K.; Thakur, R.R.S.; Donnelly, R.F. Microneedle array systems for long-acting drug delivery. Eur. J. Pharm. Biopharm. 2021, 159, 44–76. [Google Scholar] [CrossRef] [PubMed]
- Yong, L.; Guangzhi, G.; Yanni, W.; Fengsen, M. Drug delivery with dissolving microneedles: Skin puncture, its influencing factors and improvement strategies. J. Drug Deliv. Sci. Technol. 2022, 76, 103653. [Google Scholar]
- Angel, D.; Carrillo, J.; Gomerz, E.; Antonia, D.; Prieto, M.; Maria, J.; Gomez, J.R. The effect of focused MW is on the reaction of ethyl N-trichloroethyls ledene carbamatic with pyrazole derivatives. Cannizaro reaction. Synlett 1998, 10, 10. [Google Scholar]
- Zhuang, J.; Rao, F.; Wu, D.; Huang, Y.; Xu, H.; Gao, W.; Zhang, J.; Sun, J. Study on the Fabrication and Characterization of Tip-Loaded Dissolving Microneedles for Transdermal Drug Delivery. Eur. J. Pharm. Biopharm. 2020, 157, 66–73. [Google Scholar] [CrossRef]
- McElvany, K.D. FDA requirements for preclinical studies. In Clinical Trials in the Neurosciences; Karger Publishers: Basel, Switzerland, 2009; Volume 25, pp. 46–49. [Google Scholar]
- Faraji Rad, Z.; Prewett, P.D.; Davies, G.J. High-resolution two-photon polymerization: The most versatile technique for the fabrication of microneedle arrays. Microsyst. Nanoeng. 2021, 7, 71. [Google Scholar] [CrossRef]
- Celis, P.; Vazquez, E.; Soria-Hernandez, C.G.; Bargnani, D.; Rodriguez, C.A.; Ceretti, E.; García-López, E. Evaluation of ball end micromilling for Ti6Al4V ELI microneedles using a nanoadditive under MQL condition. Int. J. Precis. Eng. Manuf. Green Technol. 2022, 9, 1231–1246. [Google Scholar] [CrossRef]
- O’Toole, L.; Kang, C.-W.; Fang, F.-Z. Precision micro-milling process: State of the art. Adv. Manuf. 2021, 9, 173–205. [Google Scholar] [CrossRef]
- Tarbox, T.N.; Watts, A.B.; Cui, Z.; Williams, R.O. An update on coating/manufacturing techniques of microneedles. Drug Deliv. Transl. Res. 2018, 8, 1828–1843. [Google Scholar] [CrossRef]
- Olowe, M.; Parupelli, S.K.; Desai, S. A Review of 3D-Printing of Microneedles. Pharmaceutics 2022, 14, 2693. [Google Scholar] [CrossRef] [PubMed]
- Dabbagh, S.; Sarabi, M.; Rahbarghazi, R.; Sokullu, E.; Yetisen, A.; Tasoglu, S. 3D-printed microneedles in biomedical applications. iScience 2021, 24, 102012. [Google Scholar] [CrossRef] [PubMed]
- Han, D.; Morde, R.S.; Mariani, S.; La Mattina, A.A.; Vignali, E.; Yang, C.; Barillaro, G.; Lee, H. 4D Printing of a Bioinspired Microneedle Array with Backward-Facing Barbs for Enhanced Tissue Adhesion. Adv. Funct. Mater. 2020, 30, 1909197. [Google Scholar] [CrossRef]
- Choi, J.; Kwon, O.-C.; Jo, W.; Lee, H.J.; Moon, M.-W. 4D Printing Technology: A Review. 3D Print. Addit. Manuf. 2015, 2, 159–167. [Google Scholar] [CrossRef]
- Wan, X.; Luo, L.; Liu, Y.; Leng, J. Direct Ink Writing Based 4D Printing of Materials and Their Applications. Adv. Sci. 2020, 7, 2001000. [Google Scholar] [CrossRef]
- Papadimitriou, P.; Andriotis, E.G.; Fatouros, D.; Tzetzis, D. Design and Prototype Fabrication of a Cost-Effective Microneedle Drug Delivery Apparatus Using Fused Filament Fabrication, Liquid Crystal Display and Semi-Solid Extrusion 3D Printing Technologies. Micromachines 2022, 13, 1319. [Google Scholar] [CrossRef]
- Kumar, H.; Kim, K. Stereolithography 3D Bioprinting. In 3D Bioprinting: Principles and Protocols; Crook, J.M., Ed.; Methods in Molecular Biology; Springer: New York, NY, USA, 2020; pp. 93–108. ISBN 978-1-07-160520-2. [Google Scholar]
- Zhao, Z.; Tian, X.; Song, X. Engineering materials with light: Recent progress in digital light processing based 3D printing. J. Mater. Chem. C 2020, 8, 13896–13917. [Google Scholar] [CrossRef]
- Kafle, A.; Luis, E.; Silwal, R.; Pan, H.M.; Shrestha, P.L.; Bastola, A.K. 3D/4D Printing of Polymers: Fused Deposition Modelling (FDM), Selective Laser Sintering (SLS), and Stereolithography (SLA). Polymers 2021, 13, 3101. [Google Scholar] [CrossRef]
- Willemen, N.G.A.; Morsink, M.A.J.; Veerman, D.; da Silva, C.F.; Cardoso, J.C.; Souto, E.B.; Severino, P. From oral formulations to drug-eluting implants: Using 3D and 4D printing to develop drug delivery systems and personalized medicine. Bio-Des. Manuf. 2022, 5, 85–106. [Google Scholar] [CrossRef]
- Gittard, S.D.; Ovsianikov, A.; Chichkov, B.N.; Doraiswamy, A.; Narayan, R.J. Two-photon polymerization of microneedles for transdermal drug delivery. Expert Opin. Drug Deliv. 2010, 7, 513–533. [Google Scholar] [CrossRef]
- Tran, K.T.; Nguyen, T.D. Lithography-based methods to manufacture biomaterials at small scales. J. Sci. Adv. Mater. Devices 2017, 2, 1–14. [Google Scholar] [CrossRef]
- Ross, S.; Scoutarios, N.; Lamprou, D.; Mallinson, D.; Douroumis, D. Drug Delivery Transl. Res 2015, 5, 451. [Google Scholar]
- Lechuga, Y.; Kandel, G.; Miguel, J.A.; Martinez, M. Development of an Automated Design Tool for FEM-Based Characterization of Solid and Hollow Microneedles. Micromachines 2023, 14, 133. [Google Scholar] [CrossRef]
- Howells, O.; Blayney, G.J.; Gualeni, B.; Birchall, J.C.; Eng, P.F.; Ashraf, H.; Sharma, S.; Guy, O.J. Design, fabrication, and characterisation of a silicon microneedle array for transdermal therapeutic delivery using a single step wet etch process. Eur. J. Pharm. Biopharm. 2022, 171, 19–28. [Google Scholar] [CrossRef] [PubMed]
- Hardick, O.; Stevens, B.; Bracewell, D.G. Nanofibre fabrication in a temperature and humidity controlled environment for improved fibre consistency. J. Mater. Sci. 2011, 46, 3890–3898. [Google Scholar] [CrossRef]
- Ali, R.; Mehta, P.; Monou, P.K.; Arshad, M.S.; Panteris, E.; Rasekh, M.; Singh, N.; Qutachi, O.; Wilson, P.; Tzetzis, D. Electrospinning/electrospraying coatings for metal microneedles: A design of experiments (DOE) and quality by design (QbD) approach. Eur. J. Pharm. Biopharm. 2020, 156, 20–39. [Google Scholar] [CrossRef]
- Peng, T.; Zhu, Y.; Leu, M.; Bourell, D. Additive manufacturing-enabled design, manufacturing, and lifecycle performance. Addit. Manuf. 2020, 36, 101646. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Jiang, G.; Aharodnikau, U.E.; Yunusov, K.; Sun, Y.; Liu, T.; Solomevich, S.O. Recent Advances in Polymer Microneedles for Drug Transdermal Delivery: Design Strategies and Applications. Macromol. Rapid Commun. 2022, 43, 2200037. [Google Scholar] [CrossRef]
- Wu, C.; Cheng, J.; Li, W.; Yang, L.; Dong, H.; Zhang, X. Programmable Polymeric Microneedles for Combined Chemotherapy and Antioxidative Treatment of Rheumatoid Arthritis. ACS Appl. Mater. Interfaces 2021, 13, 55559–55568. [Google Scholar] [CrossRef]
- Yao, W.; Tao, C.; Zou, J.; Zheng, H.; Zhu, J.; Zhu, Z.; Zhu, J.; Liu, L.; Li, F.; Song, X. Flexible two-layer dissolving and safing microneedle transdermal of neurotoxin: A biocomfortable attempt to treat Rheumatoid Arthritis. Int. J. Pharm. 2019, 563, 91–100. [Google Scholar] [CrossRef] [PubMed]
- Tekko, I.A.; Chen, G.; Domínguez-Robles, J.; Thakur, R.R.S.; Hamdan, I.M.N.; Vora, L.; Larrañeta, E.; McElnay, J.C.; McCarthy, H.O.; Rooney, M.; et al. Development and characterisation of novel poly (Vinyl Alcohol)/poly (Vinyl Pyrrolidone)-based hydrogel-forming microneedle arrays for enhanced and sustained transdermal delivery of methotrexate. Int. J. Pharm. 2020, 586, 119580. [Google Scholar] [CrossRef]
- Du, G.; He, P.; Zhao, J.; He, C.; Jiang, M.; Zhang, Z.; Zhang, Z.; Sun, X. Polymeric microneedle-mediated transdermal delivery of melittin for rheumatoid arthritis treatment. J. Control. Release 2021, 336, 537–548. [Google Scholar] [CrossRef] [PubMed]
- Dangol, M.; Yang, H.; Li, C.G.; Lahiji, S.F.; Kim, S.; Ma, Y.; Jung, H. Innovative polymeric system (IPS) for solvent-free lipophilic drug transdermal delivery via dissolving microneedles. J. Control. Release 2016, 223, 118–125. [Google Scholar] [CrossRef]
- Ogbechie-Godec, O.A.; Elbuluk, N. Melasma: An Up-to-Date Comprehensive Review. Dermatol. Ther. 2017, 7, 305–318. [Google Scholar] [CrossRef] [PubMed]
- Machekposhti, S.A.; Soltani, M.; Najafizadeh, P.; Ebrahimi, S.A.; Chen, P. Biocompatible polymer microneedle for topical/dermal delivery of tranexamic acid. J. Control. Release 2017, 261, 87–92. [Google Scholar] [CrossRef] [PubMed]
- He, Y.T.; Hao, Y.Y.; Yu, R.X.; Zhang, C.; Chen, B.Z.; Cui, Y.; Guo, X.D. Hydroquinone cream-based polymer microneedle roller for the combined treatment of large-area chloasma. Eur. J. Pharm. Biopharm. 2023, 185, 5–12. [Google Scholar] [CrossRef] [PubMed]
- Shravanth, S.H.; Osmani, R.A.M.; L, J.S.; Anupama, V.P.; Rahamathulla, M.; Gangadharappa, H.V. Microneedles-based drug delivery for the treatment of psoriasis. J. Drug Deliv. Sci. Technol. 2021, 64, 102668. [Google Scholar] [CrossRef]
- Gowda, B.H.J.; Ahmed, M.G.; Hani, U.; Kesharwani, P.; Wahab, S.; Paul, K. Microneedles as a momentous platform for psoriasis therapy and diagnosis: A state-of-the-art review. Int. J. Pharm. 2023, 632, 122591. [Google Scholar] [CrossRef]
- Zhao, Z.; Chen, Y.; Shi, Y. Microneedles: A potential strategy in transdermal delivery and application in the management of psoriasis. RSC Adv. 2020, 10, 14040–14049. [Google Scholar] [CrossRef]
- Tekko, I.A.; Permana, A.D.; Vora, L.; Hatahet, T.; McCarthy, H.O.; Donnelly, R.F. Localised and sustained intradermal delivery of methotrexate using nanocrystal-loaded microneedle arrays: Potential for enhanced treatment of psoriasis. Eur. J. Pharm. Sci. 2020, 152, 105469. [Google Scholar] [CrossRef]
- Ramalheiro, A.; Paris, J.L.; Silva, B.F.B.; Pires, L.R. Rapidly dissolving microneedles for the delivery of cubosome-like liquid crystalline nanoparticles with sustained release of rapamycin. Int. J. Pharm. 2020, 591, 119942. [Google Scholar] [CrossRef]
- Demartis, S.; Anjani, Q.K.; Volpe-Zanutto, F.; Paredes, A.J.; Jahan, S.A.; Vora, L.K.; Donnelly, R.F.; Gavini, E. Trilayer dissolving polymeric microneedle array loading Rose Bengal transfersomes as a novel adjuvant in early-stage cutaneous melanoma management. Int. J. Pharm. 2022, 627, 122217. [Google Scholar] [CrossRef]
- Matadh, A.V.; Jakka, D.; Pragathi, S.G.; Rangappa, S.; Shivakumar, H.N.; Maibach, H.; Reena, N.M.; Murthy, S.N. Polymer-Coated Polymeric (PCP) Microneedles for Controlled Dermal Delivery of 5-Fluorouracil. AAPS PharmSciTech 2022, 24, 9. [Google Scholar] [CrossRef]
- Alafnan, A.; Seetharam, A.A.; Hussain, T.; Gupta, M.S.; Rizvi, S.M.D.; Moin, A.; Alamri, A.; Unnisa, A.; Awadelkareem, A.M.; Elkhalifa, A.O.; et al. Development and Characterization of PEGDA Microneedles for Localized Drug Delivery of Gemcitabine to Treat Inflammatory Breast Cancer. Materials 2022, 15, 7693. [Google Scholar] [CrossRef] [PubMed]
- Bhatnagar, S.; Bankar, N.G.; Kulkarni, M.V.; Venuganti, V.V.K. Dissolvable microneedle patch containing doxorubicin and docetaxel is effective in 4T1 xenografted breast cancer mouse model. Int. J. Pharm. 2019, 556, 263–275. [Google Scholar] [CrossRef] [PubMed]
- Moreira, A.F.; Rodrigues, C.F.; Jacinto, T.A.; Miguel, S.P.; Costa, E.C.; Correia, I.J. Poly (Vinyl Alcohol)/chitosan layer-by-layer microneedles for cancer chemo-photothermal therapy. Int. J. Pharm. 2020, 576, 118907. [Google Scholar] [CrossRef] [PubMed]
- Than, A.; Liang, K.; Xu, S.; Sun, L.; Duan, H.; Xi, F.; Xu, C.; Chen, P. Transdermal Delivery of Anti-Obesity Compounds to Subcutaneous Adipose Tissue with Polymeric Microneedle Patches. Small Methods 2017, 1, 1700269. [Google Scholar] [CrossRef]
- Rabiei, M.; Kashanian, S.; Bahrami, G.; Derakhshankhah, H.; Barzegari, E.; Samavati, S.S.; McInnes, S.J.P. Dissolving microneedle-assisted long-acting Liraglutide delivery to control type 2 diabetes and obesity. Eur. J. Pharm. Sci. 2021, 167, 106040. [Google Scholar] [CrossRef]
- Xie, Y.; Shao, R.; Lin, Y.; Wang, C.; Tan, Y.; Xie, W.; Sun, S. Improved Therapeutic Efficiency against Obesity through Transdermal Drug Delivery Using Microneedle Arrays. Pharmaceutics 2021, 13, 827. [Google Scholar] [CrossRef]
- Dangol, M.; Kim, S.; Li, C.G.; Fakhraei Lahiji, S.; Jang, M.; Ma, Y.; Huh, I.; Jung, H. Anti-obesity effect of a novel caffeine-loaded dissolving microneedle patch in high-fat diet-induced obese C57BL/6J mice. J. Control. Release 2017, 265, 41–47. [Google Scholar] [CrossRef]
- Chen, Y.; Yang, Y.; Xian, Y.; Singh, P.; Feng, J.; Cui, S.; Carrier, A.; Oakes, K.; Luan, T.; Zhang, X. Multifunctional Graphene-Oxide-Reinforced Dissolvable Polymeric Microneedles for Transdermal Drug Delivery. ACS Appl. Mater. Interfaces 2020, 12, 352–360. [Google Scholar] [CrossRef] [PubMed]
- Chen, B.Z.; Ashfaq, M.; Zhang, X.P.; Zhang, J.N.; Guo, X.D. In vitro and in vivo assessment of polymer microneedles for controlled transdermal drug delivery. J. Drug Target. 2018, 26, 720–729. [Google Scholar] [CrossRef] [PubMed]
- Garland, M.J.; Caffarel–Salvador, E.; Migalska, K.; Woolfson, A.D.; Donnelly, R.F. Dissolving polymeric microneedle arrays for electrically assisted transdermal drug delivery. J. Control. Release 2012, 159, 52–59. [Google Scholar] [CrossRef] [PubMed]
- Patel, S.R.; Edelhauser, H.F.; Prausnitz, M.R. Targeted Drug Delivery to the Eye Enabled by Microneedles. In Drug Product Development for the Back of the Eye; Kompella, U.B., Edelhauser, H.F., Eds.; AAPS Advances in the Pharmaceutical Sciences Series; Springer: Boston, MA, USA, 2011; pp. 331–360. ISBN 978-1-4419-9920-7. [Google Scholar]
- Thakur, R.R.S.; Tekko, I.A.; Al-Shammari, F.; Ali, A.A.; McCarthy, H.; Donnelly, R.F. Rapidly dissolving polymeric microneedles for minimally invasive intraocular drug delivery. Drug Deliv. Transl. Res. 2016, 6, 800–815. [Google Scholar] [CrossRef]
- Jakka, D.; Matadh, A.V.; Shankar, V.K.; Shivakumar, H.N.; Murthy, S.N. Polymer Coated Polymeric (PCP) Microneedles for Controlled Delivery of Drugs (Dermal and Intravitreal). JPharmSci 2022, 111, 2867–2878. [Google Scholar] [CrossRef]
- Barrett, C.; Dawson, K.; O’Mahony, C.; O’Riordan, A. Development of Low Cost Rapid Fabrication of Sharp Polymer Microneedles for In Vivo Glucose Biosensing Applications. ECS J. Solid State Sci. Technol. 2015, 4, S3053. [Google Scholar] [CrossRef]
- Joshi, P.; Riley, P.R.; Mishra, R.; Azizi Machekposhti, S.; Narayan, R. Transdermal Polymeric Microneedle Sensing Platform for Fentanyl Detection in Biofluid. Biosensors 2022, 12, 198. [Google Scholar] [CrossRef]
- Caliò, A.; Dardano, P.; Di Palma, V.; Bevilacqua, M.F.; Di Matteo, A.; Iuele, H.; De Stefano, L. Polymeric microneedles based enzymatic electrodes for electrochemical biosensing of glucose and lactic acid. Sens. Actuators B Chem. 2016, 236, 343–349. [Google Scholar] [CrossRef]
- Keirouz, A.; Mustafa, Y.L.; Turner, J.G.; Lay, E.; Jungwirth, U.; Marken, F.; Leese, H.S. Conductive Polymer-Coated 3D Printed Microneedles: Biocompatible Platforms for Minimally Invasive Biosensing Interfaces. Small 2023, 19, 2206301. [Google Scholar] [CrossRef] [PubMed]
- Saifullah, K.M.; Faraji Rad, Z. Sampling Dermal Interstitial Fluid Using Microneedles: A Review of Recent Developments in Sampling Methods and Microneedle-Based Biosensors. Adv. Mater. Interfaces 2023, 10, 2201763. [Google Scholar] [CrossRef]
- Amarnani, R.; Shende, P. Microneedles in diagnostic, treatment and theranostics: An advancement in minimally-invasive delivery system. Biomed Microdevices 2021, 24, 4. [Google Scholar] [CrossRef] [PubMed]
- Gowda, B.H.J.; Ahmed, M.G.; Sanjana, A. Can Microneedles Replace Hypodermic Needles? Reson 2022, 27, 63–85. [Google Scholar] [CrossRef]
- Liu, G.-S.; Kong, Y.; Wang, Y.; Luo, Y.; Fan, X.; Xie, X.; Yang, B.-R.; Wu, M.X. Microneedles for transdermal diagnostics: Recent advances and new horizons. Biomaterials 2020, 232, 119740. [Google Scholar] [CrossRef] [PubMed]
- Ali, A.A.; McCrudden, C.M.; McCaffrey, J.; McBride, J.W.; Cole, G.; Dunne, N.J.; Robson, T.; Kissenpfennig, A.; Donnelly, R.F.; McCarthy, H.O. DNA vaccination for cervical cancer; a novel technology platform of RALA mediated gene delivery via polymeric microneedles. Nanomed. Nanotechnol. Biol. Med. 2017, 13, 921–932. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, S.P.; Koutsonanos, D.G.; del Pilar Martin, M.; Lee, J.W.; Zarnitsyn, V.; Choi, S.-O.; Murthy, N.; Compans, R.W.; Skountzou, I.; Prausnitz, M.R. Dissolving polymer microneedle patches for influenza vaccination. Nat. Med. 2010, 16, 915–920. [Google Scholar] [CrossRef]
- Arshad, M.S.; Fatima, S.; Nazari, K.; Ali, R.; Farhan, M.; Muhammad, S.A.; Abbas, N.; Hussain, A.; Kucuk, I.; Chang, M.-W.; et al. Engineering and characterisation of BCG-loaded polymeric microneedles. J. Drug Target. 2020, 28, 525–532. [Google Scholar] [CrossRef]
- Xue, P.; Zhang, X.; Chuah, Y.J.; Wu, Y.; Kang, Y. Flexible PEGDA-based microneedle patches with detachable PVP–CD arrowheads for transdermal drug delivery. RSC Adv. 2015, 5, 75204–75209. [Google Scholar] [CrossRef]
- Dabholkar, N.; Gorantla, S.; Waghule, T.; Rapalli, V.K.; Kothuru, A.; Goel, S.; Singhvi, G. Biodegradable microneedles fabricated with carbohydrates and proteins: Revolutionary approach for transdermal drug delivery. Int. J. Biol. Macromol. 2021, 170, 602–621. [Google Scholar] [CrossRef]
- Escobar-Chávez, J.J.; Bonilla-Martínez, D.; Angélica, M.; Molina-Trinidad, E.; Casas-Alancaster, N.; Revilla-Vázquez, A.L. Microneedles: A valuable physical enhancer to increase transdermal drug delivery. J. Clin. Pharmacol. 2011, 51, 964–977. [Google Scholar] [CrossRef]
- Matt, C.; Hess, T.; Benlian, A. Digital transformation strategies. Bus. Inf. Syst. Eng. 2015, 57, 339–343. [Google Scholar] [CrossRef]
- Lu, S.; Cao, G.; Zheng, H.; Li, D.; Shi, M.; Qi, J. Simulation and experiment on droplet formation and separation for needle-type micro-liquid jetting dispenser. Micromachines 2018, 9, 330. [Google Scholar] [CrossRef]
- Won, Y.-W.; Patel, A.N.; Bull, D.A. Cell surface engineering to enhance mesenchymal stem cell migration toward an SDF-1 gradient. Biomaterials 2014, 35, 5627–5635. [Google Scholar] [CrossRef] [PubMed]
- Han, M.; Hyun, D.; Park, H.; Lee, S.; Kim, C.; Kim, C. A novel fabrication process for out-of-plane microneedle sheets of biocompatible polymer. Micromech. Microeng. 2007, 17, 1184. [Google Scholar] [CrossRef]
- McGrath, M.G.; Vucen, S.; Vrdoljak, A.; Kelly, A.; O’Mahony, C.; Crean, A.M.; Moore, A. Production of dissolvable microneedles using an atomised spray process: Effect of microneedle composition on skin penetration. Eur. J. Pharm. Biopharm. 2014, 86, 200–211. [Google Scholar] [CrossRef]
- Ling, M.-H.; Chen, M.-C. Dissolving polymer microneedle patches for rapid and efficient transdermal delivery of insulin to diabetic rats. Acta Biomater. 2013, 9, 8952–8961. [Google Scholar] [CrossRef]
- Dewia, H.A.; Fangben Mengb, B.S.; Guoa, C.; Norlingc, B.; Chenb, X.; Lima, S. Investigation of electron transfer from isolated spinach thylakoids to indium tin oxide. RSC Adv. 2014, 4, 48815–48820. [Google Scholar]
- Yang, S.; Feng, Y.; Zhang, L.; Chen, N.; Yuan, W.; Jin, T. A scalable fabrication process of polymer microneedles. Int. J. Nanomed. 2012, 7, 1415–1422. [Google Scholar]
- Sabbagh, F.; Kim, B.-S. Ex Vivo Transdermal Delivery of Nicotinamide Mononucleotide Using Polyvinyl Alcohol Microneedles. Polymers 2023, 15, 2031. [Google Scholar] [CrossRef] [PubMed]
- Gittard, S.D.; Ovsianikov, A.; Monteiro-Riviere, N.A.; Lusk, J.; Morel, P.; Minghetti, P.; Lenardi, C.; Chichkov, B.N.; Narayan, R.J. Fabrication of polymer microneedles using a two-photon polymerization and micromolding process. J. Diabetes Sci. Technol. 2009, 3, 304–311. [Google Scholar] [CrossRef]
- JEmer, J.; Waldorf, H. Nonhyaluronic Acid. Aesthetic Dermatol. Curr. Perspect. 2018, 29. [Google Scholar]
- Ito, Y.; Kobuchi, S.; Inoue, G.; Kakumu, E.; Aoki, M.; Sakaeda, T.; Takada, K. Dissolving microneedles for enhanced local delivery of capsaicin to rat skin tissue. J. Drug Target. 2017, 25, 420–424. [Google Scholar] [CrossRef]
- Liu, Y.; Eng, P.F.; Guy, O.J.; Roberts, K.; Ashraf, H.; Knight, N. Advanced deep reactive-ion etching technology for hollow microneedles for transdermal blood sampling and drug delivery. IET Nanobiotechnol. 2013, 7, 59–62. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.-C.; Ling, M.-H.; Lai, K.-Y.; Pramudityo, E. Chitosan microneedle patches for sustained transdermal delivery of macromolecules. Biomacromolecules 2012, 13, 4022–4031. [Google Scholar] [CrossRef]
- Nagarkar, R.; Singh, M.; Nguyen, H.X.; Jonnalagadda, S. A review of recent advances in microneedle technology for transdermal drug delivery. J. Drug Deliv. Sci. Technol. 2020, 59, 101923. [Google Scholar] [CrossRef]
- Lee, M.-T.; Lee, I.-C.; Tsai, S.-W.; Chen, C.-H.; Wu, M.-H.; Juang, Y.-J. Spin coating of polymer solution on polydimethylsiloxane mold for fabrication of microneedle patch. J. Taiwan Inst. Chem. Eng. 2017, 70, 42–48. [Google Scholar] [CrossRef]
- Kim, D.; Cao, Y.; Mariappan, D.; Bono, M.S., Jr.; Hart, A.J.; Marelli, B. A microneedle technology for sampling and sensing bacteria in the food supply chain. Adv. Funct. Mater. 2021, 31, 2005370. [Google Scholar] [CrossRef]
- Juster, H.; van der Aar, B.; de Brouwer, H. A review on microfabrication of thermoplastic polymer-based microneedle arrays. Polym. Eng. Sci. 2019, 59, 877–890. [Google Scholar] [CrossRef]
- Gao, Y.; Hou, M.; Yang, R.; Zhang, L.; Xu, Z.; Kang, Y.; Xue, P. Transdermal delivery of therapeutics through dissolvable gelatin/sucrose films coated on PEGDA microneedle arrays with improved skin permeability. J. Mater. Chem. B 2019, 7, 7515–7524. [Google Scholar] [CrossRef]
- Jin, J.; Reese, V.; Coler, R.; Carter, D.; Rolandi, M. Chitin Microneedles for an Easy-to-Use Tuberculosis Skin Test. Adv. Healthc. Mater. 2014, 3, 349–353. [Google Scholar] [CrossRef]
- Vora, L.K.; Courtenay, A.J.; Tekko, I.A.; Larrañeta, E.; Donnelly, R.F. Pullulan-based dissolving microneedle arrays for enhanced transdermal delivery of small and large biomolecules. Int. J. Biol. Macromol. 2020, 146, 290–298. [Google Scholar] [CrossRef]
- Available online: https://www.google.co.in/search?q=https%3A%2F%2Fwww.fda.gov%2Fmedia%2F107708&sxsrf=AJOqlzW0n0UDrfJpR3-O2EM2kSubcn348g%3A1678378634552&source=hp&ei=igYKZLb-HpaYseMP7tme4AM&iflsig=AK50M_UAAAAAZAoUmkCCY0_nlhfFzTSYHEJjGlT-_S9p&ved=0ahUKEwi267vSn8_9AhUWTGwGHe6sBzwQ4dUDCAg&uact=5&oq=https%3A%2F%2Fwww.fda.gov%2Fmedia%2F107708&gs_lcp=Cgdnd3Mtd2l6EAM6BwgjEOoCECdQpgZYpgZgqhBoAXAAeACAAakBiAGpAZIBAzAuMZgBAKABAqABAbABCg&sclient=gws-wiz (accessed on 9 March 2023).
- U.S. Food & Drug Adeministration. 510 (k) Devices Cleared in 2022. FDA 2023. Available online: https://www.fda.gov/medical-devices/510k-clearances/510k-devices-cleared-2023 (accessed on 1 April 2023).
- Yadav, P.R.; Munni, M.N.; Campbell, L.; Mostofa, G.; Dobson, L.; Shittu, M.; Pattanayek, S.K.; Uddin, M.J.; Das, D.B. Translation of polymeric microneedles for treatment of human diseases: Recent trends, Progress, and Challenges. Pharmaceutics 2021, 13, 1132. [Google Scholar] [CrossRef]
- Pulse Biosciences, Inc. Prospective, Open Label, Multi-Center, Non-Significant Risk Study of Nano-Pulse StimulationTM (NPSTM) Technology in Healthy Adults with Sebaceous Hyperplasia. 2020. Available online: clinicaltrials.gov (accessed on 21 February 2021).
- Yonsei University. A Study on the Effectiveness and Safety Evaluation of Combination Therapy with 1927 nm Thulium Laser and Fractional Microneedle Radiofrequency Equipment for Improvement of Skin Aging. 2020. Available online: clinicaltrials.gov (accessed on 21 February 2021).
- Zosano Pharma Corporation. Randomized, Double-Blind, Multi-Center, Parallel-Group, Dose-Ranging Comparison of the Safety and Efficacy of the ZP-Zolmitriptan Intracutaneous Microneedle Systems to Placebo for the Acute Treatment of Migraine. 2018. Available online: clinicaltrials.gov (accessed on 21 February 2021).
- University of California, Davis. The Use of Microneedles With Topical Botulinum Toxin for Treatment of Palmar Hyperhidrosis. 2017. Available online: clinicaltrials.gov (accessed on 21 February 2021).
- Prausnitz, M. A Phase I Study of the Safety, Reactogenicity, Acceptability and Immunogenicity of Inactivated Influenza Vaccine Delivered by Microneedle Patch or by Hypodermic Needle. 2019. Available online: clinicaltrials.gov (accessed on 21 February 2021).
- Radius Health, Inc. A Randomized, Double-Blind, Placebo-Controlled, Phase 2 Study of BA058 Administered Via a Coated Transdermal Microarray Delivery System (BA058 Transdermal) in Healthy Postmenopausal Women With Osteoporosis. 2020. Available online: clinicaltrials.gov (accessed on 21 February 2021).
- University of British Columbia. Controlled Comparison in Canadian Seniors of Seasonal Influenza Vaccines for 2011–2012. 2015. Available online: clinicaltrials.gov (accessed on 21 February 2021).
- Felner, E. Insulin Delivery Using Microneedles in Type 1 Diabetes. 2013. Available online: clinicaltrials.gov (accessed on 21 February 2021).
- Available online: https://www.google.co.in/search?q=https%3A%2F%2Fmembers.wto.org%2Fcrnattachments%2F2018%2FTBT%2FUSA%2F18&sxsrf=AJOqlzX9CZBzGBT7QS6wz2is2N1zZTlmdg%3A1678379222786&source=hp&ei=1ggKZO2jLZfr-QaWu5joCw&iflsig=AK50M_UAAAAAZAoW5q2YHj6V_wBM8woXfxGBuLbcRHoi&ved=0ahUKEwjt5vrqoc_9AhWXdd4KHZYdBr0Q4dUDCAg&uact=5&oq=https%3A%2F%2Fmembers.wto.org%2Fcrnattachments%2F2018%2FTBT%2FUSA%2F18&gs_lcp=Cgdnd3Mtd2l6EAMyBwgjEOoCECcyBwgjEOoCECcyBwgjEOoCECcyBwgjEOoCECcyBwgjEOoCECcyBwgjEOoCECcyBwgjEOoCECcyBwgjEOoCECcyBwgjEOoCECcyBwgjEOoCECdQ7wpY7wpg_gpoAXAAeACAAQCIAQCSAQCYAQCgAQKgAQGwAQo&sclient=gws-wiz (accessed on 9 March 2023).
- Chapter 1-Scope of the Regulations. Available online: https://www.gov.uk/government/consultations/consultation-on-the-future-regulation-of-medical-devices-in-the-united-kingdom/outcome/chapter-1-scope-of-the-regulations (accessed on 9 March 2023).
- Falo, J.L.D.; Erdos, G. Bioactive Components Conjugated to Dissolvable Substrates of Microneedle Arrays. U.S. Patent 10737083B2, 11 August 2020. Available online: https://patents.google.com/patent/US10737083B2/en?oq=US10737083 (accessed on 9 March 2023).
- Kaspar, R.L.; Speaker, T. Microneedle Arrays Formed from Polymer Films. U.S. Patent 10377062B2, 13 August 2019. Available online: https://patents.google.com/patent/US10377062B2/en?oq=US10377062 (accessed on 9 March 2023).
- Chiou, J.-C.; Hung, C.-C.; Chang, C.-W. Method for Fabricating Microneedle Array and Method for Fabricating Embossing Mold of Microneedle Array. U.S. Patent 7429333B2, 30 September 2008. Available online: https://patents.google.com/patent/US7429333B2/en?oq=US7429333 (accessed on 9 March 2023).
- Jin, T. Fabrication Process of Phase-Transition Microneedle Patch. U.S. Patent 10195410B2, 5 February 2019. Available online: https://patents.google.com/patent/US10195410B2/en?oq=US10195410 (accessed on 9 March 2023).
- Ghartey-Tagoe, E.; Wendorf, J.; Williams, S.; Singh, P.; Worsham, R.W.; Trautman, J.C.; Bayramov, D.; Bowers, D.L.; Klemm, A.R.; Klemm, S.R.; et al. Method of Vaccine Delivery via Microneedle Arrays. U.S. Patent 9498524B2, 22 November 2016. Available online: https://patents.google.com/patent/US9498524B2/en?oq=US9498524 (accessed on 9 March 2023).
- Park, J.-H.; Prausnitz, M.R. Microneedle Devices and Production Thereof. U.S. Patent 9302903B2, 5 April 2016. Available online: https://patents.google.com/patent/US9302903B2/en?oq=US9302903 (accessed on 9 March 2023).
- Allen, M.G.; Prausnitz, M.R.; McAllister, D.V.; Cros, F.P.M. Microneedle Devices and Methods of Manufacture and Use Thereof. U.S. Patent 8708966B2, 29 April 2014. Available online: https://patents.google.com/patent/US8708966B2/en?oq=US8708966 (accessed on 9 March 2023).
- Falo, L.D., Jr.; Erdos, G.; Ozdoganlar, O.B. Dissolvable Microneedle Arrays for Transdermal Delivery to Human Skin. U.S. Patent 8834423B2, 16 September 2014. Available online: https://patents.google.com/patent/US8834423B2/en?oq=US8834423 (accessed on 9 March 2023).
- Kato, H. Microneedle and Method for Manufacturing Microneedle. U.S. Patent 10682504B2, 16 June 2020. Available online: https://patents.google.com/patent/US10682504B2/en?oq=US10682504 (accessed on 9 March 2023).
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).