1. Introduction
Depressive disorders are some of the most common and disabling psychiatric conditions worldwide. Epidemiological studies have shown an increase in the degree of depression incidence for several decades, which has been linked to the growth and aging of the general population. According to the World Health Organization report, the number of people suffering from depression is increasing dramatically every year. With over 350 million patients diagnosed with depression worldwide, the economic burden has become considerable for both developed and developing countries [
1,
2].
Antidepressants remain the main therapy for depressive disorders, which are globally available. The various antidepressants target certain neurotransmitters, such as serotonin, noradrenaline, and dopamine, and work via slightly different pathways. These drugs are divided into several classes according to their mechanism of action [
2]. The first registered antidepressant was a drug from the group of monoamine oxidase inhibitors (MAO), whose mechanism of action was the oxidative deamination of biogenic and sympathomimetic amines. Around the same time, drugs called tricyclic antidepressants (TCA) were registered. These drugs owe their name to their three-benzene ring molecular core. They have a diverse pharmacological profile, with significant pharmacological action at three receptor proteins and two reuptake transporters. Some time later, selective serotonin reuptake inhibitors (SSRI), serotonin-norepinephrine reuptake inhibitors (SNRI), and atypical antidepressants were also discovered. Currently, clinicians have a huge arsenal of antidepressants with different mechanisms of action [
3].
Despite this, up to 60% of major depressive disorder patients do not respond sufficiently to the initial antidepressant treatment [
4].
Therefore, the search and study of substances with potential antidepressant activity remain urgent tasks [
5]. Recently, it is often proposed to use alternative drugs, previously developed for other purposes, for depression treatment. The examples of such therapy applications are the use of ketamine and esketamine [
6], as well as anti-inflammatory drugs (cytokine inhibitors and pioglitazone) [
7].
At the same time, much less attention has been given to studying the possibilities of neuropeptides as drugs with probable antidepressant activity. A promising agent for these purposes is neuropeptide Y (NY), which is widely distributed in the central nervous system, including the hippocampus and cerebral cortex, areas involved in the formation of affective symptoms [
8]. The effects of NY are mediated by at least four G protein-coupled receptors (Y1R, Y2R, Y4R, and Y5R) [
9]. NY performs multiple functions, including the regulation of nutrition and satiety, learning and memory, circadian rhythm, neuroprotection, and behavioral responses to stress [
10,
11,
12,
13]. NY is one of the most important regulators mediating resistance to the harmful effects of stress [
9,
10,
11,
14]. It has been hypothesized that NY provides resilience by controlling pro-stress transmitters, such as the corticotropin-releasing hormone and norepinephrine [
14]. It was previously shown that the concentration of NY can change in cerebrospinal fluid and blood during depression and antidepressant therapy [
15,
16]. When neuropeptide Y was injected into the brain structures or intraventricularly, various behavioral effects were observed, such as increased appetite and social activity, decreased anxiety, and antidepressant-like effects [
17].
The intranasal way of drug administration is being actively investigated. This method may be a viable strategy for rapid drug delivery to the central nervous system. It has several advantages, such as noninvasiveness, simplicity of administration, no hepatic elimination, and fewer possible side effects compared to peripheral administration [
12]. Therefore, this method is potentially suitable for different classes of drugs, including antidepressants. Furthermore, our recent study demonstrated that both the intranasal and intraperitoneal administration of clomipramine had similar antidepressant effect. However, clomipramine affected the total metabolome in the frontal cortex and hippocampus differently depending on the way of administration [
18]. Since metabolic processes in the brain can change dissimilarly depending on the different ways of administration, antidepressant therapy should also be evaluated from this perspective. The usage of the intranasal administration of antidepressants needs to be further explored.
Intranasal administration is crucial for peptide delivery to the brain because it represents a pathway that bypasses the blood–brain barrier via the olfactory and trigeminal neural pathways [
19,
20]. Intranasally injected, NY reaches several brain regions, including the hypothalamus, within 30 min in rodents [
21]. Furthermore, there is increasing evidence that the intranasal delivery of NY to the brain has therapeutic potential [
9,
12,
22,
23]. Neuropeptide Y produces no side effects and is well-tolerated when administered intranasally in patients with major depressive disorder [
24].
However, to our knowledge, there are no data regarding the chronic intranasal administration of neuropeptide Y, either in humans or in translational animal models.
The aim of our study was to explore the possibility of using NY, alone and in combination with the antidepressant clomipramine. We chose clomipramine for this study based on our previous experiments on the intranasal/intraperitoneal administration of clomipramine [
18]. We decided that it would be important to investigate the combined effect of an antidepressant with NY, because such research has not been performed before. However, some studies have demonstrated the successful therapeutic activity of combinations of different drugs. For example, the combined administration of orexin 1 receptor antagonist and a non-selective nitric oxide synthase inhibitor was revealed to have an antidepressant-like effect in mice [
25].
We investigated the efficacy of drugs, alone and in combination, in models of acute and chronic stress. Immobilization stress was chosen as a model of acute stress [
26], and unavoidable ultrasonic exposure was used as a chronic stress model. It is assumed that a massive information flow that carries the opposite motivational-emotional load (different US frequencies) can cause stress conditions in rats. Thus, prolonged exposure to ultrasound in animals may induce a depression-like state [
27]. Previous research has demonstrated that the ultrasound-induced depression model is equivalent to the conventional CMS protocol and meets all necessary validity criteria [
28].
2. Materials and Methods
2.1. Animals and Drug Administration
Our experiment was carried out on male Wistar rats from the Nursery of Laboratory Animals (Pushchino, Russian Academy of Sciences, Moscow, Russia). Animals adapted in the laboratory for two weeks before the experiment. At the beginning of the experiment, the age of the animals was 2 months (their weight was between 200–250 g). During the experiment, all animals were kept at a constant temperature (23 °C). Direct lighting was controlled (12/12 h), and the animals were given free access to food and water. Throughout the experiment, the animals were housed individually in transparent polycarbonate cages (42 × 26 × 15 cm) (during stress protocols and after stress during behavioral tests). Containment conditions and all experimental procedures were established and maintained in accordance with the Directive 2010/63/EU of 22 September 2010 and approved by the local ethical committee of VP Serbsky National Medical Research Center for Psychiatry and Narcology.
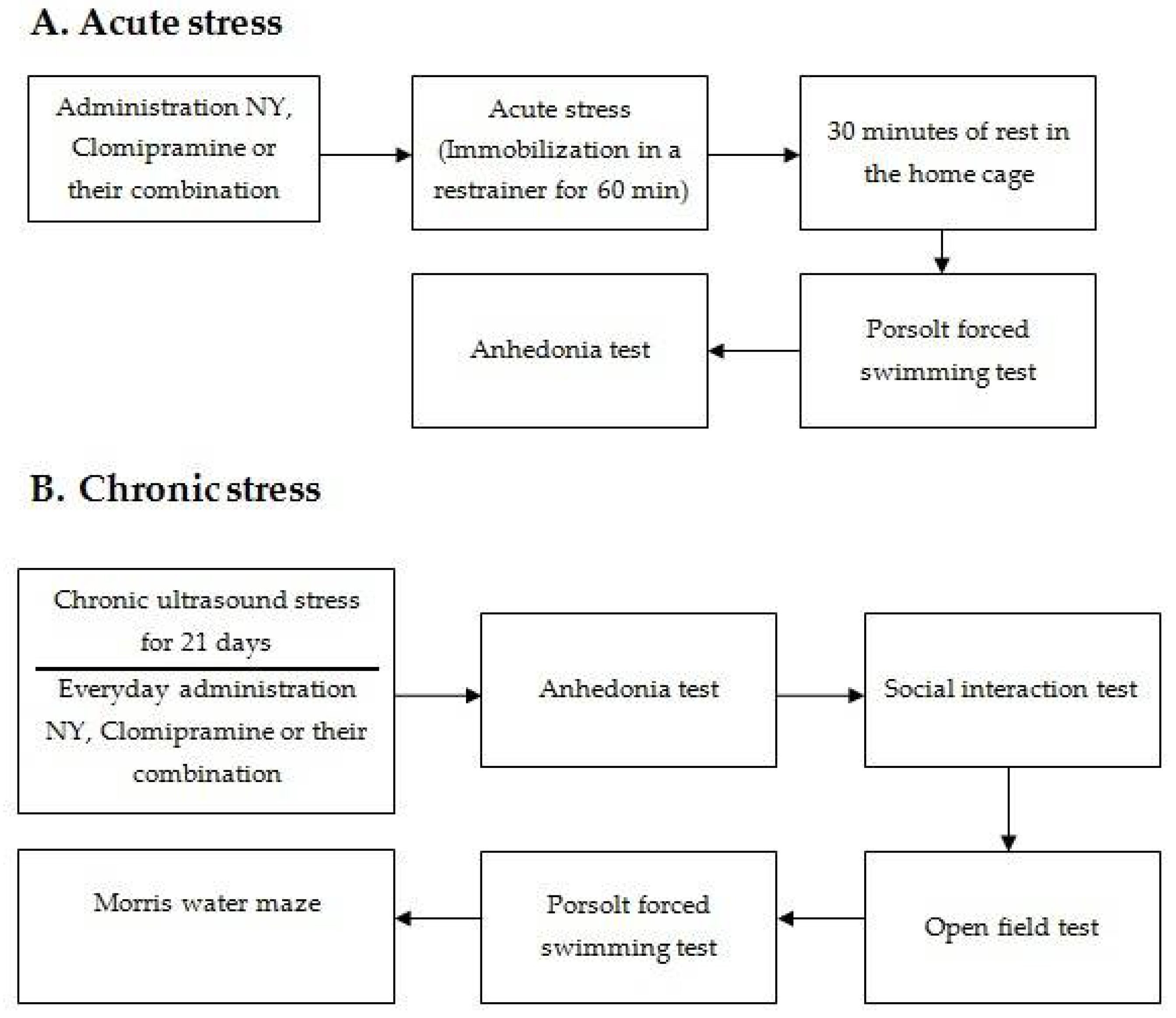
The study included two series of experiments with acute and chronic stress. The schematic of the experiments is presented in
Figure 1.
For the acute stress experiment, the rats were randomly divided into six experimental groups: the control group (n = 10), including intact rats that were acutely intranasally injected with saline; AS (n = 10)—the group with acutely stressed animals injected intranasally with a saline solution; AS + K (n = 10)—the group with acutely stressed animals administrated with clomipramine at a dose of 7.5 mg/kg; AS + NY (n = 10)—the group with acutely stressed animals administrated with NY at a dose of 100 μg/kg; AS + K + NY (n = 10)—the group with acutely stressed animals administrated with clomipramine and NY at similar doses. Immediately before the acute stress, the rats were subjected to a single intranasal injection of the drug or saline. After the acute stress procedure, the rats were rested in their home cage for 30 min. After resting, the rats were tested with a Porsolt forced swimming test, followed by an anhedonia test. Each animal was kept in an individual cage for the entire duration of the experiment.
For the chronic stress experiment, the rats were randomly divided into six experimental groups: the control group (n = 10), including intact rats that were chronically intranasally injected with saline; CS (n = 10)—the group with chronically stressed animals injected intranasally with a saline solution; CS + K (n = 10)—the group with chronic stress and intranasal therapy of clomipramine at a dose of 7.5 mg/kg; CS + NY (n = 10)—the group with chronic stress and intranasal therapy of NY at a dose of 100 μg/kg; CS + NY + K (n = 10)—the group with chronic stress and combined intranasal therapy of NY and clomipramine at similar doses. The rats were placed in individual cages and subjected to a chronic stress procedure for 21 days. During the entire period of chronic stress, the animals were injected daily with the drug or saline. The control animals were kept in individual cages for 21 days until the behavioral tests, with a daily administration of saline. All rats resided in individual cages during all behavioral testing. On the last day of chronic stress, the first behavioral test for anhedonia was conducted. Then, other tests were conducted in the following sequence: a “Social Interaction” test, an open field test, a Porsolt forced swimming test, and Morris water maze. The interval between behavioral tests was at least two days.
The following preparations were used in the experiment: clomipramine (Sigma Aldrich, PO Box 14508 Saint Louis, MO 63178, USA) and NY were synthesized by Almabion (Voronezh 394005, Russia) in the amount of 14 mg. NY was dissolved in physiological saline in an amount of 1400 μL to the concentration of 10 mg/mL. The solution was stored at −25 °C. Clomipramine was dissolved in saline just before the injection. The volume of the intranasal administration of each drug was 100 µL per rat. The single dose was 100 μg/kg for NY and 7.5 mg/kg for clomipramine [
18,
22].
During intranasal administration, the animals were placed in a plastic restrainer, which were held at an angle of approximately 45°. When the nose of the rat was open at the narrowed end of the container and the animal itself froze, the intranasal administration procedure was performed. Antidepressants were administered through 200 μL micropipettes with pipette dispensers. Drops were carefully dripped alternately into both nostrils, while a pause was made for the rat to inhale a drop into the nostril before introducing the next one. The total volume of injected fluid was 30 µL per rat for the drugs and saline. The procedure time per animal was about two minutes.
2.2. Modeling Acute Stress
Stressing was carried out by immobilizing the rats in a restrainer for 1 h. After that, the rat was placed in a cage for 30 min to rest. The rats were kept in individual cages during the stress procedure and during the behavioral testing.
2.3. Modeling Chronic Stress
A depressive-like state was modeled by the chronic continuous exposure of individually housed rats to ultrasound waves of variable frequency. The animals were continuously exposed to ultrasound waves (20–45 kHz) for 21 days, using a Weitech ultrasound generator (Wavre, Belgium). The ultrasound frequency ranges were alternated every 10 min between the following intervals: short frequencies (20–25 kHz), medium frequencies (25–40 kHz), and high frequencies (40–45 kHz). In general, the total duration of ultrasound exposure at short and medium frequencies was 35%, and at high frequencies—30%. Sound pressure was 50 dB ± 5 dB (fluctuations ± 10%) [
29].
2.4. Tests
2.4.1. Open Field Test
This test is designed to study locomotor and exploratory activity under conditions of low stress caused by bright light and open space. The test was carried out in an arena measuring 45 × 45 × 45 cm (MultiCondition (TSE Systems, Bad Homburg, Germany)). The floor of the installation was made of gray plastic, and the walls were made of black plastic under room lighting. The animal was placed in the arena near the wall at the same distance between two corners. The test time was 5 min [
28].
2.4.2. Sucrose Preference Test (Detection of Anhedonia)
In the evening (before the start of the active behavioral phase), the drinker with ordinary water was replaced with a drinker with 2% sucrose solution for 2 h, so that the rat got acquainted with the solution. The next day at 16:00 (before the evening active phase), the animals were given two drinkers—one with the ordinary water, and the other with 1% sucrose solution. The drinkers were changed every 6 h to avoid site preference. After 24 h, the drinkers were removed. The total volume of fluid consumed and the volumes of ordinary and sweetened water drunk were measured, as well as the preference index, which was calculated according to the following formula: (volume of sweet water drunk × 100%)/total volume of liquid consumed. The volumes of the consumed liquid were measured by the difference in the weight of the drinkers with liquids before and after the experiment procedure.
2.4.3. “Social Interaction” Test
To assess the social activity and depression-like state of the experimental animals, a social interaction test was performed. During the test, a 1-month-old juvenile male was placed in the home cage of the tested rat. Within 10 min, the duration and the number of social contacts with the juvenile male, as well as the duration and the number of threatening and aggressive contacts, were recorded.
2.4.4. Porsolt Forced Swimming Test
This test has been proposed as a model for some aspects of human depression, namely the feeling of hopelessness. An animal forced to swim in a confined space quickly stops active attempts to get out and assumes a state of relative immobility. Most clinically effective antidepressants increase the time of active resistance and, consequently, reduce the time of immobility. The test was carried out in a white PVC apparatus (with a diameter of 31 cm and a height of 40 cm) filled with water so that the tail of the experimental animal did not touch the bottom. The water temperature was 24 °C. The animal was placed in the installation for 8 min, and during the last 6 min, the immobility time was recorded. Immobility was considered as the absence of active movements: either when the animal completely froze, drifted along the cylinder without moving its paws or tail, or made weak movements with its hind legs to stay afloat. Due to this test being the most stressful and it could affect the performance of subsequent tests, it was carried out lastly. After the test, the animal was wiped dry and returned to the home cage.
2.4.5. Morris Water Maze
The Morris maze was a pool with gray walls, 150 cm in diameter and 60 cm high, filled with water to a depth of 40 cm. The water temperature was 23 °C. A round plastic platform matching with the color of the apparatus was placed in the center of one of the quadrants of the pool, 2 cm below the water surface. The diameter of the platform was 8 cm. The pool was located in a room with a large number of spatial landmarks. During the training session, the rats were placed in water at 8 different points at approximately the same distance from the platform. After the animal reached the platform, it was left on it for 15 s and then placed in a separate cage for 60 s. The rats that could not find the platform within 60 s were gently guided to it. In each trial, the time required to reach the platform was recorded. After 48 h, long-term memory was assessed in the experimental session.
2.5. Statistical Analysis
Analysis was performed using jamovi and RStudio software. After receiving the data, the distribution was checked for normality using the Shapiro–Wilk test, which designated the choice of statistical comparison criteria: parametric or non-parametric. In the case of a normal distribution, the mean and the standard deviation were considered; in the case of an abnormal distribution, the median and the interquartile range were calculated. Parametric analysis (if distribution was normal) was performed using ANOVA analysis of variance and Tukey’s multiple comparison test for post hoc analysis, adjusted for multiple comparisons. Non-parametric analysis (if distribution was non-normal) was performed using the Kruskal–Wallis test and the Dwass–Steel–Critchlow–Fligner multiple comparison test for post hoc analysis. Differences were considered significant at p < 0.05.
4. Discussion
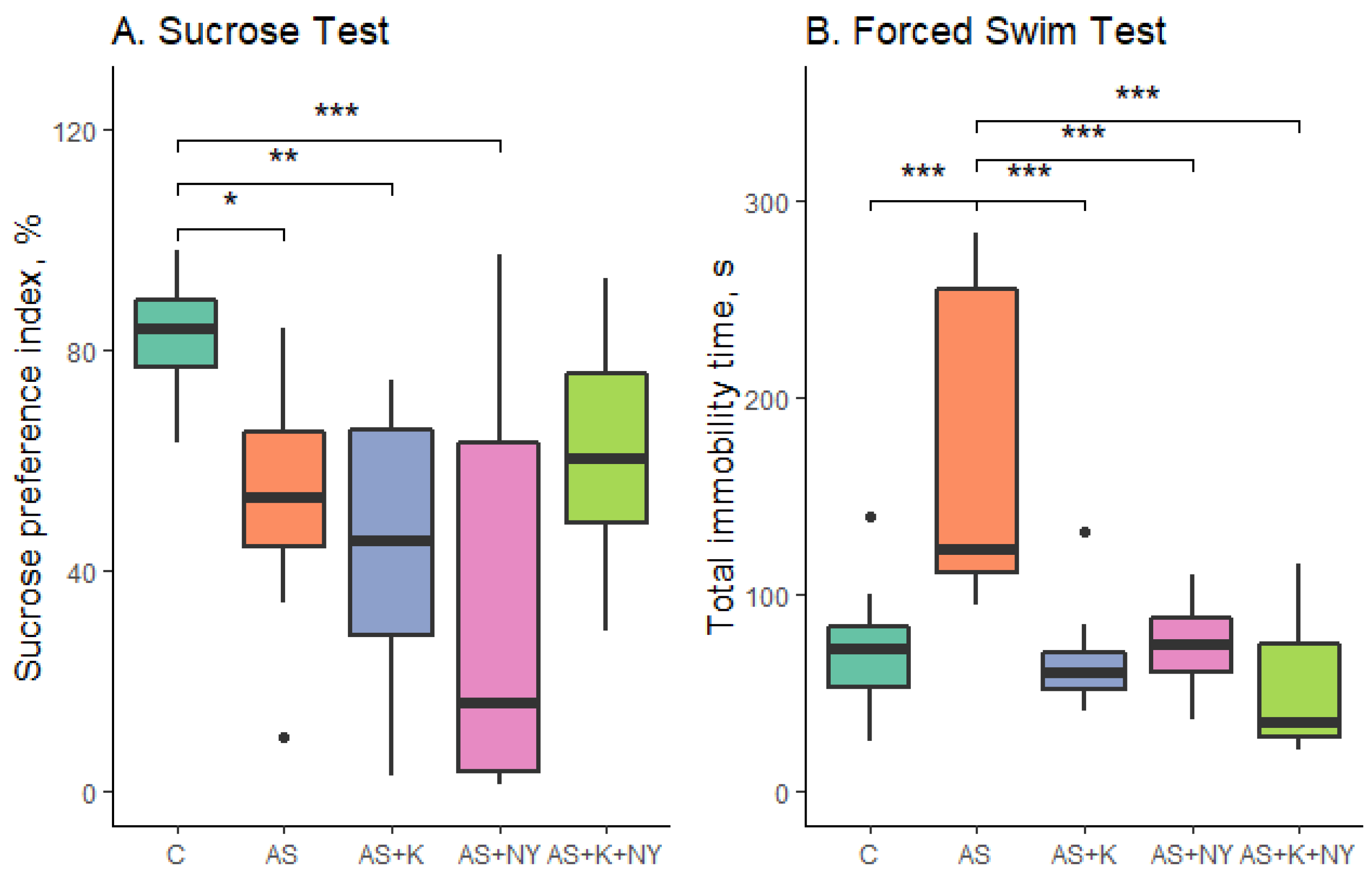
Our research demonstrated that acute immobilization-induced stress resulted in increased anhedonia and increased despair behavior in rats. All drug variants were equally successful in eliminating despair behavior in acutely stressed animals. However, only the combination of NY and clomipramine partially eliminated anhedonia, while these drugs separately were unable to reduce the level of anhedonia. Our acute stress protocol is similar to the single prolonged stress (SPS) protocol used in models of post-traumatic stress disorder (PTSD) in rodents involving prolonged immobilization. There is a fair amount of literature data describing the therapeutic activity of NY in animal models of PTSD. NY eliminated many impairments caused by traumatic stress. The intranasal administration of NY (in a single dose of 100–300 μg) not only prevented, but also reversed, SPS-elicited increases in startle response, anxiety, depressive-like behavior, and hyperarousal symptoms in several studies on rats and mice [
9,
22,
23,
30,
31]. It is also capable of improving increased anxiety, depressive-like behavior, and startle response when administered one week or more after stressful exposure to SPS [
9,
23,
30]. However, the importance of the investigation of the effectiveness of different NY doses was demonstrated, as there is evidence that low single doses of NY (50 μg) may not be effective [
23]. Nevertheless, in our study, a fairly low single dose (100 μg/kg, that is, about 20–25 μg per rat) was used, since it induced a significant effect in eliminating despair behavior in the forced swimming test, on par with the intranasally administered antidepressant. This indicates a lack of information about the effects of different NY doses, and that this needs to be studied further. In addition, our study was conducted on males, while one study showed that a single intranasal dose of NY in an effective concentration for males (300 µg) did not significantly affect the parameters of anxious behavior in the elevated plus maze or depressive behavior in the forced swimming test in females seven days after SPS [
32]. This suggests the need for the investigation of the effective doses of the drugs in different sexes, as there is a lack of such studies. Therefore, our study further demonstrated the possibility of the intranasal application of NY in a model of acute stress in animals, using a relatively small dose of the drug.
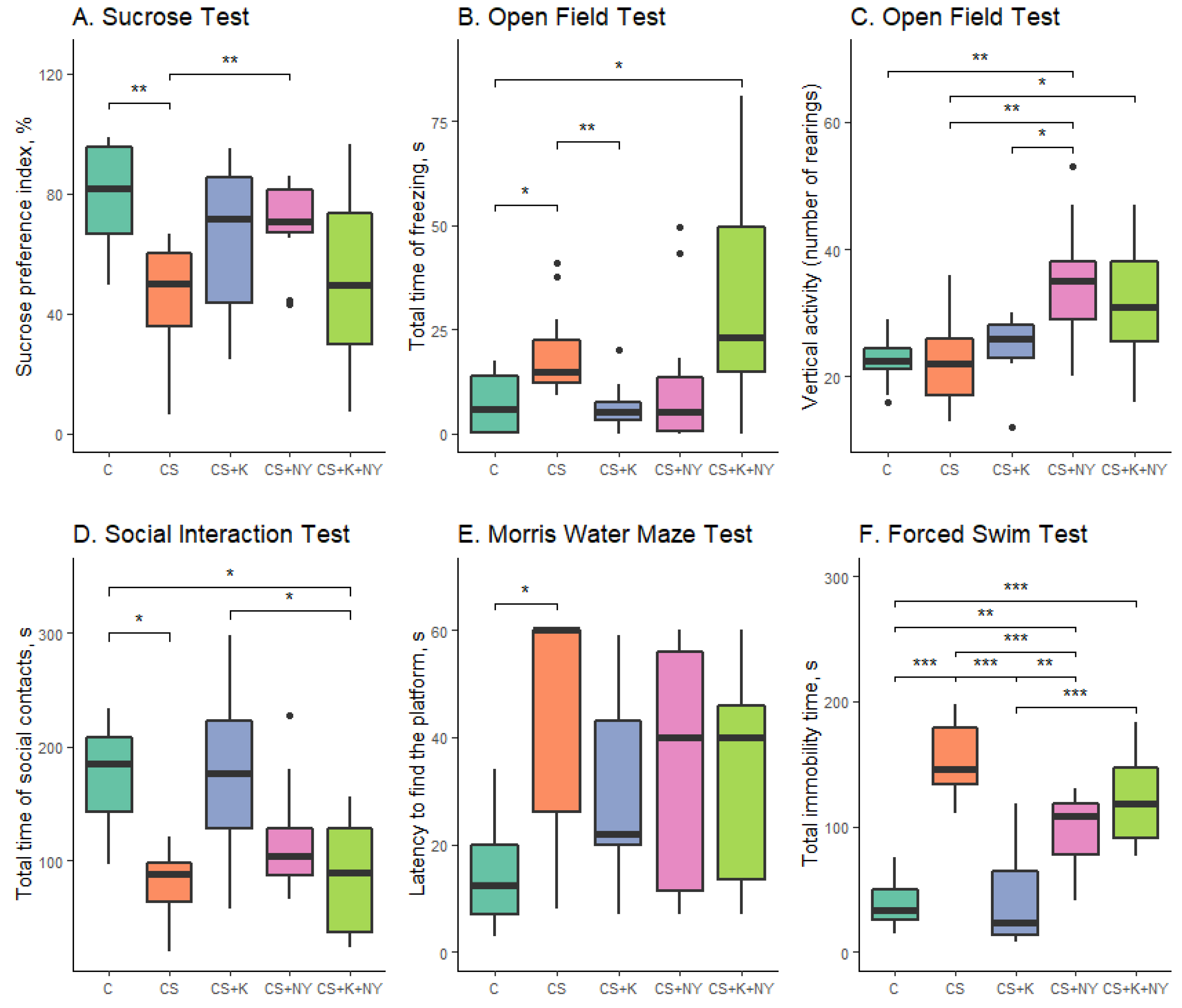
To our knowledge, in our study, the effects of chronically intranasally administered NY in an animal model of chronic stress were investigated for the first time. In our experiment, chronic stress induced by the exposure of variable frequency ultrasound caused depressive-like behavior in rats (increased anhedonia and anxiety, decreased social contact, impaired cognitive functions, and increased despair behavior). The antidepressant clomipramine significantly reduced the level of depressive-like behavior after chronic stress. In particular, its administration reduced anxiety, partially restored the level of social contact, cognitive functions, and the level of anhedonia, and completely eliminated despair behavior. The administration of NY under a chronic stress paradigm partially reduced the symptoms of a depressive-like state. It reduced anhedonia, anxiety behavior, and partially eliminated despair behavior, but did not significantly improve cognitive functions or social behavior in rats with chronic stress. In general, the injection of NY produced a therapeutic effect, but less pronounced than the clomipramine injection. The combination of the antidepressants clomipramine and NY either did not the change measures of depressive-like behavior in rats or, in the case of social behavior and anxiety, produced a negative effect.
The results of our study indicated that chronic NY usage induces some protective antidepressant effects, but less pronounced compared to clomipramine. As it is known that antidepressants have many side effects [
33], the search for drugs devoid of this drawback is an important task. Our study suggests that NY might be effective during therapy for depression, especially since it has been shown that acute intranasal NY administration in people with PTSD [
34] and depression [
24] produces some positive effects. In addition, the evidence for the successful intranasal therapy of transgenic mice with a severe ataxic phenotype was demonstrated. These mice were injected with an NY solution into the nostrils 5 days a week. After 8 weeks of treatment, the alleviation of motor and balance impairments was observed. This was consistent with a decrease in cerebellar pathology [
35]. Nevertheless, the therapeutic potential of NY in chronic stress needs to be further explored, as according to our data, there are no such studies yet.
The combination of clomipramine and NY demonstrated a better effect compared to the drugs administered alone only in the test for anhedonia under acute stress, and even in this case, the combination only partially reduced the level of anhedonia in rats after acute stress. In other conditions, the co-administration had a worse effect than clomipramine and NY alone in the same concentrations. We suggest that other dosage combinations of these drugs should be tested, or another antidepressant should be used in combination with NY and potentially make the combination more effective.
Additionally, a number of limitations of our study must be discussed. Modeling depressive-like behavior in rats has its own limitations when translating results into clinical practice, because basic symptoms, such as low mood and thoughts, cannot be modeled on rats. However, drug development requires animal model research. Depression-like models reproduce only some of the aspects of human depression. Each animal model has strengths and limitations that must be recognized in order to be used effectively in depression research. The acute stress model is suitable for anxiolytics and antidepressants screening, and reflects the response to stress. At the same time, the results cannot be directly used for depression treatment evaluation, but can help for screening out ineffective drugs. The effectiveness of drugs shown to be effective in acute stress should be investigated in other models of depression-like behavior. The US model of chronic stress possesses face, construct, and predictive validity [
28]. The US model replicates and reproduces aspects such as learned helplessness, anhedonia (the rejection of pleasant things), and social withdrawal. An important advantage of our model can be considered the reproduction of cognitive impairment, which also accompanies depression in humans. Our study demonstrated the effects of potential antidepressants on various aspects of a depression-like state. This provides a background for their further investigation for the therapy of depressive symptoms.
Another limitation of our study is the lack of research on female rats. The progression of depression in men and women may have different characteristics and gender-specific pathophysiology [
36]. Additionally, there is evidence that stress may act differently on the behavior of male and female rats [
37]. This is important to consider with the prospect of further studies on the effects of NY and its combination with an antidepressant in humans. As there is a possibility that the drugs may act differently in men and women, it is necessary to address the differences in the progression of the disease. Therefore, in further animal studies, it is necessary to identify sex differences in drug action.