Systematic Review on the Effectiveness of Essential and Carrier Oils as Skin Penetration Enhancers in Pharmaceutical Formulations
Abstract
1. Introduction
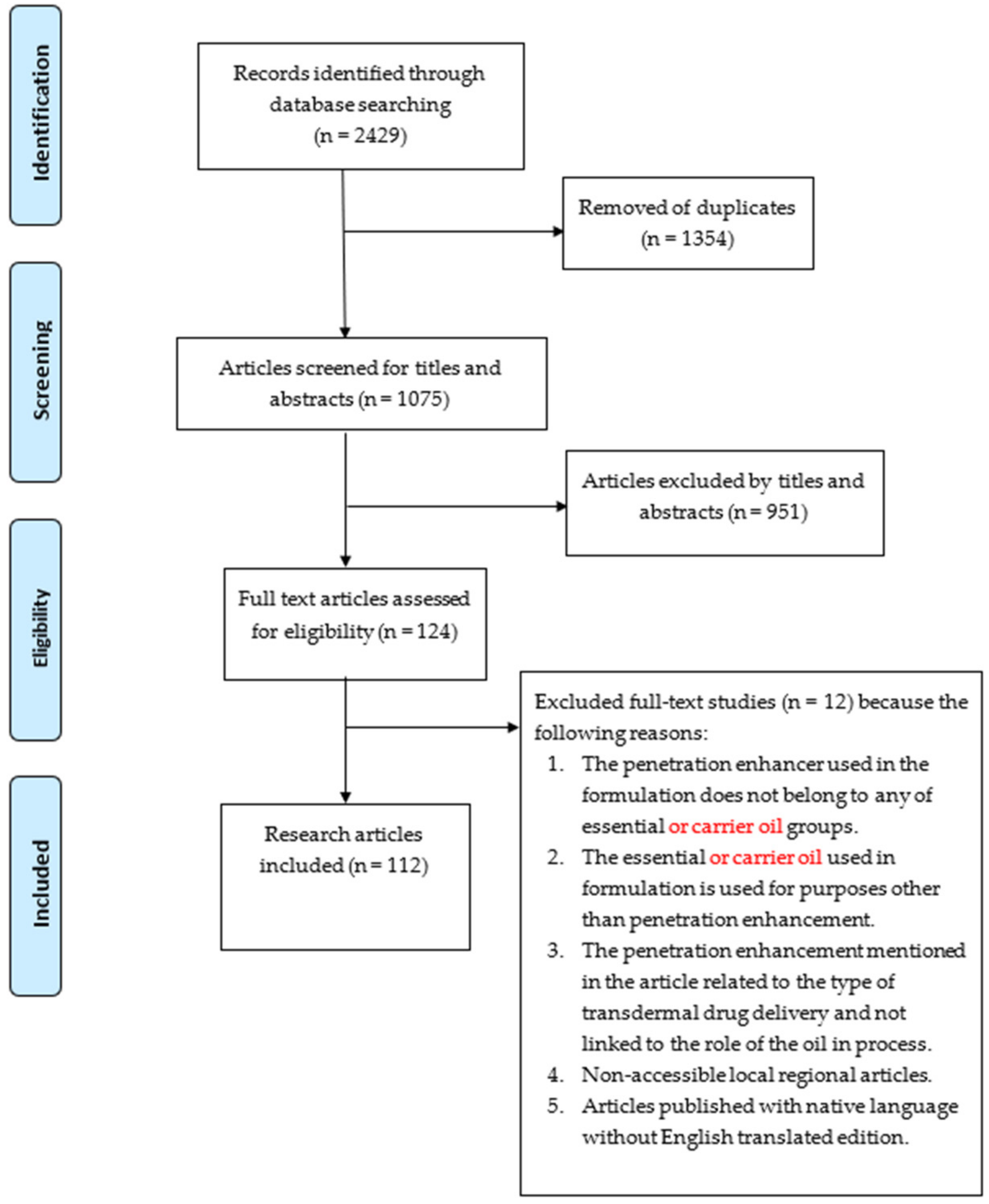
2. Materials and Methods
2.1. Search Strategy
2.2. Study Selection
2.3. Data Extraction
2.4. Quality Assessment
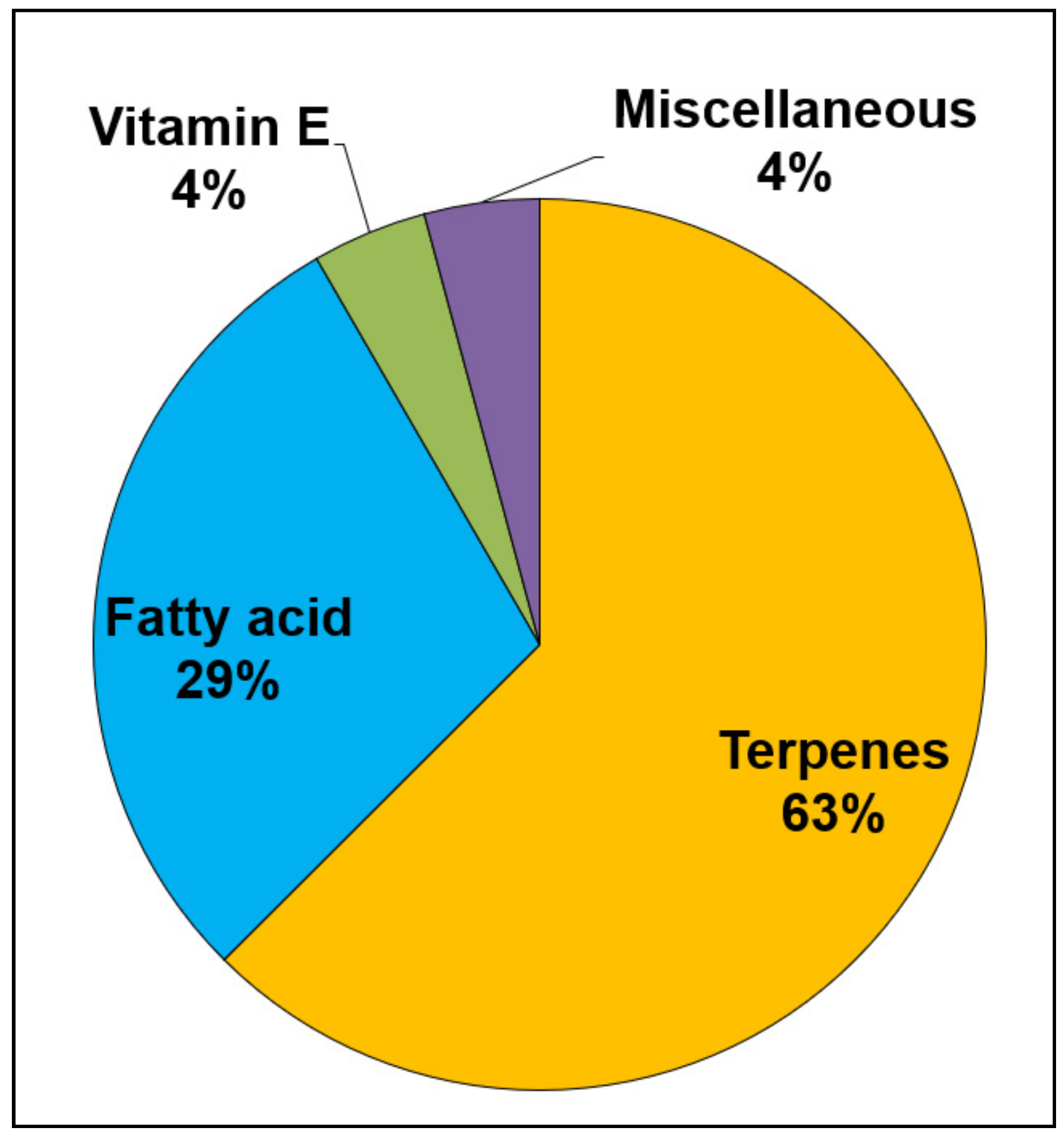
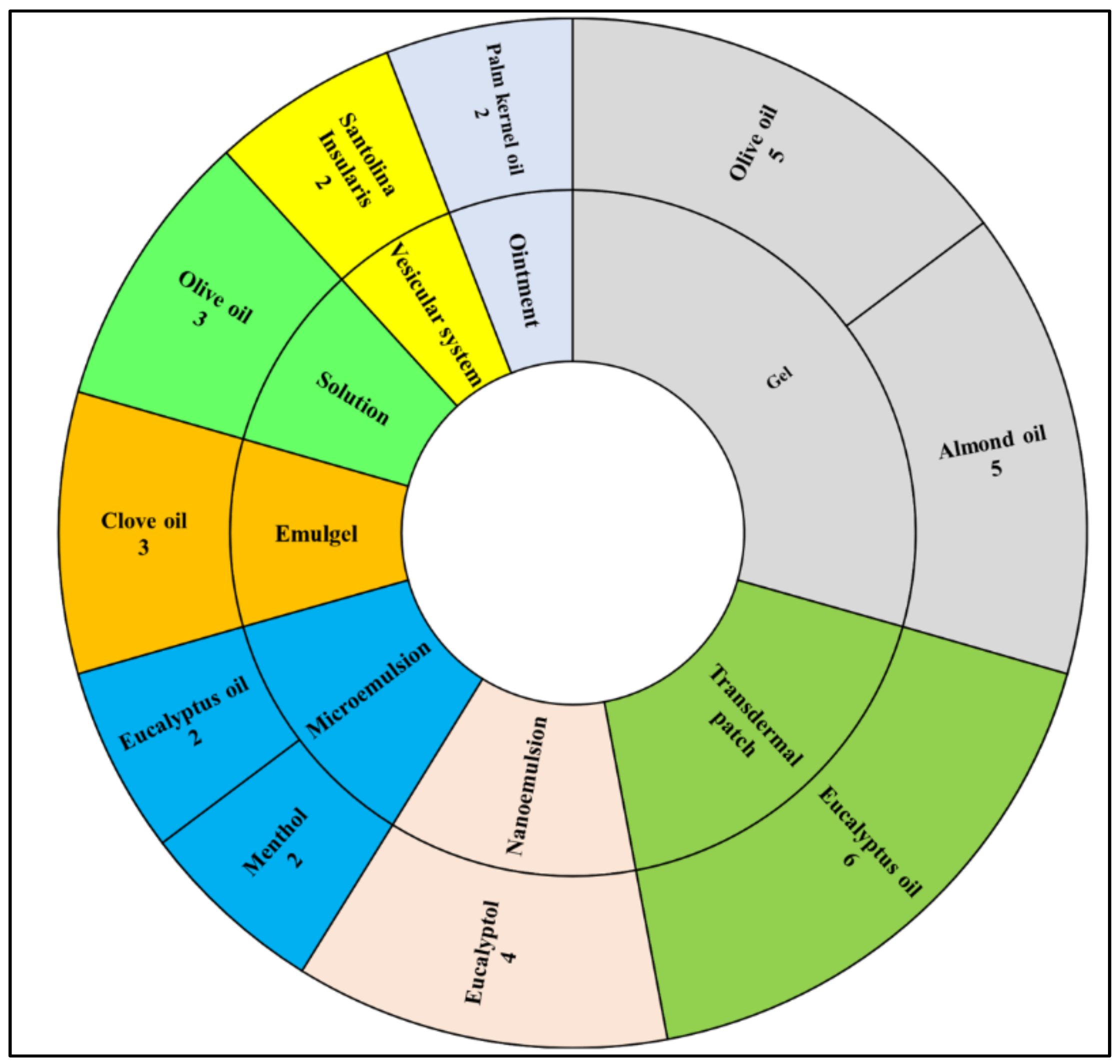
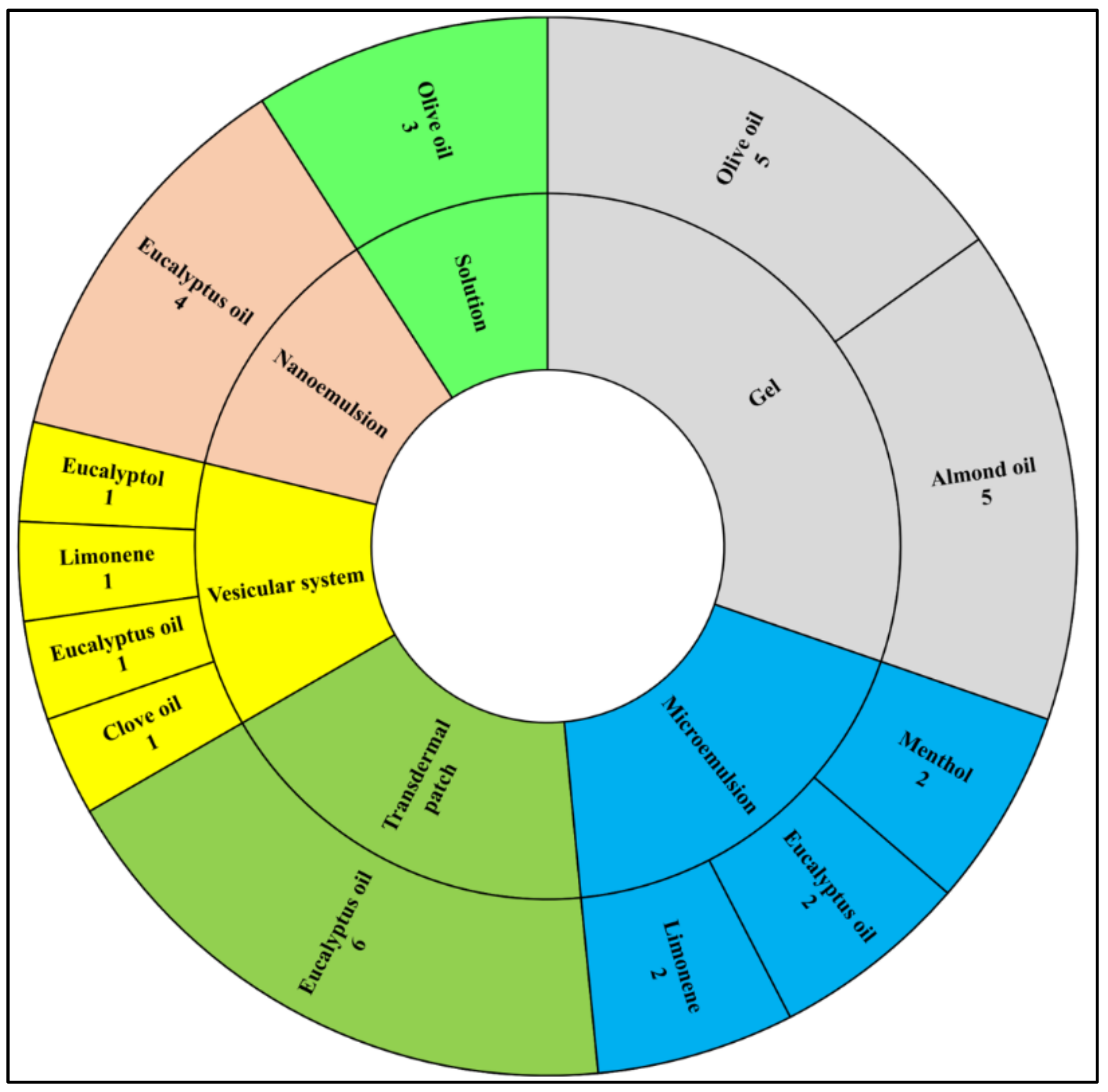
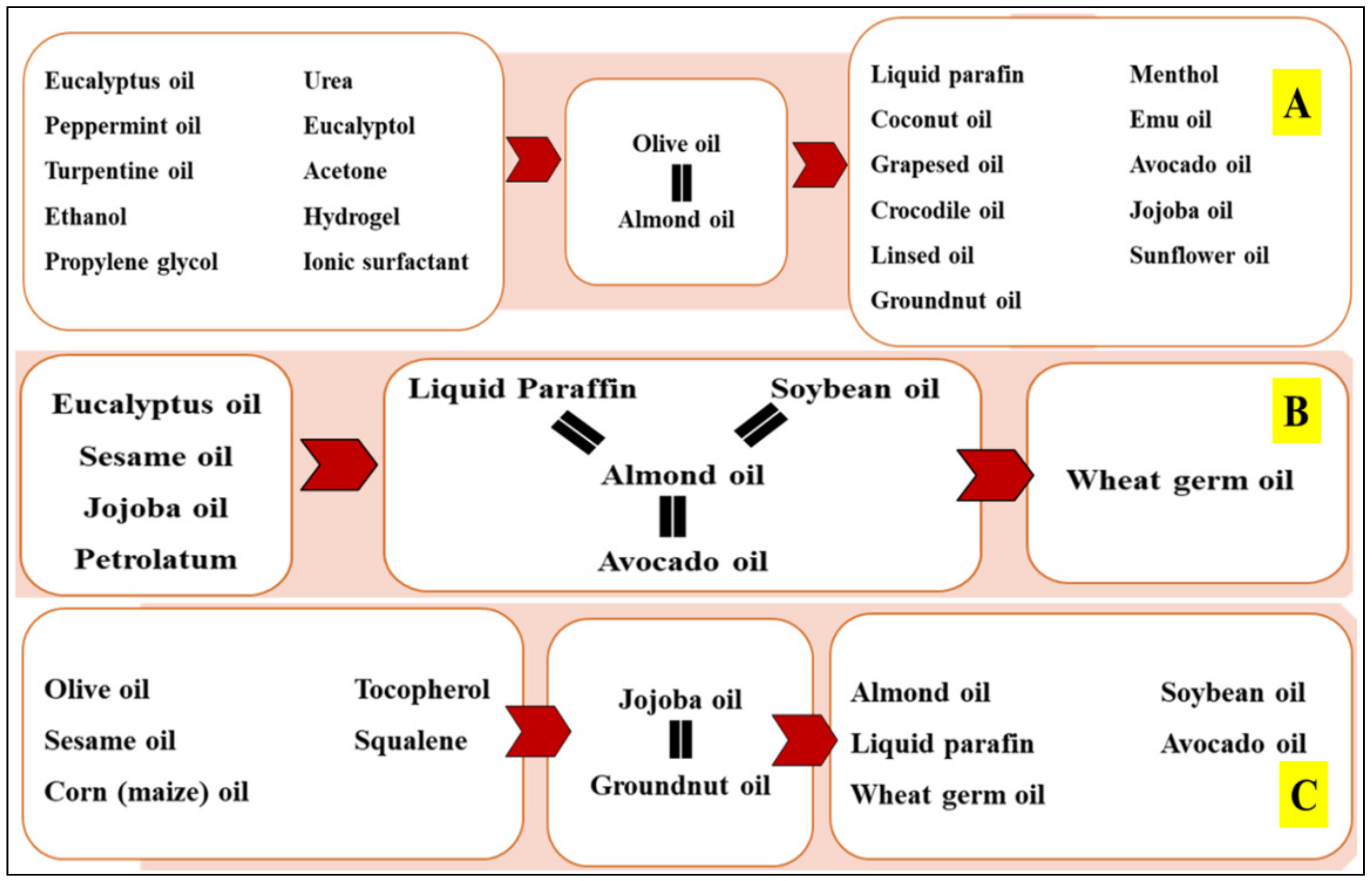
3. Results
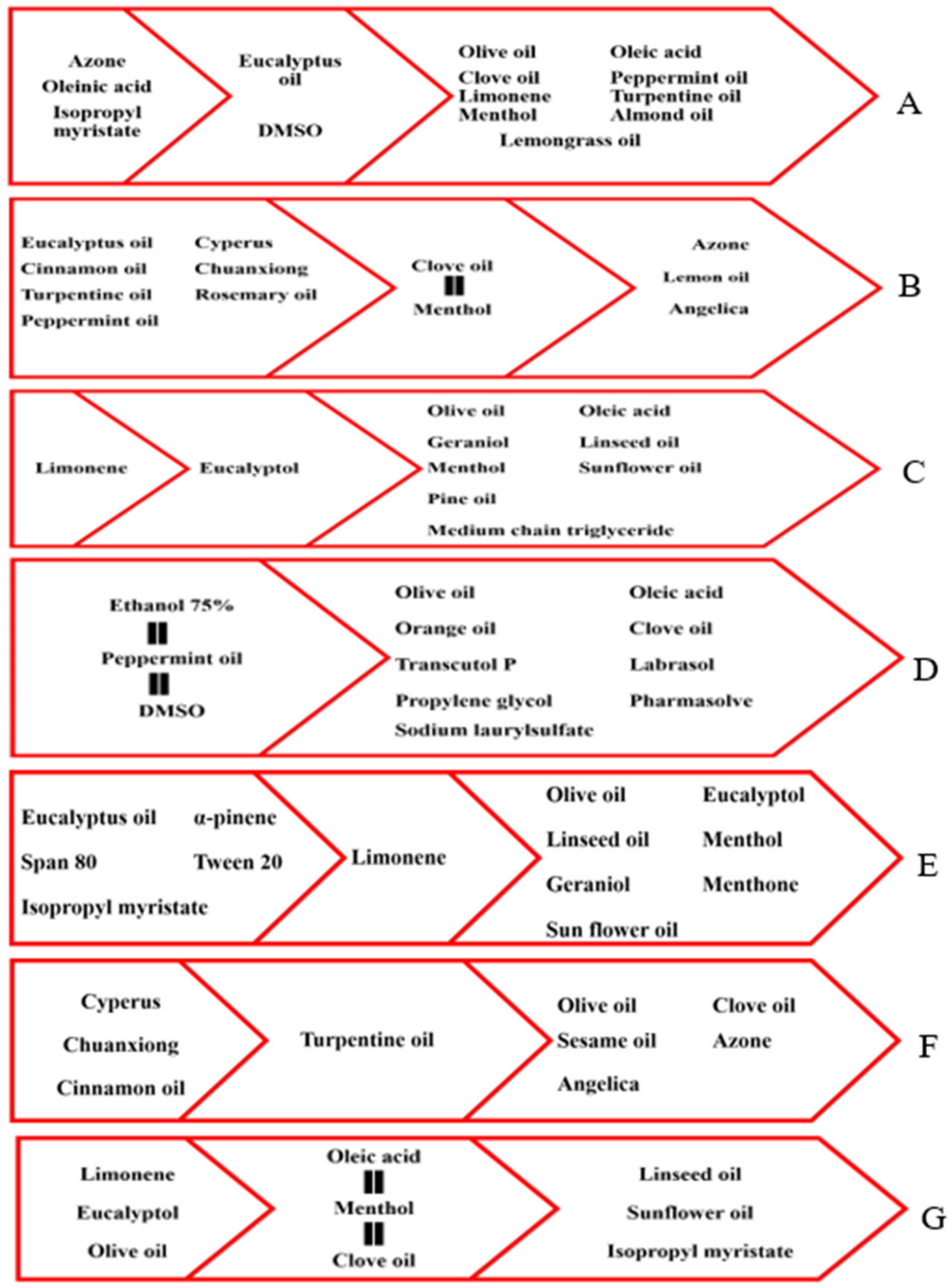
3.1. Terpenes
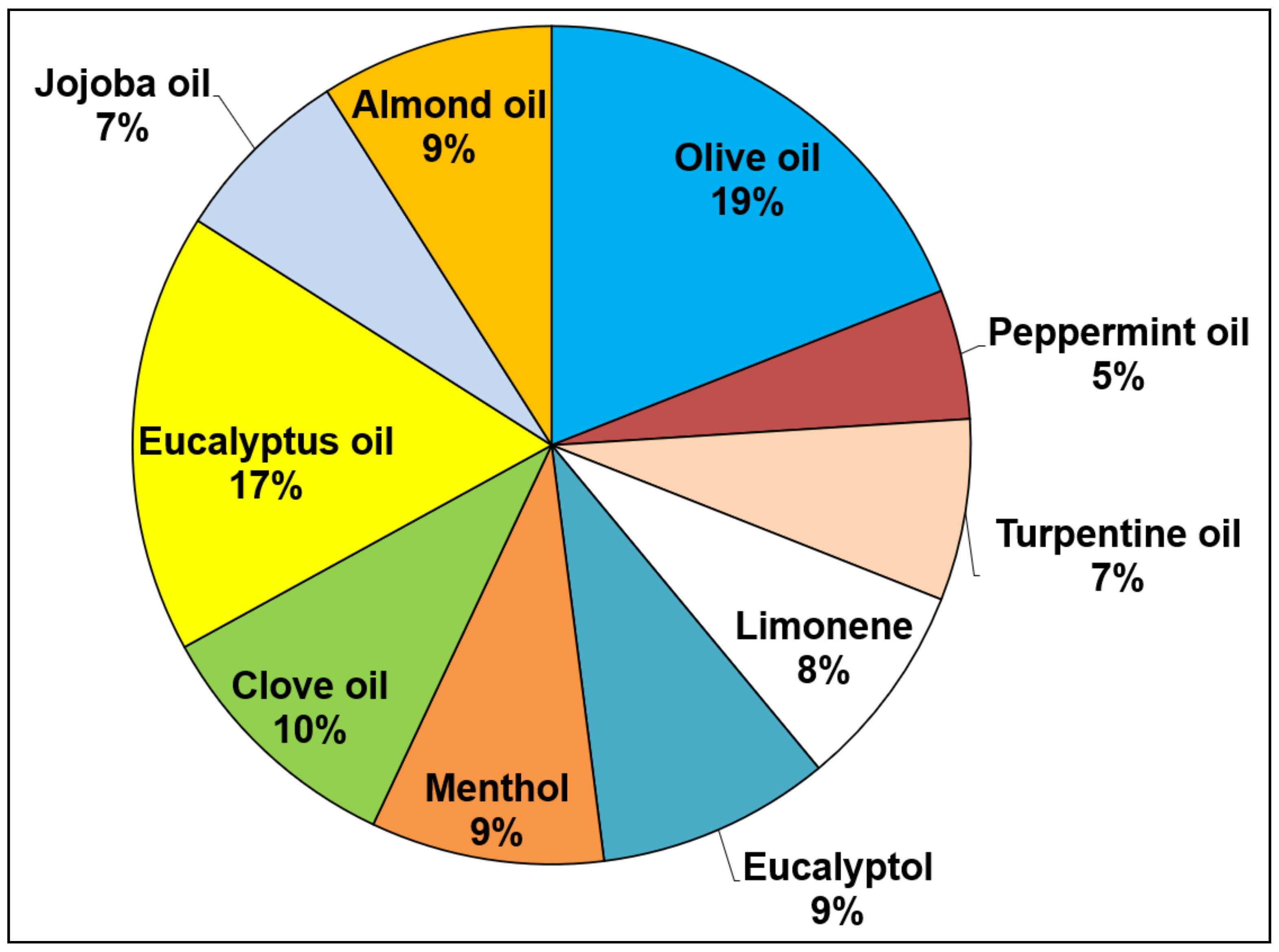
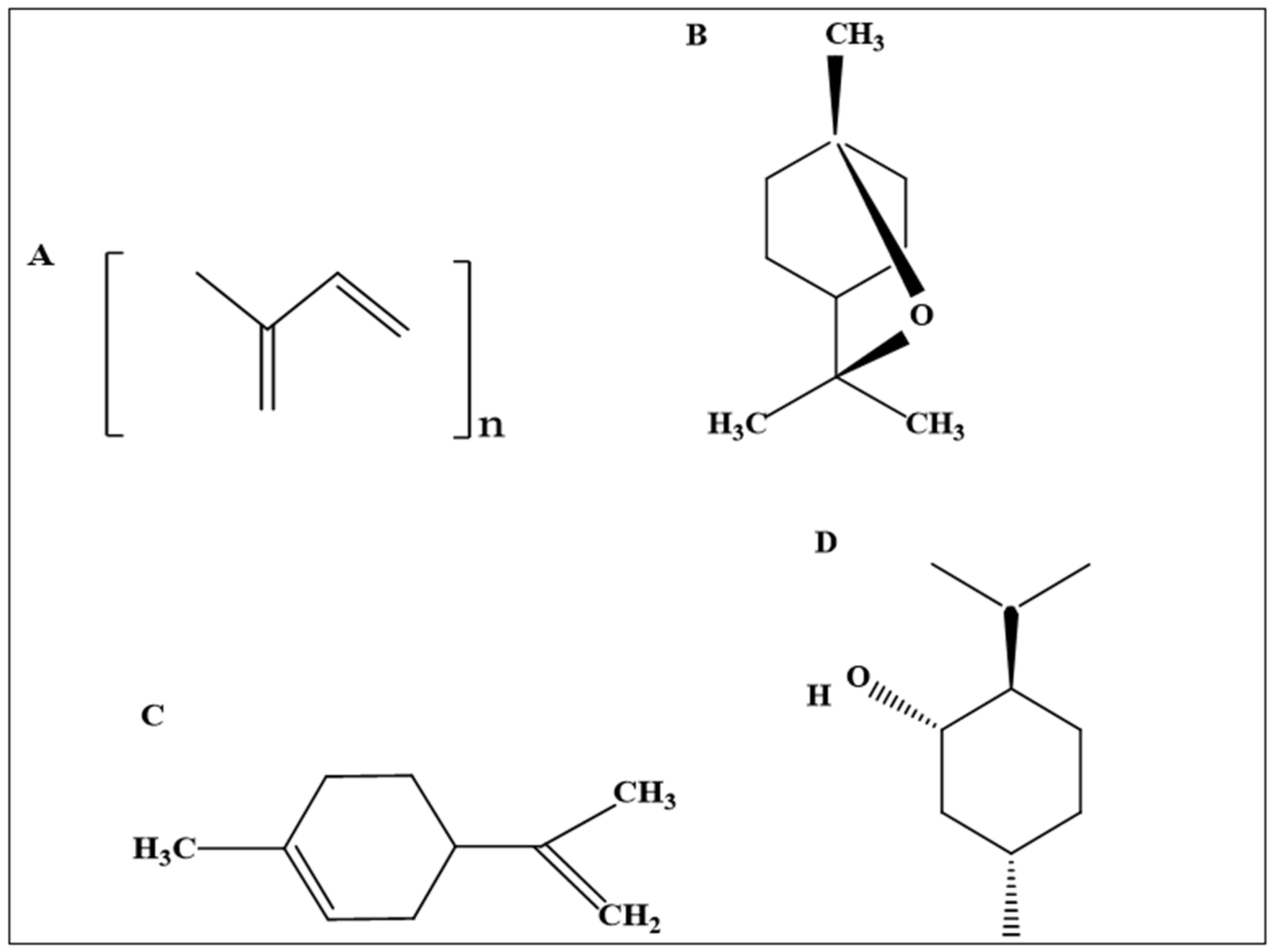
3.1.1. Eucalyptus Oil
3.1.2. Clove Oil
3.1.3. Eucalyptol
3.1.4. Peppermint Oil
Menthol
3.1.5. Limonene
3.1.6. Turpentine Oil
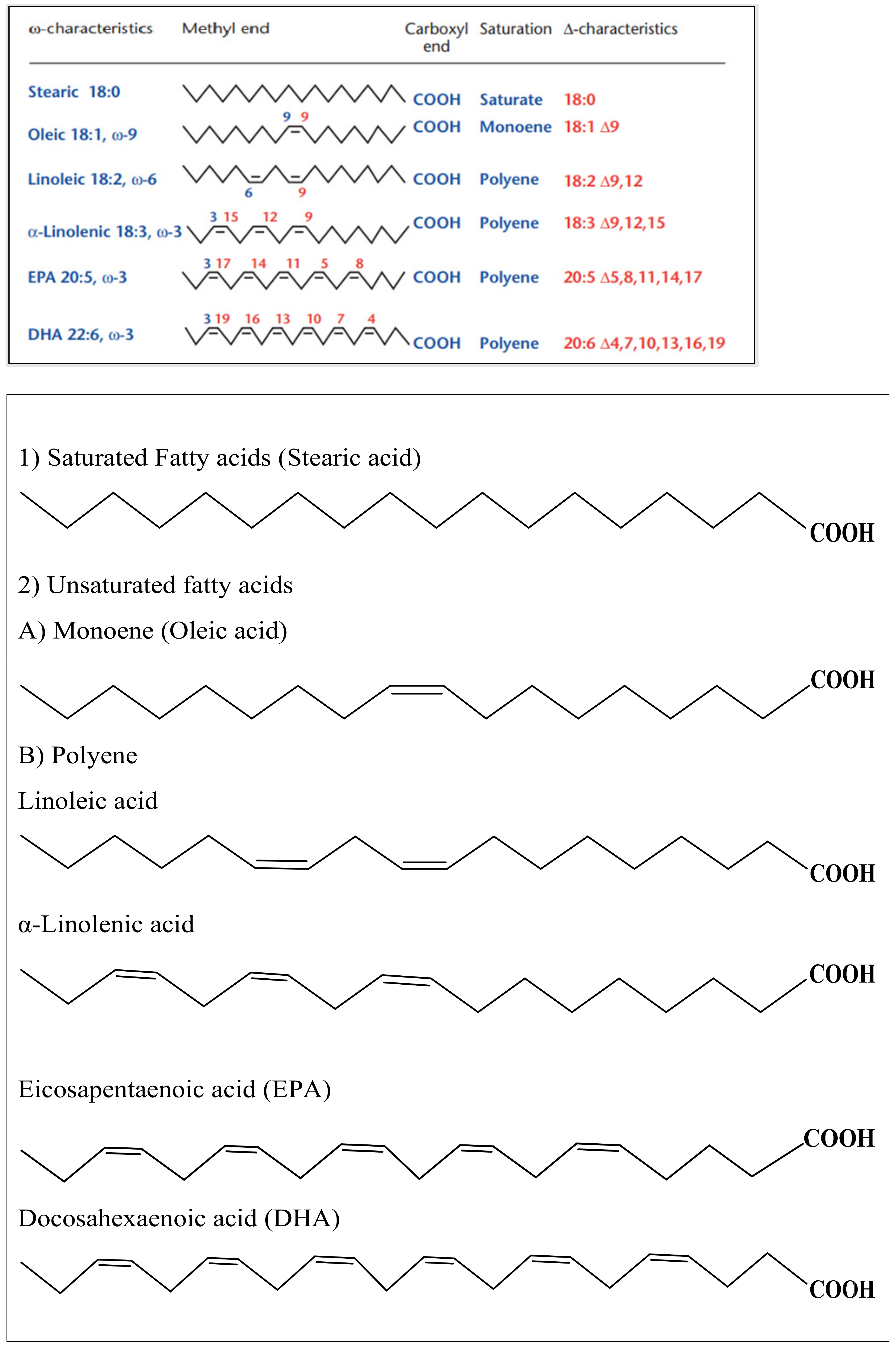
3.2. Fatty Acid Containing Vegetable Carrier Oils
3.2.1. Olive Oil
3.2.2. Almond Oil
3.2.3. Jojoba Oil
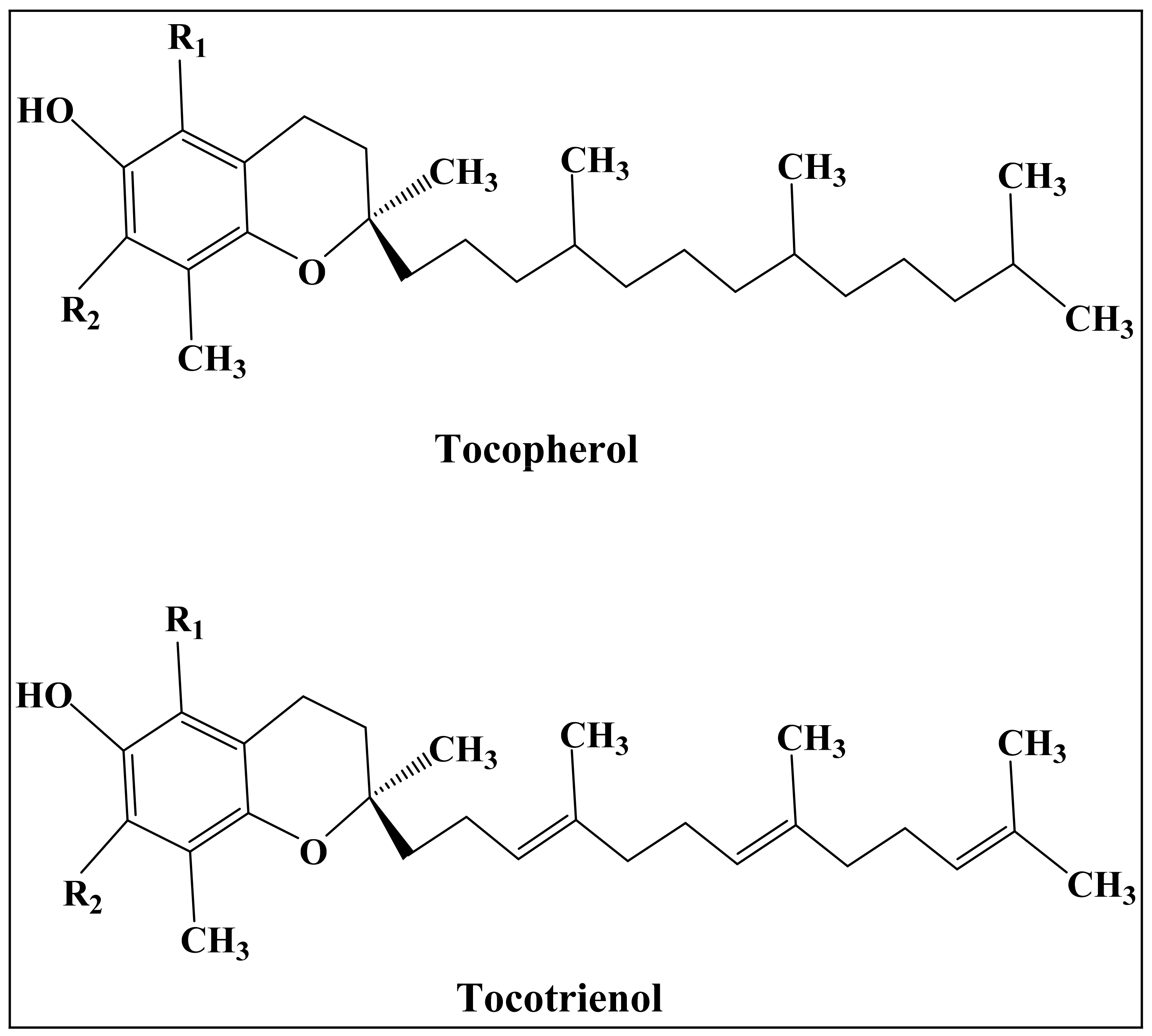
3.3. Vitamin E Derivatives
3.4. Miscellaneous Essential and Carrier Oils
3.4.1. Aloe Vera
3.4.2. Chuanxiong
3.4.3. White Mustard Seed (Sinapis alba L.) Volatile Oil
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Conflicts of Interest
References
- Aggarwal, S.; Agarwal, S.; Jalhan, S. Essential oils as novel human skin penetration enhancer for transdermal drug delivery: A review. Int. J. Pharm. Bio Sci. 2013, 4, 857–868. [Google Scholar]
- El-Nabarawi, M.; El Meshad, A.N.; Moutasim, M.Y. Assessment of bioavailability of sumatriptan transdermal delivery systems in rabbits. Int. J. Pharm. Pharm. Sci. 2013, 5, 225–240. [Google Scholar]
- Herman, A.; Herman, A.P. Essential oils and their constituents as skin penetration enhancer for transdermal drug delivery: A review. J. Pharm. Pharmacol. 2015, 67, 473–485. [Google Scholar] [CrossRef] [PubMed]
- Williams, A.C.; Barry, B.W. Penetration enhancers. Adv. Drug Deliv. Rev. 2012, 64, 128–137. [Google Scholar] [CrossRef]
- Karande, P.; Jain, A.; Ergun, K.; Kispersky, V.; Mitragotri, S. Design principles of chemical penetration en-hancers for transdermal drug delivery. Proc. Natl. Acad. Sci. USA 2005, 102, 4688–4693. [Google Scholar] [CrossRef]
- Prausnitz, M.R.; Langer, R. Transdermal drug delivery. Nat. Biotechnol. 2008, 26, 1261–1268. [Google Scholar] [CrossRef]
- Bilia, A.R.; Guccione, C.; Isacchi, B.; Righeschi, C.; Firenzuoli, F.; Bergonzi, M.C. Essential oils loaded in nanosystems: A developing strategy for a successful therapeutic approach. Evid. Based Complement. Alternat. Med. 2014, 2014, 651593. [Google Scholar] [CrossRef]
- de Matos, S.P.; Teixeira, H.F.; de Lima, A.A.N.; Veiga-Junior, V.F.; Koester, L.S. Essential oils and isolated terpenes in nanosystems designed for topical administration: A review. Biomolecules 2019, 9, 138. [Google Scholar] [CrossRef]
- El Asbahani, A.; Miladi, K.; Badri, W.; Sala, M.; Ait, A.E.H.; Casabianca, H.; Mousadik, A.; Hartmann, D.; Jilale, A.; Renaud, F.N.R.; et al. Essential oils: From extraction to encapsulation. Int. J. Pharm. 2015, 483, 220–243. [Google Scholar] [CrossRef] [PubMed]
- Mehdizadeh, L.; Moghaddam, M. Essential Oils: Biological Activity and Therapeutic Potential. In Therapeutic, Probiotic, and Unconventional Foods; Academic Press: Cambridge, MA, USA, 2018; pp. 167–179. [Google Scholar]
- Osuntokun, O.T. Prospects of essential oils in drug discovery. Adv. Cytol. Pathol. 2017, 2, 00010. [Google Scholar] [CrossRef][Green Version]
- Jiang, Q.; Wu, Y.; Zhang, H.; Liu, P.; Yao, J.; Yao, P.; Chen, J.; Duan, J. Development of essential oils as skin permeation enhancers: Penetration enhancement effect and mechanism of action. Pharm. Biol. 2017, 55, 1592–1600. [Google Scholar] [CrossRef] [PubMed]
- Sharma, R.; Mehra, G.R. Preparation, characteriszation, in vitro and in vivo evaluation of transdermal matrix of Celecoxib. Acta Pharm. Sci. 2011, 53, 67–76. [Google Scholar]
- Liu, C.-H.; Chang, F.Y. Development and characterization of eucalyptol microemulsions for topic delivery of curcumin. Chem. Pharm. Bull. 2011, 59, 172–178. [Google Scholar] [CrossRef] [PubMed]
- Mehta, N.; Patadiya, N.; Patel, J.; Shastri, D.; Shelat, P. Development and evaluation of antiarthritic herbal ointment. Res. J. Pharm. Biol. Chem. Sci. 2013, 4, 221–228. [Google Scholar]
- Shah, S.N.H.; Hussain, T.; Khan, I.U.; Asghar, S.; Shahzad, Y. Formulation study of topically applied lotion: In vitro and in vivo evaluation. BioImpacts 2013, 3, 11–19. [Google Scholar]
- Zhang, Y.; Yang, N.; Lv, J.; Song, H.; Duan, X.; Leng, J.; Bo, J.; Liu, N.; Huang, Y. A Preclinical Study of Novel Phentolamine Formulation and in Vitro/in Vivo Evaluation. J. Drug Deliv. Sci. Technol. 2014, 24, 22–26. [Google Scholar] [CrossRef]
- Pichersky, E.; Noel, J.P.; Dudareva, N. Biosynthesis of plant volatiles: Nature’s diversity and ingenuity. Science 2006, 311, 808–811. [Google Scholar] [CrossRef]
- Chen, J.; Jiang, Q.-D.; Chai, Y.-P.; Zhang, H.; Peng, P.; Yang, X.-X. Natural terpenes as penetration enhancers for transdermal drug delivery. Molecules 2016, 21, 1709. [Google Scholar] [CrossRef]
- Barry, B. Lipid-protein-partitioning theory of skin penetration enhancement. J. Control. Release 1991, 15, 237–248. [Google Scholar] [CrossRef]
- Gunstone, F. (Ed.) Vegetable Oils in Food Technology: Composition, Properties and Uses; John Wiley & Sons: Hoboken, NJ, USA, 2011. [Google Scholar]
- Fox, L.T.; Gerber, M.; Du Plessis, J.; Hamman, J.H. Transdermal drug delivery enhancement by compounds of natural origin. Molecules 2011, 16, 10507–10540. [Google Scholar] [CrossRef]
- Viljoen, J.; Cowley, A.; du Preez, J.; Gerber, M.; Du Plessis, J. Penetration enhancing effects of selected natural oils utilized in topical dosage forms. Drug Dev. Ind. Pharm. 2015, 41, 2045–2054. [Google Scholar] [CrossRef] [PubMed]
- Dubois, V.; Breton, S.; Linder, M.; Fanni, J.; Parmentier, M. Fatty acid profiles of 80 vegetable oils with regard to their nutritional potential. Eur. J. Lipid Sci. Technol. 2007, 109, 710–732. [Google Scholar] [CrossRef]
- Hussain, A.; Khan, G.M.; Khan, N.R.; Khan, A.; Rehman, S.U.; Asif, H.M.; Akram, M.; Ali, Y. Trans-dermal diclofenac potassium gels natural penetration enhancers can be effective. Lat. Am. J. Pharm. 2015, 34, 1022–1029. [Google Scholar]
- Aggarwal, G.; Dhawan, S.L.; Hari, K.S. Natural oils as skin permeation enhancers for transdermal delivery of olanzapine: In vitro and in vivo evaluation. Curr. Drug Deliv. 2012, 9, 172–181. [Google Scholar] [CrossRef] [PubMed]
- Mahmood, H.S.; Alaayedi, M.; Ashoor, J.A.; Alghurabi, H. The enhancement effect of olive and almond oils on permeability of nimesulide as transdermal gel. Int. J. Pharm. Res. 2019, 11, 1200–1206. [Google Scholar]
- Nawaz, A.; Khan, G.M.; Shah, S.U.; Shah, K.U. Preparation and Evaluation of Clotrimazole Matrix Type Patch: Effect of Olive Oil on Drug Penetration Across Rabbit Skin. Proc. Pak. Acad. Sci. 2011, 48, 95–100. [Google Scholar]
- Hasan, Z.A.; Al-Mousawy, J.M.M.; Alghurabi, H.S.K. The effect of almond oil on the permeability of ketoprofen hydrogel. Int. J. Appl. Pharm. 2019, 12, 65–69. [Google Scholar] [CrossRef]
- Aggarwal, G.; Dhawan, S.; Hari, K.S. Formulation, in vitro and in vivo evaluation of transdermal patches containing risperidone. Drug Dev. Ind. Pharm. 2013, 39, 39–50. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. Int. J. Surg. 2010, 8, 336–341. [Google Scholar] [CrossRef]
- Khizer, Z.; Sadia, A.; Sharma, R.; Farhaj, S.; Nirwan, J.S.; Kakadia, P.G.; Hussain, T.; Yousaf, A.; Shahzad, Y.; Conway, B.; et al. Drug delivery approaches for managing overactive bladder [OAB]: A systematic review. Pharmaceuticals 2021, 14, 409. [Google Scholar] [CrossRef]
- Higgins, J.P.; Thomas, J.; Chandler, J.; Cumpston, M.; Li, T.; Page, M.J. Cochrane Handbook For Systematic Reviews of Interventions; John Wiley & Sons: Hoboken, NJ, USA, 2019. [Google Scholar]
- Kmet, L.M.; Cook, L.S.; Lee, R.C. Standard quality assessment criteria for evaluating primary research papers from a variety of fields. Educ. Res. Arch. 2004, 1–4. [Google Scholar] [CrossRef]
- Nirwan, J.S.; Hasan, S.S.; Conway, B.R.; Ghori, M.U. Global prevalence and risk factors of gastro-oesophageal reflux disease [GORD]: Systematic review with meta-analysis. Sci. Rep. 2020, 10, 5814. [Google Scholar] [CrossRef] [PubMed]
- Walsh, D.; Downe, S. Appraising the quality of qualitative research. Midwifery 2006, 22, 108–119. [Google Scholar] [CrossRef]
- Korakakis, V.; Whiteley, R.; Tzavara, A.; Malliaropoulos, N. The effectiveness of extracorporeal shockwave therapy in common lower limb conditions: A systematic review including quantification of patient-rated pain reduction. Br. J. Sports Med. 2018, 52, 387–407. [Google Scholar] [CrossRef] [PubMed]
- Sapra, B.; Jain, S.; Tiwary, A. Percutaneous permeation enhancement by terpenes: Mechanistic view. AAPS J. 2008, 10, 120–132. [Google Scholar] [CrossRef]
- Perveen, S.; Al-Taweel, A. Introductory Chapter: Terpenes and Terpenoids. In Terpenes Terpenoids; BoD-Books on Demand: London, UK, 2018; pp. 1–12. [Google Scholar]
- Sahu, P.; Kashaw, S.K.; Jain, S.; Sau, S.; Iyer, A.K. Assessment of penetration potential of pH responsive double walled biodegradable nanogels coated with eucalyptus oil for the controlled delivery of 5-fluorouracil: In vitro and ex vivo studies. J. Control. Release 2017, 253, 122–136. [Google Scholar] [CrossRef]
- Akhlaq, M.; Siddiqua, A.; Ullah, H.; Akram, M.; Abdur, R.S.; Khan, M.; Nazir, R.; Imran, M.; Sherazi, M.; Baloch, M.; et al. Development of semi-solid formulation for skin administration of pioglitazone. Lat. Am. J. Pharm. 2019, 38, 771–779. [Google Scholar]
- Lin, L.; Chen, W.; Li, C.; Cui, H. Enhancing stability of Eucalyptus citriodora essential oil by solid nanoliposomes encapsulation. Ind. Crops Prod. 2019, 140, 111615. [Google Scholar] [CrossRef]
- Lu, H.; Shao, X.; Cao, J.; Ou, C.; Pan, D. Antimicrobial activity of eucalyptus essential oil against Pseudomonas in vitro and potential application in refrigerated storage of pork meat. Int. J. Food Sci. Technol. 2016, 51, 994–1001. [Google Scholar] [CrossRef]
- Higgins, C.; Palmer, A.; Nixon, R. Eucalyptus oil: Contact allergy and safety. Contact Derm. 2015, 72, 344–346. [Google Scholar] [CrossRef]
- Sugumar, S.; Clarke, S.; Nirmala, M.; Tyagi, B.; Mukherjee, A.; Chandrasekaran, N. Nanoemulsion of euca-lyptus oil and its larvicidal activity against Culex quinquefasciatus. Bull. Entomol. Res. 2014, 104, 393–402. [Google Scholar] [CrossRef] [PubMed]
- Tolba, H.; Moghrani, H.; Benelmouffok, A.; Kellou, D.; Maachi, R. Essential oil of Algerian Eucalyptus citriodora: Chemical composition, antifungal activity. J. Mycol. Med. 2015, 25, e128–e133. [Google Scholar] [CrossRef] [PubMed]
- Brochot, A.; Guilbot, A.; Haddioui, L.; Roques, C. Antibacterial, antifungal, and antiviral effects of three essential oil blends. Microbiologyopen 2017, 6, e00459. [Google Scholar] [CrossRef] [PubMed]
- Harkat-Madouri, L.; Asma, B.; Madani, K.; Said, Z.B.-O.S.; Rigou, P.; Grenier, D.; Allalou, H.; Remini, H.; Adjaoud, A.; Boulekbache-Makhlouf, L. Chemical composition, antibacterial and antioxidant activities of essential oil of Eucalyptus globulus from Algeria. Ind. Crops Prod. 2015, 78, 148–153. [Google Scholar] [CrossRef]
- Preedy, V.R. Essential Oils in Food Preservation, Flavor and Safety; Academic Press: Cambridge, MA, USA, 2015. [Google Scholar]
- Akram, M.R.; Ahmad, M.; Abrar, A.; Sarfraz, R.M.; Mahmood, A. Formulation design and development of matrix diffusion controlled transdermal drug delivery of glimepiride. Drug Des. Dev. Ther. 2018, 12, 349–364. [Google Scholar] [CrossRef]
- Yaqoob, A.; Ahmad, M.; Mahmood, A.; Sarfraz, R.M. Preparation, in vitro and in vivo characterization of hydrophobic patches of a highly water soluble drug for prolonged plasma half life: Effect of permeation enhancers. Acta Pol. Pharm. 2016, 73, 1639–1648. [Google Scholar]
- Zeng, Z.-W.; Lin, J.; Li, H.; Xi, T.; Zhou, W.; Fan, H. Effect of the matrices and penetration enhancers in elemene transdermal drug delivery system. Afr. J. Pharm. Pharmacol. 2011, 5, 879–886. [Google Scholar]
- Shen, T.; Xu, H.; Weng, W.; Zhang, J. Development of a reservoir-type transdermal delivery system containing eucalyptus oil for tetramethylpyrazine. Drug Deliv. 2013, 20, 19–24. [Google Scholar] [CrossRef]
- Madkaikar, N.; Shirodker, A.; Bhangle, S.; Gude, R. Formulation, optimization and evaluation of matrix type transdermal drug delivery system of antiemetic drug using essential oils and non-ionic surfactant as permeation enhancers. Indian Drugs 2018, 55, 77–78. [Google Scholar] [CrossRef]
- Rajan, R.; Vasudevan, D.T. Effect of permeation enhancers on the penetration mechanism of transfersomal gel of ketoconazole. J. Adv. Pharm. Technol. Res. 2012, 3, 112–116. [Google Scholar]
- Parney, S.; Dhurke, R.K. Effect of natural penetration enhancers on dermal delivery of hydrocortisone acetate. J. Pharm. Investig. 2014, 44, 365–380. [Google Scholar] [CrossRef]
- Rout, B.; Liu, C.H.; Wu, W.C. Enhancement of photodynamic inactivation against Pseudomonas aeruginosa by a nano-carrier approach. Colloids Surf. B Biointerfaces 2016, 140, 472–480. [Google Scholar] [CrossRef] [PubMed]
- El Maghraby, G.M.; Arafa, M.F.; Osman, M.A. Microemulsion for simultaneous transdermal delivery of benzocaine and indomethacin: In vitro and in vivo evaluation. Drug Dev. Ind. Pharm. 2014, 40, 1637–1644. [Google Scholar] [CrossRef] [PubMed]
- Saadatzadeh, A.; Salimi, A.; Zarooni, M. Influence of permeation enhancers on the in vitro skin permeation of ketorolac tromethamine through excised rat skin: A mechanistic study. Asian J. Pharm. Clin. Res. 2018, 11, 242–247. [Google Scholar] [CrossRef]
- Christensen, L.; Turner, R.; Weaver, S.; Caserta, F.; Long, L.; Ghannoum, M.; Brown, M. Evaluation of the ability of a novel miconazole formulation to penetrate nail by using three in vitro nail models. Antimicrob. Agents Chemother. 2017, 61, e02554-16. [Google Scholar] [CrossRef] [PubMed]
- Eid, R.K.; Essa, E.A.; El Maghraby, G.M. Essential oils in niosomes for enhanced transdermal delivery of felodipine. Pharm. Dev. Technol. 2019, 24, 157–165. [Google Scholar] [CrossRef] [PubMed]
- Kakadia, P.G. Formulation and Evaluation of Nanoencapsulated Antimicrobial Agents for Dermal Delivery; University of Huddersfield: Huddersfield, UK, 2016. [Google Scholar]
- Nurdjannah, N.; Bermawie, N.C. Handbook of Herbs and Spices; Elsevier: Amsterdam, The Netherlands, 2012; pp. 197–215. [Google Scholar]
- Surburg, H.; Panten, J. Common Fragrance and Flavor Materials: Preparation, Properties and Uses; John Wiley & Sons: Hoboken, NJ, USA, 2016. [Google Scholar]
- Barboza, J.N.; Filho, C.D.S.M.B.; Silva, R.O.; Medeiros, J.V.R.; de Sousa, D. An Overview on the Anti-inflammatory Potential and Antioxidant Profile of Eugenol. Oxid. Med. Cell. Longev. 2018, 2018, 3957262. [Google Scholar] [CrossRef]
- Arora, R.; Khan, R.; Ojha, A.; Chopra, H.; Upadhyaya, K. Formulation and characterization of terbinafine emulgel for superficial fungal infections. Res. J. Pharm. Technol. 2018, 11, 5029–5036. [Google Scholar] [CrossRef]
- Lv, X.; Liu, T.; Ma, H.; Tian, Y.; Li, L.; Li, Z.; Gao, M.; Zhang, J.; Tang, Z. Preparation of essential oil-based microemulsions for im-proving the solubility, pH stability, photostability, and skin permeation of quercetin. AAPS PharmSciTech 2017, 18, 3097–3104. [Google Scholar] [CrossRef]
- Khullar, R.; Kumar, D.; Seth, N.; Saini, S. Formulation and evaluation of mefenamic acid emulgel for topical delivery. Saudi Pharm. J. 2012, 20, 63–67. [Google Scholar] [CrossRef]
- Muţ, A.; Vlaia, L.; Coneac, G.; Olariu, I.; Vlaia, V.; Stănciulescu, C.; Mitu, M.A.; Szabadi, Z.; Lupuliasa, D. Chitosan/HPMC-based hydrogels containing essential oils for topical delivery of fluconazole: Preliminary studies. Farmacia 2018, 66, 248–256. [Google Scholar]
- Magdum, S.S.; Dounde, P.; Kamble, D.D.; Patil, S.V. Design and characterization of novel emulgel for pain management. Indian Drugs 2016, 53, 16–20. [Google Scholar] [CrossRef]
- Tripathi, A.K.; Mishra, S. Plant Monoterpenoids [Prospective Pesticides]. In Ecofriendly Pest Management for Food Security; Elsevier: Amsterdam, The Netherlands, 2016; pp. 507–524. [Google Scholar]
- PubChem. Eucalyptol Cited 2021. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/2758 (accessed on 1 December 2021).
- Galan, D.M.; Ezeudu, N.E.; Garcia, J.; Geronimo, C.A.; Berry, N.M.; Malcolm, B.J. Eucalyptol [1,8-cineole]: An underutilized ally in respiratory disorders? J. Essent. Oil Res. 2020, 32, 103–110. [Google Scholar] [CrossRef]
- Prakash, G.; Venkala, R.I.; Doddayya, H. Comparative evaluation of selected vegetable oils and terpenes on transdermal permeation of ketorolac tromethamine. Int. J. Drug. Dev. Res. 2011, 3, 178–188. [Google Scholar]
- Nikolić, I.; Mitsou, E.; Pantelic, I.; Randjelovic, D.; Markovic, B.; Papadimitriou, V.; Xenakis, A.; Lunter, D.J.; Zugic, A.; Savic, S. Microstructure and biopharmaceutical performances of curcumin-loaded low-energy nanoemulsions containing eucalyptol and pinene: Terpenes’ role overcome penetration enhancement effect? Eur. J. Pharm. Sci. 2020, 142, 105135. [Google Scholar] [CrossRef] [PubMed]
- Abd, E.; Namjoshi, S.; Mohammed, Y.H.; Roberts, M.S.; Grice, J.E. Synergistic skin penetration enhancer and nanoemulsion formulations promote the human epidermal permeation of caffeine and naproxen. J. Pharm. Sci. 2016, 105, 212–220. [Google Scholar] [CrossRef]
- Shi, J.; Cong, W.; Wang, Y.; Liu, Q.; Luo, G. Microemulsion-based patch for transdermal delivery of huperzine A and ligustrazine phosphate in treatment of Alzheimer’s disease. Drug Dev. Ind. Pharm. 2012, 38, 752–761. [Google Scholar] [CrossRef]
- Ubongkot, T.; Duangjit, S.; Rojanarata, T.; Opanasopit, P.; Ngawhirunpat, T. Ultradeformable liposomes with terpenes for delivery of hydrophilic compound. J. Liposome Res. 2012, 22, 254–262. [Google Scholar] [CrossRef]
- Abd, E.; Benson, H.A.E.; Roberts, M.; Grice, J.E. Minoxidil Skin Delivery from Nanoemulsion Formulations Containing Eucalyptol or Oleic Acid: Enhanced Diffusivity and Follicular Targeting. Pharmaceutics 2018, 10, 19. [Google Scholar] [CrossRef]
- Ngawhirunpat, T.; Thipwichai, S.; Opanasopit, P.; Rojanarata, T.; Panomsuk, S. Development and Evaluation of Ketoprofen Acrylic Transdermal Patches. Trop. J. Pharm. Res. 2012, 11, 553–560. [Google Scholar] [CrossRef][Green Version]
- Casey, A.L.; Karpanen, T.J.; Conway, B.R.; Worthington, T.; Nightingale, P.; Waters, R. Enhanced chlor-hexidine skin penetration with 1,8-cineole. BMC Infect. Dis. 2017, 17, 350. [Google Scholar] [CrossRef] [PubMed]
- Herro, E.; Jacob, S.E. Mentha piperita [peppermint]. Dermatitis 2010, 21, 327–329. [Google Scholar] [CrossRef] [PubMed]
- de Groot, A.C.; Schmidt, E. Essential Oils: Contact Allergy and Chemical Composition; Routledge: London, UK, 2021. [Google Scholar]
- PubChem. Peppermint Oil. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/6850741 (accessed on 1 December 2021).
- Maniyar, M.G.; Kokare, C.R. Formulation and evaluation of spray dried liposomes of lopinavir for topical application. J. Pharm. Investig. 2018, 49, 259–270. [Google Scholar] [CrossRef]
- Moghimi, H.R.; Jamali, B.; Farahmand, S.; Shafaghi, B. Effect of essential oils, hydrating agents, and ethanol on hair removal efficiency of thioglycolates. J. Cosmet. Dermatol. 2013, 12, 41–48. [Google Scholar] [CrossRef] [PubMed]
- Eccles, R. Menthol and related cooling compounds. J. Pharm. Pharmacol. 1994, 46, 618–630. [Google Scholar] [CrossRef] [PubMed]
- PubChem. Menthol. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/1254 (accessed on 1 December 2021).
- Stanos, S.P.; Tyburski, M.D.; Parikh, S.S. Minor and Short-Acting Analgesics, Including Opioid Combination Products. In Practical Management of Pain; Elsevier: Amsterdam, The Netherlands, 2014; pp. 508–529. [Google Scholar]
- Choulis, N. Miscellaneous Drugs, Materials, Medical Devices and Techniques. In Side Effects of Drugs Annual; Elsevier: Amsterdam, The Netherlands, 2014; Volume 36, pp. 725–746. [Google Scholar]
- Yu, Z.; Liang, Y.; Liang, W. Development and evaluation of alpha-asarone transdermal patches based on hot-melt pressure-sensitive adhesives. AAPS PharmSciTech 2013, 14, 294–300. [Google Scholar] [CrossRef] [PubMed]
- Das, G.; Ahmed, A.B. Effect of menthol on the transdermal permeation of aceclofenac from microemulsion formulation. Int. J. Appl. Pharm. 2019, 11, 117–122. [Google Scholar]
- Patel, P.M. Formulation Development and Evaluation of Bioadhesive Drug Delivery System Containing Selected Phytopharmaceuticals. Ph.D. Thesis, Gujarat Technology University, Ahmedabad, Gujarat, India, 2017. [Google Scholar]
- Wang, Y.; Cao, S.; Yu, K.; Yang, F.; Yu, X.; Zhai, Y.; Wu, C.; Xu, Y. Integrating tacrolimus into eutectic oil-based microemulsion for atopic dermatitis: Simultaneously enhancing percutaneous delivery and treatment efficacy with relieving side effects. Int. J. Nanomed. 2019, 14, 5849–5863. [Google Scholar] [CrossRef]
- Liu, C.; Hu, J.; Sui, H.; Zhao, Q.; Zhang, X.; Wang, W. Enhanced skin permeation of glabridin using eutectic mixture-based nanoemulsion. Drug Deliv. Transl. Res. 2017, 7, 325–332. [Google Scholar] [CrossRef]
- PubChem. Limonene. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/22311 (accessed on 1 December 2021).
- Ravichandran, C.; Badgujar, P.C.; Gundev, P.; Upadhyay, A. Review of toxicological assessment of d-limonene, a food and cosmetics additive. Food Chem. Toxicol. 2018, 120, 668–680. [Google Scholar] [CrossRef]
- Charoenputtakun, P.; Pamornpathomkul, B.; Opanasopit, P.; Rojanarata, T.; Ngawhirunpat, T. Terpene composited lipid nanoparticles for enhanced dermal delivery of all-trans-retinoic acids. Biol. Pharm. Bull. 2014, 37, 1139–1148. [Google Scholar] [CrossRef] [PubMed]
- Hoppel, M.; Caneri, M.; Glatter, O.; Valenta, C. Self-assembled nanostructured aqueous dispersions as dermal delivery systems. Int. J. Pharm. 2015, 495, 459–462. [Google Scholar] [CrossRef] [PubMed]
- Wattanasri, P. Development of microemulsions, microemulgels and organogels for transdermal delivery of Kaempferia parviflora extract. AAPS PharmSciTech 2016, 19, 2058–2067. [Google Scholar]
- Trease, G.; Evans, M. Pharmacopoeial and Related Drugs of Biological Origin. A Textbook of Pharmacognosy, 15th ed.; WB Saunders: London, UK, 2001; pp. 262–270. [Google Scholar]
- Mercier, B.; Prost, J.; Prost, M. The essential oil of turpentine and its major volatile fraction [α-and β-pinenes]: A review. Int. J. Occup. Med. Environ. Health 2009, 22, 331–342. [Google Scholar] [CrossRef]
- Xu, P.-G.; Lei, X.-F.; Ren, B.-D.; Lv, S.-Y.; Zhang, J.-L. Diclofenac transdermal patch versus the sustained release tablet: A randomized clinical trial in rheumatoid arthritic patients. Trop. J. Pharm. Res. 2017, 16, 477. [Google Scholar] [CrossRef][Green Version]
- Khan, N.; Khan, G.M.; Wahab, A.; Khan, A.R.; Hussain, A.; Nawaz, A.; Akhlaq, M. Formulation, and physical, in vitro and ex vivo evaluation of transdermal ibuprofen hydrogels containing turpentine oil as penetration enhancer. Pharmazie 2011, 66, 849–852. [Google Scholar]
- Wang, F.L.; Ji, H.M.; Zhu, J.Y.; Xu, G.J.; Guan, Y.Z.; Chen, Y.J. Penetration enhancement effect of turpentine oil on transdermal film of ketorolac. Trop. J. Pharm. Res. 2015, 14, 1341. [Google Scholar] [CrossRef][Green Version]
- Wang, H.B.; Yang, F.F.; Gai, X.M.; Cheng, B.C.; Li., J.Y.; Pan, H.; Yang, X.-G.; Pan, W.-S. A pH-independent instantaneous release of flurbiprofen: A study of the preparation of complexes, their characterization and in vitro/in vivo evaluation. Drug Dev. Ind. Pharm. 2017, 43, 1460–1471. [Google Scholar] [CrossRef]
- Rustan, A.; Drevon, C. Fatty Acids: Structures and Properties. In Encyclopedia of Life Sciences; Nature Publishing: London, UK, 2005. [Google Scholar]
- Kumar, L.; Verma, S.; Kumar, S.; Prasad, D.N.; Jain, A.K. Fatty acid vesicles acting as expanding horizon for transdermal delivery. Artif. Cells Nanomed. Biotechnol. 2016, 45, 251–260. [Google Scholar] [CrossRef]
- Mittal, A.; Sara, U.; Ali, A.; Aqil, M. Status of fatty acids as skin penetration enhancers-a review. Curr. Drug Deliv. 2009, 6, 274–279. [Google Scholar] [CrossRef]
- Bonnist, E.; Gorce, J.-P.; Mackay, C.; Pendlington, R.; Pudney, P. Measuring the Penetration of a Skin Sensitizer and Its Delivery Vehicles Simultaneously with Confocal Raman Spectroscopy. Skin Pharmacol. Physiol. 2011, 24, 274–283. [Google Scholar] [CrossRef] [PubMed]
- Hussain, A.; Khan, G.M.; Jan, S.U.; Shah, S.; Shah, K.U.; Akhlaq, M.; Khan, N.; Nawaz, A.; Wahab, A. Effect of olive oil on transdermal penetration of flurbiprofen from topical gel as enhancer. Pak. J. Pharm. Sci. 2012, 25, 365–369. [Google Scholar] [PubMed]
- Pelikh, O.; Stahr, P.-L.; Huang, J.; Gerst, M.; Scholz, P.; Dietrich, H.; Geisel, N.; Keck, C.M. Nanocrystals for improved dermal drug delivery. Eur. J. Pharm. Biopharm. 2018, 128, 170–178. [Google Scholar] [CrossRef] [PubMed]
- Kiritsakis, A.; Markakis, P. Olive oil: A review. Adv. Food Res. 1988, 31, 453–482. [Google Scholar]
- Tsimidou, M.; Blekas, G.; Boskou, D. Olive oil. In Encyclopedia of Food Sciences and Nutrition, 2nd ed.; Academic Press: London, UK, 2003; pp. 4252–4260. [Google Scholar]
- Calabriso, N.; Scoditti, E.; Pellegrino, M.; Carluccio, M.A. Olive Oil. In The Mediterranean Diet; Elsevier: Amsterdam, The Netherlands, 2015; pp. 135–142. [Google Scholar]
- PubChem. Olive Oil. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/24753271 (accessed on 1 December 2021).
- Nawaz, A.; Khan, G.M.; Akhlaq, M.; Zeb, A.; Khan, A.; Hussain, A.; Dayo, A. Formulation and in-vitro evaluation of topically applied curcumin hydrogel. Lat. Am. J. Pharm. 2012, 31, 671–677. [Google Scholar]
- Khan, M.R.U. Formulation design, optimization and enhancement of skin permeation of ibuprofen cream by using olive oil as permeation enhancer. Indo Am. J. Pharm. Sci. 2018, 5, 17–23. [Google Scholar]
- Mortazavi, S.A.; Pishrochi, S. Formulation and in-vitro evaluation of tretinoin microemulsion as a potential carrier for dermal drug delivery. Iran. J. Pharm. Res. IJPR 2013, 12, 599. [Google Scholar]
- Aggarwal, G.; Dhawan, S.; Harikumar, S.L. Formulation, in vitro, and in vivo evaluation of matrix-type transdermal patches containing olanzapine. Pharm. Dev. Technol. 2013, 18, 916–925. [Google Scholar] [CrossRef]
- Shokri, N.; Javari, H.A. Using a synergistic combination of two enhancers for dermal delivery of collagen in pharmaceutical and cosmetic products. J. Pharm. Res. 2015, 14, 1–6. [Google Scholar] [CrossRef]
- Ahmad, Z. The uses and properties of almond oil. Complement. Ther. Clin. Pract. 2010, 16, 10–12. [Google Scholar] [CrossRef]
- National Research Council. Diet and Health: Implications for Reducing Chronic Disease Risk; National Academies Press: Cambridge, MA, USA, 1989. [Google Scholar]
- Nawaz, A.; Jan, S.U.; Khan, N.; Hussain, A.; Khan, G.M. Formulation and in vitro evaluation of clotrimazole gel containing almond oil and tween 80 as penetration enhancer for topical application. Pak. J. Pharm. Sci. 2013, 26, 617. [Google Scholar] [PubMed]
- Hussain, A.; Khan, G.M.; Shah, S.U.; Shah, K.U.; Khan, N.; Wahab, A.; Rehman, A.-U. Development of a novel ketoprofen transdermal patch: Effect of almond oil as penetration enhancers on in-vitro and ex-vivo penetration of ketoprofen through rabbit skin. Pak. J. Pharm. Sci. 2012, 25, 227–232. [Google Scholar]
- Parveen, K.; Shrivastava, B.; Gupta, M.M.; Sharma, A.K. Optimization and preparation of solid lipid nano-particle incorporated transdermal patch of timolol maleate using factorial design. Int. J. Appl. Pharm. 2019, 11, 100–107. [Google Scholar] [CrossRef]
- Thakur, N.K.; Bharti, P.; Mahant, S.; Rao, R. Formulation and characterization of benzoyl peroxide gellified emulsions. Sci. Pharm. 2012, 80, 1045–1060. [Google Scholar] [CrossRef]
- Choe, C.; Schleusener, J.; Lademann, J.; Darvin, M.E. In vivo confocal Raman microscopic determination of depth profiles of the stratum corneum lipid organization influenced by application of various oils. J. Dermatol. Sci. 2017, 87, 183–191. [Google Scholar] [CrossRef] [PubMed]
- Patzelt, A.; Lademann, J.; Richter, H.; Darvin, M.E.; Schanzer, S.; Thiede, G.; Sterry, W.; Vergou, T.; Hauser, M. In vivo investigations on the penetration of various oils and their influence on the skin barrier. Skin Res. Technol. 2011, 18, 364–369. [Google Scholar] [CrossRef]
- Wisniak, T. Jojoba oil and derivates. Prog. Chem. Fats Other Lipids 1977, 15, 167–218. [Google Scholar] [CrossRef]
- Pazyar, N.; Yaghoobi, R.; Ghassemi, M.; Kazerouni, A.; Rafeie, E.; Jamshydian, N. Jojoba in dermatology: A succinct review. G. Ital. Dermatol. 2013, 148, 687–691. [Google Scholar]
- Vater, C.; Hlawaty, V.; Werdenits, P.; Cichon, M.A.; Klang, V.; Elbe-Burger, A.; Wirth, M.; Valenta, C. Effects of lecithin-based nanoemulsions on skin: Short-time cytotoxicity MTT and BrdU studies, skin penetration of surfactants and additives and the delivery of curcumin. Int. J. Pharm. 2020, 580, 119209. [Google Scholar] [CrossRef]
- Gogoll, K.; Stein, P.; Lee, K.; Arnold, P.; Peters, T.; Schild, H.; Radsak, M.; Langguth, P. Solid nanoemulsion as antigen and immunopotentiator carrier for transcutaneous immunization. Cell. Immunol. 2016, 308, 35–43. [Google Scholar] [CrossRef] [PubMed]
- Keen, M.A.; Hassan, I. Vitamin E in dermatology. Indian Dermatol. Online J. 2016, 7, 311. [Google Scholar] [CrossRef] [PubMed]
- Niki, E.; Abe, K. Vitamin E: Structure, Properties and Functions. In Vitamin E: Chemistry and Nutritional Benefits; Royal Society of Chemistry: London, UK, 2019; pp. 1–11. [Google Scholar] [CrossRef]
- Ingh, I.; Nair, R.S.; Gan, S.; Cheong, V.; Morris, A. An evaluation of crude palm oil (CPO) and tocotrienol rich fraction (TRF) of palm oil as percutaneous permeation enhancers using full-thickness human skin. Pharm. Dev. Technol. 2019, 24, 448–454. [Google Scholar]
- Sharma, K.; Mittal, A.; Chauhan, N. Aloe vera as penetration enhancer. Int. J. Drug Dev. Res. 2015, 7, 31–43. [Google Scholar]
- Vashisth, I.; Ahad, A.; Aqil, M.; Agarwal, S.P. Investigating the potential of essential oils as penetration enhancer for transdermal losartan delivery: Effectiveness and mechanism of action. Asian J. Pharm. Sci. 2014, 9, 260–267. [Google Scholar] [CrossRef]
- Ran, X.; Ma, L.; Peng, C.; Zhang, H.; Qin, L.-P. Ligusticum chuanxiong Hort: A review of chemistry and pharmacology. Pharm. Biol. 2011, 49, 1180–1189. [Google Scholar] [CrossRef]
- Zhang, L.-C.; Gao, L.-H.; Hu, J.-H.; Yan, C.-X.; Zhu, Q.-G. Enhancing effect of essential oils from Ligusticum chuanxiong Hort. On the permeation of flurbiprofen through isolated rat skin. Pharm. Care Res. 2006, 6, 413. [Google Scholar]
- Mitrović, P.M.; Stamenković, O.S.; Banković-Ilić, I.; Djalović, I.G.; Nježić, Z.B.; Farooq, M.; Kadambot, H.M.S.; Velijovic, V.B. White mustard [Sinapis alba L.] oil in biodiesel production: A review. Front. Plant Sci. 2020, 11, 299. [Google Scholar] [CrossRef]
- Ruan, S.; Wang, Z.; Xiang, S.; Chen, H.; Shen, Q.; Liu, L.; Wu, W.; Cao, S.; Wang, Z.; Yang, Z.; et al. Mechanisms of white mustard seed (Sinapis alba L.) volatile oils as transdermal penetration enhancers. Fitoterapia 2019, 138, 104195. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alhasso, B.; Ghori, M.U.; Conway, B.R. Systematic Review on the Effectiveness of Essential and Carrier Oils as Skin Penetration Enhancers in Pharmaceutical Formulations. Sci. Pharm. 2022, 90, 14. https://doi.org/10.3390/scipharm90010014
Alhasso B, Ghori MU, Conway BR. Systematic Review on the Effectiveness of Essential and Carrier Oils as Skin Penetration Enhancers in Pharmaceutical Formulations. Scientia Pharmaceutica. 2022; 90(1):14. https://doi.org/10.3390/scipharm90010014
Chicago/Turabian StyleAlhasso, Bahjat, Muhammad Usman Ghori, and Barbara R. Conway. 2022. "Systematic Review on the Effectiveness of Essential and Carrier Oils as Skin Penetration Enhancers in Pharmaceutical Formulations" Scientia Pharmaceutica 90, no. 1: 14. https://doi.org/10.3390/scipharm90010014
APA StyleAlhasso, B., Ghori, M. U., & Conway, B. R. (2022). Systematic Review on the Effectiveness of Essential and Carrier Oils as Skin Penetration Enhancers in Pharmaceutical Formulations. Scientia Pharmaceutica, 90(1), 14. https://doi.org/10.3390/scipharm90010014







