Ameliorating Effect of Combined Cinnamon and Ginger Oils against the Neurotoxicity of Nicotine Administration on the Prefrontal Cortex of Adult Albino Rats: Immunohistochemical and Ultrastructural Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Experimental Animals
2.2. Study Design
2.3. Biochemical Analysis
2.4. Histopathological Examination of the Brain Tissues by H&E
2.5. Immunohistochemical Examination for TNF Alpha and GFAP
2.6. Electron Microscopic Examination of the Prefrontal Cortex
2.7. Statistical Analysis
3. Results
3.1. Oxidative Stress Markers (MDA, GSH) Level in Nicotine-Exposed Rat’s Prefrontal Cortex with Combined Treatment with Ginger and Cinnamon Oils
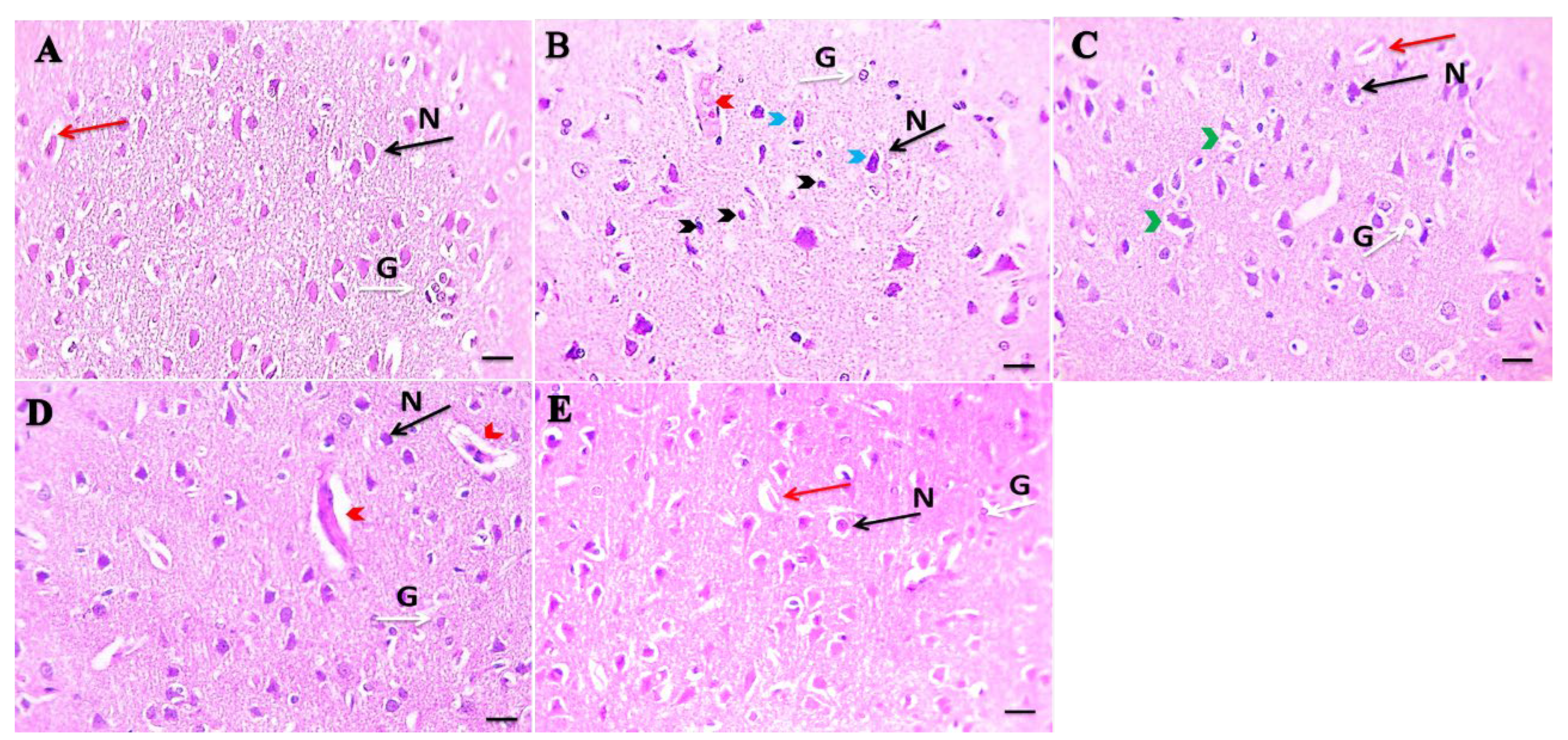
3.2. Morphological Changes in Prefrontal Cortex under Combined Treatment of Ginger and Cinnamon Oils
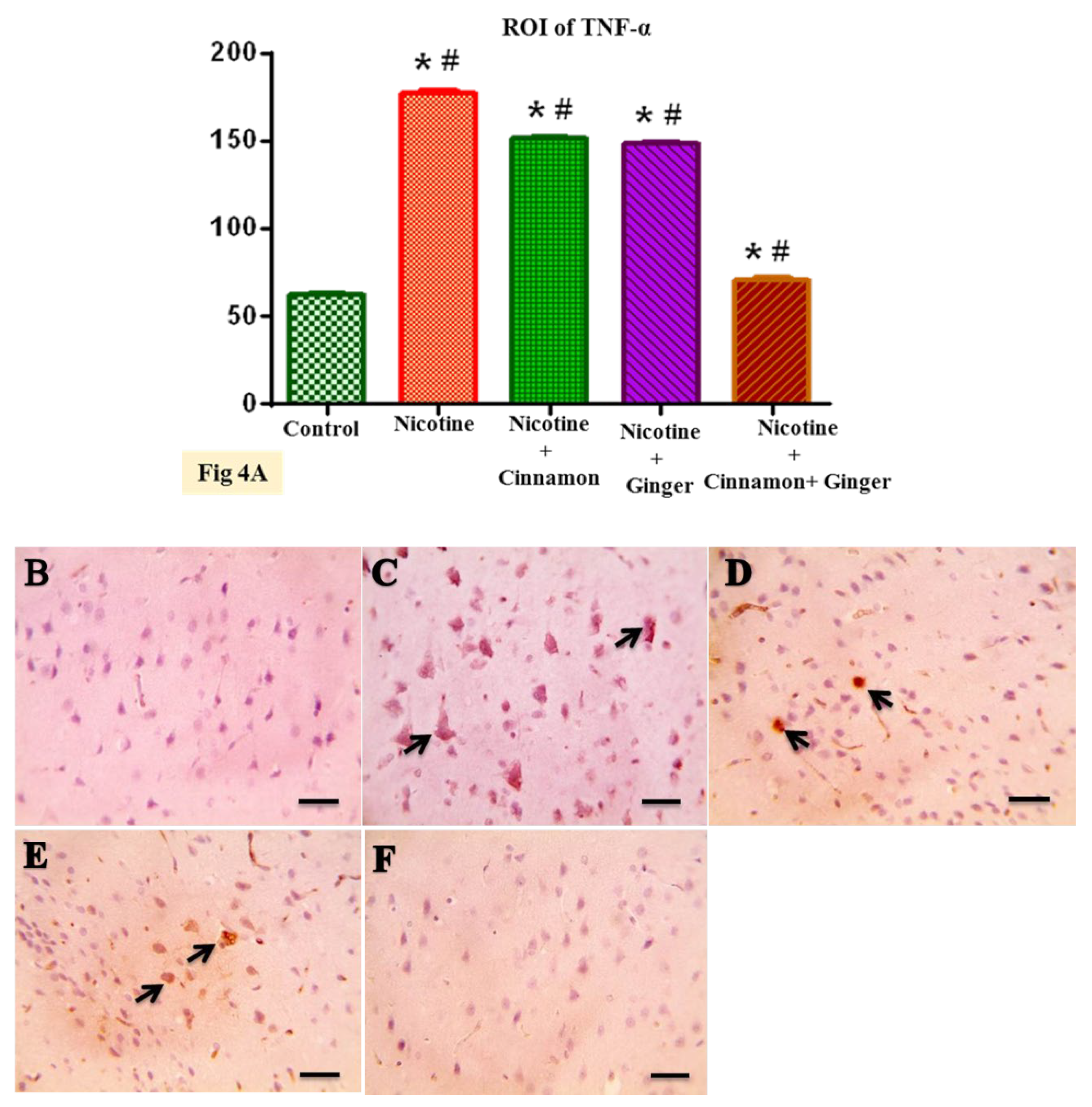
3.3. Effects of Combined Cinnamon and Ginger Oils on Proinflammatory Cytokines Marker (TNF-α) in Brain Tissues
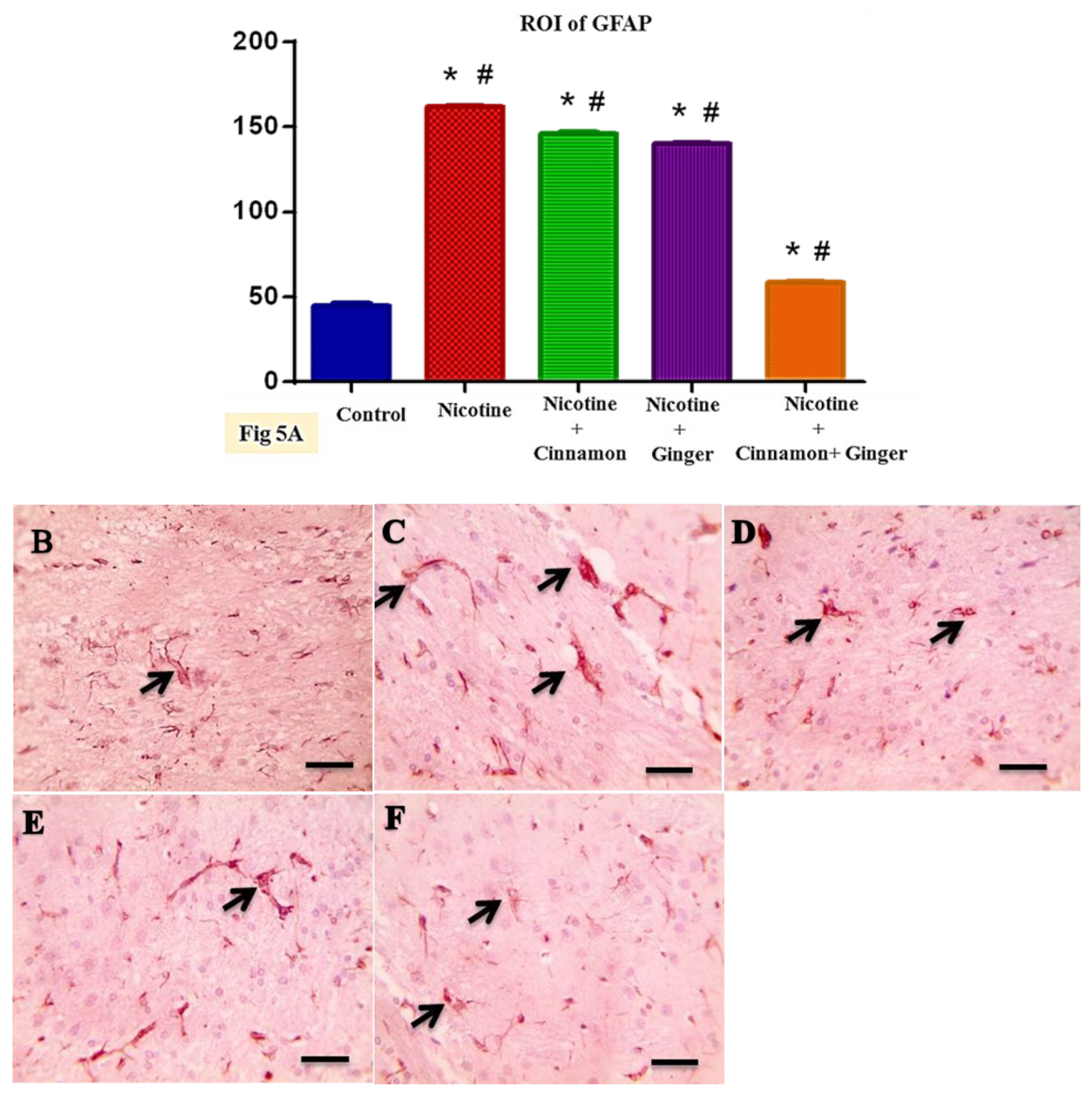
3.4. Combined Effect of Cinnamon and Ginger Oils on Marker of Astrogliosis GFAP in Brain Tissues
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Noor, K.C.; Mawaddatul, U.S.; Bahrudin, M. Effect of Electronic Cigarette on Brain Prefrontal Cortex of Male wistar Rats. J. Med. Res. 2020, 6, 98–102. [Google Scholar] [CrossRef]
- Center for Disease Control. State specific prevalence of current cigarettes smoking among adults and their proportion of adults who work in a smoke-free environment—United States, 1999. Morb. Mortal. Wkly. Rep. 2000, 200, 978–982. [Google Scholar]
- Benowitz, N.L. Pharmacology of nicotine: Addiction, smoking-induced disease, and therapeutics. Ann. Rev. Pharmacol. Toxicol. 2009, 49, 57–71. [Google Scholar] [CrossRef] [Green Version]
- Djordjevic, M.V.; Branneman, K.D.; Hoffman, D. Identification and analysis of a nicotine derived N. nitrosamine acid and other nitrosamine acids in tobacco. Carcinogenesis 1989, 10, 1725–1731. [Google Scholar] [CrossRef]
- Abel, E.L. Prenatal effects of alcohol on growth: A brief overview. Fed. Proc. 1985, 44, 2318–2322. [Google Scholar]
- Jha, P.; Chaloupka, F.J.; Moore, J.; Gajalakshmi, V.; Gupta, P.C.; Peck, R.; Asma, S.; Zatonski, W. Tobacco Addiction, Disease Control Properties in Developing Countries, 2nd ed.; The International Bank for Reconstruction and Development/the World Bank: Washington, DC, USA, 2006; Chapter 46. [Google Scholar]
- Farsalinos, K.E.; Spyrou, A.; Stefopoulos, C.; Tsimopoulou, K.; Kourkoveli, P.; Tsiapras, D.; Kyrzopoulos, S.; Poulas, K.; Voudris, V. Nicotine absorption from electronic cigarette use: Comparison between experienced consumers (vapers) and naïve users (smokers). Sci. Rep. 2015, 17, 11269. [Google Scholar] [CrossRef]
- Hecht, S.S. Tobacco carcinogens, their biomarkers and tobacco induced cancer. Nat. Rev. Cancer 2007, 3, 733–744. [Google Scholar] [CrossRef]
- Hellenbrand, W.; Seidler, A.; Robra, B.P. Smoking and Parkinson’s disease: A case-control study in Germany. Int. J. Epidemiol. 1997, 26, 328–339. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Grandinetti, A.; Morens, D.M.; Reed, D. Prospective study of cigarette smoking and the risk of developing idiopathic Parkinson’s disease. Am. J. Epidemiol. 1994, 139, 1129–1138. [Google Scholar] [CrossRef]
- Riljak, V.M.; Milotová, M.K.; Jandová, K.; Pokorný, J.; Langmeier, M. Morphological changes in the hippocampus following nicotine and kainic acid administration. Physiol. Res. 2007, 56, 641–649. [Google Scholar]
- Valentine, G.; Sofuoglu, M. Cognitive Effects of Nicotine: Recent Progress. Curr. Neuropharmacol. 2018, 16, 403–414. [Google Scholar] [CrossRef] [PubMed]
- Akkoc, R.F.; Ogeturk, M. The Prefrontal Cortex: A Basic Embryological, Histological, Anatomical, and Functional Guideline. J. Hum. Anat. Physiol. 2017, 1, 4. [Google Scholar]
- Qasim, H.; Karim, Z.A.; Rivera, J.O.; Khasawneh, F.T.; Alshbool, F.Z. Impact of Electronic Cigarettes on the Cardiovascular System. J. Am. Heart Assoc. 2017, 6, 1–14. [Google Scholar] [CrossRef]
- Shanmugam, K.R.; Ramakrishna, C.H.; Mallikarjuna, K.; Reddy, K.S. Protective effect of ginger against alcohol-induced renal damage and antioxidant enzymes in male albino rats. Indian J. Exp. Biol. 2010, 48, 143–149. [Google Scholar] [PubMed]
- Huang, C.N.; Horng, J.S.; Yin, M.C. Antioxidative and antiglycative effects of six organo sulfur compounds in low-density lipoprotein and plasma. J. Agric. Food Chem. 2004, 52, 3674–3678. [Google Scholar] [CrossRef]
- Habib, S.H.; Makpol, S.; Hamid, N.A.A.; Das, S.; Ngah, W.Z.W.; Yusof, Y.A.M. Ginger extract (Zingiber officinale) has anti-cancer and anti-inflammatory effects on ethionine-induced hepatoma rats. Clinics 2008, 63, 807–813. [Google Scholar] [CrossRef] [Green Version]
- Shanmugam, K.R.; Mallikarjuna, K.; Kesireddy, N.; Reddy, K.S. Neu-roprotective effect of ginger on anti-oxidant enzymes in streptozotocin-induced diabetic rats. Food Chem. Toxicol. 2011, 49, 893–897. [Google Scholar] [CrossRef]
- Tsakanova, G.V.; Ayvazyan, V.A.; Boyajyan, A.S.; Arakelova, E.A.; Grigoryan, G.S.; Guevorkyan, A.A.; Mamikonyan, A.A. A comparative study of antioxidant system and intensity of lipid peroxidation in type 2 diabetes mellitus and ischemic stroke aggravated and not aggravated by type 2 diabetes mellitus. Bull. Exp. Biol. Med. 2011, 151, 564–566. [Google Scholar] [CrossRef]
- Medagama, A.B. The glycaemic outcomes of Cinnamon, a review of the experimental evidence and clinical trials. Nutr. J. 2015, 14, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Rao, P.V.; Gan, S.H. Cinnamon: A multifaceted medicinal plant. Evid.-Based Complement. Altern. Med. 2014, 2014, 1–12. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dutta, A.; Chakraborty, A. Cinnamon in Anticancer Armamentarium: A Molecular Approach. J. Toxicol. 2018, 2018, 8978731. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yulug, B.; Kilic, E.; Altunay, S.; Ersavas, C.; Orhan, C.; Dalay, A.; Mehmet, T.; Nurhan, S.; Vijaya, J.; Sahin, K. Cinnamon polyphenol extract exerts neuroprotective activity in traumatic brain injury through modulation of Nfr2 and cytokine expression. CNS Neurol. Disord.-Drug Targets 2018, 17, 439–447. [Google Scholar] [CrossRef]
- Sohrabi, R.; Pazgoohan, N.; Seresht, H.R.; Amin, B. Repeated systemic administration of the cinnamon essential oil possesses anti-anxiety and anti-depressant activities in mice Iran. Iran. J. Basic Med. Sci. 2017, 20, 708–714. [Google Scholar] [CrossRef]
- Masuda, Y.; Kikuzaki, H.; Hisamoto, M.; Nakatani, N. Antioxidant properties of gingerol related compounds from ginger. Biofactors 2004, 21, 293–306. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, E.S. The use of lead citrate of high pH as an electron opaque stain in electron microscopy. J. Cell Biol. 1963, 17, 208. [Google Scholar] [CrossRef] [Green Version]
- Levin, E.D.; Rezvani, A. Nicotinic interactions with antipsychotic drugs, models of schizophrenia and impacts on cognitive function. Biochem. Pharmacol. 2007, 74, 1182–1191. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, Y.; Tran, V.H.; Duke, C.C.; Roufogalis, B.D. Preventive and protective properties of Zingiber officinale (ginger) in diabetes mellitus, diabetic complications, and associated lipid and other metabolic disorders: A brief review. Evid.-Based Complement. Altern. Med. 2012, 2012, 516870. [Google Scholar] [CrossRef] [Green Version]
- Mahmoud, F.Y.; Abou Elghait, A.T.; Abdel Aziz, H.A.; Mohamed, H. Effect of chronic nicotine administration on the anterior cingulate cortex (area 24a) of adult albino rats: A histological and immunohistochemical study. Egypt. J. Histol. 2013, 36, 149–163. [Google Scholar] [CrossRef]
- Mansour, A.; Fox, C.A.; Burke, S.; Meng, F.; Thompson, R.C.; Akil, H.; Watson, S.J. Mu, delta, and kappa opioid receptor mRNA expression in the rat CNS: An in situ hybridization study. J. Comp. Neurol. 1994, 350, 412–438. [Google Scholar] [CrossRef]
- Matta, S.G.; Elberger, A.J. Combined exposure to nicotine and ethanol throughout full gestation results in enhanced acquisition of nicotine self-administration in young adult rat offspring. Psychopharmacology 2007, 193, 199–213. [Google Scholar] [CrossRef]
- Dunnet, S.B.; Martel, F.L. Proactive interference effects on short-term memory in rats, Basic parameters and drug effects. Behav. Neurosci. 1990, 104, 655–665. [Google Scholar] [CrossRef]
- Heisham, S.J.; Kleykamp, B.A.; Singleton, E. Meta-analysis of the acute effects of nicotine and smoking on human performance. Psychopharmacology 2010, 210, 453–469. [Google Scholar] [CrossRef] [Green Version]
- Spilich, G.J.; June, L.; Renner, J. Cigarette smoking and cognitive performance. Br. J. Addict. 1992, 87, 1313–1326. [Google Scholar] [CrossRef]
- Mishra, A.; Chaturvedi, P.; Datta, S.; Sinukumar, S.; Joshi, P.; Garg, A. Harmful effect of nicotine. Indian J. Med. Paediatr. Oncol. 2015, 36, 24–31. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Toda, N.; Okamura, T. Cigarette smoking impairs nitric oxide- mediated cerebral blood flow increase: Implication for Alzheimer’s disease. J. Pharmacol. Sci. 2016, 131, 223–232. [Google Scholar] [CrossRef] [Green Version]
- Budzynska, A.; Boguszewska-Czubara, M.; Kruk-Slomka, J.; Biala, G. Mephedrone and nicotine: Oxidative stress and behavioral interactions in animal models. Neurochem. Res. 2015, 40, 1083–1093. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mons, U.; Muscat, J.E.; Modesto, J.; Richie, J.P., Jr.; Brenner, H. Effect of smoking reduction and cessation on the plasma levels of the oxidative stress biomarker glutathione—Post-hoc analysis of data from a smoking cessation trial. Free Radic. Biol. Med. 2016, 91, 172–177. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zarei, M.; Uppin, V.; Acharya, P.; Talahalli, R. Ginger and turmeric lipid-soluble attenuate heated oil-induced oxidative stress in the brain via the upregulation of NRF2 and improve cognitive function in rats. Metab. Brain Dis. 2021, 36, 225–238. [Google Scholar] [CrossRef] [PubMed]
- Ashafaq, M.; Hussain, S.; Alshahrani, S.; Madkhali, O.; Siddiqui, R.; Khuwaja, G.; Alam, M.I.; Islam, F. Role of cinnamon oil against acetaminophen overdose induced neurological aberrations through brain stress and cytokine upregulation in rat brain. Drug Chem. Toxicol. 2020, 5, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Khaki, A.; Khaki, A.A.; Hajhosseini, L.; Golzar, F.S.; Ainehchi, N. The anti-oxidant effects of ginger and cinnamon on spermatogenesis dys-function of diabetes rats. Afr. J. Tradit. Complement. Altern. Med. 2014, 11, 1–8. [Google Scholar] [CrossRef] [Green Version]
- Khanna, A.; Guo, M.; Mehra, M.; Royal, W., III. Inflammation and oxidative stress induced by cigarette smoke in Lewis rat brains. J. Neuroimmunol. 2013, 254, 69–75. [Google Scholar] [CrossRef] [Green Version]
- El-Akabawy, G.; El-Kholy, W. Neuroprotective effect of ginger in the brain of streptozotocin-induced diabetic rats. Ann. Anat. 2014, 196, 119–128. [Google Scholar] [CrossRef]
- Lee, S.C.; Wang, S.Y.; Li, C.C.; Liu, C.T. Anti-inflammatory effect of cinnamaldehyde and linalool from the leaf essential oil of Cinnamomum osmophloeum Kanehira in endotoxin-induced mice. J. Food Drug Anal. 2018, 26, 211–220. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Takarada, T.; Nakamichi, N.; Kawagoe, H.; Ogura, M.; Fukumori, R.; Nakazato, R.; Fujikawa, K.; Kou, M.; Yoneda, Y. Possible neuroprotective property of nicotinic acetylcholine receptors in association with predominant upregulation of glial cell line derived neurotrophic factor in astrocytes. J. Neurosci. Res. 2012, 90, 2074–2085. [Google Scholar] [CrossRef]
- Sofroniew, M.V.; Vinters, H.V. Astrocytes: Biology and pathology. Acta Neuropathol. 2010, 119, 7–35. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baydas, G.; Reiter, R.J.; Yasar, A.; Tuzcu, M.; Akdemir, I.; Nedzvetskii, V.S. Melatonin reduces glial reactivity in the hippocampus, cortex, and cerebellum of streptozotocin-induced diabetic rats. Free Radic. Biol. Med. 2003, 35, 797–804. [Google Scholar] [CrossRef]
- Duarte, J.M.; Agostinho, P.M.; Carvalho, R.A.; Cunha, R.A. Caffeine consumption prevents diabetes-induced memory impairment and synaptotoxicity in the hippocampus of NONcZNO10/LTJ mice. PLoS ONE 2012, 7, e21899. [Google Scholar] [CrossRef] [Green Version]
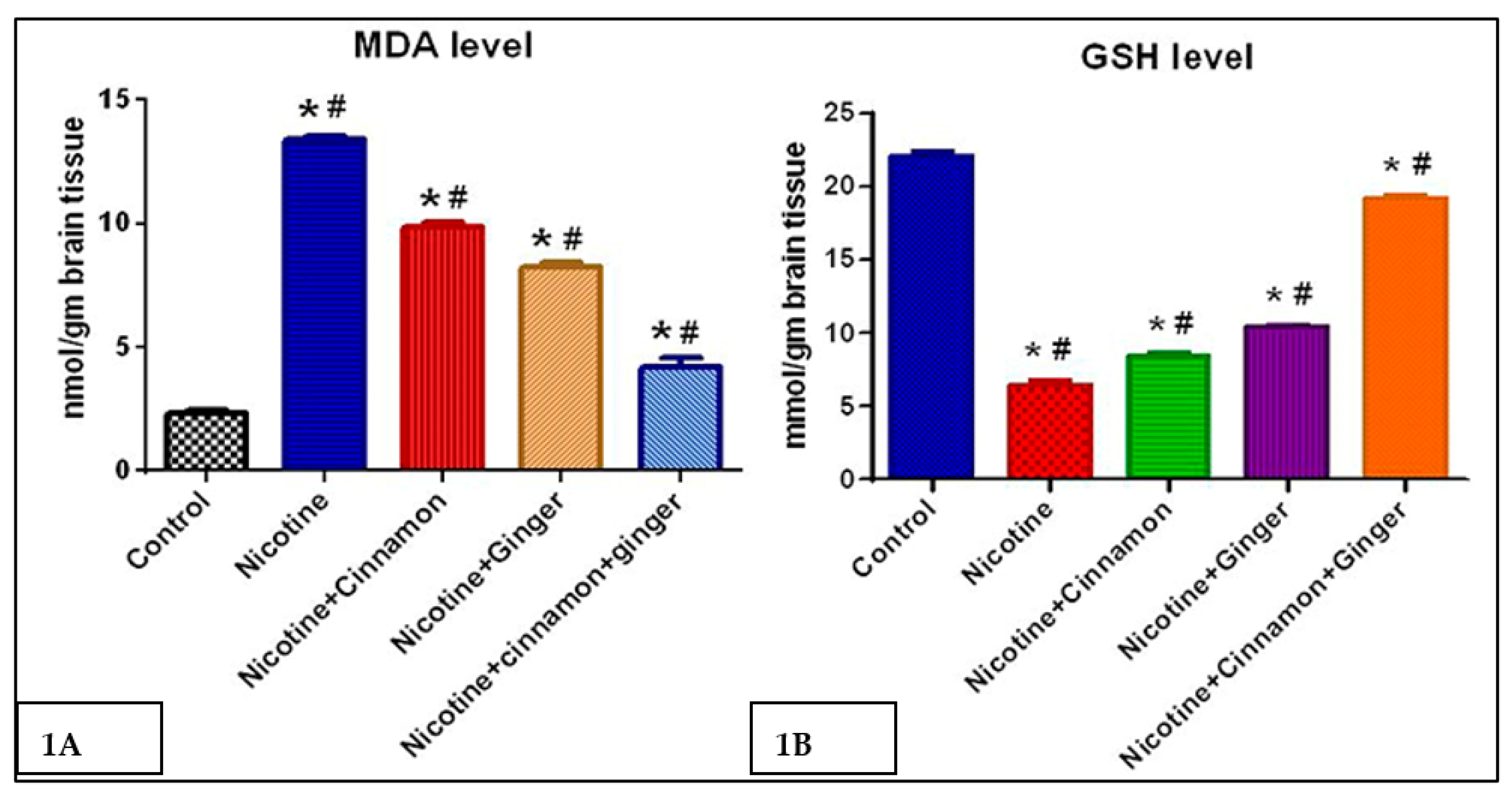

| Group | Control | Nicotine | Nicotine + Cinnamon | Nicotine + Ginger | Nicotine + Cinnamon + Ginger |
|---|---|---|---|---|---|
| Mean ± SEM | 2.317 ± 0.065 | 13.381 ± 0.060 *# | 9.850 ± 0.076 *# | 8.250 ± 0.067 *# | 4.217 ± 0.135 *# |
| Group | Control | Nicotine | Nicotine + Cinnamon | Nicotine + Ginger | Nicotine + Cinnamon + Ginger |
|---|---|---|---|---|---|
| Mean ± SEM | 22.13 ± 0.125 | 6.533 ± 0.108 *# | 8.421 ± 0.091 *# | 10.431 ± 0.061 *# | 19.28 ± 0.070 *# |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Taha, M.; Ibrahim, M.M.; Eldesoqui, M.; Iesa, M.A.M.; Baokbah, T.A.S.; Sonpol, H.M.A. Ameliorating Effect of Combined Cinnamon and Ginger Oils against the Neurotoxicity of Nicotine Administration on the Prefrontal Cortex of Adult Albino Rats: Immunohistochemical and Ultrastructural Study. Sci. Pharm. 2021, 89, 41. https://doi.org/10.3390/scipharm89030041
Taha M, Ibrahim MM, Eldesoqui M, Iesa MAM, Baokbah TAS, Sonpol HMA. Ameliorating Effect of Combined Cinnamon and Ginger Oils against the Neurotoxicity of Nicotine Administration on the Prefrontal Cortex of Adult Albino Rats: Immunohistochemical and Ultrastructural Study. Scientia Pharmaceutica. 2021; 89(3):41. https://doi.org/10.3390/scipharm89030041
Chicago/Turabian StyleTaha, Medhat, Mohie Mahmoud Ibrahim, Mamdouh Eldesoqui, Mohamed A. M. Iesa, Tourki A. S. Baokbah, and Hany M. A. Sonpol. 2021. "Ameliorating Effect of Combined Cinnamon and Ginger Oils against the Neurotoxicity of Nicotine Administration on the Prefrontal Cortex of Adult Albino Rats: Immunohistochemical and Ultrastructural Study" Scientia Pharmaceutica 89, no. 3: 41. https://doi.org/10.3390/scipharm89030041
APA StyleTaha, M., Ibrahim, M. M., Eldesoqui, M., Iesa, M. A. M., Baokbah, T. A. S., & Sonpol, H. M. A. (2021). Ameliorating Effect of Combined Cinnamon and Ginger Oils against the Neurotoxicity of Nicotine Administration on the Prefrontal Cortex of Adult Albino Rats: Immunohistochemical and Ultrastructural Study. Scientia Pharmaceutica, 89(3), 41. https://doi.org/10.3390/scipharm89030041







