The Effect of Wheatgrass Lyophilizate on Blood Clotting Time in Rats
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Substances
2.2. Sample Preparation Method for HPLC Analysis
2.3. Flavonoid Content Determination
2.4. Animals and Husbandry
2.5. Warfarin and Wheatgrass Administration
2.6. Blood Samples
2.7. Clotting Assays
2.8. Statistical Analysis
3. Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Biographical Information about Charles F. Schnabel. Available online: https://web.archive.org/web/20190118134018/https://cerophyl.net/cerophyl-1937.html (accessed on 10 August 2021).
- Marawaha, R.K.; Bansal, D.; Kaur, S.; Trehan, A. Wheat grass juice reduces transfusion requirement in patients with thalassemia major: A pilot study. Indian Pediatr. 2004, 41, 716–720. [Google Scholar] [PubMed]
- Gore, R.D.; Palaskar, S.J.; Bartake, A.R. Wheatgrass: Green Blood can Help to Fight Cancer. J. Clin. Diagn. Res. 2017, 11, ZC40–ZC42. [Google Scholar] [CrossRef]
- Das, P.; Mukhopadhyay, S.; Mandal, S.; Chakraborty, A.; Pal, A.; Sarkar, N.K.; Mukhopadhyay, A. Acute toxicity test of a natural iron chelator and an antioxidant, extracted from Triticum aestivum Linn. (wheat grass). Nat. Prod. Res. 2014, 28, 1379–1382. [Google Scholar] [CrossRef]
- Mutha, A.S.; Shah, K.U.; Kinikar, A.A.; Ghongane, B.B. Efficacy and safety of wheat grass in thalassemic children on regular blood transfusion. Cureus 2018, 10, e2306. [Google Scholar] [CrossRef] [PubMed]
- Illingworth, C.F.W. Haemorrthage in jaundice. Lancet 1939, 233, 1031–1035. [Google Scholar] [CrossRef]
- Ben-Arye, E.; Goldin, E.; Wengrower, D.; Stamper, A.; Kohn, R.; Berry, E. Wheat grass juice in the treatment of active distal ulcerative colitis: A randomized double-blind placebo-controlled trial. Scand. J. Gastroenterol. 2002, 37, 444–449. [Google Scholar] [CrossRef]
- Tirgar, P.R.; Shah, K.V.; Thumber, B.L.; Desai, R. Investigation into therapeutic role of Triticum aestivum (wheat) grass in busulfan induce thrombocytopenia. Int. J. Univers. Pharm. Life Sci. 2011, 1, 85–97. [Google Scholar]
- Shermer, M. Wheatgrass juice and folk medicine. Sci. Am. 2008, 299, 42. [Google Scholar] [CrossRef]
- Akbas, E.; Kilercioglu, M.; Onder, O.N.; Koker, A.; Soyler, B.; Oztop, M.H. Wheatgrass juice to wheat grass powder: Encapsulation, physical and chemical characterization. J. Funct. Foods 2017, 28, 19–27. [Google Scholar] [CrossRef]
- Bitra, V.R.; Rapaka, D.; Mathala, N.; Akula, A. Effect of wheat grass powder on aluminum induced Alzheimer’s disease in Wistar rats. Asian Pac. J. Trop. Med. 2014, 7S1, S278–S281. [Google Scholar] [CrossRef]
- Shyam, R.; Singh, S.N.; Vats, P.; Singh, V.K.; Bajaj, R.; Singh, S.B.; Banerjee, P.K. Wheat grass supplementation decreases oxidative stress in healthy subjects: A comparative study with spirulina. J. Altern. Complement. Med. 2007, 13, 789–791. [Google Scholar] [CrossRef]
- Das, A.; Raychaudhuri, U.; Chakraborty, R. Effect of freeze drying and oven drying on antioxidant properties of fresh wheatgrass. Int. J. Food Sci. Nutr. 2012, 63, 718–721. [Google Scholar] [CrossRef]
- Chomchan, R.; Siripongvutikorn, A.P.D.S.; Puttarak, D.P.; Rattanapon, M.R. Investigation of phytochemical constituents, phenolic profiles and antioxidant activities of ricegrass juice compared to wheatgrass juice. Funct. Foods Heal. Dis. 2016, 6, 822. [Google Scholar] [CrossRef]
- Durairaj, V.; Hoda, M.; Shakya, G.; Babu, S.P.P.; Rajagopalan, R. Phytochemical screening and analysis of antioxidant properties of aqueous extract of wheatgrass. Asian Pac. J. Trop. Med. 2014, 7S1, S398–S404. [Google Scholar] [CrossRef]
- Jangle, S.N.; Padmanabhan, P. Evaluation of phytochemicals, reducing power, antioxidant activity and in-vitro lipid peroxidation activity of wheat grass juice. Int. J. Pharm. Sci. Res. 2016, 7, 3436–3440. [Google Scholar] [CrossRef]
- Ozkose, A.; Arslan, D.; Acar, A. The comparison of the chemical composition, sensory, phenolic and antioxidant properties of juices from different wheatgrass and turfgrass species. Not. Bot. Horti Agrobot. Cluj-Napoca 2016, 44, 499–507. [Google Scholar] [CrossRef]
- Rajoria, A.; Mehta, A.; Mehta, P.; Ahirwal, L.; Shukla, S. Phytochemical analysis and estimation of major bioactive compounds from Triticum aestivum L. grass with antimicrobial potential. Pak. J. Pharm. Sci. 2015, 28, 2221–2225. [Google Scholar]
- Skoczylas, L.; Korus, A.; Tabaszewska, M.; Gedos, K.; Szczepanska, E. Evaluation of the quality of fresh and frozen wheatgrass juices depending on the time of grass harvest. J. Food Process. Preserv. 2018, 42, e13401. [Google Scholar] [CrossRef]
- Peryt, B.; Szymczyk, T.; Lesca, P. Mechanism of antimutagenicity of wheat sprout extracts. Mutat. Res. 1992, 269, 201–215. [Google Scholar] [CrossRef]
- Wagner, H.; Obermeier, G.; Chari, V.M.; Galle, K. Flavonoid-C-glycosides from Triticum aestivum L. J. Nat. Prod. 1980, 43, 583–587. [Google Scholar] [CrossRef]
- Bar-Sela, G.; Cohen, M.; Ben-Arye, E.; Epelbaum, R. The medical use of wheatgrass: Review of the gap between basic and clinical applications. Mini Rev. Med. Chem. 2015, 15, 1002–1010. [Google Scholar] [CrossRef] [PubMed]
- Alitheen, N.B.; Oon, C.L.; Keong, Y.S.; Chuan, T.K.; Li, H.K.; Yong, H.W. Cytotoxic effects of commercial wheatgrass and fiber towards human acute promyelocytic leukemia cells (HL60). Pak. J. Pharm. Sci. 2011, 24, 243–250. [Google Scholar] [PubMed]
- Rajoria, A.; Mehta, A.; Mehta, P.; Ahirwal, L.; Shukla, S.; Bajpai, V.K. Evaluation of antiproliferative and hepatoprotective effects of wheat grass (Triticum aestivum). Acta Biol. Hung. 2017, 68, 150–161. [Google Scholar] [CrossRef][Green Version]
- Hussain, A.; Gheewala, T.M.; Vas, A.J.; Shah, K.; Goala, P.; Khan, S.; Hinduja, S.; Sharma, C. Growth inhibitory and adjuvant therapeutic potential of aqueous extract of Triticum aestivum on MCF-7 and HeLa cells. Exp. Oncol. 2014, 36, 9–16. [Google Scholar]
- Shakya, G.; Balasubramanian, S.; Hoda, M.; Rajagopalan, R. Inhibition of metastasis and angiogenesis in Hep-2 cells by wheatgrass extract - an in vitro and in silico approach. Toxicol. Mech. Methods 2018, 28, 205–218. [Google Scholar] [CrossRef]
- Bar-Sela, G.; Tsalic, M.; Fried, G.; Goldberg, H. Wheat grass juice may improve hematological toxicity related to chemotherapy in breast cancer patients: A pilot study. Nutr. Cancer 2007, 58, 43–48. [Google Scholar] [CrossRef]
- Lalsolanki, M.S.J.K.; Bhaidpatel, L. Clinical efficacy evaluation of wheat grass tablets as supportive treatment in leukemia patients. Res. J. Pharm. Biol. Chem. Sci. 2015, 6, 1450–1454. [Google Scholar]
- Choi, D.H.; Piao, Y.J.; Choi, E.J.; Choi, J.S.; Burm, J.P. Effects of amlodipine on the pharmacokinetics of warfarin after oral and intravenous administration of warfarin in rats. Biomol. Ther. 2011, 19, 493–497. [Google Scholar] [CrossRef]
- Vranckx, P.; Valgimigli, M.; Heidbuchel, H. The significance of drug–drug and drug–food interactions of oral anticoagulation. Arrhythmia Electrophysiol. Rev. 2018, 7, 55. [Google Scholar] [CrossRef]
- Leite, P.M.; Martins, M.A.P.; Castilho, R.O. Review on mechanisms and interactions in concomitant use of herbs and warfarin therapy. Biomed. Pharmacother. 2016, 83, 14–21. [Google Scholar] [CrossRef]
- Nutescu, E.; Chuatrisorn, I.; Hellenbart, E. Drug and dietary interactions of warfarin and novel oral anticoagulants: An update. J. Thromb. Thrombolysis 2011, 31, 326–343. [Google Scholar] [CrossRef]
- Bohm, B.A. Introduction to Flavonoids; Harwood Academic Publishers: Amsterdam, The Netherlands, 1998. [Google Scholar]
- Dong, H.; Ma, J.; Li, T.; Xiao, Y.; Zheng, N.; Liu, J.; Gao, Y.; Shao, J.; Jia, L. Global deregulation of ginseng products may be a safety hazard to warfarin takers: Solid evidence of ginseng-warfarin interaction. Sci. Rep. 2017, 7, 5813. [Google Scholar] [CrossRef]
- Guidance for Industry Estimating the Maximum Safe—Starting Dose in Initial Clinical Trials for Therapeutics in Adult Healthy Volunteers. 2005. Available online: https://www.fda.gov/media/72309/download (accessed on 10 August 2021).
- Kiss, T.; Szabo, A.; Oszlanczi, G.; Lukacs, A.; Tõmar, Z.; Tiszlavicz, L.; Csupor, D. Repeated-dose toxicity of common ragweed on rats. PLoS One 2017, 12, e0176818. [Google Scholar] [CrossRef]
- Rahman, H.; Kim, M.; Leung, G.; Green, J.A.; Katz, S. Drug-herb interactions in the elderly patient with IBD: A growing concern. Curr. Treat. Options Gastroenterol. 2017, 15, 618–636. [Google Scholar] [CrossRef]
- Petry, J.J. Garlic and postoperative bleeding. Plast. Reconstr. Surg. 1995, 96, 483–484. [Google Scholar] [CrossRef]
- Sunter, W.H. Warfarin and garlic. Pharm. J. 1991, 246, 772–775. [Google Scholar]
- Evans, V. Herbs and the brain: Friend or foe? The effects of ginkgo and garlic on warfarin use. J. Neurosci. Nurs. 2000, 32, 229–232. [Google Scholar] [CrossRef]
- Ge, B.; Zhang, Z.; Zuo, Z. Updates on the clinical evidenced herb-warfarin interactions. Evid.-Based Complement. Altern. Med. 2014, 2014, 957362. [Google Scholar] [CrossRef]
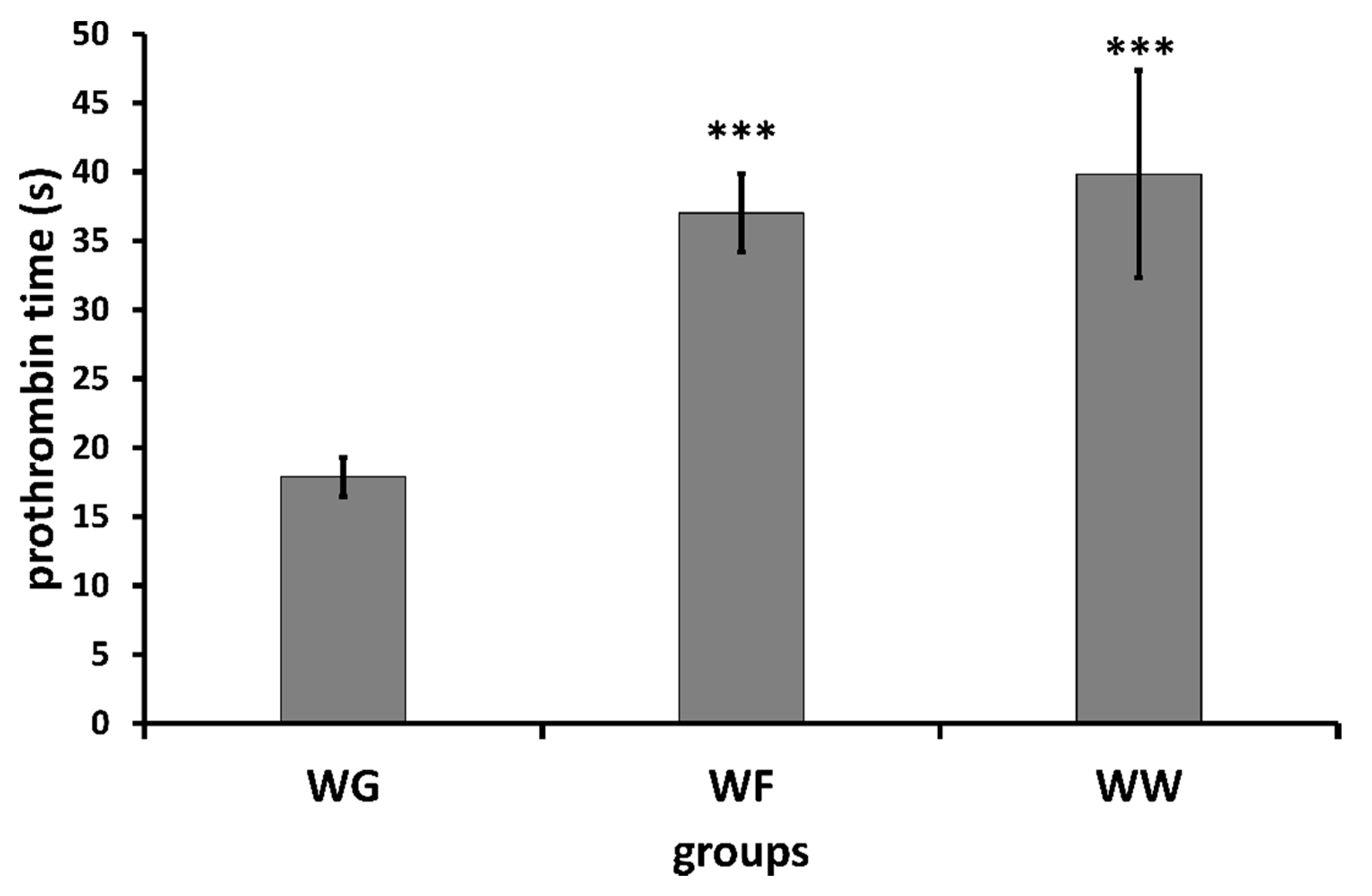
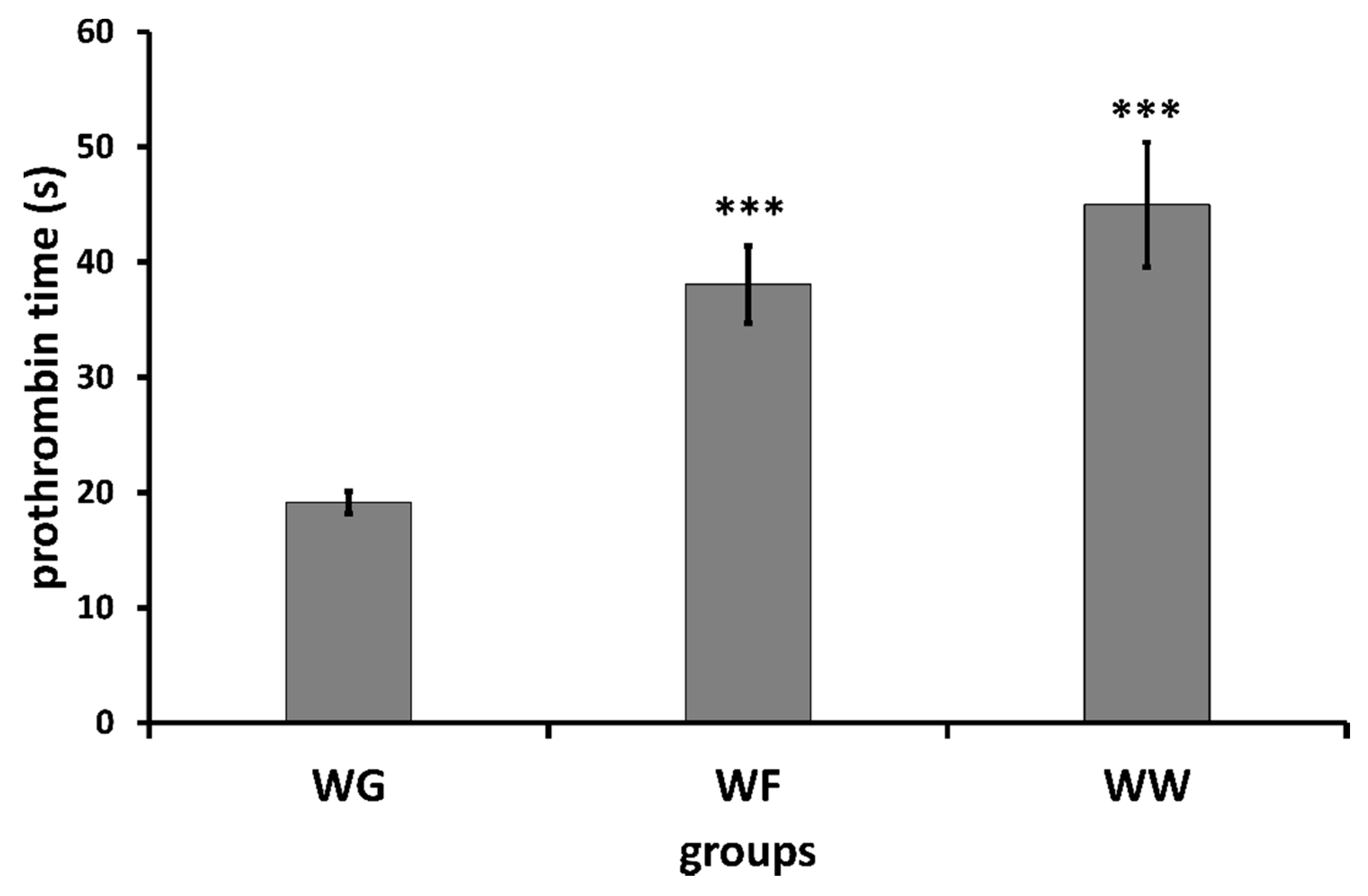
| Group | Prothrombin Time (in Seconds) Determined by Means of Mechanical Coagulometer | Prothrombin Time (in Seconds) Determined by Means of Optical Coagulometer | ||
|---|---|---|---|---|
| Before | After | Before | After | |
| WG | 16.48 ± 0.89 | 18.07 ± 1.48 ** | 19.06 ± 1.90 | 19.18 ± 1.00 |
| WF | 17.48 ± 0.62 | 38.10 ± 2.27 ** | 18.23 ± 0.55 | 38.06 ± 3.34 ** |
| WW | 17.60 ± 0.96 | 41.87 ± 9.47 * | 18.04 ± 0.74 | 44.98 ± 5.42 ** |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Takács, I.; Urkon, M.; Kiss, T.; Horváth, E.; Laczkó-Zöld, E.; Zomborszki, Z.P.; Lukács, A.; Oszlánczi, G.; Csupor, D.; Szabó, A. The Effect of Wheatgrass Lyophilizate on Blood Clotting Time in Rats. Sci. Pharm. 2021, 89, 39. https://doi.org/10.3390/scipharm89030039
Takács I, Urkon M, Kiss T, Horváth E, Laczkó-Zöld E, Zomborszki ZP, Lukács A, Oszlánczi G, Csupor D, Szabó A. The Effect of Wheatgrass Lyophilizate on Blood Clotting Time in Rats. Scientia Pharmaceutica. 2021; 89(3):39. https://doi.org/10.3390/scipharm89030039
Chicago/Turabian StyleTakács, István, Melinda Urkon, Tivadar Kiss, Edina Horváth, Eszter Laczkó-Zöld, Zoltán Péter Zomborszki, Anita Lukács, Gábor Oszlánczi, Dezső Csupor, and Andrea Szabó. 2021. "The Effect of Wheatgrass Lyophilizate on Blood Clotting Time in Rats" Scientia Pharmaceutica 89, no. 3: 39. https://doi.org/10.3390/scipharm89030039
APA StyleTakács, I., Urkon, M., Kiss, T., Horváth, E., Laczkó-Zöld, E., Zomborszki, Z. P., Lukács, A., Oszlánczi, G., Csupor, D., & Szabó, A. (2021). The Effect of Wheatgrass Lyophilizate on Blood Clotting Time in Rats. Scientia Pharmaceutica, 89(3), 39. https://doi.org/10.3390/scipharm89030039









