Enhancement of Antimicrobial and Antiproliferative Activities of Standardized Frankincense Extract Using Optimized Self-Nanoemulsifying Delivery System
Abstract
1. Introduction
2. Material and Methods
2.1. Materials
2.1.1. Chemicals
2.1.2. Microbial Strains and Cell Lines
2.2. Methods
2.2.1. Plant Extraction
2.2.2. HPLC Standardization
2.3. Formulation Development and Optimization of Fr-SNEDS
2.3.1. Screening of Different Excipients for Development of Fr-SNEDS
2.3.2. Experimental Design for Development and Optimization of Fr-SNEDS
2.4. Characterization of the Optimized Fr-SNEDS
2.4.1. Visual Inspection, DS, PDI, and Zeta Potential (ZP) Measurements
2.4.2. Assessment of Self-Emulsification Efficiency
2.4.3. Thermodynamic Stability
2.4.4. Study of Surface Morphology by Transmission Electron Microscopy (TEM)
2.5. Assessment of Antimicrobial Activity
2.5.1. Preparation of the Inoculum
2.5.2. Sensitivity Test
2.5.3. Minimum Inhibitory Concentrations (MICs) and Minimum Biocidal Concentrations (MBC)
2.6. In Vitro Antiproliferative Assay
2.7. Stability Study
3. Results
3.1. Standardization of BAs in fr-Extract
3.2. Formulation Development and Optimization of Fr-SNEDS
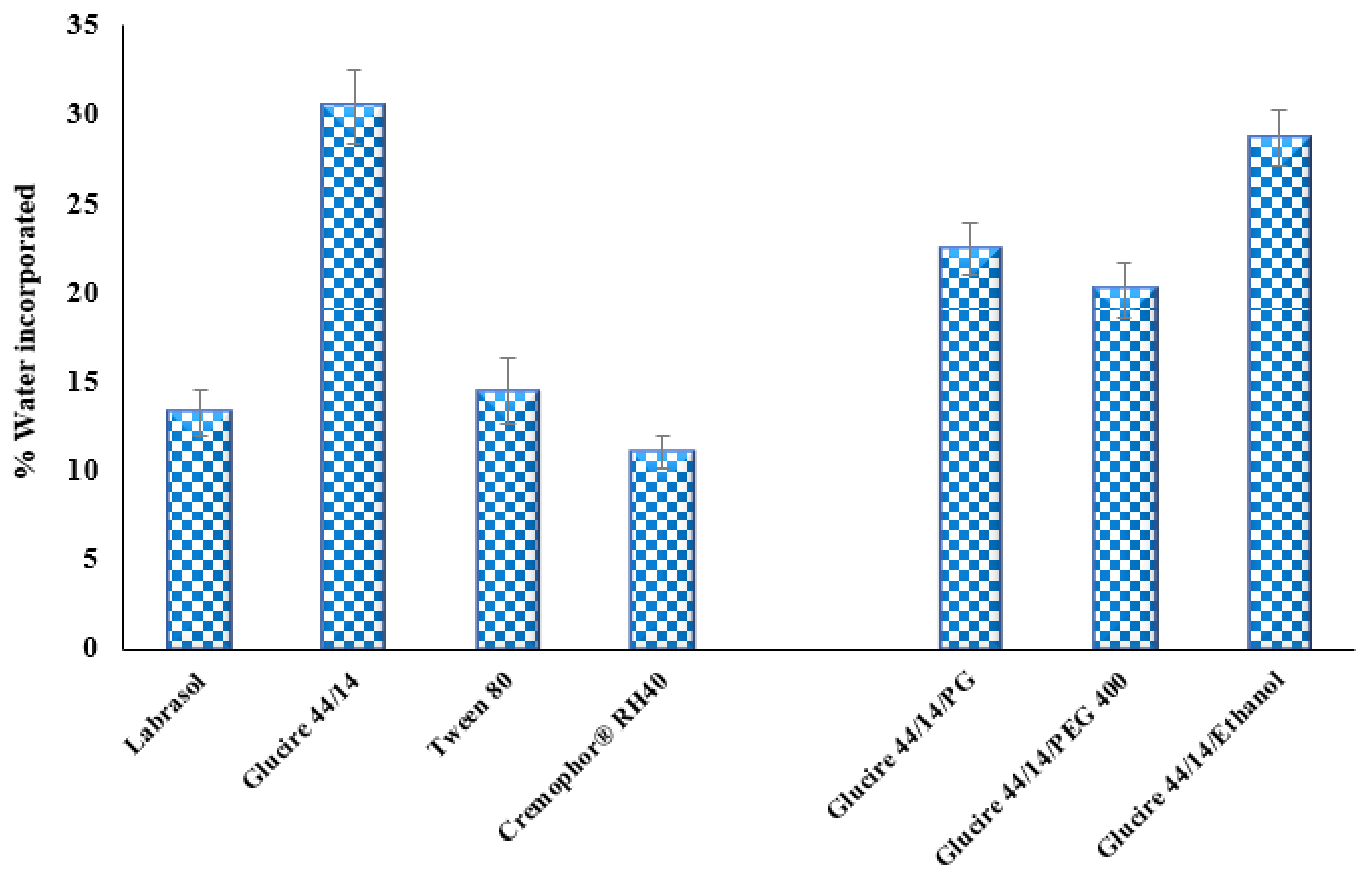
3.2.1. Screening of Excipients
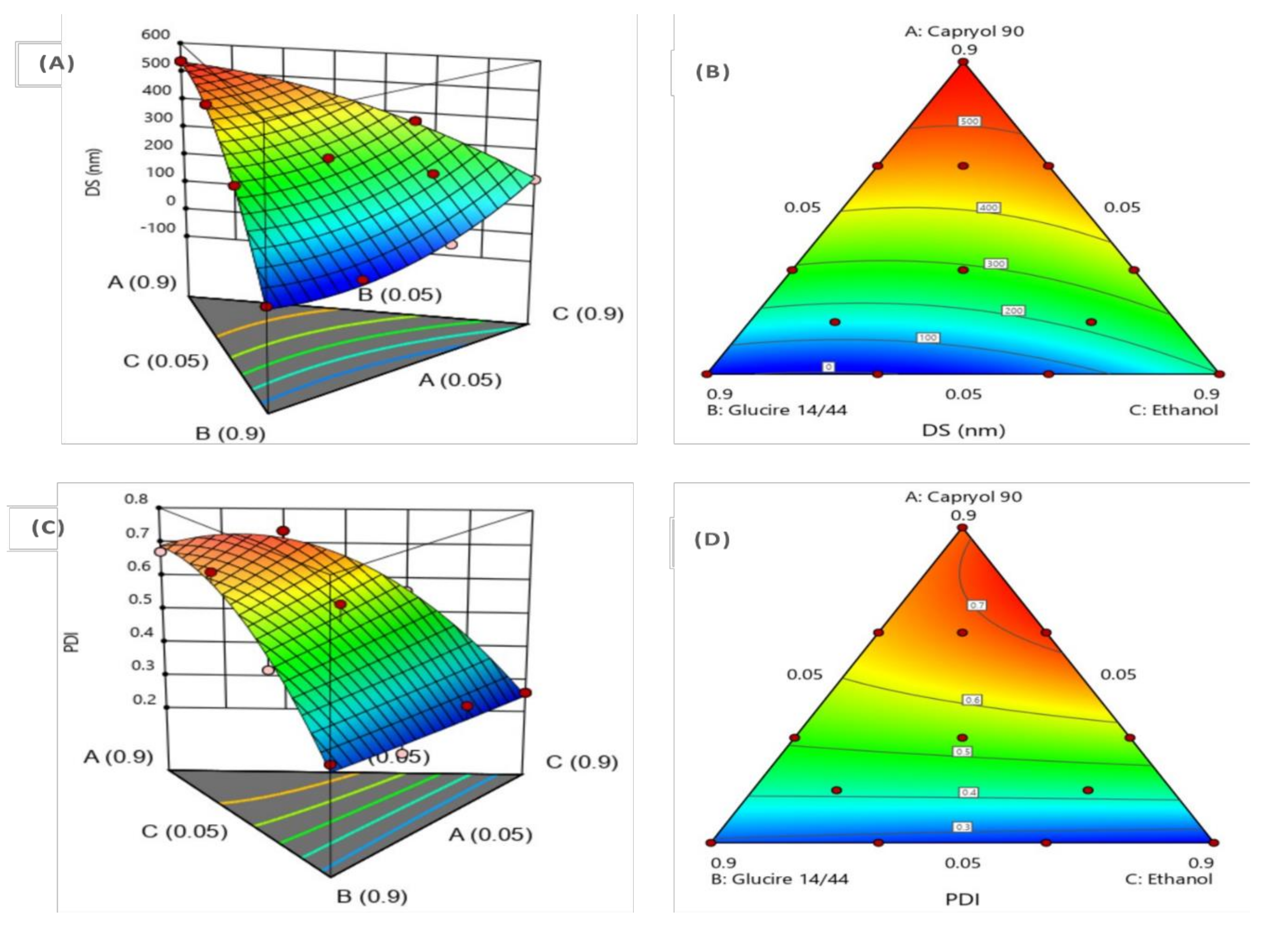
3.2.2. Experimental Design
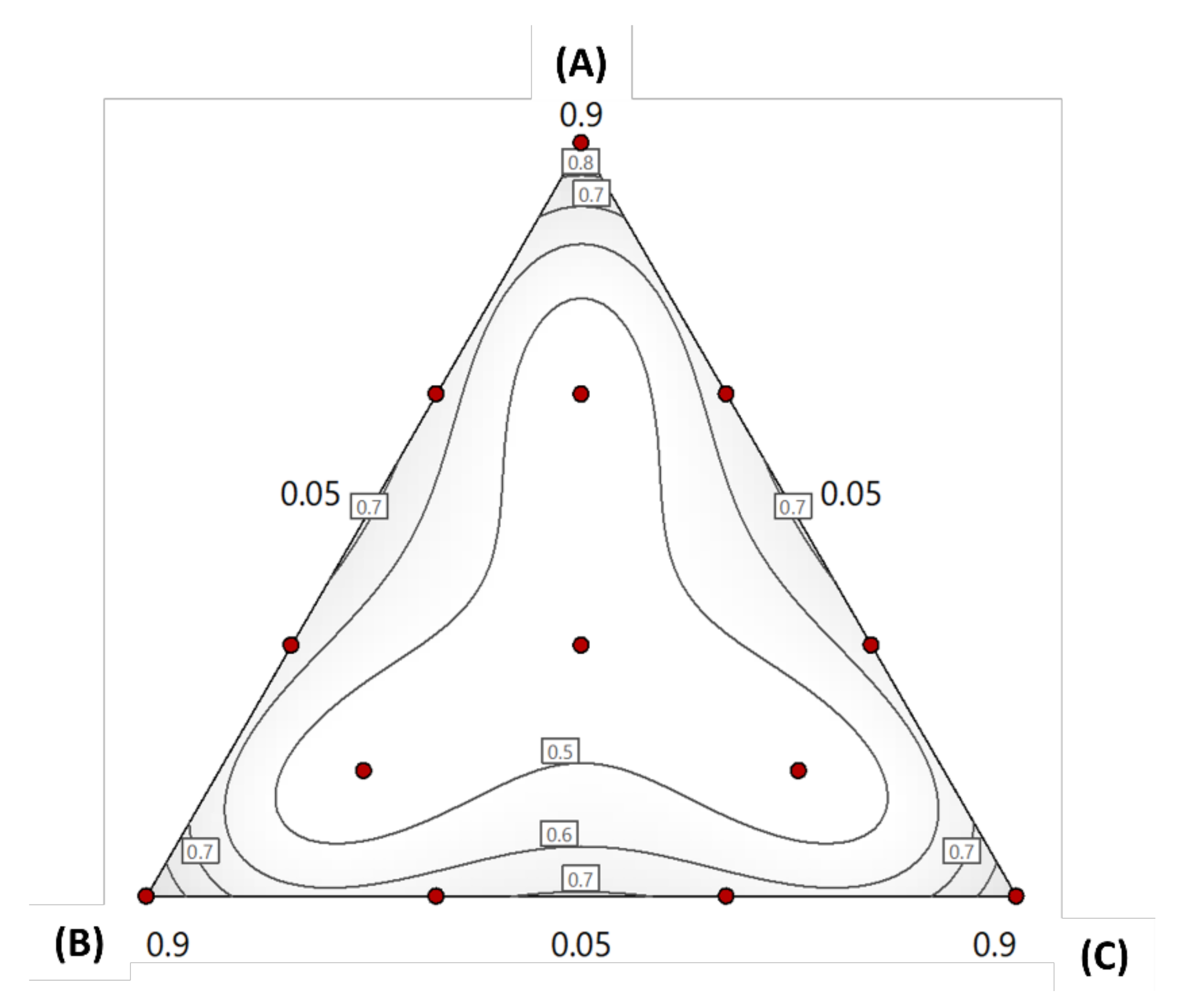
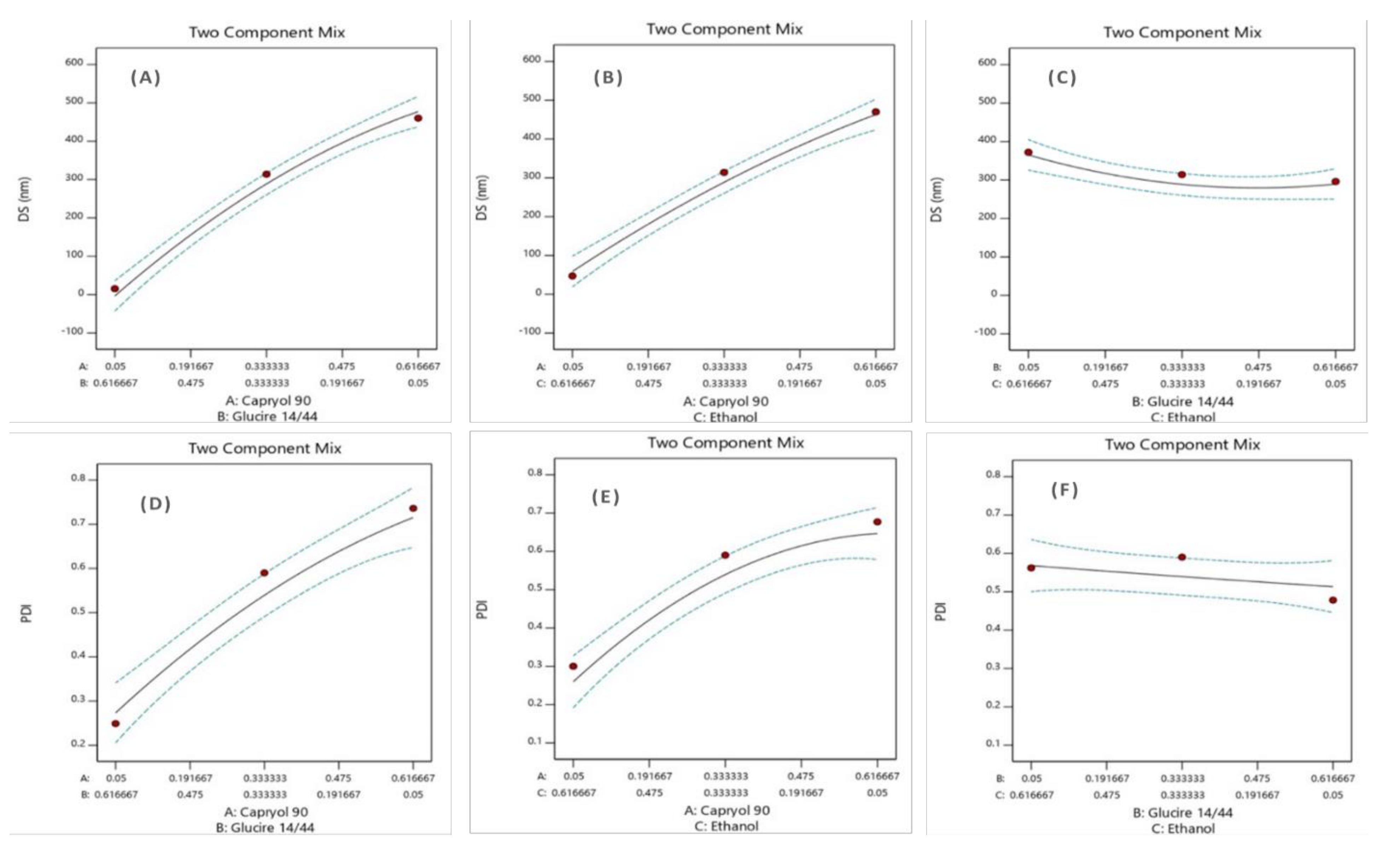
Model Statistical Analysis and ANOVA
Effect of Formulation Composition on DS
Effect of Formulation Composition on PDI
Formulation Optimization
3.3. Characterization of the Optimized Fr-SNEDS:
3.3.1. Visual Inspection, DS, PDI, and ZP Measurements and Assessment of Self-Emulsification
3.3.2. Thermodynamic Stability
3.3.3. Surface Morphology by TEM
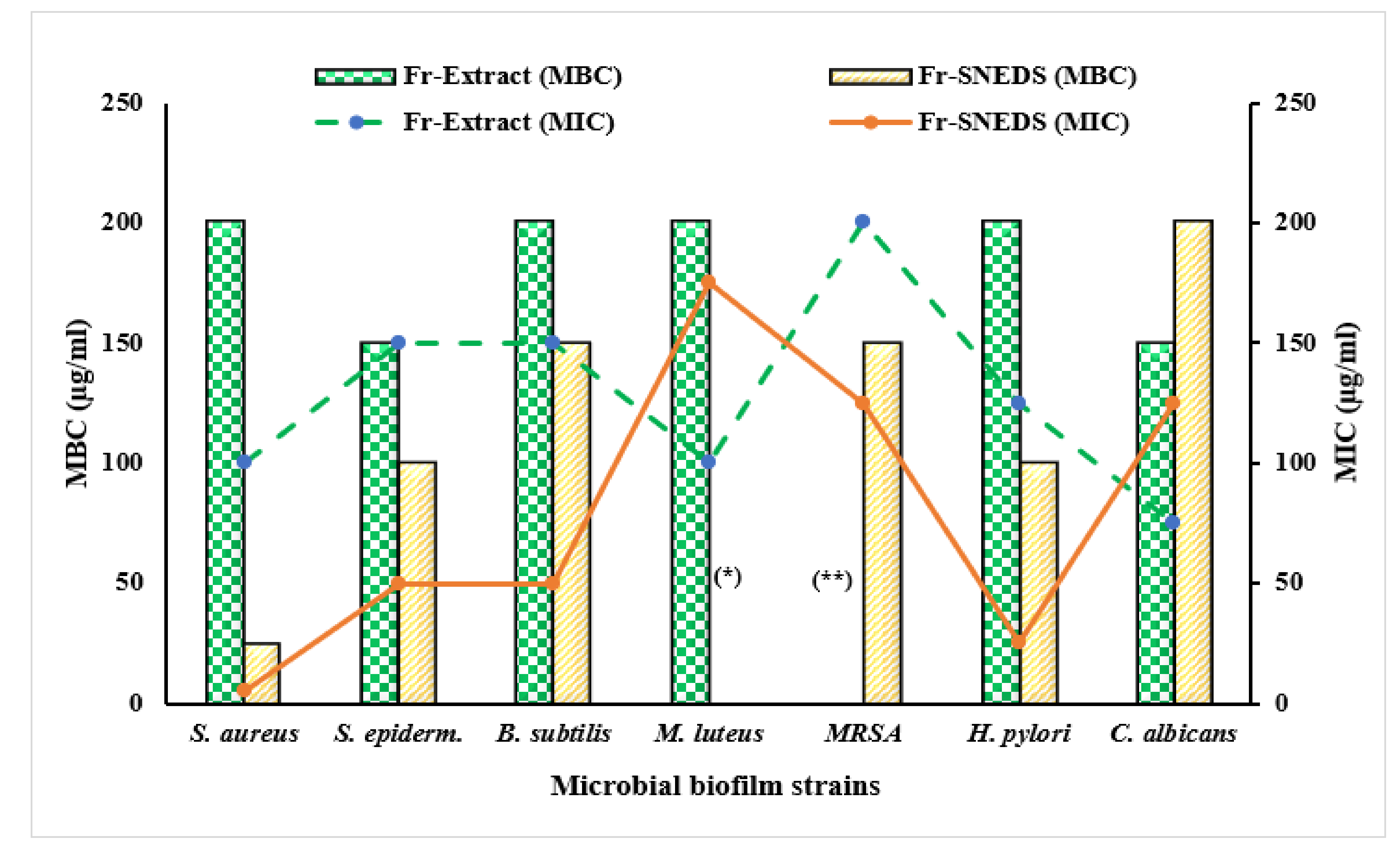
3.4. Assessment of Antimicrobial Activity
3.5. Antiproliferative Study
3.6. Stability Study
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Mannino, G.; Occhipinti, A.; Maffei, M.E. Quantitative Determination of 3-O-Acetyl-11-Keto-β-Boswellic Acid (AKBA) and Other Boswellic Acids in Boswellia sacra Flueck (syn. B. carteri Birdw) and Boswellia serrata Roxb. Molecules 2016, 21, 1329. [Google Scholar] [CrossRef]
- Chevrier, M.R.; Ryan, A.E.; Lee, D.Y.-W.; Zhongze, M.; Wu-Yan, Z.; Via, C.S. Boswellia carterii extract inhibits TH1 cytokines and promotes TH2 cytokines in vitro. Clin. Diagn. Lab. Immunol. 2005, 12, 575–580. [Google Scholar] [CrossRef]
- Büchele, B.; Zugmaier, W.; Simmet, T. Analysis of pentacyclic triterpenic acids from frankincense gum resins and related phytopharmaceuticals by high-performance liquid chromatography. Identification of lupeolic acid, a novel pentacyclic triterpene. J. Chromatogr. B 2003, 791, 21–30. [Google Scholar] [CrossRef]
- Frank, A.; Unger, M. Analysis of frankincense from various Boswellia species with inhibitory activity on human drug metabolising cytochrome P450 enzymes using liquid chromatography mass spectrometry after automated on-line extraction. J. Chromatogr. A 2006, 1112, 255–262. [Google Scholar] [CrossRef] [PubMed]
- Schmiech, M.; Ulrich, J.; Lang, S.J.; Büchele, B.; Paetz, C.; St-Gelais, A.; Syrovets, T.; Simmet, T. 11-Keto-α-Boswellic Acid, a Novel Triterpenoid from Boswellia spp. with Chemotaxonomic Potential and Antitumor Activity against Triple-Negative Breast Cancer Cells. Molecules 2021, 26, 366. [Google Scholar] [CrossRef]
- Raja, A.F.; Ali, F.; Khan, I.A.; Shawl, A.S.; Arora, D.S.; Shah, B.A.; Taneja, S.C. Antistaphylococcal and biofilm inhibitory activities of acetyl-11-keto-β-boswellic acid from Boswellia serrata. BMC Microbiol. 2011, 11, 54. [Google Scholar] [CrossRef]
- Alam, M.; Khan, H.; Samiullah, L.; Siddique, K.M. A review on phytochemical and pharmacological studies of Kundur (Boswellia serrata Roxb ex Colebr.)-A Unani drug. J. Appl. Pharm. Sci. 2012, 2, 148–156. [Google Scholar]
- Xia, L.; Chen, D.; Han, R.; Fang, Q.; Waxman, S.; Jing, Y. Boswellic acid acetate induces apoptosis through caspase-mediated pathways in myeloid leukemia cells. Mol. Cancer Ther. 2005, 4, 381–388. [Google Scholar] [CrossRef]
- Raja, A.F.; Ali, F.; Khan, I.A.; Shawl, A.S.; Arora, D.S. Acetyl-11-keto-β-boswellic acid (AKBA); targeting oral cavity pathogens. BMC Res. Notes 2011, 4, 406. [Google Scholar] [CrossRef]
- Tambe, A.; Mokashi, P.; Pandita, N. Ex-vivo intestinal absorption study of boswellic acid, cyclodextrin complexes and poloxamer solid dispersions using everted gut sac technique. J. Pharm. Biomed. Anal. 2019, 167, 66–73. [Google Scholar] [CrossRef]
- Kalhapure, R.S.; Suleman, N.; Mocktar, C.; Seedat, N.; Govender, T. Nanoengineered drug delivery systems for enhancing antibiotic therapy. J. Pharm. Sci. 2015, 104, 872–905. [Google Scholar] [CrossRef]
- Donsì, F.; Annunziata, M.; Vincensi, M.; Ferrari, G. Design of nanoemulsion-based delivery systems of natural antimicrobials: Effect of the emulsifier. J. Biotechnol. 2012, 159, 342–350. [Google Scholar] [CrossRef]
- El-Haddad, A.E.; Sheta, N.M.; Boshra, S.A. Isolation, Formulation, and Efficacy Enhancement of Morin Emulsified Carriers Against Lung Toxicity in Rats. AAPS PharmSciTech 2018, 19, 2346–2357. [Google Scholar] [CrossRef]
- Pápay, Z.E.; Kállai-Szabó, N.; Ludányi, K.; Klebovich, I.; Antal, I. Development of oral site-specific pellets containing flavonoid extract with antioxidant activity. Eur. J. Pharm. Sci. 2016, 95, 161–169. [Google Scholar] [CrossRef] [PubMed]
- Grove, M.; Müllertz, A.; Nielsen, J.L.; Pedersen, G.P. Bioavailability of seocalcitol II: Development and characterisation of self-microemulsifying drug delivery systems (SMEDDS) for oral administration containing medium and long chain triglycerides. Eur. J. Pharm. Sci. 2006, 28, 233–242. [Google Scholar] [CrossRef]
- Oh, D.H.; Kang, J.H.; Kim, D.W.; Lee, B.-J.; Kim, J.O.; Yong, C.S.; Choi, H.-G. Comparison of solid self-microemulsifying drug delivery system (solid SMEDDS) prepared with hydrophilic and hydrophobic solid carrier. Int. J. Pharm. 2011, 420, 412–418. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Dong, J.; Chen, J.; Eastoe, J.; Li, X. Design and optimization of a new self-nanoemulsifying drug delivery system. J. Colloid Interface Sci. 2009, 330, 443–448. [Google Scholar] [CrossRef] [PubMed]
- Nazari-Vanani, R.; Moezi, L.; Heli, H. In vivo evaluation of a self-nanoemulsifying drug delivery system for curcumin. Biomed. Pharmacother. 2017, 88, 715–720. [Google Scholar] [CrossRef]
- Hilbig, J.; Ma, Q.; Davidson, P.M.; Weiss, J.; Zhong, Q. Physical and antimicrobial properties of cinnamon bark oil co-nanoemulsified by lauric arginate and Tween 80. Int. J. Food Microbiol. 2016, 233, 52–59. [Google Scholar] [CrossRef] [PubMed]
- Alwadei, M.; Kazi, M.; Alanazi, F.K. Novel oral dosage regimen based on self-nanoemulsifying drug delivery systems for codelivery of phytochemicals—Curcumin and thymoquinone. Saudi Pharm. J. 2019, 27, 866–876. [Google Scholar] [CrossRef] [PubMed]
- Aldawsari, H.M.; Elfaky, M.A.; Fahmy, U.A.; Aljaeid, B.M.; Alshareef, O.A.; El-Say, K.M. Development of a fluvastatin-loaded self-nanoemulsifying system to maximize therapeutic efficacy in human colorectal carcinoma cells. J. Drug Deliv. Sci. Technol. 2018, 46, 7–13. [Google Scholar] [CrossRef]
- Batool, A.; Arshad, R.; Razzaq, S.; Nousheen, K.; Kiani, M.H.; Shahnaz, G. Formulation and evaluation of hyaluronic acid-based mucoadhesive self nanoemulsifying drug delivery system (SNEDDS) of tamoxifen for targeting breast cancer. Int. J. Biol. Macromol. 2020, 152, 503–515. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; McClements, D.J. Inhibition of lipase-catalyzed hydrolysis of emulsified triglyceride oils by low-molecular weight surfactants under simulated gastrointestinal conditions. Eur. J. Pharm. Biopharm. 2011, 79, 423–431. [Google Scholar] [CrossRef]
- Nazari-Vanani, R.; Azarpira, N.; Heli, H. Development of self-nanoemulsifying drug delivery systems for oil extracts of Citrus aurantium L. blossoms and Rose damascena and evaluation of anticancer properties. J. Drug Deliv. Sci. Technol. 2018, 47, 330–336. [Google Scholar] [CrossRef]
- Sanka, K.; Suda, D.; Bakshi, V. Optimization of solid-self nanoemulsifying drug delivery system for solubility and release profile of clonazepam using simplex lattice design. J. Drug Deliv. Sci. Technol. 2016, 33, 114–124. [Google Scholar] [CrossRef]
- Tang, B.; Cheng, G.; Gu, J.-C.; Xu, C.-H. Development of solid self-emulsifying drug delivery systems: Preparation techniques and dosage forms. Drug Discov. Today 2008, 13, 606–612. [Google Scholar] [CrossRef]
- Cui, J.; Yu, B.; Zhao, Y.; Zhu, W.; Li, H.; Lou, H.; Zhai, G. Enhancement of oral absorption of curcumin by self-microemulsifying drug delivery systems. Int. J. Pharm. 2009, 371, 148–155. [Google Scholar] [CrossRef] [PubMed]
- Sachan, A.K.; Gupta, A. A Review on Nanotized Herbal Drugs. Int. J. Pharm. Sci. Res. 2015, 6, 961–970. [Google Scholar] [CrossRef]
- El-Haddad, A.E.; Saadeldeen, A.M.; El-Emam, S.Z. Anti-angiogenic Activity of Major Phenolics in Tamarind Assessed with Molecular Docking Study on VEGF Kinase Proteins. Pak. J. Biol. Sci. 2019, 22, 502–509. [Google Scholar] [CrossRef]
- Mostafa, D.M.; Ammar, N.M.; El-Alim, S.H.A.; Kassem, A.A.; Hussein, R.A.; Awad, G.; El-Awdan, S.A.-W. Boswellia carterii liquisolid systems with promoted anti-inflammatory activity. Curr. Drug Deliv. 2015, 12, 454–463. [Google Scholar] [CrossRef]
- ICH Q8 International Conference on Harmonization (ICH) of Technical Requirements for Registration of Pharmaceuticals for Human Use 2005. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4544148/ (accessed on 3 June 2021).
- Nornoo, A.O.; Osborne, D.W.; Chow, D.S.-L. Cremophor-free intravenous microemulsions for paclitaxel I: Formulation, cytotoxicity and hemolysis. Int. J. Pharm. 2008, 349, 108–116. [Google Scholar] [CrossRef]
- Ujilestari, T.; Martien, R.; Ariyadi, B.; Dono, N.D. Self-nanoemulsifying drug delivery system (SNEDDS) of Amomum compactum essential oil: Design, formulation, and characterization. J. Appl. Pharm. Sci. 2018, 8, 14–21. [Google Scholar] [CrossRef]
- Cuiné, J.F.; Charman, W.N.; Pouton, C.W.; Edwards, G.A.; Porter, C.J.H. Increasing the proportional content of surfactant (Cremophor EL) relative to lipid in self-emulsifying lipid-based formulations of danazol reduces oral bioavailability in beagle dogs. Pharm. Res. 2007, 24, 748–757. [Google Scholar] [CrossRef]
- Gupta, S.; Chavhan, S.; Sawant, K.K. Self-nanoemulsifying drug delivery system for adefovir dipivoxil: Design, characterization, in vitro and ex vivo evaluation. Colloids Surf. A Physicochem. Eng. Asp. 2011, 392, 145–155. [Google Scholar] [CrossRef]
- Tawfick, M.M.; Alshareef, W.A.; Bendary, H.A.; Elmahalawy, H.; Abdulall, A.K. The emergence of carbapenemase blaNDM genotype among carbapenem-resistant Enterobacteriaceae isolates from Egyptian cancer patients. Eur. J. Clin. Microbiol. Infect. Dis. 2020, 39, 1251–1259. [Google Scholar] [CrossRef] [PubMed]
- Dolinsky, A.L.; Ohiro, R.K.; Fan, W.; Xiao, C.; Wu, F. National Committee for Clinical Laboratory Standards. 2000. Performance standard for antimicrobial susceptibility testing. Document M100–S10. J. Int. Med. Res. 2017, 46, 18. [Google Scholar] [CrossRef]
- Nazeam, J.A.; AL-Shareef, W.A.; Helmy, M.W.; El-Haddad, A.E. Bioassay-guided isolation of potential bioactive constituents from pomegranate agrifood by-product. Food Chem. 2020, 326, 126993. [Google Scholar] [CrossRef] [PubMed]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef]
- Elzahraa, F.; Elsayed, I.; Kamal, M.; Hassen, A.; Ibrahim, M. Response surface optimization, Ex vivo and In vivo investigation of nasal spanlastics for bioavailability enhancement and brain targeting of risperidone. Int. J. Pharm. 2017, 530, 1–11. [Google Scholar] [CrossRef]
- Thapa, P.; Hyung, D.; Soo, M.; Hoon, S. Effects of granulation process variables on the physical properties of dosage forms by combination of experimental design and principal component analysis. Asian J. Pharm. Sci. 2019, 14, 287–304. [Google Scholar] [CrossRef] [PubMed]
- Khattab, A.; Hassanin, L.; Zaki, N. Self-nanoemulsifying drug delivery system of coenzyme (Q10) with improved dissolution, bioavailability, and protective efficiency on liver fibrosis. AAPS PharmSciTech 2017, 18, 1657–1672. [Google Scholar] [CrossRef] [PubMed]
- Rota, M.C.; Herrera, A.; Martínez, R.M.; Sotomayor, J.A.; Jordán, M.J. Antimicrobial activity and chemical composition of Thymus vulgaris, Thymus zygis and Thymus hyemalis essential oils. Food Control 2008, 19, 681–687. [Google Scholar] [CrossRef]
- Mounika, P. Helicobacter pylori Infection and Risk of Lung Cancer: A Meta-Analysis. Lung Cancer Int. 2013, 2013, 1–6. [Google Scholar] [CrossRef] [PubMed]
- González, I.; Araya, P.; Roj, A. Helicobacter pylori infection and lung cancer: New insights and future challenges. Chin. J. Lung Cancer 2018, 21, 658–662. [Google Scholar] [CrossRef]
- Ganzera, M.; Khan, I.A. A reversed phase high performance liquid chromatography method for the analysis of boswellic acids in Boswellia serrata. Planta Med. 2001, 67, 778–780. [Google Scholar] [CrossRef]
- Singh, S.; Khajuria, A.; Taneja, S.C.; Johri, R.K.; Singh, J.; Qazi, G.N. Boswellic acids: A leukotriene inhibitor also effective through topical application in inflammatory disorders. Phytomedicine 2008, 15, 400–407. [Google Scholar] [CrossRef]
- Büchele, B.; Zugmaier, W.; Genze, F.; Simmet, T. High-performance liquid chromatographic determination of acetyl-11-keto-α-boswellic acid, a novel pentacyclic triterpenoid, in plasma using a fluorinated stationary phase and photodiode array detection: Application in pharmacokinetic studies. J. Chromatogr. B 2005, 829, 144–148. [Google Scholar] [CrossRef]
- Taha, E.; Samy, A.; Kassem, A.; Khan, M. Response Surface Methodology for the Development of Self-Nanoemulsified Drug Delivery System (SNEDDS) of All-Trans-Retinol Acetate. Pharm. Dev. Technol. 2005, 10, 363–370. [Google Scholar] [CrossRef] [PubMed]
- Bakr, R.O.; Tawfike, A.; El-Gizawy, H.A.; Tawfik, N.; Abdelmohsen, U.R.; Abdelwahab, M.F.; Alshareef, W.A.; Fayez, S.M.; El-Mancy, S.M.S.; El-Fishawy, A.M.; et al. The metabolomic analysis of five Mentha species: Cytotoxicity, anti-Helicobacter assessment, and the development of polymeric micelles for enhancing the anti-Helicobacter activity. RSC Adv. 2021, 11, 7318–7330. [Google Scholar] [CrossRef]
- Ahmed, I.S.; Elnahas, O.S.; Assar, N.H.; Gad, A.M.; Hosary, R. Nanocrystals of fusidic acid for dual enhancement of dermal delivery and antibacterial activity: In vitro, ex vivo and in vivo evaluation. Pharmaceutics 2020, 12, 199. [Google Scholar] [CrossRef]
- Zhang, H.; Cui, Y.; Zhu, S.; Feng, F.; Zheng, X. Characterization and antimicrobial activity of a pharmaceutical microemulsion. Int. J. Pharm. 2010, 395, 154–160. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.D.; Liang, Y.J.; Chen, B.H. Effects of tanshinone nanoemulsion and extract on inhibition of lung cancer cells A549. Nanotechnology 2016, 27, 495101. [Google Scholar] [CrossRef] [PubMed]
- Usmani, A.; Mishra, A.; Arshad, M.; Jafri, A. Development and evaluation of doxorubicin self nanoemulsifying drug delivery system with Nigella Sativa oil against human hepatocellular carcinoma. Artif. Cells Nanomed. Biotechnol. 2019, 47, 933–944. [Google Scholar] [CrossRef] [PubMed]
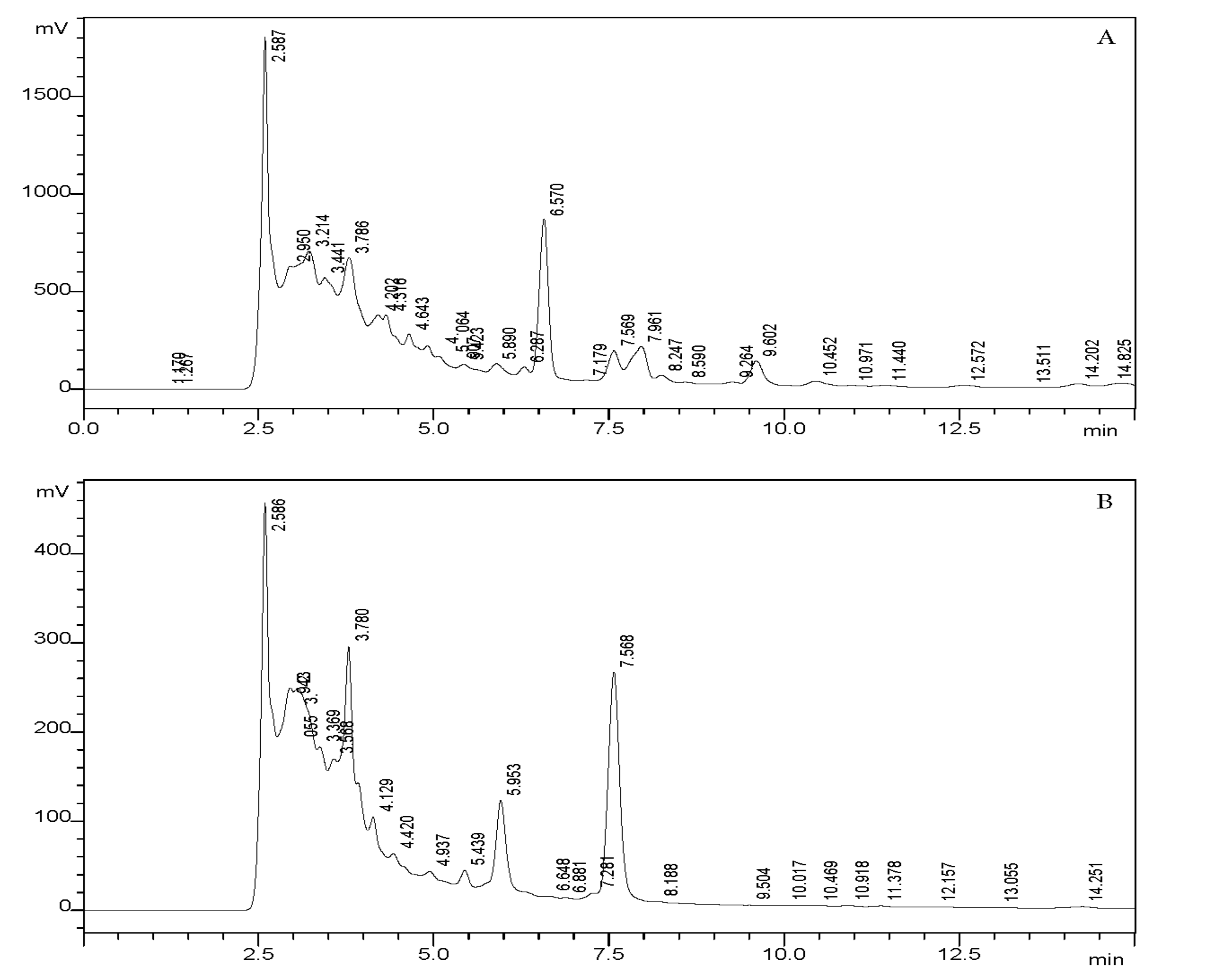

| Compound | Rt (min) | Content % |
|---|---|---|
| 11-keto-boswellic acid | 2.5 | 16.25% (major) |
| Acetyl-11-keto-boswellic acid | 3.7 | 11.8% (major) |
| α-boswellic acid | 5.8 | 2.19% (minor) |
| β-boswellic acid | 6.5 | 8.76% (major) |
| Acetyl-α-boswellic acid | 7.5 | 2.5% (minor) |
| Acetyl-β-boswellic acid | 7.9 | 3.46% (minor) |
| Total content | 44.96% |
| Excipient | Observed Solubility |
|---|---|
| Labrafil® M 1944 CS | Slightly soluble |
| Capryol™ 90 | Completely soluble |
| IPM | Insoluble |
| Myritol 318 | Insoluble |
| Tween 80 | Slightly soluble |
| Gelucire® 44/14 | Completely soluble |
| Labrasol® | Slightly soluble |
| Kolliphor® RH40 | Insoluble |
| PG | Completely soluble |
| PEG400 | Completely soluble |
| Ethanol | Completely soluble |
| Factors | Responses | ||||
|---|---|---|---|---|---|
| * Formulation No. | Oil (A) Capryol™ 90 | Surfactant (B) Gelucire® 44/14 | CoS (C) Ethanol | DS (nm) | PDI |
| 1 | 0.19 | 0.19 | 0.60 | 237 ± 6 | 0.359 ± 0.01 |
| 2 | 0.60 | 0.19 | 0.19 | 456 ± 25 | 0.67 ± 0.04 |
| 3 | 0.049 | 0.88 | 0.049 | 21 ± 0.4 | 0.311 ± 0.02 |
| 4 | 0.33 | 0.60 | 0.049 | 296 ± 6 | 0.478 ± 0.02 |
| 5 | 0.049 | 0.60 | 0.33 | 15.5 ± 0.17 | 0.249 ± 0.015 |
| 6 | 0.60 | 0.33 | 0.049 | 470 ± 15 | 0.677 ± 0.07 |
| 7 | 0.33 | 0.049 | 0.60 | 372 ± 21 | 0.562 ± 0.06 |
| 8 | 0.88 | 0.049 | 0.049 | 536 ± 28 | 0.672 ± 0.06 |
| 9 | 0.60 | 0.049 | 0.33 | 460 ± 35 | 0.736 ± 0.07 |
| 10 | 0.049 | 0.33 | 0.60 | 47 ± 0.5 | 0.30 ± 0.01 |
| 11 | 0.049 | 0.049 | 0.88 | 193 ± 2 | 0.258 ± 0.01 |
| 12 | 0.19 | 0.60 | 0.19 | 99 ± 11 | 0.40 ± 0.07 |
| 13 | 0.33 | 0.33 | 0.33 | 314 ± 5 | 0.59 ± 0.02 |
| Source | Sum of Squares | df | Mean Square | F-Value | p-Value | |
|---|---|---|---|---|---|---|
| DS | Model | 4.031 × 105 | 5 | 80620.85 | 127.14 | <0.0001 * |
| Linear Mixture | 3.763 × 105 | 2 | 1.881 × 105 | 296.68 | <0.0001 * | |
| X1 X2 | 13,167.92 | 1 | 13,167.92 | 20.77 | 0.0026 * | |
| X1 X3 | 3790.31 | 1 | 3790.31 | 5.98 | 0.0444 * | |
| X2 X3 | 7353.97 | 1 | 7353.97 | 11.60 | 0.0114 * | |
| R2 = 0.9891, Prediction R2 = 0.9813, Adjusted R2 = 0.9684, Adeq. Precision = 31.3709 | ||||||
| PDI | Model | 0.3678 | 5 | 0.0736 | 39.64 | <0.0001 * |
| Linear Mixture | 0.3238 | 2 | 0.1619 | 87.26 | <0.0001 * | |
| X1 X2 | 0.0101 | 1 | 0.0101 | 5.42 | 0.0528 | |
| X1 X3 | 0.0367 | 1 | 0.0367 | 19.79 | 0.0030 * | |
| X2 X3 | 8.837 × 10−6 | 1 | 8.837 × 10−6 | 0.0048 | 0.9469 | |
| R2 = 0.9659, Prediction R2 = 0.9415, Adjusted R2 = 0.8370, Adeq. Precision = 15.944 | ||||||
| Factors (Independent Variables) | Design Constraints | |||
|---|---|---|---|---|
| Low Limit (+0) | Upper Limit (+1) | Optimized Level | ||
| A: Capryol TM 90 | 0.05 | 0.9 | 0.050 | |
| B: Gelucire® 44/14 | 0.05 | 0.9 | 0.487 | |
| C: Ethanol | 0.05 | 0.9 | 0.463 | |
| Responses (Dependent Variables) | Constraints | 95% Prediction Interval (Low/High) | Predicted | Observed |
| DS (nm) | Minimize | −39.08/70.08 | 15.500 | 17.92 ± 0.2 |
| PDI | Minimize | 0.173/0.36 | 0.267 | 0.297 ± 0.015 |
| Microbial Species | Fr-Extract | Fr-SNEDS | Plain SNEDS | Antimicrobial Agents * | |||
|---|---|---|---|---|---|---|---|
| F | AMC | CFPM | VA | ||||
| S. aureus | 19 ± 1.1 | 23 ± 1.3 | 9 ± 0.3 | 20 ± 1.3 | 34 ± 2.1 | 25 ± 1.9 | 16 ± 0.7 |
| S. epidermidis | 19 ± 1.5 | 24 ± 1.5 | 9 ± 0.4 | 19 ± 0.9 | NA | 20 ± 1.7 | 14 ± 0.6 |
| B. subtilis | 22 ± 0.2 | 24 ± 0.7 | 9 ± 0.3 | 25 ± 1.9 | NA | 20 ± 1.4 | 17 ± 0.5 |
| M. luteus | 20 ± 0.5 | 12 ± 0.3 | NA | 14 ± 0.8 | 18 ± 0.3 | 23 ± 1.3 | 15 ± 0.8 |
| MRSA | 22 ± 1.8 | 22 ± 1.4 | 9 ± 0.3 | ND | ND | R | 17 ± 0.9 |
| H. pylori ** | 25 ± 0.2 | 28 ± 1.1 | 10 ± 0.6 | ND | ND | ND | NA |
| C. albicans *** | 22 ± 1.1 | 14 ± 1.0 | NA | NA | NA | NA | NA |
| Fr-SNEDS | DS (nm) | PDI | ZP (mV) | Assessment of Self-Emulsification | |
|---|---|---|---|---|---|
| Emulsification Time | % Transmittance | ||||
| Freshly prepared | 17.9 ± 0.2 | 0.3 ± 0.015 | −14.5 ± 0.236 | <1 min | 97.8 ± 0.2 |
| Stored for one year | 22.3 ± 0.7 | 0.5 ± 0.018 | −22.4 ± 0.781 | <1 min | 98.1 ± 0.1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
El-Mancy, S.S.; El-Haddad, A.E.; Alshareef, W.A.; Saadeldeen, A.M.; El-Emam, S.Z.; Elnahas, O.S. Enhancement of Antimicrobial and Antiproliferative Activities of Standardized Frankincense Extract Using Optimized Self-Nanoemulsifying Delivery System. Sci. Pharm. 2021, 89, 36. https://doi.org/10.3390/scipharm89030036
El-Mancy SS, El-Haddad AE, Alshareef WA, Saadeldeen AM, El-Emam SZ, Elnahas OS. Enhancement of Antimicrobial and Antiproliferative Activities of Standardized Frankincense Extract Using Optimized Self-Nanoemulsifying Delivery System. Scientia Pharmaceutica. 2021; 89(3):36. https://doi.org/10.3390/scipharm89030036
Chicago/Turabian StyleEl-Mancy, Shereen S., Alaadin E. El-Haddad, Walaa A. Alshareef, Amr M. Saadeldeen, Soad Z. El-Emam, and Osama S. Elnahas. 2021. "Enhancement of Antimicrobial and Antiproliferative Activities of Standardized Frankincense Extract Using Optimized Self-Nanoemulsifying Delivery System" Scientia Pharmaceutica 89, no. 3: 36. https://doi.org/10.3390/scipharm89030036
APA StyleEl-Mancy, S. S., El-Haddad, A. E., Alshareef, W. A., Saadeldeen, A. M., El-Emam, S. Z., & Elnahas, O. S. (2021). Enhancement of Antimicrobial and Antiproliferative Activities of Standardized Frankincense Extract Using Optimized Self-Nanoemulsifying Delivery System. Scientia Pharmaceutica, 89(3), 36. https://doi.org/10.3390/scipharm89030036








