The Chemistry and Pharmacology of Fungal Genus Periconia: A Review
Abstract
1. Introduction
2. Phylogeny of the Genera Periconia
3. Natural Product Diversity
3.1. Overview of Compounds Derived from the Periconia Fungus
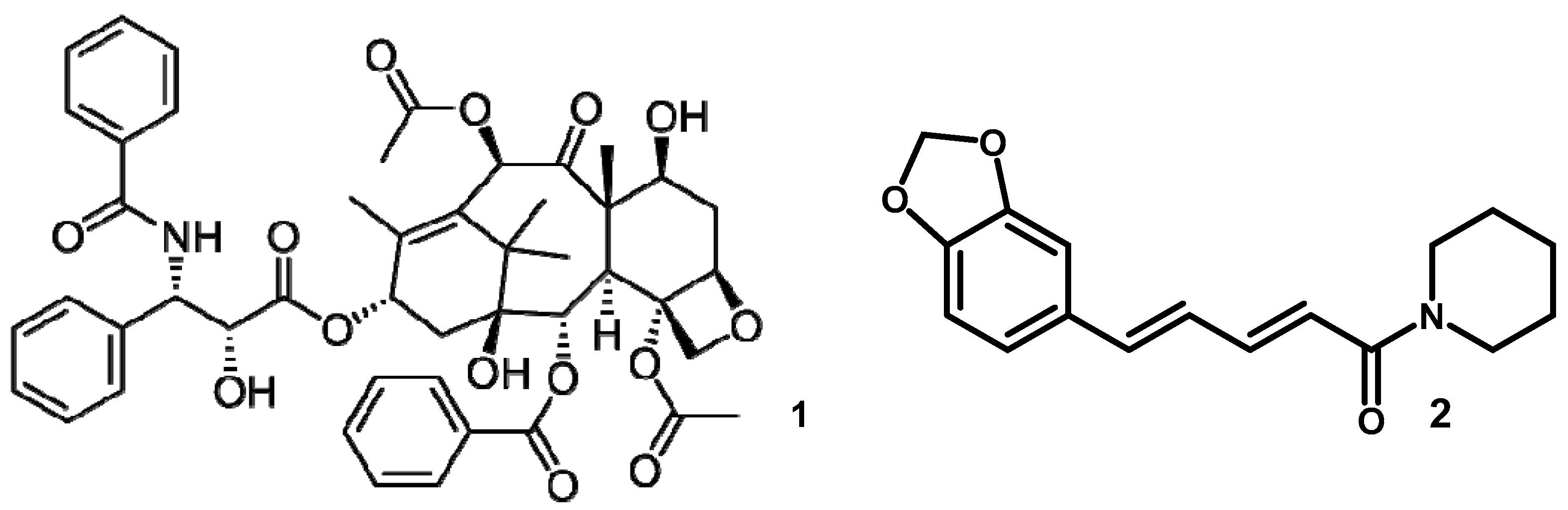
3.2. Taxol and Piperine Producing Periconia Species
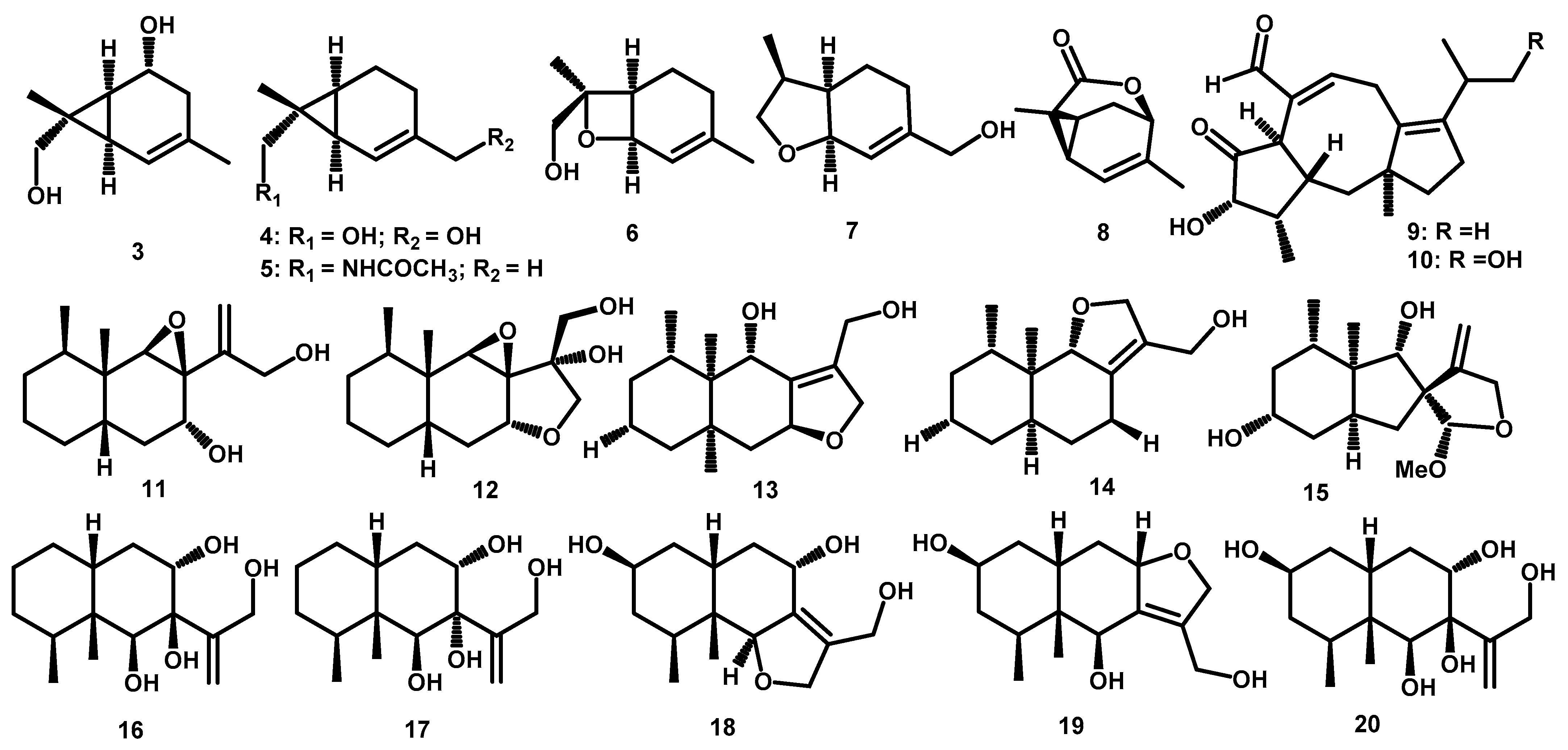
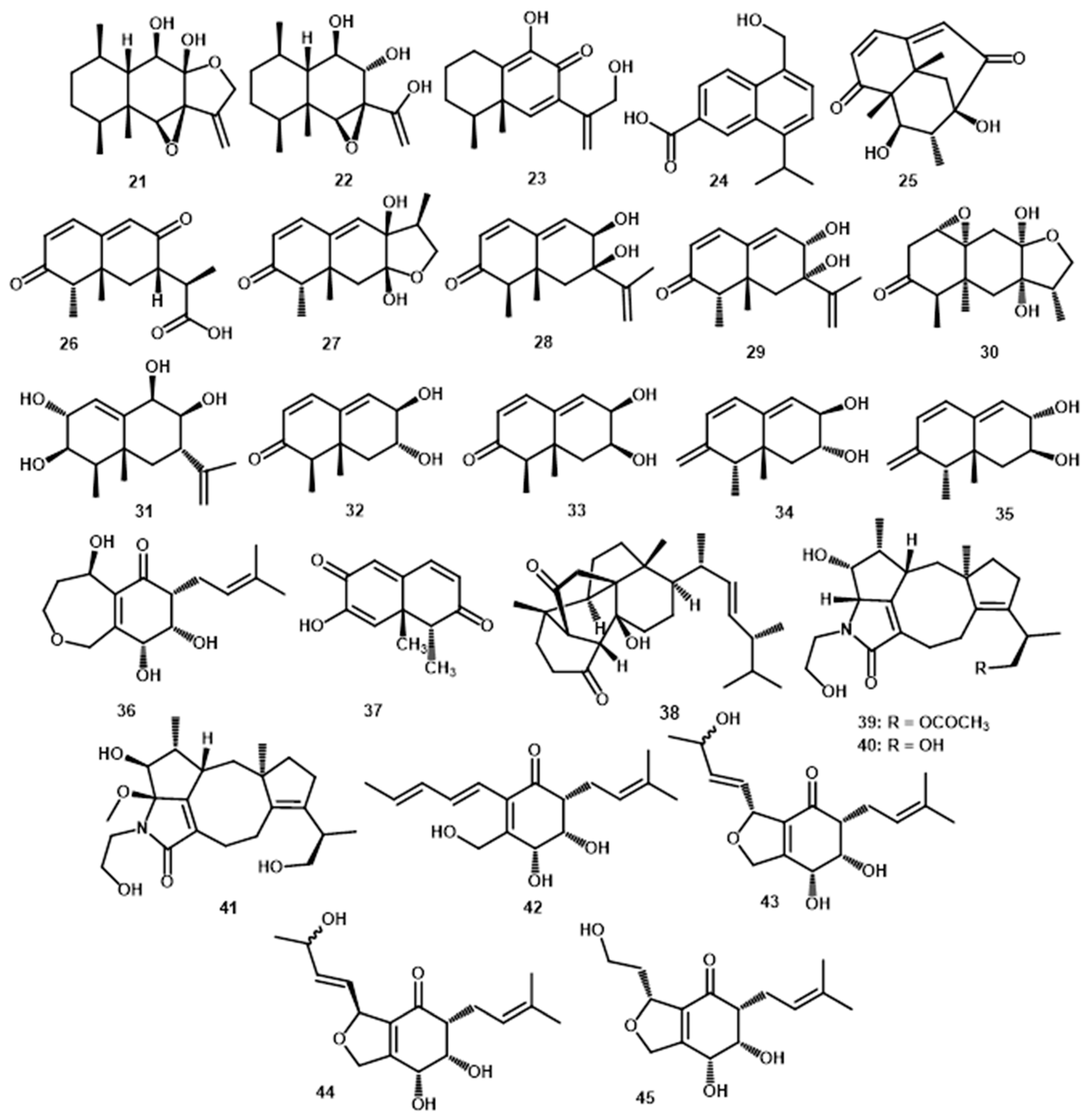
3.3. Terpenoids
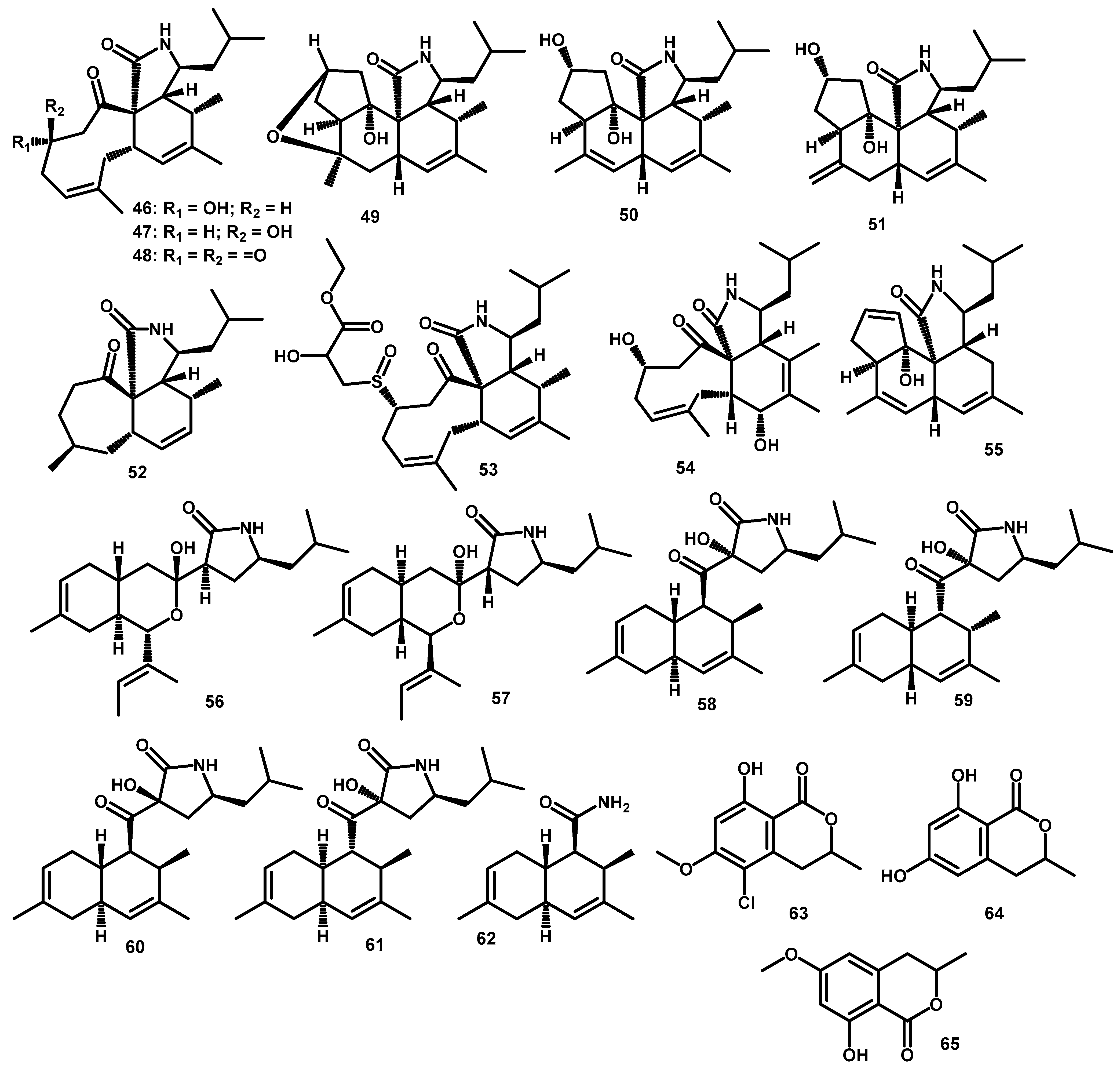
3.4. Polyketides
3.5. Aromatic Compounds and Carbohydrates Derivates
4. Pharmacological Activities
4.1. Antimicrobial Activity
4.2. Cytotoxic Activity
4.3. Anti-Inflammatory Activity
5. Conclusions and Future Perspectives
| Type of Compounds | Name of Compounds | Molecular Formula | Molecular Weights (g/mol) | MS (m/z) | Fungal Strains | Host Origin | Part of Host Origin | Ref. |
|---|---|---|---|---|---|---|---|---|
| Terpenes | ||||||||
| Monoterpene | 2-carene-5,8-diol (3) | C10H16O2 | 168.1150 | HR-ESI-MS: 151.1115 [M+H–H2O]+, 133.1011 [M +H–2xH2O]+; GC-EI-MS: 168 [M]+, 150 [M–H2O]+ | Periconia sp. F-31 | Annona muricata | Leaves | [19] |
| 2-carene-8,10-diol (4) | C10H16O2 | 168.1150 | HR-ESI-MS: 151.1112 [M+H–H2O]+, 133.1008 [M +H–2xH2O]+; GC-EI-MS: 168 [M]+, 150 [M–H2O]+ | Periconia sp. F-31 | Annona muricata | Leaves | [19] | |
| 2-carene-8-acetamide (5) | C12H19NO | 193.1467 | HR-ESI-MS: 194.1509 [M+H]+, 387.2942 [2M +H]+ | Periconia sp. F-31 | Annona muricata | Leaves | [19] | |
| 8-hydroxy-1,7-expoxy-2-menthene (6) | C10H16O2 | 168.1150 | HR-ESI-MS: 151.1112 [M+H–H2O]+; GC-EI-MS: 168 [M]+, 153 [M–CH3]+ | Periconia sp. F-31 | Annona muricata | Leaves | [19] | |
| anethofuran (7) | C10H16O2 | 168.1150 | * HR-ESI-MS: 169.1229 [M+H]+ | Periconia sp. F-31 | Annona muricata | Leaves | [19] | |
| (-)-(1R,4R,6S,7S)-2-caren-4,8-olide (8) | C10H12O2 | 164.0837 | HR-ESI-MS: 165.0914 [M+H]+ | Periconia sp. F-31 | Annona muricata | Leaves | [23] | |
| Diterpene | periconicin A (9) | C20H28O3 | 316.2038 | HR-EI-MS: 316.2046 [M+H]+ | Periconia sp. | Taxus cuspidata | Small Branches | [25] |
| periconicin B (10) | C20H28O4 | 332.1988 | HR-EI-MS: 332.2008 [M+H]+ | Periconia atropurpurea | Xylopia aromatica | Leaves | [11] | |
| Sesquiterpene | peribysin A (11) | C15H25O3 | 253.1803 | HR-EI-MS: 253.1802 [M+H]+ | Periconia byssoides | Aplysia kurodai | gastrointestinal tract | [13,34] |
| peribysin B (12) | C15H24O4 | 268.1673 | HR-EI-MS: 268.1665 [M]+ | Periconia byssoides | Aplysia kurodai | gastrointestinal tract | [13,34] | |
| peribysin C (13) | C15H22O2 | 234.1619 | HR-EI-MS: 234.1619 [M]+ | Periconia byssoides | Aplysia kurodai | gastrointestinal tract | [13,34] | |
| peribysin D (14) | C15H22O2 | 234.1619 | HR-EI-MS: 234.1618 [M]+ | Periconia byssoides | Aplysia kurodai | gastrointestinal tract | [13] | |
| peribysin E (15) | C20H30O6 | 366.2042 | HR-EI-MS: 366.0948 [M]+ | Periconia byssoides | Aplysia kurodai | gastrointestinal tract | [31] | |
| peribysin F (16) | C21H32O7 | 396.2148 | HR-EI-MS: 396.1677 [M]+ | Periconia byssoides | Aplysia kurodai | gastrointestinal tract | [13,34] | |
| peribysin G (17) | C21H32O7 | 396.2148 | HR-EI-MS: 396.1677 [M]+ | Periconia byssoides | Aplysia kurodai | gastrointestinal tract | [13,34] | |
| peribysin H (18) | C15H24O4 | 268.1673 | HR-EI-MS: 268.1673 [M]+ | Periconia byssoides | Aplysia kurodai | gastrointestinal tract | [32] | |
| peribysin I (19) | C15H24O4 | 268.1673 | HR-EI-MS: 268.1673 [M]+ | Periconia byssoides | Aplysia kurodai | gastrointestinal tract | [32] | |
| peribysin J (20) | C15H26O5 | 286.1773 | HR-EI-MS: 268.1775 [M]+ | Periconia byssoides | Aplysia kurodai | gastrointestinal tract | [33] | |
| peribysin O (21) | C15H22O4 | 266.1518 | ESI-TOF-MS: 289.1416 [M+Na]+; 555.2919 [2M+Na]+ | P. macrospinosa KT3863 | terrestrial herbaceous plant at Kanagawa prefecture in 2018 | - | [37] | |
| peribysin P (22) | C15H24O4 | 268.1675 | ESI-TOF-MS: 291.1566 [M+Na]+; 559.3230 [2M+Na]+ | P. macrospinosa KT3863 | terrestrial herbaceous plant at Kanagawa prefecture in 2018 | - | [37] | |
| peribysin Q (23) | C15H20O3 | 248.1412 | ESI-TOF-MS: 249.1496 [M+H]+; 231.1388 [M+H-H2O]+; 271.1311 [M+Na]+ | P. macrospinosa KT3863 | terrestrial herbaceous plant at Kanagawa- -prefecture in 2018 | - | [37] | |
| pericoterpenoid A (24) | C15H16O3 | 244.1099 | HR-ESI-MS: 267.1002 [M+Na]+ | Periconia sp. | Parmelia sp. | - | [8] | |
| periconianone A (25) | C15H18O4 | 262.1205 | HR-ESI-MS: 263.1271 [M+H]+ | Periconia sp. F-31 | Annona muricata | Leaves | [38] | |
| periconianone B (26) | C15H18O4 | 262.1205 | HR-ESI-MS: 263.1280 [M+H]+ | Periconia sp. F-31 | Annona muricata | Leaves | [38] | |
| periconianone C (27) | C15H20O4 | 264.1362 | HR-ESI-MS: 265.1434 [M+H]+ | Periconia sp. F-31 | Annona muricata | Leaves | [39] | |
| periconianone D (28) | C15H20O3 | 248.1412 | HR-ESI-MS: 249.1458 [M+H]+ | Periconia sp. F-31 | Annona muricata | Leaves | [39] | |
| periconianone E (29) | C15H20O3 | 248.1412 | HR-ESI-MS: 249.1478 [M+H]+ | Periconia sp. F-31 | Annona muricata | Leaves | [39] | |
| periconianone F (30) | C15H22O5 | 282.1467 | HR-ESI-MS: 283.1540 [M+H]+ | Periconia sp. F-31 | Annona muricata | Leaves | [39] | |
| periconianone G (31) | C15H24O4 | 268.1675 | HR-ESI-MS: 291.1567 [M+Na]+ | Periconia sp. F-31 | Annona muricata | Leaves | [39] | |
| periconianone H (32) | C12H16O3 | 208.1099 | HR-ESI-MS: 209.1172 [M+H]+ | Periconia sp. F-31 | Annona muricata | Leaves | [39] | |
| periconianone I (33) | C12H16O3 | 208.1099 | HR-ESI-MS: 209.1172 [M+H]+ | Periconia sp. F-31 | Annona muricata | Leaves | [39] | |
| periconianone J (34) | C12H16O3 | 208.1099 | HR-ESI-MS: 209.1172 [M+H]+ | Periconia sp. F-31 | Annona muricata | Leaves | [39] | |
| periconianone K (35) | C12H16O3 | 208.1099 | HR-ESI-MS: 209.1172 [M+H]+ | Periconia sp. F-31 | Annona muricata | Leaves | [39] | |
| (+)-(3S,6S,7R,8S)-periconone A (36) | C15H22O5 | 282.1467 | HR-ESI-MS: 283.1549 [M+H]+ | Periconia sp. F-31 | Annona muricata | Leaves | [23] | |
| Norsesqui-terpene | Dihydronaphthalene-2,6-dione (37) | C12H12O3 | 204.0786 | * HR-ESI-MS: 205.0865 [M+H]+ | Periconia sp. F-31 | Annona muricata | Leaves | [38] |
| Steroid | periconiastone A (38) | C28H42O3 | 426.3134 | HR-ESI-MS: 449.3026 [M+Na]+ | Periconia sp. TJ403-rc01 | Rosa chinensis | Leaves | [43] |
| Meroterpenes | pericolactine A (39) | C24H35NO5 | 417.2515 | HR-ESI-MS: 418.2595 [M+H]+ | Periconia sp. | Parmelia sp. | - | [7] |
| pericolactine B (40) | C22H33NO4 | 375.2410 | HR-ESI-MS: 376.2491 [M+H]+ | Periconia sp. | Parmelia sp. | - | [7] | |
| pericolactine C (41) | C23H35NO5 | 405.2515 | HR-ESI-MS: 428.2418 [M+Na]+ | Periconia sp. | Parmelia sp. | - | [7] | |
| periconone B (42) | C17H24O4 | 292.1675 | HR-ESI-MS: 293.1741 [M+H]+ | Periconia sp. F-31 | Annona muricata | Leaves | [43] | |
| periconone C (43) | C17H24O5 | 308.1624 | HR-ESI-MS: 331.1516 [M+Na]+ | Periconia sp. F-31 | Annona muricata | Leaves | [43] | |
| periconone D (44) | C17H24O5 | 308.1624 | HR-ESI-MS: 331.1516 [M+Na]+ | Periconia sp. F-31 | Annona muricata | Leaves | [43] | |
| periconone E (45) | C15H22O5 | 282.1467 | HR-ESI-MS: 283.1540 [M+H]+ | Periconia sp. F-31 | Annona muricata | Leaves | [43] | |
| Polyketides | ||||||||
| Cytochalasan | periconiasin A (46) | C22H33NO3 | 359.2460 | HR-ESI-MS: 360.2528 [M+H]+ | Periconia sp. F-31 | Annona muricata | Leaves | [49] |
| periconiasin B (47) | C22H33NO3 | 359.2460 | HR-ESI-MS: 360.2525 [M+H]+ | Periconia sp. F-31 | Annona muricata | Leaves | [48] | |
| periconiasin C (48) | C22H31NO3 | 357.2304 | HR-ESI-MS: 358.2377 [M+H]+ | Periconia sp. F-31 | Annona muricata | Leaves | [48] | |
| periconiasin D (49) | C22H33NO3 | 359.2460 | HR-ESI-MS: 360.2533 [M+H]+ | Periconia sp. F-31 | Annona muricata | Leaves | [49] | |
| periconiasin E (50) | C22H33NO3 | 359.2460 | HR-ESI-MS: 360.2533 [M+H]+ | Periconia sp. F-31 | Annona muricata | Leaves | [49] | |
| periconiasin F (51) | C22H33NO3 | 359.2460 | HR-ESI-MS: 360.2533 [M+H]+ | Periconia sp. F-31 | Annona muricata | Leaves | [49] | |
| periconiasin G (52) | C20H31NO2 | 317.2355 | HR-ESI-MS: 318.2427 [M+H]+ | Periconia sp. F-31 | Annona muricata | Leaves | [14] | |
| periconiasin H (53) | C27H41NO6S | 507.2655 | HR-ESI-MS: 508.2703 [M+H]+ | Periconia sp. F-31 | Annona muricata | Leaves | [14] | |
| periconiasin I (54) | C22H33NO4 | 375.2410 | HR-ESI-MS: 376.2474 [M+H]+ | Periconia sp. F-31 | Annona muricata | Leaves | [50] | |
| periconiasin J (55) | C22H31NO2 | 341.2355 | HR-ESI-MS: 342.2422 [M+H]+ | Periconia sp. F-31 | Annona muricata | Leaves | [50] | |
| pericoannosin A (56) | C22H35NO3 | 361.2617 | HR-ESI-MS: 384.2505 [M+Na]+ | Periconia sp. F-31 | Annona muricata | Leaves | [50] | |
| Other Polyketides | pericoannosin B (57) | C22H35NO3 | 361.2617 | FABMS: 362.2 [M+H]+; HR-ESI-MS: 384.2495 [M+Na]+ | Periconia sp. F-31 | Annona muricata | Leaves | [52] |
| pericoannosin C (58) | C22H33NO3 | 359.2460 | HR-ESI-MS: 360.2527 [M+H]+ | Periconia sp. F-31 | Annona muricata | Leaves | [52] | |
| pericoannosin D (59) | C22H33NO3 | 359.2460 | HR-ESI-MS: 360.2527 [M+H]+ | Periconia sp. F-31 | Annona muricata | Leaves | [52] | |
| pericoannosin E (60) | C22H33NO3 | 359.2460 | HR-ESI-MS: 360.2527 [M+H]+ | Periconia sp. F-31 | Annona muricata | Leaves | [52] | |
| pericoannosin F (61) | C22H33NO3 | 359.2460 | HR-ESI-MS: 360.2527 [M+H]+ | Periconia sp. F-31 | Annona muricata | Leaves | [52] | |
| pericoannosin G (62) | C14H21NO | 219.1623 | HR-ESI-MS: 220.1696 [M+H]+ | Periconia sp. F-31 | Annona muricata | Leaves | [10] | |
| 5-chloro-3.4-dihydro-8-hydroxy-6-methoxy--3-methylisocoumarin (63) | C11H11ClO4 | 242.0346 | * HR-ESI-MS: 243.0414 [M+H]+ | Periconia macrospinosa | - | - | [55] | |
| 3,4-dihydro-6,8-dihydroxy-3-methylisocoumarin (64) | C10H10O4 | 194.0579 | * HR-ESI-MS: 195.0657 [M+H]+ | Periconia macrospinosa | - | - | [54] | |
| 3,4-dihydro-8-hydroxy-6-methoxy-3-methylisocoumarin (65) | C11H12O4 | 208.0736 | * HR-ESI-MS: 209.0814 [M+H]+ | Periconia macrospinosa | - | - | [54] | |
| Circinatin (66) | C20H31O7N3 | 425 | FABMS: 426 [M+H]+ | Periconia circinata (Mangin) Sacc. | Sorghum bicolor (L.) | root and crown root | [57] | |
| peritoxin A (67) | C20H29O9N4Cl3 | 574 | FABMS: 575 [M+H]+ | Periconia circinata (Mangin) Sacc. | Sorghum bicolor (L.) | root and crown root | [56] | |
| peritoxin B (68) | C20H29O8N4Cl3 | 558 | FABMS: 559 [M+H]+ | Periconia circinata (Mangin) Sacc. | Sorghum bicolor (L.) | root and crown root | [56] | |
| periconin A (69) | C20H29O8N4Cl3 | 558 | FABMS: 559 [M+H]+ | Periconia circinata (Mangin) Sacc. | Sorghum bicolor (L.) | root and crown root | [56] | |
| periconin B (70) | C20H29O8N4Cl3 | 558 | FABMS: 559 [M+H]+ | Periconia circinata (Mangin) Sacc. | Sorghum bicolor (L.) | root and crown root | [56] | |
| Macrolide | modiolide A (71) | C10H14O4 | 198.21 | * HR-EI-MS: 199.0970 [M+H]+ | Periconia siamensis CMUGE015 | Thysanoleana lafifolia | Leaves | [60,61] |
| Macrosphelide | macrosphelide A (72) | C16H22O8 | 342.1315 | HR-EI-MS: 343.1392 [M+H]+ | Periconia byssoides OUPS-N133 | Aplysia kurodai | gastrointestinal tract | [65] |
| macrosphelide C (73) | C16H22O7 | 326.1366 | HR-EI-MS: 327.1442 [M+H]+ | Periconia byssoides OUPS-N133 | Aplysia kurodai | gastrointestinal tract | [6,63] | |
| macrosphelide E (74) | C16H22O8 | 342.1315 | HR-EI-MS: 343.1392 [M+H]+ | Periconia byssoides OUPS-N133 | Aplysia kurodai | gastrointestinal tract | [6,63] | |
| seco-Macrosphelide E (75) | C16H23O8 | 342.1315 | HR-EI-MS: 342.1315 [M]+ | Periconia byssoides OUPS-N133 | Aplysia kurodai | gastrointestinal tract | [66] | |
| macrosphelide F (76) | C16H22O7 | 326.1366 | HR-EI-MS: 327.1442 [M+H]+ | Periconia byssoides OUPS-N133 | Aplysia kurodai | gastrointestinal tract | [6,63] | |
| macrosphelide G (77) | C16H22O8 | 342.1315 | HR-EI-MS: 343.1392 [M+H]+ | Periconia byssoides OUPS-N133 | Aplysia kurodai | gastrointestinal tract | [6,63] | |
| macrosphelide H (78) | C16H22O8 | 342.1315 | HR-EI-MS: 343.1392 [M+H]+ | Periconia byssoides OUPS-N133 | Aplysia kurodai | gastrointestinal tract | [6,63] | |
| macrosphelide I (79) | C16H22O8 | 342.1315 | HR-EI-MS: 343.1392 [M+H]+ | Periconia byssoides OUPS-N133 | Aplysia kurodai | gastrointestinal tract | [65] | |
| macrosphelide L (80) | C16H22O8 | 342.1315 | HR-EI-MS: 342.1313 [M]+ | Periconia byssoides OUPS-N133 | Aplysia kurodai | gastrointestinal tract | [67] | |
| macrosphelide M (81) | C16H22O8 | 342.1315 | HR-EI-MS: 343.1392 [M+H]+ | Periconia byssoides OUPS-N133 | Aplysia kurodai | gastrointestinal tract | [33] | |
| Cyclopentene | methyl 2-allyl-3,5-dichloro-1,4-dihydroxycyclopent-2-enoate methyl ester (82) | C10H12Cl2O4 | 374.0113 | * HR-EI-MS: 375.0191 [M+H]+ | Periconia macrospinosa | - | - | [55] |
| cyclopericodiol (83) | C12H12O5 | 236.0685 | ESI-TOF-MS: 255.0399 [M+Na]+; 487.0910 [2M+Na]+; 215.0475 [M+H–H2O]+ | Periconia macrospinosa KT3863 | terrestrial herbaceous plant at Kanagawa prefecture in 2018 | - | [57] | |
| Cryptosporiopsinol (84) | C10H12Cl2O4 | 266.0113 | * HR-EI-MS: 267.0191 [M+H]+ | Periconia macrospinosa | - | - | [53] | |
| Cryptosporiopsin (85) | C10H10Cl2O4 | 263.9956 | * HR-EI-MS: 265.0034 [M+H]+ | Periconia macrospinosa | - | - | [53] | |
| Aromatic Compounds | ||||||||
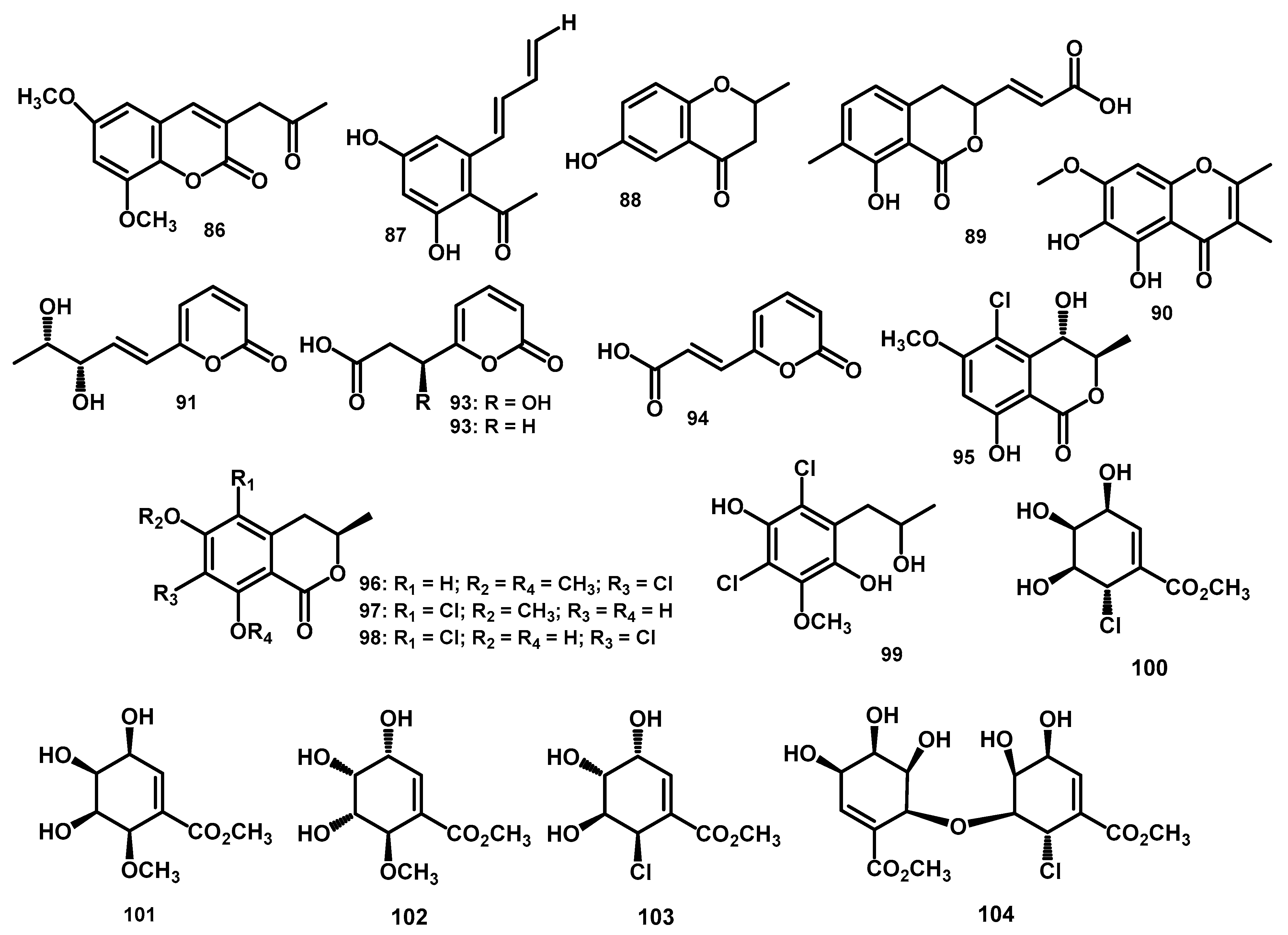
| 6,8-Dimethoxy-3-(20-oxo-propyl)-coumarin (86) | C14H14O5 | 262.0841 | HR-ESI-MS: 285.0739 [M+Na]+ | Periconia atropurpurea | Xylopia aromatica | Leaves | [11] | |
| 2,4-dihydroxy-6-[(10E,30E)-penta-1′,3′-dienyl]-benzaldehyde (87) | C12H12O3 | 204.0786 | HR-ESI-MS: 203.0708 [M–H]- | Periconia atropurpurea | Xylopia aromatica | Leaves | [11] | |
| 4-chromanone, 6-hydroxy-2-methyl-(5CI) (88) | C10H10O3 | 178.0630 | * HR-EI-MS: 179.0708 [M+H]+ | Periconia siamensis CMUGE015 | Thysanoleana latifolia | Leaves | [62] | |
| pericocin A (89) | C13H12O5 | 248.0685 | HR-ESI-MS: 271.0587 [M+Na]+ | Periconia sp. | Parmelia sp. | - | [9] | |
| pericocin B (90) | C12H12O5 | 236.0685 | HR-ESI-MS: 237.0769 [M+H]+ | Periconia sp. | Parmelia sp. | - | [9] | |
| pericocin C (91) | C10H12O4 | 196.0736 | HR-ESI-MS: 197.0814 [M+H]+ | Periconia sp. | Parmelia sp. | - | [9] | |
| pericocin D (92) | C8H8O5 | 184.0372 | HR-ESI-MS: 185.0450 [M+H]+ | Periconia sp. | Parmelia sp. | - | [9] | |
| 3-(2-oxo-2H-pyran-6-yl)propanoic acid (93) | C8H8O4 | 168.0423 | * HR-ESI-MS: 169.0501 [M+H]+ | Periconia sp. | Parmelia sp. | - | [9] | |
| (E)-3-(2-oxo-2H-pyran-6-yl)acrylic acid (94) | C8H6O4 | 166.0266 | * HR-ESI-MS: 167.0344 [M+H]+ | Periconia sp. | Parmelia sp. | - | [9] | |
| (3R,4S)-5-chloro-4-hydroxy-6-methoxymellein (95) | C11H11ClO5 | 258.0295 | ESI-TOF-MS: 259.0368 [M+H]+; 281.0187 [M+Na]+; 296.9927 [M+K]+; 539.0482 [2M+Na]+ | Periconia macrospinosa KT3863 | terrestrial herbaceous plant at Kanagawa prefecture in 2018 | - | [57] | |
| (R)-7-chloro-6-methoxy-8-O-methyl-mellein (96) | C12H13ClO4 | 256.0502 | ESI-TOF-MS: 257.0575 [M+H]+; 279.0395 [M+Na]+; 535.0897 [2M+Na]+ | Periconia macrospinosa KT3863 | terrestrial herbaceous plant at Kanagawa prefecture in 2018 | - | [57] | |
| (R)-5-chloro-6-methoxy-mellein (97) | C11H11ClO4 | 242.0346 | ESI-TOF-MS: 243.0419 [M+H]+; 265.0238 [M+Na]+; 280.9977 [M+K]+; 507.0584 [2M+Na]+ | Periconia macrospinosa KT3863 | terrestrial herbaceous plant at Kanagawa prefecture in 2018 | - | [57] | |
| pericochlorosin A (98) | C10H7Cl2O4 | 260.9721 | ESI-MS m/z 263.3 [M+H]+, 265.2 [M+2+H]+, 267.2 [M+4+H]+; HR-ESI-MS m/z 260.9723 [M-H]- | Periconia sp. F-31 | Annona muricata | Leaves | [10] | |
| pericochlorosin B (99) | C10H11Cl2O3 | 249.0085 | ESI-MS m/z 265.6 [M-H]–, 267.2 [M+2-H]–, 269.3 [M+4-H]–; HR-ESI-MS m/z 249.0080 [M+H-H2O]+ | Periconia sp. F-31 | Annona muricata | Leaves | [10] | |
| CarbohydrateDerivates | ||||||||
| pericosine A (100) | C8H11ClO5 | 222.0295 | HR-EI-MS: 223.0373 [M+H]+ | Periconia byssoides OUPS-N133 | Aplysia kurodai | gastrointestinal tract | [6,66] | |
| pericosine B (101) | C9H13O6 | 217.0712 | HR-EI-MS: 218.0790 [M+H]+ | Periconia byssoides OUPS-N133 | Aplysia kurodai | gastrointestinal tract | [6,66] | |
| pericosine C (102) | C9H14O6 | 218.0790 | HR-EI-MS: 219.0854 [M+H]+ | Periconia byssoides OUPS-N133 | Aplysia kurodai | gastrointestinal tract | [66] | |
| pericosine D (103) | C8H11ClO5 | 222.0295 | HR-EI-MS: 223.0363 [M+H]+ | Periconia byssoides OUPS-N133 | Aplysia kurodai | gastrointestinal tract | [66] | |
| pericosine E (104) | C16H21ClO10 | 408.0832 | HR-EI-MS: 409.0900 [M+H]+ | Periconia byssoides OUPS-N133 | Aplysia kurodai | gastrointestinal tract | [66] | |
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Periconia Tode. Available online: http://www.indexfungorum.org/names/NamesRecord.asp?RecordID=9263 (accessed on 4 November 2020).
- Liu, N.; Hongsanan, S.; Yang, J.; Bhat, D.J.; Liu, J.; Jumpathong, J.; Liu, Z. Periconia thailandica (Periconiaceae) a new species from Thailand. Phytotaxa 2017, 323, 253–263. [Google Scholar] [CrossRef]
- Markovskaja, S.; Kačergius, A. Morphological and molecular characterization of Periconia pseudobyssoides sp. nov. and closely related P. byssoides. Mycol. Prog. 2014, 13, 291–302. [Google Scholar] [CrossRef]
- Carmarán, C.C.; Novas, M.V. A review of Spegazzini taxa of Periconia and Sporocybe after over 115 years. Fungal Divers. 2003, 14, 67–76. [Google Scholar]
- Leukel, R.W. Periconia circinata and its relation to Milo Disease. J. Agric. Res. 1948, 77, 201–222. [Google Scholar]
- Numata, A.; Iritani, M.; Yamada, T.; Minoura, K.; Matsumura, E.; Yamori, T.; Tsuruo, T. Novel antitumour metabolites produced by a fungal strain from a sea hare. Tetrahedron Lett. 1997, 38, 8215–8218. [Google Scholar] [CrossRef]
- Wu, Y.H.; Chen, G.D.; He, R.R.; Wang, C.X.; Hu, D.; Wang, G.Q.; Guo, L.D.; Yao, X.S.; Gao, H. Pericolactines A–C, a new class of diterpenoid alkaloids with unusual tetracyclic skeleton. Sci. Rep. 2015, 5, 17082. [Google Scholar] [CrossRef]
- Wu, Y.H.; Chen, G.D.; Wang, C.X.; Hu, D.; Li, X.X.; Lian, Y.Y.; Lin, F.; Guo, L.D.; Gao, H.J. Pericoterpenoid A, a new bioactive cadinane-type sesquiterpene from Periconia sp. Asian Nat. Prod. Res. 2015, 17, 671–675. [Google Scholar] [CrossRef]
- Wu, Y.H.; Xiao, G.K.; Chen, G.D.; Wang, C.X.; Hu, D.; Lian, Y.Y.; Lin, F.; Guo, L.D.; Yao, X.S.; Gao, H. Pericocins A–D, new bioactive compounds from Periconia sp. Nat. Prod. Commun. 2015, 10, 2127–2130. [Google Scholar] [CrossRef]
- Liu, J.; Chen, M.; Chen, R.; Xie, K.; Chen, D.; Si, S.; Dai, J.J. Three new compounds from endophytic fungus Periconia sp. F-31. Chin. Pharm. Sci. 2020, 29, 244–251. [Google Scholar] [CrossRef]
- Teles, H.L.; Sordi, R.; Silva, G.H.; Castro-Gamboa, I.; da Silva Bolzani, V.; Pfenning, L.H.; de Abreu, L.M.; Costa-Neto, C.M.; Young, M.C.M.; Araújo, Â.R. Aromatic compounds produced by Periconia atropurpurea, an endophytic fungus associated with Xylopia aromatica. Phytochemistry 2006, 67, 2686–2690. [Google Scholar] [CrossRef]
- Verma, V.C.; Lobkovsky, E.; Gange, A.C.; Singh, S.K.; Prakash, S. Piperine production by endophytic fungus Periconia sp. Isolated from Piper longum L. J. Antibiot. 2011, 64, 427–431. [Google Scholar] [CrossRef]
- Yamada, T.; Iritani, M.; Minoura, K.; Kawai, K.; Numata, A. Peribysins A–D, potent cell-adhesion inhibitors from a sea hare-derived culture of Periconia species. Org. Biomol. Chem. 2004, 2, 2131–2135. [Google Scholar] [CrossRef]
- Zhang, D.; Tao, X.; Liu, J.; Chen, R.; Zhang, M.; Li, L.; Fang, X.; Yu, L.Y.; Dai, J. Periconiasin G, a new cytochalasan with unprecedented 7/6/5 tricyclic ring system from the endophytic fungus Periconia sp. Tetrahedron Lett. 2016, 57, 796–799. [Google Scholar] [CrossRef]
- Li, J.Y.; Sidhu, R.S.; Ford, E.J.; Long, D.M.; Hess, W.M.; Strobel, G.A. The induction of taxol production in the Endophytic fungus—Periconia sp. from Torreya grandifolia. J. Ind. Microbiol. Biotechnol. 1998, 20, 259–264. [Google Scholar] [CrossRef]
- Tanaka, K.; Hirayama, K.; Yonezawa, H.; Sato, G.; Toriyabe, A.; Kudo, H.; Hashimoto, A.; Matsumura, M.; Harada, Y.; Kurihara, Y.; et al. Revision of the Massarineae (Pleosporales, Dothideomycetes). Stud. Mycol. 2015, 82, 75–136. [Google Scholar] [CrossRef]
- Periconia Tode, Fungi Mecklenburgenses Selecti 2: 2. 1791. Available online: https://www.mycobank.org (accessed on 4 November 2020).
- NCBI Taxonomy Browser of Periconia. Available online: https://www.ncbi.nlm.nih.gov/Taxonomy/Browser/wwwtax.cgi?mode=Undef&name=Periconia (accessed on 4 November 2020).
- Wani, M.C.; Taylor, H.L.; Wall, M.E.; Coggon, P.; McPhail, A.T. Plant antitumor agents VI: The isolation and structure of taxol, a novel antilekemic and antitumor agent from Taxus brevifolia. J. Am. Chem. Soc. 1971, 2325–2327. [Google Scholar] [CrossRef]
- Stierle, A.; Strobel, G.A.; Stierle, D. Taxol and taxane production by Taxomyces andreanae, an endophytic fungus of Pacific yew. Science 1993, 260, 214–216. [Google Scholar] [CrossRef]
- Bach, T.J.; Rohmer, M. Isoprenoid Synthesis in Plants and Microorganisms: New Concepts and Experimental Approaches; Springer Science & Business Media: New York, NY, USA, 2012. [Google Scholar] [CrossRef]
- Liu, J.M.; Zhang, D.W.; Du, W.Y.; Zhang, M.; Zhao, J.L.; Chen, R.D.; Xie, K.B.; Dai, J.G. Four new monoterpenoids from an endophytic fungus Periconia sp. F-31. J. Asian Nat. Prod. Res. 2017, 19, 541–549. [Google Scholar] [CrossRef]
- Ge, H.L.; Zhang, D.W.; Li, L.; Xie, D.; Zou, J.H.; Si, Y.K.; Dai, J. Two new terpenoids from endophytic fungus Periconia sp. F-31. Chem. Pharm. Bull. 2011, 59, 1541–1544. [Google Scholar] [CrossRef][Green Version]
- Delfine, S.; Csiky, O.; Seufert, G.; Loreto, F. Fumigation with exogenous monoterpenes of a non-isoprenoid-emitting oak (Quercus suber, Monoterpene acquisition, translocation, and effect on the photosynthetic properties at high temperatures. New Phytol. 2000, 146, 27–36. [Google Scholar] [CrossRef]
- Kim, S.; Shin, D.S.; Lee, T.; Oh, K.B. Periconicins, two new fusicoccane diterpenes produced by an endophytic fungus Periconia sp. with antibacterial activity. J. Nat. Prod. 2004, 67, 448–450. [Google Scholar] [CrossRef]
- Ballio, A.; Chain, E.B.; De Leo, P.; Erlanger, B.F.; Mauri, M.; Tonolo, A. Fusicoccin: A new wilting toxin produced by Fusicoccum amygdali. Del. Nat. 1964, 203, 297. [Google Scholar] [CrossRef]
- Muromtsev, G.S.; Voblikova, V.D.; Kobrina, N.S.; Koreneva, V.M.; Krasnopolskaya, L.M.; Sadovskaya, V.L. Occurrence of fusicoccanes in plants and fungi. J. Plant Growth Regul. 1994, 13, 39–49. [Google Scholar] [CrossRef]
- Christianson, D.W. Unearthing the roots of the terpenome. Curr. Opin. Chem. Biol. 2008, 12, 141–150. [Google Scholar] [CrossRef] [PubMed]
- Lesburg, C.A.; Caruthers, J.M.; Paschall, C.M.; Christianson, D.W. Managing and manipulating carbocations in biology: Terpenoid cyclase structure and mechanism. Curr. Opin. Struct. Biol. 1998, 8, 695–703. [Google Scholar] [CrossRef]
- Xu, J.Q.; Hu, L.H. Five new eremophilane sesquiterpenes from Ligularia przewalskii. Helv. Chim. Acta 2008, 91, 951–957. [Google Scholar] [CrossRef]
- Yamada, T.; Doi, M.; Miura, A.; Harada, W.; Hiramura, M.; Minoura, K.; Tanaka, R.; Numata, A. Absolute stereostructures of cell-adhesion inhibitors, peribysins A, E, F and G, produced by a sea hare-derived Periconia sp. J. Antibiot. 2005, 58, 185–191. [Google Scholar] [CrossRef]
- Yamada, T.; Minoura, K.; Tanaka, R.; Numata, A. Cell-adhesion inhibitors produced by a sea hare-derived Periconia sp.: II absolute stereostructures of peribysins H and I. J. Antibiot. 2006, 59, 345–350. [Google Scholar] [CrossRef]
- Yamada, T.; Minoura, K.; Tanaka, R.; Numata, A. Cell-adhesion inhibitors produced by a sea hare-derived Periconia sp. III absolute stereostructures of peribysin J and macrosphelide M. J. Antibiot. 2007, 60, 370–375. [Google Scholar] [CrossRef]
- Athawale, P.R.; Kalmode, H.P.; Motiwala, Z.; Kulkarni, K.A.; Reddy, D.S. Overturning the peribysin family natural products isolated from Periconia byssoides OUPS-N133: Synthesis and stereochemical revision of peribysins A, B, C, F, and G. Org. Lett. 2020, 22, 3104–3109. [Google Scholar] [CrossRef]
- Koshino, H.; Satoh, H.; Yamada, T.; Esumi, Y. Structural revision of peribysins C and D. Tetrahedron Lett. 2006, 47, 4623–4626. [Google Scholar] [CrossRef]
- Angeles, A.R.; Waters, S.P.; Danishefsky, S.J. Total syntheses of (+) and (−) peribysin E. J. Am. Chem. Soc. 2008, 130, 13765–13770. [Google Scholar] [CrossRef]
- Inose, K.; Tanaka, K.; Yamada, T.; Koshino, H.; Hashimoto, M.J. Isolation of peribysins O, P, and Q from Periconia macrospinosa KT3863 and configurational reinvestigation of peribysin E diacetate from Periconia byssoides OUPS-N133. Nat. Prod. 2019, 82, 911–918. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Ge, H.; Zou, J.H.; Tao, X.; Chen, R.; Dai, J. Periconianone A, a new 6/6/6 carbocyclic sesquiterpenoid from endophytic fungus Periconia sp. with neural anti-inflammatory activity. Org. Lett. 2014, 16, 1410–1413. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Zhang, D.; Zhang, M.; Zhao, J.; Chen, R.; Wang, N.; Zhang, D.; Dai, J.J. Eremophilane sesquiterpenes from an endophytic fungus Periconia species. Nat. Prod. 2016, 79, 2229–2235. [Google Scholar] [CrossRef] [PubMed]
- Rücker, G. Sesquiterpenes. Angew. Chem. Int. Ed. Engl. 1973, 12, 793–806. [Google Scholar] [CrossRef]
- Cheng, A.X.; Lou, Y.G.; Mao, Y.B.; Lu, S.; Wang, L.J.; Chen, X.Y. Plant terpenoids: Biosynthesis and ecological functions. J. Integr. Plant Biol. 2007, 49, 179–186. [Google Scholar] [CrossRef]
- Gao, W.; Chai, C.; He, Y.; Li, F.; Hao, X.; Cao, F.; Gu, L.; Liu, J.; Hu, Z.; Zhang, Y. Periconiastone A, an antibacterial ergosterol with a pentacyclo-[8.7.0.01,5.02,14.010,15]heptadecane system from Periconia sp. TJ403-rc01. Org. Lett. 2019, 21, 8469–8472. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.M.; Zhang, D.W.; Zhang, M.; Chen, R.D.; Yan, Z.; Zhao, J.Y.; Zhao, J.L.; Wang, N.; Dai, J.G. Periconones B–E, new meroterpenoids from endophytic fungus Periconia sp. Chin. Chem. Lett. 2017, 28, 248–252. [Google Scholar] [CrossRef]
- Moraga, J.; Gomes, W.; Pinedo, C.; Cantoral, J.M.; Hanson, J.R.; Carbú, M.; Garrido, C.; Durán-Patrón, R.; Collado, I.G. The current status on secondary metabolites produced by plant pathogenic Colletotrichum species. Phytochem. Rev. 2019, 18, 215–239. [Google Scholar] [CrossRef]
- Korman, T.P.; Ames, B.; Tsai, S.C. 1.08: Structural Enzymology of Polyketide Synthase: The Structure–Sequence–Function Correlation; Elsevier: Oxford, UK, 2010. [Google Scholar] [CrossRef]
- Pfeifer, B.A.; Khosla, C. Biosynthesis of polyketides in heterologous hosts. Microbiol. Mol. Biol. Rev. 2001, 65, 106–118. [Google Scholar] [CrossRef]
- Zaghouani, M.; Kunz, C.; Guédon, L.; Blanchard, F.; Nay, B. First total synthesis, structure revision, and natural history of the smallest cytochalasin: (+) Periconiasin G. Chem. Eur. J. 2016, 22, 15257–15260. [Google Scholar] [CrossRef]
- Zhang, D.; Ge, H.; Xie, D.; Chen, R.; Zou, J.H.; Tao, X.; Dai, J. Periconiasins A-C, new cytotoxic cytochalasans with an unprecedented 9/6/5 tricyclic ring system from endophytic fungus Periconia sp. Org. Lett. 2013, 15, 1674–1677. [Google Scholar] [CrossRef]
- Zhang, D.; Tao, X.; Chen, R.; Liu, J.; Li, L.; Fang, X.; Yu, L.; Dai, J. Pericoannosin A, a polyketide synthase-nonribosomal peptide synthetase hybrid metabolite with new carbon skeleton from the endophytic fungus Periconia sp. Org. Lett. 2015, 17, 4304–4307. [Google Scholar] [CrossRef]
- Liu, J.; Zhang, D.; Zhang, M.; Liu, X.; Chen, R.; Zhao, J.; Li, L.; Wang, N.; Dai, J. Periconiasins I and J, two new cytochalasans from an endophytic fungus Periconia sp. Tetrahedron Lett. 2016, 57, 5794–5797. [Google Scholar] [CrossRef]
- Fan, Y.; Zhang, D.; Tao, X.; Wang, Y.; Liu, J.; Li, L.; Zhao, J.; Yu, L.; He, Y.P.; Dai, J.; et al. Biosynthetic hypothesis-guided discovery and total syntheses of PKS-NRPS hybrid metabolites from endophytic fungus Periconia species. Org. Lett. 2019, 21, 1794–1798. [Google Scholar] [CrossRef]
- Zhang, D.W.; Tao, X.Y.; Liu, J.M.; Chen, R.D.; Zhang, M.; Fang, X.M.; Yu, L.Y.; Dai, J.G. A new polyketide synthase-nonribosomal peptide synthetase hybrid metabolite from plant endophytic fungus Periconia sp. Chin. Chem. Lett. 2016, 27, 640–642. [Google Scholar] [CrossRef]
- Giles, D.; Turner, W.B. Perkin Transactions. Chlorine-containing metabolites of Periconia macrospinosa. J. Chem. Soc. 1969, 1, 2187–2189. [Google Scholar] [CrossRef]
- Henderson, G.B.; Hill, R.A. The biosynthesis of chlorine-containing metabolites of Periconia macrospinosa. J. Chem. Soc. 1982, 1, 3037–3039. [Google Scholar] [CrossRef]
- Macko, V.; Stimmel, M.B.; Peeters, H.; Wolpert, T.J.; Dunkle, L.D.; Acklin, W.; Bänteli, R.; Jaun, B.; Arigoni, D. The structure of circinatin, a non-toxic metabolite from the plant pathogenic fungus Periconia circinate. Experientia 1990, 46, 1206–1209. [Google Scholar] [CrossRef]
- Macko, V.; Stimmel, M.B.; Wolpert, T.J.; Dunkle, L.D.; Acklin, W.; Bänteli, R.; Jaun, B.; Arigoni, D. Structure of the host-specific toxins produced by the fungal pathogen Periconia circinate. Proc. Natl. Acad. Sci. USA 1992, 89, 9574–9578. [Google Scholar] [CrossRef]
- Inose, K.; Tanaka, K.; Koshino, H.; Hashimoto, M. Cyclopericodiol and new chlorinated melleins isolated from Periconia macrospinosa KT3863. Tetrahedron 2019, 75, 130470. [Google Scholar] [CrossRef]
- Paek, S.M.; Suh, Y.G. Synthetic studies on bioactive natural polyketides: Intramolecular nitrile oxide-olefin cycloaddition approach for construction of a macrolactone skeleton of macrosphelide B. Molecules 2011, 16, 4850–4860. [Google Scholar] [CrossRef]
- Wińska, K.; Mączka, W.; Grabarczyk, M.; Sugimoto, K.; Matsuya, Y.; Szumny, A.; Anioł, M. A macrosphelide as the unexpected product of a Pleurotus ostreatus strain-mediated biotransformation of halolactones containing the gem-dimethylcyclohexane ring, Part 1. Molecules 2016, 21, 859. [Google Scholar] [CrossRef]
- Bhilabutra, W.; Techowisan, T.; Peberdy, J.F.; Lumyong, S. Antimicrobial activity of bioactive compounds from Periconia siamensis CMUGE015. Res. J. Microbiol. 2007, 2, 749–755. [Google Scholar] [CrossRef]
- Fun, H.K.; Bhilabutra, W.; Tuntiwachwuttikul, P.; Chantrapromma, S. 5,8-Dihydroxy-10-methyl-5,8,9, 10-tetrahydro-2H-oxecin-2-one. Acta Crystallogr. Sect. E Struct. Rep. Online 2006, 62, 2478–2480. [Google Scholar] [CrossRef]
- Tsuda, M.; Mugishima, T.; Komatsu, K.; Sone, T.; Tanaka, M.; Mikami, Y.; Kobayashi, J.I. Modiolides A and B, two new 10-membered macrolides from a marine-derived fungus. J. Nat. Prod. 2003, 66, 412–415. [Google Scholar] [CrossRef] [PubMed]
- Yamada, T.; Iritani, M.; Doi, M.; Minoura, K.; Ito, T.; Numata, A. Absolute stereostructures of cell-adhesion inhibitors, macrosphelides C, E–G and I, produced by a Periconia species separated from an Aplysia sea hare. J. Chem. Soc. Perkin. Trans. 2001, 1, 3046–3053. [Google Scholar] [CrossRef]
- Nakamura, H.; Ono, M.; Yamada, T.; Numata, A.; Akita, H. Determination of the absolute stereostructure of seco-Macrosphelide E produced by a fungal strain from a sea hare. Chem. Pharm. Bull. 2002, 50, 303–306. [Google Scholar] [CrossRef][Green Version]
- Yamada, T.; Iritani, M.; Minoura, K.; Numata, A.; Kobayashi, Y.; Wang, Y.G. Absolute stereostructures of cell adhesion inhibitors, macrosphelides H and L, from Periconia byssoides OUPS-N133. J. Antibiot. 2002, 55, 147–154. [Google Scholar] [CrossRef]
- Yamada, T.; Iritani, M.; Ohishi, H.; Tanaka, K.; Minoura, K.; Doi, M.; Numata, A. Pericosines, antitumour metabolites from the sea hare-derived fungus Periconia byssoides: Structures and biological activities. Org. Biomol. Chem. 2007, 5, 3979–3986. [Google Scholar] [CrossRef] [PubMed]
- Usami, Y.; Ichikawa, H.; Arimoto, M. Synthetic efforts for stereo structure determination of cytotoxic marine natural product Pericosines as metabolites of Periconia sp. from sea hare. Int. J. Mol. Sci. 2008, 9, 401–421. [Google Scholar] [CrossRef]
- Andrus, M.B.; Shih, T.L. Synthesis of tuckolide, a new cholesterol biosynthesis inhibitor. J. Org. Chem. 1996, 61, 8780–8785. [Google Scholar] [CrossRef]
- Shin, D.S.; Oh, M.N.; Yang, H.C.; Oh, K.B. Biological characterization of periconicins, bioactive secondary metabolites, produced by Periconia sp. OBW-15. J. Microbiol. Biotechnol. 2005, 15, 216–220. [Google Scholar]
- Babu, D.C.; Rao, C.B.; Venkatesham, K.; Selvam, J.J.P.; Venkateswarlu, Y. Toward synthesis of carbasugars (+) gabosine C,(+) COTC,(+) pericosine B, and (+) pericosine C. Carbohydr. Res. 2014, 388, 130–137. [Google Scholar] [CrossRef]
- Boyd, D.R.; Sharma, N.D.; Acaru, C.A.; Malone, J.F.; O’Dowd, C.R.; Allen, C.C.; Stevenson, P.J. Chemoenzymatic synthesis of carbasugars (+) pericosines A−C from diverse aromatic cis-dihydrodiol precursors. Org. Lett. 2010, 12, 2206–2209. [Google Scholar] [CrossRef]
- Donohoe, T.J.; Blades, K.; Helliwell, M.; Waring, M.J.; Newcombe, N.J. The synthesis of (+) pericosine B. Tetrahedron Lett. 1998, 39, 8755–8758. [Google Scholar] [CrossRef]
- Garcia Ruano, J.L.; López-Cantarero, J.; Alemparte, C. Toward the synthesis of (+) pericosine B. Phosphorus Sulfur Silicon Relat. Elem. 2005, 180, 1493–1494. [Google Scholar] [CrossRef]
- Muni Raju, C.; Rao, J.P.; Rao, B.V. Stereoselective synthesis of (+) pericosine B and (+) pericosine C using ring closing metathesis approach. Tetrahedron Asymmetry 2012, 23, 86–93. [Google Scholar] [CrossRef]
- Tripathi, S.; Shaikh, A.C.; Chen, C. Facile carbohydrate-based stereocontrolled divergent synthesis of (+)-pericosines A and B. Org. Biomol. Chem. 2011, 9, 7306–7308. [Google Scholar] [CrossRef]
- Usami, Y. Synthesis of Marine-Derived Carbasugar Pericosines. In Studies in Natural Products Chemistry; Elsevier: Amsterdam, The Netherland, 2014. [Google Scholar] [CrossRef]
- Usami, Y.; Hatsuno, C.; Yamamoto, H.; Tanabe, M.; Numata, A. Synthesis of the epimer of pericosine B from (−)-quinic acid. Chem. Pharm. Bull. 2004, 52, 1130–1133. [Google Scholar] [CrossRef]
- Usami, Y.; Horibe, Y.; Takaoka, I.; Ichikawa, H.; Arimot, M. First total synthesis of (-)-pericosine A from (-)-shikimic acid: Structure revision and determination of the absolute configuration of antitumor natural product pericosine A. Synlett 2006, 10, 1598–1600. [Google Scholar] [CrossRef]
- Usami, Y.; Numata, A. Examination of the reactivity of hydroxy groups in multioxygenated cyclohexanoids: Synthetic study toward cytotoxic pericosine B. Chem. Pharm. Bull. 2004, 52, 1125–1129. [Google Scholar] [CrossRef] [PubMed]
- Usami, Y.; Ohsugi, M.; Mizuki, K.; Ichikawa, H.; Arimoto, M. Facile and efficient synthesis of naturally occurring carbasugars (+)-Pericosines A and C. Organic Lett. 2009, 11, 2699–2701. [Google Scholar] [CrossRef] [PubMed]
- Usami, Y.; Ohsugi, M.; Mizuki, K.; Ichikawa, H.; Arimoto, M. Synthesis of (−)-pericosine B, the antipode of the cytotoxic marine natural product. Org. Bio. Chem. 2009, 7, 315–318. [Google Scholar] [CrossRef] [PubMed]
- Usami, Y.; Takaoka, I.; Ichikawa, H.; Horibe, Y.; Tomiyama, S.; Ohtsuka, M.; Imanishi, Y.; Arimoto, M. First total synthesis of antitumor natural product (+)-and (−)-pericosine A: Determination of absolute stereo structure. J. Org. Chem. 2007, 72, 6127–6134. [Google Scholar] [CrossRef] [PubMed]
- Usami, Y.; Ueda, Y. Synthetic study toward antitumour natural product pericosine A. Chem. Lett. 2005, 34, 1062–1063. [Google Scholar] [CrossRef]
- Usami, Y.; Ueda, Y. Stereoselective syntheses of diastereomers of antitumor natural product pericosine a from (−)-quinic acid. Synthesis 2007, 20, 3219–3225. [Google Scholar] [CrossRef]
- Hayashi, M.; Kim, Y.P.; Hiraoka, H.; Natori, M.; Takamatsu, S.; Kawakubo, T.; Masuma, R.; Komiyama, K.; Omura, S. Macrosphelide, a novel inhibitor of cell-cell adhesion molecule. J. Antibiot. 1995, 48, 1435–1439. [Google Scholar] [CrossRef]
- Kim, H.Y.; Park, E.J.; Joe, E.H.; Jou, I. Curcumin suppresses Janus kinase-STAT inflammatory signaling through activation of Src homology 2 domain-containing tyrosine phosphatase 2 in brain microglia. J. Immunol. 2003, 171, 6072–6079. [Google Scholar] [CrossRef]
- Yang, S.; Zhang, D.; Yang, Z.; Hu, X.; Qian, S.; Liu, J.; Wilson, B.; Block, M.; Hong, J.S. Curcumin protects dopaminergic neuron against LPS induced neurotoxicity in primary rat neuron/glia culture. Neurochem. Res. 2008, 33, 2044–2053. [Google Scholar] [CrossRef] [PubMed]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Azhari, A.; Supratman, U. The Chemistry and Pharmacology of Fungal Genus Periconia: A Review. Sci. Pharm. 2021, 89, 34. https://doi.org/10.3390/scipharm89030034
Azhari A, Supratman U. The Chemistry and Pharmacology of Fungal Genus Periconia: A Review. Scientia Pharmaceutica. 2021; 89(3):34. https://doi.org/10.3390/scipharm89030034
Chicago/Turabian StyleAzhari, Azmi, and Unang Supratman. 2021. "The Chemistry and Pharmacology of Fungal Genus Periconia: A Review" Scientia Pharmaceutica 89, no. 3: 34. https://doi.org/10.3390/scipharm89030034
APA StyleAzhari, A., & Supratman, U. (2021). The Chemistry and Pharmacology of Fungal Genus Periconia: A Review. Scientia Pharmaceutica, 89(3), 34. https://doi.org/10.3390/scipharm89030034





