3.1. Preliminary Phytochemical Screening
The phytochemical study of the methanol extract of
Crocus leaves revealed the presence of various bioactive compounds (
Table 1), including polyphenols, tannins, flavonoids, xanthones, hydroxycinnamic acids, hydrolysable tannins, triterpenoids, saponins, aminoacids, and proteins. Alkaloids and coumarins were not found in this raw material.
Quantitative phytochemical analysis showed that C. sativus leaves contained the largest amount of biologically active compounds, primarily phenolic and flavonoid compounds.
Previously, Baba et al. [
14] reported that the total phenolic content of ethanolic extracts of
C. sativus leaves from Jammu and Kashmir (India) was 5.62 mg/g, using the Folin–Ciocalteu reagent method, and that the total flavonoid content was 1.61 ± 0.12 mg RE/g. According to Jadouali et al. [
13], the total phenolic content for
C. sativus leaves from Morocco was 81.69 ± 1.58 mg GAE/mg of powder crude extract, and the total flavonoids content was 50.64 ± 1.63 mg of CE/g of dry plant material. In another investigation [
15] of saffron from Morocco, the total phenolic and flavonoid content of the methanolic
C. sativus leaf extracts was 94 ± 17 mg of GA eq/g and 27 ± 4 mg of quercetin eq/g, respectively. This data is practically correlated with previous studies and is probably due to a single area of plant cultivation. In Spain, in
C. sativus leaves, the total polyphenol content was 1.5 g GAE/100 g [
11]. Thus, it can be seen that the place of cultivation and climatic factors [
27,
28,
29] have a great influence on the composition of phenolic compounds in
Crocus leaves.
The current phytochemical analysis revealed the high total phenolic content of
C. sativus leaves from Ukraine, corresponding to 5.44 ± 0.01 mg GAE/g DW and a flavonoid content of 2.63 ± 0.05 mg RE/g DW. Phenolic compounds play a key role in scavenging free radicals, inhibiting hydrolytic and oxidative enzymes, and exhibiting anti-inflammatory effects [
30]. Therefore, it is possible to expect a pronounced antioxidant effect for
Crocus leaves from Ukraine. Therefore, a deeper study of the chemical profile of
Crocus leaves was carried out.
3.2. Identification and Content of Phenolic Compounds by HPLC
The most common constituents in the leaves of the Iridaceae plant family are flavone C-glycosides, flavanols, and isoflavones derivatives [
7,
20,
31]. The most studied are leaves and rhizomes of
Iris spp. In plant derivatives of 7-methyl ester and 7,4-dimethyl ester of apigenin, derivatives of 6,7-methylenedioxyflavans, as well as various isoflavonoids and xanthones, have been found. Derivatives of myricetin, kaempferol, and quercetin were found in the leaves of different
Crocus species [
6]. However, data on the composition of flavonoids in
Crocuses are very limited.
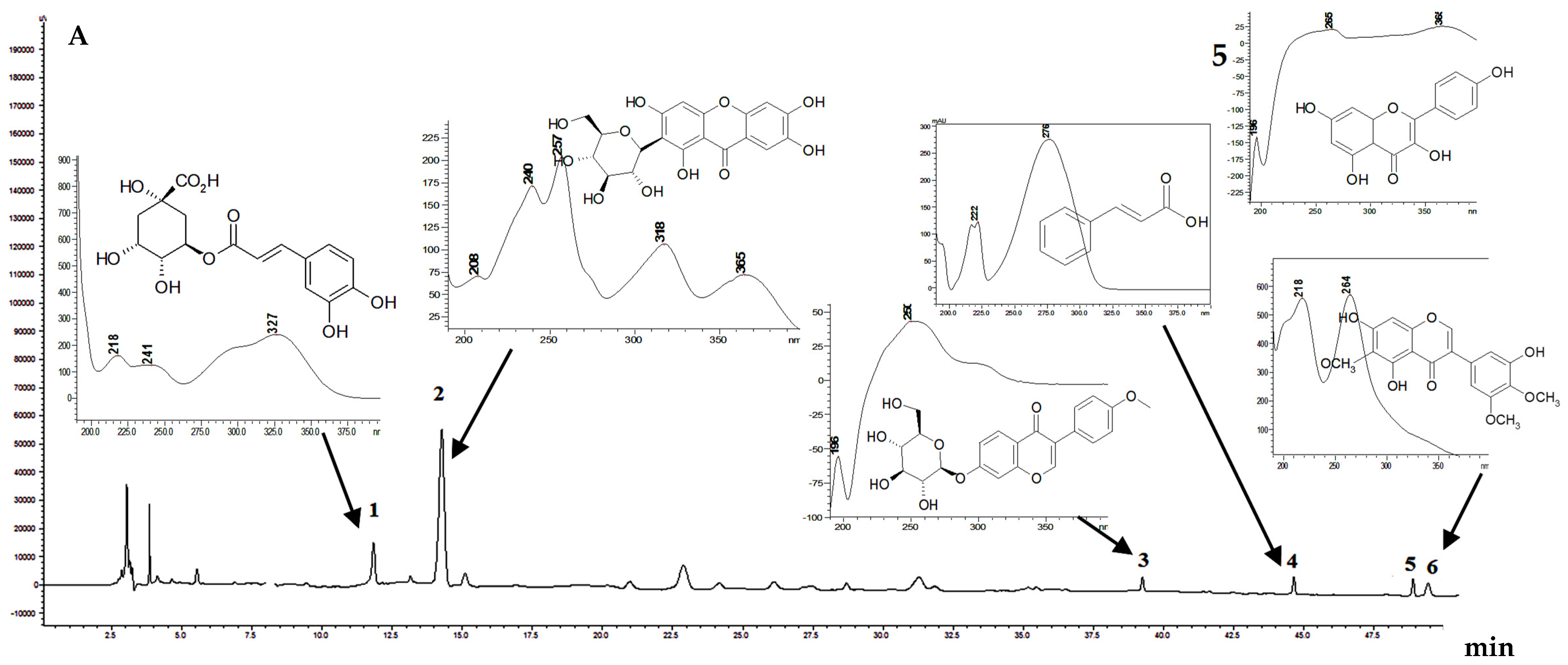
Compounds identification in
Crocus leaves extract was based on their coelution with reference compounds and on the UV/MS spectroscopic data according to the HPLC-DAD method. For the identification of phenolic compounds, a more selective and sensitive UPLC method for plants using the negative ionization mode was selected [
32]. Six phenolic compounds were detected in the studied
Crocus leaves for the first time (
Figure 1). The quantification of the phenolic compounds using the available standards was carried out (
Table 2). All identified compounds were of a minor amount, among them two hydroxycinnamic acid derivatives, i.e.,
trans-cinnamic acid (0.15 ± 0.002 mg/g) and chlorogenic acid (0.31 ± 0.004 mg/g); flavonoid kaempferol (0.032 ± 0.001 mg/g); and two isoflavones—ononin (0.04 ± 0.001 mg/g) and irigenin (0.002 ± 0.001 mg/g)—were determined. Additionally, xanthone mangiferin was found in
C. sativus leaves for the first time, and its content was the highest (1.26 ± 0.02 mg/g) among all identified compounds. As a result,
trans-cinnamic acid, chlorogenic acid, ononin, irigenin, and mangiferin were identified first in
C. sativus leaves from Ukraine.
The HPLC method used was validated for such parameters as linearity range, LOD, LOQ, accuracy, precision, repeatability, and specificity for each analyte. These are presented in
Table 3 and
Table 4. A regression equation and correlation coefficient ranging from 0.9999 to 1.000 revealed a good linearity response within the tested ranges. The series of calibration solutions were prepared and separated under the optimal conditions as described above. The LOD and LOQ values (
Table 3) indicate that the proposed method demonstrates good sensitivity for quantifying seven phenolic compounds in
Crocus leaves. The repeatability was observed in the range of 0.24–1.05%, which was satisfactory and demonstrated the good repeatability of the proposed method. The determination of the main compounds in the test solutions was carried out by comparing the retention times of peaks and UV spectrum obtained from the chromatogram of the standard solution (
Table 2). All results showed the repeatability, accuracy, high sensitivity, and good linearity of the method.
3.3. UPLC-MS/MS Analysis of Phenolic Compounds
To confirm the identified compounds in
Crocus leaves, further UPLC-MS/MS analysis of these components was performed in negative ion mode. Their retention times (t
R), calculated molecular weights, and MS/MS data are shown in
Table 2, respectively. All compounds were unambiguously identified with the reference standards comparison. Peak
1 ([M
−H]
− at
m/
z 353) was assigned to monocaffeoyquinic acids. The MS spectrum of peak
1 was characterized by the loss of one moiety of caffeic acid, due to the ester bond, which readily dissociated and was identified as 3-
O-caffeylquinic acid or chlorogenic acid in comparison with the reference standards. In the MS spectrum, peak
2 produced the [M
−H]
– ion at
m/
z 421. The MS
2 spectrum gave the predominant fragment ions at
m/
z 331 and 301 owing to the neutral loss of C
3H
6O
3 and C
4H
8O
4, respectively. The low-intensity product ion at
m/
z 259 appeared in the MS
2 spectrum due to the loss of the glucose residue from the [M
−H]
– ion. The MS
3 spectrum of the precursor ion at
m/
z 301 gave the product ion at
m/
z 273, 271, and 257 resulting from the loss of CO, CH
2O, and CO
2, respectively. The RDA fragmentation reaction was also observed in the MS/MS spectrum and corresponded with the literature data [
33]. Peak
4 produced the [M
–H]
– ion at
m/
z 147 at 44.76 min and was identified as
trans-cinnamic acid. Peaks
5 produced the [M
−H]
– ion at
m/
z 285 and were identified as kaempferol (calculated for C
15H
10O
6,
m/
z 286). They were further confirmed by comparing the retention time and the MS/MS spectra with the corresponding standards. In the MS spectrum, peak
6 produced the [M
−H]
– ion at
m/
z 459 and lost three methyl groups, showing fragments at
m/
z 344 and
m/
z 329. The isoflavonoid ononin was identified in the positive ionization mode according to the retro-Diels–Adel reaction (RDA) [
33]. Since we were able to identify almost all substances in the negative ion mode, we did not carry out a separate determination for ononin. According to Luo et al. [
33], the excimer ion peak [M
+H]
+ of ononin has a
m/
z of 431. However, we did not perform such a definition.
3.4. GF-AAS Analysis of Mineral Composition
The literature data of macro- and microelement analysis in
C. sativus leaves are limited. It is known that Jadouali et al. [
13] only established the calcium, potassium, sodium, nitrogen, iron, and zinc elements, as well as a high concentration of iron (985 mg/kg), in
C. sativus leaves from Morocco.
As a result of the element profile of
Crocus leaves from Ukraine, the presence of 15 minerals has been determined (
Table 5). Among the identified elements were three macroelements, five mezoelements, and seven ultramicroelements. It is interesting to note the presence of a significant amount of essential and conditionally essential elements, such as calcium (Ca), iron (Fe), manganese (Mn), zinc (Zn), silica (Si), phosphorus (P), and nickel (Ni).
The results of the mineral content of
C. sativus leaves from Ukraine are presented in
Table 5. In leaves, potassium and calcium were found to be the most abundant minerals with a value of 1525 mg/100 g and 550 mg/100 g, respectively. The high content of potassium in
Crocus leaves is due to the physiological function of potassium in the process of transport in plants: first and foremost, with the participation of a highly osmotic active substance in the induction of corms pressure and transport of assimilates in the leaf phloem [
34]. Calcium accumulates at the deposit places in the cell (vacuole, endoplasmic reticulum), is gradually released, and performs its physiological function [
35]. The plant has the highest calcium content in the photosynthetically active leaves vacuole in the form of oxalate or other hardly soluble salts. Calcium passes slowly through the xylem, so its content is lower than potassium. A significant decrease in the content of magnesium (6.73 mg/100 g) in
Crocus leaves is directly related to the content of potassium in the raw material and is associated with the mechanisms of absorption of these nutrients from the soil [
36,
37]. It should be noted that the crop
C. sativus is cultivated without any nitrates, pesticides, or additional growth stimulants [
29,
38]. Nitrogen, phosphorus, and potassium fertilizers are applied only in April after harvesting
Crocus corms from the field. The current study showed that
C. sativus leaves have a pronounced mineral composition without additional fertilizers.
3.5. GC-MS Analysis of Amino Acids and Short-Chained and Long-Chain Carboxylic Acids
The amino acid composition was studied only for
C. sativus stigma from Spain, Italy, Greece, and Iran, and high levels of alanine, proline, and aspartic acid were found [
39]. Fatty acids, such as myristic, stearic, palmitic, palmitoleic, oleic, linoleic, linolenic, and arachidonic acids, have also been found in
C. sativus stigmas [
40,
41].
Linoleic acid, linolenic acid, and palmitic acid were the predominant fatty acids of
C. sativus flowers [
42]. Lauric acid and hexadecanoic acid have been found in
C. sativus tepals, stamens, and stigmas [
43]. Ursolic, oleanolic, palmitic, palmitoleic, oleic, linolenic, and linoleic acids were found in
C. sativus corms [
44], as well as amino acids (aspartic acid, glutamic acid, cysteine, serine, glycine, proline, phenylalanyl, leucine, valine, and methionine) [
45]. No published data on the amino acids and fatty acids composition of
C. sativus leaves has been found.
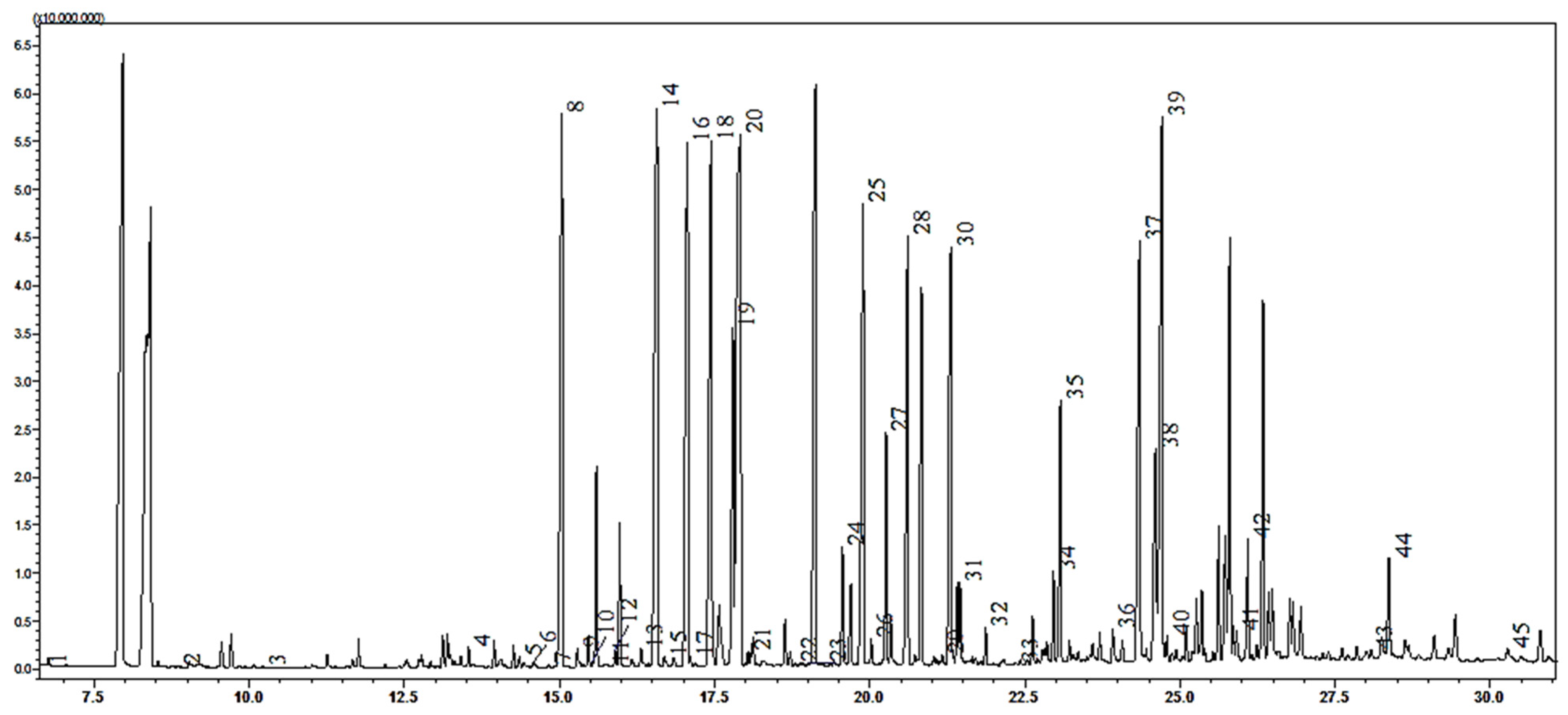
For the investigation, a method for the simultaneous study of fatty acids, short-chained carboxylic acids, and amino acids in
C. sativus leaves from Ukraine was used. As a result, 44 metabolites were found. This separation was achieved by the derivatization method. The derivatization procedure is highly dependent on the solvent used. Therefore, in our previous work [
22,
23], we developed the GC-MS method for organic acid investigation. Different times, temperatures and the effect of the reagent amount on the derivatization process were tested. The optimal conditions for the complete derivatization of most amino acids and organic acids were ultrasonic extraction for 10 min, followed by derivatization with acetonitrile and N-Methyl-N-tert-butyldimethylsilyltrifluoroacetamide MTBSTFA (1:1) for 4 h at 70 °C.
All compounds were identified by comparing the retention times of selected acids in specific MS chromatograms. The content of identified carboxylic acids and amino acids is presented in
Table 6.
In the current study, we have determined the presence and quantity of 22 amino acids, 14 short-chained carboxylic acids, 7 fatty acids, and 1 phosphorus acid (
tris-phosphoric acid) in
C. sativus leaves by the GC-MS method. According to the results, 45 different acids are present in
Crocus leaves. These are the first complete results of the composition of carboxylic and amino acids in
Crocus leaves. Furthermore, this method was used to quantify the acids content in the raw material (
Table 6,
Figure 2).
L-Glutamine, Isoleucine, L-alanine, L-leucine, L-pyroglutamic, and L-phenylalanine acids were present in large amounts—7374 µg/g, 7965 µg/g, 6744 µg/g, 6955 µg/g, 6233 µg/g, and 6032 µg/g, respectively. High amounts of L-glutamine may be due to the glutamate synthase cycle [
46], where L-glutamine is formed from simple compounds, such as ammonia, and only then are other amino acids, such as arginine, tryptophan, histidine, or asparagine, formed [
47]. The seven essential amino acids in leaves that were present in high concentration included isoleucine (7965 µg/g), L-leucine (6955 µg/g), L-lysine (866 µg/g), L-methionine (398 µg/g), L-phenylalanine (6032 µg/g), L-threonine (2701 µg/g), L-tryptophan (1186 µg/g), and L-valine (6151 µg/g). As for the total amino acids content in
C. sativus leaves, it exceeds 43,174 µg/g and shows that this raw material can be an excellent and underestimated source of proteinogenic amino acids [
48], especially glutamine, phenylalanine, isoleucine, alanine, and leucine.
For the composition of amino acids, a high content of isoleucine, L-leucine, L-phenylalanine, L-pyroglutamic acid, L-glutamine, L-alanine, and L-tryptophan was observed along with a low content of L-glycine, which indicates the plant’s response to cold stress [
49,
50] as well as consistent with previously published data for this genus [
51].
Among the organic acids of the tricarboxylic acid cycle (TCA cycle) in
C. sativus leaves were found citric acid (834 µg/g), fumaric acid (195 µg/g), and malic acid (925 µg/g), which had the highest content of all short-chain carboxylic acids. In a TCA cycle, citric acid and malic acid are generally the most accumulated organic acids in plants [
52]; at the same time,
Crocus leaves have an increased content of malic acid during acclimatization to the low temperature [
53,
54] of Ukraine in the postharvest period. In addition, the path analysis of the effects of different acids on malic acid content revealed that citric acid has a maximum direct effect on malic acid [
55]. Furthermore, 3,3-dimethylacrylic acid, 4-hydroxybutanoic acid, and lactic acid were found in amounts of 187 µg/g, 162 µg/g, and 98 µg/g, respectively. The content of other organic acids found did not exceed 50 µg/g. Additionally, polyphenols and organic acids may be responsible for the antioxidant activity of the plant extracts.
The analysis of fatty acids of C. sativus leaves showed that seven acids (myristic acid, palmitic acid, linoelaidic acid, α-linolenic acid, stearic acid, behenic acid, and lignoceric acid) were detected. The total long-chain carboxylic acids content was 63,544 µg/g of the total acids yield and is the largest part of all identified acids in the Crocus leaves. The dominant saturated acid was palmitic (C16:0) in Crocus leaves, with a content of 6997 µg/g, followed by lignocerinic acid (C24:0), with a content of 1611 µg/g. The content of unsaturated fatty acids was two times higher than that of saturated acids.
The GC-MS analyses revealed the essential fatty acids α-linolenic acid (ω3) (22,490 µg/g) and linoelaidic acid (ω6) (9880 µg/g) as major constituents among the acids that were found in
Crocus leaves for the first time. The biosynthesis of dienoic and trienoic acids 18:2ω6 and 18:3ω3 plays a defining role in the adaptation of plants to hypothermia. These polyunsaturated fatty acids regulate the “fluidity” of cell membranes in a wide temperature range and influence the resistance of plants to difficult environments. In stressful environments (low temperature, freezing, salinity, drought), the amount of unsaturated fatty acids is much higher than the saturated acids content in the plant organs [
56,
57]. The high content of linolenic acid in
Crocus leaves is probably due precisely to the plant’s adaptation to the low temperatures in Ukraine (December +3/−5 °C) during the harvested period.
The presence of
tris-phosphoric acid (1305 µg/g) in
Crocus leaves, according to Hu et al. [
58], also signals the plant’s response to temperature stress, which is expressed in shifts in the metabolite profile.
Tris-phosphoric acid could supply adequate P secondary compounds for cell energy metabolism in stress conditions. In this case, an increase of organic acids (malic acid, citric acid,
tris-phosphoric acid), amino acids (isoleucine, pyroglutamic acid, phenylalanine, alanine, glutamine), and unsaturated fatty acids (linoelaidic acid, linoleic acid) for this plant was established. Some of the phenolic compounds and organic acids might be responsible for the high antioxidant activities, antimicrobial activity, blood glucose control, and regulation of lipid abnormalities [
59,
60]. Thus, the leaves of
C. sativus from Ukraine need to be further explored.
3.6. Antibacterial Activity
The results showed that ethanolic and water extracts of
C. sativus leaves effectively suppressed the growth of the test strains of microorganisms with variable potency. As indicated in
Table 7, the ethanolic and water leaf extracts had the maximum zone of inhibition against
B. subtilis at 20.30 ± 0.36 mm and 18.00 ± 0.32 mm, respectively. Both extracts showed almost the same inhibitory effect against four pathogenic strains (
E. coli,
P. vulgaris,
P. aeruginosa,
C. albicans). Concerning
S. aureus, only ethanolic
C. sativus leaves extract showed potential activity, with an inhibition zone of 18.00 ± 0.32 mm. It has been reported previously that the methanolic extract of
C. sativus leaves from Morocco [
13] did not show antibacterial activity against
S. aureus, B. subtilis, P. aeruginosa, and
P. mirabilis; however, it had antibacterial activity against
Listeria spp. Our data confirmed that the leaf extracts of
C. sativus from Ukraine possessed significant (
p < 0.05) antibacterial activities against Gram-positive bacteria.
The data of the serial dilutions method showed that the lowest MIC values (125 µg/mL) were demonstrated by both leaf extracts against
S. aureus and
B. subtilis. In general, the ethanolic extract had lower MIC values than the water extract, due to the chemical composition of the extracts. In addition, this is consistent with the literature data that ethanolic extracts have a higher antimicrobial activity than aqueous extracts [
61,
62]. In further studies, it is necessary to select an extractant for the maximum yield of phenolic compounds, amino acids, fatty acids, and terpenes in crude extracts of
C. sativus leaves for pharmacological assessment. The established antibacterial activity of
Crocus leaves may be due to the presence of various phenolic compounds and other classes of plant compounds. Therefore, despite the well-known medicinal properties of
C. sativus (saffron) stigma, our research suggests that
Crocus leaves may also be used as a valuable plant source of naturally occurring bioactive molecules to develop new functional food and pharmaceutical ingredients, with the potential not only to promote human health but also to improve the biovalorisation of saffron.