Antihyperuricemic, Anti-Inflammatory and Antihypertensive Effect of a Dry Extract from Solidago virgaurea L. (Asteraceae)
Abstract
1. Introduction
2. Materials and Methods
2.1. Reagents and Drugs
2.2. Plant material and Extract
2.3. Phytochemical Analysis
2.4. Animals
2.5. Antihyperuricemic Effect
2.6. Anti-Inflammatory Effect
2.7. Antihypertensive Effect in Hyperuricemic Animals
2.8. Statistical Analysis
3. Results
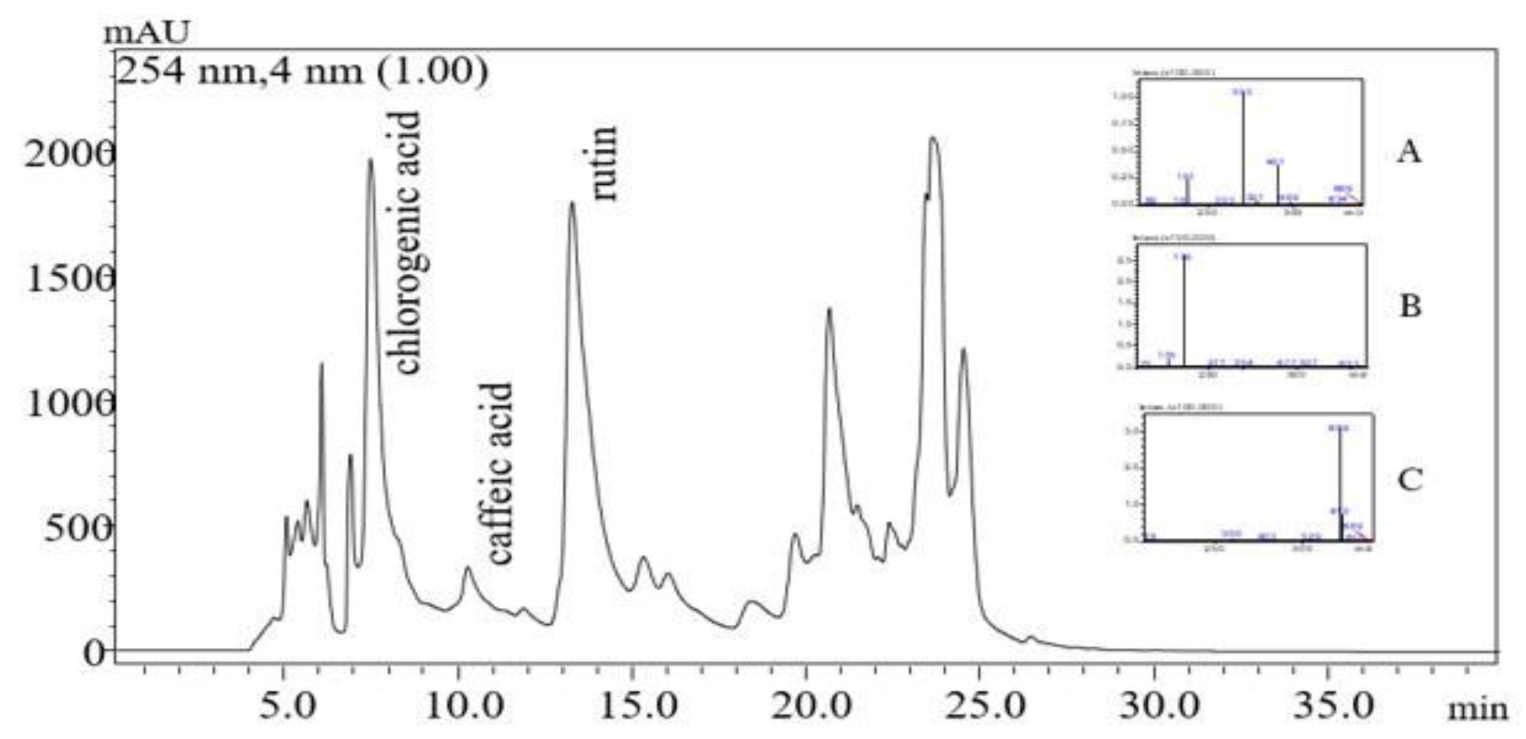
3.1. Phytochemical Analysis
3.2. Antihyperuricemic Effect
3.3. Anti-Inflammatory Effect
3.4. Antihypertensive Effect in Hyperuricemic Animals
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Roddy, E.; Mallen, C.D.; Doherty, M. Gout. BMJ 2013, 347, f5648. [Google Scholar] [CrossRef] [PubMed]
- Punzi, L.; Scanu, A.; Galozzi, P.; Luisetto, R.; Spinella, P.; Scire, C.A.; Oliviero, F. One year in review 2020: Gout. Clin. Exp. Rheumatol. 2020, 38, 807–821. [Google Scholar] [PubMed]
- Pascual, E.; Addadi, L.; Andres, M.; Sivera, F. Mechanism of crystal formation in gout. Nat. Rev. Rheumatol. 2015, 11, 725–730. [Google Scholar] [CrossRef]
- Feig, D.I. Serum uric acid and the risk of hypertension and chronic kidney disease. Curr. Opin. Rheumatol. 2014, 26, 176–185. [Google Scholar] [CrossRef] [PubMed]
- Richette, P.; Garay, R. Novel drug discovery strategies for gout. Expert Opin. Drug Discov. 2013, 8, 183–189. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.H.; Wang, C.Z.; Wang, S.Q.; Mi, C.; He, Y.; Zhang, J.; Zhang, Y.W.; Anderson, S.; Yuan, C.S. Anti-hyperuricemia effects of allopurinol are improved by Smilax riparia, a traditional Chinese herbal medicine. J. Ethnopharmacol. 2015, 162, 362–368. [Google Scholar] [CrossRef]
- Ling, X.; Bochu, W. A review of phytotherapy of gout: Perspective of new pharmacological treatments. Pharmazie 2014, 69, 243–256. [Google Scholar] [CrossRef] [PubMed]
- Corp, N.; Pendry, B. The role of Western herbal medicine in the treatment of gout. J. Herb. Med. 2013, 3, 157–170. [Google Scholar] [CrossRef]
- Havlik, J.; Gonzalez de la Huebra, R.; Hejtmanikova, K.; Fernandez, J.; Simonova, J.; Melich, M.; Rada, V. Xanthine oxidase inhibitory properties of Czech medicinal plants. J. Ethnopharmacol. 2010, 132, 461–465. [Google Scholar] [CrossRef]
- Wichtl, M.; Anton, R. Plantes Thérapeutiques, Tradition, Pratique Officinale, Science et Thérapeutique, 2nd ed.; EM Inter/Tec & Doc: Paris, France, 2003. [Google Scholar]
- European Medicines Agency (EMEA). Assessment Report on Solidago virgaurea L. Herba. 2008. Available online: https://www.ema.europa.eu/en/documents/herbal-report/assessment-report-solidago-virgaurea-l-herba_en.pdf. (accessed on 10 April 2021).
- Menkovic, N.; Savikin, K.; Tasic, S.; Zdunic, G.; Stesevic, D.; Milosavljevic, S.; Vincek, D. Ethnobotanical study on traditional uses of wild medicinal plants in Prokletije mountains (Montenegro). J. Ethnopharmacol. 2011, 133, 97–107. [Google Scholar] [CrossRef]
- Tămaș, M. Solidago species in Phytotherapy. Acta Phytother. Rom. 2000, 6, 43–44. [Google Scholar]
- El-Ghazaly, M.; Khayyal, M.T.; Okpanyi, S.N.; Arens-Corell, M. Study on the anti-inflammatory activity of Populus tremula, Solidago virgaurea and Fraxinus excelsior. Arzeim. Forsch. 1992, 42, 333–336. [Google Scholar]
- Romanian Pharmacopoeia, 10th ed.; Editura Medicala: Bucuresti, Romania, 2015.
- Plazonic, A.; Bucar, F.; Males, Z.; Mornar, A.; Nigovic, B.; Kujundzic, N. Identification and quantification of flavonoids and phenolic acids in Burr Parsley (Caucalis platycarpos L.) using high-performance liquid chromatography with diode array detection and electrospray ionization mass spectrometry. Molecules 2009, 14, 2466–2490. [Google Scholar] [CrossRef] [PubMed]
- Sugino, H.; Shimada, H. Effect of isoproterenol on renal uric acid excretion in rats. Jpn. J. Pharmacol. 1987, 45, 343–348. [Google Scholar] [CrossRef]
- Vostinaru, O.; Dinte, E.; Soran, M.L.; Lung, I.; Opris, O.; Mogosan, C. Evaluation of the anti-gout potential of an extract from Calluna vulgaris L. (Ericaceae) in rats. Rec. Nat. Prod. 2018, 12, 432–444. [Google Scholar] [CrossRef]
- Winter, C.A.; Risley, E.A.; Nuss, G.W. Carrageenan-induced oedema in the hind paw of rat as an assay for anti-inflammatory activity. Proc. Soc. Exp. Biol. Med. 1962, 111, 544–547. [Google Scholar] [CrossRef] [PubMed]
- Conea, S.; Mogosan, C.; Vostinaru, O.; Toma, C.C.; Cuc Hepcal, I.; Cazacu, I.; Pop, C.; Vlase, L. Polyphenolic profile, anti-inflammatory and antinociceptive activity of an extract from Arctium lappa L. roots. Not. Bot. Horti Agrobot. 2017, 45, 59–64. [Google Scholar] [CrossRef]
- Mazzali, M.; Hughes, J.; Kim, Y.G.; Jefferson, A.; Kang, D.H.; Gordon, K.L.; Lan, H.Y.; Kivlighn, S.; Johnson, R. Elevated uric acid increases blood pressure in the rat by a novel crystal-independent mechanism. Hypertension 2001, 38, 1101–1106. [Google Scholar] [CrossRef] [PubMed]
- Dobjanschi, L.; Paltinean, R.; Vlase, L.; Babota, M.; Fritea, L.; Tamas, M. Comparative phytochemical research of Solidago genus: S. graminifolia. Note I. Flavonoids. Acta Biol. Marisiensis 2018, 1, 18–26. [Google Scholar] [CrossRef]
- Tămaș, M.; Roșca, M. Cercetări asupra saponinelor din speciile indigene de Solidago. Farmacia 1988, 36, 167–172. [Google Scholar]
- Hiller, K.; Fötsch, G. Zur quantitativen Verteilung der Phenolglykoside Virgaureosid A und Leiocarposid in Solidago virgaurea L. Pharmazie 1986, 41, 415–416. [Google Scholar]
- Narang, R.K.; Vincent, Z.; Phipps-Green, A.; Stamp, L.K.; Merriman, T.R.; Dalbeth, N. Population-specific factors associated with fractional excretion of uric acid. Arthritis Res. Ther. 2019, 21, 234. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zhang, G.; Pan, J.; Gong, D. Novel insights into the inhibitory mechanism of kaempferol on xanthine oxidase. J. Agric. Food Chem. 2015, 63, 526–534. [Google Scholar] [CrossRef] [PubMed]
- Yan, J.; Zhang, G.; Hu, Y.; Ma, Y. Effect of luteolin on xanthine oxidase: Inhibition kinetics and interaction mechanism merging with docking simulation. Food Chem. 2013, 141, 3766–3773. [Google Scholar] [CrossRef]
- Sugino, H.; Shimada, H. The uricosuric effect in rats of E5050, a new derivative of ethanolamine involves inhibition of the tubular postsecretory reabsorption of urate. Jpn. J. Pharmacol. 1995, 68, 297–303. [Google Scholar] [CrossRef]
- Chen, Y.-S.; Hu, Q.-H.; Zhang, X.; Zhu, Q.; Kong, L.-D. Beneficial effect of rutin on oxonate-induced hyperuricemia. Pharmacology 2013, 92, 75–83. [Google Scholar] [CrossRef]
- Yoo, H.; Ku, S.-K.; Baek, Y.-D.; Bae, J.-S. Anti-inflammatory effects of rutin on HMGB1-induced inflammatory responses in vitro and in vivo. Inflamm. Res. 2014, 63, 197–206. [Google Scholar] [CrossRef]
- Serafini, M.; Peluso, I.; Raguzzini, A. Flavonoids as anti-inflammatory agents. Proc. Nutr. Soc. 2010, 69, 273–278. [Google Scholar] [CrossRef]
- Dos Santos, M.D.; Almeida, M.C.; Lopes, N.P.; De Souza, G.E. Evaluation of the anti-inflammatory, analgesic and antipyretic activities of the natural polyphenol chlorogenic acid. Biol. Pharm. Bull. 2006, 29, 2236–2240. [Google Scholar] [CrossRef]
- Khosla, U.M.; Zharikov, S.; Finch, J.L.; Nakagawa, T.; Roncal, C.; Mu, W.; Krotova, K.; Block, E.R.; Prabhakar, S.; Johnson, R.J. Hyperuricemia induces endothelial dysfunction. Kidney Int. 2005, 67, 1739–1742. [Google Scholar] [CrossRef]
- Scheepers, L.E.J.M.; Wei, F.-F.; Stolarz-Skrzypek, K.; Malyutina, S.; Tikhonoff, V.; Thijs, L.; Salvi, E. Xanthine oxidase gene variants and their association with blood pressure and incident hypertension: A populational study. J. Hypertens. 2016, 34, 2147–2154. [Google Scholar] [CrossRef]
- Agarwal, V.; Nidhi, H.; Messerli, F.H. Effect of alopurinol on blood pressure: A systematic review and meta-analysis. J. Clin. Hypertens. 2013, 15, 435–442. [Google Scholar] [CrossRef]
- Buzas, R.; Tautu, O.-F.; Dorobantu, M.; Ivan, V.; Lighezan, D. Serum uric acid and arterial hypertension-data from Sephar III survey. PLoS ONE 2018, 13, e0199865. [Google Scholar] [CrossRef]
- Perez-Vizcaino, F.; Duarte, J.; Jimenez, R.; Santos-Buelga, C.; Osuna, A. Antihypertensive effects of the flavonoid quercetin. Pharmacol. Rep. 2009, 61, 67–75. [Google Scholar] [CrossRef]
| Group (Dose) | Serum Uric Acid (mg/dL) | Urine Uric Acid (mg/dL) | Serum Creatinine (mg/dL) | Urine Creatinine (mg/dL) | Fractional Excretion of Uric Acid (%) |
|---|---|---|---|---|---|
| Oxonate control (250 mg/kg) | 1.30 ± 0.20 | 34.65 ± 3.64 | 0.72 ± 0.03 | 74.2 ± 6.42 | 25.86 |
| ESV (125 mg/kg) | 1.04 ± 0.11 | 72.80 ± 3.70 | 0.68 ± 0.07 | 107.3 ± 4.98 | 44.36 |
| ESV (250 mg/kg) | 0.86 ± 0.08 * | 101.8 ± 8.34 * | 0.53 ± 0.04 | 113.7 ± 8.01 | 55.17 * |
| ESV (500 mg/kg) | 0.60 ± 0.07 * | 125.4 ± 3.84 * | 0.48 ± 0.05 | 138.1 ± 7.12 | 72.64 * |
| Probenecid (125 mg/kg) | 0.92 ± 0.06 * | 54.67 ± 4.32 * | 0.60 ± 0.07 | 89.5 ± 8.03 | 39.83 * |
| Group | Dose | Edema 1 h (mL) | Edema 2 h (mL) | Edema 3 h (mL) | Edema 4 h (mL) |
|---|---|---|---|---|---|
| Control (vehicle) | - | 0.72 ± 0.09 | 1.03 ± 0.23 | 2.28 ± 0.41 | 3.02 ± 0.27 |
| ESV | 125 mg/kg | 0.74 ± 0.23 | 1.10 ± 0.48 | 2.04 ± 0.22 | 2.89 ± 0.36 |
| ESV | 250 mg/kg | 0.70 ± 0.14 | 1.04 ± 0.29 | 1.87 ± 0.58 | 2.74 ± 0.33 |
| ESV | 500 mg/kg | 0.66 ± 0.12 * | 0.81 ± 0.14 * | 1.48 ± 0.29 * | 1.38 ± 0.26 * |
| Diclofenac | 20 mg/kg | 0.57 ± 0.09 * | 0.73 ± 0.21 * | 1.17 ± 0.15 * | 1.30 ± 0.26 * |
| Group (Dose) | SBP Week 0 (mm Hg) | SBP Week 3 (mm Hg) |
|---|---|---|
| Oxonate control (250 mg/kg) | 111.8 ± 4.43 | 143.4 ± 4.03 |
| Oxo + ESV (125 mg/kg) | 111.4 ± 6.65 | 138.2 ± 6.18 |
| Oxo + ESV (250 mg/kg) | 114.2 ± 5.93 | 134.8 ± 4.76 * |
| Oxo + ESV (500 mg/kg) | 113.6 ± 4.27 | 124.0 ± 2.91 * |
| Oxo + Probenecid (125 mg/kg) | 113 ± 4.30 | 126.3 ± 2.13 * |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tămaş, M.; Vostinaru, O.; Soran, L.; Lung, I.; Opris, O.; Toiu, A.; Gavan, A.; Dinte, E.; Mogosan, C. Antihyperuricemic, Anti-Inflammatory and Antihypertensive Effect of a Dry Extract from Solidago virgaurea L. (Asteraceae). Sci. Pharm. 2021, 89, 27. https://doi.org/10.3390/scipharm89020027
Tămaş M, Vostinaru O, Soran L, Lung I, Opris O, Toiu A, Gavan A, Dinte E, Mogosan C. Antihyperuricemic, Anti-Inflammatory and Antihypertensive Effect of a Dry Extract from Solidago virgaurea L. (Asteraceae). Scientia Pharmaceutica. 2021; 89(2):27. https://doi.org/10.3390/scipharm89020027
Chicago/Turabian StyleTămaş, Mircea, Oliviu Vostinaru, Loredana Soran, Ildiko Lung, Ocsana Opris, Anca Toiu, Alexandru Gavan, Elena Dinte, and Cristina Mogosan. 2021. "Antihyperuricemic, Anti-Inflammatory and Antihypertensive Effect of a Dry Extract from Solidago virgaurea L. (Asteraceae)" Scientia Pharmaceutica 89, no. 2: 27. https://doi.org/10.3390/scipharm89020027
APA StyleTămaş, M., Vostinaru, O., Soran, L., Lung, I., Opris, O., Toiu, A., Gavan, A., Dinte, E., & Mogosan, C. (2021). Antihyperuricemic, Anti-Inflammatory and Antihypertensive Effect of a Dry Extract from Solidago virgaurea L. (Asteraceae). Scientia Pharmaceutica, 89(2), 27. https://doi.org/10.3390/scipharm89020027









