Michelia Essential Oil Inhalation Increases Fast Alpha Wave Activity
Abstract
1. Introduction
2. Materials and Methods
2.1. Essential Oils
2.2. Subjects
2.3. Experimental Protocols
2.4. Data Acquisition and Processing
2.5. Statistical Analyses
3. Results
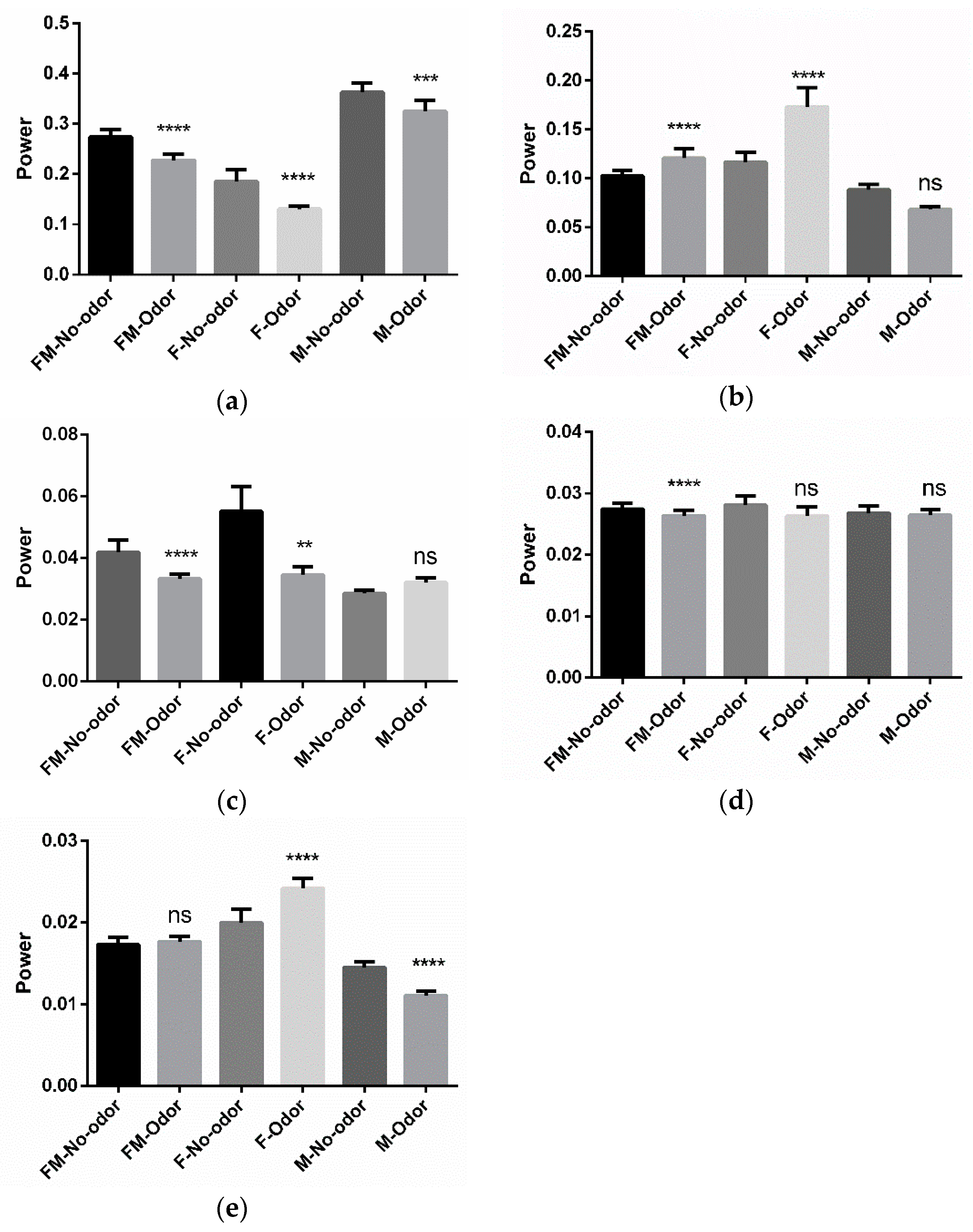
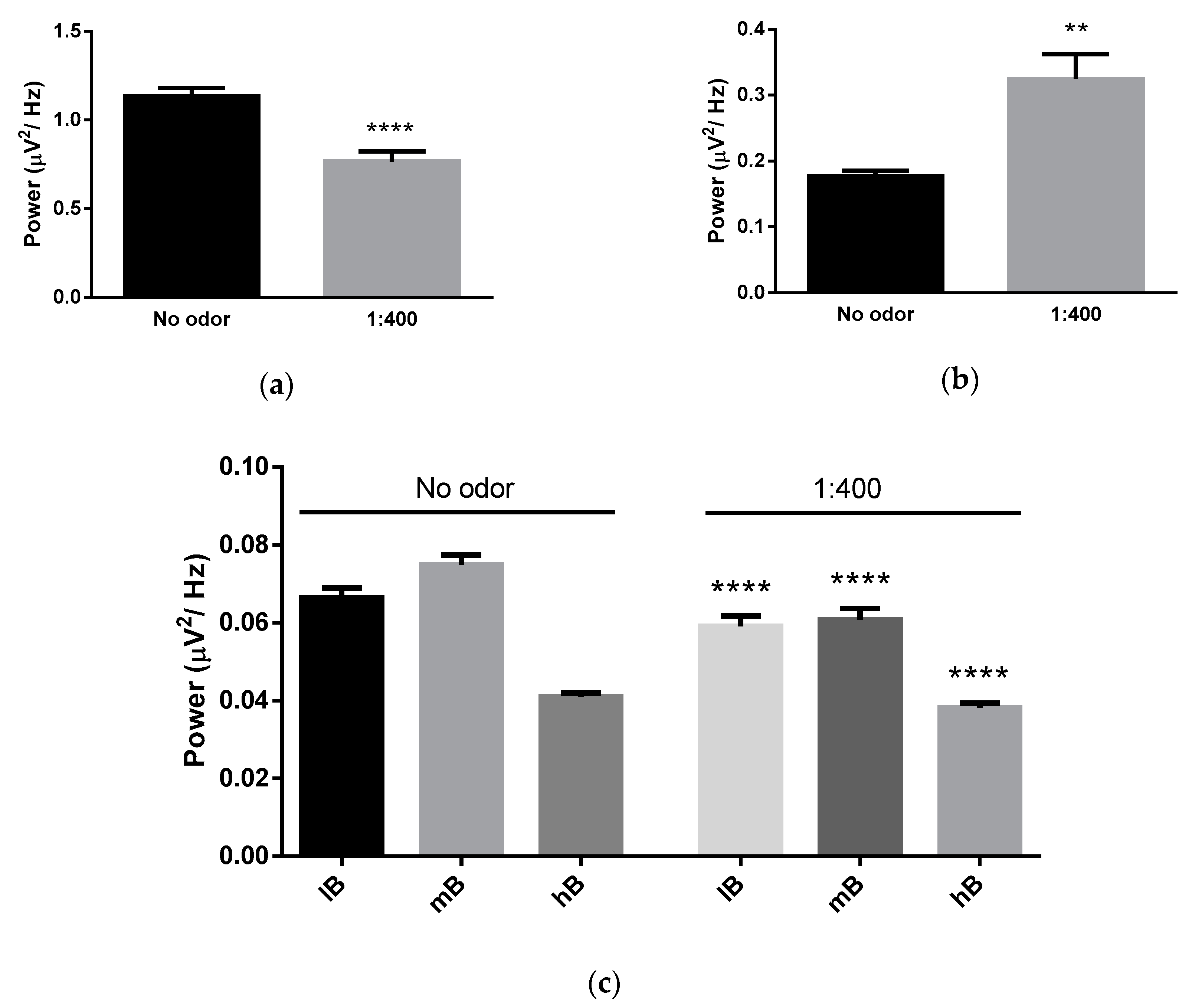
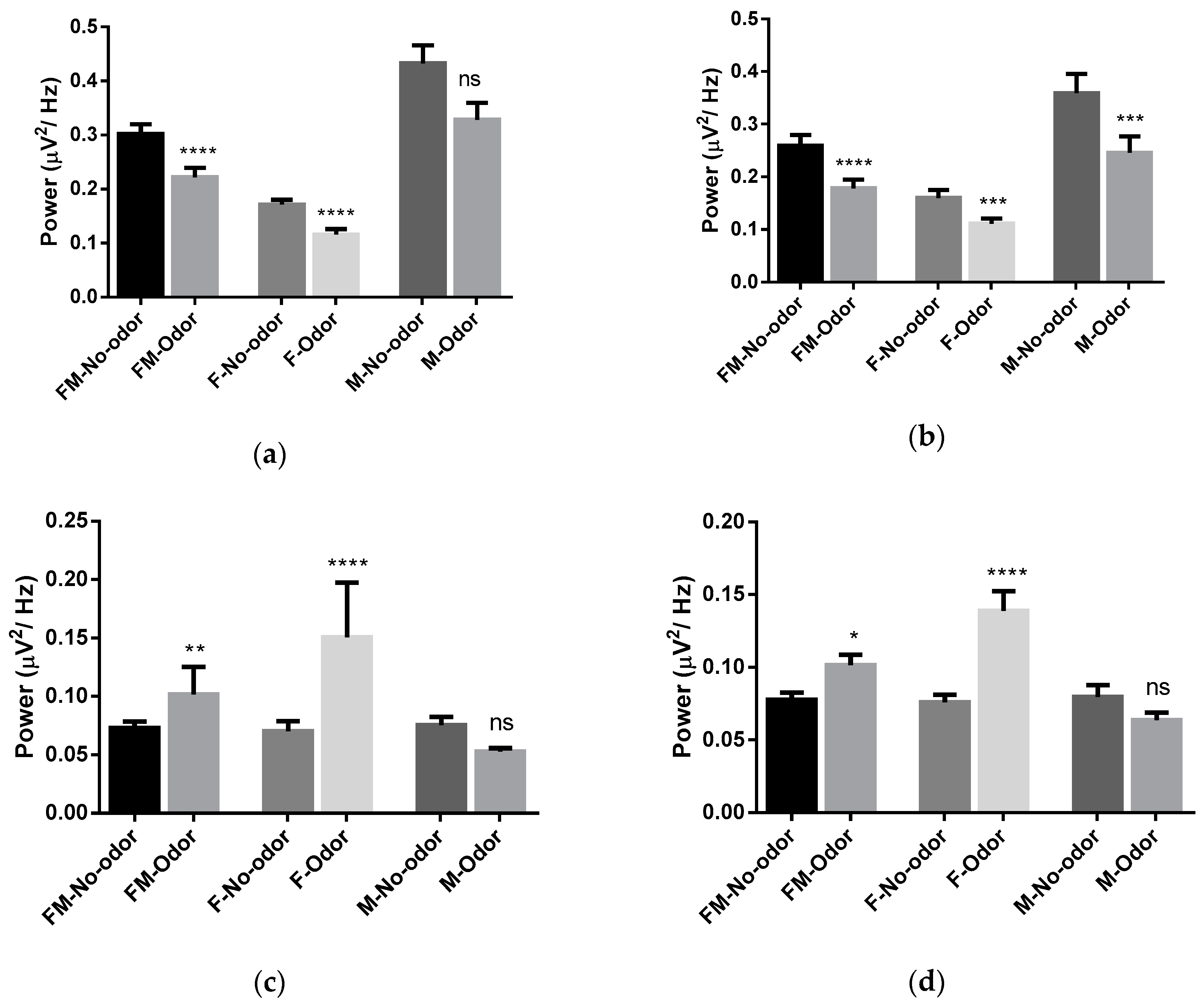
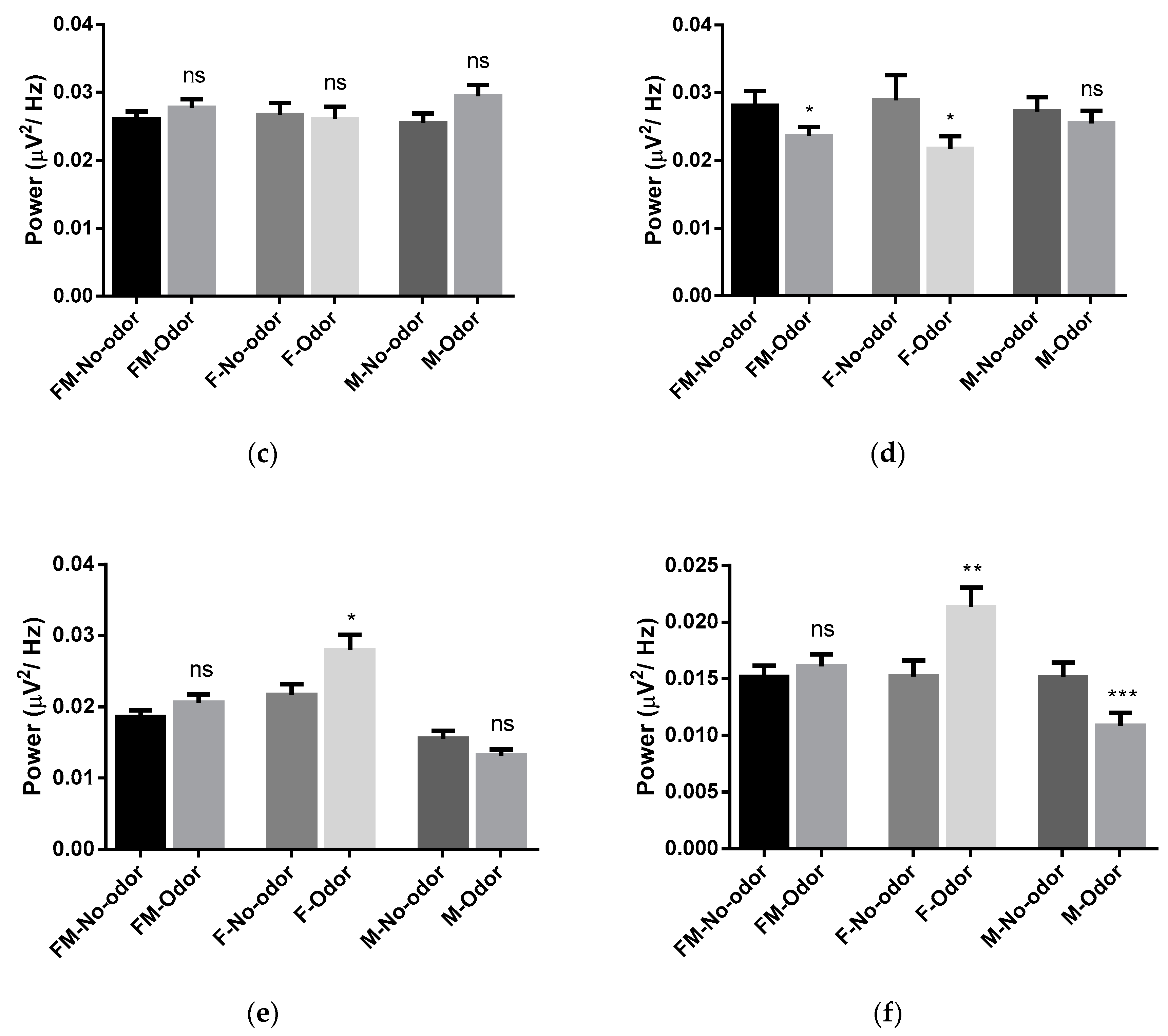
3.1. Effects of Michelia Essential Oil and Linalool Component Inhalation on Brainwaves
3.2. Brain Area Analysis
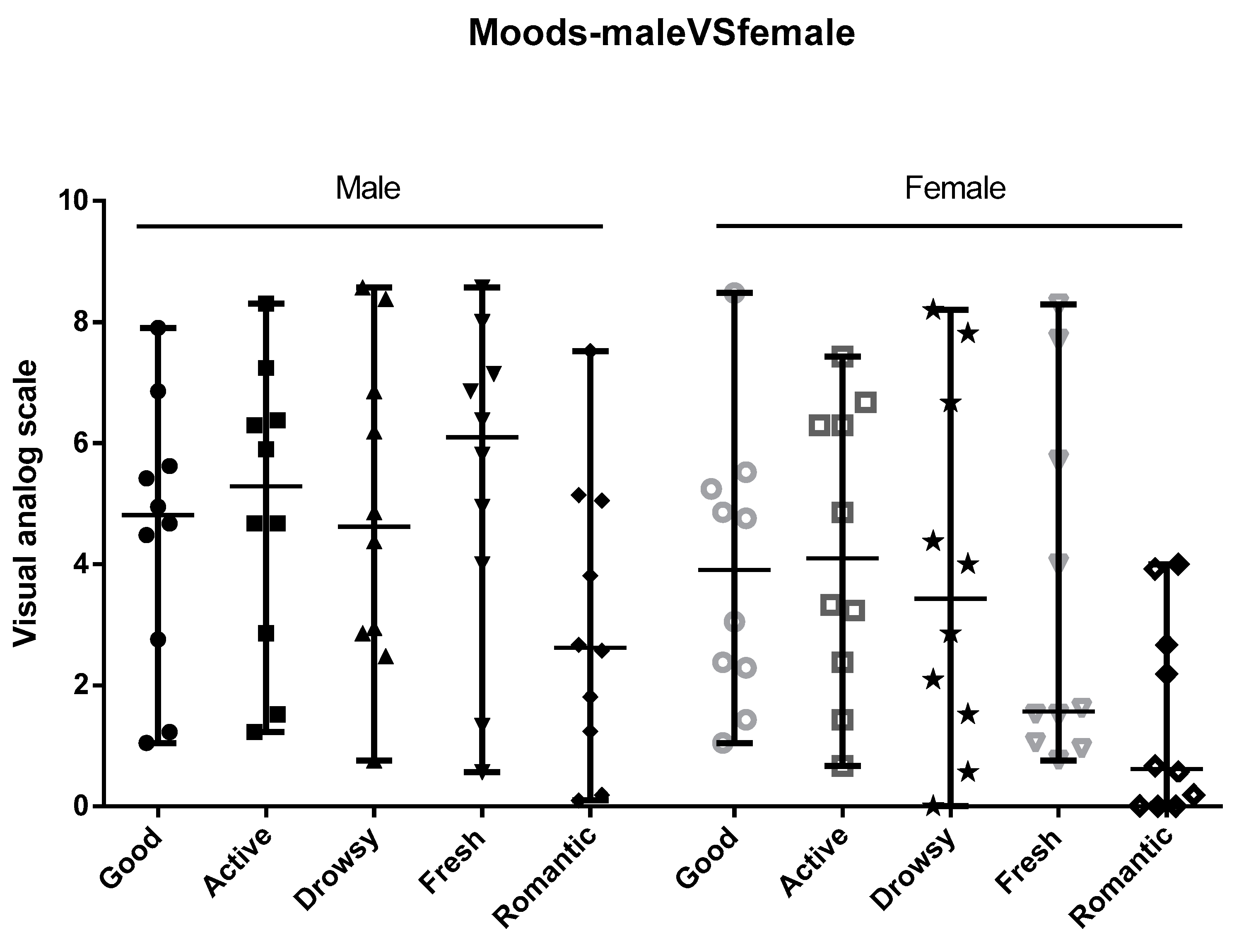
3.3. Moods and Brain-State Perception
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Esposito, E.R.; Bystrek, M.V.; Klein, J.S. An elective course in aromatherapy science. Am. J. Pharm. Educ. 2014, 78, 79. [Google Scholar] [CrossRef] [PubMed]
- Aleksic, V.; Knezevic, P. Antimicrobial and antioxidative activity of extracts and essential oils of Myrtus communis L. Microbiol. Res. 2014, 169, 240–254. [Google Scholar] [CrossRef] [PubMed]
- Amorati, R.; Foti, M.C.; Valgimigli, L. Antioxidant Activity of Essential Oils. J. Agric. Food Chem. 2013, 61, 10835–10847. [Google Scholar] [CrossRef] [PubMed]
- Gautam, N.; Mantha, A.K.; Mittal, S. Essential Oils and Their Constituents as Anticancer Agents: A Mechanistic View. BioMed Res. Int. 2014, 2014, 23. [Google Scholar] [CrossRef] [PubMed]
- Pérez, G.S.; Zavala, S.M.; Arias, G.L.; Ramos, L.M. Anti-inflammatory Activity of Some Essential Oils. J. Essent. Res. 2011, 23, 38–44. [Google Scholar] [CrossRef]
- Saad, N.Y.; Muller, C.D.; Lobstein, A. Major bioactivities and mechanism of action of essential oils and their components. Flavour Frag. J. 2013, 28, 269–279. [Google Scholar] [CrossRef]
- Sowndhararajan, K.; Kim, S. Influence of Fragrances on Human Psychophysiological Activity: With Special Reference to Human Electroencephalographic Response. Sci. Pharm. 2016, 84, 724–751. [Google Scholar] [CrossRef]
- Moss, M.; Oliver, L. Plasma 1,8-cineole correlates with cognitive performance following exposure to rosemary essential oil aroma. Ther. Adv. Psychopharmacol. 2012, 2, 103–113. [Google Scholar] [CrossRef]
- Suhem, K.; Matan, N.; Matan, N.; Danworaphong, S.; Aewsiri, T. Enhanced antifungal activity of michelia oil on the surface of bamboo paper packaging boxes using helium-neon (HeNe) laser and its application to brown rice snack bar. Food Control. 2017, 73, 939–945. [Google Scholar] [CrossRef]
- Sugawara, Y.; Hara, C.; Tamura, K.; Fujii, T.; Nakamura, K.-i.; Masujima, T.; Aoki, T. Sedative effect on humans of inhalation of essential oil of linalool:: Sensory evaluation and physiological measurements using optically active linalools. Anal. Chim. Acta 1998, 365, 293–299. [Google Scholar] [CrossRef]
- Kuroda, K.; Inoue, N.; Ito, Y.; Kubota, K.; Sugimoto, A.; Kakuda, T.; Fushiki, T. Sedative effects of the jasmine tea odor and (R)-(−)-linalool, one of its major odor components, on autonomic nerve activity and mood states. Eur. J. Appl. Physiol. 2005, 95, 107–114. [Google Scholar] [CrossRef] [PubMed]
- Masago, R.; Matsuda, T.; Kikuchi, Y.; Miyazaki, Y.; Iwanaga, K.; Harada, H.; Katsuura, T. Effects of Inhalation of Essential Oils on EEG Activity and Sensory Evaluation. J. Physiol. Anthropol. Appl. Hum. Sci. 2000, 19, 35–42. [Google Scholar] [CrossRef] [PubMed]
- Sayowan, W.; Siripornpanich, V.; Hongratanaworakit, T.; Kotchabhakdi, N.; Ruangrungsi, N. The effects of jasmine oil inhalation on brain wave activities and emotions. J. Health Res. 2013, 27, 73–77. [Google Scholar]
- Gulluni, N.; Re, T.; Loiacono, I.; Lanzo, G.; Gori, L.; Macchi, C.; Epifani, F.; Bragazzi, N.; Firenzuoli, F. Cannabis Essential Oil: A Preliminary Study for the Evaluation of the Brain Effects. Evid.-Based Compl. Altern. 2018, 2018, 11. [Google Scholar] [CrossRef]
- Ha, C.T.T.; Thai, T.H.; Diep, L.N.; Thanh, T.X.; Thu Thuy, D.T.; Tra, N.T.; Thu Ha, N.T. Chemical composition and antimicrobial activity of the essential oils from stems and leaves of Michelia alba D. C. growing in Vietnam. TAP CHI SINH HOC 2018, 2018, 40. [Google Scholar]
- Songsamoe, S.; Matan, N.; Matan, N. Antifungal activity of Michelia alba oil in the vapor phase and the synergistic effect of major essential oil components against Aspergillus flavus on brown rice. Food Control. 2017, 77, 150–157. [Google Scholar] [CrossRef]
- Leelapornpisid, P.; Chansakaow, S.; Chaiyasut, C.; Wongwattananukul, N. Antioxidant Activity of Some Volatile Oils and Absolutes from Thai Aromatic Plants; International Society for Horticultural Science (ISHS): Leuven, Belgium, 2008; pp. 61–66. [Google Scholar]
- Sowndhararajan, K.; Cho, H.; Yu, B.; Song, J.; Kim, S. Effect of inhalation of essential oil from Inula helenium L. root on electroencephalographic (EEG) activity of the human brain. Eur. J. Integr. Med. 2016, 8, 453–457. [Google Scholar] [CrossRef]
- Shipley, M.T.; Murphy, A.Z.; Rizvi, T.A.; Ennis, M.; Behbehani, M.M. Chapter 22 Olfaction and brainstem circuits of reproductive behavior in the rat. In Progress in Brain Research; Holstege, G., Bandler, R., Saper, C.B., Eds.; Elsevier: Amsterdam, The Netherlands, 1996; Volume 107, pp. 355–377. [Google Scholar]
- Klemm, W.R.; Lutes, S.D.; Hendrix, D.V.; Warrenburg, S. Topographical EEG maps of human responses to odors. Chem. Senses 1992, 17, 347–361. [Google Scholar] [CrossRef]
- Boyle, R.R.; McLean, S.; Brandon, S.; Wiggins, N. Rapid Absorption of Dietary 1,8-Cineole Results in Critical Blood Concentration of Cineole and Immediate Cessation of Eating in the Common Brushtail Possum (Trichosurus vulpecula). J. Chem. Ecol. 2005, 31, 2775–2790. [Google Scholar] [CrossRef]
- Brum, L.F.; Elisabetsky, E.; Souza, D. Effects of linalool on [(3)H]MK801 and [(3)H] muscimol binding in mouse cortical membranes. Phytother. Res. 2001, 15, 422–425. [Google Scholar] [CrossRef]
- Silva Brum, L.F.; Emanuelli, T.; Souza, D.O.; Elisabetsky, E. Effects of linalool on glutamate release and uptake in mouse cortical synaptosomes. Neurochem. Res. 2001, 26, 191–194. [Google Scholar] [CrossRef] [PubMed]
- Platt, B.; Riedel, G. The cholinergic system, EEG and sleep. Behav. Brain Res. 2011, 221, 499–504. [Google Scholar] [CrossRef] [PubMed]
- Jensen, O.; Mazaheri, A. Shaping functional architecture by oscillatory alpha activity: Gating by inhibition. Front. Hum. Neurosci. 2010, 4, 186. [Google Scholar] [CrossRef] [PubMed]

| Gender | Numbers | Age | BMI | Dominance Score |
|---|---|---|---|---|
| Male | 10 | 21.1 ± 0.18 | 22.73 ± 1.23 | 14.59 ± 2.34 |
| Female | 10 | 21.5 ± 0.17 | 21.77 ± 0.98 | 15.99 ± 3.63 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Koomhin, P.; Sattayakhom, A.; Chandharakool, S.; Sinlapasorn, J.; Suanjan, S.; Palipoch, S.; Na-ek, P.; Punsawad, C.; Matan, N. Michelia Essential Oil Inhalation Increases Fast Alpha Wave Activity. Sci. Pharm. 2020, 88, 23. https://doi.org/10.3390/scipharm88020023
Koomhin P, Sattayakhom A, Chandharakool S, Sinlapasorn J, Suanjan S, Palipoch S, Na-ek P, Punsawad C, Matan N. Michelia Essential Oil Inhalation Increases Fast Alpha Wave Activity. Scientia Pharmaceutica. 2020; 88(2):23. https://doi.org/10.3390/scipharm88020023
Chicago/Turabian StyleKoomhin, Phanit, Apsorn Sattayakhom, Supaya Chandharakool, Jennarong Sinlapasorn, Sarunnat Suanjan, Sarawoot Palipoch, Prasit Na-ek, Chuchard Punsawad, and Narumol Matan. 2020. "Michelia Essential Oil Inhalation Increases Fast Alpha Wave Activity" Scientia Pharmaceutica 88, no. 2: 23. https://doi.org/10.3390/scipharm88020023
APA StyleKoomhin, P., Sattayakhom, A., Chandharakool, S., Sinlapasorn, J., Suanjan, S., Palipoch, S., Na-ek, P., Punsawad, C., & Matan, N. (2020). Michelia Essential Oil Inhalation Increases Fast Alpha Wave Activity. Scientia Pharmaceutica, 88(2), 23. https://doi.org/10.3390/scipharm88020023




