Is the Botryococcus braunii Dry Biomass an Adjuvant for Anti-UVB Topical Formulations?
Abstract
1. Introduction
2. Materials and Methods
2.1. Growth and Production of Microalgae
2.2. Preparation of Photoprotective Formulations
2.3. In Vitro Photoprotective Activity and Photoestability
2.4. Statistical Analysis
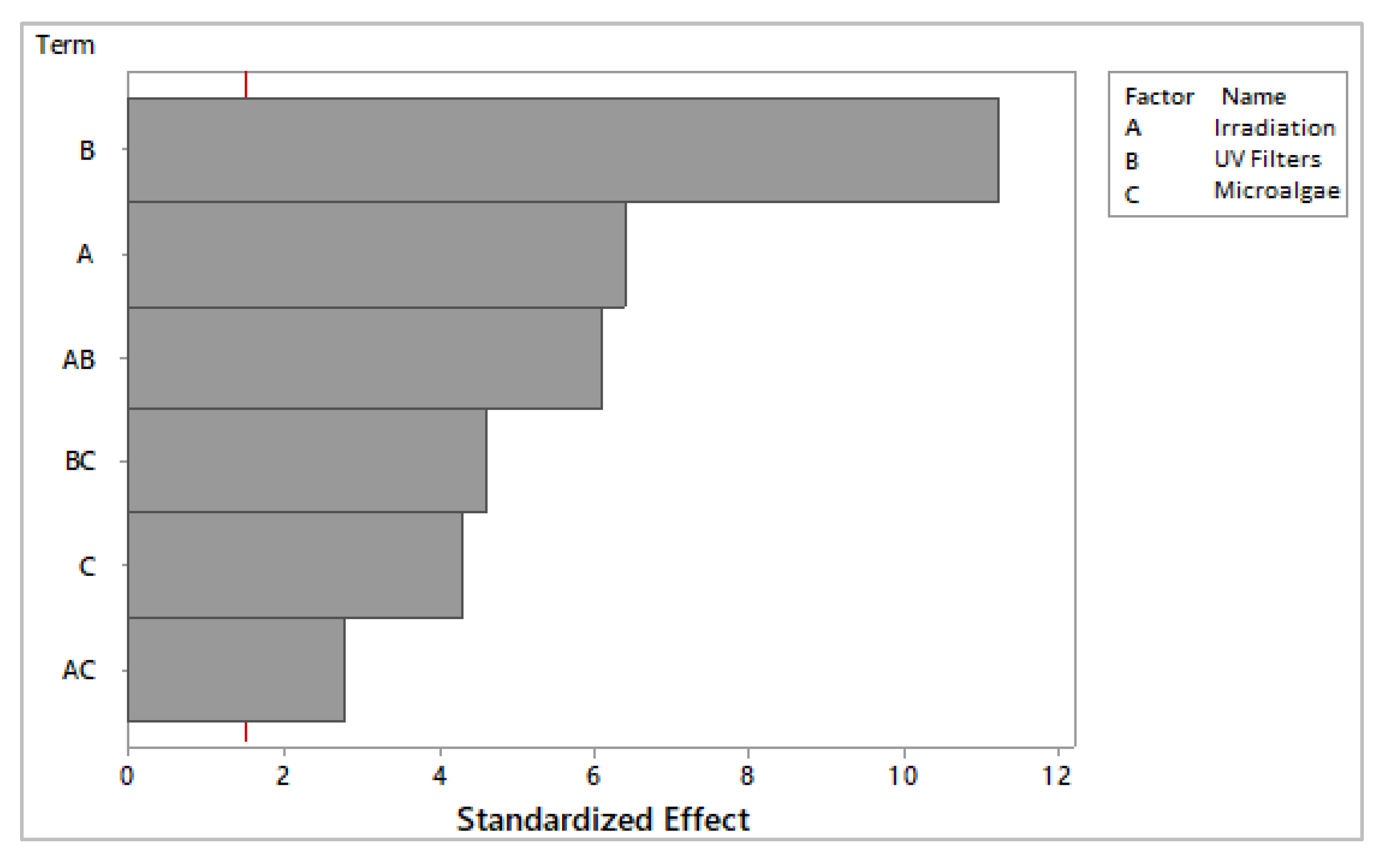
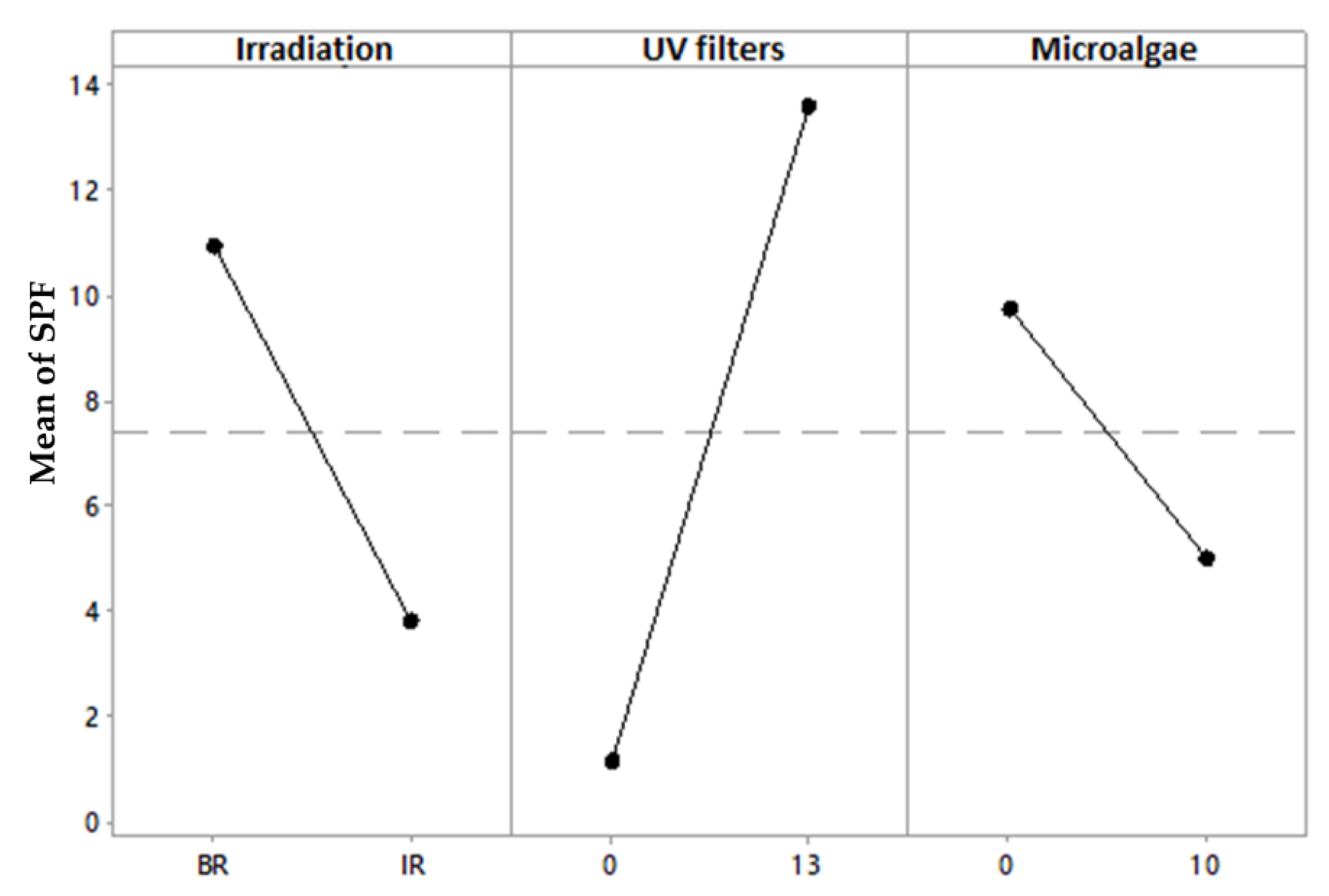
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Peres, D.A.; de Oliveira, C.A.; da Costa, M.S.; Tokunaga, V.K.; Mota, J.P.; Rosado, C.; Consiglieri, V.O.; Kaneko, T.M.; Velasco, M.V.R.; Baby, A.R. Rutin increases critical wavelength of systems containing a single UV filter and with good skin compatibility. Ski. Res. Technol. 2016, 22, 325–333. [Google Scholar] [CrossRef] [PubMed]
- Martinez, R.M.; Pinho-Ribeiro, F.A.; Steffen, V.S.; Caviglione, C.V.; Vignoli, J.A.; Barbosa, D.S.; Baracat, M.M.; Georgetti, S.R.; Verri, W.A.; Casagrande, R. Naringenin inhibits UVB irradiation-induced inflammation and oxidative stress in the skin of hairless mice. J. Nat. Prod. 2015, 78, 1647–1655. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.C.; Hwang, E.; Kim, T.; Ham, J.; Kim, S.Y.; Kwon, H.C. Nocatriones A and B, photoprotective tetracenediones from a marine-derived Nocardiopsis sp. J. Nat. Prod. 2014, 77, 2326–2330. [Google Scholar] [CrossRef]
- De Oliveira, C.A.; Peres, D.D.; Rugno, C.M.; Kojima, M.; De Oliveira Pinto, C.A.S.; Consiglieri, V.O.; Kaneko, T.M.; Rosado, C.; Mota, J.; Velasco, M.V.R.; et al. Functional photostability and cutaneous compatibility of bioactive UVA sun care products. J. Photochem. Photobiol. B Biol. 2015, 148, 154–159. [Google Scholar] [CrossRef] [PubMed]
- Hupel, M.; Poupart, N.; Gall, E.A. Development of a new in vitro method to evaluate the photoprotective sunscreen activity of plant extracts against high UV-B radiation. Talanta 2011, 86, 362–371. [Google Scholar] [CrossRef] [PubMed]
- Suh, S.; Hwang, J.; Park, M.; Seo, H.H.; Kim, H.; Lee, J.H.; Moh, S.H.; Lee, T. Anti-inflammation activities of Mycosporine-like amino acids (MAAs) in response to UV radiation suggest potential anti-skin aging activity. Mar. Drugs 2014, 5174–5187. [Google Scholar] [CrossRef] [PubMed]
- Martins, F.J.; Caneschi, C.A.; Vieira, J.L.F.; Barbosa, W.; Raposo, N.R.B. Antioxidant activity and potential photoprotective from amazon native flora extracts. J. Photochem. Photobiol. B Biol. 2016, 161, 34–39. [Google Scholar] [CrossRef]
- Saewan, N.; Jimtaisong, A. Natural products as photoprotection. J. Cosmet. Dermatol. 2015, 14, 47–63. [Google Scholar] [CrossRef]
- Lotan, A. Biologic Sunscreen Composition. WO2012093388A2, 12 July 2012. [Google Scholar]
- Kawakami, C.M.; Gaspar, L.R. Mangiferin and naringenin affect the photostability and phototoxicity of sunscreens containing avobenzone. J. Photochem. Photobiol. B Biol. 2015, 151, 239–247. [Google Scholar] [CrossRef]
- Afonso, S.; Horita, K.; Sousa, E.; Silva, J.P.; Almeida, I.F.; Amaral, M.H.; Lobão, P.A.; Costa, P.C.; Miranda, M.S.; Esteves Da Silva, J.C.G.; et al. Photodegradation of avobenzone: Stabilization effect of antioxidants. J. Photochem. Photobiol. B Biol. 2014, 140, 36–40. [Google Scholar] [CrossRef]
- Elliott, A.; Mundy, C.J.; Gosselin, M.; Poulin, M.; Campbell, K.; Wang, F. Spring production of mycosporine-like amino acids and other UV-absorbing compounds in sea ice-associated algae communities in the Canadian Arctic. Mar. Ecol. Prog. Ser. 2015, 541, 91–104. [Google Scholar] [CrossRef]
- Buono, S.; Langellotti, A.L.; Martello, A.; Bimonte, M.; Tito, A.; Carola, A.; Apone, F.; Colucci, G.; Fogliano, V. Biological activities of dermatological interest by the water extract of the microalga Botryococcus braunii. Arch. Dermatol. Res. 2012, 304, 755–764. [Google Scholar] [CrossRef]
- Wang, H.M.D.; Chen, C.C.; Huynh, P.; Chang, J.S. Exploring the potential of using algae in cosmetics. Bioresour. Technol. 2015, 184, 355–362. [Google Scholar] [CrossRef] [PubMed]
- Maadane, A.; Merghoub, N.; Ainane, T.; El Arroussi, H.; Benhima, R.; Amzazi, S.; Bakri, Y.; Wahby, I. Antioxidant activity of some Moroccan marine microalgae: Pufa profiles, carotenoids and phenolic content. J. Biotechnol. 2015, 215, 13–19. [Google Scholar] [CrossRef]
- Batista, A.P.; Gouveia, L.; Bandarra, N.M.; Franco, J.M.; Raymundo, A. Comparison of microalgal biomass profiles as novel functional ingredient for food products. Algal Res. 2013, 2, 164–173. [Google Scholar] [CrossRef]
- Hagino, H.; Saito, M. Use of Algal Proteins in Cosmetics. EP03029218A, 22 September 2010. [Google Scholar]
- Furuhashi, K.; Noguchi, T.; Okada, S.; Hasegawa, F.; Kaizu, Y.; Imou, K. The surface structure of Botryococcus braunii colony prevents the entry of extraction solvents into the colony interior. Algal Res. 2016, 16, 160–166. [Google Scholar] [CrossRef]
- Pérez-Mora, L.S.; Matsudo, M.C.; Cezare-Gomes, E.A.; Carvalho, J.C. An investigation into producing Botryococcus braunii in a tubular photobioreactor. J. Chem. Technol. Biotechnol. 2016, 91, 3053–3060. [Google Scholar] [CrossRef]
- Furuhashi, K.; Hasegawa, F.; Saga, K.; Kudou, S.; Okada, S.; Kaizu, Y.; Imou, K. Effects of culture medium salinity on the hydrocarbon extractability, growth and morphology of Botryococcus braunii. Biomass Bioenergy 2016, 91, 83–90. [Google Scholar] [CrossRef]
- UTEX Culture Collection of Algae UTEX. Available online: https://utex.org/products/chus-medium?variant=30991781953626 (accessed on 16 April 2018).
- Eroglu, E.; Okada, S.; Melis, A. Hydrocarbon productivities in different Botryococcus strains: Comparative methods in product quantification. J. Appl. Phycol. 2011, 23, 763–775. [Google Scholar] [CrossRef]
- Ariede, M.B. Aplicação do extrato da microalga Botryococcus braunii em formulações cosméticas fotoprotetoras. Master’s Thesis, University of São Paulo, São Paulo, Brazil, 2016. [Google Scholar]
- Europe Cosmetics. In Vitro Method for the Determination of the UVA Protection Factor and “Critical Wavelength” Values of Sunscreen Products; COLIPA: Auderghem, Belgium, 2011. [Google Scholar]
- United States Food and Drug Administration, Department of Health and Human Services. 21 CFR Parts 201 and 310. Labeling and effectiveness testing; sunscreen drug products for over-the-counter human use. In Fed. Regist.; 2011; 76, pp. 35620–35665. [Google Scholar]
- Scalia, S.; Mezzena, M. Photostabilization effect of quercetin on the UV filter combination, butyl methoxydibenzoylmethane-octyl methoxycinnamate. Photochem. Photobiol. 2010, 86, 273–278. [Google Scholar] [CrossRef] [PubMed]
- Peres, L.P.A.; Dias, I.L.T. Desenvolvimento e avaliação de formulações em gel para veiculação de ácido salicílico. Rev. Multidiscip. Saúde 2011, 3, 26–36. [Google Scholar]
- Adriano, J.; Netz, D.J.A.; Bresolin, T.M.B.; Freitas, R.A.; Couto, A.G. Development of a topical gel containing dried extract of Ipomoea pes-caprae brasiliensis (L.) R. Br. (Convolvulaceae). J. Med. Plants Res. 2014, 8, 1074–1080. [Google Scholar] [CrossRef]
- Zanini, M. Gel de ácido tricloroacético—Uma nova técnica para um antigo ácido. Med. Cutan. Ibero. Lat. Am. 2007, 35, 14–17. [Google Scholar]
- Daniels, R. Emulsifier-Free, Polymer-Stabilized Foam Formulations. EP2509565A1, 17 October 2012. [Google Scholar]
- Pinto, C.A.S.D.O. Influência da rutina na fotoestabilização da avobenzona (filtro UVA) e do p-Metoxicinamato de octila (Filtro UVB). Ph.D. Thesis, University of São Paulo, São Paulo, Brazil, 2014. [Google Scholar]
- Sohn, M.; Herzog, B.; Osterwalder, U.; Imanidis, G. Calculation of the sun protection factor of sunscreens with different vehicles using measured film thickness distribution—Comparison with the SPF in vitro. J. Photochem. Photobiol. B 2016, 159, 74–81. [Google Scholar] [CrossRef] [PubMed]
- Balogh, T.S.; Velasco, M.V.R.; Pedriali, C.A.; Kaneko, T.M.; Baby, A.R. Ultraviolet radiation protection: Current available resources in photoprotection. An. Bras. Dermatol. 2011, 86, 732–742. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.J.; Kim, M.J.; Im, N.R.; Park, S.N. Photolysis of the organic UV filter, avobenzone, combined with octyl methoxycinnamate by nano-TiO2 composites. J. Photochem. Photobiol. B Biol. 2015, 149, 196–203. [Google Scholar] [CrossRef]
- Kumar, V.; Jahan, F.; Kameswaran, K.; Mahajan, R.V.; Saxena, R.K. Eco-friendly methodology for efficient synthesis and scale-up of 2-ethylhexyl-p-methoxycinnamate using Rhizopus oryzae lipase and its biological evaluation. J. Ind. Microbiol. Biotechnol. 2014, 41, 907–912. [Google Scholar] [CrossRef]
- ANVISA. Resolução-RDC N° 30, de 1° de junho de 2012; Agência Nacional de Vigilância Sanitária (ANVISA): Brasília, Brazil, 2012; Volume 2012, pp. 760–763. [Google Scholar]
- Peres, D.D.; Ariede, M.B.; Candido, T.M.; Almeida, T.S.; Lourenço, F.R.; Consiglieri, V.O.; Kaneko, T.M.; Velasco, M.V.R.; Baby, A.R. Quality by design (QbD), process analytical technology (PAT), and design of experiment applied to the development of multifunctional sunscreens. Drug Dev. Ind. Pharm. 2017, 2, 246–256. [Google Scholar] [CrossRef]
- Peres, D.A.; Hubner, A.; De Oliveira, C.A.; De Almeida, T.S.; Kaneko, T.M.; Consiglieri, V.O.; Pinto, C.A.S.D.O.; Velasco, M.V.R.; Baby, A.R. Hydrolyzed collagen interferes with in vitro photoprotective effectiveness of sunscreens. Braz. J. Pharm. Sci. 2017, 53, 16119. [Google Scholar] [CrossRef]
- Stiefel, C.; Schwack, W.; Nguyen, Y.T.H. Photostability of cosmetic UV filters on mammalian skin under UV exposure. Photochem. Photobiol. 2015, 91, 84–91. [Google Scholar] [CrossRef] [PubMed]
- Sambandan, D.R.; Ratner, D. Sunscreens: An overview and update. J. Am. Acad. Dermatol. 2011, 64, 748–758. [Google Scholar] [CrossRef] [PubMed]
- Stiefel, C.; Schwack, W. Reactivity of cosmetic UV filters towards skin proteins: Model studies with Boc-lysine, Boc-Gly-Phe-Gly-Lys-OH, BSA and gelatin. Int. J. Cosmet. Sci. 2014, 36, 561–570. [Google Scholar] [CrossRef] [PubMed]
| Ingredients (Vendor/Supplier/Local) | Concentration (% w/w) | |||||
|---|---|---|---|---|---|---|
| Formulation Codes | ||||||
| F2 | F3 | F4 | F6 | F7 | F8 | |
| Aristoflex® AVC (Pharmaspecial, Brazil) | 1.0 | 1.0 | 1.0 | 0.5 | 0.5 | 0.5 |
| Crodafos® CES (Mapric, Brazil) | - | - | - | 6.0 | 6.0 | 6.0 |
| Propylene glycol (Mapric, Brazil) | - | - | - | 5.0 | 5.0 | 5.0 |
| Avobenzone (Fragon, Brazil) | 3.0 | - | 3.0 | 3.0 | - | 3.0 |
| Octyl p-methoxycinnamate (Fragon, Brazil) | 10.0 | - | 10.0 | 10.0 | - | 10.0 |
| B. braunii dry biomass | - | 10.0 | 10.0 | - | 10.0 | 10.0 |
| Caprylic/Capric Triglyceride (Mapric, Brazil) | 3.5 | - | 3.5 | 3.5 | - | 3.5 |
| Phenoxyethanol (and) methylparaben (and) ethylparaben (and) butylparaben (and) propylparaben (and) isobutylparaben (Mapric, Brazil) | 0.8 | |||||
| Purified water | * | |||||
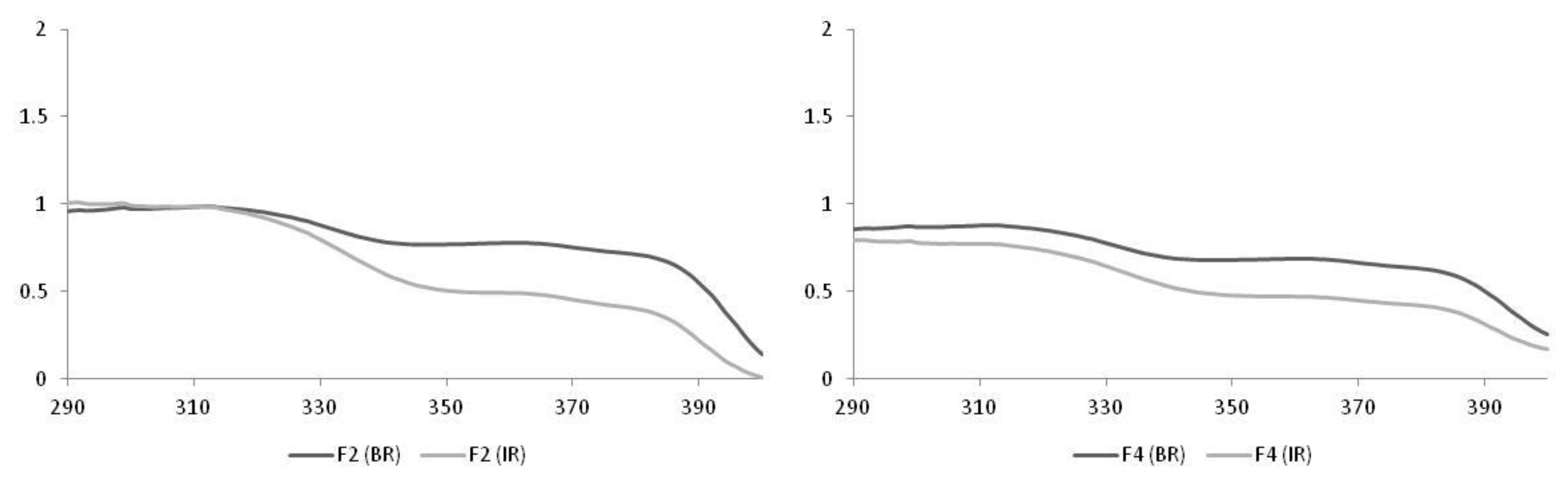
| Formulations | Irradiation | SPF |
|---|---|---|
| F2 | BR | 10.0 ± 2.1 |
| IR | 8.0 ± 4.5 | |
| F4 | BR | 7.0 ± 1.0 |
| IR | 5.6 ± 1.2 |
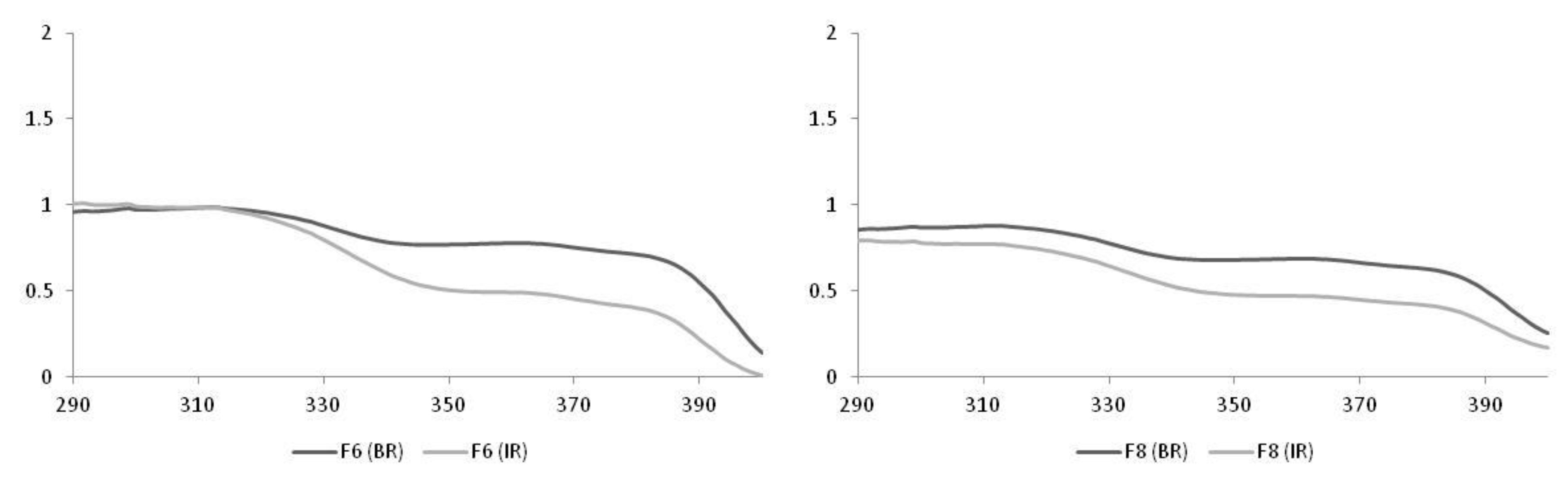
| Formulations | Irradiation | SPF |
|---|---|---|
| F6 | BR | 29.0 ± 4.7 |
| IR | 8.0 ± 1.1 | |
| F8 | BR | 12.0 ± 1.5 |
| IR | 5.0 ± 1.0 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ariede, M.B.; Morocho-Jácome, A.L.; Candido, T.M.; Lourenço, F.R.; Kato, E.T.M.; Lima, F.V.; Rosado, C.; Velasco, M.V.R.; Carvalho, J.C.M.d.; Baby, A.R. Is the Botryococcus braunii Dry Biomass an Adjuvant for Anti-UVB Topical Formulations? Sci. Pharm. 2020, 88, 22. https://doi.org/10.3390/scipharm88020022
Ariede MB, Morocho-Jácome AL, Candido TM, Lourenço FR, Kato ETM, Lima FV, Rosado C, Velasco MVR, Carvalho JCMd, Baby AR. Is the Botryococcus braunii Dry Biomass an Adjuvant for Anti-UVB Topical Formulations? Scientia Pharmaceutica. 2020; 88(2):22. https://doi.org/10.3390/scipharm88020022
Chicago/Turabian StyleAriede, Maíra Bueno, Ana Lucia Morocho-Jácome, Thalita Marcílio Candido, Felipe Rebello Lourenço, Edna Tomiko Myiake Kato, Fabiana Vieira Lima, Catarina Rosado, Maria Valéria Robles Velasco, João Carlos Monteiro de Carvalho, and André Rolim Baby. 2020. "Is the Botryococcus braunii Dry Biomass an Adjuvant for Anti-UVB Topical Formulations?" Scientia Pharmaceutica 88, no. 2: 22. https://doi.org/10.3390/scipharm88020022
APA StyleAriede, M. B., Morocho-Jácome, A. L., Candido, T. M., Lourenço, F. R., Kato, E. T. M., Lima, F. V., Rosado, C., Velasco, M. V. R., Carvalho, J. C. M. d., & Baby, A. R. (2020). Is the Botryococcus braunii Dry Biomass an Adjuvant for Anti-UVB Topical Formulations? Scientia Pharmaceutica, 88(2), 22. https://doi.org/10.3390/scipharm88020022







