Synthesis and Regularities of the Structure–Activity Relationship in a Series of N-Pyridyl-4-methyl-2,2-dioxo-1H-2λ6,1-benzothiazine-3-carboxamides
Abstract
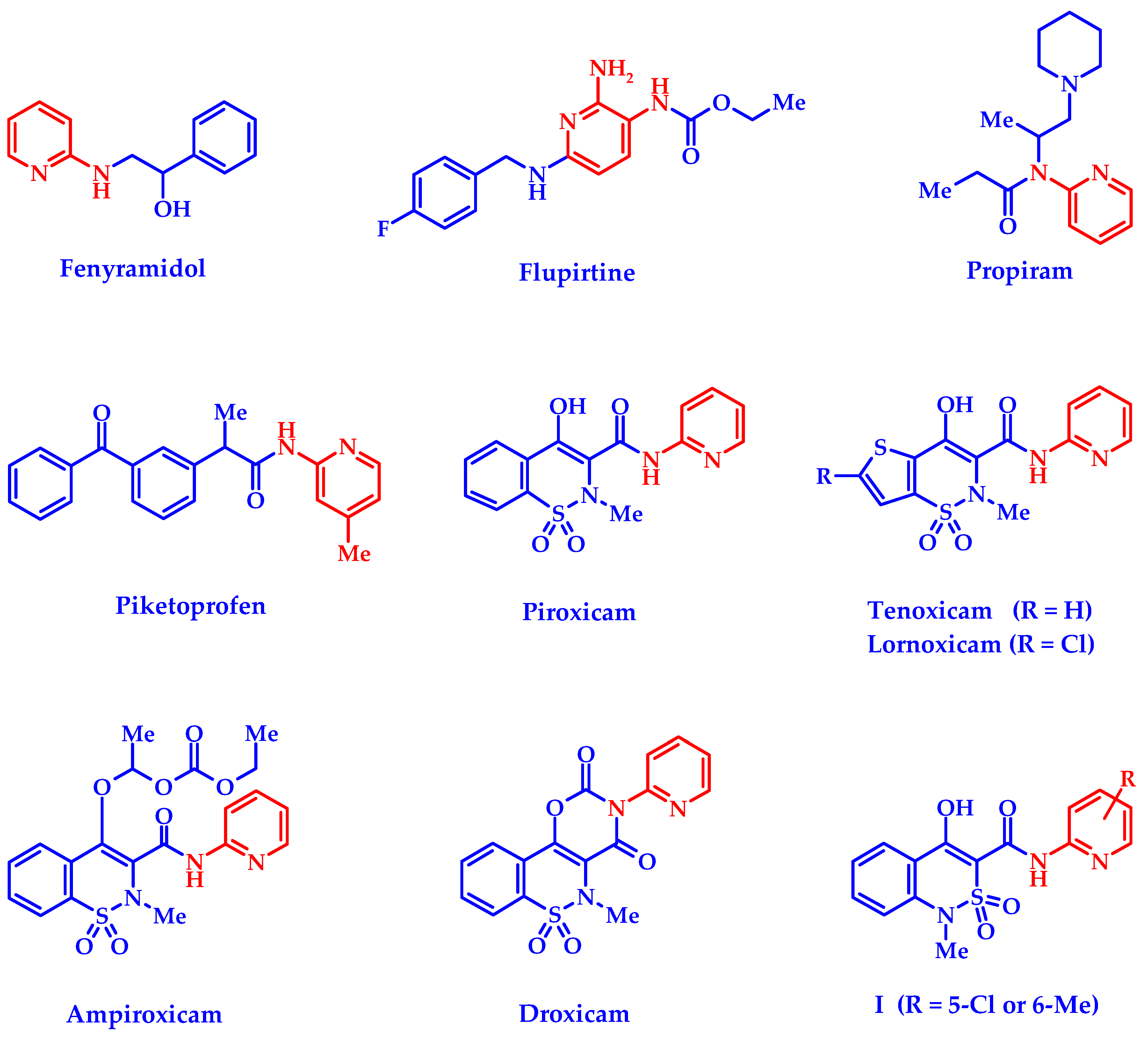
:1. Introduction
2. Materials and Methods
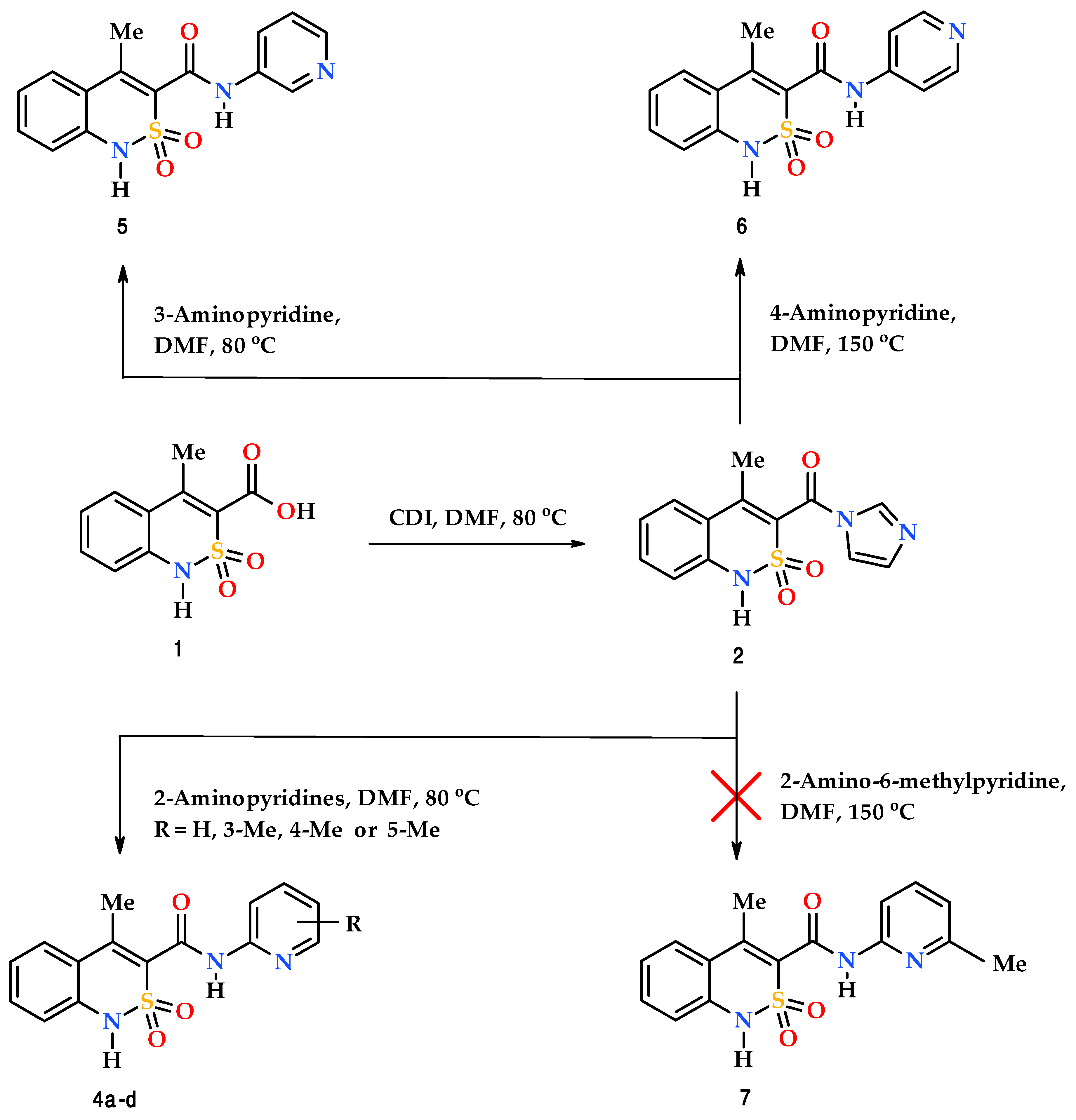
2.1. Chemistry
2.2. 4-Methyl-2,2-dioxo-1H-2λ6,1-benzothiazine-3-carboxylic acid imidazolide (2)
2.3. General Procedure for the Synthesis of N-pyridyl-4-methyl-2,2-dioxo-1H-2λ6,1-benzothiazine-3-carboxamides (4a–d and 5)
2.4. N-(Pyridin-3-yl)-4-methyl-2,2-dioxo-1H-2λ6,1-benzothiazine-3-carboxamide N,N-dimethylformamide monosolvate (5a)
2.5. N-(Pyridin-4-yl)-4-methyl-2,2-dioxo-1H-2λ6,1-benzothiazine-3-carboxamide (6)
2.6. X-ray Structural Analysis of 4-Methyl-2,2-dioxo-1H-2λ6,1-benzothiazine-3-carboxylic acid imidazolide (2)
2.7. X-ray Structural Analysis of N-(Pyridin-2-yl)-4-methyl-2,2-dioxo-1H-2λ6,1-benzothiazine-3-carboxamide (4a)
2.8. X-ray Structural Analysis of N-(3-Methylpyridin-2-yl)-4-methyl-2,2-dioxo-1H-2λ6,1-benzothiazine-3-carboxamide (4b)
2.9. X-ray Structural Analysis of N-(4-Methylpyridin-2-yl)-4-methyl-2,2-dioxo-1H-2λ6,1-benzothiazine-3-carboxamide (4c)
2.10. X-ray Structural Analysis of N-(5-Methylpyridin-2-yl)-4-methyl-2,2-dioxo-1H-2λ6,1-benzothiazine-3-carboxamide (4d)
2.11. X-ray Structural Analysis of N-(Pyridin-3-yl)-4-methyl-2,2-dioxo-1H-2λ6,1-benzothiazine-3-carboxamide-N,N-dimethylformamide monosolvate (5a)
2.12. X-ray Structural Analysis of N-(Pyridin-4-yl)-4-methyl-2,2-dioxo-1H-2λ6,1-benzothiazine-3-carboxamide (6)
2.13. Pharmacology
Analgesic and Anti-Inflammatory Tests
3. Results and Discussion
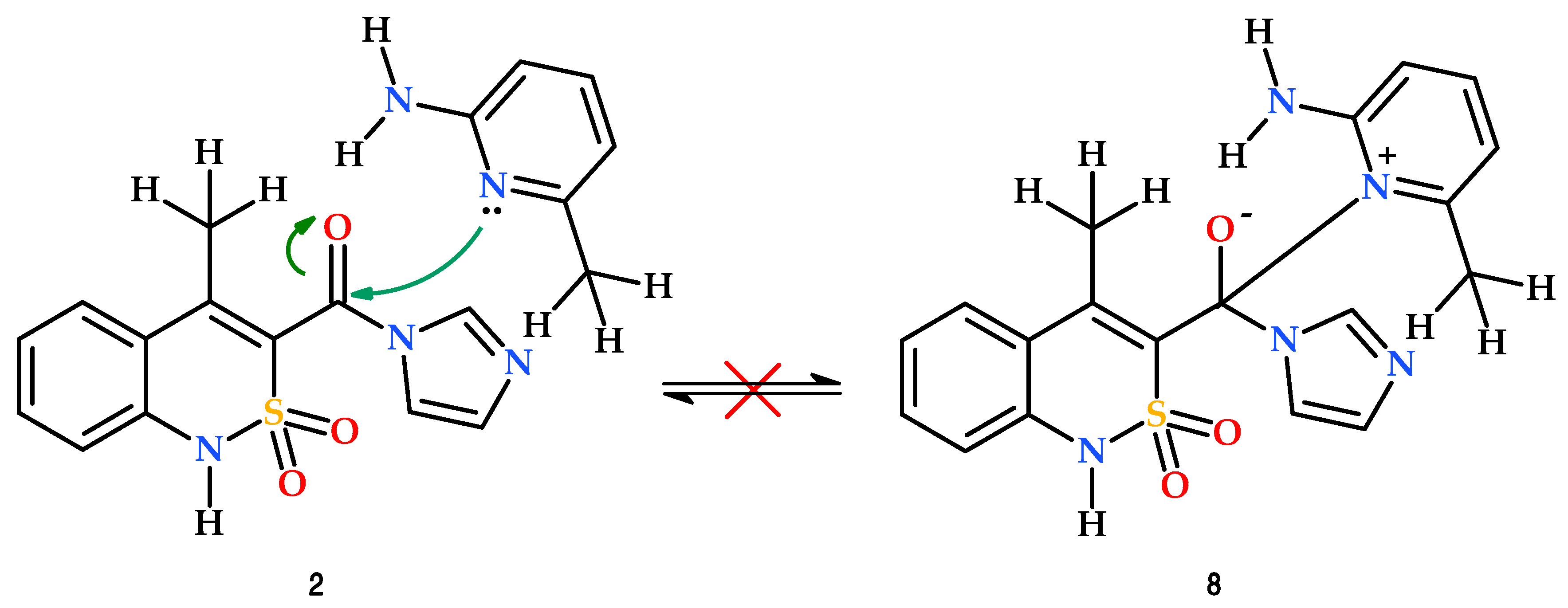
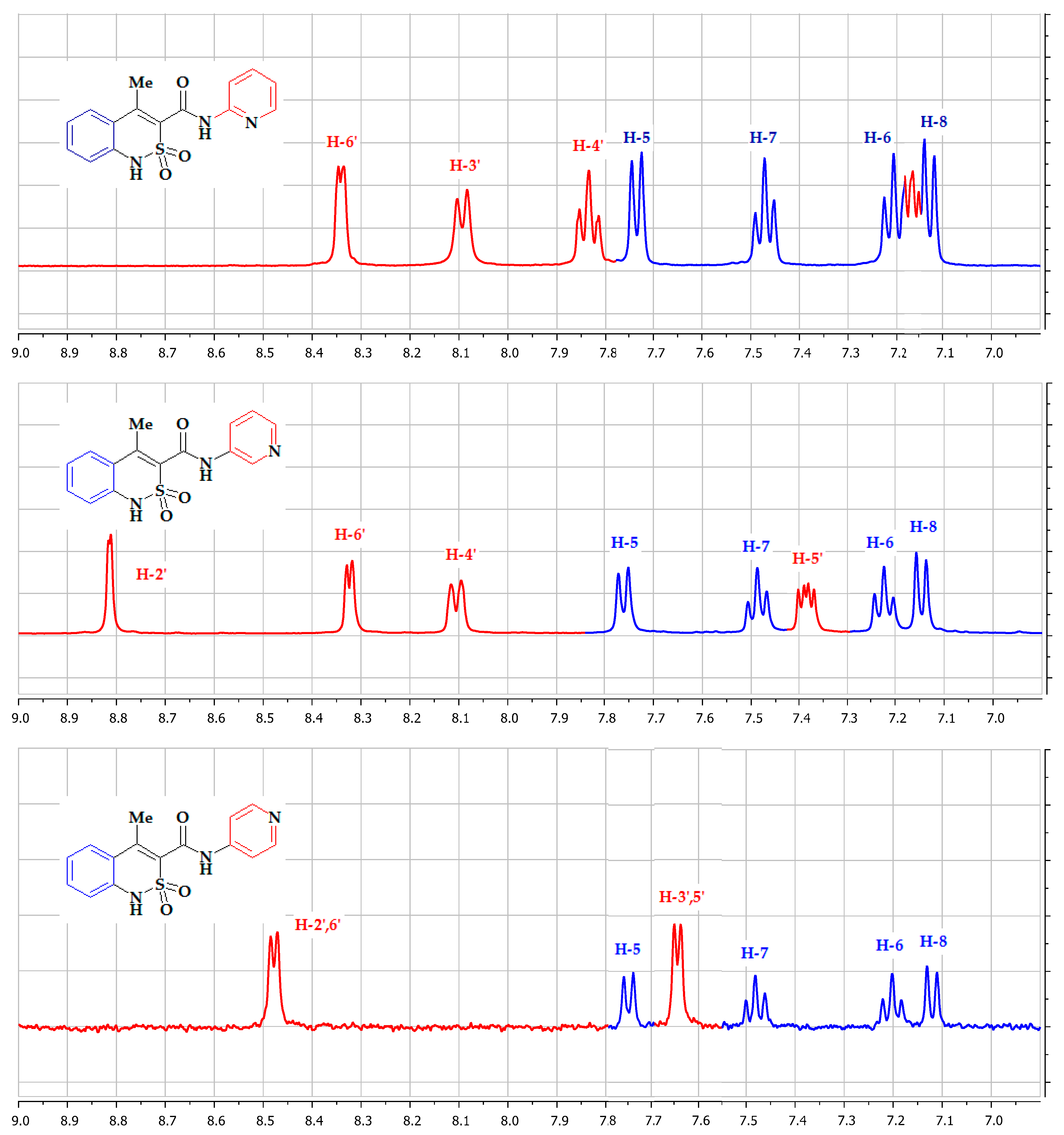
3.1. Chemistry
3.2. Evaluation of the Analgesic and Anti-Inflammatory Activity
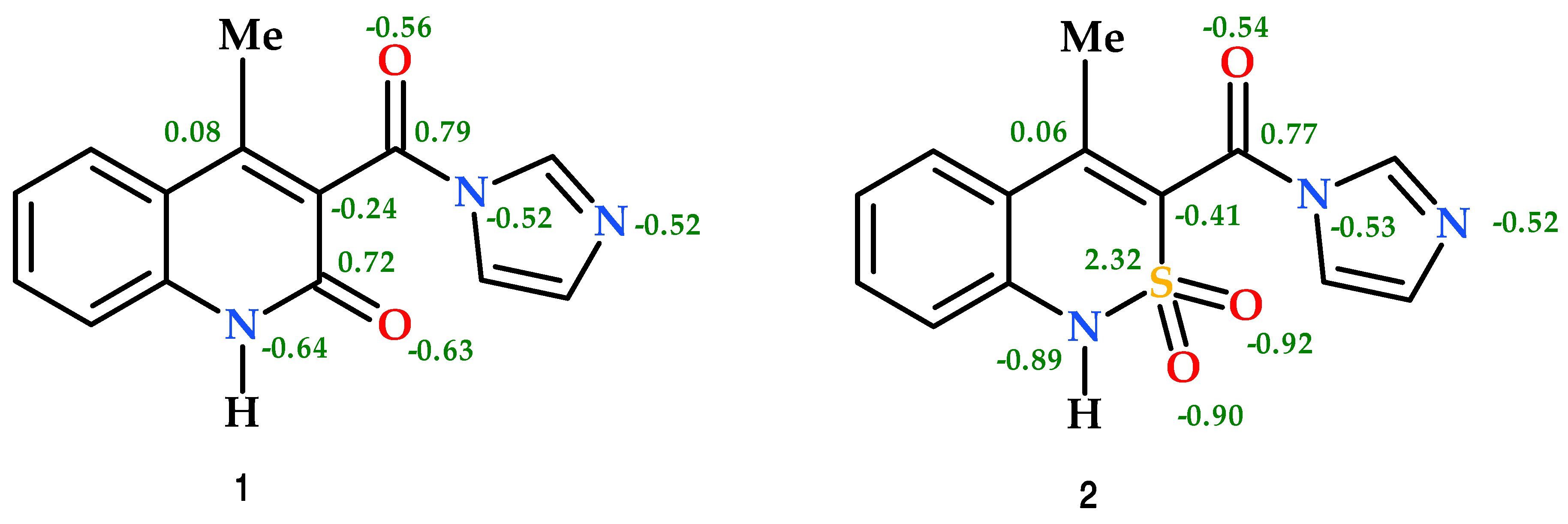
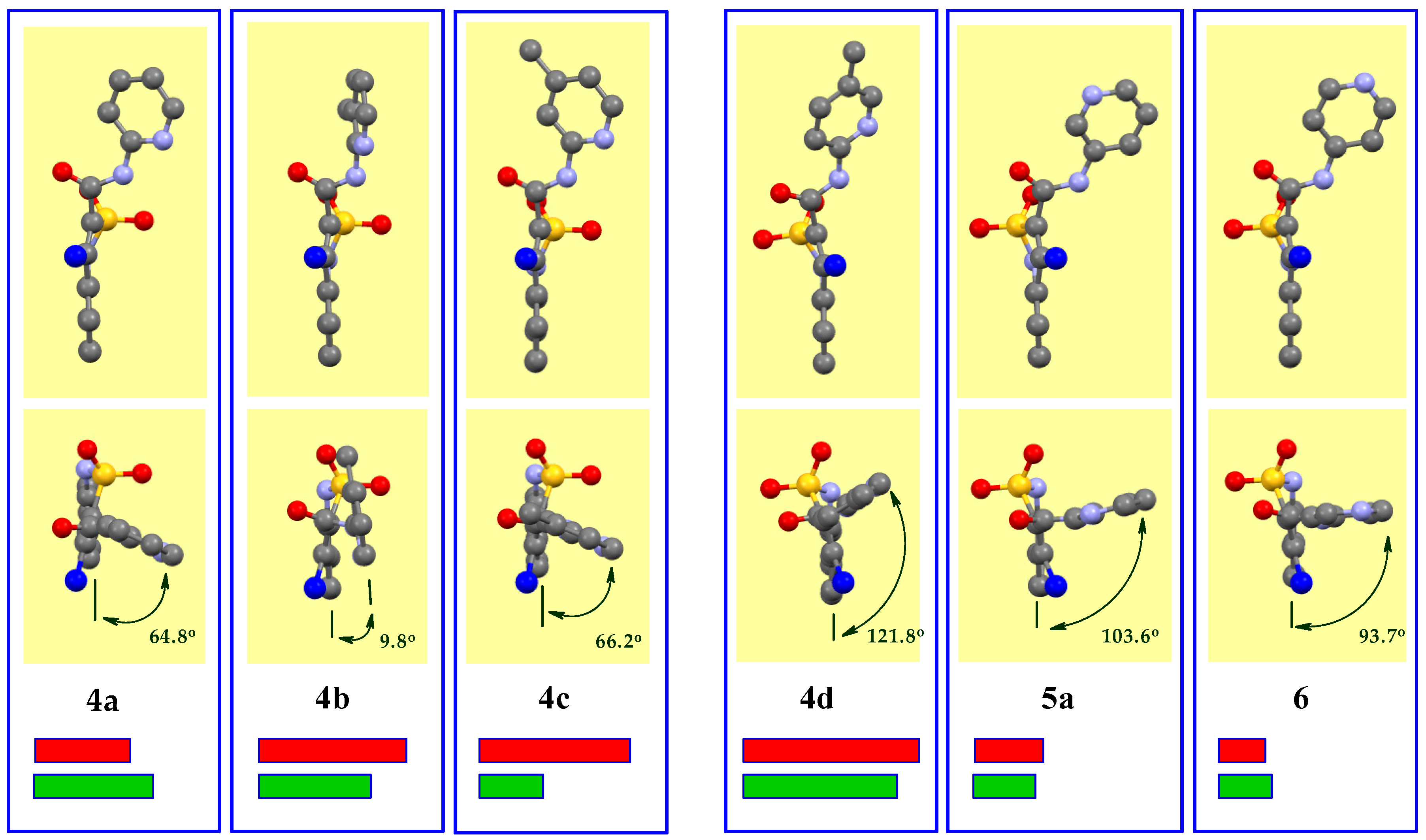
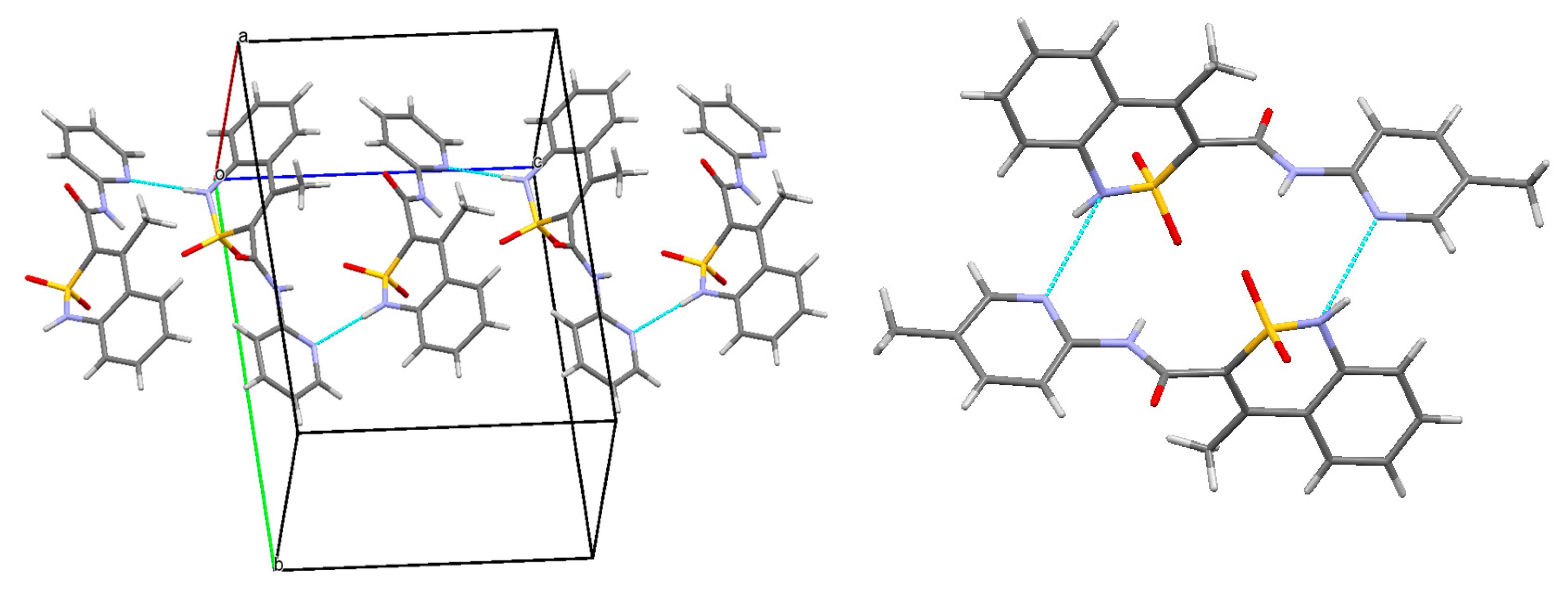
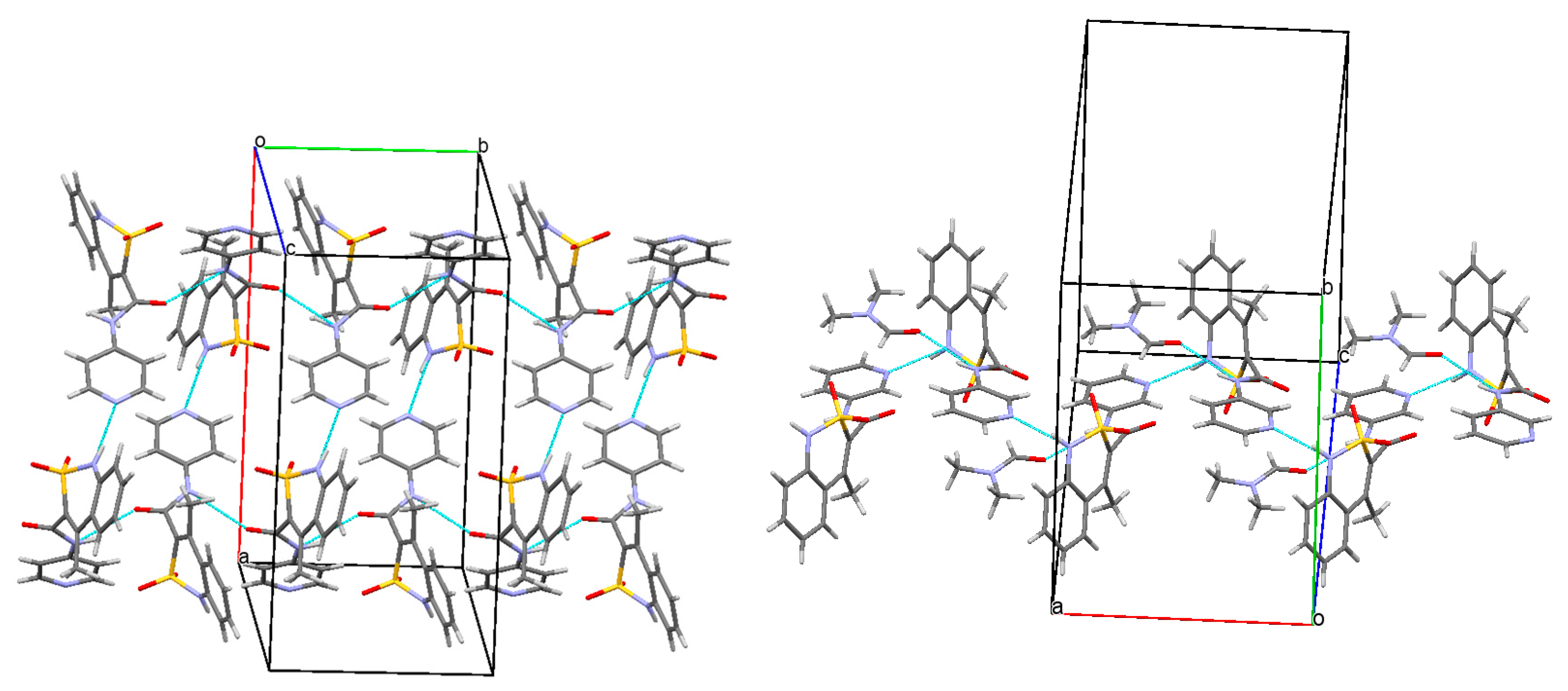
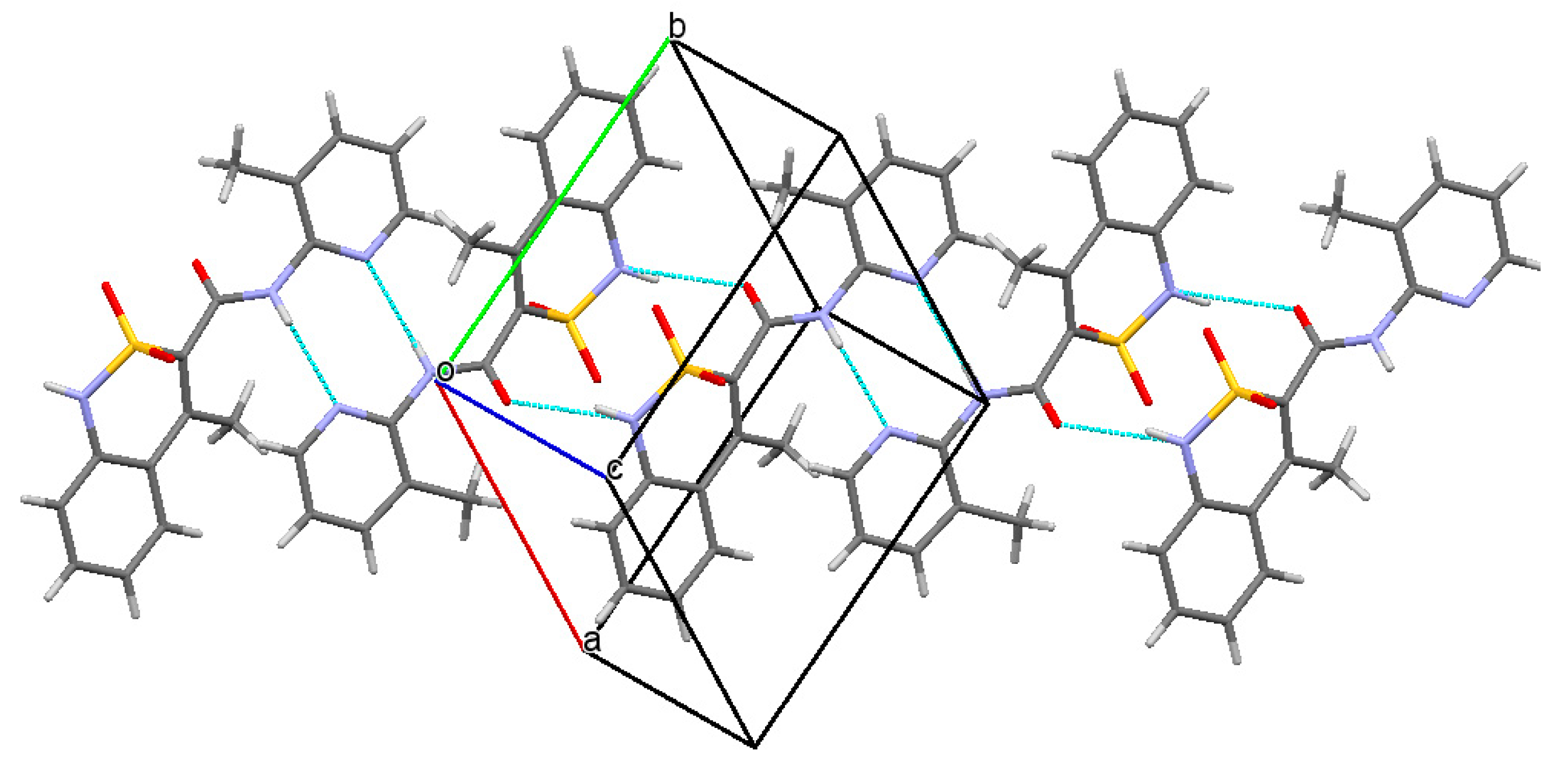
3.3. The Molecular and Crystal Structure Study
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- O’Neil, M.J.; Heckelman, P.E.; Koch, C.B.; Roman, K.J. The Merck Index: An Encyclopedia of Chemicals, Drugs, and Biologicals, 14th ed.; Merck and Co., Inc.: Whitehouse Station, NJ, USA, 2006. [Google Scholar]
- Kleemann, A.; Engel, J.; Kutscher, B.; Reichert, D. Pharmaceutical Substances: Syntheses, Patents, Applications of the Most Relevant APIs, 5th ed.; Thieme: Stuttgart, Germany, 2008. [Google Scholar]
- Ukrainets, I.V.; Gorokhova, O.V.; Jaradat, N.A.; Petrushova, L.A.; Mospanova, E.V.; Savchenkova, L.V.; Kuz’min, V.E.; Lyahovsky, A.V. 4-Hydroxyquinolin-2-ones and their close structural analogues as a new source of highly effective pain-killers. In Pain and Treatment; Racz, G.B., Noe, C.E., Eds.; InTech: Rijeka, Croatia, 2014; pp. 21–73. [Google Scholar]
- Ukrainets, I.V.; Petrushova, L.A.; Dzyubenko, S.P.; Liu, Y. 2,1-Benzothiazine 2,2-dioxides. 4. Synthesis, structure, and analgesic properties of 4-hydroxy-1-methyl-2,2-dioxo-N-(pyridin-2-yl)-1H-2λ6,1-benzothiazine-3-carboxamides. Chem. Heterocycl. Compd. 2014, 50, 564–572. [Google Scholar] [CrossRef]
- Shishkina, S.V.; Ukrainets, I.V.; Petrushova, L.A. Competition between intermolecular hydrogen bonding and stacking in the crystals of 4-hydroxy-N-(pyridin-2-yl)-2,2-dioxo-1H-2λ6,1-benzothiazine-3-carboxamides. Z. Kristallogr. 2017, 232, 307–316. [Google Scholar] [CrossRef]
- Ukrainets, I.V.; Hamza, G.M.; Burian, A.A.; Shishkina, S.V.; Voloshchuk, N.I.; Malchenko, O.V. 4-Methyl-2,2-dioxo-1H-2λ6,1-benzothiazine-3-carboxylic acid. Peculiarities of preparation, structure, and biological properties. Sci. Pharm. 2018, 86, 9. [Google Scholar] [CrossRef]
- Zhao, Y.; Truhlar, D.G. The M06 suite of density functionals for main group thermochemistry, thermochemical kinetics, noncovalent interactions, excited states, and transition elements: Two new functionals and systematic testing of four M06-class functionals and 12 other functionals. Theor. Chem. Acc. 2007, 120, 215–241. [Google Scholar]
- Kendall, R.A.; Dunning, T.H., Jr.; Harrison, R.J. Electron affinities of the first-row atoms revisited. Systematic basis sets and wave functions. J. Chem. Phys. 1992, 96, 6796–6806. [Google Scholar] [CrossRef]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Petersson, G.A.; Nakatsuji, H.; et al. Gaussian 09, Revision A.02. Gaussian, Inc.: Wallingford, UK, 2016. [Google Scholar]
- Weinhold, F. Natural Bond Orbital Methods. In Encyclopedia of Computational Chemistry; Schleyer, P.v.R., Allinger, N.L., Clark, T., Gasteiger, J., Kollman, P.A., Schaefer, H.F., III, Schreiner, P.R., Eds.; John Wiley & Sons: Chichester, UK, 1998; Volume 3, pp. 1792–1811. [Google Scholar]
- Glendening, E.D.; Badenhoop, J.K.; Reed, A.E.; Carpenter, J.E.; Bohmann, J.A.; Morales, C.M.; Weinhold, F. NBO 5.0; Theoretical Chemistry Institute, University of Wisconsin: Madison, WI, USA, 2001. [Google Scholar]
- Sheldrick, G.M. A short history of SHELX. Acta Crystallogr. Sect. A Found. Crystallogr. 2008, A64, 112–122. [Google Scholar] [CrossRef]
- Cambridge Crystallographic Data Center. Request Quoting via: CCDC 1910363. Available online: www.ccdc.cam.ac.uk/conts/retrieving.html (accessed on 16 April 2019).
- Cambridge Crystallographic Data Center. Request Quoting via: CCDC 1910364. Available online: www.ccdc.cam.ac.uk/conts/retrieving.html (accessed on 16 April 2019).
- Cambridge Crystallographic Data Center. Request Quoting via: CCDC 1910362. Available online: www.ccdc.cam.ac.uk/conts/retrieving.html (accessed on 16 April 2019).
- Cambridge Crystallographic Data Center. Request Quoting via: CCDC 1910367. Available online: www.ccdc.cam.ac.uk/conts/retrieving.html (accessed on 16 April 2019).
- Cambridge Crystallographic Data Center. Request Quoting via: CCDC 1910361. Available online: www.ccdc.cam.ac.uk/conts/retrieving.html (accessed on 16 April 2019).
- Cambridge Crystallographic Data Center. Request Quoting via: CCDC 1910365. Available online: www.ccdc.cam.ac.uk/conts/retrieving.html (accessed on 16 April 2019).
- Cambridge Crystallographic Data Center. Request Quoting via: CCDC 1910366. Available online: www.ccdc.cam.ac.uk/conts/retrieving.html (accessed on 16 April 2019).
- Ukrainian Law No. 3447-IV. On Protection of Animals from Severe Treatment. Available online: http://zakon2.rada.gov.ua/laws/show/3447-15 (accessed on 4 August 2017).
- Vogel, H.G. (Ed.) Drug Discovery and Evaluation: Pharmacological Assays, 2nd ed.; Springer: Berlin, Germany, 2008; pp. 1103–1106. [Google Scholar]
- Gregory, N.S.; Harris, A.L.; Robinson, C.R.; Dougherty, P.M.; Fuchs, P.N.; Sluka, K.A. An overview of animal models of pain: Disease models and outcome measures. J. Pain. 2013, 14, 1255–1269. [Google Scholar] [CrossRef]
- Ukrainets, I.V.; Sidorenko, L.V.; Gorokhova, O.V.; Shishkina, S.V. 4-Hydroxy-2-quinolones. 96. Synthesis and properties of 4-methyl-2-oxo-1,2-dihydroquinoline-3-carboxylic acid. Chem. Heterocycl. Compd. 2006, 42, 776–781. [Google Scholar] [CrossRef]
- Ukrainets, I.V.; Gorokhova, O.V.; Sidorenko, L.V.; Bereznyakova, N.L. 4-Hydroxy-2-quinolones. 111. Simple synthesis of 1-substituted 4-methyl-2-oxo-1,2-dihydroquinoline-3-carboxylic acids. Chem. Heterocycl. Compd. 2007, 43, 58–62. [Google Scholar] [CrossRef]
- Bochkov, A.F.; Smit, V.A. Organic Synthesis. The Aims, Methods, Tactics, Strategy; Nauka: Moscow, USSR, 1987; pp. 249–252. [Google Scholar]
- Ukrainets, I.V.; Hamza, G.M.; Burian, A.A.; Voloshchuk, N.I.; Malchenko, O.V.; Shishkina, S.V.; Grinevich, L.N.; Grynenko, V.V.; Sim, G. Molecular conformations and biological activity of N-hetaryl(aryl)-alkyl-4-methyl-2,2-dioxo-1H-2λ6,1-benzothiazine-3-carboxamides. Sci. Pharm. 2018, 86, 50. [Google Scholar] [CrossRef]
- Ukrainets, I.V.; Hamza, G.M.; Burian, A.A.; Voloshchuk, N.I.; Malchenko, O.V.; Shishkina, S.V.; Danylova, I.A.; Sim, G. The crystal structure of N-(1-arylethyl)-4-methyl-2,2-dioxo-1H-2λ6,1-benzothiazine-3-carboxamides as the factor determining biological activity thereof. Sci. Pharm. 2019, 87, 10. [Google Scholar] [CrossRef]
- Armstrong, A.; Li, W. N, N-Carbonyldiimidazole in Encyclopedia of Reagents for Organic Synthesis; John Wiley & Sons: Hoboken, NJ, USA, 2007. [Google Scholar]
- Ukrainets, I.V.; Bereznyakova, N.L.; Parshikov, V.A.; Naboka, O.I. 4-Hydroxy-2-quinolones. 142. 4-Methyl-2-oxo-1,2-dihydro-quinoline-3-carboxylic acid anilides as potential diuretics. Chem. Heterocycl. Compd. 2008, 44, 178–183. [Google Scholar] [CrossRef]
- Ukrainets, I.V.; Bereznyakova, N.L.; Parshikov, V.A. 4-Hydroxy-2-quinolones. 153. Synthesis of hetarylamides of 4-methyl-2-oxo-1,2-dihydroquinoline-3-carboxylic acid. Chem. Heterocycl. Compd. 2009, 45, 345–350. [Google Scholar] [CrossRef]
- Deady, L.W.; Stillman, D.C. Mechanisms for the acetylation of aminopyridines. Aust. J. Chem. 1978, 31, 1725–1730. [Google Scholar] [CrossRef]
- Boulton, A.J.; McKillop, A. Reactivity of six-membered rings. In Comprehensive Heterocyclic Chemistry; Katritzky, A.R., Rees, C.W., Eds.; Elsevier Science Ltd.: Oxford, UK, 1984; Volume 2, pp. 29–65. [Google Scholar]
- Lowe, P.A. Naphthyridines, pyridoquinolines, anthyridines and similar compounds. In Comprehensive Heterocyclic Chemistry; Katritzky, A.R., Rees, C.W., Eds.; Elsevier Science Ltd.: Oxford, UK, 1984; Volume 2, pp. 581–627. [Google Scholar]
- Wang, M.-X. Three Heterocyclic Rings Fused (5:6:6). In Comprehensive Heterocyclic Chemistry II; Katritzky, A.R., Rees, C.W., Scriven, E.F.V., Eds.; Elsevier Science Ltd.: Oxford, UK, 1996; Volume 8, pp. 1023–1064. [Google Scholar]
- Caprio, V. Pyridines and their benzo derivatives: Reactivity of substituents. In Comprehensive Heterocyclic Chemistry III; Katritzky, A.R., Ramsden, C.A., Scriven, E.F.V., Taylor, R.J.K., Eds.; Elsevier Science Ltd.: Oxford, UK, 2008; Volume 7, pp. 101–169. [Google Scholar]
- Uff, B.C. Pyridines and their benzo derivatives: (iii) reactivity of substituents. In Comprehensive Heterocyclic Chemistry; Katritzky, A.R., Rees, C.W., Eds.; Elsevier Science Ltd.: Oxford, UK, 1984; Volume 2, pp. 315–364. [Google Scholar]
- Pozharskii, A.F. Theoretical Fundamentals of Heterocycle Chemistry; Khimia: Moscow, USSR, 1985; pp. 126–160. [Google Scholar]
- Ukrainets, I.V.; Sidorenko, L.V.; Gorokhova, O.V. 4-Hydroxy-2-quinolones. 84. Synthesis of 5-R-5H-5,7a,12-triazabenzo[a]anthracene-6,7-diones. Chem. Heterocycl. Compd. 2005, 41, 896–904. [Google Scholar] [CrossRef]
- Ukrainets, I.V.; Bereznyakova, N.L.; Petyunin, G.P.; Tugaibei, I.A.; Rybakov, V.B.; Chernyshev, V.V.; Turov, A.V. 4-Hydroxy-2-quinolones. 120. Synthesis and structure of ethyl 2-hydroxy-4-oxo-4H-pyrido[1,2-a]pyrimidine3-carboxylate. Chem. Heterocycl. Compd. 2007, 43, 729–739. [Google Scholar] [CrossRef]
- Sykes, P. A Guidebook to Mechanism in Organic Chemistry, 6th ed.; Pearson: London, UK, 2003; pp. 203–245. [Google Scholar]
- Ukrainets, I.V.; Sidorenko, L.V.; Svechnikova, E.N.; Shishkin, O.V. 4-Hydroxy-2-quinolones. 130. The reactivity of ethyl 4-hydroxy-2-oxo-1,2-dihydroquinoline-3-carboxylates. Chem. Heterocycl. Compd. 2007, 43, 1275–1279. [Google Scholar] [CrossRef]
- Ukrainets, I.V.; Mospanova, E.V.; Davidenko, A.A. Using bioisosteric replacements to enhance the analgesic properties of 4-hydroxy-6,7-dimethoxy-2-oxo-1,2-dihydroquinoline-3-carboxamides. Pharm. Chem. J. 2016, 50, 365–368. [Google Scholar] [CrossRef]
- Ukrainets, I.V.; Mospanova, O.V.; Bereznyakova, N.L.; Davidenko, O.O. Polymorphism and the analgesic activity of N-(3-pyridylmethyl)-4-hydroxy-2-oxo-1,2,5,6,7,8-hexahydroquinoline-3-carboxamide. J. Org. Pharm. Chem. 2015, 13, 41–46. [Google Scholar] [CrossRef]
- Ukrainets, I.V.; Mospanova, E.V.; Bereznyakova, N.L.; Davidenko, A.A. Modification of the benzene moiety in the quinolone nucleus of 4-hydroxy-6,7-dimethoxy-2-oxo-N-(pyridin-3-ylmethyl)-1,2-dihydroquinoline-3-carboxamide as an attempt to enhance its analgesic activity. Pharm. Chem. J. 2018, 52, 825–829. [Google Scholar] [CrossRef]
- Ukrainets, I.V.; Mospanova, E.V. Polymorphism and analgesic properties of N-(3-pyridylmethyl)-4-hydroxy-2-oxo-1,2,5,6,7,8-hexahydroquinoline-3-carboxamide. In European Science and Technology, Materials of the V International Research and Practice Conference, Vol. I, Munich, Germany, October 3–4, 2013; Vela: Waldkraiburg, Germany, 2013; pp. 79–81. [Google Scholar]
- Ukrainets, I.V.; Shishkina, S.V.; Baumer, V.N.; Gorokhova, O.V.; Petrushova, L.A.; Sim, G. The structure of two pseudo-enantiomeric forms of N-benzyl-4-hydroxy-1-methyl-2,2-dioxo-1H-2λ6,1-benzothiazine-3-carboxamide and their analgesic properties. Acta Crystallogr. Sect. C: Struct. Chem. 2016, 72 Pt 5, 411–415. [Google Scholar] [CrossRef]
- Ukrainets, I.V.; Petrushova, L.A.; Shishkina, S.V.; Grinevich, L.A.; Sim, G. Synthesis, spatial structure and analgesic activity of sodium 3-benzylaminocarbonyl-1-methyl-2,2-dioxo-1H-2λ6,1-benzothiazin-4-olate solvates. Sci. Pharm. 2016, 84, 705. [Google Scholar] [CrossRef]
- Ukrainets, I.V.; Burian, A.A.; Baumer, V.N.; Shishkina, S.V.; Sidorenko, L.V.; Tugaibei, I.A.; Voloshchuk, N.I.; Bondarenko, P.S. Synthesis, crystal structure and biological activity of ethyl 4-methyl-2,2-dioxo-1H-2λ6,1-benzothiazine-3-carboxylate polymorphic forms. Sci. Pharm. 2018, 86, 21. [Google Scholar] [CrossRef]
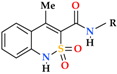
| Entry | Product | R | Pain Threshold on Damaged Extremity (g/mm2) | Pain Threshold on Non-Damaged Extremity (g/mm2) | Δ Pain Threshold | Analgesic Activity, Compared to Control (%) |
|---|---|---|---|---|---|---|
| 1 | 2 |  | 358.0 ± 25.5 | 216.0 ± 15.6 | 142.0 ± 14.0 1 | +55.3 |
| 2 | 4a | 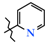 | 449.0 ± 20.5 | 291.0 ± 11.6 | 158.0 ± 4.6 1,2 | +50.3 |
| 3 | 4b |  | 350.0 ± 26.1 | 281.0 ± 27.4 | 69.0 ± 6.4 1,2 | +78.3 |
| 4 | 4c |  | 346.0 ± 27.0 | 282.0 ± 14.9 | 64.0 ± 4.9 1 | +79.9 |
| 5 | 4d |  | 362.0 ± 33.2 | 341.0 ± 35.3 | 21.0 ± 4.0 1,2 | +93.4 |
| 6 | 5 |  | 390.0 ± 34.9 | 353.0 ± 29.9 | 37.0 ± 4.3 1,2 | +88.4 |
| 7 | 5a |  | 419.0 ± 38.3 | 206.0 ± 12.6 | 213.0 ± 29.9 | +33.0 |
| 8 | 6 | 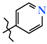 | 412.0 ± 30.9 | 172.0 ± 16.0 | 240.0 ± 15.7 1 | +24.5 |
| 9 | Lornoxicam | − | 441.0 ± 33.1 | 346.0 ± 30.2 | 95.0 ± 12.7 1 | +70.1 |
| 10 | Control | − | 593.0 ± 21.1 | 275.0 ± 32.1 | 318.0 ± 18.6 1 | 0 |
| Entry | Product | R | Volume of Damaged Extremity (mm3) | Volume of Non-Damaged Extremity (mm3) | Δ Volume (Volume Increase) | Anti-Inflammatory Activity, Compared to Control (%) |
|---|---|---|---|---|---|---|
| 1 | 2 |  | 469.1 ± 33.6 | 180.7 ± 65.5 | 288.4 ± 22.0 1 | +30.3 |
| 2 | 4a | 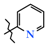 | 349.4 ± 33.5 | 197.9 ± 43.2 | 151.4 ± 16.9 1,2 | +63.4 |
| 3 | 4b |  | 330.5 ± 27.3 | 159.7 ± 8.31 | 170.8 ± 6.5 1 | +58.7 |
| 4 | 4c | 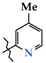 | 493.5 ± 45.0 | 218.7 ± 29.4 | 274.7 ± 21.6 1,2 | +33.6 |
| 5 | 4d | 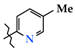 | 288.3 ± 53.5 | 212.5 ± 23.9 | 75.8 ± 7.9 1,2 | +81.7 |
| 6 | 5 |  | 243.4 ± 37.5 | 148.4 ± 28.2 | 94.9 ± 4.8 1,2 | +77.1 |
| 7 | 5a | 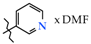 | 473.7 ± 35.3 | 209.7 ± 41.0 | 263.9 ± 14.8 | +36.2 |
| 8 | 6 |  | 427.9 ± 44.3 | 129.5 ± 36.1 | 298.4 ± 15.01 | +27.9 |
| 9 | Lornoxicam | − | 360.5 ± 82.5 | 263.9 ± 60.9 | 96.6 ± 5.7 1 | +76.7 |
| 10 | Control | − | 568.7 ± 27.3 | 154.9 ± 11.4 | 413.9 ± 32.2 1 | 0 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ukrainets, I.V.; Burian, A.A.; Hamza, G.M.; Voloshchuk, N.I.; Malchenko, O.V.; Shishkina, S.V.; Sidorenko, L.V.; Burian, K.O.; Sim, G. Synthesis and Regularities of the Structure–Activity Relationship in a Series of N-Pyridyl-4-methyl-2,2-dioxo-1H-2λ6,1-benzothiazine-3-carboxamides. Sci. Pharm. 2019, 87, 12. https://doi.org/10.3390/scipharm87020012
Ukrainets IV, Burian AA, Hamza GM, Voloshchuk NI, Malchenko OV, Shishkina SV, Sidorenko LV, Burian KO, Sim G. Synthesis and Regularities of the Structure–Activity Relationship in a Series of N-Pyridyl-4-methyl-2,2-dioxo-1H-2λ6,1-benzothiazine-3-carboxamides. Scientia Pharmaceutica. 2019; 87(2):12. https://doi.org/10.3390/scipharm87020012
Chicago/Turabian StyleUkrainets, Igor V., Anna A. Burian, Ganna M. Hamza, Natali I. Voloshchuk, Oxana V. Malchenko, Svitlana V. Shishkina, Lyudmila V. Sidorenko, Kateryna O. Burian, and Galina Sim. 2019. "Synthesis and Regularities of the Structure–Activity Relationship in a Series of N-Pyridyl-4-methyl-2,2-dioxo-1H-2λ6,1-benzothiazine-3-carboxamides" Scientia Pharmaceutica 87, no. 2: 12. https://doi.org/10.3390/scipharm87020012
APA StyleUkrainets, I. V., Burian, A. A., Hamza, G. M., Voloshchuk, N. I., Malchenko, O. V., Shishkina, S. V., Sidorenko, L. V., Burian, K. O., & Sim, G. (2019). Synthesis and Regularities of the Structure–Activity Relationship in a Series of N-Pyridyl-4-methyl-2,2-dioxo-1H-2λ6,1-benzothiazine-3-carboxamides. Scientia Pharmaceutica, 87(2), 12. https://doi.org/10.3390/scipharm87020012





