The Crystal Structure of N-(1-Arylethyl)-4-methyl- 2,2-dioxo-1H-2λ6,1-benzothiazine-3-carboxamides as the Factor Determining Biological Activity Thereof
Abstract
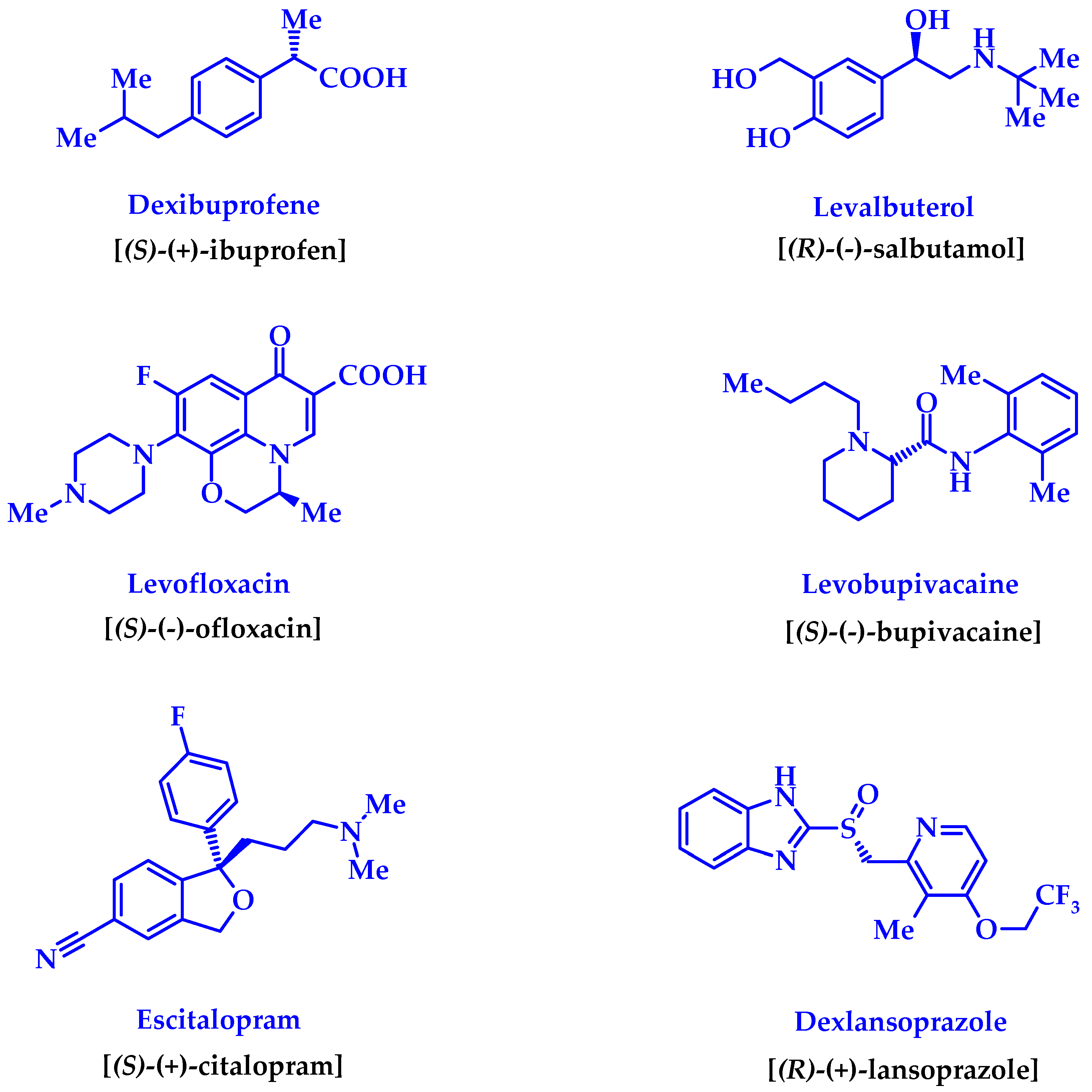
1. Introduction
2. Materials and Methods
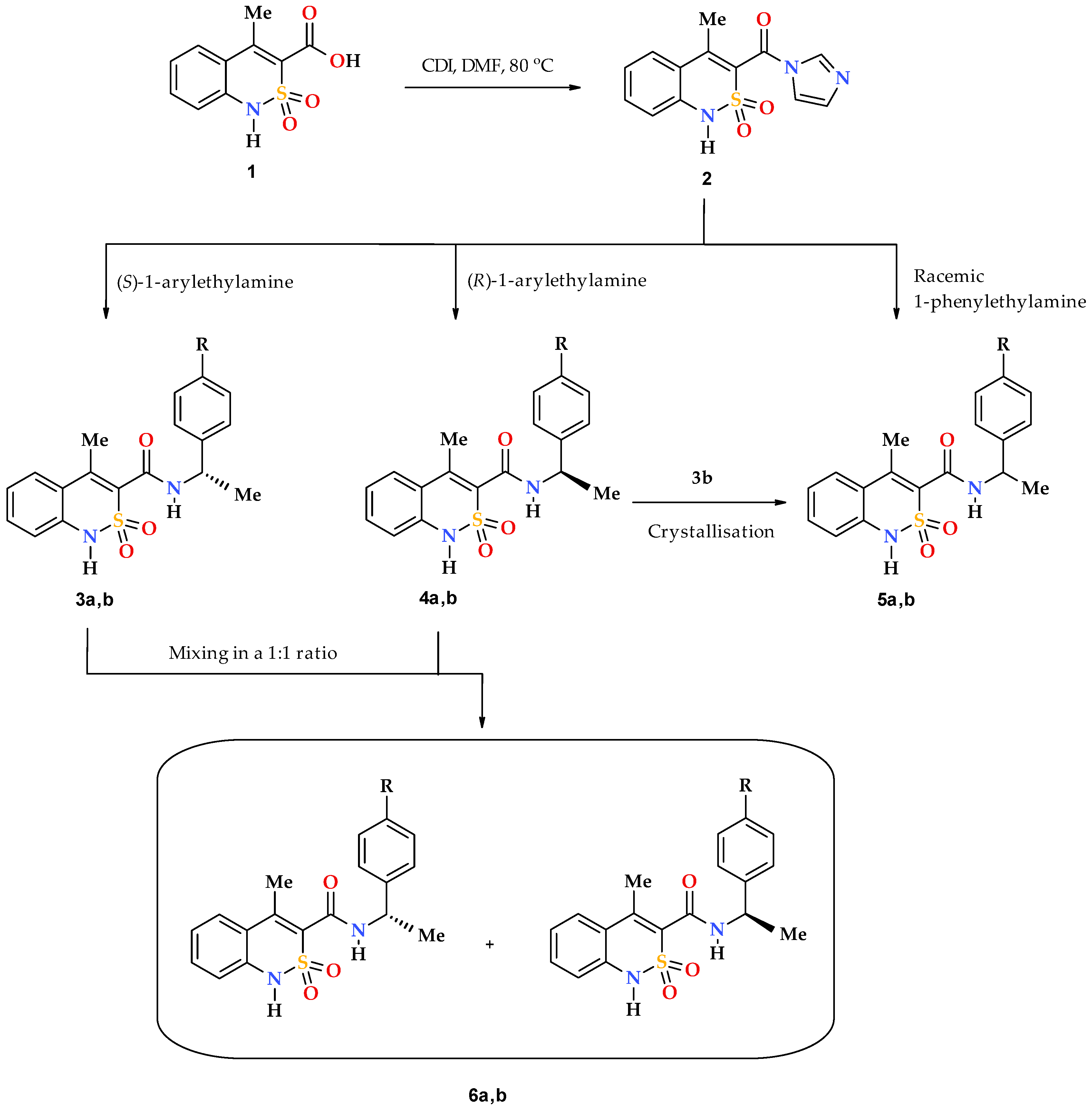
2.1. Chemistry
2.2. General Procedure for the Synthesis of N-(1-arylethyl)-4-methyl-2,2-dioxo-1H-2λ6,1-benzothiazine-3-carboxamides 3, 4 and 5a
2.3. N-[1-(4-Methoxyphenyl)ethyl]-4-methyl-2,2-dioxo-1H-2λ6,1-benzothiazine-3-carboxamide (5b)
2.4. Mechanical Racemic Mixture of Optically Active N-(1-Phenylethyl)-4-methyl-2,2-dioxo-1H-2λ6,1- benzothiazine-3-carboxamides (6a)
2.5. Mechanical Racemic Mixture of Optically Active N-[1-(4-Methoxyphenyl)ethyl]-4-methyl-2,2-dioxo- 1H-2λ6,1-benzothiazine-3-carboxamides (6b)
2.6. X-ray Structural Analysis of N-[(1S)-1-Phenylethyl]-4-methyl-2,2-dioxo-1H-2λ6,1-benzothiazine-3-carboxamide (3a)
2.7. X-ray Structural Analysis of N-(1-Phenylethyl)-4-methyl-2,2-dioxo-1H-2λ6,1-benzothiazine-3-carboxamide (5a)
2.8. X-ray Structural Analysis of N-[(1S)-1-(4-Methoxyphenyl)ethyl]-4-methyl-2,2-dioxo-1H-2λ6,1-benzothiazine-3-carboxamide (3b)
2.9. X-ray Structural Analysis of N-[(1R)-1-(4-Methoxyphenyl)ethyl]-4-methyl-2,2-dioxo-1H-2λ6,1-benzothiazine-3-carboxamide (4b)
2.10. X-ray Structural Analysis of N-[1-(4-Methoxyphenyl)ethyl]-4-methyl-2,2-dioxo-1H-2λ6,1-benzothiazine-3-carboxamide (5b)
2.11. Pharmacology
Analgesic and Anti-Inflammatory Tests
3. Results and Discussion
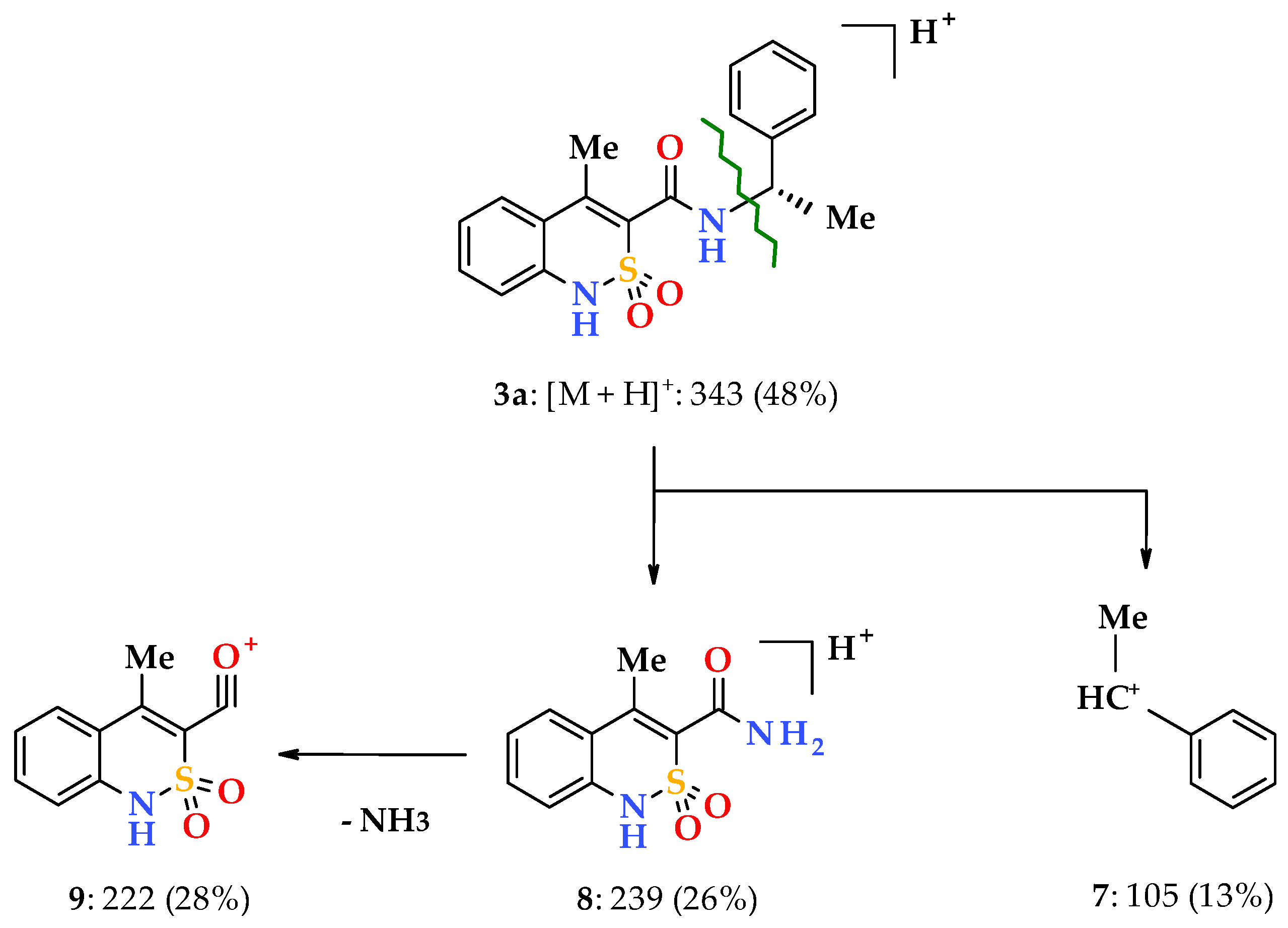
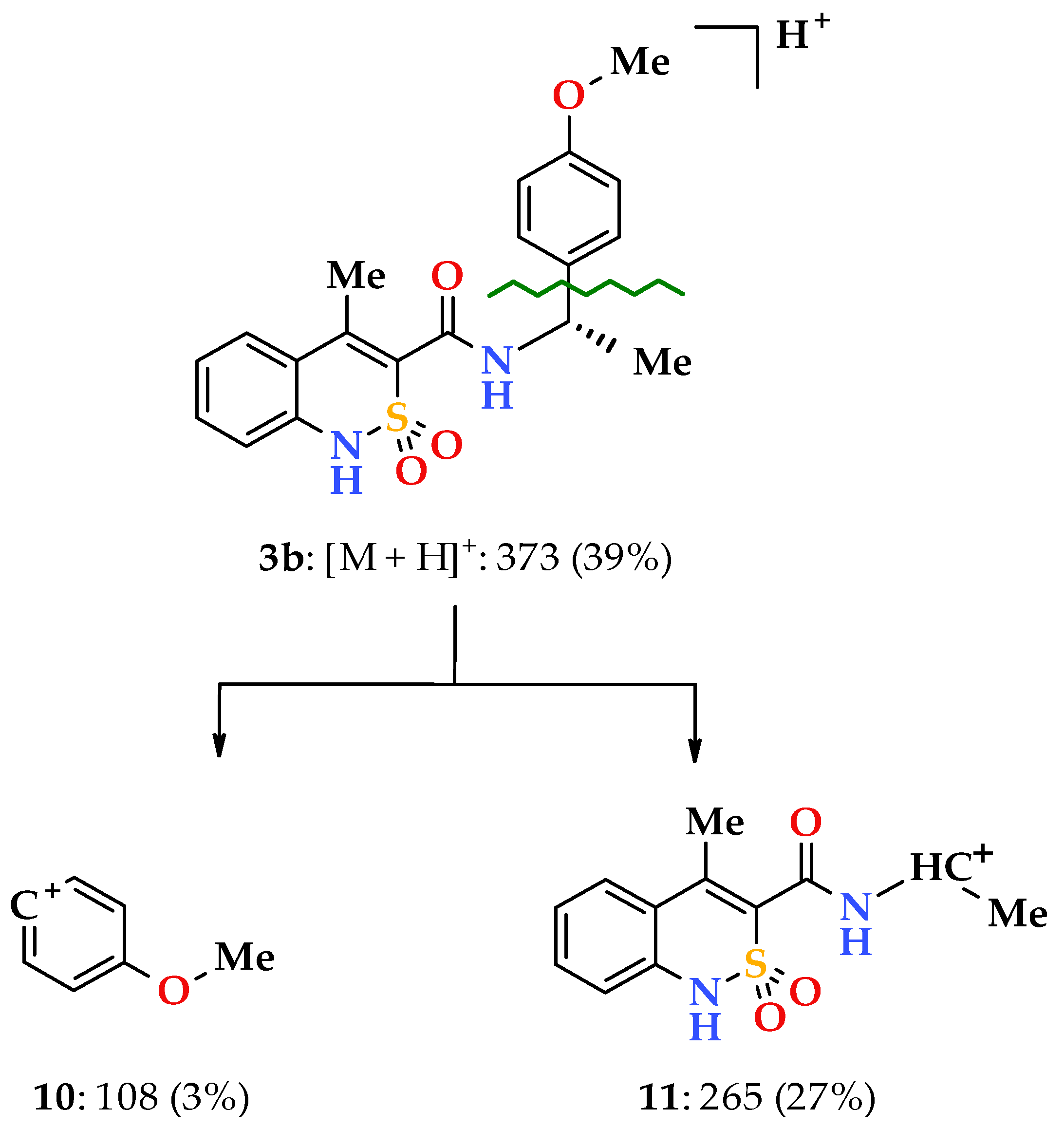
3.1. Chemistry
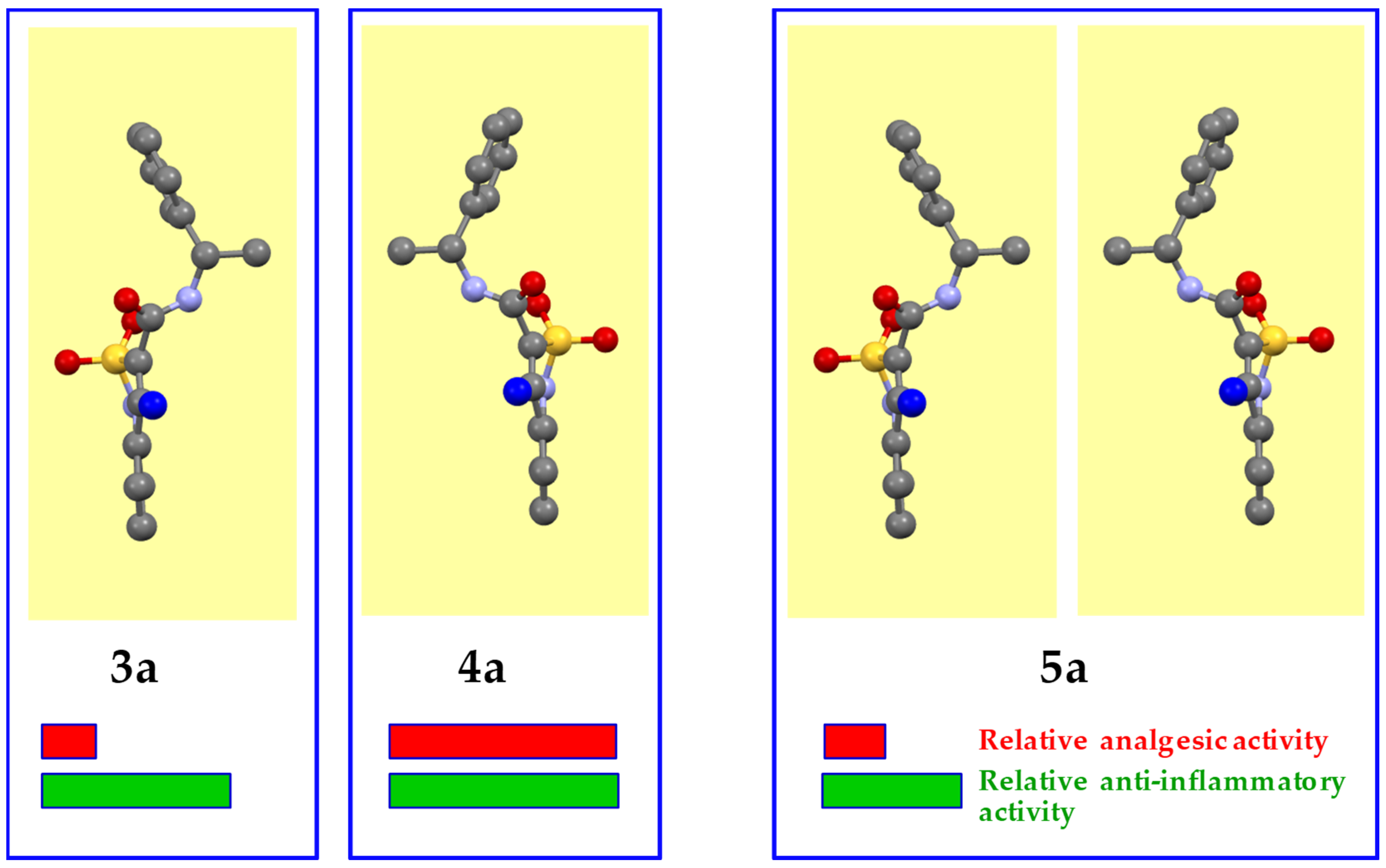
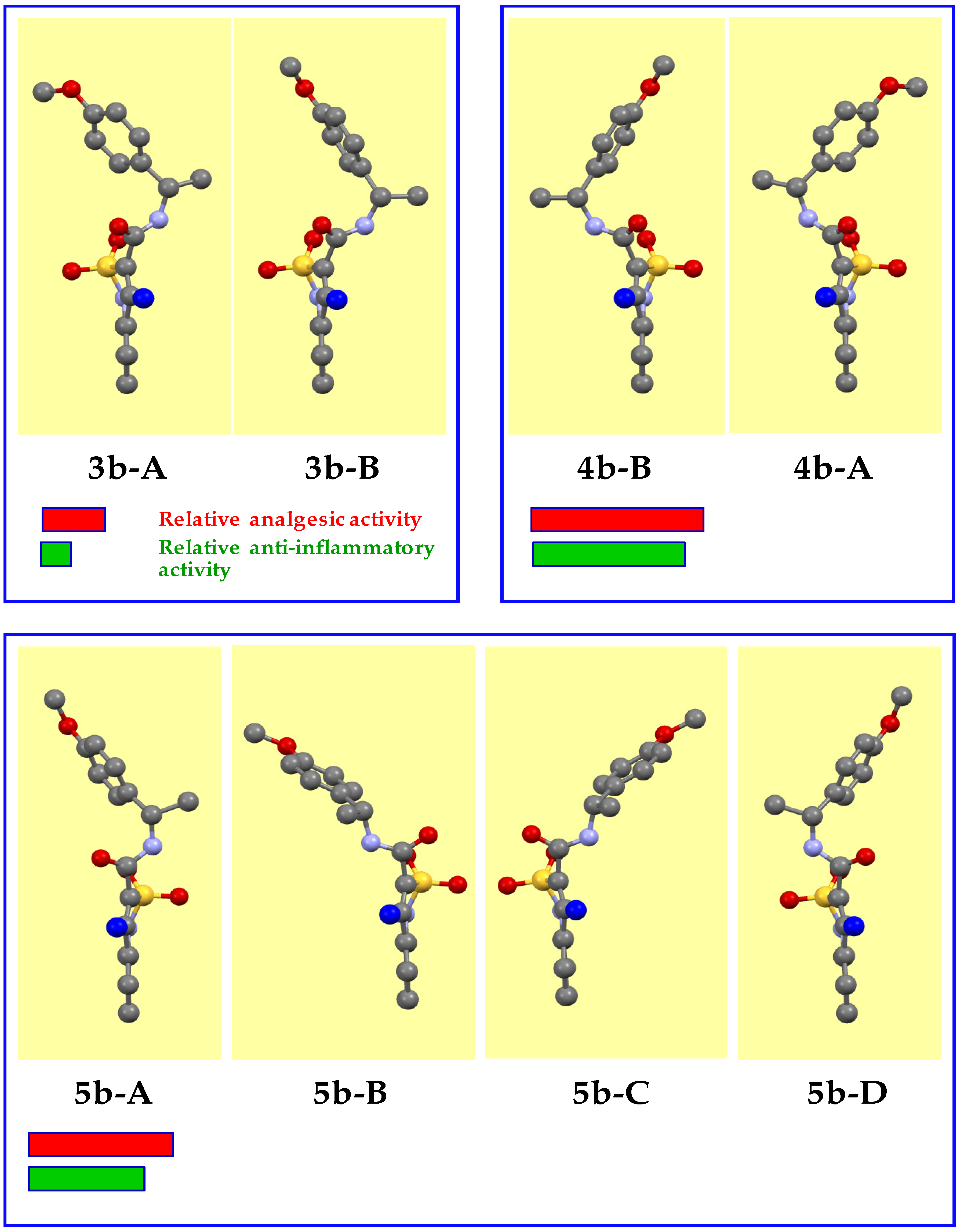
3.2. Evaluation of the Analgesic and Anti-Inflammatory Activity
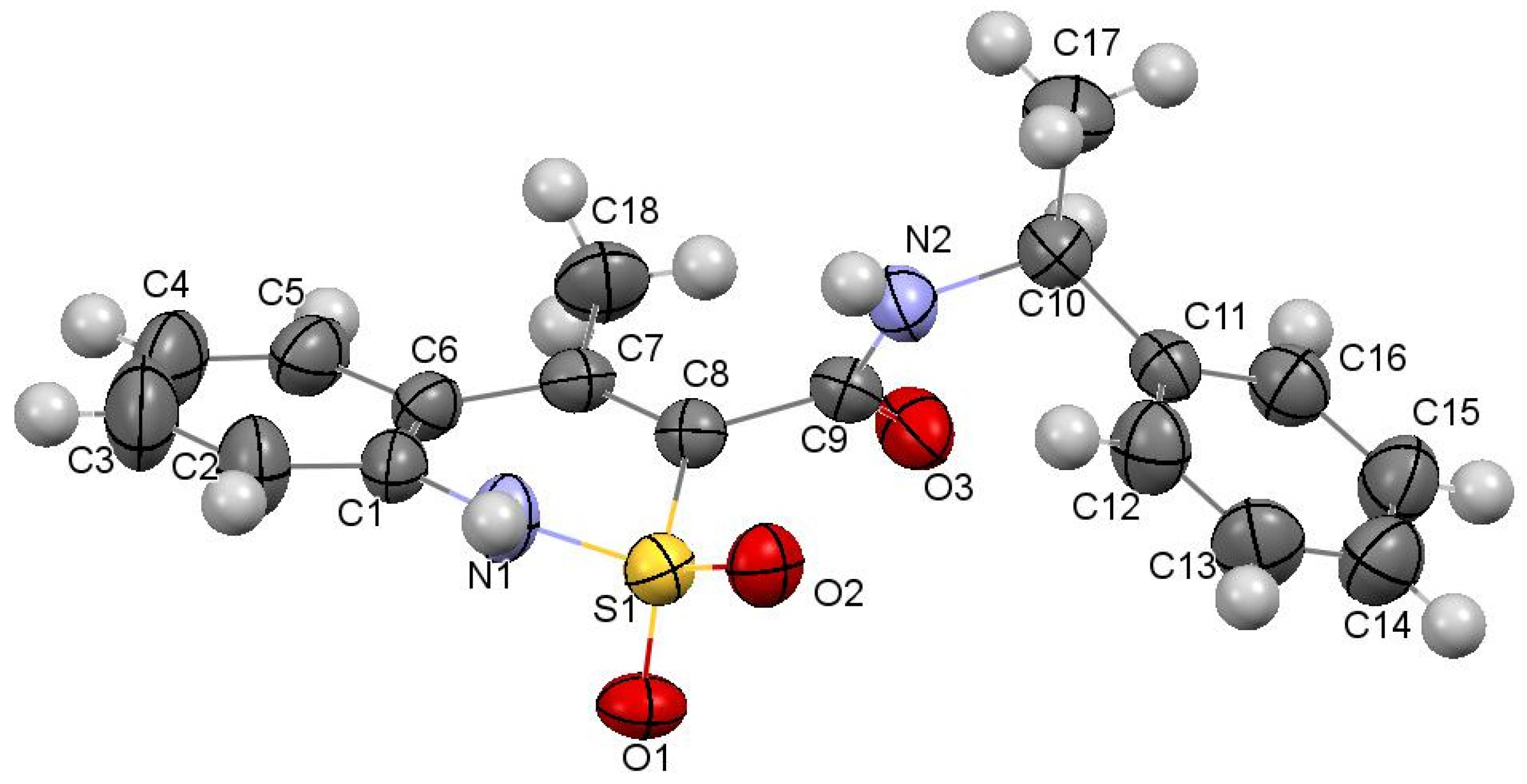
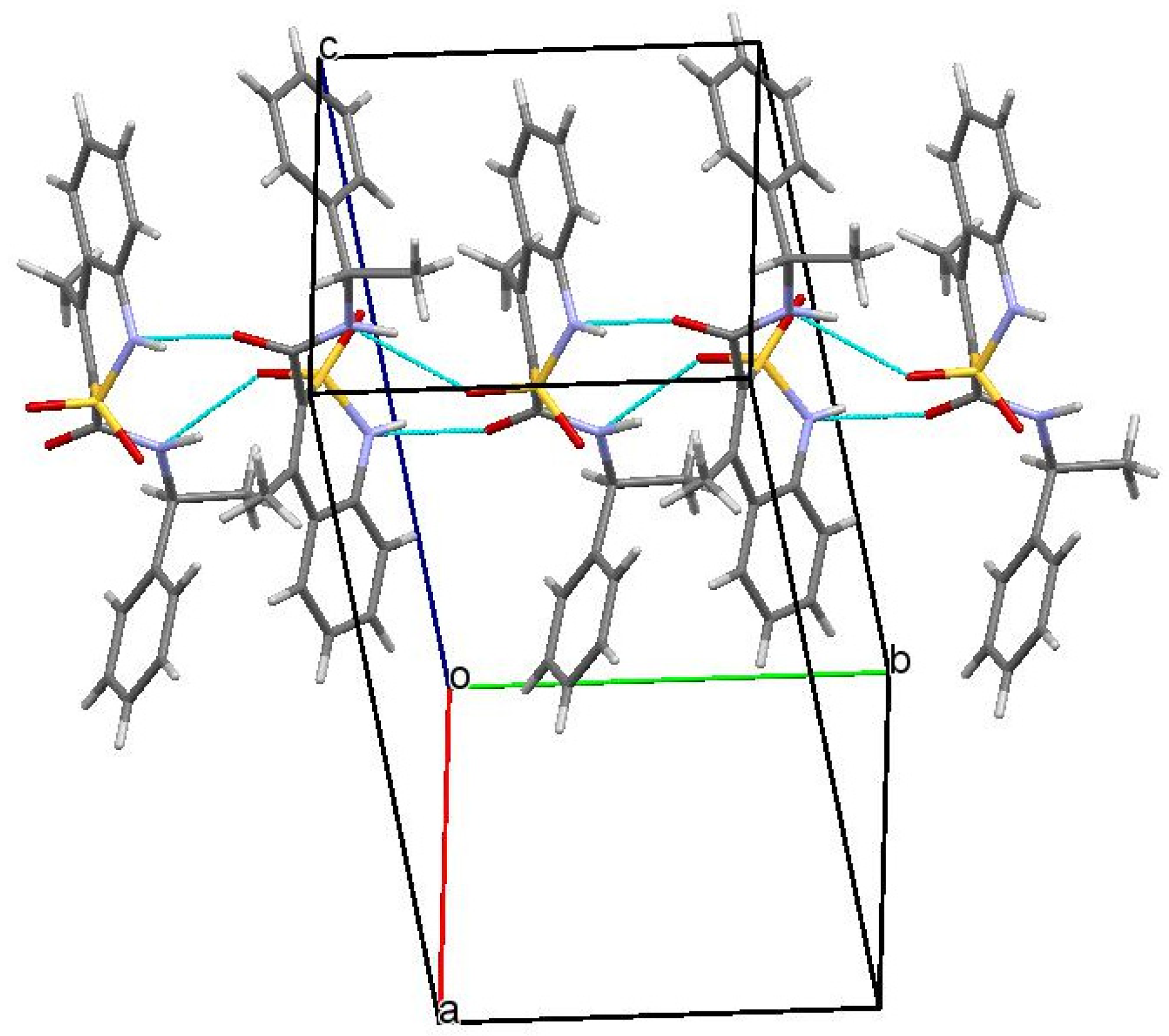
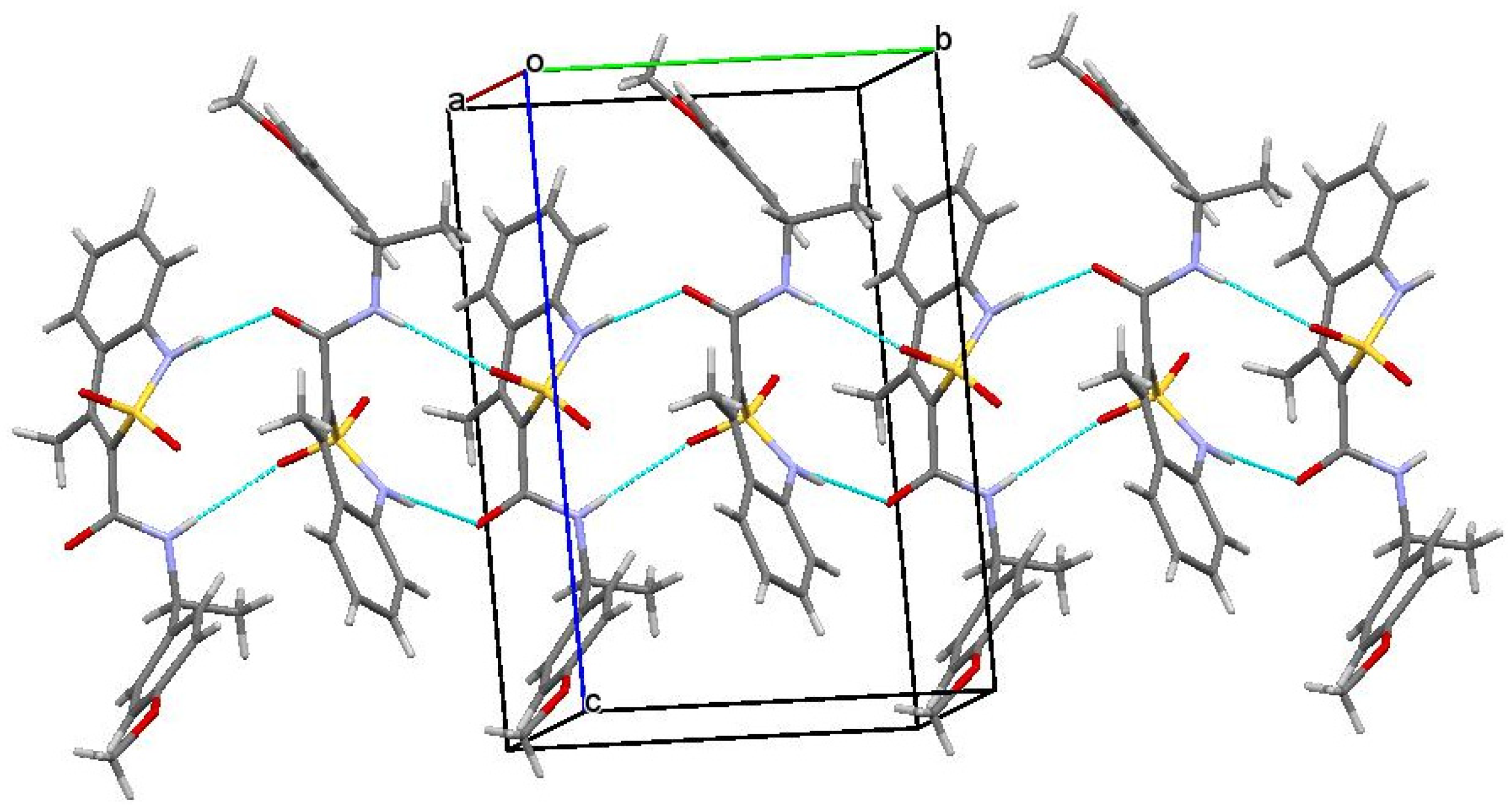
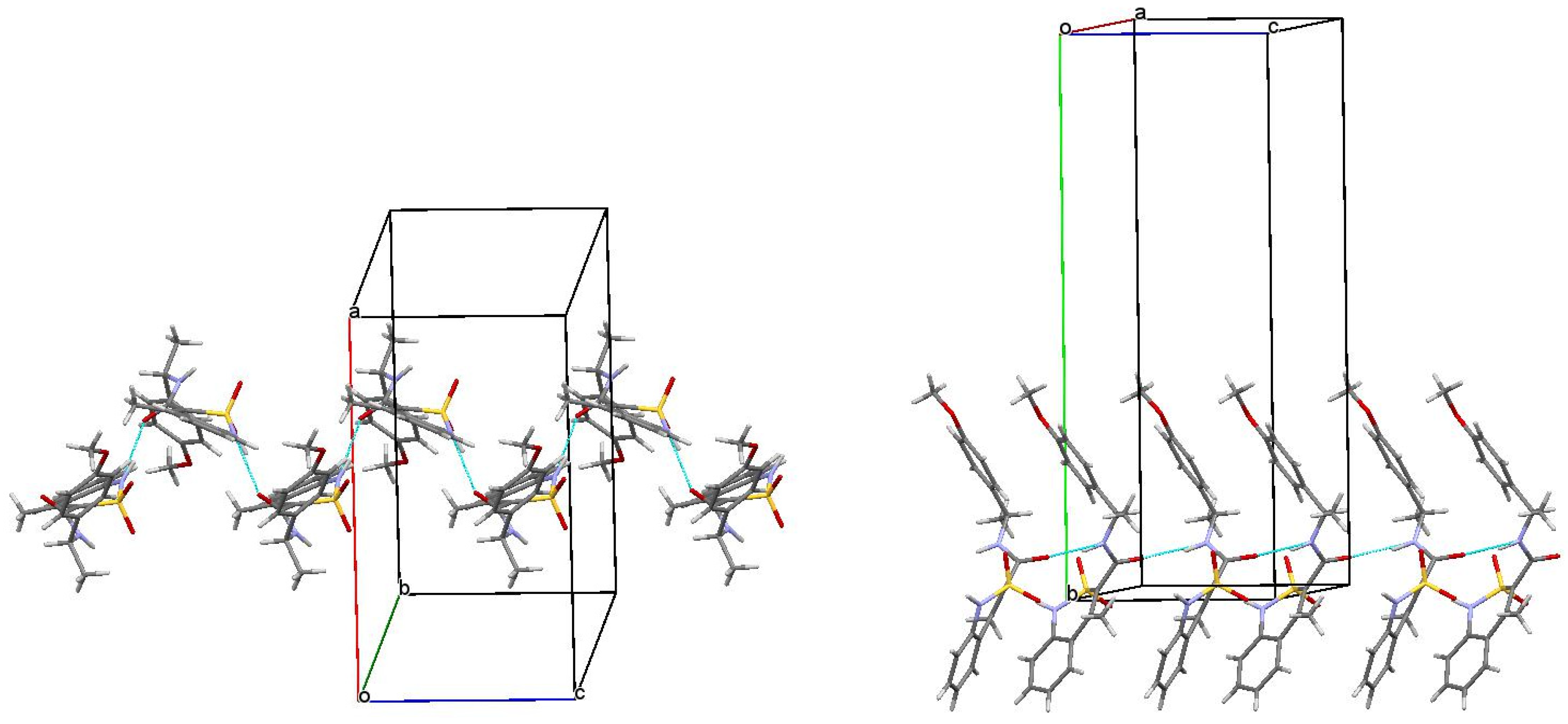
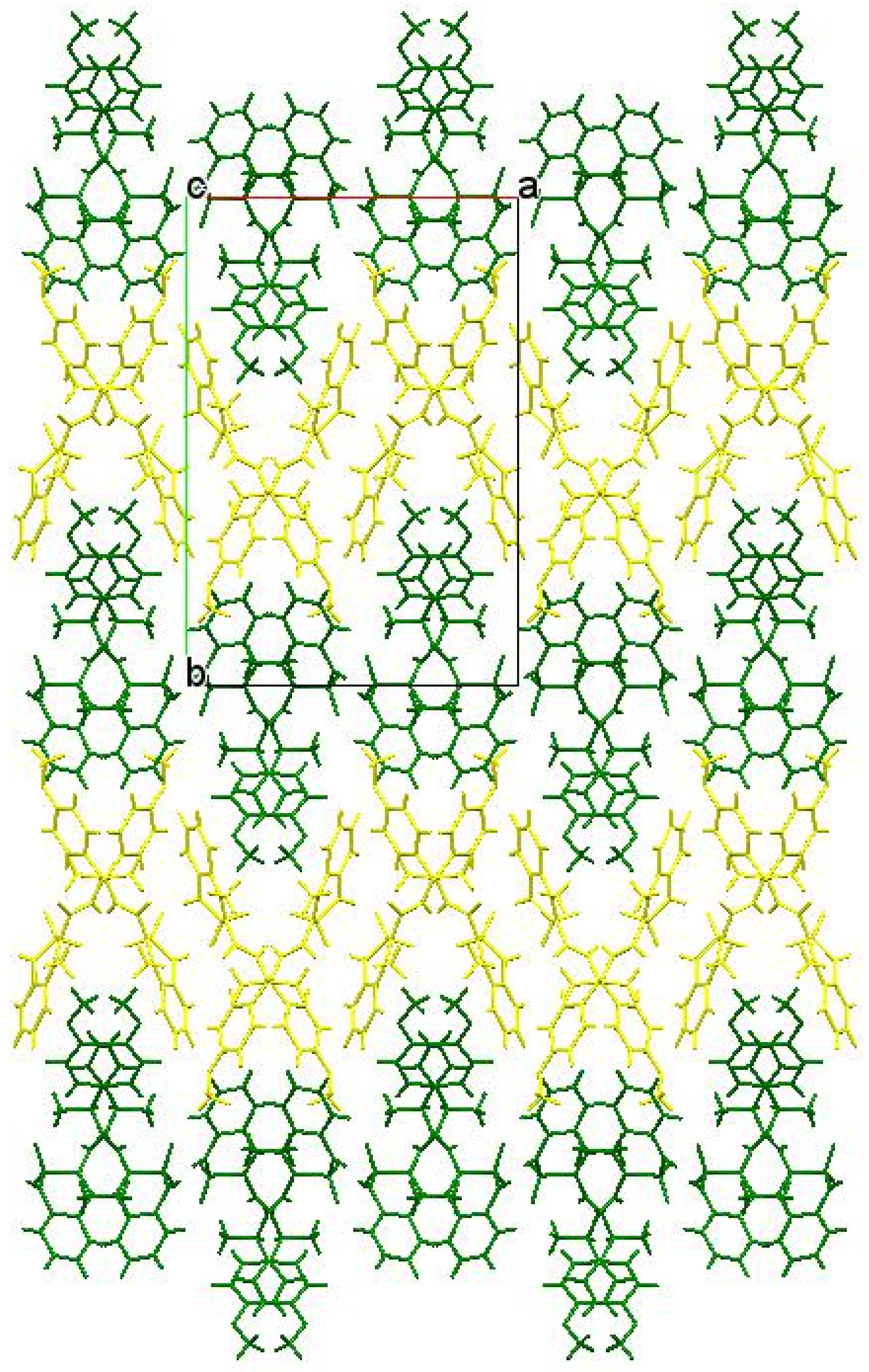
3.3. The Molecular and Crystal Structure Study
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ariëns, E.J. Stereochemistry, a basis for sophisticated nonsense in pharmacokinetics and clinical pharmacology. Eur. J. Clin. Pharmacol. 1984, 26, 663–668. [Google Scholar] [CrossRef] [PubMed]
- Kubinyi, H. In search of the new compounds-leaders for creation of drugs. Russ. Chem. J. 2006, 50, 5–17. Available online: http://www.chem.msu.su/rus/journals/jvho/2006-2/5.pdf (accessed on 26 May 2006).
- Agranat, I.; Caner, H.; Caldwell, J. Putting chirality to work: The strategy of chiral switches. Nat. Rev. Drug Discov. 2002, 1, 753–768. [Google Scholar] [CrossRef] [PubMed]
- Calcaterra, A.; D’Acquarica, I. The market of chiral drugs: Chiral switches versus de novo enantiomerically pure compounds. J. Pharm. Biomed. Anal. 2018, 147, 323–340. [Google Scholar] [CrossRef] [PubMed]
- Tucker, G.T. Chiral switches. Lancet 2000, 355, 1085–1087. [Google Scholar] [CrossRef]
- Thacker, H.P. S-amlodipine—The 2007 clinical review. J. Indian Med. Assoc. 2007, 105, 180–182. [Google Scholar] [PubMed]
- Hardikar, M.S. Chiral non-steroidal anti-inflammatory drugs—A review. Indian Med. Assoc. 2008, 106, 615–618. [Google Scholar]
- Gordon, A.L.; Lopatko, O.V.; Somogyi, A.A.; Foster, D.J.; White, J.M. (R)- and (S)-methadone and buprenorphine concentration ratios in maternal and umbilical cord plasma following chronic maintenance dosing in pregnancy. Br. J. Clin. Pharmacol. 2010, 70, 895–902. [Google Scholar] [CrossRef]
- Matera, M.G.; Calzetta, L.; Rogliani, P.; Bardaro, F.; Page, C.P.; Cazzola, M. Evaluation of the effects of the R- and S-enantiomers of salbutamol on equine isolated bronchi. Pulm. Pharmacol. Ther. 2011, 24, 221–226. [Google Scholar] [CrossRef] [PubMed]
- Yurovskaya, М.А.; Kurkin, А.V. Some aspects of the relationship between chirality and biological activity. In Proceedings of the Synthesis and Complexation Achievements, Materials of the all-Russian Scientific Conference with International Participation, Dedicated to the International Year of Chemistry, Moscow, Russia, 18–22 April 2011; p. 19. [Google Scholar]
- Kaneko, T. Troglitazone (CS-045): A new antidiabetic agent. Horm. Metab. Res. 1997, 29, 203–213. [Google Scholar] [CrossRef] [PubMed]
- De Sousa, D.P.; Nóbrega, F.F.; Santos, C.C.; de Almeida, R.N. Anticonvulsant activity of the linalool enantiomers and racemate: Investigation of chiral influence. Nat. Prod. Commun. 2010, 5, 1847–1851. [Google Scholar] [CrossRef] [PubMed]
- Raffa, R.B.; Friderichs, E.; Reimann, W.; Shank, R.P.; Codd, E.E.; Vaught, J.L.; Jacoby, H.I.; Selve, N. Complementary and synergistic antinociceptive interaction between the enantiomers of tramadol. J. Pharmacol. Exp. Ther. 1993, 267, 331–340. [Google Scholar] [PubMed]
- Ukrainets, I.V.; Hamza, G.M.; Burian, A.A.; Shishkina, S.V.; Voloshchuk, N.I.; Malchenko, O.V. 4-Methyl-2,2-dioxo-1H-2λ6,1-benzothiazine-3-carboxylic acid. Peculiarities of preparation, structure, and biological properties. Sci. Pharm. 2018, 86, 9. [Google Scholar] [CrossRef]
- Sheldrick, G.M. A short history of SHELX. Acta Crystallogr. Sect. A Found. Crystallogr. 2008, A64, 112–122. [Google Scholar] [CrossRef] [PubMed]
- Cambridge Crystallographic Data Center. Request Quoting via: CCDC 1899013. Available online: www.ccdc.cam.ac.uk/conts/retrieving.html (accessed on 23 February 2019).
- Cambridge Crystallographic Data Center. Request Quoting via: CCDC 1899015. Available online: www.ccdc.cam.ac.uk/conts/retrieving.html (accessed on 23 February 2019).
- Cambridge Crystallographic Data Center. Request Quoting via: CCDC 1899014. Available online: www.ccdc.cam.ac.uk/conts/retrieving.html (accessed on 23 February 2019).
- Cambridge Crystallographic Data Center. Request Quoting via: CCDC 1899016. Available online: www.ccdc.cam.ac.uk/conts/retrieving.html (accessed on 23 February 2019).
- Cambridge Crystallographic Data Center. Request Quoting via: CCDC 1899017. Available online: www.ccdc.cam.ac.uk/conts/retrieving.html (accessed on 23 February 2019).
- Ukrainian Law No. 3447-IV. On Protection of Animals from Severe Treatment. Available online: http://zakon2.rada.gov.ua/laws/show/3447-15 (accessed on 4 August 2017).
- Vogel, H.G. (Ed.) Drug Discovery and Evaluation: Pharmacological Assays, 2nd ed.; Springer: Berlin, Germany, 2008; pp. 1103–1106. [Google Scholar]
- Gregory, N.S.; Harris, A.L.; Robinson, C.R.; Dougherty, P.M.; Fuchs, P.N.; Sluka, K.A. An overview of animal models of pain: Disease models and outcome measures. J. Pain 2013, 14, 1255–1269. [Google Scholar] [CrossRef]
- Hsu, L.C.; Kim, H.; Yang, X.; Ross, D. Large scale chiral chromatography for the separation of an enantiomer to accelerate drug development. Chirality 2011, 23, 361–366. [Google Scholar] [CrossRef] [PubMed]
- Schuur, B.; Verkuijl, B.J.; Minnaard, A.J.; de Vries, J.G.; Heeres, H.J.; Feringa, B.L. Chiral separation by enantioselective liquid-liquid extraction. Org. Biomol. Chem. 2011, 9, 36–51. [Google Scholar] [CrossRef]
- Van Leeuwen, T.; Gan, J.; Kistemaker, J.C.; Pizzolato, S.F.; Chang, M.C.; Feringa, B.L. Enantiopure functional molecular motors obtained by a switchable chiral-resolution process. Chemistry 2016, 22, 7054–7058. [Google Scholar] [CrossRef]
- Kannappan, V.; Mannemala, S.S. Simultaneous enantioseparation and purity determination of chiral switches of amlodipine and atenolol by liquid chromatography. J. Pharm. Biomed. Anal. 2016, 120, 221–227. [Google Scholar] [CrossRef]
- Kato, N.; Mita, T.; Kanai, M.; Therrien, B.; Kawano, M.; Yamaguchi, K.; Danjo, H.; Sei, Y.; Sato, A.; Furusho, S.; et al. Assembly state of catalytic modules as chiral switches in asymmetric Strecker amino acid synthesis. J. Am. Chem. Soc. 2006, 128, 6768–6769. [Google Scholar] [CrossRef]
- Fuwa, H.; Suzuki, T.; Kubo, H.; Yamori, T.; Sasaki, M. Total synthesis and biological assessment of (−)-exiguolide and analogues. Chemistry 2011, 17, 2678–2688. [Google Scholar] [CrossRef] [PubMed]
- Umezawa, T.; Shibata, M.; Kaneko, K.; Okino, T.; Matsuda, F. Asymmetric total synthesis of danicalipin a and evaluation of biological activity. Org. Lett. 2011, 13, 904–907. [Google Scholar] [CrossRef]
- Rossi, S.; Porta, R.; Brenna, D.; Puglisi, A.; Benaglia, M. Stereoselective catalytic synthesis of active pharmaceutical ingredients in homemade 3D-printed mesoreactors. Angew. Chem. Int. Ed. 2017, 56, 4290–4294. [Google Scholar] [CrossRef] [PubMed]
- Núñez, M.C.; García-Rubiño, M.E.; Conejo-García, A.; Cruz-López, O.; Kimatrai, M.; Gallo, M.A.; Espinosa, A.; Campos, J.M. Homochiral drugs: A demanding tendency of the pharmaceutical industry. Curr. Med. Chem. 2009, 16, 2064–2074. [Google Scholar] [CrossRef] [PubMed]
- Ukrainets, I.V.; Hamza, G.M.; Burian, A.A.; Voloshchuk, N.I.; Malchenko, O.V.; Shishkina, S.V.; Grinevich, L.N.; Grynenko, V.V.; Sim, G. Molecular conformations and biological activity of N-hetaryl(aryl)alkyl-4-methyl-2,2-dioxo-1H-2λ6,1-benzothiazine-3-carboxamides. Sci. Pharm. 2018, 86, 50. [Google Scholar] [CrossRef]
- Terentev, P.B.; Stankiavichyus, A.P. Mass Spectrometry Bioactive Nitrogenous Bases; Mokslas: Vilnius, Lithuania, 1987; pp. 60–64. [Google Scholar]
- Potapov, V.M. Stereochemistry; Khimia: Moscow, Russia, 1988; pp. 282–284. [Google Scholar]
- Sykes, P. A Guidebook to Mechanism in Organic Chemistry, 6th ed.; Pearson: London, UK, 2003; pp. 340–358. [Google Scholar]
- Glusker, J.P.; Domenicano, A. Introduction to X-ray crystallography. In Accurate Molecular Structures. Their Determination and Importance; Domenicano, A., Hargittai, I., Eds.; Oxford University Press: Oxford, UK, 1992; pp. 158–210. [Google Scholar]
- Ukrainets, I.V.; Taran, S.G.; Likhanova, N.V.; Rybakov, V.B.; Gorokhova, O.V.; Nidal Amin Jaradat. 4-Hydroxy-2-quinolones. 38. Synthesis, structure, and anticonvulsant activity of optically active 1-phenylethylamides of 1-R-4-hydroxy-2-oxo-3-quinolinecarboxylic acids. Chem. Heterocycl. Compd. 2000, 36, 49–56. [Google Scholar] [CrossRef]
- Ukrainets, I.V.; Taran, S.G.; Likhanova, N.V.; Nidal Amin Jaradat; Shishkin, O.V. 4-Hydroxy-2-quinolones. 39. Structure of 6-bromo-4-hydroxy-1-isoamyl-2-oxo-3-quinolinecarboxylic acid. Chem. Heterocycl. Compd. 2000, 36, 57–61. [Google Scholar] [CrossRef]
- Zefirov, N.S.; Palyulin, V.A.; Dashevskaya, E.E. Stereochemical studies. XXXIV. Quantitative description of ring puckering via torsional angles. The case of six-membered rings. J. Phys. Org. Chem. 1990, 3, 147–158. [Google Scholar] [CrossRef]
- Zefirov, Y.V. Reduced intermolecular contacts and specific interactions in molecular crystals. Crystallogr. Rep. 1997, 42, 865–886. [Google Scholar]
- Orpen, A.G.; Brammer, L.; Allen, F.H.; Kennard, O.; Watson, D.G.; Taylor, R. Typical interatomic distances in organic compounds and organometallic compounds and coordination complexes of the d- and f-block metals. In Structure Correlation; Burgi, H.-B., Dunitz, J.D., Eds.; Wiley-VCH: Weinheim, Germany, 1994; Volume 2, pp. 741–926. [Google Scholar]
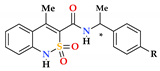
| Entry | Product | Configuration of Chiral Centre | R | Pain Thresholdon Damaged Extremity (g/mm2) | Pain Threshold on Non-Damaged Extremity (g/mm2) | Δ Pain Threshold | Analgesic Activity, Compared to Control (%) |
|---|---|---|---|---|---|---|---|
| 1 | 3a | (S) | H | 514.0 ± 33.2 | 265.0 ± 29.0 | 249.0 ± 22.1 1,2,3 | +21.7 |
| 2 | 3b | (S) | OMe | 450.0 ± 29.3 | 236.0 ± 15.6 | 214.0 ± 19.0 1,2,3 | +32.7 |
| 3 | 4a | (R) | H | 369.0 ± 15.0 | 335.2 ± 24.1 | 33.8 ± 5.2 1,2,3 | +89.4 |
| 4 | 4b | (R) | OMe | 416.0 ± 37.9 | 382.0 ± 32.2 | 34.0 ± 5.5 1,2 | +89.3 |
| 5 | 5a | (±) | H | 469.0 ± 20.1 | 228.0 ± 10.1 | 241.0 ± 42.2 1,2,3 | +24.2 |
| 6 | 5b | (±) | OMe | 408.0 ± 10.3 | 333.0 ± 18.5 | 75.0 ± 7.3 1,2,3 | +76.4 |
| 7 | 6a | (S) & (R) | H | 501.0 ± 54.4 | 282.0 ± 15.4 | 219.0 ± 29.8 1,2 | +31.1 |
| 8 | 6b | (S) & (R) | OMe | 356.0 ± 15.2 | 186.0 ± 12.6 | 170.0 ± 33.4 1,2 | +46.5 |
| 9 | Lornoxicam | − | − | 441.0 ± 33.1 | 346.0 ± 30.2 | 95.0 ± 12.7 1 | +70.1 |
| 10 | Diclofenac | − | − | 538.0 ± 23.6 | 479.0 ± 32.8 | 59.0 ± 15.3 1 | +81.4 |
| 11 | Control | − | − | 593.0 ± 62.7 | 257.0 ± 41.4 | 318.0 ± 58.2 1 | 0 |
| Entry | Product | Configuration of Chiral Centre | R | Volume of Damaged Extremity (mm3) | Volume of Non-Damaged Extremity (mm3) | Δ Volume (Volume Increase) | Anti- Inflammatory Activity, Compared to Control (%) |
|---|---|---|---|---|---|---|---|
| 1 | 3a | (S) | H | 607.8 ± 15.5 | 500.9 ± 17.6 | 106.9 ± 7.7 1,2,3 | +74.2 |
| 2 | 3b | (S) | OMe | 660.4 ± 47.4 | 311.6 ± 25.1 | 348.7 ± 22.8 1,2,3 | +15.7 |
| 3 | 4a | (R) | H | 520.9 ± 87.7 | 478.9 ± 87.3 | 41.9 ± 3.6 1,2,3 | +89.9 |
| 4 | 4b | (R) | OMe | 636.2 ± 74.9 | 552.0 ± 53.8 | 84.2 ± 6.9 1,2,3 | +79.7 |
| 5 | 5a | (±) | H | 648.6 ± 42.1 | 464.2 ± 13.9 | 184.4 ± 26.3 1,2 | +55.4 |
| 6 | 5b | (±) | OMe | 535.7 ± 64.3 | 374.5 ± 31.8 | 161.1 ± 19.3 1,2,3 | +61.1 |
| 7 | 6a | (S) & (R) | H | 687.3 ± 94.0 | 360.5 ± 32.7 | 326.8 ± 59.4 1,2,3 | +21.0 |
| 8 | 6b | (S) & (R) | OMe | 743.8 ± 98.6 | 379.1 ± 91.6 | 364.6 ± 87.5 1,2,3 | +11.9 |
| 9 | Lornoxicam | − | − | 360.5 ± 82.5 | 263.9 ± 60.9 | 96.6 ± 22.7 1 | +76.7 |
| 10 | Diclofenac | − | − | 397.6 ± 22.4 | 306.6 ± 17.6 | 91.0 ± 16.0 1 | +78.0 |
| 11 | Control | − | − | 768.7 ± 61.0 | 354.9 ± 22.9 | 413.7 ± 72.1 1 | 0 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ukrainets, I.V.; Hamza, G.M.; Burian, A.A.; Voloshchuk, N.I.; Malchenko, O.V.; Shishkina, S.V.; Danylova, I.A.; Sim, G. The Crystal Structure of N-(1-Arylethyl)-4-methyl- 2,2-dioxo-1H-2λ6,1-benzothiazine-3-carboxamides as the Factor Determining Biological Activity Thereof. Sci. Pharm. 2019, 87, 10. https://doi.org/10.3390/scipharm87020010
Ukrainets IV, Hamza GM, Burian AA, Voloshchuk NI, Malchenko OV, Shishkina SV, Danylova IA, Sim G. The Crystal Structure of N-(1-Arylethyl)-4-methyl- 2,2-dioxo-1H-2λ6,1-benzothiazine-3-carboxamides as the Factor Determining Biological Activity Thereof. Scientia Pharmaceutica. 2019; 87(2):10. https://doi.org/10.3390/scipharm87020010
Chicago/Turabian StyleUkrainets, Igor V., Ganna M. Hamza, Anna A. Burian, Natali I. Voloshchuk, Oxana V. Malchenko, Svitlana V. Shishkina, Irina A. Danylova, and Galina Sim. 2019. "The Crystal Structure of N-(1-Arylethyl)-4-methyl- 2,2-dioxo-1H-2λ6,1-benzothiazine-3-carboxamides as the Factor Determining Biological Activity Thereof" Scientia Pharmaceutica 87, no. 2: 10. https://doi.org/10.3390/scipharm87020010
APA StyleUkrainets, I. V., Hamza, G. M., Burian, A. A., Voloshchuk, N. I., Malchenko, O. V., Shishkina, S. V., Danylova, I. A., & Sim, G. (2019). The Crystal Structure of N-(1-Arylethyl)-4-methyl- 2,2-dioxo-1H-2λ6,1-benzothiazine-3-carboxamides as the Factor Determining Biological Activity Thereof. Scientia Pharmaceutica, 87(2), 10. https://doi.org/10.3390/scipharm87020010





