Influence of Fragrances on Human Psychophysiological Activity: With Special Reference to Human Electroencephalographic Response
Abstract
:1. Introduction
2. Fragrance Components
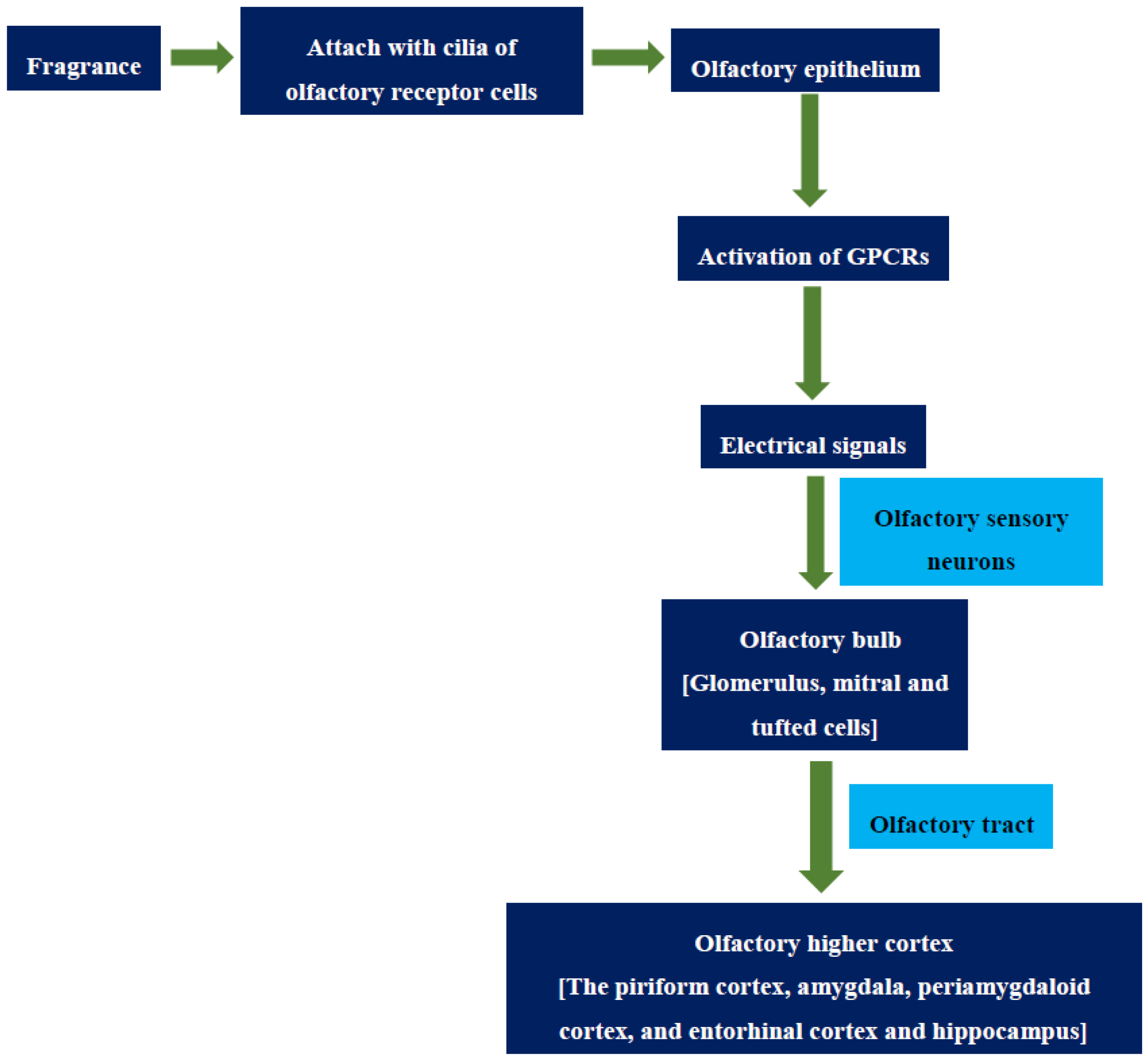
3. The Olfactory Process
4. Electroencephalography (EEG)
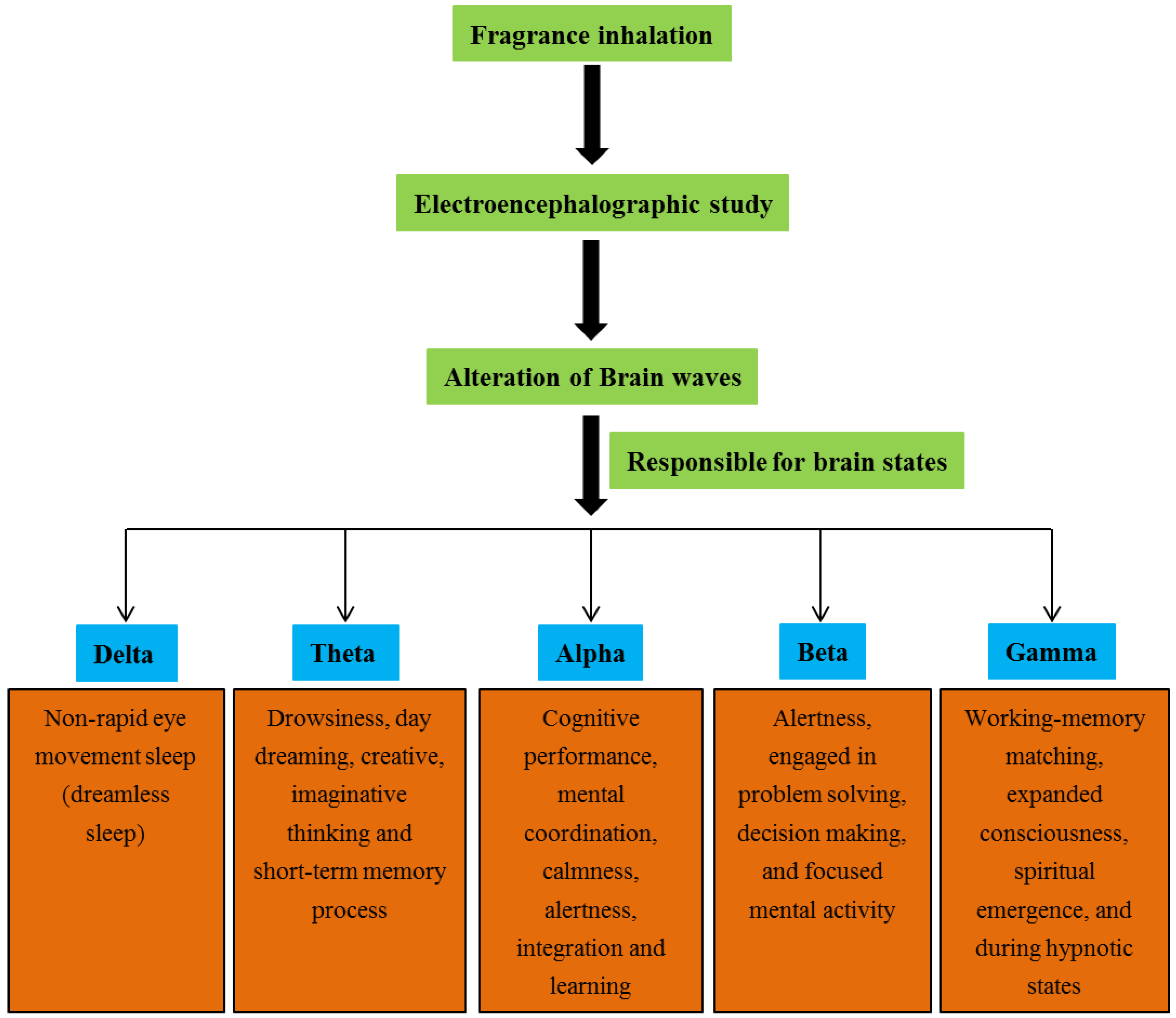
4.1. Brain Waves and Their Functions
4.1.1. Delta Waves
4.1.2. Theta Waves
4.1.3. Alpha Waves
4.1.4. Beta Waves
4.1.5. Gamma Waves
4.2. Brain Lobes (Regions) and Their Functions
4.2.1. Frontal Region
4.2.2. Parietal Region
4.2.3. Temporal Region
4.2.4. Occipital Region
4.3. Administration of Fragrances
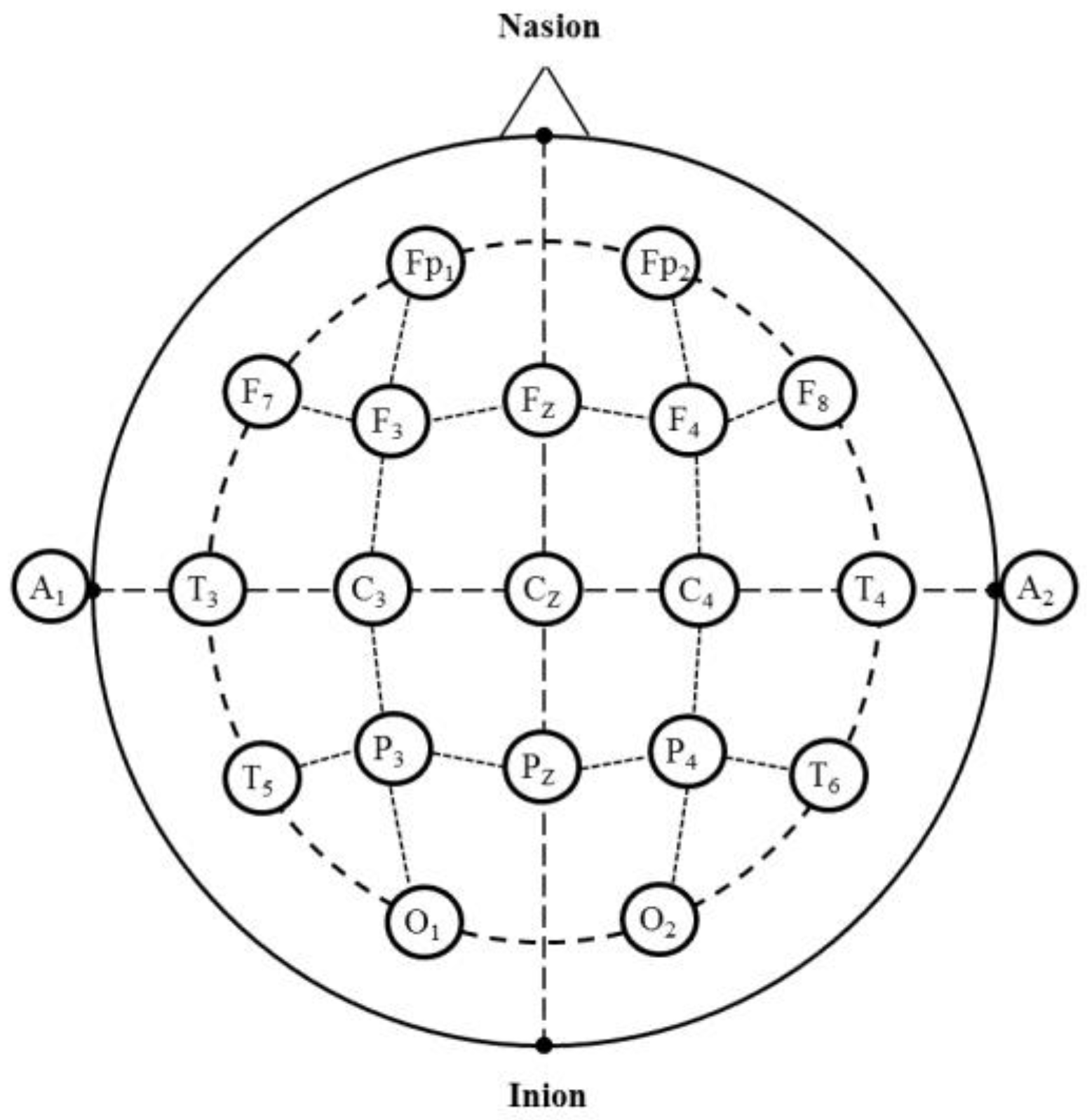
4.4. EEG Measurement
5. Effect of Inhalation of Fragrance on EEG Activity
6. Effect of Inhalation of Fragrance on Psychophysiological Activity
7. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Ali, B.; Al-Wabel, N.A.; Shams, S.; Ahamad, A.; Khan, S.A.; Anwar, F. Essential oils used in aromatherapy: A systemic review. Asian Pac. J. Trop. Biomed. 2015, 5, 601–611. [Google Scholar] [CrossRef]
- Kako, H.; Fukumoto, S.; Kobayashi, Y.; Yokogoshi, H. Effects of direct exposure of green odour components on dopamine release from rat brain striatal slices and PC12 cells. Brain Res. Bull. 2008, 75, 706–712. [Google Scholar] [CrossRef] [PubMed]
- Kiecolt–Glaser, J.K.; Graham, J.E.; Malarkey, W.B.; Porter, K.; Lemeshow, S.; Glaser, R. Olfactory influences on mood and autonomic, endocrine, and immune function. Psychoneuroendocrinology 2008, 33, 328–339. [Google Scholar] [CrossRef] [PubMed]
- Herz, R.S. Aromatherapy facts and fictions: A scientific analysis of olfactory effects on mood, physiology and behavior. Int. J. Neurosci. 2009, 119, 263–290. [Google Scholar] [CrossRef] [PubMed]
- Angelucci, F.L.; Silva, V.V.; Dal Pizzol, C.; Spir, L.G.; Praes, C.E.; Maibach, H. Physiological effect of olfactory stimuli inhalation in humans: An overview. Int. J. Cosmet. Sci. 2014, 36, 117–123. [Google Scholar] [CrossRef] [PubMed]
- Sell, C.S. The Chemistry of Fragrances—From Perfumer to Consumer, 2nd ed.; Quest International: Irvine, CA, USA, 2006. [Google Scholar]
- Kutlu, A.K.; Yilmaz, E.; Cecen, D. Effects of aroma inhalation on examination anxiety. Teach. Learn. Nurs. 2008, 3, 125–130. [Google Scholar] [CrossRef]
- Touhara, K.; Vosshall, L.B. Sensing odorants and pheromones with chemosensory receptors. Annu. Rev. Physiol. 2009, 71, 307–332. [Google Scholar] [CrossRef] [PubMed]
- Diego, M.A.; Jones, N.A.; Field, T.; Hernandez-Reif, M.; Schanberg, S.; Kuhn, C.; McAdam, V.; Galamaga, R.; Galamaga, M. Aromatherapy positively affects mood, EEG patterns of alertness and math computations. Int. J. Neurosci. 1998, 96, 217–224. [Google Scholar] [CrossRef] [PubMed]
- Field, T.; Diego, M.; Hernandez-Reif, M.; Cisneros, W.; Feijo, L.; Vera, Y.; Gil, K.; Grina, D.; Claire He, Q. Lavender fragrance cleansing gel effects on relaxation. Int. J. Neurosci. 2005, 115, 207–222. [Google Scholar] [CrossRef] [PubMed]
- Grabenhorst, F.; Rolls, E.; Margot, C. A hedonically complex odor mixture produces an attentional capture effect in brain. Neuroimage 2010, 55, 832–843. [Google Scholar] [CrossRef] [PubMed]
- Sayorwan, W.; Siripornpanich, V.; Piriyapunyaporn, T.; Hongratanaworakit, T.; Kotchabhakdi, N.; Ruangrungsi, N. The effects of lavender oil inhalation on emotional states, autonomic nervous system, and brain electrical activity. J. Med. Assoc. Thai. 2012, 95, 598–606. [Google Scholar] [PubMed]
- Schulz, H.; Jobert, M.; Hiibner, W.D. The quantitative EEG as a screening instrument to identify sedative effects of single doses of plant extracts in comparison with diazepam. Phytomedicine 1998, 5, 449–458. [Google Scholar] [CrossRef]
- Iijima, M.; Osawa, M.; Nishitani, N.; Iwata, M. Effects of incense on brain function: Evaluation using electroencephalograms and event–related potentials. Neuropsychobiology 2009, 59, 80–86. [Google Scholar] [CrossRef] [PubMed]
- Matsubara, E.; Fukagawa, M.; Okamoto, T.; Ohnuki, K.; Shimizu, K.; Kondo, R. The essential oil of Abies sibirica (Pinaceae) reduces arousal levels after visual display terminal work. Flavour Frag. J. 2011, 26, 204–210. [Google Scholar] [CrossRef]
- Skoric, M.K.; Ivan Adamec, I.; Jerbić, A.B.; Gabelić, T.; Hajnšek, S.; Habek, M. Electroencephalographic response to different odors in healthy individuals: A promising tool for objective assessment of olfactory disorders. Clin. EEG Neurosci. 2015, 46, 370–376. [Google Scholar] [CrossRef] [PubMed]
- Sowndhararajan, K.; Cho, H.; Yu, B.; Kim, S. Effect of olfactory stimulation of isomeric aroma compounds, (+)-limonene and terpinolene on human electroencephalographic activity. Eur. J. Integr. Med. 2015, 7, 561–566. [Google Scholar] [CrossRef]
- Achermann, P. EEG analysis applied to sleep. Epileptologie 2009, 26, 28–33. [Google Scholar]
- Ashour, M.; Wink, M.; Gershenzon, J. Biochemistry of Terpenoids: Monoterpenes, Sesquiterpenes and Diterpenes. In Annual Plant Reviews: Biochemistry of Plant Secondary Metabolism, 2nd ed.; Wink, M., Ed.; Wiley–Blackwell: Oxford, UK, 2010; Volume 40, pp. 258–303. [Google Scholar]
- Regnault–Roger, C.; Vincent, C.; Arnason, J.T. Essential oils in insect control: Low-risk products in a high-stakes world. Annu. Rev. Entomol. 2012, 57, 405–424. [Google Scholar] [CrossRef] [PubMed]
- Pavela, R. Essential oils for the development of eco–friendly mosquito larvicides: A review. Ind. Crop. Prod. 2015, 76, 174–187. [Google Scholar] [CrossRef]
- Bakkali, F.; Averbeck, S.; Averbeck, D.; Idaomar, M. Biological effects of essential oils—A review. Food Chem. Toxicol. 2008, 46, 446–475. [Google Scholar] [CrossRef] [PubMed]
- Llana-Ruiz-Cabello, M.; Pichardo, S.; Maisanaba, S.; Puerto, M.; Prieto, A.I.; Gutiérrez-Praena, D.; Jos, A.; Cameán, A.M. In vitro toxicological evaluation of essential oils and their main compounds used in active food packaging: A review. Food Chem. Toxicol. 2015, 81, 9–27. [Google Scholar] [CrossRef] [PubMed]
- Lange, C.; Kuch, B.; Metzger, J.W. Occurrence and fate of synthetic musk fragrances in a small German river. J. Hazard. Mater. 2015, 282, 34–40. [Google Scholar] [CrossRef] [PubMed]
- Feron, F.; Perry, C.; McGrath, J.J.; Mackay-Sim, A. New techniques for biopsy and culture of human olfactory epithelial neurons. Arch. Otolaryngol. Head Neck Surg. 1998, 124, 861–866. [Google Scholar] [CrossRef] [PubMed]
- Alan, M.S.; Royet, J.P. Structure and function of the olfactory system. In Olfaction and the Brain; Brewer, W.J., Castle, D., Pantelis, C., Eds.; Cambridge University Press: Cambridge, UK, 2006; pp. 3–27. [Google Scholar]
- Breer, K.R. Sense of smell: Recognition and transduction of olfactory signals. Biochem. Soc. Trans. 2003, 31, 113–116. [Google Scholar] [CrossRef] [PubMed]
- Strous, R.D.; Shoenfeld, Y. To smell the immune system: Olfaction, autoimmunity and brain involvement. Autoimmun. Rev. 2006, 6, 54–60. [Google Scholar] [CrossRef] [PubMed]
- Benarroch, E.E. Olfactory system: Functional organization and involvement in neurodegenerative disease. Neurology 2010, 75, 1104–1109. [Google Scholar] [CrossRef] [PubMed]
- Buck, L.; Axel, R. A novel multigene family may encode odorant receptors: A molecular basis for odor recognition. Cell 1991, 65, 175–187. [Google Scholar] [CrossRef]
- Haberly, L.B. Parallel-distributed processing in olfactory cortex: New insights from morphological and physiological analysis of neuronal circuitry. Chem. Senses 2001, 26, 551–576. [Google Scholar] [CrossRef] [PubMed]
- Dade, L.A.; Zatorre, R.J.; Jones-Gotman, M. Olfactory learning: Convergent findings from lesion and brain imaging studies in humans. Brain 2002, 125, 86–101. [Google Scholar] [CrossRef] [PubMed]
- MacDonald, D.B. Electroencephalography: Basic Principles and Applications. In International Encyclopedia of the Social & Behavioral Sciences, 2nd ed.; Wright, J.D., Ed.; Elsevier: Amsterdam, The Netherlands, 2015; pp. 353–363. [Google Scholar]
- Lorig, T.S. Human EEG and odor response. Prog. Neurobiol. 1989, 33, 387–398. [Google Scholar] [CrossRef]
- Pinto, R.J.C.; Xavier, I.P.P.P.; Calado, M.R.A.; Mariano, S.J.P.S. Analysis of the Human Reaction to Odors Using Electroencephalography Responses. Available online: http://www.iaeng.org/publication/WCE2014/WCE2014_pp243-247.pdf (accessed on 3 June 2016).
- Jackson, A.F.; Bolger, D.J. The neurophysiological bases of EEG and EEG measurement: A review for the rest of us. Psychophysiology 2014, 51, 1061–1071. [Google Scholar] [CrossRef] [PubMed]
- Desai, R.; Tailor, A.; Bhat, T. Effects of yoga on brain waves and structural activation: A review. Complement. Ther. Clin. Pract. 2015, 21, 112–118. [Google Scholar] [CrossRef] [PubMed]
- Buzsaki, G.; Watson, B.O. Brain rhythms and neural syntax: Implications for efficient coding of cognitive content and neuropsychiatric disease. Dialogues Clin. Neurosci. 2012, 14, 345–367. [Google Scholar] [PubMed]
- Halasz, P.; Bodizs, R.; Parrino, L.; Terzano, M. Two features of sleep slow waves: Homeostatic and reactive aspects—From long term to instant sleep homeostasis. Sleep Med. 2014, 15, 1184–1195. [Google Scholar] [CrossRef] [PubMed]
- Assenza, G.; Pellegrino, G.; Tombini, M.; Di Pino, G.; Di Lazzaro, V. Wakefulness delta waves increase after cortical plasticity induction. Clin. Neurophysiol. 2015, 126, 1221–1227. [Google Scholar] [CrossRef] [PubMed]
- Buzsaki, G. Theta rhythm of navigation: Link between path integration and landmark navigation, episodic and semantic memory. Hippocampus 2005, 15, 827–840. [Google Scholar] [CrossRef] [PubMed]
- Lisman, J.E.; Idiart, M.A. Storage of 7 +/−2 short–term memories in oscillatory subcycles. Science 1995, 267, 1512–1515. [Google Scholar] [CrossRef] [PubMed]
- Palva, S.; Palva, J.M. New vistas for alpha-frequency band oscillations. Trends Neurosci. 2007, 30, 150–158. [Google Scholar] [CrossRef] [PubMed]
- Klimesch, W.; Schmike, H.; Pfurtscheller, G. Alpha frequency cognitive load and memory performance. Brain Topogr. 1993, 5, 241–251. [Google Scholar] [CrossRef] [PubMed]
- Basar, E. A review of alpha activity in integrative brain function: Fundamental physiology, sensory coding, cognition and pathology. Int. J. Psychophysiol. 2012, 86, 1–24. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.C.; Lee, M.H.; Jang, C.; Kwon, J.W.; Park, J.W. The effect of alpha rhythm sleep on EEG activity and individuals’ attention. J. Phys. Ther. Sci. 2013, 25, 1515–1518. [Google Scholar] [CrossRef] [PubMed]
- Neuper, C.; Pfurtscheller, G. Event-related dynamics of cortical rhythms: Frequency-specific features and functional correlates. Int. J. Psychophysiol. 2001, 43, 41–58. [Google Scholar] [CrossRef]
- Lee, B.G.; Lee, B.L.; Chung, W.Y. Mobile healthcare for automatic driving sleep-onset detection using wavelet-based EEG and respiration signals. Sensors 2014, 14, 17915–17936. [Google Scholar] [CrossRef] [PubMed]
- Skinner, J.E.; Molnar, M.; Kowalik, Z.J. The role of the thalamic reticular neurons in alpha- and gamma-oscillations in neocortex: A mechanism for selective perception and stimulus binding. Acta Neurobiol. Exp. 2000, 60, 123–142. [Google Scholar] [PubMed]
- Wang, J.; Barstein, J.; Ethridge, L.E.; Mosconi, M.W.; Takarae, Y.; Sweeney, J.A. Resting state EEG abnormalities in autism spectrum disorders. J. Neurodev. Disord. 2013, 5, 24. [Google Scholar] [CrossRef] [PubMed]
- Schoenemann, P.T. Evolution of the size and functional areas of the human brain. Annu. Rev. Anthropol. 2006, 35, 379–406. [Google Scholar] [CrossRef]
- Bush, E.C.; Allman, J.M. The scaling of frontal cortex in primates and carnivores. Proc. Natl. Acad. Sci. USA 2004, 101, 3962–3966. [Google Scholar] [CrossRef] [PubMed]
- Siddiqui, S.V.; Chatterjee, U.; Kumar, D.; Siddiqui, A.; Goyal, N. Neuropsychology of prefrontal cortex. Indian J. Psychiatry 2008, 50, 202–208. [Google Scholar] [PubMed]
- Acuna, B.D.; Eliassen, J.C.; Donoghue, J.P.; Sanes, J.N. Frontal and Parietal Lobe Activation during transitive inference in humans. Cereb. Cortex 2002, 12, 1312–1321. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, C.L.R.; Flindall, J.W. Parietal Lobe. In International Encyclopedia of the Social & Behavioral Sciences, 2nd ed.; Wright, J.D., Ed.; Elsevier: Oxford, UK, 2015; pp. 506–510. [Google Scholar]
- Mai, J.G.; Paxinos, G.; Voss, T. Atlas of the Human Brain, 3rd ed.; Elsevier: Amsterdam, The Netherlands, 2008. [Google Scholar]
- Kiernan, J.A. Anatomy of the temporal lobe. Epilepsy Res. Treat. 2012, 2012, 176157. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Xu, C.; Cao, X.; Gao, Q.; Wang, Y.; Wang, Y.; Peng, J.; Zhang, K. Abnormal activation of the occipital lobes during emotion picture processing in major depressive disorder patients. Neural Regener. Res. 2013, 8, 1693–1701. [Google Scholar]
- Martin, G.N. Human electroencephalographic (EEG) response to olfactory stimulation: Two experiments using the aroma of food. Int. J. Psychophysiol. 1998, 30, 287–302. [Google Scholar] [CrossRef]
- Sowndhararajan, K.; Cho, H.; Yu, B.; Song, J.; Kim, S. Effect of inhalation of essential oil from Inula helenium L. root on electroencephalographic (EEG) activity of human. Eur. J. Integr. Med. 2016. [Google Scholar] [CrossRef]
- Masago, R.; Matsuda, T.; Kikuchi, Y.; Miyazaki, Y.; Iwanaga, K.; Harada, H.; Katsuura, T. Effects of inhalation of essential oils on EEG activity and sensory evaluation. J. Physiol. Anthropol. 2000, 19, 35–42. [Google Scholar] [CrossRef]
- Fernandez, M.; Hernandez-Reif, M.; Field, T.; Diego, M.; Sanders, C.; Roca, A. EEG during lavender and rosemary exposure in infants of depressed and non-depressed mothers. Infant Behav. Dev. 2004, 27, 91–100. [Google Scholar] [CrossRef]
- Watanabe, S.; Hara, K.; Ohta, K.; Iino, H.; Miyajima, M.; Matsuda, A.; Hara, M.; Maehara, T.; Matsuura, M.; Matsushima, E. Aroma helps to preserve information processing resources of the brain in healthy subjects but not in temporal lobe epilepsy. Seizure 2013, 22, 59–63. [Google Scholar] [CrossRef] [PubMed]
- Lee, I. Effects of inhalation of relaxing essential oils on electroencephalogram activity. Int. J. New Technol. Res. 2016, 2, 37–43. [Google Scholar]
- Tyner, F.S.; Knott, J.R.; Mayer, W.B. Fundamentals of EEG Technology, Basic Concepts and Methods; Raven Press: New York, NY, USA, 1983; Volume 1, pp. 136–145. [Google Scholar]
- Lopes da Silva, F. EEG: Origin and measurement. In EEG–fMRI: Physiological Basis, Technique, and Applications; Mulert, C., Lemieux, L., Eds.; Springer: New York, NY, USA, 2010; pp. 19–38. [Google Scholar]
- Lorig, T.S. On the similarity of odor and language perception. Neurosci. Biobehav. Rev. 1999, 23, 391–398. [Google Scholar] [CrossRef]
- Van Toller, S. The application of EEG measurements to the study of sensory responses to odours. Trends Food Sci. Technol. 1991, 2, 173–175. [Google Scholar] [CrossRef]
- Martin, G.N. Olfactory remediation: Current evidence and possible applications. Soc. Sci. Med. 1996, 43, 63–70. [Google Scholar] [CrossRef]
- Van Toller, S.; Behan, J.; Howells, P.; Kendal-Reed, M.; Richardson, A. An analysis of spontaneous human cortical EEG activity to odours. Chem. Senses 1993, 18, 1–16. [Google Scholar] [CrossRef]
- Min, B.C.; Jin, S.H.; Kang, I.H.; Lee, D.H.; Kang, J.K.; Lee, S.T.; Sakamoto, K. Analysis of mutual information content for EEG responses to odor stimulation for subjects classified by occupation. Chem. Senses 2003, 28, 741–749. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Field, T.; Diego, M.; Hernandez-Reif, M. Tai chi/yoga effects on anxiety, heart rate, EEG and math computations. Complement. Ther. Clin. Pract. 2010, 16, 235–238. [Google Scholar] [CrossRef] [PubMed]
- Freeman, W.J. Mechanism and significance of global coherence in scalp EEG. Curr. Opin. Neurobiol. 2015, 31, 199–205. [Google Scholar] [CrossRef] [PubMed]
- Lorig, T.S.; Huffman, E.; DeMartino, A.; DeMarco, J. The effects of low concentration odors on EEG activity and behavior. J. Psychophysiol. 1991, 5, 69–77. [Google Scholar]
- Seppalainen, A.M.; Laine, A.; Salmi, T.; Verkkala, E.; Riihimäki, V.; Luukkonen, R. Electroencephalographic findings during experimental human exposure to m-xylene. Arch. Environ. Health 1991, 46, 16–24. [Google Scholar] [CrossRef] [PubMed]
- Brauchli, P.; Ruegg, P.B.; Etzweiler, F.; Zeier, H. Electrocortical and autonomic alteration by administration of a pleasant and an unpleasant odor. Chem. Senses 1995, 20, 505–515. [Google Scholar] [CrossRef] [PubMed]
- Masumoto, Y.; Morinushi, T.; Kawasaki, H.; Takigawa, M. Spectral analysis of changes in electroencephalographic activity after the chewing of gum. Psychiatry Clin. Neurosci. 1998, 52, 587–592. [Google Scholar] [CrossRef] [PubMed]
- Konagai, C.; Hamada, M.; Nguyen, V.C.; Koga, Y. The effect of the aroma from soybeans after heating on EEG. Int. Congr. Ser. 2002, 1232, 119–123. [Google Scholar] [CrossRef]
- Iijima, M.; Nio, E.; Nashimoto, E.; Iwata, M. Effects of aroma on the autonomic nervous system and brain activity under stress conditions. Auton. Neurosci. 2007, 135, 97–98. [Google Scholar] [CrossRef]
- Klemm, W.R.; Lutes, S.D.; Hendrix, D.V.; Warrenberg, S. Topographical EEG maps of human responses to odors. Chem. Senses 1992, 17, 347–361. [Google Scholar] [CrossRef]
- Boha, R.; Molnar, M.; Gaal, Z.A.; Czigler, B.; Rona, K.; Kass, K.; Klausz, G. The acute effect of low–dose alcohol on working memory during mental arithmetic: I. Behavioral measures and EEG theta band spectral characteristics. Int. J. Psychophysiol. 2009, 73, 133–137. [Google Scholar] [CrossRef] [PubMed]
- Kawakami, M.; Aoki, S.; Ohkubo, T. A study of “fragrance” on working environment characteristics in VDT work activities. Int. J. Prod. Econ. 1999, 60–61, 575–581. [Google Scholar] [CrossRef]
- Yagyu, T.; Kondakor, I.; Kochi, K.; Koenig, T.; Lehmann, D.; Kinoshita, T.; Hirota, T.; Yagyu, T. Smell and taste of chewing gum affect frequency domain EEG source localizations. Int. J. Neurosci. 1998, 93, 205–216. [Google Scholar] [CrossRef] [PubMed]
- Morinushi, T.; Masumoto, Y.; Kawasaki, H.; Takigawa, M. Effect on electroencephalogram of chewing flavored gum. Psychiatry Clin. Neurosci. 2000, 54, 645–651. [Google Scholar] [CrossRef] [PubMed]
- Weber, B.; Mosandl, A. Sterioisomeric flavor compounds LXXV: Synthesis and structure–function relationship of 3–methylthiobutanol enantiomers. Z. Lebensm. Unters. F. A 1997, 204, 194–197. [Google Scholar] [CrossRef]
- Laska, M.; Teubner, P. Olfactory discrimination ability of human subjects for ten pairs of enantiomers. Chem. Senses 1999, 24, 161–170. [Google Scholar] [CrossRef] [PubMed]
- Brenna, E.; Fuganti, C.; Serra, S. Enantio selective perception of chiral odorants. Tetrahedron Asymmetry 2003, 14, 1–42. [Google Scholar] [CrossRef]
- Sugawara, Y.; Haraa, C.; Tamuraa, K.; Fujiia, T.; Nakamuraa, K.; Masujimab, T.; Aoki, T. Sedative effect on humans of inhalation of essential oil of linalool: Sensory evaluation and physiological measurements using optically active linalools. Anal. Chim. Acta 1998, 365, 293–299. [Google Scholar] [CrossRef]
- Sugawara, Y.; Hara, C.; Aoki, T.; Sugimoto, N.; Masujima, T. Odor distinctiveness between enantiomers of linalool: Difference in perception and responses elicited by sensory test and forehead surface potential wave measurement. Chem. Senses 2000, 25, 77–84. [Google Scholar] [CrossRef] [PubMed]
- Peng, S.M.; Koo, M.; Yu, Z.R. Effects of music and essential oil inhalation on cardiac autonomic balance in healthy individuals. J. Altern. Complement. Med. 2009, 15, 53–57. [Google Scholar] [CrossRef] [PubMed]
- Owen, C.M.; Patterson, J. Odour liking physiological indices: A correlation of sensory and electrophysiological responses to odour. Food Qual. Prefer. 2002, 13, 307–316. [Google Scholar] [CrossRef]
- Watanuki, S.; Kim, Y.K. Physiological responses induced by pleasant stimuli. J. Physiol. Anthropol. Appl. Hum. Sci. 2005, 24, 135–138. [Google Scholar] [CrossRef] [PubMed]
- Patterson, J.; Owen, C.M.; Frank, D.; Smith, R.; Cadusch, P. Flavour sensory qualities and consumer perceptions—A comparison of sensory and brain activity responses to flavour components in different populations. Int. J. Food Sci. Technol. 2004, 39, 481–490. [Google Scholar] [CrossRef]
- Woronuk, G.; Demissie, Z.; Rheault, M.; Mahmoud, S. Biosynthesis and therapeutic properties of lavandula essential oil constituents. Planta Med. 2011, 77, 7–15. [Google Scholar] [CrossRef] [PubMed]
- Koulivand, P.H.; Ghadiri, M.K.; Gorji, A. Lavender and the nervous system. Evid. Based Complement. Altern. Med. 2013, 2013, 681304. [Google Scholar] [CrossRef] [PubMed]
- Setzer, W.N. Essential oils and anxiolytic aromatherapy. Nat. Prod. Commun. 2009, 4, 1305–1316. [Google Scholar] [PubMed]
- Sanders, C.; Diego, M.; Fernandez, M.; Field, T.; Hernandez-Reil, M.; Roca, A. EEG asymmetry responses to lavender and rosemary aromas in adults and infants. Int. J. Neurosci. 2002, 112, 305–320. [Google Scholar] [CrossRef]
- Jung, H.; Choi, H. Effects of Lavandula angustifolia aroma on electroencephalograms in female adults with sleep disorders. J. Life Sci. 2012, 22, 192–199. [Google Scholar] [CrossRef]
- Cho, H.; Yu, B.; Sowndhararajan, K.; Jung, J.W.; Jhoo, J.W.; Kim, S. Effect of essential oil from San-Jo-In (Zizyphus jujuba Mill. seeds) on human electroencephalographic activity. J. Life Sci. 2013, 23, 1170–1176. [Google Scholar] [CrossRef]
- Cho, H.; Sowndhararajan, K.; Jung, J.W.; Jhoo, J.W.; Kim, S. Fragrance chemicals in the essential oil of Mentha arvensis reduce the level of mental stress. J. Life Sci. 2013, 23, 933–940. [Google Scholar] [CrossRef]
- Cho, H.; Sowndhararajan, K.; Jung, J.W.; Jhoo, J.W.; Kim, S. Fragrant Chemicals in the Supercritical Carbon Dioxide Extract of Magnolia Kobus DC. Flower Buds Increase the Concentration State of Brain Function. J. Essent. Oil Bear. Plants 2015, 18, 1059–1069. [Google Scholar] [CrossRef]
- Sayowan, W.; Siripornpanich, V.; Hongratanaworakit, T.; Kotchabhakdi, N.; Ruangrungsi, N. The effects of jasmine oil inhalation on brain wave activities and emotions. J. Health Res. 2013, 27, 73–77. [Google Scholar]
- Yoto, A.; Moriyama, T.; Yokogoshi, H.; Nakamura, Y.; Katsuno, T.; Nakayama, T. Effect of smelling green tea rich in aroma components on EEG activity and memory task performance. Int. J. Affect. Eng. 2014, 13, 227–233. [Google Scholar] [CrossRef]
- Iannilli, E.; Sorokowska, A.; Zhigang, Z.; Hähner, A.; Warr, J.; Hummel, T. Source localization of event-related brain activity elicited by food and nonfood odors. Neuroscience 2015, 289, 99–105. [Google Scholar] [CrossRef] [PubMed]
- Wada, Y.; Takizawa, Y.; Jiang, Z.Y.; Yamaguchi, N. Gender differences in quantitative EEG at rest and during photic stimulation in normal young adults. Clin. Electroencephalogr. 1994, 25, 81–85. [Google Scholar] [CrossRef] [PubMed]
- Jausovec, N.; Jausovec, K. Resting brain activity: Differences between genders. Neuropsychologia 2010, 48, 3918–3928. [Google Scholar] [CrossRef] [PubMed]
- Corsi-Cabrera, M.; Ramos, J.; Guevara, M.A.; Arce, C.; Gutierrez, S. Gender differences in the EEG during cognitive activity. Int. J. Neurosci. 1993, 72, 257–264. [Google Scholar] [CrossRef] [PubMed]
- Guntekin, S.; Basar, E. Brain oscillations are highly influenced by gender differences. Int. J. Psychophysiol. 2007, 65, 294–299. [Google Scholar] [CrossRef] [PubMed]
- Hur, M.H.; Lee, M.S.; Kim, C.; Ernst, E. Aromatherapy for treatment of hypertension: A systematic review. J. Eval. Clin. Pract. 2012, 18, 37–41. [Google Scholar] [CrossRef] [PubMed]
- Roudnitzky, N.; Bult, J.H.; de Wijk, R.A.; Reden, J.; Schuster, B.; Hummel, T. Investigation of interactions between texture and ortho- and retronasal olfactory stimuli using psychophysical and electrophysiological approaches. Behav. Brain Res. 2011, 216, 109–115. [Google Scholar] [CrossRef] [PubMed]
- Hiessl, A.K.; Skrandies, W. Evaluation of multisensory stimuli—Dimensions of meaning and electrical brain activity. Neuropsychologia 2013, 51, 1330–1335. [Google Scholar] [CrossRef] [PubMed]
- Sugawara, Y.; Shigetho, A.; Yoneda, M.; Tuchiya, T.; Matumura, T.; Hirano, M. Relationship between mood change, odour and its physiological effects in humans while inhaling the fragrances of essential oils as well as linalool and its enantiomers. Molecules 2013, 18, 3312–3338. [Google Scholar] [CrossRef] [PubMed]
- Morris, N.; Birtwistle, S.; Toms, M. Anxiety reduction by aromatherapy: Anxiolytic effects of inhalation of geranium and rosemary. Int. J. Aromather. 1995, 7, 33–39. [Google Scholar] [CrossRef]
- Sugawara, Y.; Hino, Y.; Kawasaki, M.; Hara, C.; Tamura, K.; Sugimoto, N.; Yamanishi, Y.; Miyauchi, M.; Masujima, T.; Aoki, T. Alteration of perceived fragrance of essential oils in relation to type of work: A simple screening test for efficacy of aroma. Chem. Senses 1999, 24, 415–421. [Google Scholar] [CrossRef] [PubMed]
- Lehrner, J.; Eckersberger, C.; Walla, P.; Potsch, G.; Deecke, L. Ambient odor of orange in a dental office reduces anxiety and improves mood in female patients. Physiol. Behav. 2000, 71, 83–86. [Google Scholar] [CrossRef]
- Nagai, M.; Wada, M.; Usui, N.; Tanaka, A.; Hasebe, Y. Pleasant odors attenuate the blood pressure increase during rhythmic handgrip in humans. Neurosci. Lett. 2000, 289, 227–229. [Google Scholar] [CrossRef]
- Haze, S.; Sakai, K.; Gozu, Y. Effects of fragrance inhalation on sympathetic activity in normal adults. Jpn. J. Pharmacol. 2002, 90, 247–253. [Google Scholar] [CrossRef] [PubMed]
- Bensafi, M.; Rouby, C.; Farget, V.; Bertrand, B.; Vigouroux, M.; Holley, A. Autonomic nervous system responses to odours: The role of pleasantness and arousal. Chem. Senses 2002, 27, 703–709. [Google Scholar] [CrossRef] [PubMed]
- Ilmberger, J.; Heuberger, E.; Mahrhofer, C.; Dessovic, H.; Kowarik, D.; Buchbauer, G. The influence of essential oils on human attention 1: Alertness. Chem. Senses 2001, 26, 239–245. [Google Scholar] [CrossRef] [PubMed]
- Heuberger, E.; Hongratanaworakit, T.; Bohm, C.; Weber, R.; Buchbauer, G. Effects of chiral fragrances on human autonomic nervous system parameters and self–evaluation. Chem. Senses 2001, 26, 281–292. [Google Scholar] [CrossRef] [PubMed]
- Cavanagh, H.M.A.; Wilkinson, J.M. Biological activities of lavender essential oil. Phytother. Res. 2002, 16, 301–308. [Google Scholar] [CrossRef] [PubMed]
- Motomura, N.; Sakurai, A.; Yotsuya, Y. Reduction of mental stress with lavender odorant. Percept. Motor Skill. 2001, 93, 713–718. [Google Scholar] [CrossRef] [PubMed]
- Holmes, C.; Hopkins, V.; Hensford, C.; MacLaughlin, V.; Wilkinson, D.; Rosenvinge, H. Lavender oil as a treatment for agitated behaviour in severe dementia: A placebo controlled study. Int. J. Geriatr. Psychiatry 2002, 17, 305–308. [Google Scholar] [CrossRef] [PubMed]
- Moss, M.; Cook, J.; Wesnes, K.; Duckett, P. Aromas of rosemary and lavender essential oils differentially affect cognition and mood in healthy adults. Int. J. Neurosci. 2003, 113, 15–38. [Google Scholar] [CrossRef] [PubMed]
- Burnett, K.M.; Solterbeck, L.A.; Strapp, C.M. Scent and mood state following an anxiety provoking task. Psychol. Rep. 2004, 95, 707–722. [Google Scholar] [CrossRef] [PubMed]
- Campenni, C.E.; Crawley, E.J.; Meier, M.E. Role of suggestion in odor induced mood change. Psychol. Rep. 2004, 94, 1127–1136. [Google Scholar] [CrossRef] [PubMed]
- Gedney, J.J.; Glover, T.L.; Fillingim, R.B. Sensory and affective pain discrimination after inhalation of essential oils. Psychosom. Med. 2004, 66, 599–606. [Google Scholar] [CrossRef] [PubMed]
- Goel, N.; Kim, H.; Lau, R.P. An olfactory stimulus modifies nighttime sleep in young men and women. Chronobiol. Int. 2005, 22, 889–904. [Google Scholar] [CrossRef] [PubMed]
- Lehrner, J.; Marwinski, G.; Lehr, S.; Johren, P.; Deecke, L. Ambient odors of orange and lavender reduce anxiety and improve mood in a dental office. Physiol. Behav. 2005, 86, 92–95. [Google Scholar] [CrossRef] [PubMed]
- Field, T.; Field, T.; Cullen, C.; Largie, S.; Diego, M.; Schanberg, S.; Kuhn, C. Lavender bath oil reduces stress and crying and enhances sleep in very young infants. Early Hum. Dev. 2008, 84, 399–401. [Google Scholar] [CrossRef] [PubMed]
- Howard, S.; Hughes, B.M. Expectancies, not aroma, explain impact of lavender aromatherapy on psychophysiological indices of relaxation in young healthy women. Brit. J. Health Psychol. 2008, 13, 603–617. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Kim, H.J.; Yeo, J.S.; Hong, S.J.; Lee, J.M.; Jeon, Y. The effect of lavender oil on stress, bispectral index values, and needle insertion pain in volunteers. J. Altern. Complement. Med. 2011, 17, 823–826. [Google Scholar] [CrossRef] [PubMed]
- Sakamoto, R.; Minoura, K.; Usui, A.; Ishizuka, Y.; Kanba, S. Effectiveness of aroma on work efficiency: Lavender aroma during recesses prevents deterioration of work performance. Chem. Senses 2005, 30, 683–691. [Google Scholar] [CrossRef] [PubMed]
- Rho, K.H.; Han, S.H.; Kim, K.S.; Lee, M.S. Effects of aromatherapy massage on anxiety and self–esteem in korean elderly women: A pilot study. Int. J. Neurosci. 2006, 116, 1447–1455. [Google Scholar] [CrossRef] [PubMed]
- Raudenbush, B.; Corley, N.; Eppich, W. Enhancing athletic performance through administration of peppermint odor. J. Sport Exerc. Psychol. 2001, 23, 156–160. [Google Scholar] [CrossRef]
- Raudenbush, B.; Meyer, B.; Eppich, B. The effects of odors on objective and subjective measures of athletic performance. Int. Sports J. 2002, 6, 1–15. [Google Scholar]
- Goel, N.; Lau, R.P. Sleep changes vary by odor perception in young adults. Biol. Psychol. 2006, 71, 341–349. [Google Scholar] [CrossRef] [PubMed]
- Meamarbashi, A.; Rajabi, A. The effects of peppermint on exercise performance. J. Int. Soc. Sports Nutr. 2013, 10, 15. [Google Scholar] [CrossRef] [PubMed]
- Ho, C.; Spence, C. Olfactory facilitation of dual-task performance. Neurosci. Lett. 2005, 389, 35–40. [Google Scholar] [CrossRef] [PubMed]
- Norrish, M.I.K.; Dwyer, K.L. Preliminary investigation of the effect of peppermint oil on an objective measure of daytime sleepiness. Int. J. Psychophysiol. 2005, 55, 291–298. [Google Scholar] [CrossRef] [PubMed]
- Moss, M.; Hewitt, S.; Moss, L.; Wesnes, K. Modulation of cognitive performance and mood by aromas of peppermint and ylang-ylang. Int. J. Neurosci. 2008, 118, 59–77. [Google Scholar] [CrossRef] [PubMed]
- Hongratanaworakit, T.; Buchbauer, G. Relaxing effect of ylang-ylang oil on humans after transdermal absorption. Phytother. Res. 2006, 20, 758–763. [Google Scholar] [CrossRef] [PubMed]
- Moss, M.; Howarth, R.; Wilkinson, L.; Wesnes, K. Expectancy and the aroma of Roman chamomile influence mood and cognition in healthy volunteers. Int. J. Aromather. 2006, 16, 63–73. [Google Scholar] [CrossRef]
- Kohler, M.; Pavy, A.; van den Heuvel, C. The effects of chewing versus caffeine on alertness, cognitive performance and cardiac autonomic activity during sleep deprivation. J. Sleep Res. 2006, 15, 358–368. [Google Scholar] [CrossRef] [PubMed]
- Hummel, T.; Heilmann, S. Olfactory event–related potentials in response to ortho- and retronasal stimulation with odors related or unrelated to foods. Int. Dairy J. 2008, 18, 874–878. [Google Scholar] [CrossRef]
- Yamagishi, R.; Yokomaku, A.; Omoto, F.; Misao, K.; Takada, K.; Yoshimatsu, S.; Abe, A.; Hayashi, M. Sleep-improving effects of the aromatic compound heliotropin. Sleep Biol. Rhythms 2010, 8, 254–260. [Google Scholar] [CrossRef]
- Moss, M.; Oliver, L. Plasma 1,8-cineole correlates with cognitive performance following exposure to rosemary essential oil aroma. Ther. Adv. Psychopharmacol. 2012, 2, 103–113. [Google Scholar] [CrossRef] [PubMed]
- Jun, Y.S.; Kang, P.; Min, S.S.; Lee, J.M.; Kim, H.K.; Seol, G.H. Effect of eucalyptus oil inhalation on pain and inflammatory responses after total knee replacement: A randomized clinical trial. Evid. Based Complement. Altern. Med. 2013, 2013, 502727. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.H.; Lin, T.H.; Chang, K.M. The physical effects of aromatherapy in alleviating work-related stress on elementary school teachers in Taiwan. Evid. Based Complement. Altern. Med. 2013, 2013, 853809. [Google Scholar] [CrossRef] [PubMed]
- Sugawara, Y.; Shigetho, A.; Yoneda, M.; Tuchiya, T.; Yamada, H.; Matumura, T.; Hirano, M. Versatile psychophysiological potencies of essential oils, when seen as a function of behavioral task assigned to the participants after inhalation. Inter. J. Soc. Sci. Stud. 2015, 3, 94–113. [Google Scholar] [CrossRef] [Green Version]
- Pastena, L.; Formaggio, E.; Storti, S.F.; Faralli, F.; Melucci, M.; Gagliardi, R.; Ricciardi, L.; Ruffino, G. Tracking EEG changes during the exposure to hyperbaric oxygen. Clin. Neurophysiol. 2015, 126, 339–347. [Google Scholar] [CrossRef] [PubMed]
- Kuroda, K.; Inoue, N.; Ito, Y.; Kubota, K.; Sugimoto, A.; Kakuda, T.; Fushiki, T. Sedative effects of the jasmine tea odor and (R)-(−) linalool one of its major odor components, on autonomic nerve activity and mood states. Eur. J. Appl. Physiol. 2005, 95, 107–114. [Google Scholar] [CrossRef] [PubMed]
| Plants Name | Botanical Name | Major Components |
|---|---|---|
| Bergamot | Citrus bergamia | limonene, linalool, linalyl acetate |
| Caraway | Carum carvi | carvone, limonene |
| Chamomile | Matricaria chamomilla | α-bisabolol, bisabolol oxide B, (E)-β-farnesene, α-bisabolone oxide |
| Cinnamon | Cinnamomum zeylanicum | cinnamaldehyde, cinnamyl acetate |
| Cornmint | Mentha arvensis | menthol, menthone, isomenthone, menthyl acetate |
| Eucalyptus | Eucalyptus sps. | 1,8-cineole (eucalyptol), limonene, aromadendrene |
| Geranium | Pelargonium graveolens | citronellol, geraniol, citronellyl formate, linalool |
| Jasmine | Jasminum sps. | benzyl alcohol, linalool, benzyl acetate, jasmone, geraniol |
| Juniper | Juniperus sps. | bornyl acetate, sabinene, α-pinene, limonene |
| Lavender | Lavandula angustifolia | geraniol, linalool, linalyl acetate, β-caryophyllene |
| Lemon | Citrus limon | limonene, β-pinene, γ-terpinene, p-cymene |
| Lemongrass | Cymbopogon citratus | citral (geranial), neral, myrcene |
| Oregano | Origanum vulgare | carvacrol, thymol, cymene |
| Palmarosa | Cymbopogon martinii | geraniol, geranyl acetate, linalool |
| Peppermint | Mentha piperita | menthol, menthone, 1,8-cineole, menthofuran |
| Pine | Pinus sps. | α-humulene, caryophyllene, β-pinene, β-cadinene |
| Rose | Rosa damascena | citronellol, geraniol, β-pinene, rose oxide |
| Rosemary | Rosmarinus officinalis | camphor, 1,8-cineole, α-pinene, borneol, camphene, β-phellandrene |
| Sandalwood | Santalum album | α-santalol, β-santalol, β-curcumen-12-ol |
| Spearmint | Mentha spicata | carvone, 1,8-cineole, limonene |
| Sweet basil | Ocimum basilicum | linalool, α-cadinol, α-bergamotene, γ-cadinene |
| Thyme | Thymus vulgaris | thymol, carvacrol, terpinene, cymene |
| Ylang-ylang | Cananga odorata | geranyl acetate, benzyl benzoate, eugenol, germacrene-d, geraniol |
| S. No. | Odorant Materials | EEG Wave Changes | Brain Functions | Reference |
|---|---|---|---|---|
| 1. | Galaxolide | Alpha decreased. | Odors produce divided attention even when undetected. | [74] |
| 2. | m-Xylene | Alpha increased. | Stimulating and excitatory effects. | [75] |
| 3. | Birch tar, galbanum, heliotropine, jasmine, lavender, lemon and peppermint | Increased theta for birch tar, jasmine, lavender and lemon. | Subjects differed in their subjective responses to the odors. | [80] |
| 4. | 5-α-Androstan-3-one, bangalol, white sapphire, indole, linalyl acetate, eucalyptus oil and ammonia. | Alpha increased. | From more anterior electrodes—related to psychometric responses. | [70] |
| 5. | Phenylethyl alcohol and valeric acid | Valeric acid—alpha 2 increased. | Unpleasant odor leads to a cortical deactivation. | [76] |
| 6. | Lavender and rosemary | Lavender—beta increased. Rosemary—frontal alpha and beta decreased. | Lavender—increased drowsiness. Rosemary—increased alertness. | [9] |
| 7. | Synthetic odors—almond, chocolate, spearmint, strawberry, vegetable, garlic, onion and cumin Odors of real foods—chocolate, baked beans and rotting pork | Chocolate odor—less theta activity. | Reduced level of attention. | [59] |
| 8. | Chewing of marketed gum | Alpha power increased. | Arousal psychosomatic responses. | [77] |
| 9. | Valeriana off, Lavandula off, Passiflora incarnata, Piper methysticum, Melissa off, Eschscbolzia californica, Hypericum perforatum and Ginkgo biloba | Valerian extract—delta and theta activity increased and beta activity decreased. | Self-rated tiredness increased under some of the plant extracts. | [13] |
| 10. | (R)-(−)-, (S)-(+)- and (RS)-(±)-forms of linalools | (RS)-(±)-linalool—greater decrease of the beta wave after work than before work. | (RS)-(±)-linalool and (R)-(−)-linalool -favorable impression. (S)-(+)-linalool—unfavorable impression. | [88] |
| 11. | Chewing regular gum or gum base without flavor | Alpha-2 and beta-2 increased for regular gum and decreased for gum base. | Activates different brain neuronal populations. | [83] |
| 12. | Sedative effects—lemon, lavender and sandalwood Awakening effects—jasmine, ylang-ylang, rose and peppermint | Awakening fragrances—decreased alpha and beta activities. | Sedative fragrances—improvement in productivity. Awakening fragrances—effect in mitigating the workload. | [82] |
| 13. | Lavender, chamomile, sandalwood and eugenol | Alpha 1 decreased at parietal and posterior temporal regions. | Subjects felt comfortable. | [61] |
| 14. | Chewing gum with and without flavor and flavored aromatic oil | Chewing gum with flavor and inhale aromatic oil increase alpha and beta waves. | Induce concentration with a harmonious high arousal state in brain function. | [84] |
| 15. | Enantiomers of linalools | (R)-(−)-linalool—beta decreased after hearing environmental sound. Mental work—beta increased. | Odor perception and responses—chiral dependence and also with task dependence. | [89] |
| 16. | Aroma of soybeans heated to various temperatures | Alpha wave increased—heated after immersion in fructose–glycine solution. | Amino-carbonyl reaction aroma products increase brain alpha waves. | [78] |
| 17. | β-Damascenone | Non-significant trend for left frontal differences in EEG associated with different liking responses. | Left frontal response associated with liking an odor. | [91] |
| 18. | Lavender and rosemary aromas | Induce left frontal EEG shifting in adults and infants with greater baselines than right frontal EEG activation. | Associated with greater approach behavior and less depressed affect. | [97] |
| 19. | General workers, perfume salespersons and professional perfume researchers | Professional perfume researchers respond to odors mainly in the frontal region. | Functional coupling for people—occupationally exposed to odors may be related to psychological preference. | [71] |
| 20. | Lavender and rosemary | Increased relative left frontal EEG asymmetry. | Infants of depressed and non-depressed mothers respond differently to odors. | [62] |
| 21. | Para-cresol 4-methylphenol, 2-heptanone, methional 3-methylthiopropionaldehyde and dimethyltrisulphide. | Theta wave activation in frontal region between the different populations. | Cultural differences in odor responsiveness. | [93] |
| 22. | Pleasant odor | Beta wave increased in the left frontal region. | Enhancement of left frontal brain region by a pleasant odor. | [92] |
| 23. | Neroli and grapefruit oils | Slow alpha (8–10 Hz) and theta activities increased in the occipital region. | Reduce the cortical deactivation or promote a relaxed state. | [79] |
| 24. | Low-dose alcohol | Theta power decreased in both hemispheres in the high-dose condition. | Corresponding to working memory demand. | [81] |
| 25. | Odor of incense and rose oil | Fast alpha activity increased in bilateral posterior regions during incense exposure. | Cortical and function of inhibitory processing of motor response. | [14] |
| 26. | Citrus bergamia oil | Negative percentage changes of the ratio of low to high frequency in the music, aroma and combined groups than control group. | Listening to soft music and inhaling Citrus bergamia essential oil—effective method of relaxation. | [90] |
| 27. | Abies sibirica essential oil | Increased theta activity after the visual display terminal task. | Prevention of visual display terminal—mental health disturbance. | [15] |
| 28. | Lavandula angustifolia | Good sleep quality—occipital and parietal alpha decreased, frontal theta and occipital beta increased. Poor sleep quality—theta increased in the all cranial regions. | Beneficial effect for female adults with sleep disorder. | [98] |
| 29. | Lavender oil | Theta and alpha activities increased. | Relaxing effect of inhaling lavender oil. | [12] |
| 30. | Essential oil of Zizyphus jujuba seeds | Fast alpha increased in the left prefrontal, right prefrontal and left frontal regions. | Increasing attention and relaxation. | [99] |
| 31. | Essential oil of Mentha arvensis L. f. piperascens aerial parts | Relative fast alpha increased. Gamma and the spectral edge frequency 90% decreased. | Reducing mental stress. | [100] |
| 32. | Jasmine oil | Beta wave increased in the anterior center and left posterior regions. | Increased—feeling of well-being, active, fresh and romantic. | [102] |
| 33. | Ylang–ylang essential oil | Prolonged the latencies of P300 | Not affect information processing resources in patients with TLE. | [63] |
| 34. | Essential odors—mint and lemon Commerical odors—criton-verbena, lize, melody and rozan | All odors affected the EEG waves in at least some subjects. | Essential odors stimulated more than commercial odors and women are more sensitive than men. | [35] |
| 35. | Pan-fired Japanese green tea (Koushun and Kouju) | Kouju affect the beta 1 at right frontal region. | Improve memory task performance. | [103] |
| 36. | Magnolia kobus flower | Absolute alpha decreased at left parietal region. | Awaken and increase the concentration states of brain. | [101] |
| 37. | Strawberry aroma (food) and the odor of lily of the valley (non-food) | Specific scalp potential maps for the two conditions. | Food odor—associated with the processing of rewards. Non-food odor—reflects odor characteristics excluding the reward. | [104] |
| 38. | Hyperbaric oxygen exposure | Fast delta decreased and alpha increased in the posterior regions. | Oxygen-toxicity diving-related problems. | [151] |
| 39. | Lemon, peppermint, and vanilla | Theta showed statistically significant results between different odor conditions | Stimuli can affect the frequency characteristics of the electrical activity of the brain. | [16] |
| 40. | Isomers of limonene and terpinolene | (+)-Limonene—relative high beta increased in the right temporal region. Terpinolene—relative mid beta decreased and relative fast alpha increased in the right prefrontal region. | Terpinolene—reducing the tension and increasing the relaxation and stabilization states of brain function. | [17] |
| 41. | Essential oil of Inula helenium root | Theta (in all the regions except T3), beta (Fp1) and mid beta (P4) and relative theta (Fp1, Fp2, F3 and F4) decreased. | Enhance the alertness state of brain. | [60] |
| 42. | Lavender and bergamot | The absolute theta increased at the right prefrontal region Significant differences in the relative fast and slow alpha. | Both physical and mental states became more stable and relaxed. | [64] |
| S. No. | Odorant Materials | Psychophysiological Changes | Reference |
|---|---|---|---|
| 1. | Rosemary and geranium oil | Geranium oil inhalation—both state and trait scores were significantly lowered. | [113] |
| 2. | Ylang-ylang, orange, geranium, cypress, bergamot, spearmint, and juniper | Cypress produced favorable impression after physical work and juniper produced favorable impression after mental work. | [114] |
| 3. | Orange | Relaxant effect—lower level of state anxiety, a more positive mood, and a higher level of calmness. | [115] |
| 4. | Rose, jasmine and lavender | Inhalation of favorite odors suppresses the muscle sympathetic vasoconstrictor activity and attenuates the blood pressure increase by affecting the central nervous system higher than the midbrain. | [116] |
| 5. | Enantiomers of limonene and carvone | Carvone—subjective restlessness. Prolonged inhalation of fragrances influences autonomic nervous system parameters as well as mental and emotional conditions. | [120] |
| 6. | Peppermint, jasmine, ylang-ylang, 1,8-cineole and menthol | Essentials oils or their components on basic forms of attention behavior are mainly psychological. | [119] |
| 7. | Lavender | Lavender odorants were associated with reduced mental stress and increased arousal rate. | [122] |
| 8. | Peppermint | Enhanced physical performance and generating more push-ups and running faster. | [135] |
| 9. | Isovaleric acid, thiophenol, pyridine, l-menthol, isoamyl acetate, and 1,8-cineole | Autonomic variations in response to olfactory stimuli—along two main dimensions of pleasantness and arousal. | [118] |
| 10. | Pepper oil, estragon oil, fennel oil or grapefruit oil, rose oil or patchouli oil | Fragrance inhalation of rose oil or patchouli oil caused a 40% decrease in relative sympathetic activity. | [117] |
| 11. | Lavender oil | Treatment of agitated behavior in patients with severe dementia. | [123] |
| 12. | Peppermint oil, jasmine oil and dimethyl sulfide | Peppermint odor reduced received work load and effort and increased self-evaluated physical performance and energy. | [136] |
| 13. | Lavender and rosemary oils | Lavender produced a significant decrease in performance of working memory. Rosemary produced a significant enhancement of performance for overall quality of memory. | [124] |
| 14. | Rosemary and lavender oils | Both rosemary and lavender scents were associated with lower mean ratings on the fatigue-inertia subscale, relative to the control group. | [125] |
| 15. | Lavender and neroli | Relaxing odors decreased heart rate and skin conductance, with stimulating odors producing reverse effects under equivalent conditions. | [126] |
| 16. | Lavender and rosemary oils | Alter affective appraisal of the experience and consequent retrospective evaluation of treatment-related pain. | [127] |
| 17. | Lavender oil | Increased the percentage of deep or slow-wave sleep in men and women and decreased rapid-eye movement sleep. | [128] |
| 18. | Peppermint oil | Reduced fatigue and improved mood and was rated as more pleasant, intense, stimulating, and elating than water. | [137] |
| 19. | Synthetic peppermint odor | Performance improvement in the presence of peppermint odor when the response mapping was incompatible but not in the compatible condition. | [139] |
| 20. | Jasmine tea, lavender, (R)-(−)-linalool and (S)-(+)-linalool | Jasmine tea, lavender and (R)-(−)-linalool increased positive mood state. | [152] |
| 21. | Orange and lavender | Reduced anxiety and improved mood in patients waiting for dental treatment. | [129] |
| 22. | Peppermint oil | Daytime sleepiness, peppermint oil can indeed reduce sleepiness. | [140] |
| 23. | Lavender and Jasmine | During recesses—higher concentration levels for lavender group | [133] |
| 24. | Ylang-ylang oil | More calm and more relaxed. | [142] |
| 25. | Chewing and caffeine | Caffeine led to improved speed and accuracy on cognitive tasks and increased alertness when compared with chewing. | [141] |
| 26. | Essential oil of Roman chamomile | Sedative effect | [143] |
| 27. | Lavender, chamomile, rosemary, and lemon | Aromatherapy massage exerts positive effects on anxiety and self-esteem. | [134] |
| 28. | With or without lavender-scented bath oil | Mothers—more relaxed, and smiled. Infants—cried less and spent more time in deep sleep. | [130] |
| 29. | Lavender | Increased relaxation. | [131] |
| 30. | Lavender and chocolate odors | Ortho- and retronasal odor presentation—route of odor presentation has direct implications for the enjoyment of foods and drinks. | [145] |
| 31. | Ylang-ylang aroma, peppermint aroma | Peppermint enhanced memory whereas ylang-ylang impaired it, peppermint increased alertness and ylang-ylang decreased it. | [141] |
| 32. | Aroma of heliotropin | Reduced sleepiness and improved refreshment at awakening for participants who suffered from insomniac symptoms. | [146] |
| 33. | Lavender oil | Reduced the stress levels and the pain intensity of needle insertion. | [132] |
| 34. | 1,8-Cineole following exposure to rosemary aroma | Cognitive tasksare significantly related to concentration of absorbed 1,8-cineole following exposure to rosemary aroma. | [147] |
| 35. | Eucalyptus oil | Pain and inflammatory responses after total knee replacement. | [148] |
| 36. | Bergamot essential oil and synthetic oil | Relieved work-related stress of teachers with various workloads and had a weak effect on young teachers who had a heavy workload. | [149] |
| 37. | Peppermint oil | Relaxation of bronchial smooth muscles, increased ventilation and brain oxygen concentration, and decreased blood lactate level. | [138] |
| 38. | Basil, bergamot, cardamom, cinnamon, juniper, lemon, orange, plamarosa, peppermint, sandalwood, spearmint and ylang-ylang | Essential oils may have versatile psychophysiological potencies. | [150] |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sowndhararajan, K.; Kim, S. Influence of Fragrances on Human Psychophysiological Activity: With Special Reference to Human Electroencephalographic Response. Sci. Pharm. 2016, 84, 724-751. https://doi.org/10.3390/scipharm84040724
Sowndhararajan K, Kim S. Influence of Fragrances on Human Psychophysiological Activity: With Special Reference to Human Electroencephalographic Response. Scientia Pharmaceutica. 2016; 84(4):724-751. https://doi.org/10.3390/scipharm84040724
Chicago/Turabian StyleSowndhararajan, Kandhasamy, and Songmun Kim. 2016. "Influence of Fragrances on Human Psychophysiological Activity: With Special Reference to Human Electroencephalographic Response" Scientia Pharmaceutica 84, no. 4: 724-751. https://doi.org/10.3390/scipharm84040724
APA StyleSowndhararajan, K., & Kim, S. (2016). Influence of Fragrances on Human Psychophysiological Activity: With Special Reference to Human Electroencephalographic Response. Scientia Pharmaceutica, 84(4), 724-751. https://doi.org/10.3390/scipharm84040724






