Role of Evolutionary Selection Acting on Vaccine Antigens in the Re-Emergence of Bordetella Pertussis
Abstract
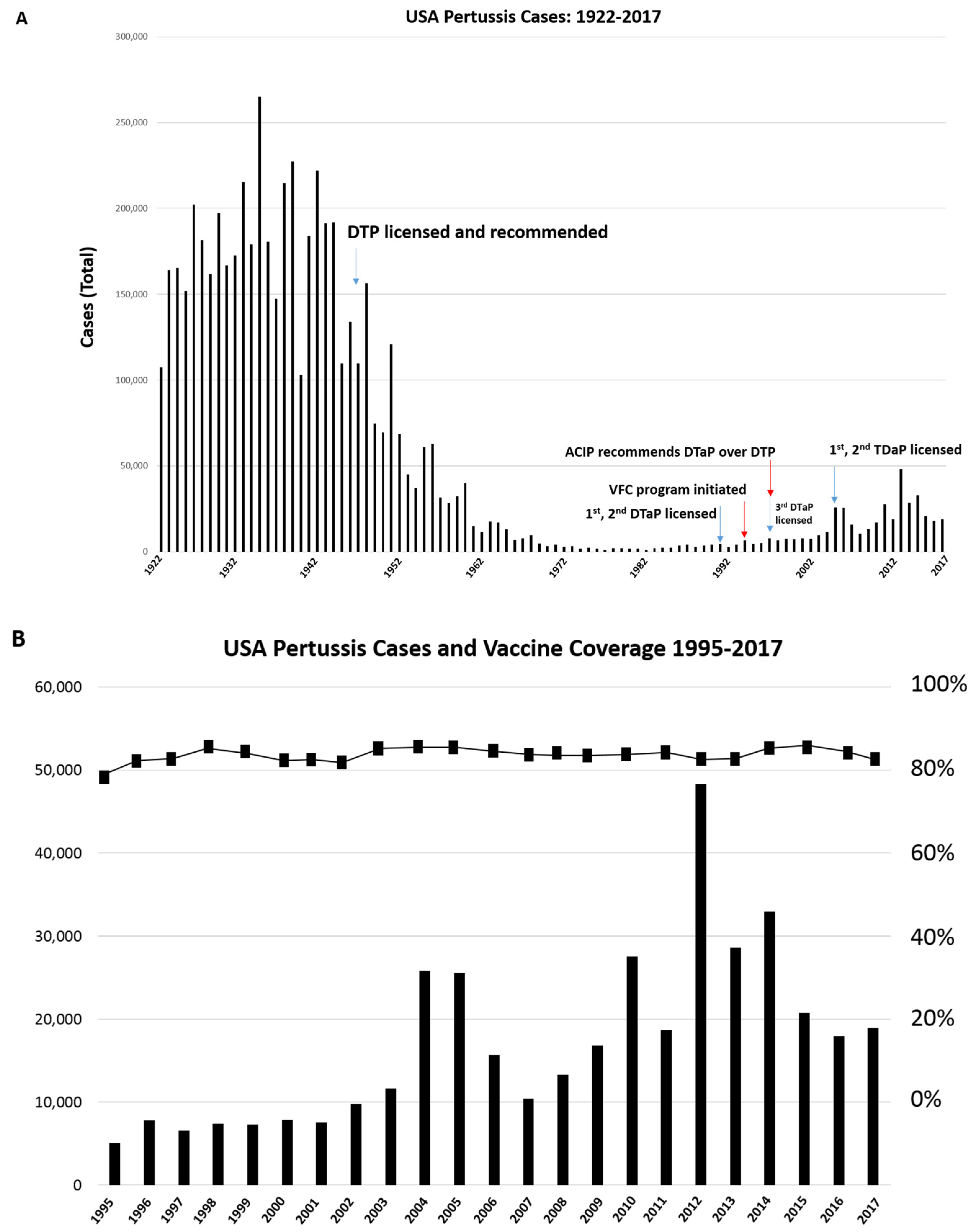
1. Introduction
2. Materials and Methods
2.1. Dataset Construction
2.2. Evolutionary Selection Analysis
2.3. Sequence, Immunoinformatic and Structural Analysis
2.4. Statistical Procedures
3. Results
3.1. Evolutionary Selection
3.2. Immunoinformatics
3.3. Protein Structure
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Cherry, J.D.; Heininger, U. Pertussis and other Bordetella infections. In Textbook of Pediatric Infectious Diseases, 6th ed.; Feigin, R.D., Cherry, J.D., Demmler-Harrison, G.J., Kaplan, S.L., Eds.; Elsevier Saunders: Philadelphia, PA, USA, 2009; pp. 1683–1706. [Google Scholar]
- Castagnini, L.A.; Munoz, F.M. Clinical characteristics and outcomes of neonatal pertussis: A comparative study. J. Pediatr. 2010, 156, 498–500. [Google Scholar] [CrossRef] [PubMed]
- Shojaei, J.; Saffar, M.; Hashemi, A.; Ghorbani, G.; Rezai, M.; Shahmohammadi, S. Clinical and laboratory features of pertussis in hospitalized infants with confirmed versus probable pertussis cases. Ann. Med. Health Sci. Res. 2014, 4, 910–914. [Google Scholar] [PubMed]
- Eidlitz-Markus, T.; Mimouni, M.; Zeharia, A. Pertussis symptoms in adolescents and children versus infants: The influence of vaccination and age. Clin. Pediatr. (Phila) 2007, 46, 718–723. [Google Scholar] [CrossRef]
- Miller, D.L.; Alderslade, R.; Ross, E.M. Whooping cough and whooping cough vaccine: The risks and benefits debate. Epidemiol. Rev. 1982, 4, 1–24. [Google Scholar] [CrossRef]
- Kretsinger, K.; Broder, K.R.; Cortese, M.M.; Joyce, M.P.; Ortega-Sanchez, I.; Lee, G.M.; Tiwari, T.; Cohn, A.C.; Slade, B.A.; Iskander, J.K.; et al. Preventing tetanus, diphtheria, and pertussis among adults: Use of tetanus toxoid, reduced diphtheria toxoid and acellular pertussis vaccine recommendations of the Advisory Committee on Immunization Practices (ACIP) and recommendation of ACIP, supported by the Healthcare Infection Control Practices Advisory Committee (HICPAC), for use of Tdap among health-care personnel. MMWR Recomm. Rep. 2006, 55, 1–37. [Google Scholar]
- Cherry, J.D.; Gornbein, J.; Heininger, U.; Stehr, K. A search for serologic correlates of immunity to Bordetella pertussis cough illnesses. Vaccine 1998, 16, 1901–1906. [Google Scholar] [CrossRef]
- Benson, D.A.; Cavanaugh, M.; Clark, K.; Karsch-Mizrachi, I.; Lipman, D.J.; Ostell, J.; Sayers, E.W. GenBank. Nucleic Acids Res. 2017, 45, D37–D42. [Google Scholar] [CrossRef]
- Stern, A.; Doron-Faigenboim, A.; Erez, E.; Martz, E.; Bacharach, E.; Pupko, T. Selecton 2007: Advanced models for detecting positive and purifying selection using a Bayesian inference approach. Nucleic Acids Res. 2007, 35, W506–W511. [Google Scholar] [CrossRef] [PubMed]
- Doron-Faigenboim, A.; Stern, A.; Mayrose, I.; Bacharach, E.; Pupko, T. Selecton: A server for detecting evolutionary forces at a single amino-acid site. Bioinformatics 2005, 21, 2101–2103. [Google Scholar] [CrossRef]
- Yang, Z.; Bielawski, J.P. Statistical methods for detecting molecular adaptation. Trends Ecol. Evol. 2000, 15, 496–503. [Google Scholar] [CrossRef]
- Sievers, F.; Higgins, D.G. Clustal Omega, accurate alignment of very large numbers of sequences. Methods Mol. Biol. 2014, 1079, 105–116. [Google Scholar] [PubMed]
- Jespersen, M.C.; Peters, B.; Nielsen, M.; Marcatili, P. BepiPred-2.0: Improving sequence-based B-cell epitope prediction using conformational epitopes. Nucleic Acids Res. 2017, 45, W24–W29. [Google Scholar] [CrossRef]
- Kolaskar, A.S.; Tongaonkar, P.C. A semi-empirical method for prediction of antigenic determinants on protein antigens. FEBS Lett 1990, 276, 172–174. [Google Scholar] [CrossRef]
- Vita, R.; Mahajan, S.; Overton, J.A.; Dhanda, S.K.; Martini, S.; Cantrell, J.R.; Wheeler, D.K.; Sette, A.; Peters, B. The Immune Epitope Database (IEDB): 2018 update. Nucleic Acids Res. 2019, 47, D339–D343. [Google Scholar] [CrossRef] [PubMed]
- Garnier, J.; Gibrat, J.F.; Robson, B. GOR method for predicting protein secondary structure from amino acid sequence. Methods Enzymol. 1996, 266, 540–553. [Google Scholar]
- Ausiello, C.M.; Urbani, F.; La Sala, A.; Lande, R.; Piscitelli, A.; Cassone, A. Acellular vaccines induce cell-mediated immunity to Bordetella pertussis antigens in infants undergoing primary vaccination against pertussis. Dev. Biol. Stand. 1997, 89, 315–320. [Google Scholar]
- Tartof, S.Y.; Lewis, M.; Kenyon, C.; White, K.; Osborn, A.; Liko, J.; Zell, E.; Martin, S.; Messonnier, N.E.; Clark, T.A.; et al. Waning immunity to pertussis following 5 doses of DTaP. Pediatrics 2013, 131, e1047–e1052. [Google Scholar] [CrossRef]
- van Gent, M.; Bart, M.J.; van der Heide, H.G.; Heuvelman, K.J.; Mooi, F.R. Small mutations in Bordetella pertussis are associated with selective sweeps. PLoS ONE 2012, 7, e46407. [Google Scholar] [CrossRef] [PubMed]
- Gzyl, A.; Augustynowicz, E.; Gniadek, G.; Rabczenko, D.; Dulny, G.; Slusarczyk, J. Sequence variation in pertussis S1 subunit toxin and pertussis genes in Bordetella pertussis strains used for the whole-cell pertussis vaccine produced in Poland since 1960: Efficiency of the DTwP vaccine-induced immunity against currently circulating B. pertussis isolates. Vaccine 2004, 22, 2122–2128. [Google Scholar] [PubMed]
- Kallonen, T.; Gröndahl-Yli-Hannuksela, K.; Elomaa, A.; Lutyńska, A.; Fry, N.K.; Mertsola, J.; He, Q. Differences in the genomic content of Bordetella pertussis isolates before and after introduction of pertussis vaccines in four European countries. Infect. Genet. Evol. 2011, 11, 2034–2042. [Google Scholar] [CrossRef]
- Mooi, F.R.; Zeddeman, A.; van Gent, M. The pertussis problem: Classical epidemiology and strain characterization should go hand in hand. J. Pediatr. (Rio. J.) 2015, 91, 315–317. [Google Scholar] [CrossRef][Green Version]
- van der Zee, A.; Vernooij, S.; Peeters, M.; van Embden, J.; Mooi, F.R. Dynamics of the population structure of Bordetella pertussis as measured by IS1002-associated RFLP: Comparison of pre- and post-vaccination strains and global distribution. Microbiology 1996, 142, 3479–3485. [Google Scholar] [CrossRef]
- Barkoff, A.M.; Mertsola, J.; Guillot, S.; Guiso, N.; Berbers, G.; He, Q. Appearance of Bordetella pertussis strains not expressing the vaccine antigen pertactin in Finland. Clin. Vaccine Immunol. 2012, 19, 1703–1704. [Google Scholar] [CrossRef]
- Bouchez, V.; Brun, D.; Cantinelli, T.; Dore, G.; Njamkepo, E.; Guiso, N. First report and detailed characterization of B. pertussis isolates not expressing Pertussis Toxin or Pertactin. Vaccine 2009, 27, 6034–6041. [Google Scholar] [CrossRef]
- Lam, C.; Octavia, S.; Ricafort, L.; Sintchenko, V.; Gilbert, G.L.; Wood, N.; McIntyre, P.; Marshall, H.; Guiso, N.; Keil, A.D.; et al. Rapid increase in pertactin-deficient Bordetella pertussis isolates, Australia. Emerg. Infect. Dis. 2014, 20, 626–633. [Google Scholar] [CrossRef]
- Otsuka, N.; Han, H.J.; Toyoizumi-Ajisaka, H.; Nakamura, Y.; Arakawa, Y.; Shibayama, K.; Kamachi, K. Prevalence and genetic characterization of pertactin-deficient Bordetella pertussis in Japan. PLoS ONE 2012, 7, e31985. [Google Scholar] [CrossRef]
- Tsang, R.S.; Shuel, M.; Jamieson, F.B.; Drews, S.; Hoang, L.; Horsman, G.; Lefebvre, B.; Desai, S.; St-Laurent, M. Pertactin-negative Bordetella pertussis strains in Canada: Characterization of a dozen isolates based on a survey of 224 samples collected in different parts of the country over the last 20 years. Int. J. Infect. Dis. 2014, 28, 65–69. [Google Scholar] [CrossRef]
- Pawloski, L.C.; Queenan, A.M.; Cassiday, P.K.; Lynch, A.S.; Harrison, M.J.; Shang, W.; Williams, M.M.; Bowden, K.E.; Burgos-Rivera, B.; Qin, X.; et al. Prevalence and molecular characterization of pertactin-deficient Bordetella pertussis in the United States. Clin Vaccine Immunol 2014, 21, 119–125. [Google Scholar] [CrossRef]
- Williams, M.M.; Sen, K.; Weigand, M.R.; Skoff, T.H.; Cunningham, V.A.; Halse, T.A.; Tondella, M.L.; Group, C.P.W. Bordetella pertussis strain lacking pertactin and pertussis toxin. Emerg. Infect. Dis. 2016, 22, 319–322. [Google Scholar] [CrossRef]
- Hegerle, N.; Dore, G.; Guiso, N. Pertactin deficient Bordetella pertussis present a better fitness in mice immunized with an acellular pertussis vaccine. Vaccine 2014, 32, 6597–6600. [Google Scholar] [CrossRef]
- Safarchi, A.; Octavia, S.; Luu, L.D.; Tay, C.Y.; Sintchenko, V.; Wood, N.; Marshall, H.; McIntyre, P.; Lan, R. Pertactin negative Bordetella pertussis demonstrates higher fitness under vaccine selection pressure in a mixed infection model. Vaccine 2015, 33, 6277–6281. [Google Scholar] [CrossRef]
- Mooi, F.R.; van Oirschot, H.; Heuvelman, K.; van der Heide, H.G.; Gaastra, W.; Willems, R.J. Polymorphism in the Bordetella pertussis virulence factors P.69/pertactin and pertussis toxin in The Netherlands: Temporal trends and evidence for vaccine-driven evolution. Infect. Immun. 1998, 66, 670–675. [Google Scholar]
- Truebestein, L.; Leonard, T.A. Coiled-coils: The long and short of it. Bioessays 2016, 38, 903–916. [Google Scholar] [CrossRef]
- Lin, C.J.; Deger, K.A.; Tien, J.H. Modeling the trade-off between transmissibility and contact in infectious disease dynamics. Math. Biosci. 2016, 277, 15–24. [Google Scholar] [CrossRef]
- Mooi, F.R.; van Loo, I.H.; van Gent, M.; He, Q.; Bart, M.J.; Heuvelman, K.J.; de Greeff, S.C.; Diavatopoulos, D.; Teunis, P.; Nagelkerke, N.; et al. Bordetella pertussis strains with increased toxin production associated with pertussis resurgence. Emerg. Infect. Dis. 2009, 15, 1206–1213. [Google Scholar] [CrossRef]
- Advani, A.; Gustafsson, L.; Carlsson, R.M.; Donnelly, D.; Hallander, H.O. Clinical outcome of pertussis in Sweden: Association with pulsed-field gel electrophoresis profiles and serotype. APMIS 2007, 115, 736–742. [Google Scholar] [CrossRef]
- Bart, M.J.; Harris, S.R.; Advani, A.; Arakawa, Y.; Bottero, D.; Bouchez, V.; Cassiday, P.K.; Chiang, C.S.; Dalby, T.; Fry, N.K.; et al. Global population structure and evolution of Bordetella pertussis and their relationship with vaccination. MBio 2014, 5, e01074. [Google Scholar] [CrossRef]
- Clarke, M.; McIntyre, P.B.; Blyth, C.C.; Wood, N.; Octavia, S.; Sintchenko, V.; Giles, L.; Quinn, H.; Hill, V.; Hanly, G.; et al. The relationship between Bordetella pertussis genotype and clinical severity in Australian children with pertussis. J. Infect. 2016, 72, 171–178. [Google Scholar] [CrossRef]
- Octavia, S.; Sintchenko, V.; Gilbert, G.L.; Lawrence, A.; Keil, A.D.; Hogg, G.; Lan, R. Newly emerging clones of Bordetella pertussis carrying prn2 and ptxP3 alleles implicated in Australian pertussis epidemic in 2008–2010. J. Infect. Dis. 2012, 205, 1220–1224. [Google Scholar] [CrossRef]
- Carbonetti, N.H.; Artamonova, G.V.; Van Rooijen, N.; Ayala, V.I. Pertussis toxin targets airway macrophages to promote Bordetella pertussis infection of the respiratory tract. Infect. Immun. 2007, 75, 1713–1720. [Google Scholar] [CrossRef]
- Kirimanjeswara, G.S.; Agosto, L.M.; Kennett, M.J.; Bjornstad, O.N.; Harvill, E.T. Pertussis toxin inhibits neutrophil recruitment to delay antibody-mediated clearance of Bordetella pertussis. J. Clin. Investig. 2005, 115, 3594–3601. [Google Scholar] [CrossRef]
- Schmidtke, A.J.; Boney, K.O.; Martin, S.W.; Skoff, T.H.; Tondella, M.L.; Tatti, K.M. Population diversity among Bordetella pertussis isolates, United States, 1935–2009. Emerg. Infect. Dis. 2012, 18, 1248–1255. [Google Scholar] [CrossRef]
- Mastrantonio, P.; Spigaglia, P.; van Oirschot, H.; van der Heide, H.G.; Heuvelman, K.; Stefanelli, P.; Mooi, F.R. Antigenic variants in Bordetella pertussis strains isolated from vaccinated and unvaccinated children. Microbiology 1999, 145, 2069–2075. [Google Scholar] [CrossRef][Green Version]
- Breakwell, L.; Kelso, P.; Finley, C.; Schoenfeld, S.; Goode, B.; Misegades, L.K.; Martin, S.W.; Acosta, A.M. Pertussis vaccine effectiveness in the setting of pertactin-deficient pertussis. Pediatrics 2016, 137, e20153973. [Google Scholar] [CrossRef]
- Glanz, J.M.; Narwaney, K.J.; Newcomer, S.R.; Daley, M.F.; Hambidge, S.J.; Rowhani-Rahbar, A.; Lee, G.M.; Nelson, J.C.; Naleway, A.L.; Nordin, J.D.; et al. Association between undervaccination with diphtheria, tetanus toxoids, and acellular pertussis (DTaP) vaccine and risk of pertussis infection in children 3 to 36 months of age. JAMA Pediatr. 2013, 167, 1060–1064. [Google Scholar] [CrossRef]
- Bolotin, S.; Harvill, E.T.; Crowcroft, N.S. What to do about pertussis vaccines? Linking what we know about pertussis vaccine effectiveness, immunology and disease transmission to create a better vaccine. Pathog. Dis. 2015, 73, ftv057. [Google Scholar] [CrossRef]
- Burns, D.L.; Meade, B.D.; Messionnier, N.E. Pertussis resurgence: Perspectives from the Working Group Meeting on pertussis on the causes, possible paths forward, and gaps in our knowledge. J. Infect. Dis. 2014, 209, S32–S35. [Google Scholar] [CrossRef]
- Esposito, S.; Principi, N. Prevention of pertussis: An unresolved problem. Hum. Vaccin. Immunother. 2018, 14, 2452–2459. [Google Scholar] [CrossRef]
- Plotkin, S.A. The pertussis problem. Clin. Infect. Dis. 2014, 58, 830–833. [Google Scholar] [CrossRef]

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Etskovitz, H.; Anastasio, N.; Green, E.; May, M. Role of Evolutionary Selection Acting on Vaccine Antigens in the Re-Emergence of Bordetella Pertussis. Diseases 2019, 7, 35. https://doi.org/10.3390/diseases7020035
Etskovitz H, Anastasio N, Green E, May M. Role of Evolutionary Selection Acting on Vaccine Antigens in the Re-Emergence of Bordetella Pertussis. Diseases. 2019; 7(2):35. https://doi.org/10.3390/diseases7020035
Chicago/Turabian StyleEtskovitz, Haley, Nicole Anastasio, Evangeline Green, and Meghan May. 2019. "Role of Evolutionary Selection Acting on Vaccine Antigens in the Re-Emergence of Bordetella Pertussis" Diseases 7, no. 2: 35. https://doi.org/10.3390/diseases7020035
APA StyleEtskovitz, H., Anastasio, N., Green, E., & May, M. (2019). Role of Evolutionary Selection Acting on Vaccine Antigens in the Re-Emergence of Bordetella Pertussis. Diseases, 7(2), 35. https://doi.org/10.3390/diseases7020035



