The Effects of Exercise on IL-6 Levels and Cognitive Performance in Patients with Schizophrenia
Abstract
1. Introduction
2. Schizophrenia and Exercise
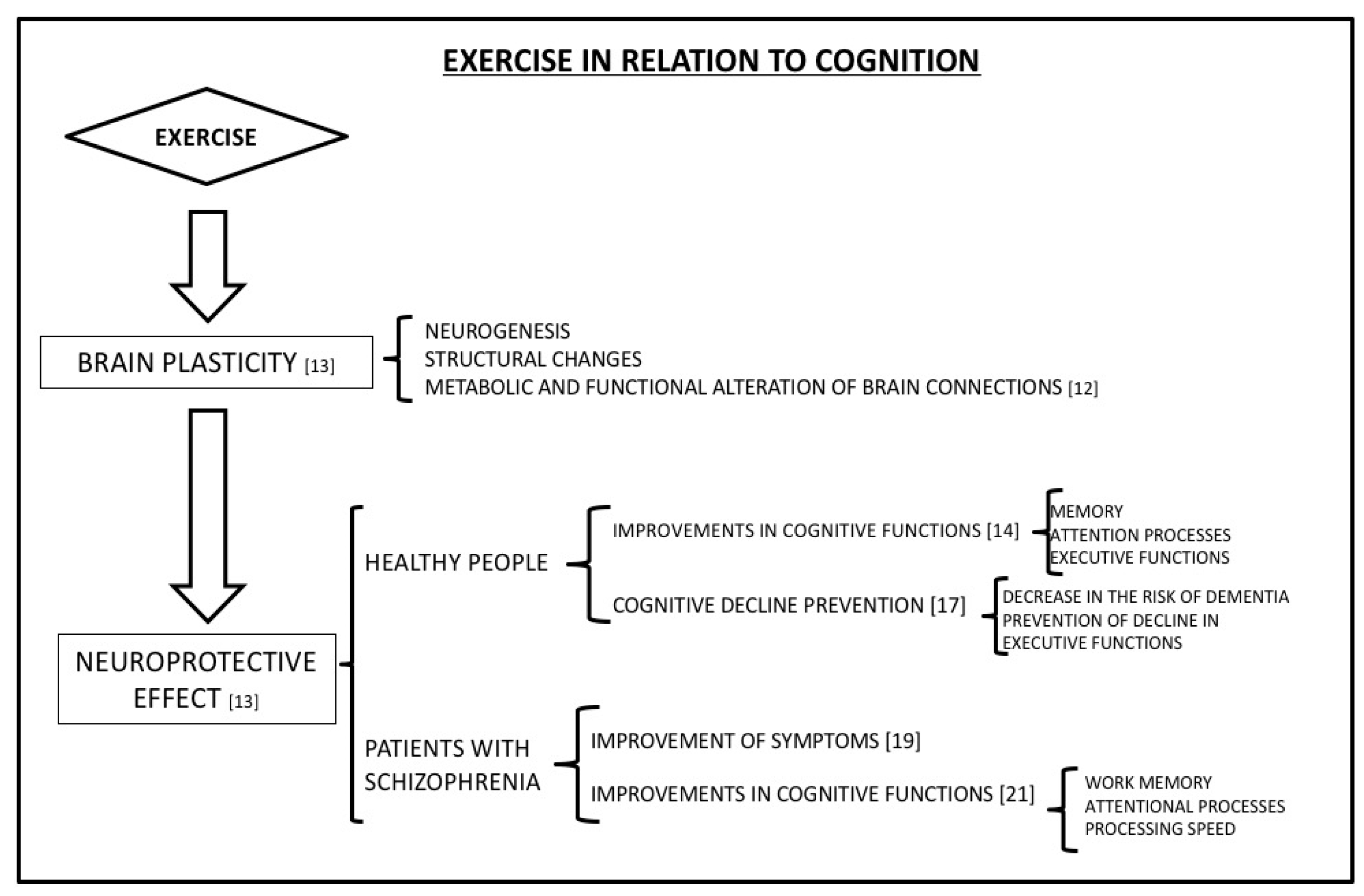
2.1. Exercise in Relation to Cognition
2.2. Schizophrenia and Exercise
3. Schizophrenia, Exercise and the Immune System
3.1. The Immune System in Relation to Cognition
3.2. The Immune System and Schizophrenia
3.3. The Immune System and Exercise
4. Schizophrenia, Exercise and IL-6
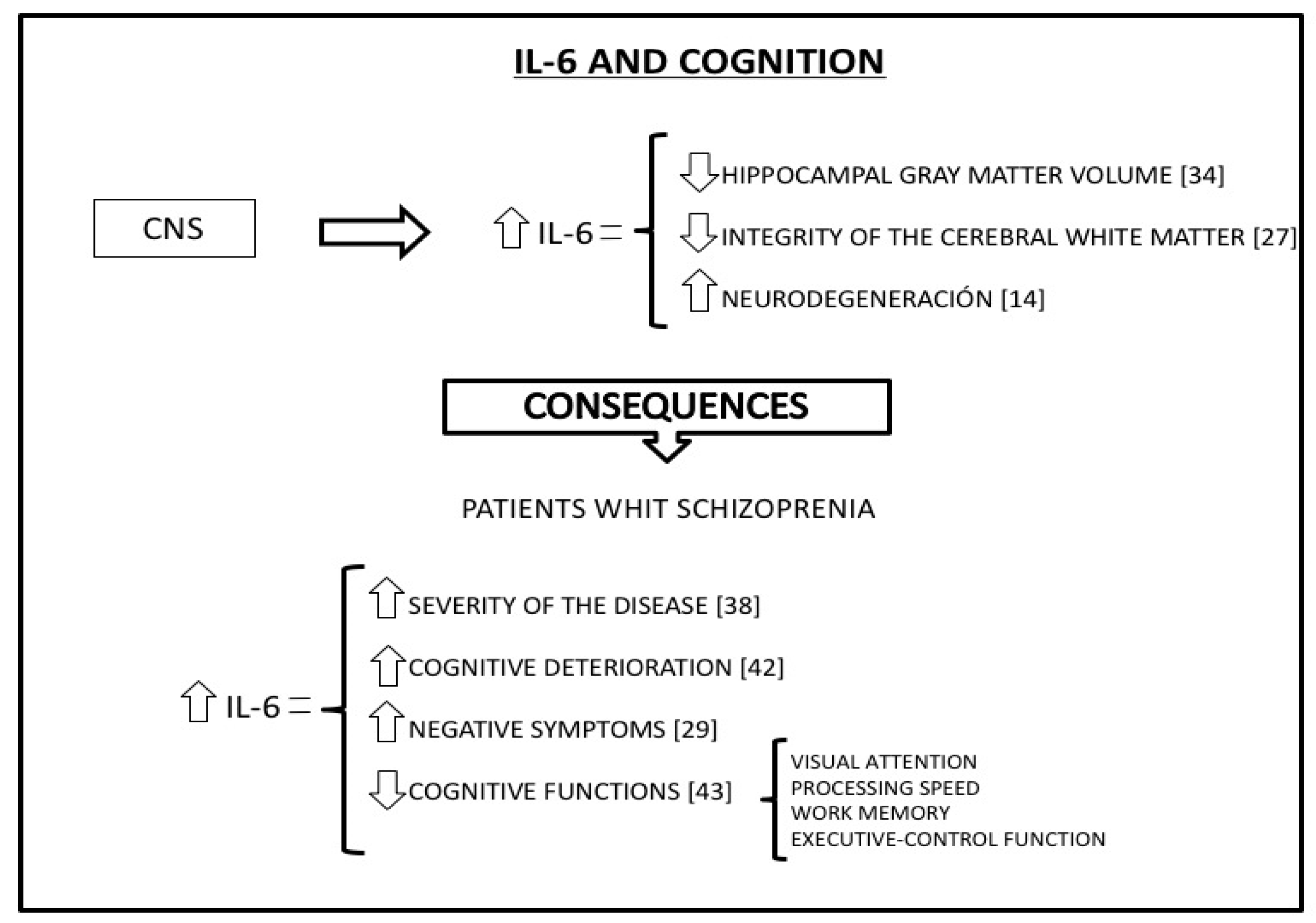
4.1. IL-6 and Cognition
4.2. Il-6 and Schizophrenia
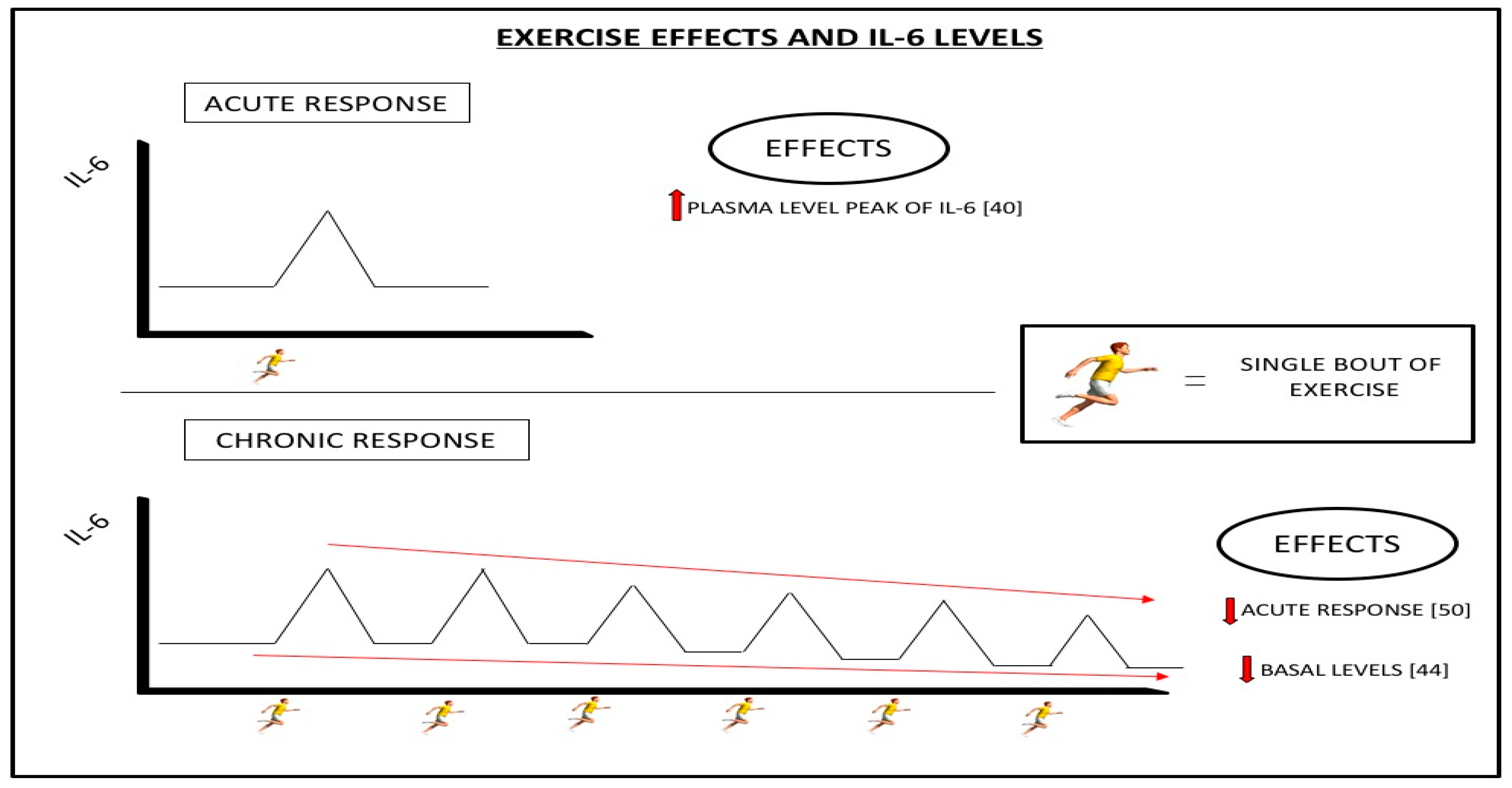
4.3. Il-6 and Exercise
5. Discussion
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disoreders, Fourth Edition(DSM-IV); American Psychiatric Organisation: Washington, DC, USA, 1994. [Google Scholar]
- Nielsen, R.E.; Levander, S.; Kjaersdam Telléus, G.; Jensen, S.O.W.; Østergaard Christensen, T.; Leucht, S. Second-generation antipsychotic effect on cognition in patients with schizophrenia-a meta-analysis of randomized clinical trials. Acta Psychiatr. Scand. 2015, 131, 185–196. [Google Scholar] [CrossRef] [PubMed]
- Van Kesteren, C.F.M.G.; Gremmels, H.; de Witte, L.D.; Hol, E.M.; Van Gool, A.R.; Falkai, P.G.; Kahn, R.S.; Sommer, I.E.C. Immune involvement in the pathogenesis of schizophrenia: A meta-analysis on postmortem brain studies. Transl. Psychiatry 2017, 7, e1075. [Google Scholar] [CrossRef] [PubMed]
- Singh, B.; Chaudhuri, T.K. Role of C-reactive protein in schizo- phrenia: An overview. Psychiatry Res. 2014, 216, 277–285. [Google Scholar] [CrossRef] [PubMed]
- Barron, H.; Hafizi, S.; Andreazza, A.C.; Mizrahi, R. Neuroinflam- mation and oxidative stress in psychosis and psychosis risk. Int. J. Mol. Sci. 2017, 18, 651. [Google Scholar] [CrossRef] [PubMed]
- Orsolini, L.; Sarchione, F.; Vellante, F.; Fornaro, M.; Matarazzo, I.; Martinotti, G.; Valchera, A.; Di Nicola, M.; Carano, A.; Di Giannantonio, M.; et al. Protein-C reactive as biomarker predictor of schizophrenia phases of illness? A systematic review. Curr. Neuropharmacol. 2018, 16, 583–606. [Google Scholar] [CrossRef] [PubMed]
- Licastro, F.; Chiappelli, M.; Ruscica, M.; Carnelli, V.; Corsi, M.M. Altered cytokine and acute phase response protein levels in the blood of children with Downs syndrome: Relationship with dementia of Alzheimer’s type. Int. J. Immunopathol. Pharmacol. 2005, 18, 165–172. [Google Scholar] [CrossRef]
- Hatzistilianou, M.; Hitoglou, S.; Gougoustamou, D.; Rekliti, A.; Tzouvelekis, G.; Nanas, C.; Catriu, D.; Kotsis, O. Serum procalcitonin, adenosine deaminase and its isoenzymes in the aetiological diagnosis of pneumonia in children. Int. J. Immunopathol. Pharmacol. 2002, 15, 119–127. [Google Scholar] [CrossRef]
- De Berardis, D.; Campanella, D.; Gambi, F.; La Rovere, R.; Carano, A.; Conti, C.M.; Silvestrini, C.; Serroni, N.; Piersanti, D.; Di Giuseppe, B.; et al. The Role of C-Reactive Protein in Mood Disorders. Int. J. Immunopathol. Pharmacol. 2006, 19, 721–725. [Google Scholar] [CrossRef]
- Rodrigues-Amorim, D.; Rivera-Baltanás, T.; Spuch, C.; Caruncho, H.J.; González-Fernandez, Á.; Olivares, J.M.; Agís-Balboa, R.C. Cytokines dysregulation in schizophrenia: A systematic review of psychoneuroimmune relationship. Schizophr. Res. 2018, 197, 19–33. [Google Scholar] [CrossRef]
- Van der Stouwe, E.C.D.; Van Busschbach, J.T.; de Vries, B.; Cahn, W.; Aleman, A.; Pijnenborg, G.H.M. Neural correlates of exercise training in individuals with schizophrenia and in healthy individuals_ A systematic review. Neuroimage Clin. 2018, 19, 287–301. [Google Scholar] [CrossRef]
- Mandolesi, L.; Polverino, A.; Montuori, S.; Foti, F.; Ferraioli, G.; Sorrentino, P.; Sorrentino, G. Effects of Physical Exercise on Cognitive Functioning and Wellbeing: Biological and Psychological Benefits. Front. Psychol. 2018, 9, 509. [Google Scholar] [CrossRef] [PubMed]
- Di Benedetto, S.; Müller, L.; Wenger, E.; Düzel, S.; Pawelec, G. Contribution of neuroinflammation and immunity to brain aging and the mitigating effects of physical and cognitive interventions. Neurosci. Biobehav. Rev. 2017, 75, 114–128. [Google Scholar] [CrossRef] [PubMed]
- Thielen, J.-W.; Kärgel, C.; Müller, B.W.; Rasche, I.; Genius, J.; Bus, B.; Maderwald, S.; Norris, D.G.; Wiltfang, J.; Tendolkar, I. Aerobic Activity in the Healthy Elderly Is Associated with Larger Plasticity in Memory Related Brain Structures and Lower Systemic Inflammation. Front. Aging Neurosci. 2016, 8, 319. [Google Scholar] [CrossRef] [PubMed]
- Ostermann, S.; Herbsleb, M.; Schulz, S.; Donath, L.; Berger, S.; Eisenträger, D.; Siebert, T.; Müller, H.-J.; Puta, C.; Voss, A.; et al. Exercise Reveals the Interrelation of Physical Fitness, In ammatory Response, Psychopathology, and Autonomic Function in Patients With Schizophrenia. Schizophr. Bull. 2018, 39, 1139–1149. [Google Scholar] [CrossRef] [PubMed]
- Niemann, C.; Godde, B.; Staudinger, U.M.; Voelcker-Rehage, C. Exercise-induced changes in basal ganglia volume and cognition in older adults. Neuroscience 2014, 281, 147–163. [Google Scholar] [CrossRef]
- Hollamby, A.; Davelaar, E.J.; Cadar, D. Increased physical fitness is associated with higher executive functioning in people with dementia. Front. Public Heal. 2017, 5, 346. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, B.K.; Febbraio, M. Muscle-derived interleukin-6—A possible link between skeletal muscle, adipose tissue, liver, and brain. Brain Behav. Immun. 2005, 19, 371–376. [Google Scholar] [CrossRef]
- Dauwan, M.; Begemann, M.J.H.; Heringa, S.M.; Sommer, I.E. Exercise Improves Clinical Symptoms, Quality of Life, Global Functioning, and Depression in Schizophrenia: A Systematic Review and Meta-analysis. Schizophr. Bull. 2016, 42, 588–599. [Google Scholar] [CrossRef]
- Stubbs, B.; Vancampfort, D.; Hallgren, M.; Firth, J.; Veronese, N.; Solmi, M.; Brand, S.; Cordes, J.; Malchow, B.; Gerber, M.; et al. EPA guidance on physical activity as a treatment for severe mental illness: A meta-review of the evidence and Position Statement from the European Psychiatric Association (EPA), supported by the International Organization of Physical Therapists in Mental Health (IOPTMH). Eur. Psychiatry 2018, 54, 124–144. [Google Scholar] [CrossRef]
- Firth, J.; Cotter, J.; Elliott, R.; French, P.; Yung, A.R. A systematic review and meta-analysis of exercise interventions in schizophrenia patients. Psychol. Med. 2015, 45, 1343–1361. [Google Scholar] [CrossRef]
- Hidese, S.; Matsuo, J.; Ishida, I.; Hiraishi, M.; Teraishi, T.; Ota, M.; Hattori, K.; Kunugi, H. Relationship of Handgrip Strength and Body Mass Index With Cognitive Function in Patients With Schizophrenia. Front. Psychiatry 2018, 9, 156. [Google Scholar] [CrossRef] [PubMed]
- Leiter, O.; Kempermann, G.; Walker, T.L. A common language: How neuroimmunological cross talk regulates adult hippocampal neurogenesis. Stem Cells Int. 2016, 2016, 1681590. [Google Scholar] [CrossRef] [PubMed]
- Hasegawa-Ishii, S.; Inaba, M.; Umegaki, H.; Unno, K.; Wakabayashi, K.; Shimada, A. Endotoxemia-induced cytokine-mediated responses of hippocampal astrocytes transmitted by cells of the brain-immune interface. Sci. Rep. 2016, 6, 25457. [Google Scholar] [CrossRef] [PubMed]
- Levin, S.G.; Godukhin, O.V. Modulating Effect of Cytokines on Mechanisms of Synaptic Plasticity in the Brain. Biochemistry 2017, 82, 264–274. [Google Scholar] [CrossRef] [PubMed]
- McAfoose, J.; Baune, B.T. Evidence for a cytokine model of cognitive function. Neurosci. Biobehav. Rev. 2009, 33, 355–366. [Google Scholar] [CrossRef] [PubMed]
- Marsland, A.L.; Gianaros, P.J.; Kuan, D.C.-H.; Sheu, L.K.; Krajina, K.; Manuck, S.B. Brain Morphology Links Systemic Inflammation to Cognitive Function in Midlife Adults. Brain Behav. Immun. 2015, 48, 195–204. [Google Scholar] [CrossRef] [PubMed]
- Scheinert, R.B.; Asokan, A.; Rani, A.; Kumar, A.; Foster, T.C.; Ormerod, B.K. Some hormone, cytokine and chemokine levels that change across lifespan vary by cognitive status in male Fischer 344 rats. Brain Behav. Immun. 2015, 49, 216–232. [Google Scholar] [CrossRef]
- Goldsmith, D.R.; Haroon, E.; Miller, A.H.; Strauss, G.P.; Buckley, P.F.; Miller, B.J. TNF-α and IL-6 are associated with the deficit syndrome and negative symptoms in patients with chronic schizophrenia. Schizophr. Res. 2018, 199, 281–284. [Google Scholar] [CrossRef]
- Noto, C.; Maes, M.; Ota, V.K.; Teixeira, A.L.; Bressan, R.A.; Gadelha, A.; Brietzke, E. High predictive value of immune-inflammatory biomarkers for schizophrenia diagnosis and association with treatment resistance. World J. Biol. Psychiatry 2015, 16, 422–429. [Google Scholar] [CrossRef]
- van der Geest, K.S.M.; Wang, Q.; Eijsvogels, T.M.H.; Koenen, H.J.P.; Joosten, I.; Brouwer, E.; Hopman, M.T.E.; Jacobs, J.F.M.; Boots, A.M.H. Changes in peripheral immune cell numbers and functions in octogenarian walkers—An acute exercise study. Immun. Ageing 2017, 14, 5. [Google Scholar] [CrossRef]
- Gjevestad, G.O.; Holven, K.B.; Ulven, S.M. Effects of Exercise on Gene Expression of Inflammatory Markers in Human Peripheral Blood Cells: A Systematic Review. Curr. Cardiovasc. Risk Rep. 2015, 9, 34. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.; Wang, R.; Grant, A.R.; Zhang, J.; Gordon, P.M.; Wei, Y.; Chen, P. Immune adaptation to chronic intense exercise training: New microarray evidence. BMC Genom. 2017, 18, 29. [Google Scholar] [CrossRef] [PubMed]
- Kline, R.; Wong, E.; Haile, M.; Didehvar, S.; Farber, S.; Sacks, A.; Pirraglia, E.; de Leon, M.J.; Bekker, A. Peri-Operative Inflammatory Cytokines in Plasma of the Elderly Correlate in Prospective Study with Postoperative Changes in Cognitive Test Scores. Int. J. Anesthesiol. Res. 2016, 4, 313–321. [Google Scholar] [CrossRef] [PubMed]
- Puzianowska-Kuźnicka, M.; Owczarz, M.; Wieczorowska-Tobis, K.; Nadrowski, P.; Chudek, J.; Slusarczyk, P.; Skalska, A.; Jonas, M.; Franek, E.; Mossakowska, M. Interleukin-6 and C-reactive protein, successful aging, and mortality: The PolSenior study. Immun. Ageing 2016, 13, 21. [Google Scholar] [CrossRef] [PubMed]
- Paine, N.J.; Bosch, J.A.; Ring, C.; Drayson, M.T.; van Zasten, J.J.C.S.V. Induced mild systemic inflammation is associated with impaired ability to improve cognitive task performance by practice. Psychophysiology 2015, 52, 333–341. [Google Scholar] [CrossRef] [PubMed]
- Bradburn, S.; Sarginson, J.; Murgatroyd, C.A. Association of Peripheral Interleukin-6 with Global Cognitive Decline in Non-demented Adults: A Meta-Analysis of Prospective Studies. Front. Aging Neurosci. 2018, 9, 438. [Google Scholar] [CrossRef] [PubMed]
- Shivakumar, V.; Debnath, M.; Venugopal, D.; Rajasekaran, A.; Kalmady, S.V.; Subbanna, M.; Narayanaswamy, J.C.; Amaresha, A.C.; Venkatasubramanian, G. Influence of correlation between HLA-G polymorphism and Interleukin-6 (IL6) gene expression on the risk of schizophrenia. Cytokine 2018, 107, 59–64. [Google Scholar] [CrossRef] [PubMed]
- Gruol, D.L. IL-6 regulation of synaptic function in the CNS. Neuropharmacology 2015, 96, 42–54. [Google Scholar] [CrossRef] [PubMed]
- Philippe, M.; Krüsmann, P.J.; Mersa, L.; Eder, E.M.; Gatterer, H.; Melmer, A.; Ebenbichler, C.; Burtscher, M. Acute effects of concentric and eccentric exercise on glucose metabolism and interleukin-6 concentration in healthy males. Biol. Sport. 2016, 33, 153–158. [Google Scholar] [CrossRef] [PubMed]
- Lavratti, C.; Dorneles, G.; Pochmann, D.; Peres, A.; Bard, A.; de Lima Schipper, L.; Lago, P.D.; Wagner, L.C.; Elsner, V.R. Exercise-induced modulation of histone H4 acetylation status and cytokines levels in patients with schizophrenia. Physiol. Behav. 2017, 168, 84–90. [Google Scholar] [CrossRef] [PubMed]
- Borovcanin, M.M.; Jovanovic, I.; Radosavljevic, G.; Pantic, J.; Minic Janicijevic, S.; Arsenijevic, N.; Lukic, M.L. Interleukin-6 in Schizophrenia—Is There a Therapeutic Relevance? Front. Psychiatry 2017, 8, 221. [Google Scholar] [CrossRef]
- Misiak, B.A.; Czykiewicz, B.O.S.; Kotowicz, K.; Rybakowski, J.K.; Samochowiec, J.; Frydecka, D. Cytokines and C-reactive protein alterations with respect to cognitive impairment in schizophrenia and bipolar disorder: A systematic review. Schizophr. Res. 2018, 192, 16–29. [Google Scholar] [CrossRef]
- Monteiro-Junior, R.S.; de Tarso Maciel-Pinheiro, P.; da Matta Mello, E.; da Silva, L.F.; Terra, R.; Carneiro, L.S.F.; Rodrigues, V.D.; Nascimento, O.J.M.; Deslandes, A.C.; Laks, J. Effect of Exercise on Inflammatory Profile of Older Persons: Systematic Review and Meta-Analyses. J. Phys. Act. Health. 2018, 15, 64–71. [Google Scholar] [CrossRef]
- Kapilevich, L.V.; Zakharova, A.N.; Kabachkova, A.V.; Kironenko, T.A.; Orlov, S.N. Dynamic and Static Exercises Differentially Affect Plasma Cytokine Content in Elite Endurance- and Strength-Trained Athletes and Untrained Volunteers. Front. Physiol. 2017, 8, 35. [Google Scholar] [CrossRef]
- Huh, J.Y. The role of exercise-induced myokines in regulating metabolism. Arch. Pharmacal Res. 2018, 41, 14–29. [Google Scholar] [CrossRef]
- Cronin, O.; Keohane, D.M.; Molloy, M.G.; Shanahan, F. The effect of exercise interventions on inflammatory biomarkers in healthy, physically inactive subjects: A systematic review. Int. J. Med. 2017, 110, 629–637. [Google Scholar] [CrossRef]
- Pruimboom, L.; Raison, C.L.; Muskiet, F.A.J. Physical Activity Protects the Human Brain against Metabolic Stress Induced by a Postprandial and Chronic Inflammation. Behav. Neurol. 2015. [Google Scholar] [CrossRef]
- Firth, J.; Cotter, J.; Carney, R.; Yung, A.R. The pro-cognitive mechanisms of physical exercise in people with schizophrenia. Br. J. Pharmacol. 2017, 174, 3161–3172. [Google Scholar] [CrossRef]
- Reihmane, D.; Dela, F. Interleukin-6: Possible biological roles during exercise. Eur. J. Sport Sci. 2014, 14, 242–250. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gómez-Rubio, P.; Trapero, I. The Effects of Exercise on IL-6 Levels and Cognitive Performance in Patients with Schizophrenia. Diseases 2019, 7, 11. https://doi.org/10.3390/diseases7010011
Gómez-Rubio P, Trapero I. The Effects of Exercise on IL-6 Levels and Cognitive Performance in Patients with Schizophrenia. Diseases. 2019; 7(1):11. https://doi.org/10.3390/diseases7010011
Chicago/Turabian StyleGómez-Rubio, Pablo, and Isabel Trapero. 2019. "The Effects of Exercise on IL-6 Levels and Cognitive Performance in Patients with Schizophrenia" Diseases 7, no. 1: 11. https://doi.org/10.3390/diseases7010011
APA StyleGómez-Rubio, P., & Trapero, I. (2019). The Effects of Exercise on IL-6 Levels and Cognitive Performance in Patients with Schizophrenia. Diseases, 7(1), 11. https://doi.org/10.3390/diseases7010011




