“Holes” in the Jaw—A Report of Two Cases of Periapical Actinomycosis
Abstract
1. Introduction
2. Case Presentation
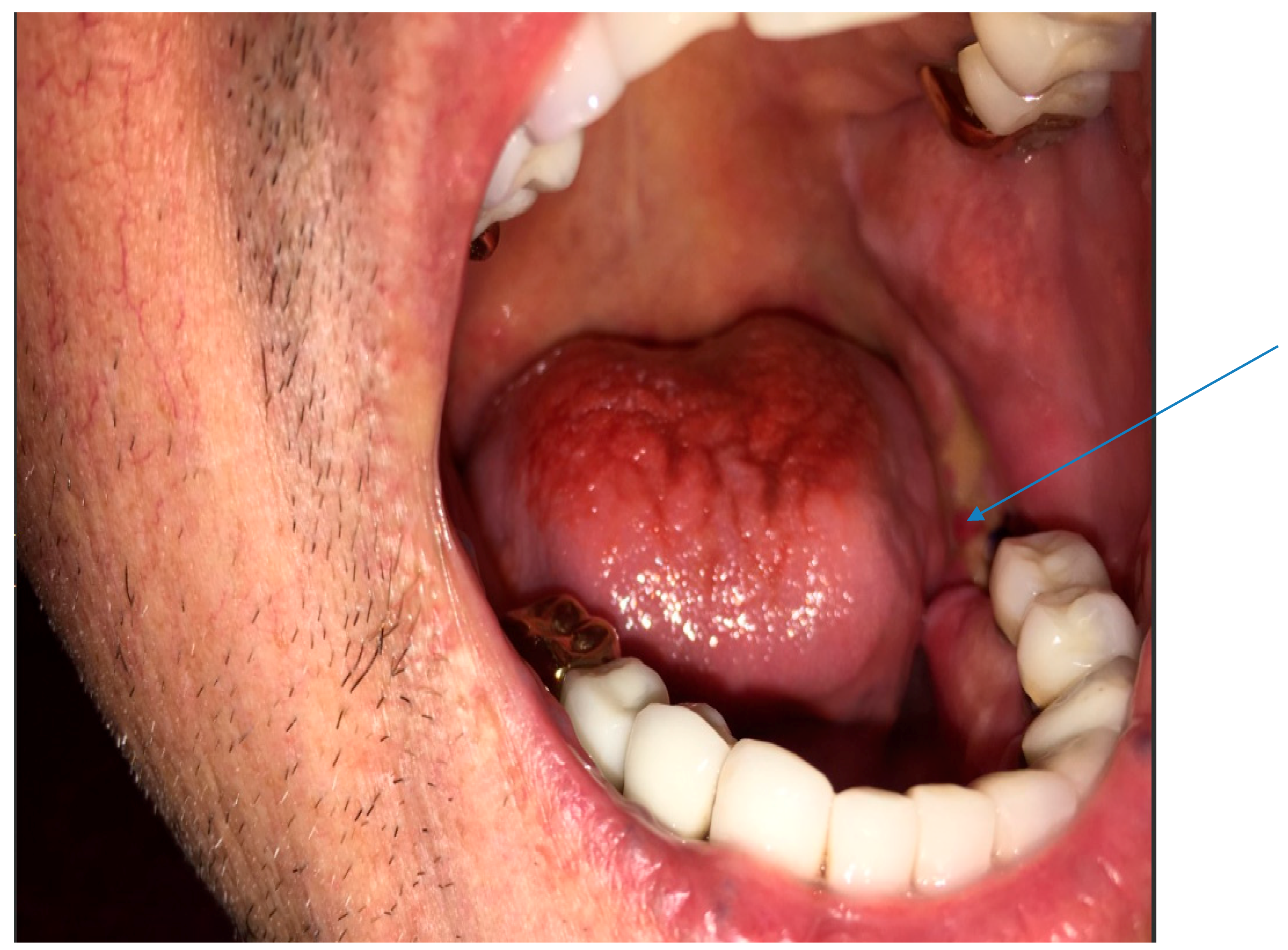
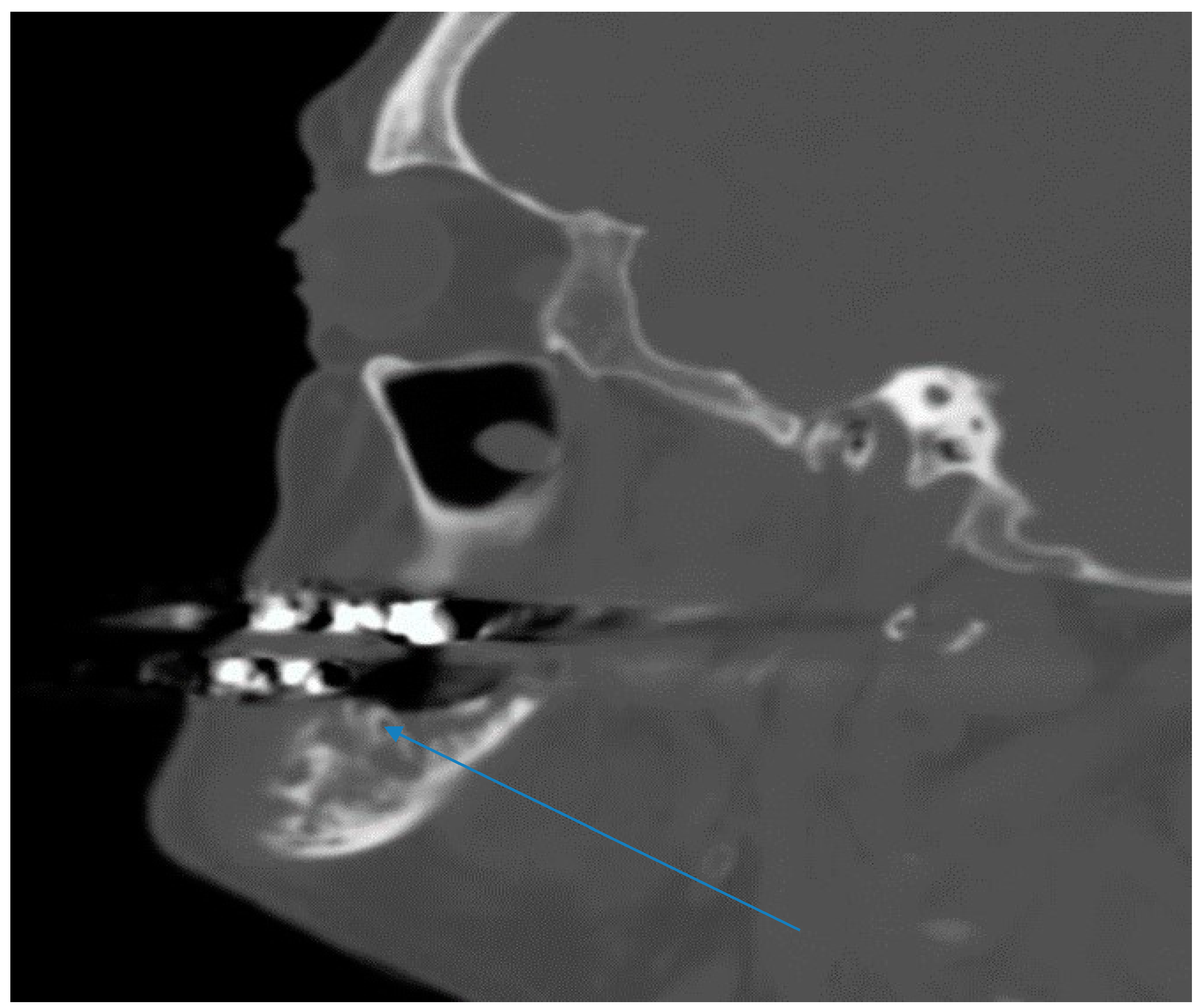
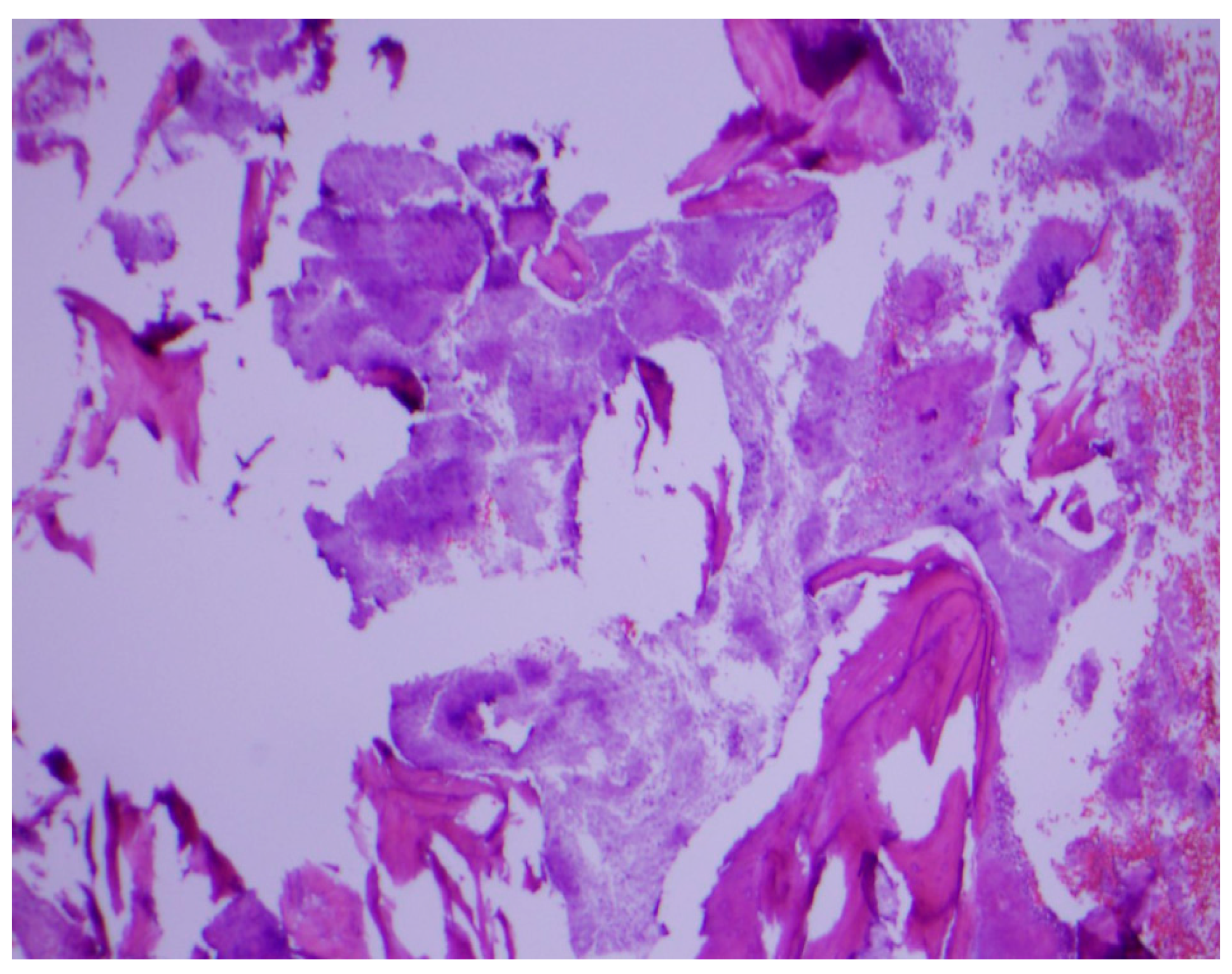
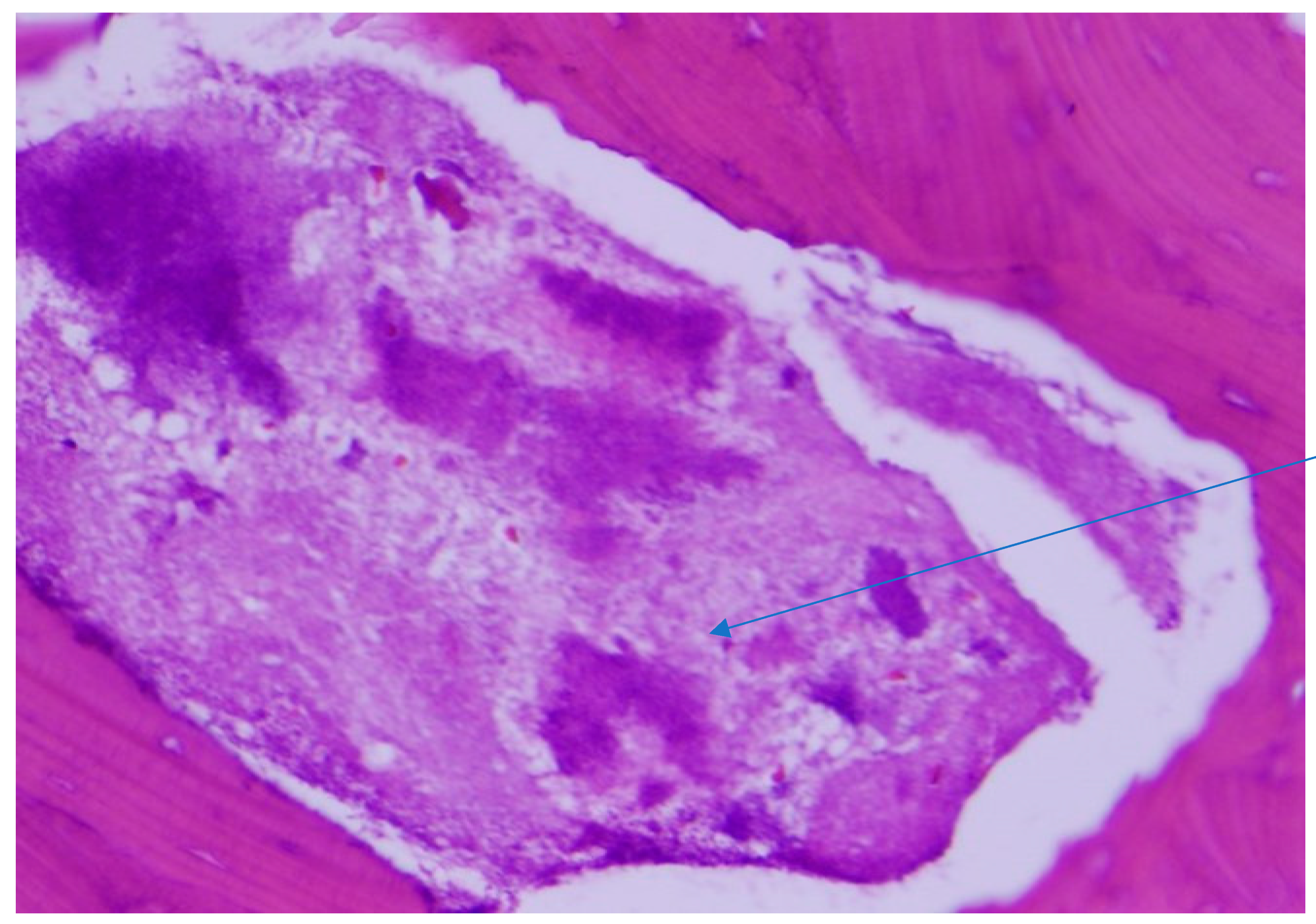
2.1. Case 1
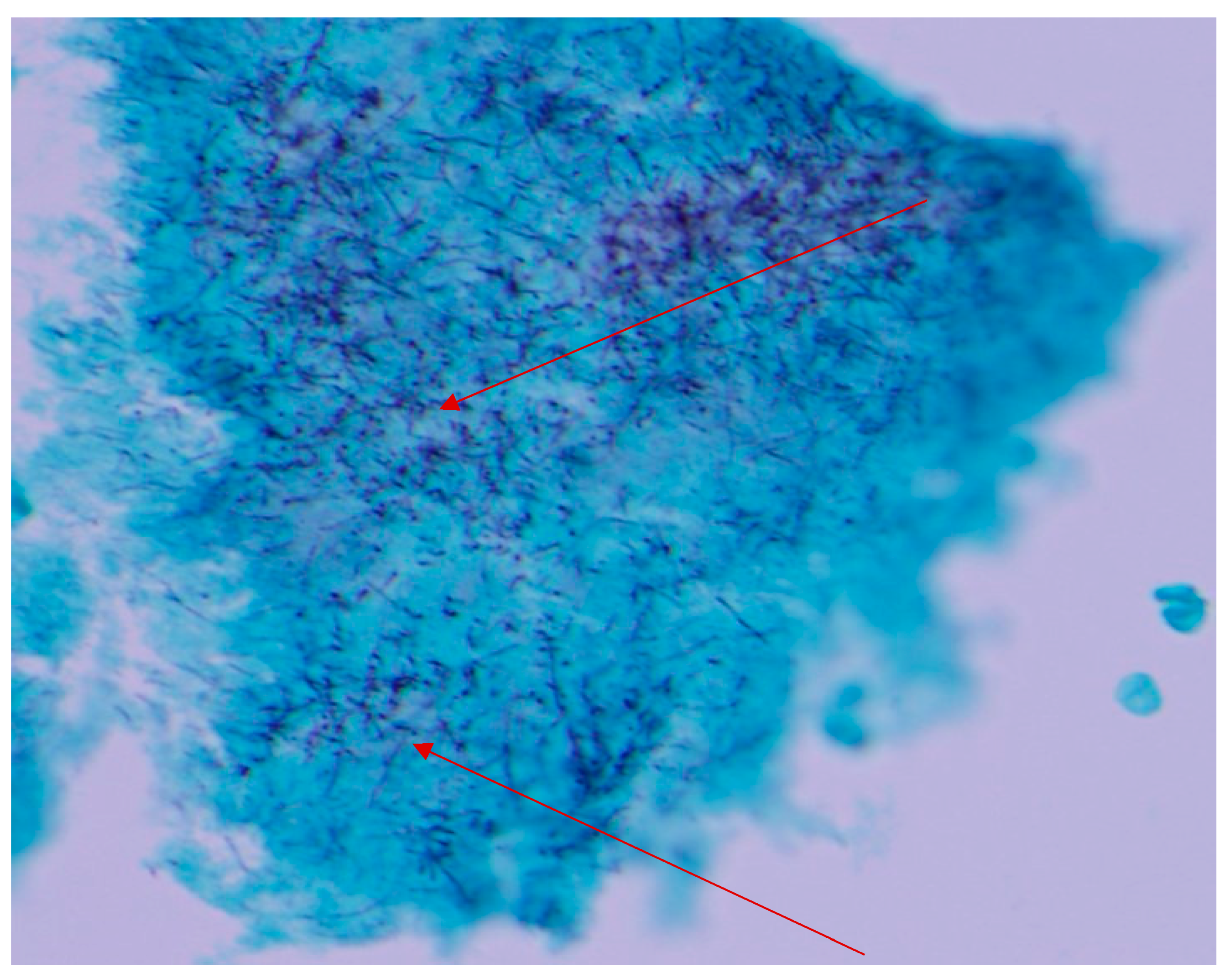
2.2. Case 2
3. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Consent
References
- Grzywa-Celińska, A.; Emeryk-Maksymiuk, J.; Szmygin-Milanowska, K.; Czekajska-Chehab, E.; Milanowski, J. Pulmonary actinomycosis—The great imitator. Ann. Agric. Environ. Med. 2017, 25, 211–212. [Google Scholar] [CrossRef] [PubMed]
- Pasupathy, S.P.; Chakravarthy, D.; Chanmougananda, S.; Nair, P.P. Periapical actinomycosis. BMJ Case Rep. 2012. [Google Scholar] [CrossRef] [PubMed]
- Ricucci, D.; Siqueira, J.F., Jr. Apical actinomycosis as a continuum of intraradicular and extraradicular infection: Case report and critical review on its involvement with treatment failure. J. Endod. 2008, 34, 1124–1129. [Google Scholar] [CrossRef] [PubMed]
- Hirshberg, A.; Tsesis, I.; Metzger, Z.; Kaplan, I. Periapical actinomycosis: A clinicopathologic study. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 2003, 95, 614–620. [Google Scholar] [CrossRef] [PubMed]
- Esteves, L.S.; Henriques, Á.C.; Silva, C.Á.; Cangussu, M.C.; Ramos, E.A.; Estrela, C.; Santos, J.N. Actinomycosis is not Frequent in the Periapex But is a Persistent Lesion. Braz. Dent. J. 2017, 28, 688–693. [Google Scholar] [CrossRef] [PubMed]
- Endo, S.; Mishima, E.; Takeuchi, Y.; Ohi, T.; Ishida, M.; Yanai, M.; Kiyomoto, H.; Nagasawa, T.; Ito, S. Periodontitis-associated septic pulmonary embolism caused by Actinomyces species identified by anaerobic culture of bronchoalveolar lavage fluid: A case report. BMC Infect. Dis. 2015, 15, 552. [Google Scholar] [CrossRef] [PubMed]
- Kim, N.R.; Park, J.B.; Ko, Y. Differential diagnosis and treatment of periodontitis-mimicking actinomycosis. J. Periodontal Implant Sci. 2012, 42, 256–260. [Google Scholar] [CrossRef] [PubMed]
- Lerner, P.I. The lumpy jaw. Cervicofacial actinomycosis. Infect. Dis. Clin. N. Am. 1988, 2, 203–220. [Google Scholar]
- Cole, M.F.; Bryan, S.; Evans, M.K.; Pearce, C.L.; Sheridan, M.J.; Sura, P.A.; Wientzen, R.; Bowden, G.H. Humoral Immunity to Commensal Oral Bacteria in Human Infants: Salivary Antibodies Reactive with Actinomyces naeslundii Genospecies 1 and 2 during Colonization. Infect. Immun. 1998, 66, 4283–4289. [Google Scholar] [PubMed]
- Cho, J.Y. The treatment of actinomycosis mimicking a retained root tip: A confusing case. J. Korean Assoc. Oral Maxillofac. Surg. 2016, 42, 205–208. [Google Scholar] [CrossRef] [PubMed]
- Gannepalli, A.; Ayinampudi, B.K.; Baghirath, P.V.; Reddy, G.V. Actinomycotic Osteomyelitis of Maxilla Presenting as Oroantral Fistula: A Rare Case Report. Case Rep. Dent. 2015, 2015, 689240. [Google Scholar] [CrossRef] [PubMed]
- Miller, M.; Haddad, A.J. Cervicofacial actinomycosis. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 1998, 85, 496–508. [Google Scholar] [CrossRef]
- Rush, J.R.; Sulte, H.R.; Cohen, D.M.; Makkawy, H. Course of infection and case outcome in individuals diagnosed with microbial colonies morphologically consistent with Actinomyces species. J. Endod. 2002, 28, 613–618. [Google Scholar] [CrossRef] [PubMed]
- Marx, R.E.; Carlson, E.R.; Smith, B.R.; Toraya, N. Isolation of Actinomyces species and Eikenella corrodens from patients with chronic diffuse sclerosing osteomyelitis. J. Oral Maxillofac. Surg. 1994, 52, 26–33; discussion 33–34. [Google Scholar] [CrossRef]
- Ledezma-Rasillo, G.; Flores-Reyes, H.; Gonzalez-Amaro, A.M.; Garrocho-Rangel, A.; Ruiz-Rodriguez Mdel, S.; Pozos-Guillen, A.J. Identification of cultivable microorganisms from primary teeth with necrotic pulps. J. Clin. Pediatr. Dent. 2010, 34, 329–333. [Google Scholar] [CrossRef] [PubMed]
- Bennhoff, D.F. Actinomycosis: Diagnostic and therapeutic considerations and a review of 32 cases. Laryngoscope 1984, 94, 1198–1217. [Google Scholar] [CrossRef] [PubMed]
- Kitajima, S.; Mizuno, S.; Kogiso, R.; Tsuji, C. Severe Maxillary Medication-related Osteonecrosis of the Jaw. Intern. Med. 2018. [Google Scholar] [CrossRef] [PubMed]
- Shigemura, T.; Nakamura, J.; Kishida, S.; Harada, Y.; Ohtori, S.; Kamikawa, K.; Ochiai, N.; Takahashi, K. Incidence of osteonecrosis associated with corticosteroid therapy among different underlying diseases: Prospective MRI study. Rheumatology 2011, 50, 2023–2028. [Google Scholar] [CrossRef] [PubMed]
- Oteri, G.; Campisi, G.; Panzarella, V.; Morreale, I.; Nucera, R.; Di Fede, O.; Picone, A.; Marcianò, A. Could the Combined Administration of Bone Antiresorptive Drug, Taxanes, and Corticosteroids Worsen Medication Related Osteonecrosis of the Jaws in Cancer Patients? BioMed Res. Int. 2018, 2018, 4021952. [Google Scholar] [CrossRef] [PubMed]
- De Ceulaer, J.; Tacconelli, E.; Vandecasteele, S.J. Actinomyces osteomyelitis in bisphosphonate-related osteonecrosis of the jaw (BRONJ): The missing link? Eur. J. Clin. Microbiol. Infect. Dis. 2014, 33, 1873–1880. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.Y.; Pien, F.D.; Suzuki, J.B. Identification and treatment of bisphosphonate-associated actinomycotic osteonecrosis of the jaws. Implant Dent. 2011, 20, 331–336. [Google Scholar] [CrossRef] [PubMed]
- Anavi-Lev, K.; Anavi, Y.; Chaushu, G.; Alon, D.M.; Gal, G.; Kaplan, I. Bisphosphonate related osteonecrosis of the jaws: Clinico-pathological investigation and histomorphometric analysis. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2013, 115, 660–666. [Google Scholar] [CrossRef] [PubMed]
- Wong, V.K.; Turmezei, T.D.; Weston, V.C. Actinomycosis. BMJ 2011, 343, d6099. [Google Scholar] [CrossRef] [PubMed]
- Lisa-Gracia, M.; Martín-Rivas, B.; Pajarón-Guerrero, M.; Arnáiz-García, A. Abdominal actinomycosis in the last 10 years and risk factors for appendiceal actinomycosis: Review of the literature. Turk. J. Med. Sci. 2017, 47, 98–102. [Google Scholar] [CrossRef] [PubMed]
- Dixon, W.G.; Abrahamowicz, M.; Beauchamp, M.E.; Ray, D.W.; Bernatsky, S.; Suissa, S.; Sylvestre, M.P. Immediate and delayed impact of oral glucocorticoid therapy on risk of serious infection in older patients with rheumatoid arthritis: A nested case-control analysis. Ann. Rheum. Dis. 2012, 71, 1128–1133. [Google Scholar] [CrossRef] [PubMed]
- Fuenfer, M.M.; Olson, G.E.; Polk, H.C., Jr. Effect of various corticosteroids upon the phagocytic bactericidal activity of neutrophils. Surgery 1975, 78, 27–33. [Google Scholar] [PubMed]
- Li, Y.; Liu, L.; Wang, X.; Zhang, C.; Reyes, J.; Hoffmann, M.; Palmisano, M.; Zhou, S. In Vivo Assessment of the Effect of CYP1A2 Inhibition and Induction on Pomalidomide Pharmacokinetics in Healthy Subjects. J. Clin. Pharmacol. 2018, 58, 1295–1304. [Google Scholar] [CrossRef] [PubMed]
- Lacy, M.Q.; Hayman, S.R.; Gertz, M.A.; Short, K.D.; Dispenzieri, A.; Kumar, S.; Greipp, P.R.; Lust, J.A.; Russell, S.J.; Dingli, D.; et al. Pomalidomide (CC4047) plus low dose dexamethasone (Pom/dex) is active and well tolerated in lenalidomide refractory multiple myeloma (MM). Leukemia 2010, 24, 1934–1939. [Google Scholar] [CrossRef] [PubMed]
- Leleu, X.; Attal, M.; Arnulf, B.; Moreau, P.; Traulle, C.; Marit, G.; Mathiot, C.; Petillon, M.O.; Macro, M.; Roussel, M.; et al. Pomalidomide plus low-dose dexamethasone is active and well tolerated in bortezomib and lenalidomide-refractory multiple myeloma: Intergroupe Francophone du Myélome 2009-02. Blood 2013, 121, 1968–1975. [Google Scholar] [CrossRef] [PubMed]
- Galustian, C.; Meyer, B.; Labarthe, M.C.; Dredge, K.; Klaschka, D.; Henry, J.; Todryk, S.; Chen, R.; Muller, G.; Stirling, D.; et al. The anti-cancer agents lenalidomide and pomalidomide inhibit the proliferation and function of T regulatory cells. Cancer Immunol. Immunother. 2009, 58, 1033–1045. [Google Scholar] [CrossRef] [PubMed]
- Emami, E.; de Souza, R.F.; Kabawat, M.; Feine, J.S. The impact of edentulism on oral and general health. Int. J. Dent. 2013, 2013, 498305. [Google Scholar] [CrossRef] [PubMed]
- Gordon, A.M.; Dick, H.M.; Mason, D.K.; Manderson, W.; Crichton, W.B. Mycoplasmas and recurrent oral ulceration. J. Clin. Pathol. 1967, 20, 865–869. [Google Scholar] [CrossRef] [PubMed]
- Taboza, Z.A.; Costa, K.L.; Silveira, V.R.; Furlaneto, F.A.; Montenegro, R., Jr.; Russell, S.; Dasanayake, A.; Rego, R.O. Periodontitis, edentulism and glycemic control in patients with type 2 diabetes: A cross-sectional study. BMJ Open Diabetes Res. Care 2018, 6, e000453. [Google Scholar] [CrossRef] [PubMed]
- Smith, A.J.; Hall, V.; Thakker, B.; Gemmell, C.G. Antimicrobial susceptibility testing of Actinomyces species with 12 antimicrobial agents. J. Antimicrob. Chemother. 2005, 56, 407–409. [Google Scholar] [CrossRef] [PubMed]
- Crossman, T.; Herold, J. Actinomycosis of the maxilla—A case report of a rare oral infection presenting in general dental practice. Br. Dent. J. 2009, 206, 201–202. [Google Scholar] [CrossRef] [PubMed]
- Alamillos-Granados, F.J.; Dean-Ferrer, A.; García-López, A.; López-Rubio, F. Actinomycotic ulcer of the oral mucosa: An unusual presentation of oral actinomycosis. Br. J. Oral Maxillofac. Surg. 2000, 38, 121–123. [Google Scholar] [CrossRef] [PubMed]
- Valour, F.; Sénéchal, A.; Dupieux, C.; Karsenty, J.; Lustig, S.; Breton, P.; Gleizal, A.; Boussel, L.; Laurent, F.; Braun, E.; Chidiac, C. Actinomycosis: Etiology, clinical features, diagnosis, treatment, and management. Infect. Drug Resist. 2014, 7, 183–197. [Google Scholar] [PubMed]
- Sezer, B.; Akdeniz, B.G.; Günbay, S.; Hilmioğlu-Polat, S.; Başdemir, G. Actinomycosis osteomyelitis of the jaws: Report of four cases and a review of the literature. J. Dent. Sci. 2017, 12, 301–307. [Google Scholar] [CrossRef]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ayoade, F.; Olayiwola, A.; Li, A. “Holes” in the Jaw—A Report of Two Cases of Periapical Actinomycosis. Diseases 2018, 6, 79. https://doi.org/10.3390/diseases6030079
Ayoade F, Olayiwola A, Li A. “Holes” in the Jaw—A Report of Two Cases of Periapical Actinomycosis. Diseases. 2018; 6(3):79. https://doi.org/10.3390/diseases6030079
Chicago/Turabian StyleAyoade, Folusakin, Ayoola Olayiwola, and Ailing Li. 2018. "“Holes” in the Jaw—A Report of Two Cases of Periapical Actinomycosis" Diseases 6, no. 3: 79. https://doi.org/10.3390/diseases6030079
APA StyleAyoade, F., Olayiwola, A., & Li, A. (2018). “Holes” in the Jaw—A Report of Two Cases of Periapical Actinomycosis. Diseases, 6(3), 79. https://doi.org/10.3390/diseases6030079




