Wine and Olive Oil Phenolic Compounds Interaction in Humans
Abstract
1. Introduction
2. Materials and Methods
2.1. Subjects and Study Design
2.2. Red Wine and Extra Virgin Olive Oil
2.3. Standards and Reagents
2.4. Extraction and Analysis of Resveratrol in Red Wine and Urine Samples
2.4.1. Red Wine
2.4.2. Urine Samples
2.5. Extraction and Analysis of Hydroxytyrosol in Extra Virgin Olive Oil, Red Wine and Urine Samples
2.5.1. Extra Virgin Olive Oil
2.5.2. Red Wine
2.5.3. Urine Samples
2.6. Ethyl Glucuronide Quantification
2.7. Statistical Analysis
3. Results
3.1. Phenolic Content in Extra Virgin Olive Oil and Red Wine
3.2. Quantification of Phenolic Compounds in Urine
3.2.1. Baseline
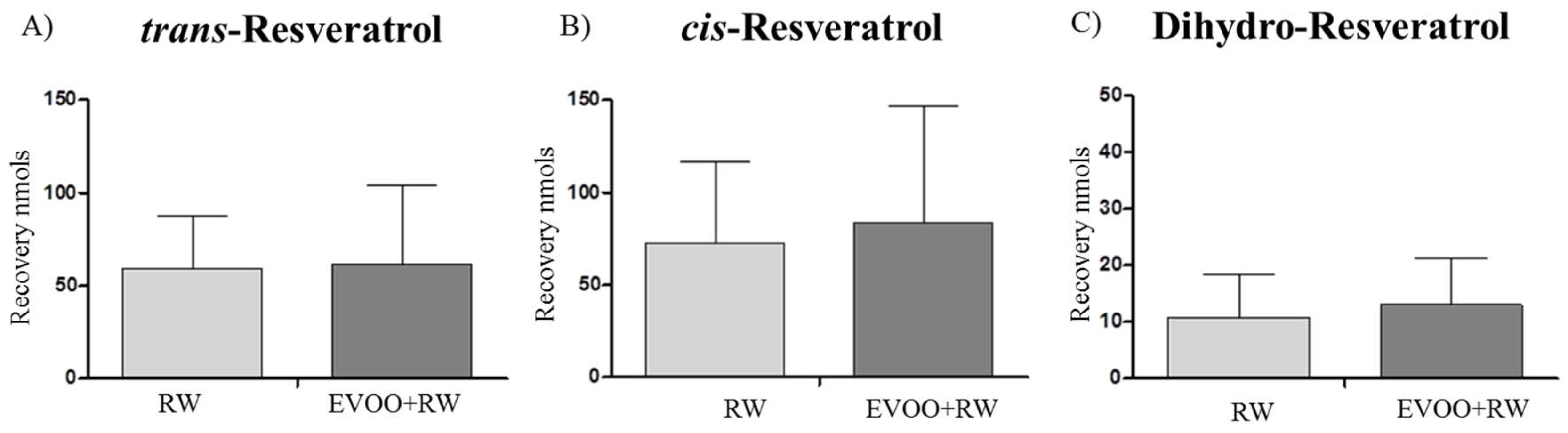
3.2.2. Resveratrol
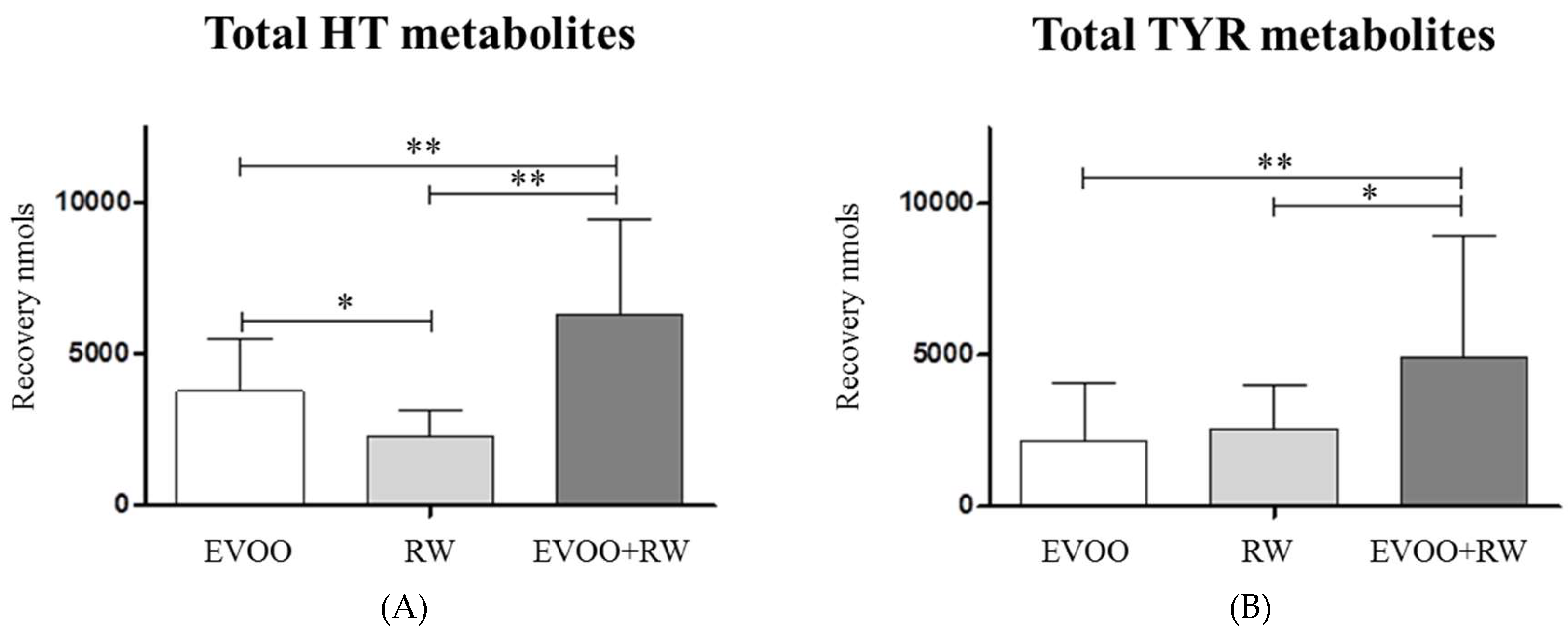
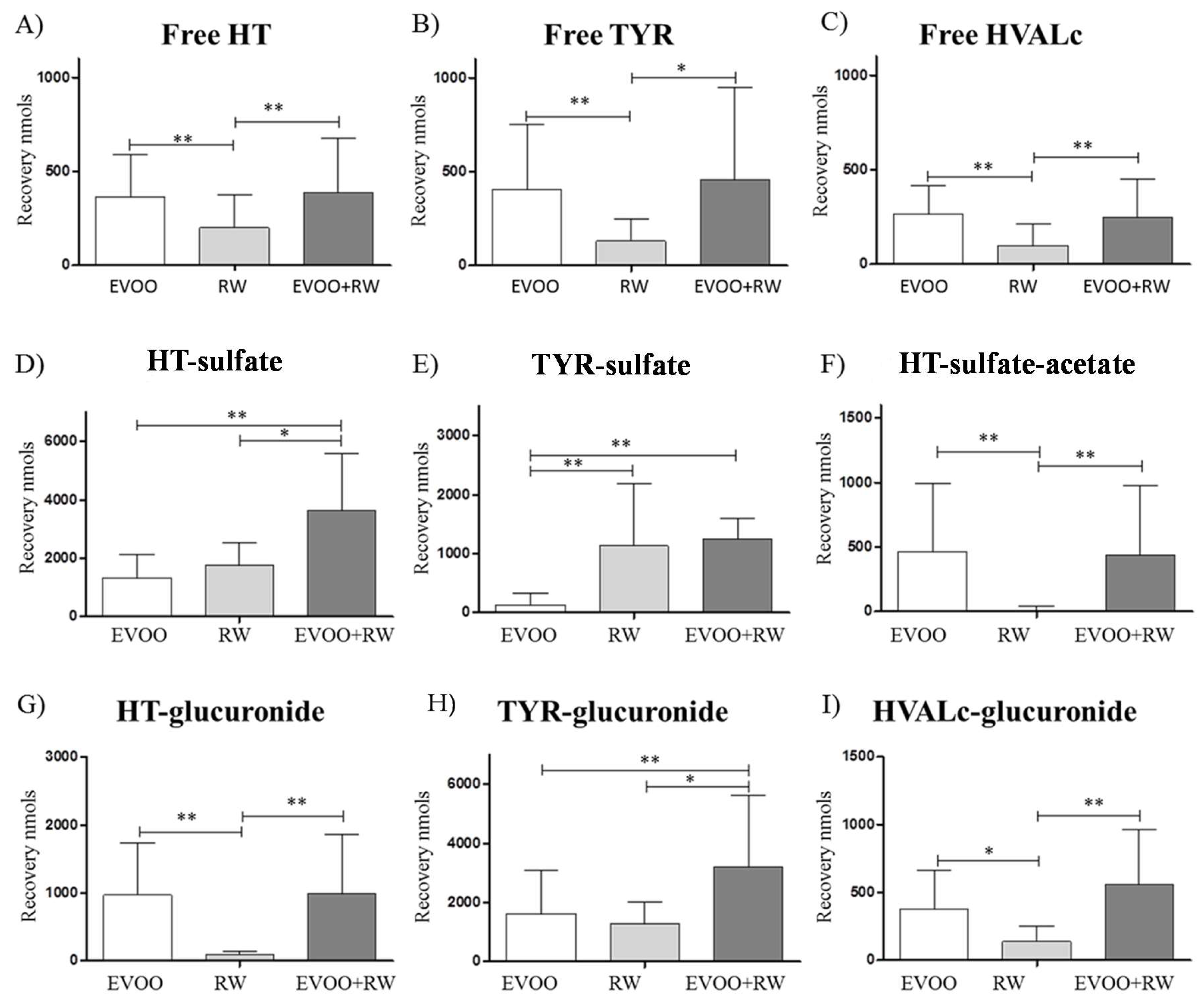
3.2.3. Hydroxytyrosol
3.3. Ethyl Glucuronide
4. Discussion
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| NH4I | ammonium iodide; |
| EFSA | European Food Safety Agency; |
| EVOO | extra virgin olive oil; |
| GC-MS | gas chromatography coupled to mass spectrometry; |
| H-COOH | formic acid; |
| HVALc | homovanillyl alcohol; |
| HCl | hydrochloric acid; |
| HT | hydroxytyrosol; |
| IS | internal standard; |
| LC-MS/MS | liquid chromatography coupled to tandem mass spectrometry; |
| MD | Mediterranean diet; |
| MSTFA | N-methyl-N-trimethylsilyltrifluoroacetamide; |
| RW | red wine; |
| RESV | resveratrol; |
| NaCl | sodium chloride; |
| NaOH | sodium hydroxide; |
| TYR | tyrosol. |
References
- Sofi, F.; Cesari, F.; Abbate, R.; Gensini, G.F.; Casini, A. Adherence to Mediterranean diet and health status: Meta-analysis. BMJ 2008, 337, 673–675. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Morató, J.; Boronat, A.; Kotronoulas, A.; Pujadas, M.; Pastor, A.; Olesti, E.; Pérez-Mañá, C.; Khymenets, O.; Fitó, M.; Farré, M.; et al. Metabolic disposition and biological significance of simple phenols of dietary origin: Hydroxytyrosol and tyrosol. Drug Metab. Rev. 2016, 48, 218–236. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Huélamo, M.; Rodríguez-Morató, J.; Boronat, A.; de la Torre, R. Modulation of Nrf2 by olive oil and wine polyphenols and neuroprotection. Antioxidants 2017, 6, 73. [Google Scholar] [CrossRef] [PubMed]
- Ragusa, A.; Centonze, C.; Grasso, M.E.; Latronico, M.F.; Mastrangelo, P.F.; Fanizzi, F.P.; Maffia, M. Composition and Statistical Analysis of Biophenols in Apulian Italian EVOOs. Foods 2017, 6, 90. [Google Scholar] [CrossRef] [PubMed]
- Ragusa, A.; Centonze, C.; Grasso, M.E.; Latronico, M.F.; Mastrangelo, P.F.; Sparascio, F.; Fanizzi, F.P.; Maffia, M. A Comparative Study of Phenols in Apulian Italian Wines. Foods 2017, 6, 24. [Google Scholar] [CrossRef] [PubMed]
- Ortuño, J.; Covas, M.I.; Farre, M.; Pujadas, M.; Fito, M.; Khymenets, O.; Andres-Lacueva, C.; Roset, P.; Joglar, J.; Lamuela-Raventós, R.M.; et al. Matrix effects on the bioavailability of resveratrol in humans. Food Chem. 2010, 120, 1123–1130. [Google Scholar] [CrossRef]
- Gambini, J.; Inglés, M.; Olaso, G.; Lopez-Grueso, R.; Bonet-Costa, V.; Gimeno-Mallench, L.; Mas-Bargues, C.; Abdelaziz, K.M.; Gomez-Cabrera, M.C.; Vina, J.; et al. Properties of Resveratrol: In Vitro and In Vivo Studies about Metabolism, Bioavailability, and Biological Effects in Animal Models and Humans. Oxid. Med. Cell. Longev. 2015, 2015, 837042. [Google Scholar] [CrossRef] [PubMed]
- Zordoky, B.N.M.; Robertson, I.M.; Dyck, J.R.B. Preclinical and clinical evidence for the role of resveratrol in the treatment of cardiovascular diseases. Biochim. Biophys. Acta 2014, 1852, 1155–1177. [Google Scholar] [CrossRef] [PubMed]
- Park, E.J.; Pezzuto, J.M. The pharmacology of resveratrol in animals and humans. Biochim. Biophys. Acta Mol. Basis Dis. 2015, 1852, 1071–1113. [Google Scholar] [CrossRef] [PubMed]
- Monika, B.S.P.; Garg, R.; Sardana, S. Research Problems Associated with Resveratrol (trans-3,5,4′-trihydroxystilbene; RSV) and Various Strategies to Overcome those Problems (Review). Curr. Drug Deliv. 2017, 14, 364–376. [Google Scholar] [CrossRef] [PubMed]
- Di Benedetto, R.; Varì, R.; Scazzocchio, B.; Filesi, C.; Santangelo, C.; Giovannini, C.; Matarrese, P.; D'Archivio, M.; Masella, R. Tyrosol, the major extra virgin olive oil compound, restored intracellular antioxidant defences in spite of its weak antioxidative effectiveness. Nutr. Metab. Cardiovasc. Dis. 2007, 17, 535–545. [Google Scholar] [CrossRef] [PubMed]
- Warleta, F.; Quesada, C.S.; Campos, M.; Allouche, Y.; Beltrán, G.; Gaforio, J.J. Hydroxytyrosol protects against oxidative DNA damage in human breast cells. Nutrients 2011, 3, 839–857. [Google Scholar] [CrossRef] [PubMed]
- Covas, M.I.; Nyyssönen, K.; Poulsen, H.E.; Kaikkonen, J.; Zunft, H.J.; Kiesewetter, H.; Gaddi, A.; de la Torre, R.; Mursu, J.; Bäumler, H.; et al. The Effect of Polyphenols in Olive Oil on Heart Disease Risk Factors. Ann. Intern. Med. 2006, 145, 333–341. [Google Scholar] [CrossRef] [PubMed]
- Vilaplana-Pérez, C.; Auñón, D.; García-Flores, L.A.; Gil-Izquierdo, A. Hydroxytyrosol and Potential Uses in Cardiovascular Diseases, Cancer, and AIDS. Front. Nutr. 2014, 1, 18. [Google Scholar] [PubMed]
- Rodríguez-Morató, J.; Xicota, L.; Fitó, M.; Farré, M.; Dierssen, M.; De La Torre, R. Potential role of olive oil phenolic compounds in the prevention of neurodegenerative diseases. Molecules 2015, 20, 4655–4680. [Google Scholar] [CrossRef] [PubMed]
- De la Torre, R.; Corella, D.; Castaner, O.; Martínez-González, M.A.; Salas-Salvador, J.; Vila, J.; Estruch, R.; Sorli, J.V.; Arós, F.; Fiol, M.; et al. Protective effect of homovanillyl alcohol on cardiovascular disease and total mortality: Virgin olive oil, wine, and catechol-methylathion. Am. J. Clin. Nutr. 2017, 105, 1297–1304. [Google Scholar] [CrossRef] [PubMed]
- De la Torre, R.; Covas, M.I.; Pujadas, M.A.; Fitó, M.; Farré, M. Is dopamine behind the health benefits of red wine? Eur. J. Nutr. 2006, 45, 307–310. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Mañá, C.; Farré, M.; Pujadas, M.; Mustata, C.; Menoyo, E.; Pastor, A.; Langohr, K.; de la Torre, R. Ethanol induces hydroxytyrosol formation in humans. Pharmacol. Res. 2015, 9596, 27–33. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Mañá, C.; Farré, M.; Rodríguez-Morató, J.; Papaseit, E.; Pujadas, M.; Fitó, M.; Robledo, P.; Covas, M.I.; Cheynier, V.; Meudec, E.; et al. Moderate consumption of wine, through both its phenolic compounds and alcohol content, promotes hydroxytyrosol endogenous generation in humans. A randomized controlled trial. Mol. Nutr. Food Res. 2015, 59, 1213–1216. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Morató, J.; Robledo, P.; Tanner, J.A.; Boronat, A.; Pérez-Mañá, C.; Chen, C.Y.; Tyndale, R.F.; de la Torre, R. CYP2D6 and CYP2A6 biotransform dietary tyrosol into hydroxytyrosol. Food Chem. 2017, 217, 716–725. [Google Scholar] [CrossRef] [PubMed]
- EFSA Panel on Dietetic Products, Nutrition and Allergies (NDA). Scientific Opinion on the substantiation of health claims related to fruits and/or vegetables (ID 1212, 1213, 1214, 1217, 1218, 1219, 1301, 1425, 1426, 1427, 1428, 1429, 1430) and to the “Mediterranean diet” (ID 1423) pursuant to Article 13(1) of Regulation (EC) No 1924/2006. EFSA J. 2011, 9, 2245. [Google Scholar]
- Rotches-Ribalta, M.; Urpi-Sarda, M.; Llorach, R.; Boto-Ordoñez, M.; Jauregui, O.; Chiva-Blanch, G.; Perez-Garcia, L.; Jaeger, W.; Guillen, M.; Corella, D.; et al. Gut and microbial resveratrol metabolite profiling after moderate long-term consumption of red wine versus dealcoholized red wine in humans by an optimized ultra-high-pressure liquid chromatography tandem mass spectrometry method. J. Chromatogr. A 2012, 1265, 105–113. [Google Scholar] [CrossRef] [PubMed]
- Miro-Casas, E.; Covas, M.I.; Farre, M.; Fito, M.; Ortuño, J.; Weinbrenner, T.; Roset, P.; de la Torre, R. Hydroxytyrosol disposition in humans. Clin. Chem. 2003, 49, 945–952. [Google Scholar] [CrossRef] [PubMed]
- Khymenets, O.; Fito, M.; Touriño, S.; Muñoz-Aguayo, D.; Pujadas, M.A.; Torres, J.L.; Joglar, J.; Farré, M.; Covas, M.I.; de La Torre, R. Antioxidant Activities of Hydroxytyrosol Main Metabolites Do Not Contribute to Beneficial Health Effects after Olive Oil Ingestion. Drug Metab. Dispos. 2010. [Google Scholar] [CrossRef] [PubMed]
- Kotronoulas, A.; Pizarro, N.; Serra, A.; Robledo, P.; Joglar, J.; Rubió, L.; Hernaéz, Á.; Tormos, C.; Motilva, M.J.; Fitó, M.; et al. Dose-dependent metabolic disposition of hydroxytyrosol and formation of mercapturates in rats. Pharmacol. Res. 2013, 77, 47–56. [Google Scholar] [CrossRef] [PubMed]
- Visioli, F.; Galli, C.; Grande, S.; Colonnelli, K.; Patelli, C.; Galli, G.; Caruso, D. Hydroxytyrosol excretion differs between rats and humans and depends on the vehicle of administration. J. Nutr. 2003, 133, 2612–2615. [Google Scholar] [CrossRef] [PubMed]
- Migliori, M.; Panichi, V.; de la Torre, R.; Fitó, M.; Covas, M.; Bertelli, A.; Muñoz-Aguayo, D.; Scatena, A.; Paoletti, S.; Ronco, C. Anti-inflammatory effect of white wine in CKD patients and healthy volunteers. Blood Purif. 2015, 39, 218–223. [Google Scholar] [CrossRef] [PubMed]
- Atzeri, A.; Lucas, R.; Incani, A.; Peñalver, P.; Zafra-Gómez, A.; Melis, M.P.; Pizzala, R.; Morales, J.C.; Deiana, M. Hydroxytyrosol and tyrosol sulfate metabolites protect against the oxidized cholesterol pro-oxidant effect in Caco-2 human enterocyte-like cells. Food Funct. 2016, 7, 337–346. [Google Scholar] [CrossRef] [PubMed]
| Treatment | Concentration (mg/L) | Dose Administered (mL) | Dose Administered (mg) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| t-RESV | c-RESV | t-Piceid | c-Piceid | HT | TYR | t-RESV | c-RESV | t-Piceid | c-Piceid | HT | TYR | ||
| EVOO | 0 | 0 | 0 | 0 | 19.8 ± 1.9 | 24.1 ± 2.8 | 25 | 0 | 0 | 0 | 0 | 0.50 | 0.60 |
| RW | 2.4 ± 0.1 | 3.0 ± 0.4 | 4.9 ± 0.2 | 3.0 ± 0.4 | 1.5 ± 0.1 | 35.0 ± 1.0 | 150 | 0.36 | 0.45 | 0.74 | 0.45 | 0.22 | 5.25 |
| EVOO + RW | NA | NA | NA | NA | NA | NA | 25 + 150 | 0.36 | 0.45 | 0.74 | 0.45 | 0.72 | 5.85 |
| Phenolic Compound (nmols) | EVOO | RW | EVOO + RW |
|---|---|---|---|
| t-RESV | 0.0 ± 0.0 | 59.2 ± 28.7 aa | 61.7 ± 42.4 aa |
| c-RESV | 0.0 ± 0.0 | 72.8 ± 44.3 aa | 83.8 ± 62.6 aa |
| Dihydro-RESV | 0.0 ± 0.0 | 10.9 ± 7.5 aa | 13.0 ± 8.3 aa |
| Phenolic Compound (nmols) | EVOO | RW | EVOO + RW |
|---|---|---|---|
| Total HT | 3788 ±1751 | 2308 ± 847 a | 6286 ± 3198 aa bb |
| Total TYR | 2180 ± 1917 | 2567 ± 1468 | 4925 ± 3993 aa b |
| Free HT | 367 ± 221 | 201 ± 173 aa | 386 ± 289 bb |
| Free TYR | 404 ± 346 | 132 ± 114 aa | 460 ± 490 b |
| Free HVALc | 269 ± 145 | 110 ± 118 aa | 247 ± 205 bb |
| HT-sulfate | 1336 ± 795 | 1767 ± 787 | 3655 ± 1926 aa b |
| TYR-sulfate | 138 ±194 | 1133 ± 1052 aa | 1252 ± 1190 bb |
| HT-acetate-sulfate | 465 ± 528 | 11.2 ± 30.9 aa | 436 ± 543 bb |
| HT-glucuronide | 974 ± 766 | 90.5 ± 56.3 aa | 1000 ± 856 bb |
| TYR-glucuronide | 1639 ± 1438 | 1301 ± 720 | 3215 ± 2421 aa b |
| HVALc-glucuronide | 376 ± 284 | 139 ± 114 a | 563 ± 401 bb |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Boronat, A.; Martínez-Huélamo, M.; Cobos, A.; De la Torre, R. Wine and Olive Oil Phenolic Compounds Interaction in Humans. Diseases 2018, 6, 76. https://doi.org/10.3390/diseases6030076
Boronat A, Martínez-Huélamo M, Cobos A, De la Torre R. Wine and Olive Oil Phenolic Compounds Interaction in Humans. Diseases. 2018; 6(3):76. https://doi.org/10.3390/diseases6030076
Chicago/Turabian StyleBoronat, Anna, Miriam Martínez-Huélamo, Ariadna Cobos, and Rafael De la Torre. 2018. "Wine and Olive Oil Phenolic Compounds Interaction in Humans" Diseases 6, no. 3: 76. https://doi.org/10.3390/diseases6030076
APA StyleBoronat, A., Martínez-Huélamo, M., Cobos, A., & De la Torre, R. (2018). Wine and Olive Oil Phenolic Compounds Interaction in Humans. Diseases, 6(3), 76. https://doi.org/10.3390/diseases6030076




